Abstract
As a consequence of the complex mixture of oils, alkalis, salts and other corrosive substances in crude oil and its products, internal corrosion of pipelines is a significant issue for the petrochemical industry, as corrosion reactions are easily triggered between these substances and the inner surfaces of conveying pipes. Corrosion can damage equipment, increasing accident risk and reducing safety procedures effectiveness. Since corrosion accounts for a significant portion of the world’s gross domestic product each year, finding methods to prevent corrosion and increase pipeline corrosion resistance has become an essential topic in oil production and industry. There is an unavoidable risk that the oil transportation system will be severely damaged during the extraction and installation of oil and natural gas wells that contain large volumes of carbon dioxide, condensate oil, hydrogen sulfide, and brine. Oil and gas are collected and transported in pipelines using a variety of fluids, including gas, water, hydrocarbons, and solids through multistage flow mediums. Due to this, as the oil and gas field develops, the transport medium becomes heavier due to the injection of water, which exacerbates the corrosion of pipelines. Thus, research institutions involved in corrosion resistance have paid close attention to pipeline internal corrosion mechanisms and technologies, and organizations seeking to improve corrosion resistance have increasingly turned to them as a focal point. This study simulates the corrosion environment of an internal pipeline based on the actual operating parameters of oil pipelines. It was intended to indicate the extent to which they could influence the development and propagation of corrosion.
Introduction
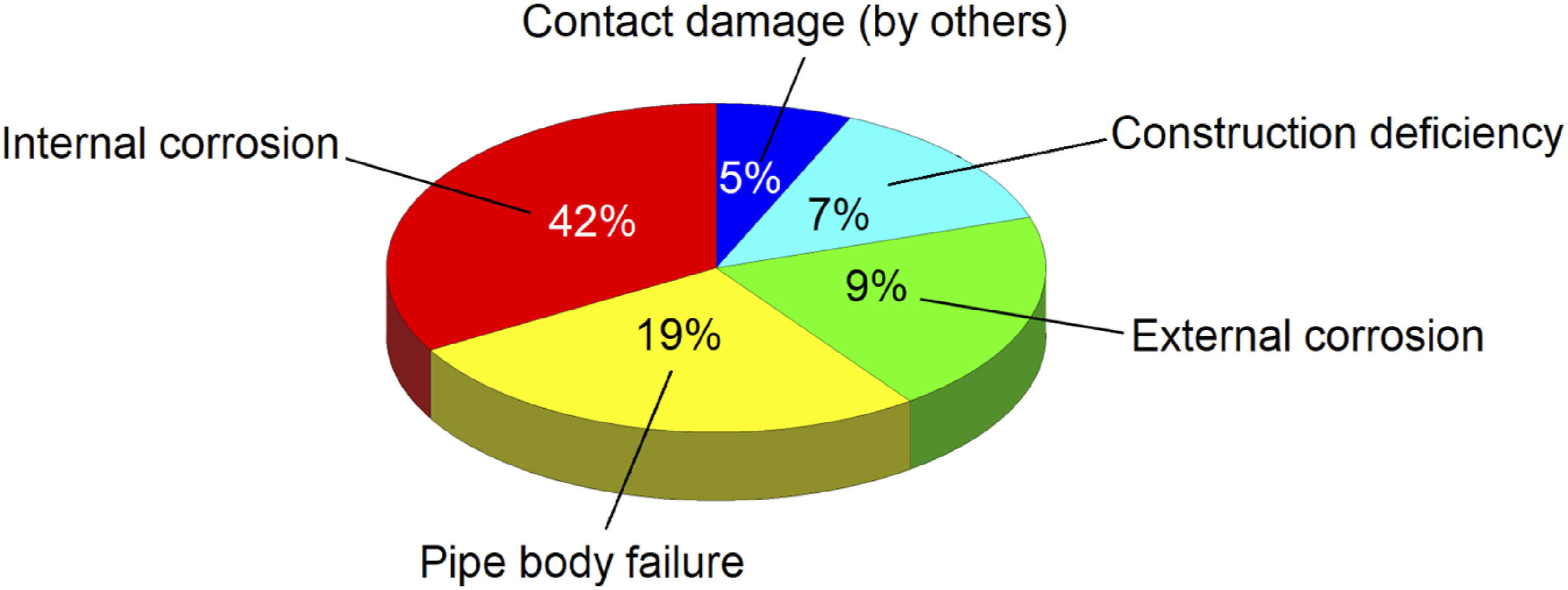
As far as the oil and gas industry is concerned, pipelines continue to be viewed as the most efficient, safest and most economically efficient form of mass transportation. However, in the meantime, even a small malfunction within a pipeline could have disastrous consequences in the future. Corrosion of pipelines, which is a gradual degradation of metal caused by chemical reactions, is a significant concern and the most common and dreaded causes of failure in many industries, including the oil, gas, and water industries. It can lead to pipeline failure, leaks, and severe safety hazards if it is not adequately controlled. Material deterioration is the outcome of a naturally occurring electrochemical reaction due to the nature of the material being transported and the material the pipeline are made of.1–8 According to a report issued by the Alberta Energy Regulator, internal corrosion of oil pipelines accounted for the majority of pipeline failures in 2023 as shown in Figure 1. Corrosion was responsible for approximately 51% of failures that year, with internal corrosion responsible for 42% and external corrosion responsible for 9%. There was multi-mechanism corrosion involved in pipe failures caused by internal corrosion as mentioned in AER report in 2023. There are also other sources of internal corrosion, including under-deposit corrosion (19%), corrosion influenced by microbes (14%), corrosion caused by CO2 (6%), and corrosion caused by internal coatings (5%).
9
As a result, it is imperative that oil and gas pipelines are properly assessed for corrosion and their remaining service life assessed. There is a risk associated with internal corrosion that must be recognized by personnel in the petroleum industry. It is possible for the corrosion of the pipeline to cause cracks in the pipe, which can pose a threat to the environment as well as to workers. For example, the produced water from the extraction of crude oil may contain highly corrosive components, such as salts, liquids, and gases. CO2 and H2S in aqueous form have long been regarded as a cause of pipeline corrosion in the oil and gas industry.10–18 The most significant types of oil pipeline failure in 2023.
