Abstract
In order to lengthen the lifespan of NdFeB materials, nickel coating was manufactured by magnetic jet electrodeposition. The surface morphology, coating adhesion, and corrosion resistance of the coating were characterized and analyzed, respectively, by scanning electron microscope, scratch tester, and electrochemical workstation. The experimental results show that the surface of nickel coating which was fabricated by magnetic jet electrodeposition had no nodularity, and as a result, the surface morphology improved; furthermore, the corrosion resistance and coating adhesion of the coating improved in the effect of magnetic field intensity. When the value of the current density was 60 A/dm2 and the intensity of magnetic field was 100 mT, the coating adhesion could reach the standard in 33.4 N, which was doubled compared to the coating without magnetic field. Corrosion of the coating morphology is no longer the regional corrosion. Magnetic field is beneficial to improve the quality of coating surface morphology and reduce the corrosion current density of the coating.
Introduction
NdFeB material 1 with excellent magnetic properties is widely known as “magnetic king” and is adeptly used in the field of wind power generators. But as a permanent magnetic material, NdFeB is easily corrupted when it is in a low temperature and humid environment. 2 Therefore, to meet the special requirements of practical application of NdFeB in industry, surface treatment methods3,4 are often used to improve this material’s character. Electrodeposition5–7 is one of the important industrial methods to fabricate coatings. When NdFeB substrate is immersed in the electrolyte for a long time, the electrolyte can penetrate into the NdFeB pores gradually and further cause an internal corrosion. 8 Although the traditional jet electrodeposition can solve this problem, the coating uniformity is unsteady due to the high current density and tip discharge of the method. In recent years, researchers have implemented composite electrodeposition processes, such as the friction auxiliary process9,10 and the magnetic field–assisted process.11–17 The friction auxiliary process, which is controlled by mechanical force, is a contact process. It has low controllability and can inhibit the overall growth of the coating. However, the magnetic field–assisted electrodeposition process is non-contact and controllable; it can accelerate the ion mass transfer and not pollute the material. Researchers have used the magnetic field to form metal coating. Li et al. 11 studied the surface morphology of nanocrystalline Co-Ni films in the domain of high magnetic field–aided electrodeposition. Zieliński12,13 analyzed Lorentz force and the effect of magnetohydrodynamics (MHD) on the growth of the alloy structure under the magnetic field. Yu et al. 14 introduced a magnetic field, which was parallelled to the electrode during the electroplating process and determined that with the magnetic field intensity strengthening, the limiting current and deposition quality gradually increase. Udagawa et al. 15 discussed the situation, which is the metal deposition mechanism of copper sulfate aqueous solutions under magnetic fields from the perspective of Lorentz force and magnetizing force. Zhao et al. 16 found that it improved the coating adhesion between the coating and the substrate when the magnetic field and the electric field were perpendicular to each other. Miao et al. 17 studied the effects of the magnetic field plating conditions on the texture and surface morphology of nickel coatings.
The above description of the magnetic field composite electrodeposition is mostly based on the strong magnetic field (2–10 T) under the influence of the groove plating deposition. The processing current density is low (0.5–5 A/dm2),11–17 known as “high magnetic field and low current density” (HMLC). However, a strong magnetic field has some environmental pollution and needs expensive requirements. There are few studies on the preparation of coatings by electrodeposition at high current densities and low magnetic fields, known as “low magnetic field and high current density” (LMHC). In this study, a low magnetic field–assisted (0.05–0.1 T) jet electrodeposition technology can be used at high current densities (30–60 A/dm2). Nickel coating is made using NdFeB by magnetic field–assisted jet electrodeposition to solve the coating uniformity and flatness problems. The mechanism analysis of the coating fabricated by LMHC shows that the coating adhesion and corrosion resistance of the fabricated coating are obviously improved under the high current densities of the weak magnetic field, and the processing efficiency is improved.
Experimental system
Experimental device
Figure 1 shows the self-developed magnetic field–assisted jet electrodeposition system, which was equipped with a constant temperature water bath control system to ensure the accuracy and stability. The workpiece was installed on the stage connected to the cathode of the power supply. The nozzle outlet structure, designed as a narrow slit shape, was connected to the anode and installed on the anode cavity of the machine tool. The cavity is provided a nickel rod and a certain amount of nickel beads. The stage moves periodically and reciprocally when the electrolyte flowed and hits the surface of the workpiece. Filtration devices were installed in electrolyte inlet and outlet for filtering electrolyte.
