Abstract
Jet electrodeposition has the characteristics of rapid deposition with high current density, but faces the problem of tip discharge, which causes a decrease in coating surface uniformity and flatness. In this paper, scanning jet electrodeposition was adopted to optimize the tip discharge. The different nanoparticles were mixed as the reinforcing phase of Ni-Co alloy coating. Nano-CeO2/Nip mixed particles enhanced Nickel-Cobalt composite coatings on 304 stainless steel by scanning jet electrodeposition. The morphologies, texture orientation, microhardness, coating adhesion, and corrosion resistance of Ni-Co-CeO2/Ni composite coatings were characterized. The addition of nano-mixed particles (CeO2/Nip) changed the morphology of Ni-Co-CeO2/Nip composite coating from large cellular protrusion structures to fine granular structures and improved the uniformity of the composite coating. Compared with Ni-Co alloy coating, the microhardness increased from 467.8 to 524.7 HV, the adhesion between the substrate and the coating increased from 20.6 to 28.9 N, and the corrosion current density decreased from 2.413 × 10−5 to 5.598 × 10−6 A·cm−2 in Ni-Co-CeO2/Nip (4 g/L) composite coating, indicating that nano-mixed particles (CeO2/Nip) improved the mechanical properties and corrosion resistance of Ni-Co alloy coating in scanning jet electrodeposition.
Introduction
Metal materials are prone to fatigue failure and corrosion shedding in daily wear, oxidation and corrosion, which directly affect the service life of metal materials. 1 Nickel-Cobalt (Ni-Co) composite coatings have been widely used in the surface protection of engineering materials in recent years with their excellent mechanical, physical and chemical properties.1–4 Compared with the methods of magnetron sputtering, 5 plasma spraying, 6 and laser melting 7 to fabricate Ni-Co alloy coatings, electrodeposition has the advantages of mature technology, wide application range, moderate cost, precise control of coating thickness and structure.8–13 There are some reports on nanoparticle-reinforced Ni-Co composite coatings doped with nano-SiO2,14,15 SiC,16,17 ZrO2, 18 CeO2, 19 but the studies above are all immersion electrodeposition where samples were completely immersed in the electrolyte.
Different from the studies above, in jet electrodeposition,20,21 the electrolyte was circulated at a high speed, and the workpiece was not immersed in the electrolyte, which was suitable for high current density deposition. The electrolyte was sprayed on the surface of the sample through the anode nozzle, which had the characteristics of localization and selective deposition. Therefore, it could be used for surface repair and remanufacturing of engineering materials, which has been widely reported in recent years.22,23
Functional metal nanoparticles have unique thermal, electrical, and flame-retardant properties. 24 Recently, Ni-Co-CeO2 composite coatings mainly focused on high-temperature oxidation resistance, microhardness and corrosion resistance.19,25,26 The Ni-Co-CeO2 composite coating has been applied to Crofer 22 APU alloy via the electroless meth and then it has good adhesion due to no gap between the coating and the substrate by observing the cross-sectional morphology. 25 Isothermal oxidation and cyclic oxidation has been studied respectively and the results showed that the coating formed on the steel surface could resist cracking and delamination and had stronger oxidation resistance. 26 In addition, Nano CeO2 was added to the nickel-cobalt alloy to study the effect of cobalt addition in the alloy on the structure and properties. It is found that the microhardness of Ni-Co-CeO2 composite coating is the highest, as high as 540 HK 50 gf, about 522 HV (Empirical formula: 1 HV = 0.968 HK) when the cobalt content is 25%. 19 However, it does not been reported that metal nanoparticles were mixed with nano CeO2 particles to prepare nano-mixed particles to deposit composite coatings. In this work, uniform nano-mixed particles (CeO2/Nip) were obtained by magnetic stirring and added to the electrolyte. Ni-Co-CeO2/Nip composite coatings were rapidly deposited by scanning jet electrodeposition. The differences between Ni-Co composite coating, single nanoparticle composite coating and nano-mixed particles composite coating at different concentrations were compared. The influence of nano-mixed particles on the surface morphologies, texture orientation, microhardness, adhesion, and corrosion resistance of Ni-Co-CeO2/Nip composite coating was researched.
Experimental of Nickel-Cobalt composite coatings
Electrodeposition system
Figure 1 indicates a schematic diagram of the scanning jet electrodeposition system. There is a power supply, control system, temperature control system, and anode jet system. There is a high-purity metal nickel rod and some metal nickel beads in the anode jet system, which can effectively improve the conductivity of the electrolyte. Before scanning jet electrodeposition, the substrate was fixed on the stage of the electrolytic cell, and the machining gap between the anode nozzle outlet and the cathode substrate was adjusted by the control system. Then, the electrolyte was pumped out of the electrolytic cell, flowed through the pipe and was sprayed and deposited on the cathode surface under the action of the external electric field. During electrodeposition, the anode nozzle moved back and forth along the horizontal direction to ensure that the electrolyte fully covered the deposition area.
