Abstract
Home-based peritoneal dialysis (PD) is less resource-intensive than in-center hemodialysis. When provided with a choice, many patients prefer home-based therapies. The Starting Dialysis on Time, At Home on the Right Therapy (START) project was a quality improvement initiative aiming to increase PD use in Alberta, Canada. The START project provided site-specific audit and feedback reports on the processes of care for PD and increased the use of PD. In this current study, we conducted a retrospective cost analysis of the START project. We used the perspective of a publicly funded healthcare system to compare the costs before and after the START intervention. We used a decision analytic model stratifying the patient cohort by age (under and over 65 years) and estimated the impact of the START intervention on the overall cost of care at 1, 3, 5, and 10 years. Sensitivity analyses were performed. We found cost savings of $CAD 1.2 million, $CAD 1.9 million, $CAD 2.4 million, and $CAD 2.7 million for the START intervention at 1, 3, 5, and 10 years, respectively. Results were robust to a variety of sensitivity and scenario analyses. Even modest increases in PD utilization led to cost savings. We found that the implementation of a quality improvement initiative to increase PD resulted in substantial cost savings over time.
Background
The global demand for dialysis is increasing, putting financial pressure on health systems.1,2 Home-based peritoneal dialysis (PD) is less resource-intensive than in-center hemodialysis (HD) 3 in most developed countries, and clinical outcomes are similar. 4 Many patients also report a preference for home-based therapies, with data supporting a higher quality of life for those on PD. 5 As a result, many dialysis programs promote PD use.
We recently reported on the Starting Dialysis on Time, At Home on the Right Therapy (START) project, 6 a quality improvement initiative that increased the use of PD in Alberta, Canada. Measuring the impact of the START intervention on overall healthcare costs is important to inform future decision-making. In this study, we conducted a cost analysis to assess the costs and savings associated with the START intervention.
Methods
Overview
We conducted a retrospective cost analysis using data from the quality improvement project to evaluate the impact of the START intervention, described previously.6,7 Briefly, the START project provided dialysis programs with high-quality, site-specific data on individual steps in the process of care (identification, assessment, determination of eligibility, offer of PD, patient choice, and receipt of PD) that drive PD utilization. Audit and feedback reports to individual clinics with tailored information allowed programs to identify site-specific barriers and then design, implement, and evaluate local interventions. Quarterly reports were provided to sites, combined with discussion of improvement strategies at innovation learning collaborative sessions and local working group meetings. During the study period, a total of 1962 patients started dialysis; 964 before the implementation of START and 998 after the implementation of the project. The START project demonstrated an absolute increase in the use of PD (at 6 months after dialysis initiation) province-wide by 5.4% (95% CI: 1.5% to 9.2%) with a level change model. Despite the increase in PD utilization, there were no changes in the rates of hospitalization, death, or probability of transfer to HD in the first year of therapy.
Decision model design and assumptions
We used data on PD use at 6 months (study primary outcome), with estimates of the costs of implementing the START project and the costs of HD and PD provision. Costs are in 2020 Canadian Dollars ($CAD), discounted at 5% per year.
We used the perspective of a publicly funded healthcare system to compare the cost of caring for a cohort of patients starting dialysis before and after the START intervention. In the 18 months before and after initiating the START project, 964 and 998 patients started dialysis, respectively. We created a decision analytic model, stratifying the patient cohort by age (under and over 65 years), and estimated the impact of the START project on the overall cost of care at 1, 3, 5, and 10 years for these two “18-month” cohorts. We conducted a Markov analysis in individuals under and over 65 years of age. Participants then transition into three of the four health states (Figure 1): alive with HD, alive with PD, or alive with a transplant. From there, they can remain in their current health state, transition to another health state, or die. Those on PD can switch to HD or receive a transplant. Those on HD can either continue on HD or receive a transplant. Individuals who have had a transplant can return to HD.
The model was developed and analyzed using best practices 8 using TreeAge Pro 2024, R2.1 Healthcare software (Williamstown, MA, USA), validated against an Excel model. The usual care and START project time periods had similar patients, and we supplemented START data (including proportion of patients on PD and PD failure rates within 12 months), with data from the Canadian Organ Replacement Registry 9 (transplant failure rates) to model the costs of the program overall. We assumed the program's effectiveness at increasing PD use would persist.
