Abstract
Poly(methyl methacrylate) (PMMA)/poly(ethylene oxide) (PEO) (90/10) nanocomposites containing various amounts of graphene nanoplatelets were fabricated by solution method and then the effects of graphene concentration on morphology, thermal, mechanical, and electrical properties of the nanocomposites were investigated. Characterization by electron microscopy and X-ray diffraction of the nanocomposites showed a relatively good dispersion of graphene sheets in the polymer matrix. The results indicated that thermal stability, glass transition temperature, and mechanical properties of PMME/PEO blend improved by increasing graphene concentration. The electrical properties of polymer nanocomposites revealed a significant improvement with increasing the amount of graphene and the percolation threshold was about 3.33 wt% of graphene.
Keywords
Introduction
Graphene, as a monolayer of carbon atoms arranged in a honeycomb network, has recently attracted tremendous attention in the fields of fundamental and applied due to its outstanding physical, chemical, and mechanical properties. 1 –5 The discovery of new nanomaterial graphene by Andre Geim 6 in 2004 with its unique properties has created a new class of polymer nanocomposites and expanded the application of polymers. 7 Polymers have an important role in the industrial applications, but the increasing demand of materials for advanced applications cannot be provided by simple homopolymers. Because of that, there are considerable scientific and industrial interests in modifying or mixing known polymer materials. Polymer blending is a very attractive, versatility, simplicity, and economical method for obtaining new polymeric materials with desirable properties. Various research groups prepared polymer blends and reinforcing materials filled polymer blends. 8 –10 Scaffaro et al. 11 prepared polyamide 6/high density polyethylene/clay nanocomposites and showed that the incorporation of nanoclay into polymer blends improve the mechanical properties. The polymer blend system can be used for preparation of polymer electrolyte by the addition of liquid electrolyte to polymer matrix. 12,13 Poly(methyl methacrylate) (PMMA)/poly(ethylene oxide) (PEO) blend has received substantial attention as high performance polymer in recent years. This is due to its interesting properties, such as good processability and optical properties, as well as by its wide applications. 14 –16 However, as an attractive candidate for further applications, PMMA/PEO blend suffers from inadequate electrical, thermal, and mechanical properties. 15 The low price and outstanding properties of graphene nanoplatelet (GnP) make it a potential choice as a reinforcement filler in the preparation of polymer nanocomposites as compared to other nanofillers such as layered silicate clay and carbon nanotubes. GnPs combine the layer structure of clays with excellent thermal, electrical, and mechanical properties of carbon nanotubes. Pinto et al. 17 have investigated the application of graphene as a reinforcement filler in poly(lactic acid) film. The maximum improvement in the final properties of graphene/polymer nanocomposites will be achieved with well-dispersed structure of graphene in the polymer matrix. 4,17 However, since the GnPs have a strong tendency to agglomerate because of the attractive van der Waals force between them, obtaining a good dispersion is a challenging task. In recent years, solution mixing method has been widely used as an effective technique for fine dispersion of nanofillers. 18 One of the main advantages of the solution mixing is low viscosity of the medium that allows good dispersion of graphene platelets in the polymer matrix. According to the previous studies, modification of graphene decreases the electrical properties as compared to neat graphene due to the presence of functionalized groups on the surface of graphene layers. Functionalization of graphene nanosheets destructs the sp2 structure and leads to the formation of defects and loss of electrical conductivity. 19 Therefore, we use neat graphene in polymer matrix to produce nanocomposites with improved properties. However, to the best of our knowledge, there are little reports about the effect of graphite nanoplatelets on electric performance of form-stable polyethylene glycol/PMMA composite phase change materials, 20 whereas the influences of neat GnP on different properties of PMMA/PEO blend have not been reported so far.
In this effort, as an extension to our previous report 21 published on the effect of oxidized GnP on the final properties of PMMA/PEO blend, we aim to contribute to the discussions surrounding the effect of different pure GnP concentrations on morphological, thermal, mechanical, and electrical properties of PMMA/PEO blend for the first time here. With this aim in view, the PMMA/PEO blend and its nanocomposites with neat GnPs were fabricated via solution blending method and analyzed by X-ray diffraction (XRD), differential scanning calorimetry (DSC), resistivity meter, and tensile test. In order to achieve optimal properties in nanocomposites, the weight percentage of graphene sheets was changed. What makes this contribution distinguished from previously published, similar ones is that the former papers used functionalized graphene rather than neat graphene to investigate the properties to attain a better dispersion, while we intend to explore the influence of neat GnP on PMMA/PEO blend.