9
Oil pipelines are subject to this type of corrosion throughout their life cycle, which poses a serious threat to their integrity. It is well known that carbon steel has a relatively low corrosion resistance, but it is commonly used for oil and gas pipelines due to its high mechanical properties, low price, and widespread availability. It has been noted that this issue has once again resurfaced in the oil and gas extraction industry as a result of the use of cutting-edge methods such as carbon dioxide injection for increased oil recovery and the extraction of deep natural gas reservoirs.19–24 As shown in Figure 2, corrosion can be exacerbated or lessened in severity depending on a number of factors. A significant point to note is that there are often interconnections between the characteristics that can be broadly categorized into eight categories and that these characteristics may have opposing effects on corrosion processes. It is of critical importance for corrosion engineers to understand and investigate the effects of each parameter, as well as identify the predominant corrosion mechanisms within a given environment. Corrosion engineers can implement the conclusions they reach or the recommendations they make through mechanical or chemical means, such as removing the source of oxygen intrusion or limiting the growth of bacteria.24–29 Internal corrosion influential factors.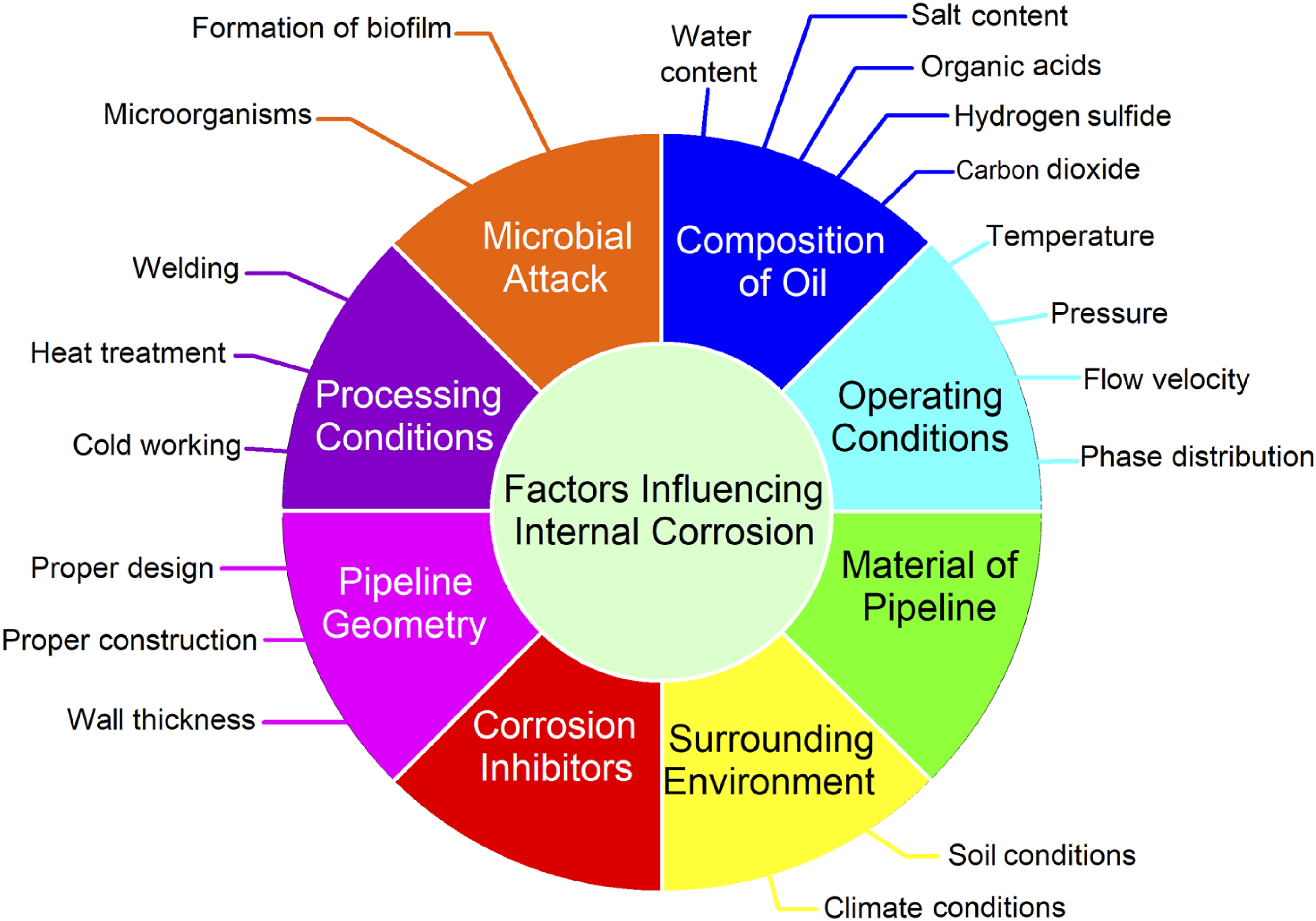
Without lining, internal pipelines are susceptible to deterioration over time. The immediate intervention is required in the case of destructive corrosion. As liners, polymers are the most effective materials. As corrosion inhibitors, polymers have been used for a long time in the oil and gas pipeline sector. As a result of the polymer, a barrier is created between the fluids being conveyed and the metallic host pipe. Depending on the flow of fluid and the state of the pipe, the type of polymer material to be used is determined. A variety of polymers find extensive use in a number of industries, including high density polyethylene (HDPE), ethylene butyl acrylate (EBA), fusion bonded epoxy (FBE), and polypropylene (PP). Oil and gas companies have utilized polymeric linings to restore ageing gravity pipes, including sewers, in the oil and gas industry. In spite of this, since these environments can have an adverse effect on polymers, such as deterioration of their mechanical and physical properties such as hardness and tensile strength, they can shorten their service life. Humidity, sea environments, broad temperature variations, and exposure to ultraviolet light are examples of these environments. The pipeline’s polymer lining is depicted in Figure 3.30–35 Pipeline lined with polymer.
30
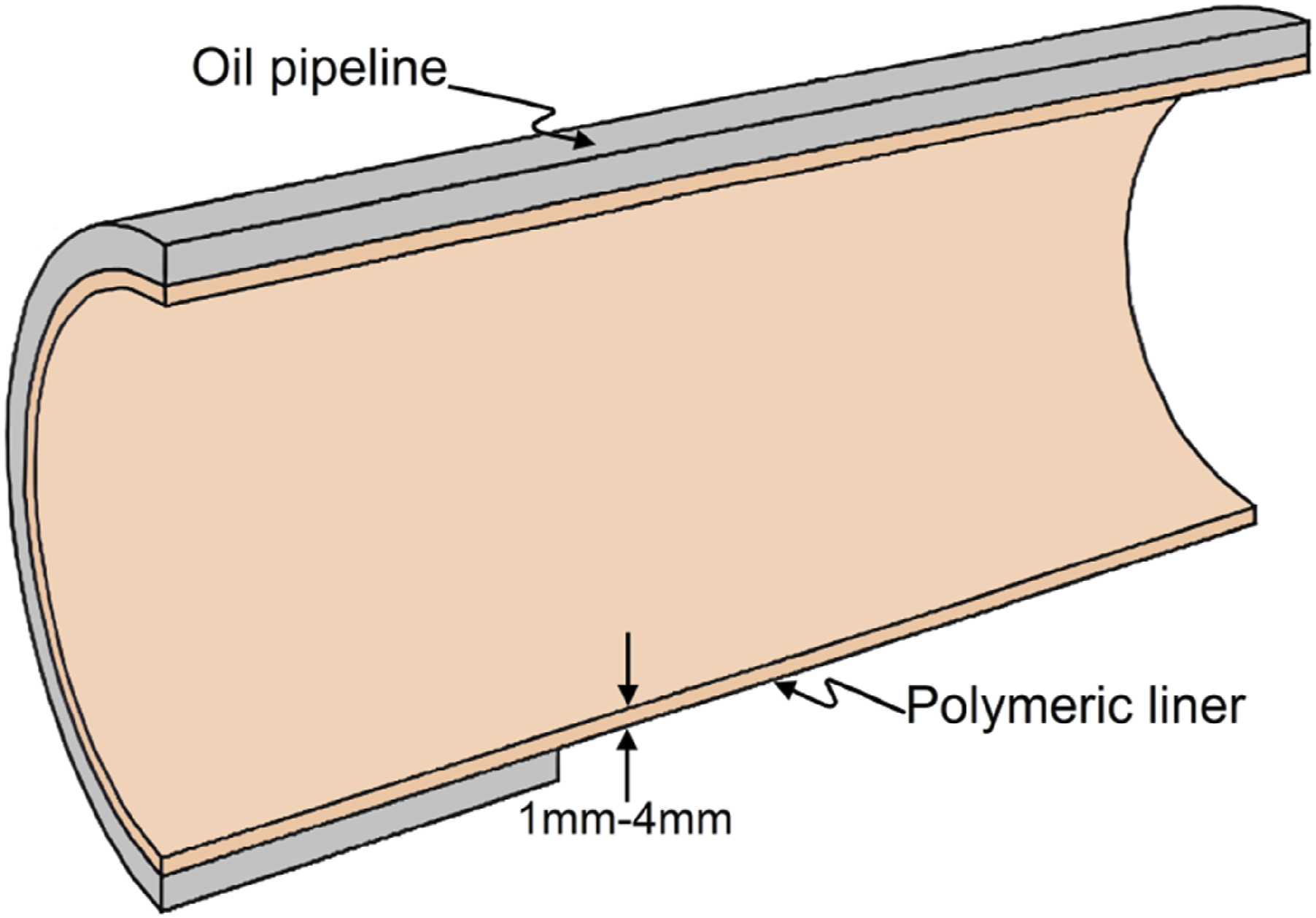
The objective of this review paper is to provide a comprehensive overview of polymeric liners designed for pipeline integrity and corrosion prevention, including their role, materials, performance, challenges, and future trends. In addition to explaining why polymeric liners are used in oil pipelines, this review will compare the various types of polymers and discuss their benefits in reducing maintenance costs.
