Abstract
Infection-related complications remain the most significant cause for morbidity and technique failure in infants, children and adolescents who receive maintenance peritoneal dialysis (PD). The 2024 update of the Clinical Practice Guideline for the Prevention and Management of Peritoneal Dialysis Associated Infection in Children builds upon previous such guidelines published in 2000 and 2012 and provides comprehensive treatment guidance as recommended by an international group of pediatric PD experts based upon a review of published literature and pediatric PD registry data. The workgroup prioritized updating key clinical issues contained in the 2012 guidelines, in addition to addressing additional questions developed using the PICO format. A variety of new guideline statements, highlighted by those pertaining to antibiotic therapy of peritonitis as a result of the evolution of antibiotic susceptibilities, antibiotic stewardship and clinical registry data, as well as new clinical benchmarks, are included. Recommendations for future research designed to fill important knowledge gaps are also provided.
This is a visual representation of the abstract.
What's New in the 2024 Update of the ISPD Pediatric Peritonitis Guidelines?
Revised, updated training recommendations (page 6) Revised, updated recommendations for early exit-site care (page 17) Revised recommendations for chronic exit-site care (page 18) Revised, updated recommendations regarding adjunctive prophylactic antimicrobial therapy (page 29) Revised recommendations for the diagnosis of peritonitis (page 33) Revised, updated recommendations for modification of therapy for gram-positive peritonitis (page 41) Revised, updated recommendations for modification of therapy for gram-negative peritonitis (page 46) Revised recommendations for treatment of culture-negative peritonitis (page 54) Revised recommendations for treatment of fungal peritonitis (page 57) New recommendations for treatment of Mycobacterial peritonitis (page 60) Revised recommendations regarding relapsing, recurrent and repeat peritonitis (page 62) Revised recommendations regarding adjunctive therapy of peritonitis (page 66) Revised criteria for diagnosis of PD catheter related infection (page 74) Revised recommendations for treatment of a PD catheter related infection (page 79) Revised, updated recommendations regarding monitoring of infection rates and risk factor assessment, and clinical benchmarks for rates of peritonitis overall, culture-negative peritonitis and percentage of PD catheter insertions followed by peritonitis within 30 days of catheter insertion (page 84)
Introduction
Infection-related complications remain the most significant cause of morbidity and peritoneal dialysis (PD) discontinuation in infants, children and adolescents who receive maintenance peritoneal dialysis (PD). The clinical, emotional and economic impact associated with these infections prompted publication of the initial, largely opinion based recommendations pertaining to the prevention and treatment of catheter-related infections and peritonitis in pediatric patients receiving PD in 2000. 1 Subsequent to that work, the International Pediatric Peritonitis Registry (IPPR) was established to collect infection related data and to evaluate the influence of implementation of the guidelines on the pediatric PD population. The information made available from the IPPR served as the foundation for the second set of guidelines published in 2012. 2 Over the past decade, further data collection by the International Pediatric Peritoneal Dialysis Network (IPPN) and the Standardizing Care to Improve Outcomes in Pediatric End-stage Kidney Disease (SCOPE) quality improvement collaborative, complemented by a variety of publications, has created a new evidence base that allowed the creation of this update to the 2012 consensus guidelines.
Methods
As was the case with the two prior sets of guidelines, the workgroup consisted of an international team of experts comprised of pediatric nephrologists, pediatric infectious diseases specialists, a pediatric dialysis nurse, and methodology experts. Pediatric pharmacologists were added to the workgroup for this set of guidelines. Panel discussions took place face-to-face, during conference calls, and by email.
The current set of guidelines was developed using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach for evidence assessment in collaboration with the Evidence Based Practice and Implementation Center (EPIC) at the University of Kansas Medical Center. The workgroup prioritized updating questions addressed in the 2012 guidelines and developed a list of additional questions for good practice and implementation using the PICO format (Population, Intervention, Comparison, Outcomes). 3 In addition to peritonitis rates, workgroup members also considered other outcomes such as PD catheter exit-site/tunnel infections, resolution of clinical symptoms, and transfer to hemodialysis in the process. Details around both sets of questions and corresponding outcomes can be found in Supplemental Tables S1 and S2.
Literature search and article selection
Ovid Medline, Embase, and Cochrane databases were searched for relevant articles published through November 2021. A combination of keywords and subject headings were used for a more comprehensive search. As an additional step, a reference list of existing peritonitis guidelines and resources suggested by workgroup members was reviewed.2,4–6 Supplemental Table S3 provides the search strings we used across databases.
All titles and abstracts of the references identified by the search strategy were screened by two independent workgroup members using the online platform Rayyan. 7 Any article that was included by at least one reviewer was included for full-text screening. A similar process was carried out for the full-text screening of articles using the software Covidence. 8 Disagreements around inclusion or exclusion of articles at the full-text screening stage were resolved through workgroup meeting discussions.
Any studies that addressed at least one of the 54 prioritized PICO questions were included. This included randomized studies, non-randomized studies with a comparison (e.g., cohort, case-control, cross-sectional), and non-randomized studies without a comparison (i.e., case series). We excluded non-English studies, studies describing only the adult population, and case reports. Supplemental Figure S1 provides detailed information about inclusion and exclusions, using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram. 9 Supplemental Table S4 provides information around the included studies.
Data collection and analysis
Two independent workgroup members extracted information in duplicate from the eligible studies using a standardized data extraction form. The two extractions were collated into one and any disagreements were resolved through discussions. For each study, general characteristics (e.g., publication year, country, study design), details about the population (age, gender, duration of PD treatment), details about the intervention (e.g., dose, route of administration), and details around outcomes (e.g., frequency of peritonitis episodes, change in dialysis modality) were extracted. When studies reported a mix of children and adults, only the results from children were included. When studies used more than one intervention and reported on the combined results of all interventions, the results of the intervention were summarized only if more than 80% of the study participants received that intervention. Since performing a quantitative synthesis of the results was not possible with the heterogeneity of the eligible studies, the study results were synthesized narratively.
Risk of bias and certainty of evidence
We used the Risk of Bias in Nonrandomized Studies of Interventions (ROBINS-I) tools 10 to appraise the risk of bias in prevalence studies and in case series. 11 Supplemental Tables S5 and S6 provide a summary of those judgments. The GRADE framework was used to assess the overall certainty of the evidence by evaluating the body of evidence for each outcome in the following domains: risk of bias, imprecision, inconsistency, indirectness, and publication bias.12,13 The GRADE summary of findings tables were developed using the GRADEpro Guideline Development Tool. 14 Supplemental Tables S7–S47 provide details around those summaries and the certainty of evidence judgments.
Unpublished data
For questions that have no or sparse published data to inform them, the guideline workgroup communicated with the leadership of the IPPN registry and the SCOPE Collaborative requesting analyses be completed to answer these questions. When feasible, published or unpublished evidence from IPPN and SCOPE was utilized to inform questions.
Evidence to recommendations
The workgroup used the Evidence-to-Decision (EtD) framework for developing recommendations using GRADEpro software. 14 This included considering the certainty of evidence and the balance between desirable and undesirable effects, in addition to other domains (e.g., feasibility, resource use, acceptability) for developing recommendations. Supplemental Tables S48–S85 provide details around the considerations and judgments for each recommendation.
These recommendations are based on contemporary evidence when such evidence is available. As per GRADE methodology, within each recommendation, the strength of recommendation is indicated as Level 1 (We recommend), Level 2 (We suggest) or Not Graded, and the quality of the supporting evidence is shown as A (high quality), B (moderate quality), C (low quality) or D (very low quality). The recommendations are not meant to be implemented in every situation indiscriminately. Each PD unit should examine its own pattern of infection, causative organisms and antibiotic susceptibilities and adapt the protocols to local conditions as necessary. Supplemental Figure S2 provides the suggested interpretation of strong and weak recommendations for the population, health care workers, and policy makers.
Statements that are not supported by a systematic review of the pediatric literature (often times as a result of the absence of pediatric literature), there is no supporting pediatric registry data, they are opinion based or the recommendation might have been extrapolated from pediatric literature published subsequent to November, 2021, are based on the adult experience without a systematic review of the supporting evidence or are based on a non-systematic review of other pertinent literature, were Not Graded (We suggest).
Finally, when possible, the workgroup attempted to harmonize the recommendations for children with the recently published adult recommendations. However, where the adult and pediatric recommendations differed, the adult recommendation has been included in the rationale which supports the pediatric recommendation following the specific guideline statement. In addition, supporting information (e.g., reporting of peritonitis rates, definitions, stability of antimicrobials) included in recent publications pertaining to adult PD patients that is equally applicable to the pediatric PD population has been included in this publication.
Guideline 1 – Training
1.1 PD training should be performed using a formalized teaching program with clear content, objectives, and consideration of the previously published ISPD syllabus for teaching peritoneal dialysis to patients and their caregivers (Not Graded).
1.2. We suggest that the training session length and total duration of training be tailored to the needs of the child on PD and their caregivers, with an emphasis on providing longer total training duration rather than shorter, whenever possible (2D).
1.3. We suggest that a formal assessment, including post-training written and demonstration competency testing, be performed as part of home PD training for children on PD and/or their caregivers (2D).
1.4. We suggest that a home visit be performed as part of the training process for children on PD (2D).
1.5. We suggest regular retraining/reassessment of all children on PD and/or their caregivers (2D).
Rationale
When training patients and caregivers to perform home PD, current guidelines suggest the use of a formalized training program that has clear objectives and criteria, and incorporates adult learning principles (Table 1).16,17 Health literacy assessments
18
and learning style assessments
19
are commonly used to tailor patient and/or caregiver education to the learning style, literacy and health literacy level of each learner. The ISPD published a syllabus for teaching PD to patients and caregivers in 2016, based on theories and principles of adult education, which is currently being updated
Peritoneal dialysis training contents.

The ISPD training syllabus suggests that “after completion of the content contained in the PD training syllabus, the PD nurse will have provided education to a patient and/or caregiver such that the patient/caregiver has the required knowledge, skills and abilities to perform PD at home safely and effectively.” 17 While this training syllabus was intended for the adult learner and the adult PD patient population, it can be modified to include pediatric specific content that the learner would need to know to be successful in providing PD care to children on PD.
The training should be performed by an experienced PD nurse, ideally with pediatric training, and should include content related to infection prevention such as hand hygiene, aseptic technique, exit-site care and appropriate treatment for contamination.16,17
Training can take place in the clinic, the patient's home, in the hospital or any location that can accommodate all of the components of PD training. There is no evidence to suggest that any location is superior to the others. 17
In a retrospective pediatric study, patients on home automated PD (APD) had their initial home visit conducted by a trained dialysis nurse. The authors suggested that home visits be performed soon after initiation of home PD and that follow-up home visits should occur every 12 months, after episodes of peritonitis, or when other concerns arise. 25 The recommendation was made that the following should be recorded as part of the home visit assessment: type of surrounding community, type of dwelling, presence of a working dialysis machine, thermometer, smoke detector, and fire extinguisher, assessment of the room where dialysis is performed each night, assessment of the space where handwashing occurs, assessment of where disposal of fluids occurs, location of storage of dialysis supplies, and a review of medications and their proper storage. A review of the findings at each home visit should then be discussed with the interdisciplinary team and recommendations made based on the findings. 25
In terms of peritonitis risk, an international survey from 2001 showed no correlation between home visits and peritonitis rates, whereas a survey from 2016 in one country demonstrated a significant correlation between home visits and peritonitis rates. 26 A recent international survey from the IPPN registry showed that there was no significant difference in the rate of peritonitis between those pediatric patients who received a home visit and those who did not, after adjusting for country gross domestic product (GDP) and center size. 22 The SCOPE Collaborative also evaluated whether compliance with a home visit was associated with risk for infection at the patient level. This assessment revealed that there was no significant difference in median days to first peritonitis or first episode of peritonitis within 3 months of PD training completion between those patients who did and did not have a home visit. 21
As part of training and home visits, an assessment should be performed regarding pets in the home. Education of children on PD and their caregivers should include risks of peritonitis when the child on PD and/or the PD equipment has direct contact with animals. The ISPD peritonitis guideline recommendations: 2022 update on prevention and treatment describes specific infectious complications and should be referred to during training, home visits and when a diagnosis of peritonitis with unusual organisms suspicious of zoonoses occurs. 27 These guidelines suggest that pets not be allowed in the room where PD exchanges takes place and where dialysis tubing, equipment and the machine are stored. 27
Data from the SCOPE Collaborative has demonstrated that provider compliance with the follow-up bundle and a regular (e.g., monthly during clinic visit) review of catheter care practice recommendations, as well as demonstration of competency with these procedures every 6 months, was associated with a significantly lower risk of peritonitis at the patient level. 24 Compliance with the individual elements of the follow-up bundle including review of hand washing, exit-site care and aseptic technique was significantly lower in those patients with peritonitis compared with the no peritonitis group.
In contrast, a recent international survey from the IPPN found no significant difference in peritonitis rates between those centers who conducted regular periodic training reassessments and those who provided a training reassessment post peritonitis only, after adjusting for GDP and center size. However, these results may be confounded by the small number of centers in the survey who regularly conducted retraining sessions. 22
Research Recommendations
● Future studies should clearly define duration of PD training and evaluate the relationship between training duration and cost, patient and caregiver satisfaction, and peritonitis rate.
● Future studies should attempt to determine the form of post-training assessment which is most informative regarding future performance of PD and the risk of peritonitis.
● Future research should be conducted to address the impact of the timing and content of retraining on peritonitis rate. Studies looking at the cost and potential cost savings associated with retraining are also needed.
Guideline 2 – Catheter Type and Placement
2.1 We suggest the use of a double-cuff PD catheter with a downward or lateral exit-site orientation (2D).
2.2 We suggest that the technique used for PD catheter placement be based on patient suitability and expertise of the operator (2D).
2.3 We suggest that perioperative intravenous antibiotics be provided prior to insertion of a PD catheter (2C).
Rationale
The theoretical benefit of a PD catheter with two cuffs in the subcutaneous tunnel, rather than one, is the additive barrier against migration of organisms from the skin to the peritoneal cavity. In fact, early data from the North American Pediatric Renal Trials and Collaborative Studies (NAPRTCS) revealed a higher risk for peritonitis in children with a PD catheter with a single cuff rather than 2.
32
Those early data influenced clinical practices, and data from the SCOPE Collaborative and the IPPN reveal that two-cuffed catheters are now placed in the majority of children on maintenance PD.30,31 However, observational studies in children on PD have failed to show a consistent benefit of a second cuff in terms of infection prevention. Data from a single center retrospective review and several analyses of SCOPE data have shown no difference in peritonitis rates and/or rate of exit-site infection between catheters with one or two cuffs.24,30,33–35 A single center study of children on PD published by Macchini et al. noted higher exit-site infection rates associated with catheters with two cuffs, while data from the IPPR Registry demonstrated that the presence of a single cuff catheter was associated with a higher rate of relapsing peritonitis and gram negative peritonitis.36–38 Data from the IPPN shows no difference in the rates of catheter revision or obstruction between single and double cuffed catheters.
31
One potential complication of a double cuff catheter is the risk of cuff extrusion, with extrusion rates between 2 and 10% reported
In adults on PD, a small, potentially underpowered randomized controlled trial showed no difference in peritonitis rates with one versus two cuffed catheters, while a large retrospective study did reveal a lower peritonitis rate associated with two cuffs.43,44 However, the impact seen in the latter study may have been era related, with no difference in peritonitis rates noted in the era after the use of prophylactic exit-site and/or intranasal antibiotics had been widely adopted. 44 In fact, previous guidelines for adults on PD emphasized that placement of a catheter with two cuffs may be particularly beneficial when prophylactic antibiotics are not used. 41 However guidelines published in 2023 suggest that no catheter design, including the number of cuffs, has been shown to be superior to another in terms of reducing the risk for catheter-related infections. 45
Early data from the NAPRTCS registry demonstrated a higher risk for exit-site infection and/or peritonitis with an upward, rather than a lateral or downward orientation of the catheter exit site. 32 Subsequently, data from the IPPN and SCOPE revealed that the majority of catheters placed in children have a downward or lateral orientation of the exit site.30,31 Data from the SCOPE collaborative support the earlier findings from NAPRTCS, with multivariable analyses revealing a higher risk for peritonitis with an upward orientation of the exit site (rate ratio, 4.2; 95% confidence interval, 1.49 to 11.89). 37 A separate analysis of SCOPE data did not, however, find an association between exit-site orientation and rate of exit-site/tunnel infection. 35 These findings support the notion that unrecognized factors, including patient factors or center preference, also likely influence catheter choice.30,31
A final consideration is the adapter that connects the catheter to the transfer set. Most PD catheters are supplied by the manufacturer with a plastic adapter, but this may be exchanged for a titanium adapter. Studies from the SCOPE collaborative have demonstrated a higher risk of peritonitis with the use of a plastic, rather than a titanium adapter.24,30
Unfortunately, there have not been any prospective trials comparing outcomes in children with PD catheters based on insertion technique. Data from observational studies include an analysis of 1106 PD catheter insertions among children enrolled in the SCOPE Collaborative which investigated risk factors for early peritonitis, defined as peritonitis in the first 90 days after catheter placement. 30 In univariate analysis, catheter placement technique was not associated with the risk for early peritonitis (p = 0.440). 30
A more recent analysis of SCOPE data included 1826 PD catheters of which 1416 were inserted using the laparoscopic technique, and 410 inserted using an open technique. In multivariable analyses, there was also no significant association between laparoscopic PD catheter placement and the occurrence of peritonitis within 7 days (aOR = 2.50, 95% CI 0.64–9.80, p = 0.19) and 30 days of PD catheter insertion (aOR = 1.20, 95% CI 0.67–2.18, p = 0.54). 51
A retrospective study by Lindley et al. compared the peritonitis rate among 10 patients who received simultaneous laparoscopic-assisted placement of a percutaneous endoscopic gastrostomy (PEG) and PD catheter placement with the rate among 23 children who received simultaneous PD catheter and gastrostomy creation using an open technique. 52 There was no significant difference in peritonitis rates per catheter year between the two groups (0.89 versus 0.59, p = 0.12). 52 This study also evaluated exit-site infection rates and catheter survival and found no difference in these outcomes based on insertion technique. 52 Whereas a higher percentage of peritonitis episodes due to Candida species was observed in the laparoscopic group, the difference was not statistically significant (27% versus 14%, p = 0.42). 52 It should be noted that this study included infection events well beyond the early post-operative period when the impact of implantation technique on the risk of infections is less obvious. 52
As stated above, there may be additional benefits of laparoscopic PD catheter insertion including improved healing, but these outcomes are not routinely captured in large national or international registries. A single center, retrospective study found no difference in catheter rates of leakage or obstruction after 25 laparoscopic and 23 open PD catheter placements in children. 50
Studies in adults with side-by-side comparisons of various PD catheter insertion techniques including percutaneous needle-guidewire with or without image guidance, peritoneoscopy, laparoscopy, and open dissection, as well as systematic reviews and meta-analyses comparing laparoscopic and open placement have not demonstrated consistent superiority of one technique with regard to infection prevention.41,45,53–63 However, a meta-analysis published in 2018 differentiated between basic and advanced laparoscopic catheter placement, with basic laparoscopic technique defined as using laparoscopy only to monitor the position of the catheter tip in the peritoneal cavity, while advanced laparoscopic placement includes various preemptive procedures to optimize catheter function such as rectus sheath tunneling, omentopexy, and lysis of adhesions.41,64,65 This meta-analysis found that advanced laparoscopic placement provided better outcomes than open dissection or basic laparoscopy, with outcome metrics including catheter tip migration, obstruction and catheter survival. 64
The PD catheter may be inserted through either a lateral or midline incision. In infants and children, a paramedian insertion is typically recommended to minimize the risk for herniation or dialysate leakage.2,61,62,67,68 An omentectomy is routinely performed at the time of PD catheter placement, to minimize the risk for PD catheter obstruction.66,68
Once inserted, the catheter should be securely anchored to optimize healing and minimize the risk for traction injury-related exit-site infection. A suture should not be placed at the exit site, as the presence of sutures increases the risk for bacterial colonization and subsequent infection. 2 The application of fibrin glue to the peritoneal cuff during implantation may be used, and was shown to reduce the risk for dialysate leakage in an open-label randomized study in a single pediatric center, but that study did not find an association between application of fibrin glue and the development of either exit-site infections or peritonitis. 69
Previous guidelines for children on PD recommended delaying catheter use for 10–15 days following catheter insertion. 2 In fact, data from the SCOPE collaborative identified use of the PD catheter for dialysis within 14 days of catheter insertion as an independent risk factor for peritonitis in the first 60 days after catheter insertion. 30 Data in adults on PD has shown an association between early use of the PD catheter and risk of dialysate leak, but uncertain effect on the risk for exit-site infection.45,70
Data in adults on PD include four randomized controlled studies that compared the use of perioperative intravenous antibiotic prophylaxis (cefuroxime, gentamicin, cefazolin, and vancomycin) versus no antibiotic treatment.72–75 Follow-up periods ranged from 10 to 28 days. The most compelling data comes from a 3 arm study published by Gadallah et al., which included 221 adults randomly assigned to receive intravenous vancomycin (1 g 12 h before the procedure, n = 86), intravenous cefazolin (1 g given 3 h before placement n = 85) or no antibiotic (n = 83). The incidence of peritonitis within 2 weeks of catheter placement was significantly lower for both vancomycin (1%) and cefazolin (7%) treated patients compared to controls (12%) (p = 0.02), and vancomycin was superior to cefazolin. 75
A subsequent systematic review published in 2017 concluded that pre- or peri- operative intravenous antibiotic prophylaxis compared to no treatment may reduce the risk of early peritonitis based primarily on the results of the study published by Gadallah et al., but found that there was no difference demonstrated in the three other studies using different antibiotics.72–76 In fact, an update to the ISPD-catheter related infection recommendations published in 2023 qualifies the 1A recommendation by stating that the effects of antibiotics administered immediately before catheter insertion on catheter-related infections are uncertain. 45
Although the studies in adults on PD suggest that cefazolin may not be as effective as vancomycin for prevention of peritonitis following PD catheter placement, given the risk for development of vancomycin resistant organisms, cefazolin is the preferred agent for perioperative prophylaxis. A single dose of cefazolin (25 mg/kg, maximum 2 g) is given within 60 min prior to the surgical incision (Table 2). It has been suggested that the efficacy of vancomycin, especially when compared to cefazolin, may be related to its relatively long half-life in the setting of end-stage kidney disease. 76 Local antibiograms should help guide antibiotic choice at the unit level, and infection history, including infection with and/or carriage of methicillin-resistant S. aureus, should guide antibiotic choice at the patient level.
