Abstract
The increased demand for non-renewable, petroleum-based plastics and their negative impacts on human and environmental sustainability has led researchers to develop bio-composites to make eco-friendly materials from renewable and natural resources. These materials minimize plastic manufacturing carbon footprints while performing well. In this study, NaOH treatment of Mimusops elengi seed shell powder (MESSP) affects density, chemical functionality and composition, surface shape, and thermal degradation. It also examines the properties of polypropylene (PP) composites incorporating untreated and NaOH-treated MESSP, with sodium hydroxide concentrations ranging from 5 to 8 wt%. A series of PP composites containing untreated and NaOH-treated MESSP, with filler loadings varied between 0 and 20 wt%, was produced via melt mixing method in an internal mixer. MESSP particle treatment with 5 wt% NaOH effectively enhanced the chemical functionality, cellulose content, and particle morphology as confirmed through FTIR and SEM analysis. The treatment of MESSP resulted in a noticeable reduction of hydroxyl (OH) functional groups, confirming a decrease in its hydrophilicity. Morphological analysis revealed increased porosity in the treated MESSP and improved adhesion of the PP matrix to MESSP. At 15 wt% loading (optimum), PP/TMESSP composite exhibited 30% and 28% enhancement in tensile strength and impact strength, respectively compared with PP/MESSP composite. Since they absorbed less water than PP/MESSP composites, PP/TMESSP composites had superior water absorption resistance. This study could lead to eco-friendly polymeric materials for packaging, automobile, and construction.
Keywords
Introduction
Fossil fuel-based plastics have been a part of our daily lives ever since their development in 1907. Various types of fossil fuel-based plastics are used in numerous household and industrial applications, primarily in packaging for bottles and plastic bags, construction materials, electronic cables and insulation, textiles, and medical devices. However, the continuously growing demand for fossil fuel-based plastics contribute to substantial environmental concerns, due to increasing waste from plastic disposal, majorly from post-consumer and industries. The environmental and societal impacts of commercial plastics are largely attributed to their non-degradable nature and high chemical stability. Overconsumption of petroleum-based plastic contributes to land scarcity due to the unmanaged disposal of plastic waste through landfilling and illegal dumping, the release of hazardous and green house gases during incineration, and the depletion of non-renewable petroleum-based resources. Growing concern over the excessive use of plastic, leading to the uncontrolled accumulation and prolonged shelf life of end-of-life plastic waste, has raised global interest in developing sustainable alternatives. This has led to an intensified focus on bioplastic, bio-composites, and natural fiber-based polymer composites as eco-friendly and sustainable alternative.1,2
The expanding functional landscape of polymers, driven by their structural tailoring-ability, diverse applications, and potential for performance enhancement, has opened new directions in material science, attracting significant academic and industrial interest.3–5 Moreover, the advantages of natural fibers over synthetic fibers, including their low density, renewability, recyclability, biodegradability, competitive strength, and environmental compatibility, have garnered significant interest among researchers and industrialists. 6 Introduction of different types of natural fibers into polymer matrix sourced from cultivated plants or agricultural waste, such as jute, sisal, kenaf, banana, basalt, hemp, coconut shell, bamboo, nutshell, and vetiver, has been reported in recent years.7–11 Natural fibers or fillers can be extracted from various parts of plants, including stems, leaves, roots, seeds, and bark. However, the cultivation of specific fiber producing plants such as bamboo, hemp, jute, kenaf, and sisal demands proper agronomic practices and extensive plantation area. Additionally, the over-reliance on wood-sourced fiber-filled composites may contribute to deforestation. 12 Consequently, there is a growing focus on incorporating agricultural waste into polymers. The key sustainability and ecological advantage of natural fillers derived from waste lies in their abundance. Utilizing agricultural waste can help mitigate the issue of unmanageable disposal while addressing the shortage of wood supply. 13
Research on natural fiber filled polypropylene (PP) composites has been extensively reported previously. PP remains the preferred thermoplastic polymers for natural fiber reinforcement due to its high versatility in both inherent properties and processing methods, aligning with industrial requirements for commodity and domestic applications.14–17 The global demand for PP continues to rise annually due to its advantageous properties and versatility, including low density, tailored mechanical properties, enhanced thermal performance (depending on grade used), ease of processing, cost-effectiveness, and excellent chemical durability. As a result, the demand for PP has increased across multiple sectors, with global market consumption in 2022 reaching approximately 79.01 million metric tons. Additionally, by 2030, global demand is projected to rise to 104.99 million metric tons. 18 Therefore, selecting appropriate fillers and ensuring their compatibility with the PP matrix are crucial for meeting current industrial requirements and addressing global concerns over sustainable development.
Over the past few decades, natural fibers have become increasingly crucial as fillers and reinforcing elements in developing polymer composites and nanocomposites. Nonetheless, a significant challenge in producing natural fiber-filled polymer composites (NFPCs) lies in the inadequate compatibility and adhesion between natural fibers and the polymer matrix, resulting in low resistance to moisture absorption, poor thermal stability and weathering, and low durability. Thus, suitable fiber selection if important in producing a NFPC with balanced production cost and overall properties. 19 The incorporation of natural fibers from various sources into PP matrices has been the subject of several investigations. Many of these studies have reported that the poor adhesion between the filler and PP matrix results in a loss of mechanical capabilities. The inherent hydrophilicity of natural fibers renders them incompatible with the hydrophobic PP matrix, ultimately deteriorating the polymer composite system.20,21 Various chemical treatments of fibers and polymer matrix have been investigated to address the incompatibility between natural fibers and polymer matrix. These treatments either reduce the hydrophilicity of the natural fibers or introduce hydrophilic functional groups into the polymer chain, which may react with the filler surface to enhance wettability and interfacial adhesion between the filler and polymer matrix. 22
Researchers tend to focus more on treating natural fibers rather than modifying polymer functional groups, owing to the simplicity of the process, cost-effectiveness, preservation of polymer properties, and reduced solvent usage, which also can cause environmental hazards. 23 Examples of chemical treatments for natural fibers include alkaline, acetylation, silane, benzoylation, peroxide, enzymatic, and many others.24,25 Among these chemical treatments, alkaline treatment has been the most widely used in treating various natural fibers due to its high effectiveness in surface modification, lowest cost and simplicity of the treatment as compared to other chemical treatments. 26 Filip et al. (2024) explored the effect of alkaline treatment on waste almond and hazelnut nutshell using NaOH solution and reported that 6% NaOH pretreatment of the waste nutshell (WN) effectively improved the crystallinity index and surface roughness, by effectively removing the unwanted constituents such as lignin, hemicellulose and pectin. 27 Madu et al. (2025) investigated the effect of alkaline treatment of palm kernel shell particles using an 8 wt% NaOH solution and reported a significant improvement in the wear and abrasion resistance of palm kernel shell reinforced polypropylene composites, making them suitable for piping applications. The study demonstrated that alkaline surface modification enhances filler–matrix interfacial interaction, thereby improving abrasion resistance, and concluded that NaOH-treated palm kernel shell particles are effective reinforcements for polypropylene in applications where high wear resistance is required. 28 Recent studies have also reported the use of various agricultural waste derived nutshell fillers, including peanut, macadamia, and pumpkin seed husks, in polypropylene matrices. In general, addition of seed and nutshell-based filler in PP causes poor mechanical and chemical performance of the composites due to the incompatibility between the hydrophobic PP matrix with the hydrophilic lignocellulose component of the natural fillers. It was also reported that, alkaline pretreatment of the nutshell filler has been widely employed to improve the interfacial adhesion, stress transfer efficiency, and overall composite performance. 29 The comparison between various conventional and unconventional chemical treatments for different types of natural fibers in developing sustainable bio-composites has been addressed in the past. The review emphasized the surface roughness enhancement, elimination of impurities, and reduction of hydrophilicity of several natural fibers, such as coconut, sisal, jute, flax, and banana fiber through alkaline treatment, which strengthens the adherence of the polymer matrix to the fiber surface. 30
In previous researches, mimusops elengi seed shell powder (MESSP) has been introduced as a sustainable filler in polypropylene (PP) and polyvinyl alcohol (PVOH).31–33 The addition of MESSP to polymers has shown great potential as an alternative natural fiber source for polymer composite production, owing to its low cost, low density, and satisfactory mechanical properties. The composition of MESSP is comparable to other agricultural waste fillers such as almond and hazelnut nutshell ranging between 30 and 50% cellulose, 28 – 35% hemicellulose and 20 – 32% lignin, which supports the functional potential of MESSP as natural filler in PP, thereby extending the current portfolio of agricultural waste-based reinforcement materials. 27 Nevertheless, MESSP showed comparable processing characteristics to those of other reported natural fiber-filled polymer composites (NFPCs). At an optimum loading of 20 wt%, MESSP improves the impact strength and tensile modulus, while retain the water absorption and thermal stability as comparable to neat PP. Chemical functionality analysis and morphological observations of the PP/MESSP composite revealed that MESSP particles are only partly compatible with the PP matrix, primarily due to the presence of hydroxyl groups, which weaken interfacial adhesion. 34 Therefore, evaluating the optimal reinforcement capability of MESSP in PP through surface treatment is crucial, as it may significantly enhance the performance and durability of PP/MESSP composites for high-performance applications.
Past research indicates that pretreatment with a 5% NaOH solution is effective in enhancing the mechanical properties of natural fiber-reinforced bio-composites by selectively removing the amorphous components of the natural fibers. However, pretreatment with highly concentrated NaOH solutions exceeding 7.5% can damage and cause the degradation of the cellulose structure, leading to inferior mechanical performance. 35 Therefore, in this study focus have be given to study the effect of different concentrations of sodium hydroxide (NaOH) aqueous solution (ranging from 5 to 8 wt%) on the chemical functionality, surface morphology, particle size distribution, elemental composition, and thermal stability of MESSP.
Furthermore, the influence of treated MESSP (TMESSP) on the thermal stability, tensile and impact properties, water absorption, and morphological characteristics of PP composites at the optimum NaOH treatment concentration and MESSP loading is evaluated and compared with those containing untreated MESSP.
Experimental
Materials
Polypropylene (PP) in the form of white pellets was procured from Lotte Chemical Titan (M) Sdn. Bhd, Malaysia. The material has a density of 0.99 g/cm3, a melt flow index (MFI) of 1.4 g/min, a melting temperature in the range of 160 – 165°C, and a water absorption rate of 0.02%. The sodium hydroxide (NaOH) pellets were obtained from Lab Medical Science, Malaysia, with a density of 2.13 g/cm3 and a melting temperature of 318°C. Analytical Grade (AR) 99.5% acetone was supplied by R&M Chemicals, with molecular weight of 58.05 g/mol. AR grade toluene with molecular weight of 92.14 g/mol and purity of ≥99.9 %, was supplied by Rank Synergy Sdn. Bhd., Selangor, Malaysia. The AR grade nitric acid with 65 – 68% purity and molarity of 14.54 – 15.22 M, and AR grade sulfuric acid with 95 – 98% purity and molarity of 14.63 – 15.10 M, were supplied by Crystal Carnation Sdn. Bhd. Selangor, Malaysia.