Historical background to polymeric liners in pipeline
Historically, the development of pipeline materials is closely related to the history of pipeline corrosion. As societies became increasingly dependent on steam, oil, and later natural gas, the need for reliable transportation increased. The growth of pipelines became a vital component of the infrastructure, however, their susceptibility to corrosion soon became a major concern. In comparison to the many types of corrosion that can occur in pipelines, internal corrosion has been a major concern for the industry since the early days of pipeline technology. Internal corrosion occurs when the materials being transported interact with the inner surfaces of a pipeline. In contrast to external corrosion, which occurs as a result of the environment outside the pipeline, internal corrosion is invisible. As pipelines were developed rapidly, concerns about internal corrosion and the problems it caused increased. This was especially true when industries began to transport corrosive liquids and gases.36–39 As an example, the Trans-Alaska Pipeline System (TAPS) suffered from internal corrosion issues despite its robust construction, requiring repair and maintenance. Moreover, in 2010, a natural gas pipeline in San Bruno, California exploded as a result of internal corrosion, resulting in fatalities and extensive property damage. Also, at the Edmonton Tank Farm, 165,000 L of crude oil spilled from a redundant pipe on August 1981 as a result of internal corrosion on a tank.40–43
In the 19th century, metal pipes began to be used in a variety of industrial sectors, particularly in the distribution of water. The first pipelines were constructed of cast iron or wrought iron, which was prone to corrosion when transporting water containing a high oxygen content or acidic properties. Corrosion of pipelines, particularly internal corrosion, first became apparent in water distribution systems in major cities, leading to reduced flow efficiency, leaks, and eventually complete collapse. As the oil industry developed rapidly in the late 1800s and early 1900s and large oil fields were discovered throughout the United States and Russia, the demand for pipelines increased rapidly. Corrosion soon became a major problem for the industry as pipelines transporting crude oil, natural gas, and other hydrocarbons were largely made of wrought iron and steel. Internal corrosion is an issue experienced by oil pipelines, which is worsened when crude oil is transported in water. In cases where the transported crude oil contains water, sulphur compounds or other impurities, which react with the pipeline material, corrosive compounds such as hydrogen sulfide are formed, causing significant internal damage that can lead to leaks in the transported material, resulting in major environmental problems.44–47
Since the mid-20th century, the oil and gas industry has experienced rapid growth, which has resulted in longer and more complex pipeline networks transporting more aggressive and diverse materials. With this growth came an increase in internal pipeline corrosion, which necessitated the development of preventive measures for control of corrosion. In the 1930s, cathodic protection and corrosion inhibitors were introduced for the protection of pipelines, which significantly reduced corrosion rates. A number of protective coatings were developed for pipelines during the 1950s and 1960s, including coal tar enamel and epoxy coatings, which could be applied on both interior and exterior surfaces, providing additional protection against corrosion, as these coatings were intended to prevent direct exposure to corrosive environments from damaging the metal.47,48
In the past few decades, synthetic polymers, such as polyethylene (PE), polyvinyl chloride (PVC), and polytetrafluoroethylene (PTFE, also known as Teflon) have opened up new possibilities for corrosion protection. There are several benefits associated with these polymeric materials, such as their chemical resistance, flexibility, and relatively low cost. Liner materials made from polymers were initially used to line pipeline interiors, providing a protective barrier between the metal inside and the fluids inside. Corrosion damage prevention improved greatly in the 1960s as sophisticated techniques were developed for identifying, detecting, and assessing damage inside pipelines, including the application of smart dye technology to inspect pipelines without excavation. As a consequence, these inspection tools are capable of detecting signs of internal corrosion, such as thinning walls or potholes, which allows for targeted maintenance and repair of structures.36,46,47
Oil and gas companies were subjected to increased regulatory scrutiny in the 1970s and 1980s due to increased environmental demands and more stringent requirements to ensure pipeline integrity and corrosion management. Pipeline operators continue to develop advanced coatings, inhibitors, and materials to extend the life of their pipelines. As a result of this development, polymers with greater durability and chemical resistance have been developed, including high-density polyethylene (HDPE), epoxy-based coatings, and cross-linked polyethylene (PEX). It is important to note that these materials were specifically designed to withstand the harsh conditions encountered within pipelines, including high pressure, temperature fluctuations, and exposure to a wide range of chemicals. Commercialization of polymer liners began with manufacturers developing specialized products to suit different pipeline types. The use of HDPE liners in pipelines carrying water and wastewater became commonplace, whereas the usage of PTFE and epoxy-based liners became prevalent in pipelines carrying chemicals and hydrocarbons that are more aggressive. The use of polymer liners became the standard solution in many industries, including oil and gas, water treatment, and chemical processing, by the late 20th century. Providing a convenient and cost-effective alternative to extensive excavation and infrastructure replacement, thermoplastic liners can be inserted into existing pipelines and expanded to form a tight fit.32,49–52
Increasingly, polymer liners are being designed to improve their performance and durability in the 21st century. As part of the innovative process, multilayer liners have been developed, which combine different polymers to enhance the chemical resistance and mechanical strength of the product. In addition, improvements have been made to installation techniques, including trenchless lining technology, which minimizes disruption during lining. Smart materials have recently been incorporated into polymer liners, allowing for the monitoring of pipeline conditions in real-time. The smart liners are capable of detecting changes in pressure, temperature, or chemical composition in order to provide valuable information that can be used to prevent failures before they occur53,54 (Figure 4). Timeline scheme for the most development of polymeric liners in pipeline.
Characteristics of polymeric liners
As crude oil is transported through pipelines, it is exposed to harsh and challenging conditions that pose a great challenge to both the materials used to construct these pipes as well as the systems that monitor and maintain them. It is important to note that sour crude oil can contain sulphur compounds, including hydrogen sulfide, which are corrosive, and prolonged exposure to these substances may cause corrosion to pipelines.32,38 A highly corrosive environment is also created by the presence of water and brine in oil as a result of drilling operations. Furthermore, drilling operations can also cause abrasive particles such as sand, silt, and other sediments to enter with the oil, which may corrode the inner walls of pipelines, especially at high speeds and pressures. Hydrogen sulfide (H2S) in sour oil is highly corrosive and will cause significant degradation of the material. It is well known that carbon dioxide (CO2) is soluble in water and forms carbonic acid, which causes steel pipelines to corrode. In addition, bacteria can contribute to the corrosion of pipes. There are also conditions associated with transport that include high pressures, extreme temperatures, and mechanical stresses. Keeping pipelines intact while preventing environmental damage requires addressing these extreme conditions. This condition is mitigated by the use of polymeric liners and protective coatings. In addition to protecting the inner surface of pipes, polymer liners also reduce maintenance costs, prevent leaks, and reduce environmental pollution by reducing the possibility of pipeline failures. Polymers must have specific properties to withstand the harsh conditions of oil transportation, including chemical exposure, temperature changes, and mechanical stresses, and the following are key properties55–65: 1. Chemical resistance: Considering it is in contact with crude oil, gas, and other hydrocarbons, it is essential that the polymer has the ability to resist degradation affected by these materials, as well as degradation caused by sulphur compounds, brine, and other oil-related chemicals. 2. High temperature resistance: It is important that the polymer has thermal stability at high temperatures, since oil transport often occurs at temperatures that reach 120–150°C. In cold climates, polymers must also remain flexible at low temperatures, especially if they are to avoid becoming brittle or cracking when frozen. 3. Excellent mechanical resistance: It is necessary that the polymer liner has high abrasion resistance in order to withstand the abrasion of materials such as sand, paraffin, and other particles as they are transported. Also, it offers high tear resistance, which prevents it from tearing when exposed to high pressures. As a result of oil flowing at high speeds, the polymer liner must also be friction and wear resistant. 4. Low permeability: Oil and gas leakage can be prevented by the polymer having a low permeability. 5. High adhesion resistance: It is imperative that the polymer liner adheres strongly to the inner surface of the pipe, particularly in high pressure environments, to prevent it from delaminating. 6. Thermal expansion: It is imperative that the polymer has a thermal expansion compatible with the pipeline material in order to avoid stress build-up that can result in cracking or separation of the pipe. 7. Durability and application flexibility: In pipelines that are long and narrow, the polymer should be durable and easy to apply or install. 8. Long-term performance and aging resistance: In order for a polymer liner to perform effectively, it must be able to withstand harsh oil and gas environments.
