Abstract
The growing demand for sustainable packaging solutions has led to the development of biodegradable materials with improved functionality. In this study, polyvinyl alcohol (PVA) and sodium caseinate (NaCAS) were blended with glycerol as a plasticizer to produce a biodegradable film matrix. Matcha tea (MT) powder was then incorporated into this matrix at varying concentrations (1%, 2%, 5%, 10 wt%) as a natural antioxidant and antimicrobial agent. To enhance structural performance, glutaraldehyde (GLA) was used as a cross-linking agent. The non-crosslinked and GLA cross-linked PVA/NaCAS/MT films were then evaluated based on their morphological, mechanical properties (tensile strength, elongation at break, Young’s modulus), water solubility, antioxidant capacity (DPPH and ABTS assays), and antibacterial activity against Escherichia coli and Staphylococcus aureus. The results showed that increasing the MT content enhanced the antioxidant capacity of the films, while GLA cross-linking suppressed this. Additionally, both MT incorporation and GLA cross-linking resulted in a decrease in mechanical strength and flexibility. On the other hand, the cross-linking effect of GLA was proven through a water solubility test. According to the antibacterial activity test, PVA/NaCAS/MT films didn’t exhibit bacterial inhibition under test conditions. After the GLA cross-linking, all films showed antibacterial activity thanks to GLA. This study, which is the first investigation on the direct incorporation of MT into a polymer matrix, has generally proven the usability of MT as an antioxidant in food packaging films.
Introduction
Today, packaging materials used in the food industry are widely produced from petroleum-based polymeric materials. However, increasing environmental pollution and the damage caused to nature by plastic waste pose a major threat. 1 Microplastics in plastic materials absorb toxic chemicals, increasing the effects of this problem and negatively affecting human health.1,2 Therefore, the use of environmentally friendly and biodegradable alternative packaging materials is gaining importance. Various studies are ongoing in the literature on the use of different biodegradable polymers as environmentally friendly packaging materials.3,4 However, biomaterials are generally sensitive to environmental conditions and may have low mechanical strength.5,6 To address these limitations and enhance the functionality of biodegradable films, recent research has focused on developing active films with additional beneficial properties. In particular, antibacterial and antioxidant films have attracted increasing interest, especially in areas such as food packaging and healthcare.3,4,7 These films are typically obtained by incorporating antioxidant and antibacterial active ingredients into polymeric matrices and referred to as an active food packaging system. These active films not only act as an inert barrier but also help suppress microbial activity, thereby extending shelf life, preserving food quality, and improving safety during transportation and storage by reducing.7–9
Among the various biopolymers investigated for such active film applications, casein has emerged as a promising candidate. It is a natural, biodegradable milk protein with a wide range of chemical functionalities, making it especially valuable in the development of food packaging films. Casein is a suitable compound for film production thanks to its high molecular flexibility and random helical structure. 9 It is converted into water-soluble sodium caseinate (NaCAS) by coagulation in acidic media and increasing its pH with sodium hydroxide. 10 While NaCAS has advantages such as high solubility, good gas barrier properties, and transparency, it also has certain disadvantages, including hydrophilicity, low mechanical strength and brittleness, and high water vapor permeability.11–13 To overcome mechanical drawbacks, glycerol (GLY), a water-soluble polyol, is commonly used as an effective plasticizer. Thanks to hydroxyl (-OH) groups in its structure, GLY increases the mobility of polymer chains by reducing intermolecular interactions, thereby increasing the flexibility, extensibility, and processability of the polymer. 14 Its low molecular weight further enhances the plasticizing effectiveness. Moreover, GLY can be used in food packaging that is in direct contact with food, as it is a chemically stable and non-toxic compound. As a result, GLY effectively reduces the brittleness of NaCAS and improves its tensile strength and overall mechanical properties.5,15 Although NaCAS-based films offer several advantages, improving their durability, water vapor barrier, and mechanical strength remains a challenge.
To improve these properties, NaCAS can be blended with other biopolymers. One such polymer is polyvinyl alcohol (PVA), a synthetic but non-toxic and biodegradable polymer. Water-soluble PVA is preferred in food packaging due to its low cost and biodegradability. 16 It also stands out with its biocompatibility, flexibility, and good film-forming properties.17,18 Key properties such as molecular weight, degree of hydrolysis, and viscosity affect PVA’s water resistance and tensile strength.19,20 However, its tendency toward high solubility and water absorption can be problematic in certain applications. To eliminate these disadvantages, NaCAS can be blended with PVA to obtain more durable biological materials.21,22 To further convert these PVA/NaCAS composite films into active food packaging materials, it is crucial to incorporate natural bioactive agents that provide functional properties such as antioxidant and antibacterial activity. One promising candidate for this purpose is matcha tea.
Matcha tea (MT) is a premium powdered form of natural green tea obtained by finely grinding tea leaves and is traditionally produced in Japan. 23 The main compounds in MT are catechin and polyphenol, which have high antioxidant properties. Among these components, catechin helps prevent cell damage and reduce the effects of free radicals in the body (antioxidant effect).24,25 Additionally, catechins also exhibit antibacterial effects, contributing to MT’s antimicrobial potential. A catechin found in matcha is epigallocatechin-3-gallate (EGCG), which not only contributes its antioxidant and antibacterial activity but also enables a bio-crosslinking effect on the polymer matrices. Thanks to these components, MT offers many health benefits such as accelerating metabolism, strengthening the immune system, and promoting the cardiovascular system.25–27 Beyond its nutritional value, MT has also emerged as a promising bio-additive for the development of active food packaging films, offering both functionality and biocompatibility. So, it can be incorporated into PVA/NaCAS films as an antioxidant and antimicrobial agent. Although green tea extract has been extensively investigated for its functional benefits in various polymer films,28–32 there is a lack of studies specifically utilizing matcha tea in such polymer matrices. Unlike conventional green tea extracts, matcha tea differs in its particle size, higher bioactive compound concentration. 33
While MT improves the functionality of PVA/NaCAS films, structural reinforcement is also necessary to enhance their durability under environmental stress and reduce water solubility. For this purpose, glutaraldehyde (GLA) can be used as a crosslinking agent. Thanks to the two carbonyl groups in its structure, it reacts rapidly with primary amine groups and forms strong and permanent covalent bonds. 34 With these bonds formed by GLA, it tightly connects polymer chains and increases the resistance of the structure to water, heat, and chemicals.12,21 Furthermore, GLA can significantly improve the mechanical, chemical, and thermal stability of biopolymer structures. Its ability to achieve rapid and effective crosslinking without requiring thermal treatment makes it more advantageous than many other crosslinking techniques. 35 There are various studies in the literature on the cross-linking of materials prepared by adding NaCAS to the PVA matrix with GLA. In the study conducted by Samatya Yılmaz et al., films were prepared by the solution casting method by adding Ag nanoparticles (Ag NP) to the PVA/NaCAS composite mixture. The dipping method in the GLA bath was applied for cross-linking of the prepared composite films. Cross-linking increased thermal stability, and as the amount of Ag nanoparticles increased, the total amount of soluble matter and water vapor permeability decreased. All the produced films showed antibacterial activity and showed potential as a food packaging material. 36 Şendil et al. produced nanofibers by the electrospinning method by directly adding GLA solution to the PVA/Sodium Caseinate (SC) mixture. Although the high amount of GLA in the nanofibers increased the antibacterial activity, it caused the emergence of toxic properties. In addition, it was stated that adding GLA directly to the mixture showed a lower binding effect compared to dipping the films in the GLA bath. Therefore, the prepared films were dipped in the GLA bath, and a cross-linking process was applied. 37 Dülger et al. (2024) performed the crosslinking process using natural lactic acid (LA) as an alternative to GLA to crosslink the biodegradable PVA/NaCAS composite films they prepared. It was reported that the antibacterial effect of the crosslinked films was high. As a result of the study, lactic acid was proven to be an environmentally friendly and effective alternative. 35
This paper includes the preparation of composite films by adding MT powder, a natural antioxidant and antimicrobial agent, at different concentrations into a PVA/NaCAS blend matrix plasticized with GLY. The study aims to produce biodegradable and environmentally friendly polymeric films with improved functionality. Additionally, the effect of GLA crosslinking on the structural and functional properties of the films was evaluated. The resulting films were comparatively analyzed for their mechanical properties (tensile strength, elongation at break, and Young’s modulus), antioxidant activity (via ABTS and DPPH assays), antibacterial activity, and water solubility. Overall, this study aims to develop functional, durable, and sustainable film materials suitable for active food packaging applications. Moreover, to the best of our knowledge, this is the first study to investigate the direct incorporation of MT powder into a polymer matrix, thereby addressing a notable gap in the literature and contributing novel insights to the development of natural active packaging materials.
Experimental part
Material
The PVA used is Kuraray Poval 49-88, coded product supplied by Kuraray Co., Ltd. Its viscosity is 45.0–52.0 mPas, degree of hydrolysis is 87.0–89.0 mol%, non-volatile content is 97.5 +–2.5%, ash content is ≤ 0.4%, pH value is 5.0–7.0. NaCAS in powder form with more than 92% protein, 6% water, and 4.5% ash content was purchased from Acros Organics. Matcha Tea powder (MT) of Fishake will be used as the additive. GLA is a 50% water product supplied by Sigma. Glycerol (GLY) is a Cas-No: 56-81-5 Emsure ACS reagent supplied by Merck. It is a Ph Eur product.
Method
Preparation of composite films
First, it was decided from the literature studies in which ratios of PVA polymer and NaCAS milk protein would be prepared. In previous studies, material production was carried out using PVA/NaCAS mixtures in 70/30 (w/w or v/v) ratios.37,38 In this study, priority was given to developing biodegradable materials using more natural raw materials.21,34–36 According to previous studies, it was decided to prepare a 60/40 (v/v) PVA/NaCAS mixture solution by volume, not by weight.
PVA was dissolved in pure water at 80°C with a magnetic stirrer at a concentration of 4 wt% for 2 h, with the mouth of the beaker tightly closed with aluminum foil and parafilm. NaCAS powder was calculated for a concentration of 4 wt%, and 25 wt% GLY was added according to the calculated solid amount and dissolved together in pure water. NaCAS/GLY solution was stirred at room temperature for 2 h with the mouth of the beaker tightly closed. At the end of 2 h, PVA and NaCAS (60/40, v/v) were mixed by volume. Then, it was stirred at room temperature for 2 h. At the end of 2 h, the prepared Pure PVA/NaCAS solution was poured into 14 cm diameter petri dishes without waiting. After that, MT powder at the ratios of 1, 2, 5, and 10 wt% was added to the PVA/NaCAS (60/40, v/v) solution according to the total solids in the solution and stirred for 18 hours. After the PVA/NaCAS/MT solutions were completely dissolved homogeneously, they were poured into 14 cm diameter petri dishes. The films prepared by the solution casting method were kept at room conditions for 72 h to dry. Some of the dried films were separated to be cross-linked in the GLA bath. The remaining films were taken into a desiccator for characterization.
