Abstract
Neonatal hemochromatosis (NH) is a rare disease of iron metabolism that starts at intrauterine period causing liver failure and extrahepatic siderozis. The etiology of NH has not been understood exactly, yet it is accepted that a maternofetal alloimmune disorder that leads to liver failure in fetus causes the illness. The prognosis of NH is generally bad and death is inevitable if left untreated. The efficiency of chelation–antioxidant coctail used in medical treatment is between 10% and 20% and these patients frequently need liver transplantation. In our study, we presented four newborn cases diagnosed as NH and treated medically. Of the four patients, one died of pulmonary hemorrhage and another died of multiorgan failure in the first week of hospitalization. The other two patients' clinical status and laboratory parameters recovered with medical treatment. However, since liver transplantation was not carried out, one of these patients died at the age of two and a half months and the other at eighth month due to sepsis. In this study, we would like to emphasize the importance of early liver transplantation in patients recovered with medical treatment.
Keywords
Introduction
Neonatal hemochromatosis (NH) is clinically defined as severe neonatal liver disease in association with extrahepatic siderosis in a distribution similar to that seen in hereditary hemochromatosis. 1 Considerable evidence indicates it to be a gestational disease with a dominant fetal liver injury feature. It has been called neonatal iron storage disease on account of abnormal accumulation of iron in liver and other tissues, and it is also called congenital hemochromatosis due to its origins before birth. 2 NH was originally described in 1957 and >100 cases have been reported. 3 Liver failure in the first 30 days of life is uncommon; neonates represent <2% of children who are listed for liver transplantation, but of these few patients, NH may be one of the most common causes of liver failure. 4
Most liveborn patients exhibit evidence of in-utero insult (intrauterine growth retardation and oligohydramnios) and many are born premature.5–7 NH is characterized by hepatocellular failure, occurring in the first days of life with coagulopathy, hypoglycemia, hypoalbuminemia, low fibrinogen, thrombocytopenia, anemia, and direct and indirect hyperbilirubinemia.2,4,8 Repeated occurrence in the offspring of women who have had one affected infant is a notable feature of NH. This apparent rate of occurrence of severe disease in siblings after the index case is 60% to 80%. 9 Counseling regarding future pregnancy has been determined by this high risk, with avoidance being often advised. 10
The current medical treatment for NH, based on antioxidants and iron chelator, is often ineffective and associated with severe adverse effects. 11 The prognosis of this condition is generally poor, and NH is a frequent indication for liver transplantation in the newborn. 11 Untreated, NH is often fatal. 12
In our article, four newborn cases with NH diagnosis and treated with chelation antioxidant treatment were presented under the light of literature. Our purpose is to emphasize the importance of early liver transplantation in patients with NH after they recovered clinically with medical treatment.
Patients
Patient 1
A 12 days newborn, born from a 34-year-old mother with repeated cesarean section (Gravida 4, para 3) as 3300 gram, was hospitalized to newborn unit of our hospital due to jaundice. In the history, there was oligohydramnios. There was a second generation kinship between the mother and father in the pedigree. Their third baby died of liver failure in the second month after birth (its etiology was not explained). The siblings living today had no health problem. In the physical examination; weight was 3500 g height was 49 cm, head circumference was 36 cm, overall condition was moderate and had spread oedema, the skin and sclera were icteric, and abdomen was soft with ascites and hepatomegaly. The other system examinations were normal. The laboratory values of the patient are given in Table 1.