Categories of polymers used in liners
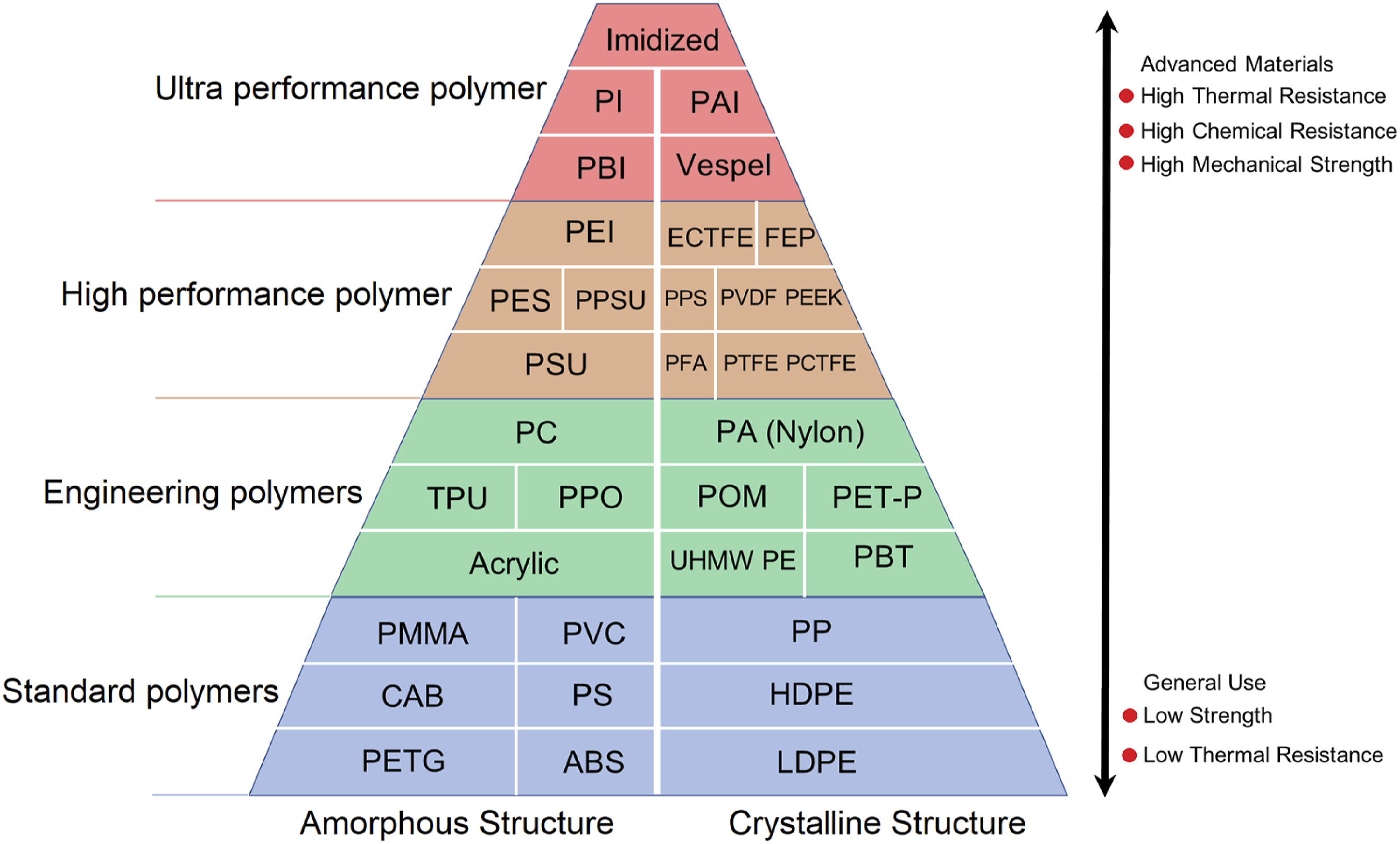
Based on their physical, chemical, and thermal characteristics, polymers are also categorized as high-performance, engineering, or standard (see Figure 5). Due to their poor mechanical properties, standard polymers, such as polypropylene, polystyrene, polyethylene, and polyvinyl chloride, should only be applied at temperatures below 100°C. It is common for them to be produced in large quantities and used in many low-temperature applications, such as packaging films, containers, clothes, and other items where their mechanical properties are not as deemed essential. In contrast to standard polymers, engineering polymers exhibit superior mechanical and thermal properties in the temperature range between 100 and 150°C. Additionally, due to their high price, they are primarily used in low-volume applications. Engineering polymers may be composed of polycarbonates, polyamides, or polyoxymethylene (POM). In addition to liquid crystallized polymers and fluoropolymers, high-performance polymer composites (HPPs) also include semicrystalline polymers such as polyphenylene sulphide and polyetheretherketone (PEI), as well as amorphous polymers such as polysulfones (PSUs) and polyetherimides (PEIs). The characteristics of high performance polymers (HPPs) include their ability to maintain desirable qualities under adverse conditions, such as high temperatures, high pressures, or corrosive environments. Polymers that resist heat, advanced engineering materials, and polymers that are resistant to high temperatures are all names given to these materials. It is necessary for a polymer to meet a series of requirements in order to be classified as high performance.69–72 Categories and types of polymers used in liners.
69
Optimal application manners for polymer liners
It is common to use heat-shrinkable polymer tapes to insulate welded pipe joints. A liquid epoxy primer coats these tapes, and their designs are often comparable to those of three-layer polyethylene pipe coatings. In spite of this, current polymer coating materials and processes do not always meet the necessary standards for making sure that polymer coatings adhere to the outside surfaces of other equipment and structures, as well as the interior surfaces of pipes. Due to the versatility of fillers and additives available in paintwork materials, it is possible to create materials for a variety of working conditions, even for the most severe ones. Recently, coatings based on materials such as urethane, epoxy, and urethane dissolved in organic solvents have become increasingly popular for painting tanks and other metal structures and insulate pipes. In most cases, coatings are applied using pneumatic or airless sprayers.73–78 Figure 6 illustrates a polymeric liner applied to a pipeline. (a) Internal liners and (b) external coatings for pipelines.
79
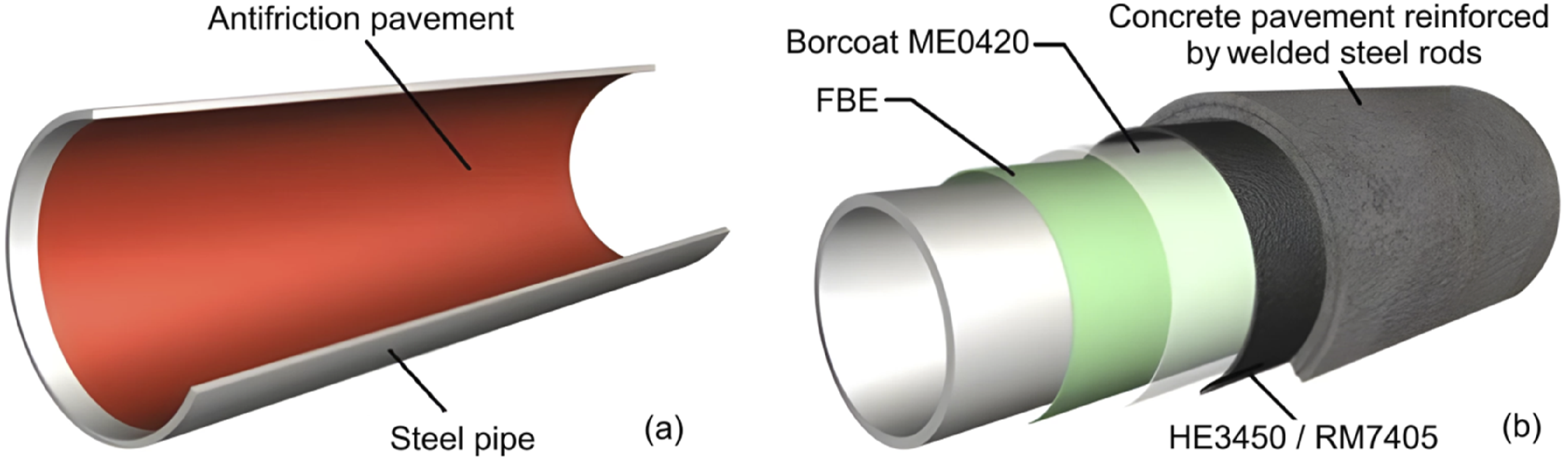
As a result of the application of thick coatings (1.5–2.5 mm), multiple layers of anticorrosive agent must be applied, allowing intermediate drying times between layers, and operating in a factory environment designed to protect workers and the surrounding environment from solvent vapors.77–83 It has been determined from both domestic and international sources that, depending on the chemical composition and composition of the mixture, “hot” airless spray application of solvent-free two-component polyurethane and epoxy-polyurethane systems at temperatures ranging from 50°C to 70°C for the “A + B” reaction mixture is the most appropriate method for the application of factory protective insulation to valves, fittings, bends, and other components. In addition, it is possible to apply coatings to metal structures that are thick enough to prevent corrosion over an extended period of time. In combination with factory-made polyethylene coatings, these coatings largely satisfy technical criteria.76,80–82 Generally, two-component polyurethanes and epoxy-polyurethane coatings are applied as follows: abrasive cleaning of the surface, application of the coating, primer drying, external spraying of polyurethane or epoxy-urethane coating, and curing. It is recommended that coatings be applied to a dry, clean surface no later than two to 3 h after cleaning has been completed. The coatings should be applied at a maximum temperature of 5°C and a humidity level of 80%. As part of the technology under consideration, layers of the same material must be deposited wet on wet, without drying in between the layers. A high degree of cohesive bond is ensured between the layers of the protective material as a result of this method. Prior to applying the outer layer of polyurethane coating, the epoxy primer must cure for a minimum of 4–25 h.83,84