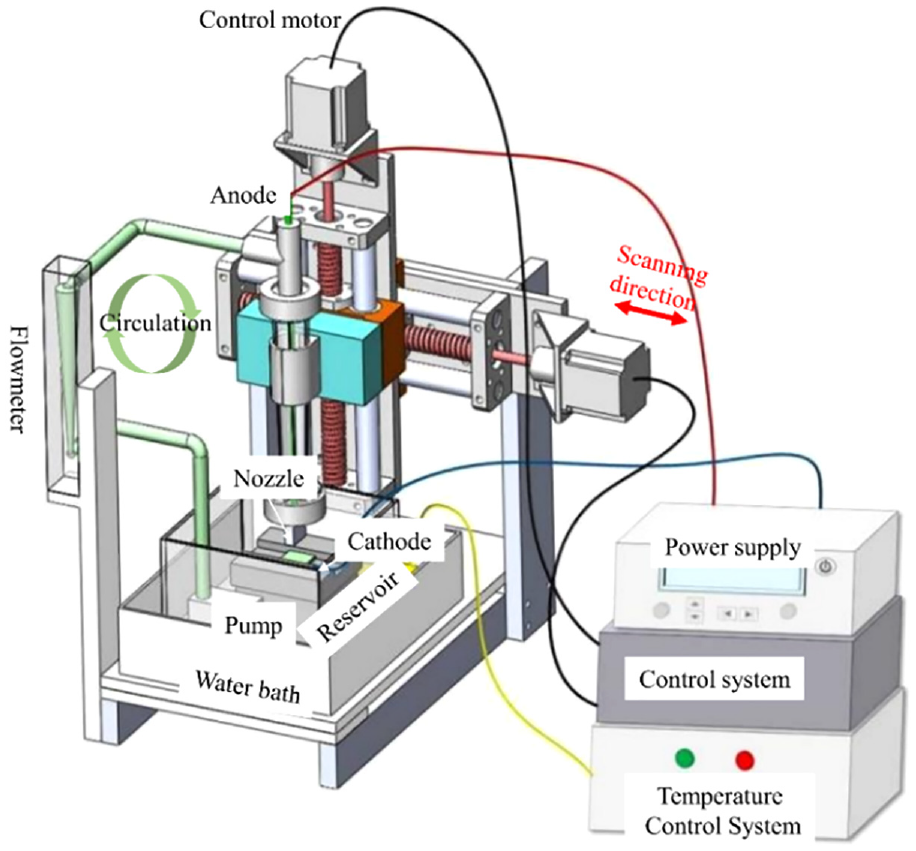
Schematic diagram of the experimental system.
Experimental parameters
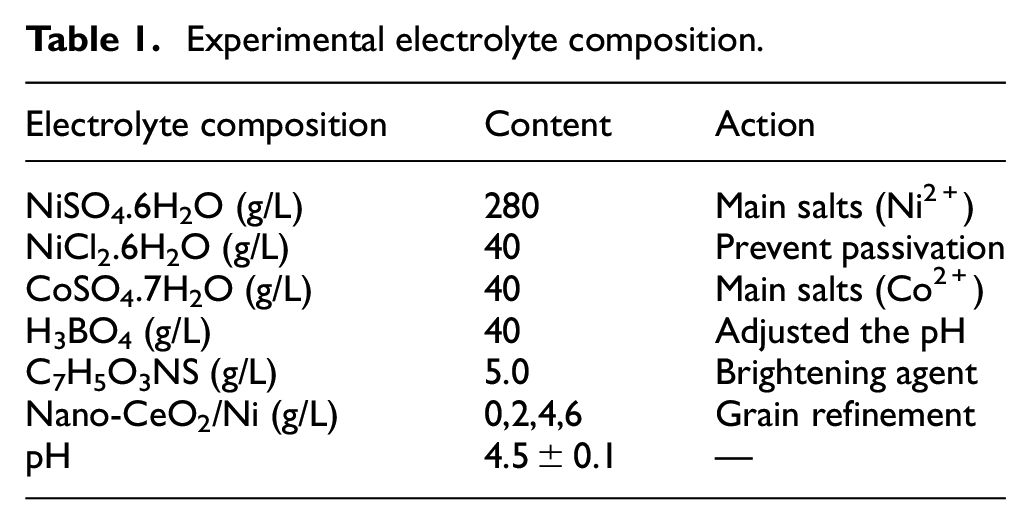
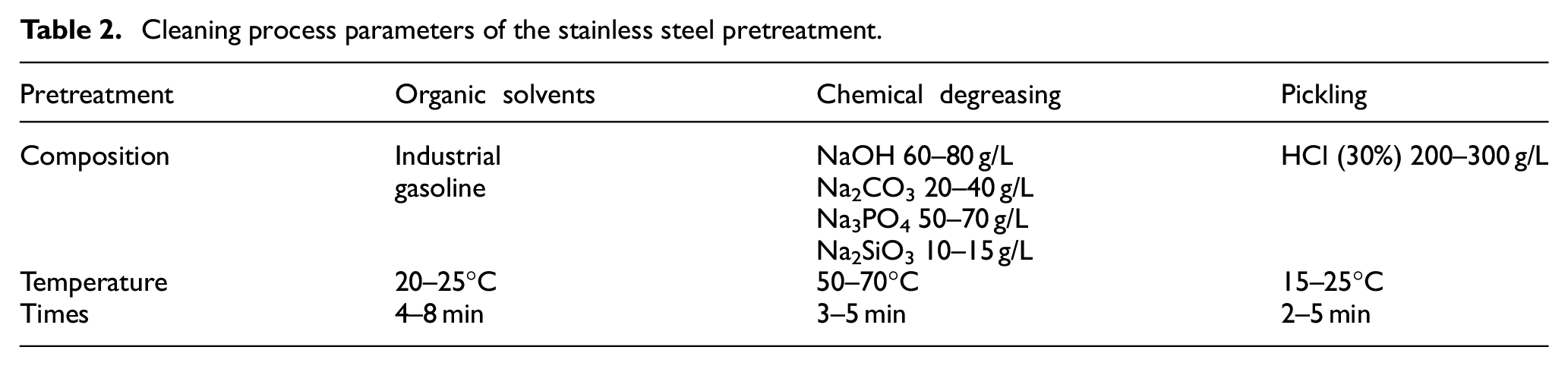
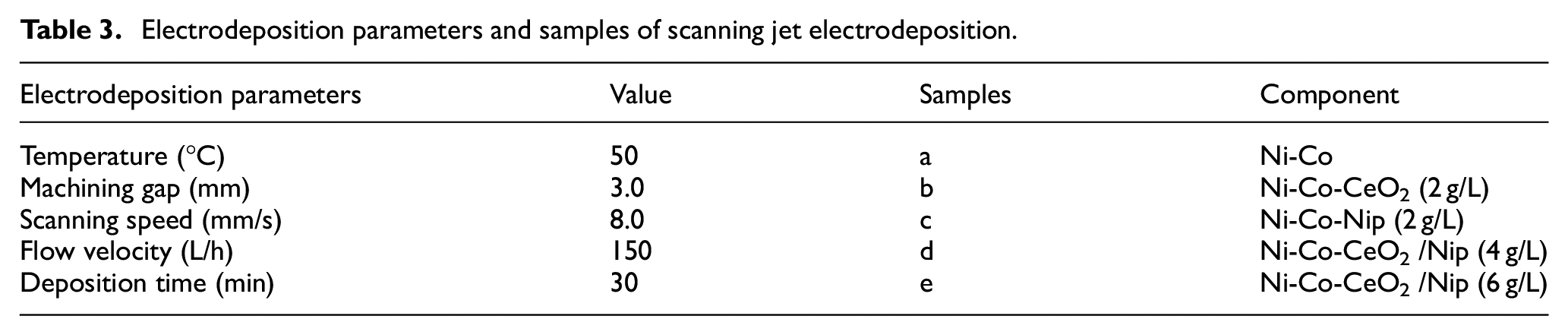
The electrolyte was prepared with an analytical reagent and distilled water, and the experimental electrolyte composition was shown in Table 1. Before electrodeposition, nanoparticles were mixed with CeO2 (20 nm) and Ni (50 nm) by magnetic stirring for 2 h. The substrate material was 304 stainless steel with a size of 20 × 20 × 2 mm 3 . Research showed that surface pretreatment had an impact on the electrodeposition of material surface.27,28 In this work, we adopted the same surface pretreatment for stainless steel samples to ensure the consistency of the impact of stainless steel pretreatment. To study the influence of nano-mixed particles on scanning jet electrodeposition, all samples should be polished with sandpaper, and then the surface oxide layer was removed by pretreatment, as shown in Table 2. Table 3 shows the electrodeposition parameters and samples of scanning jet electrodeposition. All composite coatings were prepared on the surface of 304 stainless steel by scanning jet electrodeposition, as shown in Figure 2. Scanning jet electrodeposition reduced concentration polarization, increased the current density of electrodeposition, and improved the uniformity without defects such as micro-pores and micro-cracks.