The cross-linking process in GLA bath of composites films
GLA solution bath was prepared for the cross-linking process by the dipping method according to the recipe obtained from the literature.21,34,36,37 0.15 GLA (2.25 mL GLA) (50% diluted), 0.05 M hydrochloric acid (0.255 mL HCl) (37% diluted HCl) and 147.525 mL acetone were mixed in a beaker to obtain a total of 150 mL crosslinker the GLA solution bath. The dried films were immersed in the prepared GLA bath and left at room temperature with the mouth of the beaker tightly closed for 24 h. After 24 h, the cross-linked PVA/NaCAS/GLA and PVA/NaCAS/MT/GLA films were washed. The films were first kept in ethyl alcohol for 5 min and then washed three times in phosphate buffer solution. The cross-linked and washed composite films were dried in an oven at 50°C. The PVA/NaCAS/GLA and PVA/NaCAS/MT/GLA films taken from the oven were dried at room temperature for 24 h. So, the cross-linked films with GLA were made ready for examination of their properties.
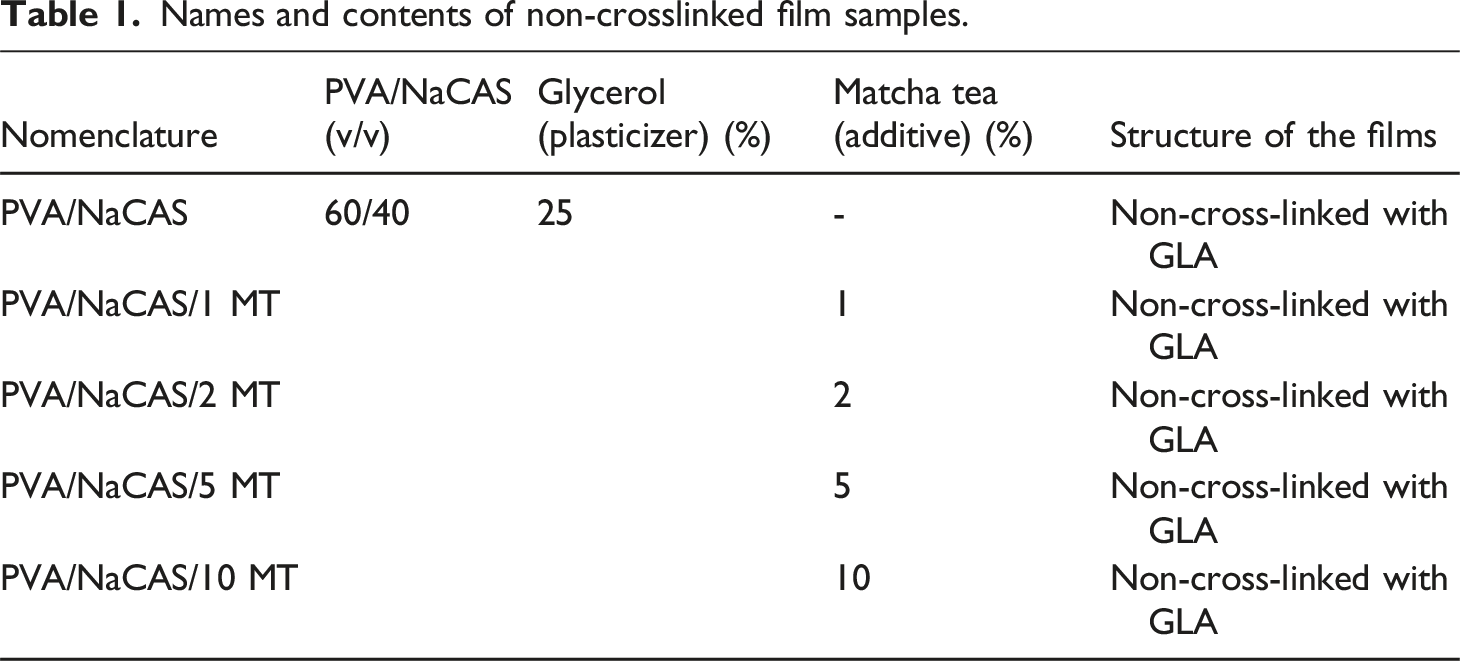
Names and contents of non-crosslinked film samples.
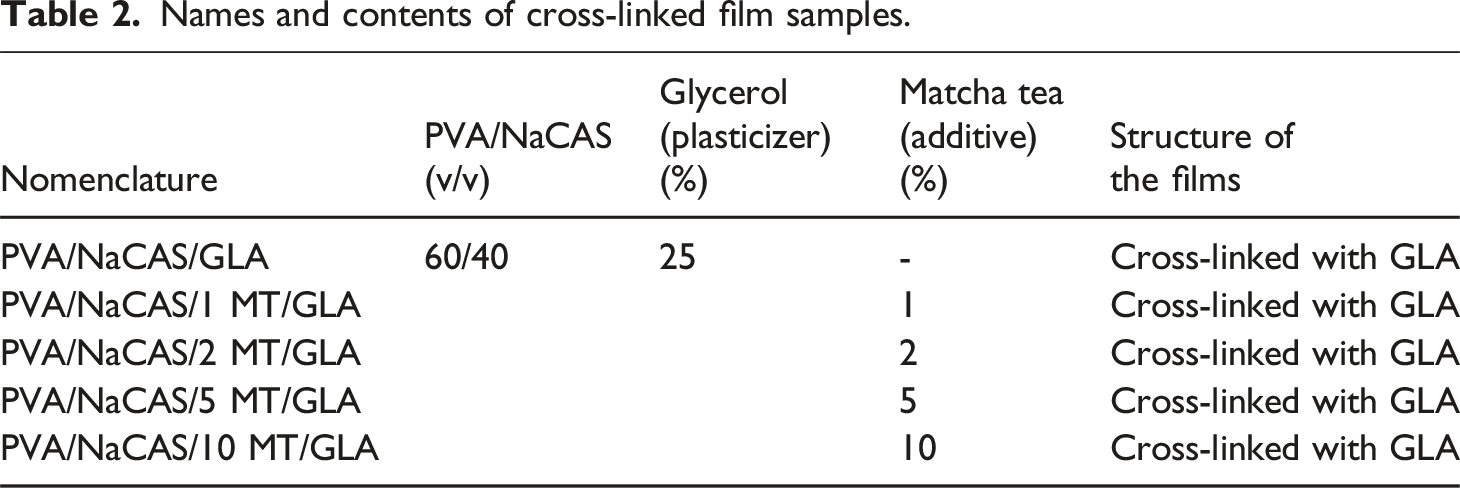
Names and contents of cross-linked film samples.
Characterization
The morphological structure of the films was examined via scanning electron microscopy (SEM) (Quanta 250 SEM). The surfaces of all films were imaged using SEM. All surfaces were coated with Au before analysis.
Fourier transform infrared spectroscopy (FTIR) analysis was performed on the film samples in the wavelength range of 650–4000 cm−1 using a Perkin Elmer Spectrum 100 infrared spectrometer. This analysis was used to examine the chemical bonds, molecular structure, and chemical interactions in the films.
In mechanical tests, tensile properties of the film samples thinner than 1 mm were measured at room temperature with an Instron brand tensile tester according to ASTM D882 standard at a strain rate of 10 mm/min. The tensile test has been repeated five times for each sample. The results were given as the mean of these values.
In the swelling capacity test, the films were cut into three 1 cm × 1 cm pieces using a method in the literature,
34
weighed, and initial weights were recorded. Then, the samples were kept in 50 mL of distilled water for 24 h. At the end of the period, the films were removed from the water and the films were weighed by removing excess water with filter paper. The swelling ratio was calculated with the following formula (1):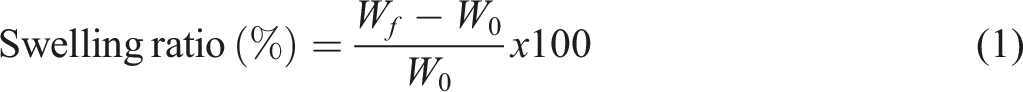
Wf = Weight of swollen the sample.
W0 = Initial dry weight of the sample.
The Total Soluble Matter (TSM) ratio test was performed by cutting three 1 cm × 1 cm film samples using a method in the literature
36
and weighing them. The samples were kept in 50 mL of distilled water for 24 h. Then, the undissolved films were dried at room temperature and weighed again after 48 h. The total soluble substance amount was calculated with the following formula (2) using the obtained data: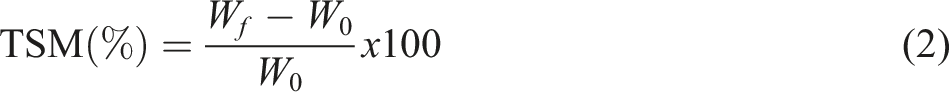
W0 = Initial weight weighed.
Wf = Remaining weight after dissolution.
Antioxidant analysis
The antioxidant capacity of the films was measured by ABTS and DPPH radical scavenging methods. Film samples (25 mg) and 5 mL of the working solution were mixed.
DPPH method
The DPPH radical-scavenging activity of each sample was determined according to the method proposed by Ajmal et al.
39
Film samples (25 mg) and 5 mL of 100 µM DPPH solution in ethanol were mixed and incubated for 30 min in the dark at room temperature. After this period, the absorbance at 517 nm was measured using a UV–visible spectrophotometer. The antioxidant activity of the films was calculated based on the following equation (3):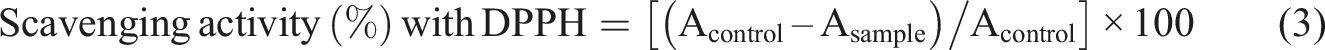
ABTS method
The ABTS radical scavenging assay was done with reference to Bozic et al.
40
The ABTS reagent was dissolved in distilled water to reach a 7 mM concentration. It was then mixed with 2.45 mM potassium persulfate solution. The solution was kept in the darkness at room temperature for 16 h. To obtain the working solution, it was diluted with phosphate buffer saline until it reached the absorbance of 0.700 ± 0.025 at 734 nm. 25 mg of film samples was added to 5 mL of the working solution. After 6 min incubation in the dark at room temperature, the absorbance was measured at 734 nm using a UV–Vis spectrophotometer. The percentage inhibitions of this radical were calculated using the following equation (4):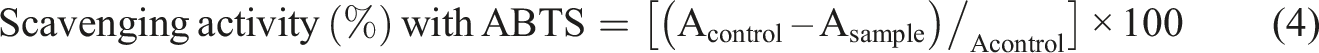
Antibacterial test
To determine the antibacterial activity of the films, the slightly modified ISO 22196 standard, which is a quantitative analysis based on agar plate counting method, was used.