Alternatives to the protective coatings previously mentioned are epoxy materials, phenolic coatings, epoxyphenolic coatings, novolac coatings, polyamide coatings, polyurethane coatings, and polyethylene coatings. Epoxy powder paints offer heat-resistant, water-resistant, and cathodic flake-resistant coatings. The disadvantages of these materials include limited impact resistance, particularly at low temperatures, higher costs, and limited usage in factories. Furthermore, the technology involved in their application is more complex and requires parts to be heated to temperatures between 220°C and 240°C, which results in an increase in energy costs. In order to extend the overhaul period of pipeline systems, it is crucial to provide effective and durable anti-corrosion coatings, since the decommissioning of equipment results in severe economic losses. It is important to choose the best anti-corrosion protection during the design stage of pipeline complex construction so that it can last for the equipment’s entire service life (15 years or more), requiring only local cosmetic repairs during the course of its lifespan. A multilayer coating consisting of epoxy primers and weather-resistant polyurethane enamels is widely used to protect the surfaces of equipment. As a result of this combination, structures are protected against corrosion over a prolonged period of time, while also maintaining a pleasing appearance over the course of their service lives. The coatings used on oil or gas terminals at the coast, as well as offshore drilling platforms, include zinc-containing epoxy primers and zinc ethyl silicate primers, which are followed by polyurethane enamels. In addition to offering protection against abrasion and wear, polyorganosiloxane compounds prevent icing on structures.81,85–88
Aging and degradation of polymeric liners in pipelines
As we know, the main difference between aging and degradation, that ageing refers to the natural breakdown of the polymer liner’s properties over time, regardless of the presence of extreme or abnormal conditions. The interaction between the material and the environment is the primary driving force behind this phenomenon. In contrast to aging, degradation is a more aggressive process which occurs as a result of chemical, physical, and mechanical factors, which may be exacerbated by environmental factors such as oil, chemicals, or mechanical forces. As polymer liners age, they become more susceptible to degradation.61,89–91 The brittleness of a liner may increase over time due to thermal aging or oxidation, increasing its susceptibility to cracking or breaking under mechanical stress or chemical exposure. For an oil pipeline system to remain intact, it is crucial to manage both aging and degradation. It is possible to mitigate these effects in a number of ways, including the use of chemical stabilizers, regular monitoring, and the choice of a polymer appropriate for the environmental conditions. In pipelines, polymers were degraded by oxidation, hydrolysis, permeation, and absorption. This failure can be attributed primarily to permeability. Hydrocarbons cause polyolefins to swell because of their similar chemical structures. The presence of water in polyamide causes it to degrade at higher temperatures.30,51,66,92–95
A polymer liner undergoes permeation when molecules from the transported fluids penetrate it, such as hydrocarbons, acidic gases (CO2, H2S), and water. In this process, the liner can be swollen, plasticized, and have the mechanical properties reduced. Due to the fact that this is a naturally occurring phenomenon, it is imperative that attention be paid to the issue. There are two factors that control permeation, (1) diffusion within molecules and (2) the solubility of the permeant within the polymer. An indicator of permeant adhesion to a polymer is its solubility. Initially, permeate is adsorbed onto the surface of the polymer, and then it diffuses from a concentration that is greater to one that is lower.51,66,92,93 Figure 7 represents the limitations of working temperatures for the main polymers. The limitations of working temperatures for the main polymers.
