Abstract
The automotive industry increasingly demands lightweight, strong materials capable of large-scale production. Therefore, the objective of this work is to develop polyamide 6 (PA6) composites reinforced with graphene nanoplatelets (GNPs) that can be processed by injection molding. This study evaluates the effect of incorporation the GNPs reinforcement on the performance of PA6. The one-way ANOVA statistical analysis was used to evaluate the mechanical properties as a function of the composition of the composites obtained. A co-rotating twin-screw extruder was used to prepare the set of PA6/GNPs composites, followed by the injection molding process. Conventional characterization techniques were used to determine the properties of the composites, such as thermogravimetrical analysis (TGA), differential scanning calorimetry (DSC), Fourier-transform infrared spectroscopy (FTIR), Field Emission Gun-Scanning Electron Microscopy (FEG-SEM), X-ray diffraction (XRD), tensile, flexural and impact strength tests. The GNPs were characterized using Raman spectroscopy, the Brunauer-Emmett-Teller (BET) method, field emission gun-scanning electron microscopy (FEG-SEM), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR) and thermogravimetrical analysis (TGA). The results suggest that the graphene used has a sparse number of stacked layers and defects, classifying it as a better-quality material. The composites showed the best tensile and flexural properties. The addition of 5.25 wt% GNPs to PA6 resulted in a 22% improvement in Young´s modulus and a 10% improvement in tensile strength compared to neat PA6. Regarding flexural properties, the incorporation of 7 wt% GNPs into PA6 generated a 34% increase in flexural modulus of elasticity and a 17% increase in flexural strength. On the other hand, GNPs reinforced PA6 composites exhibited brittle material behavior, identified by low notch impact strength compared to neat PA6.
Keywords
Introduction
In recent years, the demand for advanced materials with enhanced mechanical, electrical, and thermal properties has significantly increased, driven by the rapid evolution of industries such as aerospace and automotive, healthcare, and consumer electronics sectors. 1 The search for efficient materials, associated with high performance and low cost, has promoted the increasing use of thermoplastic composites.2–4
The addition of nanoscale fillers to enhance the properties of polymers has led to the development of nanocomposites. The discovery of nanoscale particles such as fullerene, nanoclay, carbon nanotube (CNT) and graphene (GN) has opened up the potential of a variety of new nanocomposite materials with specific properties. Graphene, is currently considered a highly potential filler for improving the mechanical, thermal, and electrical properties of polymers. Its nanometrie size, which results in a hugr surface area to volume ratio, and its unique properties have fueled intense research in materials science and engineering.5–16
The incorporation of GN into polymeric matrices has promoted the achievement of superior properties, such as increased tensile strength, thermal and electrical conductivity. 17 These properties associated with high aspect ratio and nanoscale dimension open new perspectives for the development of high-performance multifunctional materials for various industrial sectors, including automotive, construction, aerospace and packaging.18,19
Graphene is an allotrope of carbon (sp2 hybridization) with a two-dimensional structure and is defined as a single layer of graphite. Pure GN, however, is not yet mass-produced, and consists of a few layers of graphene stacked together.20,21 There is a growing interest in the use of GNPs as a reinforcement element in polymeric nanocomposites. 22 Graphene has a high aspect ratio and surface area. When incorporated into polymeric matrices, GN generates materials with excellent optical, electrical, thermal and mechanical properties superior to those of conventional materials.23–26 For these reasons, the standardization of GN and reliable quality control for industrially produced graphene materials, has now become one of the most critical prerequisites for the growing GN industry, where the fight against “fake graphene” in the market is one of the top priorities. 27
There is a significant demand to develop and implement reliable, simple, and low-cost analytical methods that provide information of bulk properties of industrially produced GN materials. Several analytical methods such as Raman spectroscopy, surface area (Brunauer-Emmett-Teller (BET) method), Field Emission Gun-Scanning Electron Microscopy (FEG-SEM), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), thermogravimetric analysis (TGA), particle size distribution (PSD) and pH titration can meet these requirements. These methods can provide valuable information about the nature of GN powders such as the definition of their crystalline and graphitic structure, surface area, chemical composition, impurities and functional groups.28,29
Polyamides, particularly PA6 or nylon 6, are widely used due to their mechanical strength, elasticity, softness, lightness, and surface reflection. The incorporation of low levels of GNPs into PA6 promotes a significant increase in mechanical, thermal and electrical properties. In addition, GNPs significantly increase the thermal stability of polyamides and affect the crystallization behavior of the polymer, acting as nucleating agents. 30
A study evaluating the incorporation of distinct grades of GNPs into polyamides showed that smaller platelets yield superior mechanical and thermal properties. Higher tensile strength values were obtained with a GNPs grade with a larger surface area. This result was attributed to the retention of the original morphology during processing and the greater degree of dispersion of the particles in the polymer. To obtain superior electrical properties, the use of GNPS with larger particle size and surface area was recommended.18,19,30
In another investigation, it was observed that nanoparticles promote an increase in interfacial shear tension between the matrix and the filler on a micrometric scale, promoting the achievement of superior physical and mechanical properties. In this study, it was shown that the incorporation of graphene oxide (GO) into PA6 presented an increase of 88% in tensile strength and 66.5% in the Young’s modulus of the composites in relation to the values of these mechanical properties presented by pure PA6 materials. 31 Another study reported that the addition of GO to PA6 improved its thermal properties, due to an increase in interfacial adhesion. 32 In turn, Tang et al. 33 found that composites containing highly dispersed GO had a higher glass transition temperature (Tg) and strength compared to those with less dispersed GO.
Recent studies demonstrate significant improvements in properties such as elongation at break and impact resistance by introducing GNPs and GO into polymer matrices.34,35 Other studies have investigated the thermal and mechanical behavior resulting from the addition of GO and reduced graphene oxide (rGO) to PA6 and PA6.6.2,36–38 Results found in the literature indicate that the incorporation of 0.1–1 wt% of rGO into PA6 promotes a reduction in plastic deformation, an increase in the modulus of elasticity and tensile strength, in addition to an increase in the crystallization temperature, crystallinity, and thermal stability.2,36–39
In general, the properties of PA6 composites reinforced with GNPs depend on the physical characteristics of the GNPs. Systematic study of the effect of several grades of GNPs on the performance of the materials obtained, showed that the particle size, as well as the surface area of the GNPs affect the mechanical behavior of the composites produced. 19 The increase in the surface area of the nanofiller promotes a more efficient transfer of stresses to the matrix, providing the formation of a more effective barrier to crack propagation and increasing the strength of the material.
There are many published works on the incorporation of CNTs into polyamides.5–8 However, there is no extensive literature on the incorporation of GNPs into these polymer matrices, as this nanomaterial with unique properties began to be incorporated into technological innovations on a commercial scale approximately 15 years ago.
It is worth noting that depending on the characteristics of GNPs, such as surface area, number of layers and defects, the properties of the materials obtained will be different. Most published works fail to address the main characteristics of the GNPs used, making it difficult to establish correlations between structure and properties. Therefore, the characterization of the different grades of GNPs and the determination of the properties of the composites obtained are crucial for the advancement of research in this area. There is a scarcity of articles describing the characteristics of the GNPs used in their work. Many authors use GN, graphite, GO, and rGO in their work and refer to all of these materials as GN. Furthermore, because the processing conditions used in most of these studies are not similar, the properties of the resulting materials are not the same. We intend to test some fully characterized grades of GNPs in order to correlate their properties with those of the resulting materials. In this context, this work presents the results obtained in the development of PA6 nanocomposites with GNPs with a surface area equal to 750 m2g−1. This work also presents the results of the characterization methods used in the quality control analysis of graphene powders, such as Raman spectroscopy, Brunauer-Emmett-Teller (BET) method, Field Emission Gun-Scanning Electron Microscopy (FEG-SEM), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR) and thermogravimetrical analysis (TGA) applied to a specific grade of graphene and a discussion on the effects of GNPs incorporation and content on the mechanical and thermal properties of PA6, aiming at its use in applications that require high performance. The effect of incorporating different contents of GNPs on the properties of PA6 was evaluated using one-way ANOVA statistical analysis.
Materials and methods
Materials
Properties of PA6.
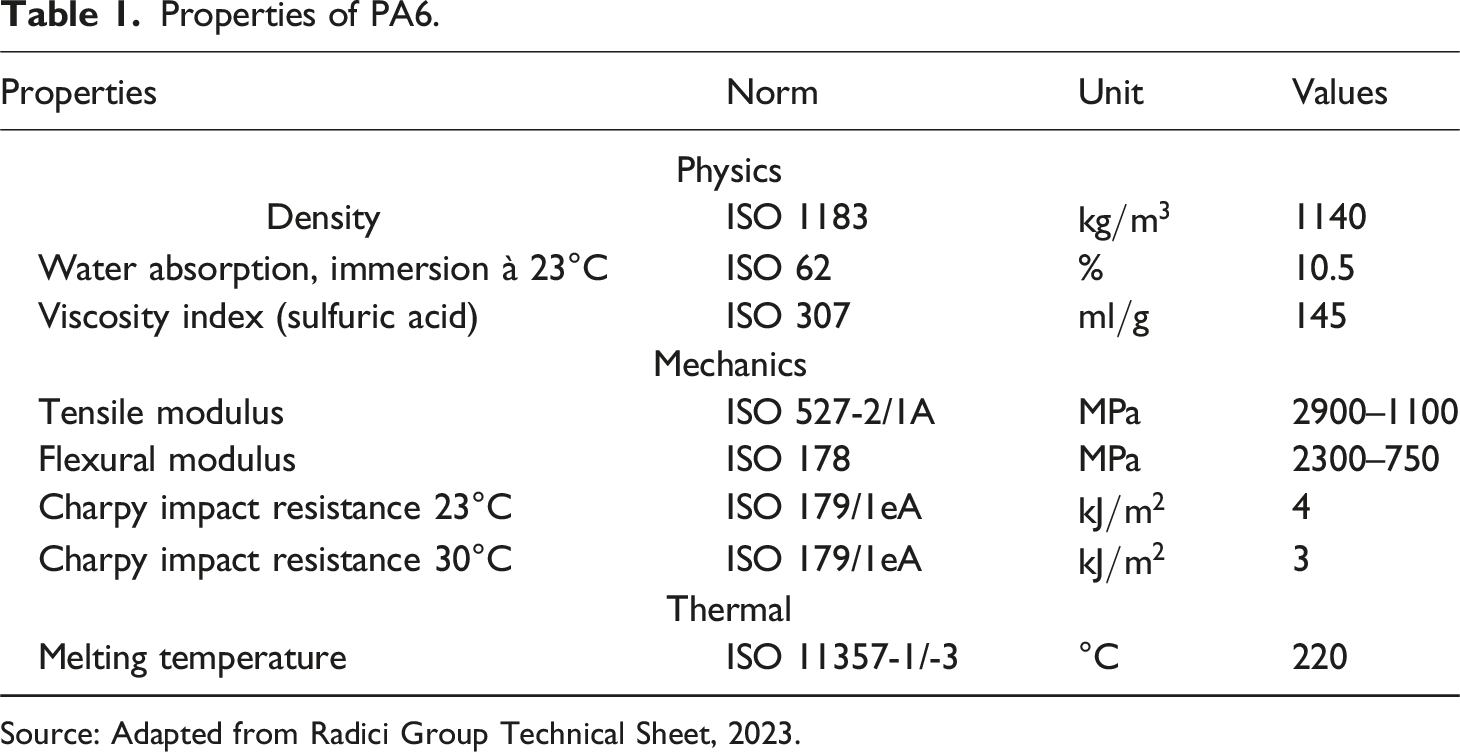
Source: Adapted from Radici Group Technical Sheet, 2023.
Experimental methods
Characterization of GNPs
The techniques and methodologies used for characterizing the GNPs are described below.
Raman spectroscopy
The Raman Spectrometer from the Laboratory of the National Institute of Metrology, Quality and Technology (INMETRO) was utilized in this study. Raman spectra were captured using a Witec Alpha 300 spectrometer with a 514.5 nm laser line, using a microscope with a 100x objective for all samples. The laser power was maintained at 0.5mW.
Brunauer–Emmett–Teller (BET) method
N2 adsorption–desorption isotherms of the prepared material were obtained at the boiling point of liquid nitrogen (77 K) using a St2 Nova 600 surface analyzer. BET method was carried out at the Laboratory of the National Institute of Metrology, Quality and Technology (INMETRO). The specific surface area was evaluated using the BET (Brunauer, Emmett and Teller) multipoint method. 40
Field emission gun-scanning electron microscopy (FEG-SEM)
The chemical and topographical characteristics of GNPs were evaluated through Field emission gun-Scanning electron microscopy (FEG-SEM) using the JEOL JSM-7100FT FEG-SEM at the Nanofab Laboratory of the State University of Rio de Janeiro.
X-ray diffraction (XRD)
X-ray diffraction measurements, using the powder method, crucial analysis to characterize GNPs were performed to identify phases in the samples, and detect potential contaminating phases introduced during powder production. For this purpose, a Bruker-AXS D8 Advance Eco diffractometer with Cu Kα radiation (40 kV/25 mA) was used at the Multiuser Laboratory of Technological Characterization (LMCT) of the Mineral Technology Center (CETEM). The experimental setup spanned an angular range of 5°–80° (2θ), scanning at 0.01° per 92 s per step with a state-of-the-art (energy discriminating) LynxEye XE silicon drift position-sensitive linear detector.
Fourier transform infrared spectroscopy (FTIR)
FTIR spectroscopy analysis of the GNPs samples was performed on a Perkin-Elmer Frontier FTIR spectrometer at the Biomaterials Laboratory of the Polytechnic Institute of the State University of Rio de Janeiro. An attenuated total reflectance (ATR) accessory, the Pike Miracle Single Reflection ATR from Technologies, was used for all analyses
Thermogravimetric analysis (TGA)
The thermal stability of GNPs was determined by thermogravimetric analysis (TGA). The analysis was performed on a TGA Universal V4.5 A (TA Instruments, USA). Analyzer at the Center of Excellence in Recycling and Sustainable Development Laboratory of the Institute of Macromolecules Professor Eloisa Mano of the Federal University of Rio de Janeiro. The test was conducted, employing approximately 6 mg of sample in porcelain capsules with a nitrogen atmosphere of 50 mL.min−1. TGA analysis comprised heating from room temperature to 700°C at a rate of 10°C/min. Equipment calibration utilized an indium (In) standard before each analysis.
