Abstract
To assess kidney damages in pregnant and lactating rats and in their suckling pups, Wistar female rats were given, through drinking water, 700 parts per million (ppm) of K2Cr2O7 from the 14th day of pregnancy until day 14 after delivery. Toxicity was objectified by a significant increase in malondialdehyde (MDA), glutathione (GSH) and nitric oxide (NO) levels in kidney of chromium-treated mothers and their suckling pups. Moreover, lactate dehydrogenase (LDH) was increased in kidney and decreased in plasma of K2Cr2O7-treated rats. Activities of superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPx) were increased in dams and decreased in their pups. Interestingly, these biochemical modifications were accompanied by higher plasma and lower urinary levels of creatinine, a specific indicator of glomerular function, and of urea than those of controls. Significant increase in creatinine clearance was also found in treated mothers and in their progeny. Histological studies showed an infiltration of mononuclear cells, necrosis and vascular congestion in kidney of pups and dams. Based on the present findings, K2Cr2O7 administrated to female rats during late pregnancy and early postnatal periods provoked kidney damages in dams and their offspring.
Introduction
Chromium (Cr) in the environment occurs predominantly in two valence states: hexavalent [Cr(VI)] and trivalent chromium [Cr (III)]. 1,2 Due to its widespread use in leather tanning, textile and pigment electroplating industries, Cr(VI) becomes one of the most abundant pollutants in aquatic and terrestrial ecosystems. 3 The deposition of Cr(VI) wastes in landfills and waterways by chromate industries affects millions of people drinking Cr-containing water, residing in the vicinity of dangerously polluted sites. 4 The wide environmental distribution in Cr leads to an increased interest of its toxicity and biological effects. 5 In reproductive toxicology, Cr is highly toxic and affects growth of fetuses and suckling pups in humans and animals. Indeed, women working in Cr industries and living around Cr-contaminated areas present gynaecological illness, 6 postnatal hemorrhage and birth complications with high levels of Cr in blood and urine. 7,8 Interestingly, it has been reported by Barceloux, 9 that Cr was transported to offspring through milk of lactating women exposed to Cr(VI). This metal could also traverse the placental barrier in rodents. 9,10 Previous findings demonstrated that Cr, administrated through drinking water for 20 days before gestation state, revealed abnormal gestational changes in mice and rats, with a decreased number of implantation sites and of viable fetuses. 11 –13 According to Junaid et al., 14 K2Cr2O7 produces embryotoxic and fetotoxic effects.
The exact mechanism of Cr(VI)-induced toxicity has not been elucidated yet; however, reduction of Cr(VI) and related free radical reactions have been postulated as possible mechanisms mediating the toxicity of Cr(VI) compounds. In fact, free radicals produce a number of toxic effects including DNA damage and lipid peroxidation; therefore, the toxic effects of Cr(VI) may be, at least in part, associated with the production of reactive species via Fenton or Haber-Weiss-type reactions. 15 Since the kidney is a main target organ of K2Cr2O7, this metal causes acute renal failure (ARF) in mammals. 16,17 Previous findings showed that chromate affected selectively the convoluted section of the proximal tubules 17 –19 and induced acute necrosis of renal tubules. 19,20
To our knowledge, no studies have been conducted on kidney of suckling rats whose mothers were treated by K2Cr2O7. Therefore, the present experiment was undertaken to evaluate biochemical parameters, lipid peroxidation, oxidative stress and histopathological changes in kidney of female rats and their progeny following subchronic exposure to K2Cr2O7 during late pregnancy and lactating periods.
Materials and methods
Chemicals
Potassium dichromate (K2Cr2O7), was obtained from Merck (Darmstadt, Germany). Sodium selenite (Na2SeO3) was purchased from Sigma (St. Louis, Missouri, USA). All other chemicals of analytical grade were purchased from standard commercial suppliers.
Animals and treatments
Experiments were performed on female Wistar rats weighing 170 ± 10 g and purchased from SIPHAT (Tunisia). After 1 week of adaptation period in a room with controlled temperature (22 ± 2°C) and lighting (12 h-light/12 h dark), female rats were mated with males. A sperm-positive vaginal smear was taken to indicate the first day of pregnancy. They were housed individually in stainless steel cages and were provided daily with standard pellet diet (SICO, Tunisia) and water ad libitum. Twelve pregnant female rats were randomly divided into two groups of six each: group I served as controls and group II received 700 parts per million (ppm) of K2Cr2O7 (equivalent to 67 mg/kg) through the drinking water from the 14th day of pregnancy until day 14 after delivery. Since the major route of Cr for the general population is the oral way, 11 the present study is designed to investigate the toxicity of K2Cr2O7 given to rats via the oral route. The dose of K2Cr2O7 and the period of treatment were selected on the basis of previous studies. 11,14,21 –23 The levels of K2Cr2O7, used in the present study, are not usually found in the environment but may be encountered in the workplace or in the vicinity of industrial establishments. 11 The day of parturition was considered as day 0 of lactation. Pups were counted, weighed and each litter was reduced to eight pups (four males and four females, if possible) as it has been shown that this procedure maximized lactation performance. 24 Daily K2Cr2O7 intake by lactating rats was determined after measuring drinking water consumption. So each lactating rat, treated with K2Cr2O7, ingested daily 26.46 ± 3.81 mg of K2Cr2O7 (Table 1). All animals were observed for signs of treatment-related effects.