Experimental
Materials
PEO (
Preparation of PMMA/PEO blend and its nanocomposites with GnP
PMMA/PEO blend and PMMA/PEO-based nanocomposites with various GnP loadings were prepared via solution casting method, using DMF as the solvent for polymers and dispersing media for GnPs. To synthesize PMMA/PEO blend, the required quantities of PMMA/PEO (90/10) were dissolved in DMF solvent, followed by stirring for 5 h at 50°C. After solution casting onto glass petri dish, the films were dried at room temperature for about 1 day and then in vacuum at 50°C for 48 h. To prepare PMMA/PEO/GnP nanocomposites, GnP was dispersed in DMF by magnetic stirring overnight and after ultrasonicating for 3 h, it was sonicated for 5 min to obtain fine dispersion. The PMMA/PEO (90/10) solution was prepared and then added into the graphene dispersion. The mixture was stirred for 5 h at 50°C. Finally, this homogeneous solution was cast onto glass petri dish and dried at room temperature for 24 h. The prepared nanocomposites were further dried at 50°C under vacuum for 48 h to remove the residual solvent completely. The obtained nanocomposites were labeled as GnP-0.3, GnP-0.8, GnP-1.5, GnP-3, GnP-5, GnP-7, and GnP-8, including 0.3, 0.8, 1.5, 3, 5, 7, and 8 wt% of GnP. According to the previous reports, various levels of graphene (from low to high) were selected to show the effects of neat GnP content on different properties of PMMA/PEO blend and to obtain the electrical percolation and optimum filler concentration for these series of nanocomposites. The PMMA/PEO blend and its composites were molded into sheets of 1 mm in thickness by a hot press at 140°C. The sheets were used for further characterization.
Characterization
The XRD analysis was performed using a Simens D5000 (Germany) X-ray diffractometer with CuK radiation at a generator voltage of 40 kV and a generator current of 40 mA. XRD scans were recorded from 2
Results and discussion
XRD analysis
Figure 1 shows the XRD patterns of pure GnP, neat PMMA/PEO, and PMMA/PEO nanocomposites with various amounts of GnP. The characteristic XRD peak of pure GnP sheets is located at 2
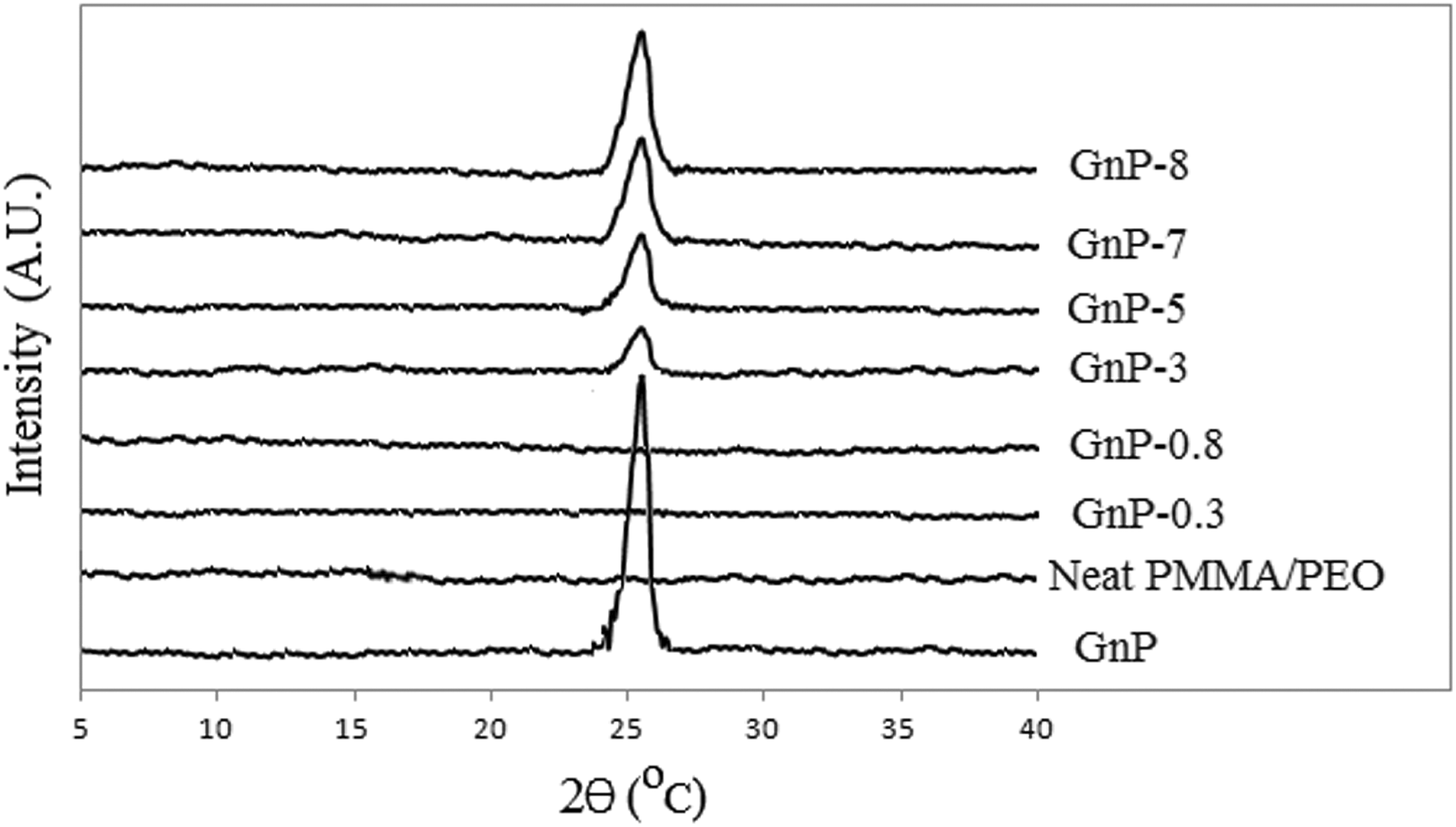
XRD patterns of GnP, neat PMMA/PEO, and PMMA/PEO/GnP nanocomposites. GnP: graphene nanoplatelet; PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide); XRD: X-ray diffraction.
