Abstract
Aim: To assess the systemic inflammatory response (SIR) and the multi-organ damage after large-volume liver radiofrequency ablation (RFA) with or without concurrent Pringle maneuver. Methods: Wistar rats were subjected to 30% liver RFA (group RFA), liver RFA under 30-min Pringle maneuver (group RFA + P), Pringle only (group P) or sham operation (group S). Serum levels of interleukin-1α (IL-1α), interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α), serum biochemical profile, multiple-organ pathology and the activity of nuclear factor-κB (NF-κB) in the liver were assessed post-operatively. Results: The levels of IL-6 and TNF-α were increased from 1h up to 1w and 6h, respectively, in both RFA groups, while IL-6 was only mildly increased at 3 h in group P. IL-6 was higher in group RFA + P compared to group RFA. Serum biochemical profile was altered more intensely in group RFA + P compared to RFA. There was tissue injury in the non-ablated liver portion as well as in adjacent and remote organs with lesions being more severe in group RFA + P. At 1 h, NF-κB was equally activated in all study groups. Conclusions: Extended liver RFA causes SIR and multi-organ injury, which are exacerbated when a concurrent Pringle maneuver is applied.
Introduction
Radiofrequency ablation (RFA), a local thermoablative method, is used for the destruction of primary and metastatic unresectable hepatic tumors. RFA is considered an effective and safe method with relatively low mortality (0.1%–4.5%) and complication rates (2.2%–9.9%).1–4 However, the application of the method on extensive liver mass has been recognized as a risk factor for the occurrence of post-RFA complications and the post-ablation syndrome.1,4–6 Furthermore, the subsequent need for performing multiple RFA cycles for the complete ablation of large volume- or multifocal hepatic tumors increases the risk of local recurrence due to electrode track seeding.4,5 So far, both animal and clinical studies have yielded controversial results on whether RFA applied on extensive liver mass (>30%) triggers a systemic inflammatory response (SIR)7–10 and/or causes remote organ injury.7,9,10
In clinical practice, a way to minimize the number of RFA sessions required for the complete ablation of a hepatic tumor, at the open-surgery or laparoscopic approach, is by concurrently applying the Pringle maneuver (temporary occlusion of both hepatic artery and portal vein). It has been shown that when hepatic blood flow is decreased thereby reducing liver tissue perfusion, the diameter of the ablated liver mass per cycle is increased.11–14 However, ischemia-reperfusion (I/R) of the liver has been shown to increase the production of proinflammatory cytokines15–17 and cause liver18–20 and remote organ injury.18,19,21,22
The aim of the present study was to assess the SIR and injury of adjacent and remote to the liver organs after large-volume liver RFA with or without the concurrent application of Pringle maneuver.
Materials and methods
Animals
A total of 168 Wistar rats, weighing 250–300 g, provided from our laboratory’s rat colony, were used. They were housed in macrolon cages, three rats per cage, at 20–22°C room temperature, on a 12-hour light:12-hour dark cycle and were provided with commercial pelleted diet and tap water ad libitum. The facilities were in accordance with Directive 86/609/EEC.
Experimental design
The animals were randomly assigned to 4 groups of 42 rats each. They were subjected to a single RFA cycle of the right median hepatic lobe (approximately 28.5% of total liver volume)
23
after midline laparotomy under general anesthesia (group RFA), RFA under 30-min Pringle maneuver (group RFA + P), Pringle alone (group P) or sham operation (group S). At various timepoints post-operation (1 h, 3 h, 6 h, 24 h, 48 h and 1 w), the levels of interleukin-1α (IL-1α), interleukin-6 (IL-6) and tumor necrosis factor α (TNF-α) and a series of biochemical parameters indicative of vital and other organs function (at 30 min, 24 h, 48 h and 1 w) were measured in blood serum. At 1 h, liver tissue specimens were excised from the non-ablated liver portion for immunohistochemical assessment of nuclear factor
Operation
The animals were anesthetized by administration of the inhaled anesthetic sevoflurane (3% in oxygen) provided by a face mask. A self-adhesive gelled grounding pad was placed at the shaved back of each animal (groups RFA and RFA + P). The surgical field was properly prepared and a midline laparotomy was performed under aseptic conditions. The median hepatic lobe was exposed and an RFA session was performed to the right median lobe. In groups RFA + P and P, a 30-min Pringle maneuver was applied by placement of a microbulldog clamp across the hepatoduodenal ligament (which includes the hepatic artery, the portal vein and the hepatic duct). In group S, a 30-min sham operation, which involved the mobilization of the median hepatic lobe after midline laparotomy, was performed. The abdominal wall was finally closed in layers using Dexon 3-0 suture.
