Abstract
Aim:
Liver radiofrequency ablation (RFA) has been shown to disrupt the mechanical component of the gut barrier. The aim of the present study was to investigate the consequences of liver RFA on the biological gut barrier in terms of the effects of bile production rate and bowel inflammatory state on intestinal microflora balance.
Method:
A total of 25 New Zealand rabbits were assigned to five groups (n = 5 per group): group CBD: subjected to common bile duct (CBD) extracorporeal bypass; group CBD-RFA: subjected to CBD bypass plus one session of open liver RFA; group RFA: subjected to liver RFA; group sham: subjected to sham operation; and group TBD: subjected to total bile deviation (TBD). In groups CBD and CBD-RFA, bile production rate was assessed for 48 h. In groups sham and RFA, measurement of biliary glycine conjugates of cholic and deoxycholic acid levels, histopathologic examination of the non-ablated liver tissue, morphometric analysis, and histopathologic examination of the terminal ileum and microbiological analysis of fecal and tissue samples collected from the jejunum and the cecum (and in group TBD) were performed at 48 h post-operation.
Results:
One session of liver RFA resulted in ablation of 18.7 ± 2.7% of liver weight. Following liver RFA, bile production rate was reduced, while the levels of biliary bile salts were not affected. There was mild injury of the non-ablated liver parenchyma, mild intestinal wall inflammation, intestinal mucosa atrophy, and intestinal microbial population overgrowth.
Conclusion:
Reduced in bile production and mild bowel inflammation secondary to liver RFA impaired the biological gut barrier as manifested by intestinal microflora imbalance.
Introduction
Radiofrequency ablation (RFA) is a contemporary method applied for the local destruction of primary and metastatic liver tumors. Although the method is considered safe, there is a small incidence (2.4–4.6%) of post-RFA complications of potentially microbial origin that may pose a life-threat to treated patients. These include hepatic and perihepatic abscesses, liver failure, peritoneal infection, unspecified sepsis, pleural effusion, pneumonia, acute respiratory distress syndrome, renal failure, fever, and the post-ablation syndrome. 1 –4 Infectious complications in other pathologic entities, such as hemorrhagic shock, trauma, acute pancreatitis, burn injury, and obstructive jaundice, have been related to migration of bacteria from the intestinal lumen through the circulation and/or the lymphatic root to extraintestinal tissues as a result of intestinal mucosa barrier disruption. 5 –9 According to recent animal studies, liver RFA at approximately 30% of rat liver parenchyma disrupted the gut barrier leading to bacterial translocation, 10,11 which offers a plausible explanation for the occurrence of septic complications.
The pathogenetic mechanism of gut barrier dysfunction following liver RFA is still under investigation. The intestinal mucosa barrier comprises of the immune, the biological, and the mechanical components. 12 The biological gut barrier is represented by a balanced intestinal microflora, which prevents invasion of pathogenic or opportunistic bacteria to extraintestinal tissues by repressing their colonization and growth, enhancing differentiation and proliferation of intestinal epithelial cells and promoting the development of gut’s mucosal immune system. 13 Among the key factors recognized to affect intestinal microflora balance are bile acids present in the bile 9 as well as the inflammatory state of the bowel. 14 Bile acid microbial modulatory function is achieved primarily by the inhibition of growth of bacterial species, such as Bacteroides, Clostridia, Lactobacillus, and Streptococci. As shown in obstructive jaundice rat models, absence of bile salts leads to gram-negative bacteria overgrowth. 9 Inflammation of the intestinal wall provides a suitable tissue substrate for certain bacterial population overgrowth, which leads to commensal microbiota imbalance. 14
The aim of the present project was to investigate whether liver RFA affects the biological gut barrier in terms of the effect of bile production rate and bowel inflammatory state on intestinal microflora balance.
Materials and methods
Animals
A total of 25 New Zealand white rabbits, aged 4 months, weighing 3.0–3.5 kg, which were provided by our inbred rabbit colony, were used in the present study. They were housed individually in stainless steel cages under controlled environmental conditions (room temperature 20–22°C, humidity 50–60%, and 12 h photoperiod). They were fed with 125 g of commercially available pelleted diet per day per animal and tap water ad libitum. The facilities were in accordance with Directive 86/609/EEC.
