Abstract
Many efforts have been made to understand the effects of hydrogen on titanium alloys, resulting in an abundance of theoretical models and papers. Titanium alloys are crucial advanced materials that provide an excellent combination of a high strength-to-weight ratio and good corrosion behaviour even though they are reasonable to corrosion attack. Titanium alloys are susceptible to hydrogen embrittlement when comes into contact with hydrogen, and galvanic pair with an active metal current, or the pH is greater than 12 or less than 3 or an impressed current. In view of the fact that hydrogen behaves differently with α and β phases, hydrogen degradation may vary markedly in titanium alloys. Hydrogen diminishes the corrosion and erosion resistance and fatigue life of in-service titanium components. A laser peening or laser shock peening is a novel technique for making the metal surfaces and sub-layers densify. It evokes that laser shock peening adoption results in yielding and plastic deformation, thereby creating high compressive residual stresses extending below the surface of the material which is desirable for hydrogen embrittlement resistance and reduction of crack initiation and growth of the component. This article is a review of information relating hydrogen embrittlement of titanium alloys and surface modification technique which influence the strength potential of titanium alloys.
Introduction
Titanium and titanium alloys have foremost useful properties, for instance, immense corrosion resistance even in aqua regia, seawater, chlorine, high strength/weight ratio and excellent biocompatibility.1–3 Concurrently, the shortcomings of titanium alloys are moderate wear and fatigue resistance, and hardness. Commercial pure (Cp) titanium grades can be as strong as steel, though 45% lighter. Titanium is alloyed with molybdenum, aluminium, iron, vanadium, among other elements, to produce lightweight alloys as well as strong alloys for aerospace,4,5 marine, medical implants,6,7 chemical process industry,8,9 nuclear industry, 10 desalination unit, hydrogen storage devices,11,12 heat exchanger systems, transuranic waste containers and seawater-cooled piping.13,14 However, the interface between hydrogen-carrying environment and titanium alloys leads to harsh and severe problems.15–20
Titanium has turned out to be an accepted metal for piping in marine industries21,22 as shown in Figure 1. Due to the better erosion and corrosion resistance combined with high strength, pipes can be made with a thinner wall and weight is appreciably reduced. The reduced diameter and close bend radii reduced to an extraordinary extent which requires less space, providing greater design flexibility. The characteristics that made titanium as an alternative in piping have also driven its use in the plate and tube heat exchangers.23–26 Nowadays, titanium exchangers are broadly used in naval industries, offshore platforms, transport vessels and commercial fishing. 27
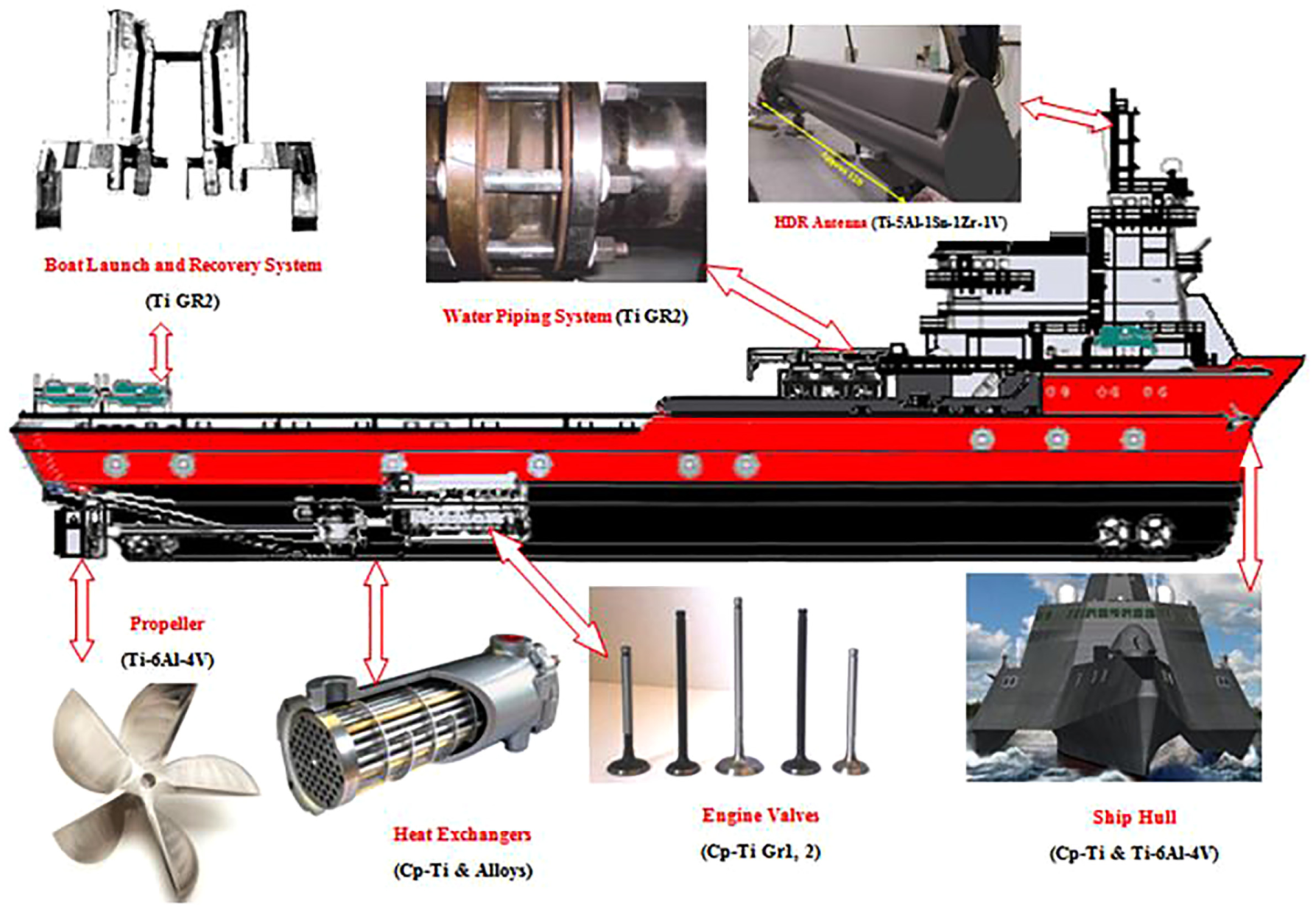
Titanium alloys for naval applications.
Cp titanium and Ti–6A1–4V alloys are widely used titanium alloys for marine and industrial applications. Corrosion of electrical apparatus uncovered to the saltwater environment is a key protection concern. Distinct from other metals, Ti alloys can withstand the severe corrosion exclusive of periodic protective coatings which make titanium a low-cost alternative to most of the applications. 28 Titanium is being used for covers, hatches, stanchions, electrical boxes, antennas, lighting fixtures, ladders and a numerous other upside components in marine industries. Corrosion of outdoor structures requires a high maintenance demand with metal choices. Because of the outstanding surface passive film formation, the continuous maintenance and corrosion resistance coatings are unnecessary, and titanium is being used for outdoor roofing, bulkheads and splash guards. Beta titanium alloys hold characteristics of excellent corrosion resistance, low modulus, high strength and formability properties which lend a hand to substitute commonly used stainless steel grades in medical implants. 29
Titanium alloys are not exceptional to hydrogen embrittlement (HE) when examined in large grained specimens at moderate and low strain rates in uniaxial tensile tests.30–33 In addition, titanium alloys are vulnerable to the occurrence of a notch, high strain rates and at low temperatures. The most recent outcome is reported as a consequence of the development of both void link-up and void nucleation at biaxial stresses or big grain sizes are the reasons for HE. The intention of this article is to review the processes that are directing for the occurrence of HE in titanium alloys and to enhance corrosion resistance under harsh hydrogen and marine environmental conditions.
Titanium and its alloys
The alloying of titanium with other chemical elements enhances toughness and tensile strength at intense temperatures. Titanium alloys are readily available, conventional engineered metals that compete with stainless steel and various steel grades, nickel-based alloys, 34 copper alloys, composites and Ti-foams.35,36 Titanium crystal structure at ambient pressure and temperature is closely packed hexagonal close packed (HCP) α-phase having a c/a ratio of 1.587. Relatively at 890 °C, titanium experiences allotropic transformation to body-centered cubic (BCC) β phase which is stable till its melting temperature. Gallium, germanium, aluminium, oxygen, carbon and nitrogen elevate the alpha-to-beta transition temperature (i.e. alpha stabilizers), whereas vanadium, tantalum, manganese, molybdenum, iron, niobium, nickel, copper, chromium, cobalt and silicon lower the transition temperature (i.e. beta stabilizers). Table 1 describes the classification of titanium alloys based on stabilizing element composition.
Typical classification of titanium alloys.

Metallurgical aspects and prominence of titanium
Corrosion resistance of titanium is admirable to natural seawater despite chemical variation and pollution effects. 40 Corrosion rates of well below 0.01 mils per year have been calculated in unmitigated programmes in splash, subsea and tidal zones. The toughness and fatigue properties of marine grade Ti alloys are unchanged to seawater conditions. The cavitations and erosion resistance make titanium a perfect metal for pumps, heat exchangers and seawater piping. 41 Heat transfer efficiency and heat transfer properties of titanium in service circumstances are fairly accurate to those of admiralty copper-nickel and brass. Although titanium and its alloys have a low coefficient of thermal conductivity, the reasons behind good heat transfer efficiency are as follows: higher strength of titanium permits the use of thin-walled equipment, less corrosion in seawater and oxide layer42,43 makes the surface for enhanced heat transfer and better erosion resistance allows considerably high service velocities.
Enhanced strength to weight ratio, low density (0.163 lb/cu) and high yield strength (40,000 psi) help in the manufacturing of titanium fitting, piping, pumps, and valves by the chemical process and service industries for more than 40 years. Trustworthy, low-cost manufacture has assisted titanium products to benefit an outstanding position in numerous markets. Similar to non-ferrous alloys, welding of titanium look forward to special care. 44 Distinct to many of the regularly used metals, titanium is truly non-toxic to public, environment and marine beings. Titanium can be employed without fear for the safety and health, for example, medical 45 and dental implants, 46 and heart valves. Industry Standards for chemical, aerospace and power generation industries have guided in promising broad acceptance of industry standards internationally. Established ASTM Standards cover tube, sheet, rod, pipe, plate, bar and so on. Cp titanium is soft adequate to be gladly shaped, and the strength titanium alloys are quite simply forged.
Admirable factors of Ti correlated to existing alloys
Stainless steels are placed low in resistance to chloride stress corrosion, and chloride attack occurs at elevated temperature processes. 47 Alloy Cu-Ni is suitable for several properties, such as thermal conductivity and superior electrical resistance to bio-fouling and ease of fabrication. On the contrary, copper is prone to attack in ammonia, sulphur, oxidizing acids, some ammonia and sulphur compounds, and oxidizing heavy metal salts. For this reason, heat exchangers made of titanium are designed with a thinner wall and near zero corrosion by minimizing the overall cost and boosted efficiency. Selected grades of titanium are employed in seawater applications; former and widespread is Grade 2, a ductile that admits easy manufacturing of parts. 48 General-purpose titanium alloy that offers higher strength is Grade 5 titanium. Its corrosion resistance is somewhat lesser than Grade 2. Grade 12 consists of 0.3% Mo and 0.8% Ni, which is a low-cost alternative to Grade 7.
Cost prevalence
Cost prevalence becomes predominantly evident when blotted out expenses such as planning, drawing changes, corrosion surveys, band-aid fixes and in-service engineering support are considered. Even though titanium is occasionally considered to be too expensive, its extended service life and greater reliability at rough seawater environment faced by the fleet can result in life-cycle cost savings. Ti alloy pipes and tubes do not need extra material for corrosion allowance, letting them be lighter and thinner than various competitors. Installation cost of titanium alloys tubes is slightly higher when compared to Cu-Ni alloy tubes, but maintenance cost of titanium alloy is extremely lower than Cu-Ni alloy tubes.
Several industries have swapped from the steel and Cu-Ni alloys to titanium. Power plants preferred to retube their condensers of the surface type with titanium Grade 2 tubes. Navy is integrating the use of titanium in onboard equipment such as doors, plate heat exchanger tanks, piping and hatches. 49 Titanium’s insusceptibility to seawater provides long-term service-proven life cycle, economic cost reliability and less maintenance.
Marine in-service conditions and requirement for novel materials
The principal components which decide the necessities for novel materials for the equipment of naval developments are as follows.50–52
Vibratory and quasi-static loading due to water wave activities
Wave loading on ship hulls has increased seriously in recent years as a result of wide research in related areas of model tests to determine dynamic,53,54 vibratory wave and quasi-static loads, 55 techniques for the theoretical calculation of wave-induced 56 bending and shear moments, 57 complete collection of ship stress data, and the collection and analysis of ocean wave records. These studies have been made necessary by the extreme changes in the characterisation of a merchant ship, particularly the bulk carriers and the top speeds of wide-ranging cargo vessels and the development of new materials for withstanding different load conditions.
Ice region pressure and decreased atmospheric temperature
For ships operating in sub-arctic and arctic waters, ice pressure and the temperature is a major threat. 58 Due to varying operating situations and uncertainties in ice conditions, an accurate estimation of the design for ice loading is difficult. The ice load is extremely localized high-pressure region phrased as critical zones. Critical zones may vary in space and time. These critical zones are categorized using parameters such as zonal area, spatial density and zonal force. The strength of the structure needs to be evaluated by various loading conditions due to uncertainties associated with ice loading and temperatures.
Repair of apparatus in the ocean conditions without heat treatment for welding
Cold repair 59 process gets rid of the need for pre-heat of component; thus, time reduction is significant in shutdown repairs. This process is intended to enhance as-welded heat-affected zone (HAZ) toughness adequately, so that reheat cracking will not take place during start-up, at maximum internal stresses. The thermal expansion dissimilarity between base and filler metal can direct to crack opening and ultimately failure. For that reason, this practice is suggested only to repair hardenable ferrous components of plant shutdowns. TBW (tempered bead welding) procedure is preferred for Ni-based wires in dissimilar material welds since they lead to fatigue cracking under cycling load. Welding of titanium alloys in such ocean conditions can be overcome by various advanced methods. 60
Titanium alloys a choice
Titanium alloys exhibit an extensive variety of strength and corrosive resistant characteristics under dynamic, static and cyclic loading conditions and high corrosion resistance towards seawater and chemical solutions for a long period. Cold resistances inside a temperature drift of up to −50 °C. Manufacturability of a material plays a vital role in the manufacture of parts, its further repair and assembly. Titanium alloys has thrice the service life than that of conventional piping materials.
Forms of hydrogen-induced damages
Hydrogen diminishes the in-service life of various metallic components working under harsh hydrogen and a few hydrogen-containing chemical environment 61 shown in Figure 2. Such diminishing may be noticeable as drop-in fatigue life, surface blisters and most commonly as an unexpected macroscopically brittle failure. The ASM Handbook of Materials listed five particular hydrogen-induced damages to metals and alloys. 62 They are (1) cracking from hydride formation, (2) crack initiation from internal hydrogen precipitates, (3) hydrogen-induced blistering, (4) hydrogen attack and (5) HE.