RFA
A Radionics Cool-tip Radiofrequency Ablation System (Tyco Healthcare, Boulder, CO, USA) consisting of a radiofrequency generator, a peristaltic perfusion pump, a grounding pad and a single-shaft 15-cm long needle electrode with a 2-cm exposure tip was used. The tip of the electrode was inserted into the hepatic parenchyma from the median surface of the right median lobe at a 90° angle. The power delivered was 10 W at a current amp 0.20 A for 2 min (group RFA) or 1 min and 15 sec (group RFA + P). The duration of RFA cycles under Pringle maneuver was determined in pilot studies so that the liver ablated area did not differ from that in group RFA. During RFA sessions, the tip of the electrode was cooled by continuous perfusion of cold normal saline delivered by the peristaltic perfusion pump.
Blood serum biochemical analysis
Venous blood samples were collected from the caudal vena cava after midline laparotomy. Blood serum was separated by centrifugation at 3000 g for 20 min and then stored at –70°C until later analyzed. The concentrations of IL-1α, IL-6 and TNF-α were measured using commercially available ELISA kits (Thermo Scientific-Pierce Biotechnology/Rockford, IL, USA) according to the manufacturers' instructions. The concentration of the following biochemical parameters were measured using an automated analyser (ILAB 600 analyser/Instrumentation Laboratory SpA, Milano, Italy); urea, creatinine, aspartate aminotrasferase (AST), alanine aminotransferase (ALT), total bilirubin, lactate dehydrogenase (LDH), γ-glutamine transferase (γ-GT) and amylase.
Histopathology—immunohistochemistry
Tissue specimens excised from the non-ablated liver portion, pancreas, spleen, kidneys and lungs were fixed in formalin and embedded in paraffin according to standard procedures. Histopathologic examination was performed in 4 μm hematoxylin-eosin stained sections. The histopathologic endpoints evaluated in each organ tissue are presented in Table 1. The severity of lesions noted was quantified according to the following scoring system: 0, none; 1, mild; 2, moderate; 3, severe. Lesion severity scores were added to obtain the histopathologic score for each organ tissue. Analysis was performed by two independent pathologists (ML, NP) who were blinded to the clinical and pathologic information and the average scores were calculated.
Histopathologic endpoints evaluated in each organ tissue

Four-micron sections of representative blocks from the paraffin embedded liver tissue specimens were deparaffinized, rehydrated and immunopositivity was detected employing the En Vision protocol (DakoCytomation, Carpinteria, CA, USA). To assess the expression of activated NF-kB in the liver tissue, a mouse monoclonal antibody against NF-kB p65 subunit (Chemicon International Inc, Temecula, CA, USA) was used. Microwave irradiation in 0.01 M citric buffer (pH 6.0) was performed as antigen retrieval method in all specimens. Slides were then incubated for 60 min with the NF-kB mouse monoclonal antibody at a 1:50 dilution. Control slides were incubated for the same period with non-immunized serum (negative control). A positive control was also included in the assay. Bound antibody complexes were stained for 10 min with 0.05% diaminobenzidine. Sections were then counterstained with Mayer’s haematoxylin, mounted and examined under a Nikon Eclipse 50i light microscope (Nikon/Tokyo, Japan) at ×200 magnification. NF-kB expression in each immunostained section was evaluated based on the number of NF-kB positive cells and was classified according to the following four-scale grading system: 0, no cell; 1, sparse cells; 2, few cells; 3, many cells. NF-kB expression was evaluated in 10 optical fields and the total score was divided by the number of optical fields to obtain the mean activated NF-kB expression score.
Statistical analysis
Data were subjected to one-way analysis of variance (ANOVA) followed by the Bonferonni test for comparisons between groups. A probability of less than 5% (p < 0.05) was considered to be statistically significant.
