Abstract
Background
Renin-angiotensin system inhibitors (RASi) offer important benefits for patients on peritoneal dialysis (PD), particularly in preserving residual kidney function and peritoneal membrane integrity. Despite these benefits, concerns about hyperkalemia and hypotension often limit clinical practice utilization.
Objectives
To achieve a 20% increase in RASi utilization among eligible patients on PD at an academic hospital in Toronto, Canada through a quality improvement initiative.
Methods
We conducted a pre-intervention analysis through retrospective chart review from July 2022 to September 2023. We implemented a “PD Passport,” a clinical documentation tool used by clinic staff in each visit to highlight missed RASi prescription opportunities. The primary outcome measure was RASi utilization at 6-month post-implementation. Process measures included PD passport completion rates, while balancing measures tracked rates of symptomatic hypotension and hyperkalemia.
Results
Among 63 patients on PD (mean age 58.7 years, 55.6% male), baseline RASi utilization was 41%. Following implementation, RASi utilization increased to 59% by October 2024, representing a 17% increase but falling short of the 20% target. There were no significant differences in mean systolic blood pressure (125.71 ± 4.19, 125.64 ± 7.02 mmHg; p = 0.653), mean serum potassium (4.34 mmol/L, 4.31 mmol/L; p = 0.662), and mean urine output (915.2 mL, 921.8 mL; p = 0.881) before and after the intervention.
Conclusions
The PD Passport initiative substantially increased RASi utilization by 17% without compromising patient safety, as evidenced by stable blood pressure and potassium levels. While falling slightly short of our 20% target, this structured documentation approach effectively bridges the gap between evidence and practice, demonstrating the value of targeted tools in enhancing guideline-concordant care for PD patients.
Introduction
Transforming peritoneal dialysis (PD) care demands innovative quality improvement (QI) strategies that bridge the gap between clinical evidence and everyday practice. Effective implementation requires both methodological rigor and practical approaches that healthcare teams can readily adopt to enhance patient outcomes. While theoretical frameworks for conducting QI in PD exist in the literature, comprehensive accounts detailing real-world implementation experiences remain limited.1,2 This presents significant challenges for PD programs seeking to systematically improve care delivery, particularly in domains fundamental to optimal PD outcomes such as preservation of residual kidney function—a key principle outlined in the International Society for Peritoneal Dialysis (ISPD) practice recommendations for prescribing high-quality goal-directed PD care.3,4 Understanding practical approaches to implementing evidence-based improvements in PD programs is essential for advancing care quality and patient outcomes in this specialized field. 1
Renin-angiotensin system inhibitors (RASi), encompassing angiotensin-converting-enzyme inhibitors and angiotensin-receptor blockers, demonstrate significant therapeutic benefits in PD patients beyond cardiovascular protection and proteinuria reduction.5–8 Observational data have shown benefits in patients on PD in terms of preserving residual kidney function (through estimated glomerular filtration rate and measured residual urine volume) as well as protecting peritoneal membrane structure.3,5,9,10 Residual kidney function is a strong predictor of morbidity and mortality in PD patients, as demonstrated by various studies but most notably the re-analysis of the landmark CANUSA study, highlighting that every 5 L/week/1.73 m incremental increase in GFR and each 250 mL increase in urine volume corresponded with a decrease in relative risk of death by 12% and 36%, respectively.3,11 Interestingly, RASi are thought to facilitate downregulation of peritoneal RAS activity induced by PD solutions, potentially mitigating membrane failure via reduced fibrosis and neo-angiogenesis.5–7,9,10,12
Although the 2015 ISPD cardiovascular and metabolic guidelines endorse RASi use for PD patients with left ventricular hypertrophy or heart failure, no formal recommendation currently exists for their use in preserving residual kidney function. 13 However, the guideline acknowledges the potential benefit and includes a weak recommendation encouraging RASi use in patients on PD with significant residual kidney function as tolerated. This is based on limited trials demonstrating a slower decline in kidney function and possible extra-renal benefits. 13 Data on RASi prescribing patterns in PD remain limited. Most large registry data, including the Dialysis Outcomes and Practice Patterns Study, focus primarily on patients receiving hemodialysis, where RASi use was reported at only 39 percent with minimal variation by cardiovascular comorbidity. 14 These findings point to broader therapeutic inertia.
Further contributing to underutilization are clinician concerns about hyperkalemia and hypotension, especially in the setting of impaired urinary, colonic, and peritoneal clearance.5,10,15,16 The recognition of RASi safety and benefit in PD is relatively recent, and variability in uptake persists across center. 17 We have observed this hesitancy within our own program. Together, these gaps in evidence and practice informed our decision to focus this QI initiative on optimizing RASi use in PD.