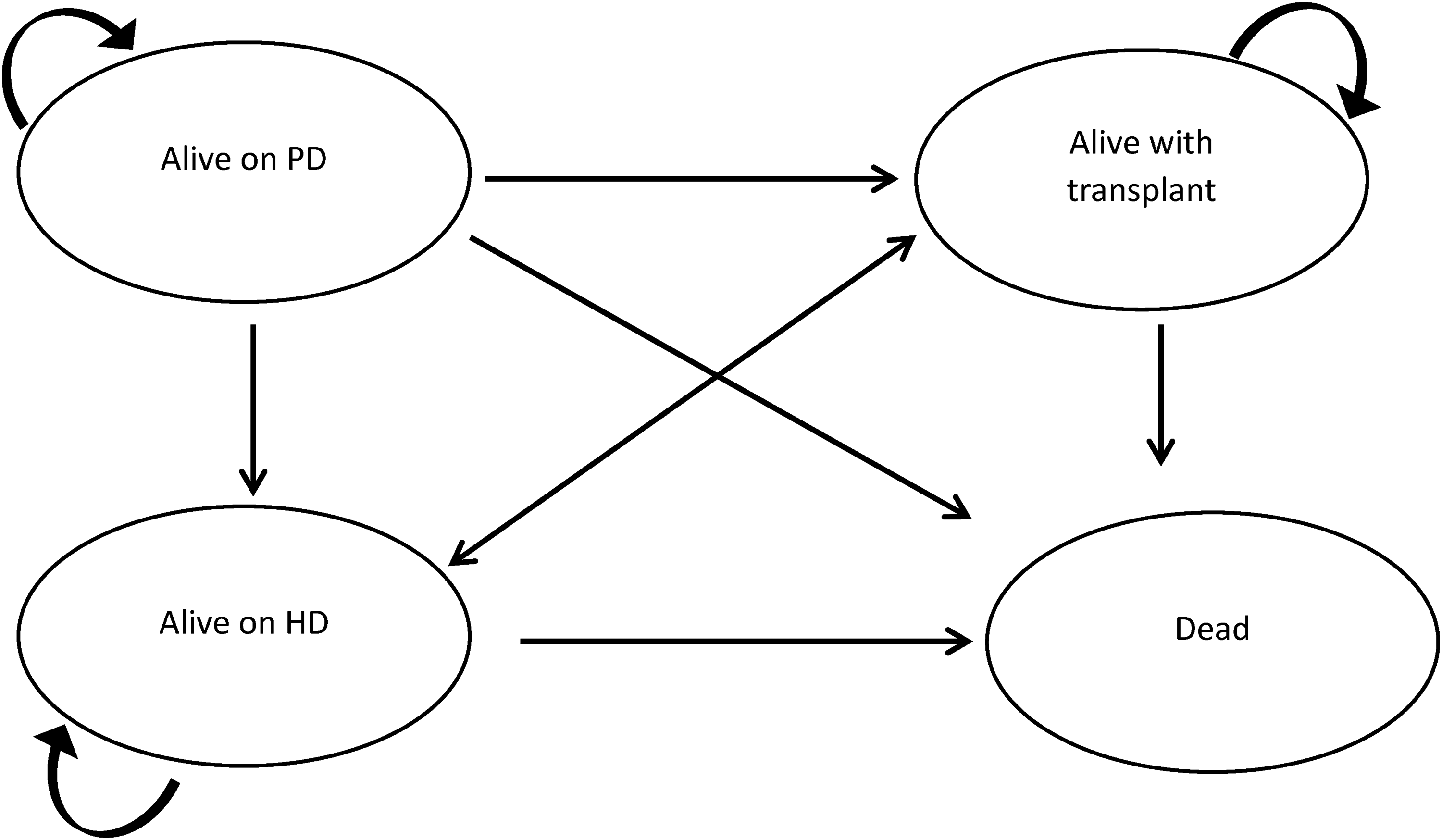
Markov model.