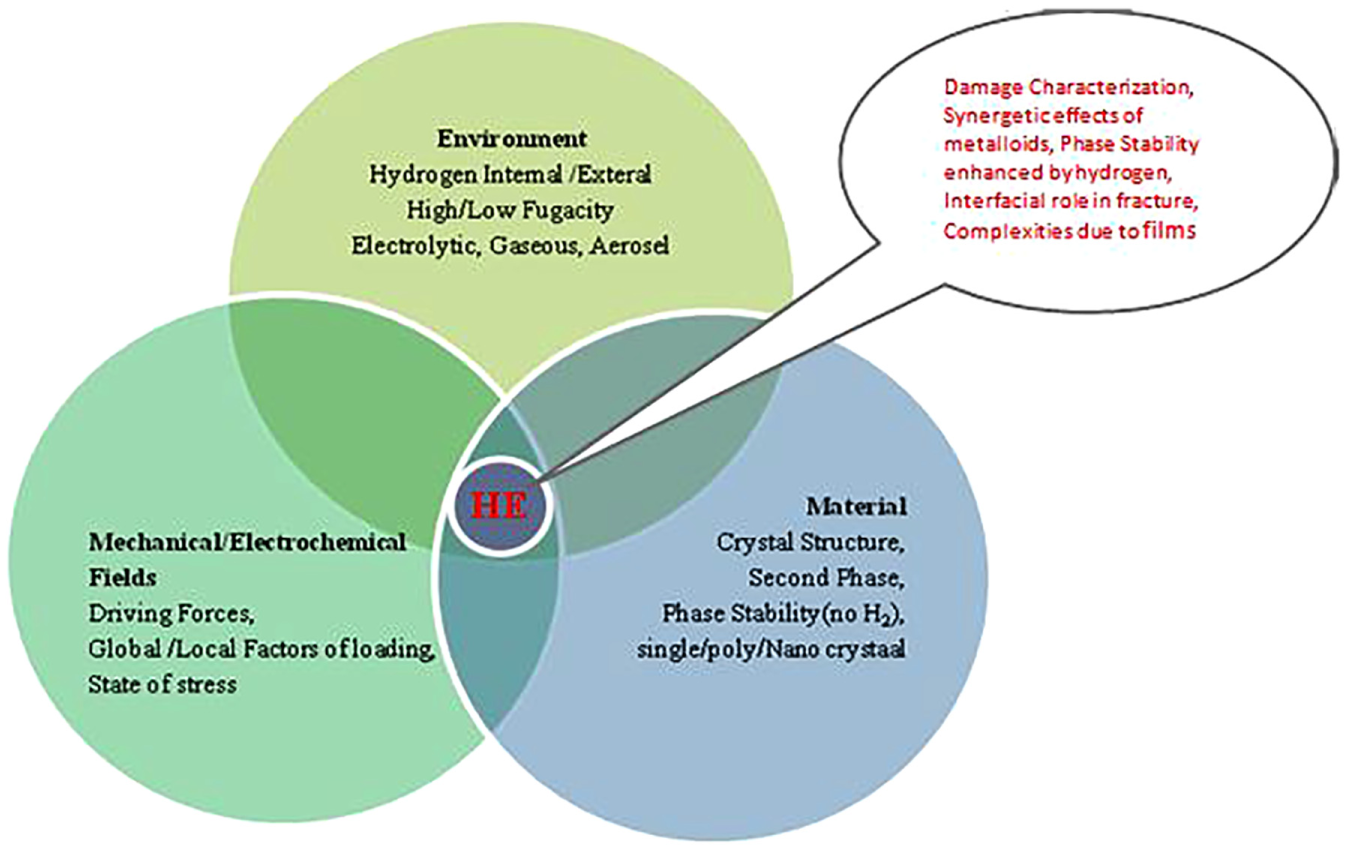
Hydrogen embrittlement interaction aspects.
Excluding HE, a phase transformation is assisted to each of the documented hydrogen-induced damage processes. Such damages are simple to recognize in a subjective fashion and can be suppressed by paying immediate attention to the damage causing process. Due to the large differences in the behaviour of hydrogen in phases (α and β) of titanium, the susceptibility of titanium-based alloys can vary markedly to a mixture of conditions and forms of HE.
Understanding the HE damage process involves unexplained challenges and taking an avoiding action for that damage process has proven a bit more difficult. Often, HE takes place in components which are in service for a time period and outcome is a distinctly intergranular surface fracture.63–68 Simply framing that root cause of failure HE or various forms of hydrogen encouraged damage is not a help to the customer except that cause is fixed with suitable recommendations that offer directions to avoid such damage in future applications.
Interaction of hydrogen with titanium
Ionic size makes hydrogen atoms very well appropriate for dissolution of solid titanium alloys. There is a solubility limit for hydrogen in titanium and its alloys. 69 Consequently, if atomic hydrogen diffusion into a titanium alloy is continued even after the hydrogen solubility limit of that alloy is attained, brittle titanium hydride phase(s) will start precipitating out. These hydride precipitates can raise the hydrogen induced cracking (HIC) of titanium alloys at lower stresses. At higher stress levels, however, an optional failure mechanism can be operational. An advance in hydrogen-assisted localized plasticity at the crack tip where the occurrence of hydrogen cuts down the stress required for plastic flow and enhanced moment of dislocation70,71 can help the HIC of titanium alloys at high stresses. The following three conditions are essential to exist at the same time for the HIC or HE of Cp titanium alloys to occur: surface hydrogen generation, temperatures above approximately 80 °C, so that the hydrogen diffusion rate becomes significant, and solution pH <3 or >12, or impressed potentials <–0.7 V (SCE). 72
However, from the above conditions, titanium alloys are fairly resistant to the ambient atmosphere; serious issues can come up when they serve in the hydrogen-carrying atmosphere which assists in picking up a huge quantity of hydrogen, particularly at elevated temperatures. The extent of the hydrogen interface with titanium alloys is instantly related to the microstructure and Ti alloying elements. Entering of hydrogen into Ti and various alloying elements start embrittlement that makes the material susceptible to cracking. Densification of metal surfaces provides slowing down of the consumption and penetration of hydrogen atoms which improves the service life of such surface-treated metals.
A similar case recently took place in 635MW CANDU heavy water reactor of Lepreau nuclear plant located in New Brunswick Canada. 73 It was reported that overpolarization of Gr.2 titanium tube sheets and tubes ends was considered as threat and given as-built coating. The coating in fact increased overpolarization at ends of tubes, which increased the protection voltage to −1.23 V Ag/AgCl. After extensive research on the tube sheets and tubes, it was reported that calcium and magnesium salts are present on unprotected tube ends. The tube walls are affected by hydride formation; in some cases, it is 80% of wall thickness as shown in Figure 3(a).

(a) Hydride formation at the inner surface of Gr.2 titanium tube and (b) crack originated at hydride zones. 73
Typically, the pH value of seawater is approximately 8 with cathodic potential of −700 mVSCE. When the potentials are driven to −1030 mVSCE and more, the pH value is approximately 13.5. Excessive cathodic potential of −1.345 to −1.36 VSCE experienced by titanium tube ends have caused the hydride formation. Generally, hydrides exhibit low ductility and are most susceptible to brittle fracture. A similar kind of cracks is noticed near thick hydriding region as shown in Figure 3(b).
The role of the passive film in retarding hydrogen absorption
A flaw free oxide film on titanium should be resistant to hydrogen. For absorption of hydrogen from various chemical and biomedical environments,74–80 it is essential to undergo redox transformations (Ti4+ + Ti3+) inside the oxide or the making of ‘hydrogen windows’ at intermetallic sites. This is a prerequisite for the threshold potential of <–0.7 V (SCE) specified by Thomas and Schutz and presented in electrochemical measurements by Vezvaie et al. 81 (see Figure 3(a)).
Figure 4(b) illustrates the oxide redox transformation-hydrogen absorption procedure, which happens at potentials tolerable negative of the flat band potential for demonstrating surface degeneracy in the oxide. 82 At these negative potentials, titanium hydrides are thermodynamically stable 83 and passive film can work as a transportation barrier shown in Figure 4. Hydrogen retention in some vanadium alloys is possible by adding the percentage titanium. 84
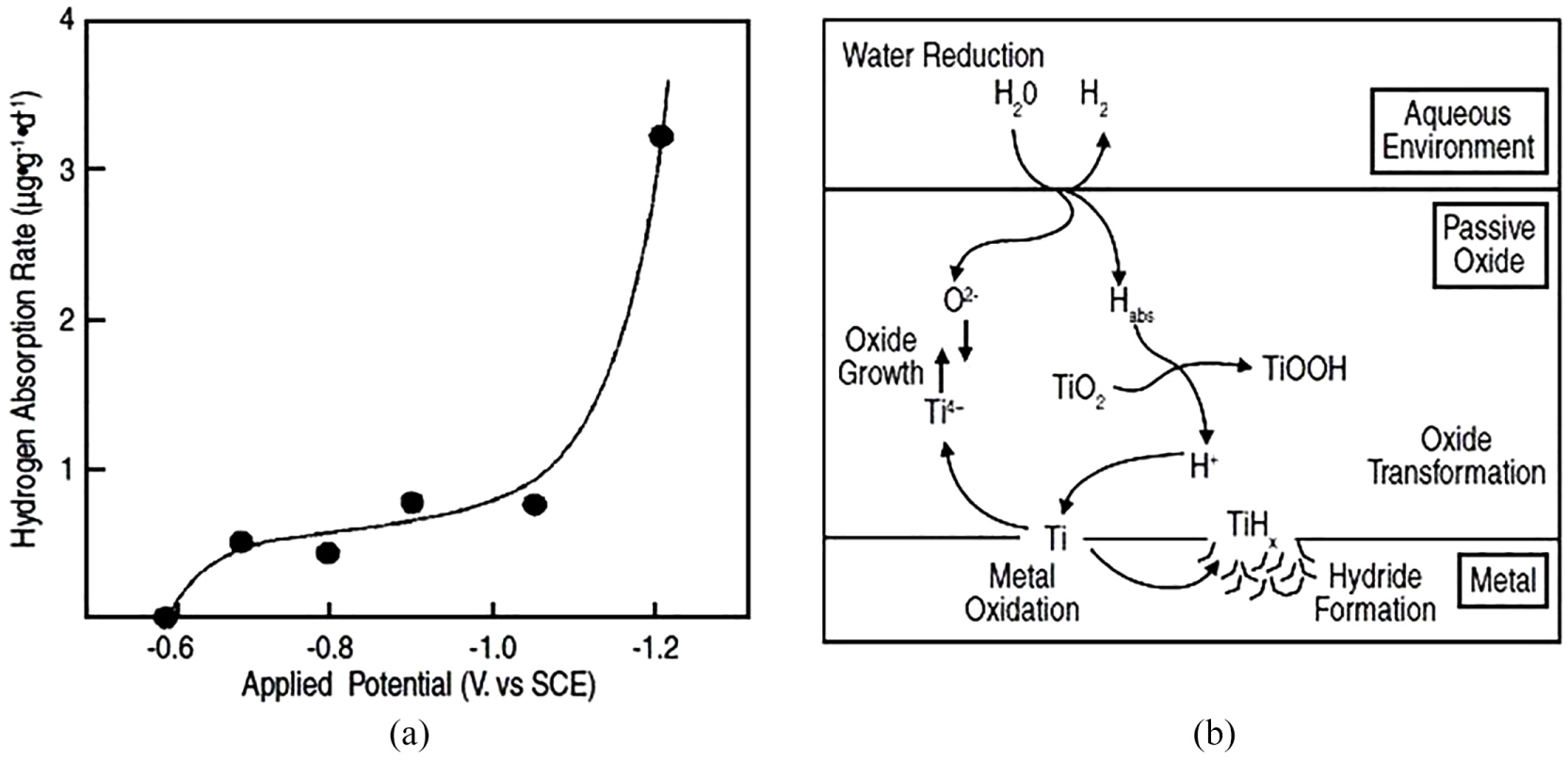
(a) Hydrogen absorption rate versus the applied potential for Ti2 and (b) schematic view of cathodic transformations which lead to the absorption of hydrogen.
Hydrogen absorption due to galvanic coupling of Ti alloys to other metals
The pairing of titanium to less active metals such as bronzes, Al and Cu-Ni alloys can also guide to improved corrosion of the pairing material.85–87 Even though no definite evidence exists, it seems unconvincing and these coupling may accomplish galvanic corrosion potentials tolerance for the cathode to make considerable hydrogen absorption by titanium. The risk of galvanic corrosion reduces noticeably when the paired metal is passive because the corrosion potentials for those metals are mostly very close.
Titanium should normally be the cathode 88 in a galvanic pair, but in some cases, 89 titanium will be the anode when paired with a dental alloys series, even though the galvanic currents are enormously low. Also, titanium grade 2 90 will be the cathode or anode when paired with various metals in hot (50 °C–90 °C) 6% NaCl. Galvanic series of metals and alloys in seawater are shown in Figure 5.
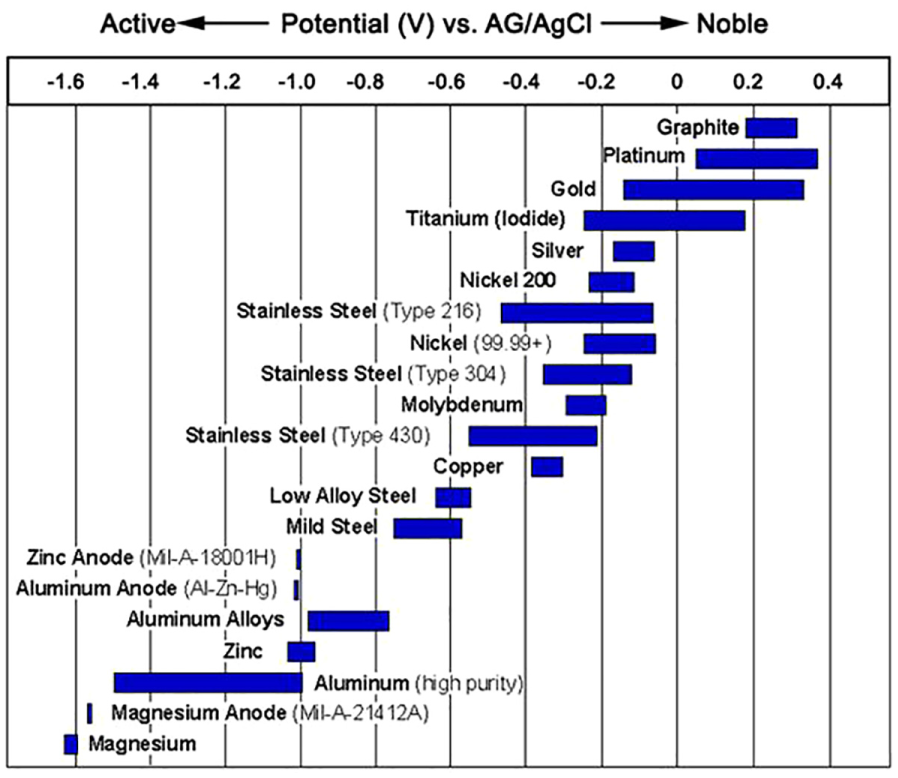
Galvanic series of metal in seawater. Potential values are shown as ranges.
Hydrogen absorption during crevice corrosion
Crevice corrosion in titanium, nickel alloys and stainless steels due to absorption of hydrogen made them severely limit their use for marine and dental applications.91–99 It can result in the failure of parts by passive film penetration and failure due to HIC improved hydrogen transport and absorption by titanium. 100 The appreciable way to cut hydrogen absorption through this route is to alloy the metal with excellent crevice corrosion–resistant alloy. Enhancement in resistant to crevice corrosion has been credited to alloying elements, which reinforce passivity. Vulnerability to crevice corrosion is reduced through the series Ti2, Ti12, Ti16.
Studies on Ti16 demonstrate that the resistance of the Pd-carrying Ti16 to crevice corrosion is taking place at 100 °C in spite of the etching of the crevice face and explicit confirmation for the acidification inside the crevice. This acidification can lead to the film breakdown/recrystallization process which takes place when the temperature is raised to >65 °C when crevice corrosion commonly initiates on the most susceptible Ti2.101,102 This is coherent with the industrial guidelines for nullifying crevice corrosion, that is, the temperature <70 °C 40 and outcomes obtained by Shibata and Zhu 103 as shown in Figure 6.