Laboratory results of the patients
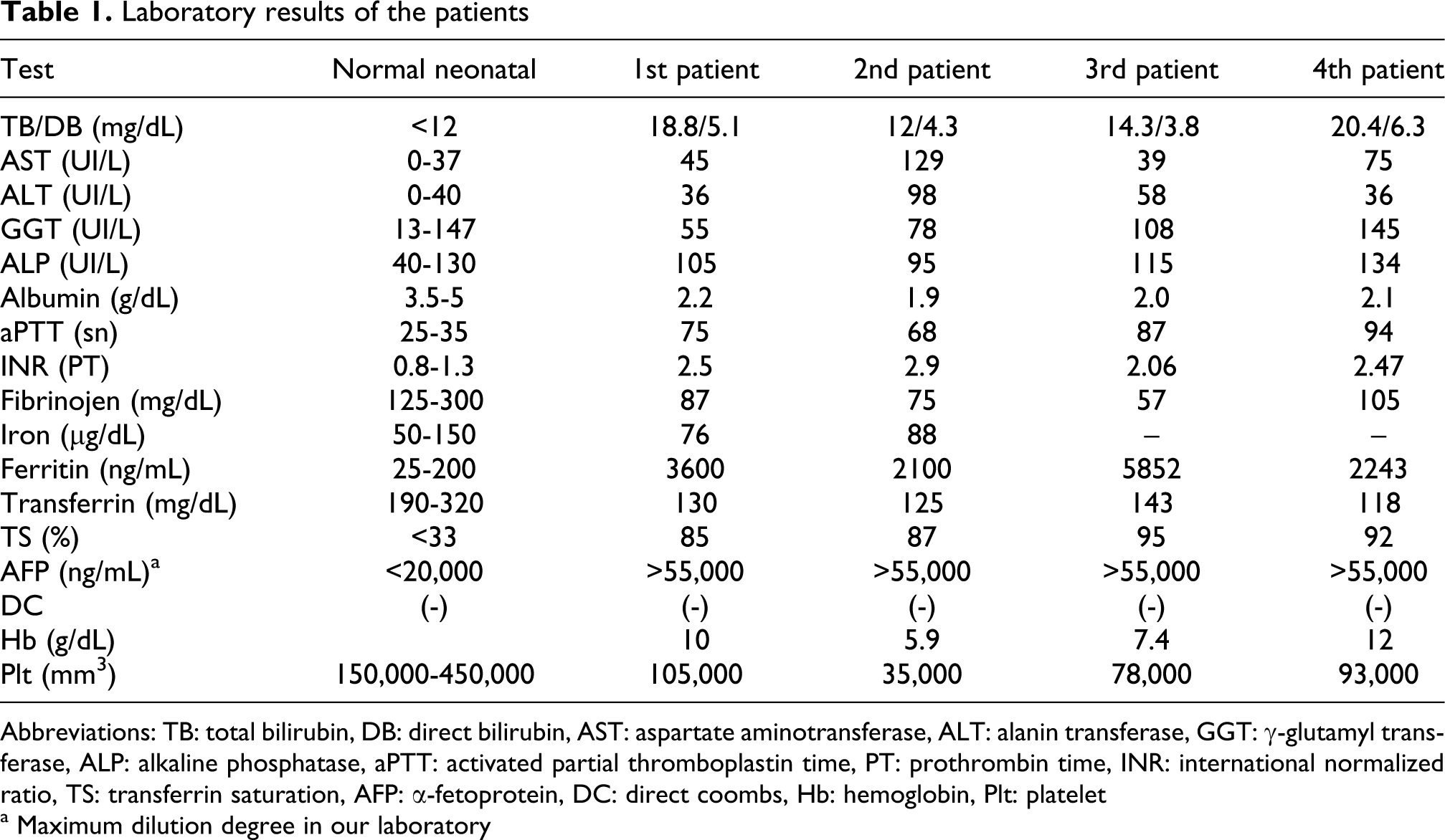
Abbreviations: TB: total bilirubin, DB: direct bilirubin, AST: aspartate aminotransferase, ALT: alanin transferase, GGT: γ-glutamyl transferase, ALP: alkaline phosphatase, aPTT: activated partial thromboplastin time, PT: prothrombin time, INR: international normalized ratio, TS: transferrin saturation, AFP: α-fetoprotein, DC: direct coombs, Hb: hemoglobin, Plt: platelet
a Maximum dilution degree in our laboratory
In ultrasonography (USG), there was homogeneous echogenicity in liver parenchyma and ascites. Gallbladder and extrahepatic bile ducts were normal. In the magnetic resonance imaging (MRI), it was defined that there was excessive accumulation of iron in liver and pancreas. HFE gene mutation was negative. Liver biopsy, carried out when coagulation test and thrombocyte count were normal, was reported to be complied with hemochromatosis. Desferrioxamine for chelation, N-acetyl cysteine as antioxidant, selenium and C and E vitamins were started. The patient, whose overall condition emeliorated after chelation-antioxidant cocktail, was followed for 8 months by pediatric gastroenterology clinic. However, the patient prepared for liver transplantation died in the eighth month of age due to sepsis.