Composites preparation
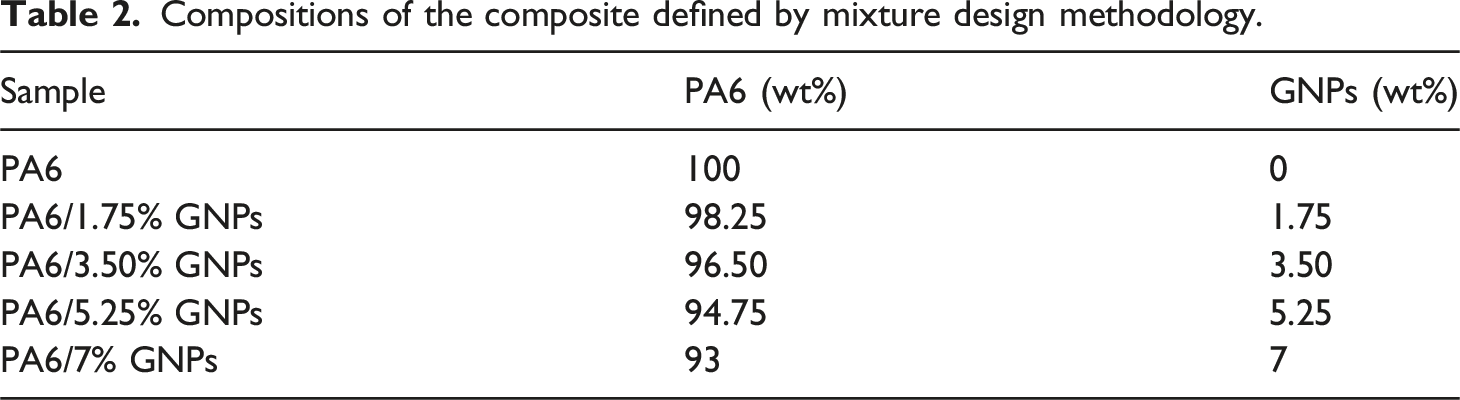
A set of PA6 compositions was formulated based on statistical planning using a Minitab 19 software. GNPs were added to PA6 in five different proportions: PA6 without GNPs (PA6), PA6 with 1.75 wt% GNPs (PA6/1.75% GNPs), PA6 with 3.50 wt% GNPs (PA6/3.5% GNPs), PA6 with 5.25 wt% GNPs (PA6/5.25% GNPs) and PA6 with 7 wt% GNPs (PA6/7% GNPs).
The selection of GNPs content was based on a comprehensive scan of their influence on the mechanical and thermal properties of PA6. This approach aims to provide support for subsequent investigations, notably the study of a ternary PA6, GNPs and short carbon fibers (SCF) system, to be conducted using the response surface methodology.
Compositions of the composite defined by mixture design methodology.
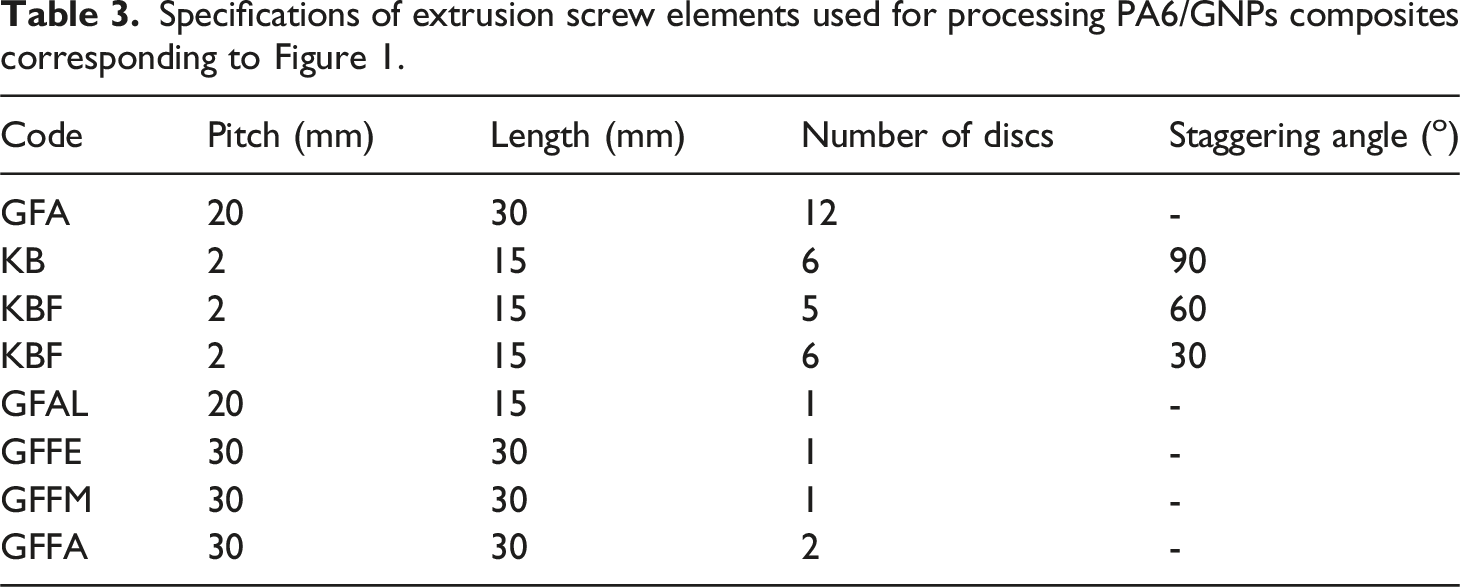
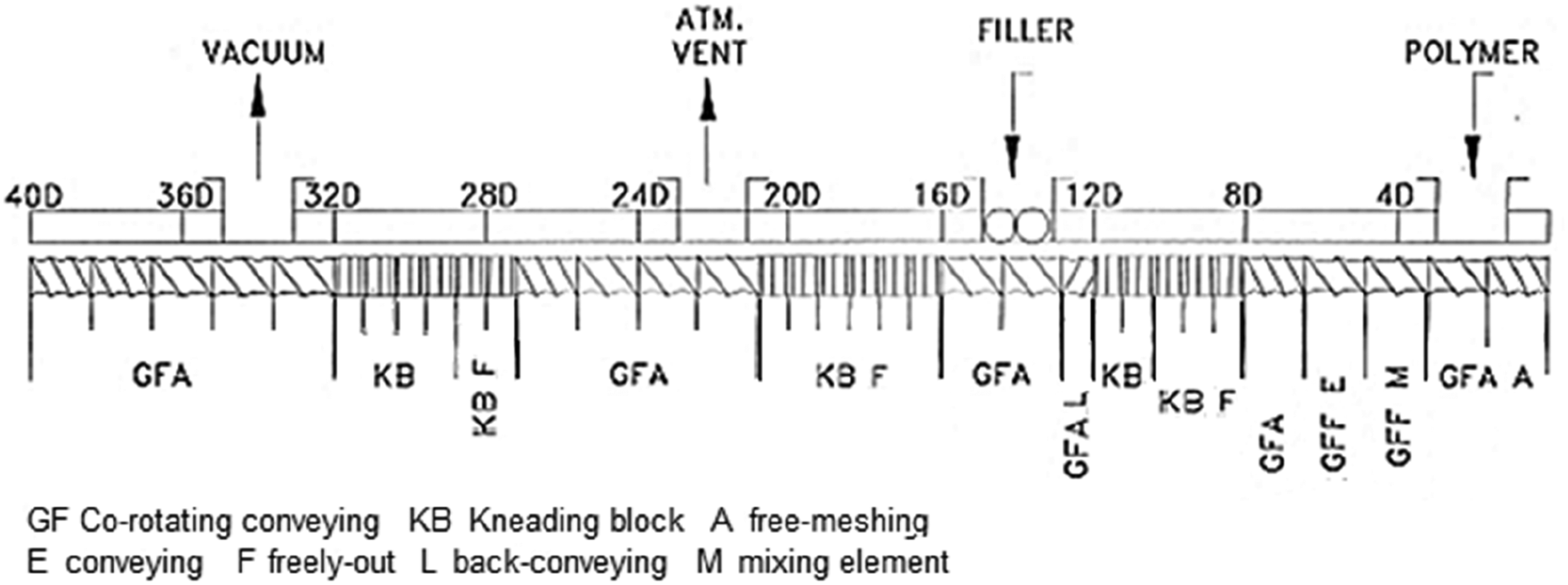
Specifications of extrusion screw elements used for processing PA6/GNPs composites corresponding to Figure 1.
Screw configuration used for processing PA6/GNPs composites.
Composites characterization
The techniques and methodologies used for characterizing the composites are described below:
Differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA)
The thermal transitions of the PA6/GNPs composites were evaluated by differential scanning calorimetry (DSC). The analyses were performed in a Perkin-Elmer (Pyris 1) equipment under a nitrogen atmosphere of 50 mL.min. Approximately 15 mg of each sample were encapsulated in porcelain crucibles used for DSC analysis. Each sample was subjected to five heating or cooling cycles: (i) heating from 50°C to 300°C at a rate of 20°C/min; (ii) isotherm at 300°C for 1 min; (iii) cooling from 300°C to 50°C at a rate of 10°C/min; (iv) isotherm at 50°C for 1 min; and (v) heating from 50°C to 300°C at a rate of 10°C/min. The equipment calibration performed before each analysis used indium (In) as standard. Parameters such as crystallization temperature (Tc), melting temperature (Tm), crystallization enthalpy (ΔHc) and fusion enthalpy (ΔHm) were obtained from the thermograms. The degree of crystallinity (χc) was calculated using equation (1): ΔHm is the melting enthalpy of PA6; ΔHm (PA6100%) is the theoretical melting enthalpy of 100% crystalline PA6 which is equivalent to (ΔHm(PA6100%) = 190.8 J.g−1)
41
; and Φ is the mass fraction of PA6 in the mixture. The thermal stability of the composites was determined by thermogravimetric analysis (TGA) and derivative thermogravimetric analysis (dTG). These analyses were performed on a TGA Q50 V6.7 (TA Instruments, USA). The TGA analysis consisted of heating the sample from room temperature to 600°C at a rate of 10°C/min. The DSC and TGA analyses were performed at the Materials Laboratory of the State University of Rio de Janeiro.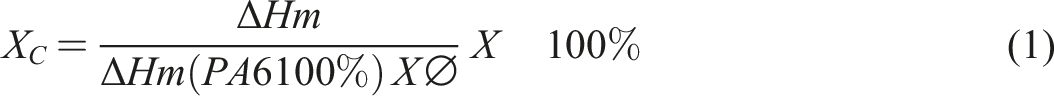
Fourier transform infrared spectroscopy (FTIR)
FTIR spectroscopy analysis of the samples was performed on a Perkin-Elmer Frontier FTIR spectrometer at the Biomaterials Laboratory of the Polytechnic Institute of the State University of Rio de Janeiro. An attenuated total reflectance (ATR) accessory, the Pike Miracle Single Reflection ATR from Technologies, was used for all analyses
Field emission gun- scanning electron microscopy (FEG-SEM)
To evaluate the progressive increase in the concentration of GNPs in the samples and the dispersion of the nanoparticles in the polymer matrix, the PA6/GNPs composites were characterized using the JEOL JSM-7100FT FEG-SEM at the Nanofab Laboratory of the State University of Rio de Janeiro.
X-ray diffraction (XRD)
To evaluate the crystalline structure of the materials, PA6/GNPs composites and pure PA6, the materials were characterized by X-ray diffraction. For this purpose, a Brucker-AXS D8 Advance Eco diffractometer with Cu Kα radiation (40 kV/25 mA) was used at the Multiuser Laboratory of Technological Characterization (LMCT) of the Mineral Technology Center (CETEM). The experimental setup spanned an angular range of 5°–80° (2θ), scanning at 0.01° per 92 s per step with a state-of-the-art (energy discriminating) LynxEye XE silicon drift position-sensitive linear detector.
Mechanical properties
Mechanical properties were evaluated by tensile, flexural and impact tests. Tensile and flexural tests were performed on a Shimadzu Brazil AG-X 100 kV Universal Testing Machine, in accordance with ASTM D 638 and ASTM D 790.42,43 For each composition, ten (10) specimens were tested. Charpy impact tests were performed with a 0.5 J pendulum on a CEAST 9050 impact tester (Instron Brazil) in accordance with ASTM D 6110. 44 The effect of incorporating varying contents of GNPs on the properties of PA 6 was evaluated using one-way ANOVA statistical analysis.
Results and discussion
Characterization of GNPs
Structural features and surface area of the GNPs obtained by raman scattering spectrum and BET method
Figure 2 shows the first and second order Raman scattering spectrum of graphene. In Figure 2, an intense D peak can be observed at approximately 1325 cm−1, indicating that there are many defects in the crystal structure of the GNPs resulting from functional groups associated with oxidation processes, edge effects, vacancies, among others. The presence of a D band is an indication of disorder in the crystal structure and does not occur in perfect crystals. The 2D band occurs at approximately 2675 cm−1 and is related to the layers present in graphene, being an indication of the nature of graphene. According to the literature, the absence of symmetry in the 2D peak suggests that multilayer flakes are present.
45
Raman spectrum of GNPs.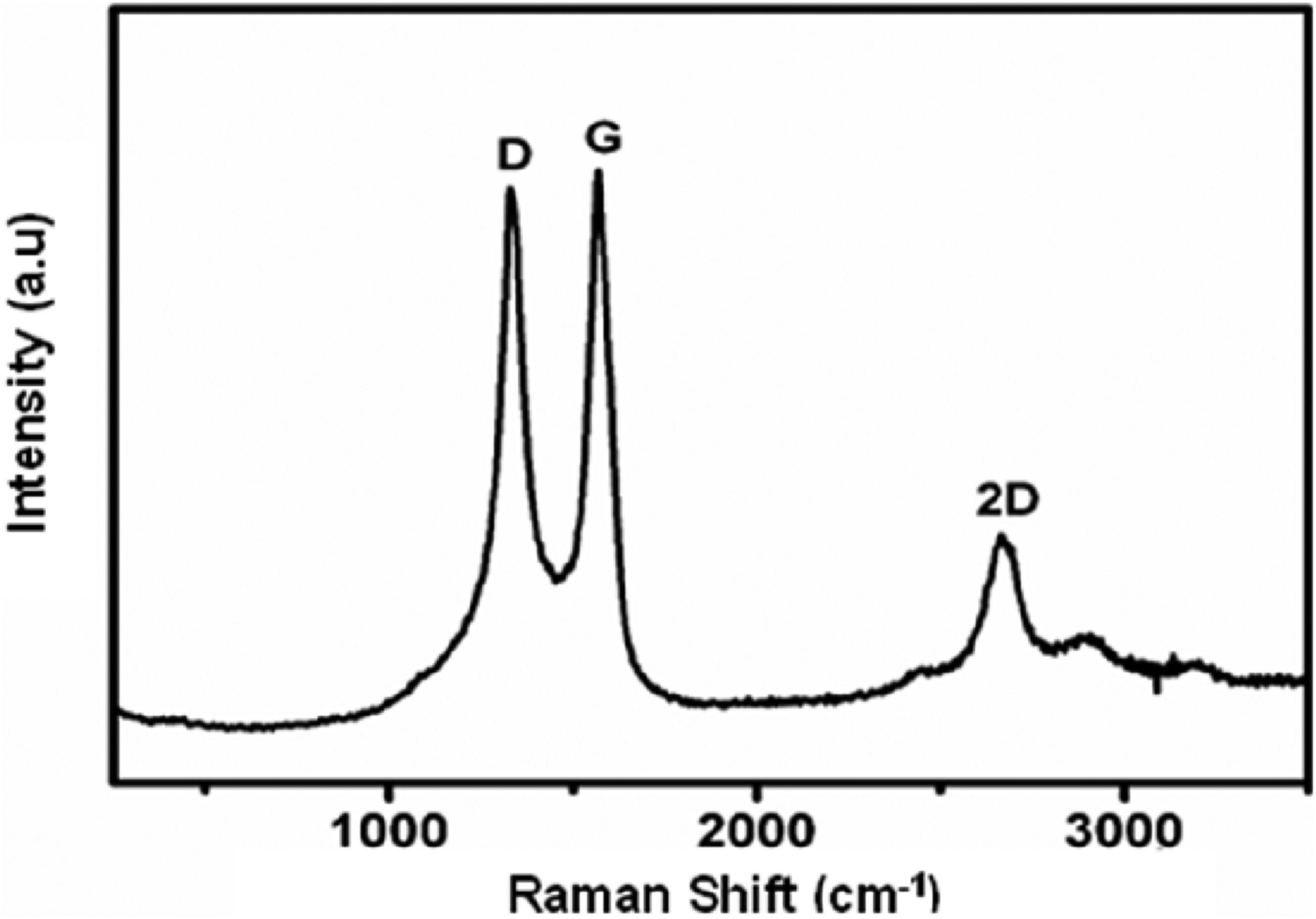
The maximum intensity of the G peak occurs at approximately 1563 cm−1. The G peak is a characteristic of ordered graphitic structures and, in the literature, a value of the ratio between the intensity of the D band and the G band (ID/IG) value close to zero is an indicative of crystalline graphite. As this value increases, the material loses its crystallinity and becomes structurally more disordered. ID/IG ratio close to zero can also be attributed to the reduction in the average size of the sp2 domains, after the reduction of graphene oxide (GO), due to the formation of several new graphite domains in the reduced graphene oxide (rGO). These new domains, although smaller in size than those found in GO, before the reduction, are more numerous.46,47
According to the Raman spectrum of the GNPs used in this work, the ID/IG ratio is 0.96, a higher ratio than that attributed to crystalline graphite. This result indicates the presence of more defects and chemical oxidation of graphite in GO. On the other hand, an I2D/IG ratio of 0.32 was observed, suggesting the presence of more than two graphene layers. This result indicates that GNPs sample has a reduced number of graphene layers, and it is plausible to infer that GNPs can be classified as a better quality few-layer graphene (FLG).46–48
The result of the analysis of the Raman spectrum of graphene showed a greater predominance of the D and G bands in the spectrum due to the presence of oxygenated groups in the crystal lattice.49,50 Similar results were found by CHEN et al. (2010) and LIU et al. (2013) for graphene oxide, where a greater intensity of the G band was observed in the Raman spectrum, which suggests that the material analyzed is graphene oxide.51,52
The surface area of graphene nanoplatelets is considered higher than that of other carbon-based nanomaterials, which contributes to maximizing the stress transfer from the polymer matrices to the filler. The specific surface area of the GNPs used in this work and obtained by the BET multipoint method was 687 m2g−1. This value was lower than that provided by the supplier, respectively equal to 750 m2g−1. However, there is no information on the method used for this determination. In any case, the value obtained is within the range of values expected for this parameter.
