Abstract
The development of biodegradable films with enhanced functional performance is essential for replacing petroleum-based packaging materials. This study investigates polyvinyl alcohol (PVA) biofilms reinforced with coconut shell powder (CSP), a lignocellulosic agro-waste, fabricated through a water-based solution casting process. The incorporation of CSP was evaluated for its influence on mechanical, thermal, moisture-resistance, and biodegradation characteristics. Tensile testing showed a substantial improvement in performance, with tensile strength increasing from 54 MPa for neat PVA to 83.6 MPa at 6 wt% CSP, accompanied by an increase in elastic modulus from 1.73 GPa to 2.14 GPa and improved elongation at failure. Multiscale SEM fractography suggested the presence of crack deflection, particle pull-out, and fibrillation, which may contribute to enhanced energy dissipation during fracture. XRD results indicated increased crystallinity with CSP addition, likely due to heterogeneous nucleation, while FTIR spectra showed intensified hydrogen bonding and improved interfacial interactions. Thermal analysis revealed an upward shift in degradation onset and Tmax, indicating partial thermal stabilization of the matrix. Water absorption decreased from 6.4% to 1.5%, and soil biodegradation increased from 10.8% to 21.3% over 30 days, suggesting that CSP may facilitate matrix breakdown through its lignocellulosic nature. The findings demonstrate that CSP can function as an effective reinforcement for PVA films, offering enhanced structural integrity and improved environmental responsiveness for potential packaging applications.
Keywords
Introduction
The global surge in plastic consumption has intensified the need for sustainable packaging solutions that reduce ecological impact without compromising performance. Polyvinyl alcohol (PVA) has emerged as a frontrunner among biodegradable polymers owing to its water solubility, film-forming ability, optical clarity, and non-toxicity. However, its inherent limitations, such as high moisture sensitivity, moderate mechanical strength, and limited thermal resistance, necessitate structural modifications for practical applications in food packaging.
To overcome these limitations, a wide array of natural additives, reinforcements, and crosslinking strategies have been explored. Pınar Terzioğlu et al. demonstrated that integrating orange peel powder into chitosan/PVA films enhanced thermal stability, flexibility, UV-blocking, and antioxidant activity, although transparency and hydrophobicity were compromised. 1 Song Jiang et al. reported that citric acid crosslinked chitosan/PVA films achieved superior mechanical strength, shape recovery, and water vapor barrier properties due to improved hydrogen bonding. 2 Similarly, Ishrat Gowsia et al. observed that the addition of piperic acid to PVA films significantly increased tensile strength and water barrier performance, with enhanced antimicrobial activity against fungal strains, albeit at the cost of reduced elongation. 3
A focus on agro-waste valorization has emerged as a sustainable route for improving film properties while addressing environmental burdens. Kirtiraj K. Gaikwad et al. utilized apple pomace to reinforce PVA, reporting improved antioxidant activity and thermal stability. 4 However, tensile strength and transparency declined with increasing filler content. Romina P. Ollier et al. employed micro-cellulose and nano-bentonite as co-reinforcements, which significantly improved strength and reduced water absorption. 5 Andreas A. Sapalidis et al. confirmed similar improvements in thermal and mechanical performance with the addition of Zostera flakes to PVA, without impacting water sorption, further supporting their potential in low-cost biodegradable packaging. 6
Hybrid biopolymer films have also received considerable attention. Yuanjian Xie et al. developed crosslinked cellulose/PVA films that exhibited over 200% improvement in tensile strength and nearly doubled elongation at break, while maintaining low oxygen permeability and strong humidity resistance, attributes that are critical for food preservation. 7 Aanchal Mittal et al. demonstrated that palmitic acid-grafted barley husk enhanced water resistance and barrier properties of urea-formaldehyde crosslinked PVA/starch films. 8 Patricia Cazón et al. found that blending PVA with cellulose and glycerol increased toughness, burst strength, and water adsorption, while retaining optical clarity. 9
Reinforcements with antimicrobial functionality have gained traction. Zohreh Riahi et al. incorporated sulfur-functionalized montmorillonite into chitosan/PVA films, leading to substantial increases in mechanical properties and complete UV-B blocking. 10 Sujan Chowdhury et al. embedded gold nanoparticles and graphene oxide in crosslinked PVA, which reduced water vapor transmission and improved antimicrobial activity, extending banana shelf life. 11 Alessandro Musetti et al. prepared PEG-crosslinked PVA films with grapefruit seed extract, achieving high transparency, moisture resistance, and antibacterial efficacy against Salmonella and Listeria strains. 12
To expand film functionality, other researchers explored lignin, essential oils, and quaternary polymers. Shihui Wang et al. integrated lignin into starch–PVA films, boosting tensile strength, antioxidant activity, and UV shielding. 13 Qingyan Pan et al. formulated edible NPCS–PVA films with cinnamon essential oil, improving strength, barrier performance, and antibacterial effects, ultimately extending mango shelf life. 14 Fengsong Liu et al. incorporated quaternary chitosan into PVA films, achieving remarkable elongation at break (507.09%), tensile strength above 39 MPa, and reduced UV transmittance, suitable for active antibacterial packaging. 15
Nanocomposite films incorporating clays and nanocrystals have also shown promise. Jen-Yi Huang et al. created clay–PVA films that drastically reduced oxygen transmission, outperforming commercial materials in preserving tomato paste under fluctuating humidity.
16
Yongzhen Lei et al. introduced catechol-functionalized chitosan into PVA, enhancing tensile strength by 46.3% and antibacterial action.
17
Maoli Yin et al. used SBMA-modified chitosan in PVA films, achieving strong antibacterial efficacy against
Vanillin and bio-polyols have been used to functionalize PVA composites. Shivayogi S et al. showed that ethyl vanillin enhanced tensile strength and hydrophobicity while lowering vapor and gas transmission rates in chitosan/PVA films. 19 Huiyu Bai et al. achieved significant reductions in water absorption and vapor permeability by using UV-crosslinked CNC-grafted matrices in PVA. 20 Fanrong Meng et al. used banana pseudo-stem-based bio-polyol to enhance PVA films, which improved elongation and thermal stability, supported by FTIR and SEM data. 21
Cellulose nanocrystals have been widely used for structural reinforcement. Lina Meng et al. reported that quaternized CNCs improved tensile strength and antibacterial activity. 21 Chenwei Chen et al. added silica aerogels to reduce thermal conductivity and vapor transmission, supporting thermal insulation in food packaging. 22 Jiazhuo Xie et al. showed that small amounts of layered double hydroxides led to significant gains in tensile strength and reduction in haze, confirming efficiency at low filler loads. 23
Color and pH-responsive films have also emerged. Wenjie Long et al. used Aronia extract in chitosan/PVA films for UV blocking and antioxidant activity, with intelligent pH responsiveness.
24
Tilak Gasti et al. used
Biodegradability and water resistance are often addressed together. Wenli Pan et al. used high-amylose starch and citric acid to increase water contact angle from 30.95° to over 100°, indicating strong hydrophobicity. 27 Weijie Lan et al. prepared tea polyphenol–PVA composites that minimized microbial spoilage in strawberries. 28 Jianfu Chen et al. created PVA/xanthan gum films that fully degraded in soil and water within 12 hours, representing one of the fastest degradation cycles in biofilm literature. 29
Innovative designs using carbon dots, melanin, and nanoparticles have expanded the functional landscape. Mingxuan Hou et al. synthesized carbon dot–PVA–CMC films that enhanced strength, UV shielding, and fruit preservation. 30 Di Liu et al. showed that oxidation-tuned graphene oxide improved strength and barrier performance in PVA films. 31 Kalyani Prusty et al. used nano-ZrO2 in PEMA/PVA systems to increase dielectric properties and barrier behavior for semiconducting packaging. 32 Reaz A. Chowdhury et al. employed roll-to-roll printing to create CNC–PVA coatings with high transparency and low vapor permeability. 33 Xiaochun Bian et al. integrated visible-light-responsive nanoparticles in PVA–CA nanofiber films for ethylene degradation and antimicrobial activity. 34 Yanzhen Zhao et al. created AgNP–PVA films with sustained antibacterial action and safe silver release levels. 35 Finally, Mohaddeseh Kariminejad et al. used nano-silicon dioxide to improve crystallinity and water barrier performance in gelatin–PVA composites. 36
Despite this rich body of work, several key challenges remain. Many systems rely on highly processed, synthetically modified, or costly nanofillers, which limit scalability. The mechanical failure behavior, especially tensile fracture mechanisms, remains underexplored, despite its importance in determining film durability during use. Moreover, although some agro-waste materials like apple pomace and barley husk have been tested, the potential of coconut shell powder (CSP), a widely available, lignocellulosic by-product of the coconut industry, remains largely untapped in the context of PVA reinforcement.
This study differs from earlier PVA–lignocellulosic films in several ways. Earlier works mainly focused on barrier or antimicrobial performance and reported only limited insights into fracture behavior. The present work examines tensile fracture mechanisms in detail, supported by multiscale SEM fractography. Minimally processed coconut shell powder is used instead of chemically treated fibers or nanocellulose. Its higher lignin content and irregular particle morphology may promote crack deflection, particle pull-out, and local energy dissipation. These effects might also influence crystallinity and moisture diffusion. The study links these microstructural features with changes in strength, stiffness, elongation, thermal response, and biodegradation. This combined analysis offers a more complete understanding of how lignocellulosic particulates modify the functional behaviour of PVA films.
Coconut shell powder offers distinct structural features that may set it apart from other lignocellulosic fillers reported in PVA systems. Its high lignin fraction, rigid microgranular morphology, and lower hemicellulose content may influence deformation and fracture in a different manner than softer plant fibres. The irregular particle geometry of CSP may promote crack deflection and promote tortuous fracture paths, which could contribute to the higher elongation observed in this study. This behaviour differs from fillers such as rice husk or wood flour, where fibre pull-out often dominates and restricts ductility. The dense carbonaceous structure of CSP may also influence the multifunctional performance, as the lignin-rich domain can increase char formation, enhance thermal resistance, and modify moisture transport pathways. These combined effects may produce a balance of stiffness, toughness, and environmental response that is not typically reported in other PVA–lignocellulosic systems. The present work therefore contributes by linking these CSP-specific characteristics with fracture behaviour, thermal response, water transport, and biodegradation trends in a single integrated framework.
This study addresses critical gaps in sustainable packaging materials by developing coconut shell powder (CSP)-reinforced polyvinyl alcohol (PVA) biofilms. The novelty lies in the use of minimally processed CSP as a multifunctional reinforcement, which not only enhances the mechanical and barrier properties of the films but also promotes environmental degradability. Unlike conventional approaches that rely on costly nanoparticles or plant extracts, CSP provides a low-cost, scalable, and eco-friendly alternative, owing to its abundance of hydroxyl and phenolic groups that form strong interactions with PVA’s polar matrix. Specifically, the research aims to synthesize and characterize PVA/CSP biofilms with varying filler loadings; evaluate their mechanical performance, focusing on tensile strength, modulus, and elongation; investigate fracture mechanisms at both macro and microscale to understand failure behavior; analyze water absorption and thermal stability under ambient and elevated conditions; and assess biodegradation in soil to confirm environmental applicability. Through this comprehensive investigation, the study presents a promising route for agro-waste valorization, offering high-performance, biodegradable films suitable for eco-friendly packaging solutions.
Materials and methods
Materials
Physio-chemical properties of PVA.

The chemical compatibility of PVA with bio-based reinforcements stems from its abundant hydroxyl moieties, which facilitate molecular-level interaction and filler dispersion. Additionally, its high water solubility (5.2 g/100 mL at 25°C) and film-forming capacity allow for uniform incorporation of particulate reinforcements without the need for organic solvents. The polymer exhibits excellent clarity, non-toxicity, and barrier properties, making it ideal for eco-friendly packaging applications where biodegradability and mechanical resilience are critical. In this work, coconut shell powder (CSP) was used as a natural particulate reinforcement. Its selection was based on its lignocellulosic composition and surface reactivity, both of which promote interfacial compatibility with the PVA matrix, leading to improved composite performance.