The neat PMMA/PEO did not show any clear peak in XRD, revealing its amorphous structure. The XRD diffraction patterns of pure PMMA/PEO and its nanocomposites (up to 0.8 wt% of GnP) are greatly similar. The disappearance of GnP peak suggests the formation of exfoliated structures and the homogeneous distribution of GnP sheets in the polymer matrix.
22,23
In contrast, the profiles of the nanocomposites with 1.5, 3, 5, 7, and 8 wt% GnP contents show the diffraction peak of GnP at 2
Thermogravimetric analysis
Thermal stability is an important factor in processability of the polymeric materials. Graphene is one of the most effective fillers, which can significantly improve the thermal stability of polymers. Thermogravimetric profiles of the neat PMMA/PEO and its nanocomposites containing 0.8 and 3 wt% of GnP are displayed in Figure 2.
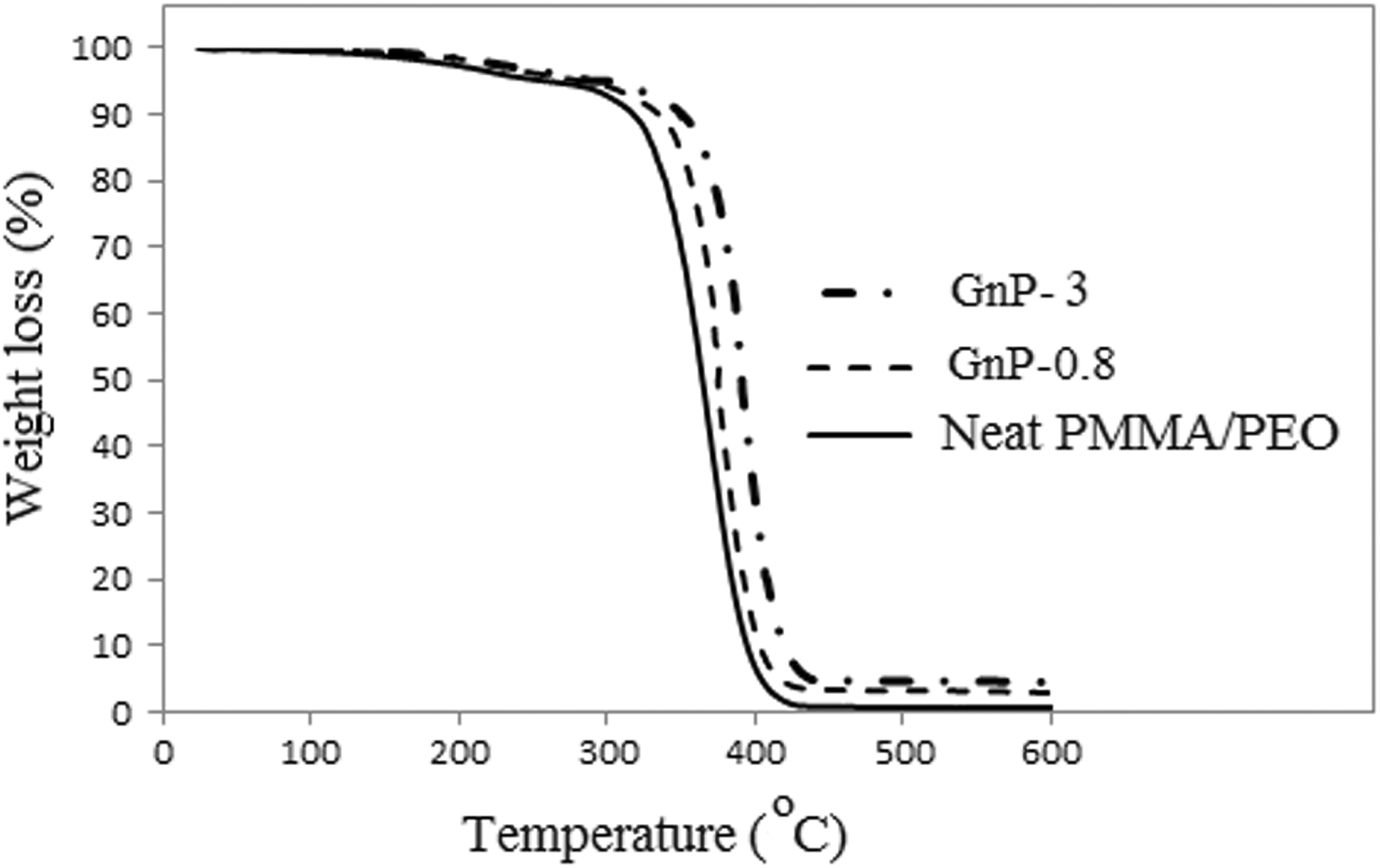
TGA thermograms of neat PMMA/PEO, PMMA/PEO/GnP-0.8, and PMMA/PEO/GnP-3 nanocomposites. GnP: graphene nanoplatelet; PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide); TGA: thermogravimetric analysis.