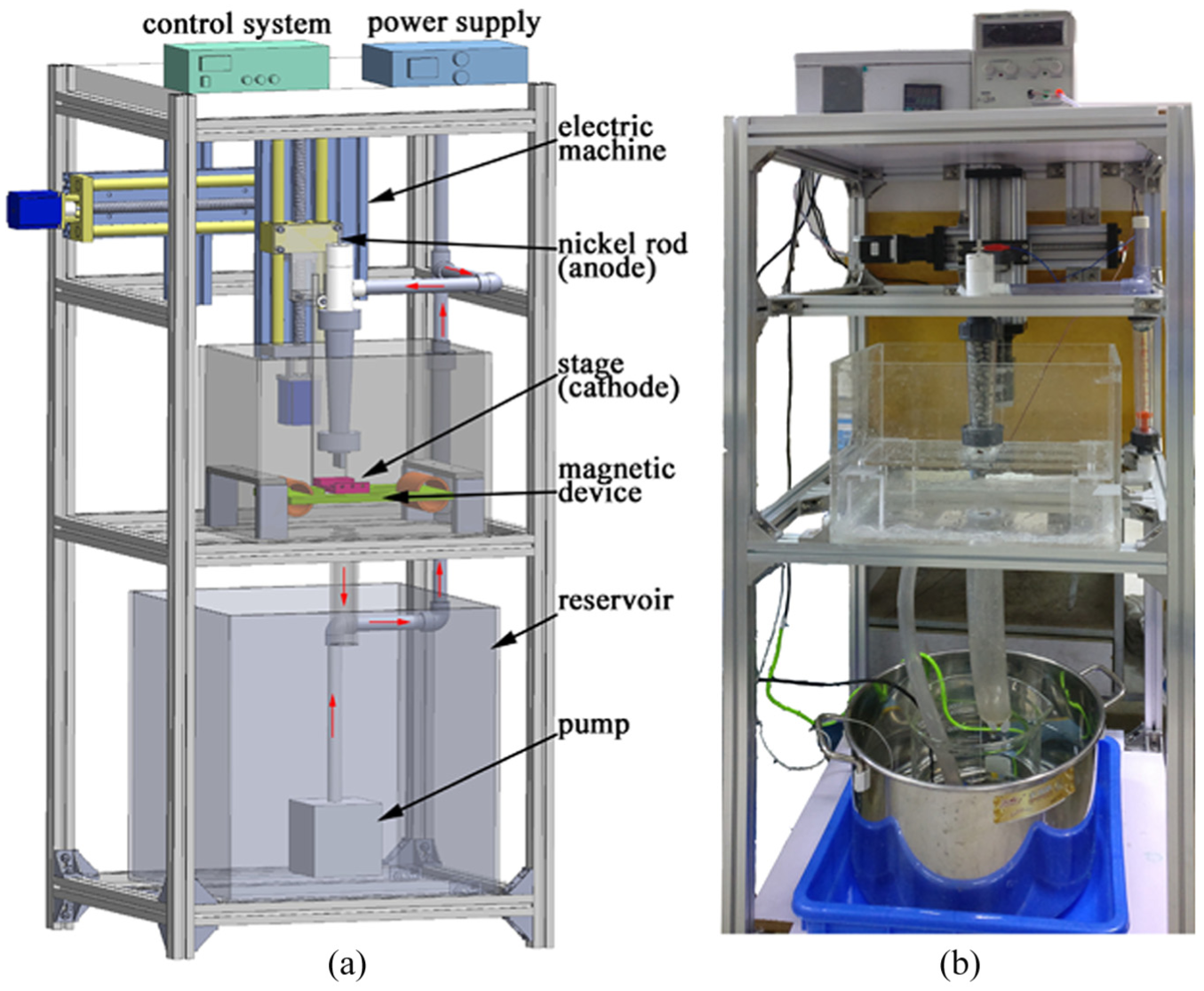
Magnetic field–assisted jet electrodeposition system: (a) model and (b) scene photograph.
Electromagnetic field device
The electromagnetic field device and its magnetic field simulation distribution are shown in Figure 2. The electromagnetic field device is mainly formed by enameled wire and silicon steel (iron core) in Figure 2(a). Figure 2(b) shows that the magnetic field intensity was basically stable in the horizontal direction of the processing region and calibrated with a Tesla meter. The electric field and magnetic field are perpendicular in the electrodeposition.