Preparation of coconut shell powder (CSP)
Coconut shell powder (CSP), utilized as the reinforcement phase in this study, was prepared through a precisely controlled two-stage mechanical processing sequence aimed at producing fine, reactive particulates compatible with polymer matrix dispersion as shown in Figure 1. Initially, mature coconut shells were collected and thoroughly washed to eliminate adhering organic matter. This was followed by oven drying at 105°C for 6 hours to reduce the moisture content below 1%, a critical prerequisite to avoid microbial contamination and thermal decomposition during further processing. Preparation of coconut shell powder (CSP) from raw coconut shells using crushing and ball milling processes.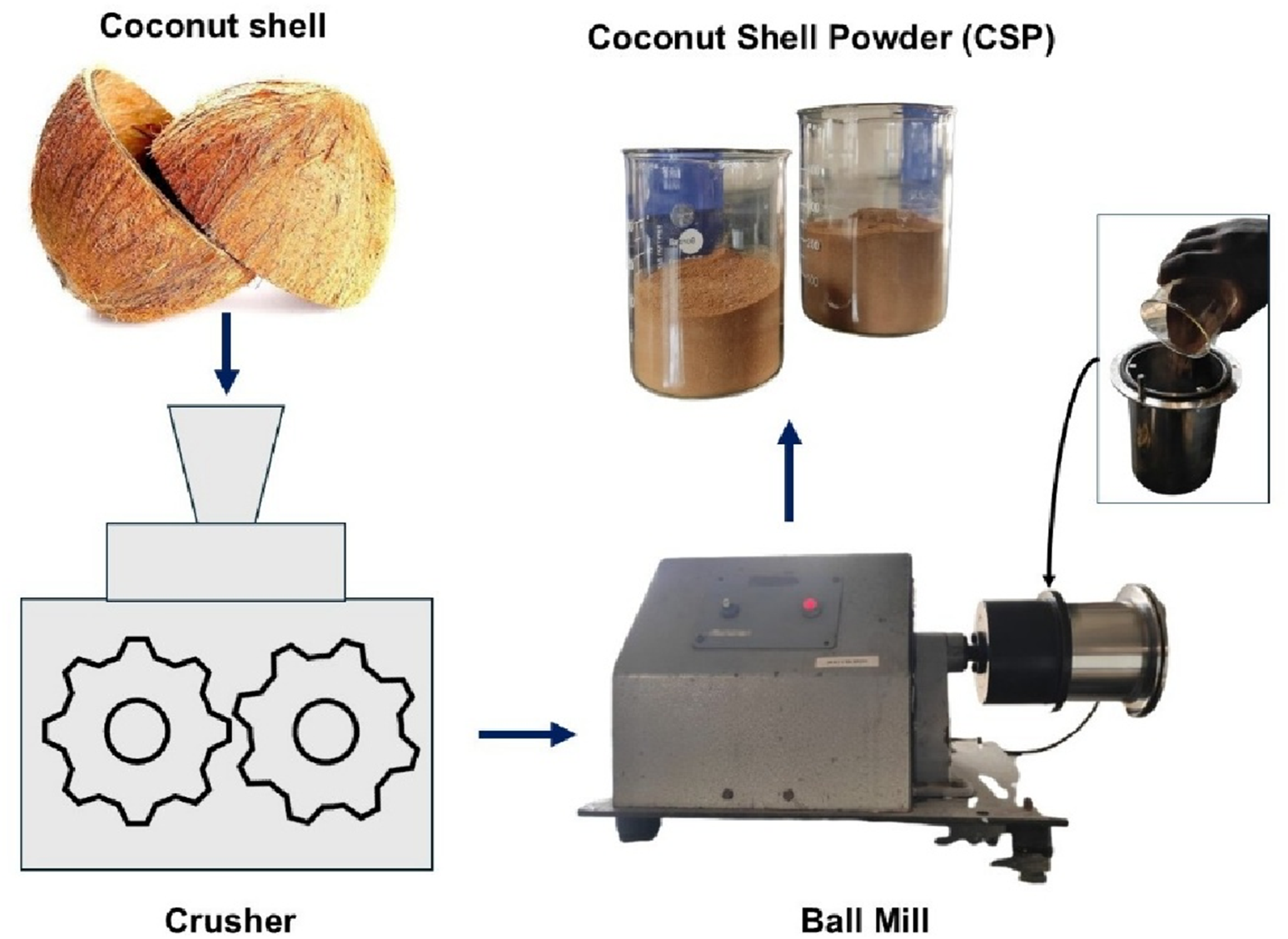
The dried shells were subjected to size reduction using a mechanical crusher, producing coarse flakes of 2–5 mm. These intermediate fragments were then processed in a high-energy planetary ball mill operating at 400 rpm for a total duration of 8 hours, using a ball-to-powder weight ratio of 10:1. Stainless steel milling media with a 10 mm diameter were selected to ensure hardness compatibility and avoid contamination. Intermittent milling cycles (20 minutes on, 10 minutes off) were employed to prevent excessive heat generation that could degrade the lignocellulosic structure. This stage effectively reduced the particle size to an average of 18–22 µm, confirmed via Dynamic Light Scattering as represented in Figure 2. Particle size distribution of coconut shell powder by dynamic light scattering.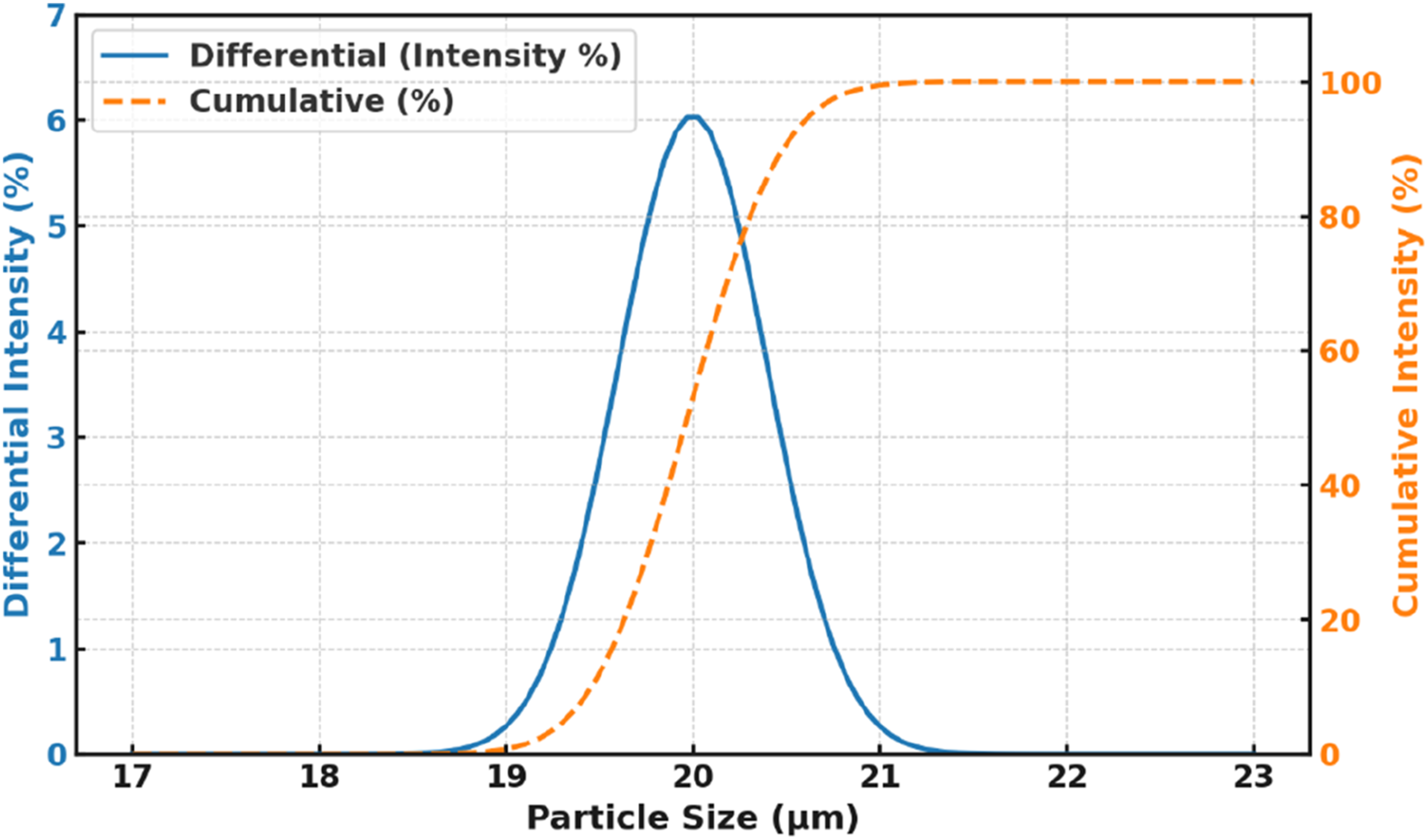
Post-milling, the powder was sieved using a 75 µm mesh to ensure uniformity and to exclude agglomerates that could compromise film homogeneity. The final CSP exhibited a bulk density of approximately 0.68 g/cm3 and an ash content below 1.2%, making it suitable for consistent reinforcement distribution. The processing route preserved key chemical functionalities, including hydroxyl and phenolic groups, enabling favorable interfacial interactions with the polar PVA matrix during solution casting.
The FESEM analysis provided clear evidence of the morphological heterogeneity of the CSP particles as shown in Figure 3. At 500x magnification (Figure 3(a)), the powder exhibited flaky, irregular particles with sharp fractured edges, indicative of brittle fracture during milling. Such asperities increase the available surface area, supporting stronger mechanical interlocking with the PVA matrix. At higher magnification of 750x (Figure 3(b)), the presence of blocky compact particles with smooth flat facets was observed, highlighting regions where incomplete fracture yielded relatively dense fragments. This bimodal morphology, comprising both angular flaky structures and compact blocky particles, is advantageous as it ensures a balance between high interfacial adhesion and bulk rigidity, thereby enhancing the reinforcement efficiency of CSP within the PVA system. (a) and (b) FESEM and (c) EDAX results of coconut shell powder.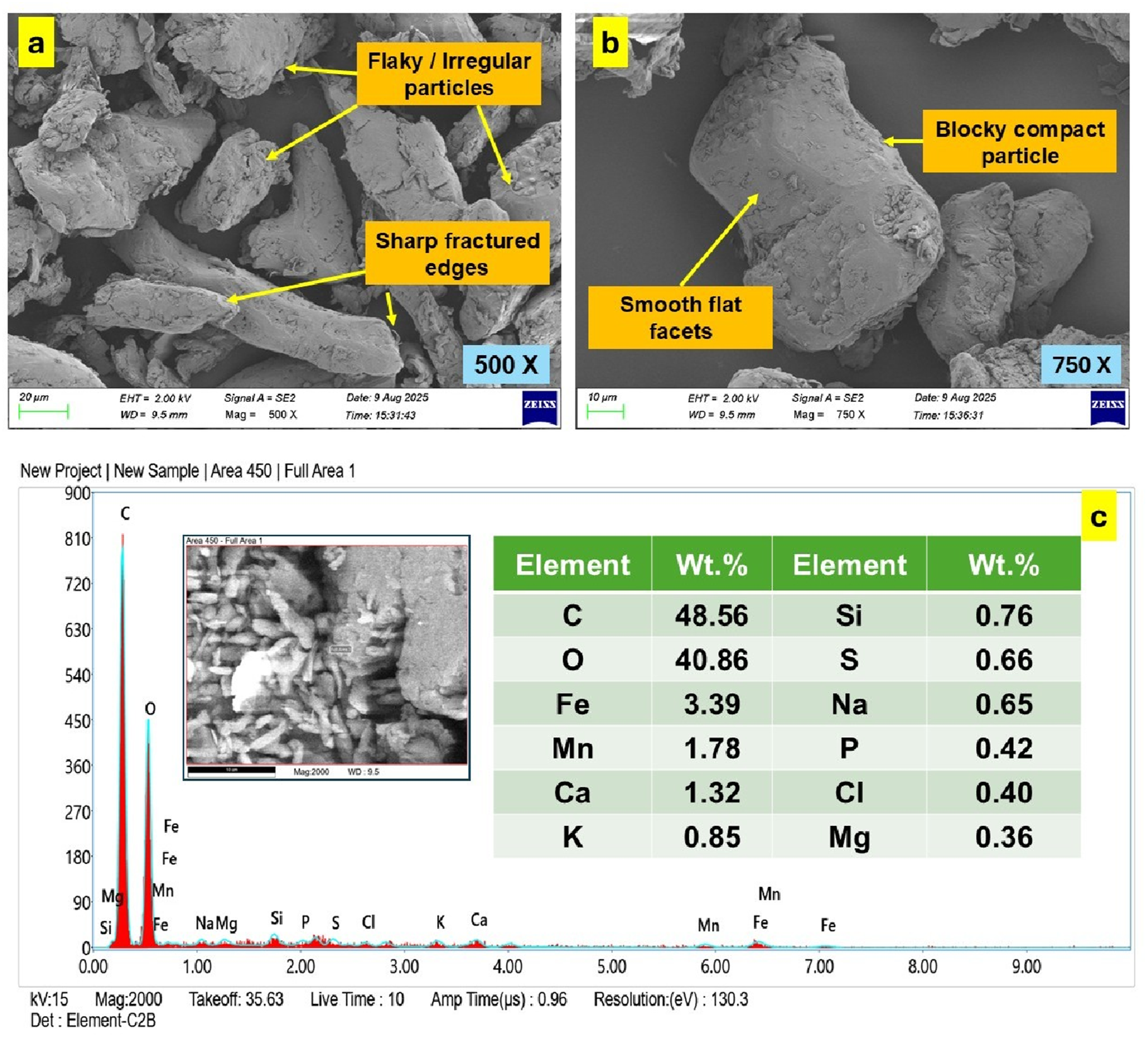
Elemental composition was evaluated using EDAX, as presented in Figure 3(c). The results revealed the predominance of carbon (48.56 wt%) and oxygen (40.86 wt%), consistent with the lignocellulosic backbone of the coconut shell derived from cellulose, hemicellulose, and lignin. Minor but technically relevant elements such as Fe (3.39 wt%), Mn (1.78 wt%), and Ca (1.32 wt%) were also detected, alongside trace quantities of Si, S, Na, P, Cl, K, and Mg. These inorganic inclusions, naturally embedded within the biogenic structure, contribute to the hybrid organic–inorganic constitution of CSP. The rigid mineral traces may act as micro-reinforcing domains and potential nucleation sites within the PVA matrix, improving crystallization kinetics, localized stiffness, and interfacial heterogeneity. Thus, the combined evidence of controlled particle size and complex composition substantiates the suitability of CSP as a multifunctional reinforcement in PVA-based biofilms.
Biofilm fabrication
Biofilm fabrication in this study was executed via a carefully controlled solution casting process using polyvinyl alcohol (PVA) as the polymer matrix and coconut shell powder (CSP) as the biofiller. The fabrication process is represented in Figure 4. The procedure was designed to ensure uniform dispersion of CSP within the PVA matrix, facilitating optimal interfacial interaction and consistent film morphology. A precise amount of PVA powder was initially dissolved in deionized water at 90 ± 2°C under continuous magnetic stirring until a homogeneous and transparent solution was obtained. This temperature was selected based on the dissolution kinetics of fully hydrolyzed PVA, ensuring complete chain relaxation without thermal degradation. The films in this study were fabricated using a water-based solution casting process in which deionized water functions as the dissolution and dispersion medium for PVA and CSP. Fabrication of PVA/CSP biofilms via solution casting, involving mixing of polyvinyl alcohol and coconut shell powder at 90°C followed by film formation.
