Abstract
In this study, a biodegradable film based on guar gum (GG) was prepared. Aloe vera gel (AG) and date seed powder (DSP) were used to modify the structure of this film (GG/AG/DSP). The physicochemical and structural properties of GG/AG/DSP films such as thickness, water solubility, water vapor permeability (WVP), moisture, color and antioxidant activity were investigated. The results indicated that the addition of AG and DSP significantly increased the thickness, color indices a and b in the films. Water solubility, transparency and L index showed a significant decrease with the addition of AG and DSP. Moisture content and WVP showed a non-significant decrease when DSP was increased. However, when AG was added, WVP showed a significant decrease and moisture content showed a significant increase. The addition of DSP had no effect on elongation at break (EAB) and tensile strength (TS), but on the contrary, both of them increased significantly with the addition of Aloe vera gel. The SEM images showed that the type and amount of variables affected the surface morphology of the films and caused cracks and crevices in the film. FT-IR results showed electrostatic interaction between the biodegradable films. XRD analysis showed less crystallinity in the biodegradable film with the addition of AG and DSP. According to the TGA results, the thermal stability of the combined and optimized film samples was better than the pure GG film.
Introduction
A biodegradable film or coating is a material used for covering or packaging. Various biodegradable films are used to increase the shelf life of packaged products and are generally edible. Biodegradable films reinforce the natural layers of the product and prevent moisture loss, while also helping to control the passage of gases such as oxygen, carbon dioxide or ethylene. Other advantages of biodegradable films include having nutritional value, not polluting the environment due to being biodegradable, preventing enzymatic and non-enzymatic browning of food during storage, and increasing the strength and integrity of the food texture. These films and coatings are made from natural polymers of animals and plants such as polysaccharides (starch, cellulose and alginates), proteins (casein, zein and gelatin) as well as lipids and vaccines, mineral oils, paraffin oil, vegetable oils, paraffin wax and beeswax.1–7 Polysaccharide films can be used to change the packaging environment of fruits and vegetables and thus reduce their respiration rate. These films provide good protection against oxygen, fat, oil and flavor compounds at low relative humidity. However, due to their hydrophilic structure, they provide little protection at high relative humidity and need to be strengthened by adding other materials to enhance their physical and chemical structure.8–10
Guar is a polysaccharide with one of the highest molecular weights of all natural water-soluble polymers. The viscosifying effect of commercial guar gum can vary greatly depending on the molecular weight of the galactomannan. The gum contains 75–85% (true hydrocolloid), 5–6% protein, and 8–14% moisture. Guar gum is mainly composed of high molecular weight polysaccharides of galactomannans, which have been synthesized by various techniques such as chemical, physical, etc. In the food industry, guar gum is used as a novel food additive in various food products to stabilize food ingredients and as a source of fiber. Guar gum is used in biodegradable films and edible coatings to prevent oxygen penetration, reduce rancidity, prevent oxidation, slow down the respiration rate in fruits, etc..11, 12
Aloe vera, scientifically known as Aloe barbadensis miller, is a perennial plant with dark green leaves that grows in dry and hot climates. 13 This plant consists of two types of cells, chlorenchyma and parenchyma. Aloe vera gel is made up of soft and slippery tissues containing parenchyma cells. It is a transparent jelly-like substance and has a complex composition. Aloe vera gel is composed of 96% water and 4% dry matter. About 22.8% of the dry matter is organic acids. It also contains 18.80% dietary fiber, 8.8% polysaccharide, 4.7% protein, 2.7% lipid and 16% ash of the dry matter. In addition to a large amount of water, approximately 1% contains bioactive compounds such as aloin, emodin (anthraquinones). Flavonoids, saponin and aloemannan, in addition to a variety of amino acids and vitamins, are found in aloe vera gel. These bioactive compounds contribute significantly to the antibacterial and antioxidant activity of aloe vera gel.14, 15
Date seed powder contains approximately 3% moisture, 5–7% protein, 7–10% fat, 12–30% crude fiber, 55–70% carbohydrate, 0.5–0.9% calcium, 0.09–0.40% phosphorus, 0.30% potassium, 0.03% sodium. Polyphenols such as ellagic acid, chlorogenic acid, epicatechin, protocatechuic acid, and phenolic acid are found in date seed powder, which provide antioxidant and anti-browning properties. The different types of flavonoids present in date kernels have diverse health benefits, including antioxidant and radical scavenging activities, reduction of some chronic diseases, prevention of some cardiovascular disorders, and certain types of cancer processes.16,17
Considering the above-mentioned issues, in this study, guar gum was modified as a biodegradable film base with aloe vera gel and date seed powder. The results showed that this film has suitable mechanical properties. Also, this film showed about 50% antioxidant properties that can be used in active packaging of oxidation-sensitive food products such as various oils.
Materials and methods
Chemicals
Aloe vera leaves were purchased fresh from a local apothecary in East Azerbaijan Province (Tabriz, Iran). Date seeds were obtained from a local market in East Azerbaijan Province (Tabriz, Iran). Guar gum was obtained from Sigma (USA). Acetic acid (99% purity) was obtained from Merck (Germany). DPPH (2,2-diphenyl-1-picrylhydrazyl), glycerin and other chemical compounds used were obtained from Merck (Germany) and Sigma (USA).