Body and relative kidney weights of suckling and adult rats, controls and K2Cr2O7 treated with 700 parts per million (ppm) administered in their drinking water from the 14th day of pregnancy until day 14 after delivery a

a Food intake and quantities of K2Cr2O7 ingested by lactating rats. The number of determinations is indicated in parenthesis. Data are shown as means ± SD.
b K2Cr2O7 treated vs controls: p < 0.05.
c K2Cr2O7 treated vs controls: p < 0.001.
d K2Cr2O7 treated vs controls: p < 0.01.
The experimental procedures were carried out according to the general guidelines on the use of living animals in scientific investigations
25
and approved by the Ethical Committee of Sciences Faculty of Sfax. The amount of ingested diet was calculated as the difference between the weight of feed that remained in the food bin (Da) and the amount placed 1 day before (Db). These data were then used to calculate the daily average feed intake, according to the formula:
Before sacrifice, urinary samples were obtained from each animal housed in a specially designed metabolic cage, where fecal contamination was avoided. They were collected into bottles within 24-h cycles. Urinary volume of 14-day-old rats was calculated by taking away the 24-h urine volume of mothers kept with their pups and those kept alone in metabolic cages. The volume of each urine sample was recorded and centrifuged at 3000 g for 5 min.
On postnatal day 14, 12 dams and 96 pups were anesthetized with chloral hydrate by intra-abdominal route. After sacrifice, blood samples were collected into heparin tubes by aortic puncture in dams and brachial artery in pups. Plasma samples were drawn from blood after centrifugation at 2200 g for 15 min. They were kept at −80°C until analysis.
Kidneys were removed, cleaned and weighed. Some samples were rinsed, homogenized (10% w/v) in phosphate buffer (pH 7.4) and centrifuged. The resulting supernatants were used for biochemical assays. Others were immediately fixed in 10% formalin solution for histological studies.
Biochemical determinations
Estimation of urea, uric acid, creatinine and creatinine clearance
The levels of urea, uric acid and creatinine in plasma and urine were estimated spectrophotometrically using commercial diagnostic kits (ref 20151, 20143, 20091, respectively) purchased from Biomagreb (Ariana, Tunisia). Creatinine clearance, an index of glomerular filtration rate, was calculated by UV/P equation 26 where U is the urinary creatinine level, V the volume of urine sample collected within 24 h and P the plasma creatinine concentration.
Lipid peroxidation
Concentration of MDA in tissues, an index of lipid peroxidation, was determined spectrophotometrically according to Draper and Hadley method. 27 A 0.5-mL aliquot of kidney extract supernatant was mixed with 1 mL of trichloroacetic acid solution and centrifuged at 2500 g for 10 min. One millilitre of a solution containing 0.67% thiobarbituric acid (TBA) and 0.5 mL of supernatant were incubated for 15 min at 90°C and cooled. Absorbance of TBA-MDA complex was determined at 532 nm using spectrophotometer. Lipid peroxidation was expressed as nmol of thiobarbituric acid reactive substances (TBARS), using 1,1,3,3-tetra-ethoxypropane as standard.
Estimation of kidney nitric oxide level
Production of nitric oxide (NO) was determined based on the Griess reaction. 28 Briefly 100 µL of deproteinized sample was incubated with 100 µL of Greiss reagent at room temperature for 10 min. Absorbance was measured at 550 nm, using a microplate reader. Nitrite concentration was measured using sodium nitrate as standard and was expressed as nmol/mg protein of tissue.
Non-protein thiols content in kidney
Kidney non-protein thiols (NPSH) levels were determined by the method of Ellman. 29 A 500 µL aliquot of supernatant was mixed with 10% trichloroacetic acid (1V/1V). After centrifugation, the protein pellet was discarded and free–SH groups were determined in a clear supernatant. A 100 µL aliquot of supernatant was added to 850 µL of 1 M potassium phosphate buffer (pH = 7.4) and to 50 µL of 5,5-dithiobis-2 nitro benzoic acid (DTNB; 10 mM). The absorbance of colorimetric reaction was measured at 412 nm.
Estimation of lactate dehydrogenase (LDH) activities
Lactate dehydrogenase activities in plasma and kidney, used as biochemical markers for renal damage, were determined by enzymatic method using commercial reagent kit (Biomaghreb, Ariana, Tunisia. Ref 20012).
Protein analysis in kidney
Protein content in kidney was determined according to Lowry et al 30 using Bovine serum albumin as standard.
Antioxidant defense system assays
The catalase (CAT) activity was determined according to the method of Aebi. 31 The H2O2 decomposition rate was followed by monitoring absorption at 240 nm. One unit of CAT activity is defined as the amount of enzymes required to decompose 1 µmol of hydrogen peroxide in 1 min. The enzyme activity was expressed as µmol H2O2 consumed/min/mg protein.
Glutathione peroxidase (GPx) activity was measured according to Flohe and Gunzler. 32 The enzyme activity was expressed as nmol of GSH oxidized/min/mg protein.
Superoxide dismutase (SOD) activity was estimated according to Beauchamp and Fridovich. 33 The reaction mixture contained 50 mM of tissue homogenates in potassium phosphate buffer (pH 7.4), 0.1 mM L-methionine, 2µM riboflavine and 75 µM Nitro Blue Tetrazolium (NBT). The developed blue color reaction was measured at 560 nm. Units of SOD activity were expressed as the amount of enzyme required inhibiting the reduction of NBT by 50% and the activity was expressed as U/mg protein.
