Abstract
Background:
Peritoneal dialysis (PD) is actively promoted, but increasing PD utilisation is difficult. The objective of this study was to determine if the Starting dialysis on Time, At Home, on the Right Therapy (START) project was associated with an increase in the proportion of dialysis patients receiving PD within 6 months of starting therapy.
Methods:
Consecutive patients over age 18, with end-stage kidney failure, who started dialysis between 1 April 2015 and 31 March 2018 in the province of Alberta, Canada. Programmes were provided with high-quality data about the individual steps in the process of care that drive PD utilisation that were used to identify problem areas, design and implement interventions to address them, and then evaluate whether those interventions had impact. The primary outcome was the proportion of patients receiving PD within 6 months of starting dialysis. Secondary outcomes included hospitalisation, death or probability of transfer to haemodialysis (HD). Interrupted time series methodology was used to evaluate the impact of the quality improvement initiative on the primary and secondary outcomes.
Results:
A total of 1962 patients started dialysis during the study period. Twenty-seven per cent of incident patients received PD at baseline, and there was a 5.4% (95% confidence interval: 1.5–9.2) increase in the use of PD in the province immediately after implementation. There were no changes in the rates of hospitalisation, death or probability of transfer to HD after the introduction of START.
Conclusions:
The approach used in the START project was associated with an increase in the use of PD in a setting with high baseline utilisation.
Introduction
Patients with kidney failure make up a small proportion of the population, but consume a disproportionate share of healthcare resources. 1 The major driver of cost is the provision of dialysis therapy. 2 While most patients with kidney failure are treated with haemodialysis (HD), peritoneal dialysis (PD) is considered equivalent with respect to important clinical outcomes, but much less expensive to provide. 3 –6 As a consequence, many jurisdictions are actively promoting PD, but increasing PD utilisation is challenging. 2,7,8
PD first policies, modality education programmes, audit-and-feedback, providing assisted PD, transitional care units and bedside catheter placement have all been associated with increased PD utilisation. 9,10 However, most historical studies involve patient- or programme-level interventions at single centres, lack control groups, are tested in programmes with low PD penetration at baseline or involve restricting patient choice and compromising the principles of shared decision-making. More recently, large-scale interventions involving audit-and-feedback, education, academic detailing and financial incentives directed at providers and facilities have not shown benefit. 11,12
The Starting dialysis on Time, At Home, on the Right Therapy (START) project provided dialysis programmes with high-quality data about the individual steps in the process of care that drive PD utilisation to identify problem areas, design and implement interventions to address them and then evaluate whether those interventions had impact. 13 In this study, the effect of the START project on incident PD utilisation, rates of hospitalisation and death and the probability of transfer to HD in the first year of therapy was evaluated.
Materials and methods
Setting and organisation of kidney care
Alberta Health Services is a province-wide, integrated health system, responsible for delivering specialty and hospital services to a population of approximately 4.3 million people. Alberta residents have universal access to hospital care and physician services. The START project was implemented between 1 October 2016 and 31 March 2018 in all Alberta Kidney Care (AKC)-South (Calgary, Lethbridge, Medicine Hat) and AKC-North regional programmes (University of Alberta Hospital, Grey Nuns Community Hospital, Royal Alexandra Hospital, and Red Deer Regional Hospital).
Data sources
Linked administrative databases housed in the Alberta Kidney Disease Network were used to conduct the study. 14 The AKC-North and AKC-South databases captured all patients requiring kidney replacement therapy in the province, their demographic characteristics, initial dialysis modality and changes in therapy. The Alberta Health Vital Statistics database was used to capture deaths. Alberta Health and Wellness data on inpatient encounters were used to identify hospitalisations (including admission and discharge dates used to calculate the number of days in hospital) and comorbidity information in conjunction with physician billing claims (Supplementary Table S1). 15 –22
Population
Patients who commenced dialysis in Alberta, Canada, between 1 April 2015 and 31 March 2018 (18 months before and after the implementation of START).
