Abstract
Poly (methyl methacrylate) block copolymer is interesting due to its good compatibility with many polymers. In this study, a novel phase-change energy storage material, n-octadecane/poly (styrene-methyl methacrylate) (OD/P(St-MMA)) microcapsules, were designed and synthesized by emulsion polymerization, in which n-OD was used as core materials, the copolymer of St and MMA as shell materials, and sodium dodecyl benzene sulfonate (SDBS) as emulsifier. The morphology, phase-change thermal properties and thermal stability of microcapsules were investigated by particle size analyzer, transmission electron microscope, thermogravimetric (TG) analyzer, and differential scanning calorimetry (DSC). The results showed that when the ratio of the two monomers, St and MMA, was 1/5 and the dosage of SDBS was 1.5 g (3% of the total mass), the particle size dispersion of microcapsules was uniform and microcapsule particles were well wrapped. TG analysis showed that the long-term use temperature of microcapsules could not exceed 131°C. DSC showed that the phase-change enthalpy of microcapsule was 148.39 J·g−1, which indicated the microcapsules had excellent energy storage property.
Introduction
At present, with the continuous improvement of science and technology, human’s activity space is expanding constantly. Cold polar, hot desert, the huge temperature difference of space, and the unknown deep-sea are the primary field of our challenge. 1,2 During these explorations, high and low temperature environment is the main difficulties and challenges. 3 In daily life, we need a more stable environment in our houses, clothes, cars, and so on. Long-term outdoor staff such as the soldiers stationed at the border, geological prospectors, workers on steel production lines, city’s traffic coordinator, and security guards, they all need to maintain their own temperature balance. 4 In addition, cold weather is easy to frail elderly infected with influenza and make recurrent arthritis in patients under suffering, and the hot weather also lets people feel dysphoria and unpleasant; thus, a comfortable and ideal environment for living is highly desirable. 5,6 Therefore, experts around the world have started paying attention to the development of thermal insulation and storage materials, 7 which are emerging endlessly, 8 –10 ranging from the simplest foam insulation 11 to the advanced electric heating 12 and from solar energy storage 13 to other energy storage materials 14 ; however, all of them have not been used widely due to their thermal and mechanical instability and high demand for their working conditions. 15 Therefore, to meet the needs of production and living, more strict requirements on thermal insulation and storage materials are put forward. 16 –18
Phase-change materials accumulate energy by substances absorbing or releasing heat when occurs phase change. 19 But it is easy to cause leakage, phase separation, volume expansion, and poor heat stability when used alone. 20 Coating a layer of membrane with excellent stable performance on the surface of phase-change material can effectively solve the problems. 21,22 As the phase-change material is wrapped in the microcapsules, it is possible to prevent the phase-change material from leaking out of the matrix due to the liquidation at high temperatures and result in polluting the environment. Consequently, it is very significant to get a phase-change microcapsule material with more uniform distribution of particle size, more tenacious capsule wall, and more latent heat. 23
In this study, n-octadecane/poly (styrene-methyl methacrylate) (OD/P(St-MMA)) microcapsules were designed and synthesized by emulsion polymerization, 24 –27 in which n-OD was used as core material, the copolymer of St and MMA as shell materials of microcapsule. These energy-storage microcapsules can be used repeatedly through phase change. 28 The influence of shell materials and the dosage of emulsifier on the morphology and thermal properties of microcapsules were discussed.
Experimental
Materials
n-OD (analytical-reagent grade (AR), colorless liquid, relative density 0.777, melting point 28.18°C, and boiling point 316.1°C) was kindly supplied by Tianjin Kermel Chemical Reagent Co., LTD (Tianjin, China). St (AR, with 5% NaOH solution to remove the polymerization inhibitor) was purchased from Tianjin Da Mao Chemical Reagent Factory (Tianjin, China). MMA (AR) was purchased from Tianjin Bodi Chemical Co., LTD (Tianjin, China). Butyl acrylate (BA; AR) was supplied by Tianjin Kermel Chemical Reagent Co., LTD (China). Potassium persulfate (AR) was purchased from Tianjin Tian Da Purification Material Factory (Tianjin, China). Sodium dodecyl benzene sulfonate (SDBS, chemical pure, HLB=10.6) was supplied by Tianjin Kermel Chemical Reagent Co., LTD (China). The other reagents used were obtained in the market.