Reduced glutathione (GSH) levels were determined by the method of Ellman 29 modified by Jollow et al 34 based on the development of a yellow color when DTNB was added to compounds containing sulfhydryl groups. Five hundred milliliters of tissue homogenate were added to 3 mL of 4% sulfosalicylic acid. The mixture was centrifuged at 1600 g for 15 min. Five hundred milliliters of supernatant were taken and added to Ellman’s reagent. The absorbance was measured at 412 nm after 10 min. Total GSH content was expressed as µg/g tissue.
Histopathological examination
Kidney histological sections of 5 µm were placed on slides and stained with hematoxylin-eosin for histopathological studies. Six slides were prepared from each kidney. All sections were evaluated for the degree of tubular and glomerular injury and necrosis. Each kidney slide was examined and assigned for severity of changes using scores on a scale of none (−), mild (+), moderate (++) and severe (+++) damages.
Statistical analysis
Comparison of mean values followed by SD between treated and control rats was made using Student’s t-test. 35 The 0.05 level was selected as the point of minimal statistical significance.
Results
Evaluation of body, absolute and relative kidney weights
During the experimental period, there are no clinical signs and no death in K2Cr2O7-treated group. Food and water consumption was reduced by 9% and 8%, respectively, in K2Cr2O7-treated dams when compared to controls (Table 1). The body weights of Cr-treated rats were decreased by 4% in mothers and 26% in their offspring as compared to controls. Relative kidney weights of treated dams were similar to those of control rats, while a marked increase in the relative kidney weight was observed in pups (+12%) when compared to corresponding controls.
Urinary volume, creatinine, urea and uric acid levels in plasma and urine
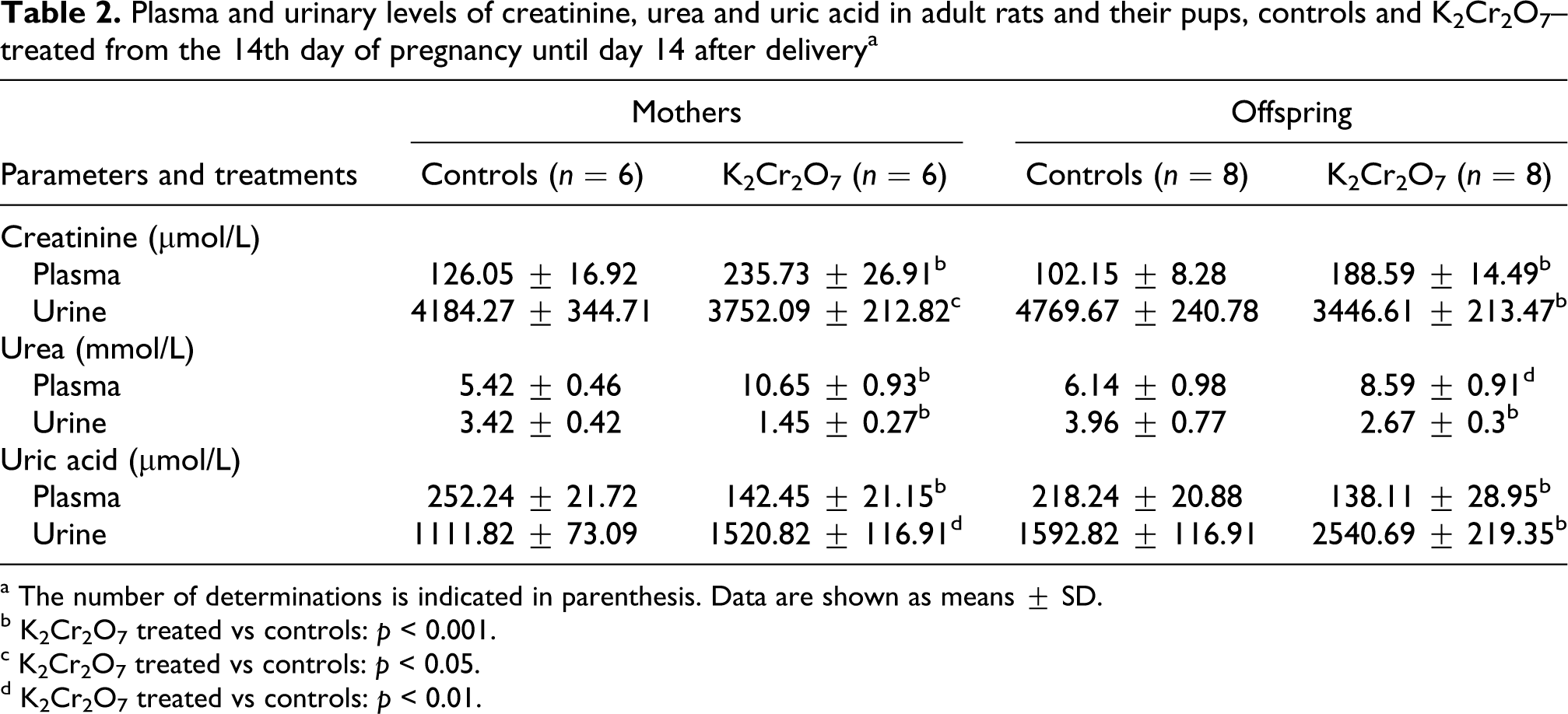
K2Cr2O7-intoxicated rats showed a constellation of disorders in renal function witnessed by increased urine output and changes in creatinine, urea and uric acid levels. In fact, the 24-h urine volume in treated mothers and in their pups was higher than in the controls (+52% and +56%, respectively; Figure 1A). Creatinine levels in K2Cr2O7-treated mothers and their pups were higher in plasma (+87%; +85%) and lower in urine (−10; −28%) than those of corresponding controls (Table 2). So we have found, after K2Cr2O7 treatment, a reduction in creatinine clearance, an indicator of glomerular dysfunction in adult rats (−32%) and in their pups (−37%; Figure 1B). Urea levels in treated mothers and in their suckling pups were higher in plasma (+86%; +40%) and lower in urine (−57%; −32%), respectively, than those of controls (Table 2). While uric acid levels were lower in plasma of K2Cr2O7-treated mothers (−44%) and of their offspring (−37%) than those of corresponding controls. Urinary excretion of uric acid, a nitrogenous waste product, was increased in K2Cr2O7-treated mothers (+37%) and their pups (+59%) compared to controls (Table 2).