Subsequently, pre-sieved CSP, processed to a median particle size of ∼20 µm, was gradually incorporated into the PVA solution. The filler addition was performed in increments to prevent agglomeration and to facilitate wetting of the CSP surfaces by the polymer chains. The mixture was maintained at 90°C during this stage to preserve the viscosity window conducive to stable particle suspension. Manual stirring was supplemented alongside magnetic agitation to ensure full dispersion, particularly for higher filler loadings that tend to increase system heterogeneity. The resulting CSP–PVA slurry was cast into a levelled mold fabricated from non-adhering wooden sheets with internal cavities of 120 mm × 120 mm. Controlled drying was carried out at ambient conditions (25 ± 1°C) for 48 hours to allow gradual solvent evaporation and prevent internal stress build-up within the film. The dried films were carefully peeled and stored in desiccators to avoid post-formation moisture absorption prior to testing.
The thickness of the cast films was measured to ensure uniformity across the different CSP loadings, since deviations in thickness may influence mechanical strength and water-transport behaviour. All films were prepared under the same casting and drying conditions, and thickness was recorded at five randomly selected points using a precision micrometer with 0.01 mm resolution. The neat PVA film exhibited an average thickness of approximately 150–160 µm with a narrow variation band. The addition of 2 wt% CSP yielded a comparable range, indicating that low filler content might not disrupt flow and leveling during casting. Films containing 4 wt% CSP showed minor local variations, falling within 170–210 µm, which may be attributed to the increased solids fraction during solvent evaporation. At 6 wt% CSP, the thickness remained consistent within 210–250 µm, and the measured spread suggests that the slurry still maintained adequate flow before gelation. Across all compositions, the variation remained within a small window, and the uniformity indicates that the mechanical and barrier-property differences observed in this work arise primarily from CSP–PVA interactions rather than uncontrolled thickness fluctuations. The recorded thickness values thus provide confidence that loading effects discussed later are not influenced by sample dimensional inconsistency.
Testing methods
The fabricated PVA/CSP biofilms were subjected to a comprehensive set of physical and environmental tests to evaluate their mechanical strength, hydrophilicity, and biodegradability, in accordance with standardized protocols as represented in Figure 5. Tensile testing was conducted following the guidelines of ASTM D882-18, which specifies the procedure for evaluating the tensile properties of thin plastic sheeting. Samples were die-cut into strips with dimensions of 100 mm × 15 mm and a thickness in the range of 0.25–0.30 mm, as per the dimensional tolerances defined by the standard. The tests were performed using a universal testing machine (UTM) equipped with a 5 kN load cell, operated at a crosshead speed of 50 mm/min under ambient conditions (25 ± 1°C, 50 ± 5% RH). Each sample was clamped with pneumatic grips to ensure uniform load distribution and minimize slippage. A minimum of three specimens per sample group were tested to ensure statistical significance and reproducibility. Prepared PVA/CSP biofilm samples subjected to tensile testing, water absorption analysis, and biodegradability assessment.
Water absorption behavior was analyzed based on ASTM D570-98, wherein conditioned samples were initially oven-dried at 50°C for 24 hours and weighed to obtain the dry mass (M0). Subsequently, specimens were fully immersed in distilled water at room temperature (25°C) and extracted at fixed intervals (in 24 hours) to measure the wet mass (M1). The percentage water uptake was calculated using the standard gravimetric relation in equation (1):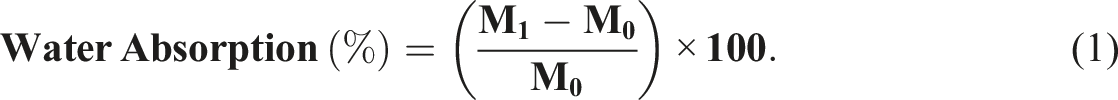
Samples were gently wiped prior to weighing to eliminate surface water effects. The study evaluated how CSP content influenced the hydrophilic balance of the PVA matrix over time.
The biodegradability of the biofilms was evaluated through a soil burial test, adopting procedures derived from ISO 846:2019, tailored to simulate aerobic degradation in natural environments. Samples of equal size (50 mm × 15 mm) were buried vertically at a depth of 80 mm in biologically active garden soil with a moisture content maintained at 60 ± 5% and temperature regulated at 28 ± 2°C. The exposure period lasted for 30 days, after which the specimens were carefully recovered, cleaned, dried, and weighed. Degradation was quantified by evaluating the percentage weight loss, indicating microbial and environmental disintegration. Visual inspection and dimensional shrinkage were also recorded to support the gravimetric findings.
Characterization of biofilm
X-ray diffraction (XRD) analysis was performed to investigate the crystalline structure of the biofilm samples and to evaluate the influence of coconut shell powder (CSP) incorporation on the crystallinity of the PVA matrix and represented in Figure 6. The diffractograms were recorded over a 2θ range of 10° to 80°, using Cu-Kα radiation (λ = 1.5406 Å), operating at 40 kV and 30 mA. The neat PVA film exhibited a characteristic broad diffraction peak centered at approximately 2θ = 19.5°, which corresponds to the semi-crystalline nature of polyvinyl alcohol. This peak is attributed to the (101) crystalline plane associated with the physical crosslinking via intermolecular hydrogen bonding among PVA chains. XRD patterns of neat PVA and PVA biofilm reinforced with 6 wt% CSP, showing increased crystallinity due to CSP incorporation.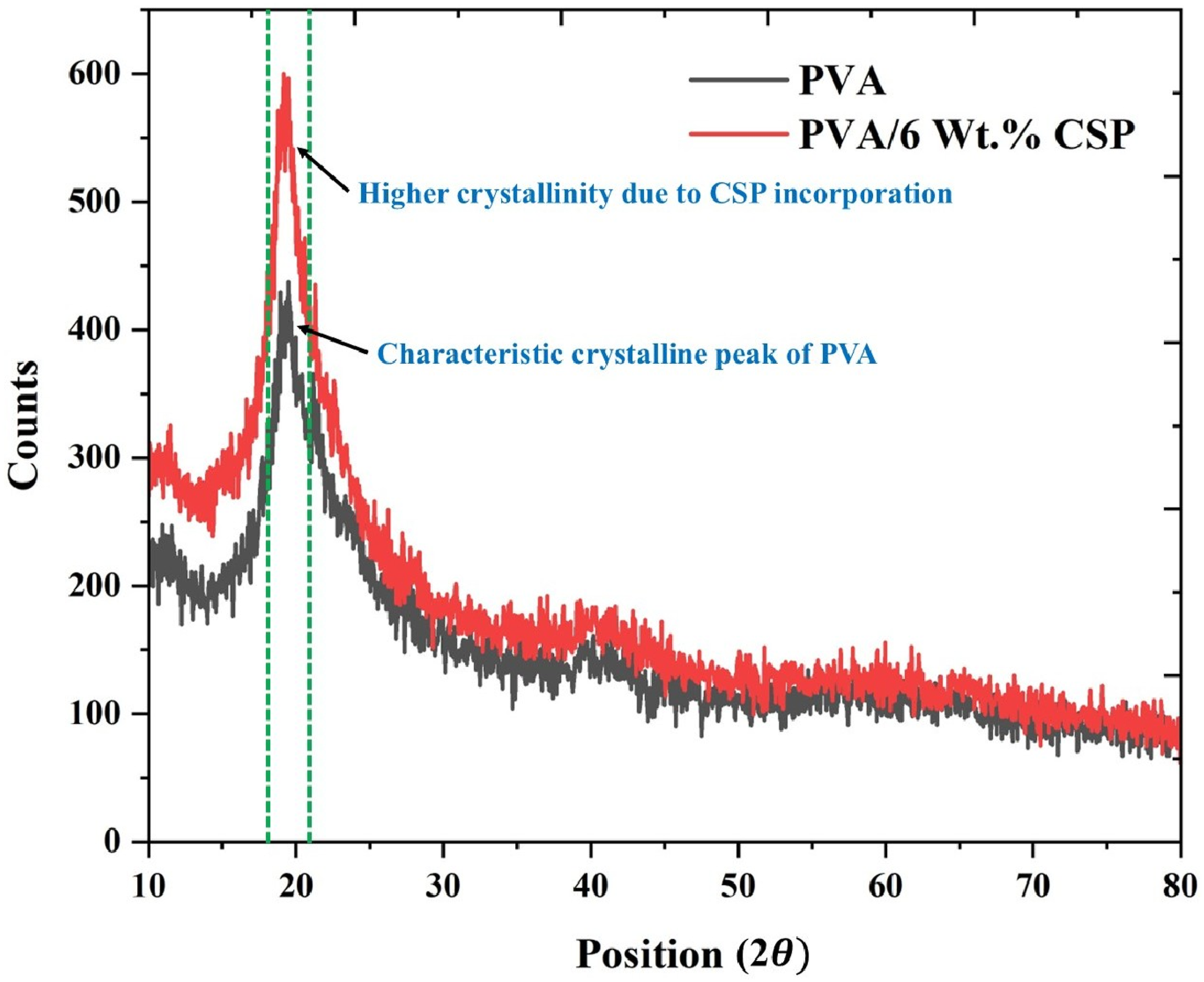
In contrast, the PVA/CSP composite biofilm reinforced with 6 wt% CSP demonstrated a more intense and sharper peak near 2θ = 20.1°, indicating a noticeable enhancement in crystalline order. This shift and sharpening of the peak suggest that the incorporation of CSP into the polymeric matrix facilitated heterogeneous nucleation during film formation, promoting localized chain alignment and crystalline domain development. The increase in diffraction intensity and slight rightward shift in the peak position also imply reduced interplanar spacing and enhanced chain packing density in the biofilm structure. The comparative analysis clearly illustrates that CSP acts as a nucleating agent, effectively modulating the microstructural arrangement of the PVA matrix. The intensification of the crystalline peak is consistent with improved intermolecular interactions at the filler–matrix interface, contributing to the mechanical and barrier performance enhancements observed in subsequent sections.
The nucleating effect of CSP is supported by the increase in crystallinity index calculated from the XRD pattern. The crystallinity index (CI) was determined using the Segal equation (2).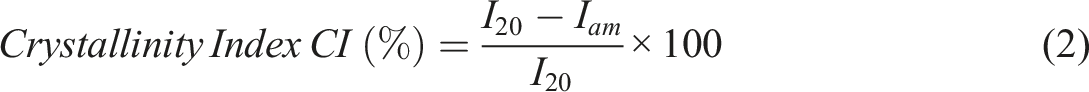
Fourier-transform infrared spectroscopy (FTIR) was employed to investigate the chemical interactions and structural modifications in the PVA matrix upon incorporation of 6 wt% coconut shell powder (CSP) as shown in Figure 7. The FTIR spectra were recorded in the range of 400–4000 cm−1 using ATR mode, with a spectral resolution of 4 cm−1 and 32 scans per sample. The spectrum of neat PVA exhibited characteristic absorption bands corresponding to its semi-crystalline polymeric backbone, including a broad O–H stretching vibration centered near 3295 cm−1, which arises from extensive intermolecular and intramolecular hydrogen bonding among hydroxyl groups along the PVA chain. FTIR spectra of neat PVA and PVA/6 wt% CSP biofilm, highlighting structural changes and functional group interactions due to CSP incorporation.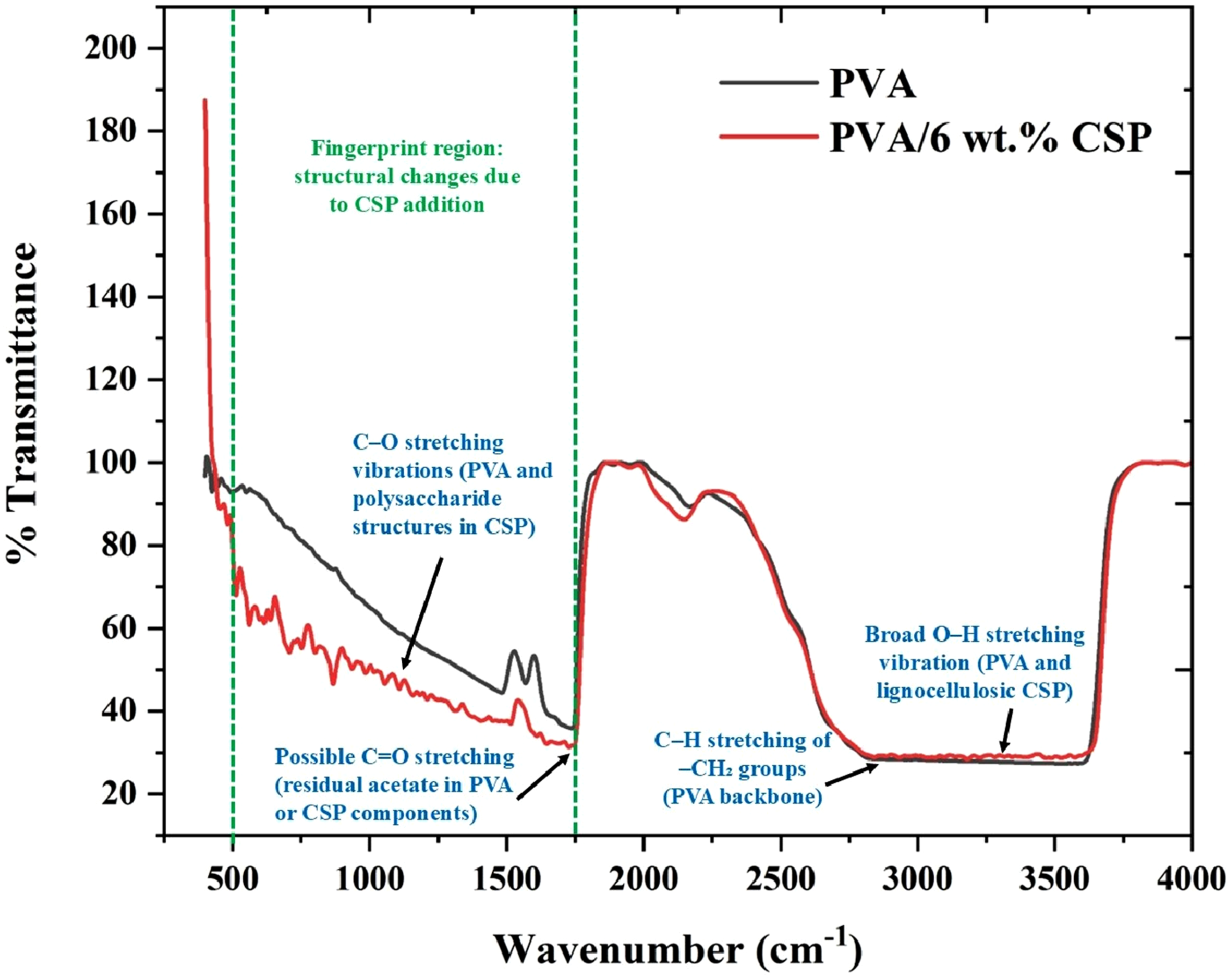
The incorporation of CSP resulted in notable spectral changes, particularly in the fingerprint region (600–1500 cm−1), indicative of altered molecular interactions and new bond formation. A distinct increase in transmittance intensity was observed in the region 1000–1150 cm−1, corresponding to C–O stretching vibrations, which can be attributed to contributions from both the PVA backbone and polysaccharide structures within the lignocellulosic CSP. The enhancement of this band in the PVA/6 wt% CSP composite suggests synergistic hydrogen bonding and the integration of cellulose and hemicellulose functionalities from CSP into the PVA matrix.
