Abstract
In this study, the sage seed gum (SSG) was electrospun as a polysaccharide polymer blended with polyvinyl alcohol (PVA) to construct nanofibrous mats for antibacterial applications. The rheological properties of the PVA/SSG solutions were investigated before electrospinning. Oscillatory, shear, and steady shear tests demonstrated that the PVA/SSG solutions had a viscous behavior, exhibiting a higher consistency coefficient (k) and a lower flow behavior index (n) at the higher PVA incorporation levels. The morphological studies by SEM images revealed that the PVA/SSG nanofibers were produced without bead defects within the diameter range of 130–300 nm. The in vitro degradation tests showed that the PVA/SSG nanofibers (at different SSG contents) were degraded by approx. 60–70% of their initial weight after one day of the degradation test. The antibacterial activity against Escherichia coli and Staphylococcus aureus microbial species and biodegradation tests also verified that the produced nanofibers could be implemented for antibacterial applications. The SSG polymer in the form of nanofibers can be, therefore, used as a natural and low-cost polymer for special antibacterial applications such as wound healing.
Introduction
Nanofibers are one of the most important areas in applied nanotechnology, which have recently attracted much attention due to their diverse applications. The nanofibers include three-dimensional (3D) fibrous structures with diameters in the range of 10–500 nm. 1 These structures have special features such as good mechanical properties, high specific surface and high porosity.2,3 Various methods such as drawing, template synthesis, phase separation, self-assembly, and electrospinning could be exploited to fabricate nanofibers. Among these techniques, electrospinning is the most efficient method to fabricate the nanofibers. 4 Different structures and features of the nanofibers can be obtained by altering some crucial and significant parameters during the electrospinning process. 5 When the diameter of the fibers fabricated from water-soluble polymers is diminished to sub-micron and nanometric scale, the resulting material with a large surface, high porosity, and 3D continuous structure will dissolve and disintegrate more rapidly in the aqueous systems. 6 Based on the literature, the interests in fabrication of the electrospun polysaccharides-based nanofibers such as chitosan, starch, alginate, xanthan, cellulose and cellulose derivatives, alongside investigating the properties of the resultant nanofibers, have been growing rapidly in recent years.7-13 The studies have been demonstrated that the electrospinning process of polysaccharides is directly affected by different factors such as their concentration, chemical structure, and pseudoplastic properties.
Many researchers have focused on finding new resources of natural hydrocolloids/polymers like plant seeds.14-18 Wild sage (Salvia macrosiphon) is endogenous to the north Mediterranean coast and is now distributed worldwide and broadly cultivated. It is found that sage seed could be used as a new source of hydrocolloid for application in pharmaceutical and food systems and has antimicrobial and antioxidant properties.19,20 Extraction optimization, physicochemical, functional, and rheological properties of the sage seed gum (SSG) have recently been comprehensively investigated.21-27 The SSG was found to be a galactomannan with a 1.78–1.93:1 mannose/galactose ratio, average molecular weight of 1.5 × 106 Da and 28.2–32.2% uronic acid. 21 Based on such properties, the SSG can be used to fabricate the biodegradable films with high thermal stability and solubility in polar solvents. However, due to some features such as the high viscosity and pseudoplastic behavior, as well as high surface tension and electrical conductivity, electrospinning of the polysaccharide-based polymers has been challenging. 11 The shear-thinning behavior of the SSG solutions at various concentrations (0.5–2 wt.%) and temperatures (25–50°C) has been reported. 24 According to dynamic rheological measurements, the flow behavior of the SSG solutions (0.5–2%, w/w) was found to be weak gel-like. 25 The pseudoplastic and weak gel-like behaviors of the SSG solutions may be connected with its molecular conformation. As long as the shear stress is low, the association of chains will be induced by hydrogen bonding, providing high viscosity. Conversely, due to successive rupture of the intra- and intermolecular hydrogen bonding at high shear stress, the decrease in apparent viscosity occurs. To overcome this problem, the combination of such materials with other polymers like polyvinyl alcohol (PVA) or polyethylene oxide (PEO) has been suggested.11,28,29 Recently, several attempts have been made to investigate the potential of the electrospinning of the extracted natural gums in combination with PVA. Serio et al. have successfully electrospun the PVA/gum Arabic nanofibers as a nanomedicine delivery system for cancer therapeutic application. 30 Ibrahim et al. also investigated the silver-loaded PVA/gum Arabic nanofiber and proposed it for food wrapping application. 31 The catechin-loaded Azivash gum (Corchorus olitorius. L)/PVA nanofibers were produced as a candidate for pharmaceutical and food packaging applications. 32 Rezaeinia et al. have worked on electrospinning of the balangu seed gum (Lallemantia royleana)/PVA blend nanofibers. 33 They encapsulated the bergamot essential oil in the electrospun fibers to create a novel fast delivery system for enhancing the flavor in food systems. Kurd et al. created novel nanofibers based on PVA/basil seed mucilage and characterized the produced fibers for their physicochemical properties. 34 They proposed the PVA/BSM nanofibers for encapsulating bioactive materials and packaging applications.