Data inputs and costs
To estimate the cost of the START project, we used local estimates from the health system (Table 1). Using the health system perspective, the cost of providing PD is nearly always less than HD, though there is variation in differences across studies. 10 In our base case analysis, we assumed PD to be half the cost of HD (i.e. $CAD 25,000 vs. $CAD 50,000), consistent with prior studies reporting ratios from 40% to 59%3,4,10,11; as well as local estimates for PD (including supplies and staffing). We included a cost of PD technique failure of $CAD 13,530, 12 which accounted for the cost of transition from PD to HD. From the START project, we used a base case absolute increase in PD uptake of 5.4% for ≤65 years (0.360 to 0.306), and 4.9% for >65 years (0.283 to 0.234).
Clinical and costing estimates used within the short-term analysis.
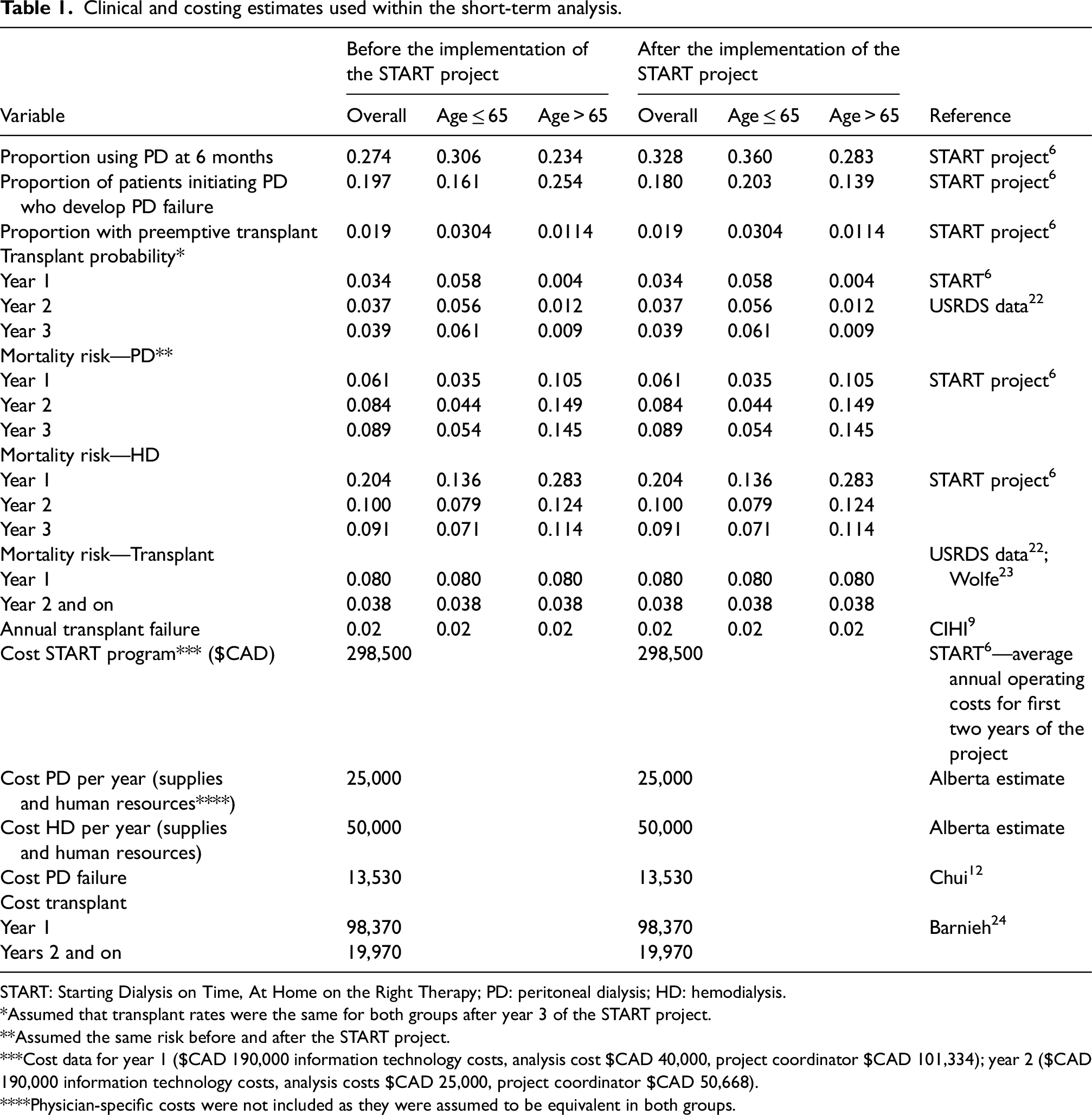
START: Starting Dialysis on Time, At Home on the Right Therapy; PD: peritoneal dialysis; HD: hemodialysis.
