Abstract
Cardiac electrophysiology utilizes nonimplantable, catheter-based devices for diagnosis and treatment of arrhythmias as well as electroanatomical mapping of cardiac chambers. Gross pathology and histopathological assessments in preclinical studies play critical roles in determining the safety and efficacy of cardiac ablation systems used to treat tachyarrhythmias. The pathologist must assess ablation sites, adjacent structures and organs, and downstream organs to characterize the effects of the ablation treatment and determine whether adverse local reactions, collateral injury, or downstream thromboembolism are present. Histopathological assessment serves as an adjunct to electroanatomical data in determining efficacy in preclinical studies. Histopathology is the standard in definitively demonstrating transmurality of ablation lesions, which is necessary for complete conduction block, as well as showing the linear or circumferential distribution of a contiguous, transmural ablation lesion necessary for electroanatomical isolation of entire target structures such as pulmonary veins and the cavotricuspid isthmus, which are involved in propagating certain arrhythmias. This article will detail gross and histological methods for the pathology assessment of preclinical studies evaluating the safety and/or efficacy of cardiac ablation catheter systems as well as discuss correlation of pathology data with other supporting evidence for safety and efficacy such as acute, electroanatomical data.
Intracardiac catheter-based ablation can be a curative therapy for cardiac arrhythmias by permanently disrupting or isolating the foci of initiation or the anatomical pathway involved in propagating the arrhythmia. Intracardiac ablation is commonly used in this manner to treat atrial fibrillation, atrial flutter and other atrial macroreentry tachycardias, atrioventricular (AV) junctional tachycardias, accessory AV connections, and ventricular tachycardias. Radiofrequency (RF) and cryothermal are the two most common energy modalities used to initiate irreversible injury to cardiomyocytes (Luik et al. 2015; Packer et al. 2013), and emerging energy-based ablative therapies have utilized ultrasound (He et al. 1995; Ohkubo et al. 1998), laser (Wagshall et al. 2002), microwave (Yiu et al. 2007), and irreversible electroporation methods (Wojtaszczyk et al. 2018).
Regardless of the energy modality used, cardiac ablation catheters have at least two basic components at the distal end which interact with the endocardium: (1) a mechanism to deliver energy and (2) electrodes used for electrocardiogram (ECG) sensing. For RF catheters, the energy, a form of alternating current, is delivered through electrodes. In contrast, cryoablation catheters contain some type of chamber which allows circulation of coolant. The intracardiac ablation procedure involves delivering the catheter to the target location, applying the energy, and assessing the effects of the energy in order to determine whether the application can be terminated or continued. Delivering the catheter to the target location is done utilizing a combination of imaging modalities, including fluoroscopy and intracardiac mapping systems, and the ECG derived from electrodes on the distal catheter. The assessment of the local effects during the ablation is done by observing amplitude reduction in the catheter derived ECG as well as drops in impedance across the catheter tip electrodes. Catheters usually contain thermocouples or thermistors which allow for real-time recording of the temperature at the tip/tissue interface.
Preclinical assessment of intracardiac catheter-based ablation systems involves demonstrating both effectiveness and safety. The preclinical pathologist is involved in both aspects of the ablation system assessment by investigating the local, collateral, and downstream effects within the animal model test system.
Effectiveness
In order for cardiac ablation to be effective, the therapy must produce a lesion within the myocardium, which eliminates the arrhythmogenic substrate and/or the continuity of the associated conduction pathway. The utilization of animal models which have cardiac arrhythmias is rare in preclinical research particularly in studies for which the objectives are to determine optimal dosing of the therapy or to show the safety of the ablation system. Similar to clinical procedures in which the patient may not be exhibiting the arrhythmia during the cardiac ablation procedure, the preclinical study approaches target ablation locations on an anatomical basis. Because the devices are designed from human, clinical application, large animal models, such as pigs, sheep, and dogs, are used for preclinical assessment. Anatomical structures which are commonly the arrhythmogenic substrates, such as the cavotricuspid isthmus (CTI) for atrial flutter and the antra/ostia of pulmonary veins (PVs) for atrial fibrillation, are targeted to model the clinical approach to therapy and demonstrate the achievement of successful ablation. These lesions may be individual/discrete lesions in which the catheter tip does not move during the power-on phase or as drag lesions in which the catheter tip is moved from point to point while dragging the catheter tip along the endocardial surface during the power-on phase.
During dosing studies, which aim at determining a relationship between ablation parameters and lesion formation, discrete lesions are typically made in a variety of cardiac locations which represent the range of tissue characteristics of potential ablation targets in the human clinical setting. This approach will include performing lesions in very thin tissue, such as venae cavae, the interatrial septum (IAS), and the lateral regions of the atria, as areas of intermediate thickness, such as PV antra, the mitral annulus, and the pulmonary outflow tract, as well as thicker cardiac tissue such as the left ventricular myocardium and interventricular septum. The surrogate of efficacy is the demonstration of a transmural ablation lesion at the target location (Chubb et al. 2017). For complex and larger anatomical structures which require multiple discrete or drag lesions, this demonstration of a transmural lesion extends longitudinally along the compound lesion line. A goal of correlating effectiveness at these locations, particularly PV antra is to show a contiguous, transmural lesion, as a break in transmurality compromises the ability to electrically isolate the anatomical structure (Koruth et al. 2015; Iwasawa et al. 2017).
Dosing studies may also be performed in an acute (nonsurvival) in vivo skeletal muscle model with the canine or porcine thigh being the most commonly used (Nakagawa et al. 1995; Yokoyama et al. 2006, 2008). In the in vivo thigh model, lesions are produced on the surface of the isolated target muscle within a recirculating, autologous, heparinized blood pool kept at a constant temperature. The catheter is fixated externally during the ablations, which allows for precise control of catheter orientation (perpendicular or parallel) and force. Data from in vivo thigh model studies are often used to show substantial equivalency or a lack thereof between the test catheter and a predicate catheter. These data can be leveraged during the regulatory submission and may shorten the time line for approval of the test catheter for human use.
Lesion Characteristics
Gross Assessment
A comparison of the treatment matrix with fluoroscopy and electroanatomic maps is useful in determining the likely anatomical locations of attempted ablation lesions. The acute ablation lesion (from nonsurvival studies) is grossly discernible on the transverse section and is characterized by a well-demarcated zone of marked color change from the surrounding unaffected myocardium (Figures 1 and 2) or skeletal muscle for the case of the in vivo thigh model. There is usually a larger central zone of light tan to brown with a narrow rim of darker tissue external to the central zone yet contrasting the unaffected bordering myocardium. The endocardial appearance of the ablation lesion is similar in color to the transverse section; however, there is wide variation in the size and discernibility due to the tissue characteristics and catheter orientation during lesion formation. Lesions formed deep in trabeculated tissue with the catheter tip in an orientation perpendicular to the endocardium may be indiscernible from the endocardial surface. However, lesions formed on smooth areas of endocardium are usually easily discernible due to the color change on the endocardial surface. On the epicardial surface, transmural lesions have a similar appearance to the transverse section; however, epicardial adipose tissue can mask their appearance. A steam pop, sometime referred to as “myocardial boiling,” is a phenomenon in which the intralesional temperature exceeds the temperature of steam formation and the resulting steam is released from the central portion of the lesion into the surrounding tissue (Cooper et al. 2004). This release of steam is often accompanied by an audible “pop” which can be heard in the treatment room. Lesions which were created from an ablation which resulted in a steam pop usually have a central cavity, sometimes with multiple clefts and a variable amount of hemorrhage.
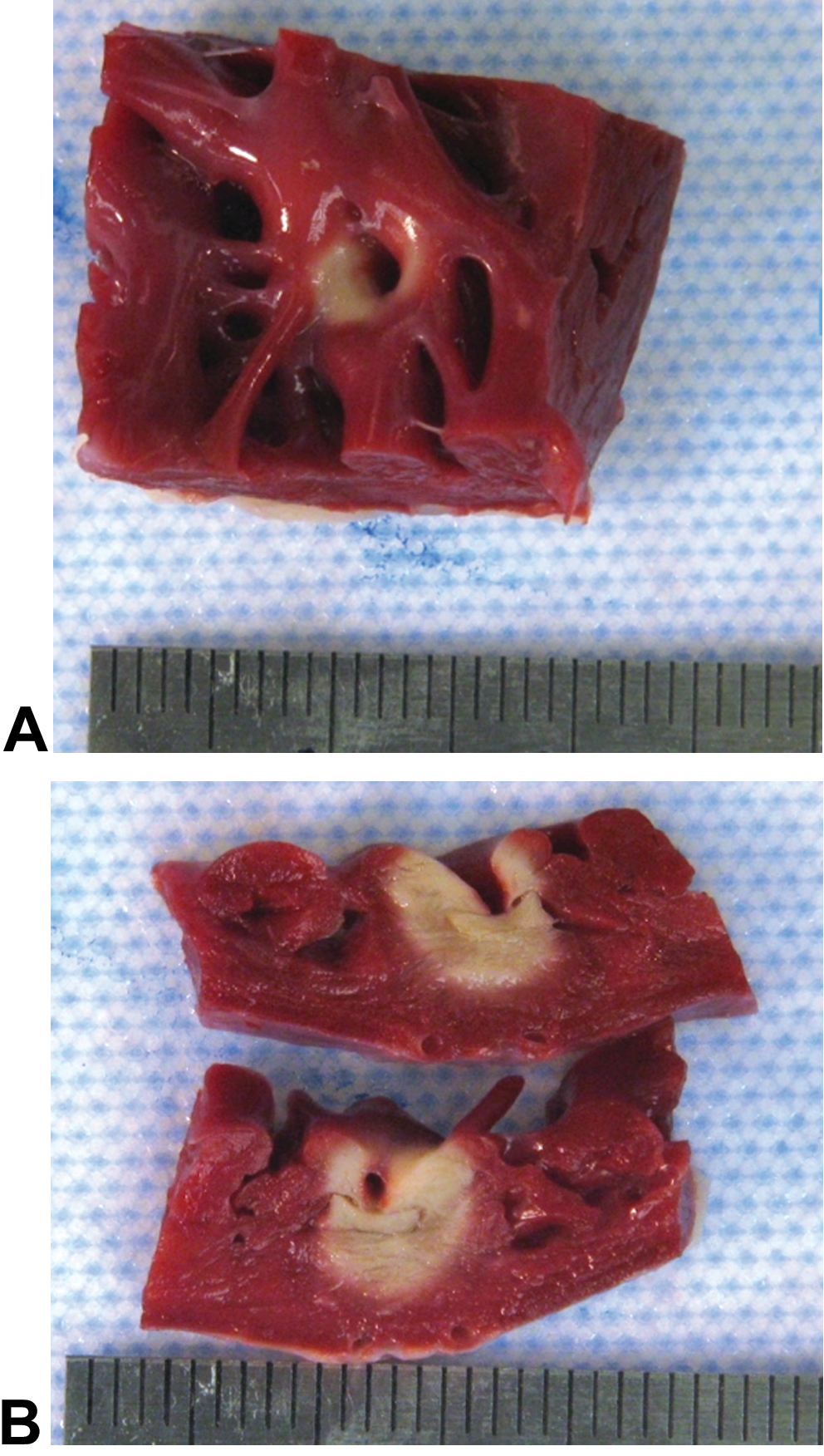
Gross appearance of a 2,3,5-triphenyltetrazolium chloride–stained cardiac ablation lesion induced by radiofrequency in the freewall of the right ventricle approximately 3 hr after creation. (A) Endocardial surface. (B) Transverse cut surface through the middle of the lesion. The catheter tip was present within the trabecula during the ablation procedure.