To the best of our knowledge, so far, no study has been taken to fabricate SSG-based nanofibers using electrospinning or to investigate their characteristics. Therefore, the purpose of the current study was to investigate the rheological properties of the PVA/SSG solutions and the fabrication of the PVA/SSG nanofibers using the electrospinning technique. We also aimed to study the impact of different factors involved in the fabrication of the nanofibers, including PVA:SSG ratio, flow rate, and applied voltage, on some physicochemical, morphological, and mechanical properties of the produced nanofibers. Antibacterial and biodegradation behavior of the PVA/SSG mats were also evaluated to verify the biological applicability of this nanofiber.
Materials and methods
Martials
PVA (M w = 72,000 Da) was purchased from Sigma-Aldrich Chemicals Co., and the sage seed was supplied by a local market in Tabriz, Iran. Phosphate buffered saline (PBS, pH = 7.4) and glutaraldehyde (biological grade, 50 wt.% in H2O) were also provided from Sigma-Aldrich Chemicals Co. The Gram-negative Escherichia coli (E. coli, ATCC 25,922) and Gram-positive strain Staphylococcus aureus (S. aureus, ATCC 25,923) were used for antibacterial activity tests.
Methods
Extraction of the sage seed gum
The sage seeds were cleaned to remove the impurities and foreign materials, including chaff, dust, and sand. The extraction of the SSG was performed according to the procedure described by Bostan et al. 23 Distilled water was added to the sage seeds in a ratio of 51:1 (w/w) at a controlled pH of 5.5, and the mixture temperature was kept at 25°C. After 20 min of soaking, the SSG solution was separated from the swelled seeds using a laboratory extractor. Finally, the extracted SSG was filtered and dried in a laboratory oven at 70°C for 24 h. The dried film was grounded and used to prepare the electrospinning solutions.
Preparation and electrospinning of PVA/SSG solutions
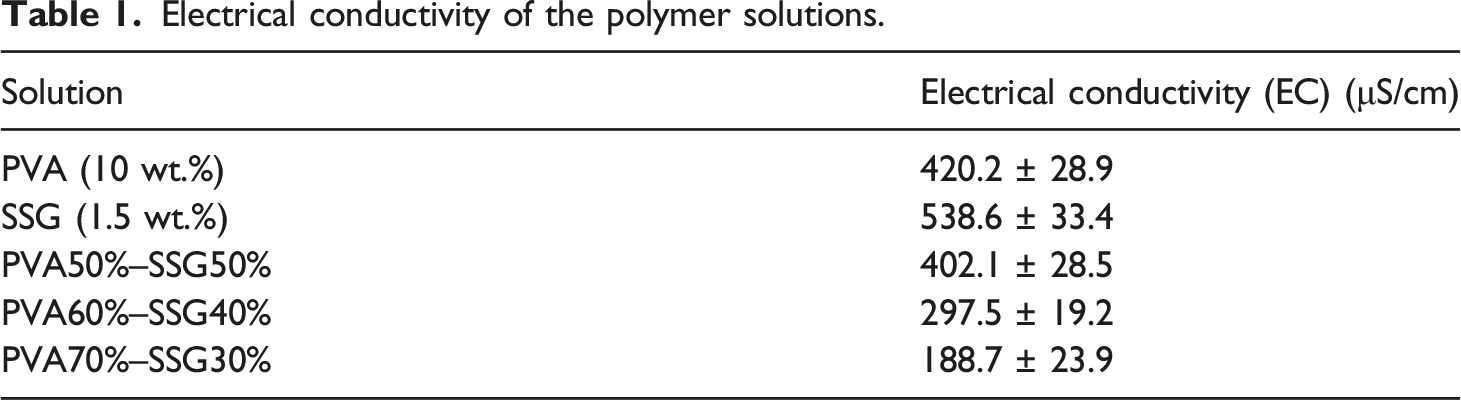
The PVA 10% w/w solution was made by dissolving PVA in distilled water and stirring for 6 h at 50°C. The sage seed gum powder was also dissolved in distilled water to obtain 1.5% w/w of the SSG solution and stirred overnight at room temperature. PVA and SSG solutions were mixed to obtain the solutions with 70:30, 60:40 and 50:50 (w/w) PVA:SSG ratios. The electrical conductivity of the polymer solutions was also measured using a conductivity meter (Crison CM35, Crison Instrument, Spain). Three tests were performed for each solution, and the values were averaged.
To find the optimum conditions of the electrospinning process, three factors were selected as the electrospinning variables; these included the PVA:SSG ratio, flow rate and the applied voltage. The electrospinning factors were adjusted at three levels of PVA:SSG (50:50, 60:40, and 70:30 w/w), electrospinning flow rate (0.5, 1.0, and 1.5 mL/h) and the applied voltage (13, 15, and 18 kV). The Taguchi method was used for the experimental design to minimize the number of the test samples by applying the Minitab 20.2.0 software.