Equipment and devices
The equipment and devices used were as follows: a sensitive balance with an accuracy of 0.001 g (model BP310S, Sartorius, Germany), a desiccator with a desiccant (silica gel), a pH meter AZ86505 (Taiwan), a Fourier transform infrared spectrometer (FT-IR) from Bruker (Germany), a bain-marie (model WD-23B, HYSC, South Korea), a scanning electron microscope (SEM) model MIRA4 from TESCAN, Czechoslovakia, a texture analyzer (TA. XT Plus, Stable Micro Systems), a ball mill (model F1100, Fidar, Iran), a digital micrometer (model 25A-3109), a homogenizer (model T 25 digital ULTRA-TURRAX, IKA, Germany), a mixer (hot plate magnetic stirrer, IKA RH basic 2, Germany), an ultrasonic bath from Iran, model 30S, and a thermal analysis weighing (TGA) model TGA2 from Mettler Toledo, Switzerland.
Extraction of aloe vera gel
Gel extraction from Aloe Vera leaves was performed by filleting. Leaves were selected with uniform maturity, size, color, and freshness and washed with tap water and then with distilled water. The outer layer of the green cortex was decolorized and the colorless gel matrix was physically removed. The extracted gel was homogenized in a blender. Then the mixture was filtered to remove impurities, pasteurized, cooled, and stabilized with acetic acid, and the prepared gel extract was stored at refrigerator temperature.
Preparation of date seed powder
After separating the seeds from the date flesh, the protective layer was removed and washed. Then the date seed were soaked in water for 2 weeks. Then they were kept in an oven for drying for 5 h at 80°C. The dried samples were converted into fine powder using a ball mill. A 0.25 mm sieve was used to ensure uniformity of the date seed powder particles. It was sterilized by autoclaving at 121°C for 15 min. The produced powder was stored in special plastic bags in the refrigerator until use.
Film preparation
List of prepared films based on central composite design.


The overall film production process (A) and example of produced composite films (B).
Film tests
Thickness measurement
The thickness of the films was measured by a digital micrometer at 10 random points of the film. This average thickness of each film was used to calculate the mechanical and water properties.
Mechanical properties
For this test, the film samples were cut into 2 × 7 cm and in order to condition the films, the samples were placed for 72 h at a relative humidity of 55% (calcium nitrate) and a temperature of 25°C. The distance between the two jaws of the device was set to 50 mm and the speed of the jaws was set to 50 mm/min, and the relevant mechanical properties (tensile strength (TS) and elongation at break (EAB)) were reported using the device.
Solubility in water
For this purpose, the films were cut into 2 × 2 cm pieces and dried and weighed after being placed in an oven at 105°C. The weighed film was immersed in 30 ml of distilled water and stirred for 3 h. Then, the entire set was passed through a Whatman filter paper with a specific weight of two microns, and the insoluble solids and filter paper were dried in an oven at 105°C for 24 h. The solubility percentage of the films was calculated according to the following equation:
Moisture content
To determine the moisture content of the prepared films, the film samples were cut into 3 × 3 cm2 pieces, then weighed and dried in an oven at 110°C until constant weight was reached, and the moisture percentage was reported according to the following equation:
Water vapor permeability (WVP)
To perform this test, the films were cut into circles with a diameter of 1 cm and placed in the lids of vials containing 3 g of calcium sulfate (RH = 0%). After initial weighing, the vials were placed in a desiccator containing potassium sulfate (RH = 97%) and then the vials were weighed at regular intervals for 72 h. The water vapor transmission rate (WVTR) (g/m2h) was calculated from the slope obtained from the regression analysis of the amount of moisture transferred to the film surface in a certain period of time using equation (3). The WVTR of the films was then used to calculate the water vapor permeability using equation (4).

WVTR: Water vapor transmission rate (g/m2h), X: Film thickness (m), A: Film surface area (m2), WVP: Water vapor permeability (g.mm/m2. Pa. s), ΔP: Water vapor pressure difference between the inner and outer surface of the film in the vials (ΔP = 3169 Pa).
Film transparency
A Canon digital camera was used to determine the surface color of the film samples. The color of the films was determined based on the Hunter lab light-dark (L), green-red (a) and blue-yellow (b) parameters. A mobile phone camera (Samsung-Galaxy A21S) and Color-Grab software were used to measurement color properties. Also, UV-Vis spectrometry was used to calculate the transparency. Here, the films were cut into 1 × 5 cm pieces and placed in the cell of the device and the film absorbance was measured. The following equations were used to measure transparency:

Antioxidant activity
The antioxidant activity of the films was tested by determining the DPPH free radical scavenging power. First, 6 cm2 of the film sample was placed in a test tube and two mL of methanol was poured into it. The contents of each tube were mixed and digested with a complete vortex. After centrifuging the solution for 10 min at 3000 rpm, 1 mL of the supernatant solution was mixed with 1 mL of methanol and 2 mL of DPPH methanol solution (0.06 mM). The control sample was also prepared by mixing 2 mL of methanol and 2 mL of DPPH methanol solution without the presence of the film samples. After 30 min at room temperature and in the dark, their absorbance was measured at a wavelength of 517 nm by spectrophotometer and the percentage of DPPH radical scavenging was obtained using the following equation: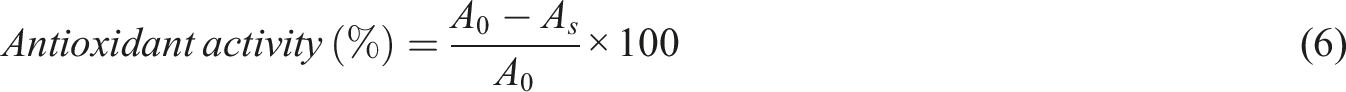
A0: absorbance of the control sample, AS: absorbance of the analyte
Scanning electron Microscopy (SEM)
The surface morphology of the films was studied using a scanning electron microscope (MIRA4, TESCAN, Czechoslovakia). In this device, electrons are irradiated to the film surface and after reflection in the detector, they are converted into photons and finally provide a visible image. Due to the insulating nature of the films, the samples were coated with gold and platinum using a polisher model SC-7620.