FEG-SEM morphological profiling of GNPs
Flexibility is a striking feature of graphene.
53
The micrographs of GNPs, obtained by FEG-SEM, are shown in Figure 3. The folds along the graphene sheets are indicative of the flexibility of this material. The morphological profile shown in Figure 3 indicates pronounced wrinkling due to the incorporation of sp3 carbon atoms, which produces the breakage of the sp2 hybridized flat graphene nanosheets.53,54 FEG-SEM of GNPs.
Analysis of X-ray diffraction (XRD) Spectrum of GNPs
Figure 4 shows the XRD spectrum of graphene. The peak at the 2θ angle of 26.30° is an indication of the (002) crystalline plane. In addition, a presence of a second peak at the 2θ angle of 43.50°, corresponding to the (101) plane, is attributed to the specific orientation of the graphene layers.
55
In fact, Figure 4 shows that the diffraction pattern of graphene does not present well-defined peaks, but rather bands, due to the spacing in the crystal lattice of graphene oxide that is not well defined, like that of highly ordered graphite. In addition, the XRD spectrum was also used to calculate the number of graphene layers, indicating that the sample consists of graphene with nine to 10 layers, i.e., few-layer graphene (FLG). This result is in good agreement with those obtained from the Raman spectra of GO.56,57 X-ray diffraction spectra of GNPs.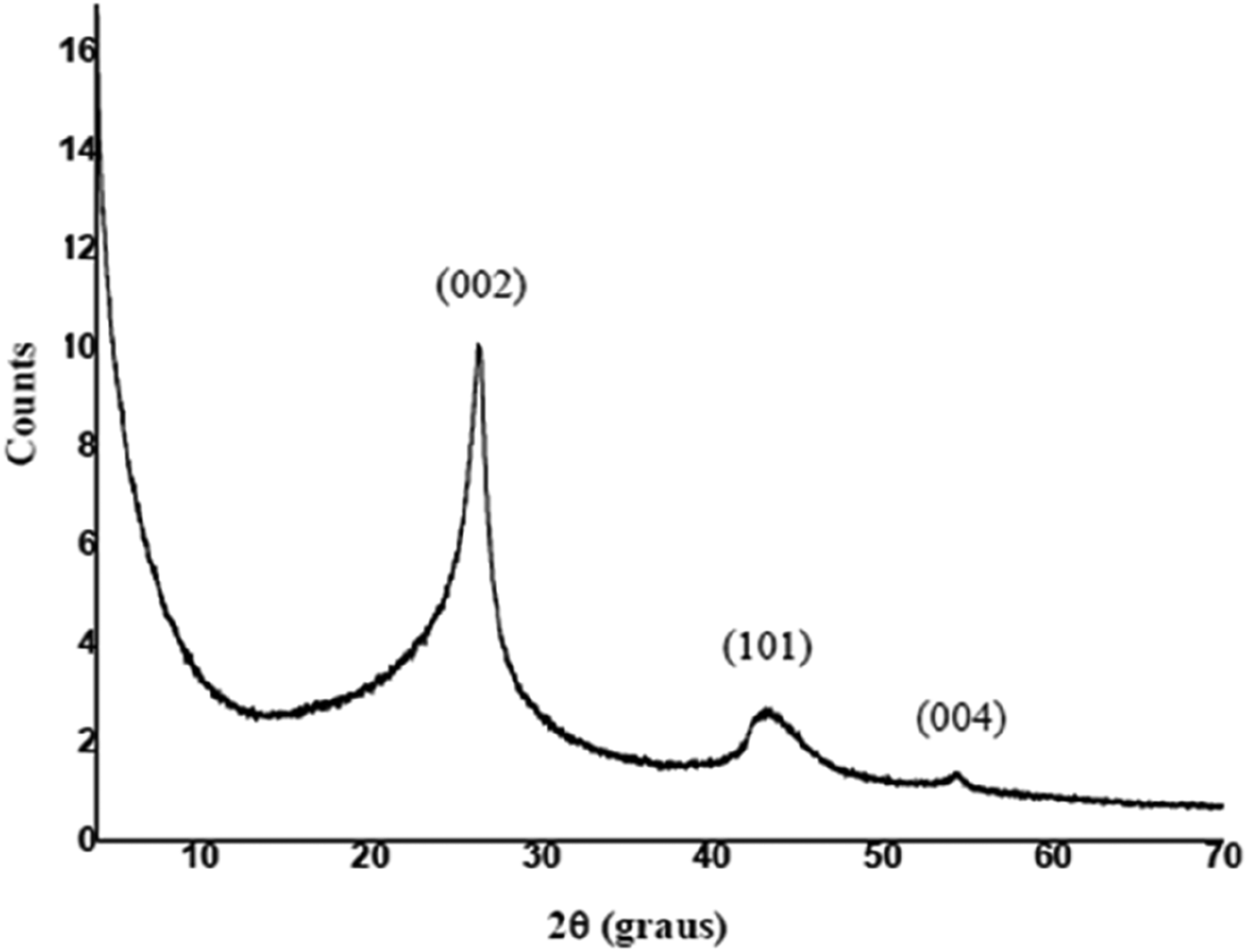
Analysis of the fourier transform infrared spectrum (FTIR) of GNPs
Figure 5 depicts the FTIR spectrum of GNPs. The following absorption bands, corresponding to the oxygenated groups derived from the graphite oxidation process were observed: 3416 cm−1 assigned to (O-H) hydroxyl group stretching vibration; 1627 cm−1 assigned to (C = O) carbonyl group and (C = O) double carboxylic bond (C = O) stretching vibrations; 1530 cm−1 assigned to carbon aromatic (C = C) vibrations; 1380 cm−1 assigned to (OH) carboxyl group stretching vibrations; 1023 cm−1 attributed to alkoxy groups vibrations.
58
Therefore, there is evidence that the acquired nanoplatelets have functional groups. Infrared absorption spectra of GNPs.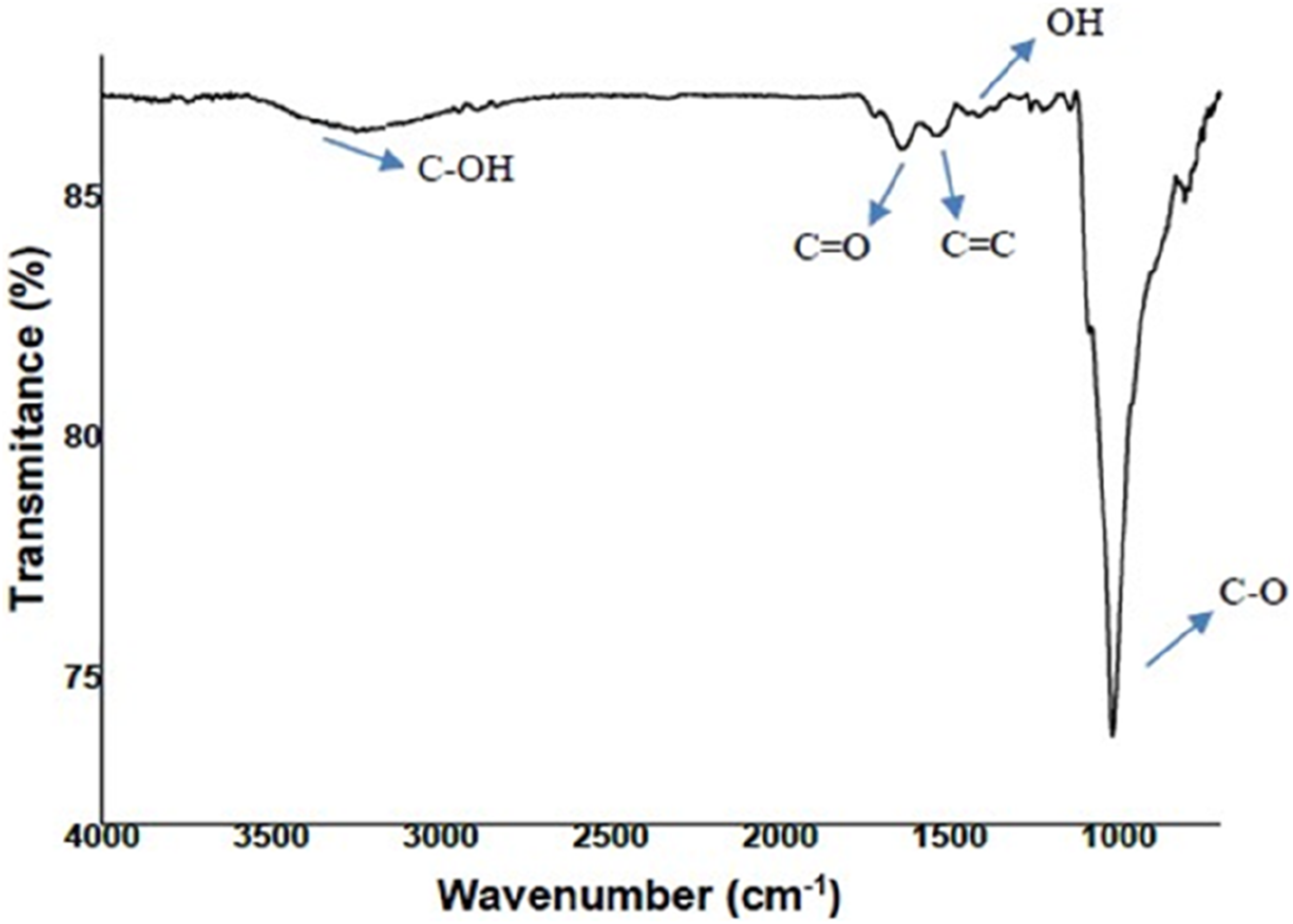
Thermogravimetric analysis (TGA) of GNPs
Figure 6 presents the TGA curve of GNPs. It is observed that GNPs present thermal stability up to approximately 700°C, where the begins combustion of the carbon structure of the sample. Up 690°C, the total percentage of mass lost was 10.9%. These results were also found by You et al.