Intervention
The START project included the development of infrastructure to support the collection of high-quality data, tied to a six-step process of dialysis modality selection (identification, assessment, determination of eligibility, offer of PD, patient choice and receipt of PD) and four main causes of exits from PD (transfer to HD, transplantation, death and other, including transfer to another programme and recovery of kidney function). Programmes conducted a structured review of all new dialysis patients at regular, multidisciplinary meetings and reported metrics related to the modality selection process and clinical outcomes. The composition of the multidisciplinary team varied from site to site but included physicians, home dialysis nurses, nurses from the kidney clinics, managers and social workers. START was supported by innovation learning collaboratives, where frontline health professionals and stakeholders met every 6 months to review results, discuss an action plan for improvement and network. Preparatory work for the START project began in March 2016 and was completed before the project went live on 1 October 2016. It involved the preparation of a business case, privacy impact assessment and signing of data sharing agreements. The data collection system and multidisciplinary meetings were implemented at all programmes in the province. The first reports were provided 6 months after the begin of data capture and the project ran for a total of 18 months. A description of the intervention has been previously published and is summarised in Figure 1. 13
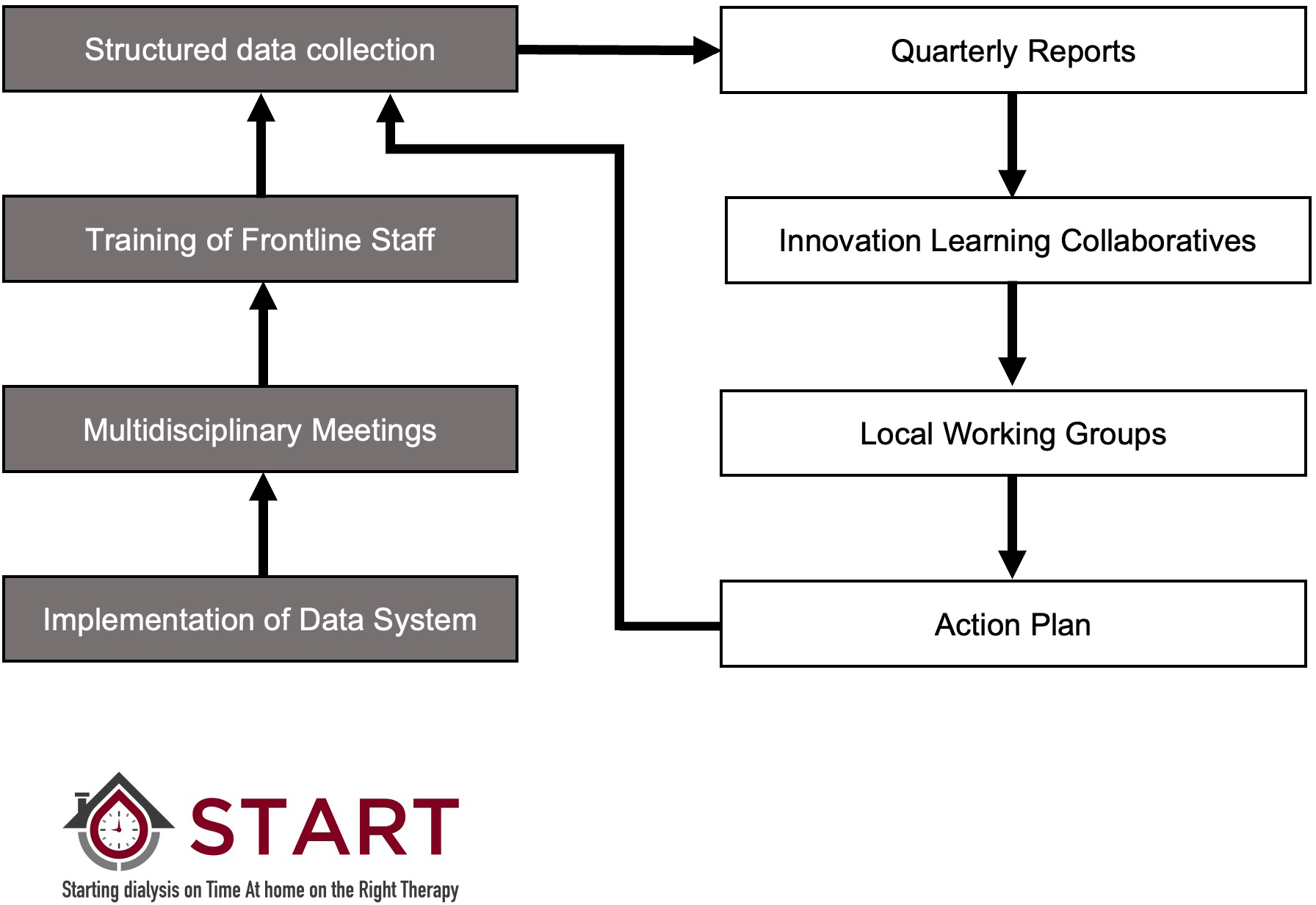
An overview of the START project. Multidisciplinary stakeholders at all sites were trained to organise and lead multidisciplinary meetings. They ensured all new dialysis starts in their programmes were identified, assessed to determine their eligibility for PD, educated about their treatment options, offered PD if they were eligible, supported to make an informed modality decision and initiated on their chosen therapy. Metrics were tied to each of these steps and entered into the data system. Participating teams were provided with site-specific reports on a quarterly basis detailing the proportion of patients who received PD within 180 days of starting dialysis overall, and by site, along with detailed metrics pertaining to the six steps in the process of modality selection and causes of loss from PD. An Innovation Learning Collaborative was implemented, consisting of three learning sessions that brought frontline staff together from across the province. Sites identified local barriers and opportunities to improve, developed action plans and implemented local interventions to address these barriers. Ongoing data collection allowed teams to determine if their interventions were effective.