Fourier transform infrared spectroscopy (FT-IR)
The FTIR test was performed using an FT-IR device (Perkin Elemer, USA). Thin film (1 mm) was obtained by mixing of film samples and dry potassium bromide in a ratio of 1:20 and a pressure of about 60 kPa. The spectrum of the samples was recorded in the range of 400-4000 cm-1 with a resolution of 1 cm-1.
X-ray diffraction (XRD)
To investigate the crystal structure of the films, an X-ray device model (Bruker AXS, Karlsruhe, Germany) was used. To perform this test, the settings of the X-ray generator were as follows: 40 kV, 3 mA, 0.02-degree step distance at a scan speed of one degree per minute and a wavelength of 0.1539 nm. The reflected radiation was collected at ambient temperature and in the angular range of 2θ = 0-80°.
Thermogravimetric analysis (TGA)
TGA test was used to study the effect of AG and DSP on biodegradable films based on GG. In order to remove volatile compounds, the samples were first placed in a vacuum oven for 1 hour at 50°C. Then, 10 mg of the samples were placed in the device and heated at a rate of 10°C/min and in the temperature range of 25 °C–605°C.
Statistical analysis
In this study, the effect of AG and DSP on the physicochemical properties of the prepared films were investigated using the central composite design (Table 1). The response surface method was used for statistical analyses. Statistical analyses were performed at a 95% probability level, and Design Expert 10 software was used for variance analysis, determining response patterns and models, and drawing graphs.
Results and discussion
Thickness and mechanical properties
The thickness of food packaging films affects several important properties, including strength, light transmission, and WVP. Therefore, biopolymers must have a thickness that provides adequate mechanical and barrier properties. The mechanical properties of films depend on the intermolecular forces of the polymer chains forming the films, the ratio of the constituent components, the additives added, and the environmental conditions. Tensile strength, the maximum stress required to break the film, is evaluated during a tensile test. Tensile strength is measured with a texture analyzer, which is based on the longitudinal stretch of the film to the point of its rupture, which is called the elongation at break.
Figure 2 shows the three-dimensional (3-D) curves, perturbation, and mathematical models of the effect of the amount of AG and DSP on the thickness, TS and EAB GG-based films. 3-D curves, perturbation and mathematical models of the effect of the amount of AG and DSP on the thickness, EAB and TS.
The 3-D curve study and the results of variance analysis of the addition of AG and DSP on the thickness of biodegradable film samples showed that the addition of AG and DSP powder each alone increased the thickness of the samples containing GG. However, in general, according to the results obtained, the highest thickness was related to the use of both substances with the highest dose (500 mg AG and 100 mg DSP) in the prepared films. This increase can be attributed to the increase in solids (DSP), which caused an interruption in the formation of the matrix network and the interruption of the biopolymer structure and the trapping of water in the network due to its fibrous properties, as well as the formation of a covalent network between GG and AG and the trapping of water between the created pores, resulting in swelling caused by them. 18
According to the curves and mathematical models related to mechanical properties, significant changes were observed with increasing the amount of Aloe Vera gel in the EAB and TS, such that an increase was observed in both of the aforementioned components. The highest value was also observed in the highest amount of AG without the presence of DSP. As explained earlier, this increase was related to hydrogen bonds, covalent connections and interactions between GG and AG, resulting in the formation of a stronger matrix network than the control sample. Also, no significant relationship was observed with the addition of DSP to the film containing GG in both tests. However, with the increase in DSP, the amount of EAB and TS decreased slightly. This decrease is probably due to their non-uniform distribution and agglomeration in the film matrix, which is also shown in the results of SEM images, which can cause a further decrease in flexibility of the films. The decrease in film flexibility can also be attributed to the anti-plasticizing behavior of DSP. The results of this study are consistent with the results of Hadi et al. (2021) who studied the addition of Aloe vera gel to a biodegradable film based on sodium alginate. 19 In another study conducted by Lawal et al. (2023), the addition of DSP to a film containing carboxymethyl cellulose reduced the tensile strength and elasticity. They attributed this decrease to the presence of cellulose in the DSP. 20
Solubility, moisture content and WVP
Water solubility is an important property of biodegradable film that determines water resistance. Because it indicates the performance of biofilm in preserving foods with high water activity, frozen and fresh. WVP is an essential parameter for packaging materials. To increase the shelf life of a product, interactions between the packaged product and its environment should be as low as possible. Factors that have a direct effect on the permeability and speed of passage of different gases include the hydrophilicity or hydrophobicity of the materials, the amount and type of materials used, the method of preparation and distribution of the added materials, the network structure created and the resulting path, the density and thickness, the presence of seams in the films, and the size of the solid particles.