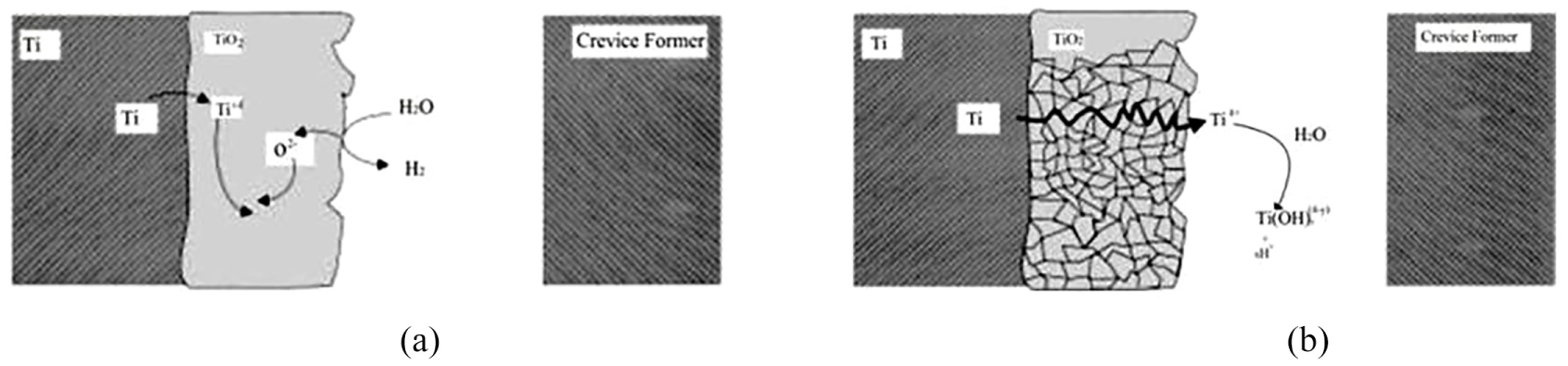
Schematic exemplifies the processes occurring within a creviced site at (a) low temperature (<65 °C) and (b) temperatures greater than 65 °C.
A study of commercial pure titanium in 1.5% HCl + 19% NaCl at 100 °C with 0.6 pH reveals that the pitting and crevice corrosion occurred on plate surface. 101 Test is performed under potential of −174 mV with saturated calomel electrode at −0.3 mA of current for 12 h of duration. The reason for the occurrence of crevice corrosion is that hydrogen gas bubbles evolve in crevice region and disperse in electrolyte present at the crevice region, which increases the resistivity leading to rise in potential drop. And it is followed by hydrolyzing of titanium ions trapped in crevice region to produce acidic local environment with pH below 1, which have chance in breaking protective film. Crevice attachment of titanium surface is shown in Figure 7.

Photograph of (a) untreated and (b) pitting and crevice corrosion attacked on crevice surfaces after treating with 1.5% HCl + 19% NaCl at 100 °C. 101
Provenience of HE
Typical mechanical ways of failures include corrosion, buckling, microfouling, 104 fracture, fatigue, creep, fatigue, thermal shock yielding, HE, aqueous stress corrosion105,106 and mechanical overload. Advanced materials like Titanium and Inconel alloys107,108 can challenge almost all these failures other than HE; the reason is that hydrogen diffusion can occur at elevated temperatures and even at relatively lower temperatures. Hydrogen damage can be done in three types: solid solution hardening, the internal defects creation and HE. 109 HE is the action by which metals such as titanium, steel and aluminium become brittle and fracture due to the subsequent diffusion of hydrogen into the metal. This was frequently a result of the unplanned introduction of hydrogen during casting and finishing operations. HE has been a serious problem for metals in various industries, especially in gas production and petroleum, where high concentrations of H2S are frequently met.
HE – internal
This form of HE occurs when hydrogen entrapped in the metal during its processing.110–112 It may lead to the morphological failure of metal, even though the metal never before exposed to hydrogen. The effect can be seen in the temperature range of 173–373 K and is almost severe at room temperature.
Environmental hydrogen embrittlement
This form of HE arises when the material is exposed to environmental hydrogen.113–115 Induced hydrogen alters the properties of the metal substantially without change in phase. 116 The effect is purely based on the applied stress and was severe at room temperature.
Stress corrosion cracking
Stress corrosion cracking (SCC)117–123 type of ecological HE acts as a particular condition where cracks are initiated and distributed under mutual effects of corrosion and stress environments. The primary corrosion may take place at maximum stress point causing the development of a microscopic crack, 124 which can be either transgranular or intergranular.125,126 Continuous exposure to the corrosion environment will spread the crack, making it a possible severe catastrophic failure. Different mechanisms have been proposed to give an explanation of HE. Actually, within a given system, based on the nature of the applied stress and source of hydrogen, the mechanism may vary. Metals such as alloyed steels,127,128 zirconium, titanium and niobium can outline stable hydrides emerge to fracture by a cleavage mechanism and induced stress hydride formation. Other mechanisms, such as adsorption decreased surface energy and high-pressure hydrogen bubble formation, have also been hinted and may play a role in particular systems.
Looking insight of failure analysis 129 of first stage of vacuum condenser boot is shown in Figure 8. The tubes and sheets of condenser are made of titanium grade 2 and carbon steel. The role of condenser is to cool the mixture of hydrocarbon in cooling water. The outlet temperature of condensate is 20 °C and pH value ranges from 3.6 to 4.1. The chloride concentration is 42–56 ppm but recommended is 10 ppm. As a result, deposits formed on the outer surface and scales inside titanium tube and tube sheets.

(a) General view of the first-step vacuum condenser. (b) The hole (10 cm) in the bottom (carbon steel boot). (c) The rusted upper part of the condenser. (d) Deposits on the outer surface of titanium tubes in the condenser. (e) Scale inside the titanium tubes and on the tube sheet (cooling water side). (f) The opposite side (exit of cooling water). (g) Aluminium sacrificial anodes in cooling water (inlet channel). 129
HE of metals
Commercial pure titanium
For an instance, absorption of hydrogen by Cp titanium 130 in acidulated phosphate fluoride (APF) solutions of different concentrations is through hydrogen thermal desorption analysis (TDA). Since titanium has higher affinity with hydrogen, breakage of titanium passive film perhaps shows the way to hydrogen absorption. The hydrogen absorption limit of alpha titanium is mostly tens of ppm mass at room temperature and it turns to hundreds of ppm mass with the rise in temperature. If titanium alloys absorb considerable amounts of hydrogen from fluoride-containing solutions for short term, HE will become a severe problem of titanium in the medical field, especially in dental application. The amount of hydrogen desorbed is being measured by hydrogen TDA. 131 Figure 9 clearly shows the increase in the concentration of APF and immersion time hydrogen desorption increased drastically.
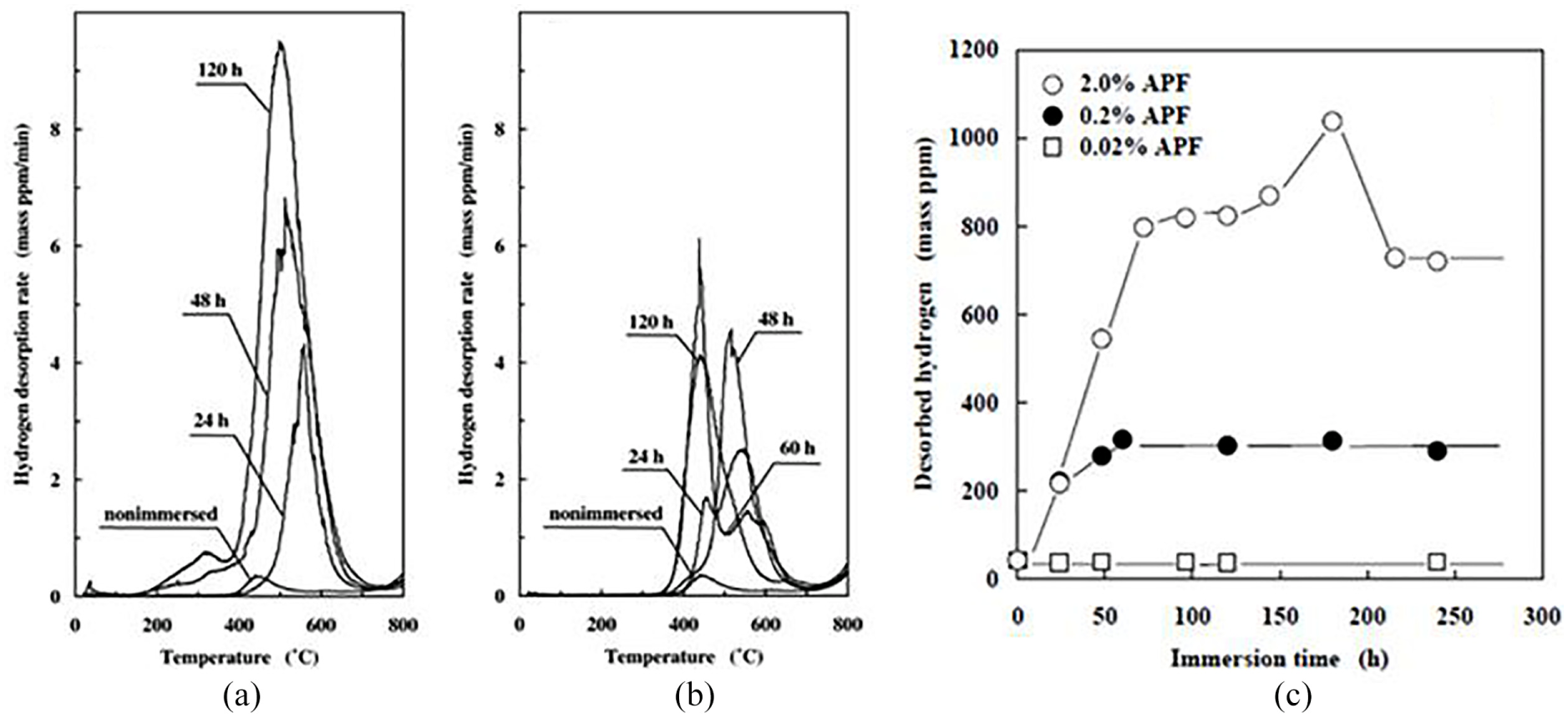
Hydrogen thermal desorption curves for various immersed time periods: (a) 2.0% and (b) 0.2% APF solutions. (c) Desorbed hydrogen obtained from thermal desorption in 2.0%, 0.2% and 0.02% APF. 131
Many recent articles have reported that commercial pure titanium grade 2 is effectively immune to hydrogen-induced cracking in 3.5% and 6% NaCl aqueous solution. All undertaking made to improve this form of cracking had no result on the elongation to failure. These comprise making the acidic solution, the temperature rise of the test solution to 70 °C, the cathodic potential of −1400 Mv (SCE), using a hydrogen recombination, and low crosshead speed which was possible with a testing machine. These results raise two critical questions. The first query is whether the titanium grade 2 is naturally immune to hydrogen cracking, if not, any other factors during experiment are preventing hydrogen from penetrating the material and causing embrittlement. The second question is why the titanium grade 3 material was much susceptible to hydrogen cracking than that of titanium grade 2 material. When grade 2 material is exposed to gaseous hydrogen at high temperatures, it is very much prone to HE. It concludes that the expected reason why embrittlement is not seen in 3.5% NaCl aqueous solution is that the kinetics of hydride precipitation is very slow to influence crack growth. The rate of hydride precipitation in titanium grade 3 is higher than in titanium grade 2 due to gaseous state of hydrogen at higher temperature, which may be the reason for hydrogen-induced cracking susceptibility of titanium grade 3.
Alpha + beta titanium
Based on the literature,132–137 most commonly used dual phase 138 alpha-beta titanium alloy is Ti-6Al-4V. Ti-6Al-4V alloy showed corrosion problems when the alloy is not surface treated and exposed to environments. Hence, there is a necessity to understand the corrosion of Ti-6Al-4V thoroughly under various environmental conditions.
A cyclic polarization technique will help in calculating the crevice and pitting corrosion resistance of Ti-6Al-4V 139 in different environments (marine, industrial and chemical) and at temperatures ranging from 25 °C to 500 °C. As a final point, SEM was accustomed to understanding the ruin of the alloy under different environmental conditions. HE has its effect towards mechanical properties of the Ti6Al4V 140 alloy if preventive measures are not taken into account. An electrochemical experiment is conducted on the uncoated and TiN-coated workpiece by immersing in 1 N of HCl for 150 h. A current of 5 mA/cm2 is applied using electrochemical analyzer G PARC273 and EG. The titanium electrode acts as a cathode where H+ was reduced to H2 gas on the electrode surface. Elastic recoil detection analysis (ERDA) is used to calculate the concentration of hydrogen in the substance. Figure 10 shows the example for ERDA spectra plotted during measurement incorporated counts of hydrogen peak incident α-particles number which is relative to the hydrogen concentration, and width of the hydrogen peak is relative to the penetration depth of hydrogen.
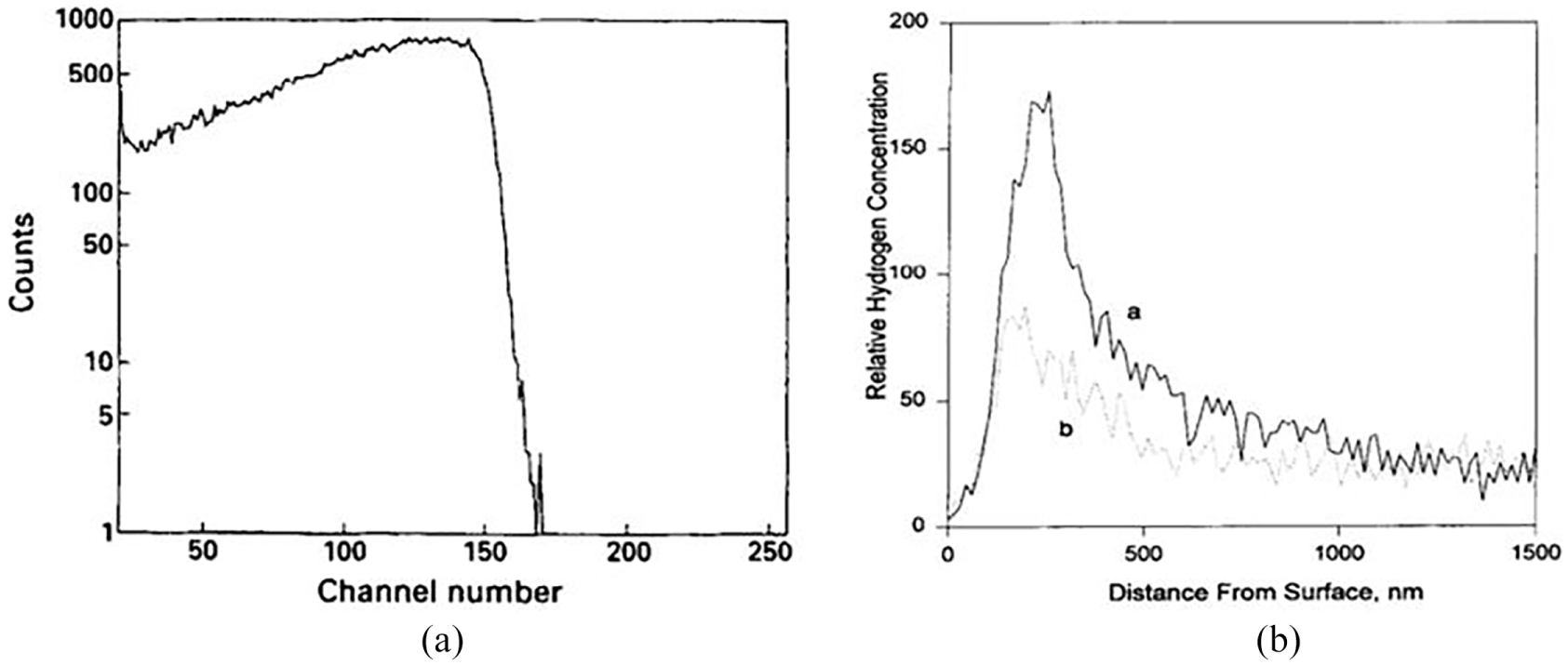
(i) ERDA results for the reference sample. (ii) Hydrogen concentration with respect to distance from the surface. (a) Uncoated and (b) TiN-coated workpiece. (B. S. Yilbas et al. 140 ).