Antifungal and antibacterial prophylaxis in peritoneal dialysis patients.
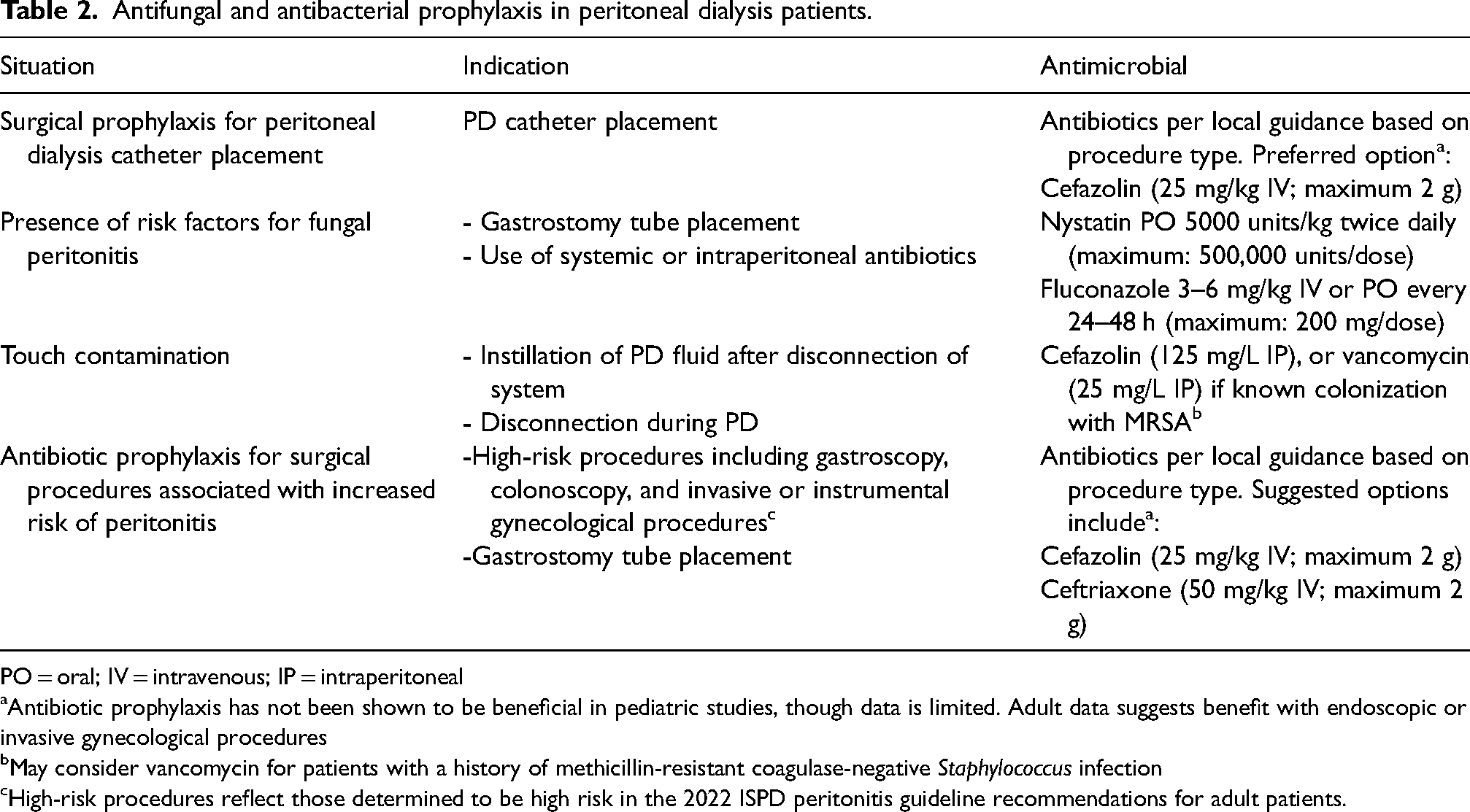
PO = oral; IV = intravenous; IP = intraperitoneal
Antibiotic prophylaxis has not been shown to be beneficial in pediatric studies, though data is limited. Adult data suggests benefit with endoscopic or invasive gynecological procedures
May consider vancomycin for patients with a history of methicillin-resistant coagulase-negative Staphylococcus infection
High-risk procedures reflect those determined to be high risk in the 2022 ISPD peritonitis guideline recommendations for adult patients.
Recently published guidelines for adults on PD suggest provision of nasal antibiotic prophylaxis when pre-PD catheter placement screening identifies nasal carriage of S. aureus (1C recommendation). 45 This suggestion is supported by data from a systematic review and meta-analysis which showed that while use of nasal antibiotic prophylaxis had uncertain effects on the risk of exit-site and tunnel infections in general, there was evidence of a significant reduction in catheter-related infections with use of intranasal mupirocin among patients who were S. aureus nasal carriers. 76 Data from SCOPE suggests that the practice of pre-PD catheter placement screening for S. aureus currently occurs in a minority of children enrolled in that collaborative, with only 529 screenings reported prior to 1998 PD catheter placements (26.5%) (B. Warady - personal communication).
Research Recommendations
● Future observational studies should seek to quantify surgical experience and explore associations between experience and PD catheter insertion outcomes.
● Studies evaluating the impact of insertion technique on risk for infection should uniformly focus on peritonitis and exit-site infections in the early post-implantation period (e.g., initial 30 days).
● Research should address the impact of pre-PD catheter placement screening for S. aureus and decolonization on the risk for PD catheter related infection in children, particularly in the early post-operative period.
Guideline 3 – Early Exit Site Care
3.1 We suggest delaying the first PD catheter dressing change for at least seven days post-PD catheter insertion unless the dressing is soiled, loose, or damp (2C).
Rationale
The suggestion has also been made that subsequent PD catheter exit-site dressings should ideally be changed only weekly until the exit site is healed as long as the dressing remains dry and intact. 80 Typically, the exit site may not be completely healed for 4–6 weeks post insertion. 79
It is generally accepted that dressing changes be performed using sterile technique by trained staff until the exit site is healed.2,81 Previously published ISPD guidelines for children on PD also recommended that the exit site be cleansed with a non irritating, nontoxic agent followed by application of a topical antibiotic cream or ointment. 2 In particular, cytotoxic agents such as povidone-iodine or hydrogen peroxide should be avoided during the healing phase.45,80 Unfortunately, there are no studies evaluating the use of specific cleansing agents or placement of any antimicrobial agent at the exit site in newly placed PD catheters in children.
Research Recommendations
● Studies evaluating the use of antimicrobial agents at the exit-site in the post-implantation period should include the use of antimicrobial impregnated dressings.
Guideline 4 – Chronic Exit Site Care
4.1 We suggest that routine PD catheter exit-site care with a sterile cleansing solution be conducted 2–3 times per week and after water exposure, vigorous exercise or soiling of the dressing (2D).
4.2 We suggest that a topical antibiotic be applied to the PD catheter exit site whenever exit-site care is performed (2D).
Rationale
Numerous exit-site cleansing agents are available including povidone-iodine, chlorhexidine solution, hydrogen peroxide and electrolyte chloroxidizing solutions. An observational study in children on PD revealed a reduction in exit-site infections, peritonitis and infection-related catheter removal when exit-site cleansing with sodium hypochlorite and application of mupirocin were routinely used, compared to application of mupirocin alone. 84
Studies in adults on PD have not consistently demonstrated superiority of one solution for prevention of exit-site infection or peritonitis. In fact, a systematic review concluded that the use of any topical disinfectant had an uncertain effect on the risk for these infections.45,76,85–93
As stated above, guidelines for adults on PD recommend daily topical application of an antibiotic cream or ointment at the catheter site. 85 This was included as a 1A recommendation in the ISPD guidelines published in 2017, although an update to these guidelines published in 2022 acknowledges that this practice varies among centers around the world.27,85 The recommendation was based on studies in adults on PD demonstrating daily application of mupirocin to the exit site as an effective strategy to reduce PD catheter-related infections.85,99–104 A meta-analysis published in 2004 found that topical administration of mupirocin to the nares of people on PD who were colonized with S. aureus significantly reduced the overall rates of exit-site and tunnel infections, but it did not reduce the rate of peritonitis. 61 Topical nasal application of mupirocin was subsequently found to have an uncertain effect on exit-site infections in a systematic review and meta-analysis published in 2017, but application of mupirocin to the exit site was grouped with oral antibiotic prophylaxis in this review. 76 More recently, a meta-analysis of six randomized controlled trials in adults on PD concluded that it was uncertain whether application of mupirocin to the exit site prevents exit-site infections, peritonitis or technical failure. 105 In light of these findings, the 2023 update to the ISPD catheter-related infection recommendations downgraded the recommendation for topical application of antibiotic cream or ointment to the catheter exit site from 1A to 1C. 45 The guideline workgroup commented that the certainty of evidence was reduced by highly variable approaches to the administration of mupirocin (intra-nasal versus exit site). 105
With the broad adoption of mupirocin as a topical prophylactic agent, early reports of mupirocin resistance and an increase in infections due to Pseudomonas spp. led to a search for alternative agents. 106 Studies in adults on PD revealed that gentamicin cream was at least as effective as mupirocin in reducing exit-site infections caused by both Pseudomonas spp. and S. aureus.101,107 However, subsequent observational studies suggested conversion from mupirocin to gentamicin was associated with an increase in Enterobacteriaceae and Pseudomonas spp. exit-site infections and that alternating between mupirocin and gentamicin may increase the risk of fungal peritonitis.108,109 A recent meta-analysis included 2 randomized controlled trials in adults on PD comparing exit-site application of gentamicin and mupirocin and found no difference in the rate of exit-site infections. 105 Guidelines published by ISPD in 2017 and 2023 suggest that topical gentamicin may be considered as an alternative to mupirocin.45,85
The HONEYPOT Study was an open-label trial in 26 centers in Australia and New Zealand in which 371 adults on PD were randomly assigned to daily topical exit-site application of antibacterial honey plus standard exit-site care versus standard exit-site care plus intranasal mupirocin in nasal carriers of S. aureus. 110 The median PD-related infection-free survival times were not significantly different in the antibacterial honey and control groups (unadjusted hazard ratio 1.12, 95% CI 0.83–1.51; p = 0.47). 110 In contrast, in a single-center case series of children on PD, the incorporation of antibacterial honey to the exit site as part of routine care was associated with a significant decrease in peritonitis rates. 76
Chronic exit-site care should also include efforts to minimize stress or tension on the exit site, including catheter immobilization. While use of a dressing is important to protect the healing exit-site, the benefit of a dressing once the exit site is healed for infection prevention has not been studied in children. Whereas current guidelines for adults on PD suggest that a dressing to cover the exit site is not mandatory, the use of a dressing in children with incontinence, including infants and toddlers, is crucial to minimize exposure of the exit site to stool and urine. 45
There is a cost associated with the use of antimicrobial prophylaxis, which varies significantly by region and the antimicrobial agent used. There are no cost-effectiveness studies in children, but a study in adults on PD demonstrated that daily application of mupirocin to the PD catheter exit site was a cost-effective preventive measure for gram-positive peritonitis. 114
Research Recommendations
● Future studies are required to clarify the relationship between infection rate (overall peritonitis rate, Pseudomonas spp. and S. aureus peritonitis rate, ESI rate) and the frequency of exit-site care.
● Ongoing studies should seek to clarify the risk and benefit of various antimicrobial agents, in terms of the efficacy of infection prevention and the development of antibiotic-resistant organisms.
Guideline 5 – Connectology
5.1 We recommend the use of flush before fill of the tubing system as part of an infection prevention strategy for children on PD (1A).
Rationale
Studies evaluating the impact of flush before fill in children have included a randomized, prospective study published by Warady et al. in which children on APD using flush before fill had a peritonitis rate of 1 infection per 16.8 patient months, whereas patients in the no flush group had a peritonitis rate of 1 infection every 12.6 patient months (p = 0.193). 117 The 1-year estimated infection-free survival was 59.7% in the flush group and 47.5% in the no flush group (p = 0.196). 117 In a retrospective observational study in children on CAPD, the traditional spike system (37 patients) and disconnect systems (16 patients) that comprised the O-set system (10 patients) and the basic Y-set system (6 patients) were compared. 118 Peritonitis rates were 1 episode every 10 months for the conventional spike system, versus 1 episode every 58 months for disconnect systems with flush before fill (p < 0.05).
The majority of the high-level evidence comes from studies in adults on CAPD, where a systematic review showed that twin-bag systems were superior to conventional single bag systems (7 trials, 485 patients, RR 0.64, 95% CI 0.53 to 0.77) in preventing peritonitis. 119 On the other hand, there was no difference in the number of patients with exit-site or tunnel infections (3 trials, 226 patients; RR, 1.00; 95% CI, 0.70 to 1.43) or in the exit-site and tunnel infection rates (2 trials, 2841 patient-months; RR 1.24; 95% CI, 0.91 to 1.69). 119 When comparing the two different systems of flush before fill, there was no significant difference between double-bag systems compared with the Y-set in terms of the number of patients with peritonitis (3 trials, 292 patients; RR 0.59; 95% CI, 0.35 to 1.01) and peritonitis rates (4 trials, 4319 patients-months; RR 0.90; 95% CI, 0.49).119–135
Research Recommendation
● Randomized controlled trials should be conducted to determine whether the reduction in peritonitis rates due to the use of the twin-bag systems is also observed in children on CAPD in low resource settings.
Guideline 6 – Ostomy Patients
6.1 We suggest placing a gastrostomy tube (GT) either prior to or concurrently with PD catheter insertion (2D).
6.2 We suggest that a gastrostomy tube can be placed using an open surgical procedure or laparoscopically for children receiving PD. In children not yet receiving PD, gastrostomy placement can be performed using either an open or laparoscopic surgical technique or by percutaneous endoscopic technique (PEG) (2D).
6.3 We suggest providing prophylactic antibiotic and antifungal therapy to children on PD in association with gastrostomy placement (2D).
Rationale
A report by Ledermann et al., showed a lower annual peritonitis rate in 15 pediatric patients undergoing PEG or Nissen fundoplication and gastrostomy (NG) or open gastrostomy (OG) prior to initiation of PD (annualized peritonitis rate: 0.6) compared with 9 pediatric patients undergoing NG or OG (annualized peritonitis rate: 1.4) and 5 pediatric patients undergoing PEG (annualized peritonitis rate: 1.0) while receiving PD. In the group of patients receiving a PEG after initiation of PD, the majority of infections occurred in the immediate postoperative period suggesting that it may not have been the timing, but the placement technique that was more important. 143 In a study by Ramage et al., no significant difference was found between nine patients who had a gastrostomy tube placed prior to commencement of PD (one episode every 7.5 patient-months) compared to 14 patients who had insertion of the gastrostomy while receiving PD. 140 There was also no statistical difference in the ESI rate of those undergoing gastrostomy tube placement prior to initiation of PD (one episode every 17.5 patient-months) and those undergoing placement while receiving PD. 140
The SCOPE collaborative investigators analyzed peritonitis rates in 204 patients who underwent gastrostomy placement before or at the same time as PD catheter insertion and compared this to the experience of 406 patients with gastrostomy placement after the initiation of PD. 51 Per multivariable analysis, timing of gastrostomy placement in relation to PD catheter insertion was not associated with risk of peritonitis within 30 days of gastrostomy placement (aOR = 1.72, 95% CI 0.87–3.38, p = 0.12). The association between gastrostomy placement after PD catheter insertion and the development of peritonitis within 7 days of gastrostomy placement did, however, reach borderline statistical significance (aOR = 3.19, 95% CI 0.90–11.28), p = 0.07), suggesting a potentially higher risk of peritonitis when a gastrostomy is placed after the PD catheter procedure. Further evidence supporting this comes from previous SCOPE data focusing on infants. 34 In this study, gastrostomy tube placement after PD catheter insertion was associated with a nearly threefold higher risk of peritonitis (OR 2.81, 95% CI 1.31, 6.01; p < 0.01). There is a further concern regarding a possible increased rate of fungal peritonitis when a gastrostomy tube, particularly a PEG, is placed after the initiation of PD. A report by von Schnakenburg, et al., showed that fungal episodes accounted for around 20% of all peritonitis episodes in those patients who had a PEG placed while receiving PD. 144 In the report by Lederman, et al., there was also a higher fungal peritonitis rate in those patients for whom gastrostomy tube placement (largely in those receiving a PEG) took place after PD had been established compared to the rate when gastrostomy placement occurred prior to PD establishment (0.36/pt-year vs. 0.093/pt-year). 143
Laparoscopy, where available, has largely supplanted open gastrostomy as the surgical technique of choice in non-CKD children.146,147 There are, however, no studies directly comparing open vs laparoscopic surgical gastrostomy placement in children on PD. In a recent retrospective study of children receiving a laparoscopic gastrostomy (11 patients) after initiation of PD, all patients were able to initiate gastrostomy use by postoperative day 2, and peritoneal dialysis was resumed in all patients between post-operative day 0 and 4. 148 The post gastrostomy peritonitis rate of 0.36 per patient year during a median follow-up time of 404 days was not significantly different than the rate experienced during the period before gastrostomy placement, and there were no fungal peritonitis episodes. In a study from New Zealand of 15 children, most of whom had a gastrostomy inserted using an open surgical procedure (2 laparoscopic) after PD had been initiated, the peritonitis rate increased from 0.6 to 1.21 episodes per patient year post gastrostomy placement. The increase did, however, not reach statistical significance and there were also no fungal peritonitis episodes in this study. Follow up before and after gastrostomy was 160 months. 149
The evidence supporting the use of prophylactic antibiotic and antifungal therapy in PD patients comes from a single comparative study of children who received a PEG after initiation of PD. 144 In this multi-center retrospective study, one-third of patients (7/21) who received antibiotic prophylaxis (with or without antifungal therapy) developed bacterial peritonitis, while 7/19 (37%) patients who received no fungal prophylaxis (with or without antibiotics) developed fungal peritonitis. One out of 8 patients who received antibiotic and antifungal therapy developed peritonitis within the first week following PEG insertion, with no fungal infections. In contrast, peritonitis occurred in 3/6 patients (2 bacterial, 1 fungal/bacterial) who did not receive any prophylactic agents. There was also an increased incidence of modality change and death on PD in patients who did not receive antifungal therapy.
