Abstract
In the present paper, the possibility to use dialkyl phosphate metabolites (DAPs) hair segmental analysis as a biomarker of past acute exposure to organophosphates is examined. Hair samples of four acute poisoning survivors were collected and segmental hair analysis was performed. The total hair samples were divided to 1 cm segments and analysed by gas chromatography-mass spectrometry (GC-MS) for the presence of four DAP metabolites, dimethyl phosphate (DMP), diethyl phosphate (DEP), diethyl thiophosphate (DETP) and diethyl dithiophosphate (DEDTP). Results were examined under the light of pesticide type and time of hair sample collection. Although DAPs were detected all along the hair shaft, higher concentrations (peaks) were detected in the segments proximate to the suicide period. It was also observed that the elevated concentrations of the present metabolites corresponded to the ones produced by the ingested parent compound. Conclusively, measurements of DAPs in the appropriate hair segments of OP-poisoned patients can be used for assessing past acute exposure to organophosphates in certain cases.
Keywords
Introduction
The wide use of organophosphates (OPs) leads to exposure of workers and the general population to these substances. Exposure may either be occupational or accidental. The consumption of contaminated food and residential application are considered as the main routes of exposure of the general population. 1 A very special group of exposed population is the people who have deliberately ingested OPs in a suicide attempt. These people suffer acute exposure usually to a substantial dose and the outcome, which is a multifactor event, is based mainly on the ingested pesticide, the dose and the time interval between the suicide attempt and hospitalization. 2,3
Many different strategies are followed for the biomonitoring of exposure to these pesticides. For the assessment of acute exposure, the most commonly used biomarker is the inhibition of plasma acetyl-cholinesterase 4,5 or the direct measurement of the concentration of pesticide in blood or urine of the patient. 6,7
The measurement of dialkyl phosphate metabolites (DAPs), non-selective metabolites of OPs, in urine and various other biological samples has been proposed as a biomarker of cumulative exposure to OPs in the last decade. 8 –11 Although hair analysis is mostly used for the assessment of low level chronic exposure to various chemicals including pesticides, 12 –15 in the present article the possibility of using segmental hair analysis for the assessment of past acute exposure to three OP pesticides is examined, through the presentation of results of segmental hair analysis for DAPs in four patients that attempted suicide by ingesting various quantities of OPs. All patients survived the attempt. Hair sampling was performed once for each patient, on one of their follow-up appointments.
Patients history
The patients were all female between 14 and 29 years of age, citizens of Sri Lanka and all had survived suicide attempts by ingesting various quantities of OP insecticides (Table 1 ).
The ingested pesticide, the produced dialkyl phosphate metabolite and total length of hair sample for each patient
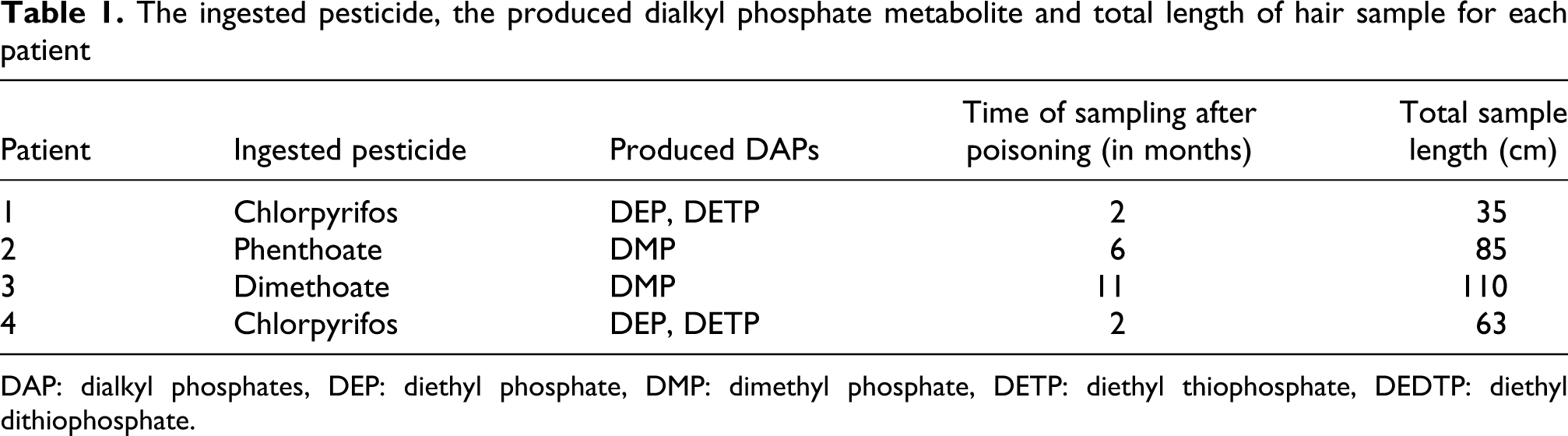
DAP: dialkyl phosphates, DEP: diethyl phosphate, DMP: dimethyl phosphate, DETP: diethyl thiophosphate, DEDTP: diethyl dithiophosphate.