Experimental design
The rabbits were randomly assigned into five groups of five animals in each group. In a first set of experiments, the rabbits were subjected to common bile duct (CBD) bypass by inserting a catheter toward the hepatic duct (proximal catheter) and another one toward the sphincter of Oddi (distal catheter) to establish an extracorporeal bile flow bypass, and then they were subjected to one session of open liver RFA (group CBD-RFA) or CBD bypass (group CBD). Bile was collected extracorporeally and its volume was measured for 48 h postoperatively. An aliquot of the collected bile (2 mL/h for 5 h/day, between 9:00 a.m. and 2:00 p.m.) was reinfused to the duodenum via the distal CBD catheter to simulate the enterohepatic circulation.
In a second set of experiments, the rabbits were subjected to either one session of liver RFA (group RFA) or sham operation (group sham) without prior cannulation of their CBD. At 48 h postoperation, (a) bile was collected by gallbladder puncture to determine biliary bile salts concentration, (b) a tissue sample was excised from the non-ablated liver portion for histopathologic examination, (c) a tissue sample was excised from the terminal ileum for morphometric analysis and histopathologic evaluation, and (d) fecal and tissue samples were collected from the jejunum and the cecum for microbiological analysis.
An extra group of rabbits was subjected to CBD single cannulation toward the hepatic duct to totally divert bile extracorporeally without reinfusion of bile to the intestine (group TBD) in order to investigate the effect of total bile deprivation on intestinal microflora. The distal part of the CBD toward the intestine was ligated. After 48 h, fecal and tissue samples were collected from the jejunum and the cecum for microbiological analysis.
At the end of the experiment, all animals were euthanized by exsanguination under general anesthesia. The experimental protocol was approved by the Animal Care and Use Committee of the local veterinary service since it complied with Directive 86/609/EEC.
Animal preparation
After 24-h food and 12-h water deprivation, anesthesia was induced by intramuscular injection of a xylazine (5 mg/kg)—atropine (0.04 mg/kg)—ketamine (50 mg/kg) mixture. Following endotracheal intubation, animals were connected to an anesthetic machine to receive 30 breaths/min of 80 mL tidal volume. Anesthesia was maintained by sevoflurane (2% in oxygen) administration. A self-adhesive gelled grounding pad was placed on a shaved surface at the back of the animals. The animals were placed at dorsal recumbency and their abdominal wall was clipped and prepared for aseptic surgery.
Common bile duct cannulation
A midline laparotomy was performed and the CBD was exposed and blindly dissected approximately 2–3 cm from the duodenal wall. After longitudinal dissection of the CBD wall, a 5-gauge umbilical catheter (Vygon/Ecouen, France) was inserted approximately 1 cm toward the liver (proximal catheter) and secured using 3-0 Vicryl ligatures (groups CBD, CBD-RFA, and TBD). A 3.5-gauge umbilical catheter was inserted toward the sphincter of Oddi (distal catheter) and secured (groups CBD and CBD-RFA). The free ends of the catheters were tunneled subcutaneously and exteriorized between the scapulae; the free end of the proximal catheter was connected to a 250-mL sterile plastic bag attached to the back of the animals to collect the bile, while that of the distal catheter was tapped and facilitated for bile reinfusion. A tailor-made jacket was placed over the rabbits to protect the catheters from being torn apart.