Patient 2
A 25 days old girl baby, born normally on time from a 19 years old mother, applied to hospital with the complaints of jaundice and distended abdomen. There was no kinship between the parents. In the physical examination, the weight was 3600 g, height was 53 cm, head circumference was 36.5 cm, overall condition was poor, tachypneic, tachycardic. In mesocardiac focus, there were 2/6 systolic murmur, peripheric oedema, hepatomegaly and ascites. The laboratory values of the patient are summarized at Table 1.
In abdomen USG evaluation, there was ascites and increase in the liver parenchymal echogenicity. MRI and tissue biopsy were not done due to the inappropriate clinical status of the patient. The patient was supported with fresh frozen plasma, packed red blood cells (RBC) concentrate and platelet concentrate. The patient diagnosed as NH was started supportive treatment consisting of chelation-antioxidant coctail. The patient died on the fifth day due to pulmonary hemorrhage. The postmortem liver biopsy complied with hemochromatosis.
Patient 3
A girl baby, born from a 21 years old mother (Gravida 2, para 1) with cesarean section due to premature birth with 30 weeks gestation time, was accepted to the newborn unit. The patient had already had hydrops fetalis in intrauterine period. The first pregnancy of the mother was ended with abortus in the 25th weeks. There was no kinship between parents. APGAR score at first minute was 3 and 5 in fifth minute. In the physical examination, the weight was 1750 g, height 41 cm and head circumference was 28 cm, overall condition was bad, there was spread oedema and severe respiratory distress. Abdomen was highly distended and there was ascites. The laboratory values of the patient are summarized in Table 1.
The patient underwent exchange transfusion due to anemia and the rapid increase in the bilirubin at the first 24 hours was observed. Immunoglobulin less then 1g/kg was given intravenously. The patient diagnosed as hemochromatosis as a result of clinical and laboratory tests was started antioxidant chelation (N-acytl sistein, selenium, C and E vitamin desferroxamine). The patient died on the fourth day due to multiorgan failure. The postmortem liver biopsy was reported as hemochromatosis.
Patient 4
The patient born from a 27 years old mother from her first pregnancy with cesarean section on the 38th week due to intrauterine meconium was admitted to our newborn unit on the third day of birth with the complaint of jaundice. APGAR score at first minute was 7 and 9 in fifth minute. On physical examination, the weight was 3000 g, height 49 cm and head circumference was 33 cm, had mild peripheric oedema, the skin and sclera were icteric and other system examinations were normal. The laboratory values of the patient are summarized in Table 1.
In USG, there was increase in the liver parenchymal echogenicity and also ascites was detected. In MRI, it was defined that there was excessive accumulation of iron in liver and pancreas. The patient was diagnosed as hemochromatosis as a results of clinical and laboratory findings and desferrioxamine for chelation, N-acetyl cysteine as antioxidant, selenium and C and E vitamins were started. The patient, whose clinical condition was not stable, was supported with fresh frozen plasma, packed RBC concentrate and platelet concentrate. The patient died on the 75th day due to sepsis. The postmortem liver biopsy was reported as hemochromatosis.