As a result, our study aims to address this gap by implementing and rigorously evaluating the “PD Passport,” a structured documentation tool designed to increase appropriate RASi prescribing by 20% in our PD program. A 20% improvement target was selected based on baseline prescribing rates, feasibility within the local context, and what was deemed a clinically meaningful and achievable change over a 12-month period. This QI initiative not only targets an important clinical need but also provides a practical framework that dialysis programs worldwide can adopt to enhance evidence-based medication use while maintaining patient safety. Through systematic implementation and evaluation, we contribute valuable methodological insights to PD care—an area where structured approaches to QI remain notably scarce in the literature.
Methods
Ethical consideration
This QI study was registered at Sunnybrook Health Sciences Centre in Toronto, Canada (QI Registration #1311761686), given that this study aligned institutional practices with established QI principles, formal ethics evaluation was not required based on institutional criteria for QI initiatives. The investigation adhered to the Standards for Quality Improvement Reporting Excellence guidelines to ensure comprehensive and transparent reporting.
Setting and participants
The study was conducted in our home dialysis clinic at Sunnybrook Health Sciences Centre, a large teaching hospital in Toronto, Canada. This clinic consists of a multidisciplinary team including a nephrologist, clinical fellow, pharmacist, dietician, social worker, and nurses. Patients typically attend clinic visits every 6–10 weeks for routine follow-up. We reviewed all patients on PD between July 2022 and October 2024. Patients were excluded if they had a primary indication for RASi therapy (such as heart failure) or no residual urine output. Patients without residual kidney function were excluded as they would not be expected to benefit from RASi with respect to preserving residual kidney function, which was the primary rationale for this intervention. Their increased risk of hyperkalemia was also taken into account.
Intervention development and implementation
To establish baseline RASi prescription patterns, we conducted a retrospective chart review using our electronic medical record system (Accuro) of all adult patients on PD followed in our clinic between July 2022 and December 2023, excluding those without residual kidney function, those no longer active in the PD program, or those who had transitioned to hemodialysis or transplant prior to the intervention. We reviewed data on patient demographics, blood pressure measurements, laboratory results, RASi use (verified through medication administration records and pharmacist review), and residual urine output volume.
Prior to implementation, we held organizational stakeholder meetings with the entire home dialysis team to conduct a root cause analysis assessing practitioners’ perspective on top barriers to prescribing RASi in PD patients and to establish consensus on RASi indications and contraindications in PD patients. Four stakeholder meetings were held with the home dialysis team on a bimonthly basis between October 2023 and April 2024 to support intervention development and implementation. The project team, consisting of staff nephrologists, nephrology trainees, and allied health members, identified clinical challenges, developed interventions, and assessed implementation strategies that could be integrated into clinic workflow.