34
During the antibacterial activity analysis, Escherichia coli ATCC 8739 and Staphylococcus aureus ATCC 6538P standard strains were used. Both bacteria were grown on Nutrient Agar (BioLife, Italy). Before starting the studies, both surfaces of the samples were sterilized with UVC for 30 min in a biosafety cabinet. Samples were transferred to 24-well plates under aseptic conditions. During the comparison of bacteria with samples, Luria Bertani Broth (LB) (Merck, Germany) medium diluted 1/250 with distilled water was used. A portion of the bacterial colonies grown on Nutrient agar medium was taken and suspended in 1/250 LB medium. Using a spectrophotometer, a turbidity equivalent to 0.5 McFarland turbidity (absorbance between 0.08 and 0.1 at 600 nm) was obtained (108 CFU/mL). These bacterial suspensions were diluted with 1/250 LB to prepare solutions containing 6 × 105 CFU/mL bacteria. 0.2 mL of this solution was taken, completed to 0.5 mL with 1/250 LB, and placed on sterile samples. Samples were incubated in a humid 37°C oven for 24 h. At the 0th and 24th h of incubation, liquid was taken from the solutions in which the samples were located and neutralized in the SCDLP solution. 0.02 mL of this neutralization solution was also taken and seeded on the NA medium surface with the help of a Drigalski spatula. The seeded petri dishes were incubated in a 37°C oven for 24 h. At the end of this period, the number of bacteria in the petri dishes was checked and counted. Antibacterial activity (%) was calculated according to the formula (5) following: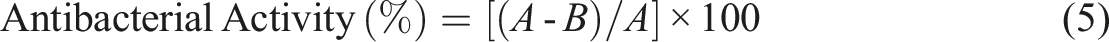
Result and discussion
Morphological properties
The morphological properties of the films were examined via SEM imaging. The surface micrographs of non-GLA cross-linked and cross-linked PVA/NaCAS and PVA/NaCAS/MT films are shown in Figures 1 and 2, respectively. PVA/NaCAS image demonstrates the film surface with no void or pit in Figure 1. The presence of casein micelles is seen as small particles in the smooth and uniform structure of PVA.
35
With the addition of MT, some morphological changes are observed. At 1 wt% MT loading, the surface exhibited roughness and the presence of particles that could be attributed to matcha powder particles or casein micelles became apparent. However, the distribution appeared to be uniform. At 2 wt% MT, the particle appearance on the surface disappeared, but deteriorations and cracks began to appear. With the increasing amount of MT, these deteriorations became more pronounced, and surface roughness and heterogeneity increased. Agglomerations and crack formations are noticeable. This situation shows that the high amount of additive negatively affects the structural integrity of the polymer matrix. It can be said that the MT additive is incompatible with the PVA/NaCAS matrix, and its distribution remains weak. These SEM images also support the mechanical property results. SEM micrographs of PVA/NaCAS and PVA/NaCAS/MT films (10 µm). SEM micrographs of PVA/NaCAS/GLA and PVA/NaCAS/GLA/MT films (10 µm).
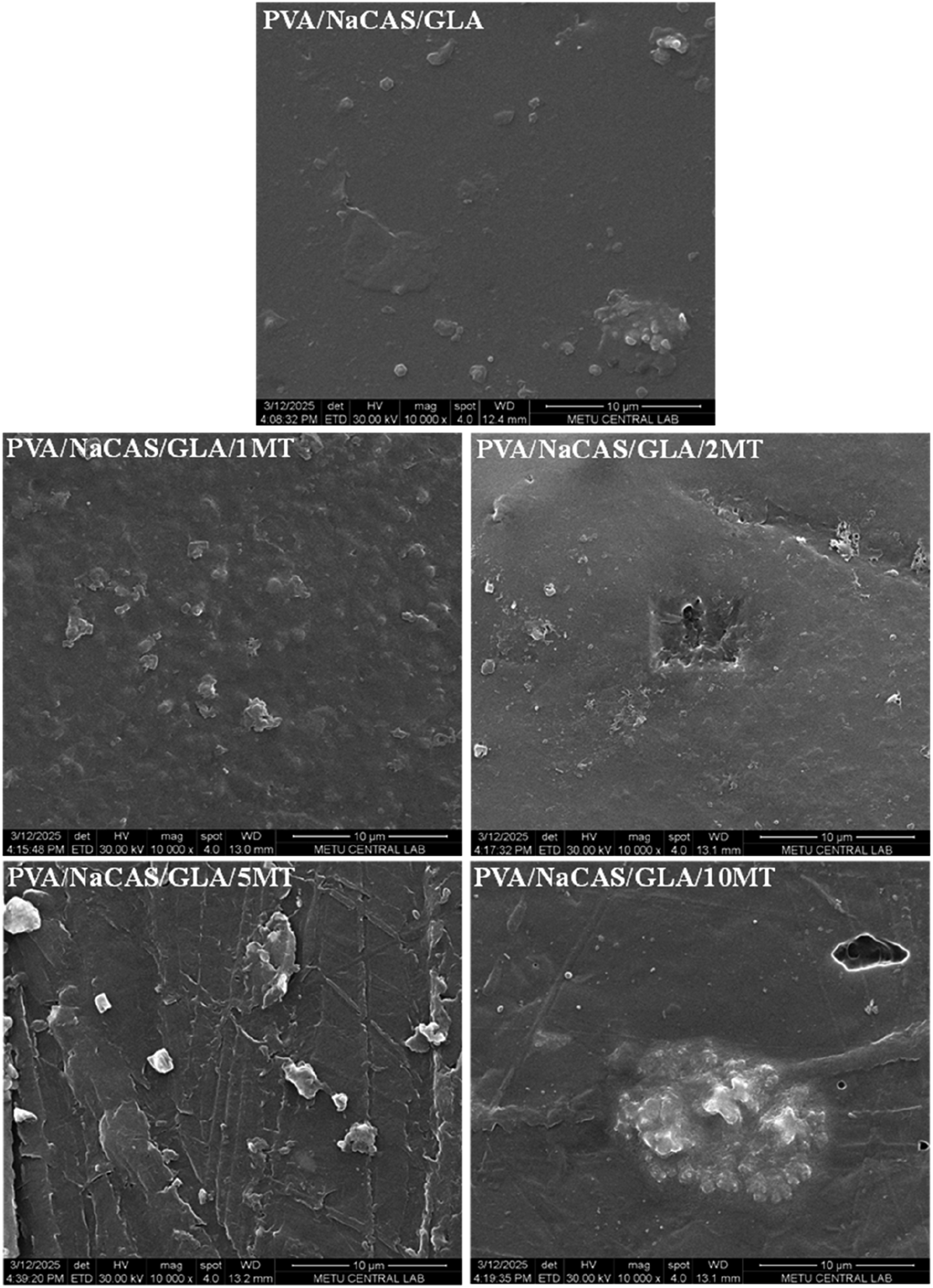
The surface morphology of PVA/NaCAS and PVA/NaCAS/MT films after GLA cross-linking can be seen in Figure 2. No significant change was observed in the PVA/NaCAS/GLA film because of cross-linking. Similar to the films that were not cross-linked with GLA, the appearance of agglomeration on the surface and crack formation became apparent with the addition and increasing amount of MT powder. Compared to non-GLA-cross-linked films, more agglomerates and occasional voids, and pits are noted on the surface of GLA-cross-linked films. Regardless of the MT powder amount, GLA cross-linking appears to exert a detrimental effect on surface morphology, leading to poorer additive dispersion and reduced interfacial compatibility between the filler and the polymer matrix. Although cross-linking is generally expected to increase structural integrity through the rearrangement of polymer chain networks, 41 GLA negatively affected structural integrity in PVA/NaCAS/MT films. It is known that the hydrophobicity of the polymer matrix increases after cross-linking with GLA. 12 This may have caused the dispersion of the hydrophilic MT powder to be weakened. As a result, worsened additive dispersion and deteriorated structural integrity due to GLA cross-linking resulted in decreased mechanical properties.