Electrospinning was performed using a rotating drum collector covered by aluminum foil with a rotation rate of 250 r/min. The distance between the needle tip (gauge 23) and the collector surface was 13 cm for all conditions. Figure 1 shows the schematic of the electrospinning of the PVA/SSG nanofibers. The schematic of the electrospinning of the PVA/SSG nanofibers.
Rheological properties of the PVA/SSG solutions
Rheological measurements of the PVA/SSG solutions were carried out before electrospinning using a stress/strain-controlled Physica MCR301 rotational rheometer (Anton Paar, Austria) equipped with a parallel-plate geometry (60 mm diameter and 250 μm gap). The strain sweep test was then implemented to find the linear viscoelastic region of the PVA/SSG solutions (70:30, 60:40 and 50:50 w/w) within the strain range of 0.01–100% and at a constant frequency of 1 Hz, at 25°C.
The steady shear flow behavior of the prepared PVA/SSG solutions was investigated at 25°C. A viscous flow behavior was observed at the strain-controlled mode with shear rates in the range of 0.1–1000 s−1 to attain the shear stress (τ) vs. shear rate
Cross-linking procedure
The PVA/SSG nanofibers were cross-linked using glutaraldehyde (GA) solution before the characterization tests to increase the stability of the nanofibrous webs in aqueous environments. The PVA/SSG nanofibers were cut in 50×50 mm dimension and then dried at 40°C to remove residual water content. The dried webs then were placed on top of a sealed desiccator that contains 20 mL of glutaraldehyde 50% w/w in H2O. The samples were kept at room temperature for 4 h to cross-link with the glutaraldehyde vapor. Finally, the cross-linked nanofibers were dried to eliminate the residual GA for two periods of 24 h at 60°C and 45°C temperatures, respectively. The two stages of the drying procedure were performed to completely remove the residual GA from the nanofibers according to the described method by Teixeira et al. 35
Characterization of the PVA/SSG nanofibers
The morphology of the electrospun nanofibers was studied using scanning electron microscopy (SEM) images. The nanofibrous webs were sputtered with a thin layer of platin; the SEM images were then captured using a MIRA3 XMU microscope at the magnification of ×6000.
The Fourier transform infrared spectroscopy (FTIR) was also applied to investigate the functional groups in the PVA/SSG nanofibers (Shimadzu 8600S, Japan), and the spectrum data were recorded in the 400–4000 cm−1 region.
To evaluate the hydrophilicity of the PVA/SSG nanofibers, the static contact angle test was carried out using the Jikan CAG-10 testing machine. The mechanical properties of the nanofiber webs were measured according to the ASTM D-882 standard method, using the Zwick universal testing machine. The specimens were cut in 50×10 mm dimensions, and the tests were carried out under a constant rate of elongation (CRE) condition. The test speed and gauge length were set at 10 mm/min and 40 mm, respectively.
The in vitro degradation behavior of the nanofibers was investigated by immersing the 2×2 cm sample in the PBS solution (pH = 7.4) and incubating it at 37°C for 1, 3, 5 and 7 days. The sample was removed after each interval, rinsed with distilled water, and dried using the vacuum oven for 24 h at room temperature. The weight loss after each interval was calculated using (equation (2)):
The antibacterial activity of the PVA/SSG nanofibers was assessed using the agar disc diffusion method by employing the Kirby–Bauer procedure against the Gram-negative (E. coli) and Gram-positive (S. aureus) microbial species. The specimens were cut in 5 mm diameter pieces and incubated for 24 h at 37°C in Petri dishes (100 mm diameter) containing a Muller Hinton agar medium cultured with the bacteria species. After the incubation period, the diameter of the zone of inhibition (ZOI) was measured using the Digimizer software in terms of millimeters.
Results and discussion
Rheological characterization
Figure 2 exhibits the strain sweep results of the PVA/SSG solutions at different polymer ratios. As it can be seen, the loss modulus (G′) of all solutions was higher than the storage modulus (Gʹ) over the strain range of 0.01–100% at the applied constant frequency of 1 Hz. The results indicated that none of the solutions had viscoelastic properties under the experimented condition (25°C, 1 Hz). As a result, the PVA/SSG solutions at the different PVA:SSG ratios showed viscous behavior. However, it was found that with the increase in SSG content, the storage modulus (Gʹ) increased and the loss modulus decreased. Accordingly, the loss tangent (tan δ = G″/G′) values obtained for the solutions containing 30%, 40% and 50% SSG were, respectively, 9.95, 3.86, and 1.08 at the strain of 1%, implying predominantly viscous behavior of the samples.
36
Viscoelastic behavior of SSG at the concentration range of 0.5–2% (w/w) has been reported by Razavi et al.