The tests of the patients in terms of metabolic diseases that would lead to liver failure (blood gas, blood ammonia and lactate levels, α-1 antitrypsin urine organic acid analysis, tandem mass spectrometry) were normal.
Discussion
The cause of neonatal hemochromatosis has not been clearly defined. It has been claimed to be linked to an autosomal recessive inheritance 13 or a gestational alloimmune disorder. 14 It has been observed that after a woman gives birth to an infant with neonatal hemochromatosis, there is approximately an 80% probability that her subsequent pregnancies will be affected as well, which is too high to consider autosomal recessive inheritance.4,13,15,16 The precise pattern of inheritance and the gene locus associated with NH remain unknown.13,15,17 Another interesting theory is that of the alloimmune origin of the disease. The alloimmune theory suggests that maternal sensitization to a fetal liver antigen leads to immune injury of the fetal liver and subsequent iron mishandling and deposition in the liver and extrahepatic tissues.14,15,18,19 Whitington and Hibbard 14 reduced the severity of the disease by treating mothers whose recent previous pregnancy had ended in NH with high-dose intravenous immunoglobulin during gestation.
NH is nearly always accompanied by severe fetal liver injury, and one of its most common presentations is late second and third trimester fetal loss as evidenced by the gestational histories of women who have had a baby diagnosed with NH. 2 The clinical presentation of neonatal hemochromatosis can be seen with complications of pregnancy such as intrauterine growth retardation, oligohydramnios, placental edema and sometimes polyhydramnios.1,8 The pregnancy frequently ends with stillborn infants or infants who are premature or small for gestational age. 1 Liver disease is generally apparent within hours of birth, and NH is one of the most commonly recognized causes of liver failure in the neonate.20,21 In rare cases, the liver disease takes a prolonged course and is manifest days to weeks after birth. 22
The presenting findings are those of liver failure and usually multiorgan failure. Affected babies are frequently diagnosed as having overwhelming sepsis of the newborn even with negative cultures. Hypoglycemia, thrombocytopenia, anemia, marked coagulopathy, hypoalbuminemia and edema with or without ascites, and oliguria are prominent features.1,2,4 Jaundice with significant elevations of both conjugated and nonconjugated bilirubin develops during the first few days after birth.2,23 Typical biochemical findings include an extremely high serum ferritin level (usually >800 ng/mL), hypersaturation of available transferrin, with hypotransferrinemia and extremely high levels of α-fetoprotein (AFP; usually >100,000 ng/mL).2,15,23 An unusual characteristic given the degree of liver injury is low or absent serum transaminases. Hepatocellular synthetic insufficiency leads to hypoglycemia, coagulopathy (factors V and VII usually less than 10% of normal, low fibrinogen), hypoalbuminemia (usually less than 2 g/dL) and decreased iron-binding capacity. Liver histology is characterized by intense fibrosis and cirrhosis without acute necrosis. However, none of these findings is diagnostic of NH.2,19,23
Diagnosis of neonatal hemochromatosis is one of exclusion after tests for other causes of liver failure have been performed and deemed negative (Table 2). In neonatal hemochromatosis, serum ferritin levels are extremely elevated, AFP levels are high, and transferrin levels are low with high percentages of iron saturation. 1 There is hepatic and extrahepatic siderosis, which spares the reticuloendothelial elements. 24 Specifically, the iron deposits are in the heart, pancreas, exocrine and endocrine organs, intestines, and gastric and salivary glands. 4 This can be verified with histological examination of a biopsy of the buccal mucosa salivary glands or MRI of the liver, pancreas, spleen, and heart.4,25,26 Hepatic siderosis is not specific for NH, rather it is observed in the normal infant liver and more prominently in a multitude of liver diseases. Therefore, liver biopsy, a hazardous procedure in the presence of coagulopathy, is not diagnostic for NH.
Differential diagnosis in neonatal liver failure4

Abbreviations: PCR: indicates polymerase chain reaction, DS-IgM EIA: double sandwich IgM enzyme immunoassay, ISAGA: IgM immunosorbent agglutination assay.