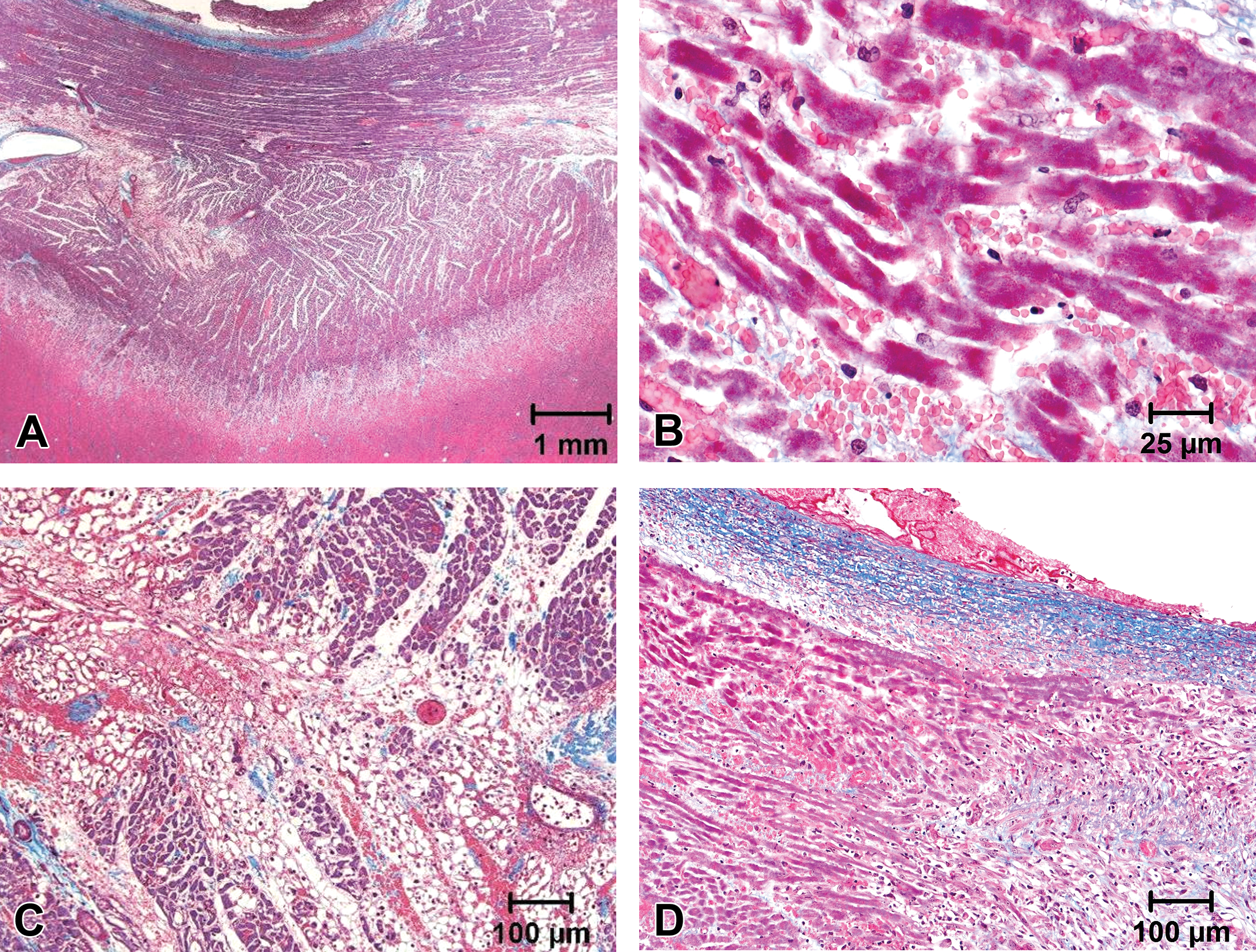
Gross and histological appearance of acute radiofrequency ablation lesions approximately 3 hr after creation in the right ventricle. (A) The mid-transverse cross section of the lesion. The myocardium is stained with 2,3,5-triphenyltetrazolium chloride. (B) Low power micrograph of the lesion in A. (C) A transmural ablation lesion in the freewall of the RV is exhibiting edema which is separating cardiomyocytes. (D) Edema and hemorrhage are separating ablated cardiomyocytes. (E) The sarcoplasm of the cardiomyocyte at the periphery of the lesion is exhibiting coagulation and loss of striations. (F) The border of the necrotic ablation lesion (below line) and the nonnecrotic adjacent myocardium (above line) are shown. All sections are stained by Masson’s trichrome method.
Vital stains which rely on a biochemical reaction to identify viable tissue are often used in gross assessments of lesions in cardiac ablation studies. The most commonly used vital stain for this purpose is 2,3,5-triphenyltetrazolium chloride (TTC) which stains viable, nonnecrotic tissue deep red due to the activity of cellular dehydrogenase (Adegboyega et al. 1997). The utilization of TTC usually adds little in aiding in the identification of ablation tissue; however, TTC perhaps provides an advantage in photographic contrast and in validating the interpretation of an area of interest as a true ablation lesion. It is important to note that TTC does not penetrate tissue; it is a surface stain only and may obscure borders of lesions in which hemorrhage is a prominent feature. The lack of surface penetration can be overcome by perfusion of TTC through the coronary arteries for hearts or through the iliac arteries for the in vivo thigh model. TTC has little advantage in assisting in the identification or contrast of ablation lesions at more chronic time points due to the staining characteristics of the remodeled lesion rich in fibrous tissue. Due to the progressive remodeling of the ablation lesion characterized by replacement of the necrotic myocardium with fibrotic tissue, ablation lesions may be less discernible from the surrounding nonaffected myocardium as the time point after treatment increases. By 30 days after treatment and beyond, the lesions are usually composed of foci of light tan tissue with less distinct borders than the acute (nonsurvival) and subacute (time point of approximately 7 days) lesions. This distinction between the ablation lesion and nonablated surrounding myocardium at chronic time points is particularly challenging when viewed from the endocardial surface. Small, focal (discrete) ablation lesions and ablation lesions in thin tissue can be more challenging to discern from surrounding nonablated tissue than larger lesions in thick tissue.
The gross identification of lesions at chronic time points in which total healing and remodeling have occurred, for instance 90 days after treatment or later, may need to rely heavily on an anatomical approach for sampling of lesions for histological assessment. Fluoroscopic and electroanatomical maps from the day of treatment should be used to confirm the treatment areas or particular PVs and to gauge other anatomical locations in which lesions should be sampled. Due to the poor discernibility of lesions on the endocardial surface at these more chronic time points due to the lack of contrast between the endocardium and fibrotic lesions, serial sectioning (breadloafing) of the anatomical areas of interest may be necessary to identify lesions.
Tissue Trimming and Histoprocessing
One of the goals of the histological assessment of cardiac ablation lesions is to determine the extent of the lesion through the myocardial wall. To successfully demonstrate transmurality of ablation lesions, transverse sections, extending from endocardium to epicardium, of the area of interest are trimmed from the myocardium with a border of unaffected myocardium around the target lesion. The midpoint of lesions which are easily discernible on the endocardial surface usually can be identified, and the grossing of discrete lesions can be done through this midpoint to sample the area of maximal mural extent. The exact origin of the slides obtained for histology should be documented. If there is doubt that this zone of maximal mural extent was sampled, additional transverse slices on each side of the original can be explored at the time of grossing, and/or multiple levels (step sectioning at specified intervals) of the resulting processed tissue block can be taken at the time of microtomy. In studies which have the purpose of validating electroanatomical data from a mapping system, the identification of zone of transmurality and mural gaps in the lesion are dependent on histological assessment of the areas of maximum tissue depth (Figure 3).
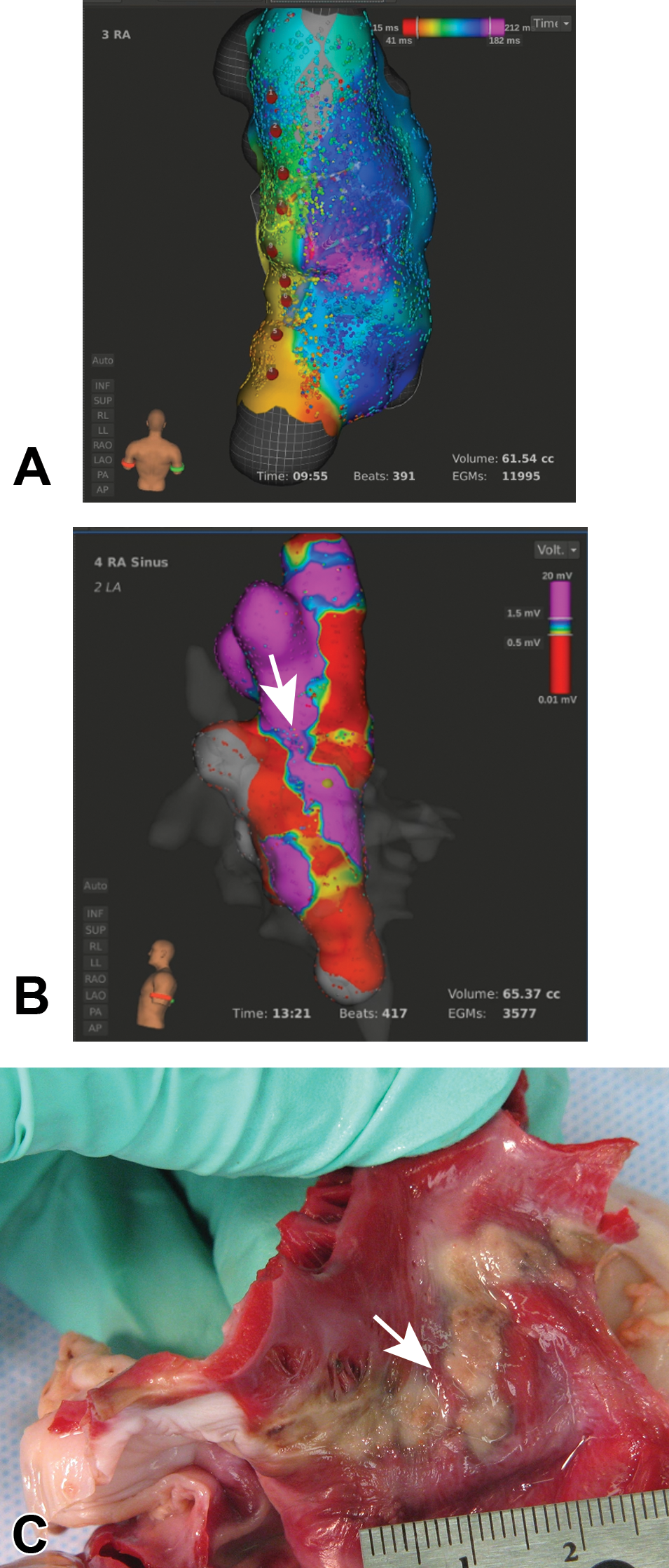
A three-dimensional electroanatomical mapping system was used to document lesions within the right atrium (RA) along a multiple point lesion line extending from the cranial vena cava to the caudal vena cava. (A) The posterior view of the activation map of the RA shows the locations of each treatment site as a red dot. (B) The voltage map of the left lateral view of the treatment line suggests a gap within the continuity of the ablation line (arrow). (C) The gross appearance of the endocardial surface shows the location of a histologically confirmed gap in the lesion line which was approximately 0.4 mm (arrow). The heart was stained with 2,3,5-triphenyltetrazolium chloride by intracoronary perfusion.