59
and corroborate with obtained in the characterization of GNPs indicates that GNPs sample can be classified it as a better-quality material.27,29,60,61 TGA curve of GNPs.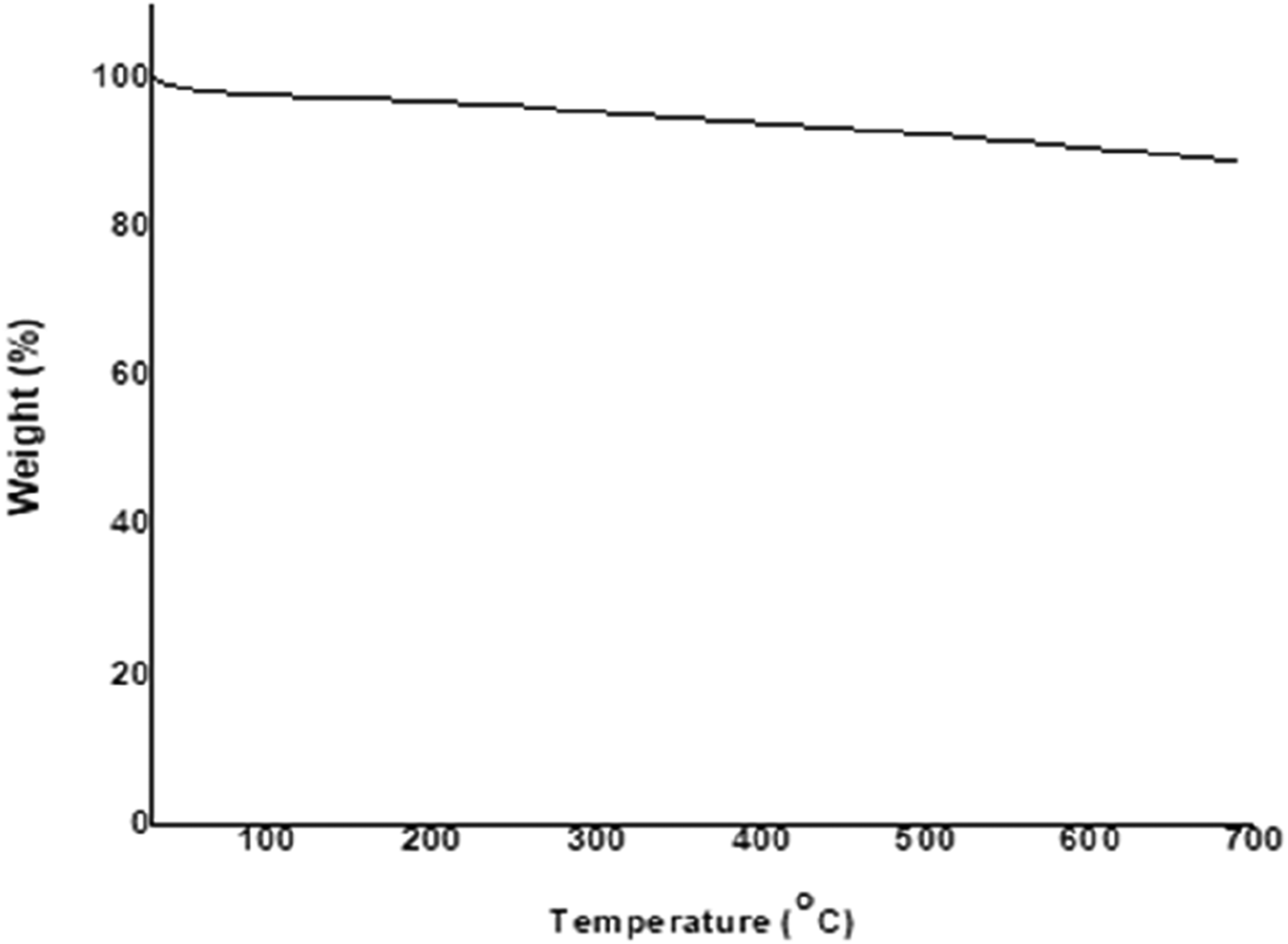
Composites characterization
Thermal stability of PA6 and PA6 composites
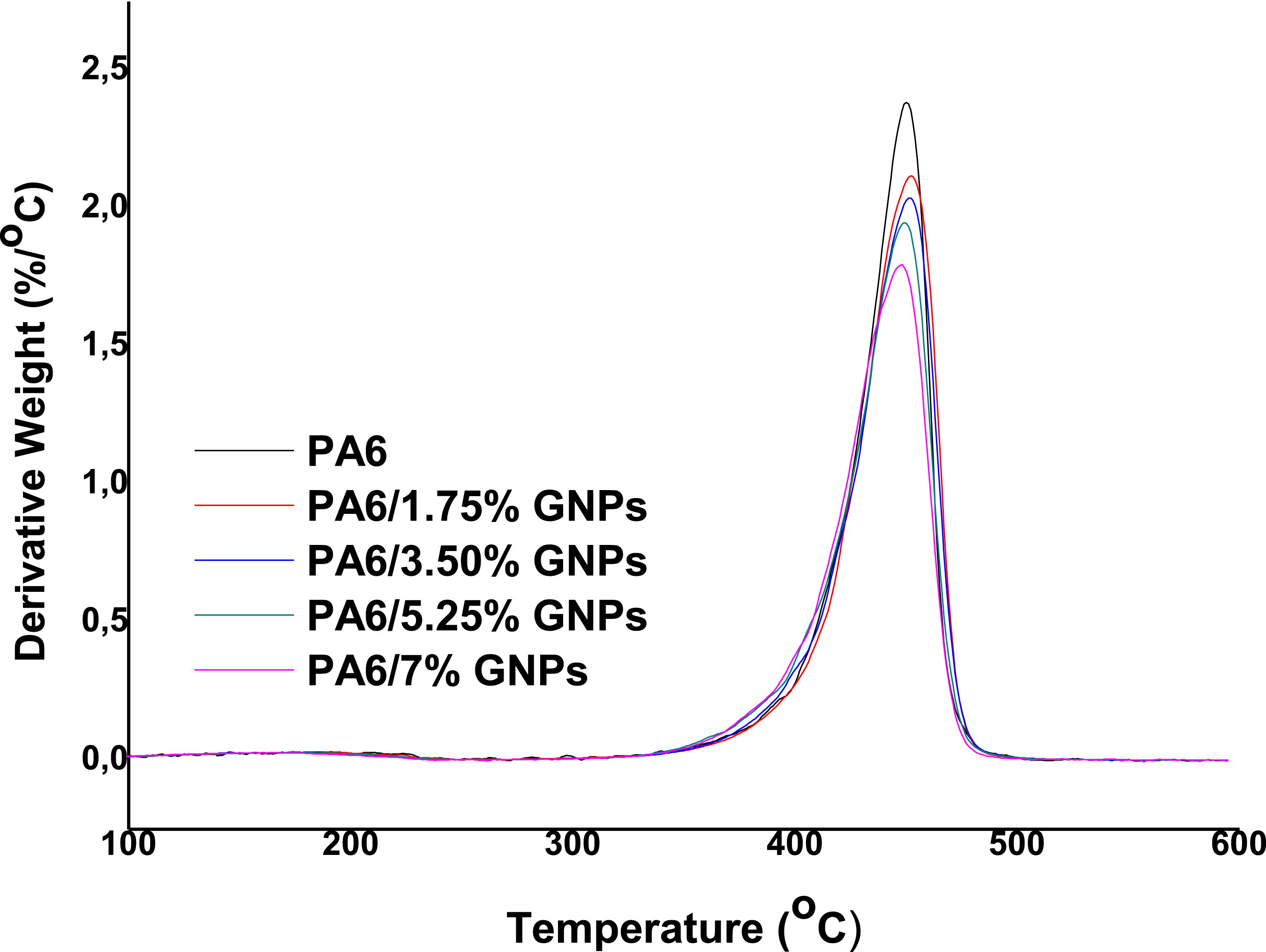
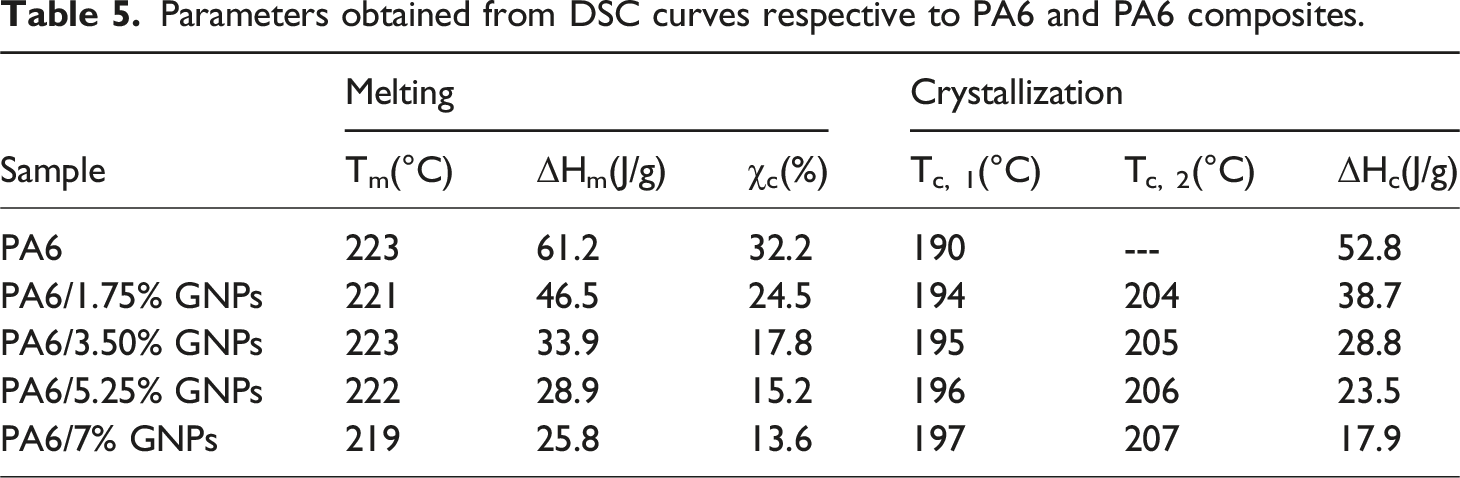
Thermogravimetric analysis (TGA) and derivative thermogravimetric analysis (dTG) of PA6 and composites were carried out to determine the effects of adding GNPs on the thermal stability of PA6. Figure 7 presents the TGA curves for all materials and Figure 8 shows the respective dTG curves. Table 4 shows the thermal stability parameters obtained from TGA and dTG curves of these materials. It is observed that the incorporation of low concentration of GNPs (up to 5.25 wt%) promotes a slight increase in the thermal stability of PA6, due to their strong secondary interaction with the polymer (Table 4).
62
The poor dispersion of GNPs particles in the polymer matrix seems to promote an adverse effect on the thermal stability of the composites obtained. In the temperature range from 50°C to 320°C, all samples present an average mass loss of 3%–4%, attributed to the elimination of residual moisture in the polymer. Subsequent mass loss occurs between 353°C (T5%) and 443°C (T50%). At 600°C, a percentage of residues (% R) is detected in each sample analyzed (see Table 4). The residual masses are consistent with the formulated filler composition for each sample. TGA curves of PA6 and PA6 composites. dTG curves of PA6 and PA6 composites. Parameters obtained from TGA curves respective to PA6 and PA6 composites.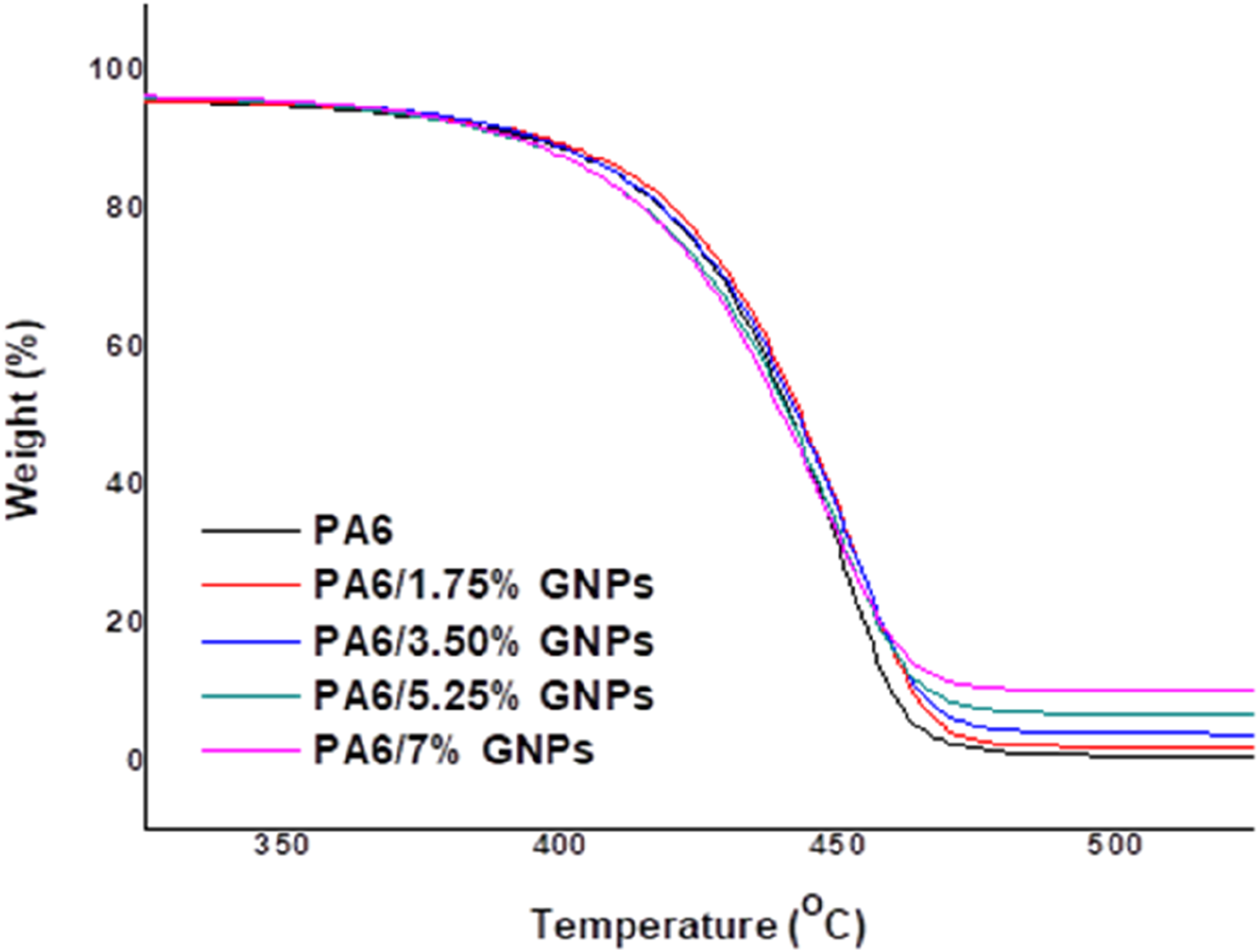

Differential scanning calorimetry (DSC) thermograms of PA6 and PA6 composites
Differential scanning calorimetry (DSC) is a thermal analysis technique commonly used in polymers to provide information on thermal transitions, such as: melting and crystallization temperatures (Tm and Tc), enthalpies of melting and crystallization (ΔHm and ΔHc) and degree of crystallinity (χc).
Figure 9 shows the DSC curves for PA6 and PA6 composites obtained during the second heating cycle (50°C–300°C). The DSC curve of PA6 shows two main melting peaks are located at 217°C and 223°C, which correspond to the melting of the γ and α-crystallites of PA6, respectively. It is well known that PA6 exhibits polymorphism behavior having three crystalline forms (α, β and γ) which generally coexist.63–65 Contrary, for the nanocomposites only a single endotherm peak appears, at the same temperature with the melting of α crystallites. This behavior indicates that the constraints imposed by GNPs on the polymeric chains favor the formation of α-phase crystals. The latter is very important as the α-crystallites constitute the more thermodynamically stable phase and exhibit better mechanical properties comparing to γ-phase crystals.
63
Furthermore, the melting peaks remain in the same position as the amount of GNPs increases, indicate that the incorporation of GNPs at concentrations up to 5.25 wt% did not significantly effect melting temperatures. However, the incorporation of a higher concentration of nanoparticles caused a decrease in the melting temperature, likely due to the higher entropy of a more disordered structure with agglomerated nanoparticles66,67 in addition to the greater restriction on the mobility of the polymer chains, which prevents the formation of strong and organized crystals. DSC Curves: Melting endotherms of PA6 and PA6 composites.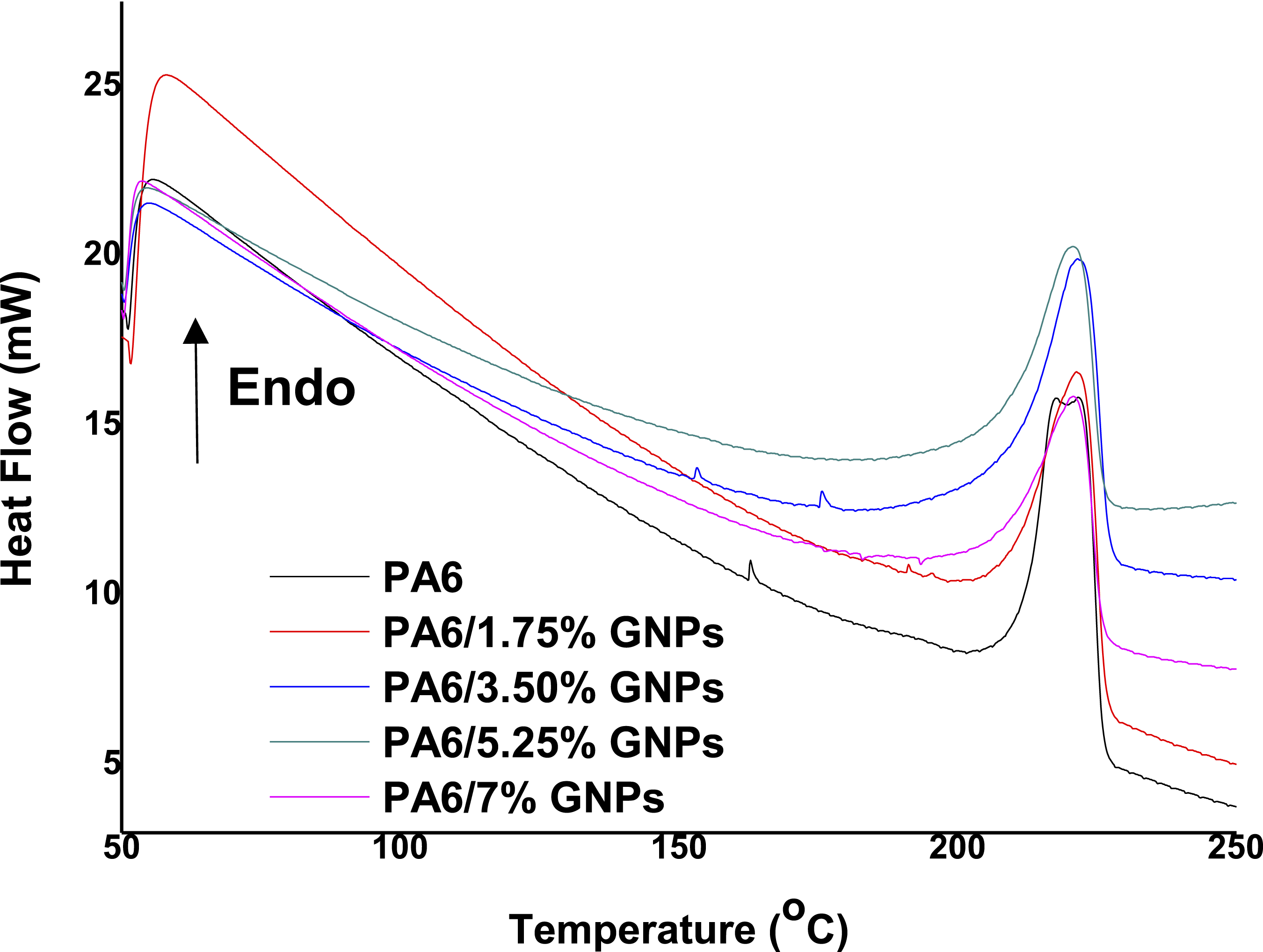
Figure 10 shows the DSC curves obtained during a cooling step from 300°C to 50°C under a nitrogen atmosphere. The PA6 thermogram shows only one crystallization peak (Tc, 1) observed at approximately 190°C. The DSC curves of PA6/GNPs composites show that the crystallization peak (Tc, 1) gradually shifts to higher temperatures as the amount of GNPs increases, indicating that GNPs act as nucleating agents. This behavior has been previously reported in the literature for nanocomposites of different matrices, such as polypropylene, polyamide 6 and polystyrene.63–67 The presence of a second crystallization peak (Tc, 2) is observed at higher temperatures only for PA6/GNPs composites, and its position gradually shifts toward higher temperatures, with a constant increase in magnitude, as the GNPs concentration increases. This behavior indicates that the second Tc, 2 peak is closely related to the presence of GNPs in the PA6 matrix. The double crystallization peak in nanocomposites may be related to two different types of crystal morphologies or to a two-step crystallization behavior.