Primary outcome – incident PD utilisation
The primary outcome was receipt of PD within 6 months of starting dialysis. While some patients start dialysis on their intended modality, others, particularly those who start urgently or in an unplanned fashion, may start on haemodialysis and then transition to PD.
Secondary outcomes – potential unintended consequences of increasing PD utilisation
When measures are implemented to increase the uptake of PD and expand the population of patients treated with home therapies, there is the potential for unintended consequences. The impact of the START project on hospitalisation, all-cause mortality and transfer to HD among incident PD patients was examined. Time series of hospitalisation, technique failure and mortality data were constructed for the cohort of patients who started dialysis in the 18 months prior to and after START. For hospitalisation, all individuals who started dialysis during each 1-month period were identified, and the rate of hospitalisation in the first year of therapy was calculated as the number of days in hospital per patient-year of follow-up. This analysis was performed including the initial hospitalisation for patients who started dialysis in hospital and excluding the initial hospitalisation in a sensitivity analysis. The process was repeated for the rate of all-cause mortality during the first year of therapy. Finally, the cohort was restricted to individuals starting PD therapy during each time period, and the proportion who experienced a permanent transfer to HD during the first year of PD therapy was calculated, defined as no return to PD within 90 days. 23 If individuals experienced death, transplantation, recovery or transfer out of province during the 90-day window, they were not counted as a permanent transfer to HD. 23
Statistical analysis
We used descriptive statistics to compare the baseline patient demographics, comorbidities and treatment-related data between patients starting dialysis before and after the implementation of START.
Interrupted time series analysis with segmented linear regression was used to evaluate the impact of the intervention. 24 The time period was divided into 1-month segments, and the proportions of patients starting dialysis during that period who received PD within 6 months of initiation were calculated. This process was repeated for 18, 1-month segments, before and after the introduction of the START project. One-month time periods were chosen to maximise the number of observations per period and allow for examination of seasonality. There were complete data for all 36 time periods. The unit of analysis was the aggregated proportion of patients receiving PD within 6 months, province-wide, during the time period of interest. This process was repeated at the level of the AKC-South and AKC-North programmes, individually. No control group was used, as the intervention was applied at the level of the entire province and impacted all dialysis programmes.
Plots of the time series were constructed to visualise the data points before and after the intervention, along with trend lines and counterfactuals (projected results in the absence of the intervention). The time series was screened for the presence of autocorrelation and seasonality. The full segmented linear regression model included an intercept, a term for the trend (slope) before and after the introduction of the intervention, as well as the level change immediately after the START project began. Insignificant terms were then removed to establish the most parsimonious final model. 24 This approach was repeated for the secondary outcomes of hospitalisation, technique failure and death.
As a sensitivity analysis, we used 2-month and 3-month time segments as well as a log-transformed model. All statistical analyses were conducted using R version 4.2.0 (R-project.org).
Ethical considerations and conduct of study
We followed the SQUIRE guidelines for the reporting of quality improvement studies, 25 and the protocol was approved by the research ethics boards at the University of Alberta and the University of Calgary, with waivers of patient consent.
Results
A total of 1962 patients started dialysis during the study period; 964 before the implementation of START and 998 after the implementation of the project. The median age was 63 years; 37% were female; and 51% started dialysis in hospital. Baseline patient characteristics were similar before and after the introduction of START (Table 1), and the median duration of follow-up from dialysis initiation was 12 months (patients were followed for a maximum of one year). A total of 15% died, 3% underwent kidney transplant and 2% left the province or were lost to follow-up.
Baseline characteristics of patients starting dialysis before and after the implementation of the START project.