Assessment of the mural extent of lesions in larger, more complex cardiac locations may require the processing of oversized sections and cutting multiple levels throughout the section. Studies that ablate PV antra often attempt to correlate electrical PV isolation with a histologically discernible transmural lesion around the entire circumference of the PV. For this assessment as well as for assessing PV stenosis resulting from endocardial hyperplasia, it is paramount to examine the transverse section of the PVs from the antra through the termination of the cardiomyocyte sleeves along the PVs in order to visualize the entire circumference. In studies which ablate a single PV or perhaps only 2 PVs which are distant to each other, the individual PVs may be trimmed from the left atrium (LA) in a manner which preserves the entire antrum and surrounding tissue and extends the entire length of the cardiomyocyte sleeves to the nonmuscular portion. If there are multiple treated PVs in the study which are too close to one another to separate the treatment zone during grossing or if there are other LA lesions which are at anatomical locations which cannot be separate from the PV lesions, such as lesions on the roof of the LA or mistral isthmus, then the entire LA can be processed as a single piece (Figures 4 and 5). To best accomplish this, the LA should be trimmed on the atrial side of the mitral annulus, as much of the left atrial appendage (LAA) as possible should be removed, and processing sponges should be used within the PVs and within the lumen of the LA in order to maintain shape and patency. The identification of individual PVs in the resulting dissected LA should be certain at the time of processing, and documentation of the identity of the PVs should be made in a manner which allows tracking of orientation throughout embedding, microtomy, and slide preparation. Photographs, grossing diagrams, and tissue dyes are useful in maintaining the orientation and identification of the PVs.
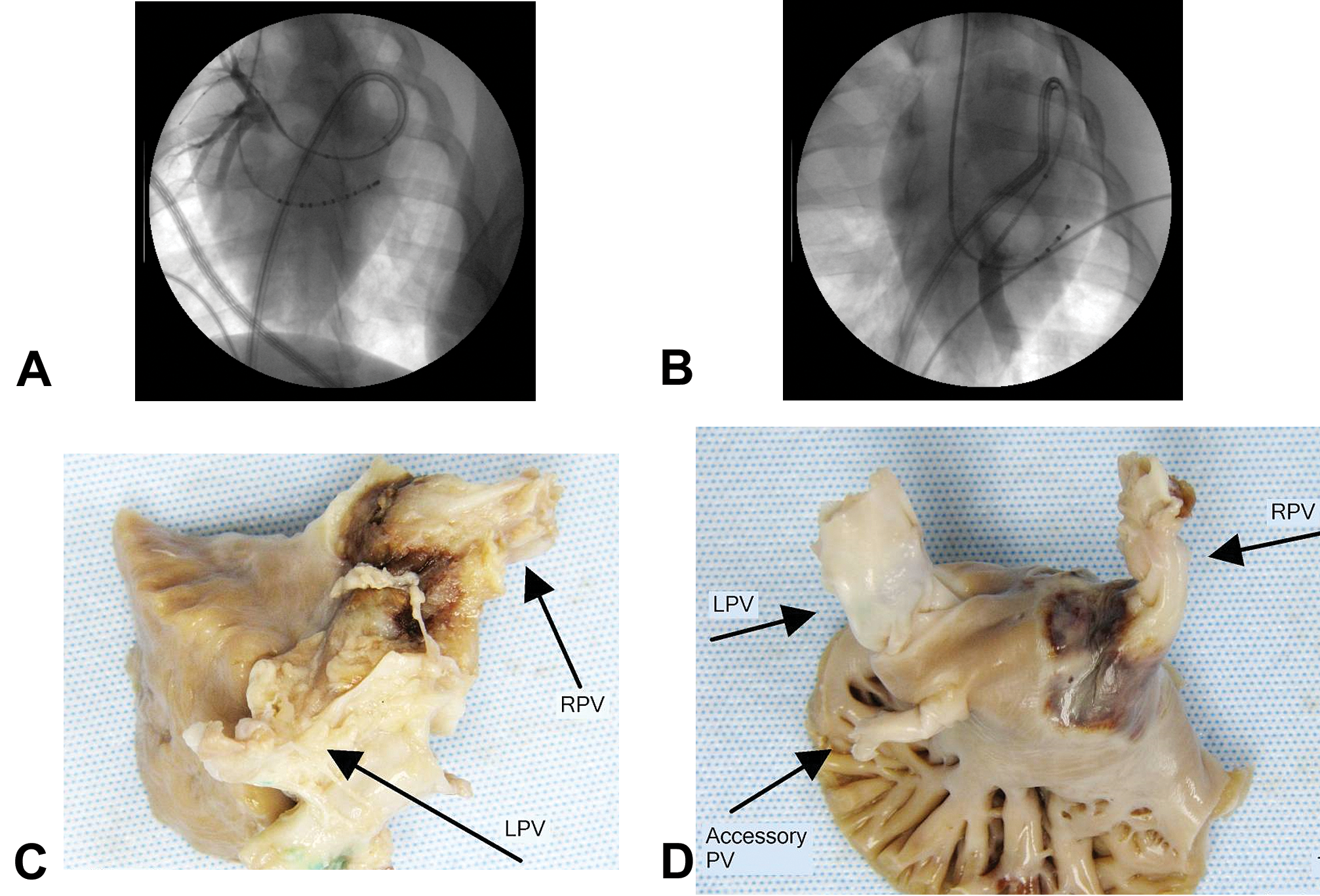
Fluoroscopy at the time of treatment and the postfixation left atrium (LA) of a pig treated with a cryoablation balloon catheter. (A) The fluoroscopy shows the balloon inflated in the right pulmonary vein (RPV) with contrast venogram during a sham/control treatment. (B) The fluoroscopy shows the balloon inflated in the left pulmonary vein (LPV) during a 1.5-min treatment cycle which was performed twice. (C) The epicardial surface of the LA shows discoloration around the RPV 8 days after treatment. (D) The endocardial surface of the LA with the pulmonary veins (PVs) inverted shows discoloration indicative of the ablation lesion at the antrum and ostium of the RPV.
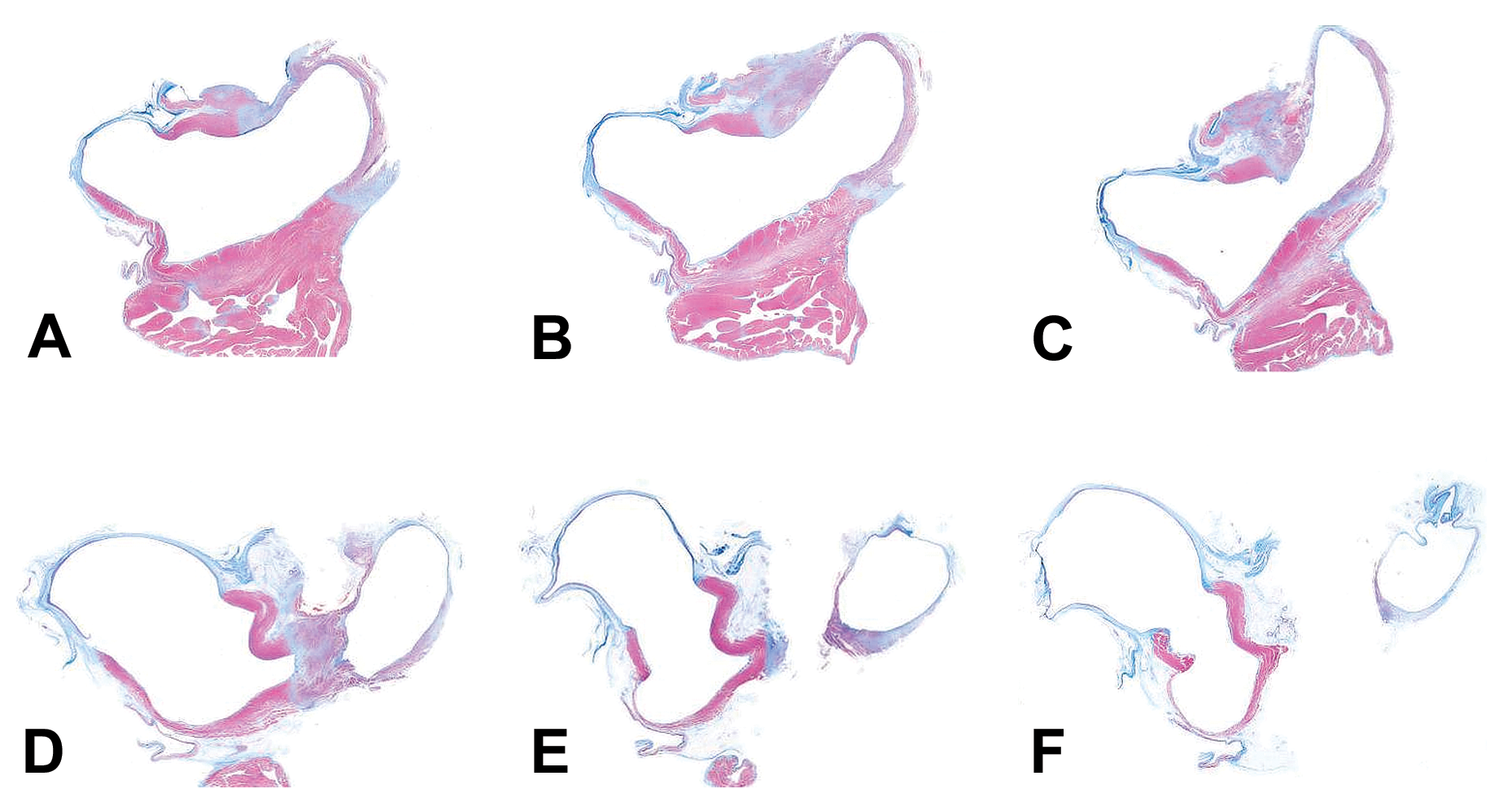
Multiple levels of the left atrium (LA) shown in Figure 4 show the low-power (subgross) histological appearance of the treatment zone within the antrum and ostium of the right pulmonary vein (RPV) 8 days after cryoablation. The trimmed LA was processed as a single piece and embedded in paraffin; (A) is closest to the mitral annulus and (F) is most proximal in the pulmonary vein (PV). The RPV is on the right side of each image. The spacing between adjacent levels is 1,000 μm, and each section was stained with Masson’s trichrome method. The thin, blue tissue represents native, fibrous venous wall architecture which is prevalent on the left side. Red represents nonablated cardiomyocytes, and the ablation lesion in the ostium of the RPV is gray/purple with blue at the periphery.
Studies in which the identification of lesion gaps within line lesions is critical, such as CTI drag lesions or multiple point lesions between the cranial and caudal venae cavae in the right atrium (RA), also benefit from the approach of oversized sections with step-sectioned levels in order to perform a histological assessment across the lesion line for continuity or gaps. Ideally, the entire longitudinal length of the ablation lesion line should be dissected free from the surrounding tissue with a border of grossly unaffected myocardium. However, linear lesions are rarely a straight line and often have multiple angles and segments; therefore, it may be necessary to split the lesion line into multiple segments for histoprocessing in order to survey the entire lesion. If the line is split into more than a single unit, each transection should be within the middle portion of a discrete focus of ablation in order to not potentially miss or misinterpret a gap between foci of ablation.
Routine processing of cardiac ablation lesions appropriate for the size of the resulting blocks into paraffin and typical microtomy at 5-μm thickness is adequate for histological assessment. Intervals of 1,000 μm for step sectioning of levels are usually satisfactory for complete assessment across discrete lesions, LA/PV blocks, or lesion lines (Figure 5). A typical microtomy scheme for assessing a lesion line in its entirety will be able to discern the presence of gaps in lines or the circumferential extent of transmurality for PV isolations. Further characterization may be necessary from unstained sections taken at 500-μm interval between the 1,000 μm levels at the time of microtomy to ensure complete characterization or to answer questions that may arise during the initial assessment. Another approach used with whole LA preparations for PV assessment is to assess a fixed number of levels. Twenty levels within the LA block, given that there is not an excessive amount of unnecessary annular tissue, have been used successfully to characterize the circumferential and mural extent of PV antral ablations. In the aforementioned scenario, the interval between levels would be determined by the height of the postprocessed block, and intervals close to 1,000 μm are typical for a canine or porcine LA.