Patient 1
A 14-year-old female patient was presented to the study hospital 4.5 hours after ingesting 150 ml of chlorpyrifos. She was presented to a nearby peripheral hospital where she spontaneously vomited. Forced emesis was done at peripheral hospital and she was also given a single 0.6 mg bolus of atropine. On admission to study hospital, she was symptomatic with vomiting and low blood pressure (BP) 90/60. Her admission pulse was 100 regular, respiratory rate (RR) 26, pupil 4/4, Glasgow Coma Scale (GCS) 6/15 and had bilateral crepitating in the lungs. On 5th hospital day, she was sent to intensive care unit (ICU) for few hours. During her hospital stay, she received I.V. N/S (normal saline), atropine, pralidoxime and antibiotic, and single dose of activated charcoal. On day 11, she was discharged home. Hair sampling was performed during her follow-up appointment, 2 months after the attempt.
Patient 2
A 29-year-old female patient was presented to study hospital 2 hours after ingesting 50 ml of phenthoate. She was presented to a nearby peripheral hospital where she had vomited several times and forced emesis was done at peripheral hospital before transfer. On admission to study hospital, she was conscious and her GCS was 15/15. On examination, her admission findings included BP 110/80, pulse 72, RR 16, pupils 3/3 and had clear lungs. She had vomited on admission, had diarrhea and abdominal pain. She was given a single dose of activated charcoal and observed for change in her clinical signs. During her hospital stay, she was given 1 g pralidoxime (PAM), 6 hourly for first 2 days. In addition to this, she was on atropine infusion for few hours. She also received antibiotic. She was discharged on 5th hospital day. Hair sampling was performed during her follow-up appointment, 6 months after the attempt.
Patient 3
A 21-year-old female was admitted to study hospital with the history of ingestion of 60 ml dimethoate 5 hours earlier. She was initially presented to a peripheral hospital where she vomited several times and forced emesis was performed. On admission to study hospital, her examination findings were pulse 88, BP 100/60, RR 28, pupil 3/3, GCS 15/15 and had clear lungs. She had vomited on admission but no decontamination was performed. During her hospital stay, she was given few bolus doses of atropine and antibiotic. She was discharged on 4th hospital day. Hair sampling was performed during her follow-up appointment, 11 months after the attempt.
Patient 4
A 27 year old female admitted to study hospital after 4 hours of ingesting 15 ml of chlorpyriphos. She was initially presented to a peripheral hospital where forced emesis was induced and immediately transferred to study hospital. She had spontaneously vomited at peripheral hospital. On admission to study hospital, her GCS was 15/15 and other clinical parameters included BP 110/80, RR 22, pulse 130, pupil 4/4 and had clear lungs. No gastrointestinal decontamination was performed. During her hospital stay, she was given few bolus doses of atropine and normal saline. She was discharged on 4th day. Hair sampling was performed during her follow-up appointment, 2 months after the attempt.
Materials and methods
Sampling
Hair samples of the four patients were removed on one of their follow-up appointments. An informed written consent form was filled by the patients on their follow-up appointment before obtaining the hair samples.
The hair sample was posted to the Laboratory of Toxicology, Medical School University of Crete from the School of Medicine University of Peradeniya, Sri Lanka. The samples were cut to 1 cm segments, each segment corresponding to one month of exposure, 16,17 and the first 20 segments of each sample were analysed for the presence of dimethyl phosphate (DMP), diethyl phosphate (DEP), diethyl thiophosphate (DETP) and diethyl dithiophosphate (DEDTP), in order to obtain the DAPs profile along the hair shaft (Table 1).
Materials
DMP (98%) was purchased from Acros Organics (Geel, Belgium, NJ, USA), DEP (98.9%) from Chem Service (West Chester, USA), O,O-diethylthiophosphate potassium salt (98%; DETP) and diethyldithiophosphate salt (95%; DEDTP) from Sigma-Aldrich (USA). Toluene (99.5%) and potassium carbonate (K2CO3) were obtained from Merck (Darmstadt, Germany). Acetonitrile and dibutyl phosphate (97%) were purchased from Roth (Karlsruhe, Germany) and Fluka (Steinheim, Germany), respectively. The derivatizing agent 2,3,4,5,6 pentafluorobenzylbromide (PFBBr; 99%) was obtained from Sigma-Aldrich (Steinheim, Germany).