Beta titanium
Contradictory opinions are found towards the influence of hydrogen on mechanical properties and microstructure of β-titanium alloys. 141 Three commercial alloys Alloy-C, Ti-10-2-3 and Ti-21S are charged with hydrogen gas phase. To differentiate the extrinsic and intrinsic effects, the hydrogen charging is carried in the second step of two-step heat treatment in vacuum. In order to create hydrogen charging, the hydrogen release and uptake in the form of gas phase should be calculated, applying, for example, volumetry and thermogravimetry.
Thermogravimetry permits to monitor the increase in weight of a specimen with hydrogen uptake from He/H2 gas mixture. Normally, the solubility of hydrogen decreases rapidly with the rise in temperature, representing the exothermic nature of the uptake reaction. Figure 11(a) shows the hydrogen concentration as a function of square root of temperatures and hydrogen partial pressure. The most prominent hydrogen effect is a swing of curves to less ΔK values in combination with a considerable decrease in the threshold of fatigue crack as indicated in Figure 11(b). Four-point bend fatigue crack growth measurements on notched alloy C are shown in Figure 11(c) for 0.1 stress ratio R. The crack propagation rate da/dN is plotted with respect to stress intensity factor ΔK.
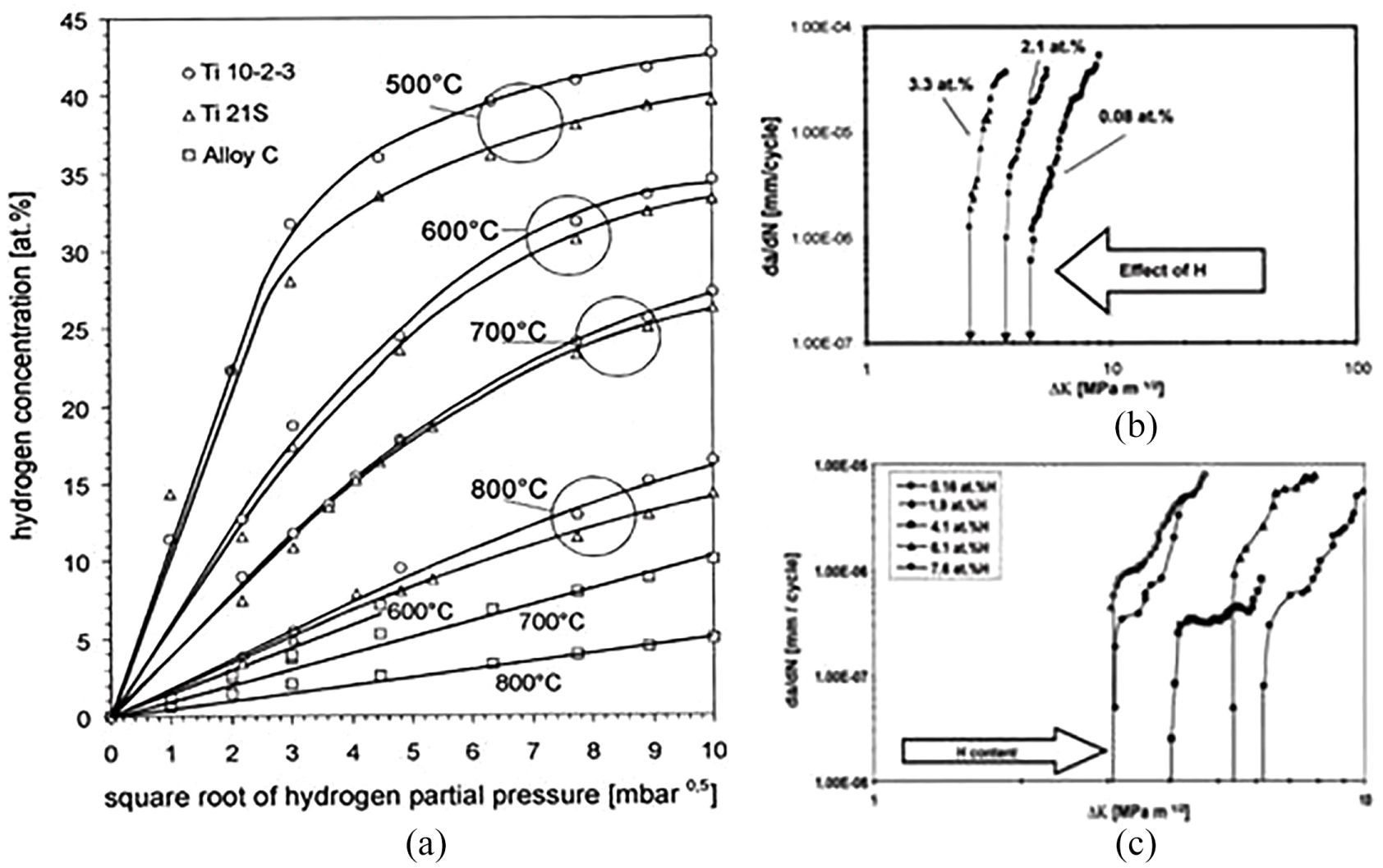
(a) Dissolved hydrogen concentrations with respect to hydrogen partial pressure. (b) Fatigue crack propagation of alloy C with various hydrogen concentrations at R = 0.1. (c) Fatigue crack propagation rate of Ti 10–2–3 with various hydrogen concentrations at R = 0.1. 141
On observation, it can be stated that the rise of hydrogen concentration in alloy C causes an embrittlement directly indicating the decrease in fatigue crack propagation threshold. Compared to the performance of alloy C fatigue crack propagation rate, threshold value of Ti 10–2–3 raises with the increase in hydrogen content. This behaviour is because of dissolved hydrogen showing a suppressing effect for the formation of α-Precipitate in the β matrix during cooling at room temperature. The suppression effect by hydrogen helps in reduction of crack tip local stress, which can be attributed to low yield strength where the α-Precipitate is slightly suppressed. Conflicting to the results on alloy C, hydrogen changes the transition range to lower temperature.
Methodology for suppressing HE
Among the multiple approaches shown in Figure 12, elimination of residual stress by surface modification is a most significant step. There are mainly three types of surface modifications: surface modification without changing material surface chemically,142,143 with changing material surface chemically 144 and adding new material to the surface (coating).
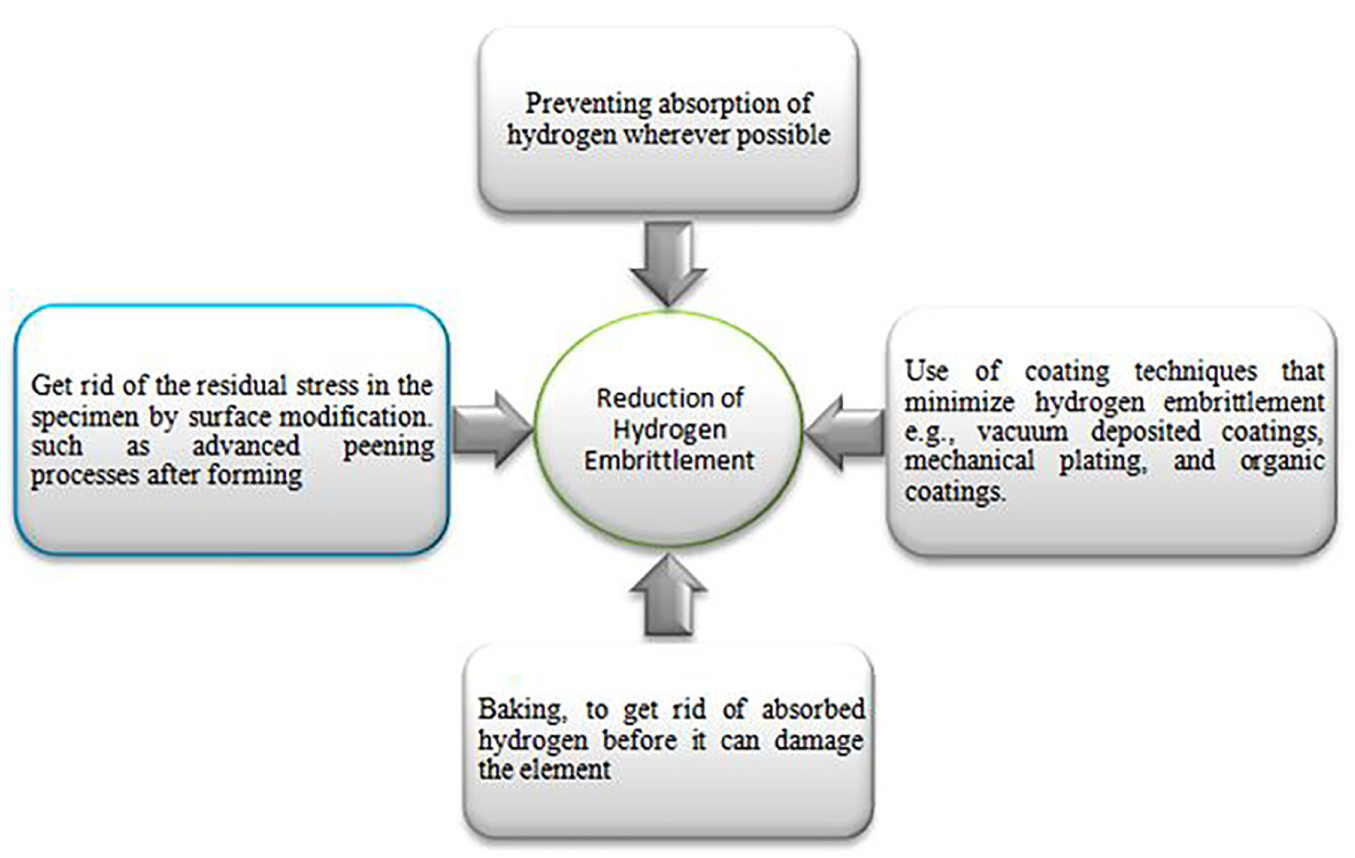
The line of actions for reducing HE.
Role of compressive stresses in suppressing hydrogen-induced damage
Compressive stresses can be induced into a material by means of conventional shot peening, cavitation peening and advanced pulsed laser peening (LP) processes. The role of compressive stresses in suppressing the hydrogen causing damages in various materials is reviewed briefly.
There is a well-built linear correlation between compressive stresses depth profile and hydrogen effect in austenitic stainless steel. 145 When the material is cavitation peened prior to hydrogen charging in 0.5 mol/L sulphuric acid solution at temperature 50 °C, the compressive residual stress which is beyond or nearly equal to yield stress significantly decreases the attack by hydrogen. The suppression of hydrogen attack increases with a rise in the compressive residual stress. And when the material is processed with LP 146 under similar conditions, the compressive stresses induced by laser shock peening (LSP) lead to a considerable decrease in average grain size and increase in dislocation density helped in reducing HE susceptibility and improved tensile strength. Comparing the results of two peening processes, LP provides an enhanced peening effect and microstructure in steel grades. Similarly, conventional shot peening–induced compressive stresses 147 towards reducing HE in low carbon steel 148 purely depends on harsh hydrogen environment, testing conditions and for stainless steel 304 it depends on phase transformation promoted by cold working. The compressive stresses show a remarkable effect of retarding hydrogen damage towards advanced materials. Hastelloy C-22 149 when subjected to annealing as well as laser peened prior to hydrogen and subjected to the tensile test. The hydrogen-assisted fracture caused by internal hydrogen calibrated with the tensile test is brought down by LP compared with the annealed specimen. The role of HE in laser-peened TC4 150 titanium alloy is examined by a performance test. The results showed an increase in hardness and decrease in compressive stresses of laser peened after electrochemical hydrogen charging. Elongation is reduced in both peened and unpeened TC4 samples with the evidence of an increase in toughness and HE susceptibility reduction.
Surface modification of metals
Surface modification is the step of altering the surface of a bulk material by enhancing mechanical, tribological, 151 chemical and biomedical 152 properties unlike from parent. The modifications can be made by various methods with the horizon to altering a wide range of characteristics of the surface, such as surface charge, surface energy, roughness, reactivity and biocompatibility.153–158 Among all these surface modification processes, the surface modification without changing material chemically is most desirable. In mechanical surface modification, the treatment process is based on the elastic and plastic cold working of the surface.
The compressive residual stresses are induced in the material and surface layers of metal are work hardened, thus the surface gets resistance against crack initiation and growth, friction fatigue increases or corrosion fatigue significantly and therefore improves the structural performance under fatigue loading. There are numerous mechanical surface treatment processes as shown in Figure 13.
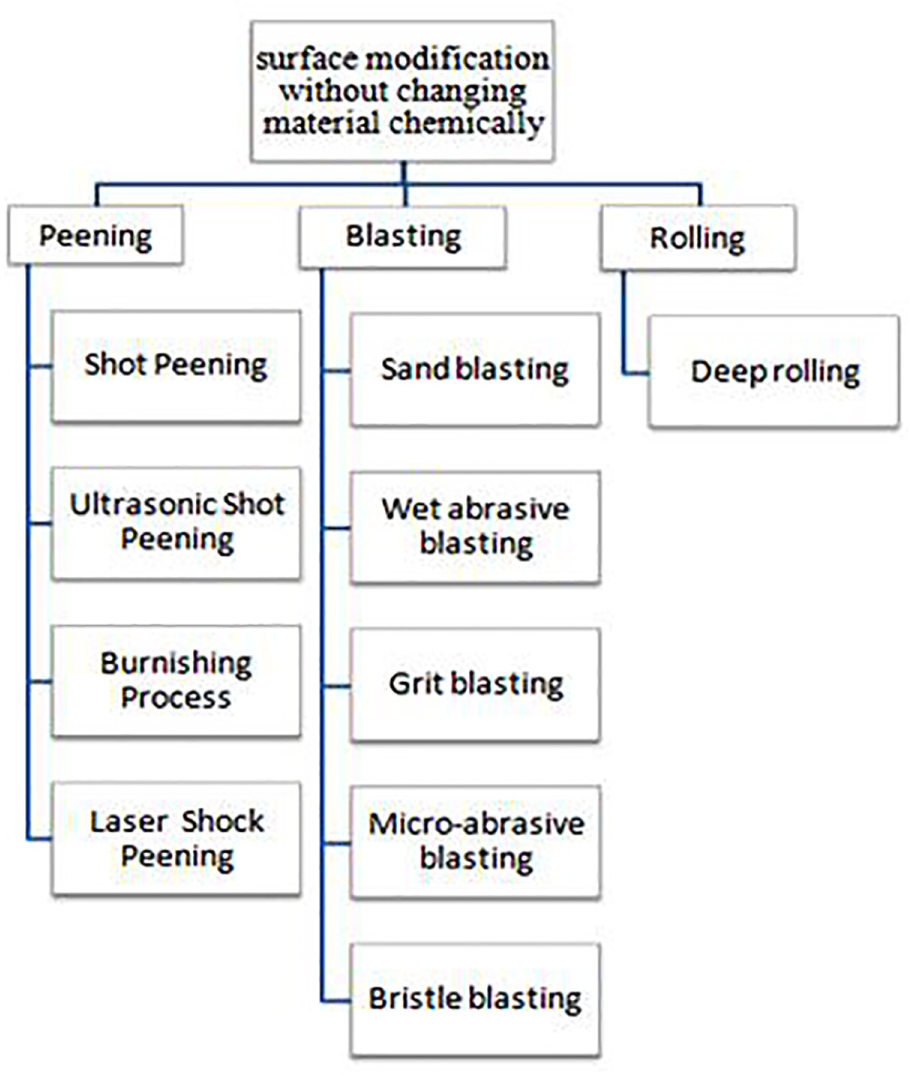
Classification of surface modification without changing material chemically.