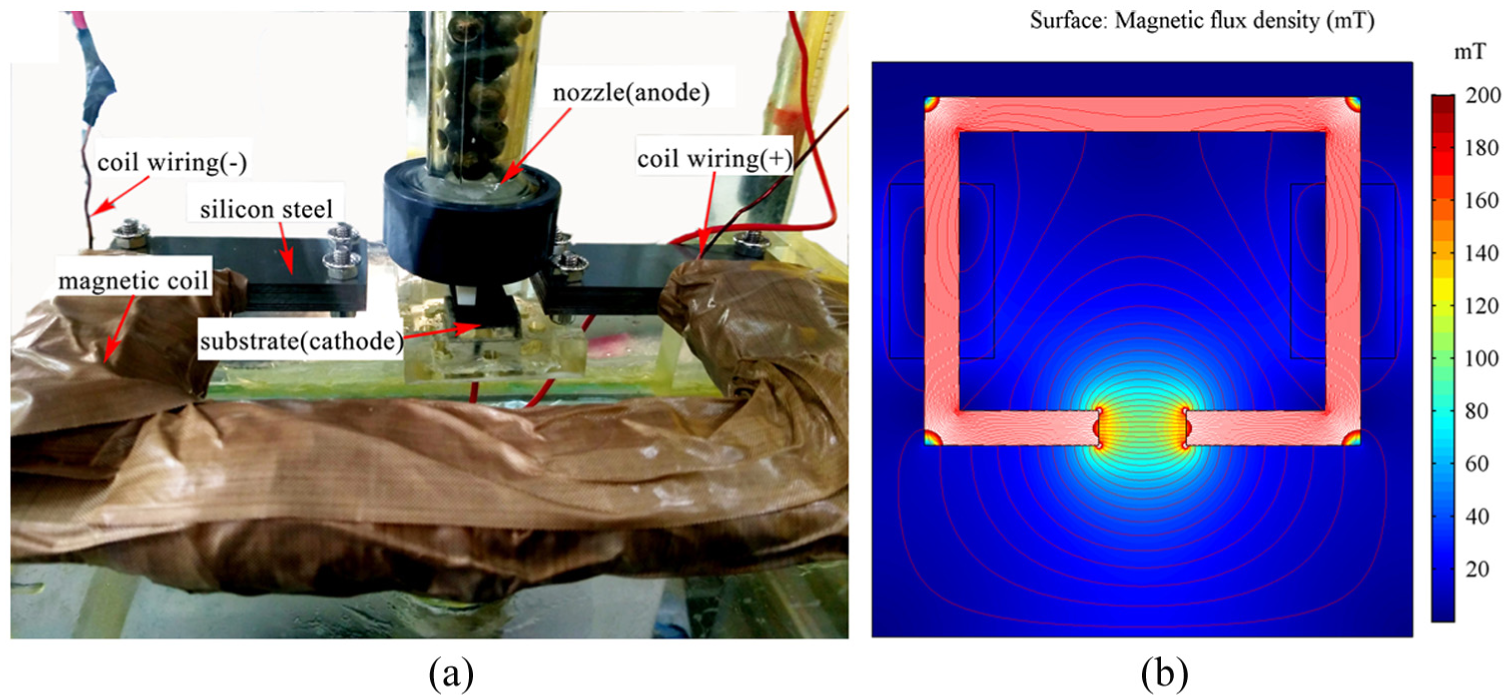
(a) Electromagnetic field device and (b) magnetic field distribution.
Principle of magnetic field–assisted electrodeposition
Figure 3 is a schematic diagram of ions adsorption by jet electrodeposition with magnetic field. Metal ions (Ni2+) were mainly affected by the electric field force adsorption without magnetic field in Figure 3(a). Metal ions were adsorbed on the surface of the cathode to grow metal layer; in the meantime, hydrogen ions adsorbed on the surface to generate bubbles, which would form air holes in the coating, unless they discharged smoothly. However, when the magnetic field was assisted, metal ions (Ni2+) are also affected by the electric field force and Lorentz force. 18 Figure 3(b) shows that metal ions (Ni2 +) changed from the original direction and started rotating and then micro-stirring the electrolyte. At the same time, the high-speed flow of electrolyte enhanced the deflection, kept the metal ions concentration of the reaction interface stable, and improved the covering ability of the cathode surface. The effect of magnetic field on electrodeposition, that was MHD accelerated the flowability of the reaction interface, increased the electrochemical deposition rate, and assisted the bubbles escape. In addition, hydrogen is a diamagnetic element and oxygen is a paramagnetic element. The magnetic field causes the hydrogen–oxygen bond (O-H) to elongate, which decreased the surface tension. 19 The coating grown uniformity as the state of ions hydration were improved. 20 Therefore, the magnetic field directly affected the exchange and transfer adsorption of ions in the electrolyte, so as to improve the quality of the coating.