Synthesis of OD/P(St-MMA) microcapsules
The OD/P(St-MMA) microcapsules were synthesized using OD as the core, P(St-MMA) as the shell, deionized water (50% of the total mass) as medium, potassium persulfate (1% of the total mass) as initiator, and SDBS (3% of the total mass) as surfactant. The specific steps are that the required amount of St, MMA, OD, and potassium persulfate was charged into a well-dried 250-mL, three-necked flask equipped with a condenser, a stirrer, and a gas inlet, the surfactant and deionized water were added subsequently. The flask was immersed into a water bath at 20°C and made the reagents scatter and emulsify for 10 min in a stirring speed of 1500 r min−1 under a nitrogen atmosphere, and then kept for 5 h in a stirring speed of 300 r min−1 at 75°C, the samples were filtered and separated repeatedly using distilled water, until the residues of reactants had been removed. Finally, the white superfine powder particles, OD/P(St-MMA) microcapsules, were obtained after being dried at 80°C. The synthesis process of OD/P(St-MMA) microcapsules is shown in Figure 1.
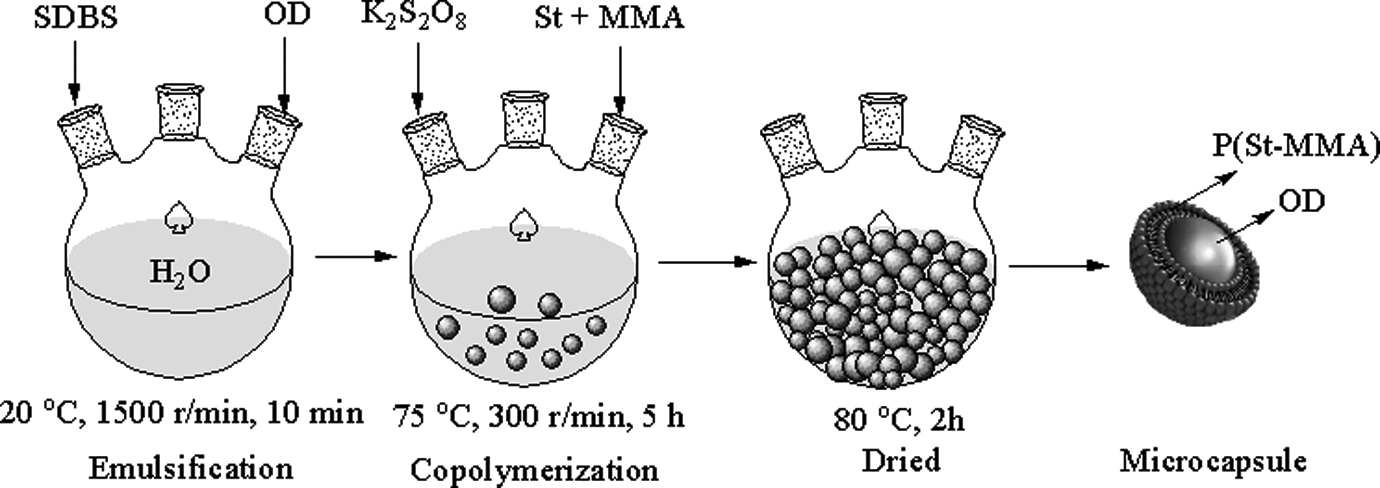
Preparation process of n-octadecane/poly (styrene-methyl methacrylate) (OD/P(St-MMA)) microcapsules.