66
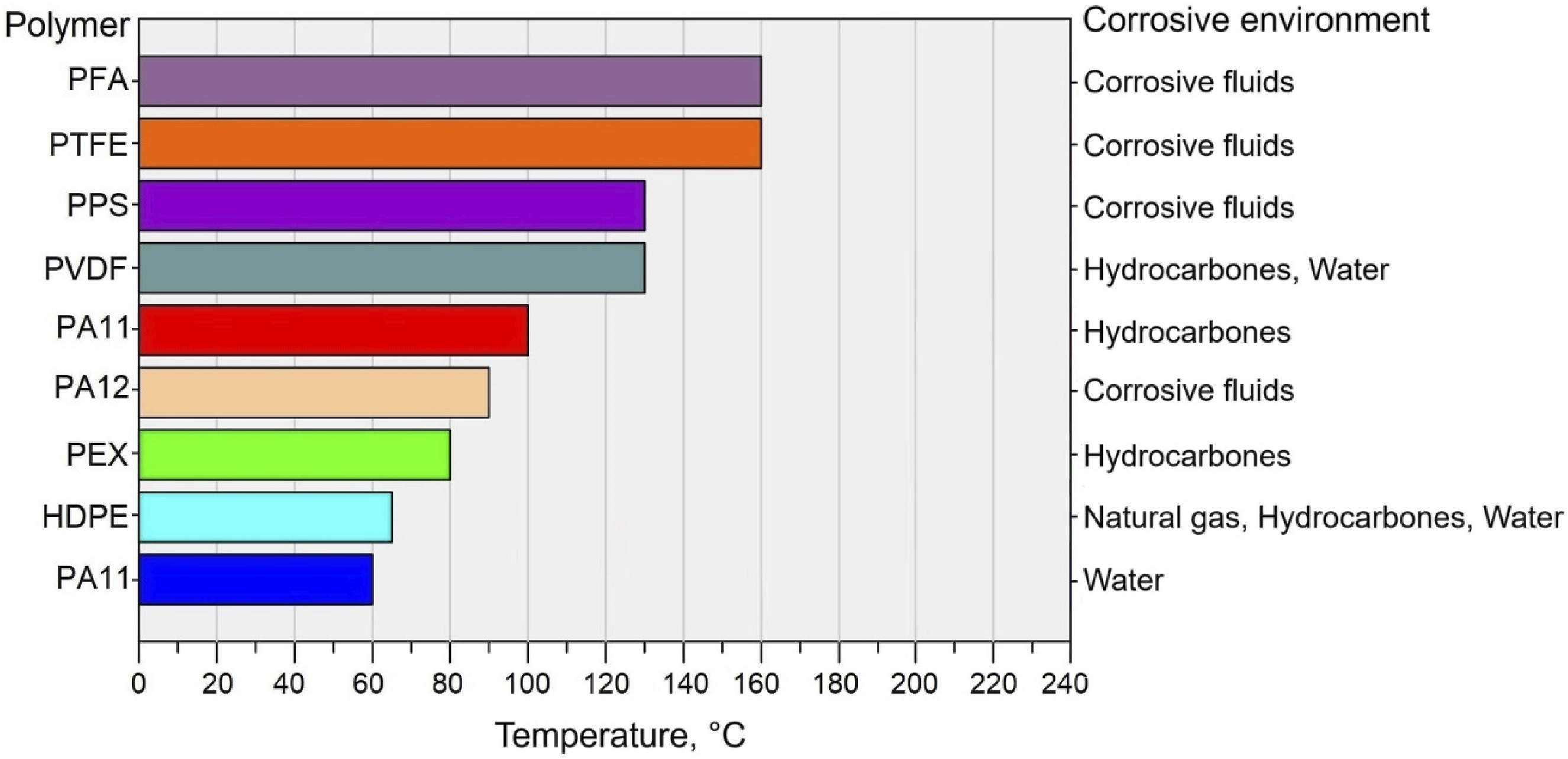
As a result of thermal expansion and hydrocarbon swelling, stresses can develop in the liner material, resulting in buckling or collapse. Despite the fact that thermoplastic materials are flexible, gases, vapours, and water can freely pass through them. It is important to note that the most important parameters that determine permeability are the liner thickness, partial pressure, crystallinity percentage, crosslinking, size of the permeant, and the distance between the liner and the host steel pipe. There is a strong correlation between temperature and permeation. It has been shown that temperature has a significant impact on gas permeability. There is a direct correlation between diffusion and permeation characteristics and temperature. Temperature-related increases were observed, whereas solubility decreased.94–99
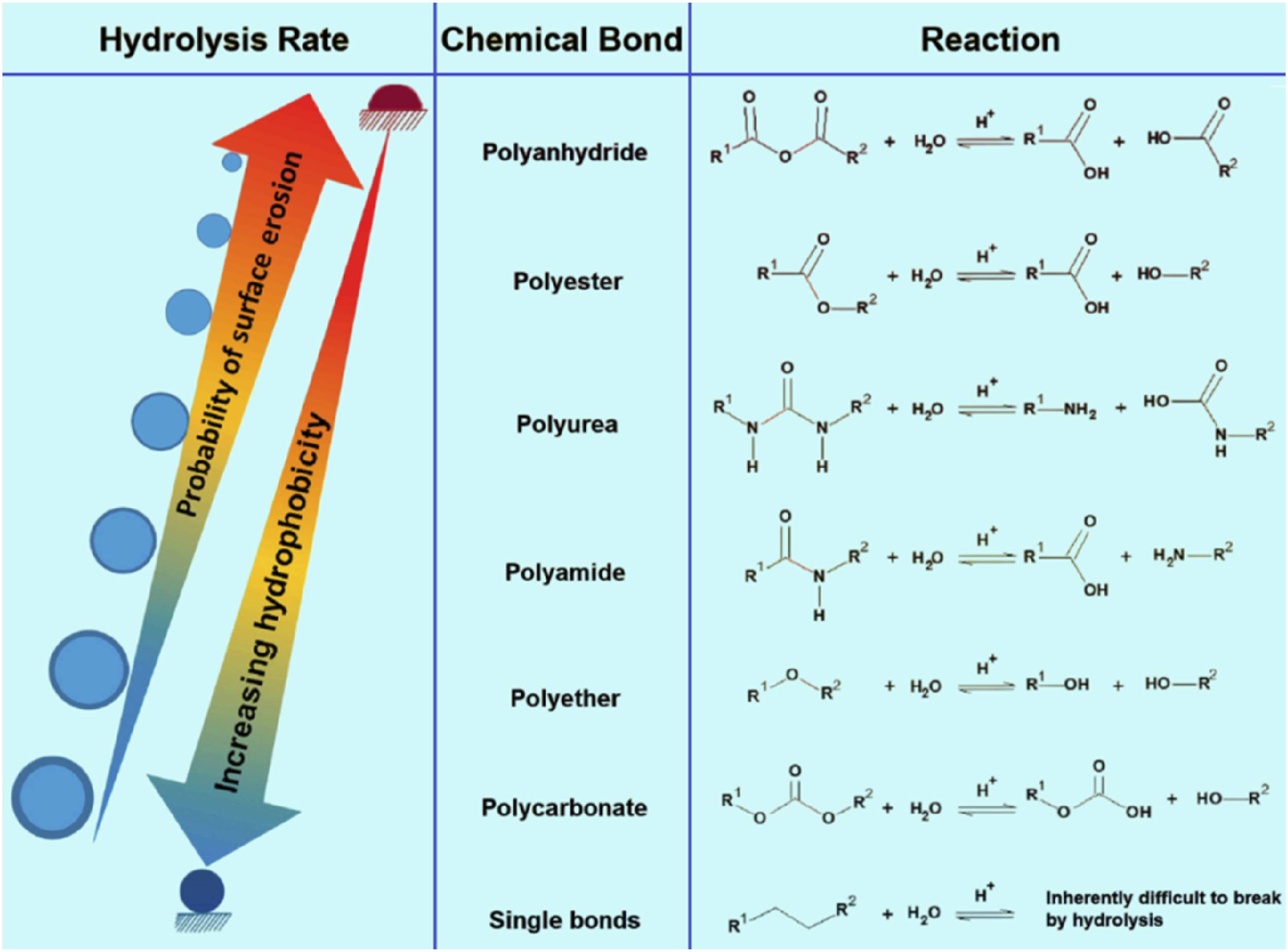
Meanwhile, hydrolysis of linings in oil pipelines occurs when water or moisture in the pipeline reacts with the polymer lining material, which results in the polymer chains being broken when water molecules interact with the polymer backbone or side groups. The chemical bonds between the polymers are broken as a result of this interaction, which in many cases occurs as ester or amide bonds. Polyamides, such as PA11 and PA12, are closely related to hydrolysis. During hydrolysis, the molar mass of the lining decreases, water permeability increases, and mechanical properties are impaired. The lining is adversely affected when fluids are transported inside the pipeline, such as oil or gas, causing corrosion and other damages. Utilizing polymers with higher water stability, such as cross-linked polyethylene (PEX) or fluoro-polymers, can reduce hydrolysis risks.30,66,100,101 The hydrolysis rates of the main polymers susceptible to degradation when exposed to moisture are ranked in Figure 8. Hydrolysis rates causing degradation of major polymers upon moisture exposure.
102
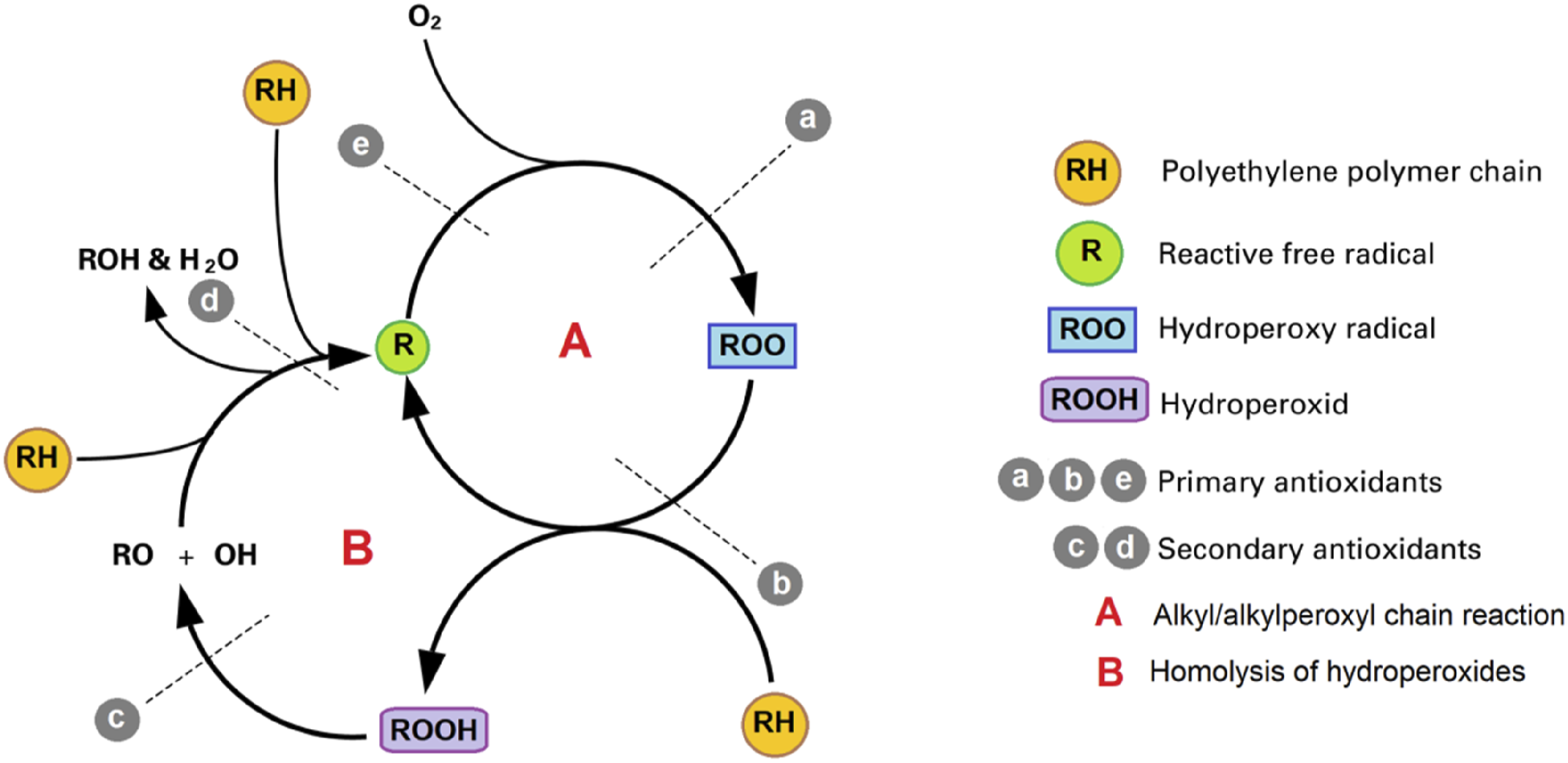
Oxidation of pipeline liners can lead to significant integrity and performance issues because of its impact on pipelines. It is true that polymers wear over time, which causes liners to deteriorate, providing a barrier to corrosion and friction reduction. The oxidation process causes polymers to undergo chain scission (breaks in their molecular chains) leading to the formation of hydroxyl, carboxyl, or ester groups. This process causes the material to become brittle and more susceptible to cracking as a result.30,103,104 As a general rule, polymer liners undergo three main stages of oxidation: initiation, propagation, and termination. Free radicals are formed during the initiation stage when heat, UV light, or chemical agents react with each other. A high temperature can cause polymer chains to collapse and reactive species to form, resulting in thermal degradation. The oxidation process is inter the propagation stage during which free radicals react with oxygen (O2) in the environment, resulting in peroxy radicals being produced. Through the reaction between these peroxy radicals and the polymer chains, hydroperoxides and additional radicals are produced, which continue to degrade the polymer. A chain reaction of oxidative reactions may eventually result in stable oxidized products by recombination of free radicals or by the combination of free radicals and stabilizing agents contained in the material. And this is the termination stage. It is important to note that several factors determine how quickly and extensively pipeline liners undergo oxidation, including (1) elevated temperatures, which result in accelerated polymer degradation due to increased oxidation rates; (2) high levels of oxygen can promote oxidation since they provide more reactants for the process; (3) the presence of chemicals and additives such as hydrogen sulfide, carbon dioxide, and antioxidants; (4) the type and characteristics of polymer used as a liner.51,52,61,105–108 Figure 9 shows the oxidation cycle of polyethylene. The oxidation cycle of polyethylene.