We developed a clinician-targeted “PD Passport” tool in collaboration with the nursing team, pharmacist, and nephrologists (Figure 1). The passport required documentation of RASi status, heart failure history, and reasons for non-implementation at each visit. Implementation began in May 2024, with the tool being used at each patient's quarterly clinic visits. The PD Passport was completed by either nursing staff or nephrology medical practitioners at any point in the visit assessment, but often with review of medications. After a 2-week pilot period, we gathered qualitative feedback from staff nephrologists, clinic nurses, and nephrology trainees to assess initial implementation and no challenges were reported. Implementation of the PD passport was ongoing for the subsequent 6 months.
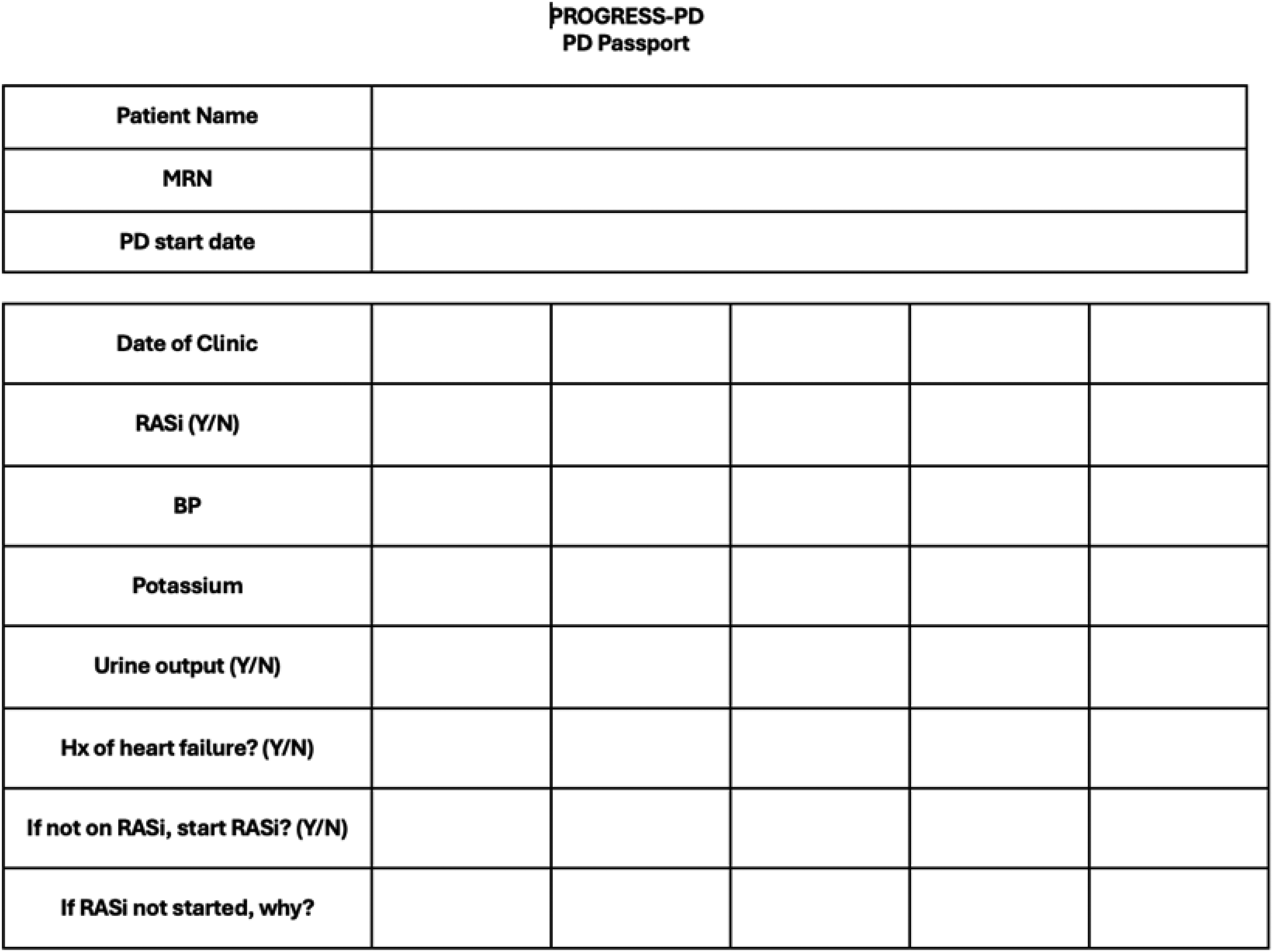
The peritoneal dialysis passport created by our team. The passport serves as a standardized documentation tool to track RASi prescription status and relevant clinical parameters during routine clinic visits such as 24-h urine collection and urine output monitoring. Abbreviations: PD: peritoneal dialysis; MRN: medical record number; RASi: renin-angiotensin system inhibitor; BP: blood pressure; Hx: history.
Outcome measures
Primary outcome measures included the percentage of eligible PD patients prescribed RASi at 6-month post-implementation. This was a serial cross-sectional analysis comparing RASi prescribing rates at baseline and again at 6 months among all active PD patients in the clinic during each respective time point. While RASi prescriptions were reviewed on a monthly basis, the primary outcome was defined as the proportion of patients on RASi at 6 months following implementation. This interval was chosen to allow for 2–3 clinic visits to evaluate practice change sustainability. Residual urine output was quantified through patient-performed 24-h urine collections.
We tracked process measures through the completion rate of PD passports at each clinic visit. Balancing measures included the incidence of adverse effects: symptomatic hypotension (defined as blood pressure less than 100/60 mmHg) and hyperkalemia (serum potassium greater than 5.5 mmol/L), documented during routine clinic visits or hospitalizations. Hypotension was defined as a systolic blood pressure <100 mmHg, consistent with prior dialysis literature, 18 where arterial hypotension is commonly defined at this threshold to identify patients at risk of medication intolerance and adverse events. Patients were monitored for 6 months following implementation in May 2024, tracking both RASi initiation and these potential adverse effects through quarterly clinic visits and hospital admissions.
Statistical analyses
Paired t-tests were used to compare pre- and post-intervention systolic blood pressure, serum potassium levels, and urine output among patients with available data.
Results
Baseline characteristics
Among 63 patients on PD, the mean age was 58.67 ± 16.89 years, with males comprising 55.6% (n = 35). The median dialysis vintage was 944 days (IQR 427–1143). Automated PD was the predominant modality (92.1%, n = 58), with a mean daily dialysate volume of 8291.83 ± 2356.42 mL. Diabetes was the primary cause of end-stage kidney disease (39.7%, n = 25), followed by glomerulonephritis (25.4%, n = 16), other causes (19.0%, n = 12), hypertension (9.5%, n = 6), reflux/obstructive kidney disease (4.8%, n = 3), and polycystic kidney disease (1.6%, n = 1) (Table 1).
Baseline demographic and clinical characteristics of patients on peritoneal dialysis (n = 63).