Characterization
The samples for transmission electron microscope (TEM, Hitachi H7650, Tokyo, Japan, acceleration voltage 100 kV) were prepared by reacted with 85% hydrazine in a closed vessel at 45°C for 2 h to selectively convert the ester groups of the acrylic modifiers to hydrazide. Then, the samples were placed in a vacuum oven at 50°C for 12 h to remove moisture and then exposed to OsO4 vapor to stain the hydrazide groups for 10 min. 29 Finally, the samples were diluted and dipped onto carbon-coated copper grids and then observed the morphology with TEM after dried.
Particle size distribution was measured using a laser particle size distribution tester (OMEC LS800, Xiangzhou, Zhuhai, Guangdong, China) and added 1–2 drops of diluted suspension of microcapsule into sample bottle before testing.
The samples were made by grinding 1∼2 mg microcapsules and 200 mg KBr together (both be dried), put in the mold, pressed into slice with a hydraulic press under 5–10 MPa. The Fourier transform infrared (FTIR) spectra of samples were collected after 256 scans at 4 cm−1 resolution in the region of 4000 to 400 cm−1 with a Spectrum one FTIR spectrometer (Perkin Elmer Co., Massachusetts, Waltham, USA).
The samples were tested with thermogravimetric (TG) analyzer (NETZSCH STA449F3 Jupiter, Freistaat Bayern, Germany) in the temperature range 20– 600°C using a heat rate of 5°C min−1 under nitrogen.
Differential scanning calorimetry (DSC) thermal analyses were performed with DSC tester (Malvern 204F1, Malvern, UK), nitrogen purging, and gas flow is set as 50 mL min−1, the temperature ranged from −50°C to 100°C for the samples at a heat rate of 5°C min−1.
Results and discussion
Effect of various shell materials on microcapsules morphology
The influence of different components as shell materials on microcapsules morphology is shown in Figure 2.

Transmission electron microscope images of microcapsules with St/butyl acrylate (a) and St/MMA (b) as shell materials.
TEM analysis showed that the microcapsules synthesized by monomer St and MMA were dispersed evenly and hardly adhered together, while the dispersion of those synthesized by St and BA was not good and the particles were agglomerated. In Figure 2(a), it can be observed that the black outer walls of the microcapsules are adhered together, so the particles cannot be dispersed and result in uneven distribution of the particle size of the microcapsules. In Figure 2(b), this phenomenon has been significantly improved, the microcapsules are individually dispersed, the outer walls are homogeneously wrapped, and the particle size is similar and no big difference. From this, we can see that the monomer MMA is more suitable for shell materials of the microcapsules than BA.
Effect of shell component on particle size of microcapsules
The heat storage capacity of phase-change microcapsules depends on the phase-change latent heat of the core material. The more core material is encapsulated in the microcapsule preparation process, the stronger heat storage capacity of the microcapsules have. Thus, the choice of appropriate particle size is very important for the formation of microcapsules, and an important factor affecting the coverage rate of microcapsules is the feed ratio of core and shell materials. The effect of shell component on particle size of microcapsules is shown in Figure 3.