Radiofrequency ablation
A Radionics Cool-tip RFA System (Valleylab/Tyco Healthcare, Gosport, UK) consisting of a radiofrequency generator, a peristaltic perfusion pump, a grounding pad, and a single-shaft, 15 cm long, needle electrode with a 2-cm exposure tip was used. After midline laparotomy, the left lateral hepatic lobe was exposed. The tip of the electrode was inserted into the hepatic parenchyma from the caudal surface of the lobe at a 90° angle. The power delivered was 60 W for a 3-min period per session. The final tissue temperature reached between 60 and 70°C. During the RFA session, the tip of the electrode was cooled by continuous perfusion of ice-cold distilled water delivered by the peristaltic perfusion pump. Sterile gauzes soaked in cold normal saline were placed around the liver lobes to prevent transmission of heat to the surrounding tissues. Finally, the abdominal wall was closed in layers using 2-0 Vicryl sutures. During autopsy, both total liver and the ablated liver portion were weighed after being dissected from the rest of the liver. The percentile portion of the ablated liver weight was calculated with the following equation
Determination of bile salts concentration
Bile samples were kept at −80°C until analyzed. Cholic (CA) and deoxycholic acids (DCA) were determined in the form of glycine conjugates (glycine cholic acid (GCA) and glycine deoxycholic acid (GDCA), respectively) on a ultrahigh-performance liquid chromatography coupled with quadrupole time-of-flight–mass spectrometry (UPLC-qTOF-MS) system (Waters,Milford, Massachusetts, USA). The system, comprising of an acquity UPLC and a qTOF Ultima MS system (Waters, Milford, Massachusetts, USA), operated on a MassLynx platform in negative electrospray ionization with the following parameters: capillary: 2.20 kV; cone: 25 V; source temperature: 120°C; desolvation temperature: 350°C; desolvation gas flow: 800 L/h; TOF: 9.10 kV; Micro-channel plate (MCP): 1950. Chromatographic separations were performed in an acquity UPLC high strength silica T3 column using a binary gradient solvent system consisting of 0.1% (volume per volume; v/v) formic acid in high-performance liquid chromatography grade water (solvent A) and 0.1% (v/v) formic acid in acetonitrile (solvent B) with the following program: 30% solvent A constant for 1 min and then linear increase to 100% solvent B within 15 min; isocratic 100% solvent B for 1 min and back to initial condition of 30% solvent B, where the system was held isocratic for 4 min for column equilibration prior to the subsequent injection. The flow rate was 0.4 mL/min and the injection volume was 5 µL. GCA and GDCA gave characteristic ions at 464.27 m/z and 448.27 m/z, respectively. To enable the quantification, GCA and GDCA injections of reference standards were used to construct linear calibration curves in the range of 100 ng/mL–100 µg/mL. Unknown samples were analyzed thrice and their average was used for quantification. Samples were diluted at the appropriate ratio (500- to 4000-fold) in order to bring their concentration within the linear dynamic range.
Histopathology—histomorphometric analysis
Tissue specimens excised from the non-ablated liver portion (right median lobe) and the terminal ileum were fixed in formalin and embedded in paraffin according to standard procedures. Histopathologic examination was performed at 4-µm hematoxylin–eosin stained sections. The endpoints evaluated for (a) liver tissue were hyperemia/distension of sinusoidal space, hepatocellular degeneration/steatosis, distention of bile ducts, portal infiltration, and necrosis and (b) ileal tissue were neutrophil granulocyte, lymphocyte, and plasma cell infiltration, edema, hyperemia/vascular dilatation, and hyperplasia. The severity of lesions was quantified according to the following scoring system: 0: none; 1: mild; 2: moderate; 3: severe. Lesion severity scores were added to obtain the histopathologic score.
Intestinal mucosal morphometric characteristics were assessed by the measurement of villous height and density. Villous height was measured in 20 well-preserved villi per sample using the Nikon Digital Sight DC-L1 software (Nikon Eclipse 50i microscope, Kawasaki, Japan) and expressed as average villous height. Villous density was measured in 10 low-power optical fields (10×) per sample, and their average was expressed as number of villi per optical field. All examinations were performed in a blinded fashion.
Microbiological analysis
Fecal samples collected aseptically from the jejunum and the cecum were homogenized in sterile buffered peptone water. Tissue samples, 3 cm long, were also aseptically excised from the jejunum and the cecum. Tissue samples were washed twice with sterile buffered peptone water and vortex-mixed to break down bacterial clumps and remove loosely attached bacteria and then homogenized in 5 mL sterilized buffered peptone water using a tissue grinder. Fecal and tissue samples were subjected to serial dilutions. The following tests for microbiological analysis were performed: (i) total aerobic counts in plate count agar (Fluka, Buchs, Switzerland) at 30°C for 48 h, (ii) staphylococci in Baird Parker egg yolk tellurite medium (Fluka) at 37°C for 48 h and confirmed by a positive coagulase test, (iii) coliforms in violet–red bile agar (Fluka) after incubation at 30°C for 24 h, (iv) enterobacteria in violet–red bile glucose agar (Fluka) at 37°C for 24 h, (v) streptococci in bile esculin azide agar after incubation at 37°C for 24 h (Fluka), (vi) lactobacilli (gram (+), catalase (–)) in acidified MRS agar (MRS: de Man, Rogosa, and Sharpe, named after the inventors; Fluka) at 37°C for 48 h anaerobically (Anaerobic Jar, Anerocult C, Merck, Germany) and confirmed by Gram staining and catalase test, (vii) lactococci in M17 agar (Fluka) at 30°C for 24 h, and (viii) yeasts and molds in malt agar (Fluka; pH was adjusted to 4.5 by sterile solution of 10% lactic acid) at 30 C for 48 h. All incubations were further extended up to 120 h, but no extra colonies were observed. Results were presented as logarithm of mean colony-forming units on solid media culture plates containing between 30 and 300 colonies/g of fecal sample or tissue.