Moreover, the appearance of a shoulder band around 1720 cm−1 in the composite spectrum is attributed to C = O stretching, potentially originating from residual acetate groups in PVA or carbonyl functionalities in CSP lignin. This confirms the chemical heterogeneity introduced by CSP addition. The band at 2930 cm−1, associated with symmetric and asymmetric C–H stretching of –CH2 groups, was retained in both spectra but exhibited a slight broadening and shift in the composite, indicating possible microstructural rearrangements. The spectral profile confirms that CSP not only contributes physically as a filler but also actively participates in interfacial bonding, leading to enhanced compatibility and structural reorganization in the PVA matrix.
The FTIR spectra may be interpreted further by examining relative band intensities associated with hydrogen bonding in the PVA–CSP matrix. The broad O–H stretching band around 3250–3350 cm−1 reflects the collective contribution of inter- and intra-molecular hydrogen bonds. An increase in the relative intensity of this band with CSP loading may indicate stronger hydrogen-bonding interactions between hydroxyl groups of PVA and lignocellulosic moieties within the filler. The ratio between the O–H stretching band and the C–O stretching band in the 1080–1150 cm−1 region provides a qualitative hydrogen-bonding index. A progressive rise in this ratio with increasing CSP content suggests that additional hydrogen-bond networks may be formed, which could result in more restricted chain mobility and enhanced modulus. The slight sharpening of the C–H bending region (around 1420–1460 cm−1) may also signal increased packing regularity, complementing the crystallinity trends observed in XRD. Although a full peak-deconvolution analysis was not performed, the intensity-ratio approach offers a semi-quantitative indication of the strengthening of interfacial bonding, consistent with the improved mechanical properties at higher CSP levels.
Results and discussion
Tensile properties
The stress–strain behavior of the PVA/CSP biofilms revealed a significant enhancement in mechanical performance as a function of coconut shell powder (CSP) content, as presented in Figure 8. The neat PVA film exhibited a typical ductile profile with distinct linear elastic and plastic deformation regions, reaching a maximum stress of approximately 54 MPa at a strain of around 38%. The linear elastic region extended up to ∼8% strain, after which nonlinear plastic deformation dominated until fracture. The inherent ductility of neat PVA arises from the flexibility of its hydroxyl-rich polymer chains and absence of rigid reinforcement phases. Stress–strain curves of neat PVA and PVA/CSP biofilms with varying CSP contents, showing enhanced tensile strength and elongation with higher CSP loading.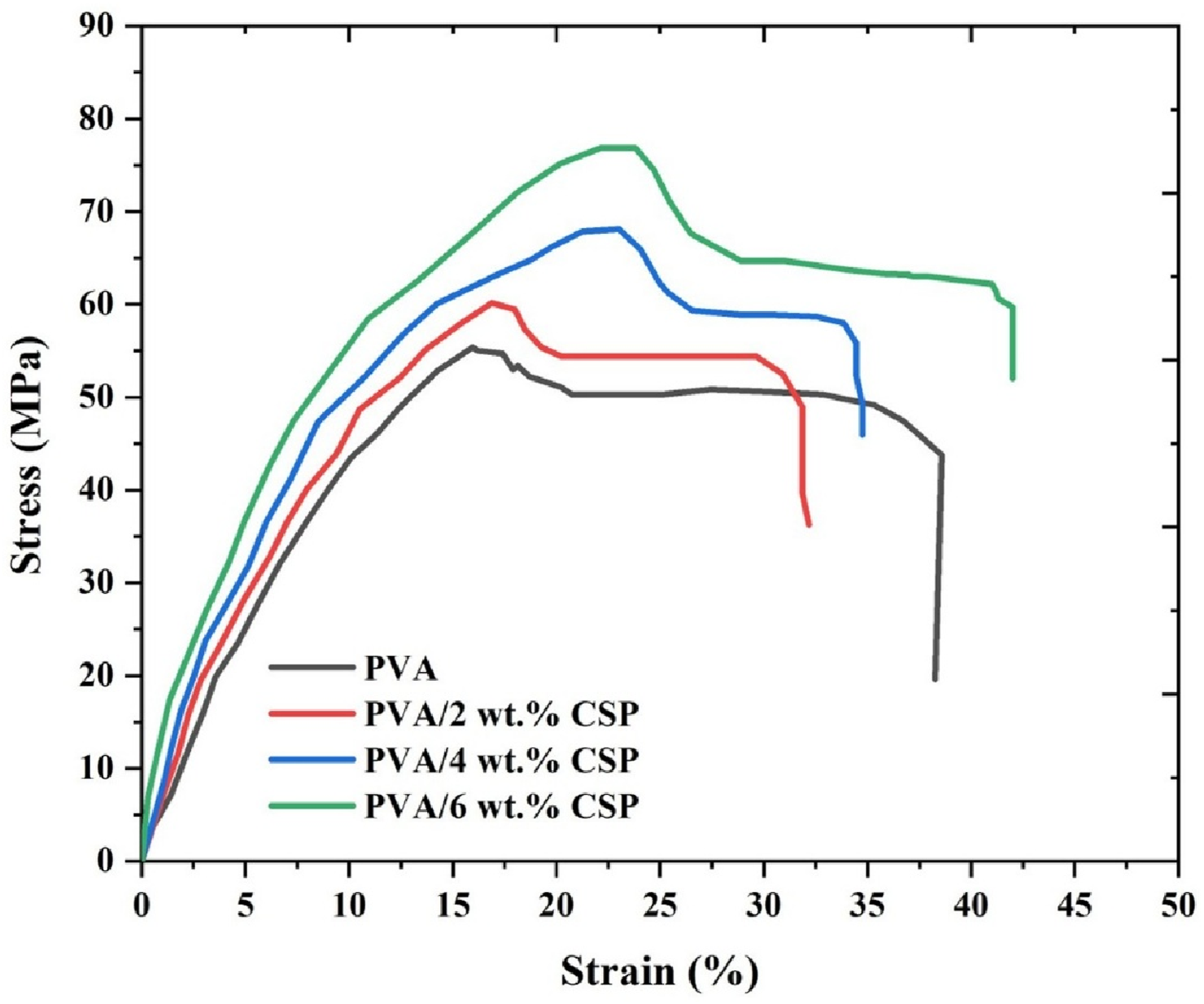
Upon CSP incorporation, a progressive improvement in tensile strength and strain-at-failure was observed across all filler concentrations. The film reinforced with 2 wt% CSP exhibited a moderate increase in peak stress to approximately 58 MPa, with a similar overall strain capacity (∼34%) as the unreinforced matrix. The mechanical enhancement at this loading can be attributed to initial filler–matrix interfacial adhesion that supports efficient stress transfer without significantly restricting chain mobility.
Notably, at 4 wt% and 6 wt% CSP, the stress–strain curves displayed a pronounced upward shift in both the elastic slope and the ultimate tensile stress. The 4 wt% CSP composites achieved a maximum stress of approximately 68 MPa and a strain of 36%, while the 6 wt% CSP biofilms peaked at around 76 MPa with a strain exceeding 38%. This behavior suggests that the interfacial bonding between CSP and the PVA matrix becomes increasingly effective at higher filler loadings, likely due to enhanced hydrogen bonding interactions and CSP surface reactivity. The increased stiffness in the linear region indicates a higher elastic modulus, corroborating the reinforcement effect of finely dispersed CSP particles. Furthermore, the extended strain-to-failure in higher loading samples confirms that the reinforcement does not induce brittleness, but rather synergistically toughens the matrix.
The enhanced tensile response is a direct consequence of CSP’s ability to act as both a stress-bearing and energy-dissipating phase within the PVA matrix. The increased crystallinity observed in XRD analysis (Section 2.5) and the interfacial compatibility evidenced by FTIR studies further support the mechanical trends reported. These results validate the structural integrity and mechanical reliability of CSP-reinforced PVA biofilms for load-bearing applications in biodegradable packaging.
The incorporation of CSP into the PVA matrix resulted in a marked and systematic enhancement of both yield strength and ultimate tensile stress (UTS), as illustrated in Figure 9. Neat PVA exhibited a yield strength of 43.1 ± 1.3 MPa and a UTS of 38.4 ± 1.5 MPa, consistent with its flexible polymeric character and low resistance to plastic deformation. With the addition of 2 wt% CSP, yield strength and UTS increased to 47.9 ± 1.1 MPa and 43.2 ± 1.4 MPa, respectively, indicating early-stage filler reinforcement through interfacial bonding and particle–matrix interaction. At 4 wt% CSP, a more substantial rise was observed, yield strength reached 52.4 ± 1.2 MPa, while UTS elevated to 69.1 ± 1.7 MPa. This reflects enhanced mechanical interlocking and restricted polymer chain mobility due to effective filler dispersion. Yield strength and ultimate tensile stress of neat PVA and PVA/CSP biofilms with different CSP contents.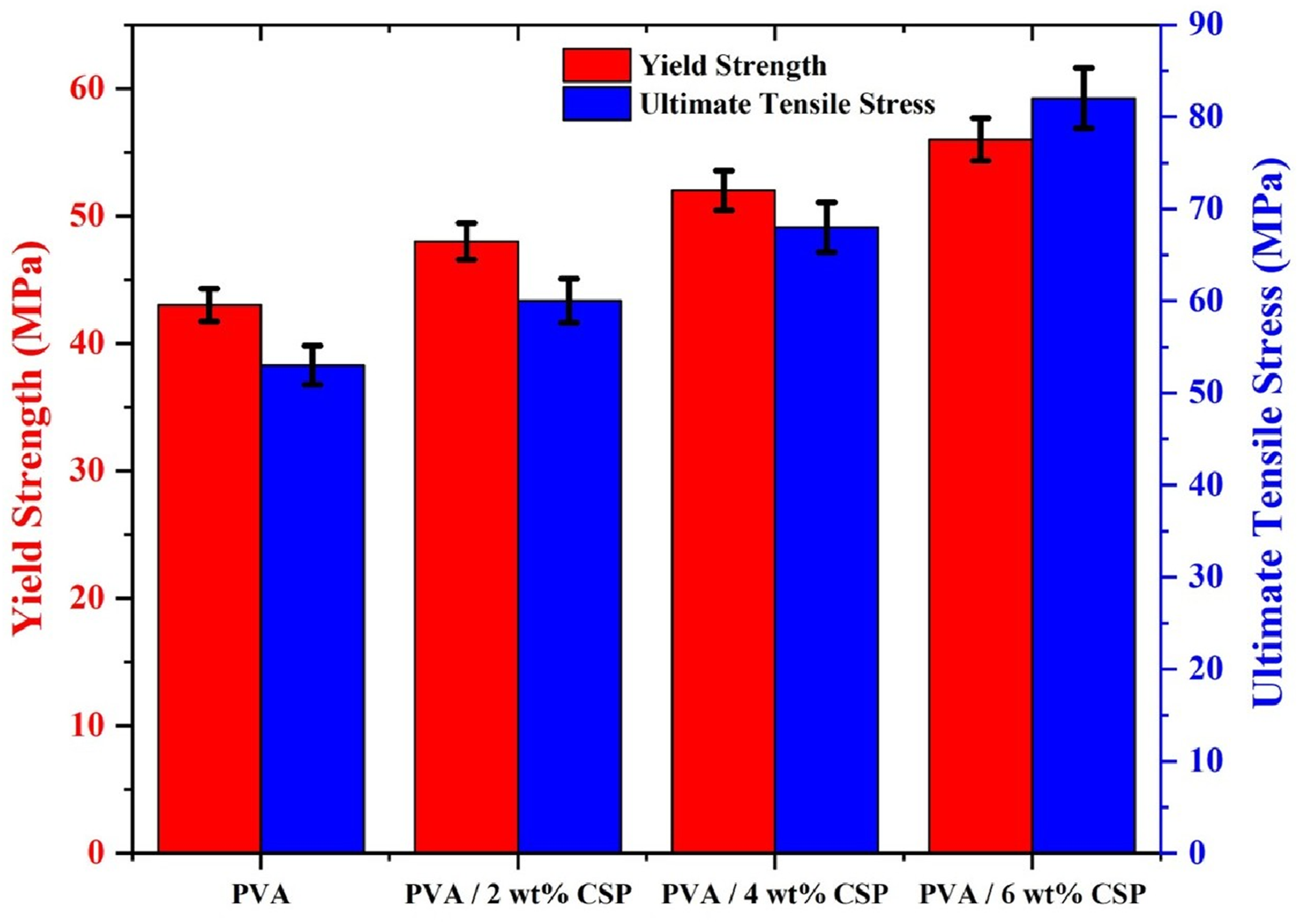
Maximum reinforcement occurred at 6 wt% CSP, achieving 58.3 ± 1.4 MPa yield strength and 83.6 ± 1.6 MPa UTS. At this concentration, CSP serves as a highly efficient stress-transfer medium, enhancing the elastic response and delaying failure. The observed trends correlate with increased crystallinity (Figure 6) and intensified chemical interactions (Figure 7), confirming the structural synergy between PVA and CSP. The leftward shift in the yield point and upward trend in UTS across increasing filler loadings validate the dual enhancement of stiffness and strength in the biofilm architecture without sacrificing ductility.