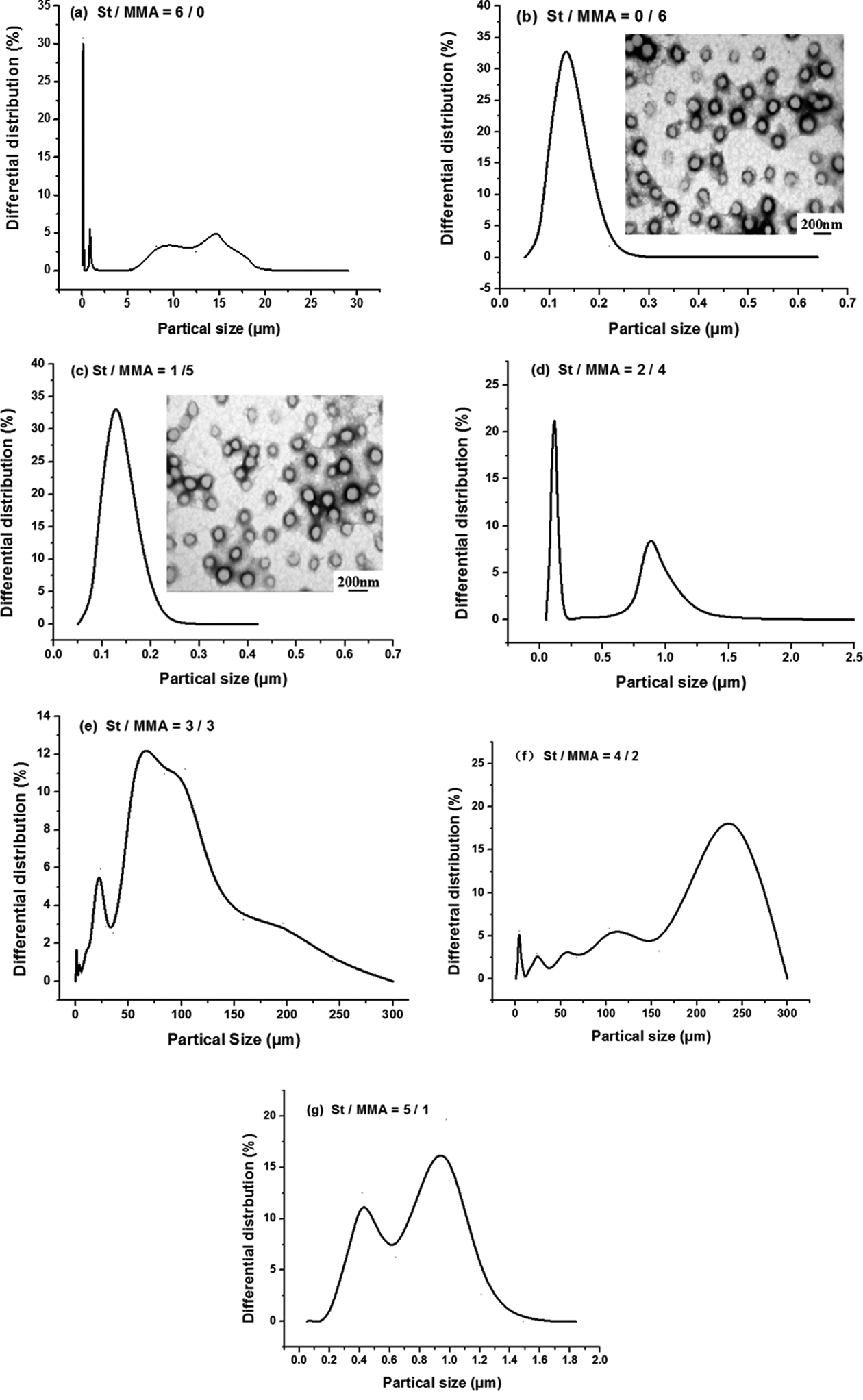
Effect of shell component on particle size of microcapsules.
The particle size distribution of Figure 3 showed that multi-peak obviously appeared in all curves when the mass ratio of St/MMA was 6/0, 2/4, 3/3, 4/2, and 5/1, it was found that the size of microcapsules was very uneven and distributed widely. Interestingly, when the ratio of St/MMA was 0/6 and 1/5, the curves present single-peak and all particle size were about 0.11μm, which indicates that the coating ratio of shell layer is the best. In addition, it can be seen from TEM figures that when the ratio of St/MMA is 0/6, the local aggregation of the black area is more obvious, which indicates that the microcapsules are more concentrated on adhesion or overlap. However, the microcapsules with a St/MMA ratio of 1/5 were not found to overlap and had little adhesion to each other. In Figure 3(b), for the sample with St/MMA ratio of 0/6, the size of the microspheres is not very uniform and shell coating is not uniform, meanwhile, more microcapsule morphology is not spherical or ellipsoid. For St/MMA ratio in 1/5 of the sample in Figure 3(c), particle size is basically uniform and shell coating is evenly, the vast majority of microcapsule morphology is spherical. In conclusion, the microcapsule dispersion effect, shell coating, and microcapsule morphology of St/MMA ratio of 1/5 are the best. This is probably due to the reaction rate of polymerization reaction is too fast when only use MMA as shell material, resulting in the phenomenon of polymer agglomeration, so that the product cannot disperse and get together. On the contrary, the addition of St slowed the polymerization rate of MMA, so that there was enough time for the shape adjustment and the dispersion of the particles during the reaction.