Chemical structure properties
FTIR spectroscopy was used to determine the chemical structure and functional groups in the pure MT powder and prepared films, and potential chemical interactions between the film components. The FTIR spectra of MT powder, PVA/NaCAS film, and PVA/NaCAS/MT films without and with GLA were depicted in Figures 3 and 4, respectively. FTIR spectra of pure MT powder, PVA/NaCAS film and PVA/NaCAS/MT films. FTIR spectra of pure MT powder, PVA/NaCAS/GLA film, and PVA/NaCAS/MT/GLA films.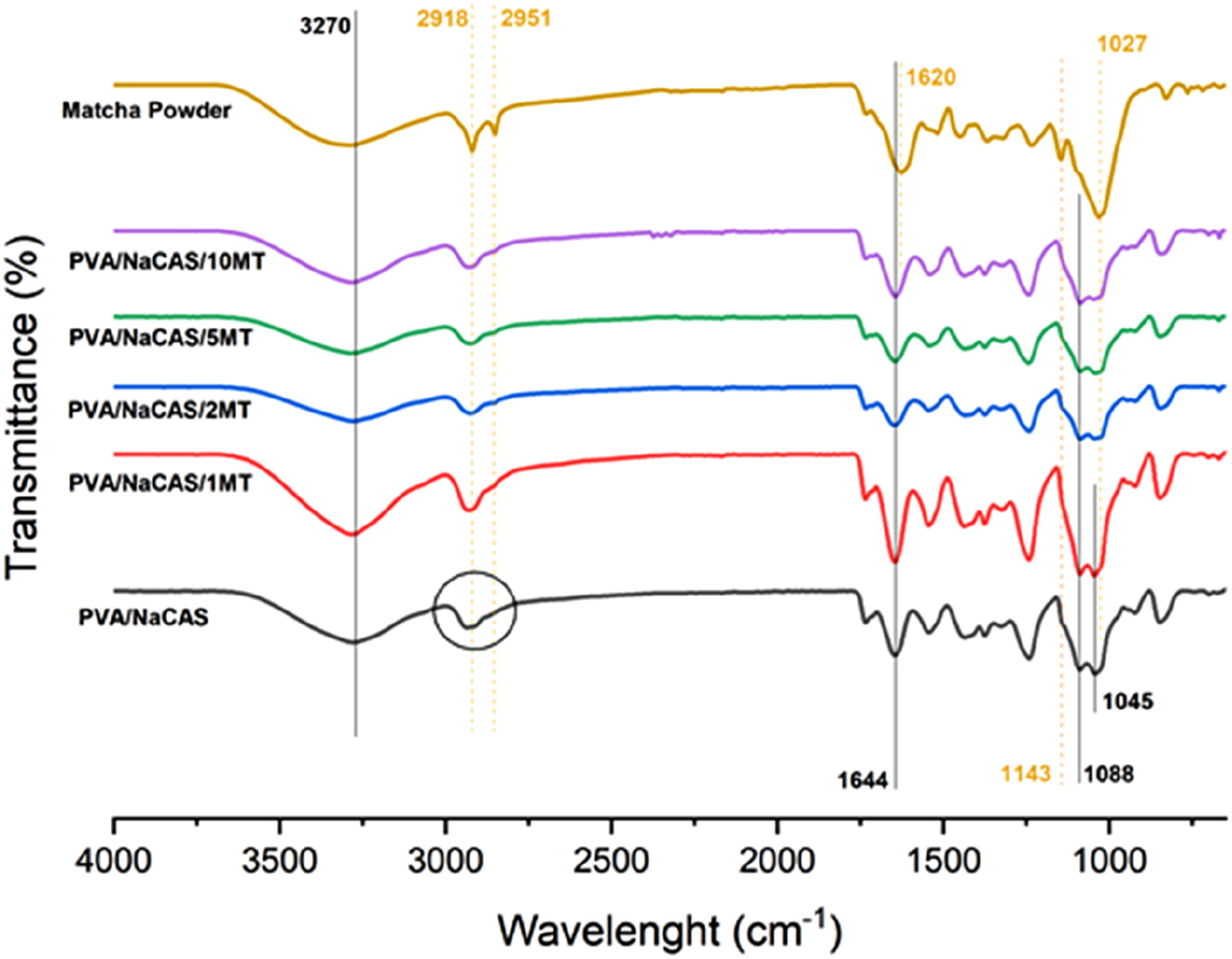
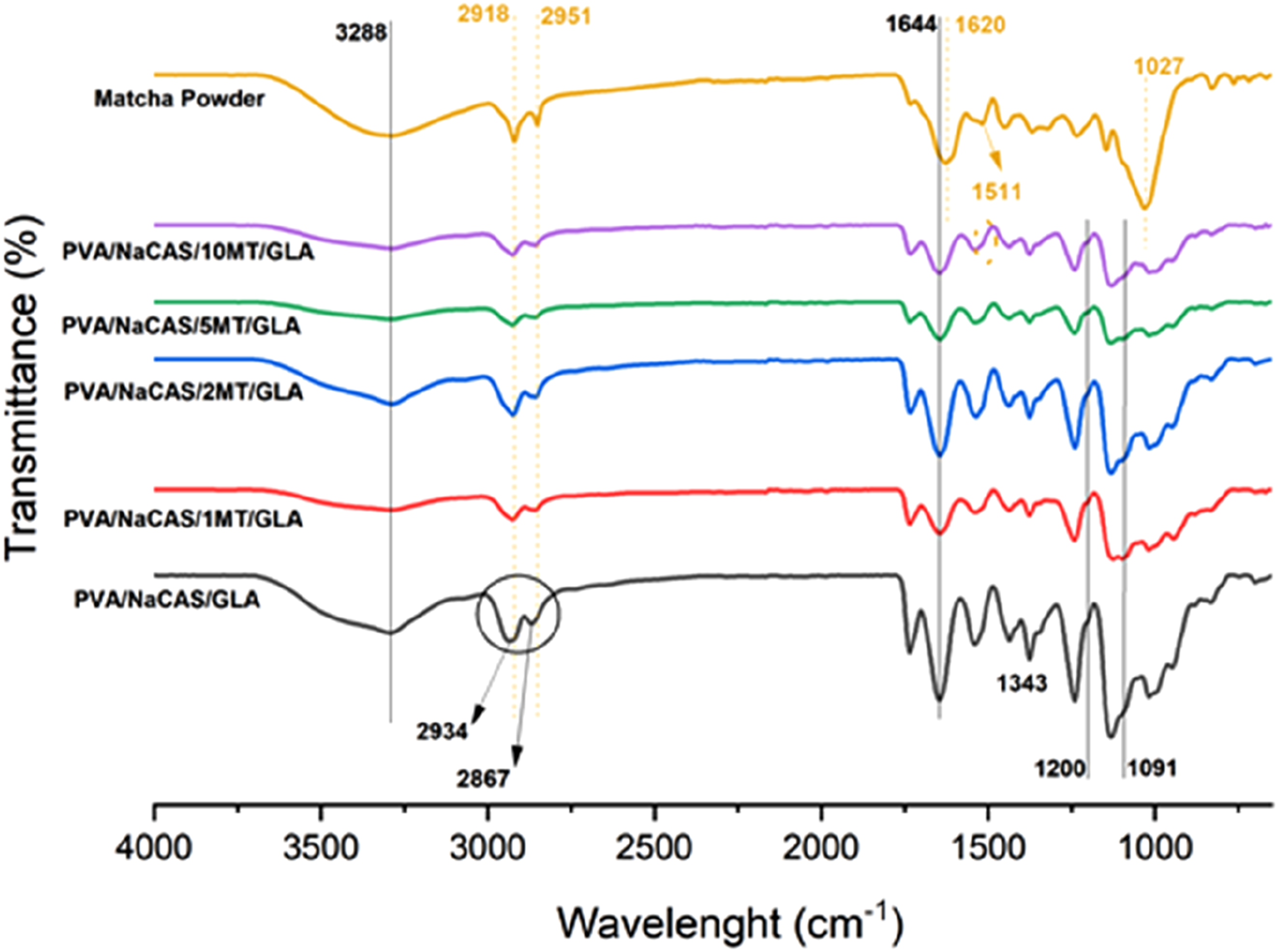
The FTIR spectrum of PVA/NaCAS film in Figure 3 exhibited the characteristic absorption bands of pure PVA and NaCAS. In our previous studies, the spectra of pure PVA and NaCAS were reported.22,37 In the spectrum of PVA/NaCAS film, a peak at 3288 cm−1 is mainly attributed to the hydroxyl group (-OH) of PVA. The peaks at 2934 and 2867 cm−1 belong to the asymmetric and symmetric stretching of the -CH bond, respectively. The carbonyl group (C = O) peak of PVA was seen at 1734 cm−1. Also, the characteristic of the amide I (C = O) bond peak of NaCAS was observed at 1644 cm−122,37. The peaks at 1045 and 1088 cm−1 can be assigned to C-C-C and C-O stretching vibrations.37,42 In addition, it has been observed in studies that the peak at 1200 cm−1 is the characteristic peak of glycerol. 43 In the spectrum of MT powder given in Figures 3 and 4, the hydroxyl group (-OH) peak was observed at a wavelength of 3288 cm−1. The peaks at 2918 and 2951 cm−1 belong to C-H stretching vibrations. The peak at 1620 cm−1 wavelength indicated -OH bending and stretching vibrations. Also, other characteristic peaks of MT powder were observed at the wavelength of 1511 cm−1 (C = O), 1367 cm−1 (C = O), 1233 cm−1 (C-OH) and 1027 cm−1 (C-O-C bond).44,45 In this case, the common peak value for both materials was extracted. When the spectra of PVA/NaCAS/MT films are examined, it is seen that the addition of MT to the PVA/NaCAS structure does not cause any new peak formation that could indicate a chemical reaction. On the other hand, small shifts in wavelengths and decreases in peak intensities can be attributed to the presence of some Van der Waals and hydrogen bond interactions.
PVA/NaCAS/MT films were prepared, and then the films were cross-linked in the GLA bath. According to the FTIR spectra of GLA cross-linked films that were shown in Figure 4, the cross-linking process of PVA/NaCAS film with GLA reduced the -OH groups by the formation of possible acetal bridges. When the spectra in Figure 2 are examined, it is seen that the PVA/NaCAS/GLA film shifts towards the wavelength of 3288 cm−1 and the hydroxyl (-OH) peak intensity decreases due to the cross-linking effect. The hydroxyl groups in the structure of PVA/NaCAS film react with the aldehyde groups of GLA. This is because the -OH bond density is consumed by the cross-linking reaction. 12 The peak belonging to -C-NO2 at 1343 cm−1 was observed as a new peak due to the cross-linking effect. 34 If the spectra of non-cross-linked and GLA-cross-linked PVA/NaCAS/MT films are compared, the hydroxyl group (-OH) peak observed in the cross-linked PVA/NaCAS/GLA film decreased with the addition and increasing amount of MT, and it was thought that matcha tea interacted with GLA. The binding of PVA/NaCAS/GLA film with GLA caused the reduction of hydroxyl groups. The peak belonging to matcha powder was slightly evident at 1511 cm−1 wavelength of PVA/NaCAS/GLA/10 MT film. This is because the amount of matcha was the highest in this film sample. In addition, a decrease in peak intensities was observed as the amount of matcha powder increased for all film samples, indicating that matcha tea affected the film properties.