68
Furthermore, the crystallization peak of PA6 is sharper compared to those of nanocomposites. The incorporation of GNPs in the PA6 matrix leads to a broader crystallite size distribution and therefore the crystallization peaks become wider. DSC curves: Crystallization exotherms of PA6 and PA6 composites.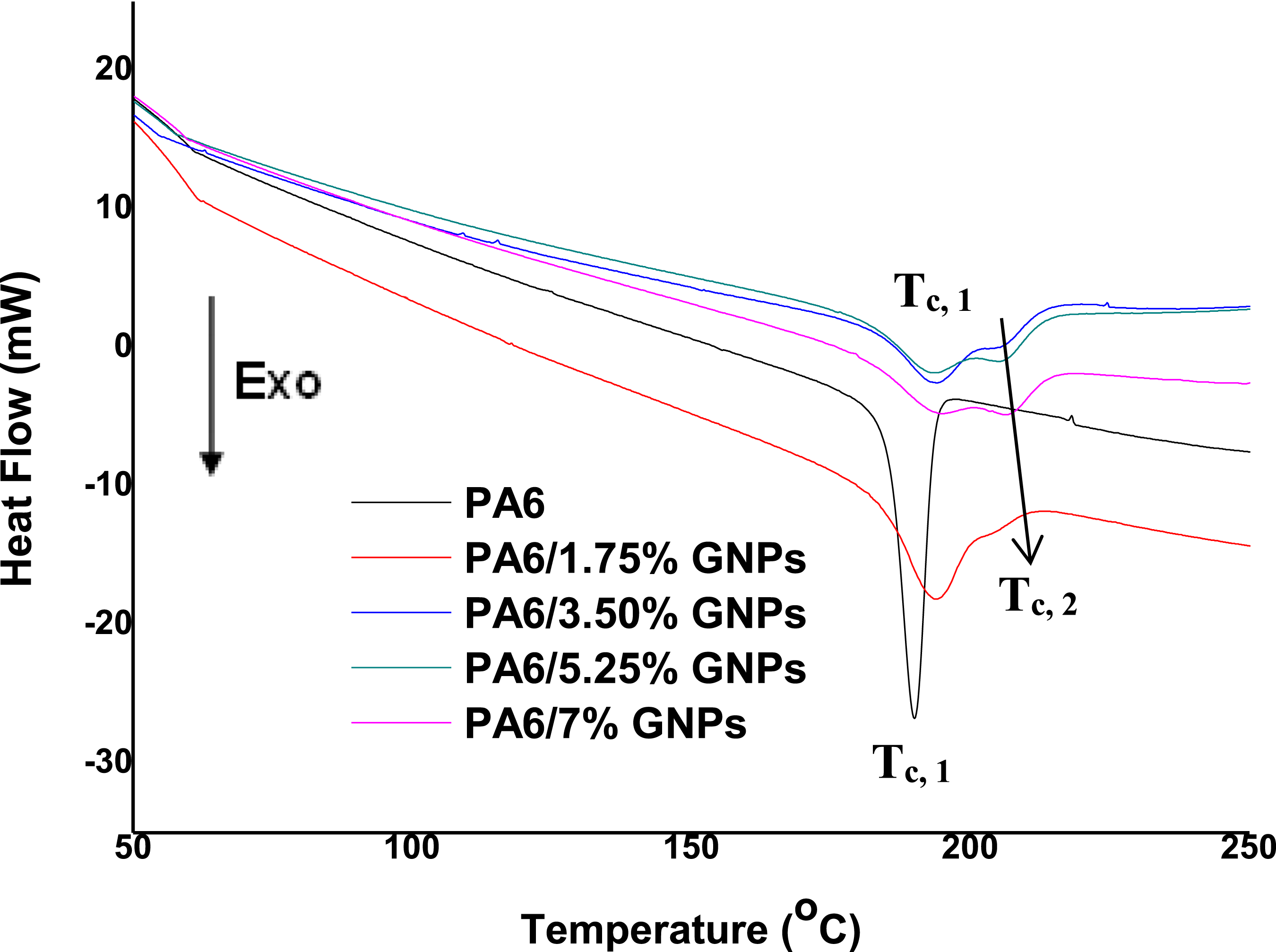
Parameters obtained from DSC curves respective to PA6 and PA6 composites.
Analysis of the fourier transform infrared spectrum (FTIR) of PA6 and PA6 composites
Figure 11 depicts the FTIR spectrum of PA6, revealing absorption bands at distinct positions: 3294 cm− 1 for asymmetric axial stretching vibrations of the [N–H] free group; 2931 cm− 1 and 2862 cm− 1 for asymmetric and symmetric stretching of the [C–H] group in CH2, respectively; 1638 cm− 1 for stretching of the [C = O] group of the primary amide; 1543 cm− 1 for folding of the [N–H] group and stretching of the [C–N] group of the secondary amide.69,70 It is possible to observe characteristic bands of the α phase of PA6 in the bands 930 cm−1 and 1200 cm−1 (Figure 12), corresponding to the CO-NH interaction in the crystallographic plane of PA6.
71
Infrared absorption spectra of PA6 and PA6 composites. Cut in FTIR spectra of PA6 and PA6 composites.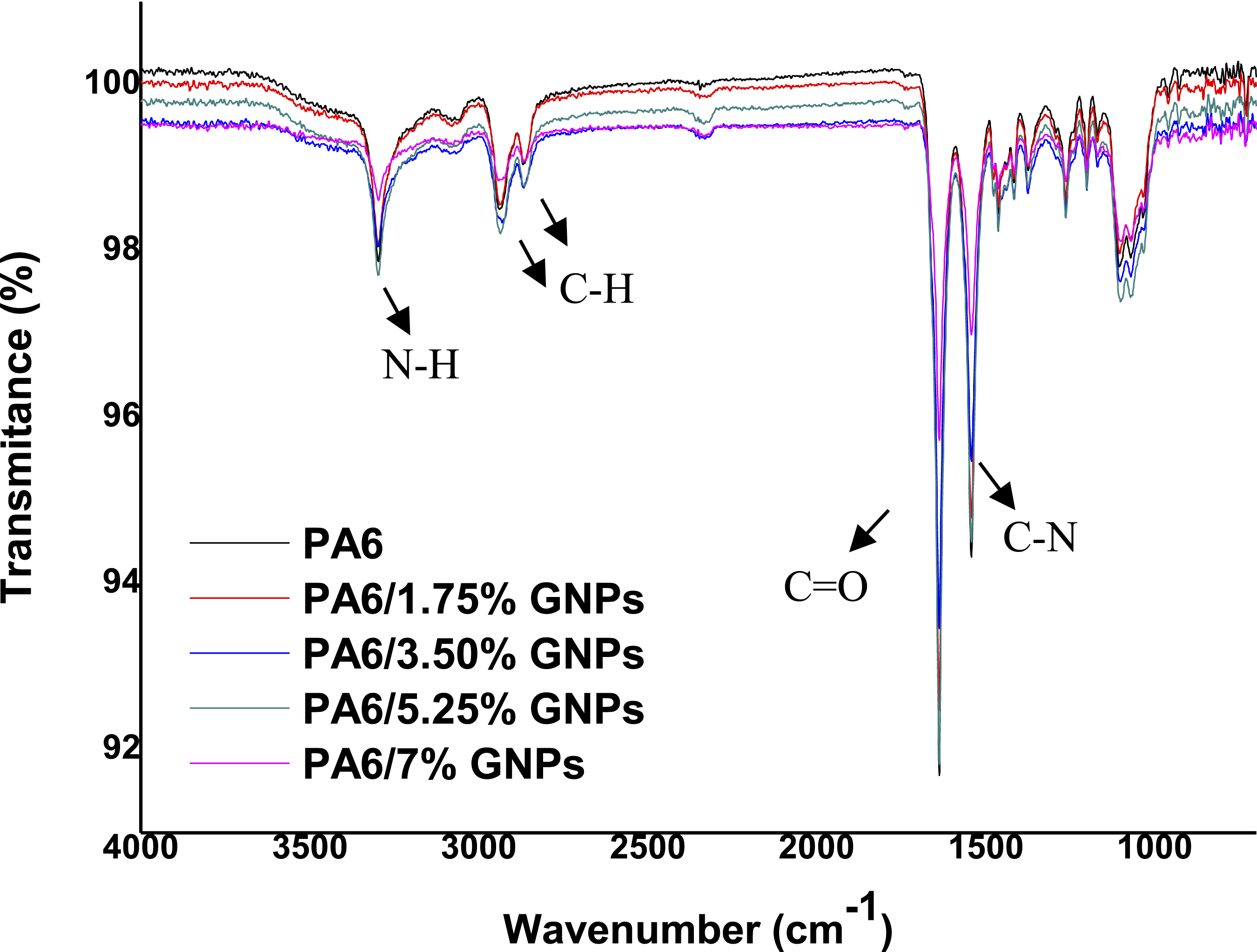
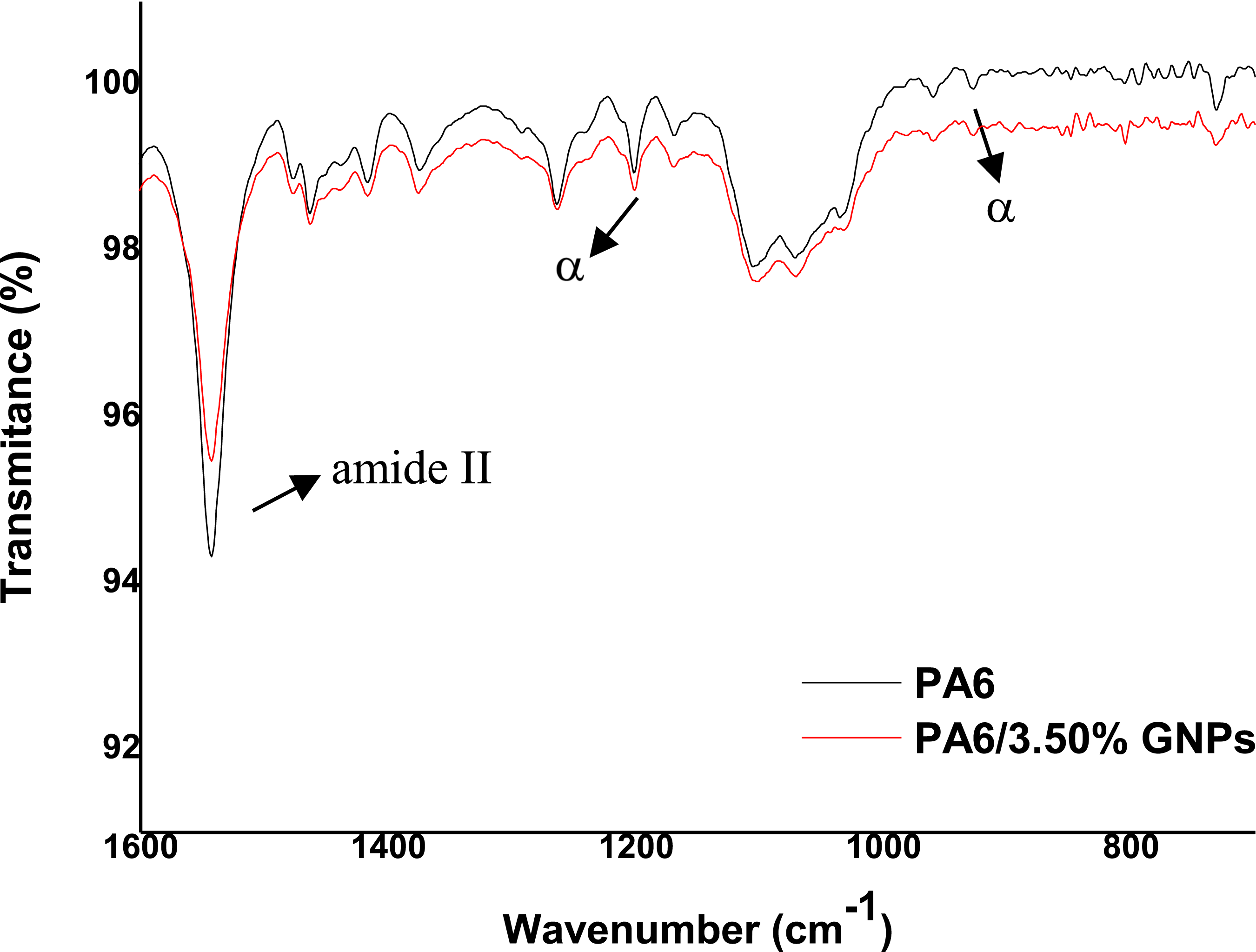
For PA6 with GNPs samples, there were no major changes in the spectrogram compared to the pure PA6 sample. This can be explained by the low percentage of GNPs added to the polymer. Figure 12 presents a section of the FTIR spectrogram of the pure PA6 sample and with GNPs sample, where a reduction in intensity of the entire spectrum of the with GNPs composite and a slight shift of the secondary amide band (1543 cm−1), which demonstrates the interaction of GNPs with PA6 through hydrogen bonds. 69 This interaction highlights the effective integration of GNPs into the polymeric matrix, contributing to the understanding of changes in the final properties of the compound.
