Abstract
Water (W) and oil (O) normally mix during production and while passing through valves and pumps to form a persistent water-in-oil (W/O) emulsion, which is a serious restriction in oil production and transporting and refining processes. The objective of this work is to treat emulsions of two crude oil samples labeled KD1 and DGH2 using commercial polymers and surfactants which are also known as demulsifiers. Hydrophile–lipophile balance (HLB) in the demulsifier structure has demonstrated a great effect on breaking W/O emulsion. Emulsion breakers with low HLB value showed better reduction in the dynamic IFT, high diffusivity at the W/O interface, and accelerated coalescence of water droplets. Concomitantly, high emulsion temperatures were found to reduce the interfacial film viscosity and accelerate water droplets coalescence. A maximum water separation efficiency (WSE) of 97% was achieved in the case of KD1 and 88% for DGH2, and using a (1:1) polymer blend demulsifier further increased WSE to 99% after 100 min.
Introduction
During crude oil production and processing, unfavorable formation called emulsion is formed due to dispersion of a liquid (dispersed phase) in the form of droplets, entirely in another liquid (continuous phase). Emulsion is an unwanted formation that causes serious corrosion in pipelines, processing equipment, and increase in operational and capital cost. Emulsion is composed of three phases: thoroughly distributed droplets form the internal or discontinuous phase; the matrix that sustains the droplets in suspension forms the external or continuous phase; and the stabilizer (interphase) that keeps the emulsion stable connects the internal and external phase together, preventing droplets from aggregating and coalescing. 1 –3 Moreover, there are plenty of factors that boost emulsion formation firmness during crude oil production. Among which is the continuous turbulence and agitation of the produced crude oil at subsurface along the flow stream. This turbulence may also happen at surface components such as transporting and refining facilities. Furthermore, existence of natural emulsifiers such as asphaltenes and resins in the crude oil also promotes stability of the emulsion formation. Asphaltenes, however, are dark brown to black macromolecules consisting of polycyclic, heterocyclic, and aromatic condensed sheets carrying side aliphatic chains as well as traces of metals like vanadium and nickel. These constituents confer polarity and amphiphilic characteristics to the asphaltene macromolecule. 4 –6
Asphaltenes form a rigid active interfacial film-like aggregations due to overlapping of their fused aromatic rings, hydrogen bonding, and Lewis acid–base interactions. These aggregations in turn adsorb at the surface of water droplets, which prevents the water droplets from coaslescing, and hence, stabilizes the emulsion. 6,7 Resins, on the other hand, have low-molecular-weight structures similar to that of asphaltenes, a lower content of aromatics, and rich in heteroatoms, mainly oxygenates. In addition, it is believed that these low-molecular-weight structures are able to solvate the asphaltene macromolecules into smaller aggregates, and hence, stabilize the colloidal particles of asphaltenes in oil. 8
In general, emulsions can be categorized depending on which phase is the dispersed (discontinuous) and which one is the dispersion (continuous). These categories are water-in-oil (W/O; water is the dispersed phase), oil-in-water (oil is the dispersed phase), and water-in-oil-in-water multiple emulsion. W/O crude oil emulsion is essentially the major type of emulsion that is formed during crude oil production and processing. However, shear mixing imposed on the crude oil during production and the presence of natural surfactants in the petroleum’s composition induces the formation of W/O emulsion. 9 –11 The presence of such emulsions can substantially increase pumping and transportation costs and pipeline corrosion. 12
Given these emulsion disadvantages, demulsifiers are required to break up crude oil emulsion into water and oil phases. Indeed, there are numerous demulsification methods, such as mechanical, electrical, and chemical, which are used in the oil and gas industry. Nowadays, polymers are found to demonstrate effective demulsification efficiency. Polymer-based demulsifiers are active substances that consist of both hydrophilic and hydrophobic portions. These amphiphilic materials can reside at the water/oil interface whereby the hydrophilic part adsorbs onto water drops, while the hydrophobic part attaches to the oil component. This dual action results in resolving the interfacial barrier which is formed due to water–oil emulsion; it destabilizes and breaks the emulsion to yield a drier oil, cleaner water, and sharp interface. 13,14
Demulsifiers, generally, can be classified into three main types: nonionic, cationic, and anionic. Chandran stated that the demulsifiers that have positive charge on the active portion of the molecule are basically cationic and can be classified as follows: quaternary ammonium compounds and amine salts (primary, secondary, and tertiary). 13 Anionic demulsifiers such as sodium dodecyl sulfate are surfactants that dissociate in water into amphiphilic anions and cations. 15 Nonionic demulsifiers have no positive or negative charges because of the balance between the hydrophobic and the hydrophilic part. The advantages of this type are the very low foaming and stability against pH changes or water hardness. The demulsification mechanism is a two-step process: flocculation and coalescence (see Figure 1). Flocculation is the process by which water droplets clump together to form flocs. The rate of flocculation depends on water content, temperature and viscosity of oil, density difference, and electrostatic field. Coalescence is the process by which water aggregations break up or fuse to form a larger drop. Coalescence may be enhanced by a number of factors such as high rate of flocculation and high water content.
