Abstract
Organophosphate (OP) poisoning continues to represent an important medical issue through its high prevalence among toxic pathologies and through its severity. In diagnosing this toxicological disorder, the most frequently utilized and available laboratory test remains the assessment of plasma cholinesterase – butyrylcholinesterase (BChE) – activity. Despite the reluctance of many researchers on the usefulness of serum BChE for kinetic analysis in OP intoxications, we have tested a recently proposed protocol, which is safe, non-expensive, easy to perform, appropriate to distinguish between an aged cholinesterase and a still reactivable one. Our aim was to validate the usefulness of this protocol, studying a series of 23 consecutive patients acutely intoxicated with OP, admitted in a regional Emergency Hospital, over a 1-year period. Introducing the proposed test in the routine of monitoring OP-intoxicated patients has resulted in the identification of a pattern with a funnel aspect, consequence of the initial possibility to increment the degree of BChE activity. This funnel shape defines the presence of reactivability, while its absence demonstrates the lack of obidoximes effect, due to cholinesterase’s ageing process. This method consisted in an advantage for the diagnosis, having the potential of improving prognostic evaluation and therapeutic orientation in OP intoxications.
Introduction
Organophosphate (OP) poisoning continues to be the leading cause of fatality among acute non-drug toxic pathologies, based on its high prevalence and its severity. 1 Despite the existence of efficient antidotes, the prognosis in this type of intoxication still remains reserved. 2
According to major clinical toxicology textbooks, the diagnosis of acute OP poisoning is suggested by history and physical findings and confirmed by a depressed serum or red blood cells cholinesterase (acetylcholinesterase) level. 3
However, measurement of acetylcholinesterase is not a standard practice. By contrast, determination of plasma butyrylcholinesterase (BChE) is a routine test for monitoring the benefit of oxime antidote therapy in OP poisoning.4–6
A limitation of this method results from the very wide interval of values for cholinesterase (ChEs) activity included in the range of normal. Due to this, in medical practice, a relatively low value may represent the normal, while a so-called normal value may be detected when inhibition of the cholinesterase activity is present.7–10
A newly described protocol capable to evaluate the reactivability of BChE activity level showed interesting perspectives for the diagnosis and optimization of antidote therapy compared with the simple detection of BChE level. 11
The aim of this study was to assess the usefulness of currently used test of BChE activity, modified according to the protocol mentioned above, for the diagnosis and management of OP acute poisoning.
Methods
Subject selection
In this study, serum samples from 23 consecutive patients, admitted with a diagnosis of OP acute poisoning in “Sf.Ioan” Emergency Clinic Hospital, Iasi (a regional Emergency Hospital which assist poisonings in North-East Romania and have a strong expertise in diagnosis and management of OP intoxications), over 1-year period has used.
Inclusion criteria
Diagnosis of acute OP poisoning and patients aged above 18 years.
Exclusion criteria
Age less than 18 years, mixed poisonings, refusing of patient or patient’s family to sign the informed consent, associated chronic therapies with ibuprofen, procainamide, phenazopyridine or L-Dopa (which are substances known to interfere with the spectrophotometric method of detection of the levels of activity of BChE used in the study). 12
Method
In our hospital, the method used to determine the level of BChE activity is a colorimetric technique, and relay on a Vitros System Chemistry 5,1/FS. In this method 11 mL drop of sample are evenly distributed on a dry, multilayered and analytical element coated type of slide. BChE brought in the reaction from the sample will hydrolyze butyrylthiocholine to thiocholine. The liberated thiocholine will reduce potassium hexacyanoferrate II, as shown below:

The rate of color loss is monitored by reflectance spectrophotometry (400 nm wavelength, multiple point rate test type) and is proportional to the amount of cholinesterase activity present in the sample. Reference interval is 4.65–10.44 U/ml (females) and 5.90–12.22 U/ml (males).
The modified protocol 11 used determined the level of BChE activity on 2 samples of 0.5 ml serum obtained from the same blood specimen. One sample was analyzed without any preparation, using the conventional method, and the second, after a preliminary incubation with Toxogonin®. The mixture was kept half an hour at 37°C on a water thermostat – to reproduce, at least at minimum, the conditions within the human body. We have diluted 0.5 ml Toxogonin® (from the 1 ml ampoule manufactured by Merck Pharma GmbH, containing as active ingredient – obidoxime chloride, 250 mg/ml) with 4 and 5 ml NaCl 0.9%. Five micro liters of this 1/10 dilution contains 0.125 mg Toxogonin®.