Urinary volume (A) and creatinine clearance (B) in adult rats and their pups: controls and K2Cr2O7 treated with 700 parts per million (ppm) administered in the drinking water from the 14th day of pregnancy until day 14 after delivery. K2Cr2O7 treated versus controls: ** p < 0.01; *** p < 0.001. Data are shown as means ± SD. The number of determinations is indicated above columns of each diagram.
Plasma and urinary levels of creatinine, urea and uric acid in adult rats and their pups, controls and K2Cr2O7–treated from the 14th day of pregnancy until day 14 after delivery a
a The number of determinations is indicated in parenthesis. Data are shown as means ± SD.
b K2Cr2O7 treated vs controls: p < 0.001.
c K2Cr2O7 treated vs controls: p < 0.05.
d K2Cr2O7 treated vs controls: p < 0.01.
Kidney malondialdehyde content
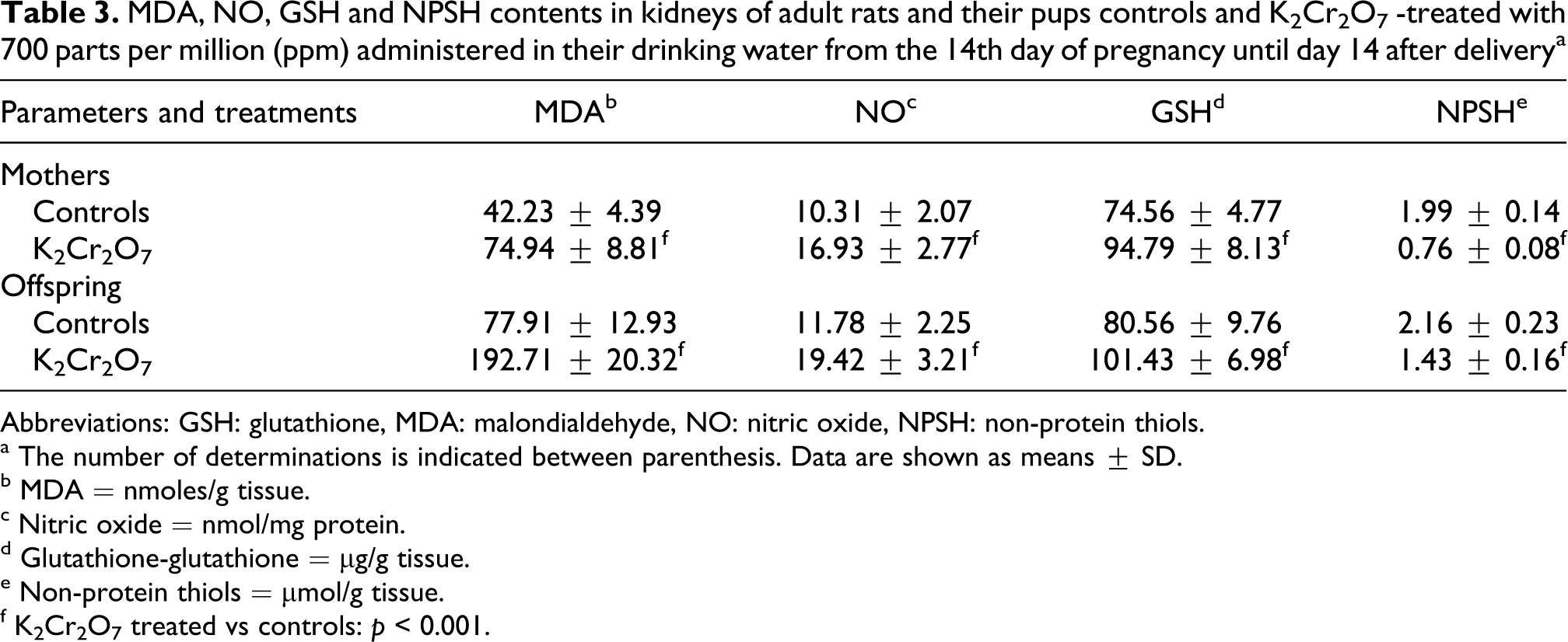
Subacute toxicity of K2Cr2O7 provoked a significant increase in kidney malondialdehyde (MDA) levels in dams (77%) and in their offspring (1.5-fold). The rate of lipid peroxidation in kidney was more pronounced in pups than in their mothers (Table 3).