Histological Stains and Interpretation
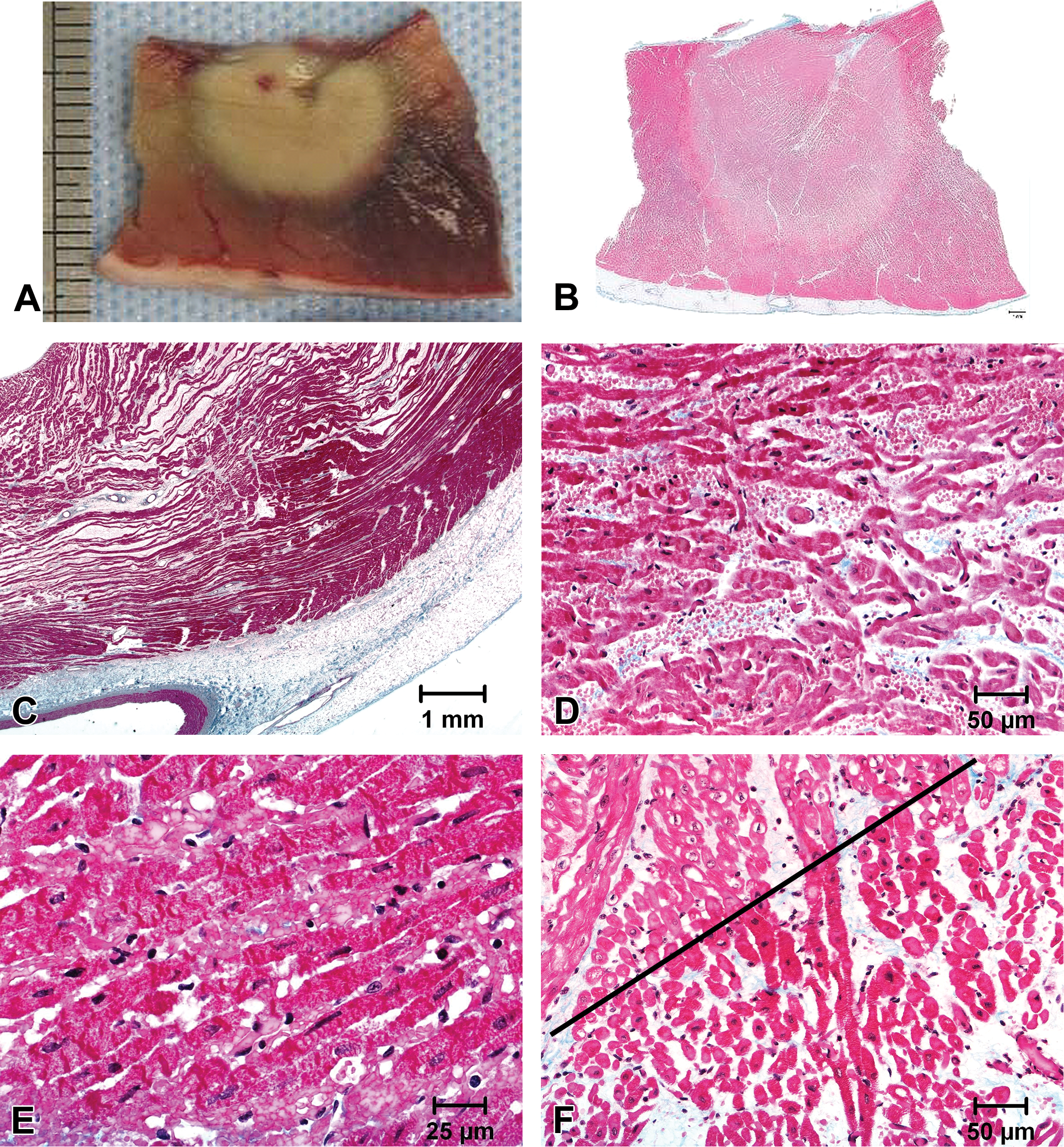
A trichrome stain is paramount in the assessment of cardiac ablation lesions. Masson’s trichrome technique is adequate for general high contrast identification of cardiomyocyte changes at acute and subacute time points as well as documentation of fibrosis at more chronic time points. If the histological assessment includes the determination of the amount of endocardial hyperplasia, such as with the qualitative or morphometric assessment of PV stenosis, techniques that combine a trichome stain with Verhoeff’s elastin technique, such as Gomori’s elastin trichrome or Masson’s elastin trichrome, or Movat’s pentachrome techniques are useful (Figure 6). We have found that the Gomori’s elastin trichrome can effectively tinctorially delineates changes to all tissue components affected in acute and subacute thermal lesions. Hematoxylin and eosin (HE)-stained sections are often assessed at each level alongside the trichrome slides; however, the HE slide usually provides little to no advantage over the trichrome slide for acute; it’s utility is largely in subacute to chronic lesions for micrographic documentation of lesional features which have poor contrast with the trichrome stain, such as inflammatory cell type differentiation and mineralization.
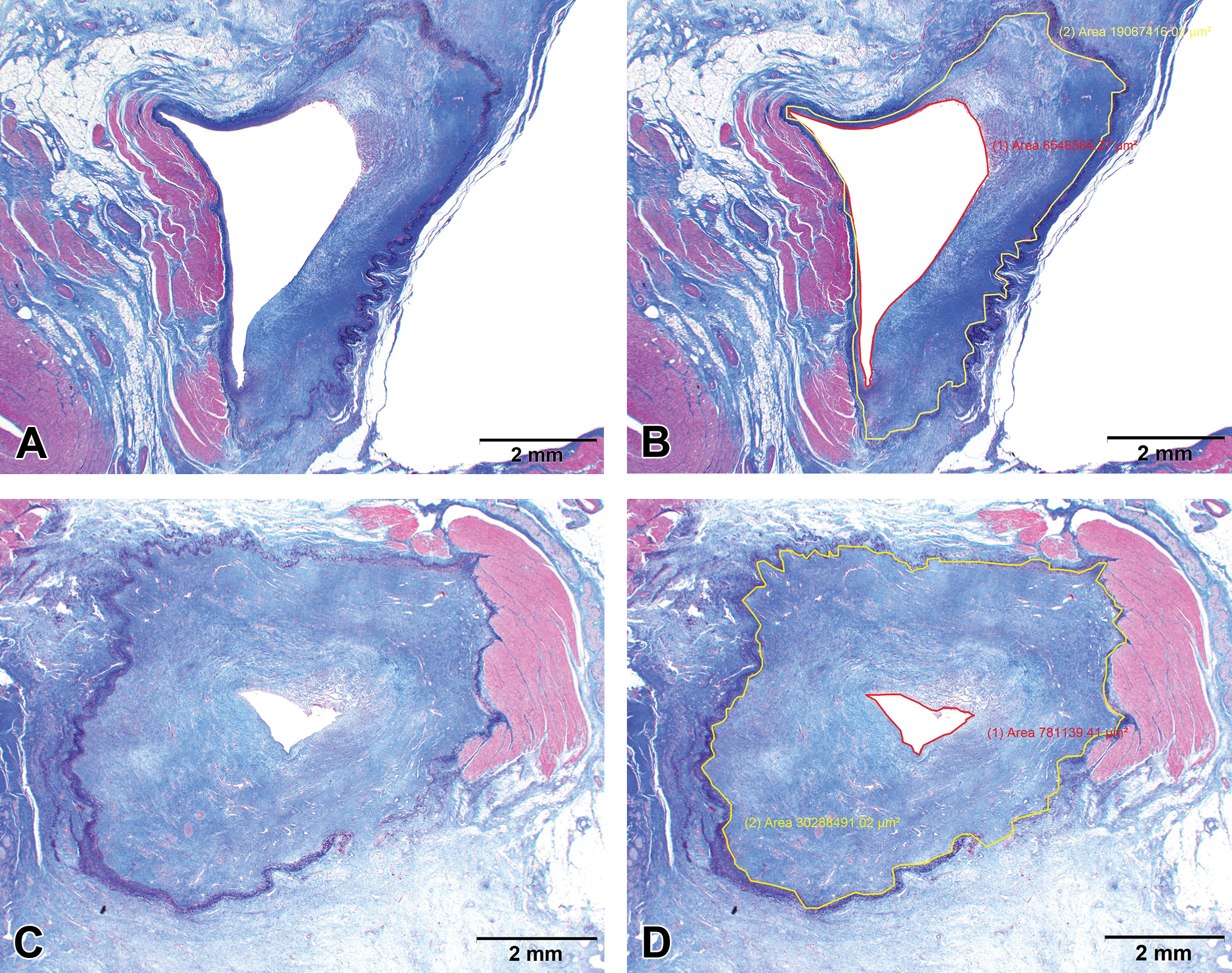
Right pulmonary veins from pigs treated with radiofrequency ablation 60 days prior to euthanasia are shown. The images on the left show Masson’s elastin trichrome-stained cross sections of each pulmonary vein (PV) and the images on the right are identical with the addition of the morphometrical marking to define the luminal area (red) and the elastin area (yellow). (A and B) The PV contains a segmental area of replacement of cardiomyocyte sleeves with fibrosis. The endocardial area adjacent to the zone of fibrosis is exhibiting hyperplasia resulting in stenosis of approximately 65%. (C and D) A segment of the cardiomyocyte sleeve remains, but the entire circumference of the PV is markedly affected by endocardial hyperplasia resulting in stenosis of approximately 97%.
Histological characteristics and the time course of lesion remodeling are highly consistent among species. Due to lesion formation through fast processes of thermal injury on the cardiomyocytes within the zone of ablation, the histological appearance of lesions created by RF or cryoablation is easily discernible with an elastin trichrome stain and characterized by homogenization of the cytoplasm of affected cardiomyocytes. Under optimal staining conditions, thermally denatured collagen shows swelling and loss of microfibrillar texture (hyalinized) and may stain slightly basophilic or black with hematoxylin or Gomori’s elastin trichrome, respectively (not observed with cryoablations). The acute and subacute appearance of the ablated cardiomyocytes is indistinctive from coagulative necrosis; however, some pathologists reject using the term necrosis due to the lesion being formed through acute thermal-induced coagulation/denaturation of cellular components rather than through the classical events which define necrosis. Reference to the lesion, though, is usually “necrosis” or “coagulative necrosis.” Although the purists who avoid using the term “necrosis” may be correct from the standpoint of mechanism, it is our opinion that the terms “necrosis” and “coagulative necrosis” clearly portray the features of the lesion and the use of the term “necrosis” is present in the literature dealing with histological characterization of ablation lesions (Aupperle et al. 2005).
In the acute to subacute ablation lesion, the appearance of a distinct zone of coagulative necrosis is accompanied by variable amounts of edema and hemorrhage separating individual cardiomyocytes (Figures 2 and 7). The periphery of the acute and subacute ablation lesion usually contains a zone of coagulative myocytolysis (“contraction band necrosis”). The transition between the zone of contraction coagulative myocytolysis and nonnecrotic myocardium is sharply demarcated representing the transition from isotherms capable of producing cardiomyocyte degeneration and resulting in complete electrical block to those which do not result in cellular degeneration, at less than 50°C for RF lesions (Haines and Verow 1990; Nath, DiMarco, and Haines 1994). Endothelial denudation of the endocardium is usually observed in acute lesions. Decreased endothelialization of the endocardial surface is rare beyond 7 days after treatment; however, it may be observed if the study protocol involves an intracardial procedure, such as electroanatomical mapping, intracardiac pacing, or contrast pulmonary venograms, on or around the day of euthanasia. The loss of endothelium is usually primarily procedurally related rather than specifically related to the ablation treatment.
Masson’s trichrome-stained sections of a discrete radiofrequency ablation lesion in the right ventricle (RV) at 10 days after ablation. (A) The partial mural lesion is well demarcated and emanating from the endocardial surface (top). The well-demarcated staining characteristics of the periphery of the lesion are due to a rim of fibroplasia between the zone of necrosis and the nonnecrotic myocardiocytes. (B) Edema and hemorrhage are separating cardiomyocytes which are exhibiting marked sarcoplasmic coagulation. (C) Secondary bundles of cardiomyocyte and individual cardiomyocytes are separated by edema, and hemorrhage is present in coalescing areas. The endocardial surface of the edge of the zone of necrosis is shown. There is a thin thrombus attached to the endocardial surface.