*Assumed that transplant rates were the same for both groups after year 3 of the START project.
**Assumed the same risk before and after the START project.
***Cost data for year 1 ($CAD 190,000 information technology costs, analysis cost $CAD 40,000, project coordinator $CAD 101,334); year 2 ($CAD 190,000 information technology costs, analysis costs $CAD 25,000, project coordinator $CAD 50,668).
****Physician-specific costs were not included as they were assumed to be equivalent in both groups.
To estimate the cost of the START project, we used actual cost data, including Information Technology infrastructure ($CAD 190,000/year) this includes the comprehensive cost of a programmer, technical support, legal costs, insurance costs, etc.; data validation and analysis ($CAD 40,000 in year 1 and $CAD 25,000 for sustainment), as well as a project coordinator salary ($CAD 101,334 in year 1 and $CAD 50,668 for sustainment). Staff and nursing costs to attend project-related meetings were provided in-kind by the programs and were not included in the costing. We averaged the two-year costs ($CAD 298,500) and modeled over a period of 10 years. Costs of additional interventions that might be introduced based on the barriers identified by the START program have not been included, as they were assumed to be part of standard clinical care. While staff nursing time specifically dedicated to the START intervention was not considered for this costing analysis, we estimated it to be 762 h from 24 staff for the project duration (24 staff to attend a full-day session and monthly 1 h staff meetings over the two years). Within the local context, nursing time spent improving care was considered part of their job within quality improvement, but this expectation may vary in other contexts. It was also estimated that it would require a 0.6 full-time equivalent across all sites of nursing time to maintain data entry for sustainability.
Sensitivity analysis
We explored the impact of uncertainty within important variables over the entire time period. In sensitivity analyses, we varied the increase in PD use (base case 5.4%; range 2.7 to 10.8%), baseline proportion using PD (base case 27%; range 13% to 54%), absolute PD failure rates (base case 20%; range 10% to 40%), differences in cost between PD and HD (base case $CAD 25,000; range $CAD 15,000 to $CAD 35,000), differences in the cost of the START program (base case $CAD 298,500; range $CAD 74,625 to $CAD 597,700) and cost of PD failure (base case $CAD 13,530; range $CAD 6765 to $CAD 27,060) were performed. The proportion of patients who were ≤65 years and >65 years was 58% and 42%, respectively. In turn, no adjustments were made in the sensitivity analysis. We also determined the break-even point, that is, the change in difference in use of PD that is required for the increased costs associated with implementing the START project to be offset by reductions in dialysis modality cost.
Results
We confirmed calibration, face, and interval validity of the model. Model validity was internally validated with primary clinical data, and results were confirmed using an Excel spreadsheet approach. We modeled the impact of the START project (compared to the status quo) on overall healthcare costs. Table 2 shows the base case analysis. We found cost savings of $CAD 1.3 million, $CAD 1.9 million, $CAD 2.4 million, and $CAD 2.7 million at 1, 3, 5, and 10 years, respectively.
The cost of caring for new kidney failure patients in Alberta with the START project, compared to without the START project.

*Assuming a dialysis program that initiates dialysis with 964 patients.
** Funds are in $CAD.
Sensitivity and scenario analysis
We explored parameter uncertainty with sensitivity analysis (Figure 2). Results were robust to plausible changes in these variables. We found two scenarios where the START project was not cost-saving. The overall cost of patients managed with the START project was noted to be higher than the status quo when the absolute use of PD at 6 months was <2.7% higher (base case was 5.4%), or when PD failure rates were increased to 40% (base case was 20%). The additional cost of the START project would be offset even if absolute PD use (within 6 months after dialysis initiation) increased by only 4.3% (break-even point).