Dorman et. al. studied 11 children who received a laparoscopic gastrostomy after initiation of PD. 148 All patients received antibiotic and antifungal prophylaxis, and after a median follow-up of 404 days, the post-gastrostomy peritonitis rate of 0.36 episodes per patient year was not significantly different than the rate experienced during the period before gastrostomy insertion in the same group of patients. There were no episodes of fungal peritonitis.
A retrospective review from New Zealand reported on 15 children who received either an open gastrostomy (90%) or a laparoscopic gastrostomy after initiation of PD. 149 All patients received prophylactic antibiotics, but not prophylactic anti-fungal therapy. There was no statistically significant increase in the peritonitis rate post gastrostomy insertion, and there were no fungal infections. The follow up period was 160 months before and after insertion.
Analysis of data from the SCOPE Collaborative revealed that in 76 patients who underwent gastrointestinal procedures (type not specified) while on PD, there was no difference in the peritonitis rate within 7 or 30 days of the procedure in the 56 patients who were given antibiotics prior to these procedures when compared to those who did not receive prophylactic therapy. 51 Nevertheless, because of the devastating consequences of fungal peritonitis, this workgroup remains in favor of prophylactic antifungal therapy in addition to antibiotic prophylaxis in patients who require a gastrostomy and who are already receiving maintenance PD. Whereas the use of prophylactic antibiotics generally raises concern regarding antibiotic resistance and side effects, the risks appear to be low based on adult data regarding prophylactic antibiotics for PEG placement. 152
Regardless of the gastrostomy placement technique used, PD should preferably be withheld for a period of time after gastrostomy placement. The optimal duration the patient should be maintained off PD is not known, but various publications have reported 0–4 days.143,148 If the gastrostomy is inserted using a PEG technique, a longer time off PD is probably warranted. 145 It is generally advisable to reinitiate dialysis with a lower PD fill volume and then gradually increase to the maintenance volume over the next 5–7 days. In the patient receiving CAPD, consideration may also be given to temporarily changing the PD modality to APD, with a diminished or absent daytime fill volume.
The presence of a concurrent colostomy in infants on PD is becoming more common in centers where neonatal maintenance PD is practiced. Recent data from the IPPN database identified 20 children on PD who also had a colostomy. 154 In this report, children with a colostomy had a significantly higher annualized peritonitis rate than a matched control group (1.13 vs 0.7). The authors of this report suggest that alternative strategies may be necessary to reduce the risk of infectious complications, such as placement of the PD catheter exit-site on the chest wall.
Research Recommendations
● Additional data should be sought that provides direct comparisons between laparoscopic and open surgical procedure for gastrostomy placement with respect to infection risk, longevity of a functional gastrostomy and patient well-being.
● Future research should be conducted to determine the optimal antibiotic and antifungal prophylactic regimen for surgically placed gastrostomies (laparoscopic or open) in children receiving PD.
● Infection prevention strategies should be developed to decrease the risk of peritonitis in children on PD with a concurrent colostomy.
Guideline 7 – Adjunctive Prophylactic Antimicrobial Therapy
7.1 We suggest the use of prophylactic nystatin or fluconazole therapy for children on PD receiving intraperitoneal or systemic antibiotic therapy (2D).
7.2 We suggest using prophylactic antibiotics for pediatric PD patients undergoing certain GI (eg. colonoscopy) or GU procedures (e.g., invasive gynecological), in accordance with local guidelines (2D).
7.3 We suggest NOT using prophylactic antibiotics for pediatric PD patients undergoing dental procedures (2D).
7.4 We suggest that prophylactic antibiotics be administered after wet contamination of the PD system to prevent the development of peritonitis (2D).
Rationale
A number of historically controlled studies, including one pediatric study and several studies in adults, have shown a decrease in fungal peritonitis after introduction of antifungal prophylaxis during the administration of antibiotics.160–164 Two randomized controlled trials in adults on PD have also demonstrated a reduction in fungal peritonitis in patients prescribed antifungal prophylaxis during the course of antibiotic therapy.165,166 In one study, patients were randomized to receive placebo or nystatin for the duration of any antibiotic administration, regardless of the indication for antibiotics. Those randomized to nystatin experienced fewer episodes of Candida peritonitis (1.9/100 vs. 6.4/100; p < 0.05). 165 In the other trial, patients were randomized to receive fluconazole or a placebo while they were treated with antibiotics for peritonitis. 166 There were significantly fewer episodes of fungal peritonitis (3 vs 15, p = 0.005) in the group receiving fluconazole. Of note, only 4 of 10 cases of Candida peritonitis were fluconazole susceptible, raising concerns about the potential for the development of resistance. A Cochrane review of the use of antimicrobials for the prevention of peritonitis in patients on PD found a significant benefit to the administration of antifungal therapy during treatment with antibiotics. 76 More recent data from SCOPE and IPPN did not identify a difference in the risk for development of fungal peritonitis between children who did and did not receive antifungal prophylaxis during episodes of bacterial peritonitis; however, 77% of 586 patients in SCOPE and 95% of 2000 patients in IPPN received antifungals during antibiotic treatment for peritonitis.167,168 Therefore, there were relatively few patients in the unexposed groups, which, combined with the rarity of fungal peritonitis, limits the conclusions that can be drawn from these data.
When evaluating the impact of prophylactic antibiotics on peritonitis rates with dental procedures, no difference was observed. This finding is consistent with the recommendation not to administer prophylactic antibiotics for the prevention of subacute bacterial endocarditis in most children with underlying heart disease when they undergo a dental procedure. 169
The adult ISPD guideline recommends prophylactic antibiotics prior to colonoscopy and invasive gynecological procedures based on rates of peritonitis for these procedures being as high as 8.5% and 38.5%, respectively. With consideration of the combination of the pediatric and adult data, we also suggest that antibiotic prophylaxis may be indicated for certain GI and GU procedures such as colonoscopy and invasive gynecological procedures. However, the local setting experience should help guide the decision regarding which procedures should be conducted with antibiotic prophylaxis. (Table 2)
The main harms associated with the use of antibiotics include common adverse drug events including Clostridium difficile infection, for which even a single dose of antibiotics can put an individual at significant risk. 170 Furthermore, as antibiotic resistance becomes more prevalent, data has demonstrated that each additional day of antibiotic use confers a 4% risk for the development of resistance. 171 On the other hand, the loss of the peritoneum as a modality for dialysis can have significant consequences to a child, so weighing the risk and benefits of the use of prophylactic antibiotics for GI and GU procedures is not trivial.
In children receiving PD, touch contamination is a well recognized risk factor for the development of peritonitis. The risk of peritonitis following touch contamination in children was significantly higher in the SCOPE collaborative (rate ratio 2.22, 1.44–3.34) based on data collected from 734 children enrolled in the collaborative. Touch contamination can be divided into two categories. In the adult ISPD guidelines and in this guideline, the type of touch contamination, wet or dry, determines the need for the administration of prophylactic antibiotics.
Data supporting the use of antibiotics as prophylaxis following wet contamination include a single center study of 548 episodes of touch contamination in adults on PD, in which the overall rate of peritonitis following touch contamination was relatively low (3.1%) and all episodes of peritonitis (N = 17) occurred after wet contamination (5.6%). 172 Among these cases of peritonitis, only one received prophylactic antibiotics following the wet contamination and prophylactic antibiotic use following touch contamination was associated with a significantly lower risk for peritonitis. In this study, the episodes of peritonitis following touch contamination were culture negative or caused by MSSA, methicillin-susceptible and methicillin-resistant coagulase negative Staphylococcus and gram-negative bacteria such as Pseudomonas aeruginosa and Acinetobacter baumanii.
The optimal antibiotic to be administered following an episode of touch contamination has not been established. Given the predominance of culture-negative peritonitis, MSSA, and methicillin-susceptible coagulase-negative Staphylococcus peritonitis following touch contamination, and weighing the risks of broader or longer antibiotic treatment, a single dose of intraperitoneal (IP) cefazolin is reasonable for most patients. IP vancomycin could be considered in patients known to be colonized with MRSA.
The SCOPE collaborative data is limited as non-specific GI, GU, and dental procedures were included in the multivariate model demonstrating that prophylactic antibiotics are not beneficial in preventing peritonitis.
Research Recommendations
● Research is needed to evaluate the safety, efficacy, and risk of development of antifungal resistance associated with the use of antifungal prophylaxis in children with administration of antibiotics for indications other than peritonitis.
● Further research is needed to compare the efficacy of nystatin vs. fluconazole as prophylactic agents in PD patients and to better understand the optimal duration of antifungal prophylaxis during treatment with systemic antibiotics.
● More research is needed, specifically in children undergoing GI and GU procedures, to determine which invasive procedures should be preceded by prophylactic antibiotics.
Guideline 8 – Diagnosis of PD-Related Peritonitis
8.1 Cloudy peritoneal effluent should be sent for cell count, differential count, gram stain and culture when the diagnosis of peritonitis is suspected (Not Graded).
8.2 An empiric diagnosis of peritonitis should be made when at least two of the following are present: effluent white blood cell count greater than 100/mm3, with at least 50% of the WBCs as polymorphonuclear leukocytes, clinical signs and symptoms (fever, abdominal pain) and growth of microorganisms in PD effluent culture (Not Graded).
8.3 We suggest that inoculation of blood culture bottles be used for bacterial culture of PD effluent, with the addition of centrifugation to enhance the bacterial yield (Not Graded).
Rationale
Microscopy is essential to confirm the presence of white blood cells (WBCs), because cloudy fluid can also be a result of the presence of eosinophilic peritonitis, blood, chyle, fibrin,
As an early screening test for the presence of WBCs, leukocyte esterase reagent test strips have been used at some centers in patients suspected of having peritonitis.175,176
For patients on CAPD or APD with a daytime exchange, the first cloudy bag or the manual drain should be sent for cell count, differential count, gram stain, and culture. For the child on APD without a daytime dwell, the fill volume should be instilled for a minimum of 2 hours, with the subsequent effluent being sent for cell count, differential count, and culture.
To guide empiric therapy, it is useful to perform a gram stain on all samples; however, the sensitivity of a gram stain is low. Cytocentrifugation of samples is recommended to increase the sensitivity of gram staining.177,178 Despite a large number of WBCs, microorganisms may not be visible or may be low in yield because of their sequestration within phagocytes. Still, the gram stain could be the first clue to a fungal infection, because budding yeast may be seen.
The abdominal pain in peritonitis is typically generalized and is often associated with guarding and rebound tenderness. The degree of pain is variable, being mild to moderate in coagulase-negative Staphylococcus (CoNS) peritonitis and more severe in infections involving Streptococcus, gram-negative rods, and S. aureus. If the pain and tenderness are localized, acute appendicitis must be considered. If subsequent peritoneal fluid cultures grow multiple organisms, viscus perforation must be excluded.
A Disease Severity Score, defined by the sum of points for pain (0 = no pain; 1 = moderate pain, or nausea not requiring specific therapy; 2 = severe pain usually requiring analgesic therapy, or vomiting; 3 = peritoneal pain with a tense abdomen or paralytic bowel) and fever based on oral temperature (0 = <37.5°C; 1 = 37.5°C – 38.9°C; 2 = >38.9°C) has been used to objectively evaluate the severity of the clinical status.182,183
After a dwell time of at least 2 hours, a peritoneal effluent WBC count of more than 100/mm3 in an uncentrifuged specimen, with a differential count of at least 50% neutrophils, is highly suggestive of peritonitis.
For the child on APD without a daytime dwell, the absolute WBC count may not fulfill the standard diagnostic criteria if the dwell time is too short, as in the case of many patients receiving APD; in this case, the presence of 50% or more neutrophils, even if the total cell count is less than 100/mm3, is highly suggestive of peritonitis. In equivocal cases, or in patients with systemic or abdominal symptoms in whom the effluent appears clear, a second exchange with a dwell time of at least 2 hours should be performed and a second effluent sample evaluated.
In an IPPR report, 2.8% of clinical peritonitis episodes had peritoneal effluent WBC counts less than 100/mm3, and 8.5% of cases had less than 50% neutrophils. 181 If the eosinophil count exceeds 10%, a diagnosis of eosinophilic peritonitis should be considered, especially if the peritoneal fluid cultures are negative. 173
Specimens should be sent to the laboratory and processed within 6 hours. Should there be any delay in either transport or processing for culture, effluent samples must be refrigerated at 4°C until processed. A delay of more than 12 hours is unacceptable and will likely generate spurious results.184,186 Patients who reside in areas far from medical facilities should be taught the recommended technique for collecting the cloudy peritoneal effluent and either inoculating it in blood culture bottles for transport or for refrigerating (not freezing) the effluent bag until the sample can be brought to the dialysis center for transport to the laboratory.
Centrifuging a large volume (50 mL) of peritoneal effluent at 3000 g for 15 minutes to obtain sediment for culture 187 has increased the yield by 5–10 times . The sediment is resuspended in 6–10 mL of sterile normal saline and inoculated directly on to solid-culture media and into standard blood-culture media.27,188 At a minimum, blood and chocolate agar plates should be inoculated with re-suspended sediment; ideally, MacConkey agar plates should also be plated. The solid-culture media should be incubated in aerobic conditions. Anaerobic cultures may also be performed as required. Concentration techniques such as this one have yielded a culture-negative rate of less than 5%.188,189
Rapid blood culture techniques such as Bactec (Becton–Dickinson, Franklin Lakes, NJ, USA), Septi-Chek (Becton–Dickinson), and BacT/Alert (bioMérieux, Marcy l’Etoile, France) are useful in reducing the time to identification of microorganisms. The total number of microorganisms recovered has been found to be significantly better from inoculated blood culture bottles than from routine culture on solid media. 190 Bedside-inoculated bottles have yielded similar results compared to laboratory-inoculated bottles.
Using the foregoing culture techniques and associated concentration methods, most cultures will become positive within 24 hours. Certain situations, such as suspicion of a slow growing organism per clinical history, may call for additional incubation time. Agar plates should be incubated at 35˚C to 37˚C in 5% CO2 for 24 hours. Anaerobic plates should be incubated at 35˚C to 37˚C under anaerobic conditions for a minimum of 7 days. Blood culture bottles should be incubated in a blood culture system for 5 days. A microbiologic diagnosis can be obtained in more than 75% of specimens by 72 hours.
Polymerase chain reaction can be a sensitive method for identifying causative organisms. Broad-spectrum polymerase chain reaction with sequencing, and quantitative bacterial DNA polymerase chain reaction assays can complement, but not replace, culture methods in the diagnosis of peritonitis, especially if the patient is receiving antibiotic therapy.191,192
Research Recommendations
● The factors contributing to elevated rates of culture-negative peritonitis (>15%) in pediatric centers should be explored.
● Prospective trials should be used to compare the sensitivity and specificity of the various diagnostic technologies available in the setting of PD-related peritonitis.
Guideline 9 – Empiric Antibiotic Therapy
9.1 Empiric antibiotic therapy should be initiated as soon as possible after appropriate microbiological specimens for the diagnosis of peritonitis have been obtained (Not Graded).
9.2 Center-specific antibiotic susceptibility patterns and patient history should help guide the selection of empiric antibiotic therapy (Not Graded).
9.3 We suggest using either a) intraperitoneal cefepime monotherapy or b) gram-positive coverage with a first-generation cephalosporin or vancomycin AND gram-negative coverage with ceftazidime (preferred) or an aminoglycoside (if ceftazidime is unavailable). In patients with features of sepsis, intravenous antibiotics should be used (2D).
Rationale
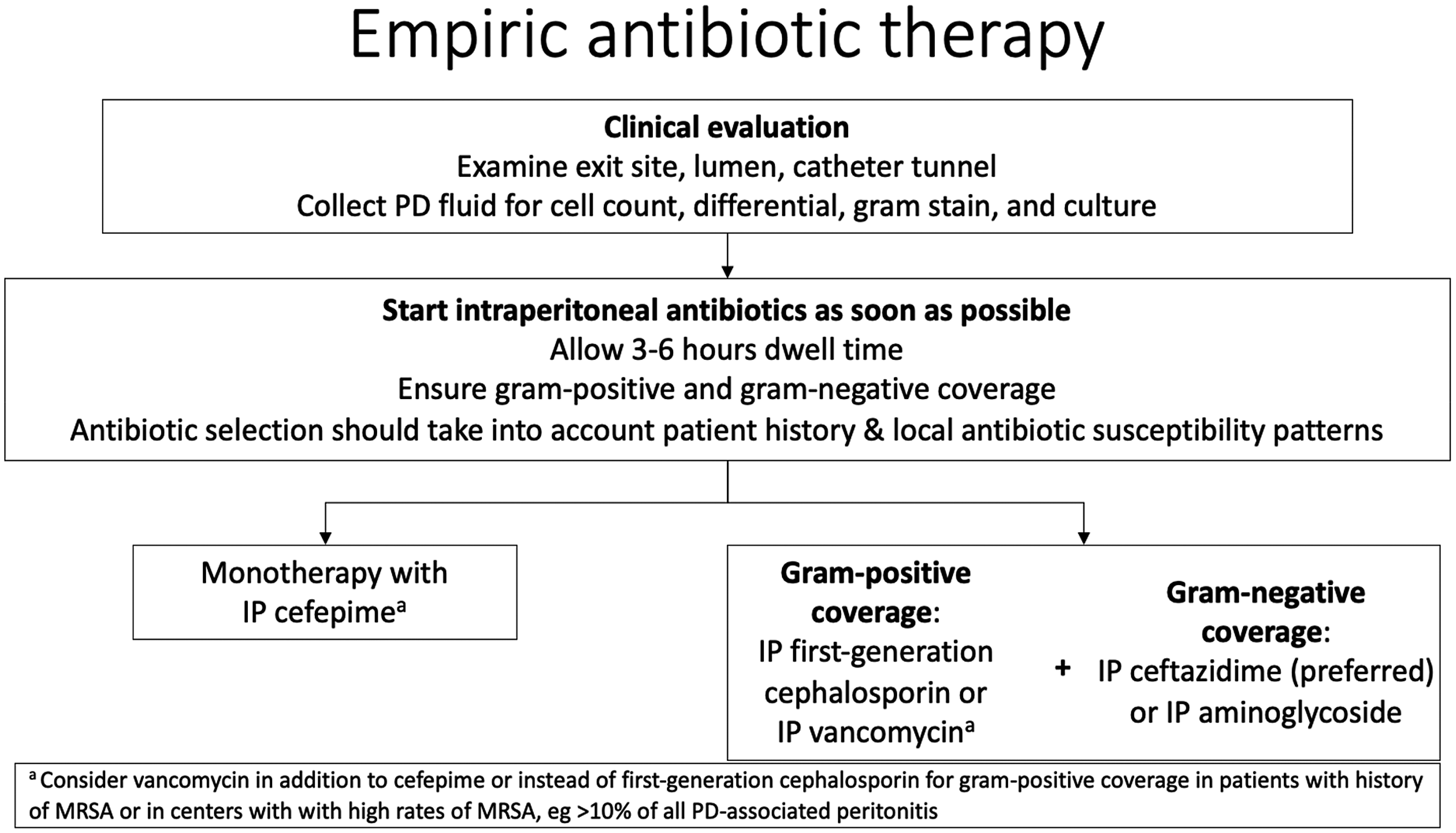
Management algorithm for empiric antibiotic therapy.