Lesion sites should be assessed for endocardial erosions and histologic changes consistent with partial mural or transmural perforation. Lesions associated with steam pops have an acute histological appearance which correlates with their gross appearance; there is marked cavitation of the lesion with disarray of the remaining cardiomyocytes, and frank hemorrhage is usually a marked feature.
At 5 to 7 days after treatment, fibroplasia can be observed in its early stages. Fibroplasia begins at the edge of the ablation lesion at the border of the zone of coagulative myocytolysis with the viable, nonaffected myocardium. The fibroplasia progresses internally with increased time points after treatment. At 30 days after treatment, a well-defined zone of fibrosis with possibly retained islands of necrotic cardiomyocytes, depending on the size of the lesion, is typical (Figure 8). At 60 days after treatment, the presence of retained necrotic cardiomyocytes is rare, and at 90 days after treatment, the ablated zone is totally replaced with fibrosis. The definition of the zone of coagulative myocytolysis disappears with increasing time points beyond 7 days after treatment due to remodeling from the fibroplasia and fibrosis. Nonnecrotic cardiomyocytes bordering remodeled, fibrotic ablation lesions tend to be mildly to moderately shrunken and often contain sarcoplasmic vacuoles.
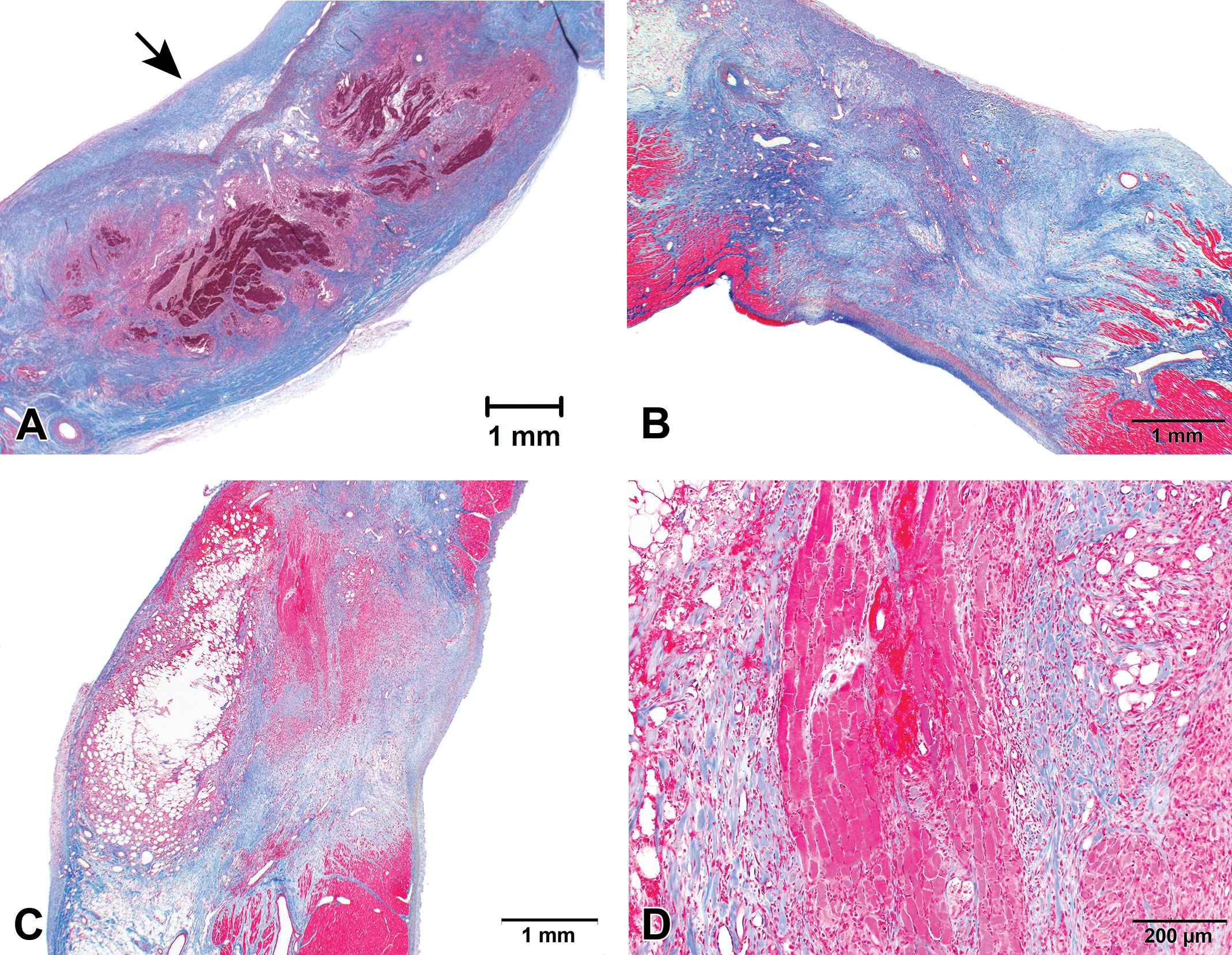
Masson’s trichrome-stained sections of cardiac radiofrequency ablation lesions 30 days after treatment. (A) The ablation lesion is transmural through the cavotricuspid isthmus (CTI) with a zone of endocardial hyperplasia (arrow). Coalescing islands of necrotic cardiomyocytes are retained within the central area of the lesion. (B) The discrete lesion on the posterior left atrium is composed of a transmural zone of fibrosis. (C) The transmural ablation lesion in the CTI near the atrioventricular groove is composed of fibrosis with a focal area of necrotic myocardiocytes. (D) The necrotic cardiomyocyte focus in C is shown surrounded by a zone of fibroplasia.
Inflammation can be variable in severity but is usually mild. The inflammatory component of a subacute lesion tends to be lymphohistiocytic at the edges of the lesion with lesser numbers of granulocytes admixed throughout the lesion. Inflammation usually decreases over time as healing occurs; however, certain lesions can be rich in multinucleated giant cells, particularly if there are clefts of mineralization, which is a variable feature of ablation lesions. Foci of multinucleated giant cells should always be investigated for foreign material as exfoliated polymers or fragments of other material from catheters or ancillary devices (wires and sheaths) can occasionally be present at the sites of ablation lesions.
Lesions should be assessed for endocardial fibrin/platelet thrombi due to their potential for embolization. Thin regions of fibrin and platelet aggregation are commonly observed on the endocardium of acute and subacute lesions and are rarely present at time points beyond 7 days (Figure 7). However, similar to the effects on endothelial coverage of the endocardium, small, thin thrombi which tend to be platelet rich are commonly observed instrumentation of the heart has occurred on or around the day of euthanasia. Endocardial hyperplasia should be assessed at chronic time points particularly in PV lesions as PV stenosis is a safety concern. Endocardial hyperplasia is usually negligible to mild; however, it can be marked particularly within PV ostia or more proximal areas of the PVs.
The histological assessment of multiple levels of PV lesions or lesion lines must be done in a manner in which there is confidence in following the distribution of the lesion serially across the levels in order to conclude whether or not anatomical PV isolation, defined by a contiguous transmural lesion around the circumference of the PV antrum, has been achieved (Figure 5). The presence of a circumferential, transmural lesion in a single level is rare; however, that does not mean a circumferential, transmural lesion is not present and assessment of continuity relies on step levels as necessary. The histological levels from PV antrum through the termination of the cardiomyocyte sleeves should include a system for tracking lesions across levels. A straightforward method is to assess the PVs in sectors analogous to a clockface and to stack the lesional areas of the individual sections to determine whether a circumferential, transmural lesion is present. This method usually allows for confidence of the assessment of circumferentiality of the lesion, allowing for identification of the location of gaps with an estimate of any gaps relative to the circumference of the PV. Assessment of lesion lines should involve tracking any gaps in continuity or transmurality across step sections at designated intervals. Since the objective of these assessment of lines is often to validate data from electroanatomical mapping systems, the minimum dimensions of and gaps in continuity and transmurality should be determined morphometrically.
Morphometrical Assessment
Dosing studies and equivalency studies which aim to correlate ablation system parameters with lesion characteristics rely on morphometry of the gross and/or histological sections of the resulting ablation lesions. For these purposes, morphometry should be performed on the midlongitudinal cross section of each lesion in an appropriate target. Directly measured dimensions which have been used for comparison include maximum lesion depth, maximum lesion width, and cross-sectional lesional area (Yokoyama et al. 2006). Comparing the volume of lesions has been a commonly employed approach for equivalency studies particularly in the in vivo thigh model. The method for estimating volume utilizes formulas which approximate the volume of an ellipsoid based on the measurements presented in Figure 9. Many studies and data for regulatory submissions have utilized the formula
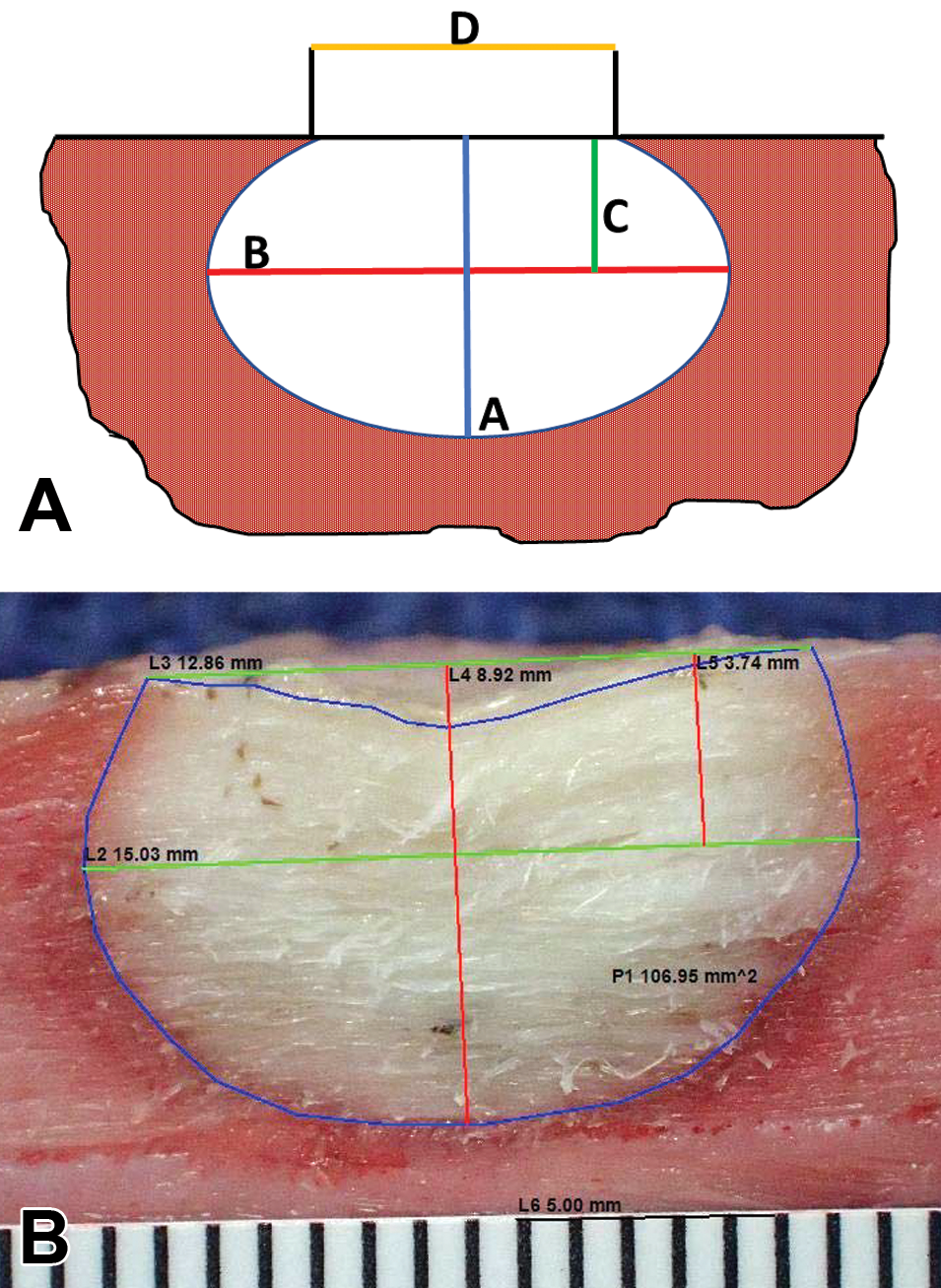
(A) The morphometrical measurements to estimate the volume of an ellipsoidal ablation lesion are shown. A = maximum depth, B = maximum width, C = depth from the surface to the plane of the maximum width, and D = width at the surface. (B) An example of an ablation lesion from an acute in vivo porcine thigh model with measurement depth (red lines) and width (green lines) measurement overlaid on the photograph. The lesion was made with a radiofrequency catheter 3 hr prior to euthanasia. The blue line denotes the area of total nonstaining of 2,3,5-triphenyltetrazolium chloride of the lesion.