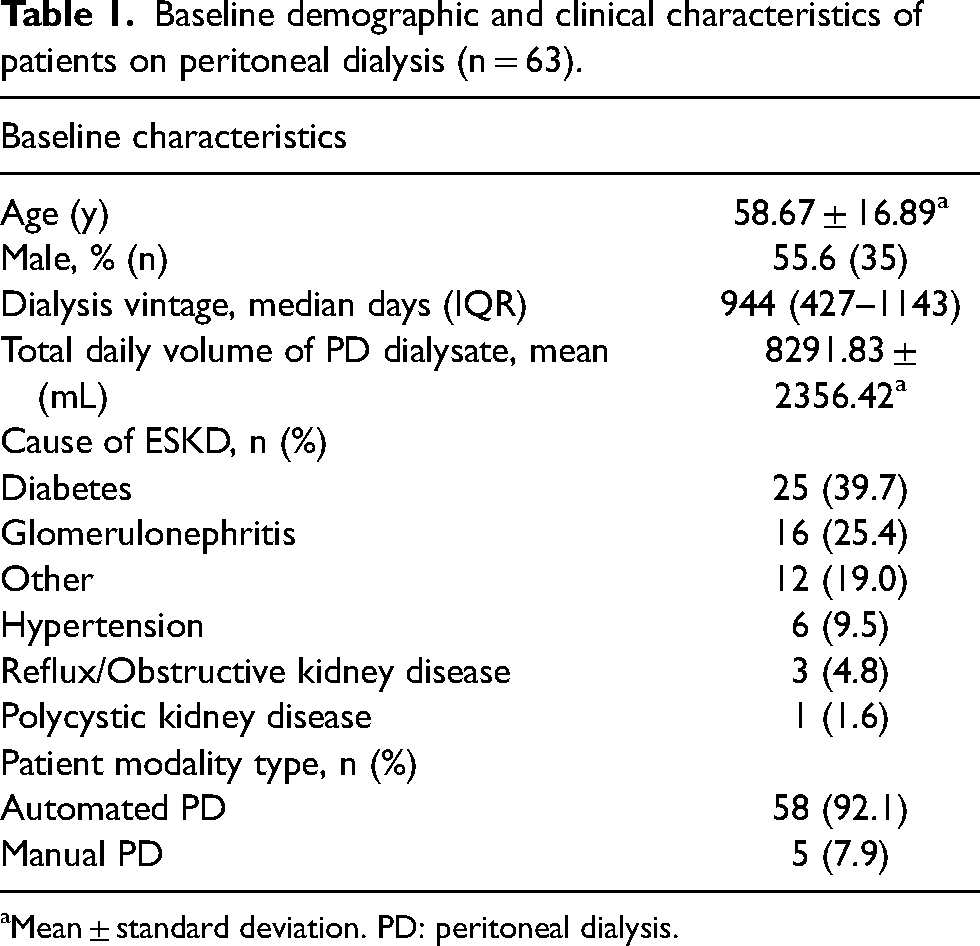
Mean ± standard deviation. PD: peritoneal dialysis.
Pre-intervention analysis
The Pareto analysis revealed key barriers to RASi prescription in the PD population. RASi blockade discontinuation at dialysis initiation represented the most frequent barrier (45.5%, n = 5), followed by preference for alternative blood pressure medications (18.2%, n = 2) and lack of clinical prioritization (18.2%, n = 2). Less common barriers included uncertainty about residual urine output (9.1%, n = 1) and concerns regarding side effect profiles (9.1%, n = 1). The cumulative analysis demonstrated that addressing the three most frequent barriers would impact 81.8% of cases where RASi therapy was not prescribed (Figure 2).