We suggest the use of ceftazidime over the aminoglycosides due to concerns for adverse events including ototoxicity and nephrotoxicity/loss of residual kidney function associated with aminoglycoside therapy. However, from observational studies and a randomized controlled trial, empiric IP aminoglycoside therapy has not been shown to negatively impact residual kidney function.198–200 Ototoxicity has, however, been observed in patients receiving IP aminoglycosides and in adults the use of N-acetylcysteine appears protective.201–205 Furthermore, individuals with specific variants in the MT-RNR1 gene should not receive aminoglycosides due to the risk of ototoxicity with just a single dose. The impact of this gene in patients receiving IP aminoglycosides is not known. In some areas, individuals can be tested for this specific gene variants. 206 Furthermore, the United States Clinical Laboratory Standards Institute (CLSI) and the European Committee on Antimicrobial Susceptibility Testing (EUCAST) no longer recommends gentamicin for the treatment of P. aeruginosa infections due to emerging data that gentamicin is inferior for treating this pathogen.207,208 CLSI recommends amikacin for P. aeruginosa only in urinary tract infections. The aminoglycoside with the best activity for invasive P. aeruginosa infections is tobramycin. If aminoglycosides are used, intermittent daily IP dosing is preferred to continuous dosing. This strategy takes advantage of the concentration-dependent mechanism of action of aminoglycosides by allowing for high peak concentrations and lower trough concentrations to minimize toxicity. Studies in adults have shown no difference in efficacy between continuous and intermittent dosing strategies, but have shown higher serum troughs in patients receiving continuous dosing, which are associated with a higher risk for ototoxicity.
Intraperitoneal administration of cephalosporins has also been associated with systemic absorption. Most importantly, neurotoxicity has been observed with IV cefepime in the form of seizures, encephalopathy and depressed consciousness. Primary risk factors have included the elderly and those with kidney dysfunction and requiring intensive care. 209 There is a report of cefepime-associated neurotoxicity in 2 patients receiving IP cefepime; in both cases, symptoms resolved with dose reduction or discontinuation of the antibiotic. 210 In a larger study of 87 episodes of peritonitis treated with IP cefepime, it was very well tolerated with no evidence of neurotoxicity. 196
In institutions where MRSA accounts for a high percentage of peritonitis episodes (for example, > 10% of all episodes of peritonitis), the center should consider vancomycin instead of cefepime or cefazolin for gram-positive coverage. The workgroup also suggests that providers choose vancomycin as part of the empiric treatment regimen in patients with a personal history of MRSA infection. In these cases, vancomycin should be combined with ceftazidime for gram-negative coverage.
Finally, intravenous therapy should be used initially in patients who are critically ill with signs of sepsis so as to ensure the achievement of therapeutic antibiotic blood levels.
Data are limited on the rate of systemic absorption of IP aminoglycosides and cephalosporins in children and the potential for associated adverse effects.
Research Recommendations
● More comparative clinical outcome data are needed to further evaluate the different empiric antibiotics regimens utilized to ensure their efficacy and safety.
● Given the recent recommendations to avoid the use of gentamicin and amikacin in the treatment of P. aeruginosa, more data are needed to evaluate the impact of this empiric antibiotic recommendation on clinical outcomes in patients with P. aeruginosa peritonitis.
Guideline 10 – General Principles of Antibiotic Therapy for Peritonitis
10.1 Antibiotic therapy should be adjusted once a causative pathogen is identified, and antibiotic susceptibility data should guide the definitive antibiotic selection (Not Graded).
10.2 Providers should select the narrowest effective agent and treat for the shortest effective duration to reduce the risks for development of resistance, antibiotic-associated adverse events, and time at risk for fungal peritonitis (Not Graded).
10.3 The IP route of antibiotic administration should be preferred as long as the compatibility and stability of the IP antibiotics allow, unless the patient has features of sepsis, in which case intravenous antibiotics should be used (Not Graded).
Rationale
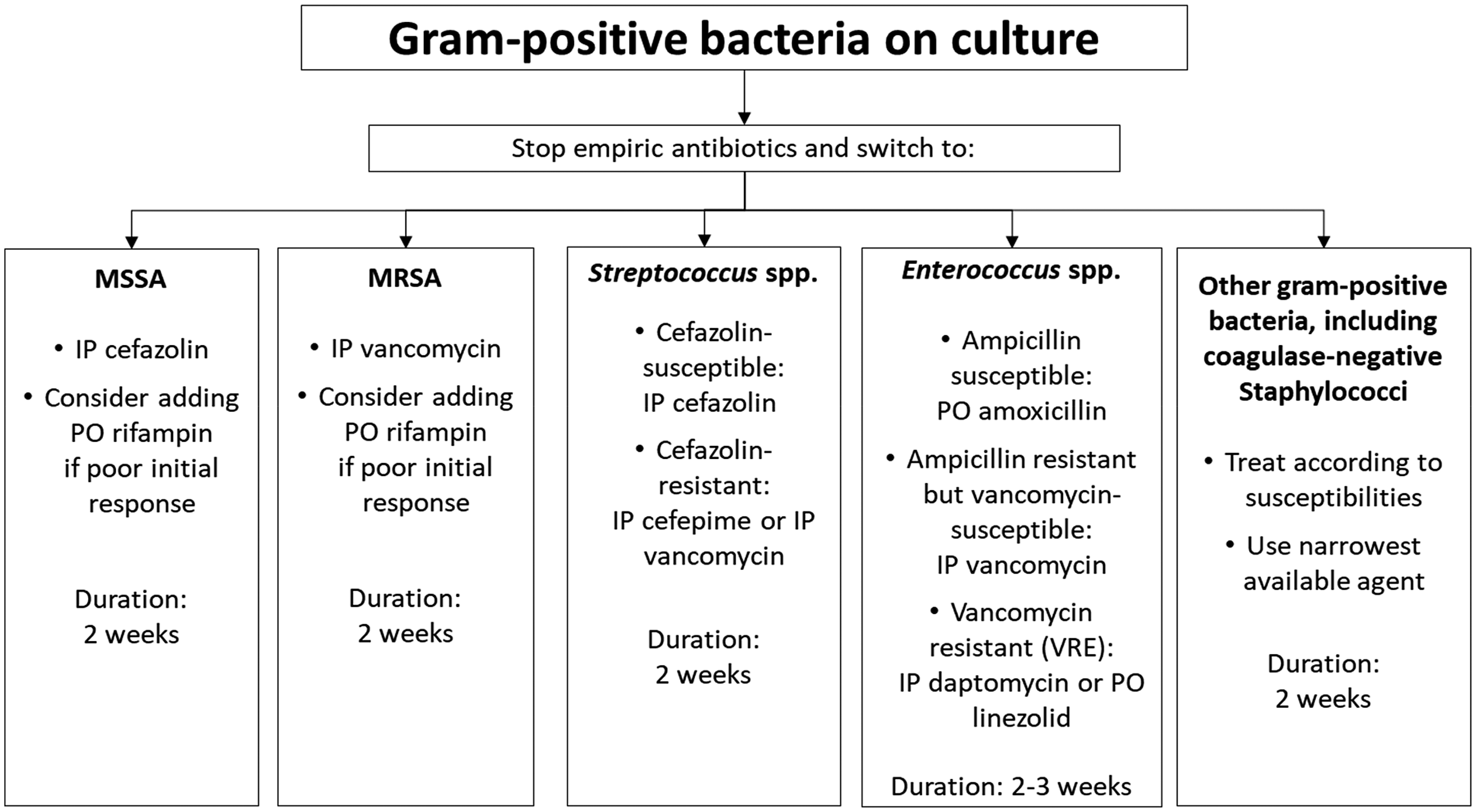
Management algorithm for gram-positive bacteria on culture.

Management algorithm for gram-negative bacteria on culture.
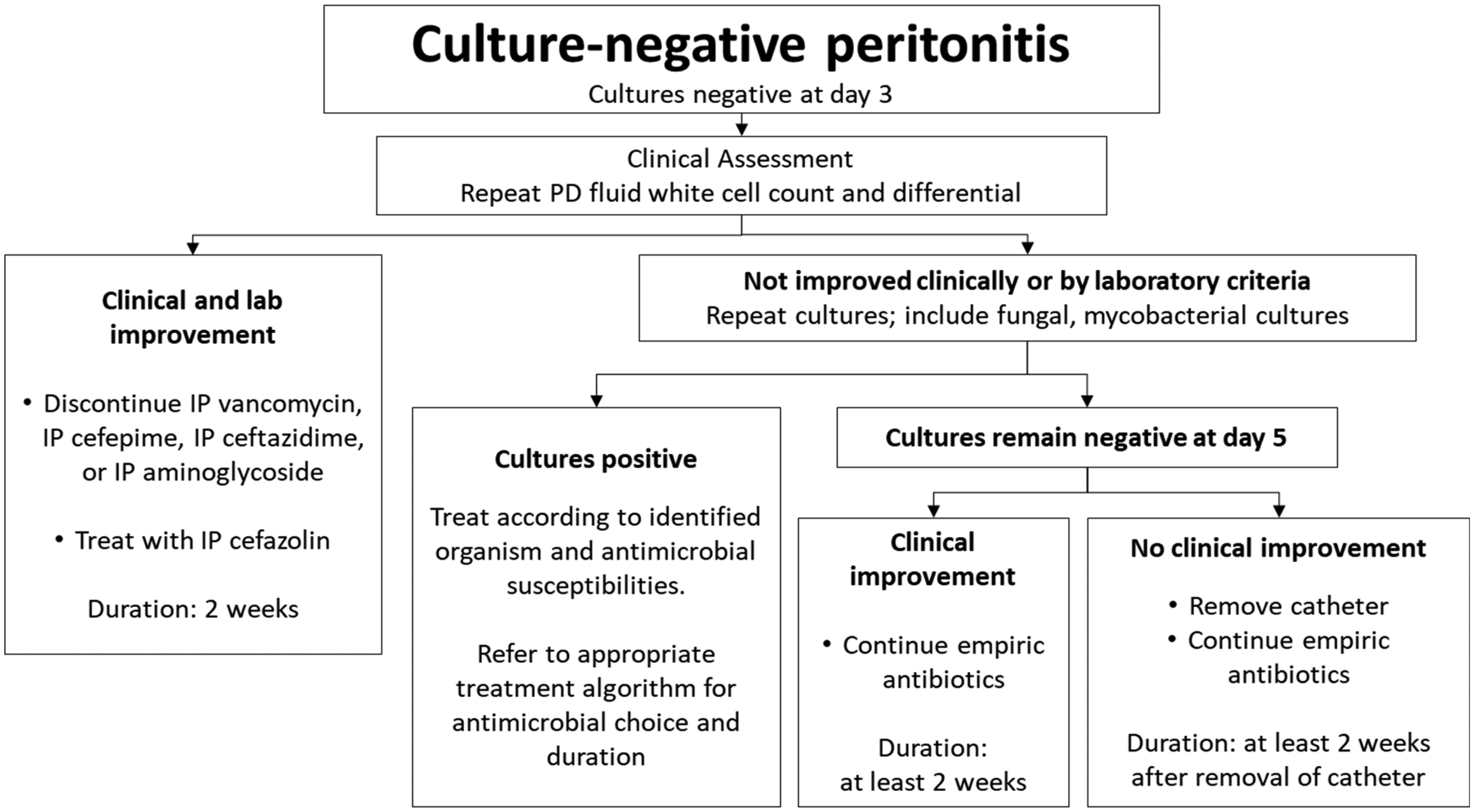
Management algorithm for culture-negative peritonitis.
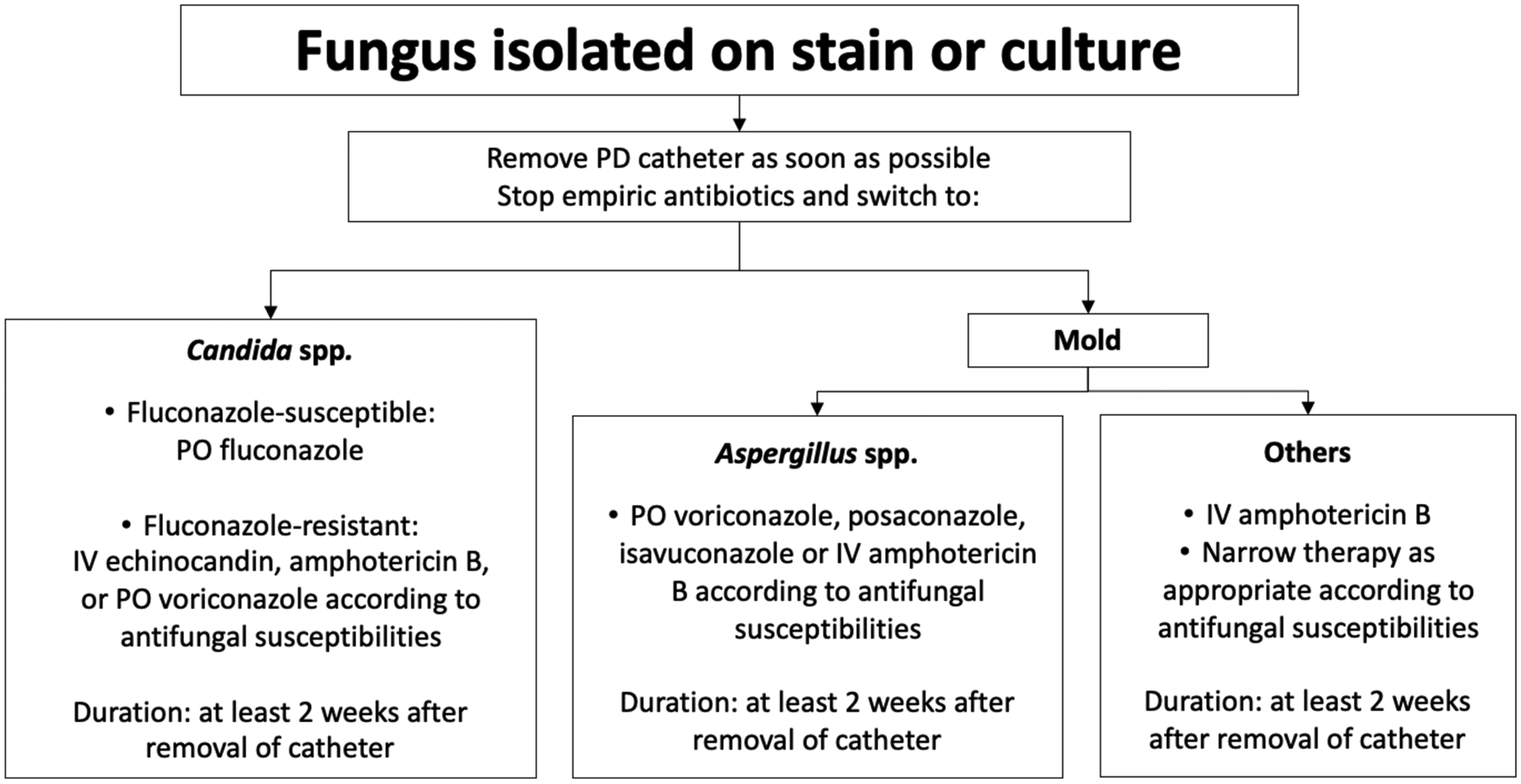
Management algorithm for fungal peritonitis.
Intraperitoneal antibiotic dosing recommendations for treatment of peritonitis.
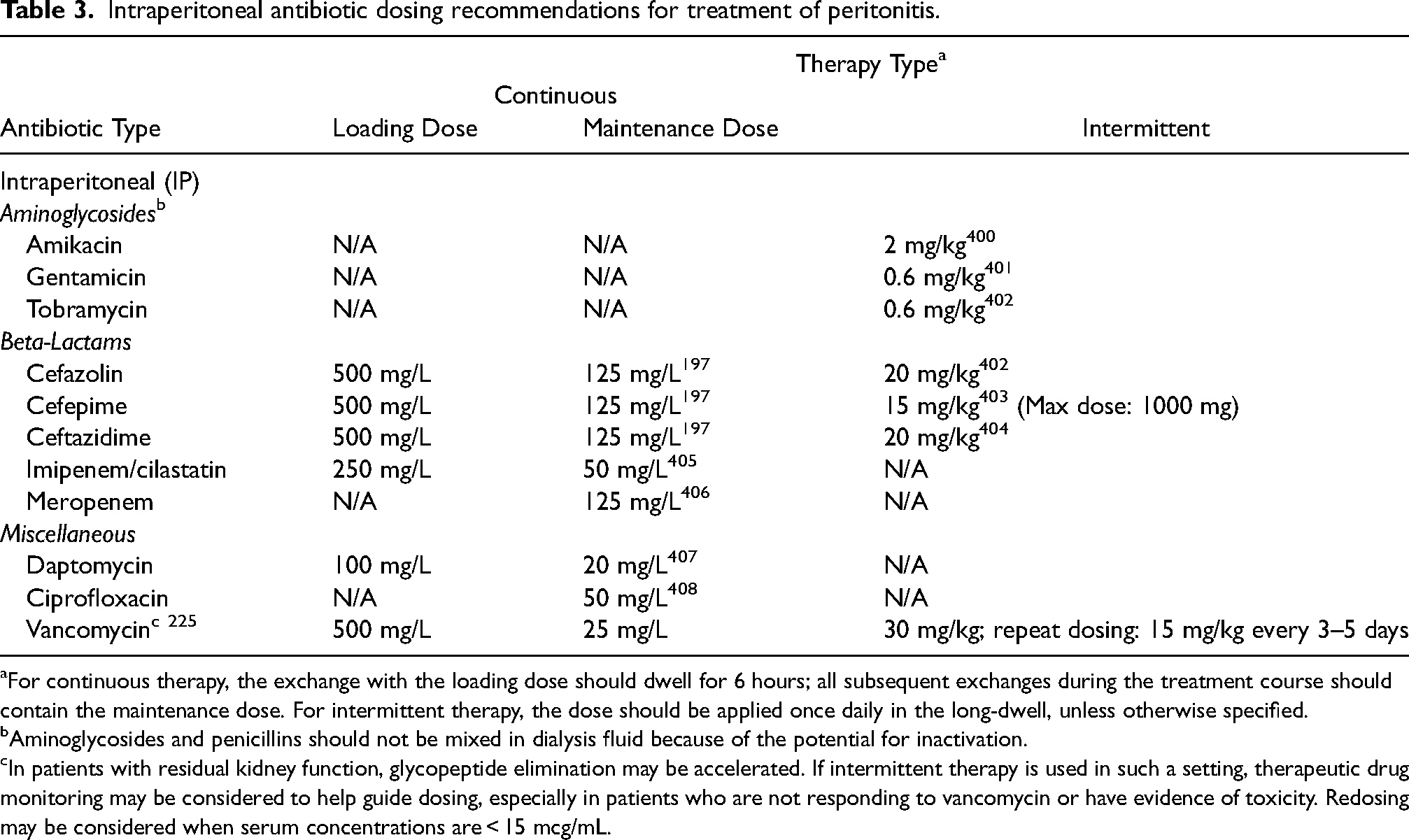
For continuous therapy, the exchange with the loading dose should dwell for 6 hours; all subsequent exchanges during the treatment course should contain the maintenance dose. For intermittent therapy, the dose should be applied once daily in the long-dwell, unless otherwise specified.