Effect of emulsifier dosage on microcapsules morphology
In this work, SDBS was used as emulsifier. Dispersing the emulsifier in water to form micelles, the lipophilic side of the emulsifier enters into the phase-change material, and the hydrophilic end is dispersed in the continuous phase water so as to realize the encapsulation of the oil phase droplets and to disperse and protect the core material. The initiator used in this experiment is potassium persulfate, water-soluble initiator, which can make the shell materials in the water to participate in the reaction and then deposited in the core material to form the shell materials. The influence of the amount of SDBS on the microcapsules morphology is shown in Figure 4.
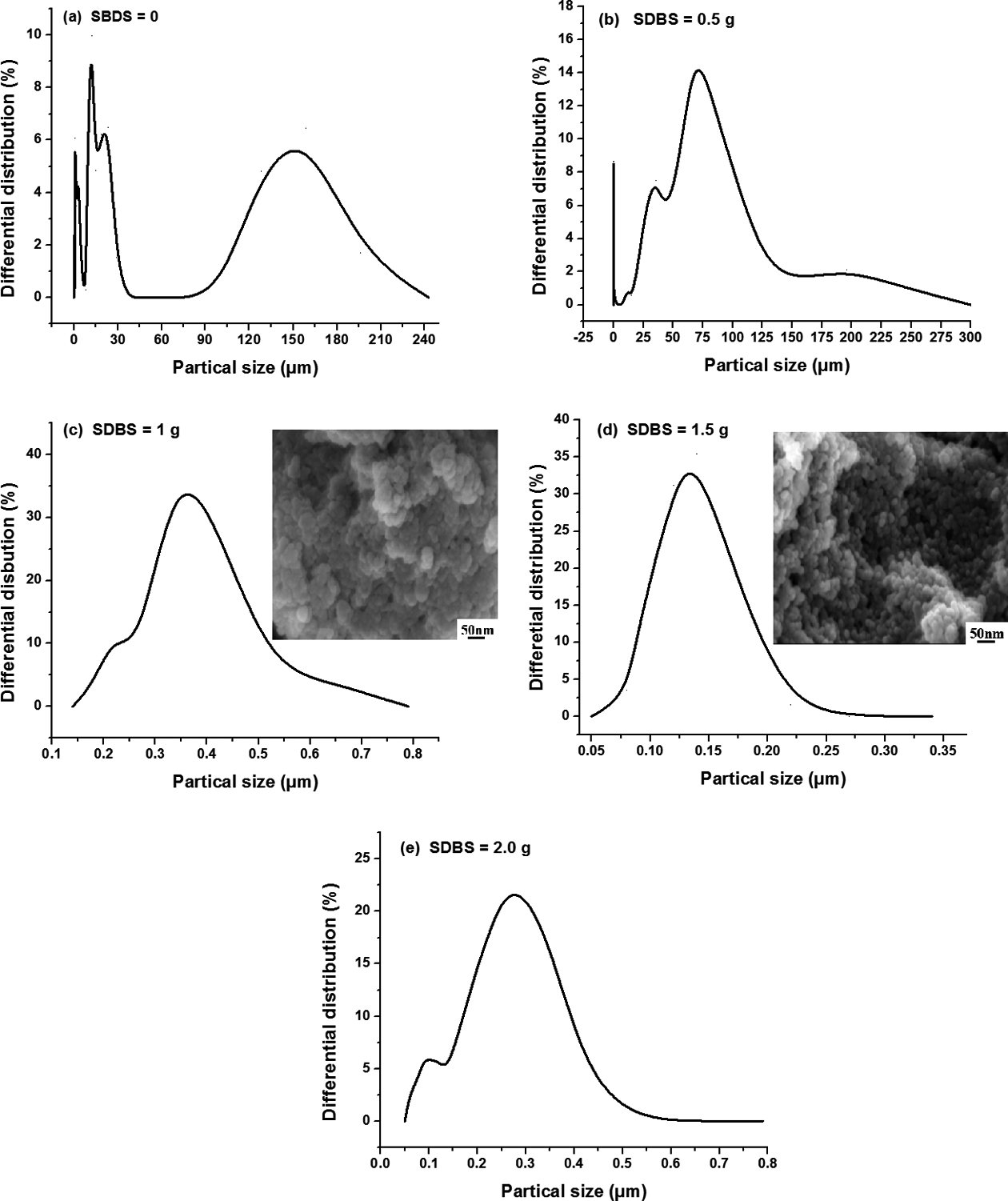
Particle size distribution maps of core-shell particles with different content of sodium dodecyl benzene sulfonate (SDBS).