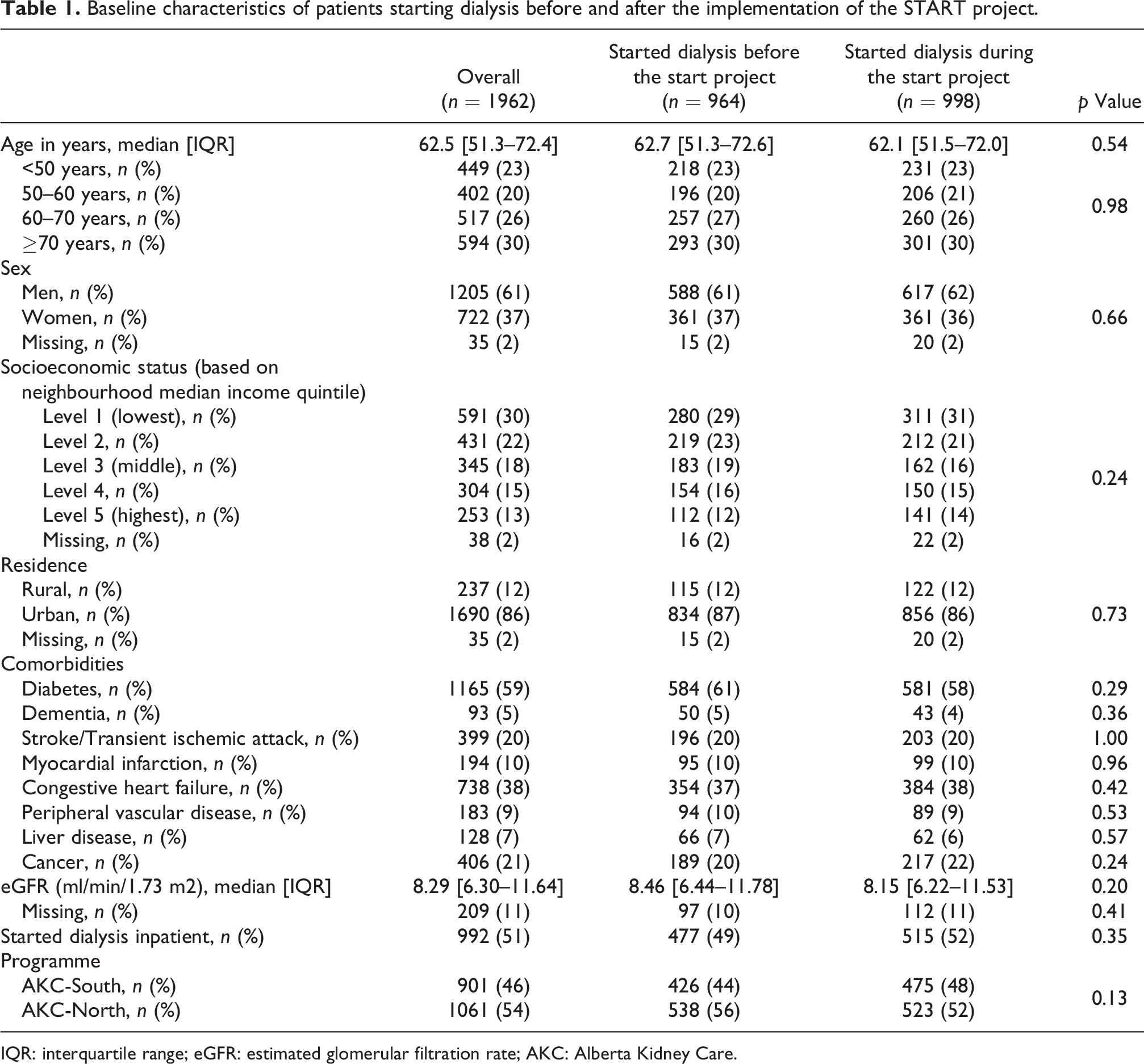
IQR: interquartile range; eGFR: estimated glomerular filtration rate; AKC: Alberta Kidney Care.
The proportion of patients who received PD within 6 months of initiating kidney replacement therapy is shown in Figure 2. There was no evidence of autocorrelation or seasonality in the data. Twenty-seven per cent of patients received PD prior to START compared to 33% after. Overall, there was no significant trend (slope) for PD utilisation before or after START, so these variables were removed from the final model. However, the level change model showed that immediately after the implementation of START, the use of PD increased province-wide by 5.4% (95% confidence interval (CI): 1.5%–9.2%).