Experimental electrolyte composition.
Cleaning process parameters of the stainless steel pretreatment.
Electrodeposition parameters and samples of scanning jet electrodeposition.
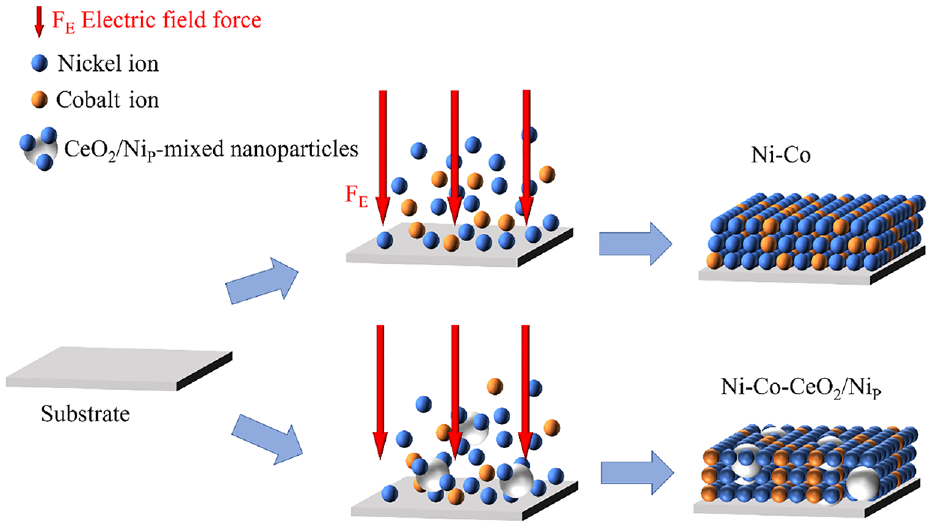
Schematic diagram of Ni-Co-CeO2/Nip composite coating by scanning jet electrodeposition.
Characterization
The surface morphologies of the Ni-Co alloy composite coating were observed by the field emission scanning electron microscope (Regulus8230). The element distribution was determined by energy dispersive spectroscopy (Oxford INCA). The surface profiles of the composite electrode were observed by a 3D laser measurement microscope (VK-X250). The texture orientation of the composite coating was tested by the X-ray diffractometer (DMAX-2500PC) with 40 kV and 150 mA by Cu-Kα radiation (λ = 1.5406 Å). Measuring five positions on each sample with a microhardness tester (HVS-1000A) as the average microhardness with a load of 0.25 kg and a dwell time of 15 s. Coating adhesion was evaluated using a scratch tester (WS-2005). Taking five scratches at different positions on each sample to get the average value, setting a load on 40 N with the scratch length of 3 mm. The corrosion resistance was evaluated in a 3.5 wt% NaCl corrosive medium using an electrochemical workstation (CHI760E). The counter electrode was a platinum electrode and the reference electrode was a saturated KCl electrode.