It can be seen from the particle size distribution diagram in Figure 4 that when the content of emulsifier is little, the microcapsules particle size analysis figure shows a great number of peaks, indicating that the dispersion effect of the OD is not good mainly because the emulsifier of the content is too small to wrap OD completely and resulting in a dispersed particle size in a state of confusion. With the increase of emulsifier content, the particle size tends to be uniform. However, when the amount of emulsifier is increased by a certain amount, the particle size will gradually decrease. As the emulsifier increases, the micelles formed by the emulsifier in the water also increase, and the monomer with stirring spread to the micelle so that the number of monomer droplets increases; due to the total amount of monomer is constant, the average particle size of the emulsion will gradually decrease with the increase of monomer droplet number. Therefore, when the amount of emulsifier is 2 g, the particle size is too small. For the samples prepared using 1.5 g SDBS, particle size analysis showed a single peak and the average particle size of microcapsules is 0.12 μm, meeting the needs of the product. In addition, found from scanning electron microscope (SEM), when the emulsifier is 1 g, the agglomeration of microcapsules is serious and the original spherical shape has been changed. While the emulsifier is 1.5 g, it can be observed that the overall still retains the spherical structure with little change to its original shape. Therefore, the choice of the amount of emulsifier used in this experiment is 1.5 g.
The TG analysis was performed in order to study the effect of the dosage of emulsifier on the thermal stability of phase-change energy-storage microcapsules.
It can be observed in Figure 5 that the weight loss of the sample at 1.0 g and 1.5 g of SDBS consisted of two stages, the first stage was 131–316°C and the second stage was 316–476°C. Compared to the first stage, the thermal degradation rate of second stage was faster. For the microcapsules added with 1.0 g and 1.5 g SDBS, both showed a decrease in mass at 131°C due to the volatilization of OD, but the rate of decrease of the former was high, indicating that the volatilization rate of OD was very fast and also proving that its package effect is not good. The boiling point of OD is 316°C, the curves appear rapidly declining after 316°C, and it can be attributed to the thermal decomposition of shell materials. The sample was completely decomposed at 476°C. In summary, microcapsules have the best thermal properties when SDBS content is 1.5 g.