Two steps of degradation can be distinguished in the thermograms of all the samples. According to the previous reports,
24
the first weight loss step is corresponded to the monomer evolution initiated at unsaturated chain ends and the second step is related to the decomposition of PEO and PMMA.
25
It is obvious from Figure 2 that thermal stability of the nanocomposites is improved as compared to the neat PMMA/PEO, which is related to the hindering effect of GnP layers on the diffusion of small gaseous molecules throughout the composite material.
26
–28
For an in-depth analysis, the detailed thermal analysis data, for example, 5% (
Thermal properties of PMMA/PEO nanocomposites.
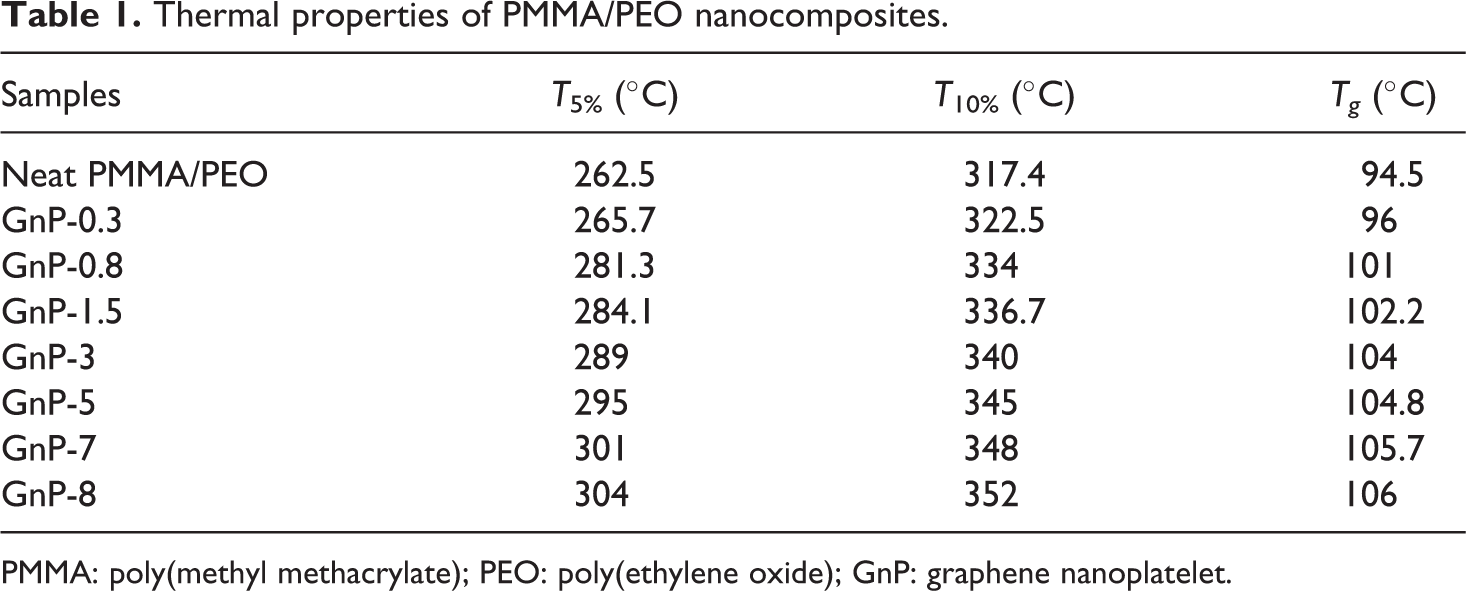
PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide); GnP: graphene nanoplatelet.
DSC analysis
DSC analysis is used to investigate the influences of different GnP contents on the thermal properties of PMMA/PEO/GnP nanocomposites. Figure 3 shows the DSC thermograms of the samples and the related values are presented in Table 1.