Statistical analysis
Data were expressed as mean ± SD. After normality of data was tested with the Kolmogorov–Smirnov test, these were subjected to analysis of variance. The Bonferoni test was used for multiple comparisons among groups and the Student’s t test for comparisons between pairs of groups. A p < 0.05 was considered statistically significant.
Results
One session of liver RFA resulted in the ablation of 18.7 ± 2.7% (range 15.4–22.7%) of total liver weight.
Bile production rate
Bile volume produced per day was corrected for body weight by adjusting the daily volume of bile to a standard body weight (4 kg). Animals subjected to liver RFA produced significantly less bile per day (p < 0.01) at 24 and 48 h compared with CBD bypassed animals (Figure 1).

Daily rate (mean, n = 5) of bile production following CBD bypass (group CBD) or CBD bypass plus liver RFA (group CBD-RFA). Error bars represent SD. *p < 0.05 versus CBD, **p < 0.01 versus CBD. CBD: common bile duct; RFA: radiofrequency ablation.
Bile salt concentration
No differences in the concentration of biliary GCA and GDCA were found between groups RFA and sham (Table 1).
Bile salts (GCA and GDCA) concentration (means ± SD, n = 5) following sham operation (group sham) or liver RFA (group RFA).
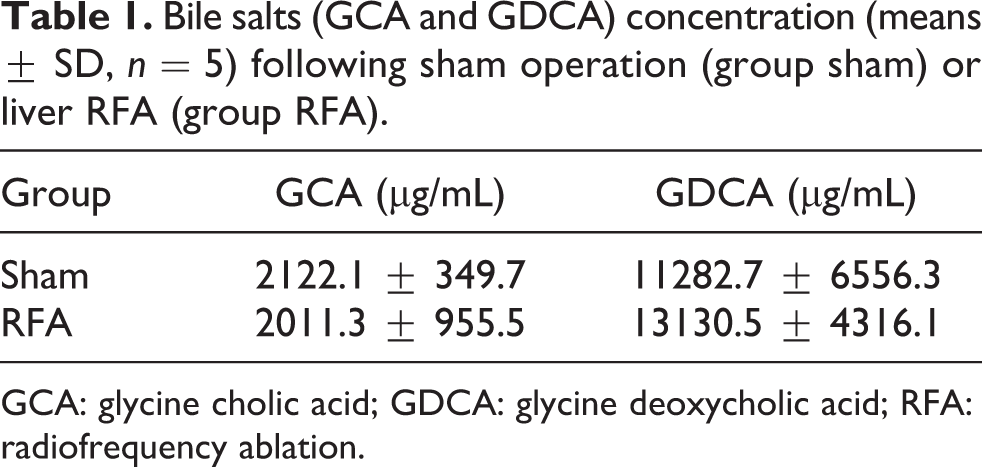
GCA: glycine cholic acid; GDCA: glycine deoxycholic acid; RFA: radiofrequency ablation.
Liver histopathology
Liver RFA induced tissue injury to the non-ablated liver parenchyma. Histopathologic lesions were mild and included hyperemia/distension of the sinusoidal space, distention of bile ducts, portal infiltration, hepatocellular degeneration/steatosis and necrosis (Table 2; Figure 2). The histopathologic score in group RFA was significantly increased (p < 0.01) compared with that of the sham-operated animals (Table 2).
Severity of histopathologic lesions (means ± SD, n = 5) in the non-ablated liver tissue following sham operation (group sham) or liver RFA (group RFA).

HSS: hyperemia/distention of sinusoidal space; HD/S: hepatocellular degeneration/steatosis; DBD: distention of biliary ducts; PI: portal infiltration; N: necrosis; RFA: radiofrequency ablation.
a p < 0.01 versus group sham.