Three of our patients were hospitalized into the newborn unit due to jauindice and the other one was hospitalized due to hydrops developed as a result of heavy intrauterin effect. In the laboratory examination of the patients' anemia, thrombocytopenia, increased direct and indirect bilirubin level, normal and slightly increased aminotransferase levels, abnormality in coagulation tests, low transferrin, increased transferrin saturation with overincreased ferritin and AFP levels were compatible with NH laboratory findings indicated in literature.
There was no positive finding in USG except homogeneous increases of echogenicity in the liver parenchyma and ascites. As the result of MRG, excessive iron load in liver and pancreas (the indicator of extrahepatic siderosis) was detected in two patients. The liver biopsies done after coagulopathy and thrombocytopenia findings were recovered and reported to be complied with hemochromatosis. Since the clinical and laboratory tables of the other two patients were not stable, they did not undergo MRG and tissue biopsy processes. However, postmortem biopsies of these two patients were complied with hemochromatosis.
Prognosis of NH is variable; although some cases of spontaneous remission have been reported, most infants do not survive without treatment.16,27,28 Treatment with either anti-oxidant/iron-chelator cocktail (Table 3) or exchange transfusion and intravenous immunoglobulin (IVIG) has been reported to be successful in small series of patients, but these therapies have not been systematically evaluated. The effectiveness of the therapy is a matter of controversy. Survival rates with this treatment have been generally reported as low.4,6,15,16,19,20,24,29 In the patients studied by Murray and Kowdley 4 and Sigurdsson et al., 24 chelation-antioxidant therapy was not efficacious. Rodrigues et al. 20 concluded in their study that this therapy did not appear to modify the outcome in NH, at least in severe cases, and recommended liver transplantation as the treatment of choice. Nevertheless, Flynn et al. 30 observed a good response to the therapy in patients with a less severely affected liver. A recent study carried out by Grabhorn et al. 12 reported survival rates of up to 80% after exclusive chelation-antioxidant therapy even in cases of severe NH.
The cocktail for treating neonatal hemochromatosis 15

Liver transplantation has been successful in treating NH. All of the studies undertaken to date confirm the effectiveness of this treatment, with good long-term results in comparison with those of medical therapy alone.4,12,21,24,30 NH is one of the most commonly recognized causes of liver failure in the neonate and it often leads to liver transplantation in the first 3 months of life.
The patients were supported with fresh frozen plasma, vitamin K, packed RBC concentrate and platelet concentrate for existing anemia, thrombocytopenia and coagulopathy. All patients received 1 gram/kg IVIG support. One of our patients was born as hydrops fetalis the result of heavy influence of intrauterin. The patient had deep anemia and was given exchange transfusion twice since bilirubin levels increased. All 4 patients having NH diagnosis as the result of clinical and laboratory findings received N-acetyl sistein, selenium and E and C vitamins as antioxidant and desferroxoamine treatment (chelation-antioxidant therapy) was started to decrease ferritin level. One of the patient that had chelation-antioxidant treatment died within the first week of hospitalization due to pulmonary hemorrhage and another one due to multiorgan failure. The other patients showed clinical and laboratory recovery due to implemented treatment. Liver transplantation was planned for these 2 patients. However, since there was no suitable donor, the transplantation was not done. One of the patients died when she was 2.5 months old and the other in 8 months due to sepsis.
NH is a disease with high mortality despite the applied medical treatment. Although there are various success levels related to the medical treatment in literature, overall treatment success is stated to be around 10%–20%. We lost two of our patients at early stage despite the medical treatment. We had response for the others with medical treatment; however, since early stage liver transplantation could not be carried out, we lost them. We believe that medical treatment in NH patients should be started as soon as possible, and after clinical success is achieved liver transplantation should be done in a short time so that the chance of pulling through can be increased.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not for-profit sectors.