MDA, NO, GSH and NPSH contents in kidneys of adult rats and their pups controls and K2Cr2O7 -treated with 700 parts per million (ppm) administered in their drinking water from the 14th day of pregnancy until day 14 after delivery a
Abbreviations: GSH: glutathione, MDA: malondialdehyde, NO: nitric oxide, NPSH: non-protein thiols.
a The number of determinations is indicated between parenthesis. Data are shown as means ± SD.
b MDA = nmoles/g tissue.
c Nitric oxide = nmol/mg protein.
d Glutathione-glutathione = µg/g tissue.
e Non-protein thiols = µmol/g tissue.
f K2Cr2O7 treated vs controls: p < 0.001.
Lactate dehydrogenase activities in kidney and plasma
In K2Cr2O7 group, lactate dehydrogenase activity, a marker of kidney cellular damage, was increased in plasma by 53% and 57% and decreased in kidney extracts by 61% and 47%, respectively, in adult rats and in their suckling pups (Figure 2).

Lactate dehydrogenase (LDH) activities in plasma (A) and in kidney (B) of adult rats and their pups: controls and K2Cr2O7 treated with 700 parts per million (ppm) administered in their drinking water from the 14th day of pregnancy until day 14 after delivery. K2Cr2O7 treated versus controls: *** p < 0.001. Data are shown as means ± SD. The number of determinations is indicated above columns of each diagram.
Kidney NO, GSH and NPSH levels
In K2Cr2O7 group, NO and GSH levels in kidney homogenates were increased by 64% and 27%, respectively, in adult rats and by 65% and 26% in their suckling pups (Table 3), while, NPSH levels decreased significantly by 47% in mothers and by 62% in their offspring (Table 3).
Effects of K2Cr2O7 on kidney antioxidant enzyme activities
K2Cr2O7 treatment led to a significant increase in CAT, SOD and GPx activities in kidney homogenates by 86%, 65% and 22%, in mothers and a decrease of these parameters by 50%, 47% and 48% in their pups, respectively, when compared to those of control group (Figure 3).
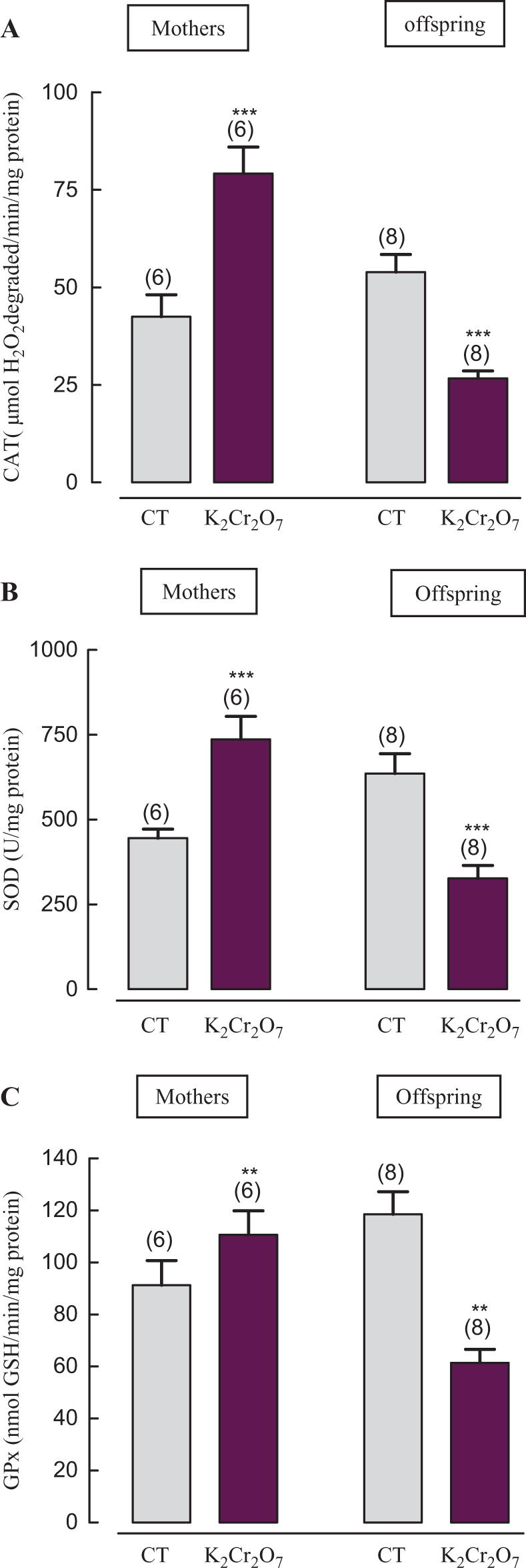
Antioxidant enzyme activities CAT (A), SOD (B) and GPx (C) in kidney of adult rats and their pups: controls and K2Cr2O7 treated with 700 parts per million (ppm) administered in their drinking water from the 14th day of pregnancy until day 14 after delivery. A: catalase (CAT), B: superoxide dismutase (SOD), C: glutathione peroxidase (GPx). K2Cr2O7 treated versus controls: *** p ≤ 0.001. Data are shown as means ± SD. The number of determinations is indicated above columns of each diagram.