The morphometrical determination of PV stenosis relies on measuring the luminal area of the histological sample as well as the endocardial elastin area which defines the pretreatment luminal area (Figure 6). The formula utilizing those measurement is Stenosis = (1 − (Luminal Area − Elastin Area))100.
Safety
The goal of the preclinical safety assessment is to characterize the potential adverse events associated with the intended use of the cardiac catheter ablation system. Intracardiac ablation catheters have inherent properties which can make them “unsafe.” Perforation of the heart, production of lesions at inappropriate cardiac locations, and production of collateral lesions on adjacent tissues and organs are possible with every RF and cryoablation system, as they can be induced by improper catheter handling and ablation parameters (Peichl et al. 2014; Zellerhoff et al. 2010; Andrade et al. 2014). Intracardiac ablation also relies on ancillary devices which have their own safety risks which can confound the assessment of the safety of the test system. Wires and sheaths can induce mechanical trauma along the usage pathway. Diagnostic catheters are usually used for the acquisition of electrograms and possibly pacing during the ablation procedure and have similar potential for trauma (Mansour et al. 2004).
Access to the heart is gained from the RA via the venae cavae, and in order to access the left side of the heart, perforation of the IAS and sheath placement through the perforation is necessary. This transseptal access is a particular source of adverse events from ancillary equipment, as a misdirected perforation needle can perforate the atrium and/or perforate the aorta (McElderry and Yamada 2009). Therefore, the preclinical safety study is dependent on therapy being delivered by an experienced catheter ablationist with the parameters of lesion generation tightly controlled and within the normal instructions for use (IFU) which were determined by time/energy dosing studies during the research and development phase of the system. For RF-based systems, these parameters include power (for power-controlled systems), temperature (for temperature-controlled systems), duration at each discrete location, force (for systems with force sensing), and saline flow rate (for open-irrigated [OI] systems). The role of ablationist in preclinical safety studies is almost exclusively fulfilled by a physician electrophysiologist (EP) with extensive clinical experience ablating the anatomical targets of the study. The catheter handling experience of the electrophysiology and the ability to interpret electrograms from a cardiac recording system and electroanatomical data from a cardiac mapping system are paramount in ensuring the success of the preclinical safety study.
Time points of the preclinical safety study of cardiac ablation catheters must be chosen in order to assess the short-term, subacute effects of the therapy as well as the maturation, remodeling, and healing of lesions at a chronic time point. The short-term, subacute time point allows morphological characterization of lesions resulting from the therapy which might resolve and heal over time. Such transient lesions, which include endocardial erosions and endocardial-attached fibrin/platelet thrombi, are, however, important in determining the safety profile of the ablation system. Additionally, the chronic time point not only shows healing and the remodeling process of ablation lesions but also identifies other possible negative outcomes of collateral damage, such as esophageal fistulas and/or PV stenosis (Van Driel et al. 2014). In order to develop an accurate safety profile, studies are commonly designed with time points of 7 and 30 days after treatment. The 7-day time point allows for local, collateral, and downstream lesions to easily be identified, as the maximum histological changes are often present at this time point. While the lesions in these areas of interest are not as distinct at the 30-day time point due to healing and remodeling, the 30-day time point captures a snapshot of the resolution and remodeling of lesions without extending the study to a later time point, such as 90 days, in which healing would be more complete. When time points longer than 30 days are elected, it is usually to increase the confidence in assessing maximal remodeling of PVs and maximal PV stenosis.
As mentioned above, the preclinical safety study for cardiac ablation catheters must assess the local, collateral, and downstream effects of the therapy. Table 1 summarizes the approach to the pathology component of the study in assessing the effects at these regions. Local effects are those which result at the tissue–catheter interface as well as the subjacent myocardial tissue. Collateral effects occur as a result of energy transfer to tissues beyond the target of ablation, and downstream effects are secondary to thromboembolic events.
Pathology Approaches to Aid in the Determination of Safety of Cardiac Ablation System.
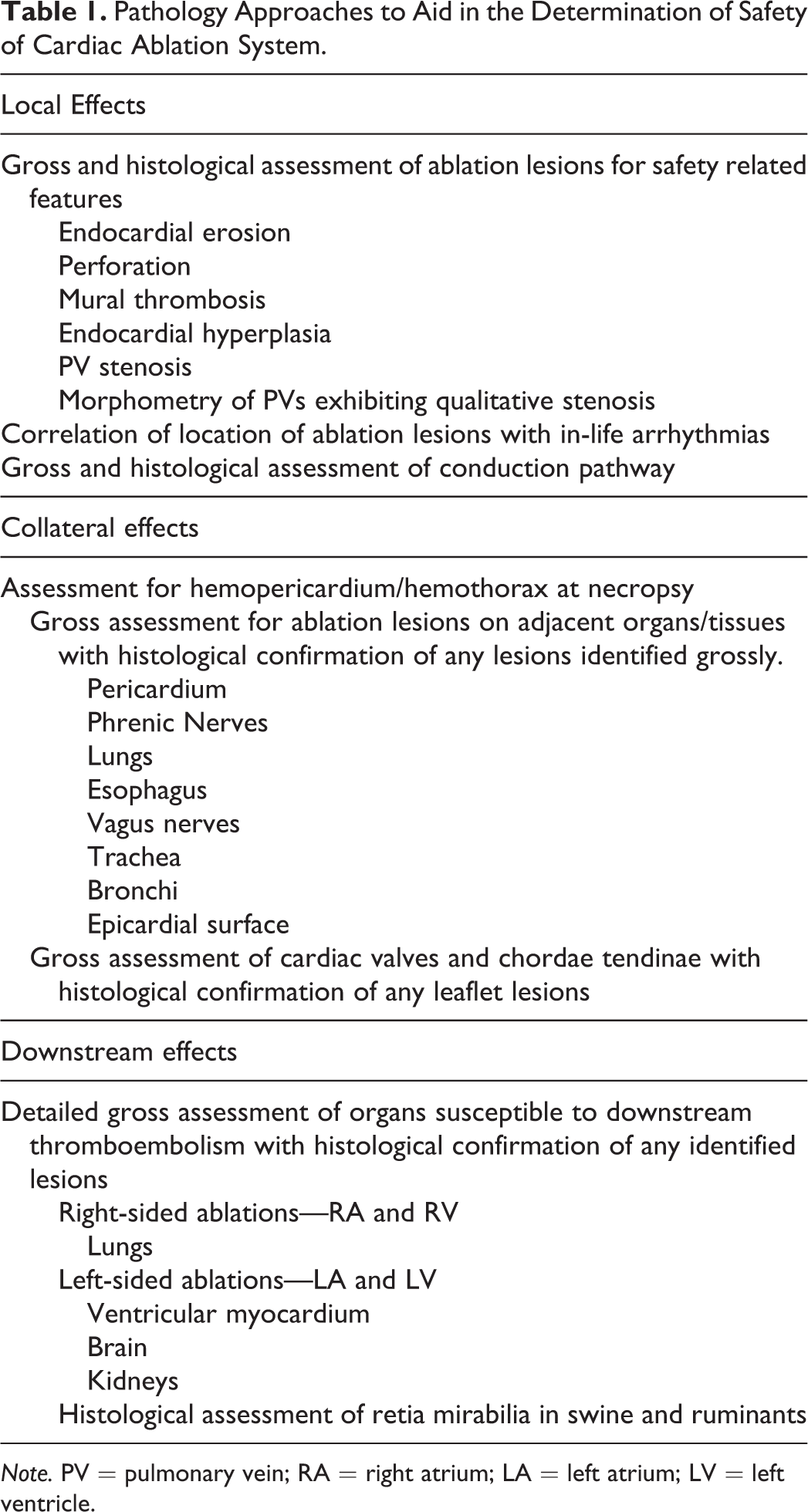
Note. PV = pulmonary vein; RA = right atrium; LA = left atrium; LV = left ventricle.
Every medical device safety study either directly or indirectly has morbidity and mortality as an end point. The study pathologist must approach necropsy and histological assessment of each early death animal and each animal which exhibits in-life adverse events from the standpoint of determining whether a cause of death or the in-life adverse event is related to the local, collateral, or downstream effects of the Test Article. This involves the study pathologist reviewing the animal record and being aware of any ablation treatment complications, in-life adverse events, and the physical and neurological examination results. The study pathologist must also have knowledge of the procedures and instrumentation which were used and on what study days. This allows interpretation of any lesions in light of the specific location of any ancillary catheters used for electrogram acquisition and pacing as well as interpretation of any lesions with regard to time points relative to instrumentation use. For instance, intracardiac instrumentation on the day of euthanasia may result in acute lesions which would be inappropriate for a chronic time point study.
Local Safety Assessment
The local safety assessment involves gross and histological evaluation of each ablation lesion site. Upon opening the thorax, the pericardium and thorax should be assessed for blood and effusions. The volume of any hemopericardium or hemothorax should be measured, and there should be an interrogation of the epicardial surface for potential sources of perforation. Sites of ablation lesions which are obvious on the endocardial surface are not the only potential sources of hemopericardium. Perforations from wires, transseptal needles, and ancillary catheters are also potential sources. In cases of hemopericardium or hemothorax, the transseptal perforation site should always be examined and the adventitial surface of the aorta interrogated for small perforations. Examination of the fresh heart at necropsy in the preclinical safety study is usually best limited to the epicardial surface especially for acute and subacute studies when lesions can be easily disrupted and dimensions distorted by excessive handling and trimming prior to fixation. Fixation of the heart by perfusion through cannulation of the aorta and perfusion through the coronary arteries provides the advantage of firming the myocardium and makes ablation lesions more contrasting to the surrounding nonablated tissue. If staining with a vital stain, such as TTC, is desired, it can also be done through coronary perfusions and should be done after adequate rinsing of the vasculature and prior to formalin perfusion fixation. Heparinization of the animal prior to euthanasia facilitates successful intracoronary perfusion, and recirculation of TTC during the color development phase aids in uniform staining throughout the myocardium.
The endocardial, epicardial, and transverse myocardial appearance of each ablation lesion should be photographed at the time of lesion trimming, and the characteristics and location of each lesion should be described and put into context of the location relative to anatomical structures. Lesion sites should be dissected free of the myocardium in a manner appropriate for the lesion type as described above. The gross patency of treated PVs should be assessed.
Of the histological parameters which are typically assessed for cardiac ablation lesions, endocardial erosion, perforation, and the presence of endocardial thrombi are the most relevant to the safety assessment for discrete lesions. All parameters discussed in the previous section should be scored in order to ascertain whether there are significant histological differences between the ablation lesions created by the Test Article compared to the Control Article. Endocardial hyperplasia of PV ablation sites is a unique safety concern at those locations (Taylor et al. 2000). Any endocardial hyperplasia resulting in appreciable PV stenosis should be documented with micrographs. Thresholds of significance for PV stenosis within the acceptance criteria of preclinical safety studies have typically been greater than 70% stenosis (Packer et al. 2005). Morphometry should be performed on the elastin trichrome-stained section of any PV level which exhibits qualitatively significant stenosis.