Results
Mortality
All animals survived the experimental period.
Serum cytokines
IL-1α
No changes were noted in IL-1α serum levels in all groups studied (Figure 1A).

Serum levels (means, n = 6) of (A) interleukin
IL-6
In groups RFA and RFA + P, IL-6 serum levels were increased throughout the study period (1 h–1 w) compared to sham operated animals. In group RFA + P, the levels were significantly higher than those in group RFA from 3-h to 48-h post-ablation; a peak was noted at 3 h, while, thereafter, levels were gradually decreased until 1 w. In group P, only a moderate increase was noted at 3-h post-operation (Figure 1B).
TNF-α
TNF-α serum levels were increased in groups RFA and RFA + P from 1 h to 6 h compared to sham-operated animals. Peak levels were observed at 6 h in group RFA, while at 3 h in group RFA + P. At 6-h post-ablation, TNF-α concentration in group RFA was significantly higher than that in group RFA + P (Figure 1C).
Blood serum biochemistry
In groups RFA and RFA + P, the levels of AST, ALT, bilirubin, LDH and γ-GT were increased by 30 min (AST, ALT, LDH) or 24 h (bilirubin, γ-GT) post-ablation. At 1 w, ALT, LDH and γ-GT levels returned to normal. The levels in group RFA + P, although higher, were not significantly different from those in group RFA. In group RFA + P, a transient increase was also noted in urea at 24 h (non-significant) and amylase concentration at 30-min post-ablation (Table 2). In group P, no alterations in the biochemical profile were noted apart from a transient increase in LDH concentration at 30-min post-operation (Table 2).
Blood serum biochemical profile after liver RFA (group RFA), liver RFA under Pringle (group RFA + P), Pringle alone (group P) or sham operation (group S)

AST, aspartate aminotrasferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; γ-GT, γ-glutamine transferase; RFA, radiofrequency ablation; Means (standard deviation).
a For each timepoint p < 0.01 vs groups P and S.
b For each timepoint p < 0.05 vs groups P and S.