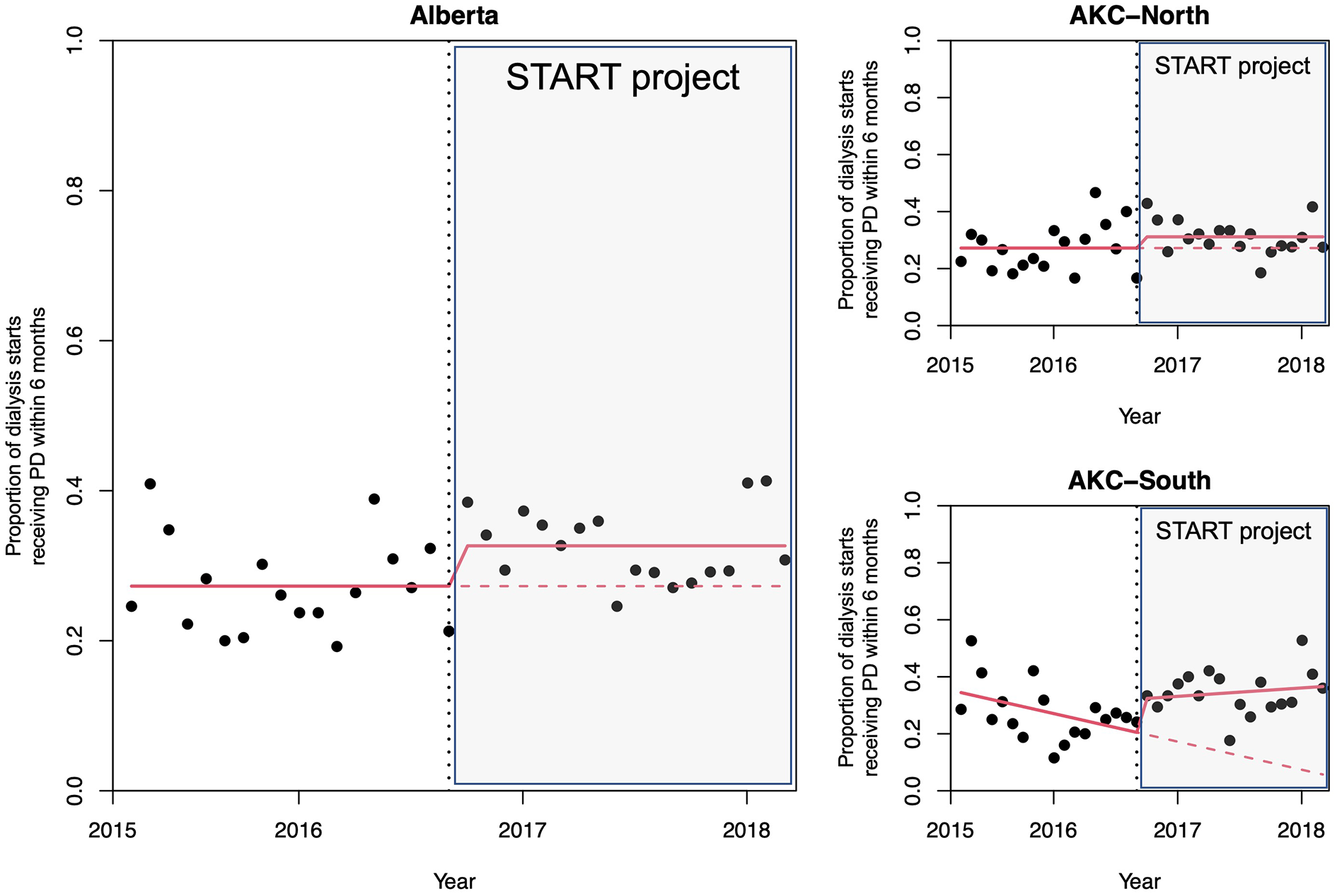
The proportion of patients receiving PD within 6 months of starting therapy. The proportion of patients receiving PD within 6 months of starting dialysis therapy is calculated for each 1-month interval before and after the introduction of the START project. Each dot represents the average of all patients who started dialysis during that month. There was a stepwise increase in PD utilisation of 5.4% that occurred immediately after implementation of START, overall. In AKC-South, there was a decreasing trend in PD utilisation before START and a 11.9% stepwise increase after implementation. In AKC-North, START had no significant impact on PD utilisation.
When data were stratified by region, there was a significant 11.9% level increase (95% CI: 0.9% to 23.0%) in PD utilisation in AKC-South immediately after implementation. Prior to START, there was a significant, decreasing trend in PD of −9.9% per year (95% CI: −19.3% to −0.4%) and a stable trend during the intervention (3%; 95% CI: −6.5% to 12.4%). In AKC-North, the level change model showed a non-significant increase in PD utilisation immediately after implementation (4%; 95% CI: −0.9% to 8.8%), and there was no significant trend in PD utilisation for before or after START. Sensitivity analyses using 2-month and 3-month time segments as well as the log transformed model showed similar results.
Individuals initiating dialysis had a rate of 33 days in hospital and 0.2 deaths per patient-year of follow-up, overall. If the initial hospitalisation was excluded, the rate was 17 days per patient-year of follow-up. There were no differences in the rates of hospitalisation or death after the implementation of START overall or when rates were examined separately for AKC-North and AKC-South (Figure 3). A total of 19% of patients initiating PD experienced a transfer to HD during the first year of therapy. The risk of transfer to HD was not different after the START project. Results did not meaningfully change for any of the secondary outcomes if a level change only model was used.