105
In oil and gas pipelines, absorption refers to the process of liquids, gases, or chemicals permeating and diffusing into the liners. Additionally, it can greatly affect the longevity of a liner, in addition to its performance, chemical resistance, and mechanical properties. According to the type of polymer, absorption can take place either physically or chemically. A polymer matrix can be penetrated by small molecules, such as water and hydrocarbons, and diffuse into its bulk during physical absorption. Polymers absorb substances based on their porosity and their interaction with those substances. In contrast, aggressive environments can result in swelling, degradation, or plasticization (softening) of the liner when chemicals such as acids, bases, or solvents react chemically with it. In this case, the process is known as chemical absorption. As a result of this type of absorption, the chemical structure of the liner can be altered and its effectiveness reduced. Oil and gas pipeline liners can absorb substances depending on a variety of parameters, including polymer structure, crystallinity, additives, temperature, concentration of absorbates, pressure, and aggressive chemicals. Liner materials can absorb a wide variety of substances, such as water, moisture, hydrocarbons and gases (such as CO2, H2S).69,91,109–111
Polymeric liners in flexible pipelines vs. rigid steel pipelines
Generally, polymer liners are used on flexible pipelines in the same way that rigid pipelines are, providing a protective inner layer against internal corrosion caused by the fluids transported (such as crude oil, gas, hydrocarbons, acids, etc.). As a result of the polymer lining, the transport pipe is protected from corrosion, which extends its usefulness. Despite this, flexible pipelines differ in terms of their design and application, as polymer linings enable the pipeline to maintain its flexibility under dynamic conditions such as seabed movement, thermal expansion, and external pressure. In rigid pipelines, the polymer lining does not impact the structural integrity of the pipe as it bears the full mechanical load. Flexible pipeline liners are generally made from polyethylene (PE), polyamide (PA), or polyvinylidene fluoride (PVDF). Materials selected for this application are flexible, chemically resistant, and capable of withstanding a range of temperatures and pressures. A polymer liner is placed in contact with the fluid at the innermost layer, which consists of multiple layers. Additional layers, such as tensile armor, pressure armor, and an outer sheath, can provide mechanical protection for dynamic environments. A rigid pipeline’s lining is typically made of HDPE (high-density polyethylene) or PTFE (polytetrafluoroethylene). Liner materials selected for their chemical resistance, low permeability, durability and effectiveness in harsh environments are usually bonded or glued together as a single layer on the inside of conveyor pipes to prevent corrosion.66,112–116 A typical structure of a flexible oil pipe is shown in Figure 10. Table 2 represents a summary of the types of polymers used in flexible and rigid pipelines. Typical structure of flexible pipeline.
66
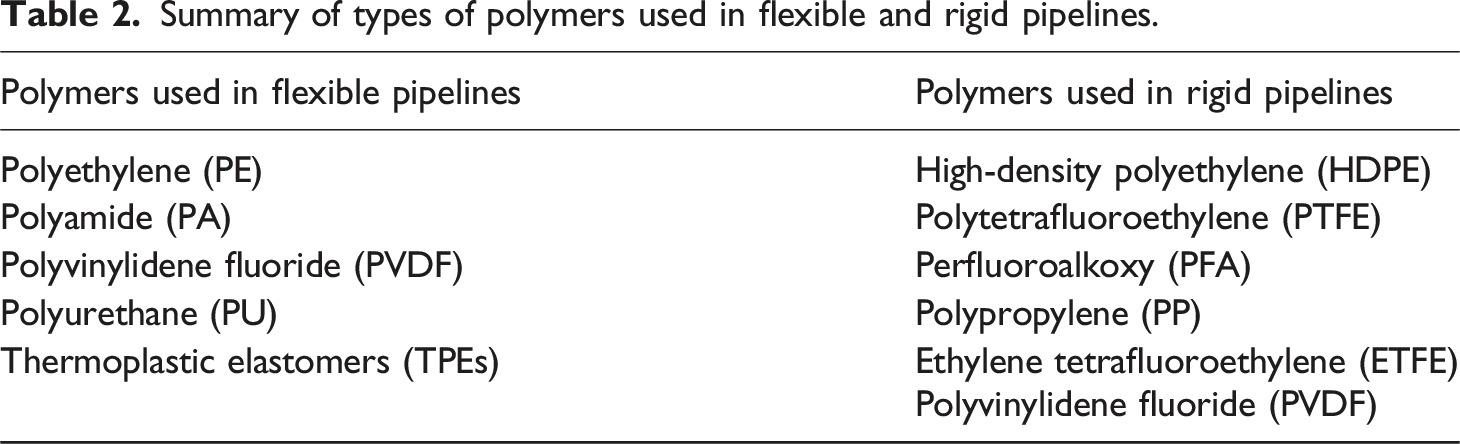
Summary of types of polymers used in flexible and rigid pipelines.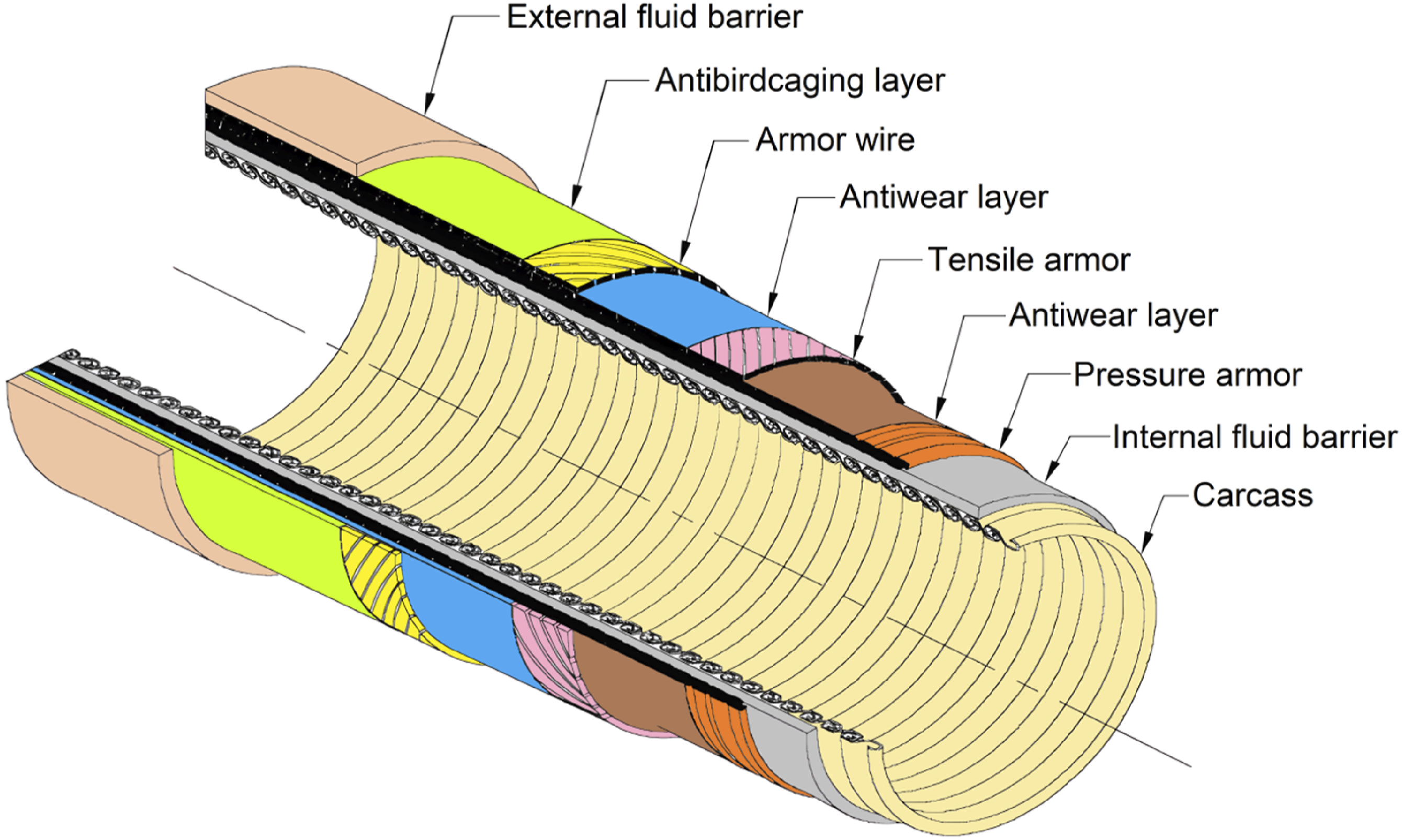
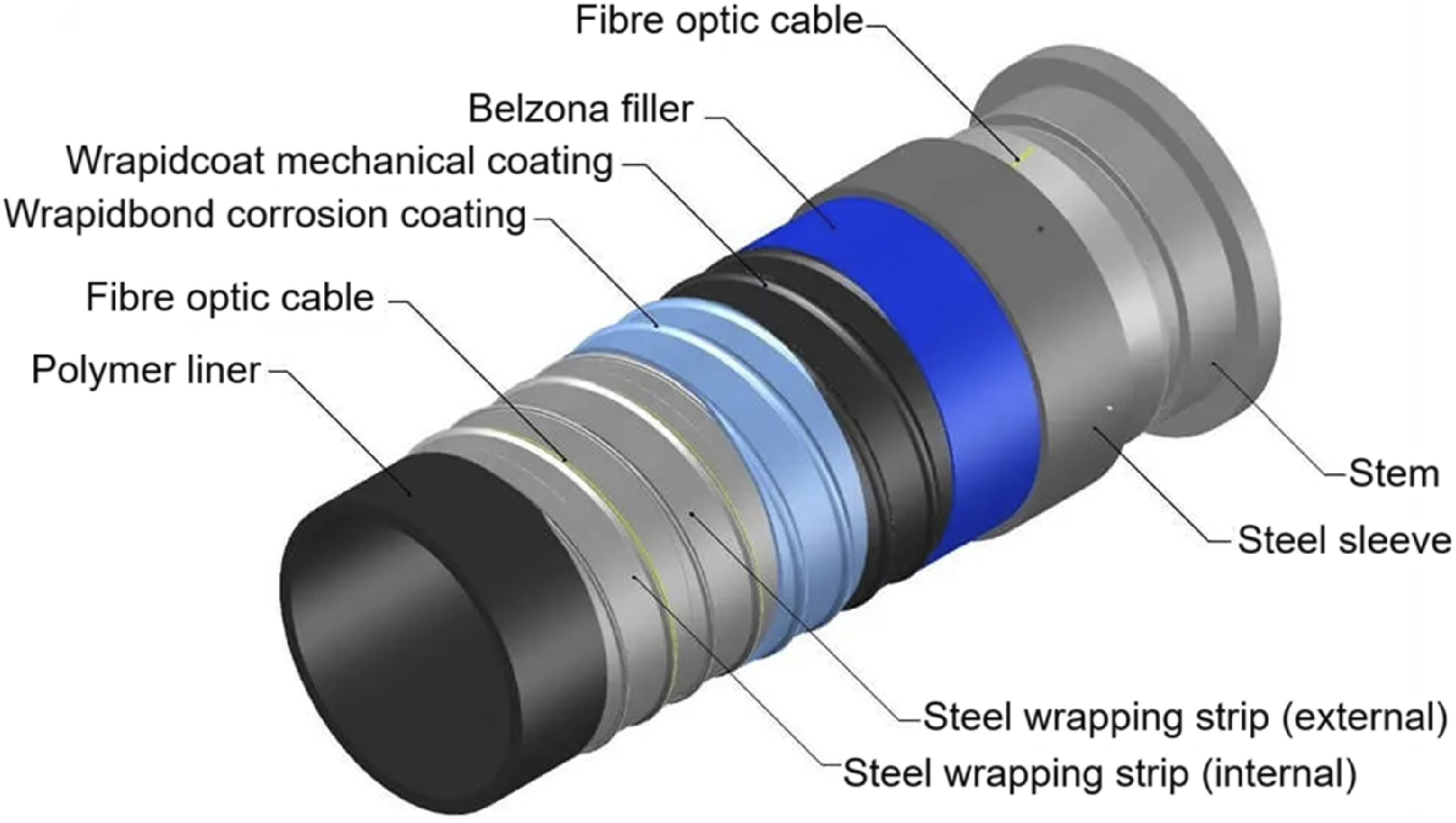
Liners of mobile automated spiral intelligent pipe (MASiP)