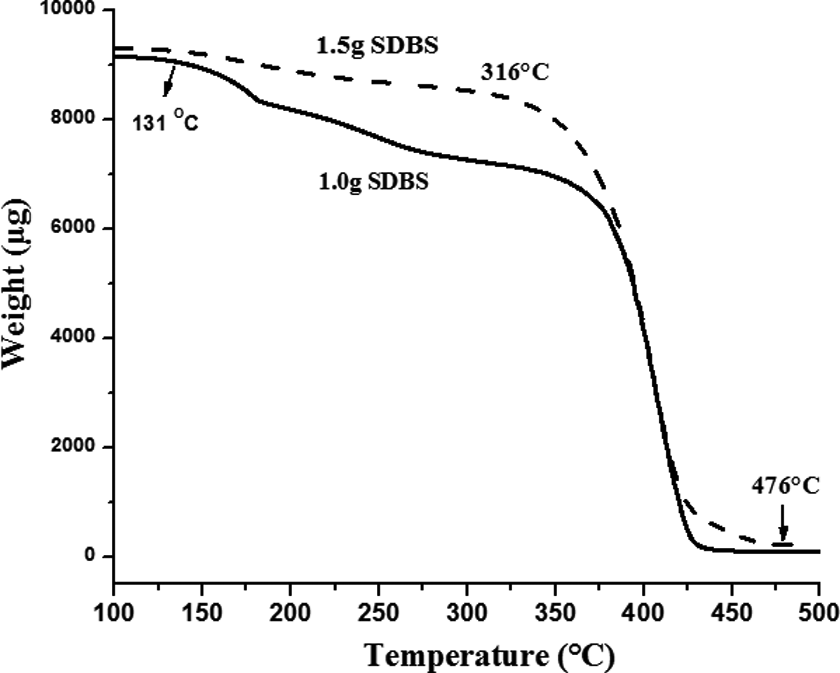
Thermogravimetric curves of microcapsules with 1.0 g and 1.5 g SDBS. SDBS: sodium dodecyl benzene sulfonate.
Infrared (IR) analysis
Compared with curve
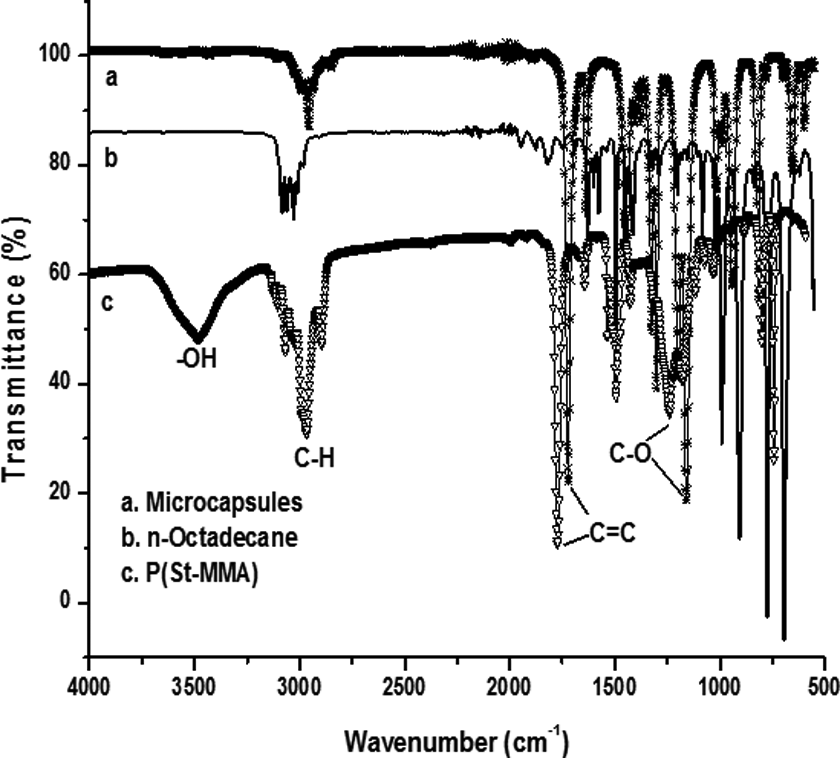
Infrared spectra of shell, core, and microcapsules.
DSC analysis of microcapsules
Figure 7 shows the differential thermal analysis of the microcapsules and OD. The exothermic peak onset temperature of OD/P(St-MMA) phase-change energy-storage microcapsules was 34.28°C, the termination temperature was 23.9°C, and the peak temperature was 29.48°C with the gradual decrease of temperature. The phase-change enthalpy of microcapsules calculated by the endothermic peak area was 148.39 J·g−1. The melting temperature of OD is 28.18°C, while it can be seen from DSC curves that the melting temperature of the microcapsules is retarded due to the phase-change materials. It illustrates that microcapsules possess good energy storage effect.