Figure 3 shows the 3-D curves, perturbation and mathematical models of the effect of the AG and DSP on the solubility in water, moisture content and WVP of the films. According to the solubility results, the solubility of the prepared films decreased by about 10% with increasing the amount of aloe vera gel, and also by increasing the amount of DSP, a 20% decrease was observed compared to the sample without AG and DSP. However, the greatest decrease occurred when the AG and DSP were simultaneously involved in the formation of the film with the highest amount. The reason for this can be the creation of crosslinks between the functional groups of GG, AG and DSP, as well as the interaction between the proteins of aloe vera gel and the carboxyl groups of GG. These factors reduced the availability of hydrophilic agents and also reduced the power of water to break the bonds, resulting in a decrease in the solubility of the films. These results were not consistent with a study in which the addition of Aloe vera gel to films based on guar gum and ε-polylysine was carried out. In that study, the increase in hydrophilic sugars and proteins was considered the reason for this.
21
The moisture content of the prepared samples according to the results related to moisture was between 26 and 60% depending on the amount of AG and DSP. Analysis of variance showed that the ratio of AG and DSP had a significant effect (p < 0.05) and their interaction had a non-significant effect on the moisture content. By increasing the amount of AG from 0 to 500 mg, the moisture content of the films increased significantly. The increase in moisture content resulting from the combination of AG with GG can be explained by the presence of OH and hydrophilic functional groups in their structure and polyphenol in the structure of AG and the formation of hydrogen bonds with water that lead to the formation of a matrix network, which is the same factor that maintains moisture in the pores. Similar results were reported by Girgin and Tugrul (2023) when adding AG to a biodegradable film based on dried walnut shells.
22
Also, the relationship between DSP and moisture content was inverse. As the amount of DSP increased, the moisture content of the film decreased. This decrease was due to the increase in the amount of solids and the reduction of voids in the combination of GG and DSP. These results were consistent with the research by Ramaraj and Poomalai (2006) regarding the addition of coconut shell powder to a biodegradable composite film based on polyvinyl alcohol.
23
3-D curves, perturbation and mathematical models of the effect of the amount of AG and DSP on solubility, moisture content and WVP.
According to the results of WVP, the addition of DSP caused a decrease in WVP. By adding DSP up to 100 mg, the permeability decreased from 0.35 to 0.21. The reason for this can be attributed to the increase in solids, the decrease in the availability of hydroxyl groups, and the increase in the matrix network. However, the greatest effect on the reduction in water permeability occurred when the maximum amount of AG (500 mg) was used without the presence of DSP. As previously mentioned, the interaction effect between GG and AG causes the formation of a regular network, reducing the knots of GG, which has also been proven in the SEM test. Previous research on the effect of adding AG on the film containing starch and chitosan partially confirms the results of the present study. 24
Film color and opacity