Total soluble matter and swelling ratio results
The total soluble matter (TSM, %) amounts of films cross-linked and uncross-linked with GLA are shown in Figure 5. It was determined that PVA/NaCAS and PVA/NaCAS/MT films cross-linked with GLA showed a very low TSM ratio. Especially pure PVA/NaCAS/GLA exhibited a TSM ratio that was almost insoluble, with a ratio of 0.94%. This result indicates that pure PVA/NaCAS film gained high insolubility against water by cross-linking with GLA. However, there was a slight increase in TSM values of GLA cross-linked films after MT was added to the structure. A linear TSM increment was noticed, especially at low MT ratios (1% and 2%). It was thought that MT is a hydrophilic natural additive with a water-soluble structure
46
and therefore exhibits dissolution behavior by attracting water. Thus, the dissolved MT additives increased the amount of soluble matter in GLA cross-linked films. In this study, the -OH group observed in the FTIR analysis of matcha powder proved the presence of hydrophilic groups in the structure. On the other hand, when the MT amount increased to 5 and 10 wt% in GLA cross-linked films, a decrease in the TSM value was observed. Especially, the GLA cross-linked 5 MT film showed almost insoluble behavior with a value of 1.03%. This reduction can be associated with the cross-linking effect of MT. It has been reported in the literature that some ingredients in the green tea structure also showed an effect as crosslinkers.47,48 However, the TSM value of the GLA cross-linked 10 MT film increased slightly compared to the GLA cross-linked 5 MT film. This phenomenon can be attributed to the dual behavior of high MT content, while it may partially contribute to cross-linking through its polyphenolic groups, its inherently hydrophilic nature also promotes water solubility. The resistance of these two opposite behaviors resulted in the 10 MT film cross-linked with GLA, showing a TSM value of 2.57%. TSM values (%) of PVA/NaCAS and PVA/NaCAS/MT Films.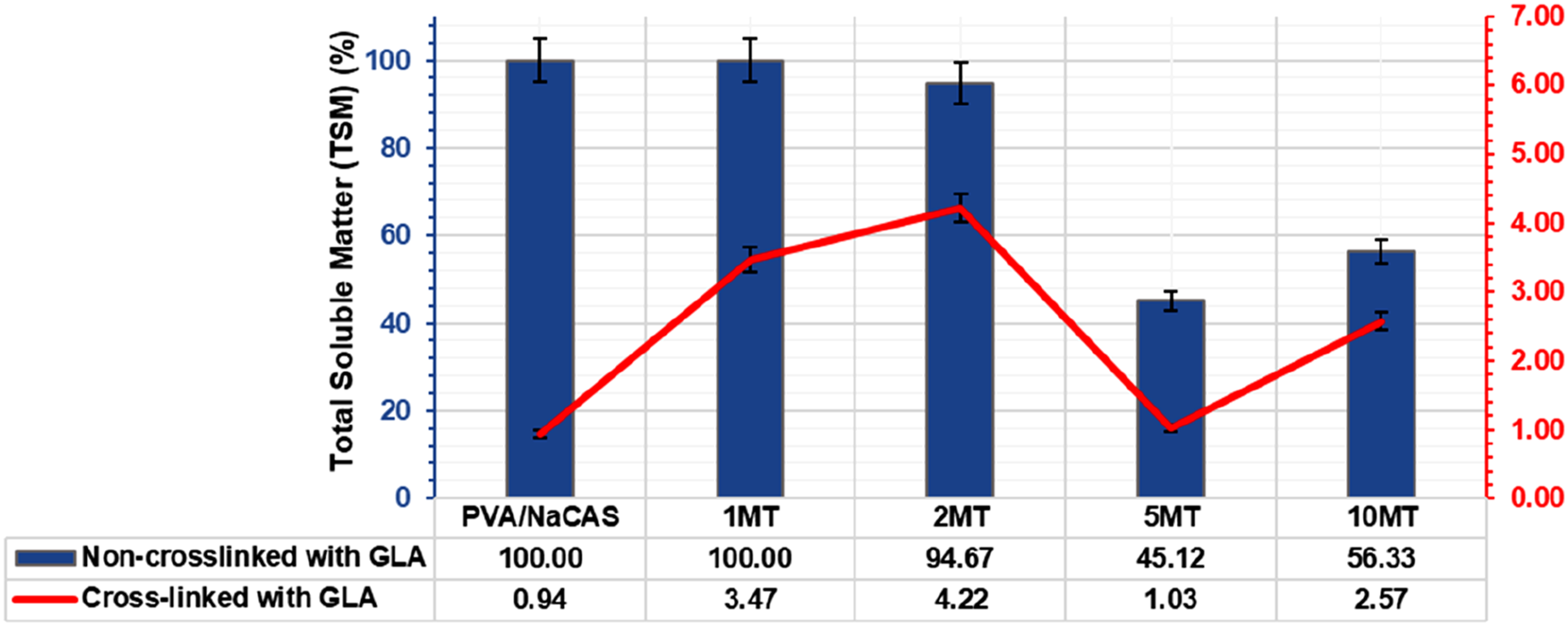
The potential cross-linking effect of MT was indirectly supported by the relatively lower TSM values observed in non-GLA-crosslinked films containing 2–5–10 wt% MT, compared to the pure PVA/NaCAS film. Non-GLA-crosslinked PVA/NaCAS and PVA/NaCAS/1 MT films were completely dissolved and showed 100% total soluble matter content. It was observed that the PVA/NaCAS/2 MT film maintained its existence in the water environment to a certain extent, as 5.33%. It was indicated that especially non-GLA-crosslinked 5 MT and 10 MT films maintained their existence in the water environment by approximately half at the end of 24 h. It was also determined that the TSM value of the non-crosslinked 10 MT film was higher than that of the 5 MT film. This result was found to be similar to the TSM behaviour of 10 MT film crosslinked with GLA.
The swelling ratios of the films cross-linked with GLA are given in Figure 6. Since the non-crosslinked films dissolved in water, they were not suitable for swelling tests, and the measurements were therefore terminated. The effect of cross-linking with GLA on the PVA/NaCAS/GLA and PVA/NaCAS/MT/GLA film samples is consistent across both TSM and swelling test results. Notably, higher swelling ratios were associated with increased TSM values, indicating lower cross-linking density. As can be seen in Figure 6, the incorporation of the MT additive into the matrix increased the swelling ratio compared to that of the pure PVA/NaCAS/GLA film. This trend can be attributed to MT impairs the crosslinking efficiency of GLA, likely due to poor dispersion and possible interactions between MT polyphenols and aldehyde. In particular, a sharp increase in swelling amount was determined for the GLA cross-linked 10 MT film. This significant increment suggested that the MT additive has a water absorption capacity
31
and caused a disruption in crosslinked network formation. Similarly, PVA/NaCAS/1 MT/GLA and PVA/NaCAS/2 MT/GLA films also exhibited a higher swelling ratio. Since 1 and 2 wt% ratios were insufficient to reveal the cross-linking effect of MT, the presence of hydrophilic MT decreased the cross-linking density and thus increased the swelling ratio. In the film with 5% MT, the swelling ratio decreased and was found almost the same as that of the pure PVA/NaCAS/GLA film. Therefore, it can be interpreted that the cross-linking effect of both GLA and MT additives was observed to be optimum in the presence of a 5 wt% MT additive ratio. Swelling ratio (%) of PVA/NaCAS and PVA/NaCAS/MT Films.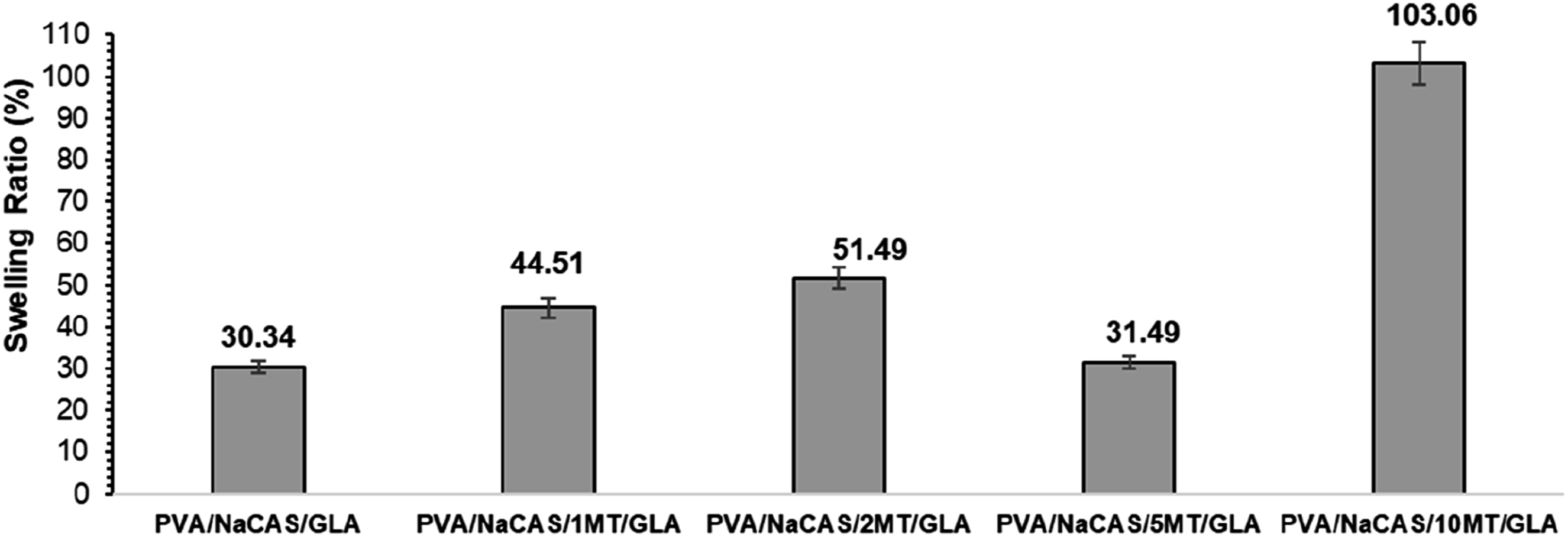
Mechanical properties
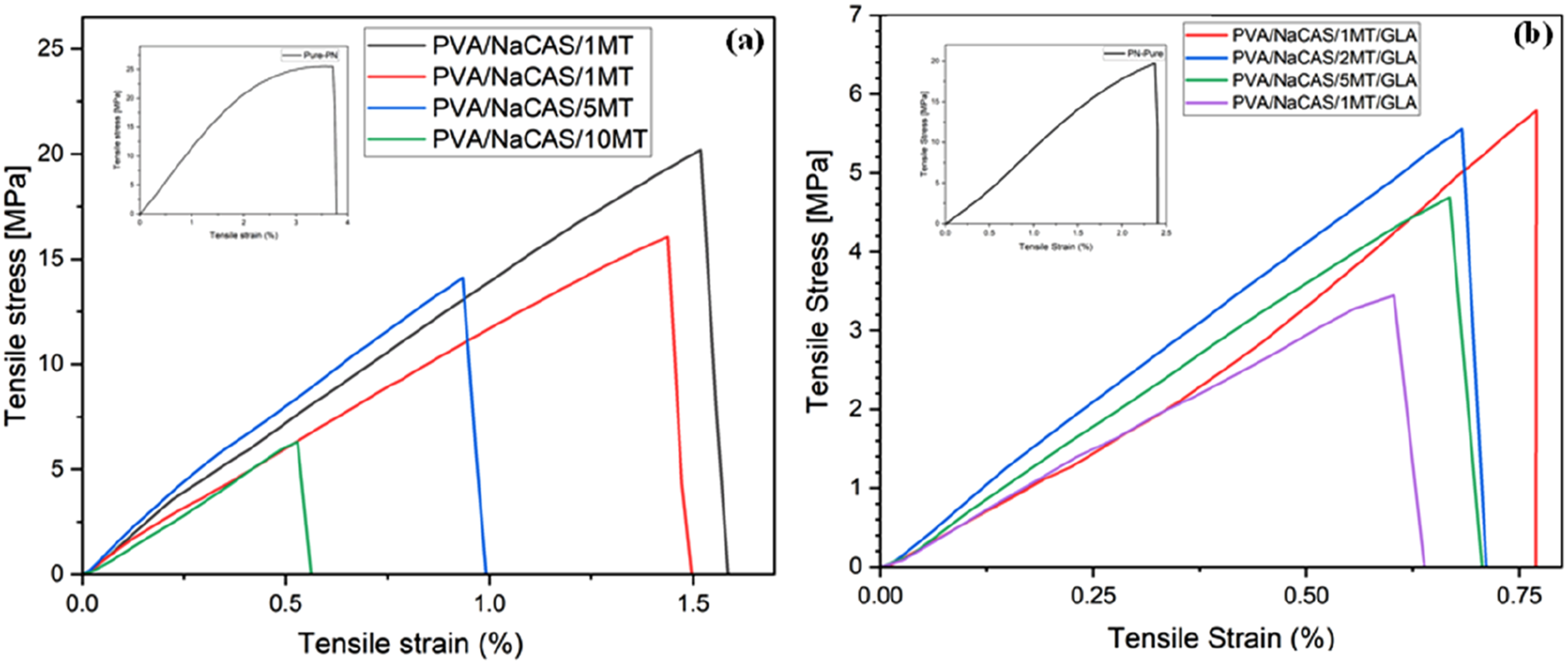
The mechanical properties of PVA/NaCAS and PVA/NaCAS/MT films with and without GLA were assessed via tensile test. The tensile strength, Young’s modulus, and strain at maximum load values of the samples are presented in Figure 7. Also, the stress-strain curves of films are shown in Figure 8 (a) and (b). The tensile test result indicated that the addition of MT powder and GLA cross-linking process remarkably changed the mechanical properties of PVA/NaCAS film. Mechanical properties of films (a) Tensile Strength (b) Young's Modulus (c) Strain at max load. Stress-Strain curves of (a) non-crosslinked (b) GLA-crosslinked films.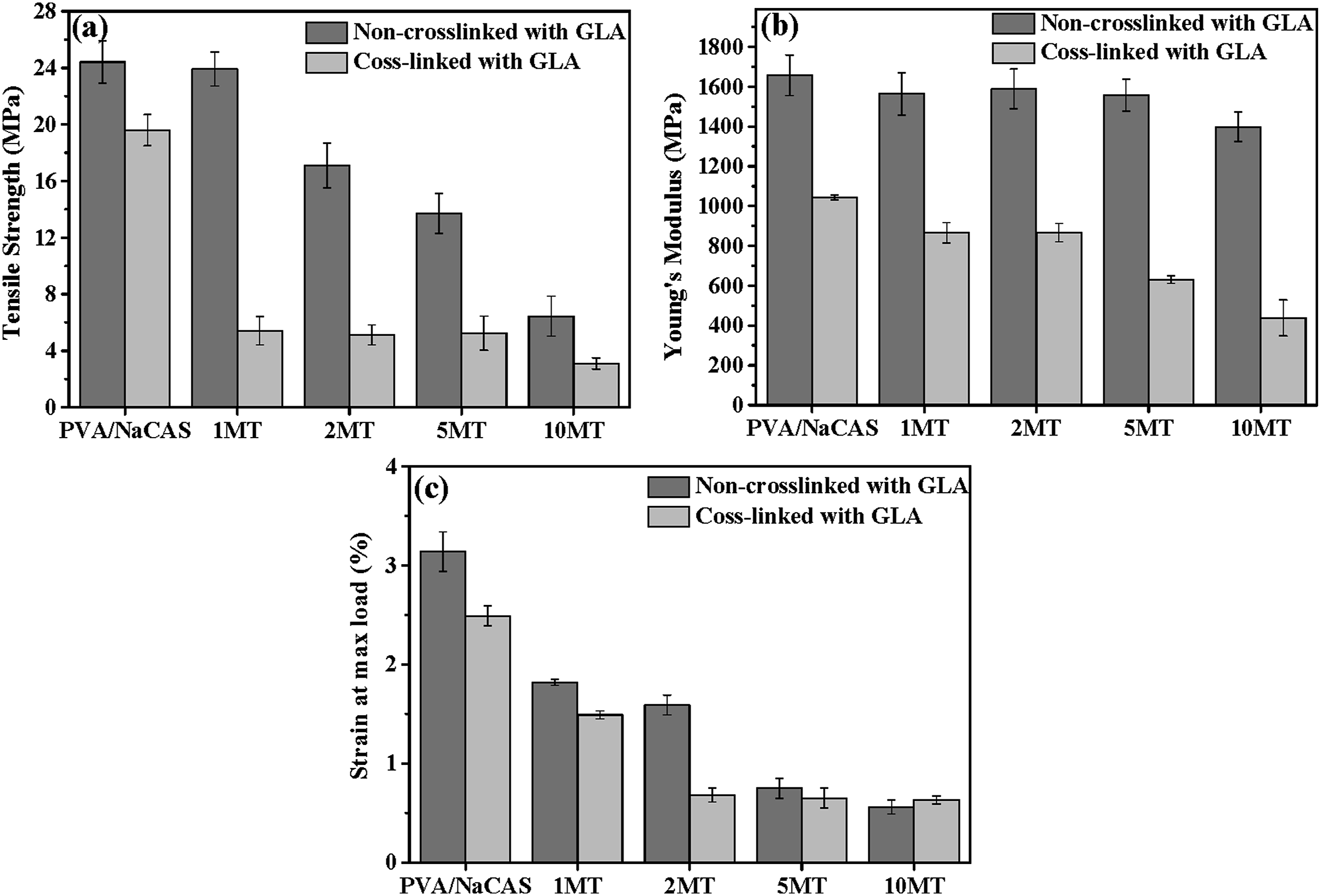