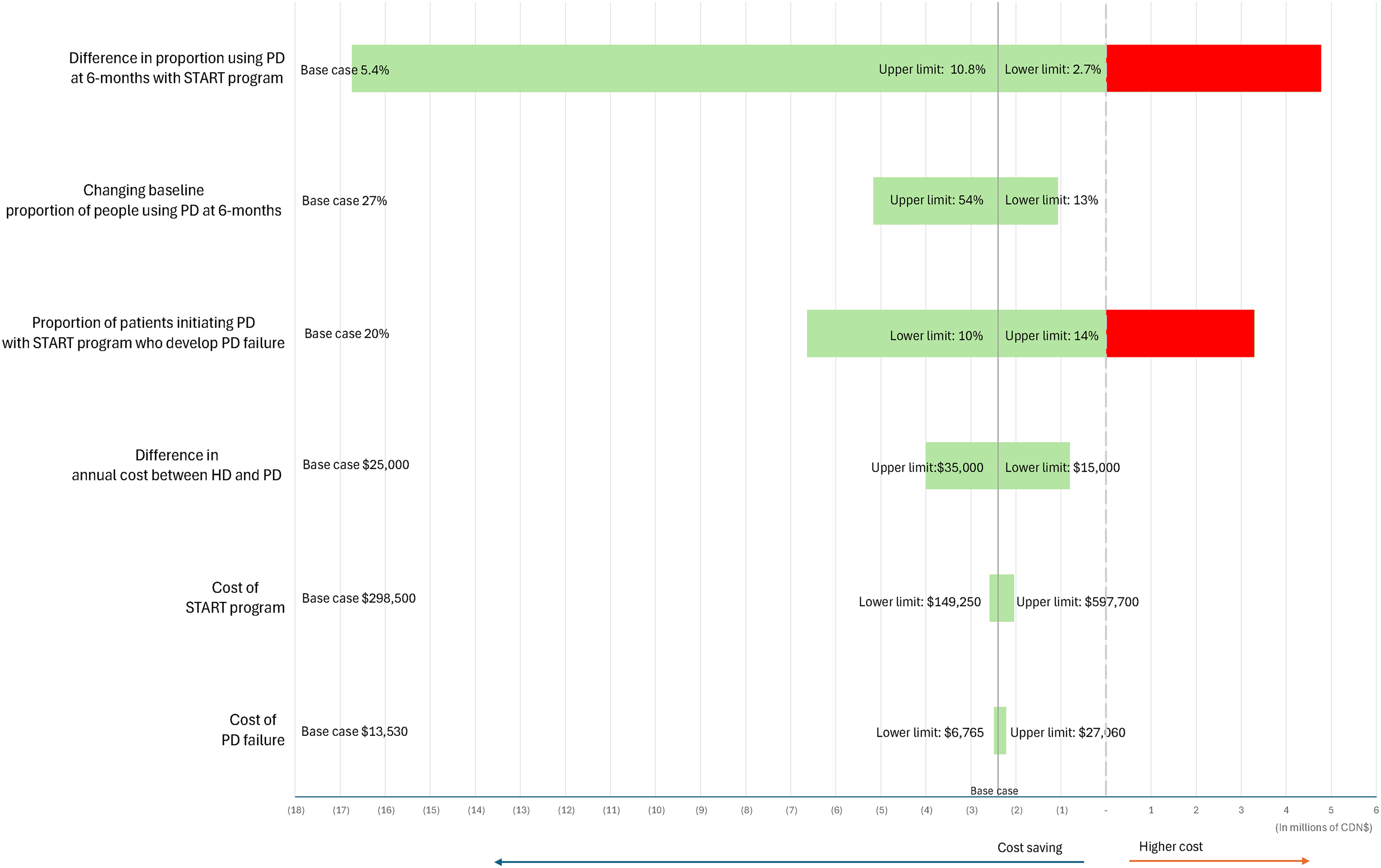
Incremental cost of the START project—one-way sensitivity analysis at 5 years.
Discussion
We found that the implementation of a quality improvement initiative to increase PD was cost-effective, resulting in substantial cost savings over time. We explored plausible changes in uncertain variables, and the findings remained. Even modest increases in PD utilization of 4.3% would lead to cost savings.