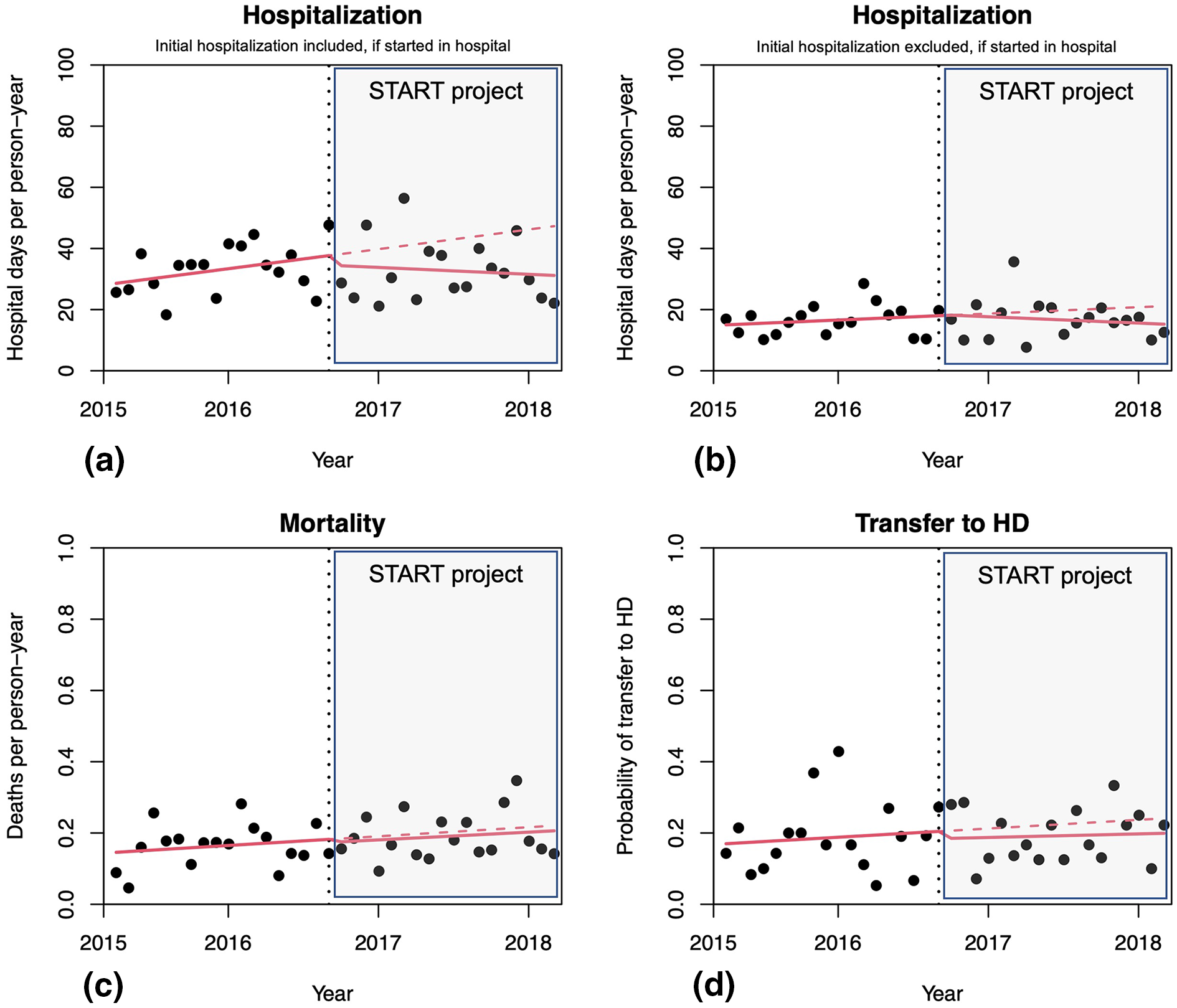
The rates of hospitalisation, mortality and transfer to HD. Rates of hospitalisation (days per person-year of follow-up), including (panel (a)) and excluding (panel (b)) the initial hospitalisation in patients who were hospitalised when they initiated dialysis. START had no impact on rates of hospitalisation. Similarly, rates of mortality (panel (c)) and transfer to HD (panel (d)) were not different before or after the START project.