Histopathological studies
Abnormalities in kidney histological pictures, detected in glomeruli and in convoluted tubules, were shown in mothers (Figure 4B1, B2, B3 and B4) as well as in their pups (Figure 5D1, D2, D3 and D4) compared to those of corresponding controls (Figures 4A and 5C). The severity of changes was assessed for each slide by scoring using a scale of no (−), mild (+), moderate (++) and severe (+++) damage. The kidney of K2Cr2O7-treated mothers exhibited vascular congestion inside glomeruli and between tubules and narrowed Bowman’s space. Furthermore, convoluted tubules were dilated (Figure B1, B2). Tubular cells showed vacuoles formation, which indicates a necrosis step (Figure 4B2). Infiltration of lymphocytes and polynuclear cells particularly between tubules was also observed (Figure 4B3, B4). Moreover, in their pups, we have observed tubular dilatation and cell necrosis (Figure 5D2, D4). Nonetheless, these effects are more pronounced in mothers. The histopathological changes were graded and summarized in Table 4 .

Kidney histological sections of adult rats: controls (A) and treated (B1, B2, B3 and B4) who have received 700 parts per million (ppm) K2Cr2O7 in their drinking water from the 14th day of pregnancy until day 14 after parturition. Optic microscopy: HE (×200) A, B1 and B3; HE (×400) B2 and B4. Arrows indicate: intra-glomeruli hemorrhage, tubular dilation, glomeruli showing large Bowman's space, markedly lobulated glomeruli, infiltration of mononuclear cells, necrosis, vacuolation, vascular congestion.
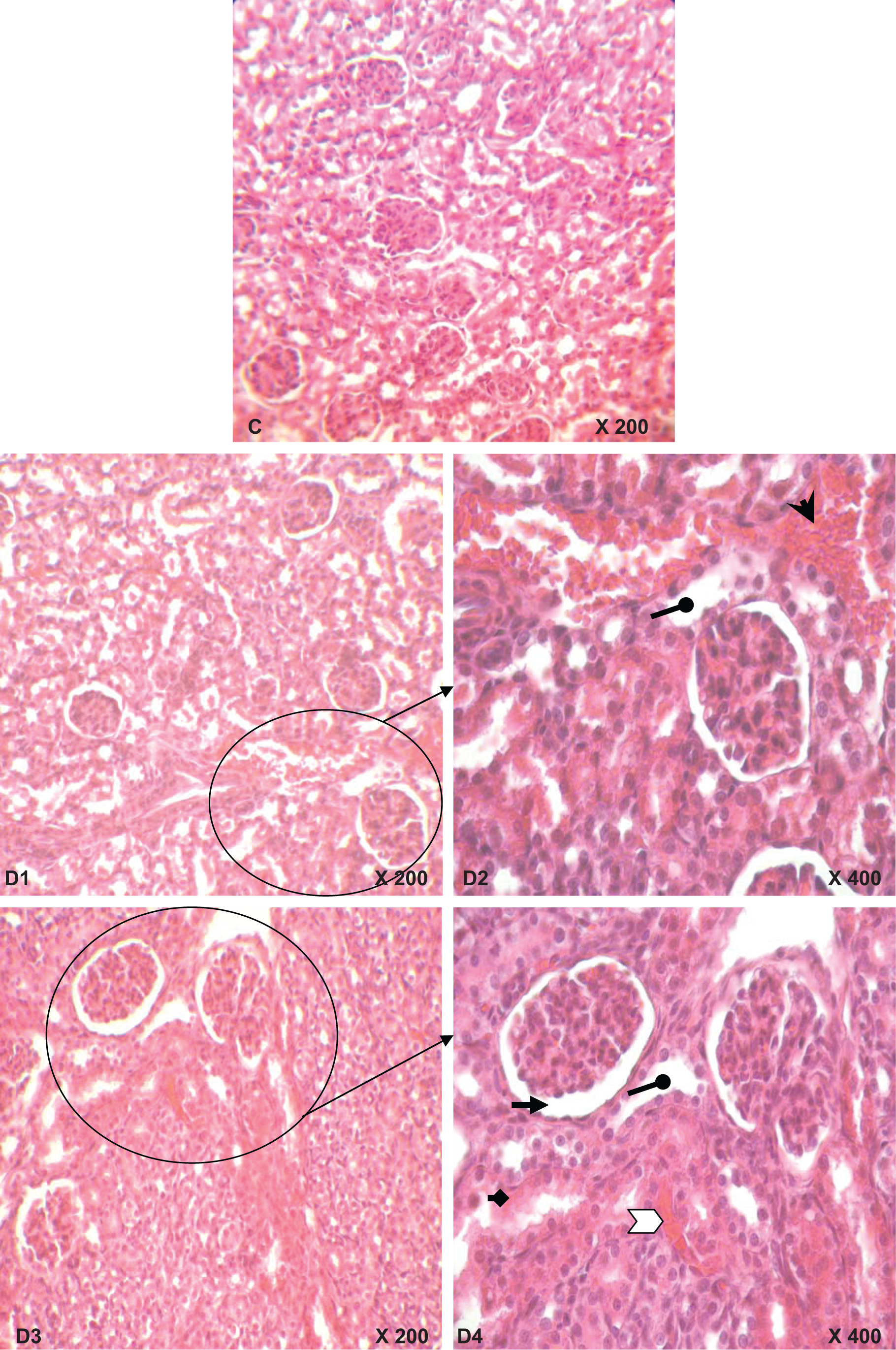
Kidney histological sections of 14-day-old rats: controls (C) and whose mothers have received K2Cr2O7 (D1, D2 D3 and B4) in their drinking water from the 14th day of pregnancy until day 14 after parturition. Optic microscopy HE (×200) C1, D1 and D3; HE (×400) D2 and D4. Arrows indicate: hemorrhage, tubular dilation, glomeruli showing large Bowman’s space, necrosis, vascular congestion.