25
The effect of dynamic strain on the viscoelastic modulus (Gʹ and G″) of the PVA/SSG solution at different ratios.
The steady shear flow behavior of PVA/SSG solutions is depicted in Figure 3(a). As it is evident, the apparent viscosity of all solutions decreases with increasing shear rate, exhibiting non-Newtonian pseudoplastic behavior. At the shear rate of around 10 s−1, which is attributed to the deformation rate felt in the needle,
37
the increase in the ratio of PVA from 50 to 70%, is resulted in an increase in the apparent viscosity of the solutions from 0.14 to 1.92 Pa.s. Similar results have been reported for the mixed aqueous solutions of agar/PVA.
38
Pseudoplastic behavior in the electrospinning process has been considered a limiting factor, as a very high shear force is applied to the polymer solution at the tip of the needle.37,39 (a). Flow behavior of the PVA/SSG solutions and (b). Values of power-law model parameters (k and n) for PVA/SSG solutions at different polymer ratios.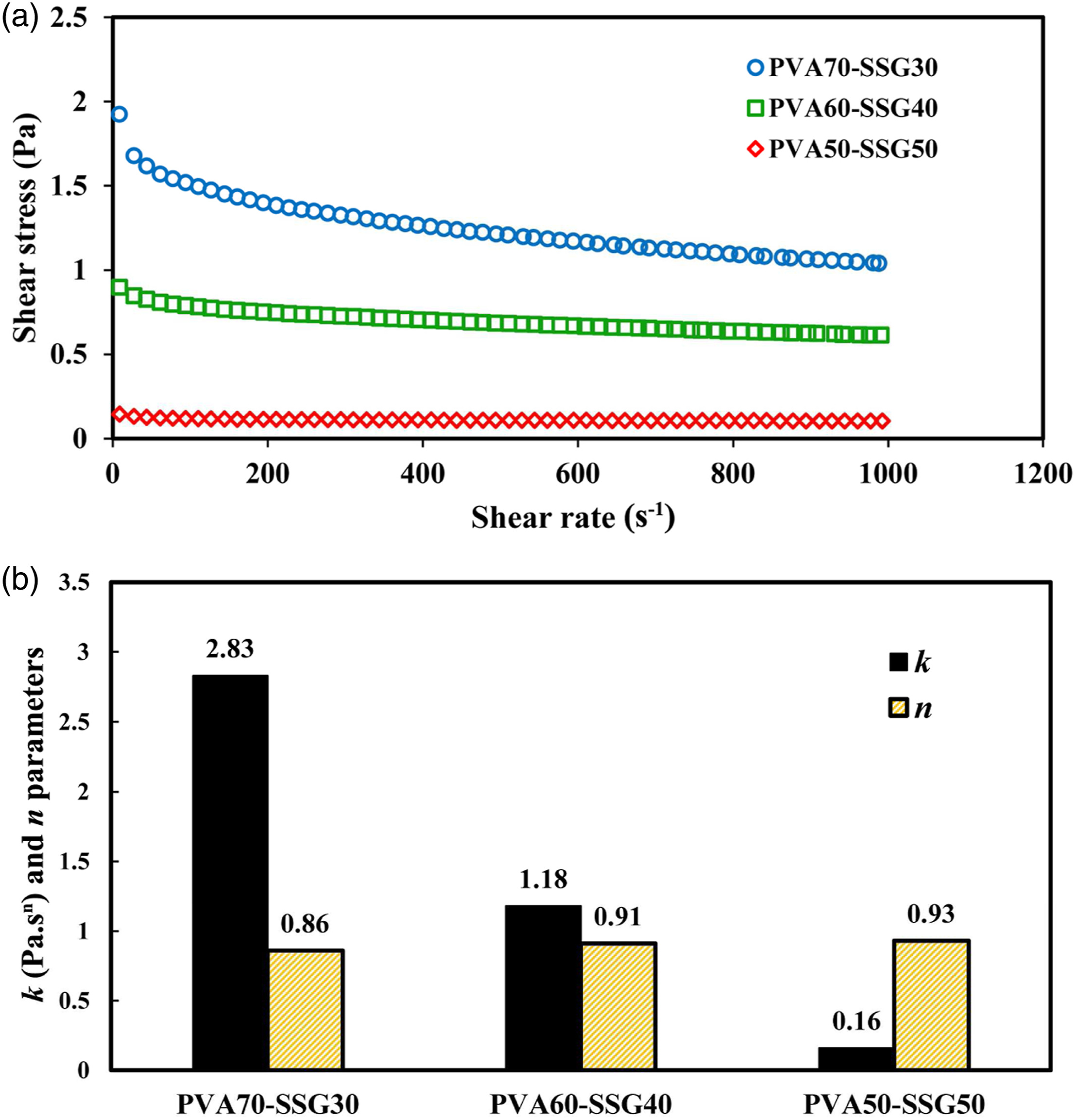
The magnitudes of rheological parameters (k and n) calculated from the power-law model (equation (1)) are summarized in Figure 3(b). The fitting results revealed that this model fitted well the stress–shear rate data (R 2 > 0.99). As the concentration of SSG in the solutions increased from 30 to 50%, the consistency coefficient (k) of PVA/SSG solutions diminished (from 2.83 to 0.16 Pa.sn), while the flow behavior index (n) enhanced (from 0.86 to 0.93), which indicates the higher tendency to Newtonian flow behavior at higher levels of SSG incorporation in the solutions. The shear-thinning behavior of SSG solutions at various concentrations (0.5–2%, w/w) and temperatures (20–50°C), as well as in the presence of different co-solutes, has recently been reported.24,27 Opting an appropriate blend of biopolymers and synthetic polymers, in terms of their types and ratios, can be considered as an essential step in a successful electrospinning process. The relaxation time for polymeric solutions with higher viscosities is longer, 40 so it will be challenging to flow such solutions out of the needle jet towards the collector. Consequently, the balance between the viscosity and the pseudoplasticity of the solution can be a substantial matter in the formation of the stable Taylor cone at the tip of the needle and the production of electrospun fibers. 11
Electrical conductivity of the polymer solutions
Electrical conductivity of the polymer solutions.