Extraction and alkaline treatment of MESSP
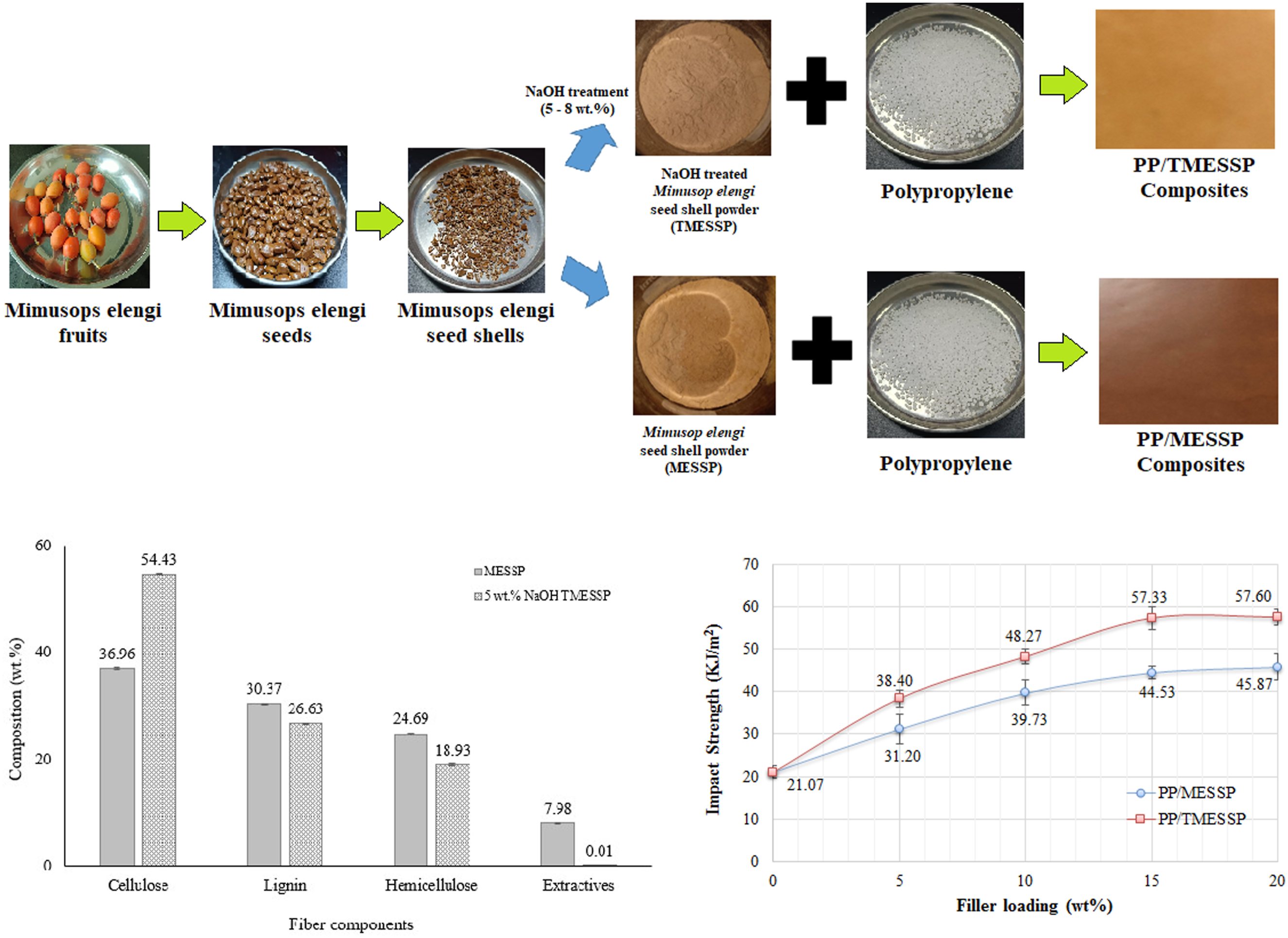
The seed shell of ripened mimusops elengi fruits were used to produce the MESSP. The fruits were collected from trees located in Taman Kampar Perdana, Kampar, Perak, Malaysia (Google map coordinates: 4°20′20″ N, 101°9′9″ E). The seeds were extracted by gently squeezing the fruits and then dried at 80°C for 24 hours using an oven (Memmert, Schwabach, Germany), until the seeds turned brownish color, indicating completely dried seeds. The seeds were subsequently crushed using a compression molding machine (GT-7014-A30C, GOTECH Testing Machines Inc., Taichung City, Taiwan), operated at room temperature, processing approximately 100 g per batch. The seed kernels were then manually removed to obtain the seed shells, which were ground using a universal powder mill grinder (Golden Bull SY-116, Selangor, Malaysia) and sieved to a particle size <45 µm using a RO-TAP® Sieve Shaker Model RX-29–10 (W.S Tyler’s, United States). 31 The alkaline treatment of MESSP was performed according to Ref. 36. MESSP was chemically treated with NaOH solutions at concentrations of 5, 6, 7, and 8 wt%. The MESSP-to-NaOH ratio was maintained at 1:15, and the mixture was left to stand at room temperature for 24 hours. Following treatment, the excess solution was removed by washing the MESSP with deionised water until a neutral pH (pH = 7) was achieved. The treated MESSP was then oven-dried at 80°C for 24 hours (Memmert, Schwabach, Germany).
Composite preparation
Compounding formulation.
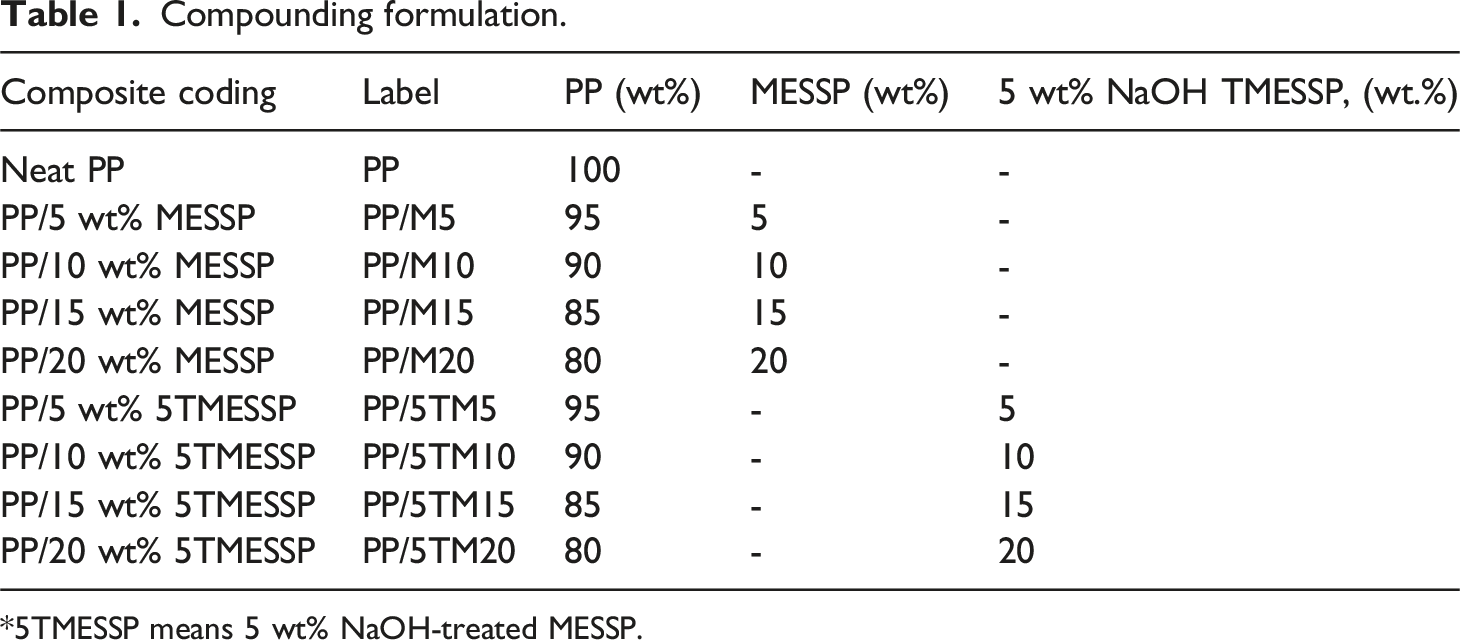
*5TMESSP means 5 wt% NaOH-treated MESSP.
Preparation of PP/MESSP and PP/TMESSP composites.
Characterizations and testing
Fourier transform infrared spectroscopy (FTIR) analysis
Fourier Transform Infrared (FTIR) analysis of MESSP and TMESSP was performed using FTIR spectrometer (Spectrum RX1, Perkin Elmer Inc., Norwalk, CT, USA) to identify and compare the chemical functionalities of MESSP before and after NaOH treatment. FTIR spectra were recorded in the range of 400 – 4000 cm−1 at a resolution of 4 cm−1, using four scans per sample. KBr pellets was thoroughly mixed with MESSP and finely ground to obtain a uniform powder, which was then compressed into a thin pellet for FTIR analysis.
Particle size analysis
Malvern Mastersizer particle size analyzer model: Hydro 2000 MU (Malvern Instrument Ltd, Malvern, UK) was used to performed the analysis on untreated and treated MESSP to evaluate their particle size distribution, uniformity, mean particle size and specific surface area. Prior to the measurement, the refractive indices of both materials were determined using a digital refractometer (Model 300034, Sper Scientific Ltd, Scottsdale, AZ, USA), yielding values of 1.3335 for untreated MESSP and 1.2635 for NaOH-treated MESSP.
Scanning electron microscope analysis
The surface morphology of both untreated and NaOH-treated MESSP particles, and the tensile fracture surfaces of the resulted PP composites, was examined using a scanning electron microscope (SEM), Model: JOEL JSM 6701-F (Jeol, Akishima, Japan) operated at an accelerating voltage of 4 kV. Prior to imaging, samples were mounted on a carbon-taped specimen holder and coated with a thin layer of platinum using a vacuum sputter coater (JFC-1600, Jeol, Akishima, Japan) to prevent electron charging.
Density test
Helium was used as the displacement gas in a Micromeritics gas pycnometer (AccuPyc II 1340, Impexron GMBH, Malaysia) to measure the densities of MESSP and TMESSP. Using a 10 cm3 sample chamber and 10 purging cycles before measurement, the test was performed in five repetitions.
Chemical composition analysis
All chemical composition analysis was conducted using five independent replicates (n = 5) for each sample. The number of extractives in MESSP was evaluated using the solvent extraction method, employing a Soxhlet HT2 extractor and acetone as the extraction solvent. A total of 1 g of MESSP was combined with 150 mL of acetone. The extraction was carried out for a total of 4 hours, with each rising stage lasting for 20 minutes, while the temperature was maintained at 90°C. The sample was then dried in an oven at 105°C for 24 hours. The content of extractives (A) was determined from equation (1)37,38:
The cellulose content on a dry basis was determined using the Kurschner and Hoffer method, which involves a nitric acid-ethanol mixture in a 1:4 ratio.39,40 One g of MESSP was mixed with 100 mL of solvent, consisting of 80 mL (95% ethanol) and 20 mL (concentrated nitric acid).
41
The mixture was refluxed in a water bath maintained at 90°C for three cycles, each lasting for an hour. After refluxing, the residue remaining was oven-dried for 24 hours at 105°C and then cooled in a desiccator before weighing the cellulose (Wcellulose).42,43 The cellulose content (B) in percentage was calculated using equation (2).
The Klason lignin method
44
was used to evaluate the lignin content in MESSP. In this procedure, a 2 hours hydrolysis of MESSP was carried out in a water bath (temperature maintained between 19 and 21°C), by mixing 1 g of MESSP with 15 mL of 72% sulfuric acid. The acid mixture was then diluted to 3% by adding 545 mL of distilled water. Using a reflux condenser, the diluted mixture was then continuously boiled for another 4 hours. The residue remaining after reflux was then cooled to room temperature and filtered. The filtered residue was then placed in a pre-weighed crucible (Wcrucible), and dried in an oven set at 105°C for 24 hours.