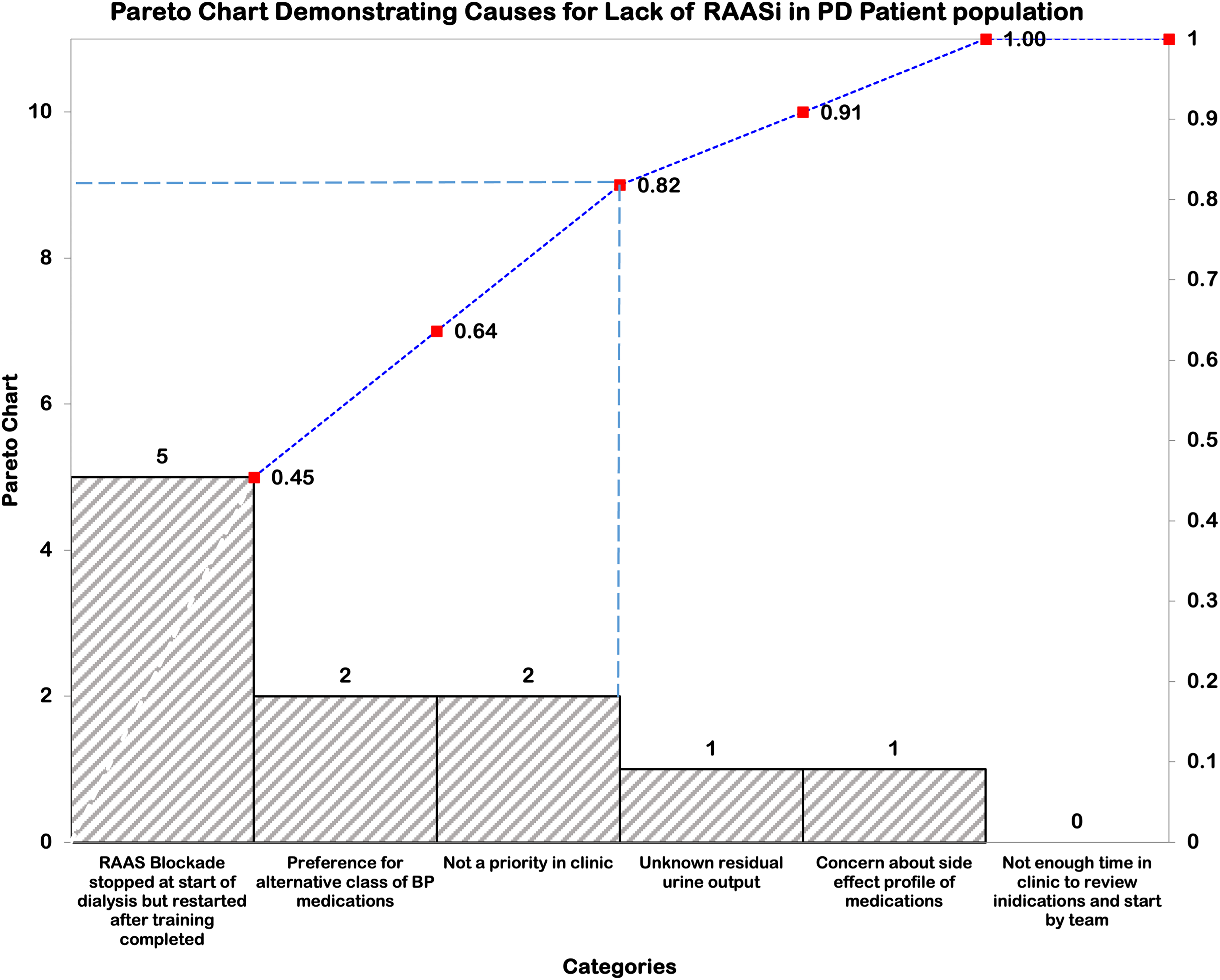
Pareto chart illustrating barriers to RASi prescription identified through multidisciplinary team feedback of all patients on peritoneal dialysis in our program. The blue bars represent individual frequencies (left y-axis), while the dotted line tracks cumulative percentages (right y-axis). The dashed vertical line marks the 80/20 principle threshold at 81.8%. Abbreviations: RASi: renin-angiotensin aldosterone system inhibitor; PD: peritoneal dialysis; BP: blood pressure.
Outcome measures
The statistical process control chart demonstrated notable changes in RASi utilization from July 2022 to October 2024. The baseline period showed a mean utilization of 41% with established control limits (UCL 51.13%, LCL 38.07%). A marked decline was observed from December 2023 to March 2024, reaching a low of 33.4%. The improvement trajectory accelerated from April 2024, with the final measurement reaching 59.49% in October 2024. Special cause variation was identified when two of three consecutive data points appeared near a control limit in the outer one-third region, indicating a statistically significant shift in process performance (Figure 3).