The blood samples were centrifuged immediately after collection. Serum was separated from the clot after centrifugation (technical specifications of the kit producer being an interval of maximum 4 hours) and processed after a maximal period of 24 hours (the kit producer recommendations are less than 7 days), while stored in refrigerator (at 2–8°C, never frozen).13–15
The dilution was constituted at the moment of inclusion in the study for each patient, and used for all the tests of reactivability performed in that patient, at different moments during the week of monitoring. The prepared dilution was stored in a brown glass bottle. 11
Every patient included in the study was evaluated in 14 different moments during the first week of hospitalization: on admission, at three moments of the day (7 a.m., 3 p.m. and 11 p.m.) in the first 72 hours, and at 7 a.m. during the following 4 days. These relatively fixed moments were adopted in order to allow comparative analyses of the dynamic in BChE reactivability. No modification was made in the therapeutic approach due to the study. Atropinization therapeutic effect was defined by a heart rate of 90–110 beats/min, dry skin and midriasis.
All aspect of the clinical study were made with regard to the bioethics rules endorsed by European Union legislation: Directive of the EU Council no. 609 from 24 November 1986, recommendations from the “Declaration of Helsinki”; recommendations from EU Directive 2001/20/CE, as well as the Romanian regulations for good clinical practice and clinical trials mentioned in Law 95/2006 (Chapter/Title “Medicine”), OMSP 904/28.07.2006, OMSP 906/28.07.2006. The study protocols have the approval of the Bioethics Commission of the “Grigore T. Popa” University of Medicine and Pharmacy Iasi and of the “Sf. Ioan” Emergency Clinic Hospital Iasi.
Statistical analysis
Comparisons of BChE levels of activity at each time between the two methods used were assessed with
Results
We studied 23 consecutive patients poisoned with OP pesticides, 9 males aged 38.7 ± 17.4 and 14 females aged 39.6 ± 18.7 years. The route of entry of toxin was digestive after ingestion in suicide attempt (20 patients) or after involuntary ingestion (3 patients). The quantity of ingested toxin was difficult to estimate in all patients, caused by lack of accurate information provided by patients, patient’s families or Emergency Medical Services personnel. We have approximated that the amount of toxin involved was between a minimum of 15 ml and a maximum of 400 ml. We have confirmation about the specific OP involved in just three cases and these were Neguvon (two subjects) and Furadan (one subject). Demographic, clinical parameters and outcome of patients included in study are represented in Table 1.
Demographic, clinical symptoms and outcome of patients included in study