40
Upon drying, the weight of the crucible containing the dried residue (Wcr) was then measured. The correction for ash content in lignin was performed by placing the crucibles and residue in the muffle furnace (LH 120/12, Nabertherm GmbH, Germany) at 575°C for 4 hours. The sample was then cooled in a desiccator for 1 hour before the weight of the crucible containing ash (WAsh) were recorded. The content of acid-insoluble lignin (C) was determined from equation (3).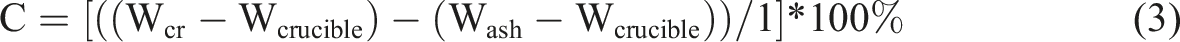
The hemicellulose content was determined by subtracting the measured amounts of extractives, lignin, and cellulose from the total biomass. As shown in equation (4), it was assumed that the biomass consisted solely of cellulose, hemicellulose, extractives, and lignin.45,46 To compare the compositional changes, the same procedure was repeated for TMESSP.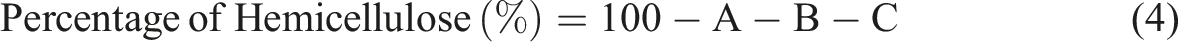
Thermogravimetric analysis
A Mettler Toledo thermal analyser (TGA/SDTA851e, Mettler-Toledo International Inc., Schwarzenbach, Switzerland) was used to determine the thermal stability of MESSP, TMESSP, and the composites based on weight loss due to thermal decomposition through thermogravimetric analysis (TGA). The test was performed from 30°C to 600°C at a heating rate of 20°C/min under a nitrogen atmosphere with a flow rate of 50 mL/min.
Tensile test
A lightweight tensile tester (Tinius Olsen H10KS-0748, Salfords, UK) was used to evaluate the tensile properties of the PP composites using the ASTM D638 test method. Five test specimens with three replications for each composition were prepared using a dumbbell cutter (Leader Technology Scientific (M) Sdn. Bhd., Balakong, Malaysia). The crosshead speed was set at 20 mm/min using a 450 N load cell.
Impact test
Using a pendulum impact tester (model 5101, Zwick, Germany), the Izod impact test was conducted according to the ASTM D256 test method to determine the impact strength of PP composites containing untreated and NaOH-treated MESSP at various loading. Five specimens with dimensions of 64 mm in length, 12.7 mm in width, and 3 mm in thickness were prepared, with three replications for each composition. The test was performed using a pendulum with a swing energy of 12.5 J, swinging at an average velocity of 3.54 m/s. The impact strength was then determined using equation (5).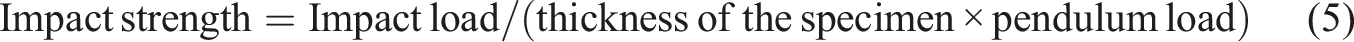
Water uptake
The percentage water uptake by PP composites was evaluated through a water swelling test, following the ASTM D570-98 test method. Five test specimens, with three replications for each composition, were prepared using a dumbbell cutter (Leader Technology Scientific (M) Sdn. Bhd., Balakong, Malaysia). The specimens were dried in an oven at 50°C for 24 hours and then cooled in a desiccator to room temperature. The initial dry weight of each specimen (Wi) was measured using a Sartorius M-Pact precision balance, Model AX224 (Sartorius AG, Germany). The weighed composite specimens were then placed in a glass bottles containing fixed volume of distilled water and kept under a dark environment for 24 hours. Upon immersion, the test specimens were carefully taken out, gently wiped with dried cloth or tissue paper, and then weighed to obtain the swollen weight (Wf). Using equation (5), the water uptake percentage was evaluated.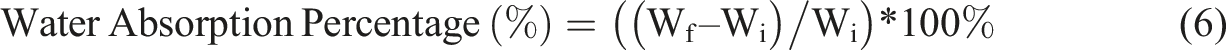
Results and discussion
FTIR analysis of MESSP and TMESSP
FTIR spectra of untreated MESSP and NaOH-treated MESSP (TMESSP) with various concentrations of NaOH are shown in Figure 2. A comparison between the spectra of untreated and NaOH-treated MESSP reveals a large reduction in the peak intensity of at 3332 cm−1, corresponding to the OH (hydroxyl) functional group, after NaOH treatment. Additionally, this peak shifted from 3332 cm−1 to 3337 cm−1, reflecting the reduction of OH groups and a weakening of hydrogen bonding within the cellulose structure of MESSP.
1
During the NaOH treatment, hemicellulose constituents, such as acetyl groups, which contribute to methyl C-H stretching, are removed, as evidenced by the reduced intensity of the peak at 2920 cm−147 The removal of carbonyl (C=O) groups from hemicellulose is confirmed from the absence of peak at 1730 cm−1 after treatment.
48
This finding aligns with the study by Kamran et al. (2022), which reported the removal of hemicellulose and lignin from natural fibers, indicated by the disappearance of the 1730 cm−1 peak.
49
FTIR spectra of MESSP and TMESSP at different NaOH concentrations.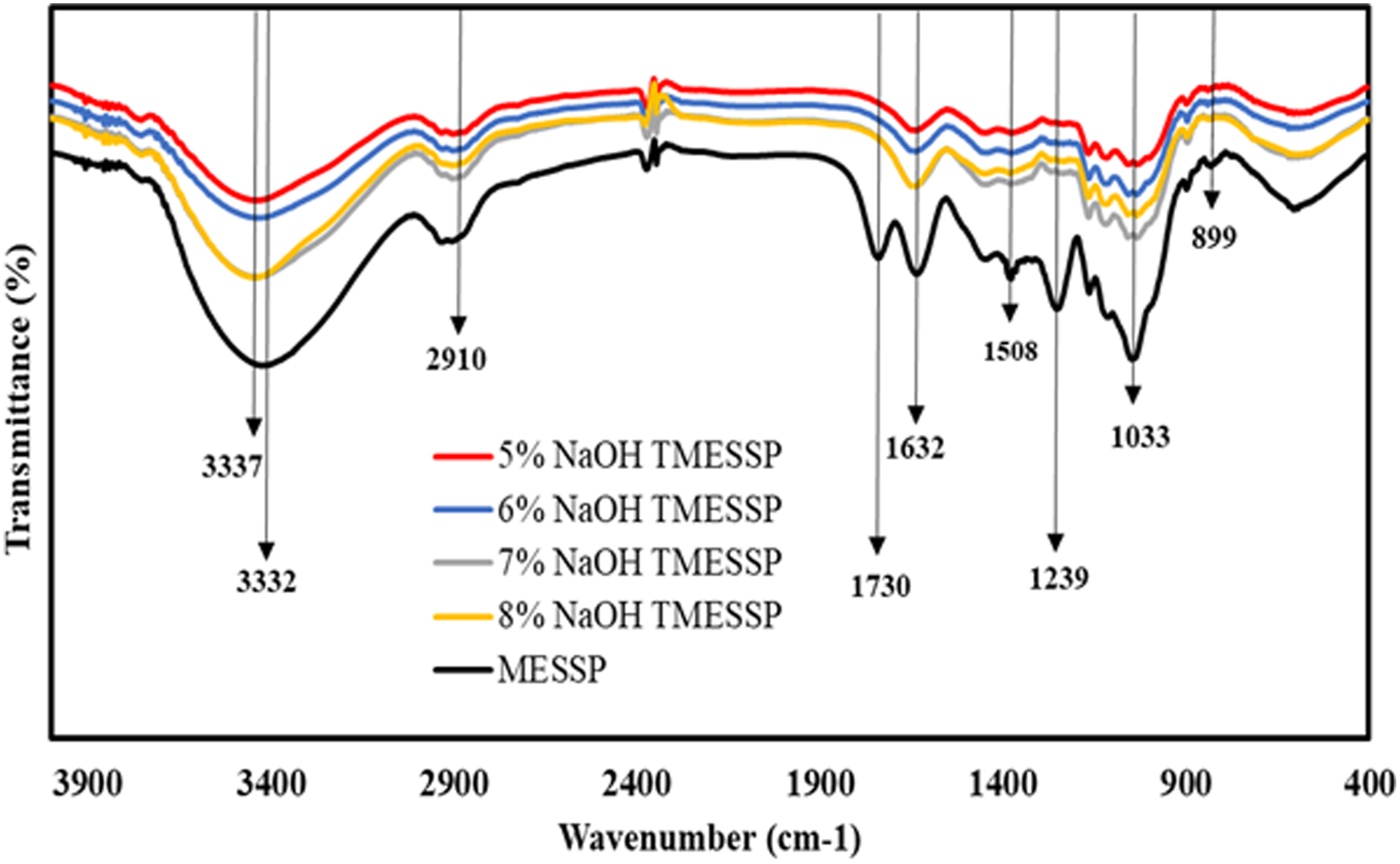
Moreover, the hydrophilicity of MESSP was also reduced following alkaline treatment, as indicated by the decreased intensity of the OH-related peak from absorbed moisture at 1632 cm−11 suggesting that TMESSP has a lower water absorption capacity than MESSP. A minor peak at 1508 cm−1, attributed to aromatic ring stretching and C-C vibrations in lignin, was observed in MESSP but absent in TMESSP, indicating lignin removal. Similarly, the strong peak at 1239 cm−1 in MESSP, associated with lignin and hemicellulose was not present in TMESSP, supporting the partial elimination of these components through alkaline treatment. 49 Furthermore, the lower band intensity at 1033 cm−1 in TMESSP compared to MESSP suggests the loss of carboxylate substituent from hemicellulose or lignin. 50 The disappearance if the TMESSP peak at 899 cm−1 indicates the dissolution of amorphous cellulose during chemical modification. 51
Notably, treatment with 5 wt% NaOH resulted in the most significant reduction in the intensity of the peaks at 3332 cm−1 and 1632 cm−1, both associated with OH groups in cellulose. Compared to treatment with 6, 7, and 8 wt% NaOH, this confirms that 5 wt% NaOH is the optimal concentration for reducing the hydrophilicity and hygroscopic characteristics of MESSP.
Particle size analysis and density MESSP and TMESSP
The particle size distribution curves for MESSP before and after treatment with various NaOH concentrations are shown in Figure 3, while Table 2 presents the results of particle size analysis and density comparison. A comparison between untreated MESSP and MESSP treated with 5 wt% NaOH reveals that both exhibited a similar particle size distribution, characterized by a narrower range and high uniformity. However, the uniformity of treated MESSP decreased significantly at higher NaOH concentrations (6–8 wt%), indicating a broader particle size distribution. According to Khoaele et al. (2024), surface treatment of natural fibers removes cellulose extractives, results in a cleaner surface, reduces microvoids, and thereby enhances fiber uniformity. However, excessive chemical treatment can create more microvoids and increased surface roughness, thereby reducing uniformity and broadening the particle size distribution.
52
The uniformity results shown in Table 2 align with this finding, confirming that higher NaOH concentrations reduce particle size uniformity due to the formation of microvoids. On the other hand, the specific surface area of MESSP increased after treatment with 5 wt% NaOH but decreased at higher NaOH concentrations (6–8 wt%). The effective removal of hemicellulose, lignin, pectin, wax, and oil-coating components during alkaline treatment reduces particle size while increasing the specific surface area and roughness of the fiber particles.