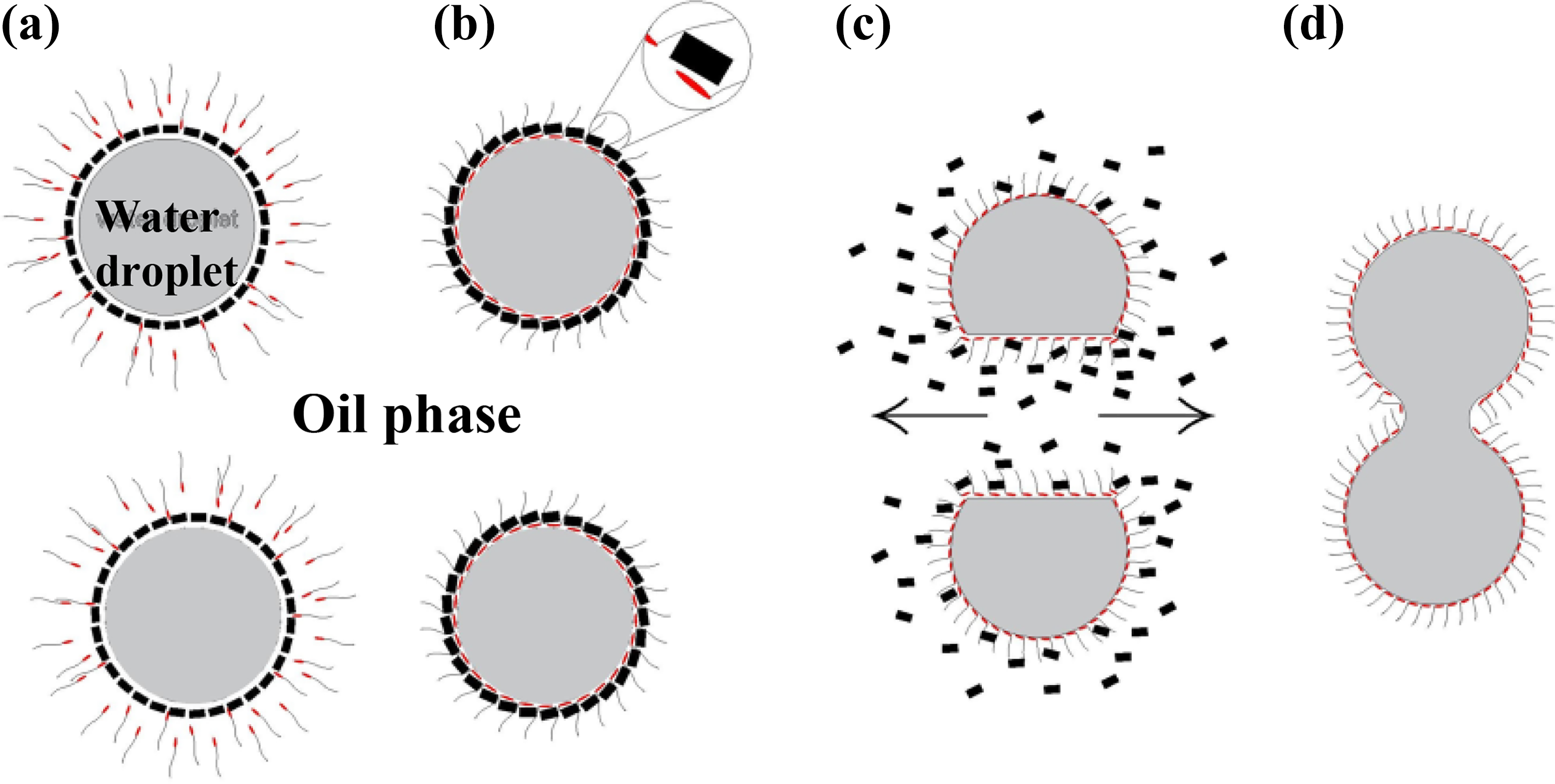
The demulsification mechanism of water-in-oil emulsions: (a) the demulsifier approaching the water drops through the voids between asphaltene aggregates, (b) the demulsifier adsorbs at the interface, (c) the asphaltene repelled from the water drop surface, and (d) the water drops coalesce. 10
Owing to the emulsion problems encountered in the oil fields located in Northern Iraq, this work focuses on a series of polymer and surfactant demulsifiers in order to chemically treat these oil emulsions. Crude oil samples labeled KD1and DGH2 were obtained from two oil fields located in Erbil city of Northern Iraq. The demulsification method performed in this study is a bottle test method. The emulsion model was synthesized by thoroughly mixing of 70% crude oil samples with 30% brine solution with a concentration of 0.5, 2, and 3.5 g/L. The effect of salinity, temperature, and demulsifier dose were all examined. Blends of polymer demulsifiers were also conducted to monitor their impact on the phase separation efficiency.
Materials and methods
Five demulsifiers labeled PG35, PG62, PP32, HA61, and PA31 were kindly obtained from Petro Fann (Houston, Texas) and Baker Hughes (London, United Kingdom) companies (see Table 2). Sodium chloride (Sigma-Aldrich, Taufkirchen, Germany) and a Milli-Q water (resistance less than 18.2 MΩ/cm) were used for the preparation of brine solutions of desired concentration. Toluene, ethanol, and benzene obtained from Merck (KGaA, Darmstadt, Germany) were used to dissolve crude oil samples. The crude oil samples used in this study were collected from two oil fields located in Erbil city of Northern Iraq. Density and viscosity of the studied samples were measured using density meter (hydrometer