Results and discussion
Morphologies
The surface morphologies of the composite coatings changed with different nanoparticles, as shown in Figure 3. The surface is relatively flat, and there is cellular protrusion on the surface of Ni-Co alloy coating in Figure 3(a). Figure 3(b) shows that there were few protrusions on the surface of Ni-Co-CeO2 (2 g/L) composite coating, so the flatness is further improved, which is related to the grain refinement in the composite coating with the addition of nano-CeO2 particles. Figure 3(c) indicates the morphologies of Ni-Co-Nip (2 g/L) composite coatings, there is a larger cellular protrusion, which may be caused by the addition of nano nickel particles (Nip) to the electrolyte. Compared with nano CeO2 particles, Nickel particles are more likely to serve as the growth points of composite electrodeposition, thus forming a larger cellular protrusion. Figure 3(d) shows the Ni-Co-CeO2/Nip (4 g/L) composite coating had a better flat surface when the nano-mixed particles are added at 4 g/L. The nano-mixed particles provide more growth points for cation deposition and refined the grain growth in the Ni-Co-CeO2/Nip (4 g/L) composite coating. However, in Figure 3(e), there are some micro-pores and cellular protrusions on the surface morphology of the Ni-Co-CeO2/Nip (6 g/L) composite coating with the nano-mixed particle concentration added to 6 g/L, it is prone to tip discharge at a high current density of scanning jet electrodeposition, leading to the rapid growth of metal cations (Ni2+, Co2+) in the electrolyte with the agglomerated nanoparticles as growth points, then the nearby area was shielded, resulting in a decrease in the flatness and uniformity of the composite coating, especially at a high concentration of nano-mixed particles.

Morphologies of the composite coatings: (a) Ni-Co, (b) Ni-Co-CeO2 (2g/L), (c) Ni-Co-Nip (2g/L), (d) Ni-Co-CeO2/Nip (4g/L), (e) Ni-Co-CeO2/Nip (6g/L).
Figure 4 shows the 3D profiles of the composite coatings. The surface roughness of Ni-Co-CeO2 composite coating was the lowest at 0.451 μm, while the surface roughness of Ni-Co-Nip (2 g/L) composite coating increased to 0.869 μm when the nano nickel particles were added at 2 g/L. The surface roughness decreased to 0.595 μm in Ni-Co-CeO2/Nip (4 g/L) composite coating. However, the surface roughness of Ni-Co-CeO2/Nip (6 g/L) composite coating increased to 0.617 μm when the concentration of the nano-mixed particles reached 6 g/L, which indirectly confirmed the morphologies of the composite coating in Figure 3.
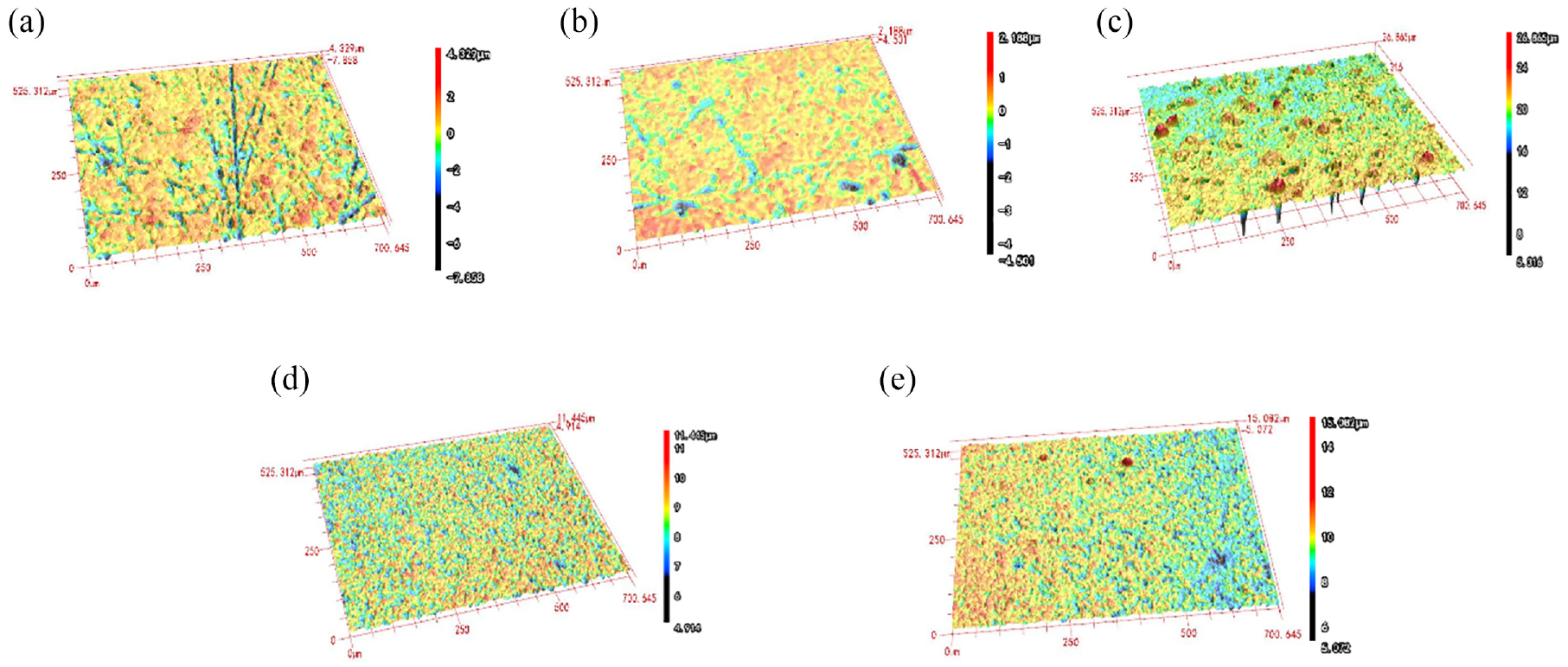
3D profiles of the composite coatings: (a) Ra 0.853 μm, (b) Ra 0.451 μm, (c) Ra 0.869 μm, (d) Ra 0.595 μm, and(e) Ra 0.617 μm.
To better observe the growth thickness of the coating, we used the scanning electron microscope to observe the direction of the coating section. Figure 5 shows the section morphology of Ni-Co-CeO2/Nip (4 g/L) composite coating along the growth direction by separating the composite coating from the substrate. On one hand, it can be seen from the growth section that the composite coating was uniform and flat. On the other hand, the growth thickness of Ni-Co-CeO2/Nip (4 g/L) composite coating was up to 30.3 μm within 30 min of electrodeposition, which indirectly reflected the deposition efficiency of the coating. Compared with electrodeposition, 11 scanning jet electrodeposition was more suitable for deposition under a high current density within a high-speed flowing electrolyte, which could timely supplement the ions concentration on the electrode surface. Therefore, scanning jet electrodeposition had the characteristics of fast and efficient manufacturing.