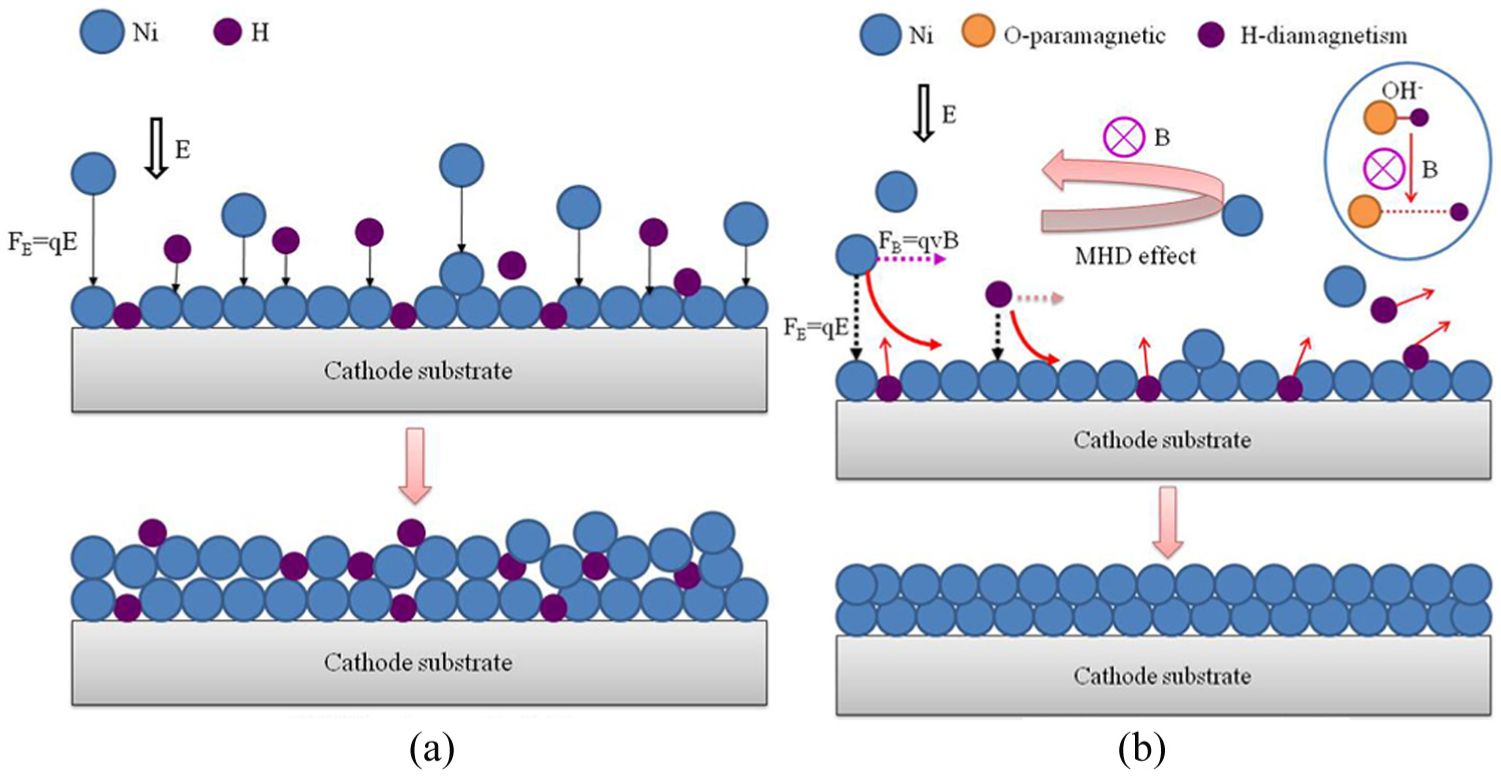
Schematic diagram of jet electrodeposition process: (a) without magnetic field and (b) with uniform magnetic field.
Experimental process
Electrolyte configuration
The electrolyte was composited with NiSO4.6H2O (280 g/L), NiCl2.6H2O (40 g/L), H3BO4 (40 g/L), and C7H5O3NS (5 g/L). The electrolyte was configured with deionized water. The pH was controlled at 4 ± 0.1, and the temperature of the electrolyte was set at 50°C. The processing time of each group of samples was 40 min, and the flow rate was 200 L/h.