The PVA/NaCAS film has a tensile strength of 24 MPa, which decreased with the addition of MT powder. PVA/NaCAS/1 MT films exhibited a tensile strength of 23.9 MPa, and this value decreased to around 17, 13, and 6 MPa with the increasing amount of MT. This reduction can be attributed to the incompatibility between the PVA/NaCAS matrix and MT particles, as well as the nonhomogeneous dispersion of MT powder particles,as mentioned in Figure 1. Moreover, it may be assigned to the reduction of intermolecular interactions between the PVA and NaCAS chains and forming some voids and pits in the matrix with the incorporation of MT. PVA/NaCAS/10 MT films showed the lowest tensile strength value due to agglomeration of MT powder and cracks. These cracks and agglomerates act as stress concentration points and cause poor stress transfer. Thus, the mechanical properties of the matrix are weakened. Furthermore, the FTIR results indicated that the blending is physical rather than chemical. Similar trends were reported for PVA/tea polyphenol films 30 and polycaprolactone-polylactic acid-green tea extract films. 28 If the effect of GLA cross-linking on the tensile strength of PVA/NaCAS and PVA/NaCAS/MT films is examined, a similar reduction trend can be seen. The tensile strength of the PVA/NaCAS/GLA film was recorded as 19.6 MPa. As the MT amount in the film matrix increased, a decrease was observed in the tensile strength of the cross-linked film samples. GLA cross-linked films containing 1, 2, 5, and 10 wt% MT powder showed significantly lower tensile strengths than the PVA/NaCAS/GLA film with values of 5.42 MPa, 5.12 MPa, 4.59 MPa, and 3.09 MPa, respectively. The decrease in tensile strength observed after GLA cross-linking suggests that an effective network structure was not obtained by the cross-linking process, and the load transfer was weakened. Similar results were reported by Samatya Yılmaz et al. 36 and Nath. 49
The modulus of elasticity, also known as Young’s modulus, is an important property that shows the durability of the material. The higher the value of the modulus, the more durable the material is. The modulus of elasticity, which can be expressed as the amount of stress applied per unit extension, provides information about the resistance of the material to deformation. The non-crosslinked PVA/NaCAS film exhibited 1657 MPa Young’s modulus, and this value decreased to 1044 MPa after the GLA cross-linking. Like tensile strength, GLA cross-linked and non-crosslinked film samples containing 1, 2, 5, and 10 wt% MT showed lower Young’s modulus than PLA/NaCAS and PLA/NaCAS/GLA films. The reduction with the addition of MT was more pronounced for GLA cross-linked films. The addition of MT and cross-linking with GLA affect the chemical structure and also the chain mobility and crystalline structure of the polymeric matrix. Both MT and GLA negatively affected mechanical durability.
The strain at max load or elongation (%) value of the PVA/NaCAS film was measured as 3% which dropped to 2.5% with GLA cross-linking. The MT powder inclusion in the structure causes a reduction in the elongation of both non-crosslinked and GLA-crosslinked films. When MT was added and the amount of MT increased in the PVA/NaCAS structure, a gradual decrease was observed in the elongation. Among the tested samples, the highest elongation amount was seen in the film sample containing 1 wt% MT, which was the closest value to the PVA/NaCAS/GLA samples. The elongation amounts of the remaining film samples with 2, 5, and 10 wt% MT content remained approximately the same but decreased slightly. This reduction is due to the incompatibility between the filler and matrix and heterogeneous dispersion of MT powder, as can be seen in SEM micrographs.
When the stress-strain graph is examined, it is understood that the toughness of the PVA/NaCAS/GLA film has higher values than the other MT-added films. After the PVA/NaCAS/GLA film, the highest toughness was observed in the films containing 1 wt% MT. It can be said that this result is related to the modulus values of the MT-added films. The 2 wt% and 5 wt% MT-added films, which have similar elongation values, are also films with almost the same toughness. While the 1 wt% MT film has the highest tensile strength values, the lowest tensile strength was observed in the film containing 10% matcha, despite the highest matcha amount. This situation was also observed in the elongation and the elasticity modulus values. The mechanical strength and elongation at break values were comparable with PVA/apple pomace films, 50 but lower than those of commercial polymers like low-density polyethylene and biodegradable PVA/lignin film. 18 On the other hand, endowing films with functional properties such as antibacterial and antioxidant activity, which will be discussed in the following sections, may offer a sustainable alternative for applications where mechanical strength is not critical, and where short-term use and active protection are prioritized.