Section morphology of Ni-Co-CeO2/Nip (4 g/L) composite coating along the growth direction.
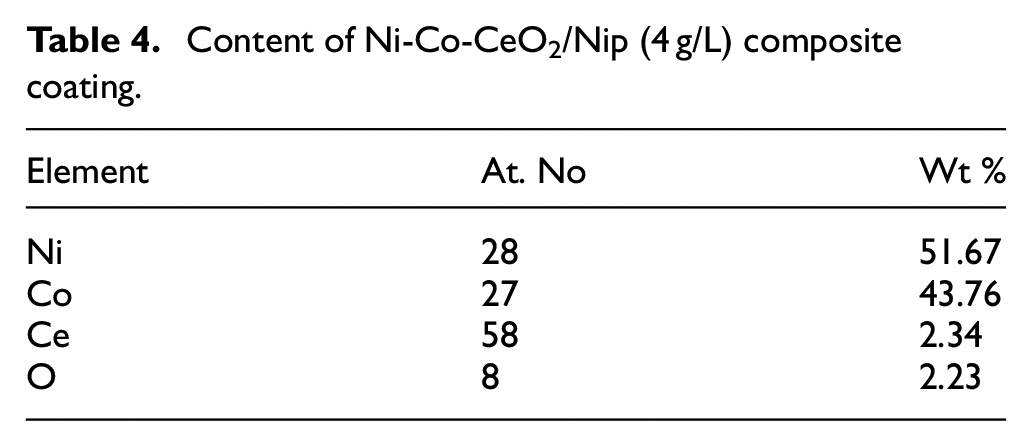
Figure 6 shows the composition of Ni-Co-CeO2/Nip (4 g/L) composite coating along the growth direction by EDS mapping. The distribution of all elements (Ni, Co, Ce, O) is uniform in the Ni-Co-CeO2/Nip (4 g/L) composite coating, which confirms the existence of nano CeO2 deposited successfully by scanning jet electrodeposition. Table 4 lists the element content ratio of Ni-Co-CeO2/Nip (4 g/L) composite coating. In scanning jet electrodeposition, there is an abnormal co-deposition of nickel ions (Ni2+) and cobalt ions (Co2+) due to Co2+ being deposited much easier than Ni2+ in the electrolyte. 29 Therefore, the content ratio of Ni and Co in the alloy composite coating is much lower than that in the electrolyte, as shown in Table 4. In Ni-Co co-deposition, the side reaction hydrogen evolution causes a slight increase in pH near the cathode substrate. The active metal Co2+ forms Co(OH)2 colloid easily under alkaline conditions due to the increase of pH near the cathode surface, it would inhibit the deposition of Ni2+, but Co2+ can continue to deposit thus affecting the Ni-Co content ratio of the composite coating. 30

Element distribution of Ni-Co-CeO2/Nip (4 g/L) composite coating.
Content of Ni-Co-CeO2/Nip (4 g/L) composite coating.
XRD analysis
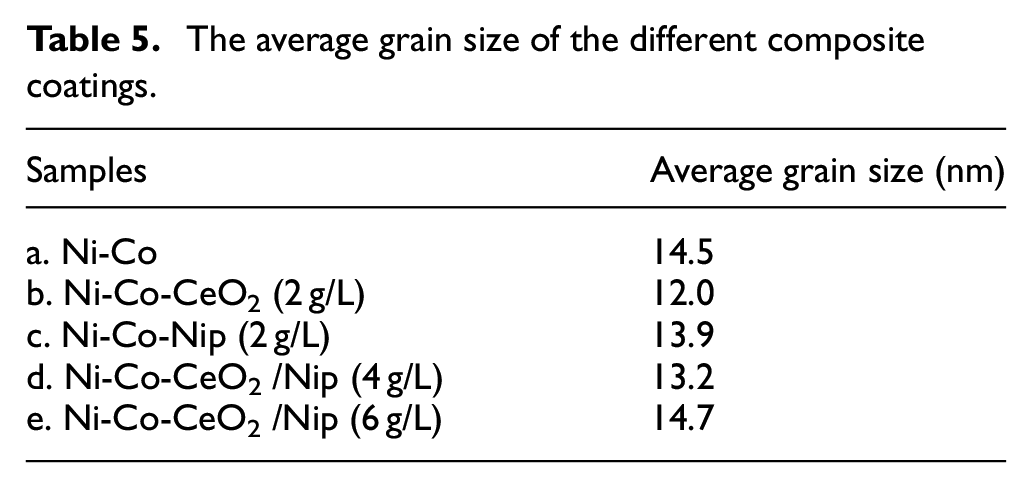
Figure 7 shows the XRD patterns of the Ni-Co alloy composite coatings. All the alloy composite coatings were face-centered cubic (FCC) structures. The preferred growth plane is (111) orientation corresponding to Ni and Co elements in all alloy composite coatings. The Co atoms enter the lattice of Ni atoms to form Ni-Co solid solution in the composite coating due to the radius of Ni atoms and Co atoms being similar. 4 Table 5 lists the average grain size of the alloy composite coatings obtained by Scherrer’s formula. Compared with the average grain size of Ni-Co alloy coating, the addition of nanoparticles refines the average grain size of the composite coating. 31 During the deposition, the nanoparticles act as the growth points of metal ions reduction deposition, thus refining the growth of grains. The grain size of Ni-Co-CeO2/Nip (4 g/L) composite coating is as low as 13.2 nm, which may be due to the grain refinement of nano-mixed particles. Besides, the grain size of the composite coating increase with the concentration of nano-mixed particles, which is caused by the agglomeration of nano-mixed particles.