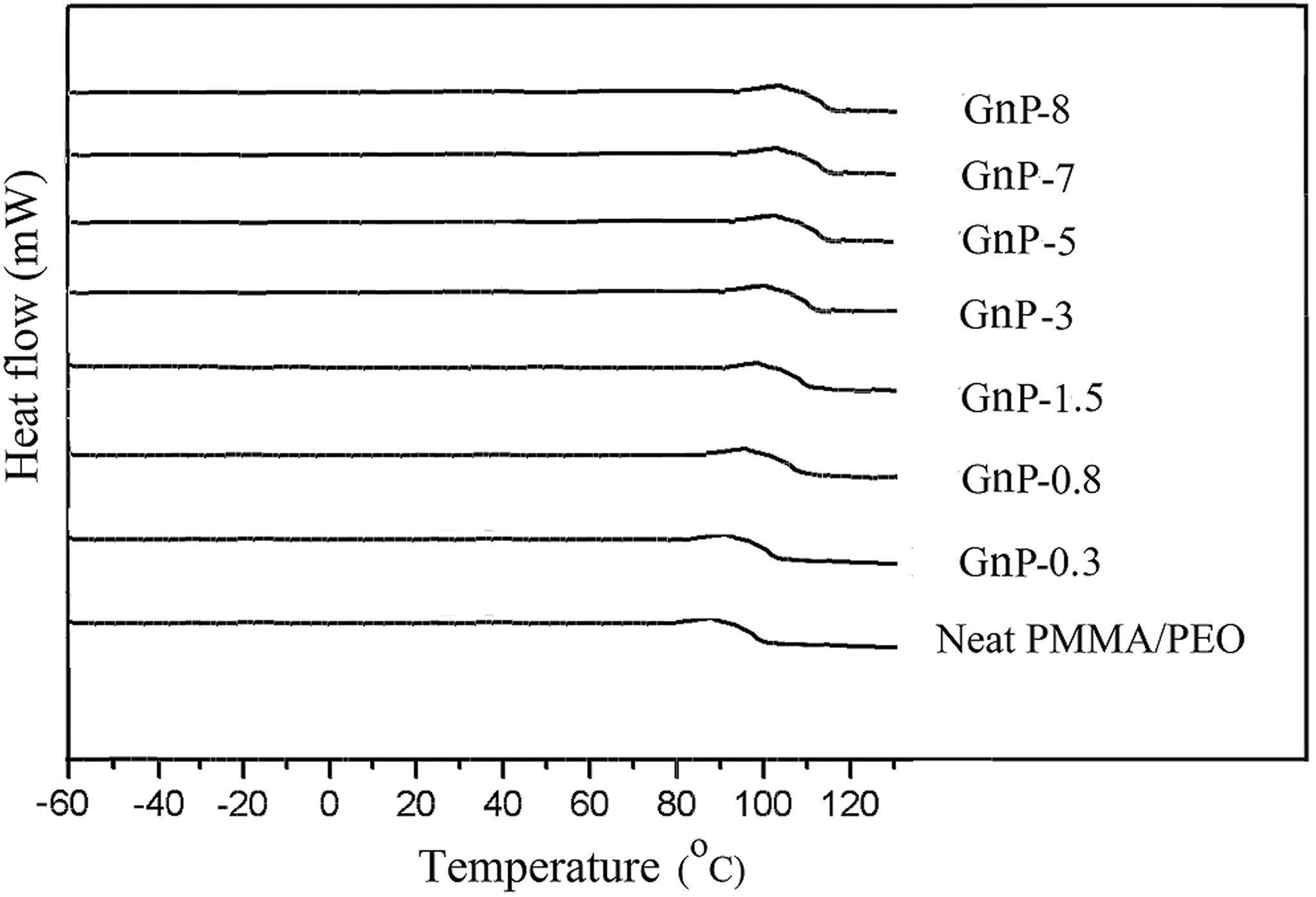
DSC thermograms of neat PMMA/PEO and its nanocomposites. DSC: differential scanning calorimetry; PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide).
The temperature corresponding to the middle point of the temperature range in which the specific heat capacity jumps from the value of glassy state to that of rubbery state is defined as
Mechanical properties
The effect of adding different amounts of GnPs on the mechanical properties of PMMA/PEO composites is shown in Figure 4 and the results are summarized in Table 2.