Morphology of the PVA/SSG nanofibers

The diameter parameters of the PVA/SSG nanofibers.

The results showed that the diameter of the PVA/SSG nanofibers was in the range of 130–300 nm. Figure 4 represents the SEM images and diameter histogram of the electrospun samples. The histograms of the diameter distribution were plotted using Minitab 20.2.0 software. The electrospinning of the PVA/SSG nanofibers was successfully performed at different conditions. Although there were almost no bead defects visible in different webs, SEM images showed that the produced PVA/SSG nanofibers at the PVA:SSG ratio of 50:50 were more non-uniform and contained broken fibers with a variable diameter (Figures 4(g) to 4(i)). This was also verified according to the CV% value of the PS551, PS552, and PS553 samples, as reported in Table 2. The SEM images of the PVA/SSG nanofibers at different electrospinning conditions and PVA:SSG ratios: (a) to (c): 70:30, (d) to (f): 60:40, and (g) to (i): 50:50.
The coefficient of variation of the nanofibers at 50:50 w/w PVA:SSG ratio was in the range of 19–23%, which was greater than that of other samples. This could be attributed to the more SSG content in the polymer jet during electrospinning. SSG is a polysaccharide polymer exhibiting high surface tension and viscosity. Thus, the flow of the polymer solution through the syringe capillary would be disrupted, leading to the formation of the broken fibers and non-uniformity in the diameters of the resulting nanofibers. This could also be seen for 60:40 (w/w) PVA/SSG nanofibers, as compared to the 70:30 samples. More SSG content led to more variation in the diameter of the nanofibers.
Skewness represents the distribution symmetry of the measured diameters. The negative value of skewness indicates the left-skewed distribution and vice versa. The normal distribution is defined by zero skewness; thus, the closer the skewness to zero, the more the normal distribution. According to Table 2, the sample PS732 had a more normal diameter distribution, with a skewness value of 0.14. The SEM image in Figure 4(b) also shows more uniform fibers for the PS732 sample.
Statistical evaluation was also performed to investigate the significance of the effective parameters on the diameters of the PVA/SSG nanofibers. One-way analysis of variance (ANOVA) test at the 95% confidence level (α=0.05) was then used to study the effect of flow rate, applied voltage and the PVA:SSG ratio on the diameter of the nanofibers. The results showed that only the PVA:SSG ratio had a significant effect on the diameter of the PVA/SSG nanofibers (p-value = 0.003 < 0.05). The results of the ANOVA test also indicated that the increase in the SSG content led to a decrease in the diameter of the nanofibers from 250 to 180 nm. However, the nanofibers had a less uniform morphology compared to the lower contents of the sage seed gum. According to EC values of the PVA/SSG solutions (Table 1), the increase in the SSG content raised the electrical conductivity of the solution. Thus, the charge carrying capacity of the polymer jet was increased and the Taylor jet would be subjected to greater tensile forces in the electric field, which could lead to a significant decrease in the diameter of the nanofibers.