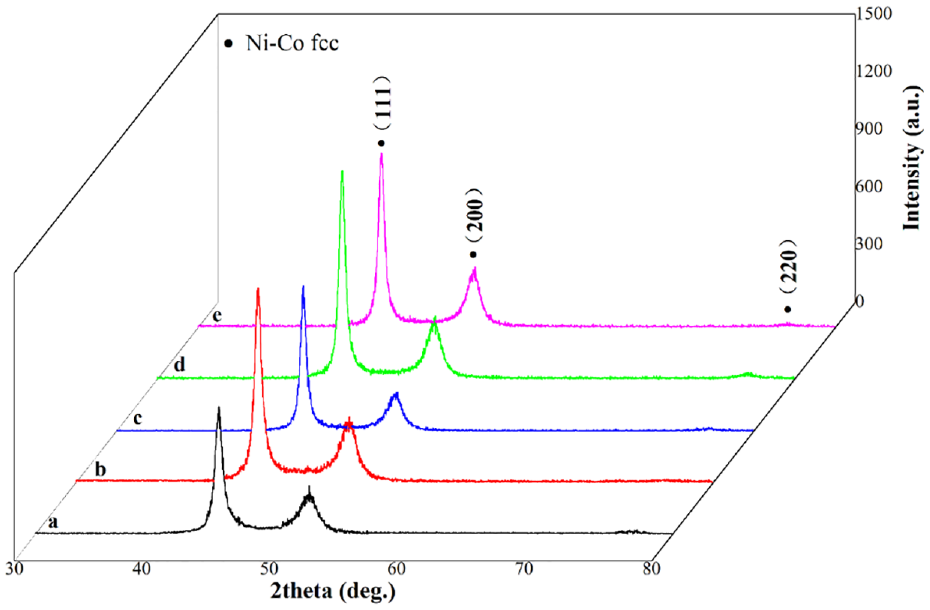
XRD patterns of the different composite coatings.
The average grain size of the different composite coatings.
Microhardness and adhesion
Figure 8 indicates the microhardness and coating adhesion of the different composite coatings. The microhardness and coating adhesion of Ni-Co-CeO2 composite coating is improved with the addition of nano-mixed particles (CeO2/Nip) compared with that of Ni-Co alloy coating. However, the Ni-Co-CeO2/Nip (4 g/L) composite coating has the highest microhardness at 524.7 HV, which is higher than that of Ni-Co-CeO2 composite coating, 19 and the best coating adhesion was at 28.9 N. While the microhardness of the Ni-Co-CeO2/Nip (6 g/L) composite coating decreases to 475.5 HV, and the coating adhesion is reduced to 19.5 N. It is related to the agglomeration of the nano-mixed particles at high concentrations and the surface roughness of the composite coating. The agglomeration of nano-mixed particles causes micro-cracks and pores on the surface of the composite coating, resulting in a decrease in the mechanical properties of the Ni-Co-CeO2/Nip (6 g/L) composite coating.
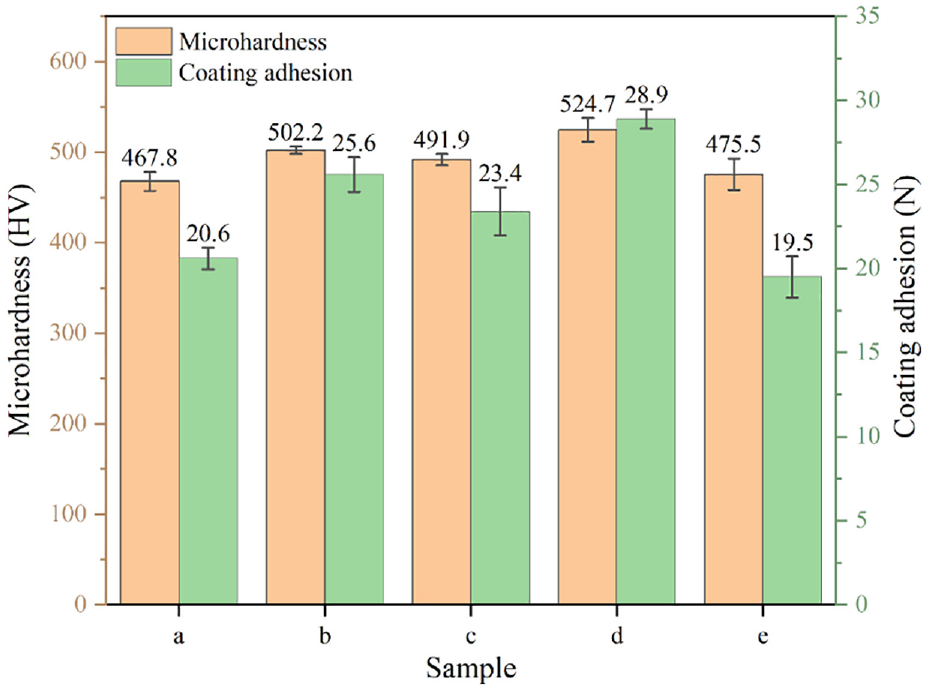
Microhardness and coating adhesion of the different composite coatings.
Generally, adhesion depends a lot on the surface state, 32 the micro unevenness of the substrate surface could increase the adhesion between the coating and the substrate. Firstly, the hooking effect and riveting effect increased the adhesion between the coating and the substrate caused by the deposition of coating metal on the uneven substrate.33–35 The other is that high surface roughness and large real surface area of the substrate resulted in a large contact area between the substrate and the coating, then enhancing adhesion. 36 To improve the adhesion between the substrate and the coating, the surface roughness of the substrate should be appropriately increased whereas the high-roughness surface also led to poor coating flatness. In this work, the micro uneven structure of the surface was polished with sandpaper to achieve a consistent flatness after the uniform pretreatment of the stainless steel substrate.