Dialkyl phosphate extraction from hair
Upon receipt, each sample was decontaminated by washing twice in water (for 10 min) and methanol (for 2 min) and drying in the oven at 36°C. It was subsequently cut into approximately 1 cm segments, based on the assumption that each centimeter of hair corresponds to 1 month of exposure and analysed. The extraction procedure has been described before. 18 Briefly, each segment was pulverized in a ball mill homogenizer (Retsch-MM 2000, Bioblock Scientific, B.P.111, F67403 Illkirch Cedex) and the powder was transferred in a test tube with 2 ml of methanol and 25 μl of DBP (10 μg/ml). Hair was incubated at room temperature in an ultrasonic bath for 4 hours and liquid-solid extraction was performed for 0.5 hour by mechanical shaking. The mixture was centrifuged at 4000 rpm for 5 min. The supernatant was transferred through an econofilter 0.2 μm, 25 mm (Agilent Technology) to a test tube containing 15 mg K2CO3 and methanol was evaporated to dryness under a gentle nitrogen stream at room temperature. Fifteen milligrams (15 mg) of K2CO3 was added to the residue, reconstituted in 1 ml of acetonitrile and 0.1 ml solution of PFBBr in acetonitrile (1:3 v/v) and incubated at 80°C in a water bath for 30 min with occasional swirling. The mixture was then brought to room temperature and the acetonitrile was evaporated to dryness under nitrogen at 35°C. The residue was finally dissolved in 50 μl of toluene and 2 μl was injected to gas chromatography-mass spectrometry (GC-MS).
Analysis of DAPs
Electron ionisation mass spectrometric analysis was performed on a GC MS QP-2010 Shimadzu system equipped with a BPX5 (30 m × 0.25 mm × 0.25 μm) capillary column (SGE). Pure helium (99.99%) with a column flow of 1 ml/min was used as a carrier gas. The column temperature was initially held at 60°C for 1 min, raised to 180°C at 20°C/min, held for 1 min, raised to 250°C at 4°C/min, held for 1 min and was finally raised to 300°C, at 25°C/min, where it remained stable for 2 min. Under these conditions, the retention times of DMP, DEP, DETP, DEDTP and DBP were 17.34, 19.84, 23.40, 24.97 and 26.98 min, respectively. The injector temperature was 270°C. The interface temperature was set at 300°C. The ion source temperature was 230°C.
Quantitative analysis was achieved in selected ion monitoring (SIM) mode with a scan time of 0.2 s. The quantification target ions (m/z) for each DAP were 306 for DMP, 258 for DEP, 350 for DETP and 366 for DEDTP and the qualifier ions were 110 and 306 for DMP, 334 and 258 for DEP, 274 and 350 for DETP, 185 and 366 for DEDTP.
Statistical analysis—Presentation of results
Levels of metabolites in hairs were expressed as means ± standard deviation (SD). Scatter plots were produced using Microsoft Excel 2007. Examination of “peaks” was performed using one-sample t-test setting significance level at 0.05. PASW Statistics 18.0 (former SPSS) was used for statistical purposes.
Results
Method evaluation
Blank hair samples were spiked with known concentrations of DMP, DEP, DETP or DEDTP in concentration range from 0 to 500 pg/mg and used as spiked samples for the calibration curves. The calibration curves obtained from the spiked hair samples were linear with coefficients of linearity greater than 0.99. The limit of quantification (LOQ) was defined as the concentration level that gave a peak with a signal to noise ratio of 10 and determined at 20 pg/mg for DMP, 10 pg/mg for DEP, and DETP and 5 pg/mg for DEDTP. The mean recoveries ranged from 84.3% for DMP to 116.1% for DEP. 18
Biological monitoring
The maximum and mean concentration ± SD of the four analysed DAPs for each patient are summarized in Table 2 . The concentration profiles of the four analysed DAPs in the hair of each patient are depicted in Figures 1–4. In order to facilitate the processing of results, all segments with non-detectable levels of DAPs were assigned the value of LOQ for each metabolite.