Koehler) (Koehler, 85 Corporate Drive Holtsville, New York 11742, USA) and Auto Kinematic Viscosity (AVK-202, TANAKA) (TANAKA, Fukushima, Tokyo, Japan), respectively. Energy-dispersive X-ray fluorescence spectroscopy was used to analyze the total ppm level of sulfur in the studied crude samples. The samples were classified as moderate crude oil KD1 and heavy crude oil DGH2 (Table 1).
Physical properties of the studied crude oil samples (KD1 and DGH2).

SP.G: Specific Gravity; API: American Petroleum Institute; BS&W: bottom sediment and water.
The physical properties of the crude samples as well as demulsification procedures were measured at the laboratories of KAR Group (Oil Refining Companies) in Kurdistan Iraq. Asphaltene content in the studied crude oil was determined by agitating 1 g of the studied crude oil with excess amounts of n-pentane at room temperature. Subsequently centrifuged at (2000 r/min) for 10 min and then allowed to settle overnight; the solid was filtered and dried at 38°C under vacuum until no change in mass was observed. The obtained asphaltenes were weighed to calculate the mass percentage. W/O emulsion models were prepared by mixing 70 v/v% crude oil and 30 v/v% brine solution of different w/v concentrations. The agitation was achieved by FM-200 (Fluko Corp., Shanghai Co., Ltd. China) mixer homogenizer. For further elucidation, the stability of W/O emulsion was tested at different mixing speeds (500–6500 r/min) for a period of 60 min in the case of KD1 and 40 min in the case of DGH2 at ambient temperature. The emulsion formulation was subsequently left for 3 h to monitor whether a stable emulsion is formed (Figure 2).

Stable water-in-oil emulsions (70:30 v/v% crude to brine water) for KD1 (right) and DGH2 (left), after 3-h settling time.