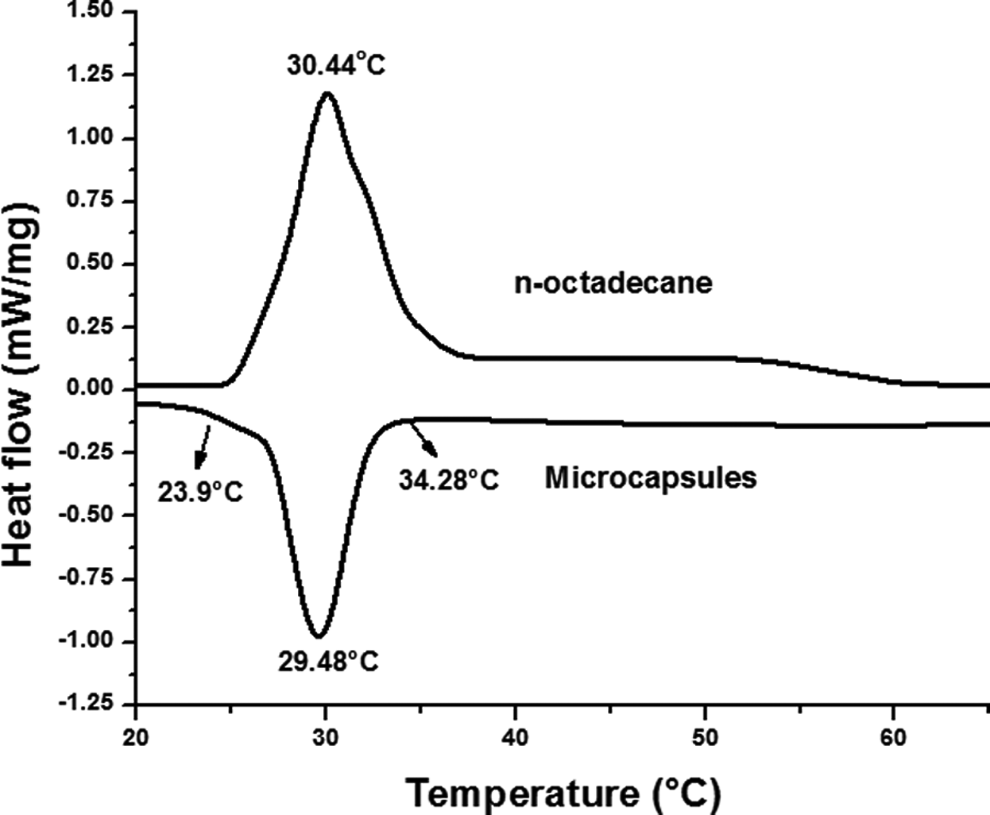
Differential scanning calorimetry curve of microcapsules.
Conclusions
In this work, a novel phase-change energy-storage microcapsules were prepared by emulsion polymerization, in which OD was used as core material and copolymer of St and MMA was used as shell materials. When the ratio of St to MMA was 1/5, the coating of shell formed was the best. When the dosage of SDBS was 1.5 g, the size of microcapsules was uniform, the dispersibility of particles and the encapsulation of shell materials were good. DSC represents that the enthalpy of phase change of microcapsules is 148.39 J.g−1, indicating that the microcapsules have excellent energy storage property. TG analysis shows that the long-term use temperature of microcapsules cannot exceed 131°C; and also, IR analysis shows that the microcapsules contain core materials and shell materials, and the shell materials consist of the copolymer of St and MMA.
Footnotes
Acknowledgement
This work was supported by 2014 graduate student innovation research of Qiqihar University, China (YJSCX2014-001X).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