52
As shown in Table 2, treatment with 5 wt% NaOH results in a reduced mean particle size and increased specific surface area. However, at higher NaOH concentrations (6–8 wt%), the mean particle size increased, while the specific surface area decreased gradually. These observations suggest that 5 wt% NaOH is the optimal concentration for effective surface treatment of MESSP, yielding smaller particle sizes and higher surface area compared to untreated MESSP. Although there is a general trend of decreasing specific surface area with increasing NaOH concentration beyond 5 wt%, the maximum surface area was recorded for the 5 wt% NaOH TMESSP. Distribution by number of particles in percentage of MESSP and TMESSP. Particle size analysis data of MESSP and TMESSP.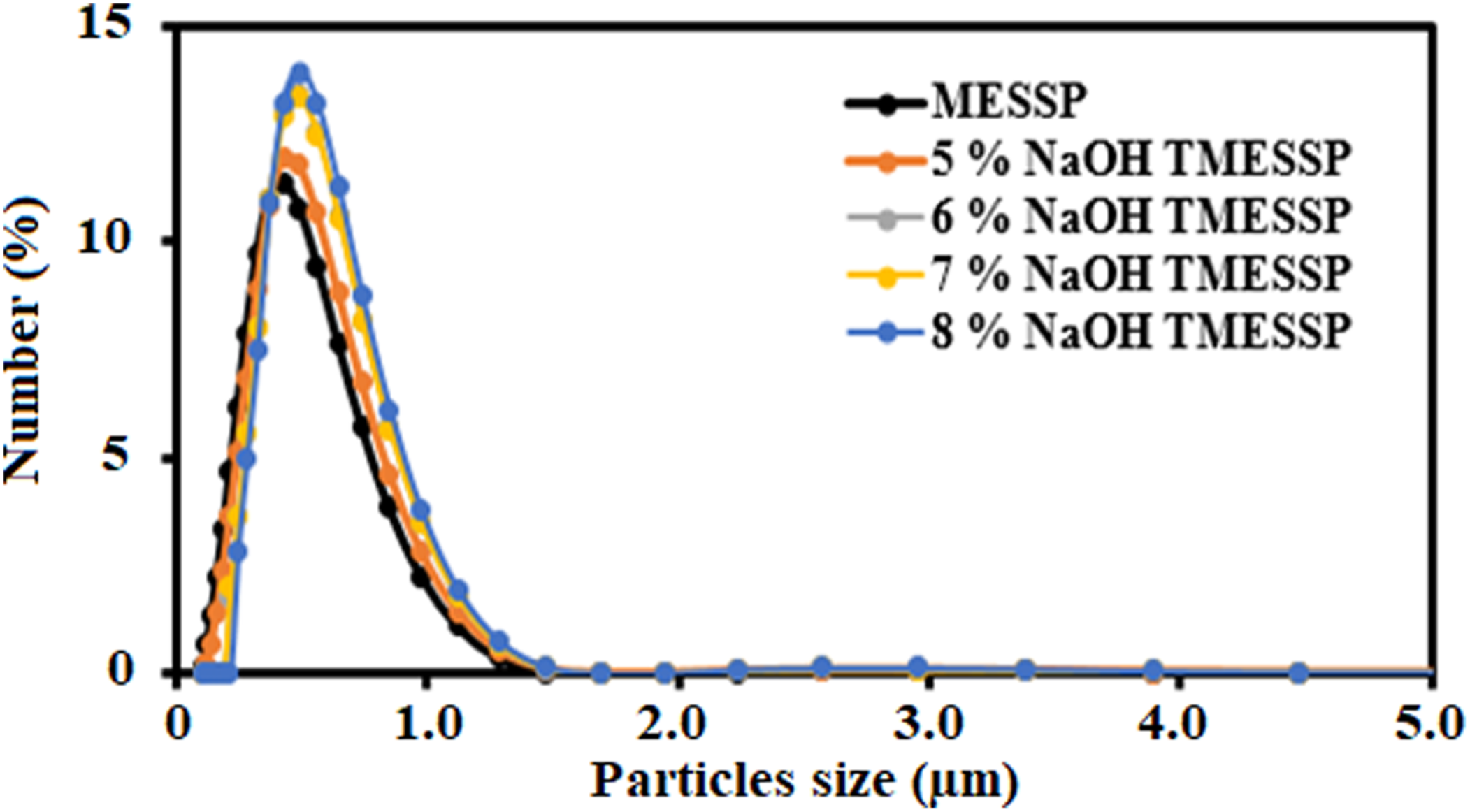
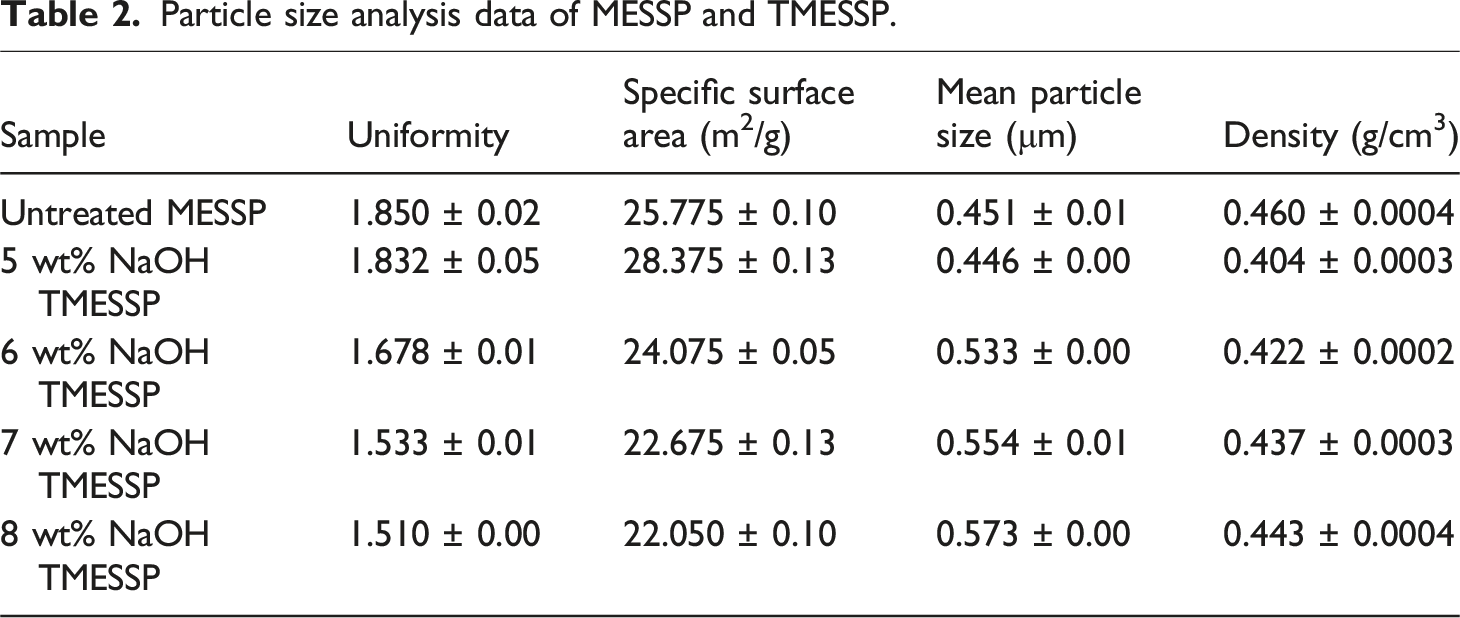
Generally, the removal of cementing elements, such as lignin, hemicellulose, and pectin, and contaminants through effective alkaline treatment reduces the mean particle diameter of natural fibers. 53 Although alkaline treatment can cause natural fibers to expand and increase in particle size; the extent of this effect depends on the type of fiber and the specific treatment condition. 54 In the case of MESSP, alkaline treatment with higher NaOH concentrations (6–8 wt%) resulted in an increase in particle size and a corresponding reduction in specific surface area. This may be due to the swelling of MESSP particles in the aqueous NaOH solution, which leads to particle expansion and a decrease in surface area. Mahmood et al. (2024) reported similar swelling behavior in jute fibers treated with aqueous NaOH, where jute fiber disintegration and particle expansion were observed. 55 Likewise, Hubbe et al. (2024) reported that the expansion of cellulosic fibers during chemical treatment can permanently alter their geometry and size, leading to increased particle dimensions when the fibers fail to shrink after drying. 56 The results observed for TMESSP suggest a comparable phenomenon by which the cellulose in MESSP swells in NaOH and retains its expanded dimension after drying. As a result, TMESSP treated with a 6–8 wt% NaOH exhibited larger particle sizes and smaller specific surface area compared to both untreated MESSP and TMESSP at 5 wt% NaOH. Additionally, the increased particle size and formation of microvoids following NaOH treatment led to a reduction in the bulk density of TMESSP, as shown in Table 2. Overall, alkaline treatment of MESSP caused a decrease in density, with the lowest value recorded for the 5 wt% NaOH TMESSP. A similar observation was reported by, 52 confirming the reduction in bulk density as a consequence of decreased particle size and the formation of microvoids in natural fibers following alkaline treatment. Furthermore, all TMESSP samples showed lower density than untreated MESSP following NaOH treatment.
Scanning electron micrograph of MESSP and TMESSP
Scanning electron microscopy was used to analyze the morphology and surface texture of untreated MESSP and 5 wt% NaOH TMESSP, as shown in Figure 4. Both untreated and treated MESSP exhibit irregular shape and size with a randomly oriented distribution, as seen in Figure 4(a) and (b). However, the untreated MESSP particles tend to form stronger aggregate structures compared to the 5 wt% NaOH TMESSP, which appear more individualized and well dispersed. Micrographs of (a) MESSP and (b) 5 wt% NaOH TMESSP (300x Magnification).
The removal of the hydroxyl (OH) functional groups from the surface of 5 wt% NaOH TMESSP during alkaline treatment reduces its hydrophilicity and weakens the interaction in between the particles. As a result, TMESSP particles show a lower tendency to aggregate. This observation aligns with the larger mean particle size observed for untreated MESSP compared to 5 wt% NaOH TMESSP. Furthermore, the interaction between fiber particles was reduced following alkaline treatment due to the removal of fiber constituents such as hemicellulose, lignin and other elements, resulting in a cleaner particle surface and minimizing aggregation and agglomeration tendencies.52,57
Figure 5(a)–(e) show the SEM analysis of individual untreated MESSP and treated MESSP particles with 5 – 8 wt% NaOH, respectively, at a higher magnification. The SEM analysis confirms that untreated MESSP exhibits a smoother surface texture with no visible microvoids, as shown in Figure 5(a). This observation is attributed to the presence of fatty acids, wax layer, and surface impurities on the fiber.
58
In contrast, the surface of treated MESSP become cleaner and rougher with increasing NaOH conctration from 5 to 8 wt%, as seen in Figure 5(b)–(e). Microvoids area clearly visible on the surface of all TMESSP samples (highlighted by white circle in Figure 5(b)–(e)). The removal of cementing materials, including pectin, lignin, hemicellulose, and other constituents such as wax, impurities, and hydroxyl ions results in increased surface roughness and the formation of microvoids. Similar surface modifications have been reported for other natural fibers after alkaline treatment, showing increased surface roughness and microvoids formation, resulted from the removal of fiber extractives and impurities.
52
The increased surface roughness and microvoids presence in TMESSP improved the exposed surface area of the particles, promoting better matrix adhesion and potential physical interlocking with the polypropylene (PP) matrix. The presence of microvoids significantly contributes to the increased specific surface area, prominently evident at 5 wt% NaOH treatment. Abdullah et al. (2019) reported a comparable observation, noting that the surface roughness of oil-palm empty fruit bunch particles increased after NaOH treatment. Consequently, the rough surface provides anchoring points between the fiber and matrix, enhancing the adherence between the filler and polymer matrix, as well as promoting physical interaction.