Non-ablated liver tissue section. (a) Sham operation, normal liver parenchyma; (b) Liver RFA, focal steatosis (arrow 1), distension of bile ducts and portal infiltration (arrow 2), and distension of blood vessels and parenchymal infiltration (arrow 3; hematoxylin and eosin, ×100.) RFA: radiofrequency ablation.
Intestinal histopathology
Histopathologic lesions of mild severity characterizing the inflammatory state were noted in all layers of the intestinal wall of rabbits subjected to liver RFA (Figure 3). Overall, the histopathologic score in group RFA was significantly higher (p < 0.001) than that in group sham (Table 3).

Ileal tissue section. (a) Sham operation, normal ileal tissue; (b) Liver RFA, stroma hyperemia (arrow 1), intestinal crypt infiltration (arrow 2), mucosa infiltration (arrow 3; hematoxylin and eosin, ×40.) RFA: radiofrequency ablation.
Severity of histopathologic lesions (means ± SD, n = 6) of ileal tissue following sham operation (group sham) or liver RFA (group RFA).
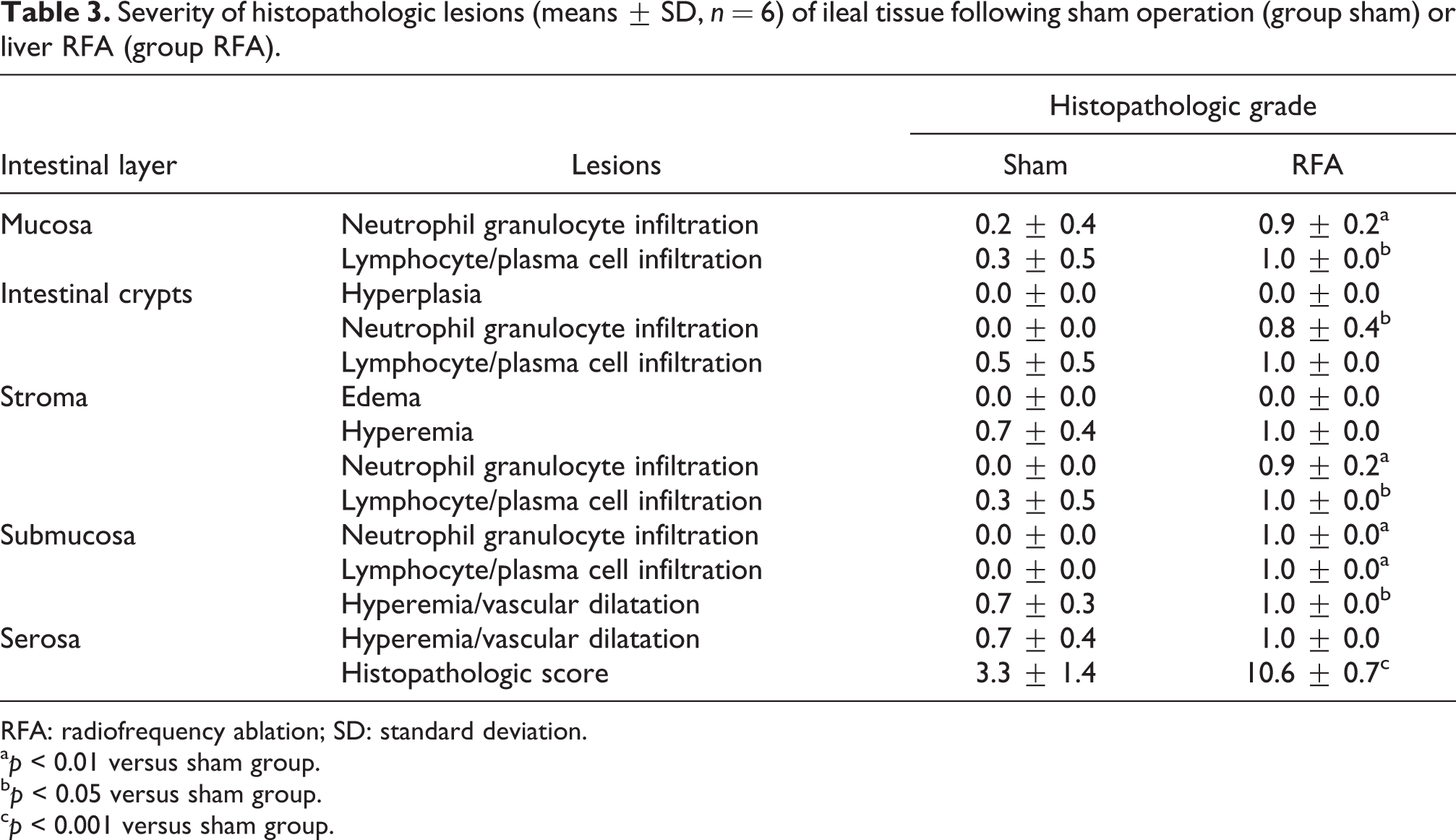
RFA: radiofrequency ablation; SD: standard deviation.