Assessment of the gross and histological response of the cardiac ablation lesions should be made in light of any arrhythmias identified after treatment. Correlation of postablation arrhythmias with cardiac lesions requires an understanding of the conduction pathway and potential arrhythmogenic substrates. Unintended ablation of sensitive conduction pathway structures usually results in a form of conduction block when the responsible lesion is below the sinoatrial (SA) node and various forms of SA dysfunction occur when the SA node is affected (Calkins et al. 1999; Ong et al. 2005). A complete assessment of the cardiac conduction system should be performed on any early death due to sudden cardiac death or any early death for which there is no discernible cause of death based on necropsy findings. Thorough histological assessment involves assessment of multiple levels of the SA node, lateral atrionodal bundle, AV node, His bundle, right bundle branches, and left bundle branches. Arrhythmias and conduction disturbances resulting from misplaced ablation lesions are usually considered operator error and do not preclude the conclusion of safety of an ablation system. Documentation of ablation lesions associated arrhythmias is critical in identifying trends with certain protocols as well as for being able to document a procedural-related cause for an adverse event.
Intracardiac lesions secondary to catheter-induced trauma rather than ablation need to be documented and reported as part of the preclinical safety study. Valvular lesions are of particular interest as the ability for the ablation catheter to be placed at distinct locations within the cardiac chambers without significant damage to the leaflets of the right (tricuspid) and left (mitral) AV valves is necessary. Any entrapment within chordae tendinae is also of particular concern.
Collateral Safety Assessment
Lesions in organs and tissues adjacent to sites of cardiac ablation can occur due to thermal conduction or transfer of the energy of the particular modality of ablation. The necropsy should involve investigating all structure within the thorax for potential collateral ablation lesions. Typical sites of collateral ablation lesions include the pericardium, lungs, phrenic nerves, esophagus, trachea, and bronchi (Zellerhoff et al. 2010; Pappone et al. 2004; Andrade et al. 2014). The location of the adjacent cardiac structure should be documented in order to correlate the collateral lesion with a particular cardiac ablation lesion. The presence of collateral ablation lesions does not necessarily preclude the conclusion of safety of the ablation system at the parameters used in the study. The relevance of collateral ablation lesions to safety is usually made within the context of whether or not the lesion has the potential to be clinically significant. The study pathologist plays a key role in this determination. Ablation lesions to certain structures, such as the pericardium, are inherently clinically silent. The clinical relevance of collateral lesions in other organs is dependent on the severity and extent of the lesions. All collateral lesions identified at necropsy should be photographed and sampled for histology in order to document the extent of the lesion within the organ and to aid in the determination of clinical relevance. The conclusion of clinical relevance within the preclinical study can be aided by supporting data such as physical examination results, weight gain, and clinical pathology.
Collateral lesions to the lung are common and usually result from atrial or right ventricular freewall ablations and may be associated with left atrial and PV ablation lesions. Acute and subacute collateral pulmonary lesions are well demarcated and characterized by necrosis and hemorrhage extending from the pleural surface typically only a short depth into the parenchyma due to the marked insulating properties imparted by the parenchymal architecture of the lungs (Figure 10A and B). At chronic time points of 30 days and beyond, collateral pulmonary lesions are usually poorly discernible and consist of small foci of pleural and subpleural fibrosis.
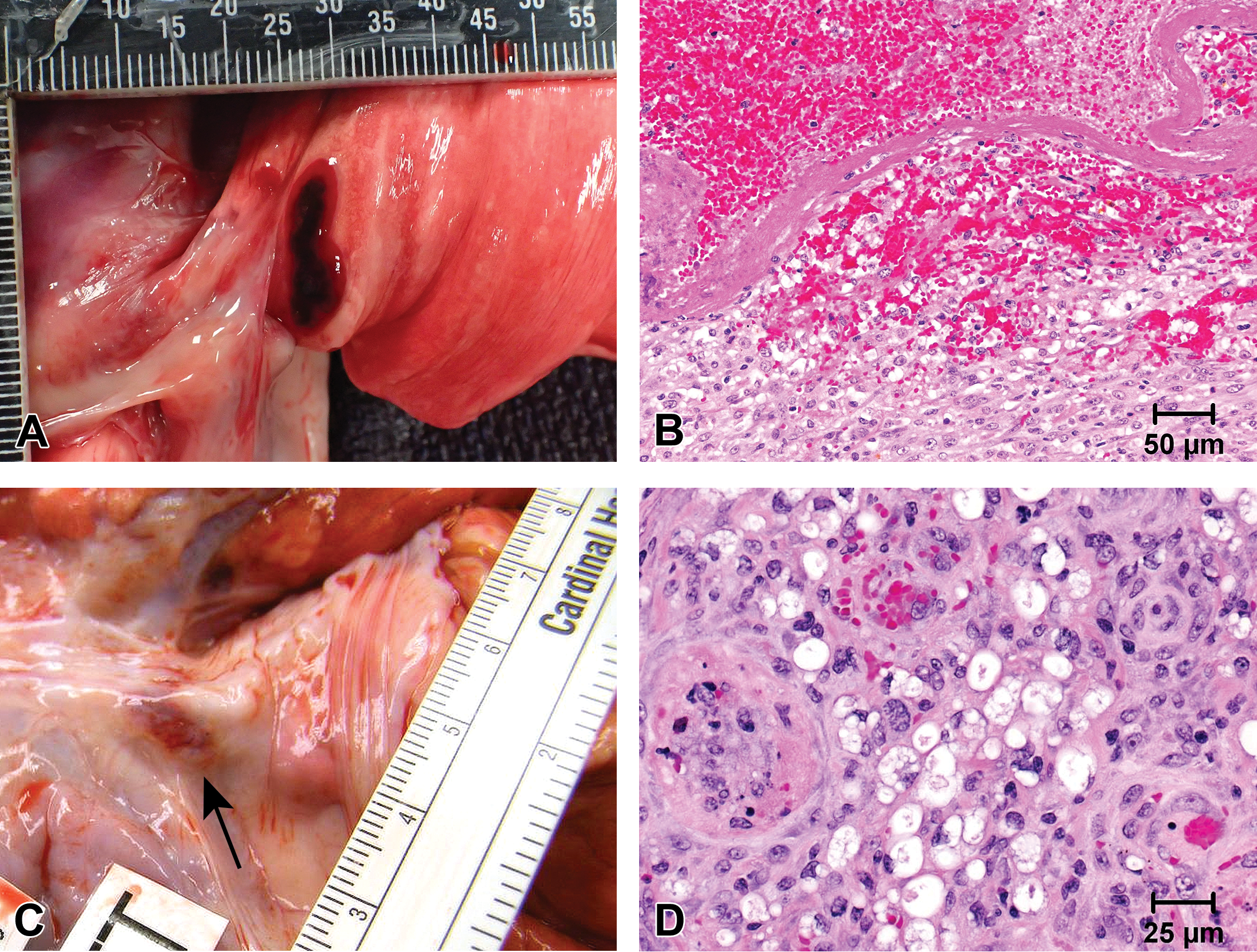
Gross and histological appearance of collateral ablation lesions. (A and B) The medial pleural surface of a lung lobe of a dog which was treated with radiofrequency ablation in the left atrium 7 days prior exhibits a collateral ablation lesion consisting of hemorrhage and necrosis. (C and D) The cross section of the left phrenic nerve (arrow) of a pig exhibits vacuolation, Schwann cell hyperplasia, mild inflammation, and hemorrhage 7 days after cryoablation of the left pulmonary vein (LPV) and right pulmonary vein (RPV).
Collateral lesions to the phrenic nerves, when present, are nearly always secondary to ablation at atrial sites. Collateral phrenic nerve lesions are of particular interest in the determination of safety due to the ability to result in phrenic nerve palsy, diaphragmatic dysfunction, and dyspnea (Kuehne et al. 2013). Acute and subacute phrenic nerve lesions are identified grossly as discrete foci of hemorrhage and edema within the pericardium subjacent to the affected nerve. The histological neurodegenerative changes are highly variable and include intrafascicular hemorrhage and edema, vacuolation, degeneration, and minimal inflammation (Figure 10C and D). At more chronic time points, the phrenic nerve lesions are usually only evident as subtle foci of pericardial fibrosis and are composed histologically of intrafascicular and perifascicular fibrosis. A detailed gross assessment of the diaphragm should be made in each case of collateral phrenic nerve injury, and correlation of pathology data to in-life assessments of diaphragmatic function should be made.
Collateral lesions to the esophagus, trachea, and bronchi usually are secondary to left atrial ablations and are of particular interest due to their potential to form fistulas with the cardiac ablation lesion (Pappone et al. 2004). The acute and subacute appearance of collateral lesions in the esophagus, trachea, and bronchi consists of well-demarcated foci of serosal hemorrhage and discoloration (Figure 11C–F). The mucosal surface of the lesional area should be assessed grossly to document any transmurality of the lesion. The histological assessment should detail the extent of the lesion through the wall of the affected organ and the documentation of any tracts through the affected wall.
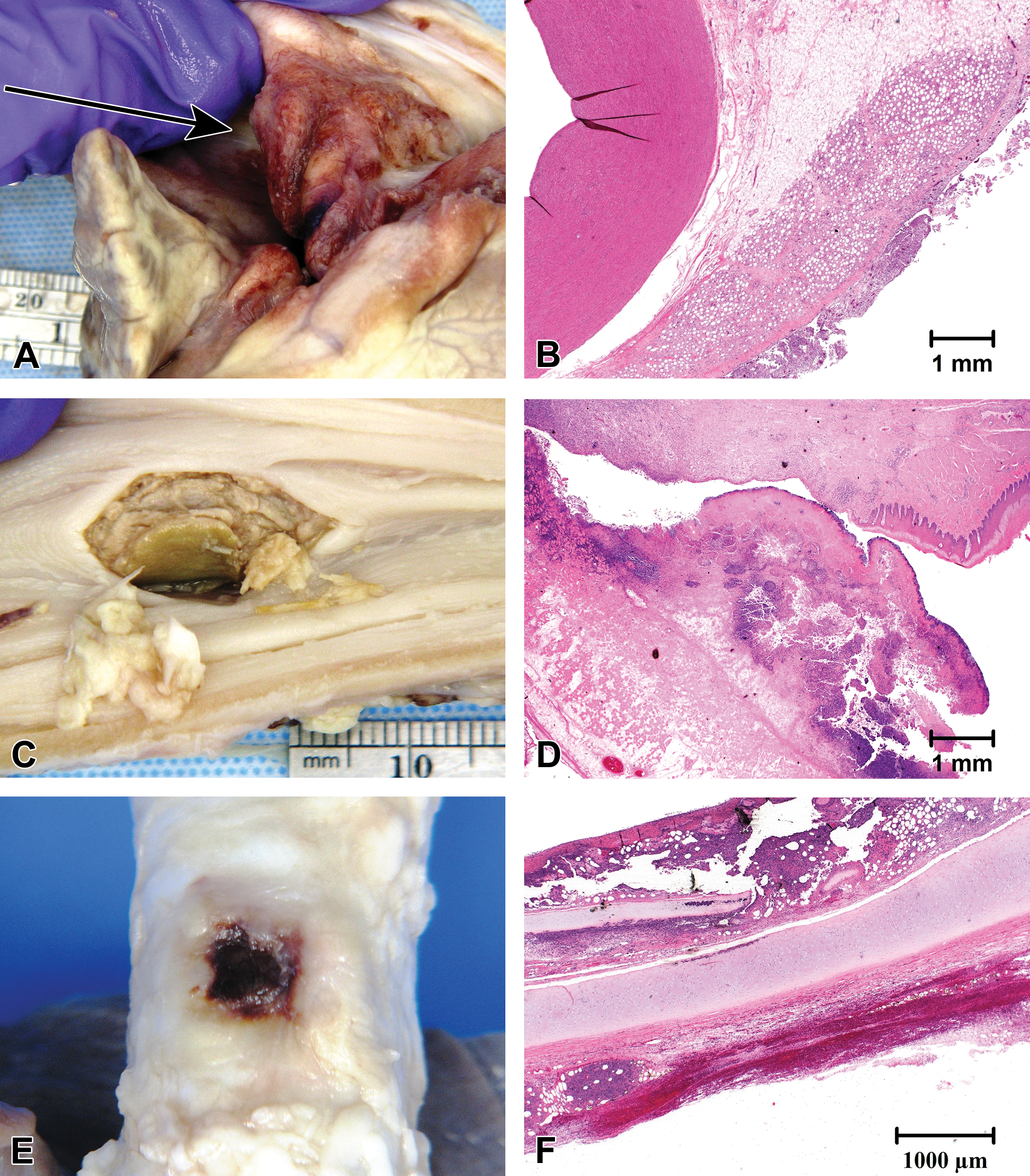
Gross and histological appearance of collateral ablation lesions. (A and B) There is hemorrhage, inflammation, and adventitial proliferation of the aorta 7 days after treatment of the pig with cryoablation in the right pulmonary vein (RPV) and left pulomonary vein (LPV). (C and D) A fistulous tract is present in the esophagus, as viewed from the mucosal surface of a pig 7 days after cryoablation in the RPV and LPV. (E and F) Transmural necrosis with serosal hemorrhage is present in the trachea of a dog 7 days after radiofrequency ablation at multiple locations in the left atrium and right atrium. All histological samples are hematoxylin and eosin stained.