59
SEM micrographs of (a) MESSP, and TMESSP with (b) 5 wt% NaOH, (c) 6 wt% NaOH, (d) 7 wt% NaOH and (e) 8 wt% NaOH (2500x magnification).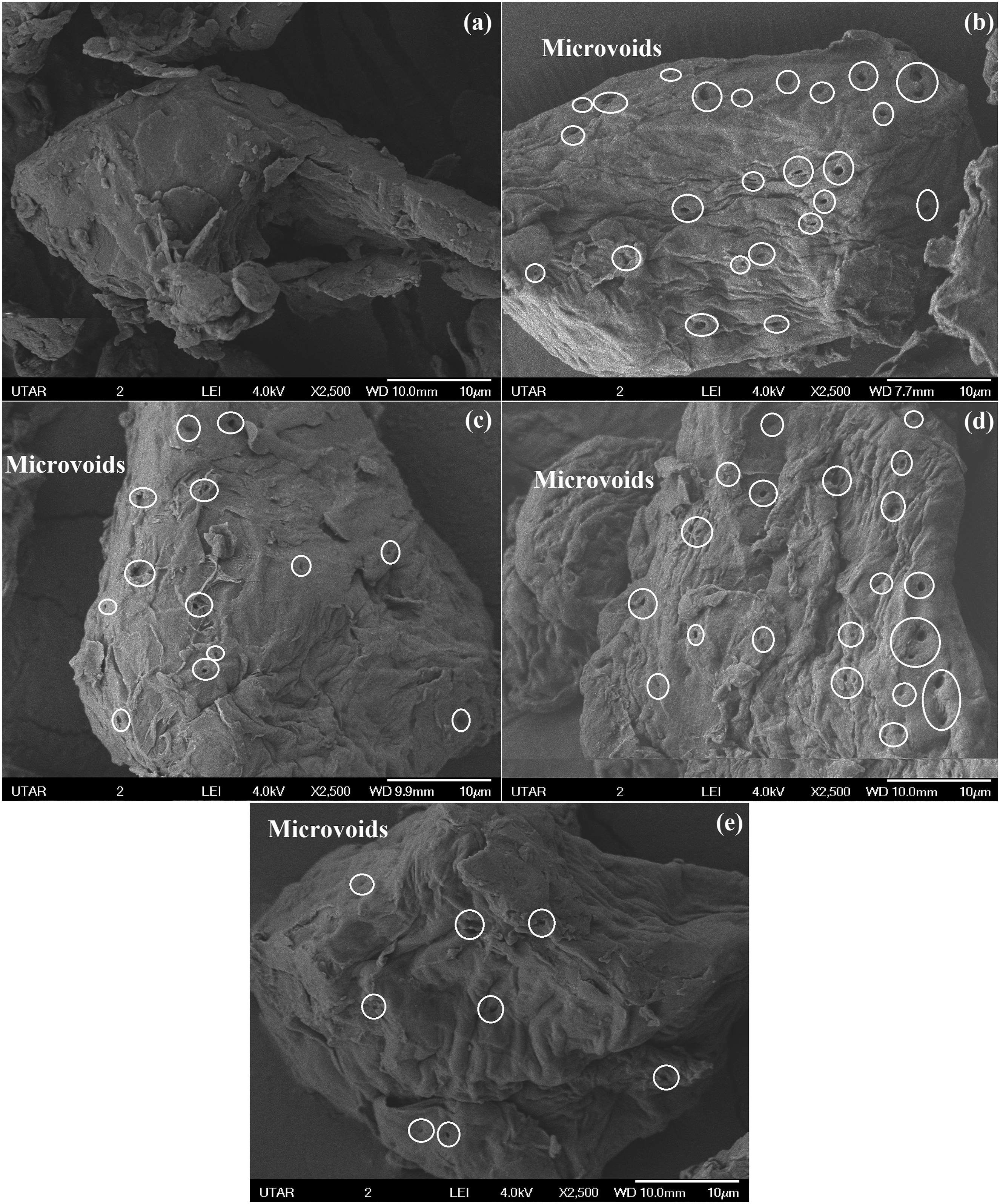
As a summary, although alkaline treatment is generally reported to increase surface roughness and reduce bulk density, the extent of these changes is highly dependent on treatment severity which includes the NaOH concentration, temperature, and duration of exposure. In the present study, mild alkaline treatment conditions were employed, which primarily induced surface chemical modification rather than extensive physical or morphological alteration of the MESSP particles. The FTIR spectra confirm the removal of impurity components and modification of hydroxyl groups, indicating an effective chemical interaction between NaOH and the filler surface. However, the modification only occurs at the molecular level, which may not necessarily translate into detectable changes in bulk particle size distribution, density, or SEM observed surface morphology. This observation is consistent with prior literature by Gao et al. (2025) reported that dilute alkaline treatment significantly altered the surface chemical composition and macrostructural arrangement of cotton fibers, as evidenced by FTIR analysis, while preserving the overall microstructure and particle integrity when observed by SEM. This results in reduced hydrophilicity of cotton fibers without significantly changing its particle size, density, or bulk morphology. 60
In the context of alkaline treatment of MESSP with 5 – 8 wt% NaOH solution, MESSP treated with 5 wt% NaOH was selected for the production of TMESSP-filled PP composites with varying filler contents ranging from 0 to 20 wt%. Based on the characterization of MESSP treated with different NaOH concentrations, 5 wt% NaOH was found to provide the optimal reduction in hygroscopic properties of MESSP compared to treatment with 6 - 8 wt% NaOH. This observation was further confirmed by FTIR analysis, which showed the greatest reduction in the intensity of the hydroxyl group associated with cellulose, indicative if reduced hygroscopic properties of MESSP for TMESSP treated with 5 wt% NaOH. Moreover, particle size analysis demonstrated that 5 wt% NaOH TMESSP exhibited comparable particle size uniformity to that of untreated MESSP, along with the largest specific surface area, the smallest mean particle size and lowest density among all samples, including untreated MESSP and those MESSP treated with 6 – 8 wt% NaOH. These findings indicate that 5 wt% NaOH is the most effective concentration for treating MESSP, resulting to stronger interaction with the PP matrix.
Chemical composition analysis of the MESSP
Figure 6 presents a comparison of fiber components - cellulose, hemicellulose, lignin, and other extractives, in MESSP and 5 wt% NaOH TMESSP. All chemical composition values presented in Figure 6 represent the mean of five independent measurements (n = 5). In MESSP, cellulose, lignin, and hemicellulose accounted for 36.96 wt%, 30.37 wt%, and 7.98 wt%, respectively. In contrast, TMESSP contained only 0.01 wt% extractives and was composed of 54.43 wt% cellulose, 26.63 wt% lignin, and 18.93 wt% hemicellulose. The primary component of all-natural fibers derived from plants is cellulose. However, other elements, such as glucose, hemicellulose, lignin, pectin, fatty acids, wax, and other smaller extractives or impurities, are also present in plant-based natural fibers. These non-cellulosic components often results in weak adhesion between natural fibers and the polymer matrix, leading to poor performance of natural fiber polymer composites (NFPC).30,61 It is noteworthy that 5 wt% NaOH TMESSP become virtually free of extractives and exhibits significantly higher cellulose content. The cellulose content is a crucial factor that influence the mechanical properties of NFPC, attributed by the stiffer properties of cellulose as compared to other fiber constituents.
62
According to Rafiqah et al. (2023), alkaline treatment results in more cellulose exposure on the fiber surface, enhancing interaction through physical interlocking with the polymer matrix and resulting in superior mechanical properties of the composite.
63
Furthermore, alkaline treatment has been proven to increase the resistance of water absorption of natural fibers compared to untreated fibers.
64
In summary, the alkaline treatment substantially reduced the non-cellulosic components, particularly hemicellulose and lignin in MESSP, resulting in an increase cellulose content from 36.96 wt% to 54.43 wt%. Fiber composition comparison of MESSP and TMESSP.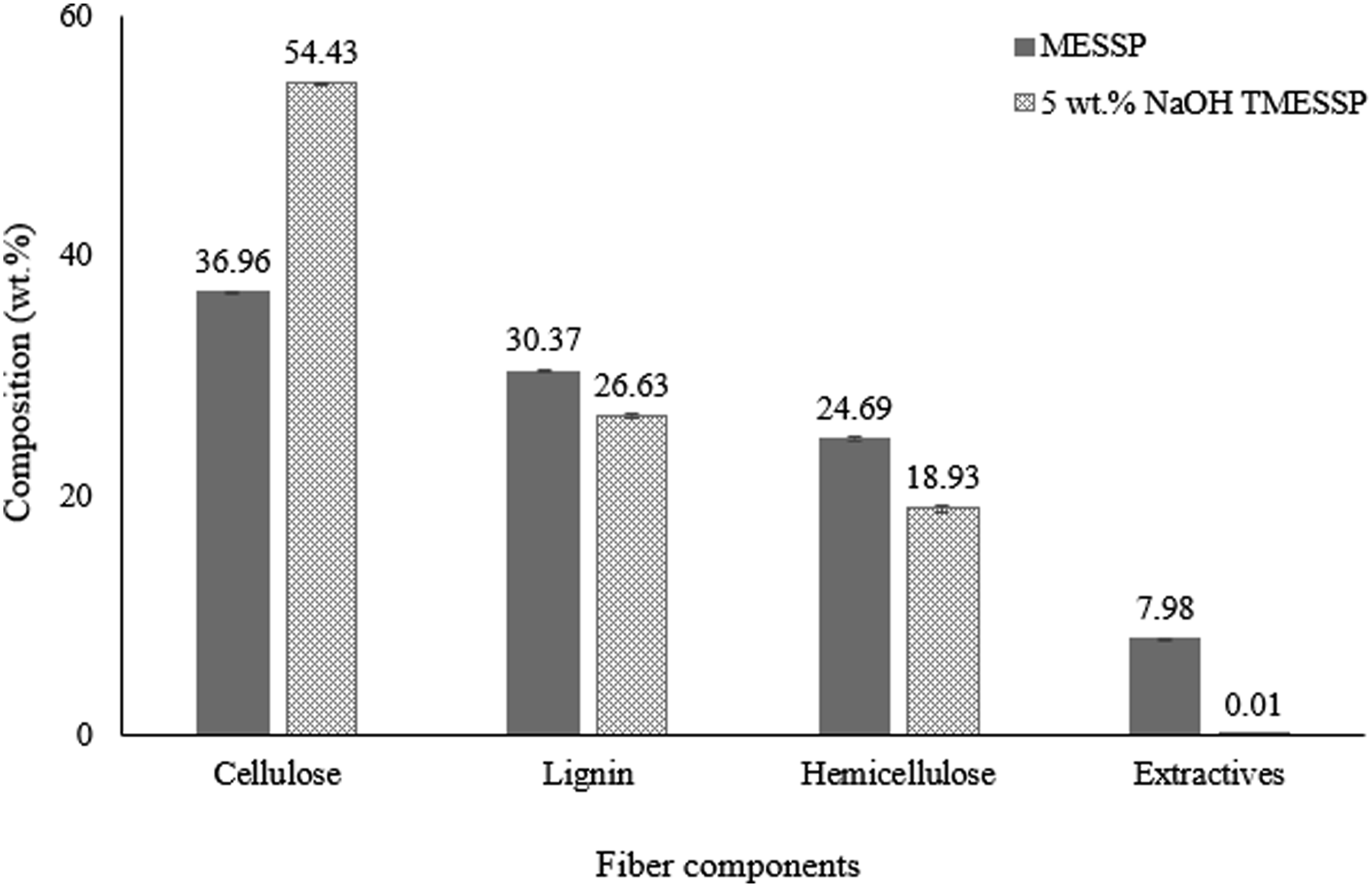
Thermogravimetric analysis
Figure 7(a) and (b) illustrate the thermal decomposition and derivative thermogravimetric (DTG) curves of MESSP and 5 wt% NaOH TMESSP, respectively. As shown in Figure 7(a), both MESSP and 5 wt% NaOH TMESSP exhibit a similar trend in weight loss with increasing temperature. Two stages of thermal decomposition are observed for both particles, occurring in the ranges of 80 – 120°C, and 220 – 480°C, respectively. The initial stage of weight loss is attributed to the removal of absorbed moisture by the fillers, with MESSP showing slightly higher weight loss compared to TMESSP. This difference is due to the removal of hydroxyl (OH) functional groups from the cell walls of MESSP during alkaline treatment, which reduced moisture absorption capacity of TMESSP compared to the untreated MESSP.