By comparing the adhesion and the surface roughness of the corresponding composite coating, it shows that the adhesion would be higher with a lower surface roughness of composite coating. 37 There were few micro-cracks or cellular bulge structures on the flat coating. However, once the composite coating had some micro-cracks and cellular bulge structures, it was easy to break and slide under external load, and then the adhesion decreased, which was similar to the previous report. 38 According to the Hall-Petch formula, 31 the refined grain was beneficial to improve the microhardness of the alloy composite coating. The addition of nano-mixed particles (CeO2/Nip) inhibited grain growth of the Ni-Co alloy composite coating. The results show that the dislocations pass through the crystal arrangement in the alloy grain until they reach the grain boundary during plastic deformation. The mismatch of large atoms between different grains also produces dislocation resistance. The reduction of grain size led to higher grain boundary density, which further resists dislocation movement, thus strengthening the material. 39 Based on the dispersion strengthening mechanism of nanoparticles, the refinement of grain size directly increased the effective contact area between the coating and the substrate, improving the stress condition of the coating under external load, strengthening the deformation resistance of the material, and thus improving the alloy composite coating adhesion. 40
Corrosion resistance
Figure 9 shows the Tafel curve of the alloy composite coatings, and the self-corrosion potential and corrosion current density are listed in Table 6. Corrosion current density is a kinetic parameter that reflects the corrosion resistance properties of the coatings. The corrosion resistance of Ni-Co alloy composite coating is improved with the addition of nano-mixed particles, especially the corrosion current density is as low as 5.598 × 10−6 A·cm−2 in Ni-Co-CeO2/Nip (4 g/L) composite coating, which indicates a strong corrosion resistance. However, the corrosion current density of the Ni-Co-CeO2/Nip composite coating increases at 1.384 × 10−5 A·cm−2 when the nano-mixed particles (CeO2/Nip) reach 6 g/L. Figure 10 indicates the Nyquist diagram of the Ni-Co alloy composite coating, and the corresponding arc radius also further confirms the corrosion resistance change of the Ni-Co alloy composite coating, the Ni-Co-CeO2/Nip (4 g/L) composite coating had the strongest corrosion resistance due to the largest arc radius of the Nyquist diagram.
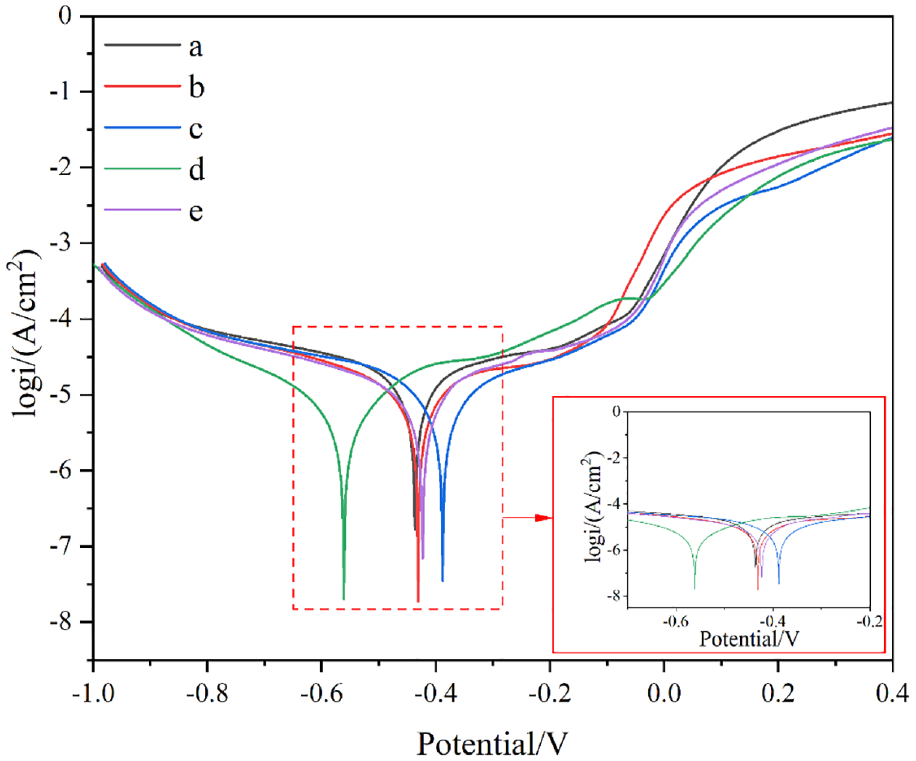
Polarization curves of the different composite coatings.
Electrochemical data from the Tafel curves.