FEG-SEM morphological profiling of PA6 and PA6 composites
The screw configuration used for composites processing shown in Figure 1 is constituted by conveying and mixing elements, as well as kneading blocks, that enhance distributive and dispersive mixing. This configuration provides better dispersion and distribution of nanofillers in polymer matrices, as evidenced in our previous work, in which polypropylene reinforced with alumina nanoparticles and short carbon fibers nanocomposites were obtained. 72 Then, we expected to obtain a suitable mixture of PA6 and graphene.
In Figure 13, micrographs of PA6 are presented at different GNPs contents: without GNPs (a), with 1.75 wt% (b) and 3.50 wt% (c). In image (a), the PA6 without GNPs displays a smooth surface, without any encrustations, while in the images (b and c), coating of the PA6 surface with GNPs is noticeable. Notably, a more evident concentration of GNPs is observed in the PA6 samples with 3.50 wt% addition, indicating a progressive increase in the presence of GNPs in the samples and a satisfactory dispersion of the material. An increase in roughness was also observed with increasing of graphene content. These results suggest that the Pa6/GNPs mixture was effective. Atomic force microscopy (AFM) will be used in the future to gain further insight into the resulting morphology. FEG-SEM of PA6 without GNPs (a), PA6 with 1.75 wt% GNPs (b) and PA6 with 3.50 wt% GNPs (c).
Analysis of X-ray diffraction (XRD) spectrum of PA6 and PA6 composites
Figure 14 shows the diffractogram of PA6 and PA6 composites. The diffractogram of PA6 and PA6/GNPs composites shows the diffraction planes at 2θ = 20° (α1) and 2θ = 24° (α2) of pure PA6, and refer to the planes (200) and (002, 202) of the α crystalline phase of the polymer, respectively.
73
The maximum diffraction at 26° for GNPs appears in the PA6/GNPs composites, which may mean high stacking and disordering of the materials lamellae, therefore indicating that the graphene is not well dispersed and distributed throughout the matrix.
74
A change in the intensity of α1 and α2 was also observed for the PA6/GNPs composites, when compared to pure PA6. This behavior indicates that the crystalline behavior of the polymer was affected by the addition of graphene, that is, the hydrogen bonds between the PA6 chains suffered disturbance in their crystalline regularity.
69
X-ray diffraction spectra obtained of PA6 and PA6 composites.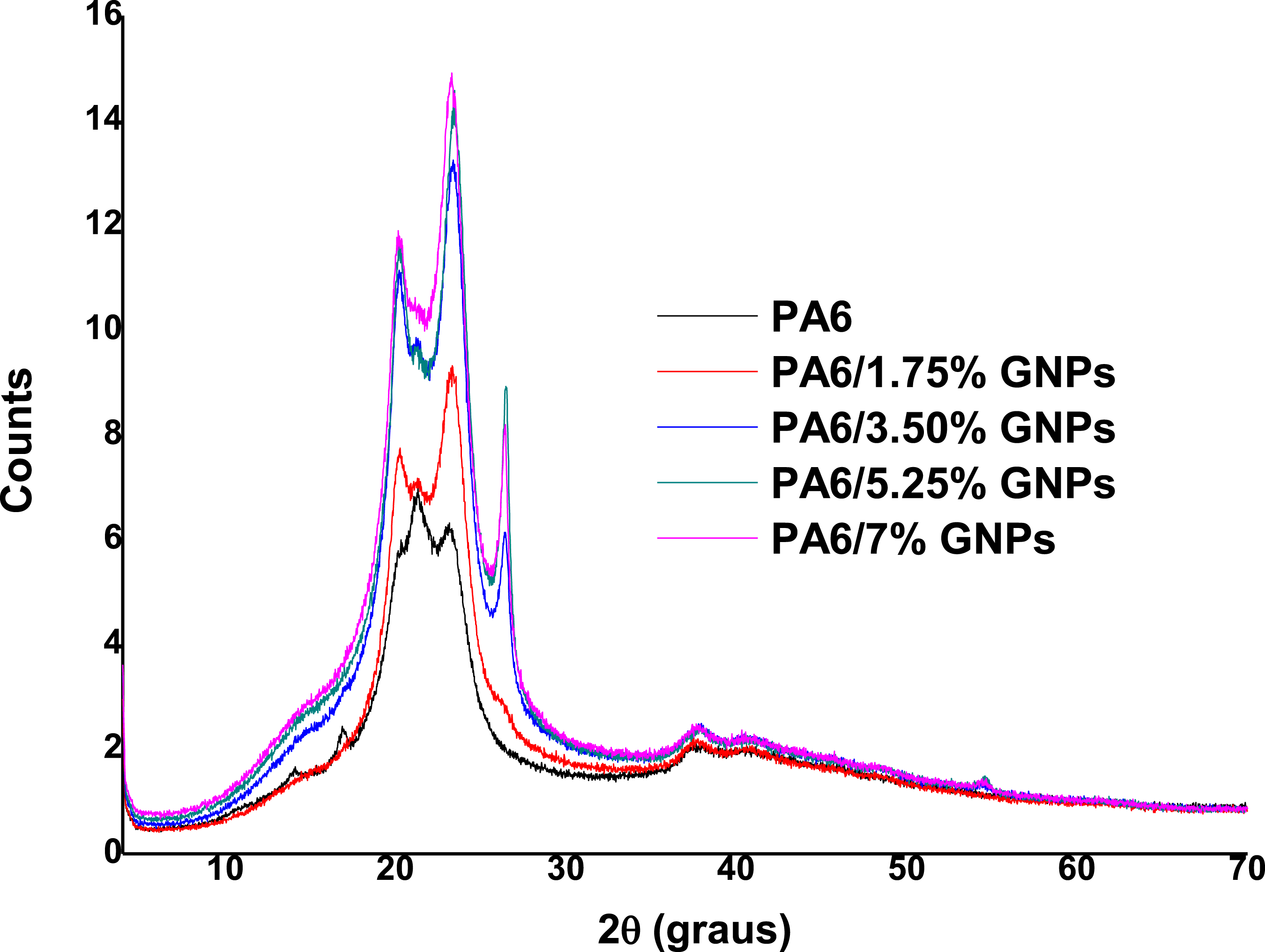
Mechanical Properties
Tensile properties
The development of PA6 composites reinforced with carbon nanomaterials such as GN, GNPs, GO, rGO, CNTs, and CF has received considerable attention due to the significant improvements in mechanical properties, especially in strength and tensile modulus. The combination of the high stiffness of these reinforcements and the control of their dispersion in the polymer matrix are decisive for the improvements obtained.6,7,75
Damacena et al. 35 reported that surface modification of CF with GO led to an approximately 27% increase in tensile strength and a 30% increase in tensile modulus in PA6/CF composites, attributed to improved interfacial adhesion between the matrix and the reinforcement. Similar results were observed by Cheng et al., 76 who investigated PA6 composites containing GO-modified carbon fibers (GO-KCF). The tensile strength reached approximately 128 MPa, representing a 21% increase compared to the unmodified PA6/KCF composite. The effect of adding GNPs has also been investigated. 77 The incorporation of 3.0 wt% GNPs into PA6 increased the tensile strength from 62 MPa (pure PA6) to 82 MPa, corresponding to a gain of approximately 32%. The tensile modulus also showed a significant increase, from approximately 1.6 GPa–2.2 GPa. The incorporation of particularly multi-walled carbon nanotubes (MWCNTs)78,79 into PA6 been highlighted for their potential for significant improvements in modulus and tensile strength when good dispersion, optimized interface, functionalization, or surface treatment are achieved. PA6 nanocomposites reinforced functionalized carbon nanotubes (CNTs) and with a treated interface, can improve tensile strength in ranges similar to those of GNPs in some cases, but excellent modulus gains are often reported, sometimes exceeding those obtained with GNPs. 78 Furthermore, the hybrid incorporation of micro- and nanoscale reinforcements has shown synergistic effects. Siddikali et al. 79 studied additively manufactured PA6 composites containing CF (15.0 wt%) and MWCNTs (0.5 wt%). The authors observed an increase of approximately 45% in tensile strength and 38% in modulus, demonstrating the potential of hybrid reinforcement. In another investigation, Dong et al. 80 evaluated different types and contents of CF in PA6 composites, achieving tensile strengths of up to 210 MPa and tensile modulus exceeding 12 GPa, values much higher than those of pure PA6 (62 MPa and 1.6 GPa, respectively). In summary, the results confirm that the addition of GN and CF promotes consistent increases in the tensile properties of PA6 composites. Critical factors include the optimal reinforcement concentration, the type of surface modification employed, and the synergy between fillers of different scales.
The results show that the addition of GNPs into PA6 results in a higher Young´s modulus in comparison to pure PA6 (Figure 15). A filler content of 5.25 wt% leads to an improvement of Young´s modulus by 22% compared to pure PA6. But an increase of the filler content causes a decrease of Young´s modulus, due to possible agglomeration of graphene, which could reduce the reinforcing effect of the graphene and therefore Young´s modulus does not further increase with increasing filler content. Russo et al.
77
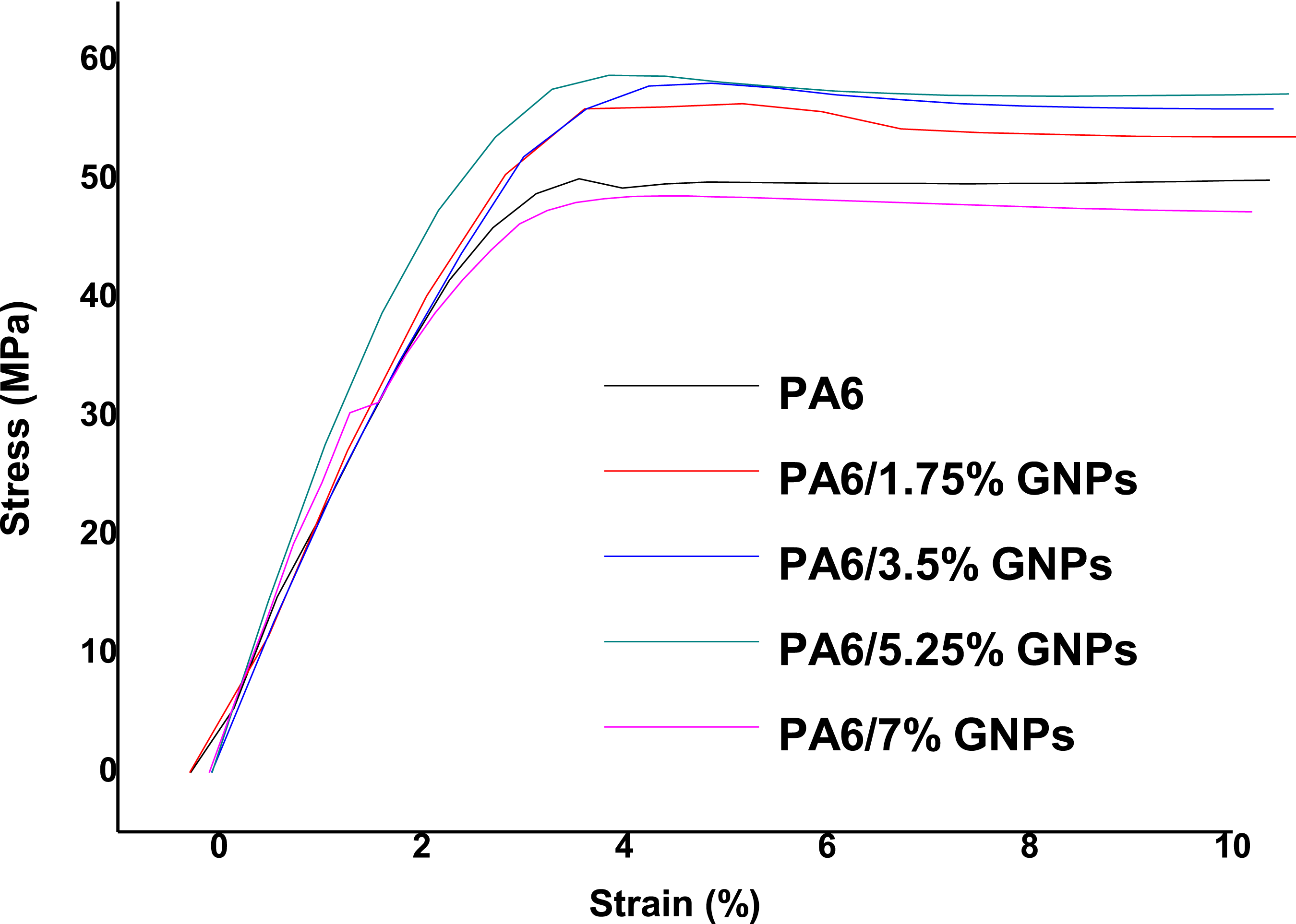
found an increase in the elastic modulus of PA6/GNPs composites with increasing graphene content. The results can be explained by considering that the modulus, among other factors, depends on the stiffness of the load and the crystallinity of the matrix used in the composite. The stress strain curves from the tensile tests of the composites are shown in Figure 16. As seen from Figure 16, all composites failure in brittle manner. Young´s modulus of PA6 and PA6 composites. Stress-strain curves from the tensile tests of PA6 and PA6/GNPs composites.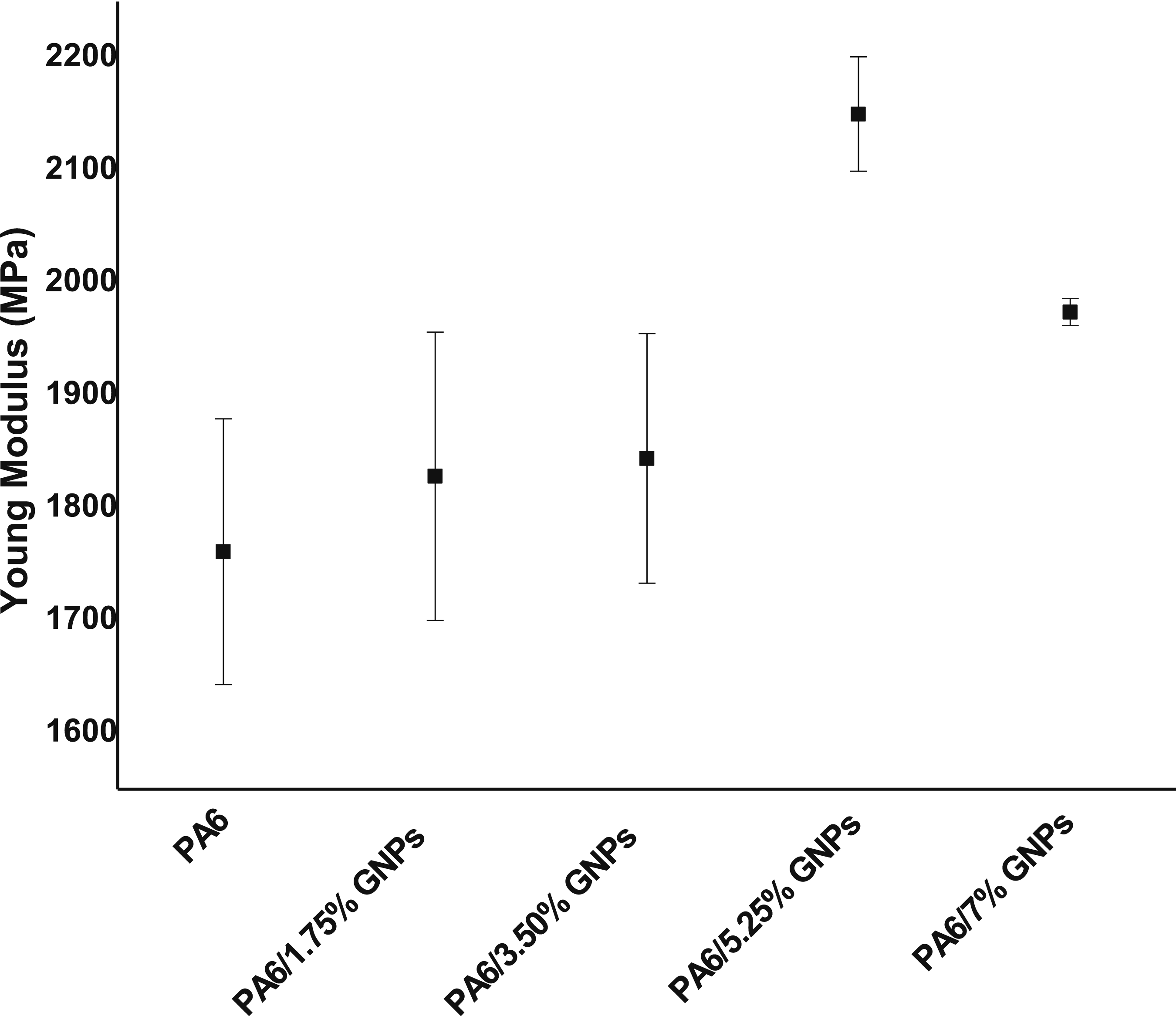
From the one-way ANOVA statistical analysis of the results of the elastic modulus of the composites, it was possible to conclude that only PA6/5.25% GNPs sample presents a significant difference from the other samples. This is an indirect indication of the great restriction on the mobility of polymer chains imposed by graphene. Up to 3.50 wt% of GNPs there seems to be no significant changes in relation to pure PA6, while a GNPs content above 5.25 wt% introduces a deleterious effect on the stiffness of the material.