64
The second stage of weight loss occurred between 220 and 480°C, which corresponds to the decomposition of fiber elements, such as cellulose, hemicellulose, and lignin. These components decompose over wide range of temperature: cellulose and hemicellulose between 220 and 340°C, whereas lignin between 360 and 480°C.
65
(a) Thermogravimetric (TGA) and (b) derivative thermogravimetric (DTG) curves of untreated MESSP compared with 5 wt% NaOH treated MESSP.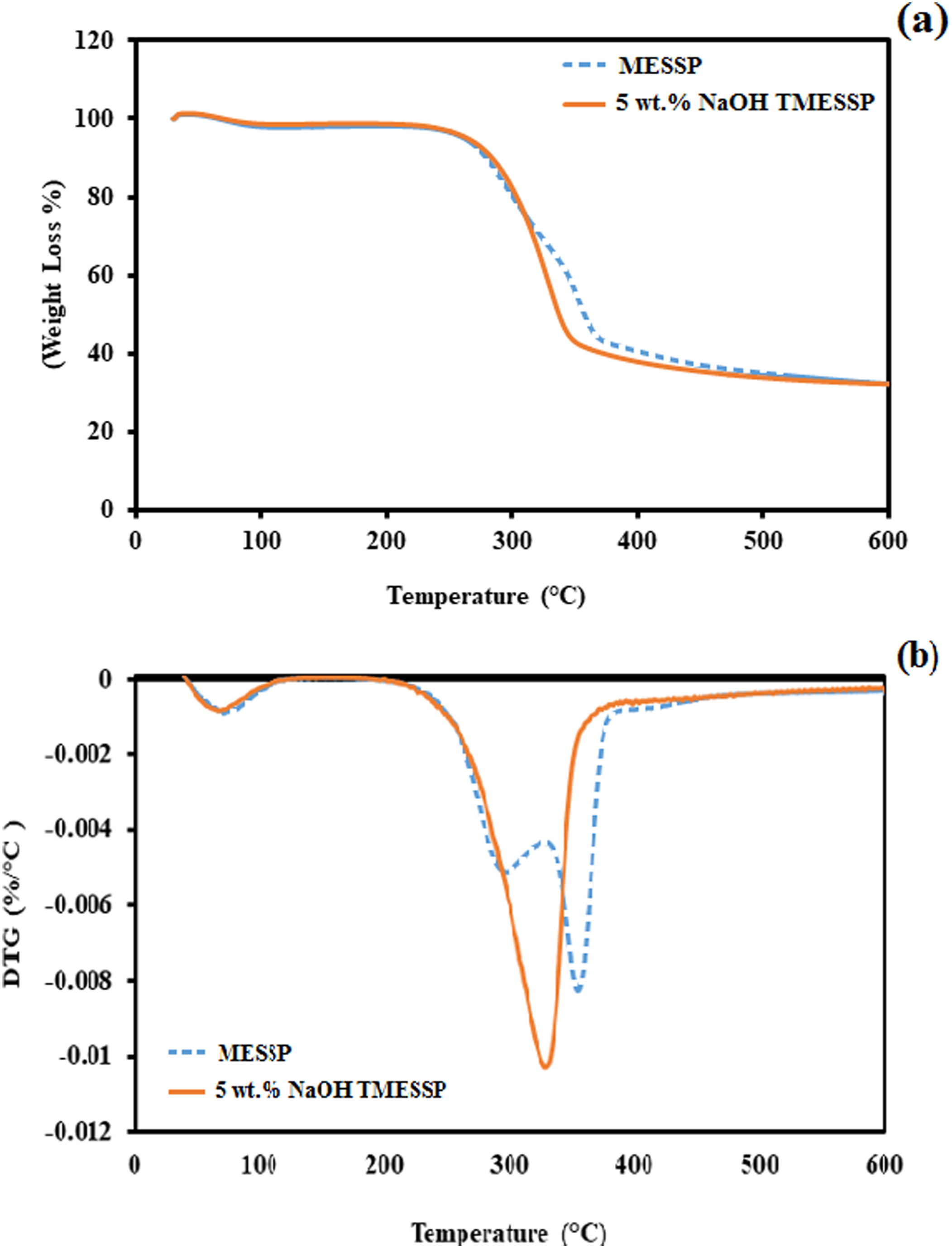
The stages of fiber element decomposition can be seen in Figure 7(b). MESSP exhibits two stages of decomposition, whereas 5 wt% NaOH TMESSP showed a single-stage decomposition between 220 and 480°C. The removal of low-thermally stable lignin and hemicellulose during alkaline treatment results in a shift in the DTG peak for 5 wt% NaOH TMESSP above 300°C, indicating improved thermal stability. The removal of non-cellulosic components of MESSP during alkaline treatment was also confirmed by the disappearance of the second DTG peak, which occurred around 250–320°C for 5 wt% NaOH TMESSP. A study by Kar et al. (2023) reported similar observations, where the occurrence of a single DTG peak between 260 and 320°C, was due to the elimination of non-cellulosic components of Calamus tenuis cane fibers after alkaline treatment. 66 NaOH treated MESSP showed better performance in terms of thermal stability and the results aligns with the FTIR and SEM, indicating reduced hydrophilicity and improved exposed surface area for better interaction with PP matrix. Moreover, the DTG profile confirms that PP composite filled with 5 wt% NaOH TMESSP is able to maintain thermal stability up to 320°C, supporting the composite’s processability and application in low- to medium-temperature environments. However, at temperature above 320°C, the presence of lignin in untreated MESSP contributes to higher thermal stability compared to TMESSP, suggesting that PP filled with untreated MESSP may be more suitable for high-temperature processing and applications. 65
TGA results of neat PP and PP composites at varying filler loadings.
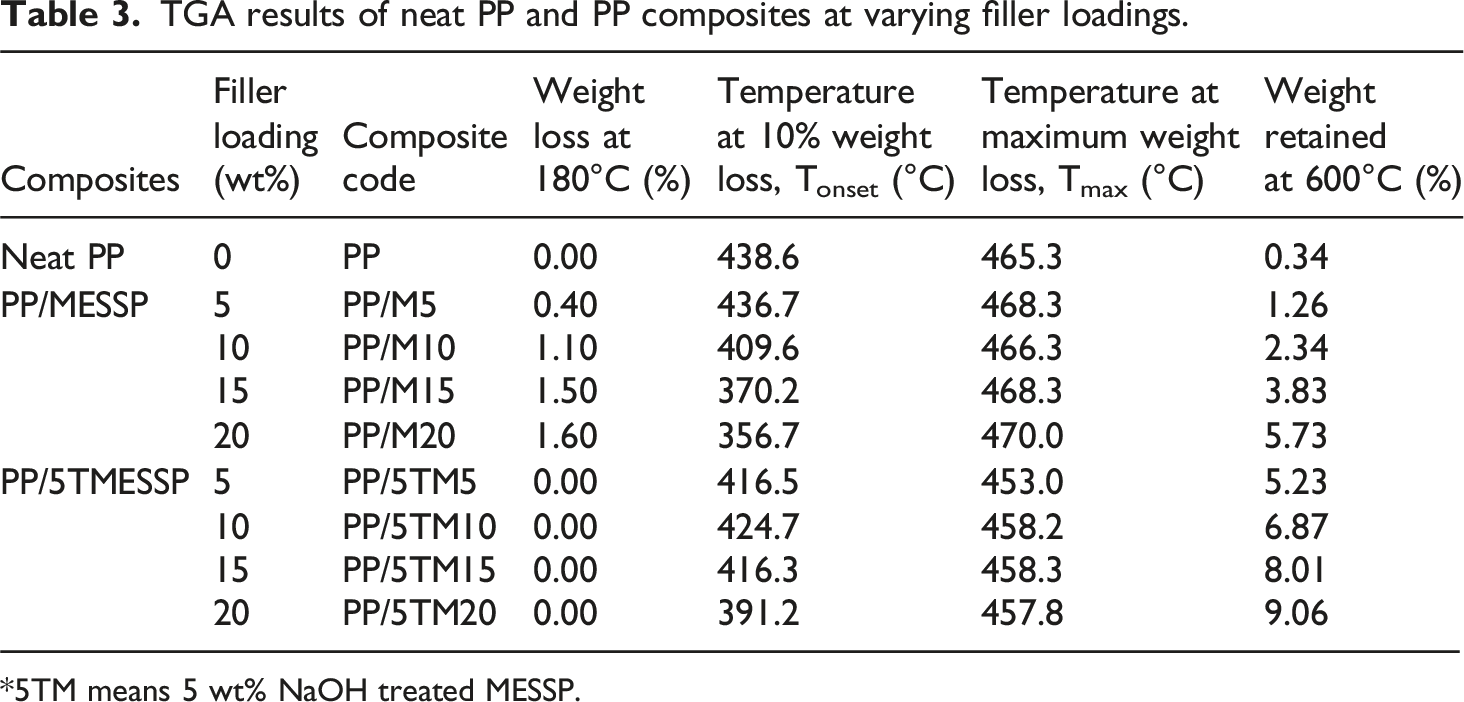
*5TM means 5 wt% NaOH treated MESSP.
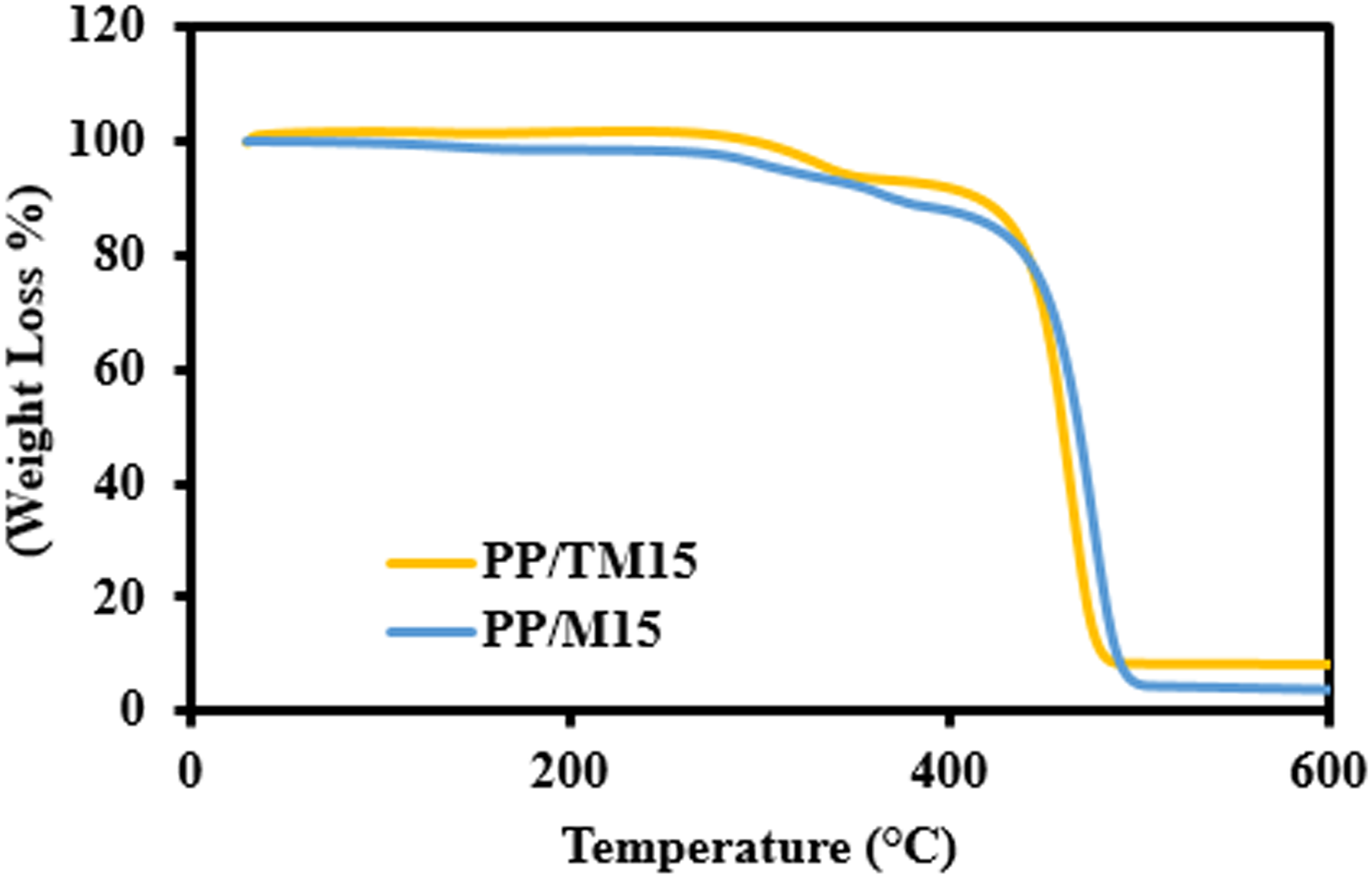
TGA of PP/M15 and PP/TM15 composites.