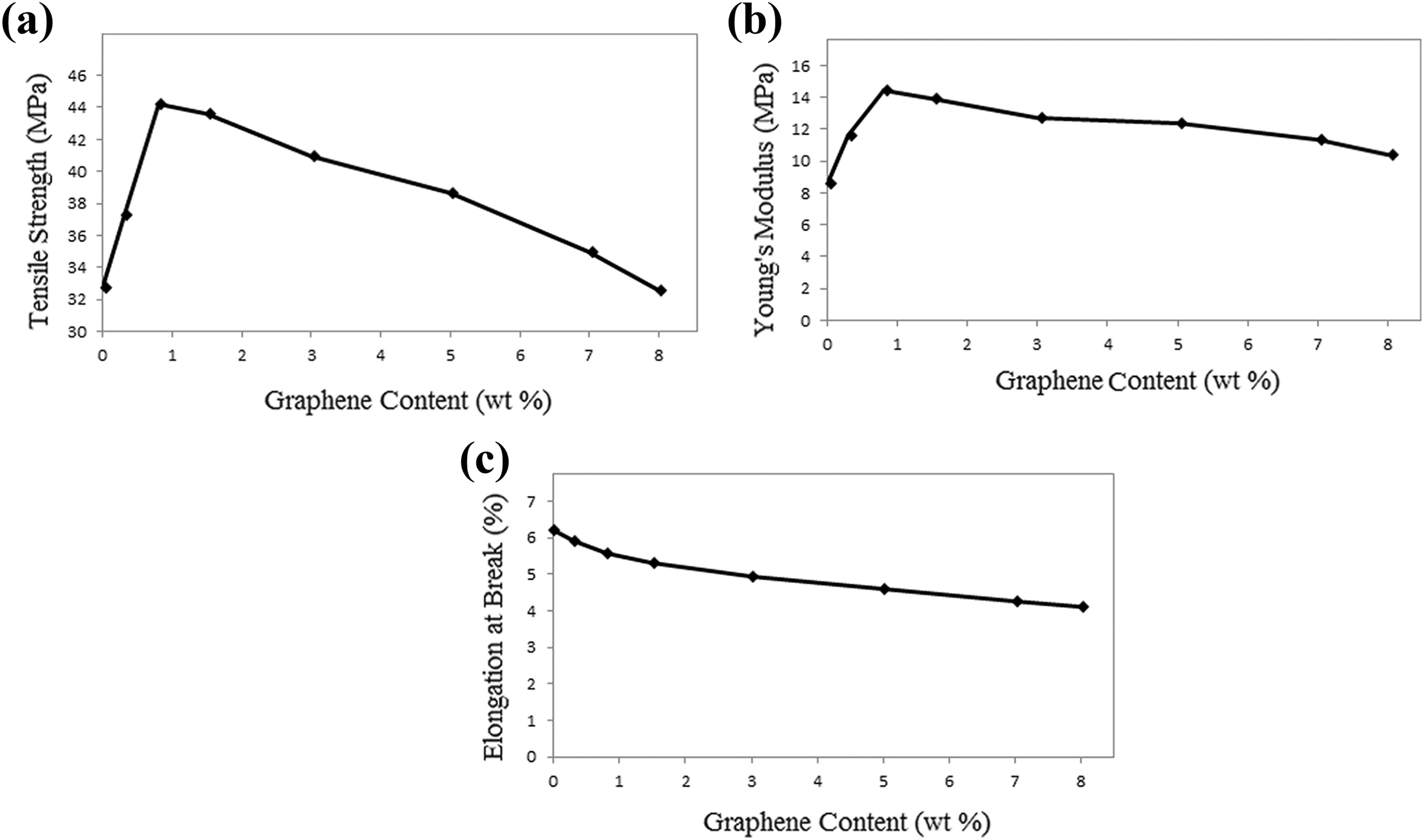
Mechanical properties of pure PMMA/PEO and PMMA/PEO/GnP composites: (a) tensile strength, (b) modulus, and (c) elongation at break. GnP: graphene nanoplatelet; PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide).
Mechanical properties of PMMA/PEO nanocomposites.
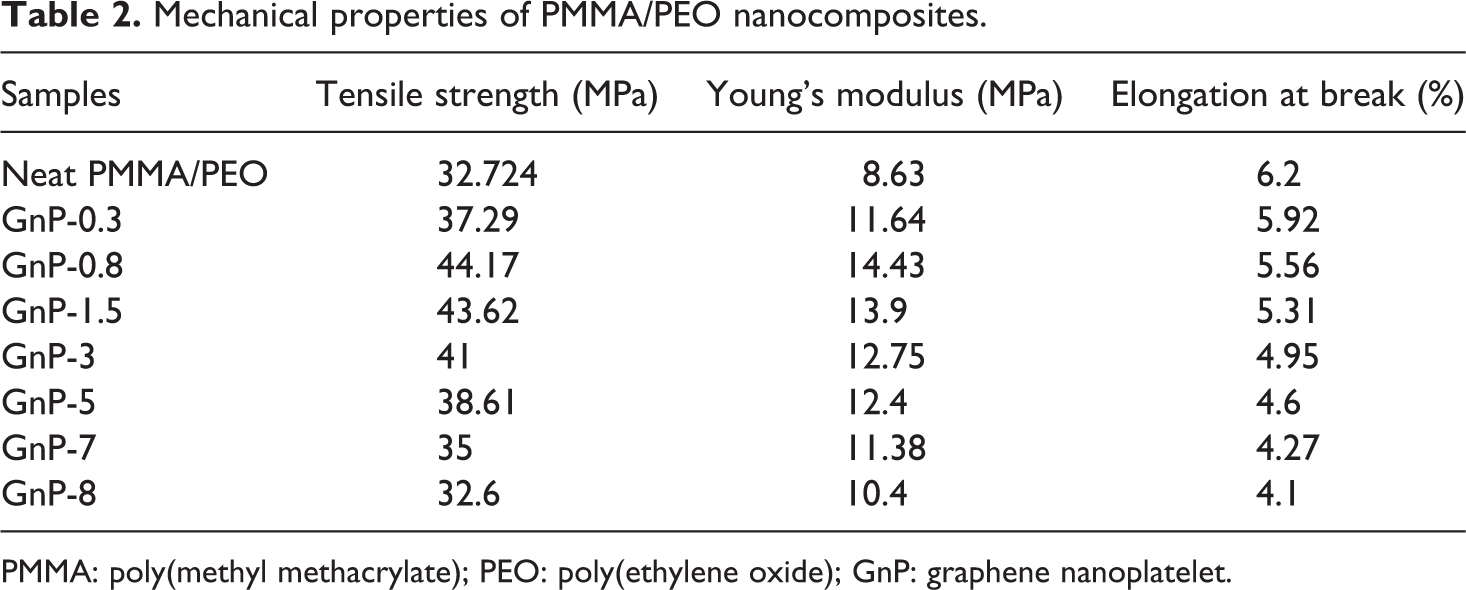
PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide); GnP: graphene nanoplatelet.