Test material
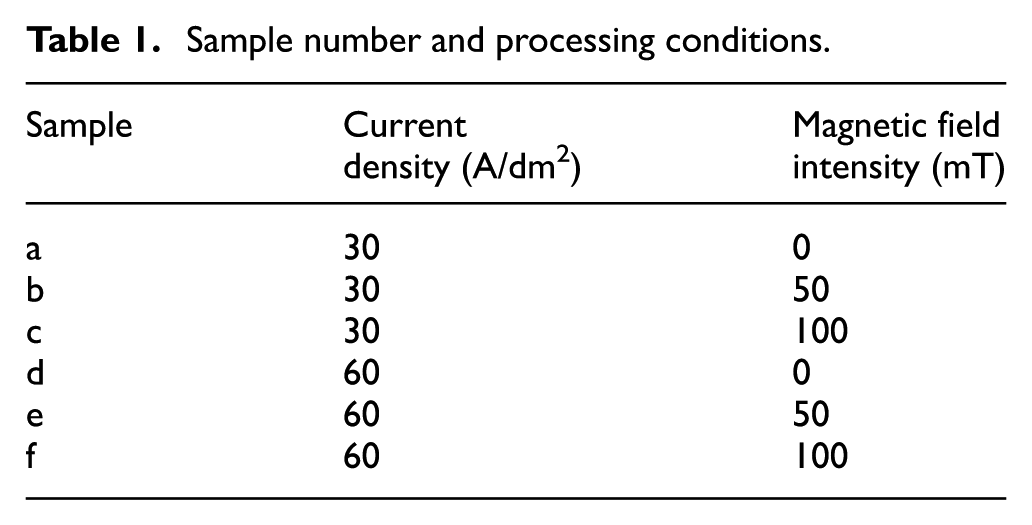
NdFeB material (N35) of a size of 20 × 30 × 3 mm was selected as the cathode material. The sample number and corresponding processing conditions are shown in Table 1. First, the sample was ground and polished to remove surface scratches by precision plane polishing machines (ZDHP-30). Second, the sample was sealed with the zinc stearate. Finally, after polishing and cleaning, the sample was dried for the experiment.
Sample number and processing conditions.
Coating performance characterization
The surface morphology of the nickel coating was analyzed using the scanning electron microscope (S-4800) by Hitachi Ltd. The coating adhesion was tested using the coating adhesion automatic scratch tester (WS-2005) made by Lanzhou Zhongke Kaihua Technology Development Co. The corrosion resistance was tested by an electrochemical workstation test (CHI660E) by Shanghai Chenhua Instrument Co Ltd.
Results and discussion
Surface morphologies
The surface morphology of the coating is shown in Figure 4. In the absence of a magnetic field, the coating surface has many protuberances, pit defects, and even with black spots in Figure 4(a). Edge effect is easy to occur in jet electrodeposition,21–23 when the surface of the coating grown with micro-protrusions, it would continue to grow and form larger bulges. So the coating flatness and uniformity was poor. Figure 4(b) shows no black spot on the coating surface, and the corresponding protuberances and pits are reduced obviously with the aid of magnetic field (50 mT). As mentioned above, the MHD brought by a magnetic field caused micro-stirring, the concentration polarization of the boundary layer was reduced, the thickness of the diffusion layer was reduced, and more metal ions (Ni2+) were homogeneously adsorbed by the surface of the cathode, which slowed down the original edge effect, prevented cell growth from continuing to grow, and reduced the protuberances. As the intensity of the magnetic field increases to 100 mT, the protuberances and pits are reduced further, and the surface smoothness is improved in Figure 4(c). Correspondingly, the coating surface is shown in Figure 4(d) when the current density is increased to 60 A/dm2. It is not difficult to find that higher current density can improve the surface quality of the coating better under the same magnetic field intensity.

SEM images of the coatings under different parameters: (a) 30A/dm2,0T, (b) 30A/dm2,50mT, (c) 30A/dm2,100mT, (d) 60A/dm2,0T, (e) 60A/dm2, 50mT and (f) 60A/dm2,100mT.
The magnetic field disturbance was relatively strong when it was at 100 mT. It was difficult to ensure real-time replenishment of metal ions adsorbed and deposited on the surface of the cathode at low current density (30 A/dm2). Therefore, the quality of the growing metal coating is easily optimized. The current density was an important parameter affecting the electrochemical deposition reaction, 24 it provided theoretical basis for the timely deposition of metal cations in the electrolyte when it was in a high current density (60 A/dm2). Of course, it provided a better reaction foundation for the sufficient reaction of metal ions in the electrolyte, due to the high-speed flow of the electrolyte, and confirmed that the high-speed jet process was more suitable for electrodeposition at high current density. 21
Figure 5 shows the nickel coating growth mechanism model of the jet electrodeposited under different modes. There is mainly crystal nucleation and growth in electrodeposition. 9 In the absence of a magnetic field, the metal ions were mainly affected by the electric field force. If micro-protrusions or pits were generated at a certain point during the deposition process, the discharge of micro-protrusions were more concentrated and grown faster, while the pits grown slower. As shown in Figure 5(a), the uneven distribution of electric field lines would affect the homogeneity of coating growth directly. Then, these two extremes would be gradually enlarged, resulting in the coating had less dense coating, lots of protuberances and pits. After the magnetic field was applied, metal ions were affected by the electric field force and Lorentz force. It reduced the shielding effect and improved the covering capacity of the electrolyte, and the electric field distribution in the growth interface tends to be uniform, as shown in Figure 5(b), which was in favor of uniform nucleation. On one hand, the accelerated growth of atoms adsorbed on the protruding points was limited. The pits could grow quickly to compensate for defects. On the other hand, the hydrogen bubbles generated in the interfacial reaction can also be discharged with the aid of magnetic field micro-disturbance. Therefore, the coating has good high density and flatness.