a p < 0.01 versus sham group.
b p < 0.05 versus sham group.
c p < 0.001 versus sham group.
Intestinal mucosa morphometry
Both intestinal villous height and density were significantly decreased in the rabbits subjected to liver RFA (Table 4).
Intestinal mucosa morphometric characteristics (means ± SD, n = 5) following sham operation (group sham) or liver RFA (group RFA).

RFA: radiofrequency ablation.
a p < 0.05 versus sham group.
b p < 0.01 versus sham group.
Intestinal microflora
In the jejunal content, the populations of Enterobacteria, Lactobacilli, and Lactococci were increased in group RFA, of yeasts and molds in group TBD, while of Coliforms in both groups TBD and RFA. In the cecal tissue and content, total aerobic counts, Enterobacteria, Coliforms, Lactobacilli, Lactococci, yeasts, and molds were increased in both groups TBD and RFA (Figure 4). No change was noted in the populations of Streptococci and Staphylococci (data not shown).

Intestinal microflora (means, n = 5) of animals subjected to sham operation (group sham), total bile deviation (group TBD), or liver RFA (group RFA). Error bars represent SD. Jejunal-T, jejunal tissue; Jejunal-C, jejunal content; Cecum-T, cecal tissue; Cecum-C, cecal content. a,b,cFor each intestinal site, means with different letters differ significantly (p < 0.05). RFA: radiofrequency ablation.
Discussion
The present experimental study contributes to the understanding of the pathogenetic mechanism of gut barrier dysfunction following liver RFA. It provides evidence supporting the view that reduced bile production and mild bowel inflammation secondary to liver RFA alter intestinal microflora and therefore disturb the biological gut barrier.
As previously demonstrated, RFA at approximately 30% of liver mass in the rat resulted in the disruption of the mechanical component of the gut barrier leading to translocation of intraluminal bacteria and endotoxins to proximal and distal to the intestine organs. Atrophy of the intestinal mucosa was attributed to increased crypt cell apoptosis related to induction of oxidative stress. 10 In the present liver RFA rabbit model, we investigated the potential relation of altered bile production with dysfunction of the biological gut barrier. The daily rate of bile production was decreased in rabbits subjected to RFA at approximately 15% of their liver mass. This effect was anticipated since (a) a functional liver volume, equivalent to that necrotized by ablation, was deprived and (b) the non-ablated liver parenchyma, which undertook bile production, was injured as evidenced histologically in the current study confirming previous experimental work. 15 –17
DCA is the main bile acid (90%) in the rabbit, followed by the CA (8%). 18 It has been estimated that the bile acid pool in the rabbit depletes in 5 days when bile flow to the duodenum is deviated. 19 In light of the impaired enterohepatic circulation of bile acids due to extracorporeal diversion of bile flow and only partial reinfusion of bile to the duodenum, bile salt levels were determined in bile samples collected directly from the gallbladder of animals with intact CBD. The concentrations of biliary deoxycholic and cholic salts were not affected. However, given the reduction in bile production rate, the quantity of bile acids ultimately reaching the duodenum was decreased.