Discussion
The START project provided dialysis programmes with high-quality data about the individual steps in the process of care that drive PD utilisation to identify problem areas, design and implement interventions to address them and then evaluate whether those interventions had impact. After the implementation of the programme, there was a 5.4% increase in the use of PD in the province, with a more pronounced increase in AKC-South, as compared to AKC-North. Despite the increase in PD utilisation, there were no changes in the rates of hospitalisation, death or probability of transfer to HD in the first year of therapy. Due to the short duration of the project, the longer-term impact of the programme was not evaluated.
Several interventions that have been associated with an increase in PD utilisation. PD first policies, such as those implemented in Hong Kong and Thailand, have been associated with growth in the numbers of patients treated with PD. 26 –28 In those jurisdictions, the provision of PD allowed many individuals who would not otherwise have received dialysis to be treated and were applied to populations with very low PD penetration at baseline. PD first policies ensured they were able to treat the most patients with a limited pool of resources, but these observations are of limited generalisability to jurisdictions where haemodialysis is already widely available. There is additional concern that home-first policies run counter to the spirit of shared decision-making and involve restricting access to in-centre HD that could have other, unintended consequences, such as increasing the numbers of patients who may unwillingly choose conservative care. 29 In terms of patient-level interventions, a randomised controlled trial of a modality education intervention increased the proportion of patients who intended to do home dialysis, but it was not clear if that translated into an actual increase in PD utilisation. 30 A more recent, large-scale, cluster-randomised trial at 55 chronic kidney disease clinics in Canada showed no benefit of a four-component intervention to increase home dialysis utilisation that involved a phone survey, audit-and-feedback, education about home dialysis and academic detailing. 11 There have also been recent studies of provider- and centre-level interventions. In the United States, a large randomised trial of financial incentives (End-Stage Renal Disease Treatment Choice (ETC) model) for facilities and clinicians to increase the use of home therapies showed no benefit. 12 A follow-up study showed that ETC sites had similar rates of home dialysis utilisation to non-ETC sites, although the rate of increase may have been slightly higher. 31
The existing literature highlights the challenges faced when trying to increase PD utilisation at scale and suggests that it may require a different approach because of its complex, multi-dimensional nature. It is important to simultaneously optimise the numbers of incident patients treated with PD, maximise the amount of time that they spend on therapy and minimise loss. 13 Within each of these areas, there are a number of potential issues that may need to be addressed, and the challenges can vary substantially from programme to programme, requiring solutions that are specific to the local context. For example, if the proportion of patients choosing PD is already high, there will be little benefit of revamping modality education, even if prior studies have suggested benefit. 30 If a programme is struggling to get patients who have chosen PD on therapy in a reasonable timeframe, addressing ways to get PD catheters placed more quickly and shortening the wait-time for PD training may be worthwhile. Understanding local barriers to growing PD is, therefore, important for the design and implementation of targeted interventions that are most likely to lead to improvement. Applying a generic intervention without first documenting if there is a problem, what that problem is and whether or not the intervention is likely to fix it may not be the best use of limited resources. In addition, performance can change over time and the drivers of PD utilisation are dynamic, so ongoing monitoring is required. This may also explain why prior attempts at audit-and-feedback have been unsuccessful – simply providing a high-level performance metric, without the ability to explain what is driving local performance may not be enough to effect change when dealing with complex systems.
The START project had three components: infrastructure to support the collection of high-quality data collection tied to a six-step process of modality selection; structured review of all new dialysis patients (including regular multidisciplinary meetings to review new starts) and the reporting of metrics tied to modality selection and outcomes; and the use of an innovation learning collaborative approach, where frontline health professionals and stakeholders met regularly to review results, discuss an action plan for improvement and network. 13 The implementation of this programme was expected to have immediate and longer-term benefits. A more structured and transparent approach to the process of patient identification, assessment for PD eligibility and ensuring that individuals had timely access placement, underwent training and were started on therapy likely led to immediate returns that were observable during the 18-month time period that the START project was active. Indeed, incident PD utilisation increased by more than 5% immediately after the implementation of START overall and by almost 12% in AKC-South. In fact, only one out of seven centres saw a decrease in PD utilisation during the START programme. 32 While PD utilisation increased overall, there were clearly regional differences in the ability to address the issues that were identified during START as well as the willingness to address them. There was no meaningful mechanism for holding programmes to account for their performance and no consequences when targets were not met. This may have partially explained the variability in results by region, and while START was successful, they may underestimate the potential impact in an environment with increased accountability for performance.