Aminoglycosides and penicillins should not be mixed in dialysis fluid because of the potential for inactivation.
In patients with residual kidney function, glycopeptide elimination may be accelerated. If intermittent therapy is used in such a setting, therapeutic drug monitoring may be considered to help guide dosing, especially in patients who are not responding to vancomycin or have evidence of toxicity. Redosing may be considered when serum concentrations are < 15 mcg/mL.
Systemic antibiotic dosing recommendations for treatment of peritonitis.
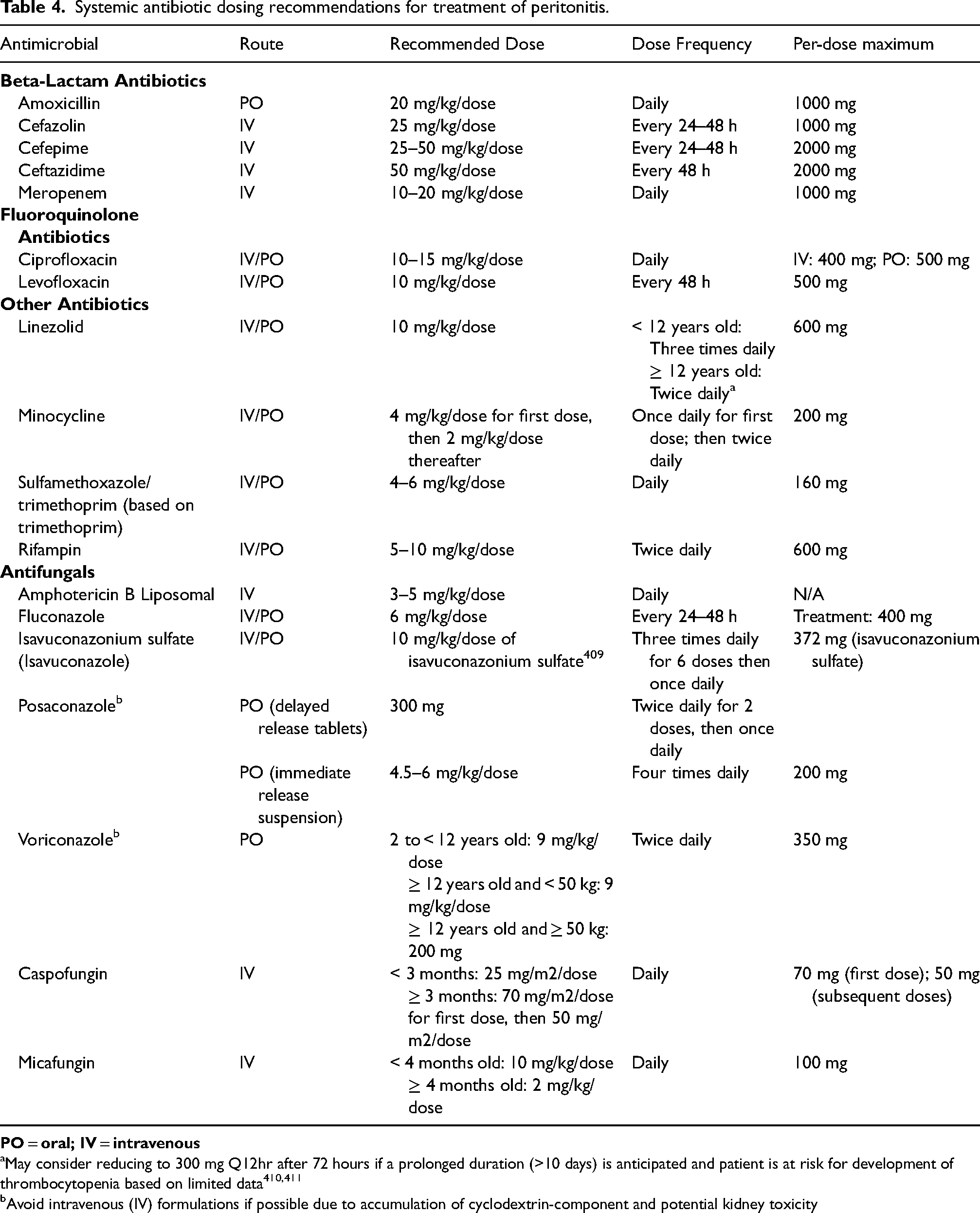
May consider reducing to 300 mg Q12hr after 72 hours if a prolonged duration (>10 days) is anticipated and patient is at risk for development of thrombocytopenia based on limited data410,411
Avoid intravenous (IV) formulations if possible due to accumulation of cyclodextrin-component and potential kidney toxicity
The approach to management of children with penicillin allergies has changed over time. While 5–10% of people report penicillin allergies, only about 10% of those who undergo testing have a true penicillin allergy. Children with penicillin allergy labels are more likely to receive guideline-discordant antibiotics that are broader than necessary and are more likely to experience antibiotic-associated adverse events. 212 It is, in turn, important to remove inaccurate penicillin allergy labels and multiple organizations recommend a proactive approach to penicillin allergy delabeling when appropriate.213,214
Even for patients with a true penicillin allergy, most cephalosporins can be used safely. Cross-reactivity between penicillins and cephalosporins was initially thought to be as high as 8% but is now understood to be much lower.215,216 Patients with non-anaphylactic penicillin allergies can receive any cephalosporin without additional testing. Patients with anaphylaxis to penicillin can safely receive cephalosporins with side chains that are different from penicillin, in particular cefazolin. Children with penicillin allergies, including anaphylaxis, can receive carbapenems when clinically appropriate. 217 If a child is thought to have an allergy to a preferred agent or an antibiotic with a similar side chain that would preclude use of the preferred agent, we recommend involvement of an allergist to facilitate optimal prescribing, rather than using second-line antibiotics.
Guideline 11 – Modification for Gram-positive Peritonitis
11.1 We suggest that peritonitis caused by methicillin-susceptible S. aureus (MSSA) be treated with IP cefazolin for a duration of 2 weeks (2D).
11.2 We suggest that peritonitis caused by methicillin-resistant S. aureus (MRSA) be treated with IP vancomycin for a duration of 2 weeks (2D).
11.3 We suggest the addition of PO rifampicin for peritonitis caused by either MSSA or MRSA if there is a poor initial response (2D).
11.4 We suggest that coagulase-negative staphylococci should be treated with IP cefazolin or vancomycin, according to susceptibilities, for a period of 2 weeks (Not Graded).
11.5 We suggest that peritonitis caused by enterococci should be treated according to susceptibilities with PO amoxicillin (for ampicillin-susceptible enterococci), IP vancomycin (for ampicillin-resistant, but vancomycin-susceptible enterococci), or with IP daptomycin or PO linezolid (for vancomycin-resistant enterococci) for a duration of 2–3 weeks (Not Graded).
11.6 We suggest that peritonitis caused by Streptococcus spp. should be treated with IP cefazolin, if susceptible, or IP cefepime or IP vancomycin if not susceptible to cefazolin, for a duration of 2 weeks (Not Graded).
11.7 We suggest that other gram-positive bacteria should be treated with the narrowest available agent for a duration of 2 weeks (Not Graded).
Rationale
Gram-positive organisms are the most commonly identified pathogens in pediatric PD-associated peritonitis, accounting for 50–60% of cases in which an organism is identified.24,48 Coagulase-negative Staphylococci are most common, followed by Staphylococcus aureus, then Enterococci and Streptococci. Outcomes for gram-positive peritonitis in children tend to be quite good, with resolution as characterized by the lack of impairment in ultrafiltration capacity and absence of clinically apparent intraperitoneal adhesions in 85% of episodes. Treatment regimens, both antibiotic choice and duration, should be tailored to the identified organism (Figure 2).
Previous concerns about worse outcomes in children with PD-associated peritonitis treated with cefazolin have not persisted in analyses of more recent data with improved cefazolin dosing and are consistent with data in other serious MSSA infections, where cefazolin is preferred. Cefazolin also has a narrower antibiotic spectrum than other cephalosporins or glycopeptides, reducing the risk for development of resistance. It is very well tolerated, and its use avoids the risks of nephrotoxicity associated with vancomycin or the neurotoxicity associated with cefepime, and it is inexpensive. Allergy to cefazolin is rare, and there is little cross-reactivity between cefazolin and other beta lactams, so it can be used safely even in children with reported allergy, including anaphylaxis, to penicillins or other cephalosporins. Therefore, in children with PD-associated peritonitis confirmed to be caused by MSSA, we suggest transition to IP cefazolin alone for treatment.
Intraperitoneal vancomycin is the treatment of choice for MRSA infections. Use of teicoplanin is not recommended because its activity against MRSA biofilm is impaired in PD solutions. 224 Optimal dosing for IP vancomycin is not clear. Several studies, including those with pharmacokinetic modeling, have demonstrated significant inter-individual variability in serum levels of vancomycin with fixed IP dosing strategies. Previous iterations of this guideline recommended higher loading doses of IP vancomycin for patients undergoing continuous PD, but more recent literature has demonstrated that lower loading doses may be utilized to minimize potential toxicity.225,226 It is also unclear whether there is a role for modifying dosing based on therapeutic drug monitoring (TDM). Whereas the utility and optimal parameters for IP vancomycin TDM are not clearly defined, clinicians may consider TDM in consultation with pharmacy experts for some patients, including those at high risk of toxicity or with poor clinical response.
The 2012 ISPD pediatric peritonitis guidelines and the ISPD peritonitis guideline recommendations: 2022 update on prevention and treatment for adult patients, recommend 3 weeks of therapy for S. aureus peritonitis. The recommendation is based on limited retrospective data from two studies in adult patients with S. aureus peritonitis, which suggested better outcomes in patients treated with 3 weeks of therapy in one study compared to worse outcomes in another study in which patients received an average of 2 weeks of therapy.2,27,227,228 No definitive conclusions can be drawn from this comparison across studies that were conducted in different geographic locations (Hong Kong vs. Australia) and across different time periods (1994–2005 and 2003–2006). Importantly, data from the IPPN registry did not show a difference in outcomes for patients with S. aureus peritonitis treated with 2 vs 3 weeks of antibiotics. 168
A comparison of 106 patients with MSSA peritonitis who received antibiotic therapy for a median of 14 days (range 12–17) to 94 patients treated for a median of 21 days (range 19–24), revealed no difference in full functional recovery (p = 0.91), PD discontinuation (p = 0.83) or catheter removal (p = 0.97).
Among 73 patients with MRSA peritonitis treated with vancomycin maintenance therapy (42% intermittent IP, 25% continuous IP, 33% iv), full functional recovery was achieved in 95% of 22 episodes treated for a median of 14 days (range 12–17 days), as compared to 73% of 51 episodes treated for a median of 21 (range 19–24) days (p = 0.03). The relapse rate was similar in both groups (4% vs. 5%, p = 0.91).
While the data from IPPN are also limited by their retrospective nature, this does provide important evidence that 2 weeks of therapy appears to be safe and effective for children with both methicillin susceptible and methicillin resistant S. aureus peritonitis.
In a retrospective, observational single-center study of 245 S.aureus episodes that occurred in 152 adult patients, primary response rate was similar between patients with and without adjuvant rifampicin therapy (82.4 v. 89.8%; P = 0.11), as was the complete cure rate (77.9 versus 72.9%; P = 0.4). 227 However, adjuvant rifampicin treatment was associated with a significantly lower risk for relapse or repeat S.aureus peritonitis than was treatment without rifampicin (21.4 versus 42.8%; P = 0.004). Adjuvant rifampicin treatment resulted in a 49.9% relative risk reduction in relapse or repeat S. aureus peritonitis (95% CI 14.6 to 70.6%). The effect of rifampicin remained substantial even after exclusion of cases with early relapse (within 4 weeks after completion of antibiotics). Adjuvant rifampicin significantly reduced the risk for repeat peritonitis (23.3 v. 38.0%; P = 0.012).
Many enterococci are susceptible to ampicillin, which was the first-line recommendation for treatment of susceptible isolates in the 2012 guideline. However, newer data suggest that ampicillin's activity is significantly impaired by PD fluid and so IP administration of ampicillin is not recommended. 238 In turn, we suggest that treatment of ampicillin-susceptible enterococci consist of oral amoxicillin. In children who are unable to tolerate or absorb PO amoxicillin, IV ampicillin/amoxicillin may be used. When there are viable alternatives, we do not recommend vancomycin for treatment of ampicillin-susceptible enterococci due to a higher risk for colonization with vancomycin-resistant enterococci (VRE). For enterococcal isolates that are resistant to ampicillin but susceptible to vancomycin, the group recommends treatment with IP vancomycin.
Treatment of VRE can be challenging and so we encourage consultation with a local infectious diseases specialist in this case. Intraperitoneal daptomycin appears to have acceptable pharmacokinetics and there are case reports of successful treatment of VRE with IP daptomycin in adults.239,240 Both oral and IV linezolid appear to achieve adequate IP concentrations and have also been used successfully for treatment of VRE peritonitis, although IP administration of linezolid is not recommended due to similar concerns to ampicillin regarding impaired activity in PD fluid and unknown dosing regimens.241,242
The optimal duration of therapy for enterococcal peritonitis is not clear. While the ISPD peritonitis guideline recommendations: 2022 update on prevention and treatment for adult patients, recommend 3 weeks of therapy for enterococcal peritonitis, large case series of adults with enterococcal peritonitis reveal that most are treated with 2 weeks of antibiotics and there are no comparative data suggesting that 3 weeks is superior.234,235 In turn, we suggest treatment with antibiotics for 2–3 weeks, with longer durations considered for patients who are slower to improve.
Research Recommendations
● Additional research is needed to determine the optimal dosing strategy for intraperitoneal vancomycin and whether there is a role for therapeutic drug monitoring, either universally or in a subset of children.
● Further evaluation of oral amoxicillin vs. vancomycin or other strategies for Streptococcal peritonitis should be conducted.
Guideline 12 – Modification of Therapy for Gram-Negative Peritonitis
12.1 We suggest that peritonitis caused by gram-negative organisms that are not otherwise specified below (i.e., not Pseudomonas aeruginosa, Acinetobacter sp., Stenotrophomonas maltophilia, Enterobacter cloacae, Klebsiella aerogenes, Citrobacter freundii) and that do not have specified markers of resistance, extended-spectrum beta-lactamases (ESBL) or carbapenem-resistant Enterobacterales (CRE), be treated according to susceptibilities with either IP cefazolin or IP ceftazidime for a duration of 2 weeks (2D).
12.2 We suggest that peritonitis caused by Extended-spectrum beta-lactamase producing Enterobacterales (ESBL-E) be treated with IP or IV meropenem or IP or PO ciprofloxacin according to susceptibilities for a duration of 2 weeks (2D).
12.3 We suggest that peritonitis caused by organisms with high risk for AmpC beta-lactamase production, defined as Enterobacter cloacae, Klebsiella aerogenes, and Citrobacter freundii, be treated with IP cefepime if susceptible, or IP or IV meropenem or IP or PO ciprofloxacin if not, for a duration of 2 weeks (2D).
12.4 We suggest that peritonitis caused by Carbapenem-resistant Enterobacterales (CRE) be treated according to susceptibilities, in consultation with an infectious diseases expert, for a duration of 2 weeks (2D).
12.5 We suggest that peritonitis caused by Pseudomonas aeruginosa be treated according to susceptibilities with a single agent, either IP ceftazidime or cefepime if susceptible, or IP or IV meropenem or IP or PO ciprofloxacin if not, for a duration of 3 weeks (2D).
12.6 We suggest that peritonitis caused by Acinetobacter spp. be treated according to susceptibilities with a single agent, either IP cefepime, IP ceftazidime, or IP or IV meropenem, for a duration of 3 weeks (Not Graded).
12.7 We suggest that peritonitis caused by carbapenem-resistant Acinetobacter baumannii (CRAB) be treated according to susceptibilities with at least two active agents when possible, including high-dose IV ampicillin-sulbactam + IV or PO minocycline/tigecycline, IV polymyxin B, or IV cefiderocol, for a duration of 3 weeks. Consultation with an infectious diseases specialist is also suggested (Not Graded).
12.8 We suggest that peritonitis caused by Stenotrophomonas maltophilia be treated with PO trimethoprim-sulfamethoxazole in combination with another active agent, such as IV or PO levofloxacin, minocycline, tigecycline, or cefiderocol, for a duration of 3 weeks (Not Graded).
Rationale
Outcomes for gram-negative peritonitis tend to be worse than for gram-positive peritonitis. Previous analysis of data from the IPPR showed that full functional recovery was achieved in 86% of episodes of gram-negative peritonitis. 38 Treatment of gram-negative infections can be particularly challenging due to rising resistance and the numerous different mechanisms of resistance that can make antibiotic selection complex. Studies comparing different treatment strategies for specific gram-negative causes of peritonitis are lacking, so most recommendations are extrapolated from general principles for the management of gram-negative infections. (Figure 3) 244 As antibiotic resistance continues to increase worldwide, it is increasingly important to prioritize therapies that limit negative consequences (i.e., narrow-spectrum antibiotics, shorter treatment time) for the treatment of susceptible organisms, as long as these are not associated with inferior outcomes.
Although previously reported data from the IPPR cohort suggested that cefazolin monotherapy was associated with a higher risk of relapse as compared to treatment with a glycopeptide, ceftazidime or aminoglycoside monotherapies, the published analysis did not specify the type of causative bacteria. 37 In addition, the reason for inferior outcomes with cefazolin for treatment of organisms that are in vitro susceptible, might be attributed to the previously advocated lower dosing of cefazolin that we now recognize to be inadequate. This association did not hold true in more recent analyses in patients receiving higher doses of cefazolin. Given the risk for development of resistant organisms (ESBL, AmpC-producing bacteria) with use of a third generation cephalosporin, cefazolin use for the treatment of susceptible gram-negative bacteria is a reasonable option. 245 In addition, cefazolin is well-tolerated and inexpensive, in addition to having a narrow spectrum of activity making it less likely to induce broader antimicrobial resistance.
The suggested duration of therapy for most gram-negative organisms, with the exceptions of Pseudomonas aeruginosa, Acinetobacter baumanii, and Stenotrophomonas maltophilia, is 2 weeks. Data from the IPPN registry found no difference in full functional recovery from peritonitis in cases of non-Pseudomonas gram-negative peritonitis in patients treated with 2 vs. 3 weeks of antibiotics.