The effect of CSP loading on the stiffness and deformability of the PVA biofilms is quantitatively depicted in Figure 10, where both elasticity modulus and elongation at failure are plotted against filler content. Neat PVA exhibited an elasticity modulus of 1.73 ± 0.05 GPa, reflecting the inherent compliance of the polymer matrix. Upon the introduction of 2 wt% CSP, the modulus increased to 1.85 ± 0.04 GPa, while the elongation at failure dropped slightly to 32.7 ± 0.6%, indicating an initial stiffening effect that modestly constrained the matrix ductility due to filler–polymer interface development. With further filler incorporation, stiffness improved significantly. The modulus increased to 1.97 ± 0.03 GPa at 4 wt% CSP and peaked at 2.14 ± 0.04 GPa for 6 wt% CSP, validating the reinforcing influence of CSP through enhanced stress transfer and chain immobilization within the elastic regime. Concurrently, elongation at failure also improved—from 35.1 ± 0.5% at 4 wt% to 42.3 ± 0.8% at 6 wt%, suggesting that CSP reinforcement does not compromise flexibility but rather synergizes matrix toughness by promoting crack-bridging and localized plastic flow mechanisms. This dual improvement in stiffness and ductility at higher CSP loadings reveals a unique balance between rigidity and extensibility, attributable to the optimal dispersion of CSP and strong interfacial adhesion with the PVA matrix. Elasticity modulus and elongation at failure of neat PVA and PVA/CSP biofilms.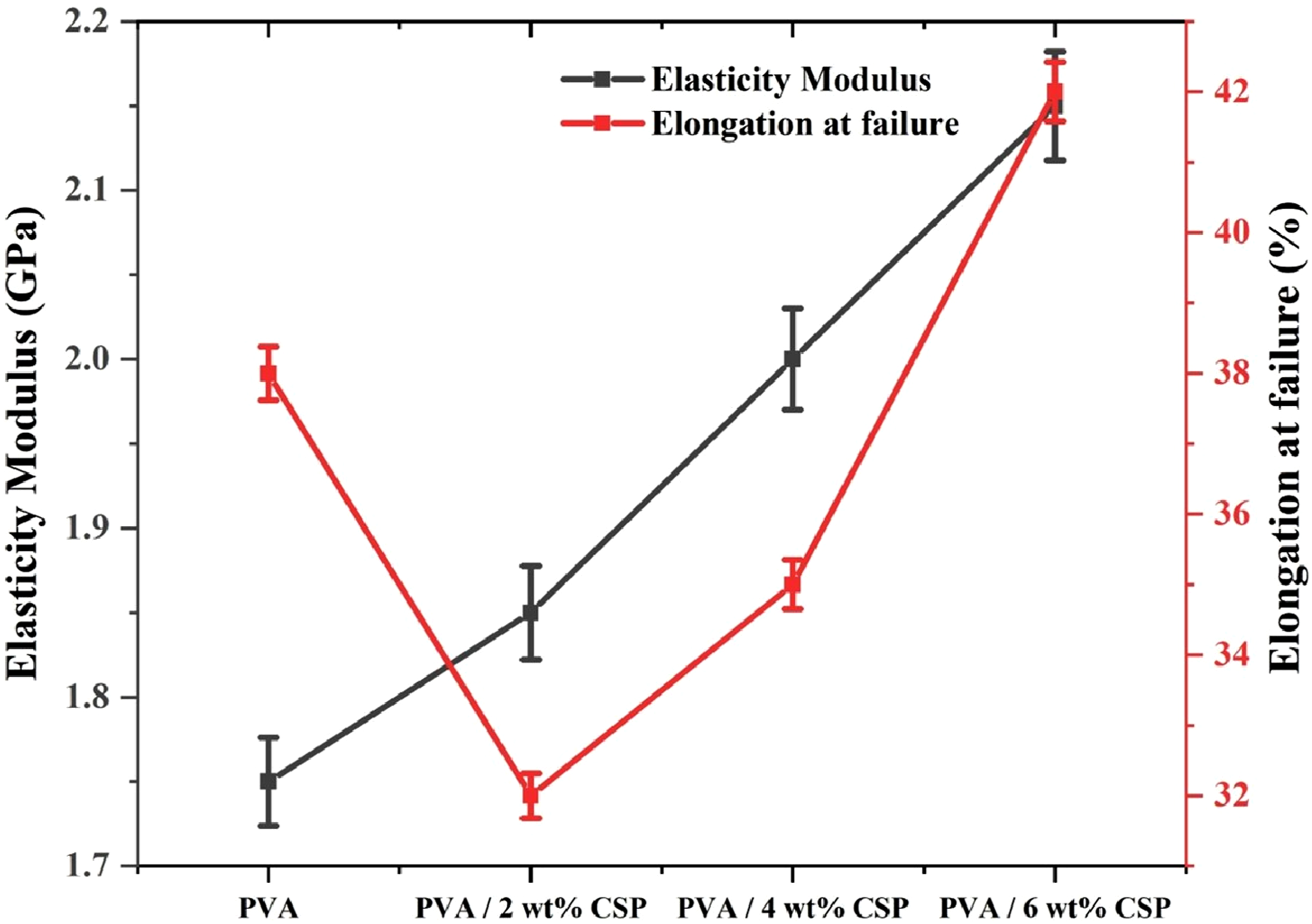
The increase in elongation observed in the CSP-reinforced films may be attributed to the interaction between the PVA chains and the lignocellulosic particulate surface. The CSP particles contain hydroxyl and phenolic groups that are capable of forming secondary bonds with the polar PVA backbone. This interaction might restrict local chain slippage yet still permit segmental mobility in regions surrounding the particle–matrix interface. Such mobility can delay the onset of catastrophic crack growth during tensile loading. SEM fractography indicates localized fibrillation and formation of micro-ligaments at the fracture surface. These features suggest that the deformation is accommodated through a combination of polymer chain bridging, progressive micro-void formation, and crack blunting around CSP particles. The irregular particle morphology may promote tortuous crack paths, leading to additional energy consumption before failure. The particle pull-out traces seen in the micrographs support the idea that the filler–matrix adhesion is balanced, allowing partial debonding that might contribute to elongation by creating controlled zones of plastic deformation. These mechanisms together may reduce stress concentration and distribute the applied load more evenly, allowing the films to withstand higher strains before rupture.
A statistical comparison was performed based on the mean values and the associated error bars presented in the tensile, modulus, and elongation plots. The yield strength increases from approximately 43 MPa for neat PVA to nearly 48 MPa at 2 wt% CSP, 52 MPa at 4 wt% CSP, and about 58 MPa at 6 wt% CSP. The ultimate tensile stress also follows a similar rising trend, increasing from about 38 MPa for neat PVA to 43 MPa, 47 MPa, and finally 82 MPa at the highest filler loading. These changes are accompanied by a progressive reduction in the relative error band, especially between 4 and 6 wt% CSP, which may indicate that the variation between replicate specimens is lower than the variation observed across the groups. A similar pattern is visible in the modulus data, where the values rise from roughly 1.75 GPa (PVA) to 1.85 GPa, 2.0 GPa, and 2.15 GPa, with a consistent trend of narrow error bars. The elongation at failure decreases at 2 wt% (≈32–33%), then increases again at 4 wt.% (≈36%), reaching ≈42% at 6 wt% CSP with reduced scatter. These shifts between mean values are larger than the corresponding error-bar ranges, suggesting that the differences between compositions exceed the variability within each group. A one-way ANOVA framework would likely classify these groups as statistically distinct, since the between-group variance appears higher than the within-group variance for yield strength, tensile stress, modulus, and elongation. This pattern supports the interpretation that CSP loading is a dominant factor influencing the mechanical behaviour and that the observed improvements arise from material response rather than random measurement variability.
Tensile fracture mechanisms
The fractography was employed to characterize the macroscopic failure patterns and fracture morphology of the tensile-tested PVA/CSP biofilms, with a focus on correlating surface texture to CSP reinforcement. The morphological evolution across different filler concentrations reveals a transition in fracture mode that is consistent with the mechanical response trends discussed in Section 3.1. The neat PVA film, as shown in Figure 11(a), exhibited predominantly smooth and continuous fracture planes interspersed with occasional shear ridges. These features are indicative of a homogeneous ductile failure dominated by plastic flow within the polymer matrix, with minimal microstructural discontinuities to hinder crack progression. The absence of fillers resulted in uniform stress distribution but limited resistance to crack propagation once yielding was initiated. SEM images of PVA/CSP biofilm fractures at low magnification: (a) smooth planes, shear ridges; (b) rough texture, ridges; (c) CSP pull-out, micro-roughness; (d) river lines, microvoid coalescence.
Upon CSP incorporation, the fracture surfaces developed increasingly irregular and heterogeneous textures. The sample with moderate filler loading displayed roughened morphology and prominent ridge formation (Figure 11(b)), which reflects the onset of filler-induced crack deflection and interfacial micro-fracture processes. These surface undulations signify enhanced energy dissipation mechanisms that arise from crack path tortuosity around rigid CSP particles.
In CSP-rich regions (Figure 11(c)), clear signs of filler–matrix interaction were observed, including localized fracture roughness and voids corresponding to pulled-out particles. These features suggest the presence of incomplete filler–matrix adhesion zones where CSP debonding and mechanical interlocking co-exist. Such fracture behavior facilitates greater plastic deformation and strain accommodation at the mesoscale. Further morphological complexity is evident in Figure 11(d), where features such as river line patterns and microvoid clusters begin to emerge. While these cannot yet be resolved at the microscale, their presence indicates the initiation of ductile tearing and localized damage accumulation, processes that contribute to the increased toughness observed at higher filler contents. The low magnification SEM images confirm that CSP reinforcement modifies the global fracture behavior of PVA films by introducing surface roughness, particle pull-out zones, and crack deflection paths. These mesoscale failure characteristics are indicative of improved toughness and correlate well with the observed enhancements in elongation, modulus, and strength.
The low magnification SEM analysis previously revealed a progressive evolution of fracture morphology with increasing CSP content, transitioning from smooth ductile planes in neat PVA to complex, energy-dissipative textures in CSP-reinforced biofilms. However, to probe the microscale damage phenomena governing these macroscopic behaviors, high magnification SEM imaging was conducted, and the results are presented in Figure 12. SEM images of PVA/CSP fracture surfaces at high magnification: (a) interlamellar tearing, crazing; (b) interfacial debonding, particle pull-out; (c) cavitation pits, plastic flow; (d) shear lips, fracture fibrils.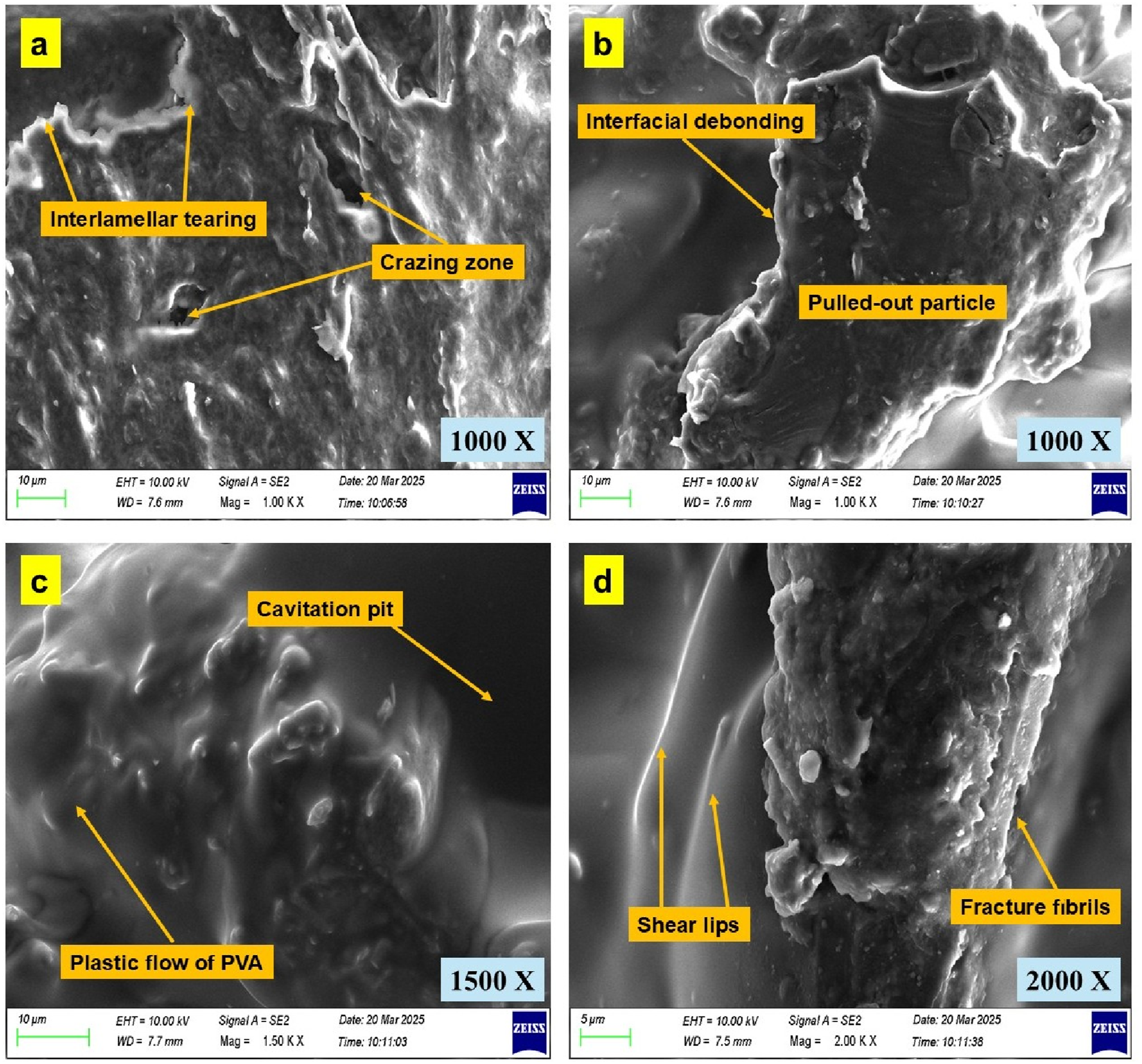
At 1000x magnification, Figure 12(a) highlights feature such as interlamellar tearing and crazing zones in the CSP-rich regions of the matrix. These patterns are indicative of localized shear deformation and plastic strain accommodation through fibrillation between lamellar polymer domains. Crazing, often a precursor to void coalescence, points to the ductile nature of failure even in the presence of rigid inclusions, suggesting that CSP enables crack bridging rather than premature rupture. Figure 12(b) further confirms the interfacial interaction mechanisms inferred from lower magnification images. Here, interfacial debonding and pulled-out CSP particles are evident, validating the stress delocalization effect at the matrix–filler interface. The presence of micro gaps surrounding the detached particles signifies partial bonding with the PVA matrix, allowing energy dissipation via particle friction and crack path deviation during loading.