c For each timepoint p < 0.05 vs other groups.
NF-κB activity
Activated NF-kB expression score was significantly increased (p < 0.01) in hepatocytes in all study groups (mean ± SD, group RFA, 1.40 ± 0.55; group RFA + P, 1.80 ± 0.45; group P, 1.83 ± 0.41) compared to sham operated animals (0.30 ± 0.25). No differences were noted among the study groups (Figure 2).
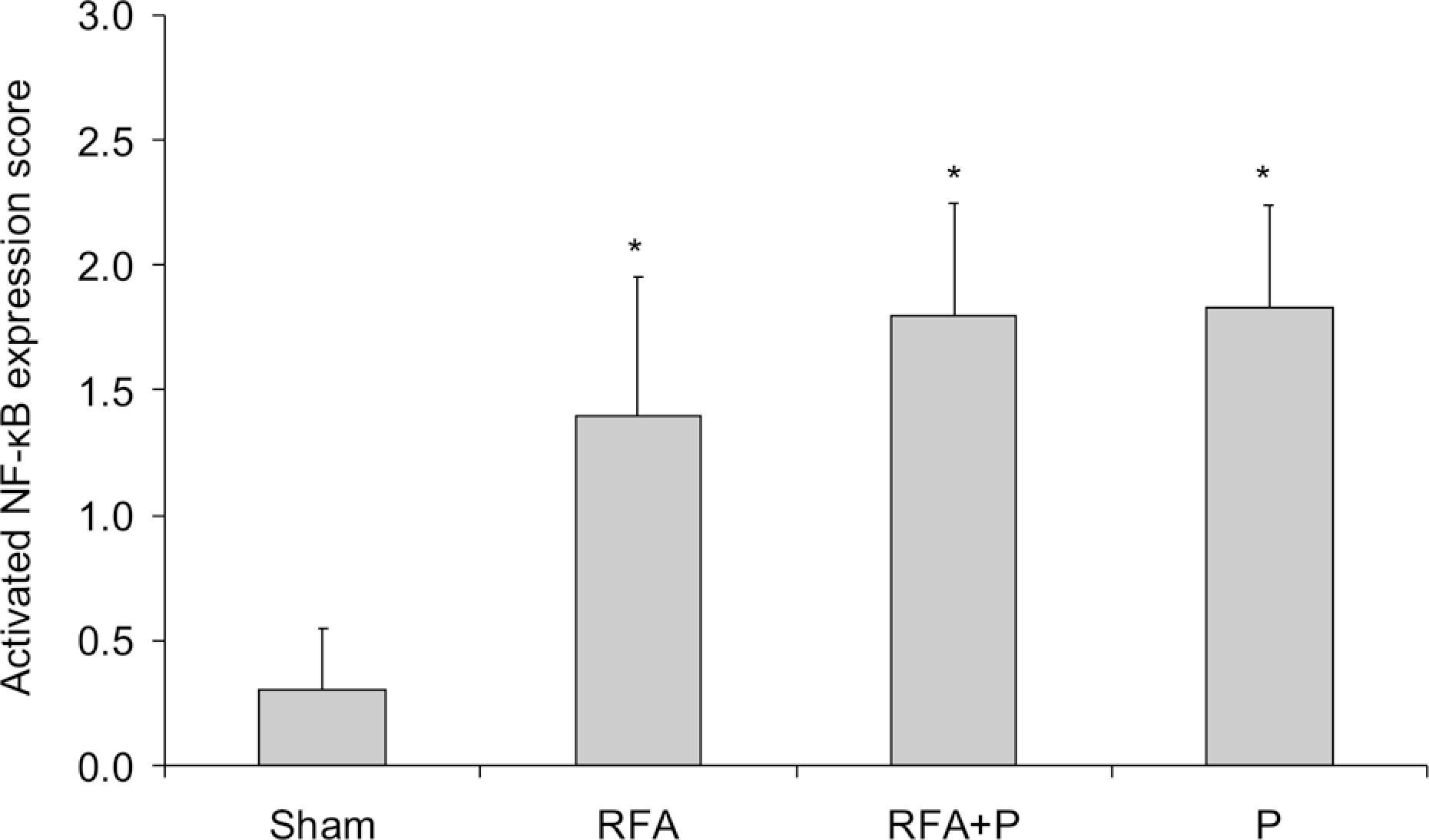
Activated nuclear factor-κB (NF-kB) expression score (means, n = 6) in hepatocytes after liver radiofrequency ablation (group RFA), liver RFA under Pringle (group RFA + P), Pringle alone (group P) or sham operation (group S). * p < 0.01 vs group S.
Organ pathology
In group RFA, histopathologic score was increased in all organs studied from 24 h to 48 h (liver) or 1 w (pancreas, spleen, lungs and kidneys) with a peak at 48-h post-ablation. In group RFA + P, tissue damage was evident by 30-min up to 1-w post-ablation. Lesion severity was higher in group RFA + P than that in group RFA and peaked at 48 h (liver, spleen and kidneys) or 1 w (lungs). A substantial deterioration in pancreatic tissue pathology was noted by 30-min post-ablation. In group P, histopathologic score was increased at all time points studied. Lesion severity was similar or lower than that seen in group RFA + P (Figure 3).

Liver, spleen, pancreas, kidney and lung histopathologic scores (means, n = 6) after liver radiofrequency ablation (group RFA), liver RFA under Pringle (group RFA + P), Pringle alone (group P) or sham operation (group S). a-d For each timepoint, means with different letters differ significantly (p < 0.05).
Discussion
The present experimental study provides evidence that extended liver RFA increases proinflammatory cytokine production and causes multiple-organ injury. Furthermore, the concurrent application of Pringle maneuver exacerbates SIR and organ damage. This effect was related to aggravation of hepatic tissue injury in the non-ablated liver portion.
