Abstract
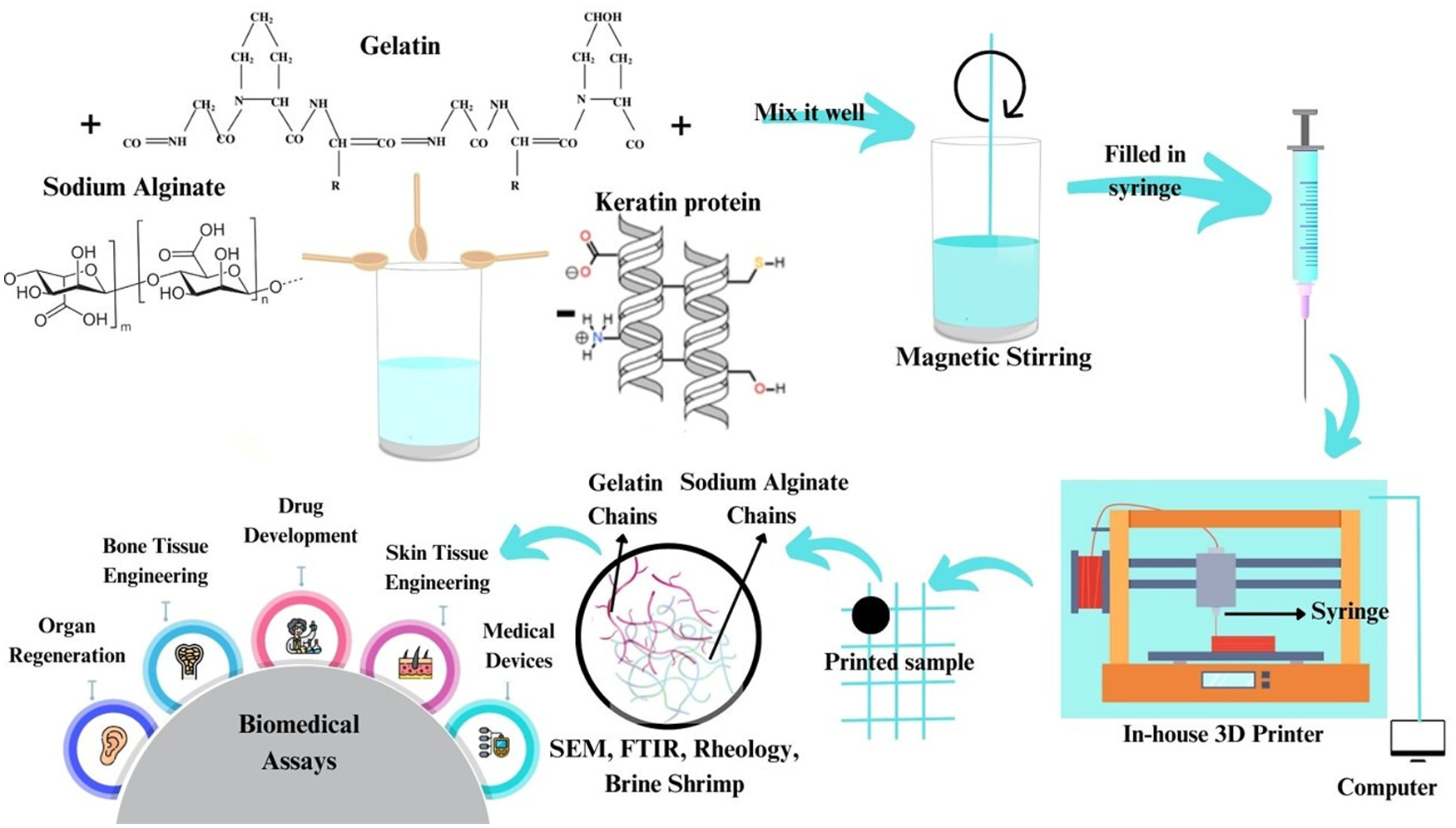
The advancement of 3D printing technology offers transformative potential in developing functional skin substitutes for scar prevention and tissue regeneration. This research investigates optimized bio-ink formulations comprising sodium alginate, gelatin, and keratin protein, evaluating their structural, mechanical, and biological properties through comprehensive characterization techniques. Scanning Electron Microscopy (SEM) revealed the structural morphology of raw materials and printed constructs, with sodium alginate exhibiting irregular shapes (100–200 µm diameter), gelatin displaying elongated particles (900–1000 µm diameter), and keratin featuring fibrous structures (300–500 µm diameter), all contributing to scaffold integration. Fourier Transform Infrared (FTIR) spectroscopy confirmed the retention of functional groups and formation of new molecular bonds during crosslinking. Rheological analysis highlighted an 81.8% increase in maximum shear stress, from 550 Pa in sodium alginate to 1000 Pa in sodium alginate-gelatin composites, demonstrating enhanced mechanical robustness and optimal shear-thinning behavior critical for printing. Tensile testing analysis revealed composition-dependent mechanical reinforcement, with the incorporation of 20% keratin leading to a maximum tensile strength improvement of approximately 64% relative to the alginate–gelatin matrix. Swelling and degradation studies indicated controlled hydration and improved structural stability of keratin-reinforced scaffolds over 48 h. In vitro biocompatibility was validated through brine shrimp toxicity tests, with keratin-enriched bio-inks achieving a 98% survival rate in seawater, a 5.55% improvement over sodium alginate-gelatin-only solutions. These findings underscore the synergistic role of gelatin in reducing toxicity and keratin in enhancing cellular attachment and tissue regenerative capabilities. This study concludes that the sodium alginate-gelatin-keratin bio-ink formulation offers superior platform for skin tissue engineering, combining enhanced printability, structural integrity, and biocompatibility.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