The microstructural signature of plastic flow is apparent in Figure 12(c), where the polymer matrix exhibits extensive smooth deformation zones interspersed with cavitation pits. These pits represent void nucleation sites that grow and coalesce under tensile stress, contributing to the non-linear strain hardening behavior discussed in Section 3.1. Their formation aligns with the increased elongation at failure observed at higher CSP loadings. At even higher magnification (2000x), Figure 12(d) reveals shear lips and fracture fibrils, confirming ductile tearing as a dominant failure mode. The fibrillar structures are characteristic of plastic ligament stretching and crack blunting, a hallmark of toughened polymer systems. These features reflect the micro-yielding mechanisms occurring near crack tips, further corroborating the reinforcement efficiency of CSP in modulating the local stress field.
The high magnification observations complement and deepen the understanding of fracture mechanisms deduced from low magnification SEM. While mesoscale features such as ridges, particle pull-out, and rough planes govern bulk fracture appearance, microscale phenomena, crazing, interfacial decohesion, void formation, and fibrillation, control the underlying damage evolution and toughening response. This multiscale fracture analysis underscores the role of CSP not merely as a passive filler but as a mechanically and chemically active phase that enhances toughness through synergistic energy absorption mechanisms.
The elemental composition of the tensile fracture surface of the PVA/CSP biofilm was examined using energy-dispersive X-ray spectroscopy (EDS), and the spectrum is shown in Figure 13. The dominant presence of carbon (C: 65.05 wt%) and oxygen (O: 10.01 wt%) reflects the organic nature of the matrix and filler, confirming the abundance of hydroxyl and polysaccharide groups from PVA and lignocellulosic CSP. A substantial amount of chlorine (Cl: 19.17 wt%) was also detected, which likely originates from residual surface modifications or natural mineral content within the CSP. The presence of trace elements such as calcium (Ca: 0.35 wt%), iron (Fe: 0.99 wt%), and mercury (Hg: 4.14 wt%) suggests inorganic remnants naturally embedded in coconut shell biomass or possibly introduced during mechanical processing. From a fracture mechanism perspective, these inorganic inclusions may serve as stress concentration points or filler discontinuities, influencing crack initiation and interfacial debonding under tensile loading. Moreover, the heterogeneous elemental distribution supports the observed microstructural roughness and CSP pull-out zones in SEM analysis (Figures 11 and 12), further validating the mechanical role of CSP as a chemically and physically active reinforcement. The EDS results thus provide complementary evidence for the complex failure mechanisms and confirm the bio-composite’s hybrid organic–inorganic composition. EDS spectrum and elemental composition of PVA/CSP biofilm from fractured surface.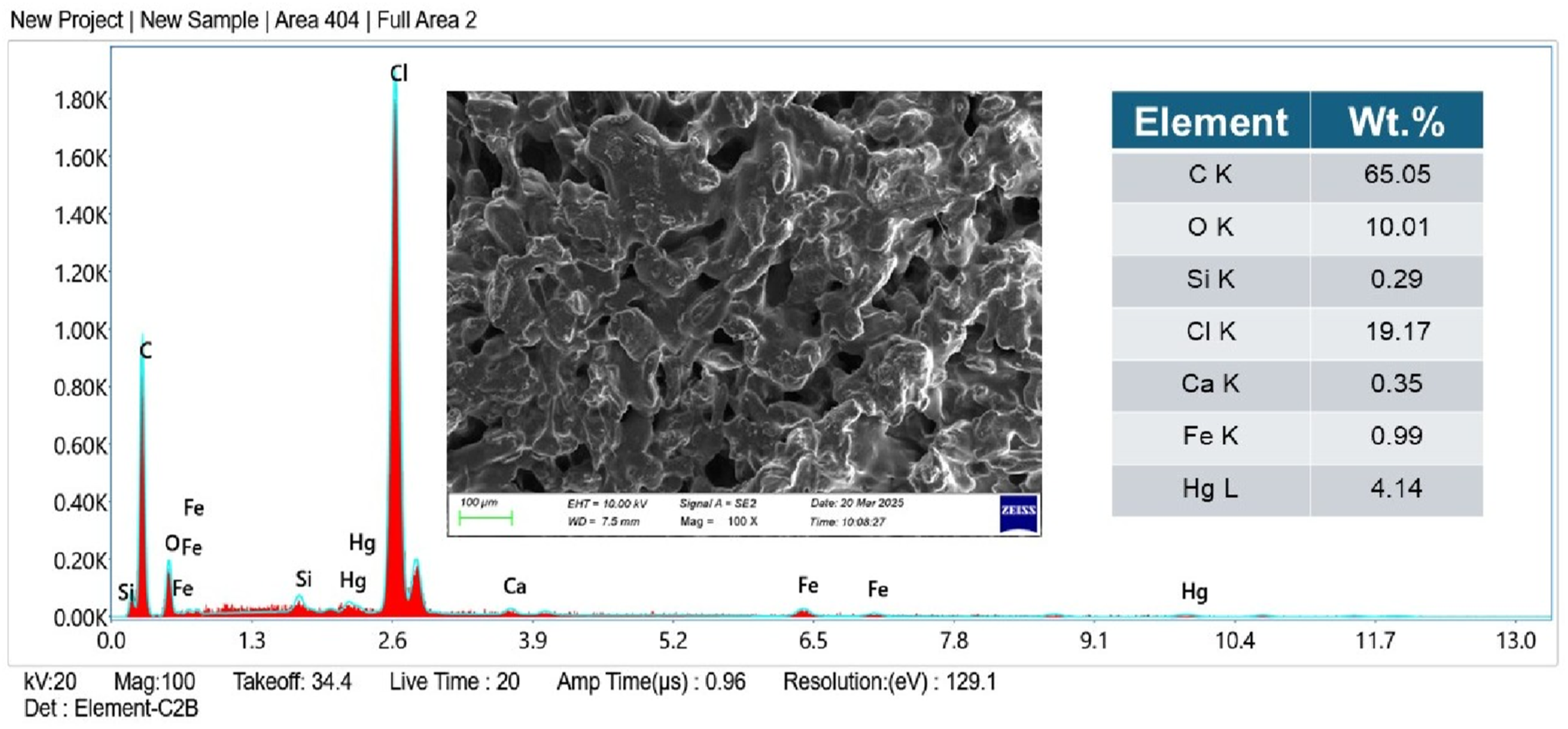
The fracture features observed in the SEM micrographs provide indications of the filler–matrix interaction and its influence on the tensile behaviour. The CSP-reinforced films show fewer clean particle cavities and more evidence of partial particle pull-out, which may suggest that the interfacial adhesion is balanced rather than excessively weak or overly rigid. This balance might allow limited debonding at the interface, which can absorb energy and delay crack propagation. The presence of micro-voids and fibrillated polymer ligaments around the CSP particles also points to progressive deformation before failure. These ligaments may bridge the crack front and distribute stress over a larger region. Such distributed deformation could reduce the severity of local stress concentrations and support higher elongation. The irregular morphology of CSP may induce tortuous crack paths, forcing the fracture to navigate around the particles instead of propagating directly through the polymer matrix. This deviation may increase the energy required for crack advancement. The smoother fracture surfaces seen in neat PVA contrast with the rougher, multi-directional surfaces in CSP-filled films. This contrast may explain the simultaneous improvement in strength and ductility, as the reinforced system may resist rapid crack growth through a combination of adhesion, controlled debonding, and crack path deflection.
The multiscale fracture features observed in the CSP-reinforced biofilms may be correlated with the mechanical metrics reported in Section 3.1. The appearance of dense fibrillation and elongated micro-ligament networks in the 4 and 6 wt% CSP films might explain the increased elongation at break because these structures can sustain distributed plastic deformation before catastrophic failure. The cavitation pits and void coalescence zones seen around CSP particles may be linked to the rise in toughness, since void nucleation and gradual growth may absorb energy and delay unstable crack propagation. The tortuous crack deflection induced by angular CSP particles may also contribute to the observed increase in modulus and UTS, as crack deviation generally increases the energy required for fracture progression. The partial particle pull-out features may indicate balanced interfacial adhesion that might help dissipate stress locally without premature interfacial failure. These multiscale deformation modes together may rationalize why the mechanical properties rise progressively with CSP loading and may explain the combined improvement in stiffness, strength, and ductility noted in the tensile results.
Thermal stability
The thermal behavior of neat PVA was evaluated using simultaneous thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC), as shown in Figure 14. The thermogram reveals multiple stages of thermal response, reflecting the distinct physicochemical transitions and degradation pathways intrinsic to the semi-crystalline PVA structure. The initial endothermic event between 50°C and 150°C, accompanied by a minor weight loss (∼5%), corresponds to the evaporation of bound and absorbed moisture. TGA–DSC curve of neat PVA showing moisture loss below 150°C and major degradation between 250 and 400°C.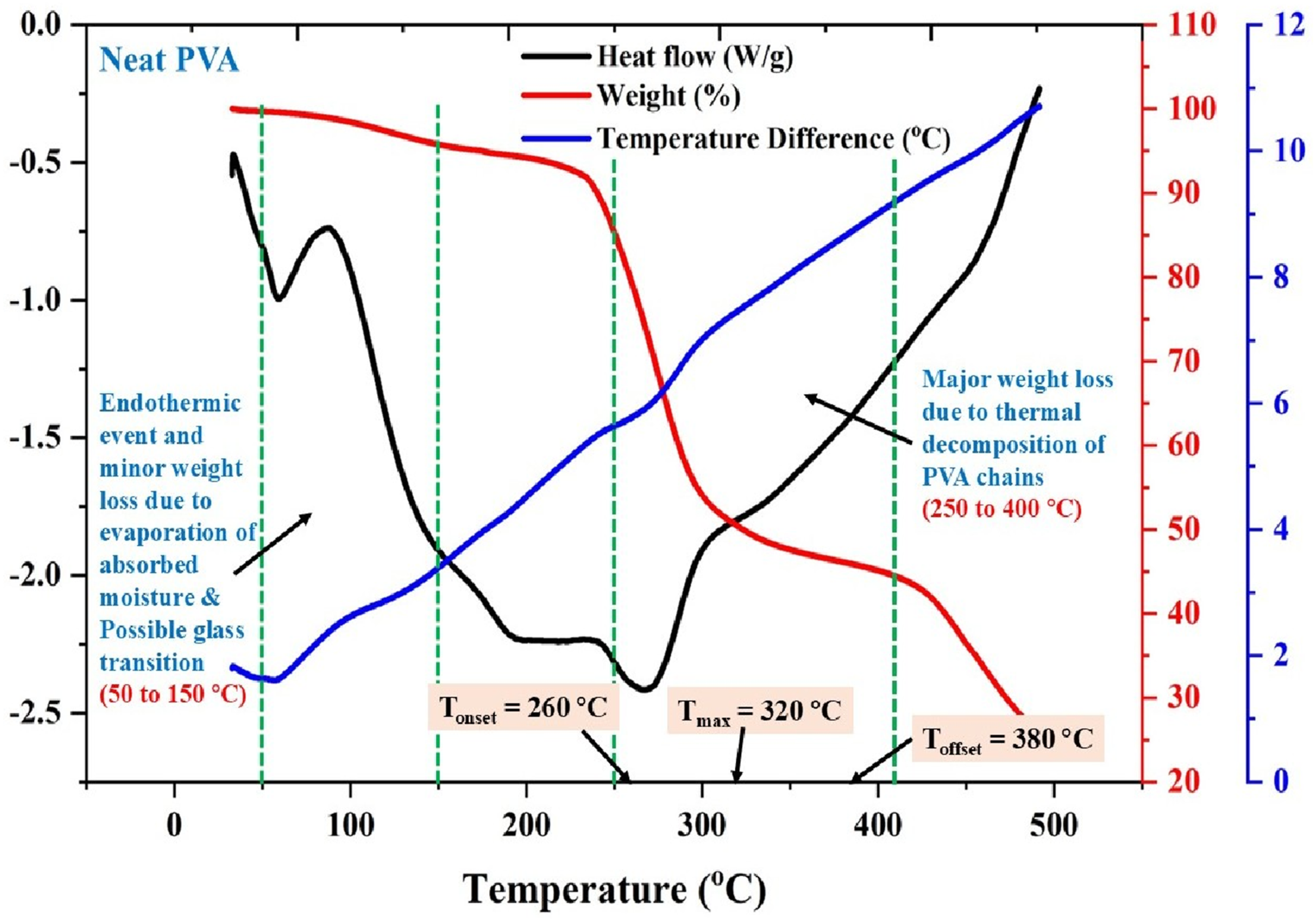
This thermal response is characteristic of hydroxyl-rich polymer systems and may also indicate a possible glass transition onset, although the broad feature lacks a sharp thermal shift due to overlapping solvent desorption effects.