With growing experience, RFA is applied to progressively larger hepatic tumors. There are reports of patients undergone RFA on liver volume exceeding 3000 ml. 24 However, extended liver RFA has been recognized as a risk factor for the occurrence of post-RFA complications.1,4–6 The fact that these may involve the dysfunction of organs, such as the lungs, the heart and the kidneys, suggests damage of remote organs. Furthermore, the volume of necrosis produced by RFA affects the incidence and duration of the post-ablation syndrome, which is characterized by fever, malaise, chills, pain and nausea and lasts 2–7 days but may be prolonged up to 3 weeks.4,25
It is not clear whether extended liver RFA induces SIR. In a clinical study, no increase of cytokine production was noted in patients subjected to open RFA of up to 58% of their liver mass. 8 On the other hand, there are recent reports of IL-6 serum level elevation by the early post-RFA period26,27; it should be noted, though, that a number of patients enrolled in these studies had been subjected to concurrent Pringle maneuver of variable duration, complicating the interpretation of the results. Experimental studies have also given controversial results. RFA on 30% of liver mass in a porcine model 9 and 50% or 60% in a rat model, 10 initiated early SIR, while other researchers have reported no change in TNF-α serum levels during the first 6 h after 35% liver RFA in rats. 7 According to the results of our study, RFA on approximately 30% of rat liver volume led to a sustained increase of IL-6 serum levels from the first hour post-ablation until the end of the 1-week follow-up period and an elevation of TNF-α levels during the first 6 h.
So far, experimental studies have yielded conflicting results on whether extended liver RFA causes lung injury9,10 or not, 7 while there were no histopathologic findings in the renal and the non-ablated portion of liver tissue.9,10 According to our results, 30% liver RFA caused damage to both adjacent (pancreas, spleen) and distant to the liver organs (kidneys, lungs) which peaked mostly at 48-h post-ablation. Thereafter, histopathologic findings, although their severity tended to decrease, were still present at 1-week post-ablation. A striking deterioration of pancreatic tissue histopathologic profile by 30-min post-ablation underscored the role of potential direct thermal injury in organs adjacent to the liver. Multi-organ injury was associated with damage of hepatic tissue of the non-ablated liver portion characterized by activation of NF-κB in hepatocytes and accompanied by a rise in serum proinflammatory cytokines and transaminases. However, one must not rule out the possibility that the necrotic ablated tissue that remained in situ might have acted as a cytokine production site itself.
Hepatic Kupffer cells have been shown to play an important role in the inflammatory response secondary to hepatic injury by producing oxidants and cytokines. The cascade of events of liver injury caused by I/R has been well-described in two distinct phases. An initial phase involves the release of reactive oxygen species by activated Kupffer cells 28 and the activation of NF-κB. 29 At a second phase, Kupffer cells and hepatocytes produce proinflammatory cytokines, such as TNF-α, which initiates a mediator cascade leading to hepatic neutrophil recruitment.30,31 TNF-α upregulates the expression of intercellular adhesion molecule-1 (ICAM-1) on hepatic vascular endothelium 32 and neutrophil- attracting ELR-positive CXC chemokines in liver parenchyma 33 which promote the recruitment of hepatic neutrophils 30 ; then, hepatic tissue injury takes place by release of reactive oxygen species and proteases. 34 Transient occlusion of hepatic artery and portal vein blood flow may be applied during liver RFA to improve its efficacy. This is achieved by hepatoduodenal ligament clamping (Pringle maneuver) at the open or laparoscopic approach, or alternatively by selective angiographic baloon occlusion or embolization during transdermal RFA. 35 By this manipulation the ablated liver volume per cycle is increased and hence the number of RFA cycles necessary to effectively ablate a tumor is reduced11–14; in this way the risk for tumor seeding due to repeated electrode insertion is limited. Furthermore, transient liver blood flow reduction minimizes the heat sink effect. The latter phenomenon may be encountered in cases of RFA on tumors adjacent to major hepatic vessels and results to inadequate ablation of perivascular tissue due to perfusion-mediating tissue cooling.36,37
Two points had to be considered during the design of our study with regard to Pringle maneuver. Since a single RFA cycle lasting 2 min ablates around 30% of rat liver, 38 the duration of Pringle maneuver had to be extended to simulate clinical conditions in which multiple or large size tumors have to be destructed by repeated overlapping RFA cycles. A standard period of 30 min Pringle maneuver was selected. Second, in order to study the effect of Pringle maneuver per se on the RFA-induced side effects we had to exclude the possibility that these were due to ablation of a larger hepatic volume. For this reason, we defined in pilot experiments the time needed to ablate equal liver volumes with (1 min 15 sec) or without Pringle maneuver (2 min).