One of the important factors in the selection, acceptability, and physical quality of the product is the color and appearance of films. In general, the more transparent a polymer film is and the more similar it is to plastics made from synthetic polymers, the more its acceptance and use will increase. Because the appearance characteristics of the packaged product will be fully visible in it. Most biodegradable packaging films are transparent and colorless. However, in some cases, due to the sensitivity of the food product to light and the loss of its nutritional compounds due to photo oxidation, as well as the color matching of the contents with the packaging material in order to attract the consumer’s attention, the use of light-blocking and color-creating compounds in the matrix of the packaging material seems necessary. Figure 4 shows the 3-D curves, perturbation, and mathematical models of the effect of the amount of AG and DSP on the color characteristics and transparency of the films. 3-D curves, perturbation and mathematical models of the effect of AG and DSP on the color properties and transparency of the films.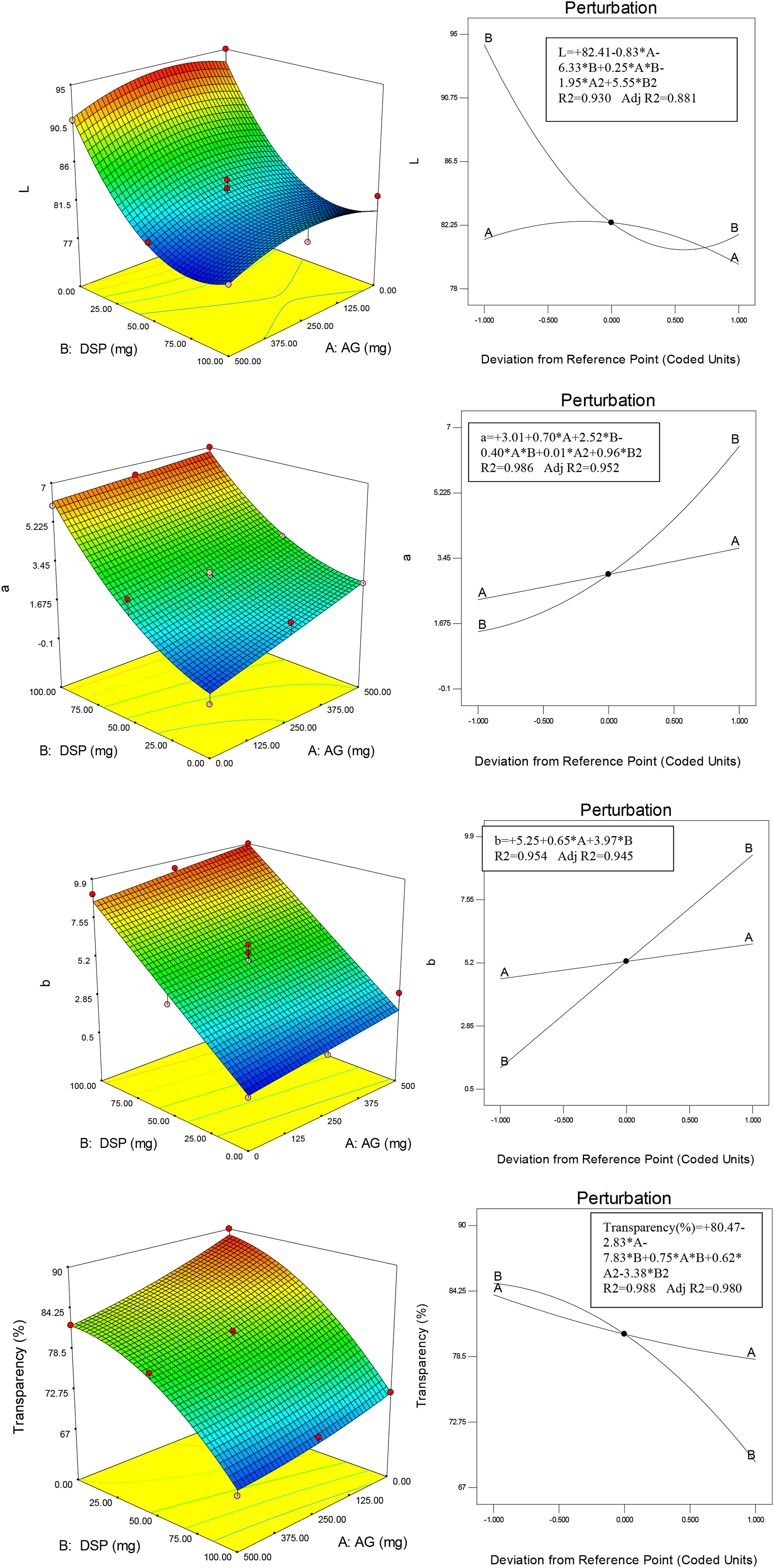
According to the curves and mathematical models obtained, the effect of adding AG on the L index was insignificant and on a and b) was significant (p < .05). The L index decreased from 95 in the control sample to 91 in the sample containing the maximum amount of AG, which was not a significant effect. Also, the addition of AG showed a significant increase in the a and b indices. So that the film containing the maximum amount of AG showed a tendency towards a dark green color. The reason for this can be attributed to the color of AG, which is green. The results obtained were consistent with the research conducted by Hadi et al. (2021) on the sodium Alginate-Aloe vera bio composite film. 19 The effect of adding DSP on all three color parameters was significant. With increasing amount of DSP, the L index decreased and the a and b parameters showed a significant increase. This is due to the brown pigments of the DSP and its distribution throughout the film. The effect of adding anthocyanin on carboxymethyl cellulose/agar based smart packaging film, which was investigated by Roy et al. (2021), also resulted in a decrease in the L index and an increase in a and b. 25 In this study, UV-vis spectrum at 600 nm was used to measure the transparency of the films. The transparency and opacity of the films are directly related to the amount of light that passes through them. In addition, consumer perception is affected by the transparency of a film. In addition, food packaging materials can reduce the degradation of light-sensitive food components. The transparency of the films is determined by the mixing and compatibility of the polymer blend. As a result, measuring the film transparency can provide valuable information about the film microstructure. The effect of adding AG and DSP each alone is significant. The control films showed the highest transparency (90%), while the film containing the highest amount (500 mg) of AG had the lowest transparency (79%). By adding DSP to the film containing GG and AG with the maximum amount, the transparency in the film decreased to (67%) and the resulting turbidity caused a decrease in the light transmission in the mentioned films. This could be due to the absorption and scattering of light by AG and DSP as well as the oxidation of phenols. These results were consistent with the data reported by Amin et al. (2019) on composite films prepared from beeswax-chitosan and Aloe vera gel. 26
Antioxidant property
DPPH assay is often used to evaluate the antioxidant activity or hydrogen donating capacity of phytochemicals or bioactive components present in biodegradable films. DPPH activity depends on the extent to which the DPPH radical (dark purple) is reduced by the antioxidant (hydrogen donor) present in the film to a light yellow compound. DPPH has a characteristic absorption at a wavelength of 517 nm when dissolved in alcohol. Antioxidant activity varies depending on the chemical nature of the phenolic compounds present in the starting materials. For example, hydroxyl groups along the aromatic ring can potentially donate hydrogen or electrons to free radicals, while aromatic rings can stabilize and relocate unpaired electrons. However, the addition of antioxidants reduces the absorption value.