Although the flow rate and the applied voltage parameters had statistically no significant effect (p-value > 0.05) on the diameter of the nanofibers, it is observed from Figure 5(d) that the diameter of the PVA/SSG nanofibers increases with the increase in the flow rate of the polymer solution, except for flow rate of 1.5 mL/h. The increase in the solution flow rate increases the size of the Taylor cone, thus resulting in a higher diameter of the nanofibers. Figures 5(a) and 5(b) show the surface plot and the interaction graph for the effect of the PVA:SSG ratio and voltage value on the diameter of the PVA/SSG nanofibers, respectively. The results indicated that there was also an apparent trend for the effect of the applied voltage on the diameter of the nanofibers. It could be concluded that the increase in the applied voltage from 13 to 18 kV led to the decrease in the diameter of the nanofibers. According to Table 2, the average diameter in different voltages was decreased from 235 to 206 nm. The increase in the applied voltage led to the increase in Taylor jet stretching and formation of a strong electric field. Consequently, the polymer jet could be more drawn in the electric field, leading to the production of thinner fibers. The surface plot and the interaction graph for the effects of the (a), (b) the PVA:SSG ratio and the applied voltage and (c), and (d) the PVA:SSG ratio and the flow rate on the diameter of the PVA/SSG nanofibers.
Figure 6 shows the morphology of the PVA/SSG nanofibers (PVA:SSG 70:30, 15 kV, 1 mL/h) before (Figure 6(a)) and after the cross-linking with the glutaraldehyde (Figure 6(b)). It is observed that the cross-linking has changed the surface morphology of the nanofibers. However, the structure and interconnections of the nanofibers have been retained. The results indicated that there were no apparent defects and failures in the fibers and the integrity of the nanofiber mat was preserved after the cross-linking procedure. The SEM images of the PVA/SSG nanofibers: (a): before and (b): after the cross-linking with GA.
FTIR results
The Fourier transform infrared spectroscopy was used to investigate the chemical structure and functional groups of the PVA/SSG nanofibers. Figure 7(a) shows the FTIR results for PVA, SSG, and PVA/SSG nanofibers at different polymer ratios. Other parameters of the electrospinning except the applied voltage were the same, as compared to the results, based on the SSG content (flow rate: 1.0 mL/h, distance to collector: 13 cm). The FTIR results for (a). The pure PVA, SSG and PVA/SSG nanofibers and (b). The cross-linked PVA/SSG nanofibers with glutaraldehyde vapor (the red-colored region) at different PVA:SSG ratios (70:30, 60:40 and 50:50).
The FTIR spectra of the PVA show the peaks at 1098, 1850, 2941, and 3384 cm−1, corresponding to the resonance bands of C-C and C-O bonds and CH2 and OH groups, respectively.
The SSG FTIR spectrum showed the typical peaks of the pectic polysaccharide polymers in the 800–1200 cm−1 region, known as the fingerprint of the carbohydrates (the violet highlighted region in Figure 7(a)). 44 The peak observed in 890 cm−1 could be attributed to the presence of β-D-mannopyranose units, which could be regarded as the characteristic of the branched polysaccharides. 45 The characteristic peaks in the regions between 950 and 1150 cm-1 corresponded to the C-O, C-C, and C-O-H vibration bands of the glycosidic bonds. The results were, therefore, in agreement with the previous data reported in the literature.44,46,47 The peaks of 1415 and 1604 cm−1 were also assigned the symmetric and asymmetric stretching bands of the C-O-O bonds, respectively. The peaks observed in the 2800–3400 cm−1 region could be attributed to the C-H stretching (2930 cm−1, including CH, CH2, and CH3) bending vibration of the methyl groups that sometimes overlapped with the asymmetric stretching vibration carboxyl groups. The strong peak at 3420 cm−1 also reflected the O-H stretch vibration of the hydroxyl groups.
The FTIR spectra of the PVA/SSG nanofibers represented all characteristic peaks of PVA and SSG. The results showed that the characteristic peaks of SSG in the 800-1200 cm−1 region became more robust with the increase of the SSG content from 30 to 50% w/w. It was also observed that the peak of the stretch vibration of the O-H bond at 3420 cm−1 was stronger due to the synergy effect caused by the presence of hydroxyl groups in two polymers (the green highlighted region).
Glutaraldehyde (GA) is generally used as a cross-linking agent for polymers containing OH groups (mainly polysaccharides) to increase their ability to have intermolecular reactions with PVA, thus increasing the integrity in aqueous environments. 48 The FTIR spectra of the cross-linked PVA/SSG nanofibers with the GA vapor are presented in Figure 7(b) (the red-colored region). The cross-linking of the PVA/SSG nanofibers with GA could be verified by the decrease in the vibration band of the OH group at 3380–3420 cm−1. Stronger peaks were also observed in the 1098–1120 cm−1 region (the green zone in Figure 7(b)). These signals could be attributed to the C-O-C stretching mode of acetal bonds formed due to reaction of the hydroxyl groups in the SSG and PVA with aldehyde groups of the glutaraldehyde. The acetal bonds can also make intermolecular bridges between PVA and polysaccharide chains in SSG. There were also intense bands at 1720 cm−1 and 2820 cm−1, which are corresponded to C=O and C-H stretching, respectively. The observed spectra, therefore, revealed the cross-linking performance of GA between PVA and SSG within the nanofiber structure. 49
Contact angle results
The static contact angle test was also conducted to investigate the hydrophilicity of the PVA/SSG nanofibers. Figure 8 shows the contact angles of the nanofibers produced with different SSG contents. The increase in the content of the sage seed gum was led to a decrease in water contact angle and an increase in the hydrophilicity of the PVC/SSG nanofibers. The ANOVA test at 95% confidence level (α=0.05) was also performed to evaluate the effect of the SSG content on the surface water contact angle of the samples. The contact angle of the PVA/SSG nanofibers at different SSG contents.