168
Among 140 patients treated for a median of 14 (range 12–17) days, 91% experienced full functional recovery versus 88% recovery among 106 patients receiving therapy for a median of 21 (range 19–24) days (p = 0.45). When performing a multivariate analysis controlling for age, presence of preceding exit-site infection, severity of symptoms at presentation and early treatment response, there was still no difference in full recovery between patients treated with short vs. long durations of therapy.
168
Prior versions of this guideline and current guidelines for
ESBLs are enzymes that inactivate most penicillins, cephalosporins, and aztreonam. Most ESBL-E remain susceptible to carbapenems and non-beta lactams (fluoroquinolones, TMP/SMX, aminoglycosides), although some may also have additional mechanisms of resistance that lead to broader resistance. ESBLs are most common in Escherichia coli, Klebsiella pneumoniae, Klebsiella oxytoca, and Proteus mirabilis, although they can be found in any gram-negative organism. Most clinical laboratories do not perform ESBL testing, so non-susceptibility to ceftriaxone is used instead as a proxy for ESBL production. While some organisms may be resistant to ceftriaxone for reasons other than ESBL production, Enterobacterales that are resistant to third generation cephalosporins (ceftriaxone, cefotaxime, or ceftazidime) should be presumed to be ESBL-E. 244
A large randomized clinical trial in adults found higher mortality in patients with ESBL-E bacteremia treated with piperacillin/tazobactam compared to meropenem. 250 IDSA guidelines recommend extending the preference for carbapenems for treatment of serious infections with ESBL-E to other sites, including intraabdominal infections, particularly for initial treatment. Intraperitoneal administration is preferred, and with optimal dosing for intermittent administration in children unclear, this would require continuous administration. If this is not possible, intravenous administration can be used.
Oral or IP fluoroquinolones can be used as step-down therapy in patients who have shown clinical improvement, or may be reasonable options for first-line treatment in children who are not critically ill.251,252 Both ciprofloxacin and moxifloxacin appear to be compatible with PD solutions and to have bactericidal activity in PD fluid.238,253,254 Fluoroquinolones are highly orally bioavailable and thus when given orally, can achieve adequate IP concentrations.255,256 If given orally, it is important to note that fluoroquinolone absorption is impaired by calcium-, magnesium-, aluminum-, zinc-, or iron-containing products (eg, antacids, dairy products, tube feeds) as well as phosphorus binders, and so administration of fluoroquinolones should be timed to be several hours before or after receipt of these products. Fluoroquinolones have been used extensively in children with very rare reports of musculoskeletal adverse events, primarily tendinitis and tendinopathy that is generally short-term and reversible.257–259 Observational data in adults suggest fluoroquinolones may be associated with an increased risk of aortic aneurysm or dissection.260–264 Unless no other treatments are available, fluoroquinolones should be avoided in children known to have an aortic aneurysm or who have risk factors for aneurysm, such as Marfan syndrome or Ehlers Danlos syndrome. 265
There is no evidence to suggest that resistant infections require longer durations of treatment than susceptible ones, as long as they are ultimately treated with an effective antibiotic. 244 Since these patients may have been started on empiric treatment with an antibiotic to which their isolate was ultimately found to be non-susceptible, it is recommended that they complete 2 weeks of treatment with an effective agent.
For the three groups of organisms with the highest risk for inducible AmpC expression, E. cloacae, K. aerogenes, and Citrobacter freundii, even if in vitro susceptibility testing indicates susceptibility to third generation cephalosporins, these antibiotics should be avoided. Intraperitoneal cefepime is recommended because it is a weak inducer of AmpC beta lactamases and can withstand hydrolysis by AmpC beta lactamases. For organisms that are resistant to cefepime, the workgroup recommends use of either IP or IV carbapenem or an IP or PO fluoroquinolone. Carbapenems are also resistant to hydrolysis by AmpC beta lactamases and fluoroquinolones are not susceptible to beta lactamases.
Knowledge of a specific CRE isolate's mechanisms of resistance, including whether it produces a carbapenemase and, if so, which carbapenemase, is critically important for determining optimal treatment. 244 For example, the most common carbapenemases in the United States are K. pneumoniae carbapenemases (KPCs), which can be found in many Enterobacterales in addition to K. pneumoniae. Novel beta lactam/beta lactamase inhibitor combinations such as ceftazidime-avibactam, meropenem-vaborbactam, and imipenem-cilastatin-relebactam are active against most Enterobacterales that produce KPC enzymes.
Because management of carbapenem-resistant infection is complex and nuanced and may vary according to local susceptibility patterns and antimicrobial availability, consultation with an infectious diseases expert is recommended. In general, beta lactam/beta lactamase inhibitor combinations with activity against the isolated organisms are preferred over aminoglycosides and colistin due to improved efficacy and decreased toxicity with such regimens. 244
Combination therapy with cefepime or ceftazidime and a second agent that has a different mechanism of action (for example, fluoroquinolone or aminoglycoside) and to which the bacteria is susceptible was advocated in the 2012 pediatric ISPD guidelines and treatment with 2 agents is also recommended in the ISPD peritonitis guideline recommendations:
In contrast, a more recent analysis of Pseudomonas aeruginosa peritonitis episodes reported to the IPPN registry between 2012 and 2020 revealed no difference in outcome between one- and two-agent therapy. 168 Out of 70 episodes, 29 were treated with one anti-pseudomonas agent (ceftazidime, gentamicin, fluoroquinolone or cefepime). The full functional recovery rate was 79%, as compared to 56% in patients treated with two anti-pseudomonas agents (p = 0.04). Patients who received two anti-pseudomonas agents presented with more severe signs and symptoms of infection at 60–72 hours of empiric therapy and more commonly had a preceding ESI (26 vs. 21%). However, multivariate analysis showed no difference in outcome between the groups after adjusting for age, disease severity score, 60–72-hour treatment response and treatment duration.
This is in line with IPPR data from 2008, where although the outcome of infections caused by Pseudomonas spp. tended to be least favorable compared to other pathogens, it was influenced independently by the concurrent presence of an exit-site infection and the initial response to treatment. 38 This is also consistent with data from gram-negative infections of other body sites that show no benefit associated with double coverage with multiple antibiotics, including infections secondary to Pseudomonas aeruginosa. 279 Current guidance from the IDSA recommends against combination therapy, even for highly resistant Pseudomonas aeruginosa. 244 If there is suspicion of an extremely resistant infection based on patient history or local susceptibilities, an aminoglycoside and beta lactam may be considered as combination therapy initially to broaden coverage and increase the likelihood that at least one agent will cover the organism. Once susceptibility to a beta lactam is confirmed, there is no evidence that continued therapy with two agents improves outcomes. 279 Also noteworthy, combination therapy is associated with an increased risk for adverse events, particularly kidney injury and potential loss of residual kidney function.
Considering the possible risks of adverse events and additional costs associated with combination treatment and the lack of discernible benefit, the balance between desirable and undesirable effects favors definitive treatment with one anti-pseudomonas agent, preferably a beta lactam when possible. We suggest treatment with IP ceftazidime or cefepime if susceptible, or IP or IV meropenem if not. If, once antimicrobial susceptibilities are available, there are no options for IP beta lactams for treatment, PO fluoroquinolones may be a reasonable alternative, especially for stepdown therapy after initial improvement.
Importantly, recent updates to The United States Clinical Laboratory Standards Institute (CLSI) recommendations have eliminated breakpoints for gentamicin based on a review of clinical and pharmacokinetic/pharmacodynamic data showing that there is no safe aminoglycoside dosing regimen that can achieve adequate killing of P. aeruginosa. Gentamicin has therefore been removed as a treatment option for P. aeruginosa. 280 Although the CLSI review focused on systemic treatment rather than IP administration of antibiotics, these changes will impact reporting of susceptibilities for bacteria grown from peritoneal fluid as clinical laboratories are recommended to no longer report P. aeruginosa susceptibility to gentamicin, and to only report amikacin for urine isolates. Beta lactams are preferred for treatment of P. aeruginosa, including novel beta lactam/beta lactamase inhibitors for highly resistant P. aeruginosa isolates. If an aminoglycoside must be used due to lack of susceptibility to a beta lactam, IP tobramycin is preferred over gentamicin or amikacin and may be considered in combination with a beta lactam in consultation with an infectious diseases specialist.
The suggested length of therapy for Pseudomonas aeruginosa peritonitis is 3 weeks. In the IPPN database, full functional recovery was achieved in 75% of patients with Pseudomonas peritonitis treated for a median of 21 (range 19–24) days and in 61% of those treated for a median of 14 (range 12–17) days (p = 0.28). 168 Although the difference was not statistically significant due to low patient numbers, there was a trend towards worse outcomes in those treated with shorter durations of therapy. In light of this, as well as the generally poor outcomes associated with Pseudomonas aeruginosa peritonitis compared to other etiologies, including other gram-negative organisms, the workgroup favors 3 weeks of therapy.
We recommend consultation with an infectious diseases specialist for peritonitis caused by carbapenem-resistant Acinetobacter baumannii (CRAB), which can be very difficult to treat. It often displays resistance not just to carbapenems, but also to other agents that may be active against wild-type A. baumannii. In concordance with IDSA guidance, we suggest that treatment regimens should include combination therapy with two active agents. 244 While it is not clear as to whether combination therapy results in better outcomes when compared to treatment with a single agent, combination therapy is suggested because data identifying the optimal single agent to use for treatment are lacking. The high degree of resistance of most CRAB isolates means that in general, if patients are not on appropriate therapy initially, they may in turn have a high burden of disease by the time CRAB is identified, and CRAB may develop resistance to antibiotics during the course of treatment.
We also suggest that regimens include ampicillin/sulbactam, even if the isolate is resistant to ampicillin/sulbactam in vitro. Sulbactam is a potent beta lactamase inhibitor that saturates PBP1z/1b and PBP3 of A. baumannii and has unique activity against A. baumannii. Even if an isolate is resistant to ampicillin/sulbactam, the sulbactam may saturate altered PBP targets and allow the second agent to be more effective. Multiple studies have demonstrated that sulbactam-containing regimens are associated with lower mortality in critically ill patients with CRAB. The optimal dosing of sulbactam in pediatric patients undergoing PD with an active CRAB infection is unknown and should be further discussed with local infectious diseases and pharmacy experts. When used for treatment of CRAB, ampicillin/sulbactam should be given in combination with minocycline, tigecycline, polymixin B, or cefiderocol.
There were too few episodes of Acinetobacter baumannii peritonitis in the IPPN database to draw any conclusions about the optimal duration of therapy for Acinetobacter spp. Therefore, we have extrapolated the 3-week duration recommended for Pseudomonas aeruginosa to Acinetobacter spp.
TMP-SMX has historically been the preferred treatment for S. maltophilia infections and most isolates are susceptible to TMP-SMX (>90% in the US), although there is increasing recognition of resistance.281,282 Existing data, which are limited to small retrospective studies or observational studies using administrative databases, conflict as to the optimal approach to treatment (eg. combination vs. monotherapy).283–286 For serious infections, IDSA guidance recommends combination therapy to ensure that at least one antibiotic will be active. Despite the lack of robust data, we suggest inclusion of PO TMP-SMX in combination antibiotic treatment strategies due to the abundance of clinical experience with its use for S. maltophilia and no clear evidence of lack of efficacy. We suggest treatment with two agents initially, with consideration of using PO TMP-SMX alone for step down therapy in patients who have exhibited marked clinical improvement or in those with relatively mild infection. If TMP-SMX cannot be used due to resistance, toxicities, or allergy, we recommend the use of combination therapy with two other active agents.
As with Acinetobacter spp., there are no data to identify optimal duration of therapy for S. maltophilia peritonitis; we recommend a treatment duration of 3 weeks due to its propensity to form biofilms, extrapolated from the recommended treatment duration for P. aeruginosa.
Research Recommendations
● Since multicenter, controlled studies on gram-negative peritonitis treatment schemes are usually not feasible in pediatrics due to very low patient numbers per center and country, more information should be recovered from international pediatric dialysis registries.
● Further research into the pharmacokinetics and feasibility of intraperitoneal use of carbapenems and fluoroquinolones is needed to help meet the increasing demand for treatment of peritonitis caused by resistant gram-negative organisms.
● Additional research is needed comparing outcomes in patients treated with a single agent versus two agents with different mechanisms of action for Pseudomonas aeruginosa.
Guideline 13 – Modification of Therapy for Culture-Negative Peritonitis
13.1 We suggest that antibiotic therapy for culture negative, non-relapsing peritonitis be continued for 2 weeks (2D).
13.2 We suggest that antibiotic therapy be switched to cefazolin monotherapy if the initial PD cultures remain sterile at 72 h and signs and symptoms of peritonitis are improved (2D).
Rationale
There are no published pediatric data regarding the optimal treatment duration for culture-negative peritonitis. An analysis of 289 culture-negative peritonitis episodes reported to the IPPN database revealed a detrimental effect of shortening treatment duration below 14 days. The PD discontinuation rate was 15% for 48 peritonitis episodes treated for shorter than 14 days as compared to 5% for 241 episodes treated for at least two weeks (p = 0.008, adjusting for age and disease severity). 168 In light of the above data, it appears safe to continue the initial empiric therapy, which provides gram-positive and gram-negative coverage, for a complete treatment course (at least 14 days) to potentially lower the risk of treatment failure and relapse. (Figure 4)
In centers in which the culture-negative peritonitis rate exceeds 15% of peritonitis episodes, sampling and culture techniques should be reviewed with the dialysis staff and the laboratory. 27
Research Recommendations
● Since randomized controlled studies comparing different treatment durations for culture-negative peritonitis are presumably not feasible, more information should be recovered from international pediatric peritonitis registries on the treatment and outcome of culture-negative infections.
● Further research should be conducted to determine if improved culture strategies result in a decrease in the percentage of culture-negative peritonitis episodes.
Guideline 14 – Modification of Therapy for Fungal Peritonitis
14.1 We suggest immediate catheter removal when fungal elements are identified in PD effluent (2D).
14.2 We suggest antifungal therapy be administered for at least 2 weeks after catheter removal in children with PD-associated fungal peritonitis (2D).
14.3 Clinicians should select an appropriate agent based on antifungal susceptibility results, with oral fluconazole preferred for infections caused by susceptible Candida species (Not Graded).
Rationale
Fungal peritonitis (FP) is rare, accounting for 2–8% of episodes of peritonitis episodes in children, but is associated with poor outcomes, including high rates of PD discontinuation.155,158,159 The two largest series of pediatric fungal peritonitis, from NAPRTCS and SCOPE, demonstrated worse outcomes for FP relative to non-fungal peritonitis; however, in contrast to the adult experience, there does not appear to be a significant increase in mortality associated with FP in children relative to bacterial peritonitis. Retrospective case series have demonstrated mortality rates associated with FP in children of 0–2.6%, compared to 9–28.7% in adults.155,158,159,289–291 (Figure 5
There are conflicting data regarding the optimal timing for catheter removal. In the NAPRTCS series in which 90% of children underwent catheter removal, 17% were removed within one day of diagnosis and 39% were not removed for four or more days after diagnosis. 155 There was no association between conversion to HD and time of catheter removal following the diagnosis of FP. In three observational studies in adults that evaluated the association between timing of catheter removal and mortality, two found that removal after 24 hours was associated with increased mortality, while the third found no difference in mortality between patients with catheter removal within the first 5 days compared to later removal.289,290,292 The conflicting results may be explained by different time frames defining early vs. late removal.
As noted above, whereas multiple studies in adults with FP have demonstrated an association between failure to remove the PD catheter or delayed removal (more than 24 hours after diagnosis) and mortality, these studies were observational and were not controlled for confounding by indication, i.e., the fact that patients who are sicker and more likely to fail treatment are also more likely to be unable to have their catheter removed or to have their catheter removed later.
The choice of antifungal agent should be driven by species identification and susceptibility testing. The vast majority of fungal infections are due to Candida spp., many of which can be effectively treated with oral fluconazole. Fluconazole is highly orally bioavailable and achieves excellent IP concentrations. 294 For organisms that are not susceptible to fluconazole, including filamentous fungi such as Aspergillus spp., voriconazole, posaconazole, and isavuconazole may be chosen, generally in consultation with an infectious diseases specialist. When possible, IV voriconazole and posaconazole should be avoided in PD patients due to the risk of accumulation of cyclodextrin, which may contribute to kidney toxicity. PO voriconazole and posaconazole achieve adequate IP concentrations and are appropriate alternatives in children with adequate absorption. 295 Intravenous echinocandins (micafungin, caspofungin, anidulafungin) may be used for fluconazole-resistant Candida spp. Intraperitoneal administration of echinocandins is discouraged due to concerns about impairment of activity against Candida biofilm by PD fluid.296,297 IV amphotericin B does not penetrate the peritoneum well and is generally reserved for infections that are not susceptible to azoles or echinocandins. 294 Intraperitoneal administration of amphotericin is poorly tolerated with significant peritoneal irritation and abdominal pain.
Research Recommendation
● Further research dedicated to the optimal treatment strategy for management of FP in children is needed. Because catheter removal has become the standard of care, there is likely not equipoise to conduct a prospective study evaluating retention of the PD catheter compared to removal. However, a larger retrospective series could provide some answers regarding the need and optimal timing for catheter removal in children.
Guideline 15 – Mycobacterial Peritonitis
15.1 We suggest that antituberculosis therapy without PD catheter removal be provided as the primary treatment of peritonitis caused by Mycobacterium tuberculosis (2D).
15.2 We suggest that non-tuberculous mycobacteria (NTM) be treated with both effective antibiotics and catheter removal (2D).
Rationale
The classical clinical presentation is a refractory, culture-negative peritonitis that may or may not be associated with an exit-site infection.303,304,306,308 In such cases, one should have a high-index of suspicion, and the laboratory should be notified to prolong the incubation time of standard bacterial cultures to 7 days, and/or use selective culture media such as Lowenstein-Jensen medium. 303 Consultation with an infectious diseases physician with expertise in the diagnosis and management of mycobacterial disease is recommended. Identification of NTMs at the species level is important because bacterial susceptibility to antimicrobial drugs is often closely predicted from characterization of the isolated species. 302
Most NTMs are resistant to standard anti-tuberculous drugs and the majority of antimicrobials, with the exception of amikacin.303,304,312 The majority of patients require catheter removal as most NTMs have a tendency to exhibit growth in biofilms. 304
A previous study had reported topical gentamicin for exit-site care as a potential risk factor for acquiring NTM peritonitis; however, a more recent report failed to validate that association.304,313
Research Recommendations
● Global registry data should be collected to determine the frequency, clinical manifestations and treatment of children with mycobacterial peritonitis.
Guideline 16 – Relapsing, Recurrent and Repeat Peritonitis
16.1 Following an initial episode of peritonitis, subsequent episodes should be characterized as relapsing, recurrent or repeat to help determine a treatment plan and prognosis (Not Graded).
16.2 We suggest that empiric antibiotic therapy of relapsing/repeat peritonitis be guided by the center specific protocol with consideration of the susceptibilities of the original bacteria (2C).
16.3 Post-empiric antibiotic therapy of relapsing/repeat peritonitis should be guided by in vitro susceptibility results (Not Graded).
16.4 We suggest that the duration of antibiotic therapy for most episodes of relapsing/repeat peritonitis be based on the standard treatment recommendations for the identified organism (2C).
16.5 We suggest that removal of the PD catheter should occur as soon as peritonitis is controlled by antibiotic therapy in the setting of relapsing/repeat peritonitis associated with a persistent or recurrent tunnel infection, or a second peritonitis relapse (2C).
Rationale
Terminology for peritonitis.