The mechanical performance of the PMMA/PEO/GnP nanocomposites is increased as compared to the neat PMMA/PEO. For instance, with the incorporation of 0.8 wt% GnP, the tensile strength increases from 32.72 to 44.17 MPa (Figure 4(a)), and the Young’s modulus increases from 8.63 to 14.43 MPa (Figure 4(b)). This progressive increase in tensile strength and Young’s modulus is a manifestation of the reinforcing ability of graphene in PMMA/PEO matrix. According to the previous studies, the enhancement in the tensile strength and modulus can also be related to a large aspect ratio and lamellar structure of the graphene sheets, which allow better wettability and improve interactions between filler and matrix, thereby leading to efficient stress transfer from matrix to the filler. 30,34 However, a further increase in the GnP concentration (1.5–8 wt%) results in a decrease in tensile strength and Young’s modulus. In fact, the presence of interactions between graphene sheets, mainly by van der Waals force, stacked the sheets together and produced GnP agglomerates. These agglomerates decreased the aspect ratio of the graphene and formed some defects in nanocomposites, which led to the fracture at the interface between PMMA/PEO matrix and GnP nanofillers. This phenomenon was also reported elsewhere. 35,36 It is notable from Figure 4(c) that the addition of graphene into polymer matrix decreases the elongation at break of the nanocomposite samples. This result can be attributed to the presence of graphene within PMMA/PEO matrix, which restricts the mobility of PMMA/PEO chains during extension. 28,35
Electrical properties
It is well known that electrical properties of the polymer composites are considerably dependent on the aspect ratio, dispersion state, concentration, and filler geometry. Compared with the carbon black or carbon fiber as conductive fillers, graphene layers can form the effective conductive pathway in nanocomposites at low volume fraction because they have high aspect ratio. 1,5 Figure 5 shows the electrical conductivity versus filler concentration for the PMMA/PEO/GnP nanocomposites.
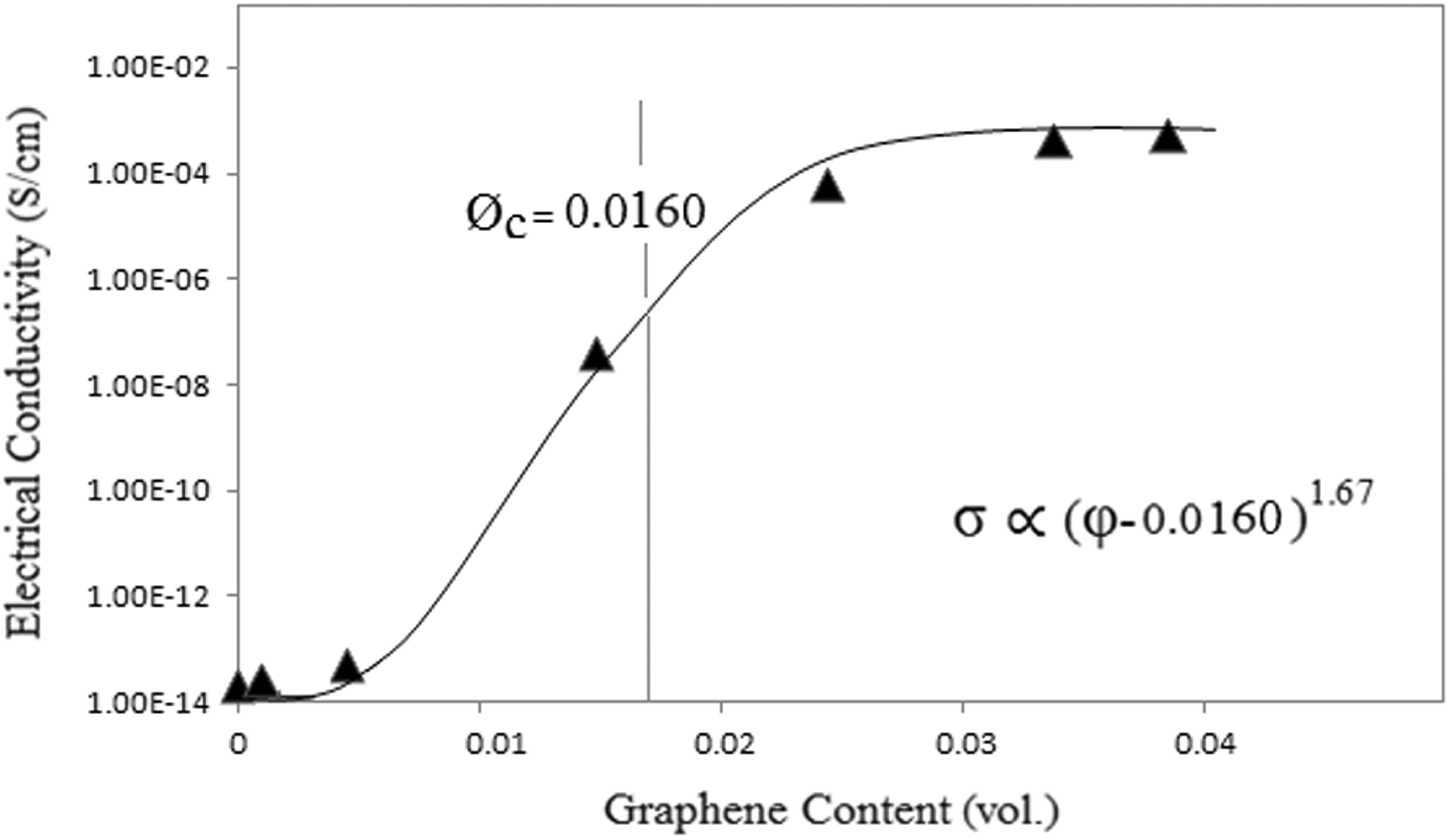
Electrical conductivity of PMMA/PEO/GnP nanocomposites as a function of GnP content. GnP: graphene nanoplatelet; PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide).
It is obvious from Figure 5 that the incorporation of GnP into PMMA/PEO blend significantly enhances the electrical conductivity of PMMA/PEO with a sharp transition from insulating to electrically conducting. For example, as the volume fraction of GnP increases from 0 to 0.0385 vol., the electrical conductivity increases by 10 orders of magnitude from 2 × 10−14 to 8 × 10−4 S cm−1. According to the percolation theory, the electrical conductivity (

where
Morphological analysis