With the high cost of dialysis, there have been calls to action for strategies to improve care and reduce costs. In the US, current legislation 13 calls for a significant increase in the use of home dialysis. However, successful strategies to increase PD use are not well established. PD-first policies, such as those in Hong Kong and Thailand,14–16 have led to increased use of PD, especially in regions where PD penetration was very low. These strategies enabled broader access to dialysis in resource-limited settings. However, their relevance may be limited in areas where HD is already widely accessible. Some argue that prioritizing home-based therapies may conflict with shared decision-making principles and could unintentionally steer some patients toward conservative care by limiting access to in-center HD. 17
In terms of strategies at the patient level, a randomized trial 18 showed that education about dialysis options increased interest in home therapies, but it's unclear if this led to more PD use. A more recent, larger Canadian trial 19 across 55 clinics tested a multi-part intervention—including surveys, feedback, and education—but found no significant impact on home dialysis uptake. Similarly, in the US, a major trial 20 offering financial incentives to promote home dialysis showed no clear benefit, with home dialysis rates remaining comparable to non-incentivized sites. A 2017 scoping review 21 identified a paucity of studies to increase PD use, with the highest quality evidence emerging for patient-targeted, modality education. Emerging strategies such as incremental and assisted PD may offer cost benefits but have not been rigorously studied.
For strategies to be effective, it is important to understand local barriers to growing a PD program. Targeted, high-quality data are a key step to identifying gaps that require attention. The START project worked to streamline data and education processes to ensure that PD was offered to all eligible patients initiating dialysis. While this approach did not restrict choice for patients, it resulted in a considerable increase in PD.
While this study was conducted in a real-world setting, it does have limitations. The duration of the START project was only 18 months; hence, we were limited to the data available from this project and timeframe. It is unclear if the effectiveness of the program would be sustained or wane over time, though our work looked at cost-effectiveness in two 18-month patient cohorts, as in the START project, with costs modeled out to 3, 5, and 10 years. Future work could explore the implementation of the START program indefinitely, with varying approaches on timing and dose of the provision of data reports to maintain initial gains. The one-way sensitivity analysis may not accurately reflect uncertainty, but our results were robust to variation in almost all variables. While there may be scenarios that would not be cost-saving, it should be noted that the START intervention could be used to identify gaps. This approach of using data to identify gaps and report to sites could also be applied to other areas, such as the timely initiation of dialysis, incremental dialysis, and transplant education/processes.
While the population of patients on dialysis is relatively small, they consume a disproportionate share of healthcare resources. Implementing successful strategies to offer and increase the uptake of less costly home-based therapies is worthwhile when attempting to honor patients’ choices and reduce costs.
Footnotes
Acknowledgements
We want to thank Alberta Kidney Care for all their support and in-kind contributions.
Author contributions
MA, BM, and FA drafted the manuscript. FA performed the economic analysis. All authors reviewed and contributed to the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study is based in part on data provided by Alberta Health and Alberta Health Services. The interpretation and conclusions contained herein are those of the researchers and do not necessarily represent the views of the Government of Alberta or Alberta Health Services. Neither the Government of Alberta, nor Alberta Health, nor Alberta Health Services expresses any opinion in relation to this study. Rob Quinn is the co-inventor of the DMAR™ System and is the co-owner of the intellectual property associated with it. He has received speaking fees and attended advisory boards for Baxter Healthcare Corporation. Matthew Oliver is co-inventor of the DMAR™ System. He received speaking fees from Baxter Healthcare. He has attended advisory boards for Janssen and Amgen. Scott Klarenbach is Director of the Real-World Evidence Consortium and Alberta ug and Therapeutic Evaluation Consortium (Universities of Alberta, Calgary, and Institute of Health Economics); these entities receive funding from decision-makers and industry to conduct research. All research funding is made to the academic institution; the investigator retains full rights of academic freedom and the right to publish. This relationship is not related to the current work. Robert Pauly, Braden Manns, Marni Armstrong, and Flora Au have no relevant disclosures.
Ethical approval
Research ethics boards at the University of Alberta and the University of Calgary (REB20-1478).
Informed consent to participate
Not required by the ethics board. Waiver of consent obtained.
Informed consent to publish
Not applicable.
Trial registration
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The START Project was primarily funded through Alberta Health Services’ Kidney Health Strategic Clinical Network (SCN). Other sources of support included funds from the University of Calgary, Cumming School of Medicine (Division of Nephrology). Braden Manns was supported by a Canadian Institutes of Health Research Foundation Grant.