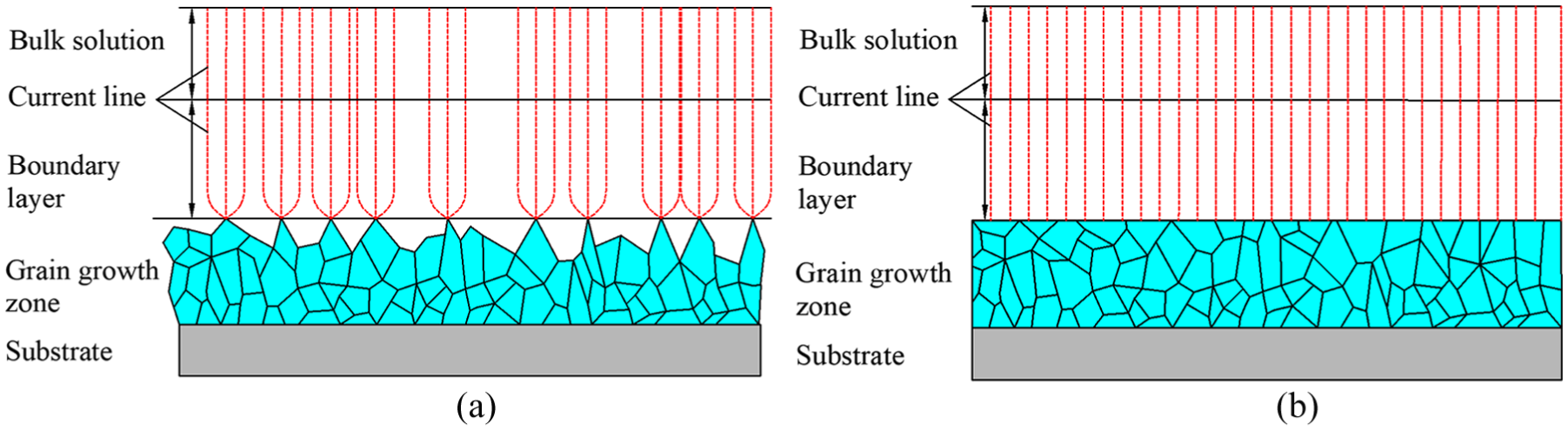
Growth mechanism model of jet electrodeposition: (a) without magnetic field and (b) with uniform magnetic field.
Coating adhesion
The WS-2005 scratch tester was used to test the coating adhesion between the nickel coating and the NdFeB substrate. The test parameters were the same (dynamic load was 40 N and scratch length was 3 mm). The test method was friction test. The measuring principle of friction testing technology was as follows: when the coating broke down, the friction force would change suddenly. The load applied at this point was the critical load. The greater the critical load, the stronger the coating adhesion strength.25,26 The coating adhesion is shown in Figure 6. The coating adhesion of the nickel by magnetic jet electrodeposition was obviously superior to that without magnetic field. As the magnetic field intensity increased, the coating adhesion was further enhanced. The coating adhesion reached 33.4 N when the magnetic field intensity was 100 mT, compared with the coating adhesion of 16.68 N without magnetic fields at the current density of 60 A/dm2.
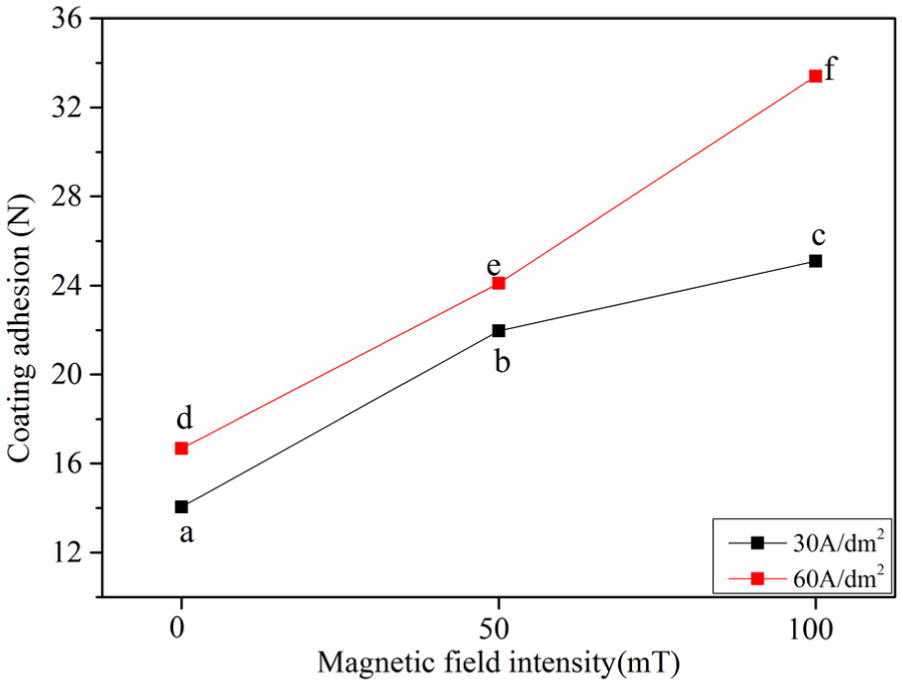
Coating adhesion of different samples.