Table 3 illustrate the temperature recorded at the maximum decomposition rate (Tmax) corresponding to the highest weight loss rate due to thermal decomposition of PP composites containing untreated MESSP and 5 wt% NaOH treated MESSP. The Tmax of neat PP was recorded at 465.33°C, while that of the PP/M composites increased with rising MESSP loading, reaching a maximum of 470.00°C at 20 wt% MESSP loading. The presence of MESSP results in formation of char layer as the protective barrier which delayed the release of volatile substances and supress further decomposition of PP composite at elevated temperature compared to the neat PP. 34 Conversely, the Tmax of PP composites was reduced with the addition of TMESSP, with the Tmax value recorded between 453 and 458°C. The removal of fiber constituents, such as hemicellulose, lignin, and other extractives, during alkaline treatment results in greater weight loss at lower temperatures and a corresponding shift in the Tmax to lower value. 67 As a result, PP/5 TM composites experience more pronounced thermal decomposition at lower temperatures compared to PP/M composites at equivalent filler loadings. Similarly, the reduction of Tmax of chemically treated natural fibers filled polymer composites was reported earlier confirming the claim that the removal of low thermally stable components such as lignin, wax, fatty acids, oil and hemicellulose during alkaline treatment contributes to greater thermal decomposition at lower temperature and a reduced Tmax.65–67
As shown in Table 3, at approximately 600°C, the PP/5TM composites exhibited significantly higher thermal stability compared to both neat PP and PP/M composites at all filler loadings. This is evidenced by the greater residue remaining after thermal decomposition for the PP composites filled with 5 wt% NaOH TMESSP. The residue content for the PP/5TM composites increased with higher filler loading, with the maximum residue recorded at 20 wt% 5TMESSP. Specifically, the residue amount increased nearly 26-fold compared to neat PP (from 0.34% to 9.06%) and 1.58 times higher than that of the PP/MESSP with 20 wt% MESSP (from 5.73% to 9.06%). Surface treatment of MESSP with 5 wt% NaOH substantially improved the thermal decomposition behavior of PP matrix. This can be evident from the increased residue content (percentage weight retained at 600°C) for PP/TMESSP compared to neat PP and PP/M composites at comparable loading, reflecting improved thermal stability of PP/TMESSP composites. The removal of lignin during alkaline treatment and the formation of a condensed polycyclic aromatic char structure from cellulose through chemical rearrangements during decomposition is responsible for the thermal stability enhancement. These structural decomposition at elevated temperature and imparts greater thermal resistance to the polymer composite. 68
Tensile properties and fracture surface analysis
Comparison of tensile properties of PP composites containing untreated MESSP (PP/M) and 5 wt% NaOH-treated MESSP (PP/5TM), at varying filler loadings.
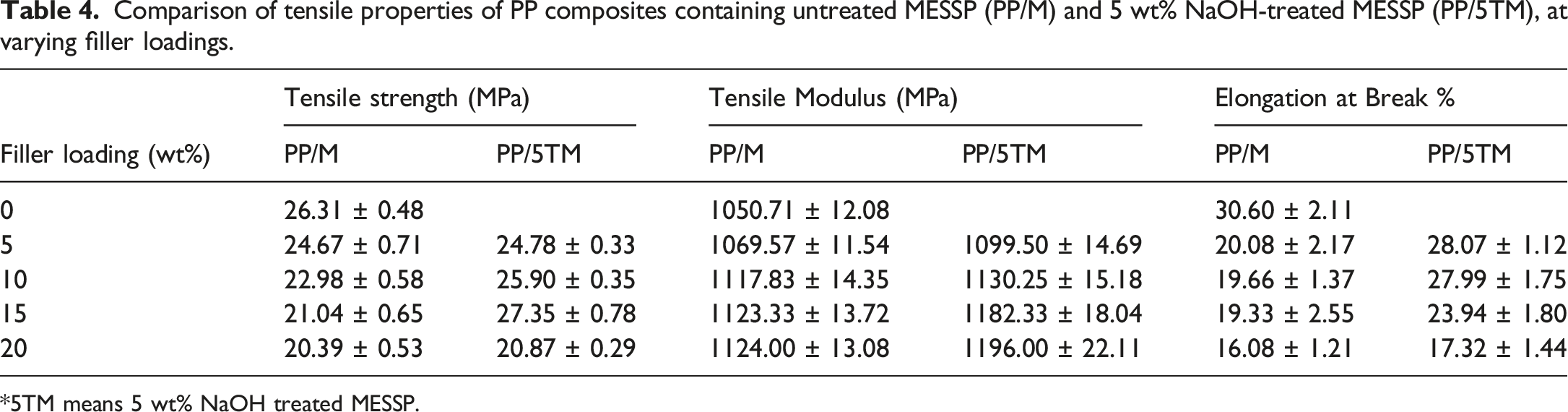
*5TM means 5 wt% NaOH treated MESSP.

SEM morphology of tensile fractured surface of PP composites containing (a) 5 wt% MESSP, (b) 5 wt% TMESSP, (c) 10 wt% MESSP, (d) 10 wt% TMESSP, (e) 15 wt% MESSP and (f) 15 wt% TMESSP, at 2500x magnification.
During alkaline treatment, hydrophilic components of natural fibers, such as lignin, pectin, wax, and hemicellulose are removed, which indirectly reduces the intensity of structural OH functional groups responsible for the hydrophilicity of the fiber. 6 FTIR analysis, chemical composition analysis, and SEM observations confirmed the optimal reduction of OH groups, improved cellulose content, and presence of microvoids, respectively, following treatment of MESSP with 5 wt% NaOH. As shown in Table 4, the tensile strength initially decreases with the addition of 5 wt% TMESSP. This reduction is attributed by the low MESSP content, which is not sufficient to facilitate stress transfer between PP matrix and MESSP. 31 However, when the TMESSP loading is increased to 15 wt%, the tensile strength improves, whereas a further increase to 20 wt% loading results in a decline. The reduction in tensile strength at higher TMESSP loading is attributed to the tendency of the particles to aggregate, leading to poor filler dispersion and the formation of stress concentration points within the PP matrix. Moreover, it was reported in previous study that 20 wt% represents the saturation point for MESSP addition into the PP matrix, beyond which particle agglomeration and poor wettability occur, leading to negative impact on the mechanical performance of PP composite. 34
The surface morphology observed through SEM confirms that the adhesion between TMESSP and PP has improved, as depicted in Figure 9(b), (d) and (f). The 5 wt% NaOH-treated MESSP particles can be seen strongly embedded in the PP matrix on the fractured surfaces, even with increasing TMESSP loading. In additional, numerous string-like structures are visible on the surface of TMESSP, indicating matrix tearing due to stronger interfacial adhesion. The formation of microvoids and the reduction in particle size with 5 wt% NaOH treatment improve the specific surface area of TMESSP, thereby improving the tensile strength of PP. Furthermore, tensile property analysis suggests efficient stress transfer at the fiber and matrix interface, facilitated by strong chain interlocking between PP and TMESSP particles, as shown in Figure 9(d). 69
The tensile strength improvement was most pronounced for PP composite containing 15 wt% TMESSP, with an increase of 1.3-fold compared to the PP composites with 15 wt% untreated MESSP, and 1.04-fold increase relative to neat PP. The NaOH treatment of MESSP eliminated the cementing elements that had covered the filler surface, thereby exposing more surface area for polymer contact. The etching effect produced a rougher surface and increased the exposed surface area of TMESSP. This observation is evidenced by the SEM micrographs in Figure 5, which show enhanced physical interaction between TMESSP and the PP matrix. 4 Comparing the PP composites at similar filler loading, improved adhesion between the TMESSP and PP produced composites with better tensile strength as compared to the PP composites containing untreated MESSP. Furthermore, the extraction of hemicellulose, lignin and wax increased the exposure of cellulose on the fiber surface, enabling stronger interaction with the PP chains. 1 This observation aligns with the increased cellulose content of MESSP after treatment with 5 wt% NaOH, as discussed in section 3.4. Moreover, the enhanced compatibility and adhesion in the PP composite containing 15 wt% TMESSP, compared to other TMESSP loadings, were further confirmed through SEM analysis, as shown in Figure 9(d). The PP matrix was observed to adhere firmly to the TMESSP surface. The presence of string-like structure (matrix tearing) on the filler surface indicates stronger adhesion and physical interaction, which contributed to the highest improvement in tensile strength.
Comparing both PP/M and PP/5TM composites, both showed similar trend of reducing elongation at break and increasing tensile modulus with increasing filler loading. With the addition of TMESSP, the composite system become more rigid, as indicated by higher tensile modulus of PP/5TM compared to PP/M composites. As mentioned earlier, stronger adhesion between PP and TMESSP, relative to untreated MESSP, facilitate efficient stress transfer and reduced plastic deformation of the composite. 70 Overall, alkaline treatment of MESSP with 5 wt% NaOH produced PP composite with enhanced tensile properties compared to neat PP and PP/M composites, particularly at the optimum filler loading of 15 wt%.
Impact properties
The impact strength comparison between PP containing untreated MESSP and 5 wt% NaOH-treated MESSP at varying filler loading is illustrated in Figure 10. From the past studies, it was reported that the addition of natural fillers or fiber into polymer matrix generally reduced the impact strength of the associated composites, attributed to their poor interfacial adhesion and incompatibility between the natural fiber and polymer matrix.