Shot peening
Shot peening (SP) is a traditional process applied to harden surface by inducing compressive residual stress in sub-layers and altering surface properties of various metals.159–162 The major surface alterations due to SP are microstructures, residual stresses, cracks, crystallographic texture, 163 surface roughness, dislocation density164–169 and hardness by work hardening as shown schematically in Figure 14. The process leads to a collision of a surface with a shot (ceramic particles, metal balls and glass whiskers) with a sufficient and high kinetic energy 170 to produce elastic and plastic deformation, and every piece of shot that hits the metal acts as a minute peening hammer contributing to the surface a tiny dimple. To make a dimple on a metal surface, the sub-layers should yield in tension and try to return its original shape, thereby producing concave of the cold-worked portion below dimple with extremely stressed in compression.
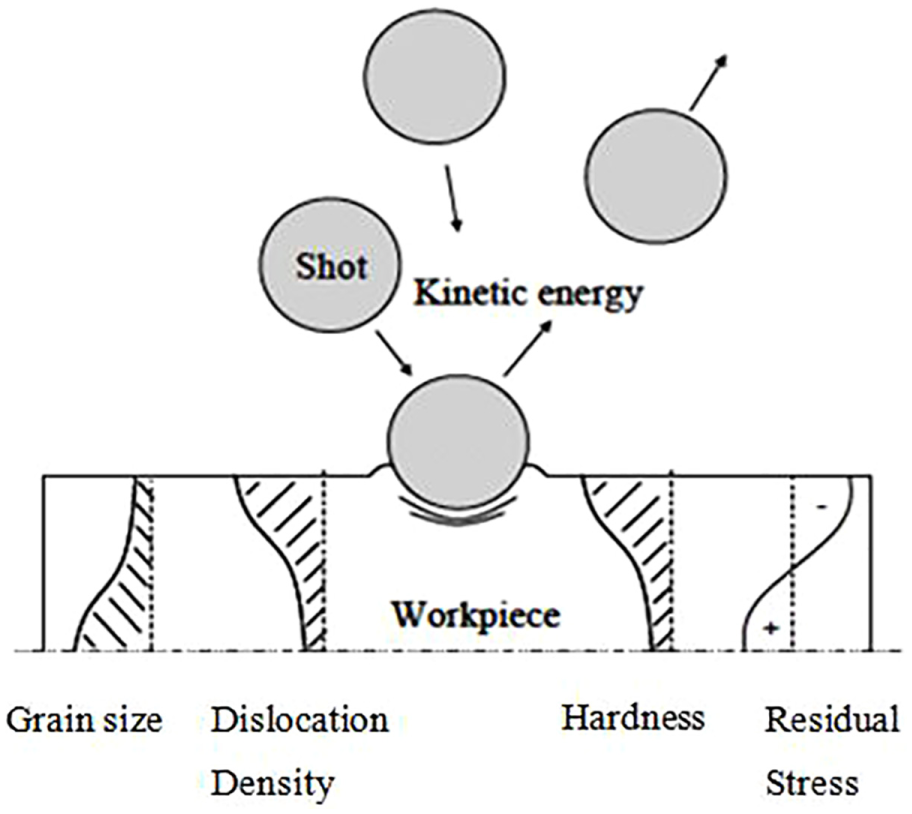
Schematic representation of the shot peening.
Ultrasonic shot peening
In recent times, ultrasonic shot peening (USP) 171 came into existence to enhance the durability of in-service parts of various materials.172–178 USP is different from shot peening where compressed air actuates the shot, and the shot is energized at an ultrasonic frequency by a sonotrode vibrator. One of the USP devices developed by SONTAS in France, called Stressonic, is illustrated in Figure 15. The process is carried out by keeping shot into a random spinning motion within a peening chamber to impact subjected metal surface. The effect of the peening action depends on the placement of the sonotrode in relation to the target.
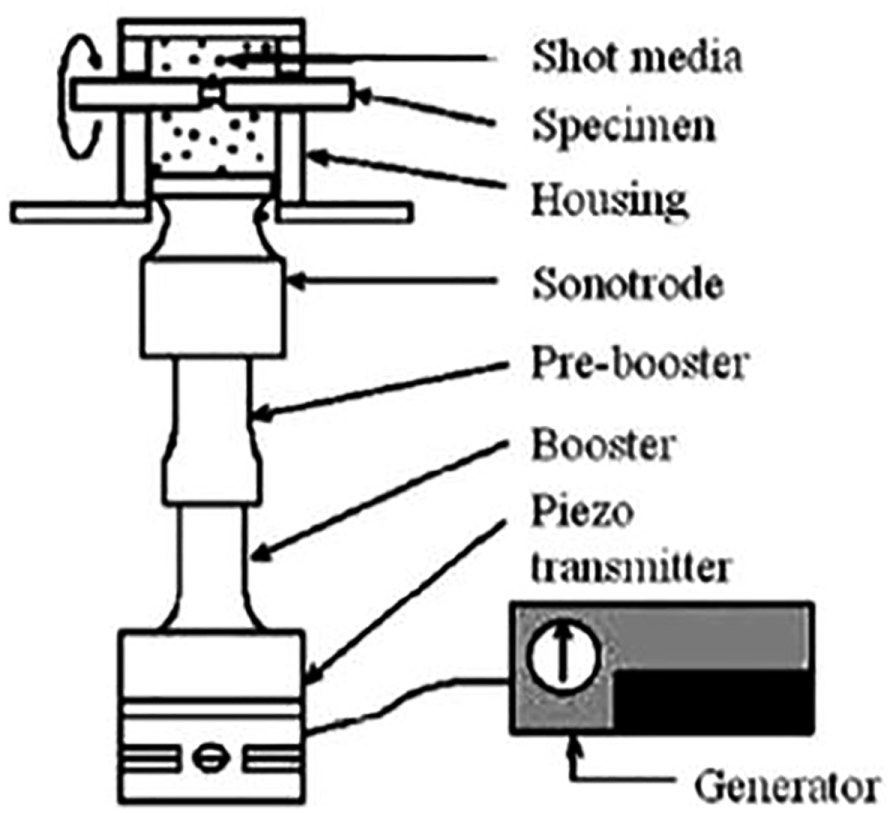
Schematic representation of the ultrasonic shot peening.
The outcome of USP process is smooth surface as compared to shot peening because of using smoothened large diameter ball. Moreover, it can provide a large amount of energy in little time because of high frequency. 179
Burnishing process
Burnishing is a mechanical process in which specimen is dynamically loaded to enhance the fatigue strength. It can be performed using roll (roll burnishing), 180 laser burnishing 181 or balls (ball-burnishing). Ball-burnishing (BB) process helps with the ball rolling on the surface exerting a normal force on the surface of the workpiece182–184 as shown in Figure 16.
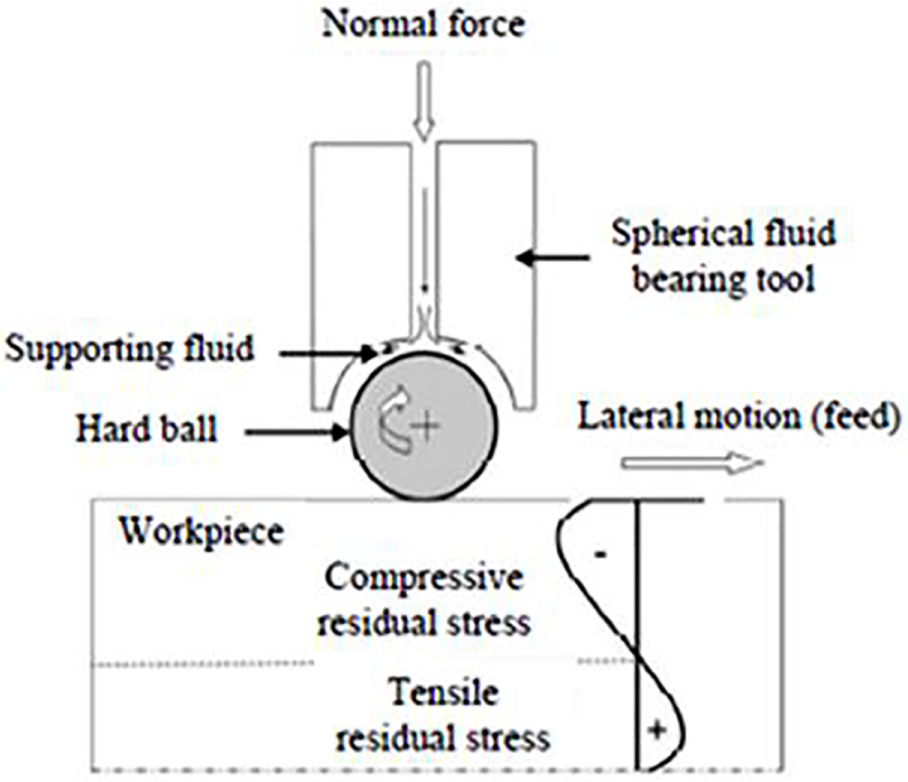
Schematic representation of the burnishing.
The pneumatically or hydrostatically pressurized ball can generously spin in all directions even at high speed inside the ball retainer. Also, it can be efficient with computer numerical controlled (CNC) milling, drilling, lathes and physically operated machines. After passing the yield point of a material, plastic flow takes place in original asperities. This process points towards a smoother surface 185 and a significant amount of induced compressive residual stress than that of the SP and USP. 186 Induced compressive residual stress will tag a sequence of a favourable effect on mechanical properties and improve fatigue, 187 corrosion resistance 188 and strain hardening of different metals under dynamic loads.
LSP
LSP or LP 189 is one of the advanced peening processes for making the metal surfaces and sub-layers densify and for changing surface chemical activities. To the surprise of the engineers, in contrast to water jet peening, 190 shot peening, cold rolling, high-pressure torsion, laser work hardening, in terms of compression layer depth, mechanical properties enhancement, and high level of induced stresses stability in metal191–198 made the LSP the most effective surface modification technique. LSP will set up a beneficial microstructural transformation in the material surface and enhances the mechanical performance of a component. It has been employed to a series of metals and alloys used in aero-engines, airframes, cryogenics199–205 and some engineering applications to enhance the compressor blades leading edges to damage tolerance, automobiles parts, dies and medical implants206–209 and so on.
The principle of LSP is explained in Figure 17. Laser pulse from a Q-switched laser with high-power density strikes a metal surface inducing shock waves in surface and subsurface layers of the metal.210,211 The shock waves diffuse in an exponential manner creating a plastic deformation in the subjected volume. 212 Before LSP, the surface of the metals or alloys is commonly coated with a black tape or paint and inertial tamping stream (water) is flown on coating as shown in Figure 17.
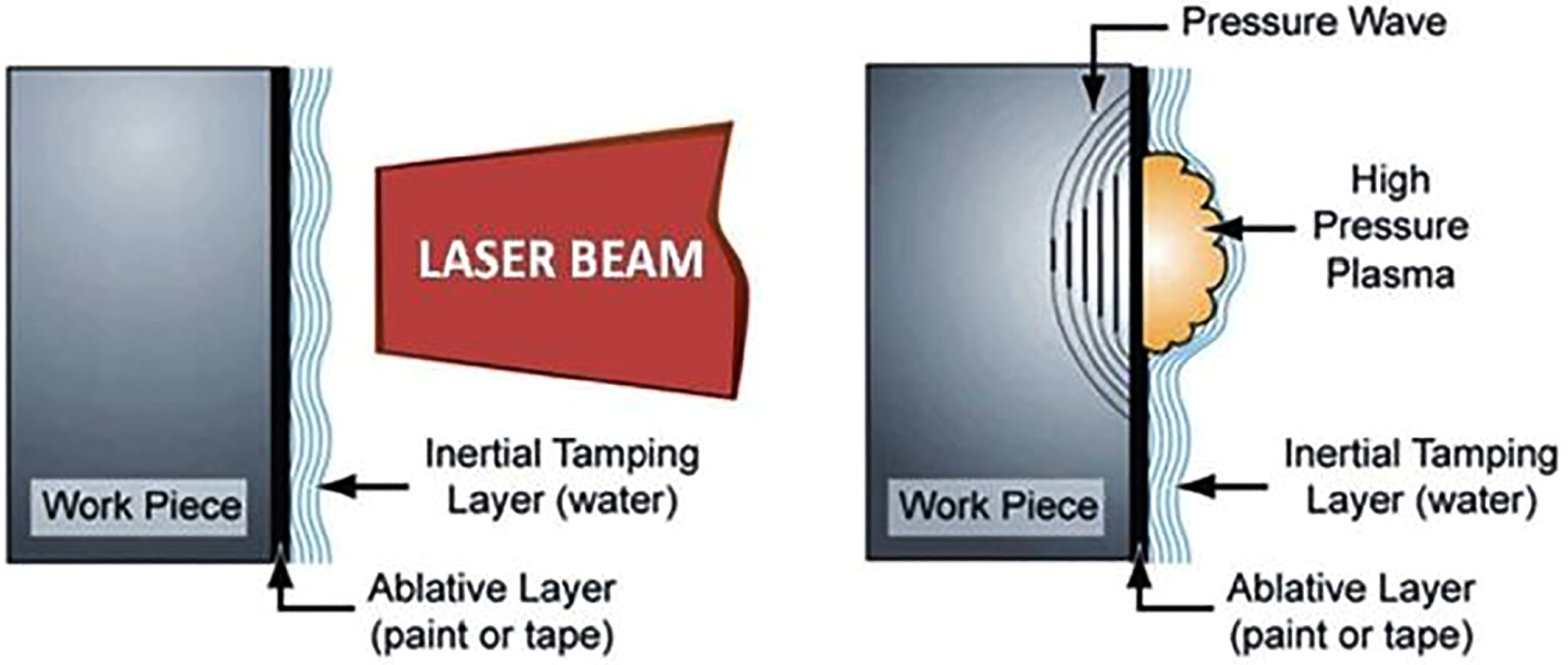
Schematic view of the laser shock peening.
When the pulsed laser beam is guided onto the metal surface, it passes through the inertial tamping layer (water) and strikes the ablative (taped or coated) sample. Due to absorption of laser beam energy, the black tape or coating is heated and vapourized. The leftover radiation of laser beam is absorbed by vapour and gives rise to plasma plume. The rapidly intensifying plasma is pinned between the transparent inert layer (water) and metal, creating an enormous pressure on the surface, which diffuses into the metal as a shock wave as in Figure 17.