A growing body of evidence highlights the important role of bile acids in preserving a balance among intestinal bacterial populations. While bile acids inhibit the growth of gram-positive bacteria, they have little effect on gram-negative ones. 20 Therefore, an overgrowth of gram-negative bacteria, in the absence of bile acids, is regarded as a consequence of intestinal microbiota imbalance. Gram-negative bacteria have variable endotoxic activity due to lipopolysaccharide contained in their outer cell membrane. Bile salts also bind directly to intraluminal endotoxins and bacteria, to form poorly absorbed detergent-like complexes, preventing them from being translocated through the gut barrier. 9
In vivo studies on the effect of bile acid deprivation on intestinal microflora typically use obstructive jaundice rat models induced by CBD ligation 21,22 with inevitable regression of bile to the liver and concurrent pancreatitis due to pancreatic ducts obstruction. We preferred bile flow extracorporeal deviation to avoid complications derived from CBD ligation. In addition, by determining microbial growth not only to fecal samples but also to intestinal tissue, the true number of microbia adhered to the intestinal mucosa was evaluated. Bile deprivation altered intestinal microflora as manifested by overgrowth of total aerobic counts, Enterobacteria, Coliforms, Lactobacilli, Lactococci, yeasts and molds, especially in the cecum. The same microbial populations were increased in those animals subjected to liver RFA, recognizing thus the reduction in bile production as a determining factor for the changes noted in the intestinal microflora.
Small intestine bacterial overgrowth (SIBO) was noted in the liver RFA group as evidenced by an increase in the population of Enterobacteria, Coliforms, Lactobacilli, and Lactococci in the jejunal content. Bacterial overgrowth in the small bowel may be a consequence of impaired intestinal motility. Attenuation or interruption of sweeping contractions that propagate from the stomach or the duodenum to the terminal ileum allow proliferation of distal gut bacterial populations into the small intestine. 23 As shown in animal studies, cutaneous burn injury leads to impaired small bowel transit. 24 This could offer a plausible explanation for the liver RFA-induced SIBO, since ablation of the liver results in severe burn trauma of the hepatic parenchyma.
Injury of the gut wall characterized by mild inflammation of all intestinal layers was noted in the rabbits at 48 h post liver RFA. Previous experimental work in rats also reported the presence of inflammatory cells confined mainly in the intestinal mucosa up to 48 h after liver RFA, further expanding to the submucosa at 72 h. 10 It has been suggested that the composition of gut microbial populations is related to the physiological state of the intestine, although little is known about the effect of inflammatory mediators of the intestinal epithelium, such as pro-inflammatory cytokines and chemokines, on intraluminal bacterial balance. 23 Inflammation of the intestinal mucosa has been shown to promote overgrowth of certain bacteria of the Enterobacteria family whose fitness is enhanced in the inflamed gastrointestinal tract. 14 Conversely, an imbalance of commensal microbiota has been proven to activate the immune response of the host and deteriorate the intestinal inflammatory state through a decrease in beneficial bacterial metabolic end products and a concurrent increase in detrimental bacterial toxic products. This host–intestinal microbiota interaction has been well illustrated in chronic inflammatory diseases, such as the inflammatory bowel disease and the irritable bowel syndrome. 23
Intestinal mucosa atrophy, suggesting dysfunction of the mechanical gut barrier, was noted in the liver RFA group. Although the pathogenetic mechanism of this phenomenon is multiparametric, reduced bile production and disturbed intestinal microflora, documented by the present study, have been shown to contribute toward this direction. Bile exerts a trophic effect on the intestinal mucosa by promoting intestinal epithelial cell proliferation and is also important for the maintenance of the integrity of enterocyte tight junctions. 9 Short-chain fatty acids produced by intraluminal bacteria stimulate differentiation and proliferation of epithelial cells in the small and large intestine. 13
In conclusion, the findings of the present study shed some light to the pathogenetic mechanism responsible for the dysfunction of the gut barrier following liver RFA. A decreased bile production rate and mild inflammation of the bowel impaired the biological gut barrier as manifested by alteration of the intestinal microflora. Malfunction of the gut barrier could justify the occurrence of infectious complications related to this tumor ablating method that narrow the safety limits of the procedure. Based on these results, future studies may focus on designing effective preventive or therapeutic strategies involving bile acid supplementation, anti-inflammatory drug administration, or intestinal microbiota balance modulators.
Footnotes
Acknowledgments
The authors wish to thank Drs Eirini Papageorgiou and Constantinos Garoufas and Mr Ioannis Maragos for their help in conducting the experiments; Mrs Marianthi Sidira for helping with the microbiological analysis; and Associate Professor Georgios Theodoridis for helping with bile salt analysis.
Conflicting Interests
The authors declared no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.