The ANOVA results indicated that the SSG content had a significant effect (p-value = 0.001 < 0.05) on the hydrophilic properties of the PVA/SSG nanofibers. The mean of the contact angle was decreased from 92° to 53° with the increase in the SSG content from 30% to 50%. The sage seed mucilage contains functional groups with a high degree of hydrophilicity. Thus, the increase of the SSG content exposed more functional groups (especially hydroxyl groups) to attach to water molecules and increase the hydrophilicity of the surface.
Mechanical properties of the PVA/SSG nanofibers
Mechanical properties of the PVA/SSG nanofiber mats were measured using the universal Zwick tensile tester. Accordingly, five tests were performed for each specimen, and the results were averaged. Figures 9(b) and 9(c) show the main mechanical properties of the nanofiber webs, including the ultimate tensile stress and ultimate tensile strain values, respectively. The results showed that the ultimate stress was increased with the decrease in the SSG content. Natural biopolymers and macromolecules have a wide range of advantages such as the bioactivity, biomimetic nature, non-toxic products, biodegradability, and biocompatibility. Natural polymers have a good performance in mimicking the extracellular matrix and interaction with tissues. These polymers contain different functional groups that can form the attachment sites for cells, and enhance the cell adhesion and proliferation. However, the poor mechanical properties of these materials have limited their application.
50
The sage seed gum could be classified as a natural polysaccharide polymer. Thus, the increase in the SSG content could lead to a significant drop in the mechanical properties of the PVA/SSG nanofibers (from 30 to 50 wt.% of the SSG content). Mechanical properties of the PVA/SSG nanofibers: (a). The stress-strain curves, (b). The mean values of the ultimate stress, and (c). The mean values of ultimate strain at different SSG contents (30, 40 and 50 wt.%).
Figure 9(a) represents the stress–strain profiles of the PVA/SSG nanofibers at different polymer ratios. Clearly, specific trends could be observed at different SSG contents. The PVA/SSG nanofibers at the higher values of polyvinyl alcohol acted as a viscoelastic material with a wide plastic area (magnified box). In contrast, the PVA/SSG nanofibers at 50 wt.% of SSG showed no plastic region at the failure point. The results showed that the increase in the PVA content caused the increase in the ultimate stress of the blend nanofibers. The combination of natural and synthetic polymers is a strategy to improve the mechanical properties and take advantage of the positive properties of both polymers in the form of blended structures. 51 The PVA is a synthetic polymer with good mechanical properties. 52 The PVA has been used both as a carrier for SSG and as an enhancer for the mechanical properties of the blended nanofibers.
The PVA/SSG nanofibers at 50 wt.% of the SSG content could suffer greater strains under the same stress, in comparison to the samples containing 40 and 30 wt.% of the sage seed gum. The observed trend was in agreement with the results obtained by Golkar et al. 53 for the mechanical properties of Plantago major seed mucilage/PVA nanofibers. They also observed an approximately linear mechanical behavior (without the plastic region) for the fibers containing the herbal mucilage. Although, the mechanical properties of the nanofibers are decreased with an increase in SSG content, the obtained values are in a reasonable range for intended applications. 54 The obtained values for ultimate stress and strain of the PVA/SSG nanofibers with the highest SSG content was lied in the range of 5.94–6.67 MPa and 6.04–7.5%, respectively. These values are competitive with the mechanical parameters obtained for nanofibers containing natural gums and polysaccharides.31,53,55
The results of the ANOVA test for the effect of the SSG content on the mechanical properties of the PVA/SSG nanofibers.

Biodegradability test results
The degradation behavior of the PVA/SSG nanofibers in the PBS solution (pH=7.4) was evaluated after cross-linking at 37°C at different intervals (1, 3, 5, and 7 days) to simulate the body fluid environment. The PVA nanofiber mat was electrospun using PVA 10 wt.% solution at the voltage of 15 kV and flow rate of 1.0 mL/h; it was used to evaluate the effect of the SSG on the degradation behavior of the blend nanofibers. Figure 10 represents the degradation behavior of the neat PVA and cross-linked PVA/SSG nanofibers. The in vitro degradation of the PVA/SSG nanofibers at different SSG contents (30, 40 and 50 wt.%).