Relapsing peritonitis follows approximately 5–20% of primary peritonitis episodes in adult and pediatric series.48,157,314 The largest pediatric experience with relapsing peritonitis has come from the IPPR where out of 490 episodes of non-fungal peritonitis, 52 were followed by a relapse, for a relapse rate of 11%. 37 Recurrent peritonitis is less common than relapsing peritonitis. A multicenter registry study of over six thousand adult PD patients showed that an episode of peritonitis occurring within 4 weeks of completion of treatment of a prior episode is 2 times more likely to be a relapse than a recurrence (14% vs. 6%). 314
Relapsing, recurrent and repeat peritonitis episodes may be caused by different spectra of bacteria.314–318 The diagnosis of a relapse / repeat peritonitis episode should not rely solely on the genus and species, but also on the antibiotic susceptibilities of the cultured organism. When available, strain identity can be confirmed by genotype analysis. Gram-positive organisms, Staphylococcus aureus and coagulase-negative Staphylococci, are more commonly responsible for episodes of relapsing and repeat peritonitis.314,316,317 In the IPPR experience, there was no significant difference in the distribution of causative organisms between cases of relapsing peritonitis and non-relapsing peritonitis. Overall, relapsing episodes consisted of 46% gram-positive organisms, 21% gram-negative organisms and 33% culture-negative cases. 37
The most recent data from the IPPN registry shows that in 217 (8.9%) of 2425 peritonitis episodes, PD had to be discontinued. Slightly more than one-quarter (25.8%) of the latter cases were either recurrent (n = 22; 10%), repeat (n = 20; 9%) or relapsing (n = 14; 6.4%) episodes. Additionally, while the incidence of recurrent peritonitis (5%) was half that of relapsing or repeat episodes, the rate of PD discontinuation was more than double (19.6%) following recurrent peritonitis compared to relapsing or repeat infections. 168
Post-empiric antibiotic therapy should be guided by in vitro susceptibility results. Based on prior IPPR data, the subsequent prescription of monotherapy with a first-generation cephalosporin based on culture and susceptibility results was associated with a higher relapse rate (23% vs. 0 to 9%; p = 0.02) compared to other antibiotic choices. 37 However, it is speculated that these inferior results associated with cefazolin monotherapy could have resulted from the use of intermittent vs. continuous therapy, as well as suboptimal antibiotic dosing in that cohort of patients. Nonetheless, it is suggested that there be close monitoring of clinical response in patients with relapsing peritonitis being treated with cefazolin monotherapy.
In contrast, in a retrospective evaluation of 232 peritonitis episodes in adults caused by coagulase negative Staphylococci (CoNS), Szeto et al. found that compared with the conventional 2-week treatment course, 3 weeks of antibiotic therapy for relapsing or repeat episodes was associated with a significantly higher complete cure rate (83.3 vs 46.7%; p = 0.047). 232 However, data from the IPPN failed to show similar results in children as there was no statistical difference between 15 episodes of coagulase negative Staphylococcus (CoNS) relapsing peritonitis treated for a median of 2 weeks compared to 20 episodes treated for a median of 3 weeks (80% vs. 73%; p = 0.64).
Research Recommendations
● Additional research should determine if extending (from 2 to 3 weeks) the therapy for relapse/repeat peritonitis secondary to coagulase-negative Staphylococci (CoNS) in children results in improved outcomes.
Guideline 17 – Adjunctive Therapy
17.1 We suggest that the use of IVIG be considered for adjunctive treatment in selected patients with repeated peritonitis episodes and/or associated sepsis and accompanying hypogammaglobulinemia
17.2 We suggest that cauterization using silver nitrate sticks, in addition to intensified local care with antiseptics (other than with povidone-iodine) and/or a local antibiotic cream be used for treatment of isolated exit-site granulation tissue
17.3 Systemic antibiotics should not be used for treatment of isolated granulation tissue at the catheter exit site without features of infection
Rationale
The role of low serum IgG levels as a potential risk factor for the development of peritonitis in patients receiving PD was evaluated in different case series involving infants and children with discrepant results. Although Neu et al. did observe a relationship between low serum IgG levels and the development of peritonitis, others did not.324–327 The only available longitudinal study, which included a small sample size, demonstrated that the mean serum IgG levels of infants who developed peritonitis (n = 8) versus those who did not (n = 18) were not different (p = 0.39). 327 An adult prospective study showed that low serum IgG levels at the time of PD initiation predicted the occurrence of PD related peritonitis. 328 The mean IgG level at PD start was lower in patients who subsequently experienced peritonitis and an increased IgG level at PD onset was associated with a reduced risk of peritonitis. 328 Significantly elevated IgG levels after IVIG therapy is evidence of the effectiveness of IVIG in correcting hypogammaglobulinemia in infants receiving PD. 327
Despite the scarcity of evidence, there are several other adjunctive therapies that may be considered. Bacteria trapped in biofilm are less susceptible to antibiotics, such that substantially higher concentrations (100–1000 times) of antibiotics are required to eradicate them. 329 Additionally, viable bacteria may also be engulfed by and persist in peritoneal mesothelial cells. 314 In turn, fibrinolytic agents (alteplase or urokinase) have been tried as a treatment designed to expose trapped bacteria within fibrin or biofilms to the action of the antibiotics. Adult randomized trials showed inconsistent results regarding the beneficial effect of adjunctive urokinase over standard antibiotic treatment in the management of refractory or relapsing peritonitis.322,330–333 Two randomized controlled trials in adults failed to show any benefit of IP urokinase in the treatment of refractory or relapsing peritonitis in terms of the rates of complete cure, catheter removal, relapsing episodes, or overall mortality when compared to standard antimicrobial treatment..330,331 However, one showed a benefit of urokinase in peritonitis resolution and the prevention of recurrences. 332 In particular for CoNS, intraluminal urokinase 100,000 IU for 2 hours and oral rifampicin 600 mg daily for 3 weeks, in addition to conventional antibiotics, resulted in a catheter salvage rate of 64%. 334 Similarly, the beneficial effect of adjunctive t-PA (6 mg/mL for 6 hours, multiple times) was observed in the treatment of repeated peritonitis episodes due to CoNS in four patients. 335 Another randomized trial, however, showed that simultaneous catheter removal and replacement was superior to IP urokinase in reducing relapsing peritonitis episodes. 322 Despite limited data, a Cochrane review concluded that for relapsing or persistent peritonitis, simultaneous catheter removal and replacement was better than urokinase at reducing treatment failure rates. 323
In the only pediatric study, Klaus and colleagues successfully used intraluminal high-dose urokinase (5000 IU/mL) and antibiotic instillation in nine children with relapsing peritonitis. No second relapse occurred in the treated patients. In contrast, 75% of patients from an untreated historical control group experienced a second relapse. 333 In practice, installation of t-PA several times per week during peritonitis treatment to prevent recurrence of peritonitis in children with suspected biofilm may allow PD continuation without having to replace the catheter (B Begin, personal communication). However, its net effect in relapsing peritonitis to prevent treatment failure or persistent/recurrent infection remains unknown.
Thus, although there is no fully convincing pediatric data, prior to proceeding to catheter replacement, a trial of a fibrinolytic agent as part of the management of the first relapsing peritonitis episode not caused by fungi, Pseudomonas aeruginosa and S. aureus, and not explained by extraluminal pathology such as a tunnel infection or intra-abdominal abscess, may be considered.
Both urokinase and t-PA may be cheaper relative to the cost of catheter replacement. Regulations in the country and/or specific institution, availability of the agents, and insurance coverage may impact the decision regarding usage.
Several other adjunctive measures in the management of peritonitis include IP heparin, initial low fill volume for pain relief, analgesic usage, and icodextrin.
Heparin presumably has an inhibitory effect on fibrin clot formation and also has effects beyond anticoagulation, having antiangiogenic and antiinflammatory properties. 336 Although hypercoagulability and hypofibrinolysis were shown during CAPD peritonitis in a small pediatric patient group, it was not confirmed in a larger adult study.337,338 Whereas the rationale for routine IP heparin (500 units/L IP) use in the treatment of peritonitis is not strong because of limited clinical evidence, it is often prescribed as an adjunctive option to prevent occlusion of the catheter with fibrin clot in severe peritonitis with turbid dialysate.
The fill volume can be slightly (<25%) lowered during the initial 24–48 h of therapy until clinical symptoms improve and then should subsequently be increased to the standard prescription to prevent a prolonged period of underdialysis. Some patients require analgesics for pain control. Pain not improved by alteration of the fill volume and the initiation of antibiotic therapy, or complicated by emesis and volume depletion, may mandate hospitalization. 2
In the recent adult guideline, icodextrin use was suggested as a means to control volume overload during acute peritonitis. Since peritoneal permeability typically increases during peritonitis and reduced ultrafiltration may result in fluid overload, temporary use of hypertonic fluids or icodextrin solution during acute peritonitis have been suggested as potential interventions. In a randomized controlled study, the primary cure rate of peritonitis was similar between the icodextrin and original glucose-based dialysis solution treatment groups. However, volume status was better controlled in the icodextrin group. 339
In a pilot trial in adults, 44 patients with exit-site granulation tissue were equally and randomly allocated to receive treatment with 2% aqueous chlorhexidine swabstick or silver nitrate cauterization. After 6 weeks of follow-up, both agents had similar success rates. Whereas use of the chlorhexidine swabstick was associated with a longer time to resolution of the granulation tissue (13 vs 33 days), both treatments were well tolerated by patients in terms of pain, burning sensation and skin discoloration. 340
The presence of granulation tissue over the exit site without other features of infection does not typically require systemic antibiotic treatment. Intensified local care is usually effective, although local application of an antibiotic cream has also been recommended. 85
There is scarce data on children pertaining to the management of exit-site granulation tissue. In one case series, 34 patients used antibacterial honey as a prophylactic agent in routine exit-site care. The effect of topical antibacterial honey as it relates to exit-site granulation tissue was evaluated in a subgroup comprised of 8 patients aged 6 months to 15 years. Although improvement in poor exit-site conditions was reported, only two of 8 patients had isolated exit-site granulation tissue; one was treated solely with medicinal honey and the other with medicinal honey and silver nitrate. 115
Research Recommendations
● In patients with repeated peritonitis episodes or in those with signs of sepsis, the use and efficacy of IVIG as an adjunctive agent to antibiotics should be evaluated.
● Further studies are needed to determine the long-term efficacy of fibrinolytic agents in treating and preventing relapsing peritonitis in pediatric PD patients.
● Future studies should evaluate the use of chlorhexidine swabstick as an alternative to silver nitrate to treat exit-site granulation tissue in children.
Guideline 18 – Catheter Removal and Replacement
18.1 We suggest removal of the PD catheter for refractory bacterial peritonitis (2D).
18.2 We suggest removal of the PD catheter following a diagnosis of fungal peritonitis (2D).
18.3 We suggest PD catheter removal in patients with an exit-site or tunnel infection in conjunction with peritonitis with the same bacteria (2D).
18.4 We suggest a minimum period of 2–3 weeks between PD catheter removal and insertion of a new catheter following fungal and refractory bacterial peritonitis (2D).
18.5 We suggest simultaneous removal and replacement of the PD catheter be considered after clearing of the peritoneal effluent (white blood cells < 100/mm3) following a second episode of relapsing bacterial peritonitis (2D).
18.6 We suggest simultaneous removal and replacement of the PD catheter be considered for management of a refractory exit-site or tunnel infection (2D).
Rationale
Indications for PD catheter removal.

The suggestion that a cut-off of 5 days be used when making a decision regarding PD catheter removal should be considered as being somewhat arbitrary. Data from the IPPN registry showed that catheter removal at <7 days (vs. > 7 days) was associated with a greater likelihood of PD continuation (100% vs. 91%; p = 0.05). The trajectory of the effluent white cell count and clinical course during the first five days of treatment should also be taken into consideration before making the decision to remove the catheter. There is substantial variation in the rate at which the PD effluent WBC count declines after antibiotic treatment initiation.345,346 In a large observational study of 644 peritonitis episodes in adults, one-fifth of the cases showed a delayed response with a 34% reduction of effluent WBC count by day 5, and without the need for PD catheter removal. 346 Accordingly, the 2022 ISPD adult guidelines suggest, and we agree, that if the WBC count of the PD effluent is decreasing towards normal by day 5, PD catheter removal could be postponed. 27 On the other hand, the catheter should be removed sooner than 5 days if the patient's condition is deteriorating.
While removal of the PD catheter in patients with refractory peritonitis will necessitate initiation of hemodialysis in most patients, this approach is necessary to prevent excess morbidity and mortality, and to possibly mitigate the possibility of long-term peritoneal membrane damage resulting from prolongation of ineffective therapy.
As referred to above, although catheter removal should be considered in most cases when an exit-site and/or tunnel infection and peritonitis occur secondary to the same organism, the same aggressive approach might not be necessary when a catheter-related infection and peritonitis is attributable to coagulase negative Staphylococcus (CoNS). CoNS peritonitis is generally milder and is usually readily responsive to antibiotic treatment.184,232
Re-insertion of a new catheter should ideally be performed using a laparoscopic or mini-laparotomy approach so that adhesions can be directly visualized and addressed during the procedure. In all cases, recommendations regarding the duration of antibiotic therapy and the timing of catheter replacement may require modification based on the clinical response of the patient.
The removal and replacement procedure has been performed successfully in pediatric and adult patient populations.39,352–355 In data collected by the Italian pediatric PD registry, simultaneous removal and replacement procedures were successfully performed in 76% of catheter removals. 67 In an adult study, simultaneous catheter removal and replacement was successful in all 22 patients with relapsing peritonitis. 354 In a small, randomized study, a recurrence rate of only 7% was associated with the use of this approach to treat recurrent CoNS and culture-negative peritonitis. 322 However, this procedure should not be attempted in patients with fungal peritonitis, active or refractory peritonitis, or with intra-abdominal adhesions.348,356
Regardless of the indication for simultaneous catheter removal and replacement, antibiotic therapy should be continued for 2–3 weeks after the procedure.353–355 Most clinicians have chosen to insert the new catheter before removing the old catheter, but the alternative approach has been performed with similar results.353–355
It is noteworthy that simultaneous catheter removal and replacement is recommended even for treatment of an ESI or tunnel infection secondary to P. aeruginosa that persists or progresses despite prolonged and appropriate antibiotic treatment. In a series comprised of 37 adult patients with refractory P. aeruginosa exit-site infection, simultaneous catheter removal and insertion of a new PD catheter without interruption of PD was successful in all patients. 357 Late recurrence of P. aeruginosa exit-site infection occurred in only 8% of the patients within the first year after the procedure. 357
The advantages of the simultaneous removal and replacement of a PD catheter and the recommended post-procedure management for patients receiving CAPD are the same as discussed under Guideline 18.5.
Research Recommendations
● Data should be collected to help determine the optimal timing of catheter removal in patients with refractory peritonitis as it relates to preservation of peritoneal membrane function.
● Future research should be carried out to help define the optimal duration between catheter removal and insertion of a new catheter when simultaneous removal and replacement is contraindicated.
Guideline 19 – Diagnosis of Catheter-Related Infection
19.1 We suggest that catheter-related infection (exit-site and tunnel) rates, including organism-specific rates, be separately reported (Not Graded).
19.2 We suggest that an objective scoring system be used to monitor the status of the PD catheter exit site (2D).
19.3 We suggest that a PD catheter insertion-related exit-site and/or tunnel infection be defined as an episode of exit-site or tunnel infection that occurs within 30 days of PD catheter insertion (Not Graded).
19.4 We suggest that a definitive diagnosis of a catheter exit-site infection be made in the presence of purulent discharge at the catheter-epidermis interface (2C).
19.5 We suggest that a tunnel infection be defined by the presence of redness, edema, and tenderness along the subcutaneous portion of the catheter with or without ultrasonographic evidence of a pericatheter fluid collection and with or without purulent drainage from the exit site (2C).
Rationale
A SCOPE collaborative report showed that cultures were obtained in 83% of ESIs, with 92% of cultures growing bacterial isolates. Methicillin-susceptible S. aureus (MSSA) was the most common organism (35%), followed by Pseudomonas spp. (18%). Methicillin-resistant S. aureus (MRSA) occurred in 6.5% of isolates. Culture-negative cases have accounted for 8–11% of infections in pediatric and adult series.35,358 Catheter-related infection rates should be determined as the organism-specific number of ESI or TI episodes divided by the number of patient years at risk, reported as episodes per patient year. Time at risk begins immediately after PD catheter insertion, irrespective of whether or not PD is being performed. Organism-specific reporting will help guide treatment strategies including empiric antibiotic selection, and will facilitate continuous quality improvement activities. 45
Exit-site scoring system. 183
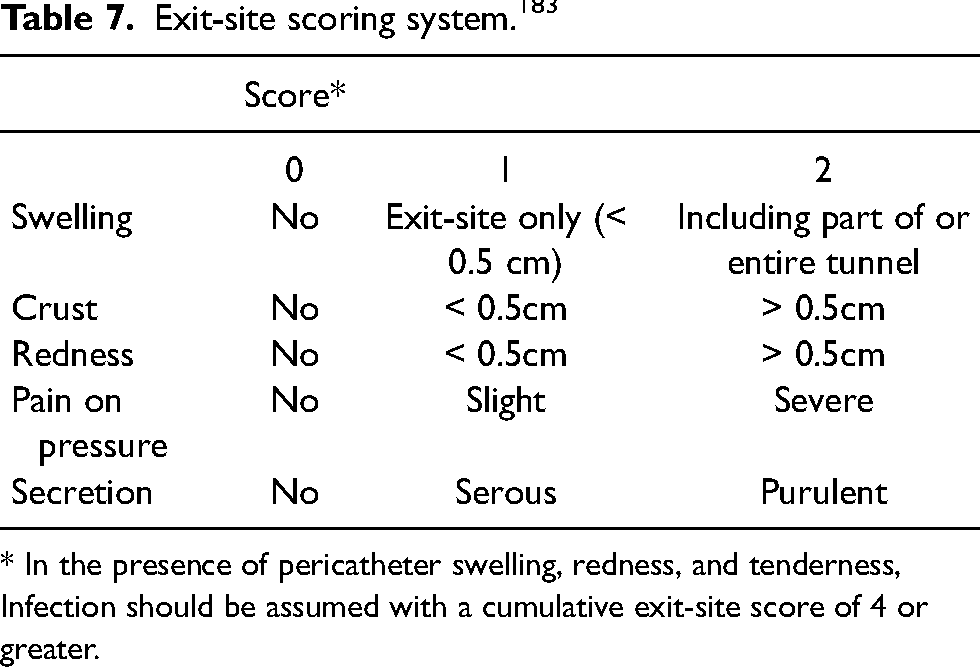
* In the presence of pericatheter swelling, redness, and tenderness, Infection should be assumed with a cumulative exit-site score of 4 or greater.
On the other hand, the scoring system outlined by Twardowski et al. is based on photo images. Also, recent pediatric data from the SCOPE Collaborative demonstrated poor reproducibility of both exit-site scoring systems when used simultaneously by two experienced care providers, especially when at least one provider characterized the exit site as abnormal. 362 Agreement and disagreement rates were similar when using either the Schaefer et al. or the Twardowski et al. criteria. This has prompted recognition of the need for additional educational strategies targeted to providers to improve exit-site characterization, both by in person visual inspection and by commonly used smartphone photographs. Thus, any scoring system must complement —but not substitute for— clinical judgment. Despite its limitations, use of a scoring system continues to be suggested for regular monitoring of the exit site and for monitoring patient response to therapy.