The addition of the nanofillers causes an increase in tensile strength (Figure 17). The use of GNPs tends to higher tensile strengths compared with pure PA6, because load is transferred from matrix to the filler via high specific surface of the filler. At a filler content of 5.25 wt% the tensile strength is improved from 53.4 MPa (pure PA6) to 58.5 MPa (PA6/5.25% GNPs) on average, which is an increase of 10%. Russo et al.
77
found results that show that in samples with concentrations above 3.0 wt% of GNPs, maximum stress tends to be lower than in samples with lower concentrations. In this context, it is difficult to predict the trend of maximum stress, since it depends not only on the crystallinity of the matrix and the size of the aggregates of the dispersed phase but is also influenced by the polymer-filler interaction.
81
Tensile strength of PA6 and PA6 composites.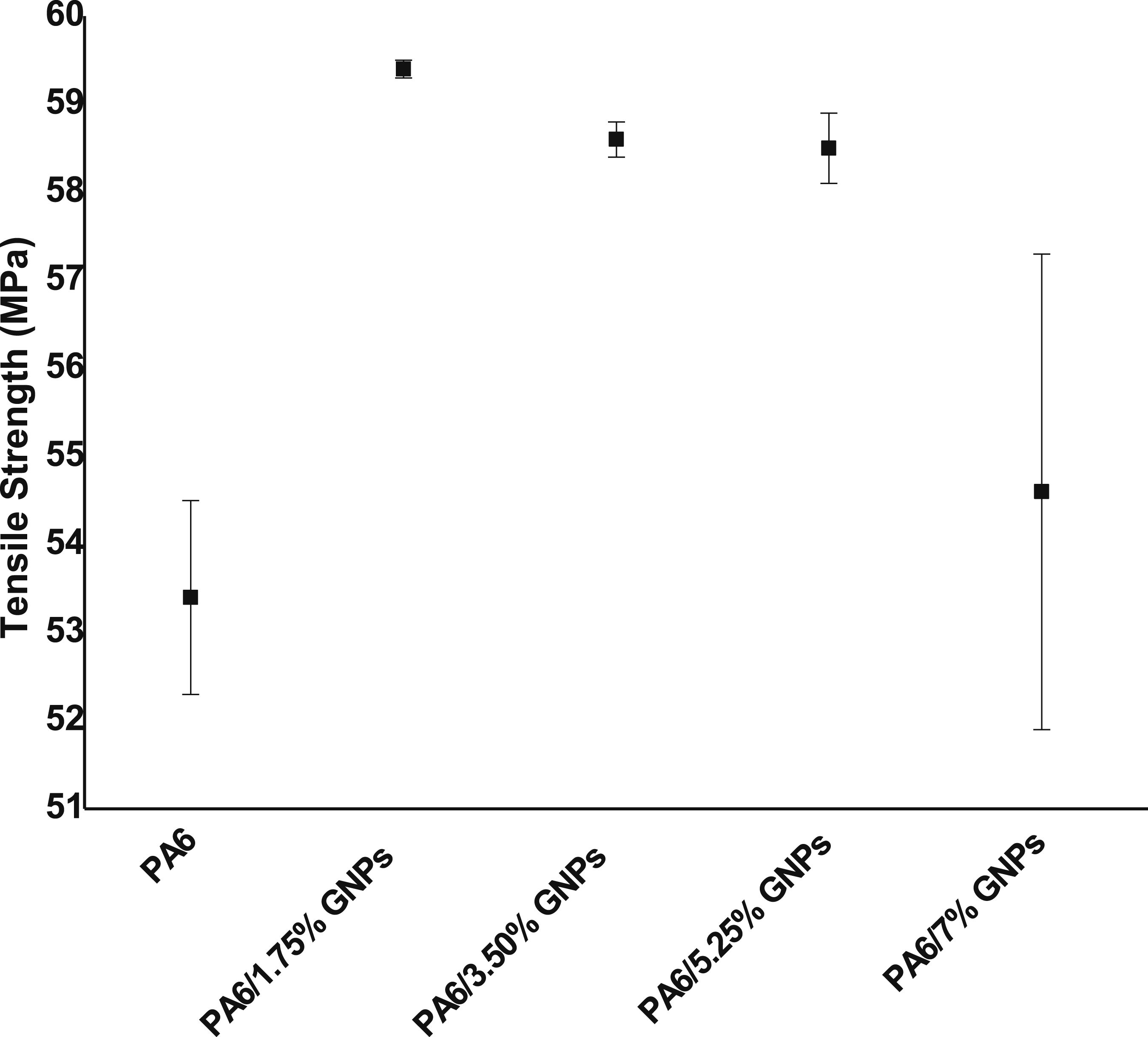
From the one-way ANOVA statistical analysis of the tensile strength results, it is possible to conclude that the PA6/1.75% GNPs sample differs significantly from the pure PA6 sample and PA6/7% GNPs sample. Other samples are equivalent from a statistical point of view and, therefore, a desirable improvement in tensile strength is only achieved up to a maximum of 1.75 wt% GNPs.
Flexural properties
In recent years, various studies have highlighted significant advances in the flexural properties of PA6 composites reinforced with carbon-based fillers such as GN, GNPs, GO, rGO, CF, and CNTs. Flexural performance has been shown to be highly dependent on the type of reinforcement, the concentration used, and the quality of the matrix-reinforcement interface.
Damacena et al. 35 investigated PA6 composites with SCF coated with GO. Their study improved flexural strength and modulus (flexural strength-22%, modulus-25%), compared to unmodified PA6/SCF composites, evidencing the positive role of surface modification in interfacial adhesion. Siddikali et al. 79 evaluated PA6 hybrid composites produced by 3D printing, containing 15.0 wt% CFs and 0.5 wt% MWCNTs. The hybrid reinforcement promoted an increase of approximately 42% in flexural strength compared to pure PA6, indicating a synergistic effect between micro- and nanometric reinforcements. Cheng et al. 76 analyzed PA6 composites containing GO-KCF. The materials showed flexural strength of approximately 180 MPa, representing an increase of 38.5% compared to unmodified PA6/CF composites. Studies with PA6/GNPs composites observed a significant increase in flexural modulus: from approximately 1996 MPa (pure PA6) to 3482 MPa (PA6/3.0 wt% GNPs), representing a 75% gain. Flexural strength also increased from 46 MPa (pure PA6) to 61 MPa (PA6/3.0 wt% GNPs), equivalent to a 33% increase. However, higher concentrations (5.0 wt% GNPs) showed a decrease, suggesting an optimal dispersion threshold. 77 Another study showed that PA6/CF composites, depending on the fiber type and content, achieved flexural strength values of approximately 224 MPa, an increase of approximately 230% compared to the pure polymer. The flexural modulus reached 14.6 GPa, representing an increase of over 600%. 80 Overall, the results confirm that the choice of reinforcement type (nano- or microstructural), concentration, and surface modification are determining factors in optimizing the flexural performance of PA6 composites.
In Figure 18, it is possible to verify that the elastic modulus in flexural increased with the increase in the graphene concentration. Unlike the results found in the tensile properties analyzed previously, the nanoplatelet content can be extended up to 7.0 wt%, which generated an increase of 34% in relation to the PA6 sample without graphene. Since graphene is a very rigid material, its incorporation into the polymer matrix provides a reinforcing load effect, which translates into an increase in the materials rigidity. Flexural modulus of PA6 and PA6 composites.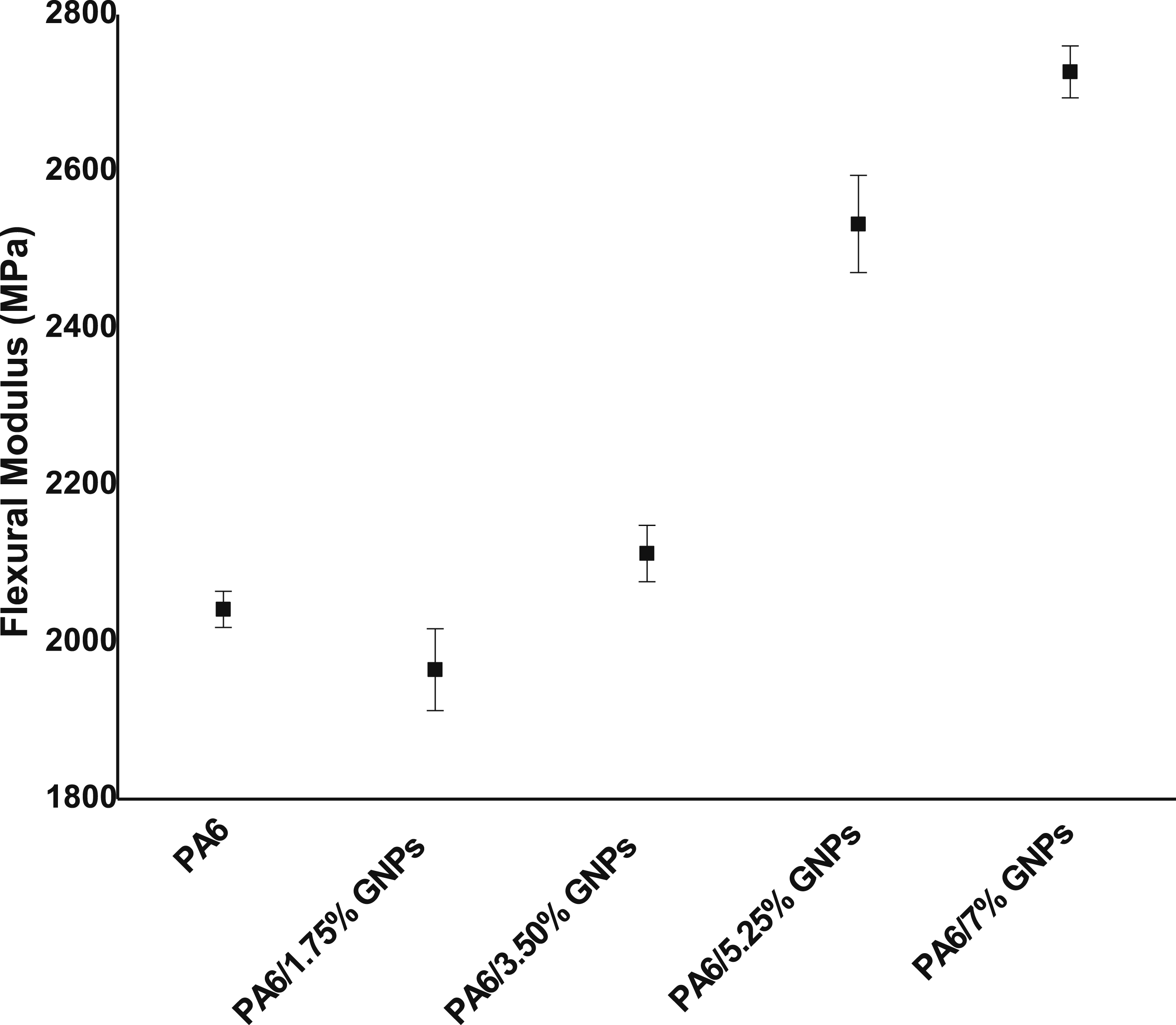
According to the statistical analysis the results of the elastic modulus in flexural of the materials, it was possible to conclude that all the samples differ from each other. Therefore, if an improvement in the elastic modulus in flexural is desirable, the graphene content can be extended up to 7.0 wt%. The stress strain curves from the flexural tests of the composites are shown in Figure 19. Stress-strain curves form flexural tests of PA6 and PA6/GNPs composites.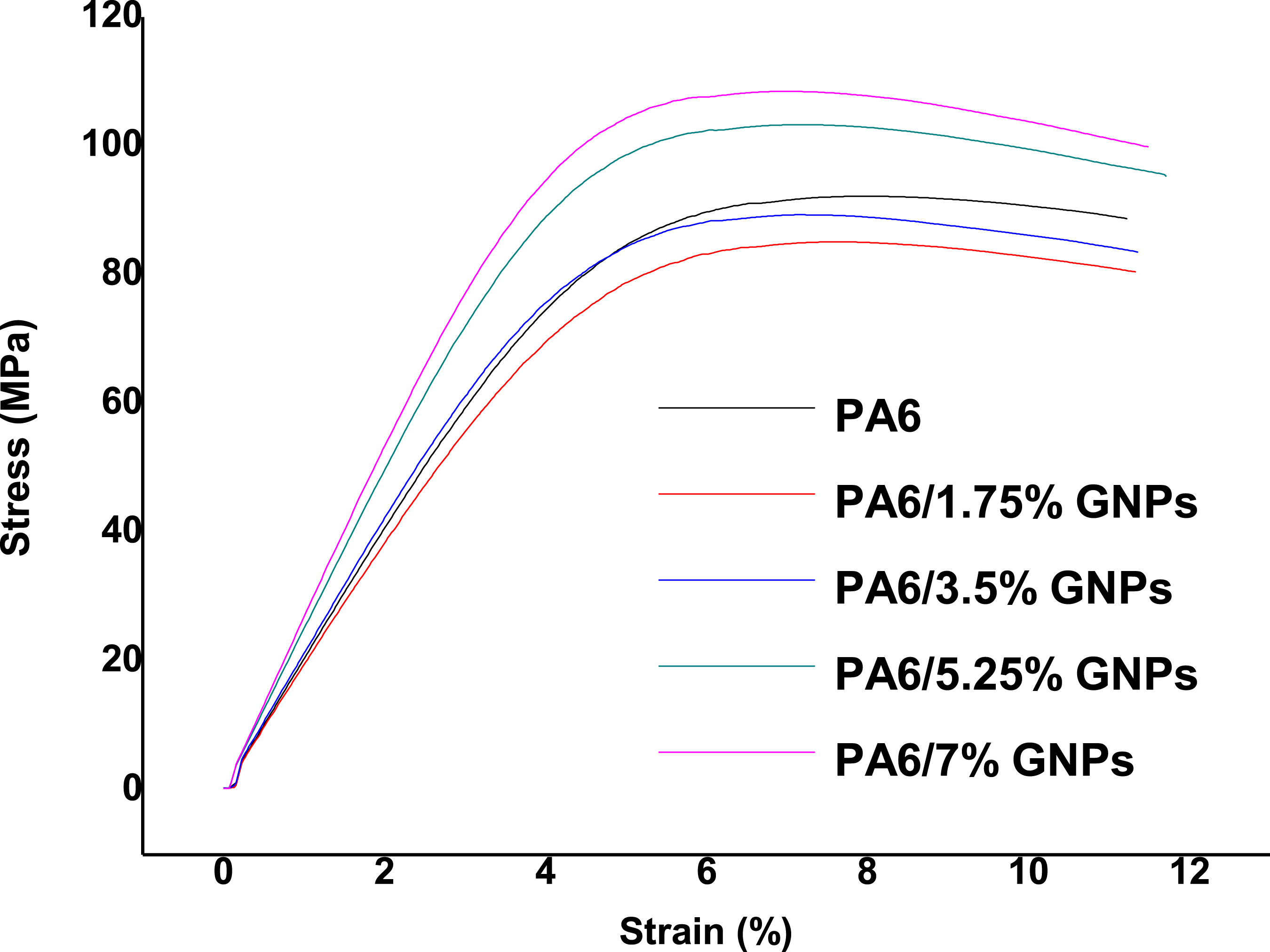
It was observed that the composites containing 1.75 wt% and 3.50 wt% of GNPs showed a slight decrease, 2.5% and 8%, in flexural strength when compared to the pure PA6 sample (Figure 20). In turn, the composites containing 5.25 wt% and 7.0 wt% of GNPs showed flexural strength values 12% and 17% higher, compared to the composite without the incorporation of GNPs. Flexural strength of PA6 and PA6 composites.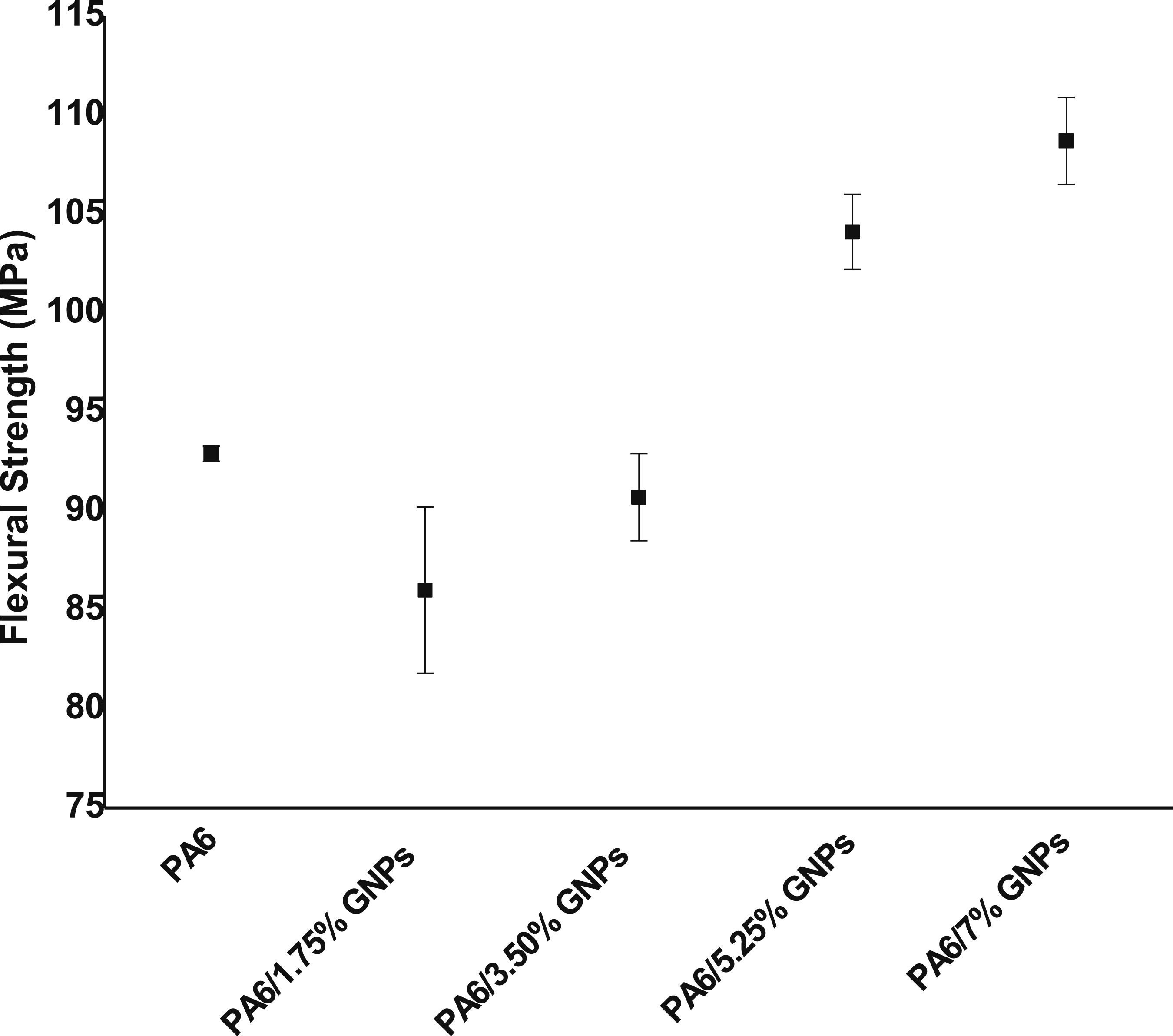
From the one-way ANOVA statistical analysis of the flexural strength results of the composites, it was possible to conclude that the flexural strength only has a real increase when the GNPs content reaches at least 5.25 wt%. Lower filler contents do not satisfactorily alter the property in relation to the pure polymer.
For Russo et al., 77 the flexural strength of the composites is always greater than that of pure PA6. This parameter follows a trend like that observed for tensile strength: it maintains an almost constant value with 1.0 wt% load, increases by approximately 30% for the composite with 3.0 wt% GNPs; and, then, there is a reduction in this parameter with an increase in load incorporation. 81
Impact strength
PA6 is widely used when good mechanical strength, wear resistance, and moderate toughness are required. The incorporation of carbon-based fillers, GN, GO, rGO, MWCNTs, and CF, has been investigated to improve the mechanical, electrical, and thermal properties of PA6 composites. However, the effect of these fillers on impact properties is complex and strongly depends on: (i) the type and functionalization of the filler; (ii) the morphology and dispersion in the matrix; (iii) the processing method; and (iv) the filler content. Recent reviews have observed both improvements and deteriorations in impact toughness, depending on these factors.14,82,83