Demulsification using bottle test method was implemented at constant pH to evaluate the demulsifier effectiveness. The dehydration rate or water separation efficiency (WSE) is given as the volumetric ratio of water separated to the original water content of the crude oil sample (see equation (1))

where W s is the fraction of water separated, V ws is the volume of water separated, and V o is the original water content in the crude oil. The demulsifiers were prepared with the desired concentration of 10, 40, and 100 ppm. It follows that the emulsion stability is given by 1 − W s, that is

For performing the bottle test method, the emulsion formulation was heated at 55°C using a water bath (WN-745 memmert) (Memmert GmbH , D-91126 Schwabach, Germany) prior to the demulsification process. Subsequently, the potential demulsifiers were diluted with ethanol and then added to a graduated cylinder containing 100 ml of the thermostated emulsion system. The system was then shaken thoroughly for 1 min to homogenize the demulsifier and the emulsion system. Water separation (in milliliters) was observed and recorded at (0–100 min) time intervals.
Results and discussion
Emulsion stability test
Figure 3 shows the influence of mixing rate on the stability of pre-synthesized W/O emulsion of KD1 and DGH2 crude oils. The synthetic W/O emulsions were prepared by mixing (70:30 v/v%) crude oil to brine solution (3.5 w/v%) according to the procedure outlined in the “Materials and methods” section. The Marangoni–Gibbs effect 10 is the most convenient concept to explain the stabilization of W/O emulsion. This phenomenon defines that if two water droplets approach each other, a surface deformation between the droplets allows a thin liquid intermediary film (continuous phase) to remain between them. The formation of such a configuration is due to various competing attractive and repulsive forces which have a different dependence on the separation of the droplets. By capillarity, the film tends to leak out and some emulsifiers depart away from the interface. At this stage, an interfacial tension (IFT) gradient is generated, which is related to the distributed surface energy. In addition, a reverse flux occurs in order to compensate for the lost emulsifier concentration at the interface. Thus, the intermediary film thinning stops and coalescence between the droplets is prevented.
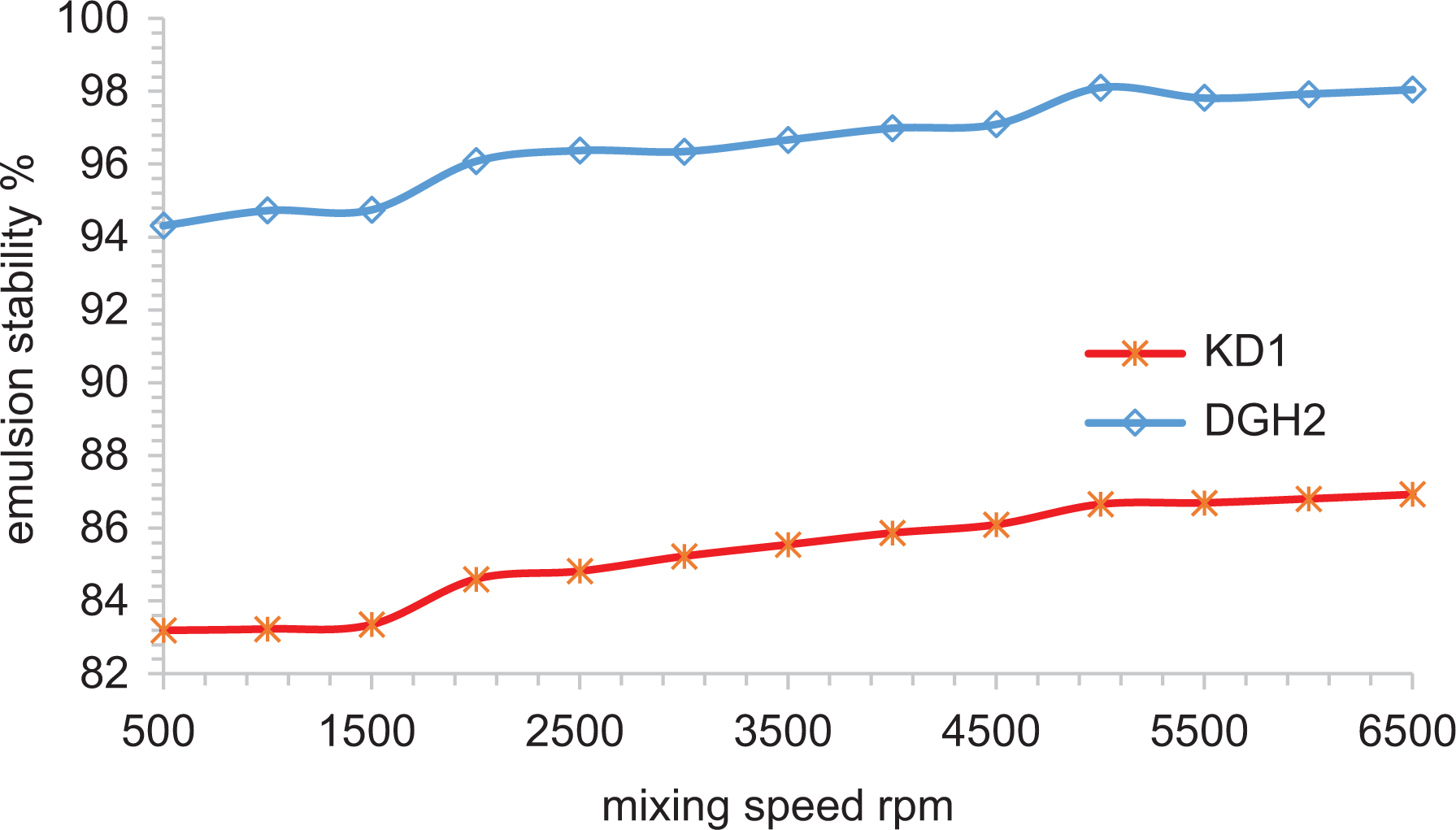
Testing the stability of KD1 and DGH2 water-in-oil emulsions.