Grading of the histopathological changes in the kidney sections a
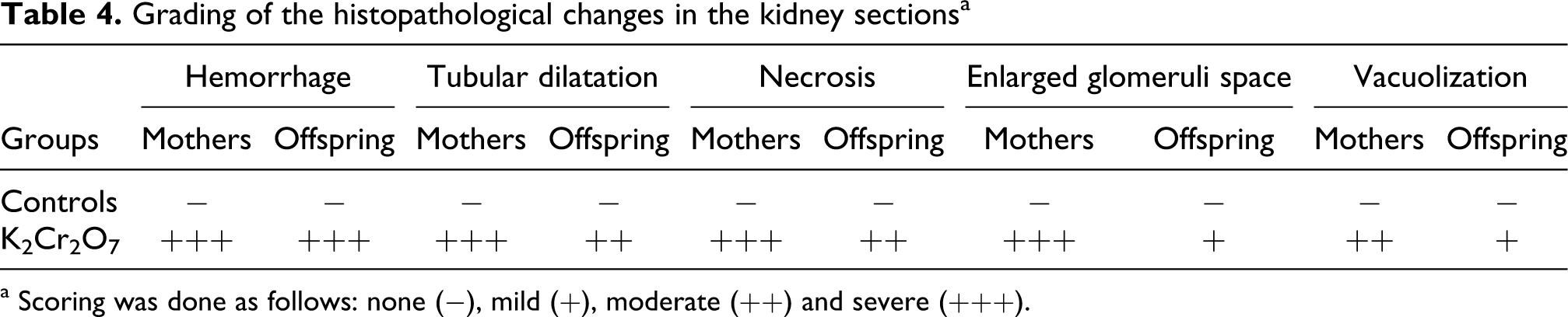
a Scoring was done as follows: none (−), mild (+), moderate (++) and severe (+++).
Discussion
Throughout the course of gestation to parturition, females' biological systems were deeply modified in order to ensure embryo-fetal development and well-being. 36 These disorders threatened both mothers and their fetuses. Impairment of gestation and complications during pregnancy and childbirth have been reported in female workers exposed to Cr at the workplace. 7,8 The same disorder was also observed in mice exposed to Cr(VI) during gestation period. 11,14,21,22,37 In the present study, we have focussed our attention on K2Cr2O7 potential causing alterations in kidney function of female rats and their pups during the suckling period.
The results, presented in this work, clearly showed several modifications, induced by K2Cr2O7 during late pregnancy and early postnatal periods, resulted in nutritional status and metabolism. In fact, ingestion of Cr(VI), through drinking water, induced a significant decrease in food consumption and in water intake by female rats. So, body and kidney weights of their offspring were altered. These findings might be attributed to a Cr transfer through both placenta and milk as demonstrated either by Barceloux, 9 or/and by cell growth inhibition of the cell cycle. 38 Similar results were observed by Gilbert et al. 39 in humans exposed to chromium picolinate.
In the present study, K2Cr2O7 strongly affected plasma and urinary parameters such as the 24-h urine volume, creatinine levels and creatinine clearance. In the current study, oral administration of K2Cr2O7 to lactating rats induced an increase in 24-h urine output both in mothers and in their offspring. Polyuria might be explained by the inhibition of antidiuretic hormone secretion, which normally provoked water reabsorption across the collecting ducts after exposure to metals. 40 In the present investigation, a reduction of the 24-h creatinine excretion and of creatinine clearance indicated kidney impairment since these parameters were used as qualitative and quantitative indexes of the alteration in glomerular filtration rate (GFR).
In addition, urea, a major nitrogen-containing metabolic product of protein metabolism, was increased in blood of K2Cr2O7-treated rats. These results correlated with an increased protein catabolism in mammals and/or with the conversion of ammonia to urea as a result of increased synthesis of arginase enzyme involved in urea production. Another biochemical marker, used in this study to evaluate kidney function, was uric acid levels in plasma and urine. In fact, many drugs could affect plasma uric acid levels by influencing the net reabsorption of uric acid in the proximal tubules of the nephron. 41 Under our experimental conditions, K2Cr2O7 poisoning in adult and young rats resulted in a decrease in plasma uric acid levels and an increase in its urinary excretion. Kidney dysfunction could probably occur via kidney oxidative damage. In fact, uric acid is a potent scavenger of peroxynitrite. 42
Once inside the cell, Cr(VI) is rapidly reduced in order to generate ROS, 43,44 so reactive Cr intermediates can directly interact with cellular constituents. Indeed, Cr V, Cr IV and the more stable Cr III are all able to generate free radicals such as superoxide, nitrogen species like peroxynitrite, NO and hydroxyl causing damage consistent with oxidative stress. 45 Free radicals can damage virtually all types of macromolecules including lipids, causing an increase of kidney MDA content, 5 the major product of lipid peroxidation. In fact, in our study, we have found that Cr(VI) treatment increased the amount of MDA levels in kidney tissue of mothers and their pups, suggesting oxidative stress in kidney and nephrotoxicity. Our results corroborated with previous findings cited above. Additionally, lipid peroxidation levels were found to be higher in young rats than in mothers, this may be due to deleterious changes as transfer of K2Cr2O7 from the mothers to their fetuses through placenta and to their newborns through milk. 9 Our results confirmed an intoxication of pups during a lactating period which was necessary for their development.