All patients presented moderate or severe types of toxicity based on clinical signs and symptoms. Diagnosis of acute OP poisoning was suggested by history and physical exam, and confirmed by a decreased level of BChE activity, as well as by a positive atropine tolerance test.
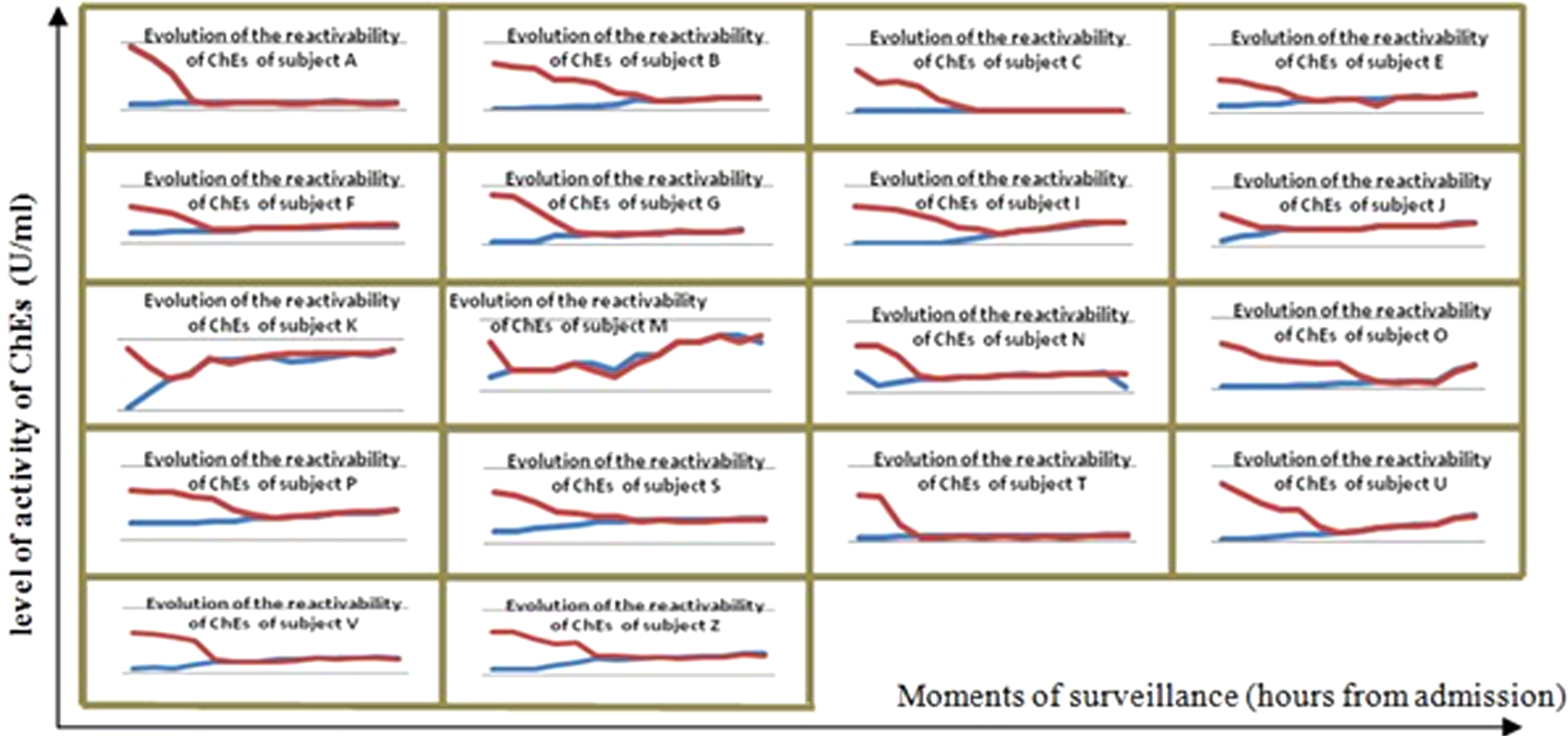
Evolution of the BChE reactivability assessed by 7 days monitoring of BChE levels of activity (determined using the conventional method and the Toxogonin®-sensitized protocol) is presented in Table 2 and Figures 1 and 2.
Evolution of the reactivability of cholinesterase (ChEs) as suggested by a 7 days monitoring of the levels of ChEs activity

Abbreviations: Cm, conventional method; Tm, Tox-serum incubation method; ns, without statistical significance.
a Letters A to Z were assigned to the patients at the inclusion in the study.
Evolution of butyrylcholinesterase

Lack of Tox-induced butyrylcholinesterase
All patients were treated with aggressive supportive care and antidotal therapy with a favorable outcome in 22 of 23 subjects and were discharged from the hospital after 8.5 ± 2.3 days.
The deceased patient (subject T) was a 43-year-old male with a history of severe ethylic psychiatric disorder and liver cirrhosis presenting at admission in the hospital with a profound coma (Glasgow Coma Score 5) and aspiration bronchopneumonia.
Discussion
The toxic mechanism of OP is based on their interaction and irreversible inhibition of acetylcholinesterase, BChE, carboxylesterases etc. by phosphorylation and phosphonylation of their active site serine. Inhibition of acetylcholinesterase is responsible for the characteristic symptoms of OP poisoning.4,16 Therapeutic strategies are directed to competitively antagonize overstimulation of muscarinic receptors by atropine, and to reverse ChEs inhibition by reactivation with oximes such as obidoxime and pralidoxime. Oximes should be administered in appropriate dosage as early as possible and effective concentrations should be maintained as long as the reactivation is possible. 17
Despite the reluctance of some researchers on the usefulness of serum BChE for kinetic analysis in OP intoxications, it is largely accepted that the practical utility of a correct identification of cholinesterases behavior regarding oximes therapy, both in human and in experimental studies.4,18
In this view, efforts for conceiving new protocols on ChEs through obidoxime-induced reactivability testing are reasonable to be undertaken and they will associate practical value, if they consist in affordable modifications to an already existing laboratory methodology. This is especially the case of developing countries, where investments in new medical technologies are not always easy to afford, these countries being also those where the prevalence of OP intoxication is the highest.