The performance of liver RFA under Pringle maneuver led to exacerbation of SIR and multi-organ injury compared to the RFA group. Systemic inflammatory response was characterized by a multifold increase of serum IL-6 levels that peaked at 3-h post-ablation. Despite the important role of TNF-α in the molecular mechanism of liver and remote organ injury after a hepatic I/R insult, no significant differences were detected in TNF-α serum levels between animals subjected to RFA or RFA plus Pringle apart from a 3-h shift in peak levels. On the other hand, aggravation of organ injury was related to a multifold increase of IL-6 serum levels which emerged as a SIR marker predicting multi-organ damage. Interleukin-6 possesses both pro- and anti-inflammatory properties. While it up-regulates the production of acute-phase proteins in the liver, 39 it has been shown to be hepatoprotective against liver I/R-induced inflammation by downregulation of TNF-α production. 40
The histopathologic profiles of adjacent as well as distant to the liver organs were deteriorated in group RFA plus Pringle compared to RFA only. It is of note that while severity of histopathologic lesions, after a peak, tended to decrease by 1-week post-ablation, in pulmonary tissue these were gradually increased. Aggravation of organ pathology was related to deterioration of histopathologic lesions severity in the non-ablated liver portion highlighting the role of hepatic tissue pathology on remote organ injury. Our histopathologic findings on liver tissue are in contrast with the only available relevant study in which biopsies from the viable hepatic parenchyma after RFA under Pringle maneuver in the pig revealed normal architecture and cell viability. 14 This difference could be attributed to the smaller size of the rat liver, which may have contributed in the intensification of the non-ablated liver tissue damage, especially when combined with concurrent liver blood flow occlusion.
At the early period following liver I/R, activation of NF-κB takes place20,29,31; this is considered the primary transcriptional mechanism for the production of proinflammatory cytokines. 15 According to our immunohistochemical analysis, NF-κB was activated in hepatocytes at 1-h post-operation in all study groups (RFA, RFA plus Pringle and Pringle alone). Nevertheless, its expression did not differ among groups suggesting that, at this phase, NF-κB activation did not determine the magnitude of SIR and that of subsequent organ damage.
The side effects noted in the RFA plus Pringle group were only partially attributed to I/R insult of the liver. In terms of SIR, 30 min of Pringle maneuver led to only mild increase of IL-6 serum levels at 3-h post-operation, while no change was observed in TNF-α and IL-1α concentrations. In contrast, previous studies report increased cytokine production, including TNF-α and IL-1β, after 30 min 19 or longer periods of liver ischemia (45 min or more) using rodent models.16,21 In addition, although there were histopathologic lesions in all organs studied, their severity was lower than that in the RFA plus Pringle group with the exception of kidneys; renal tissue was equally damaged in both groups. In other animal studies, considerably longer periods of liver ischemia (90 min or longer) caused liver, lung, heart and intestinal tissue damage.18,21,22 Lung injury was attributed to the upregulation of local expression of liver-derived TNF-α, which promotes the production of neutrophil attracting CXC chemokines and ICAM-1.31,41
Medical surgeons use Pringle maneuver in order to improve the efficacy of liver RFA especially in cases of large-volume or multifocal hepatic tumors or those adjacent to large hepatic blood vessels. Our experimental work provides evidence that this manipulation may compromise the safety of the method in terms of SIR and multi-organ injury. According to our results, hepatic tissue injury in the non-ablated liver portion was recognized as a key event that triggered SIR and led to subsequent development of adjacent and remote organ damage after extended liver RFA with or without concurrent Pringle maneuver. Aggravation of multi-organ damage was associated with the severity of non-ablated liver injury, which was increased when RFA was combined with liver I/R insult conferred by concurrent Pringle maneuver. Furthermore, increased IL-6 serum levels emerged as a SIR marker of the severity of organ injury. We believe that further elucidation of the pathogenetic mechanism involved in this phenomenon may prove useful in designing preventive strategies and hence widen the safety limits of this method.
Footnotes
The authors state that there is no conflict of interest.
The authors confirm that this research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