Figure 5 shows the 3-D curves, perturbation and mathematical models of the effect of the amount of AG and DSP on the antioxidant properties of the films. According to the results, the addition of AG and DSP had a significant effect on the antioxidant properties. In the control sample, the amount of antioxidant activity was 12%. While this amount increased to 46% when the maximum amount of AG was added. When the highest weighted amount of DSP was added to the matrix film containing GG and AG with the highest amount, it reached 48%. In a previous study, it was reported that the phenolic compounds present in AG contribute to the radical scavenging activity.
18
The fact that the GG film containing AG and DSP has about 50% antioxidant properties indicates that this film is considered as an active film and can be used in the packaging of food products sensitive to oxidation. For example, this film can be used to package various oils that are subject to oxidative deterioration. 3-D curves, perturbation, and mathematical models of the effect of the amount of AG and DSP on the antioxidant properties of the films.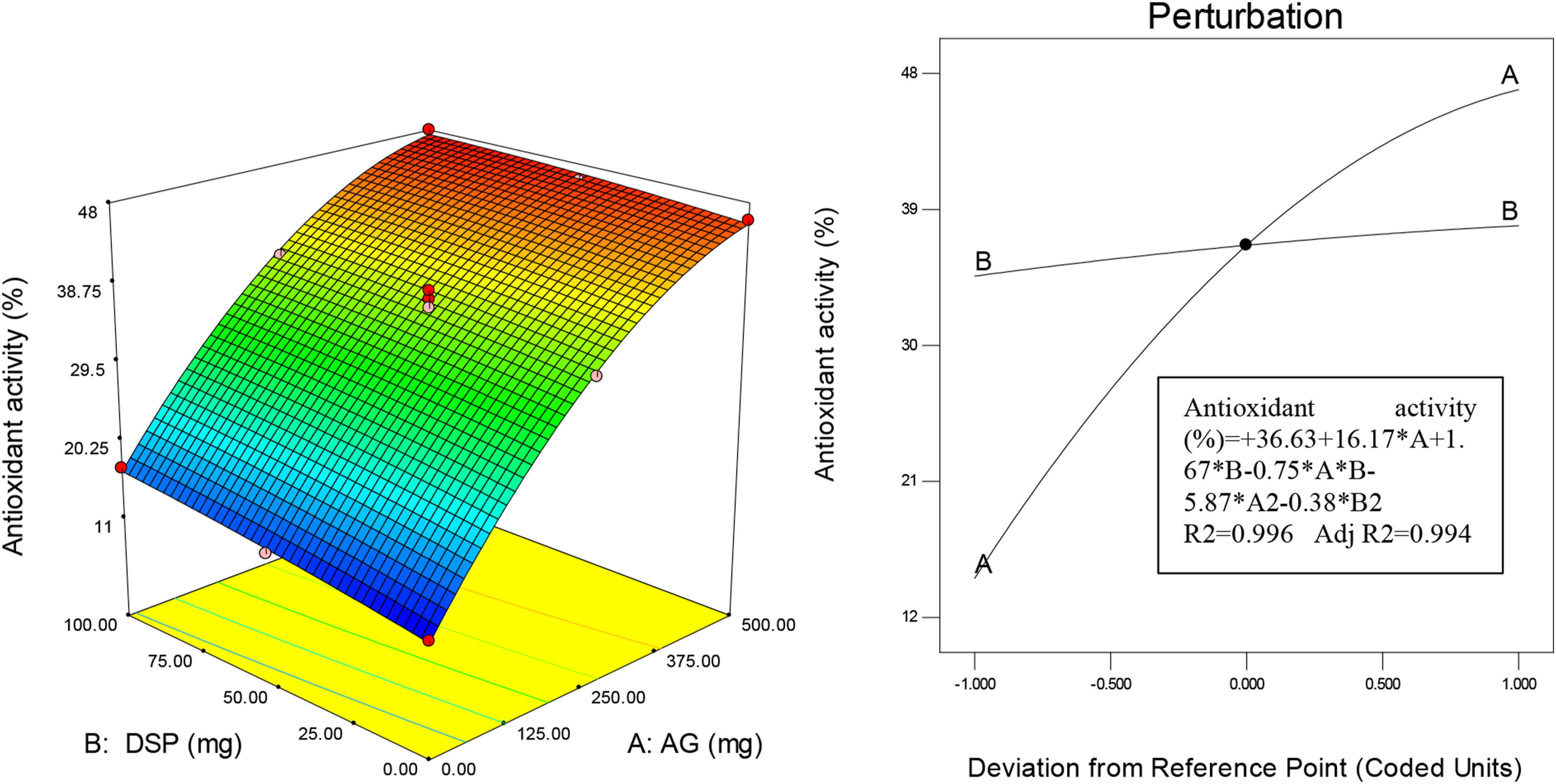
SEM images and FTIR spectra
Figure 6 shows SEM images and FTIR spectra of GG films and its various composites. According to the SEM images, a knotted structure is observed in all films, which can be due to the formation of granules or micelles in the film-forming solution. The presence of GG, which is responsible for creating the spatial mass, may have arisen due to its branched structure. In the images of the composite films, cracks are seen, which indicate GG/GG and GG/AG interactions and hydrogen bonds between GG and AG. Also, small and large holes are observed in these images, which can be related to the bursting of bubbles through the surface during water evaporation, which increases the surface roughness. The images of the composite of GG/AG/DSP show the presence of AG and DSP, which has a rough surface compared to other film samples, in which the DSP caused the formation of fine pores and also prevented further bonding of AG with GG, which caused the formation of more white knots in the film and also caused the dispersion of aloe vera gel and reduced cracks caused by hydrogen bonds. SEM images (A) and FTIR spectra (B) of GG films and its various composites.