The neat PVA nanofiber mat lost approximately 95% of its weight after one day of incubation. PVA is a highly hydrophilic and water-soluble synthetic polymer consisting of hydroxyl functional groups that can easily attach to water molecules, causing rapid degradation.56,57 The results showed a slower degradation rate for the PVA/SSG nanofibers. The PVA/SSG nanofibers (at different SSG contents) were degraded by approx. 60–70% of their initial weight after one day of the degradation test. It could be, therefore, concluded that the cross-linking of the PVA/SSG nanofibers using glutaraldehyde (GA) increased the stability of the PVA/SSG nanofibers in the aqueous environment. There was no significant difference in the degradation rate of nanofiber mats with different SSG contents. The degradation of the PVA/SSG nanofibers proceeded with the incubation time up to approx. 95% at a slower rate than observed in the initial degradation step.
Antibacterial activity assay
The antibacterial activity of the PVA/SSG nanofibers was examined against Gram-negative (E. coli) and Gram-positive (S. aureus) species. Figures 11(a) and 11(b) represent the inhibition zone of the cross-linked PVA/SSG nanofibers at different SSG contents against the E. coli and S. aureus bacteria, respectively. The neat PVA nanofiber mat was used as the negative control. PS732, PS642, and PS552 mats were selected for the antibacterial assay in the same way done for the degradation tests (produced at the flow rate of 1.0 mL/h). The diameter of the zone of inhibition (ZOI) was measured using the Digimizer software. Three measurements were performed, and the values were averaged. The mean diameter of the ZOI of different samples is shown in Figure 11(c). The inhibition halo diameter of the PVA/SSG nanofibers against E. coli and S. aureus bacteria were in the range of 9.2–9.6 mm and 11.4–12.8 mm, respectively. The results, therefore, revealed that the PVA/SSG nanofibers exhibited antibacterial effects against both Gram-positive and Gram-negative species at different polymer ratios. The observed results were agreed with the reported antibacterial and antioxidant properties of the sage seed gum.19,20 It has been reported that polysaccharides display their antibacterial activity by damaging the cell membrane, making it permeable and causing the leakage of the vital contents, leading to cell death.58-60 The antibacterial activity results of the PVA/SSG nanofiber mats: (a, b). Disc diffusion test against E. coli and S. aureus bacteria, respectively and (c). The measured diameter of the zone of inhibition against Gram-positive (S. aureus) and Gram-negative (E. coli) bacteria at different SSG contents.
The antibacterial assay also indicated that the SSG containing nanofibers had better activity against S. aureus (Gram-positive) bacteria (Figure 11(c)). Sardarodiyan et al. reported the same results for antibacterial activity of the water-soluble polysaccharides extracted from Balangu seed (Lallemantia royleana) gum against Gram-positive bacteria. 61 It was also found that there was no significant difference in the ZOI of the nanofiber mats for different contents of SSG against the same bacteria.
Conclusion
In this study, a novel polysaccharide-based nanofibers containing PVA and sage seed gum (SSG) extract was electrospun. The PVA/SSG solutions were evaluated for rheological properties. Electrospinning was performed at different conditions, and PVA/SSG nanofibers were cross-linked by glutaraldehyde (50 wt.%) to increase the integrity of the mats in the aqueous environments. The physical and chemical properties of the produced mats were investigated using SEM, FTIR, contact angle and tensile testing methods. The in vitro biodegradation and antibacterial activity assays were also performed to evaluate the bioactivity of the PVA/SSG nanofibers. The rheological studies also revealed that the PVA/SSG solutions had a predominantly viscous behavior (at 1 Hz and 25°C), exhibiting a more pseudoplastic behavior at the higher PVA incorporation levels. The results showed that the electrical conductivity of the PVA/SSG solutions increased with an increase in the sage seed gum content, due to the high negative zeta potential of the SSG in the solution. The SEM results indicated that the PVA/SSG nanofibers could be produced at the selected conditions without bead defects within the range of 130–300 nm in diameter. The SEM images also demonstrated that although the morphology of the nanofibers was changed by cross-linking, the integrity of the structure and form of the fibers was preserved. The ANOVA analysis also indicated that the SSG content of the nanofibers had a significant effect on the nanofiber diameter at the 95% significant level. The contact angle tests also verified the hydrophilicity of the specimens containing high levels of SSG (contact angle = 53°–92°). Further, mechanical testing indicated that the SSG content had a significant effect on the mechanical aspects of the PVA/SSG nanofibers. The increase in the SSG content to 50 wt.% was led to changing the mechanical response of the nanofiber mats to semi-linear stress-strain curves. The degradation test also demonstrated that the cross-linking of the PVA/SSG nanofibers with GA resulted in the slower degradation rate (60–70% of the initial weight after 24 h) and increased the stability of the mats in the PBS solution. The antibacterial activity assay also reflected the bactericidal performance of the PVA/SSG nanofibers against Gram-positive (S. aureus) and Gram-negative (E. coli) species. The PVA/SSG nanofibers can be, therefore, considered as a candidate for use in antibacterial applications such as wound healing.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to express their sincere thanks to the deputy of research of the University of Bonab for the financial support.