Collateral lesions to the aorta may also occur with ablation in the LA and PVs and are usually composed of hemorrhage with mural to partial mural necrosis and adventitial inflammation (Figure 11A and B). Similar collateral lesions are possible in pulmonary arteries. Degeneration of the adventitial and tunica media can occasionally be observed in pulmonary arteries histologically when the arteries remain part of the processed tissue block with examination of PV ablation lesions.
Similar to the phrenic nerves, branches of the vagus nerve dorsal to the heart, particularly at locations associated with the esophagus, are sites of potential collateral ablation lesions. Collateral lesions to the esophagus or in-life clinical signs of gastroparesis should trigger interrogation of the vagus nerves in the mediastinum (Shah et al. 2005).
Collateral ablation lesions can occur on the epicardial surface of the basilar portion of the ventricles secondary to lesions made in the atrial appendages, CTI, and caudal vena cava. The lesions are often identified by well-circumscribed foci of hemorrhage within the adipose tissue of the AV groove. The lesions usually only extend a short depth into the myocardium and usually do not involve elements of the cardiac conduction system. Intracardiac lesions secondary to catheter-induced trauma rather than ablation need to be documented and reported as part of the preclinical safety study. Valvular lesions are of particular interest as the ability for the ablation catheter to be placed at distinct locations within the cardiac chambers without significant damage to the leaflets and chordae tendinae of the right (tricuspid) and left (mitral) AV valves is necessary.
Downstream Safety Effects
Organs downstream in arterial flow from the chambers of ablation are sensitive to thromboembolic events which may arise from thrombi deposited on the endocardial surface of the site of ablation and potentially at the transseptal access site or from coagulum formed around the catheter tip at the time of ablation (Matsudaira et al. 2003; Doppalapudi, Yamada, and Kay 2009). This coagulum also sometimes referred to as “soft thrombus” is formed from the heating of the blood pool around the catheter tip. The formation of coagulum with RF ablation is greatly reduced with irrigated catheter tips which cool the tip through the flow of saline during the ablation (Weiss et al. 2001).
The organs of particular interest to the effects of downstream thromboembolism include the ventricular myocardium, kidneys, and brain for left-sided procedures and the lungs for right-sided procedures and arguable for left-sided procedures via the bronchial arteries. The approach for assessment for potential thromboembolic lesions in these organs involves detailed gross examination of thin transverse slices (“breadloafing”) as well as detailed surface examination. The breadloafing procedures are best carried out after fixation in the kidneys, brain, and ventricular myocardium. Fixation facilitates the ability to ascertain sections ideally at a maximum of 3 to 4 mm thickness. The macroscopic assessment of the lungs may be made at necropsy or after intratracheal perfusion fixation of the lung parenchyma. Examination of the pulmonary arteries for thrombi is best accomplished by longitudinal incision prior to breadloafing. Breadloafing prior to fixation of the lungs is challenging due to the consistency of the parenchyma. The color changes of the lung parenchyma from formalin fixation provide less contrast between normal and abnormal regions than can be observed in the fresh tissue.
Any gross lesions identified from the detailed breadloafing should be submitted for histological assessment (Figure 12). The histological reporting should aim to conclude whether or not the identified lesions are consistent with downstream thromboembolism as opposed to background lesions consistent with the test system. The strategy for regulatory approval may involve assessment of a certain number of samples from organs susceptible to downstream thromboembolism regardless of the presence of gross lesions. Specific regulatory agencies have also requested additional samples of the celiac, cranial mesenteric, and caudal mesenteric arteries for intraluminal thrombi as well as samples of their associated lymph nodes. If swine or ruminants are the test system, then the retia mirabilia should be sampled for histology regardless of lesions, as gross evidence of intravascular thrombi within the retia mirabilia is difficult to discern. The assessment of the retia is not a substitute for evaluation of the brain, as small thromboemboli can flow through the retia unfiltered.
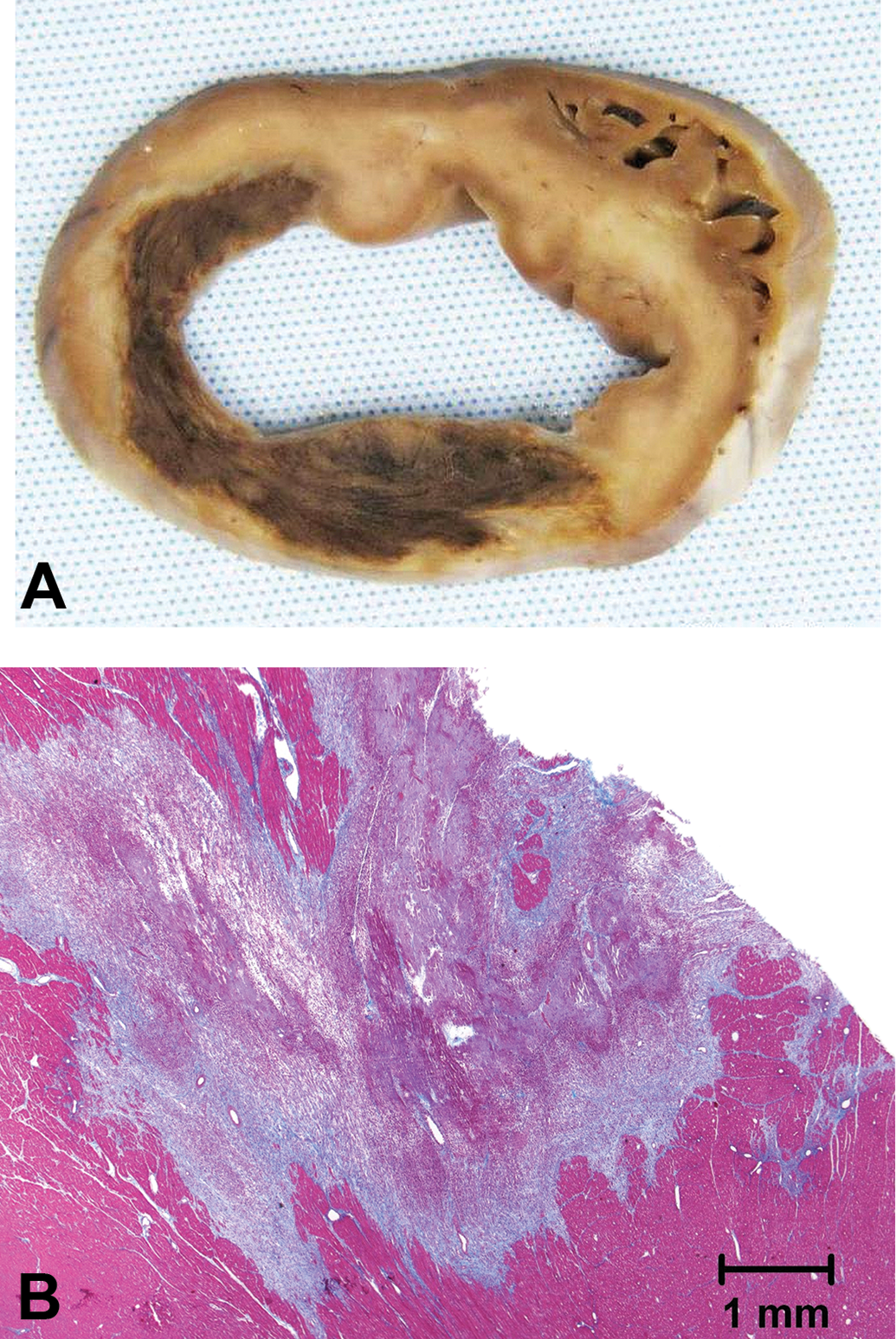
The gross (A) and histological (B; Masson’s trichrome) appearance of a myocardial infarct secondary to downstream thromboembolism 8 days after treatment of the pig with a cryoballoon ablation catheter in the left pulmonary vein (LPV) and right pulmonary vein (RPV).
Additional Elements of the Safety Evaluation
OI RF ablation catheters utilize a continuous flow of saline with exits ports at or around the catheter tip in order to cool the tissue/tip interface and prevent coagulum formation. Saline flows at a continuous low rate when the power is off and a higher continuous rate when the power is on. Fluid overload and resultant pulmonary edema have been identified as a safety concern by regulatory agencies for OI RF catheters. The addition of a histological sample regardless of the presence of gross lesions from each lung lobe can be used as an adjunct for investigations into possible fluid overload resulting in secondary pulmonary edema in acute, nonsurvival, or short-term subacute studies. Determination of the percentage of lung water can also be performed in studies which aim to definitively determine any relationship between the volume of irrigation saline infused and pulmonary fluid.
The pathway of the RF ablation catheter should not be ignored. The gross evaluation should also involve assessment of the caudal vena cava along its entire length from the junction with the heart to the iliofemoral veins (Kent et al. 1994). Perforation of the caudal vena cava should be considered in cases of rapid development of hemodynamic instability during ablation procedures especially in protocols in which catheters are removed and reintroduced into the animal often.
Conclusions
Preclinical studies of intracardiac ablation system are highly dependent on pathology to assess correlates of effectiveness and safety. Pathology assessment of cardiac ablation sites is the gold standard for determining the presence and extent of the ablation lesion. These data can be used in investigations of titration of ablation parameters including power, energy, temperature, time, contact force, and irrigation flow rate. Histological results can also be used to determine the usefulness of certain data acquired during ablation lesion formation or electroanatomical mapping data in predicting a successful ablation lesion. After the research and development phase has identified the final catheter and ablation system design as well as system parameters for intended use, the safety of that final design system operated within the confines of the upper limits of the IFU is evaluated in a large animal model prior to regulatory approval for use in a clinical trial or market release. The pathology component of the preclinical safety study must determine the local, collateral, and downstream effects of the ablation system, and the pathologist must interpret those findings in context of relationship to any in-life adverse events as well as clinical risk assessment. With continued investigations of alternate ablation energy modalities as well as parameters to decrease total case time and improve the success rate of intracardiac ablation therapy, preclinical pathological assessment will continue to play a critical role in R&D and regulatory approval strategies and pathways for emerging intracardiac ablation technologies.
Footnotes
Author Contributions
Authors contributed to conception or design (WS, SR, MR); data acquisition, analysis, or interpretation (WS, SR, MR); drafting the manuscript (WS); and critically revising the manuscript (SR, MR). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