71
In contrast, incorporating MESSP into PP led to gradual improvement in impact strength as the filler loading increased from 5 to 20 wt%. This improvement can be related to the higher energy required to detach the MESSP particles from the PP matrix, due to partial compatibility between MESSP and PP. Similarly, the impact strength of PP/TMESSP composites increased with TMESSP loading from 5 to 20 wt%. Moreover, at comparable filler loadings, PP/TMESSP composites exhibited higher impact strength than PP/MESSP composites. Comparison of impact strength of PP composites containing untreated MESSP and 5 wt% NaOH-treated MESSP, at varying filler loadings.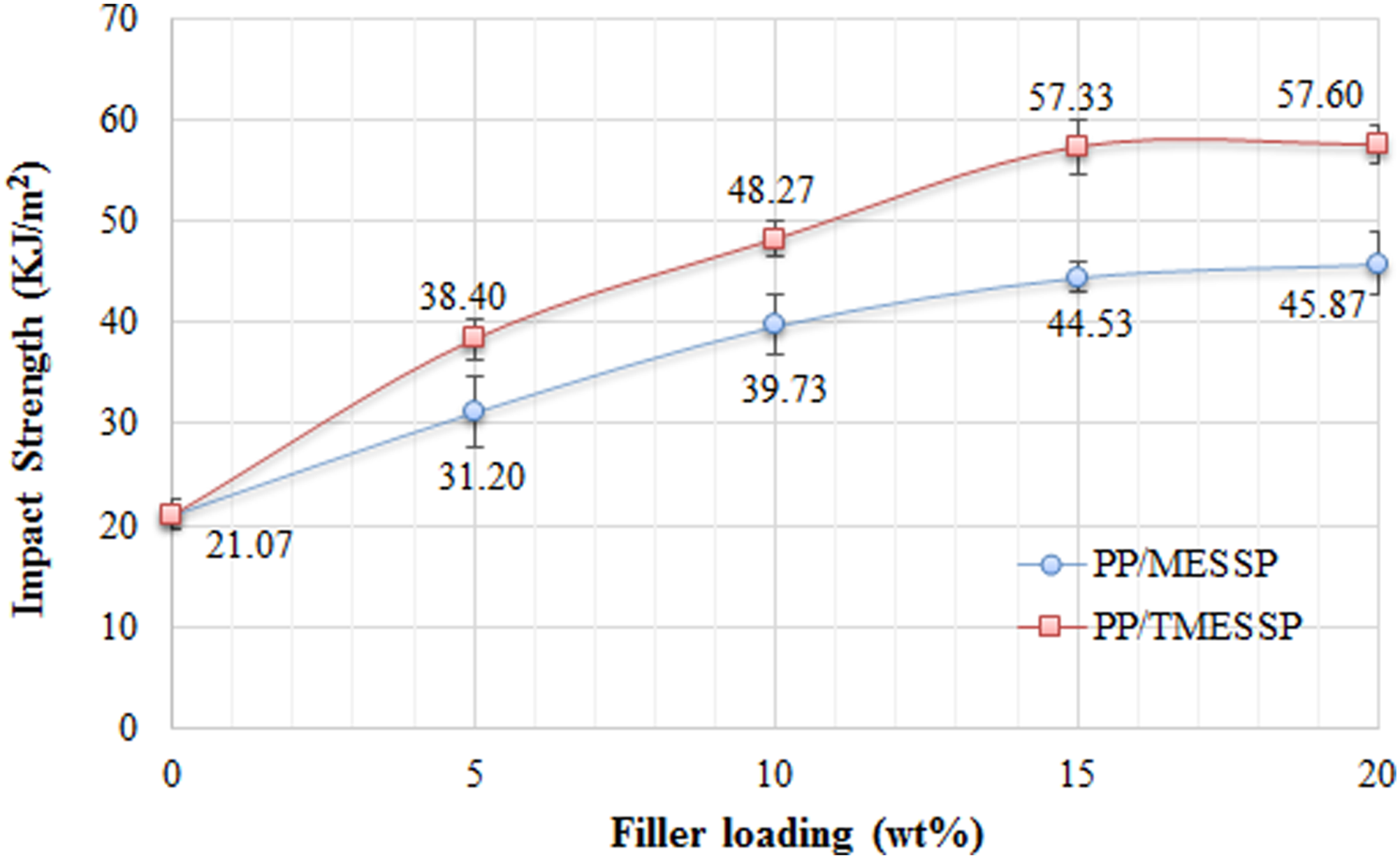
The impact strength of PP/TMESSP composites increased gradually with the increasing filler loading, reaching a maximum of 12.8 KJ/m2 at 15 wt%. The increase in impact strength can be related to the removal of amorphous fiber components such as lignin and hemicellulose during the alkaline treatment, thereby increasing the surface roughness and strengthening the interfacial adhesion between TMESSP and PP matrix. This results in higher energy absorption capacity and improved stress transfer during impact. Furthermore, the reduction in particle size and increased specific surface area of TMESSP promote stronger filler-matrix adhesion, producing superior energy absorption compared with both neat PP and PP/MESSP composites.
The findings align with past studies reporting on the improved impact strength of alkaline-treated natural fiber filled polymer composites, attributed to enhanced interfacial adhesion and higher energy absorption capacity. Study by Gupta et al. (2024) reported that better impact and toughness properties in polymer composite systems can be achieved through effective energy transfer between the filler and matrix at the interface. 72 Similarly, Norshafika et al. (2022) observed that alkaline-treated seaweed fibers reinforced PP composites exhibited higher impact properties compared with untreated fiber, due to stronger interfacial adhesion. 73 Unlike the past studies, however, the present finding highlights the combined effect of alkaline treatment of MESSP and particle size reduction in maximizing the impact performance of PP composites, suggesting a synergistic reinforcement mechanism.
Water absorption
The comparison between the water absorption percentage of neat PP and PP composites containing untreated MESSP and 5 wt% NaOH-treated MESSP (TMESSP) at varying filler loading is shown in Figure 11. Neat PP (0 wt% filler loading) showed the lowest water absorption percentage, which increased gradually with filler loading, for both MESSP and TMESSP. However, at comparable filler loadings, PP/TMESSP composites exhibited higher water absorption resistance than PP/MESSP composites. The removal of cellulosic components from natural fibers during alkaline treatment reduces the intensity of OH groups, which are responsible for forming hydrogen bonds with water molecules and thus contribute to water absorption and entrapment between polymer chains. As the natural fiber content increases, the hydrophilicity of the composite system also increases, allowing greater water absorption.
74
Comparison of water absorption percentage of PP composites containing untreated MESSP and 5 wt% NaOH-treated MESSP, at varying filler loadings.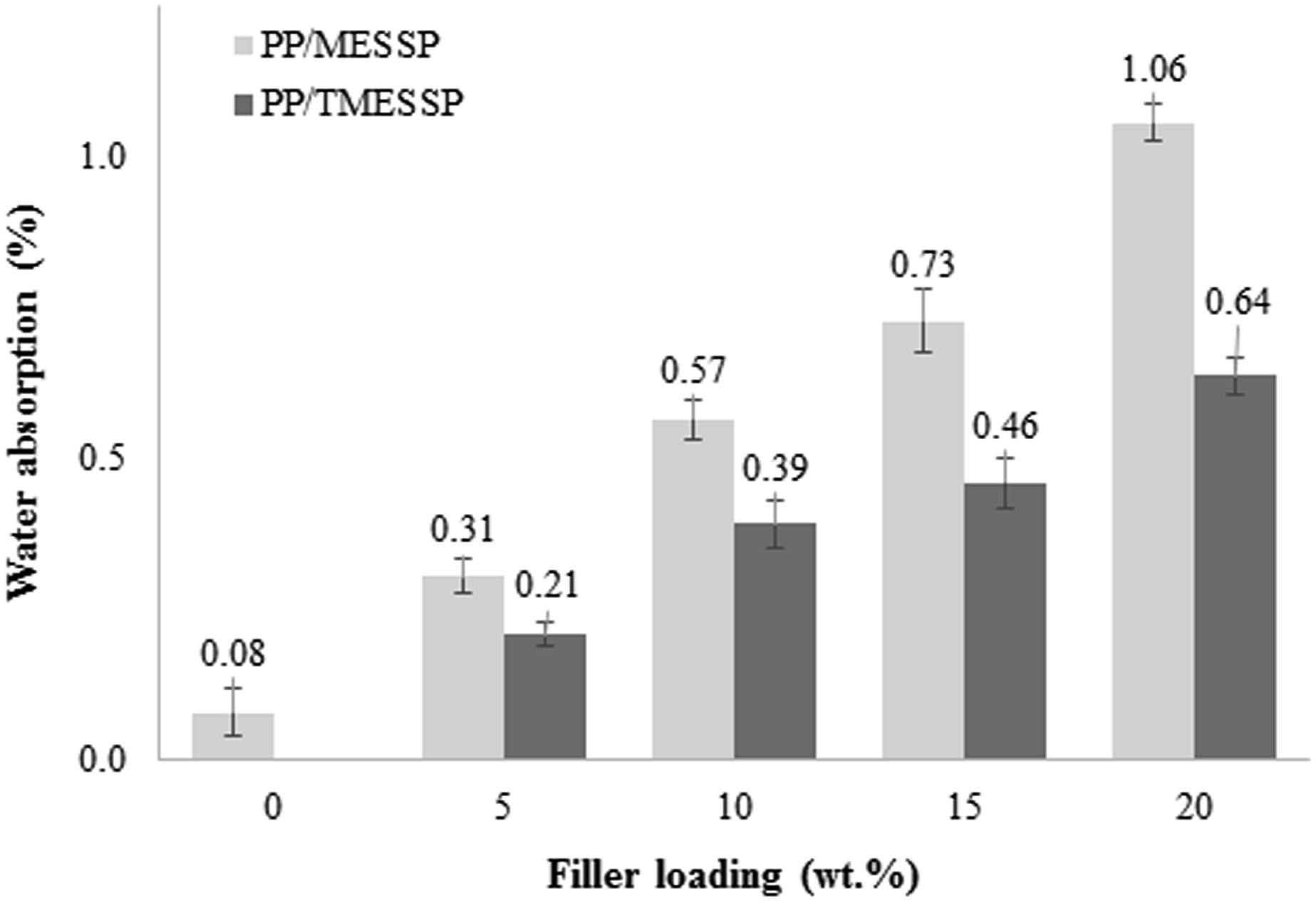
In the presence of TMESSP in PP, the water absorption resistance of PP was enhanced relatively compared to MESSP in PP. This improvement can be attributed to the removal of lignin and hemicellulose during alkaline treatment, reducing the abundance of hydroxyl groups in MESSP. The decrease in hydrophilic characteristics of MESSP, enhances the compatibility with PP matrix, thereby strengthening the interfacial adhesion and maximize the resistance for water penetration into the composite. Thus, the likelihood of water absorption and entrapment within the PP matrix is reduced. This explanation aligns with the FTIR and chemical composition analysis, as well as the observed improvement in tensile properties and impact strength of PP/TMESSP composites, all of which collectively confirms the enhanced adhesion between TMESSP and PP. However, the water absorption reported in this study corresponds to the short-term water uptake, which could reflect only the initial water diffusion to the composite system and does not represent the equilibrium water absorption saturation point. Thus, longer exposure period would provide a better insight of the equilibrium water absorption properties and a long-term durability, which will be addressed in future studies. 75
Conclusions
Alkaline treatment effectively reduced the hydrophilicity and increased the surface roughness of MESSP by removing fiber constituents such as hemicellulose, lignin, and extractives, thereby enhancing its suitability as filler for PP matrix. Among the studied pretreatment conditions, 5 wt% NaOH yielded the most favorable filler characteristics, leading to improved interfacial adhesion between MESSP and PP matrix. Consequently, improving the tensile, impact and thermal properties, as well as water absorption resistance of PP composites, particularly at TMESSP loadings of 5 – 15 wt%. The improved compatibility and interfacial adhesion were further corroborated by SEM morphological analysis, which were consistent with the observed improvements in mechanical properties. The results indicate great potential of treated natural fillers such as MESSP to improve the performance of PP composites, in line with the circular economy objectives and development of sustainable material. However, this study is limited to narrow NaOH concentration range and the absence of long-term durability, aging, and environmental impact assessment. Future work should therefore focus on optimizing the treatment conditions, including the addition of compatibilizer, as well as evaluating the processability, aging behavior, and life cycle or environmental impact study to better establish the potential application of TMESSP filled PP composites.
Footnotes
Acknowledgements
The authors would like to thank Universiti Tunku Abdul Rahman for providing adequate facilities to complete this research work.
Author contributions
All authors are equally contributed to Conceptualization, Methodology, Writing - original draft, Writing - review & editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the authors used the Grammarly tool for language editing. Following its use, the authors carefully reviewed and manually edited the content as necessary, taking full responsibility for the integrity and accuracy of the final manuscript.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