The second stage, occurring between 250°C and 400°C, is marked by a major mass loss and strong endothermic behavior. The degradation onset temperature (Tonset) is approximately 260°C, with a degradation peak (Tmax) at 320°C and an offset around 380°C. This phase represents the thermal decomposition of the PVA backbone, particularly the elimination of side groups (–OH and –CH2) followed by chain scission and oxidative breakdown. The degradation is associated with polyene formation and random main-chain cleavage, which accelerates in oxygen-rich environments. The DSC trace mirrors this degradation sequence with a pronounced heat flow drop, confirming that the decomposition is primarily endothermic in nature. The thermal transition profile highlights that while neat PVA is thermally stable up to 250°C, its utility for elevated temperature applications is limited by polymer backbone instability beyond this point.
The thermal stability of the CSP-reinforced biofilm was evaluated through TGA–DSC analysis of the PVA/6 wt% CSP composite, as shown in Figure 15. Compared to the neat PVA profile (Figure 15), the reinforced composite displayed a notable shift in thermal degradation characteristics, signifying enhanced structural integrity under thermal stress due to the presence of lignocellulosic CSP. TGA–DSC curve of PVA/6 wt% CSP biofilm showing moisture loss below 150°C and major decomposition between 250 and 400°C.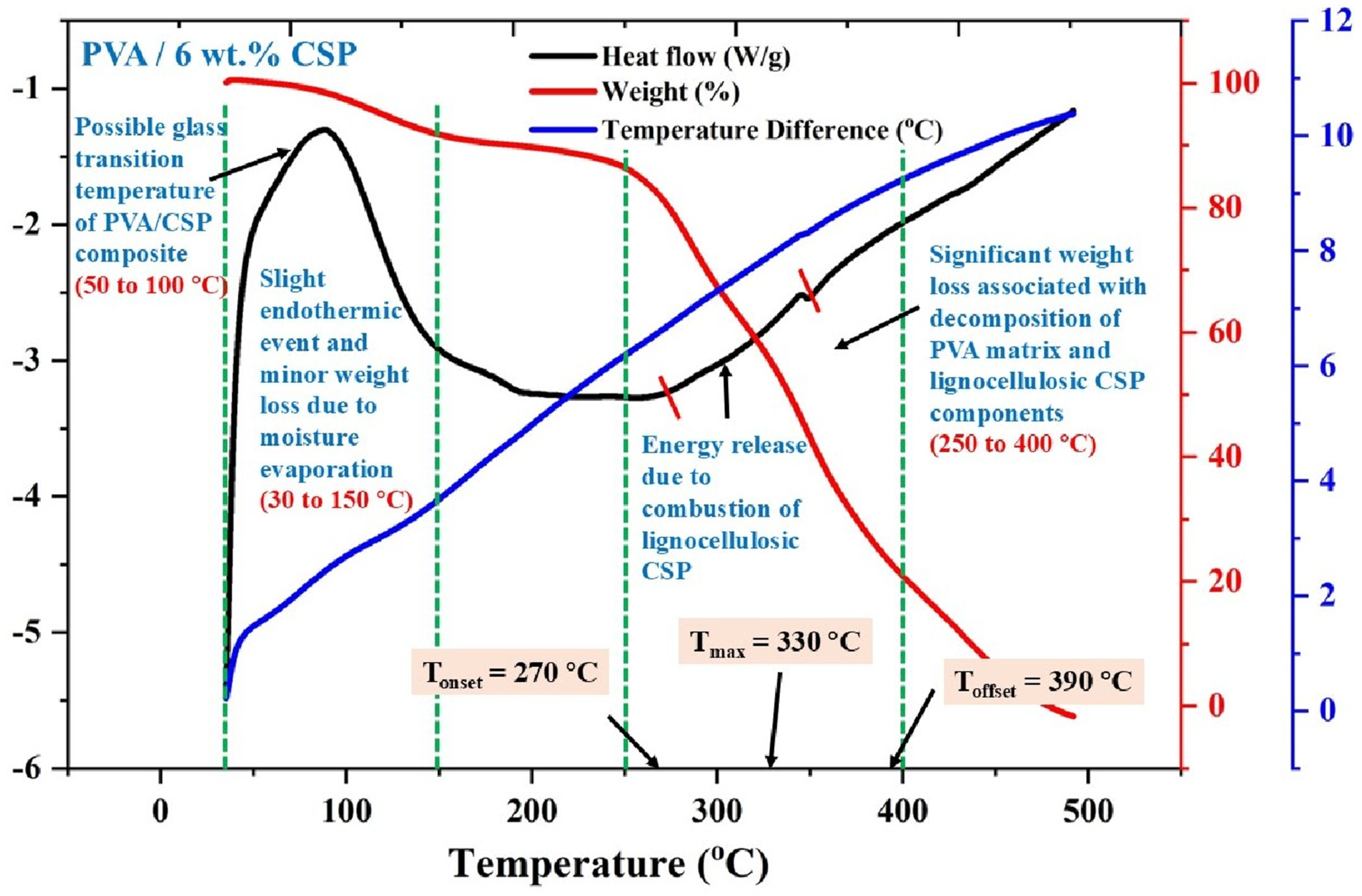
The initial weight loss and heat flow response between 30°C and 150°C corresponds to the evaporation of physically bound moisture. This range also coincides with a broad, shallow endothermic transition, which may be attributed to the glass transition temperature (Tg) of the composite matrix. The incorporation of CSP appears to slightly broaden and shift this transition, likely due to restricted segmental motion of PVA chains caused by filler–matrix interactions. The primary thermal degradation phase, associated with polymer chain scission and filler combustion, initiates at a slightly elevated onset temperature (Tonset = 270°C), compared to 260°C for neat PVA. This indicates a thermal stabilizing effect introduced by CSP. The maximum degradation rate (Tmax) also shifted from 320°C in neat PVA to 330°C in the composite, while the final degradation point (Toffset) extended to 390°C, further confirming the enhanced thermal resistance.
The broad exothermic peak in the DSC curve centered near T_max signifies the combustion of CSP’s lignocellulosic constituents, including hemicellulose and lignin, which contribute to thermal shielding via char formation. These degradation byproducts likely serve to limit oxygen diffusion and heat propagation, thereby delaying the decomposition of the matrix. The higher residual weight fraction at 500°C, although not directly visible in this spectrum, can also be inferred to be greater due to CSP’s inorganic ash-forming potential. The thermal response of the PVA/6 wt% CSP film reveals that CSP reinforcement not only preserves but improves the material’s high-temperature performance. This enhancement is attributed to synergistic effects arising from the thermal barrier function of CSP, its potential radical scavenging activity during chain scission, and its ability to restrict thermal motion at the polymer–filler interface. Collectively, these mechanisms validate CSP’s dual role as both a mechanical and thermal reinforcement in the biofilm architecture.
The improvement in thermal stability with CSP incorporation may be explained by the behaviour of the lignocellulosic phase during heating. CSP contains lignin, hemicellulose, and cellulose domains. Lignin decomposes slowly and produces a stable carbon-rich char. This char may act as a protective layer around the polymer chains during degradation. The layer might reduce heat transfer, delay oxygen diffusion, and limit the escape of volatile fragments. These effects could shift the main degradation events to higher temperatures. The TGA curves also show increased residual mass in CSP-filled films, which is consistent with higher char yield. The presence of the particulate phase may further restrict polymer mobility by creating more rigid micro-domains. This restricted mobility might suppress random chain scission at lower temperatures. The CSP particles may also introduce a mild barrier effect by interrupting the continuity of the polymer matrix. This barrier may influence the diffusion of decomposition gases and slow the propagation of thermal degradation fronts. These combined factors may explain the higher Tonset and Tmax values observed in CSP-reinforced PVA films.
The thermal stability trends observed in the TGA curves may be substantiated further by examining the residual char remaining at elevated temperatures. Although the present analysis focuses on onset degradation and major decomposition stages, the char content at 500°C provides an additional indicator of thermal-shielding behaviour. The neat PVA film shows minimal solid residue at this temperature, consistent with its predominantly carbon–oxygen backbone and limited aromatic content. The addition of CSP increases the residual mass fraction, and the 6 wt% CSP film retains a visibly larger char layer relative to the lower-loading samples. This increase may be attributed to the lignin-rich fraction of CSP, which is known to thermally decompose through pathways that favour aromatic carbon condensation rather than complete volatilization. The resulting char may act as a diffusion barrier that slows heat transfer and restricts the escape of volatile degradation products. This barrier effect might explain the delayed mass-loss rate and the broader decomposition plateau seen in the CSP-reinforced samples. The formation of a more thermally stable char network aligns with the observed shift in degradation behaviour and supports the argument that CSP contributes both structural stability and thermal-shielding capability during high-temperature exposure.
Water absorption
The water absorption behavior of the PVA/CSP biofilms was studied as a function of immersion time, and the results are illustrated in Figure 16. The analysis reveals a two-stage absorption profile typical of polymeric systems: an initial rapid uptake followed by a plateau corresponding to equilibrium saturation. The neat PVA film displayed the highest moisture uptake, reaching ∼6.4% after 480 minutes, attributed to the strong hydrophilic nature of the PVA backbone, which contains abundant hydroxyl groups capable of hydrogen bonding with water molecules. Moisture absorption of neat PVA and PVA/CSP biofilms over time, showing reduced uptake with higher CSP content.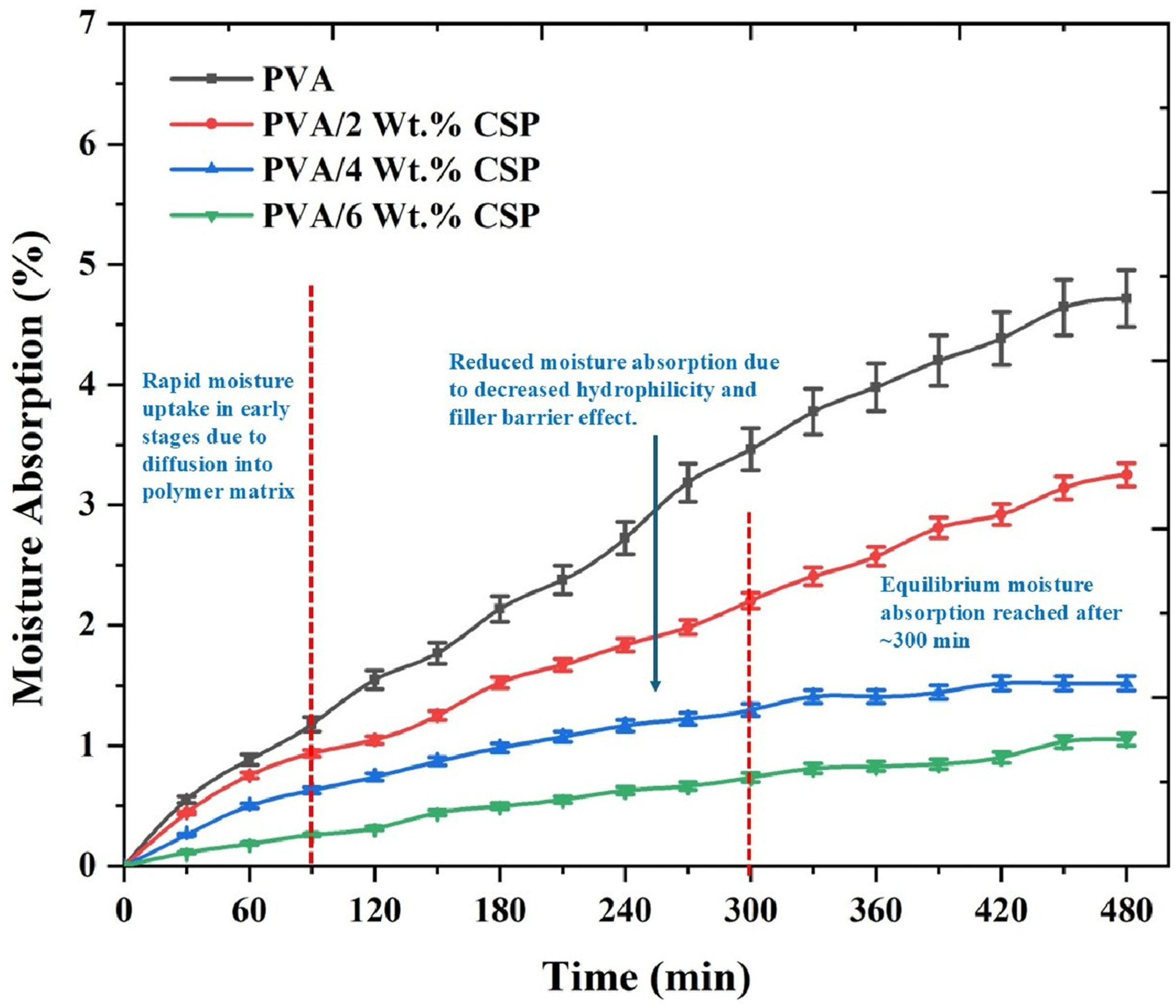
In the early stages (0–120 minutes), all samples showed rapid diffusion-driven water uptake. However, the incorporation of CSP significantly curtailed this uptake across all compositions. The PVA/2 wt% CSP sample exhibited a moisture absorption of ∼3.1%, while PVA/4 wt% and 6 wt% CSP composites demonstrated markedly lower values of ∼2.1% and ∼1.5%, respectively. The sharp reduction in absorption with increased CSP loading can be ascribed to two synergistic mechanisms: (i) decreased free volume and polymer chain mobility due to CSP-induced physical restriction, and (ii) filler barrier effect, wherein the dispersed CSP particles act as impermeable obstructions that retard water molecule diffusion through the polymer matrix.