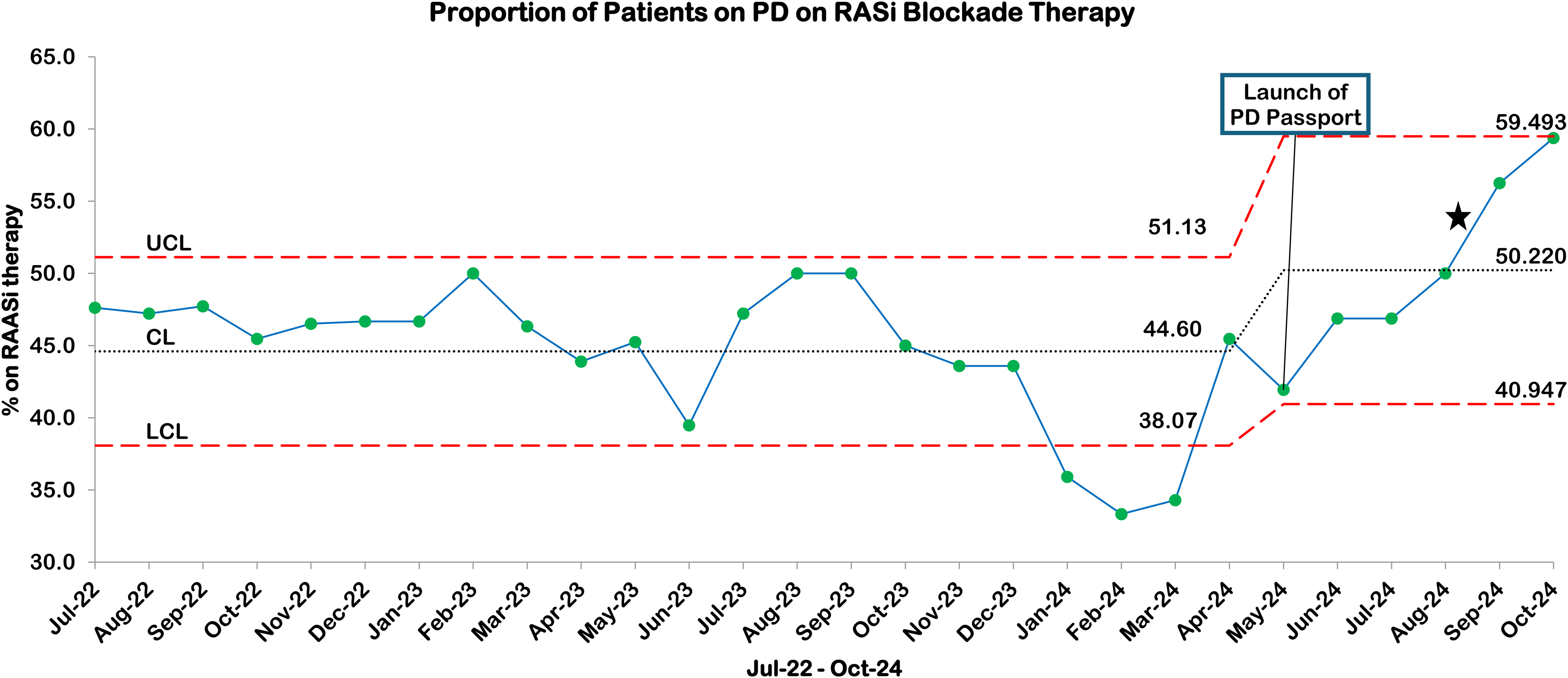
Statistical process control chart showing the proportion of patients on peritoneal dialysis receiving RASi therapy from July 2022 to October 2024. The baseline period demonstrated a mean (CL) of 44.60%* with upper and lower control limits (UCL, LCL) of 51.13% and 38.07%, respectively. Following PD Passport implementation in May 2024, a sustained improvement was observed with recalculated control limits (UCL 59.49%, LCL 40.95%) and a new mean of 50.22%. The star (★) denotes special cause variation with two out of three consecutive points near a control limit (outer one-third), signifying a significant shift in process performance. UCL: upper control limit; LCL: lower control limit; CL: center line (mean). *Our baseline RASi is higher than previously cited in our manuscript because here our software includes the full duration of the study to calculate a baseline rate rather than the timeframe specified for our pre-intervention analysis. RASi: renin-angiotensin system inhibitor; PD: peritoneal dialysis.
Balancing measures
Serial monitoring of clinical parameters showed stability across multiple measures. Mean serum potassium values demonstrated minimal variation between pre-intervention (4.34 ± 0.15 mEq/L) and post-intervention periods (4.31 ± 0.14 mEq/L) (p = 0.662). Mean systolic blood pressure remained consistent between pre-intervention (125.71 ± 4.19 mmHg) and post-intervention (124.75 ± 4.25 mmHg) periods (p = 0.653). Similarly, mean urine output showed stability from pre-intervention (915.2 ± 2.8 mL) to post-intervention (921.83 ± 3.1 mL) (p = 0.881) (Figure 4).