If the metal surface is sufficiently protected from the intensifying plasma by an absorbing layer, the LP becomes strictly mechanical. When the shock wave generation takes place together with surface ablation and melting of metal, the treatment becomes thermo-mechanical process. 213 High compressive residual stresses induced are worthy of enhancing fatigue and wear life of metal.214–216 The LSP is capable of enhancing resistance to crack growth after foreign object damage (FOD) with postponed crack initiation which is precious for aero-engine compressor blades.217–219 And supportive in fixing spinal implant, for a given fatigue strength Ti rods get better in flexibility by LSP. 220 Ti alloys are desirable for high-speed centrifugal compressor motor rotors, in which LSP process helps in retarding fatigue crack growth rate. 221 Advances in laser technology have transformed with high-throughput production using new femtosecond lasers, though the level of work hardening is alike as older nanosecond laser-based peening.222,223
The important downside of LSP is it needs a complicated laser system particularly intended for production functions with large pulse energy up to 100 J and high power. From another point of view, LSP without coating uses a compact laser system with pulse energy of 0.1 J and smaller power to reduce thermal effects can be available commercially.224,225
Shock wave propagation
The volume of plastic deformation in metal is accurately based on laser magnitude and pressure pulse. As the shock wave advances through the workpiece, its magnitude decreases with respect to the attenuation rate. Beyond assured depth, the magnitude is under the proportional limit which helps for elastic deformation. The concluding effect of the development is a residual stress in the workpiece. The surface region of the workpiece has compressive stresses and the tensile region beneath it. The dynamic yield strength of a material is a function of temperature, strain and strain rate.
When a shock wave of a magnitude beyond the dynamic yield strength propagates through a material as shown in Figure 18, it will take a definite shape based on the material properties and pressure magnitude (Adapted from Meyers MA, Dynamic behaviour of materials and Kolsky H, Stress waves in solids).226,227 An important parameter that defines the shape is the Hugoniot elastic limit (HEL), and it is defined as the axial stress necessary for a metal to deformation plastically in a uniaxial strain state. The correlation for the dynamic yield stress (σyd) and the HEL (σHEL) is

where v is Poisson’s ratio.
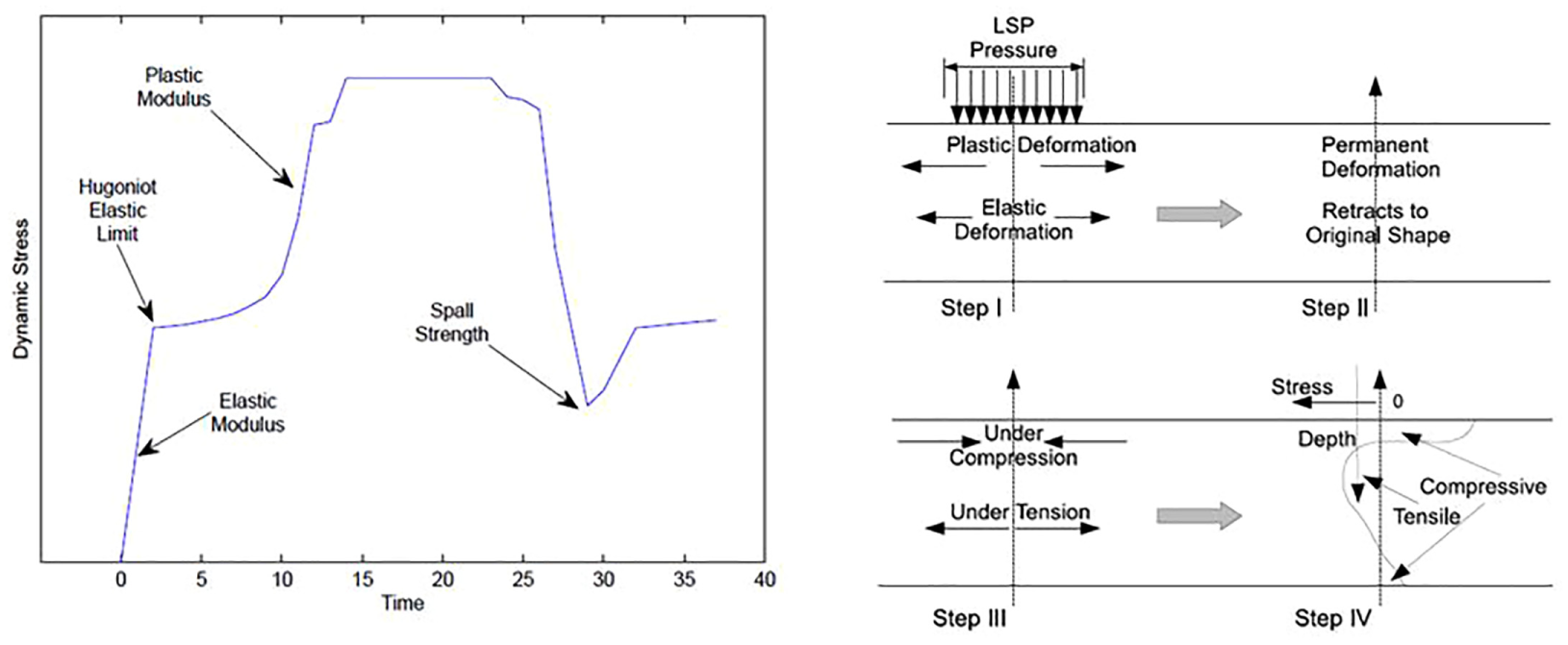
Shock wave in a material and schematic of residual stress generation.
On applying laser shock wave pressure, it will generate two types of stress waves, namely, plastic and elastic, in the material (Adapted from Meyers MA, Dynamic behaviour of materials and Kolsky H, Stress waves in solids). According to the theory of stress wave propagation, the speed of the plastic (Vp) and elastic (Ve) waves are given in equations (2) and (3), respectively
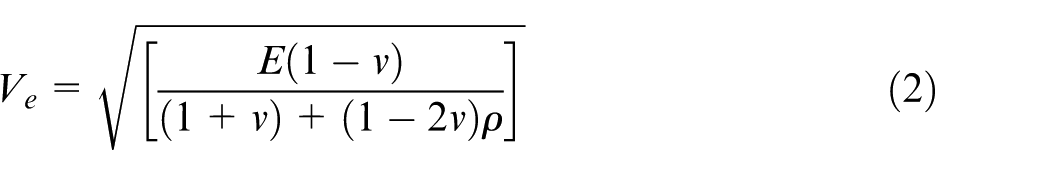

where
Residual stress generation
The complex relations between waves in the metal create compressive and tensile stress regions. Based on the applied magnitude of the pressure, the sub-layer under the surface layer also starts expanding as shown in Figure 18, but the expansion is elastic and normal to the pressure applied. Depending on the rate of attenuation, the expansion reduces with depth and beyond a certain depth, the expansion is completely elastic. The elastically deformed region of the workpiece tries to spring back to its original shape, but plastic deformation is permanent.
The compressive stresses build up on the surface region of the metal,
228
and below the region of compressive stresses, there exists a tensile stress region. Depending on metal properties, pressure pulse properties and component geometry, the tensile region may be in between the two compressive regions. Depending on the component dimensions and the material properties, the compressive region sometimes may or may not present at the bottom. Compressive stress at surface region helps to lessen the chances of crack tip opening and growth. In the absence of external force, the tensile stress region is created to balance the compressive stress for equilibrium. The physics behind the formation of stresses is explained in Figure 18. The compressive stresses depth is correlated with the depth of plastically affected zone. An empirical expression for surface residual stress
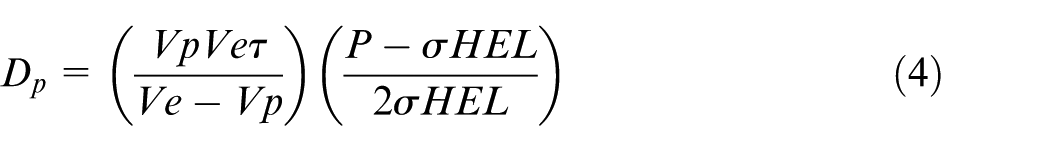
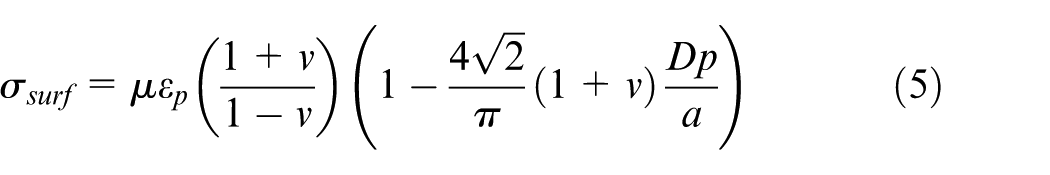
Here, Vp, Ve, P,
Authors have investigated the distribution of residual stress in Ti-2.5Cu after subjected to LP without coating. 229 Ti-2.5Cu samples are peened with Q-switched Nd:YAG laser with 1064 nm wavelength, beam diameter of 0.8 mm, pulse duration of 10 ns, repetition rate of 10 Hz and pulse energy of 350 mJ. Every increase in 10% overlap rate of laser beam would mean increase in compressive residual stress deeper in the material till 1000 μm. A similar trend has seen for all overlap rates for single power density 6.97 GW/cm2, with change in intensity of compressive residual stress as shown in Figure 19. There is no sign of tensile residual stress till 1000 μm, and sample peened with 70% laser spot overlap rate is considered as optimal work hardening parameter. By increasing the laser spot overlap percentage or laser power density, the deformation may increase beyond the favourable limits. In-depth review of LP over titanium alloys and its effects is discussed in Table 2.
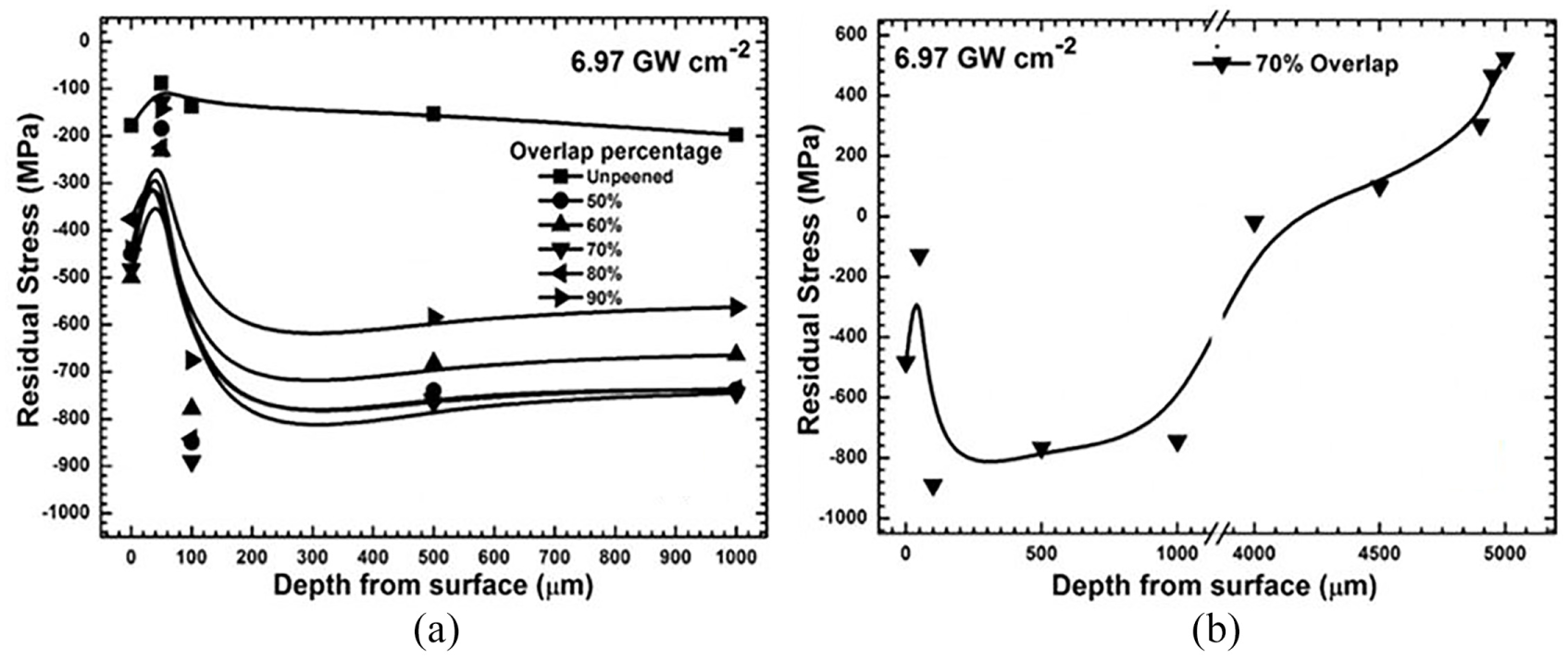
(a) Distribution of residual stress in samples laser peened with various laser spot overlap percentage and (b) in-depth residual stress measurement of sample peened with 70% laser spot overlap. 229
Effect of LSP on titanium and various alloys.
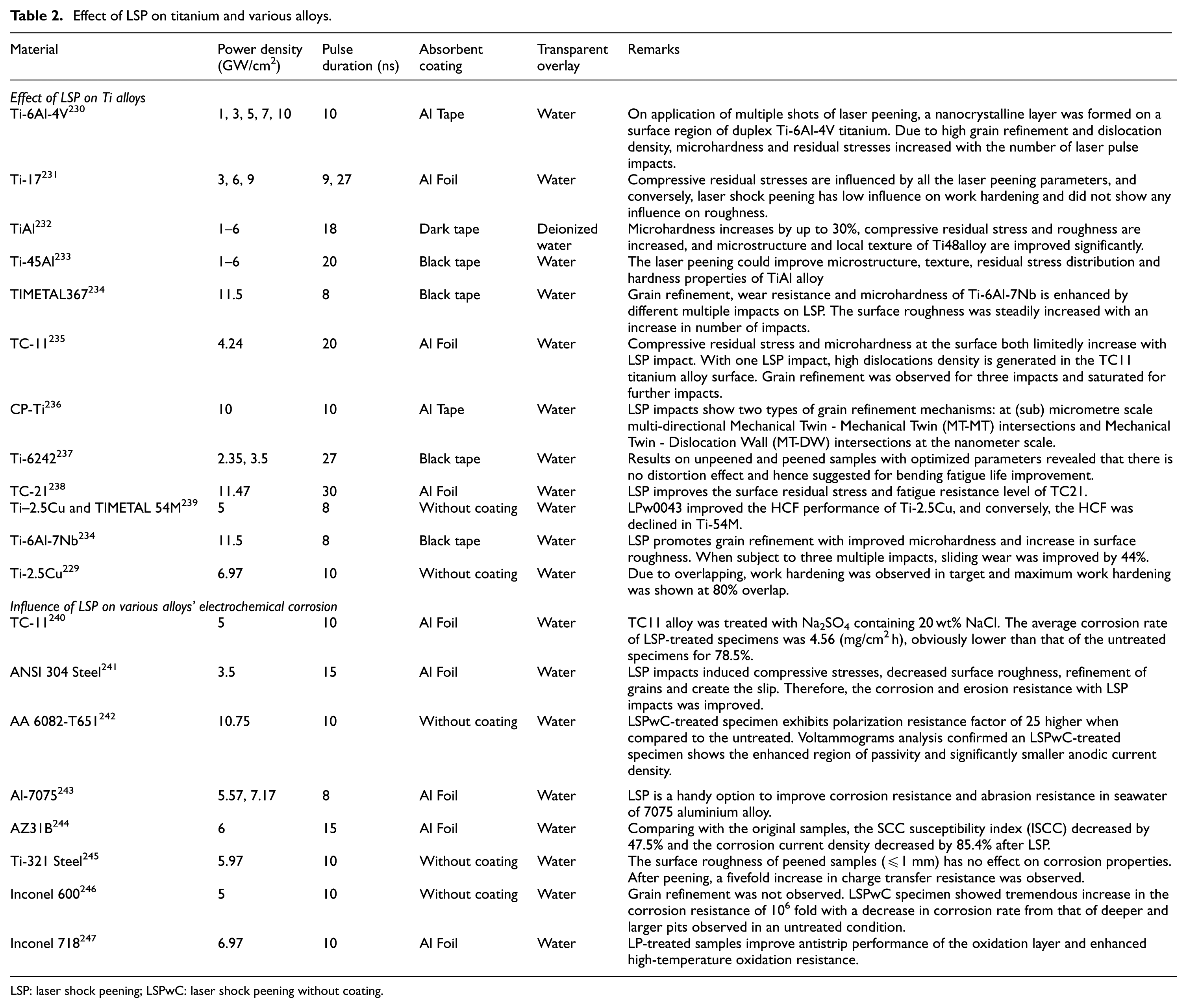
LSP: laser shock peening; LSPwC: laser shock peening without coating
Role of LSP in shielding HE
One of the earliest patents on LSP of titanium alloys 248 directed towards improvement of shielding HE. In this patent, authors disclosed that a particular titanium alloy Ti6Al4V laser peened with the beam comprising an 18 ns pulse width and at power density of 10 GW/cm2. Two layers of LSP were applied to all sides of the coupon. Cathode process is carried out by immersing both as-received and laser-peened specimens into H2SO4 at ambient temperature. The power supply was connected by three electrodes and provided a constant, uniform current density of 10 mA/cm2 to the specimen over a charging of 144 h.