In diagnosing OP poisoning, the most frequently utilized laboratory test remains the quantitative assessment of BChE activity through its effect on butyrylthiocholine as substrate, using a colorimetric evaluation.
Toxogonin®-sensitized method is based on the reactivatory effect of Toxogonin®, which increases the level of ChEs activity, when OP blocks the enzyme. According to the literature, this effect is possible only in the initial period after intoxication.16,17 Shortly after, OP blocks irreversibly the enzyme, through an “ageing process,” a dealkylation of the complex cholinesterase – organophosphate. During this interval, generally the first 48–72 hours after intoxication, obidoximes are efficient in recovering cholinesterases from their complexes with OP. This period could be longer if the absorption still continues – i.e. additional ignored sites where absorption of poison persist (head hair, fat tissue, entero-enteric circuits, etc.) – or shorter (depending on the type of involved organophosphate, modalities in which the poisoning was produced, etc.).19–21
An advantage of this test is the possibility to differentiate if the enzyme is still responsive or not to Toxogonin® and constitutes a justification for the continuation of Toxogonin® treatment, an expensive and even harmful one if the cholinesterases complex is ‘‘aged” (instead of unbinding the OP from the enzyme by linking with it, Toxogonin® will bind the enzyme, decreasing even more its level of activity). 22
We consider as strong point of our research the fact that we have tested a protocol easy to implement in the majority of medical emergencies institutions, being based on a method that is already widely used. Our research was a prospective, randomized clinical study undertaken on serum from human subjects, intoxication being caused by OP agents more close to day-to-day reality, not by nerve agents OP. Even if OP was not clearly identified in all the cases, they all belong to the common group used as pesticides, which are more currently involved, as individual poisonings, in the pathologies of an emergency department. Also, another strong point is the fact that the research did not use BChE after being frozen (at –80°C), as the technical specification of the kit producer clearly underlines.
Studying, the evolution of the reactivability of BChE, we have established, in 18 from 23 cases of OP poisoning, a pattern with the aspect of a funnel, consequence of the initial possibility to increment the degree of BChE activity, due to Toxogonin® reactivatory effect (Figure 1). Statistical analysis showed that in 15 of 18 cases with this pattern of reactivation of BChE, data obtained were significant (
The isthmus of the funnel shape indicates from which point further BChE is no longer reactivable through Toxogonin®. As observed in Figure 3(representing, due to considerations of visibility in the graphic, only the subjects – A, B, C, E and F), the loss of BChE Toxogonin®-induced reactivability does not appear in a predetermined moment of the intoxication. For all 23 subjects included in study, the documented extremes were first 8 hours in subject M versus 64 hours in subject Z. Thus, this important moment in the evolution of the OP poisoning cannot be predicted other than by monitoring the subjects with the Toxogonin®-sensitized method. Due to the necessity for an easiness of application and a clear study design, the graphical representation in Figure 3 have used as starting point the moment of admission and not the moment of intoxication, as it has been more scientifically rigorous. Despite this, the exposed conclusion was not significantly affected because the general influence of time between ingestion and the admission to hospital was minimal (99 ± 85 min—see Table 1) in the studied series of cases.

Evolution of the reactivability of butyrylcholinesterase
In conclusion, in case of a suspected OP poisoning, an important new perspective to the diagnostic evaluation and management of antidote therapy appears to be assessment of BChE reactivability, as compared with only the simple detection of the BChE activity.
In a specific “in vitro” testing protocol, adding Toxogonin® to the serum sample will generate a recovery of the BChE activity in circumstances of acute OP poisoning.
A Toxogonin®-induced reactivability of BChE activity, highlighted with an “in vitro” test will add more trust to the practitioner that he is facing the correct diagnosis and will have the potential for a better quantification of the severity, and prognosis for this toxic situation.
Analyzing ‘in vitro’ the effect of Toxogonin® on restoring the level of BChE activity resulted in a safe, non-expensive, easy-to-perform, quick (<1 hour) test capable to offer new, interesting perspectives on organophosphate intoxications.
Footnotes
We are grateful to the ‘‘Grigore T. Popa” University of Medicine and Pharmacy Iaşi (grant no. 6820 obtained in 2009 competition) for funding this study under the theme “New original in vitro method of assessing cholinesterase reactivability for optimizing the antidote treatment in organophosphate poisoning”.