The results of the START project have important clinical and policy implications. First, improvements in PD utilisation were demonstrated at scale, rather than in a single centre, in a province with relatively high baseline use of PD, and with rigorous methodology. Second, the improvements were realised without restricting patient-choice. Individuals were identified, educated about their treatment options, and then allowed to choose the dialysis therapy that was best for them, in line with the principles of shared decision-making. Finally, the programme is likely to be cost-effective. While the population of patients with end-stage kidney disease is relatively small, they consume a disproportionate share of healthcare resources, and the major driver of this cost is the provision of dialysis therapy. 5,33 If the cost savings associated with PD relative to HD is conservatively estimated at $39,000 per patient, 5 and the median time on PD in Alberta was 24 months per year when all forms of loss from therapy are accounted for (transfer to HD, death, recovery of kidney function, transplantation and transfer out of the programme), 23 the implementation of the START project would lead to savings of approximately $78,000 for every patient who received PD and would otherwise have been treated with HD. Even modest increases in PD utilisation could lead to substantial cost savings and make this type of approach cost-effective.
Our study has limitations. This quality improvement project was conducted in a real-world setting. The nature of the START project and the practicalities of implementing it, as well as the small number of programmes that provide dialysis care, made a cluster-randomised trial or stepped-wedge design challenging. While high-quality, granular data were collected during the START project, they were not available prior to its introduction or after it ended, so the evaluation of the project was necessarily based on administrative data sources. In addition, the approach taken requires buy-in from participating centres, and while PD utilisation increased across the province, there were regional differences in performance. Participating programmes must be motivated to improve and able to use the information provided to fuel improvement initiatives. Finally, the duration of the START project was only 18 months. This likely is not sufficient to thoroughly evaluate its impact and observe any delayed effects of providing information about local barriers to PD utilisation that might take time to address.
In conclusion, the START project was conceived to provide dialysis programmes with high-quality data about the individual steps in the process of care that drive PD utilisation in an attempt to identify problem areas, design and implement interventions to address them and then evaluate whether those interventions had impact. It was associated with an immediate increase in incident PD utilisation in a large Canadian province, but whether further growth in PD would have been observed over a longer time horizon is unknown.
Supplemental material
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231225013 - The impact of the Starting dialysis on Time, At home on the Right Therapy (START) project on the use of peritoneal dialysis
Supplemental Material, sj-docx-1-ptd-10.1177_08968608231225013 for The impact of the Starting dialysis on Time, At home on the Right Therapy (START) project on the use of peritoneal dialysis by Robert R Quinn, Matthew J Oliver, Alix Clarke, Farah Mohamed, Scott W Klarenbach, Braden J Manns, Danielle E Fox, Nairne Scott-Douglas, Louise Morrin, Anita Kozinski, Tracy Schwartz and Robert Pauly in Peritoneal Dialysis International
Footnotes
Acknowledgement
None.
Author contributions
All authors fulfill criteria for authorship in that they (i) made a substantial contribution to the concept or design of the work; or acquisition, analysis or interpretation of data; (ii) drafted the article or revised it critically for important intellectual content; and (iii) approved the version to be published. All authors meet all three criteria of authorship listed above.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study is based in part on data provided by Alberta Health and Alberta Health Services. The interpretation and conclusions contained herein are those of the researchers and do not necessarily represent the views of the Government of Alberta or Alberta Health Services. Neither the Government of Alberta, nor Alberta Health or Alberta Health Services express any opinion in relation to this study. Dr Rob Quinn is the co-inventor of the DMAR™ System and is the co-owner of the intellectual property associated with it. He has received speaking fees and attended advisory boards for Baxter Healthcare Corporation. Dr Matthew Oliver is co-inventor of the DMAR™ System. He received speaking fees from Baxter Healthcare. He has attended advisory boards for Janssen and Amgen. Dr Scott Klarenbach is Director of the Real World Evidence Consortium, and Alberta Drug and Therapeutic Evaluation Consortium (Universities of Alberta, Calgary, and Institute of Health Economics); these entities receive funding from decision-makers and industry to conduct research. All research funding is made to the academic institution; investigator retains full rights of academic freedom and right to publish. This relationship is not related to the current work. He was supported by the Kidney Health Research Chair and the Division of Nephrology at the University of Alberta. Dr Nairne Scott-Douglas has received speaking fees and attended advisory boards for Bayer, Boehringer Ingelheim and Astra Zeneca and attended Advisory Boards Janssen, Amgen and Otsuka. Dr Robert Pauly, Dr Braden Manns, Danielle Fox, Anita Kozinski, Farah Mohamed, Louise Morrin, Tracy Schwartz and Alix Clarke have no relevant disclosures.
Ethical approval
Research ethics boards at the University of Alberta and the University of Calgary.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the START project was provided by Alberta Health Services. There was no funding provided for this study.
Informed consent to participate
Not required by ethics board. Waiver of consent obtained.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