The parameters for the secondary ion mass spectroscopy (SIMS) measured are as follows: a specimen 250 µm × 250 µm in size was mastered on the surface of the coupon, to a depth of 85 µm. The ions collected were compared with a standard reference material (352c) containing hydrogen concentration of 49.0 + 09 µg/g in unalloyed titanium. Four main ions recorded in all samples are 16O−, 1H1−, (48Ti+1 6O)− and O3−. Recorded data compared with the standard laser-peened sample shows almost 10 times the ratio of H1− ions. Since it is required to compare the H penetration depth as received and laser peened (LP) samples, for this purpose step scan method is utilized. Step-scan test data show that the H-Charged laser peened plus (LP + HC) sample contained considerably less hydrogen than the unpeened sample. LP + HC sample contained same hydrogen content as the uncharged samples, whereas the HC coupon contained approximately thrice the usual level of hydrogen. Balancing diagram shown in Figure 20 can explain how LSP helps in balancing the damages caused by hydrogen.
Remedies for hydrogen embrittlement with laser peening.
Authors have contributed a part of work to literature by studying the effect of LP of titanium stabilized 321 steel and Inconel super alloy. 245 Titanium stabilized 321 steel is subjected to LP without coating technique and analysed the electrochemical properties in acidic solution. LP is performed using Nd:YAG laser delivering 1064 nm wavelength, repetition rate of 10 Hz, pulse duration of 10 ns with 0.8 mm laser spot diameter and 0.3 J of laser pulse energy. Samples are tested in 0.05 M H2SO4 + 0.01 M KSCN solution to analyse the effect of LP on electrochemical corrosion. The samples peened with higher pulse density that has smaller compressive stress showed good corrosion results than those of samples peened with lower pulse density. This contradictory result implies that there are other factors that influence the corrosion apart from compressive residual stresses. Samples laser peened with high pulse density show large charge transfer resistance compared with samples peened with lower pulse density. On the other hand, untreated sample showed very low resistance in transferring the charge when compared to both peened samples. Figure 21(a) illustrates the plot between Zre and Zim which are real and imaginary parts of complex impedance arising from resistance and capacitance of cell. The phase angle shift of untreated sample in bode plot is evident for high frequency range with poor corrosion resistance to that of laser-peened samples. Bode plot between phase angle and frequency log f is shown in Figure 21(b).
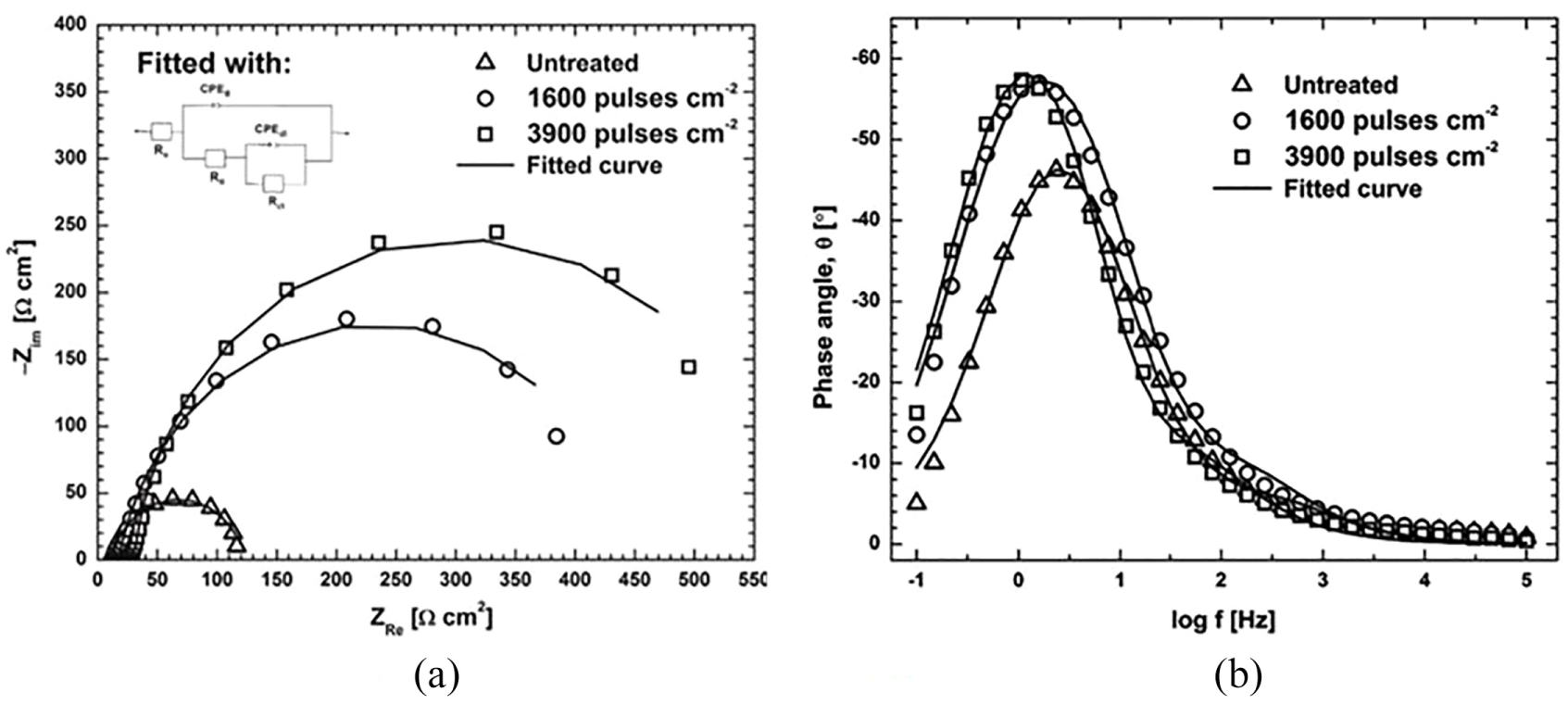
(a) Nyquist plots and (b) Bode plot. 245
Effect of LSP on the mechanical properties of near-α titanium alloy
Near-α titanium alloys are widely used in aero-engines as compressor blades and discs because of their enhanced creep properties almost at 600 °C and low weight. Jia et al. 249 investigated the effect of LSP on the high-cycle fatigue life, microhardness and residual stress of Ti834 alloy. LSP leads to high dislocation density near to surface of the metal, which is attributed to enhance microhardness of Ti834 alloy. Recurring shock waves have an extremely favourable effect on surface hardening.
The compressive residual stresses are yielded in the shocked region. After single and dual shocks, the hardness is enhanced about 364 and 366 HV, respectively, which is about 10% higher than that of the unpeened material. The extreme residual stress value grows with the multiple number of laser shocks at a point. The increase in microhardness is supposed to be caused by a grain refinement or increase in dislocation density during the LSP. The compressive stresses decrease with the increase in depth from the surface, based on the rate of attenuation of the shock wave. It is clear from the Weiju Jia investigation that the extreme compressive residual stresses are created in the surface layer of the metal, and stresses can further put forward with a multiple number of shocks at a single point as shown in Figure 22. Especially, the multiple shocking at a single point for yielding residual stress can be noticeably up to a depth of 200 μm, where the compressive stress is approximately −550 MPa for two shocks. Due to the induced compressive stresses after LSP, the high-cycle fatigue life of Ti834 alloy is increased, which can set back the fatigue crack to initiate and growth. Umapathi and Swaroop 250 investigated the effect of LP on TC6 titanium alloy with laser power densities 3, 6 and 9 GW/cm2. In X-ray diffraction (XRD) analysis, softening effect caused by LSP is observed in alpha phase due to adiabatic heating in samples peened with 6 GW/cm2 above with one and number of impacts. Increase in microhardness and surface roughness (23%, 12%–14% respectively) of peened samples directly depends on laser power density. Compressive residual stress was increased with the rise in the power density of LP. α to β phase transformation due to thermal softening was investigated with Synchrotron radiation diffraction studies which result in an increase in synchrotron radiation with raise in power density. Microstructure analysis was carried out using optical microscope correlated to SR-XRD patterns, which revealed that α to β phase transformation volume fraction steadily increased as raise in the power density.
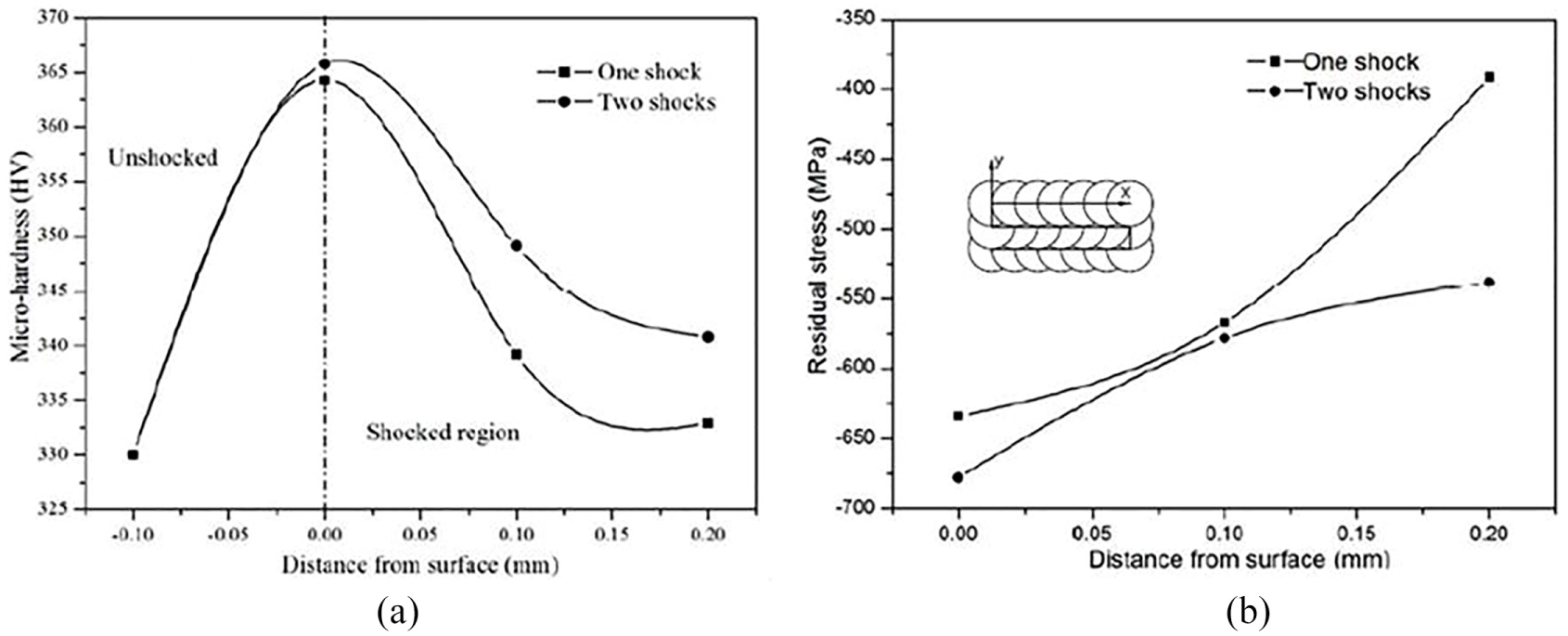
(a) Profiles of the in-depth microhardness of the specimens. (b) Profiles of the in-depth residual stress of the specimens. 249
Effect of LSP on the beta titanium alloy
TC6 titanium alloy is one of the most important titanium alloys used in aero-engines in China. TC6 titanium alloy samples were LSP processed with different LSP impacts, and the microstructure and mechanical properties were investigated by Nie et al. 251 Half of the specimens were laser shock peened processed with three impacts. Fatigue tests were carried out at the maximum stress, resonant frequency and room temperature, in the open air, to test the fatigue limits with and without LSP at 107 cycles. According to the up-and-down method of material fatigue, the fatigue test results without and with LSP are shown in Figure 23.
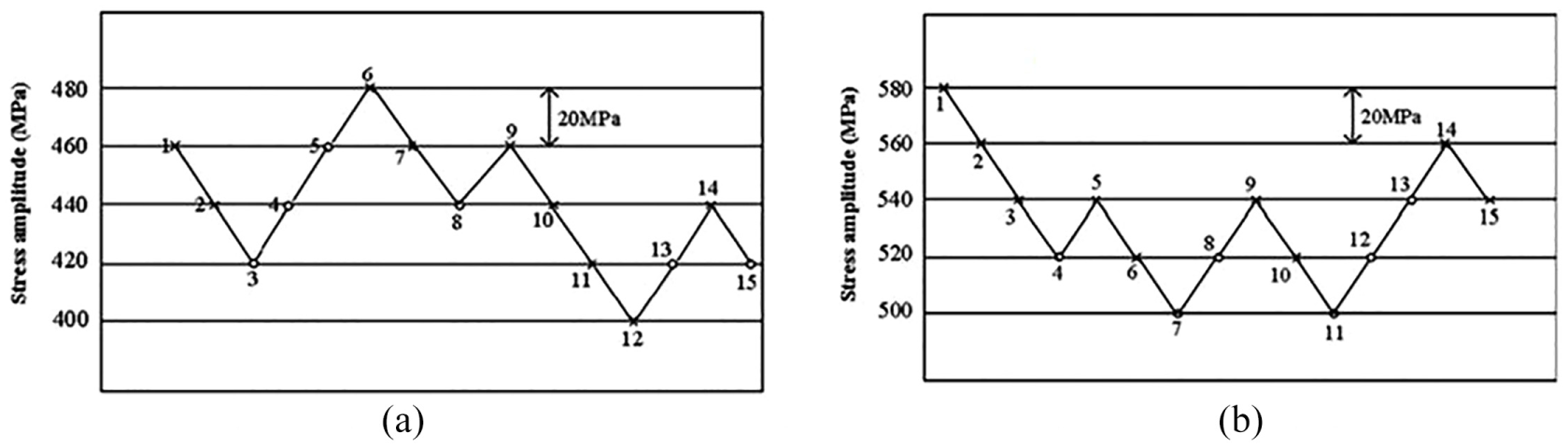
(a) Fatigue data of specimens with LSP and (b) fatigue data of specimens without LSP. 251
The amplification mechanism was pointed by analysing the effects of the changes in mechanical properties and microstructure for fatigue behaviour. Lattice micro-strains of grain refinement and dislocations lead to peak broadening. Diffraction patterns and TEM photographs specify that high strain rate deformation induced by LSP can produce nanocrystals and high-density dislocations. LSP can recover the microhardness and compressive residual stress efficiently. They increase with the LSP impact, but the rise for each impact decreases with the LSP effect. Remaining to the comprehensive action of high amplitude residual compressive stress and surface nanostructure, the fatigue limit of standard vibration specimen is improved.