The mean ± SD and maximum levels (pg/mg) of each dialkyl phosphate metabolite (DMP, DEP, DETP and DEDTP) for all patients

SD: standard deviation, DAP: dialkyl phosphates, DEP: diethyl phosphate, DMP: dimethyl phosphate, DETP: diethyl thiophosphate, DEDTP: diethyl dithiophosphate.

Dialkyl phosphate metabolites (dimethyl phosphate [DMP], diethyl phosphate [DEP], diethyl thiophosphate [DETP] and diethyl dithiophosphate [DEDTP]) hair profile across the hair shaft from root to distal end for patient 1.

Dialkyl phosphate metabolites (dimethyl phosphate [DMP], diethyl phosphate [DEP], diethyl thiophosphate [DETP] and diethyl dithiophosphate [DEDTP]) hair profile across the hair shaft from root to distal end for patient 2.

Dialkyl phosphate metabolites (dimethyl phosphate [DMP], diethyl phosphate [DEP], diethyl thiophosphate [DETP] and diethyl dithiophosphate [DEDTP]) hair profile across the hair shaft from root to distal end for patient 3.
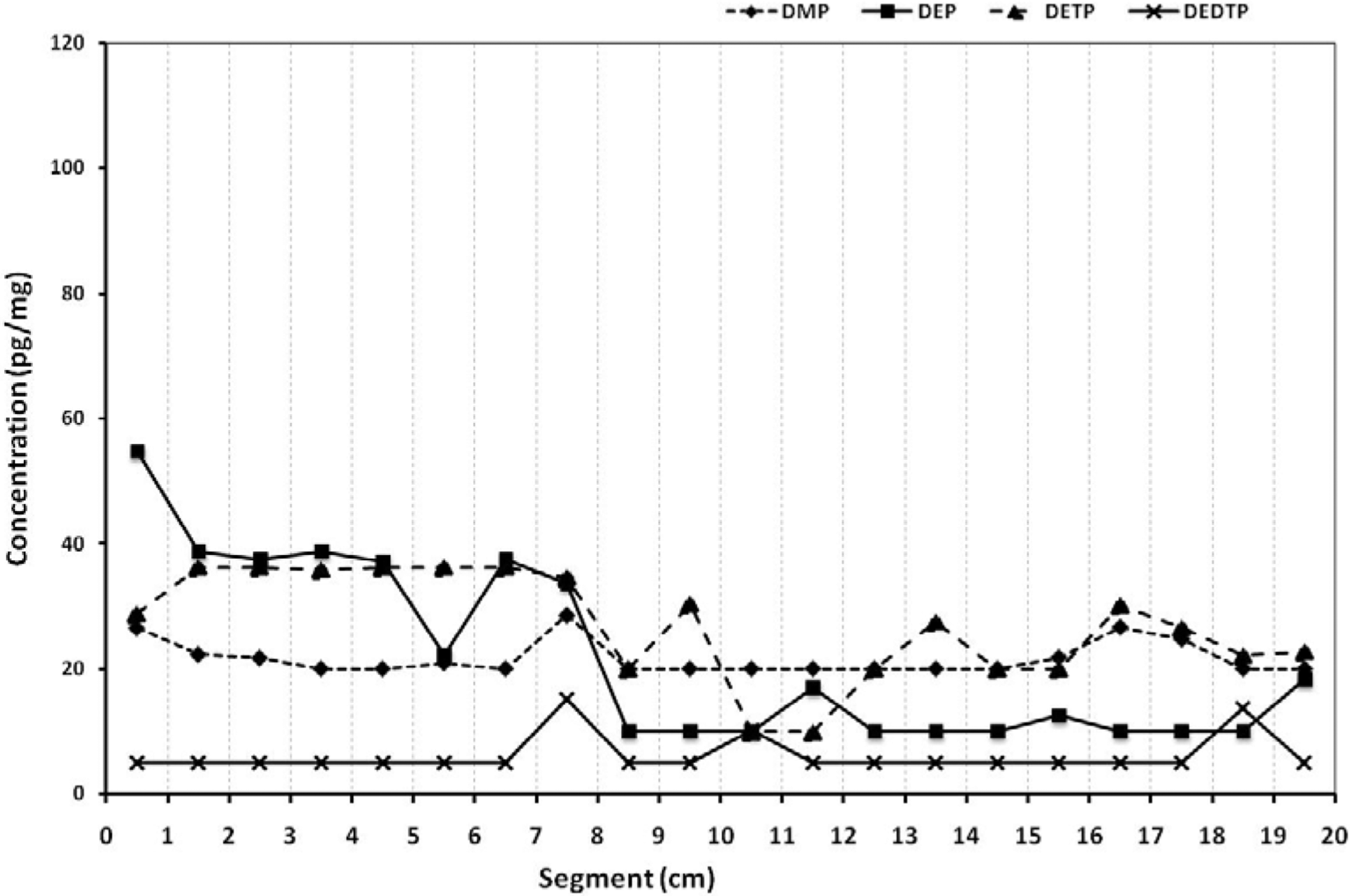
Dialkyl phosphate metabolites (dimethyl phosphate [DMP], diethyl phosphate [DEP], diethyl thiophosphate [DETP] and diethyl dithiophosphate [DEDTP]) hair profile across the hair shaft from root to distal end for patient 4.