According to the results of FTIR spectroscopy, the transmittance pattern of all samples was in the range of 400 to 4000 cm-1. No significant difference was observed in the peaks in the spectrum of films containing AG and DSP. In the film sample pattern, the O-H and C-H stretching vibrations of the alkane groups in the polymer chain of the gum were found in the range of 3000 to 3400 cm-1and 2922 cm-1, respectively. 27 In addition, the region of 3000 to 3400 cm-1contains the N-H stretching vibration peak, which overlaps with the O-H group. Also, the peak at 2922 cm-1 indicates the presence of aliphatic groups and NH3 of free amino acids. 28 The absorption peak at 1743 cm-1 indicates the presence of C = O stretching and vibrational groups or carboxylic acid ester groups, and the other peak at 1590 cm-1 is related to the asymmetric O-C-O stretching vibrations of the gum. The peak found at 1370 to 1373 cm-1 was attributed to the CH2 ester sulfate group of GG. The peak at 1232 cm-1 represents the stretching vibration of the C-O group of esters and phenols. Also, the peaks in the range of 1100 to 1102 cm-1 can be due to the C-O stretching of the polysaccharides of GG and aloe vera gel in C-OH bonds. The peaks between 717 and 926 cm-1 are related to the C = C and C-H bonds of the aromatic ring, which in the case where DSP was added, many peaks were created, indicating an increase in the aforementioned bonds. 29 The peak at 845 cm-1 can be attributed to the presence of mannose and galactose in GG, which is related to the presence of glycosidic bonds. Finally, by comparing the spectra of different films, it is observed that the same peaks in different spectra have shifted to higher or lower wavenumbers, which indicates physical connections and electrostatic interactions between the components of the composite.
XRD and TGA
The physical state (crystalline or amorphous) of the films affects other parameters such as their optical, mechanical and barrier properties. For this reason, X-ray diffraction analysis was performed on the film samples produced in the study. Figure 7 shows the XRD spectra and TGA curves of GG films and its various composites. XRD spectra (A) and TGA curves (B) of GG films and its various composites.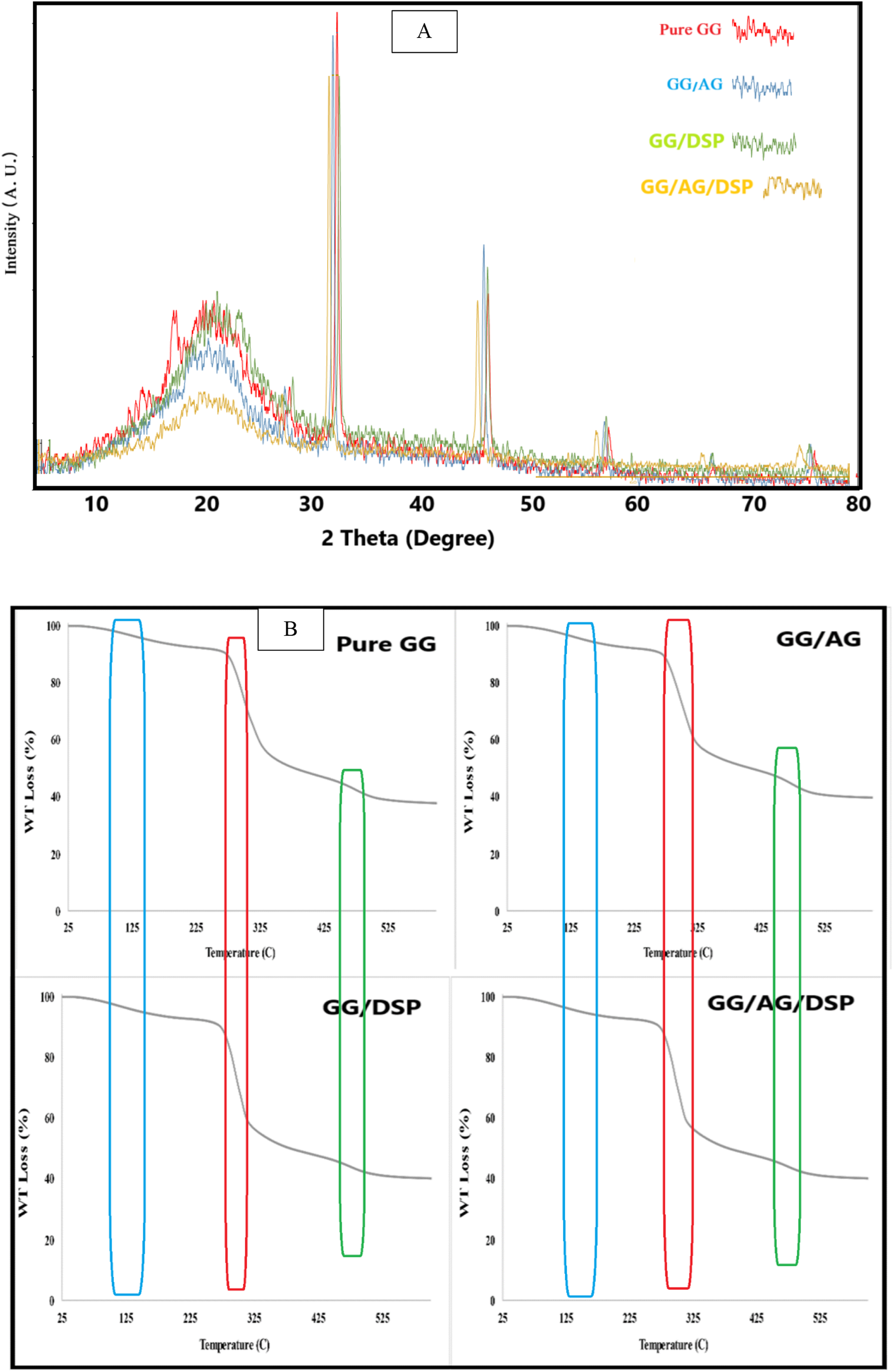
According to the obtained XRD spectra, in the spectrum of the pure GG film, peaks at 2θ = 18°, 32°, 46°, 57° and 76° were observed. The peaks at the 2θ of 32° can be attributed to the interactions of GG molecules, which confirms the semi-crystalline structure of GG. The peaks at 46° and 57° can also be related to the presence of glycerol and acetic acid in the film. 30 According to the spectrum of other films formed, the addition of single AG and DSP, as well as the addition of both of them in GG-based films, did not have much effect on changing the position of the peaks. In addition, they caused a decrease in the intensity of the peaks at angles 18° and 32°, which indicated a disruption in the formation of a regular and crystalline network due to possible interactions between GG and AG, which partially destroyed the initial crystalline structure. The reason for this could be due to the amorphous structure of AG and DSP or their low concentration, which did not have much effect on changing the position of the peaks. The results were consistent with the reports of Bhatia et al. (2024) on XRD analysis of biodegradable films based on GG and calcium caseinate combined with clary sage oil and the study of Gürler (2022) on the addition of Aloe vera gel on a composite film based on gelatin, chitosan and starch.31,32