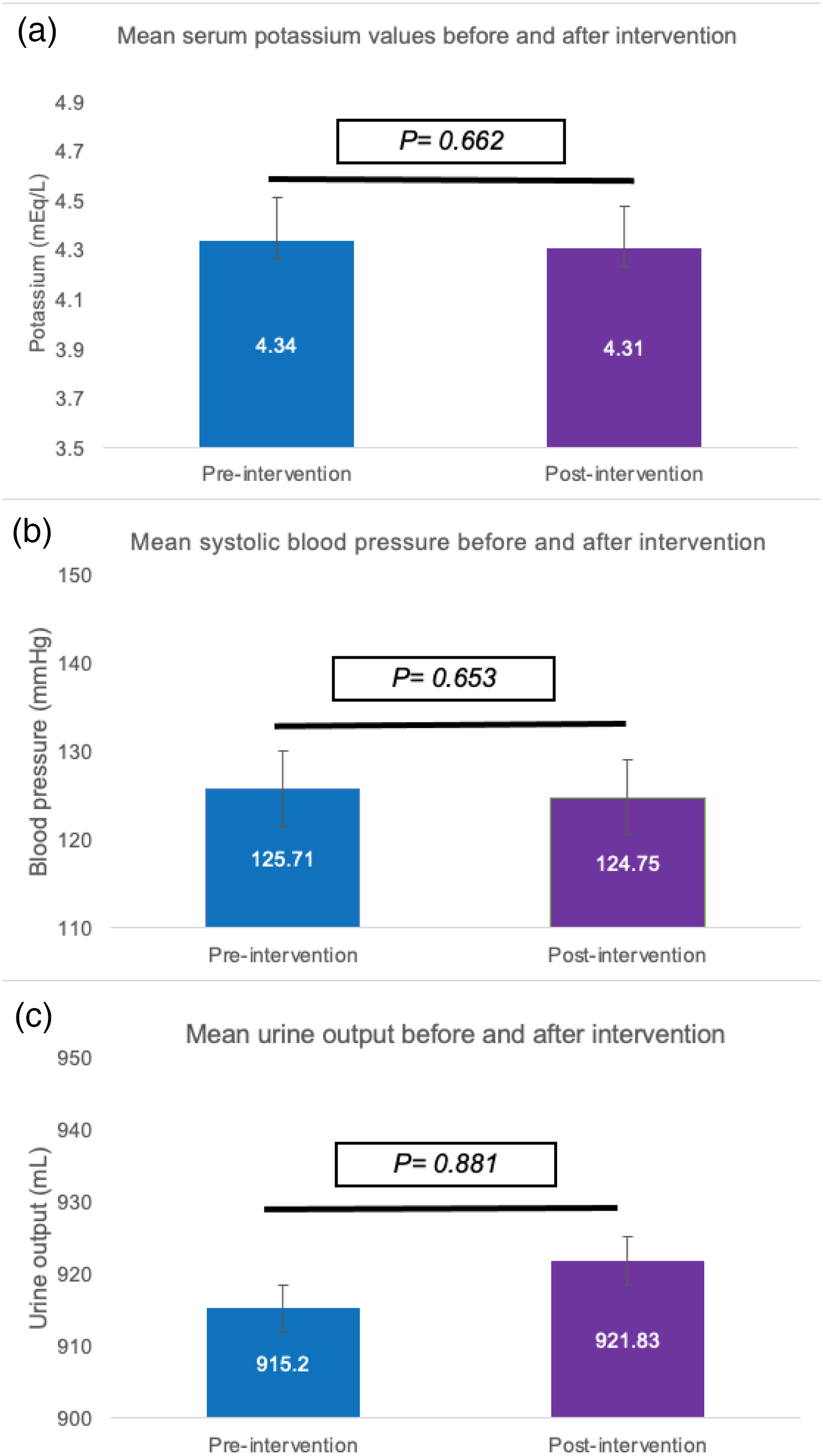
Comparison of key clinical parameters before and after PD passport implementation. (a) Mean serum potassium levels showed no significant difference between pre-intervention (4.34 ± 0.15 mEq/L) and post-intervention (4.31 ± 0.14 mEq/L) periods (p = 0.662). (b) Mean systolic blood pressure remained stable from pre-intervention (125.71 ± 4.19 mmHg) to post-intervention (124.75 ± 4.25 mmHg) periods (p = 0.653). (c) Mean urine output measurements demonstrated minimal variation between pre-intervention (915.2 ± 2.8 mL) and post-intervention (921.83 ± 3.1 mL) periods (p = 0.881). Error bars represent standard error of the mean. Statistical significance was determined using paired t-tests with p < 0.05 considered significant. PD: peritoneal dialysis.
Discussion
Implementation of the PD passport, a simple documentation tool alerting healthcare teams to missed RASi prescription opportunities, led to a meaningful 17% improvement in RASi utilization in our program. Though slightly below our 20% target, this intervention successfully increased appropriate RASi prescribing without increasing adverse events such as hypotension or hyperkalemia. There was no change to residual urine output and by proxy residual kidney function with our PD Passport intervention. Our study provides a practical demonstration of QI methodology in PD care, illustrating how a structured documentation approach used by both nursing and physician providers can effectively translate evidence into practice. To our knowledge, this represents the first evaluation of a QI intervention specifically targeting RASi utilization in the PD population.
The PD passport intervention increased RASi utilization but did not achieve our 20% target. Several factors likely contributed to this limited increase: the absence of residual urine output in some patients (raising concerns about hyperkalemia), acute illnesses, competing comorbidities, and our relatively high baseline RASi utilization (41%). We observed a potential Hawthorne effect between March and April 2024, with an 11% increase in RASi use following project announcement but before implementation, a phenomenon well-documented in QI literature. 19 The significant decrease in RASi use between September 2023 and February 2024 likely represents statistical variance, as no major changes occurred in our center during this period. Notably, our baseline utilization aligns with United States Renal System data, which reported 44% RASi use among Medicare-covered patients on PD from 2007 to 2011. 20 Importantly, our intervention did not increase the incidence of hyperkalemia or hypotension in clinic visits or hospital admissions, while residual urine volume remained stable. This safety profile is particularly relevant given that these side effects often deter RASi use in patients on PD, especially those with residual urine output.5,10,16 Our findings align with previous studies showing no increased risk of hyperkalemia 16 or hypotension5,21 with RASi use in PD patients.