Evaluation of 35, 354 exit-site assessments during the first year of dialysis in 3297 adult patients from 122 institutions revealed that the agreement between the diagnosis of an ESI based on purulent discharge and the clinical score comprised of hyperemia, edema, pain, scab, and granuloma was 60.6%. These data support the concept that the presence of purulent drainage is sufficient to diagnose an ESI, while the use of scoring systems using other clinical signs does not contribute a great deal more to the diagnosis, but provides a resource for longitudinal assessment of the exit- site appearance. 364 With more than 10,000 patient-months of follow-up data in children on PD, the SCOPE collaborative showed that a higher exit- site score at a routine follow-up visit was strongly associated with a subsequent ESI within 8 weeks. The percentage of visits that had a subsequent ESI increased from 0.8% to 7.4% if the exit site was scored 3, compared to 0 (9.25 times increase). 35
In the presence of an ESI, ultrasound evaluation allows for the diagnosis of a concomitant TI with greater sensitivity than by clinical parameters alone. A hypo/anechoic collection with a diameter of >1–2 mm, and a color Doppler signal located between the catheter wall/cuff and the surrounding tissues, supports the diagnosis of a TI. 365 In addition, ultrasound evaluation may help to more accurately distinguish TIs that will likely resolve with oral antibiotic therapy from those that may require a more aggressive therapeutic approach including intravenous antibiotic therapy, surgical revision of the tunnel or removal of the catheter.365,366
In children, 6–12% of ESIs attributable to S. aureus and P. aeruginosa are associated with a concomitant TI and subsequent peritonitis related to the catheter infection; in those cases, treatment of the catheter-related infection should be particularly aggressive, because the associated peritonitis episode can be resistant to treatment.35,38 These particular organisms may also form a biofilm in the catheter, thereby precluding successful antibiotic management without catheter removal. 367
Research Recommendations
● Studies should be conducted to assess the diagnostic accuracy of scoring systems in pediatric PD patients following targeted education of care providers.
● Strategies should be developed to improve the early detection of ESIs (i.e., by using automated image analysis and machine learning approaches).
● Prospective studies should assess the usefulness of ultrasonography to diagnose and follow catheter-related infections in pediatric patients.
Guideline 20 – Treatment of PD Catheter-Related Infection
20.1 We suggest empiric oral antibiotic treatment for PD catheter exit-site infection (ESI) with appropriate methicillin-susceptible S. aureus (MSSA) coverage, such as a first-generation cephalosporin or anti-staphylococcal penicillin, unless the patient has a prior history of infection or colonization with methicillin-resistant S. aureus (MRSA) or Pseudomonas spp.; in these cases we suggest the use of vancomycin (or clindamycin if prior isolates are susceptible) or an antipseudomonal antibiotic, respectively. Treatment should subsequently be modified as required based on culture results and susceptibilities
20.2 We suggest that in most cases, effective antibiotic treatment be continued for 7–14 days depending on the clinical response and microbiological results
20.3 We suggest that an exit-site infection caused by Pseudomonas spp. be treated with at least 3 weeks of effective antibiotic therapy
20.4 We suggest that an exit-site infection caused by Nontuberculous Mycobacteria (NTM) be treated in consultation with an infectious diseases specialist
20.5 We suggest that empiric oral antibiotic therapy with methicillin susceptible S. aureus (MSSA) coverage be initiated for treatment of a tunnel infection, unless the patient has a history of infection with MRSA or Pseudomonas spp., in which case empiric therapy should consist of IP vancomycin or an IP, oral or IV antipseudomonal antibiotic, respectively
20.6 We suggest that effective antibiotic therapy be administered for at least 3 weeks for any catheter tunnel infection
20.7 Refractory catheter-related infection should be defined as an infection that has failed to resolve after 2 weeks of effective antibiotic therapy and intensified exit-site care
20.8 We suggest that surgical salvage interventions or catheter removal be conducted as part of the treatment for refractory catheter-related infections
20.9 We recommend catheter removal if a catheter-related infection progresses to or occurs in conjunction with peritonitis due to the same organism
Rationale
Successful treatment of a catheter-related ESI is important because treatment failure may lead to catheter removal or peritonitis. In most cases, oral antibiotic therapy is effective (Table 8).45,370,371 The ideal approach is to tailor the choice and duration of treatment based on susceptibility results and clinical response.
Oral antibiotic recommendations for treatment of exit-site and tunnel infections.

May consider reducing to 300 mg Q12hr after 72 hours if a prolonged duration (>10 days) is anticipated and patient is at risk for development of thrombocytopenia based on limited data.410,411
Should not be used as monotherapy, or used routinely in areas in which tuberculosis is endemic.
The empiric antibiotic regimen should primarily cover MSSA, favoring a first-generation cephalosporin or an antistaphylococcal penicillin. If the patient has a prior history of infection or colonization with MRSA, vancomycin (or clindamycin if known to be susceptible) can be used. To prevent development of resistance and vancomycin-associated adverse events, glycopeptides should be reserved for MRSA infections and not be used for routine treatment of ESIs secondary to other gram-positive bacteria. Despite the absence of high-quality evidence, oral rifampin may be added to the treatment of S. aureus infections that fail to improve or resolve promptly. Oral rifampin should never be given as monotherapy. In the case of a prior history of infection or colonization with Pseudomonas spp., antipseudomonal antibiotics are appropriate. Concomitant antifungal prophylaxis should be considered to mitigate the risk of fungal peritonitis.27,45 (see Guideline 7)
Therapy should be continued until the exit site appears completely normal. The duration of antibiotic administration should balance the risk of recurrence due to incomplete bacterial eradication against the potential emergence of resistant strains from unnecessarily long antibiotic exposure. The recommended treatment duration should count from the day of initiation of effective antibiotic therapy based on susceptibility results. High quality data to guide optimal treatment durations are lacking. Consistent with IDSA guidelines for skin/soft tissue infections, 372 recent guidelines for management of ESIs in adults suggest adjusting the treatment duration based on clinical response; 7–10 days of effective antibiotic therapy is reasonable in most uncomplicated ESIs, except for those caused by P. aeruginosa and nontuberculous mycobacteria (NTM), if resolution of infection is confirmed by clinical evaluation at around 1 week following treatment initiation.45,372 Data from a survey conducted by the Japanese Study Group of Pediatric Peritoneal Dialysis revealed that among 130 patients less than 15 years of age, the post-treatment relapse rate was 15%; the relapse rate was 40% among infections caused by MRSA. 373 Therefore, we suggest at least 7 days of effective post-empiric antibiotic treatment in uncomplicated ESIs, if there is a good clinical response. If there is a lack of resolution after 7 days of therapy, administration of the effective antibiotic for 2 weeks is reasonable (Figure 6).
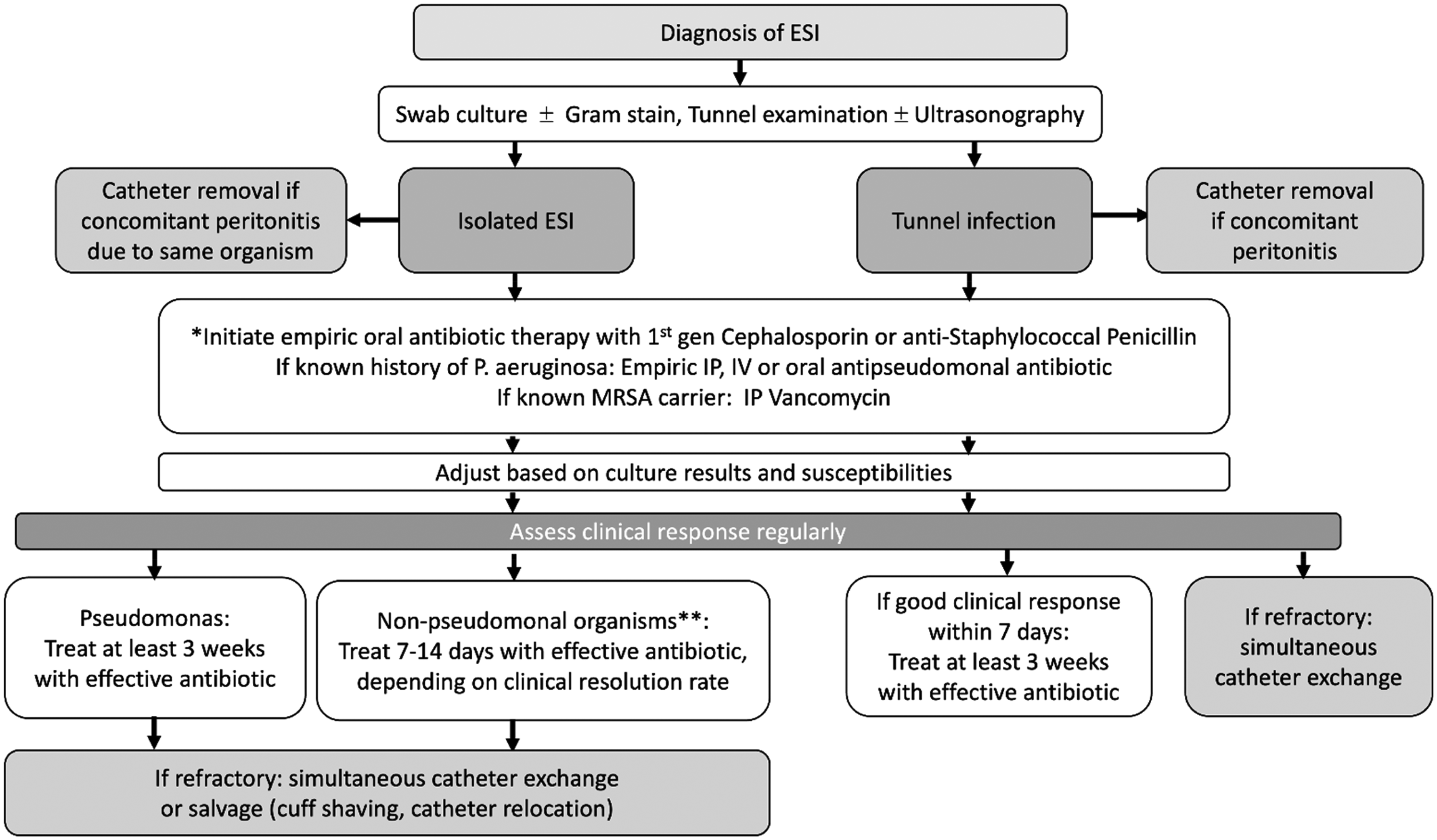
Management algorithm for PD catheter exit-site and tunnel infections.
Treatment of an ESI will also likely benefit from an increased frequency of exit-site care and at least daily ESI inspection and cleansing as long as purulent discharge continues; however, there is no evidence-based data to support this practice. This approach does facilitate early recognition of worsening infection, including the development of a tunnel infection, and review of the need for additional therapeutic options. Granulomas over the exit site may require additional exit-site care because they are associated with exacerbated breakdown of the exit site.
In cases without inner cuff involvement and without accompanying peritonitis, catheter salvage interventions can be attempted. 368 Adult studies have shown that simultaneous removal and reinsertion of the peritoneal dialysis catheter in the opposite quadrant is an acceptable surgical intervention with negligible recurrences rates when conducted for treatment of a refractory ESI or TI (see Guideline 18.6). The rate of ESI refractoriness to conservative treatment leading to catheter removal is highest in cases caused by Mycobacteria (up to 40%), S. aureus (35%), and P. aeruginosa (28%). 380 Patients with an ESI due to P. aeruginosa and those with tunnel infections may benefit from being kept on antibiotics for 1–2 weeks post catheter salvage procedure to decrease the risk for a relapse.354,357,381
Catheter removal is suggested when catheter infection occurs simultaneously with peritonitis due to the same organism, with no attempted reinsertion of the PD catheter until at least 2 weeks after catheter removal and complete resolution of peritonitis. Catheter salvage procedures should not be attempted in these cases.
Research Recommendations
● Adequately powered trials to better define the optimal treatment durations for exit-site and tunnel infections by specific microorganisms.
● Assessment of minimum waiting times for catheter reinsertion following removal due to a refractory exit-site or tunnel infection.
● Contribution of point-of-care ultrasound assessment for monitoring treatment response to antibiotic treatment of tunnel infection.
Guideline 21 – Evaluation of Primary Response
21.1 We suggest that in addition to visual inspection of the dialysis effluent for cloudiness, an objective standardized measure, such as the Disease Severity Score, be used to monitor clinical response at 72 hours after initiation of antibiotic therapy
Rationale
In some cases, use of objective, standardized response criteria can be helpful to avoid unnecessary premature changes of treatment and to rather promptly detect an insufficient treatment response. As noted in Guideline 8.2, a prospective randomized study of 168 episodes of peritonitis in 152 pediatric PD patients used a Disease Severity Score (DSS) to monitor clinical response. 183 The DSS was defined as the sum of points for pain and fever (Table 9) and the study demonstrated excellent agreement between improvement in the DSS score and final patient outcome. More recently, the IPPN has evaluated a modified DSS associated with 1060 episodes of peritonitis at 3 days after treatment initiation, in which along with assessment of effluent cloudiness, 0–2 points could be given for abdominal pain and fever, respectively. A good clinical response was characterized by a DSS <=2 and clear effluent, while complete resolution was associated with a DSS = 0 with clear effluent. Full functional recovery, as defined by continuation of PD without functional impairment, occurred in 88.6% of episodes with a good 3-day response vs. 76% of those with a poor 3-day response (p < 0.0001). 168
Disease severity score. 183

* Recent DSS calculations by IPPN based on 0–2 points for pain and 0–2 points for fever
Research Recommendation
● Data should be collected regarding the relationship between treatment modifications based on the 3-day DSS and patient outcome.
Guideline 22 – Monitoring of Infection Rates and Risk Factor Assessment
22.1 Every program should monitor the incidence and outcomes of peritonitis and exit-site infection on at least an annual basis to inform quality improvement activity
22.2 The parameters monitored should include the PD-related peritonitis and exit-site infection rates, organism-specific peritonitis and exit-site infection rates, antimicrobial susceptibilities of the infecting organisms, and peritonitis outcomes
22.3 The rate of peritonitis and exit-site infections should be reported as the number of episodes per patient-year
22.4 Culture-negative peritonitis should be reported as a percentage of all peritonitis episodes per unit time
22.5 An apparent cause analysis (ACA) should be conducted following each peritonitis episode
22.6 We suggest that the overall peritonitis rate should be no more than 0.4 episodes per year, the proportion of culture-negative peritonitis episodes should be less than 15% of all peritonitis episodes and < 5% of PD catheter insertions should be followed by peritonitis within 30 days of catheter insertion
Rationale
SCOPE has also determined overall annualized peritonitis rates by age and found them to be 0.40 based on 342 infections in children < 2 years, and 0.37 based on 1191 infections in children 2 years of age and greater. The IPPN found an annualized rate of 0.32 based on 2183 infections in children > 2 years of age. Thus, like the ISPD peritonitis guideline recommendations: 2022 update on prevention and treatment for adult patients, we suggest that the overall peritonitis rate be no more than 0.40 episodes per year at risk. 27
The ISPD Catheter-related Infection Recommendations: 2023 Update for adult patients state that the proportion of PD catheter insertion related infections within 30 days of PD catheter insertion should be less than 5% of all catheters inserted. 45 When this frequency is exceeded, the surgical and medical teams should closely evaluate operative and post-operative management, ideally incorporating quality improvement principles. Data collected from SCOPE also revealed that less than 5% of PD catheter insertions were followed by peritonitis within 30 days, prompting a similar benchmark for pediatric PD programs, and the recommendation for similar actions should the benchmark be exceeded. 389
Research Recommendations
● Further research should be conducted to better define risk factors for peritonitis and potential mitigation strategies through the assessment of ACAs.
Supplemental Material
sj-pdf-1-ptd-10.1177_08968608241274096 - Supplemental material
Supplemental material, sj-pdf-1-ptd-10.1177_08968608241274096
Footnotes
Acknowledgements
The workgroup wishes to thank biostatisticians Troy Richardson, PhD and Natalie Grills for their assistance in accessing data from the SCOPE collaborative. The authors would also like to thank Kristy Peto for her secretarial assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Appendix – PD Modality and Antibiotic Dosing
Challenges exist with selecting IP antibiotic dosing regimens for patients undergoing APD, as the majority of pharmacokinetic studies for IP antibiotics were derived from patients undergoing CAPD. 390 Various pharmacokinetic and pharmacodynamic properties of the antibiotic may be impacted during APD resulting in underexposure to antibiotics. For example, shorter dwell times and more frequent exchanges utilized during APD result in increased antibiotic clearance and shorter half-lives. For time-dependent antibiotics, such as beta-lactams, this may result in less-than-optimal antibiotic exposure as the antibiotic concentrations will not remain above the minimum inhibitory concentration (MIC) for an adequate portion of the dosing period. 390 For vancomycin, the duration of the dwell time may influence the dose of vancomycin provided as longer dwell times will allow for more complete peritoneal absorption compared to shorter dwell times. Patients with more rapid exchanges may require more frequent vancomycin dosing. 391 Overall, there is limited guidance for dosing of peritoneal antibiotics in pediatric patients undergoing APD, but dwell times, frequency of exchanges, and pharmacokinetic/dynamic properties of the antibiotic should be considered. Research is needed to explore whether necessary modifications are required (e.g., antibiotic concentration modifications to those with low fill volumes or with residual kidney function) to ensure optimal treatment.
Appendix B – Drug Delivery and Stability
Stability and compatibility of antimicrobials in PD solution is an important factor when considering treatment options (Table 10). Drug stability is determined by the degradation of the drug in solution, which is influenced by temperature and humidity and should generally be less than 10% for a drug to be considered stable. 392 Data suggest that many antibiotics are stable for variable times when added to available dialysis solutions. Vancomycin (25 mg/L) is stable for 28 days in dextrose-based dialysis solution stored at room temperature, although higher ambient temperatures will significantly reduce the duration of stability to just 1 day. Gentamicin (8 mg/L) is stable in dextrose-based and icodextrin-based solutions for 14 days at a wide range of temperatures, but the duration of stability is reduced by admixture with heparin. Conversely, the stability of gentamicin in pH-neutral PD solutions is <24 hours at room temperature. Cefazolin (500 mg/L) is stable in dextrose-based solutions for at least 8 days at room temperature, or for 14 days refrigerated; addition of heparin has no adverse influence. Ceftazidime is less stable; concentrations of 125 mg/L are stable for 4 days at room temperature in dextrose-based solutions and 2 days in icodextrin-based or pH neutral solutions. Cefepime is stable in dextrose-based dialysis solution for 14 days, if the solution is refrigerated. 393 These data are derived from duration-of-stability studies. However, it is important to recognize that because of significant differences in the constitution of current PD solutions in terms of buffers, osmotic agents, and pH, stability data cannot be extrapolated from one PD solution to another. 394
Compatibility studies of multiple drugs in PD solutions supports the admixture of gentamicin with vancomycin or cefazolin, but aminoglycosides should not be combined with penicillins due to chemical incompatibility.(Table 11) Newer studies have demonstrated that meropenem added to dextrose-based PD solutions achieves longer stability under refrigeration when heparin is also added to the solution.395,396 The addition of heparin to PD solution has also been studied individually with cefazolin, ceftazidime and vancomycin and compatibility was evident in all cases. 397
Additional studies are needed to confirm the stability of antibiotics in various PD solutions at the concentrations used and the temperatures encountered (37 °C) in clinical practice to ensure optimal outcomes. Information is also needed on newer agents that will be necessary to treat the emergence of resistant organisms that may no longer respond to agents with known stability data.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