Furthermore, a previous study of Fujihara et al. 46 showed that a basal amount of NO might be important to maintain normal renal function. However, increasing evidence suggested that excessive production of NO played a major role in oxidative stress and tissue damage in the pathophysiology of acute renal failure. 47 Under our experimental conditions, kidney NO levels increased significantly in K2Cr2O7-treated mothers and in their offspring when compared with those of the control group. Thus indicated that Cr might lead to the induction of inducible nitric oxide synthase, resulting in an increased production of NO, leading to the formation of toxic peroxynitrite. 48 An enhancement of NO was reported by Bagchi et al. 49 in peritoneal exudates cells, mainly the macrophages from sodium dichromate-treated rats.
The increased oxidative stress presented the opposite picture to the changes in antioxidants status. In fact, the impairment of the antioxidant defense system was considered as a critical event in Cr-induced nephrotoxicity. 50 Exposure of Cr was characterized by the depletion of tissue and circulating non-enzymatic anti-oxidants including GSH and NPSH. 51 Indeed, GSH was considered as the essential compound which maintained cell integrity because of its reducing properties and its participation in the cell metabolism. Current data revealed that K2Cr2O7 caused a significant increase in GSH level in mothers and their progeny. This finding might well be a compensatory response to protect tubular cells from the ravaging effects of lipid peroxidation by increasing the levels of endogenous GSH. Our results corroborates with previous study of Cupo and Wetterhahn 52 who reported that Cr(VI)-induced an increase of GSH level in chicken embryo hepatocytes. Furthermore, It has been suggested that low level of GSH enhanced Cr(VI) toxicity by promoting the formation of Cr(V), while high concentrations of glutathione and ascorbate would reduce rapidly Cr(VI) to the final product Cr(III), thus yielding lower steady state concentration of Cr(V) and scavenge Cr(VI) as Cr(III)-GSH complex. 53
In addition, another product NPSH, considered as one of the important primary defenses that counteract oxidative stress, decreased significantly in kidney of treated mothers and of their offspring when compared to the control group. Depletion of NPSH following Cr(VI) exposure might be due to either induction of oxidative stress and to consumption of GSH in the Cr(VI) reduction process 54 or to the excretion of Cr(III) in the urine as a complex with GSH. 55
Antioxidant enzymes are frequently used as markers of oxidative stress. 56 Among the first line of cellular defense, SOD, CAT and GPx are important in the preservation of homeostasis for normal cell function. According to Avti et al., 57 deviations in the physiological concentrations of these enzymes has drastic effects on the cellular resistance. Our findings showed a significant increase in the activities of SOD, GPx and CAT in kidneys of mothers and a decrease in those of pups. As these enzymes have a protective role against oxygen free radical-induced damage, their induction can be understood as an adaptative response to oxidative stress. Sengupta et al. 58 observed also an increase of antioxidant enzyme activities in adult rats exposed to hexavalent Cr. Similar results, concerning a decrease of the enzymes cited above, have been found by others in suckling mice whose mothers were treated by heavy metals such as fluoride. 59 The inhibition of most antioxidant enzymes, induced by Cr, may be due either to the direct binding of the metal to their active site or to their increased usage in scavenging free radicals induced by the metal, thus causing irreversible inhibition of their activities. The difference between deviation in concentration of these enzymes between mothers and pups could be also explained by the fact that K2Cr2O7 has different mechanisms depending on the age of animals, route and period of administration.
These biochemical modifications corresponded histologically. In fact, histological changes, seen in the kidney of mothers and their pups treated with K2Cr2O7, were characterized by necrosis, hemorrhage and tubular dilation. Besides, in mothers, extensive vacuolization and intra-glomeruli hemorrhage were observed. Changes in the histoarchitecture were also observed by Pedraza-Chaverri et al. 50 after K2Cr2O7 treatment. This might be due to the increased amount of ROS product which might attack membranes, lipid and proteins and affect kidney histoarchitecture. 60
In conclusion, we found that a high concentration of K2Cr2O7 in drinking water, ingested by pregnant and lactating rats, induced an oxidative stress in renal tissue of both adult rats and their pups, leading to lipid peroxidation. Balance between oxidative and antioxidant systems was disturbed in K2Cr2O7-treated rats. High K2Cr2O7 intake by lactating rats appeared to have pronounced toxic effects in their suckling pups. These evidences, presented in our experimental data, show for the first time the occurrence of nephrotoxicity after exposure of rats to K2Cr2O7 during the late pregnant and lactating period, make clear the need for attention of pregnant women on K2Cr2O7 exposure not only during pregnancy but also during lactation period.
Footnotes
Acknowledgments
The authors are indebted to Miss Dalenda Kchaou for her assistance in histolological techniques.
This work was supported by the DGRST grants (Appui à la Recherche Universitaire de Base ARUB 99/UR/08-73), Tunisia.