Figure 7 shows the scratch morphology of the coating at different magnetic field intensities. Figure 7(a) appears peeled off in the area near the middle of the scratch and has obvious burst area without magnetic field. Figure 7(b) has a few micro-cracks at the end of the scratch when the magnetic field is 50 mT, so the coating adhesion is slightly improved. 27 It is more obvious that the sample surface has no micro-cracks and is relatively good in Figure 7(c), when the magnetic field is raised to 100 mT.

Surface morphology after coating adhesion test: (a) 60A/dm2,0T, (b) 60A/dm2,50mT and (c) 60A/dm2,100mT.
Figure 8 is a cross-section morphology of nickel coating on NdFeB. The nickel coating has an interface with the NdFeB substrate, even peeling off, as shown in Figure 8(a). In Figure 8(c), the nickel coating is most closely related to the bonding interface between coating and NdFeB substrate. Thus, the magnetic jet electrodeposition can improve the coating adhesion. It was difficult to overcome the internal stress of the coating in jet electrodeposition, the coating adhesion was low. 8 Nickel metal is a ferromagnetic material, and nickel ions are subjected to a magnetizing force in magnetic field. In order to reduce the free energy, there is a tendency of spontaneous orientation in the direction of the magnetic field.28–30 In addition, Lorentz force interacts with the micro-current generated by the flow of the metal ions and the reduction point in the electrochemical deposition reaction. Magnetohydrodynamics (MHD) causes a change in the flow of electrolyte ions under the action of a magnetic field. It was more prominent in jet electrodeposition because it had the characteristics of high current density and high velocity flow of the electrolyte, and improved the microstructure of the nickel coating then increased the coating adhesion of the nickel coating and NdFeB substrate.

Cross-section morphology of nickel coating on NdFeB: (a) 60A/dm2,0T, (b) 60A/dm2,50mT and (c) 60A/dm2,100mT.
Corrosion behavior
Figure 9 shows the polarization curve of the coating in a 3.5 wt% NaCl solution. The corrosion potential (Ecorr) and corrosion current density (icorr) were obtained by Tafel curve extrapolation,31,32 as shown in Table 2. The nickel coating corrosion potential was −0.656 V and corrosion current density was 10.81 μA cm−2 by jet electrodeposition (60 A/dm2, 0 T). Nevertheless, the corrosion potential increased to −0.554 V, and the corrosion current density decreased to 5.073 μA cm−2 of coating with magnetic jet electrodeposition.
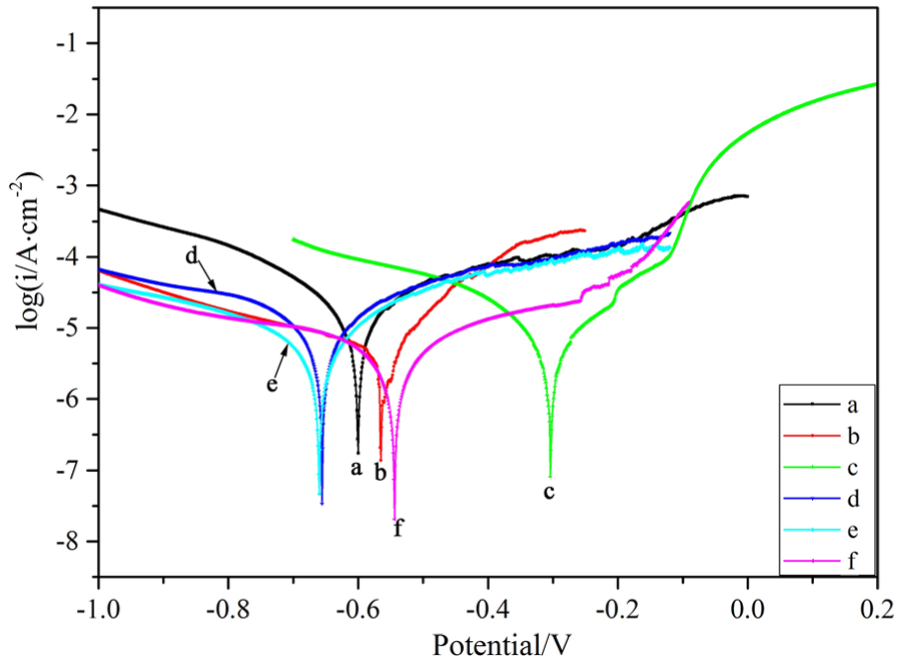
Potentiodynamic polarization curves of the nickel coating.