Effect of LSP on alpha + beta titanium alloys
Ti–6Al–4V alloy is extensively used in the turbine blade of a compressor, where metal facing fatigue life breakdown. The LP shows effect on the three-point bending fatigue, microhardness and operation residual stress of Ti–6Al–4V alloy. 252 The work hardening by LSP process will assist in the modification of alloy surface microstructure. The microhardness of bare Ti–6Al–4V alloy can be enhanced through LSP, which is associated with high dislocation density next to the surface of the metal after LP. The compressive stresses with a comparatively uniform distribution are yielded at laser shocked region. The metal undergone three laser overlapping spots show evidence of higher fatigue life than the other incremental laser overlapping spots as shown in Figure 24. This phenomenon may be associated with many reasons. Among them the first reason may be the aluminium absorbing layer, and Al element used to shield the metal surface from thermal ablation can degrade when exposed to incremental overlapped spots. In such case, the metal may be subjected to severe twinning or slipping off the surface layer, correlated to microstructural degradation and fatigue life decline. Second, the surface quality of the sample is related to the fatigue life in the laser-peened region. The rough surface may exist at the edge of the overlapped region, which may take admission to the degradation of fatigue life.
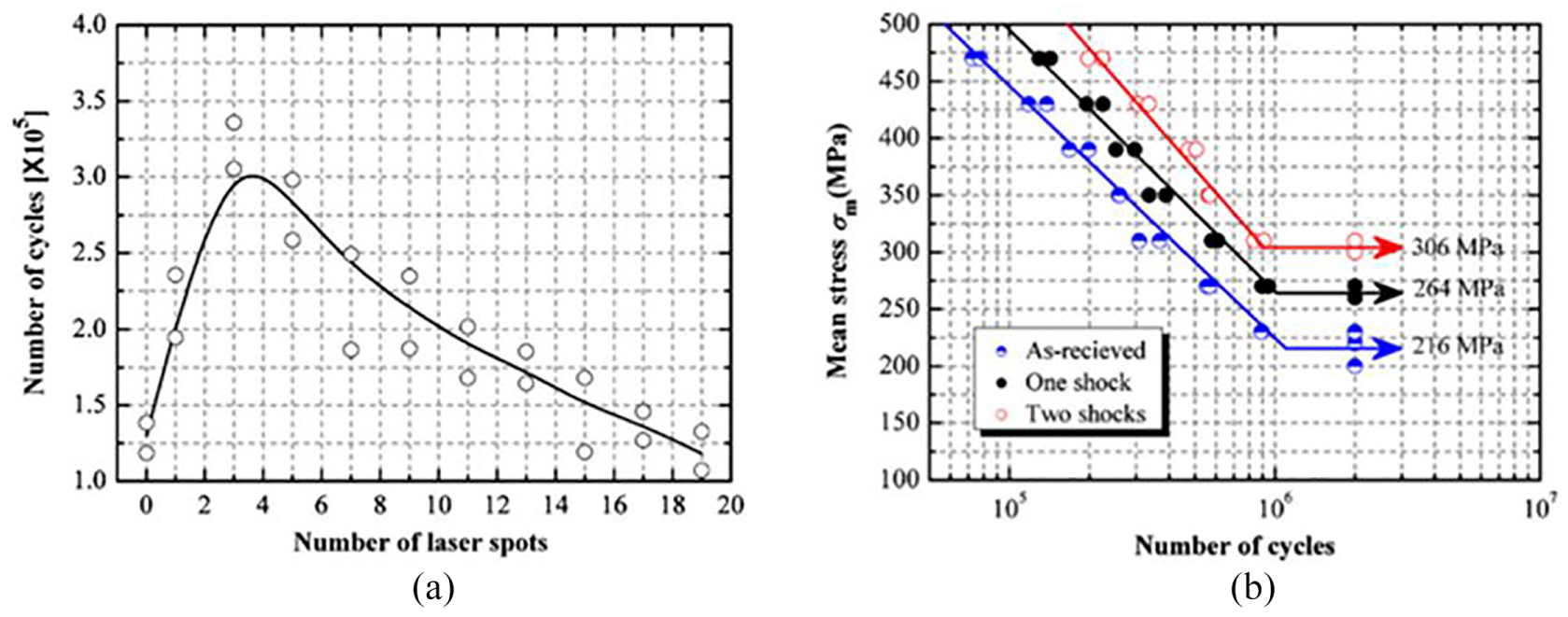
(a) Variations of the fatigue lives of the laser-peened specimens with the number of overlapped spots. (b) Fatigue lives of laser-peened and as-received Ti–6Al–4V specimens at different applied stress levels. 252
If the number of overlapped spots is more than 17, then the laser shock peened and as-received metal show equal fatigue life. With the increase in distance depthwise from the surface, the compressive stress falls exponentially and switch to tensile. The effect of LP intensity on the fatigue life of peened Ti–6Al–4V alloy is indistinct; it varies with a number of overlapped spots. When the number of overlapped spots is maintained to be three, the treatment offers a clear increase in fatigue life when compared with the un-peened metal.
Authors have performed the LP with various parameter levels on Ti6Al4V to analyse the electrochemical properties. The samples are tested in 0.05 M HCl electrolyte at room temperature. Tafel plots of unpeened and laser shock peening without coating (LSPwC) performed Ti6Al4V samples are shown in Figure 25(a) and (b). Peened samples are made as a working electrode to examine potential variation among the samples peened under various conditions. It confirms that sample LSP without coating with 532 nm has an anodic shift when compared with the potentials of the unpeened sample supporting open circuit potential (OCP) results. Potentiodynamic polarization revealed corrosion current density (Icorr) and corrosion (Ecorr) which are summarized in Table 3. And on analysing the results, samples LSPwC with 532 nm show a significant anodic shift while on the other hand the samples LSPwC with 1064 nm exhibit the equal amount of cathodic shift. A great increase and decrease in Rcorr can be observed in samples 10, 11 and 12 and 4, 5 and 6, respectively.
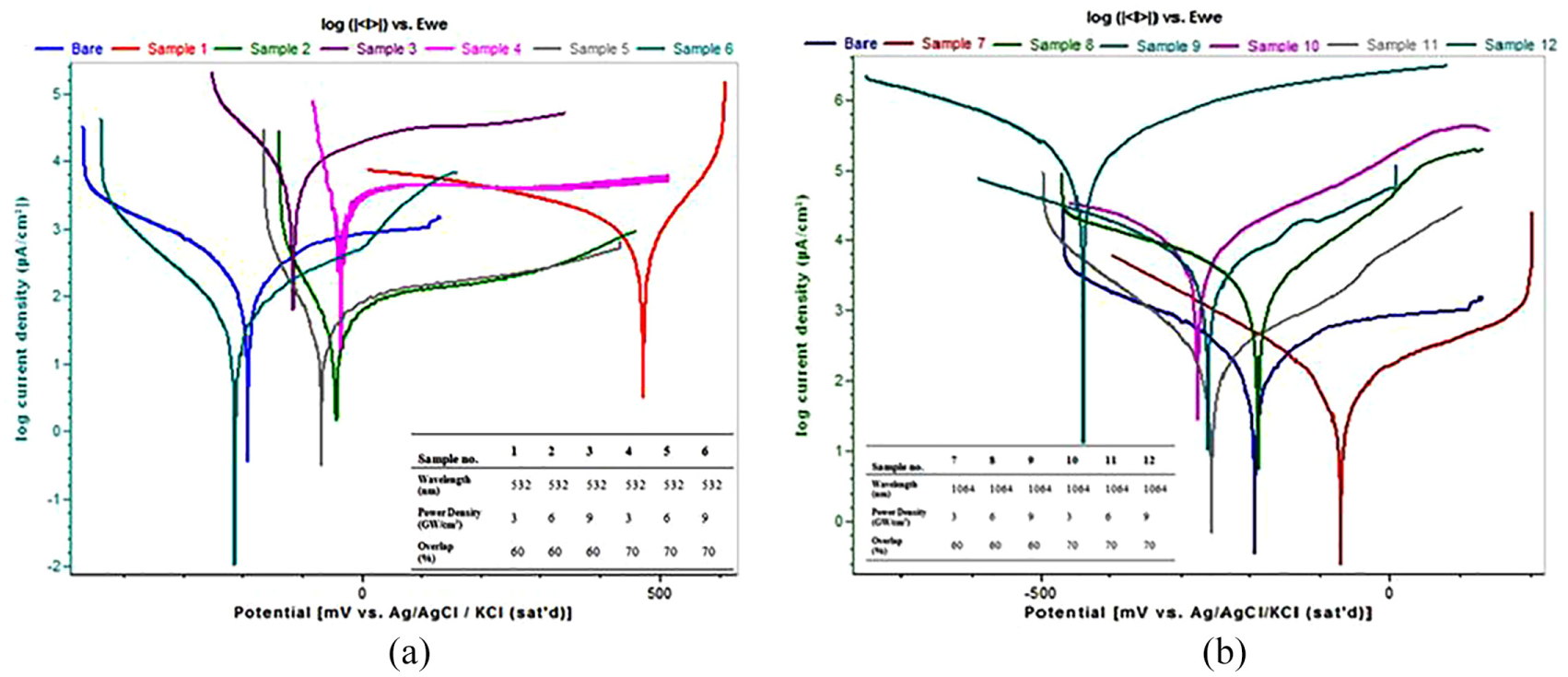
(a) and (b) Tafel plots of LSPwC samples.
Ecorr, Icorr and Rcorr values of LSPwC samples obtained from Tafel plots.
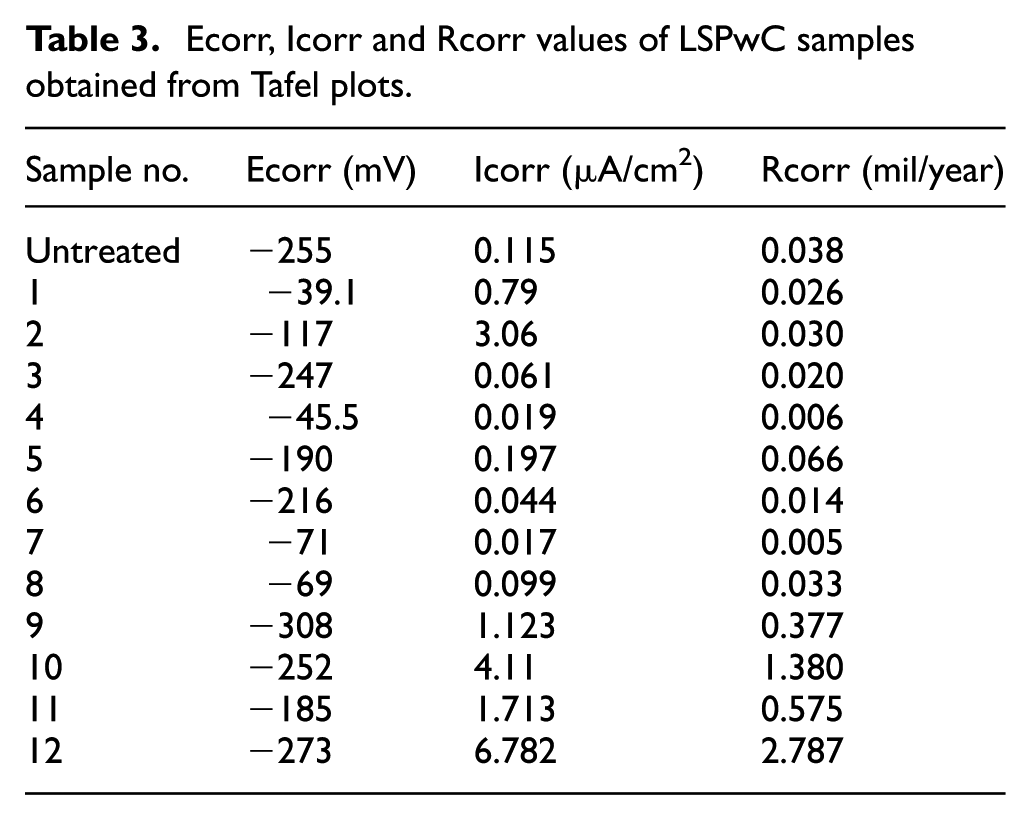
Potentiodynamic polarization result indicates the enhanced corrosion resistance only for the samples LSPwC with 532 nm wavelength at all power densities and overlap rates. The corrosion rate escalates in samples peened with 1064 nm wavelength and other considered parameters assisted in elevating corrosion rate. The corrosion is least for samples peened with 532 nm, 70% overlap, 3 GW/cm2 and 1064 nm, 60% overlap, 3 GW/cm2 laser parameter set.
Summary
In this article, the surface modification techniques are reviewed with a focus on the effect of HIC or HE on titanium alloys. Hydrogen is capable of breaking titanium oxide film with cathodic transformation at specified potentials. This transformation further leads to pits or intergranular crack propagation into the subsurface. Certain surface modification processes can help in delaying the absorption of hydrogen atoms at harsh hydrogen working environment making the surface tough and to uphold the brittle fracture. Limited articles are found in support of surface modifications that are capable of retarding HE in steel and aluminium alloys and very less towards titanium alloys. Shot peening and cavitation peening are capable of decreasing the average grain size and increasing dislocation density in various steel and aluminium alloys which helps in retarding the hydrogen atoms to settle at grain boundaries.
LSP is one of continuing advancement in production applications, technology development and scientific research. LSP exhibits significant advantages over formal shot and cavitation peening relative to the depth of compression layer, tensile and fatigue strength improvement, and process control. LSP does not involve in physical contact with the treated specimen and is not limited by geometry or surface finish. Deep compressive residual stresses of LSP process inhibit the crack propagation and hydrogen absorption even in hard materials like titanium and Inconel alloys.
Remarkable enhancements in fatigue life, HE and SCC behaviour of various materials via LSP have been thoroughly reported. The process is recognized for the induced compressive residual stresses and refined microstructure. In addition, this LP is expected to improve the corrosion and wear resistance in the biological environment of biocompatible materials. The experimental results and discussion from the work will be published in due course. Hence, it is recommended that in future, greater focus should be made on the laser developed surfaces to attain increased durability of components functioning in a corrosive environment.
Path forward
Studies associated with the interaction of titanium-hydrogen taking place in early 50s to sort out the various problems caused by hydrogen. Numerous surface modifications are practised with partial satisfactory results. After a decade of research, in early 60s laser interaction with materials is initiated and geared up to industry created by the end of 90s. Researchers all around the world reported that LSP is the best surface modification in enhancing mechanical and corrosion properties which included hydrogen-related issues. It has been carried out with some fixed parameters on widely used materials. Conversely, there are many future scopes for this process, which are recommended below:
Past a decade, LSP process is fashioned with low-energy LSP, high-energy LSP, femtosecond LSP and LSP without coating. All these new fashions are to be explored with altering parameter sets to get remarkable set of parameters for the process.
The literature reports that conventional shot and ultrasonic peening are capable of altering surface properties of steel and aluminium alloys but lagging towards titanium alloys. However, the LSP is best in all aspects compared to conventional peening, and it needs an investigation for its process best towards titanium alloys.
Scope to find out the optimum set of parameters among the vast parameters that help in establishing the best surface integrity during the LSP of titanium alloys.
LSP is found to have significant effect towards SCC and HE in steels and aluminium alloys. Since phase transformation is associated with titanium alloys, further research is to be carried out to check the significance levels of LSP towards titanium alloys SCC and HE.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