The Mobile Automated Spiral Smart Pipe (MASiP) is an advanced method of repairing damaged or old pipelines without the need for costly excavations. This technique involves creating a new pipe (see Figure 11) within an existing and damaged pipe using an automated spiral lining system. For protection, this type of pipe is also lined with polymeric material. A number of factors influence the choice of polymer lining for MASiP pipeline systems, including the type of fluid being transported, operating temperature, pressure inside the pipe, chemical compatibility, and cost. Polymers are currently used as linings for these pipes, including: high density polyethylene, polyvinylidene fluoride, polytetrafluoroethylene, polypropylene, ethylene tetrafluoroethylene, perfluoroalkoxy, and polyvinylidene dichloride.60,117–119 Typical structure of MASiP.
117
Design considerations for liners using with steel pipelines
There are several important design considerations that need to be taken into account when using polymer liners in steel pipelines to address issues related to the coefficient of thermal expansion (CTE). Due to their higher CTE than steel, polymers tend to stretch and contract more with temperature variations. Polymer liners are most likely to have CTEs that are comparable to or manageable within steel pipeline environments. It is also important to consider the installation method when talking about the expansion/contraction of the polymer liners, where the most effective method is to use a tight fitting or slip liner installation technique, in which the polymer liners are pulled or pushed into the steel pipe and then expanded to fit snugly. Moreover, because polymer liners expand more than steel, if not managed appropriately, they may buckle or wrinkle. Designing expansion gaps or sliding so that the liner is allowed to expand without excessive stress is an essential part of allowing for differential expansion. In addition, axial movement may be prevented by using liner end restraints or anchors. Keeping the operating temperature below the polymer softening point while considering thermal cycling is the most critical factor in minimizing the risk of liner failure. The internal pressure of the liner must also be controlled, as the liner expands when the temperature rises, which may cause a debonding or collapse of the liner. For this reason, the steel pipe and liner should be connected by a venting system or a pressure relief mechanism. Finally, regular pigging and inspection can detect degradation or displacement of the liner caused by time.120–125
Conclusions
The properties of polymers make them ideal materials for lining internal pipelines in the oil and gas industry, where they can reduce or prevent corrosion. A key problem that needs to be investigated to enhance mechanical and thermo-chemical properties is limitations. The development of multifunctional and effective polymeric coatings for oil pipelines is important scientific and practical work. Solving individual problems can save oil and gas companies money on repairing and replacing pipes, fittings, equipment, and structures. Although polymer linings have many benefits, their applications are limited by factors like temperature sensitivity, chemical compatibility, mechanical damage susceptibility, and pressure restraints. Moreover, they are limited in their application, especially in harsh or extreme environments. This is due to difficulties associated with installation, bonding, cost, and inspection. These limitations make it important to weigh the benefits and limitations of polymer liners. This must be done by considering the pipeline’s specific operating conditions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