Antioxidant results with ABTS and DPPH radical scavenging methods
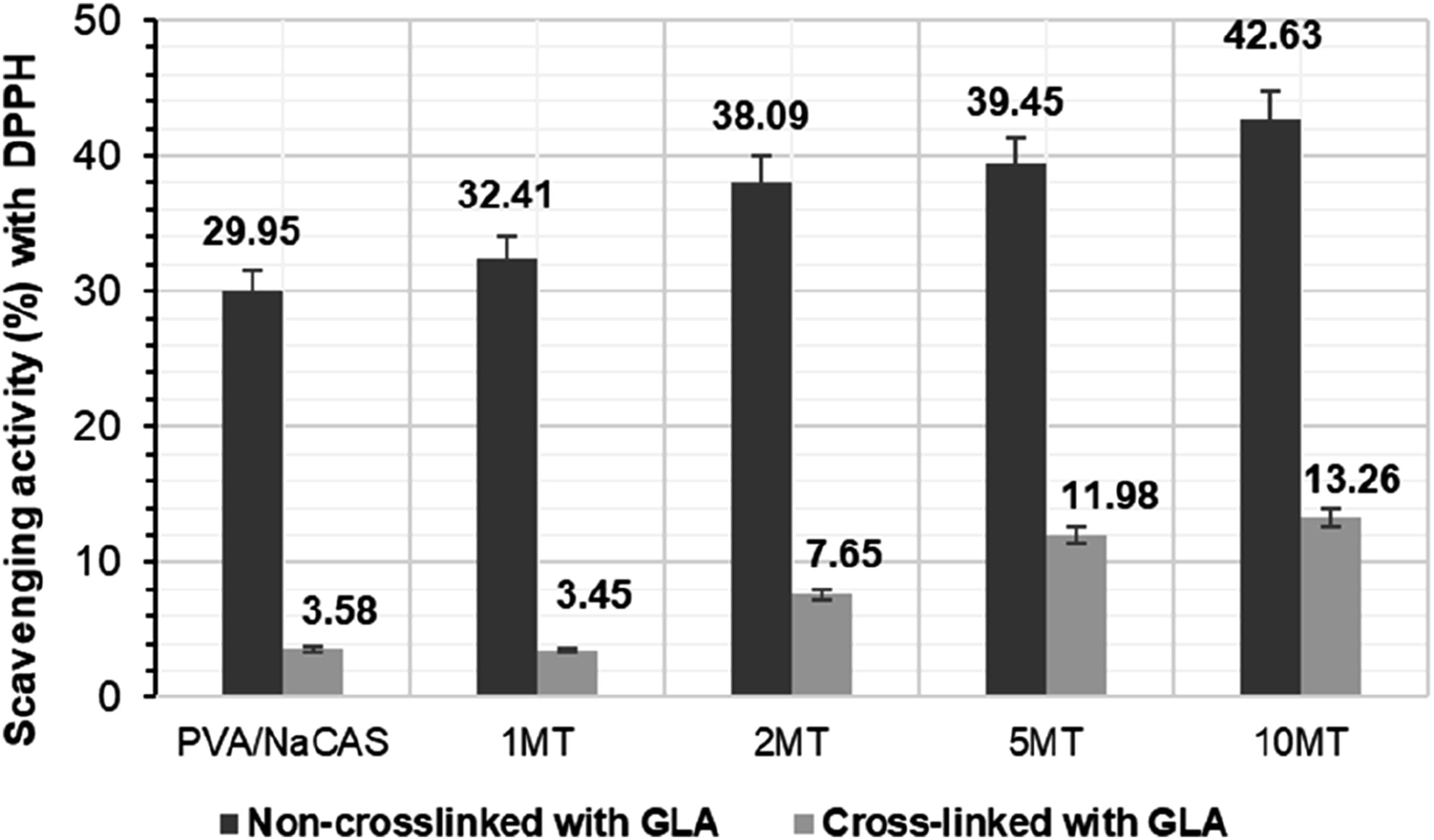
Scavenging activity (%) of GLA cross-linked and non-crosslinked PVA/NaCAS films by ABTS and DPPH methods are given in Figures 9 and 10, respectively. Scavenging activity (%) with ABTS of non-crosslinked and GLA-crosslinked films. Scavenging activity (%) with DPPH of non-crosslinked and GLA-crosslinked films.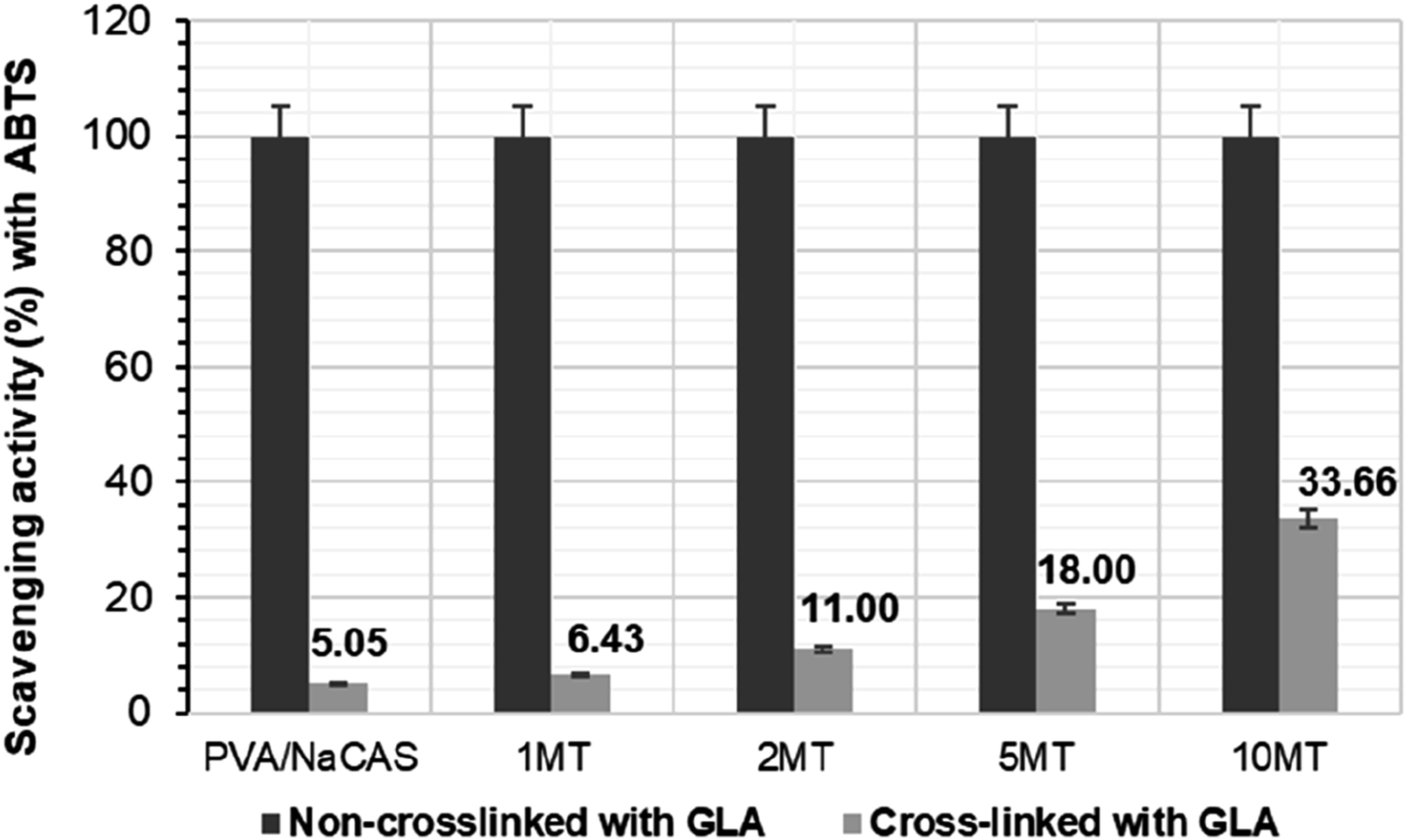
Matcha tea exhibits strong antioxidant properties due to its high levels of catechins (especially EGCG), caffeine, phenolic acids, rutin, quercetin, vitamin C, chlorophyll, and
While the ABTS scavenging activity of pure MT powder used in this study was obtained as 99.39%, the DPPH scavenging activity was found as 51.35%. This difference in the results of ABTS and DPPH methods is largely due to the chemical properties of the reagents used in both methods. DPPH is a stable nitrogen radical that can interact with peroxidic radicals that play a role in the lipid peroxidation process and tends to react especially with lipophilic fractions. On the other hand, ABTS reagent can interact with both hydrophilic and lipophilic compounds. Therefore, the DPPH test has a limited effect area, mainly in the evaluation of lipophilic antioxidant activity.52,53
ABTS scavenging activity of pure PVA/NaCAS film, non-crosslinked with GLA, and all MT-doped films were calculated as 99.44%. Here, it was stated that the antioxidant effect of MT was revealed especially in MT-doped films, and full-performance ABTS scavenging activity behaviour was observed even at the lowest MT content. It has been reported in previous studies that PVA film alone does not show any antioxidant effect.50,54–56 On the other hand, it has been confirmed by the literature that the high antioxidant activity observed in pure PVA/NaCAS film comes from NaCAS. NaCAS protein exhibited different scavenging activity rates for both ABTS and DPPH methods. In the study conducted by Tang et al. 57 in 2023, the scavenging activity of pure NaCAS according to the ABTS method was determined to be approximately 22%, while the scavenging activity in the DPPH method was determined to be approximately 16%. In another study in 2025, Kheynoor et al. 58 reported that the scavenging activity of pure NaCAS according to the ABTS method was approximately 14%, while the scavenging activity in the DPPH method was approximately 33%. These differences were associated with the different NaCAS concentrations used in the studies. It was noticed that the antioxidant properties changed as the NaCAS content and concentration changed, and different scavenging activity values were recorded in different antioxidant measurement methods.
In recent work, the scavenging activity of the non-crosslinked pure PVA/NaCAS film in the ABTS method was quite high (99.44%), while the scavenging activity in the DPPH method was reported as 29.95%. The scavenging activity of GLA cross-linked and non-cross-linked films in the DPPH method also increased as the MT content increased. In particular, the DPPH scavenging activity of the 10 MT film without cross-linking with GLA was found to be 42.63%. Here, the effect of MT powder is revealed by the 12.68% difference between the values. However, pure PVA/NaCAS film and all films with MT additives showed a sharp decrease in DPPH scavenging activity values after crosslinking with GLA. While the DPPH scavenging activity of pure PVA/NaCAS film cross-linked with GLA was recorded as 3.58%, that of 10 MT film cross-linked with GLA was determined as 13.26%. Since a similar difference (9.68%) was found between them before crosslinking with GLA, it was determined that the presence of GLA suppressed the antioxidant activity of NaCAS in particular. A similar behavior to the trend of DPPH scavenging activity values after cross-linking with GLA was also seen in the ABTS scavenging activity of the films. All films exhibited sharply lower values in ABTS scavenging activities after cross-linking with GLA. Here also, it was stated that GLA primarily limited the antioxidant activity of NaCAS.
It has been reported in the literature that antioxidant properties will be increased due to the interaction between NaCAS and epigallocatechin gallate (EGCG). The covalent grafting between EGCG and sodium caseinate (NaCAS) occurred via phenolic hydroxyl groups and resulted in the formation of NaCAS–EGCG complexes with strong antioxidant properties. Phenolic hydroxyl groups have the capacity to stabilize free radicals and play an important role in antioxidant activity thanks to these properties. The increased antioxidant activity of NaCAS has been associated with the formation of new active groups in the protein chain and structural changes that allow these groups to interact more easily with free radicals. In addition, it is thought that the changes in the secondary structure of NaCAS (protein) due to EGCG also support this activity. The analyses performed have shown that EGCG grafting significantly increases the antioxidant capacity of NaCAS. The radical scavenging and metal ion binding properties of oxidized EGCG also form a protective layer on the protein surface, reinforcing the increase in antioxidant activity. 57
According to this information, it has been determined that the antioxidant activity of NaCAS decreases because of the decrease in the hydroxyl groups in the NaCAS structure by cross-linking with GLA. Similarly, the decrease in the antioxidant activity of Matcha was understood from the decrease in ABTS and DPPH scavenging activity values, as the hydroxyl groups in EGCG in Matcha tea decreased in the presence of GLA. However, it has been reported that the presence of other active ingredients that are not affected by GLA in the MT structure ensures the sustainability of antioxidant activity. Overall, it is evident that GLA crosslinking inhibited the free radical scavenging performance of the films. While the non-crosslinked PVA/NaCAS/MT films demonstrated full ABTS radical scavenging capacity, their DPPH activity remained comparable to values reported in the literature. For instance, compared to PVA/apple pomace composite films, which exhibit similar mechanical properties, the developed films showed superior antioxidant activity. 50 Similarly, the DPPH scavenging activity was found to be comparable to, or even higher than, that of previously reported PVA/chitosan/Ocimum tenuiflorum extract 54 and PVA/green tea extract-based films. 29 These findings highlight the functional potential of the MT-loaded PVA/NaCAS films, especially when antioxidant efficacy is prioritized.