Corrosion potential and corrosion current density of coating.
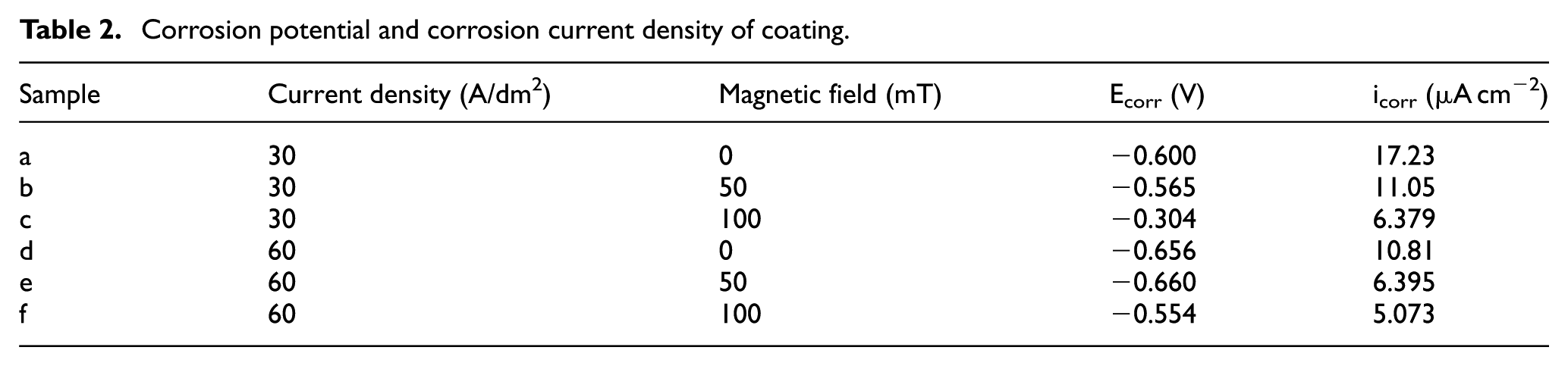
Figure 10 shows the morphology of electrochemical corrosion. Figure 10(a) is obvious regional corrosion and has lot of corrosion pits. In Figure 10(b), the corrosion pits decrease, when the magnetic field intensity was 50 mT. Furthermore, there are a few corrosive pits in Figure 10(c).

Surface morphology after coating corrosion: (a) 60A/dm2,0T, (b) 60A/dm2,50mT and (c) 60A/dm2,100mT
Obviously, the corrosion of the material began on the material surface.8,9 It was easy for the electrolyte to destroy the surface of the coating and deeply penetrate the interior when the coating surface had protuberances and pits. Magnetic jet electrodeposition, improved flatness and uniformity of the coating, and there was no longer a large area of protuberances and pits. The coating was not corroded as the electrolyte was difficult to penetrate into the coating. Thus, the corrosion resistance of nickel coating is improved and NdFeB material is protected.
Conclusion
In this study, magnetic jet electrodeposition was discussed, the LMHC processing was realized, the uniformity of the coating by traditional jet electrodeposition was solved, and the coating prepared on the NdFeB substrate was analyzed. The lifespan of NdFeB material showed obvious improvement and research significance. From this work, the following conclusions are drawn:
The magnetic field jet electrodeposition can improve the coating surface morphology, suppresses the edge effect of jet electrodeposition and improve the coating flatness and uniformity.
When the current density was 60 A/dm2 and the magnetic field intensity was 100 mT, the coating adhesion between the nickel coating and the NdFeB substrate reached 33.4 N, almost double that under the same condition without magnetic field.
The corrosion resistance of nickel coating was better by magnetic jet electrodeposition, no longer showed regional corrosion on the surface. The corrosion potential increased to −0.554 V, and the corrosion current density decreased to 5.073 μA cm−2.
Footnotes
Acknowledgements
W.J. and L.S. have equally contributed to this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation of China (Grant No.: 51475235, 51105204, and 51575271). We also extend our sincere thanks to all who contributed in the preparation of these instructions.