According to the TGA curves for all samples, a weight loss of about 11% was recorded at temperatures between 60 and 235°C. This loss indicates the loss of moisture and its evaporation from the film samples. The moisture content is crucial for the formation of intact thin films, as it helps in the plasticization of GG. The slight decrease in moisture loss due to -OH groups prevents further loss and the escape of water vapor. 33 The next loss occurred in the temperature range between 210 and 315°C, resulting in a loss of 35%. This stage can be considered as pyrolysis, where glycerol and other volatile compounds are decomposed. 21 In the third stage, weight loss happens in temperatures from 325 to 600°C, which indicates the decomposition of the polysaccharide structure. In the film samples containing AG and DSP, the resistance to decomposition was higher than in the control sample in the third stage, which indicates the interaction of AG and DSP with GG. The results obtained were consistent with the study of Karim et al. (2021) regarding film samples based on polyethylene-starch modified with Aloe vera gel and palm oil. 34
Conclusion
The results of the effect of two variables, AG and DSP, on the physicochemical properties of biodegradable films based on GG showed that the thickness of the films increased significantly when AG and DSP were added. Adding AG to films prepared from GG without the presence of DSP increased the EAB and TS, but the presence of DSP caused a non-significant decrease. Water solubility showed a significant decrease with the addition of AG and DSP. Regarding the WVP property, the effect of DSP on the reduction of the mentioned component was not significant, but in films containing AG, a significant decrease in the WVP was observed. The moisture content decreased insignificantly with the increase in DSP, but with the increase in AG, the moisture content of the films increased significantly. With the addition of AG, the L index decreased and the a and b indices increased, which led the prepared films to a dark green color. Also, the addition of DSP caused a significant decrease in the L index and an increase in a and b. Regarding transparency, with the addition of AG and DSP, the transparency decreased and the films became relatively turbid. According to SEM images, the addition of AG and DSP caused the formation of seams and holes on the film surface. FT-IR results showed electrostatic interaction between the biodegradable film sample containing GG, AG and DSP. XRD analysis showed that adding AG and DSP did not change the position of the peaks much, but rather reduced their intensity, which reduced the crystalline structure in the film and led them to an amorphous structure. According to the TGA results, the combined and optimized film had better relative thermal stability than the control sample, which could be due to the interaction between the three main components of the films. The general conclusion that was obtained was that by increasing the amount of AG and DSP, the antioxidant properties of the prepared films increased, and the produced film with suitable mechanical properties and strong antioxidant properties has the ability to be used as an active film in the packaging of food products sensitive to oxidative spoilage.
Footnotes
Author contribution
Sajad Pirsa conceived of the presented idea, Sajad Pirsa developed the theory and performed the computations. Sajad Pirsa verified the analytical methods. Ramin Hossein Pour discussed the results and contributed to the final manuscript. Ramin Hossein Pour out the experiment. Sajad Pirsa and Samira Alizadeh wrote the manuscript and revised it.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