Antibacterial results
The antibacterial activity of non-crosslinked and GLA cross-linked films was carried out by an agar plate counting method, which is a quantitative method. Visuals of bacterial colonies counted on plates at the end of 24 h to determine the antibacterial activity of non-crosslinked are given in Figure 11. The antibacterial effect of matcha is attributed to the various compounds it contains. Prominent among these compounds are polyphenols, epigallocatechin gallate, flavonoids, theanine and phenolic acids. Polyphenols act by disrupting bacterial membranes and neutralizing toxins. Epigallocatechin gallate both weakens the cell wall and disrupts bacterial signaling pathways. While flavonoids penetrate the cell membrane and act from the inside, theanine provides an antibacterial effect by increasing membrane permeability and affecting metabolism. Studies show that matcha is more effective against gram-positive bacteria. This is because gram-positive bacteria have more porous and permeable, simple-structured cell walls, whereas the cell wall of gram-negative bacteria is more complex and the outer layer is less porous. Edraki et al.
45
reported in a study conducted in 2022 that pure Matcha green tea powder showed high antibacterial properties. However, in this study, it was determined that the non-crosslinked films had no antibacterial activity against either Gram-negative E. coli or Gram-positive S. aureus bacterial strains. The highest MT content incorporated into the PVA/NaCAS films was 10 wt%. However, this amount was observed to be insufficient to achieve complete bacterial inhibition under the conditions tested. It was suggested that antibacterial properties be re-evaluated at higher MT content. Another approach was evaluated as the dissolution of PVA/NaCAS/MT films within 24 h and the release of NaCAS, a milk protein, into the medium, which acted as a nutrient for bacteria and supported their proliferation. The antibacterial activity of non-crosslinked films.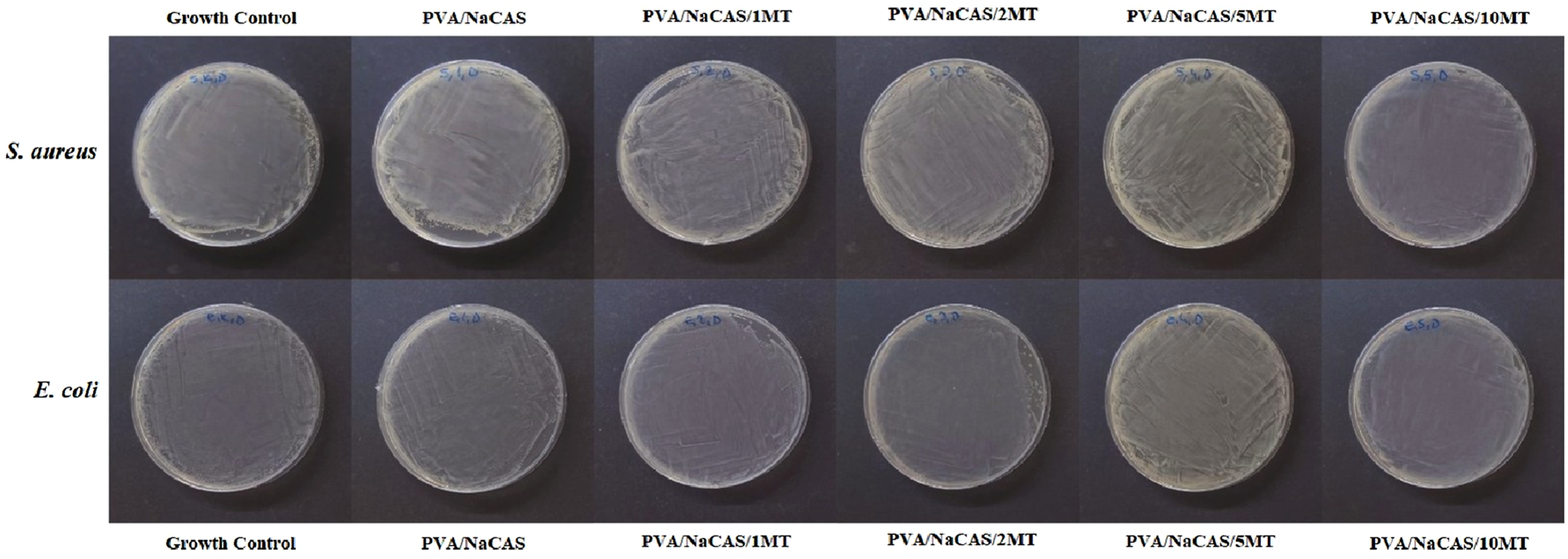
Images of the antibacterial activity of films cross-linked with GLA are given in Figure 12. While the non-crosslinked film with the highest MT content had no antibacterial activity, it was found that the cross-linked PVA/NaCAS/1 MT/GLA film with the lowest MT content showed 100% antibacterial activity against both Gram-positive S. aureus and Gram-negative E. coli bacteria. This is associated with the high antibacterial activity of GLA. It was reported that the bacteria died when the aldehyde groups, which were not cross-linked but were present on the film surface, were released into the environment.
21
In conclusion, while the GLA-crosslinked films exhibited strong antibacterial activity, particularly against both E. coli and S. aureus, their antioxidant activities were notably lower compared to the non-crosslinked counterparts. This inverse relationship suggests that GLA-crosslinked PVA/NaCAS/MT/GLA films may be more appropriate for applications where antibacterial protection is prioritized over antioxidant function. Therefore, such films hold potential for use in antibacterial surface coatings, inner liners, or active food packaging layers where microbial contamination is a major concern but prolonged oxidative stability is not essential. The antibacterial activity of GLA-crosslinked films.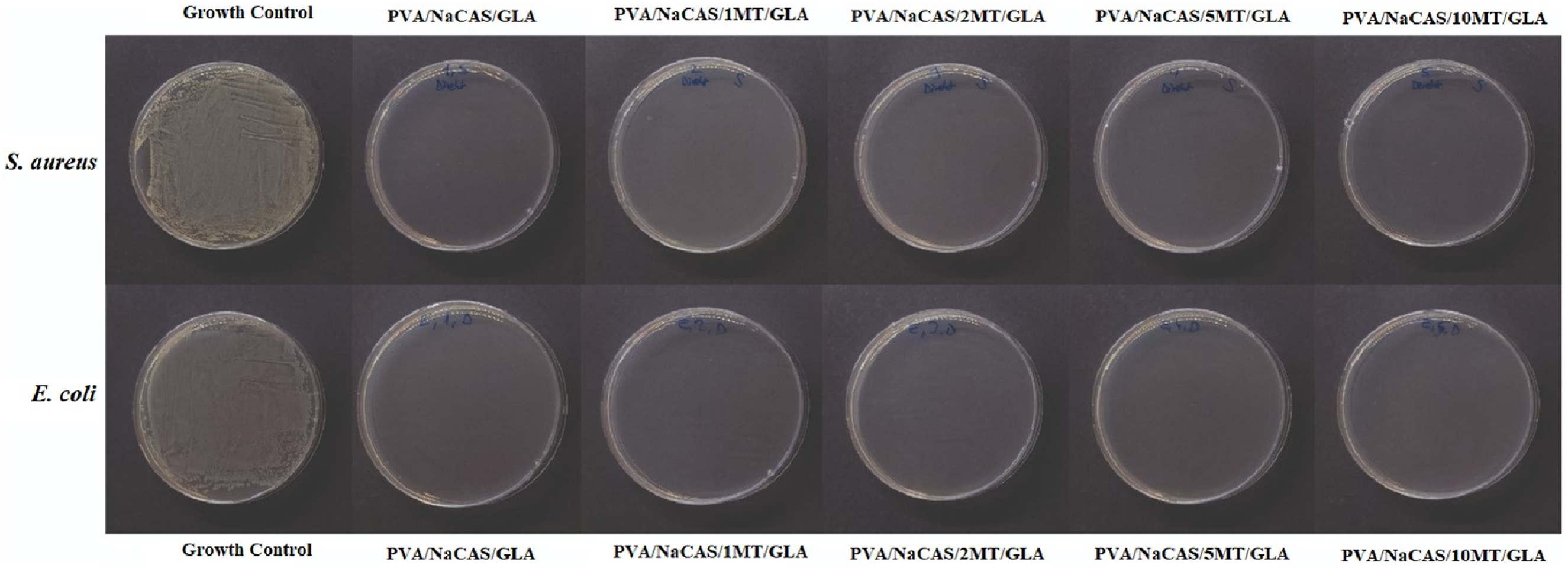
Conclusion
In this study, biodegradable composite films based on PVA/NaCAS were successfully developed by incorporating MT powder as a natural antioxidant and antimicrobial agent. The films were further modified via GLA cross-linking to enhance water resistance and antibacterial performance. The results demonstrated that increasing the MT amount particularly enhanced DPPH antioxidant scavenging activity, whereas GLA crosslinking significantly reduced this functionality due to the loss of hydroxyl groups. Despite the reduced antioxidant activity, GLA crosslinking effectively improved water resistance and antibacterial properties by introducing reactive aldehyde groups. On the other hand, it was found that MT had no antibacterial activity. Moreover, both MT incorporation and GLA cross-linking led to a notable decline in mechanical strength and flexibility, primarily due to weak interfacial compatibility and filler agglomeration. These findings reveal that the optimum film formulation must balance antioxidant/antibacterial activity, water resistance, and mechanical performance based on the intended application. While current mechanical properties may limit stretch-demanding packaging use, the films show potential for short-shelf-life food packaging, tray liners, or antimicrobial wraps, where sustainability and functional activity are prioritized. Additionally, this work presents the first reported direct incorporation of matcha tea powder into a biodegradable film matrix, offering a novel pathway in the development of green, active packaging systems. Future studies could explore compatibility enhancers or bio-based crosslinkers to overcome mechanical limitations and further expand the industrial applicability of these films.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Kocaeli University Scientific Research Projects Coordination Unit. Project number: FAA-2024-4265.