RASi offer established cardiovascular and kidney benefits. In the PD population, they are associated with preservation of residual urine output3,4—a key mortality predictor—and may reduce peritoneal membrane sclerosis and in turn improve PD technique survival.5,6,12 Despite clinical guidelines supporting RASi use in PD, prescribing hesitancy persists due to patient variability and the complexities of managing end-stage kidney disease. 4 Our intervention using the PD passport aimed to systemically address barriers to RASi prescription by targeting clinical hesitancy and treatment initiation inertia. Anecdotally, clinic members were more aware of RASi indications and contradictions in their patients and benefitted from having a standardized tool during busy visitations.
These findings have broader healthcare system implications as well. Using cost-minimization frameworks, PD has shown to be the most efficient dialysis modality internationally. 22 In Canada, PD maintenance has been found to be more cost-effective ($83,76/QALY) than hemodialysis ($104,880/QALY) over a 12-year time frame. 23 These costs benefits have been redemonstrated in many other countries and in studies accounting for patient comorbidity variability.22,24 Our QI framework demonstrates practical approaches to optimizing PD care, addressing both cost and morbidity concerns in end-stage kidney disease management.
In our publicly funded healthcare system, increased RASi utilization added minimal direct costs, with monthly RASi expenses ranging from $1–10 for patients without drug coverage. 25 While the intervention's direct costs were limited, we acknowledge potential indirect costs, particularly the time burden of completing the PD passport during clinic visits. Although we did not formally assess documentation fatigue among our team members, this remains an important consideration for intervention sustainability. 26
Our study has several important limitations. First, the cohort consisted was relatively small and predominantly male, with a median dialysis vintage of 2.6 years, and a high prevalence of diabetic kidney disease. The overwhelming use of automated PD (over 90%) may also limit generalizability to centers with different populations or modality distributions. Second, the 6-month follow-up period, while sufficient to evaluate short-term safety with respect to hyperkalemia and hypotension, was not long enough to assess the durability or increased RASi uptake or its impact on residual kidney function over time. Third, we did not assess patient-reported outcomes such as satisfaction, perceived medication burden, or adherence—all of which may shape the real-world success of implementation efforts, particularly in the context of polypharmacy. Lastly, we did not systematically collect data on concurrent diuretic use, which may influence prescriber confidence in initiating or maintaining RASi therapy, especially given the common “as needed” prescribing pattern in our program. Despite these limitations, this study provides actionable insights into the feasibility and short-term safety of a structured intervention to improve RASi prescribing in PD and highlights important areas for future QI and research.
Conclusion
In this QI initiative, we demonstrated that a simple documentation tool can transform clinical practice by substantially increasing RASi utilization in PD patients. The 17% improvement achieved—nearly reaching our ambitious 20% target—confirms that structured approaches to medication optimization can successfully bridge the evidence-practice gap, even in centers with relatively high baseline utilization. Importantly, this increased RASi prescribing occurred without compromising patient safety, as evidenced by stable blood pressure and potassium levels throughout the implementation period. Our findings extend beyond medication optimization to provide a practical blueprint for implementing evidence-based recommendations in PD care. By systematically addressing prescribing barriers through a standardized documentation approach, improving clinical hesitancy, and prompting treatment guidance, we demonstrate how targeted QI can align clinical practice with ISPD guidelines for preserving residual kidney function—a critical determinant of patient outcomes. While longer-term studies are needed to evaluate the impact on preservation of kidney function, this work establishes a foundation for other dialysis programs seeking to enhance evidence-based care through systematic QI methodology.
Footnotes
Acknowledgements
The authors gratefully acknowledge the support of the Sunnybrook Health Sciences Centre home dialysis team for their collaboration in implementing and utilizing the PD passport. We thank our multidisciplinary team members for their expertise in patient advocacy and care optimization, and express our appreciation to the clinic nurses, pharmacists, dieticians, and patients who contributed to the success of this initiative.
Authors’ contributions
Study design and concept done by BLA; data acquisition done by MS, AD, MA, and CG; supervision done by BLA; data analysis/interpretation: all authors. CG contributed to data analysis. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Declaration of conflicting interests
BLA has received speaking honoraria from Baxter Healthcare. All other authors declare that they have no relevant financial interests.
Ethical approval
This QI study was registered at Sunnybrook Health Sciences Centre in Toronto, Canada (QI Registration #1311761686). Given that this study aligned institutional practices with established QI principles.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This QI project was supported by the Home Dialysis Baxter–Oreopoulos Grant from the University of Toronto's Division of Nephrology.
Informed consent to participate
NA.
Informed consent to publish
Yes.