MWCNTs increase stiffness and tensile strength at low contents, but their effect on impact toughness is divergent. When homogeneously dispersed and functionalized to improve matrix-filler adhesion, they can increase impact strength through nanotube pull-out mechanisms and crack deflection dissipation. However, CNT aggregates act as fracture initiators and often reduce impact energy if dispersion is insufficient. Therefore, processing optimization (melt-mixing with compatibilizers, chemical treatments) is crucial.6–8,75,78,79 GN and its variations show increased modulus and static strength at low concentrations; however, multiple studies document a reduction in unnotched impact strength in the presence of untreated GN or GO, attributed to increased stiffness and the formation of stress concentrations that favor brittle fracture. In contrast, functionalizations (e.g. compatibilizing groups, H-bonding interactions with PA6) and the use of well-exfoliated GNPs or hybrids (GNPs/flexibilizing polymer) mitigate the loss of toughness and, in some cases, recover or even slightly improve the energy absorbed.14,16,76,77 CFs significantly increase flexural strength and impact resistance, especially when the fiber/matrix interface is treated (silane, GO coatings). However, composites with a high SCF content can reduce the plastic deformation capacity of the assembly, reducing the energy absorbed during impacts. Surface treatment of fibers with GO or nanotubes shows synergistic effects, improving load transfer and impact resistance when well bonded.9,35,76,80 Comparing filler types: CNTs tend to favor dynamic properties when dispersion and interface are excellent; GN/GNP/rGO increase modulus but often penalize unnotched impact toughness without toughening strategies; CF improves strength in notched tests but can reduce energy absorption in high plastic deformation impacts if the matrix becomes excessively brittle. Many recent studies present contradictory results due to differences in processing, filler nature, mass content, and test protocols (Izod vs Charpy, notched vs unnotched), which reinforces the need to compare studies with standardized protocols.
The impact strength is reduced due to the adding of GNPs (Figure 21). There are several causes that can influence the impact resistance of composites, such as: decoupling, deformation, and fracture of loads. Poor adhesion between the filler and matrix can generate crack propagation at the filler-matrix interface. On the other hand, strong adhesion can cause restriction of the mobility of the polymer matrix, making its deformation difficult and, consequently, its tenacity when subjected to sudden tensions, making them brittle.14,82,83 Impact strength of PA6 and PA6 composites.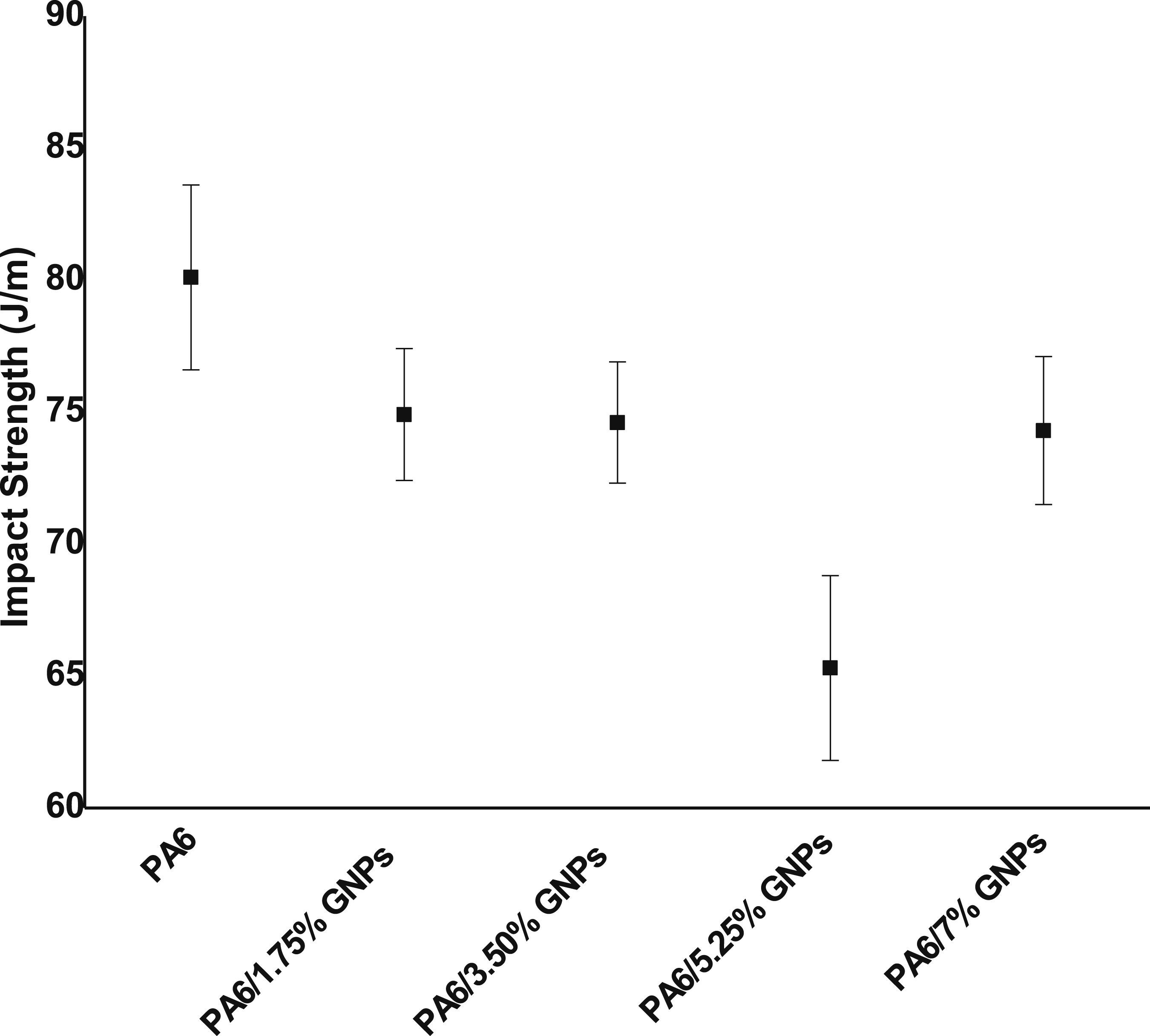
The composite containing 5.25 wt% of GNPs showed a significant decrease of 22.5% compared to the thermoplastic without the incorporation of GNPs. This result may be related to poor adhesion between GNPs and PA6, which facilitated the propagation of cracks at the filler-matrix interface and hindered energy dissipation, resulting in a composite more susceptible to breakage. 83 The remaining samples taken two by two were equivalent for the 95% confidence interval used.
Summarizing, the addition graphene results in higher Young´s modulus, tensile and flexural strength. The reinforcing effect is lower compared to the abovementioned publications. This may occur because the degree of graphene dispersion was low.
Conclusion
In this study, the effect of incorporating GNPs into a PA6 matrix on the mechanical and thermal properties of the polymer was evaluated, and the following results were obtained. (1) The incorporation of GNPs contributes to a reduction of the crystallinity of PA6 probably due to the introductions of defects in the crystal lattice and the restriction of the mobility of the polymer chains that prevent the formation of strong and organized crystals. (2) The adding of GNPs to PA6 leads to an improvement of Young´s modulus, tensile and flexural strength, with a loss of impact strength. (3) Given the need to obtain a material that presents mechanical resistance, lightness and ease of processing, samples with up to 5.25 wt% of GNPs presented better results. (4) The characterization results of GNPs suggest that the graphene used has a low number of stacked layers and defects, which classifies it as a better-quality material.
The incorporation of graphene into the PA6 matrix leads to the production of materials with superior mechanical properties. Despite their lower toughness, the resulting materials exhibit high rigidity and potential for automotive applications, including connectors and interior parts (greater fatigue resistance), fuse boxes (greater wear resistance), electronic component mounts (improved rigidity and better heat dissipation), motor amplifiers (thermal resistance and reduced deformation), and electrical and electronic system components (less static charge buildup). These results highlight graphene as a promising material for the development of innovative composites with enhanced performance and functionality.
Footnotes
Acknowledgements
The authors would like to thank the funding agencies FAPERJ and CNPq, and the Research Support Program of the State University of Rio de Janeiro (PAPD Pesquisa), for their financial support for this work. This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior - Brasil (CAPES) - Finance Code 001. The authors would like to thank the Laboratory of the National Institute of Metrology, Quality and Technology (INMETRO) for Raman spectroscopy and BET method tests, the Biomaterials Laboratory of the Polytechnic Institute of the Rio de Janeiro State University for Fourier transform infrared spectroscopy (FTIR) test support, the materials Laboratory of the Rio de Janeiro State University for thermal test, the Multiuser Laboratory of Nanofabrication and Characterization of Nanomaterials (NANOFAB) for assistance in scanning electron spectroscopy (FEM-SEM) tests, the Multiuser Laboratory of Technological Characterization (LMCT) of the Mineral Technology Center (CETEM) for assistance in X-ray diffraction (XRD) tests. DSc. Marisa Cristina Guimarães Rocha would like to thank CNPq for the financial support from process no 401982/2023-8.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