Figure 6 shows SEM micrographs of the fractured surface of PMMA/PEO blend and its nanocomposites containing 0.8, 1.5, and 3 wt% GnP.

SEM images of (a) neat PMMA/PEO, (b) PMMA/PEO/GnP-0.8, (c) PMMA/PEO/GnP-1.5, and (d) PMMA/PEO/GnP-3 nanocomposites. GnP: graphene nanoplatelet; PMMA: poly(methyl methacrylate); PEO: poly(ethylene oxide); SEM: scanning electron microscopy.
The presence of graphene nanosheets influences the morphological structural changes of the blend. In the simple polymer blend system (Figure 6(a)), we observe the relatively smooth fractured surface showing the miscible nature of PEO with PMMA. As can be seen in Figure 6(b), GnP is dispersed more homogeneously in the PMMA/PEO matrix as compared to Figure 6(c) and (d). We also use TEM to determine the dispersion state of GnP sheets in the PMMA/PEO matrix. Figure 7(a) is the representative TEM image of a nanocomposite with 0.8 wt% GnP content. It is seen from Figure 7(a) that the graphene layers are dispersed homogeneously in the PMMA/PEO blend. The black curly lines are the edges of the graphene nanosheets. The typical TEM images of PMMA/PEO/GnP-1.5 and PMMA/PEO/GnP-3 nanocomposites (Figure 7(b) and (c)) show that some of the graphene sheets were aggregated and some of them were dispersed throughout the polymer matrix. This is related to the interactions between GnPs, mainly by van der Waals force, stacked the sheets together and formed GnP agglomerates. The presence of dark areas indicates the relatively large thickness of graphene sheets due to the formation of agglomerates in the PMMA/PEO matrix. 37

TEM images of (a) nanocomposite with 0.8 wt% GnP, (b) nanocomposite with 1.5 wt% GnP, and (c) nanocomposite with 3 wt% GnP. TEM: transmission electron microscopy; GnP: graphene nanoplatelet.
Conclusion
Graphene-reinforced PMMA/PEO nanocomposites were successfully prepared via solution casting method. Various amounts of graphene nanosheets were incorporated into the PMMA/PEO blend to study their effects on thermal stability, electrical, mechanical, and morphological properties of the nanocomposites. From XRD patterns and electron microscopy images, it was understood that graphene sheets were well dispersed and exfoliated in the nanocomposite matrix at concentrations lower than 1.5 wt% of GnP, while they formed aggregates at higher GnP contents. The mechanical properties of the PMMA/PEO/GnP composites increased when graphene nanosheets were added to the polymer matrix. Thermal properties of the nanocomposites increased significantly as compared to neat PMMA/PEO. This is related to good matrix–GnP interaction, high thermal conductivity, and the barrier effect of GnP. Electrical conductivity of the nanocomposites was improved dramatically by the incorporation of GnP. The percolation threshold for electrical conduction of PMMA/PEO/GnP nanocomposites was found at 3.33 wt% GnP loading.