In patient 1, the levels of DEP and DETP, the two metabolites produced by chlorpyrifos, peaked at the segment corresponding to the time of poisoning and were significantly different (p < 0.001) from the mean values. In patient 2 that was exposed to phenthoate there were no observable differences in the examined metabolites. Patient 3, after acute exposure to dimethoate, presented a peak of DMP in the segment corresponding to exposure. Patient 4, after exposure to a small quantity of chlorpyrifos, presented a peak of DEP at the segment corresponding to acute exposure.
Discussion
DAP metabolites are highly polar compounds, excreted mainly in the urine of the patient following exposure to OPs. Animal and human studies have shown that following exposure to OPs, 90% of the compound is eliminated in the urine between 6 and 24 hours after exposure. There is evidence though that after self-poisoning, patients may still be excreting detectable levels of metabolites up to 14 days later. 19 As a result, the metabolites are circulating in the blood stream and are deposited in the newly forming hair shaft at the hair follicle.
Monitoring of these non-specific metabolites in various biological matrices has been suggested for the assessment of low level exposure to OPs. Hair analysis for DAPs has been recently used as a bio-monitoring tool for assessment of low level cumulative exposure to OP pesticides, in a study conducted in our lab. 20 There are also studies that measured the levels of parent pesticides and their metabolites in the hair of rural populations and pregnant women in Philippines and have indicated that it is possible to use hair as a biomarker of exposure to these pesticides. 21,22
In the present article, the possibility of using DAPs hair analysis as a bioindicator of past acute exposure to organophosphorus pesticides is examined, bearing in mind that some pesticides give only certain DAPs and that maybe an increased concentration of these metabolites in a specific segment could correspond to a high acute exposure. Kintz and coworkers have illustrated that multi-sectional hair analysis may indicate a single exposure to benzodiazepines and rape drug. 23,24
Hair analysis for biomonitoring acutely exposed patients to pesticides is a completely novel topic. Several points were addressed for the method evaluation: (a) the detection of the produced metabolite of the parent compound, (b) possible peaks associated with time of ingestion and (c) possible correlations of observed peak(s) with dose of ingested parent compound.
In total four cases of patients that survived OP poisoning are presented. Patient 1 ingested 150 ml of chlorpyrifos solution and hair was obtained 2 months after the attempt. While the levels of DMP and DETP remain fairly constant across the hair shaft, the levels of DEP and DETP, the metabolites produced following chlorpyrifos exposure, peak approximately on the 1st to 3rd section, that correspond to 1–3 months after the attempt.
Patient 2 ingested 50 ml of phenthoate, a pesticide producing DMP. The fluctuation of the measured metabolites across the hair shaft does not present any peak. Patient 3, having ingested 60 ml of a dimethoate solution, presents a significant peak of DMP, on the 11th section of the hair shaft, the one that corresponds to the time of the attempt. Patient 4, ingested 15 ml of chlorpyrifos solution and hair was obtained 2 months after the attempt. A peak of DEP is detected on the second segment of the hair shaft, but this is not followed by a corresponding peak of DETP.
Overall, the results indicate that although DAPs are detected all along the hair shaft, they do give a peak in the segments that correspond to the high-dose acute exposure time.
The use of measuring a DAP metabolite profile across the hair shaft has two major advantages. First of all, it provides a good idea of the cumulative exposure of the individual to pesticides that produce these metabolites. Secondly, a peak of certain DAP in a segment that differs from the background levels of similar DAP could indicate an acute exposure to an OP pesticide. Conclusively measurements of DAPs in appropriate hair segment of OP-poisoned patients can be used for assessing past acute exposure to OPs in certain cases.
Footnotes
Acknowledgments
Authors would like to express their gratitude to Maravgakis George and Kavvalakis Matthaios, PhD candidates for their useful advices on the preparation of the manuscript.
The study was co-funded by the grants of Toxicology laboratory 2600, 2590 and 2598, University of Crete Research Committee.