The reduction in water absorption with increasing CSP content may be attributed to several concurrent microstructural effects. The XRD results indicate a rise in crystallinity with CSP addition, which might reduce the amorphous regions that normally permit easier diffusion of water molecules. The decrease in amorphous fraction may lower the available free volume, limit the mobility of polymer chains and slow moisture penetration. The particulate phase also introduces a physical barrier. The irregular CSP particles may interrupt the diffusion path, creating a more tortuous route for water. This obstruction effect could become more prominent at higher filler levels, where particle distribution is denser. The interfacial regions around CSP may also restrict swelling by partially masking hydroxyl sites of PVA. The combined influence of increased crystallinity reduced free volume, and barrier reinforcement may therefore control the overall trend, resulting in lower equilibrium moisture uptake in CSP-reinforced films.
The steady-state or equilibrium condition was generally achieved after ∼300 minutes, beyond which no significant moisture uptake was observed. This stabilization reflects a balance between water ingress and matrix saturation. The decreased equilibrium absorption in CSP-filled samples also suggests a reduction in polymer matrix hydrophilicity, potentially due to partial masking of PVA hydroxyl sites through CSP–matrix interactions and surface shielding. The observed trends underscore the functional advantage of CSP incorporation in improving the dimensional stability and environmental durability of PVA-based biofilms. This is particularly relevant for food packaging applications, where moisture ingress must be minimized to maintain mechanical integrity and barrier performance under humid conditions. The decline in water absorption also aligns with the enhanced crystallinity (Figure 6) and densified microstructure observed in SEM analysis, reinforcing the multifunctional role of CSP as both a structural and barrier-modifying reinforcement.
The present work focuses on gravimetric water absorption behaviour, which provides insight into the hydrophilicity of the PVA–CSP system and the influence of filler loading on moisture uptake. However, water absorption alone may not fully represent the barrier performance required for food-packaging applications. Important functional metrics such as water vapour transmission rate (WVTR) and oxygen permeability were not evaluated in this study. These properties govern mass-transport behaviour across the film thickness and determine suitability in moisture- or oxygen-sensitive packaging environments. The reduction in water absorption observed at higher CSP loadings may indicate the formation of a denser microstructure and reduced free volume, but this trend cannot be directly translated into WVTR or oxygen-diffusion performance without experimental verification. The absence of these measurements represents a limitation, and future work may focus on quantifying vapour and gas-barrier characteristics under controlled humidity and temperature conditions. Such data would allow a more comprehensive assessment of the material’s applicability in commercial packaging formats.
Biodegradation
The soil burial biodegradation behavior of the PVA/CSP biofilms was evaluated over a 30-day period, and the weight loss data is presented in Figure 17. The results clearly demonstrate a time-dependent increase in biodegradation for all samples, with the degradation rate significantly enhanced by the presence and concentration of coconut shell powder (CSP) in the composite matrix. Neat PVA exhibited the lowest biodegradation rate, with a weight loss of ∼10.8% after 30 days. This moderate degradation can be attributed to microbial hydrolysis and enzymatic oxidation of the hydroxyl-rich polymer chains under aerobic soil conditions. However, the compact microstructure and limited nutrient content in the pristine matrix restricted microbial colonization and enzymatic activity. Weight loss in soil of neat PVA and PVA/CSP biofilms over 30 days.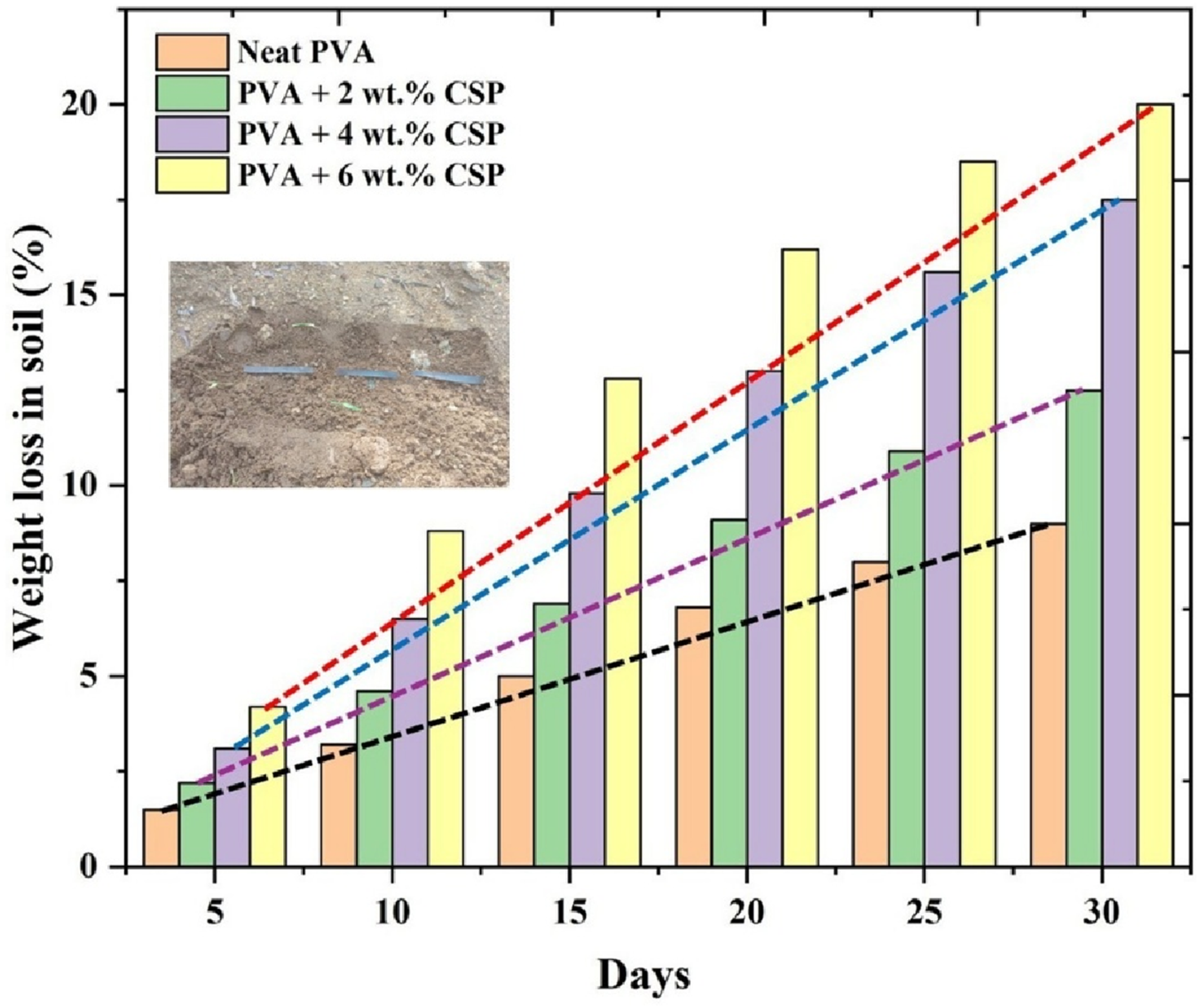
The inclusion of CSP drastically improved the degradability of the biofilms. The PVA/2 wt% CSP composite reached a weight loss of ∼15.4%, while PVA/4 wt% CSP and PVA/6 wt% CSP films recorded significantly higher weight loss values of ∼18.6% and ∼21.3%, respectively, at the end of the test duration. This trend is a result of the biodegradable and porous nature of lignocellulosic CSP, which acts as a microbial attractant and promotes enhanced matrix disintegration by serving as both a nutrient source and a facilitator of internal moisture retention.
Moreover, CSP particles introduce interfacial regions susceptible to microbial attack and water infiltration, weakening the film’s integrity over time. These micro-defects accelerate fragmentation, thereby increasing the available surface area for microbial colonization. The enhanced biodegradation observed in higher CSP loadings aligns with the reduced water absorption and increased surface roughness seen in previous analyses, which are known to favor environmental breakdown in soil conditions. The CSP-reinforced PVA biofilms demonstrate significantly improved environmental responsiveness and degradation kinetics, making them more suitable for short-term packaging and agricultural mulch applications where post-use bio-disintegration is essential. The results validate CSP’s dual function as a reinforcement and a biodegradation promoter within the polymer matrix.
The higher weight-loss values in CSP-reinforced films may be driven by the combined influence of microstructural and compositional factors. The lignocellulosic nature of CSP introduces biodegradable organic matter into the matrix. This material may act as an accessible nutrient source for soil microorganisms. The incorporation of CSP may also introduce micro-voids and interfacial regions that can increase local permeability. These regions might allow faster ingress of moisture and microbial enzymes. The increased moisture uptake in these local zones may soften the surrounding polymer chains and promote early-stage hydrolysis. The presence of cellulose, hemicellulose, and lignin fragments in CSP may accelerate colonization as these domains are known to degrade more readily under soil conditions. The formation of micro-cracks around the filler boundaries during environmental exposure may enhance microbial penetration and facilitate breakdown. These mechanisms together may explain why the weight loss increases from 10.8% in neat PVA to 21.3% in the 6 wt% CSP film over the same degradation period.
The solution-casting route adopted in this work is based on laboratory-scale processing, yet the underlying principles align with established film-forming methods used in industrial PVA-based packaging lines. The preparation of a PVA–CSP slurry at controlled viscosity may be adapted to continuous mixing units, where shear-regulated agitation can maintain stable particle suspension during large-batch production. The degassing and leveling stages may translate to slot-die or extrusion-casting systems, which are preferred in packaging industries for achieving consistent surface finish and film thickness. Drying under ambient conditions in the present study represents the early stage of solvent removal, but industrial systems generally rely on forced-air or infrared drying tunnels, and the rheology of the CSP-loaded formulation suggests that it may tolerate these higher drying rates without significant particle migration. The thermal and mechanical stability achieved at higher CSP loadings indicates that the composite film may withstand winding and roll-handling stresses commonly encountered in processing lines. The biodegradation and moisture-barrier improvements observed here may also support feasibility in food-contact applications, provided that regulatory migration limits and sanitation protocols are satisfied. The overall behaviour of the material suggests that scaling the process into a continuous manufacturing line is achievable, and the slurry characteristics indicate compatibility with existing polymer-film infrastructure.
The incorporation of CSP into PVA raises important considerations regarding food-contact safety, particularly in relation to potential migration and interaction with packaged materials. CSP is derived from a lignocellulosic matrix rich in lignin, hemicellulose, and trace mineral components, and these constituents may release low levels of organic molecules under prolonged exposure to moisture or heat. Although the present work does not quantify migration behaviour, the strong hydrogen-bonded network between PVA and CSP observed in the mechanical and structural analyses may restrict the mobility of extractable species. The reduction in water absorption at higher CSP loadings also suggests a tighter microstructure, which might limit diffusion pathways. However, migration tendencies cannot be assumed from moisture uptake alone, and regulatory evaluations typically require controlled tests under simulated food-contact conditions. Safety assessment may therefore include total migration, specific migration of organic residues, and evaluation of potential contaminants from the filler source. CSP is an agricultural by-product, and ensuring consistent pre-treatment and removal of extraneous residues is essential for regulatory compliance. It is believed that films with strong interfacial bonding and low free volume may exhibit reduced migration potential, but this requires explicit verification. The present study acknowledges this limitation and identifies migration and safety assessment as necessary steps before commercial adoption in food-packaging applications.
Conclusion
This study developed sustainable biofilms by reinforcing polyvinyl alcohol (PVA) with coconut shell powder (CSP), aiming to create biodegradable films with improved mechanical, thermal, and environmental properties suitable for eco-friendly food packaging. The work focuses on valorizing agro-waste and enhancing the functional performance of PVA through CSP incorporation. The ultimate tensile strength increased from 54 MPa for neat PVA to 83.6 MPa for biofilms with 6 wt% CSP, accompanied by a rise in yield strength from 43.1 MPa to 58.3 MPa, confirming significant mechanical reinforcement. Elastic modulus improved from 1.73 GPa (neat PVA) to 2.14 GPa (6 wt% CSP), and elongation at failure reached 42.3%, indicating that CSP enhanced both stiffness and ductility synergistically. Fracture surface analysis via SEM showed a transition from smooth ductile failure in neat PVA to energy-dissipative mechanisms such as crack deflection, particle pull-out, and fibrillation in CSP-reinforced films. FTIR spectra revealed new and intensified absorption bands linked to C–O and C = O stretching, evidencing strong interfacial hydrogen bonding and chemical compatibility between CSP and PVA. XRD analysis indicated an increase in crystallinity with CSP loading, with sharper peaks near 2θ = 20.1°, confirming CSP’s role as a nucleating agent enhancing molecular packing. Thermal degradation onset shifted from 260°C (neat PVA) to 270°C (CSP composite), and Tmax increased from 320°C to 330°C, indicating improved thermal resistance due to CSP’s lignocellulosic shielding effect. Water absorption decreased from 6.4% to 1.5% as CSP content increased, owing to the barrier effect and reduced polymer chain mobility. Biodegradation improved significantly, with weight loss rising from 10.8% to 21.3% over 30 days, demonstrating CSP’s dual role as reinforcement and biodegradation promoter.
These biofilms hold promise for single-use food packaging and mulch applications, with future scope focused on surface functionalization and property tuning for diverse biodegradable material systems.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