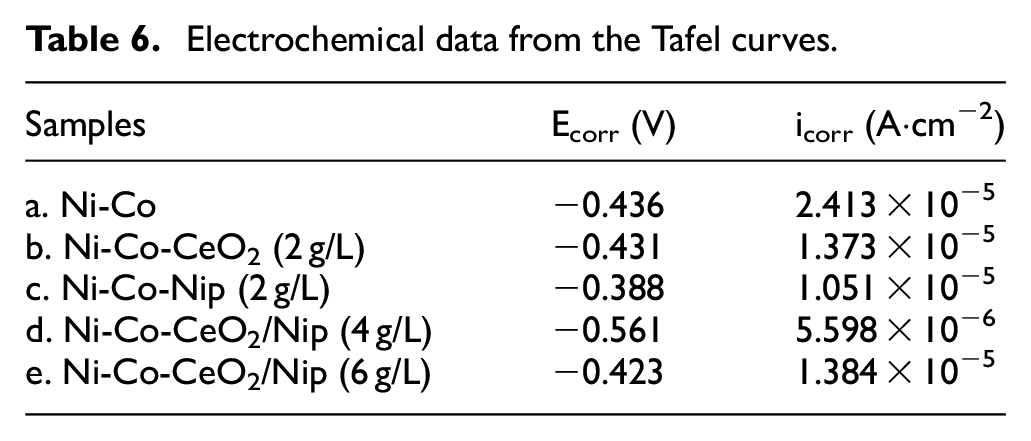
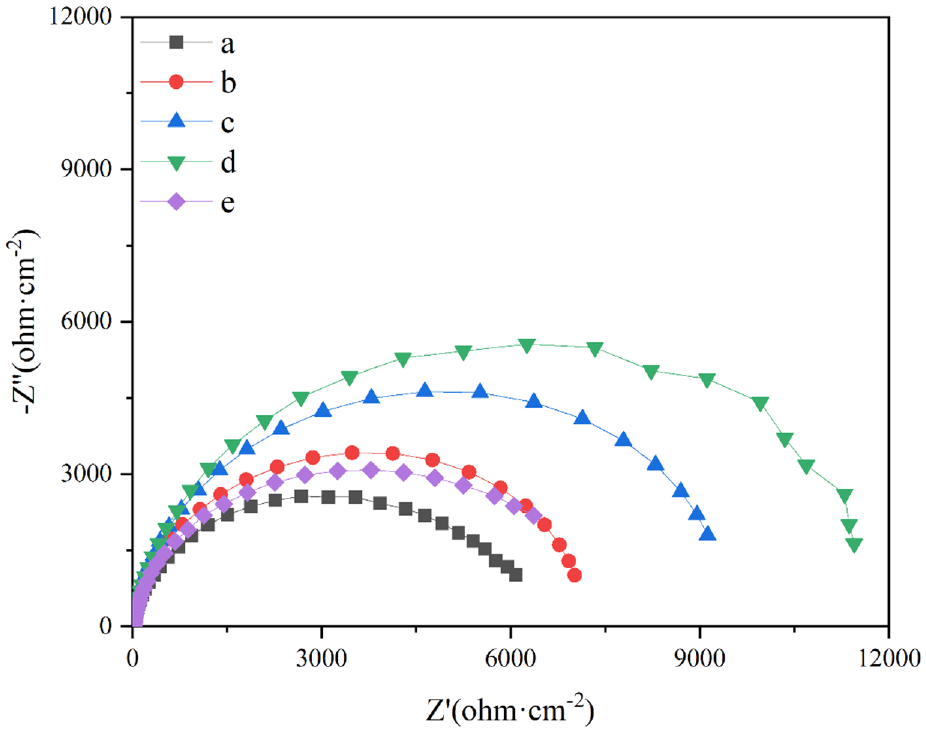
Nyquist diagram of the different composite coatings.
The corrosion of the material was that the corrosive medium penetrated and corroded through the micro-pores and micro-cracks on the composite coating, then destroyed the material structure. 41 Therefore, the surface flatness, uniformity and defects affected the corrosion resistance of the composite coating. 22 The surface flatness and uniformity of the Ni-Co alloy coating improved with the addition of nano-CeO2 particles, so the corrosion resistance of Ni-Co-CeO2 composite coating was better than that of Ni-Co alloy coating. 16 On one hand, nano-CeO2 particles change the morphology of the composite coating, improve the surface flatness and uniformity, and reduce surface defects of the composite coating, which makes it difficult for the corrosion medium to penetrate the interior of the composite coating through the surface. 42 There is no agglomeration and cellular protrusion structure on the surface of the Ni-Co-CeO2/Nip (4 g/L) composite coating when the nano-mixed particles are added at 4 g/L, which further improves the corrosion resistance of the composite coating. However, the surface of the Ni-Co-CeO2/Nip (6 g/L) composite coating has many micro-pores and protrusions due to the agglomeration of nano-mixed particles at high concentrations. 4 These micro-pores provide many corrosion channels for the penetration of Cl- in the electrolyte. The corrosion medium is easy to penetrate the interior of the surface corrosion coating through these micropores, so the corrosion resistance of the Ni-Co-CeO2/Nip (6g/L) composite coating was weakened.
Conclusions
In summary, this paper studied the effect of nano-mixed particles on the Ni-Co alloy composite coating in scanning jet electrodeposition. Compared with single nano CeO2 and nano nickel particles, it proved that nano-mixed particles (CeO2/Nip) could further improve the mechanical properties and corrosion resistance of Ni-Co alloy coatings.
The nano-mixed particles affected the morphology and surface smoothness of the composite coating. In scanning jet electrodeposition, the surface roughness of Ni-Co alloy coating is 0.853 μm, while the surface roughness of Ni-Co-CeO2/Nip composite coating is reduced at 0.595 μm when nano CeO2 and nano nickel particles are mixed at 4 g/L, which is related to the grain growth refined by nano-mixed particles and the improvement of the composite coating surface quality.
The average grain size of the Ni-Co-CeO2 composite coating is closely related to the nanoparticles. The average grain size of the Ni-Co-CeO2 composite coating is 12.0 nm when with the addition of nano CeO2 particles at 2 g/L. The average grain size of the Ni-Co-Nip composite coating is 13.9 nm with the addition of nano nickel particles at 2 g/L. The average grain size of Ni-Co-CeO2/Nip composite coating decreases to 13.2 nm when the concentration of nano-mixed particles (CeO2/Nip) was 4 g/L.
Ni-Co-CeO2/Nip composite coating exhibited excellent mechanical properties and corrosion resistance. Compared with Ni-Co alloy coating, the microhardness increases from 467.8 to 524.7 HV, and the coating adhesion is strengthened from 20.6 to 28.9 N. It is also worth noting that the Ni-Co-CeO2/Nip (4 g/L) composite coating indicated excellent corrosion resistance, its current density was 5.598 × 10−6 A·cm−2. Therefore, nano-mixed particles could further enhance the mechanical properties and corrosion resistance of the Ni-Co alloy coating.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project is supported by the Fundamental Research Funds for the Central Universities (No. JZ2022HGQA0158, No. JZ2022HG TA0323) and the Innovation and Entrepreneurship Training Program for College Students in Anhui Province (No. S202210359028). Y. Lao wishes to express his sincere gratitude to the Guangxi scholarship fund of the Guangxi education department (GCD2019).