The graphical results in Figure 3 demonstrated clear correlation between mixing shear rate and stability of W/O emulsion. As mixing shear rates increase, the stability of the studied W/O emulsions increases. These results are in good agreement with previously published work. 16 Nonetheless, a quantitative variation in the stability of the KD1 and DGH2 crude emulsions was observed. This trend variation, however, may be interpreted on the basis of the Marangoni–Gibbs effect described above. From the data in Table 1, one can notice that crude sample DGH2 is more viscous and has more emulsifying agents such as asphaltene than crude sample KD1. The high viscosity of DGH2 allows the homogenizer blade to shear off water droplets into further smaller drops. Consequently, a larger surface is created, which effectively increases the interfacial free energy due to the larger surface area contact between the two phases, and droplets approach each other generating a tension gradient at the interface. In conjunction, the intermediary film thinning and film drainage stop due to the reverse flux that occurs to replace emulsifier loss at the interface. Thus, water coalescence is retarded and emulsion stability increases. By contrast, the poor emulsion stability of KD1 oil despite high mixing speed may be attributed to inefficient distribution of water droplets due to low viscosity. This allows for larger water droplets and consequently, the rate of intermediary film thinning and film drainage by capillarity increases, that is, water coalescence is activated and emulsion stability decreases.
Optimizing demulsifier dose
W/O emulsions are produced during the production of crude oil, and these emulsions are stabilized by the interfacial film that is formed. The stability of this film is strongly dependent upon the emulsifier’s adsorption–desorption kinetics, solubility, and interfacial rheological properties such as elasticity, IFT gradient, and interfacial viscosity. 17 Breaking oil emulsion, however, is essential before transportation through pipelines and prior to refining. Chemical demulsifiers have demonstrated capability to change the rheological properties of the interfacial film and to destabilize the surfactant-stabilized emulsion films. 18 Polymer surfactants are promising demulsifiers owing to their capability to adsorb at the water–oil interface and increase film thinning by suppressing the tension gradient at the interface. 10
In this context, five demulsifiers having concentrations of 10, 40, and 100 ppm were added to break W/O emulsion of two crude oil samples labeled KD1 and DGH2. Table 2 transcribes some characteristics and supplier name of the demulsifiers used in this work. The bottle test method was implemented to optimize the demulsifier dose and to calculate the WSE. The crude emulsion system was prepared by mixing 70 v/v% of target oil with 30 v/v% of brine water (3.5 g/l) in a 100-ml graduated cylinder. The demulsification processes were performed at an optimum temperature of 55°C, 100-min settling time, and constant pH. The maximum WSEs of potential demulsifiers are shown in Figures 4 and 5.
List of demulsifiers used in this work.

HLB: hydrophile-lipophile balance; HL: Hydrophilic-Lipophilic.

Optimizing demulsifiers dose for breaking KD1 crude emulsion at 55°C.
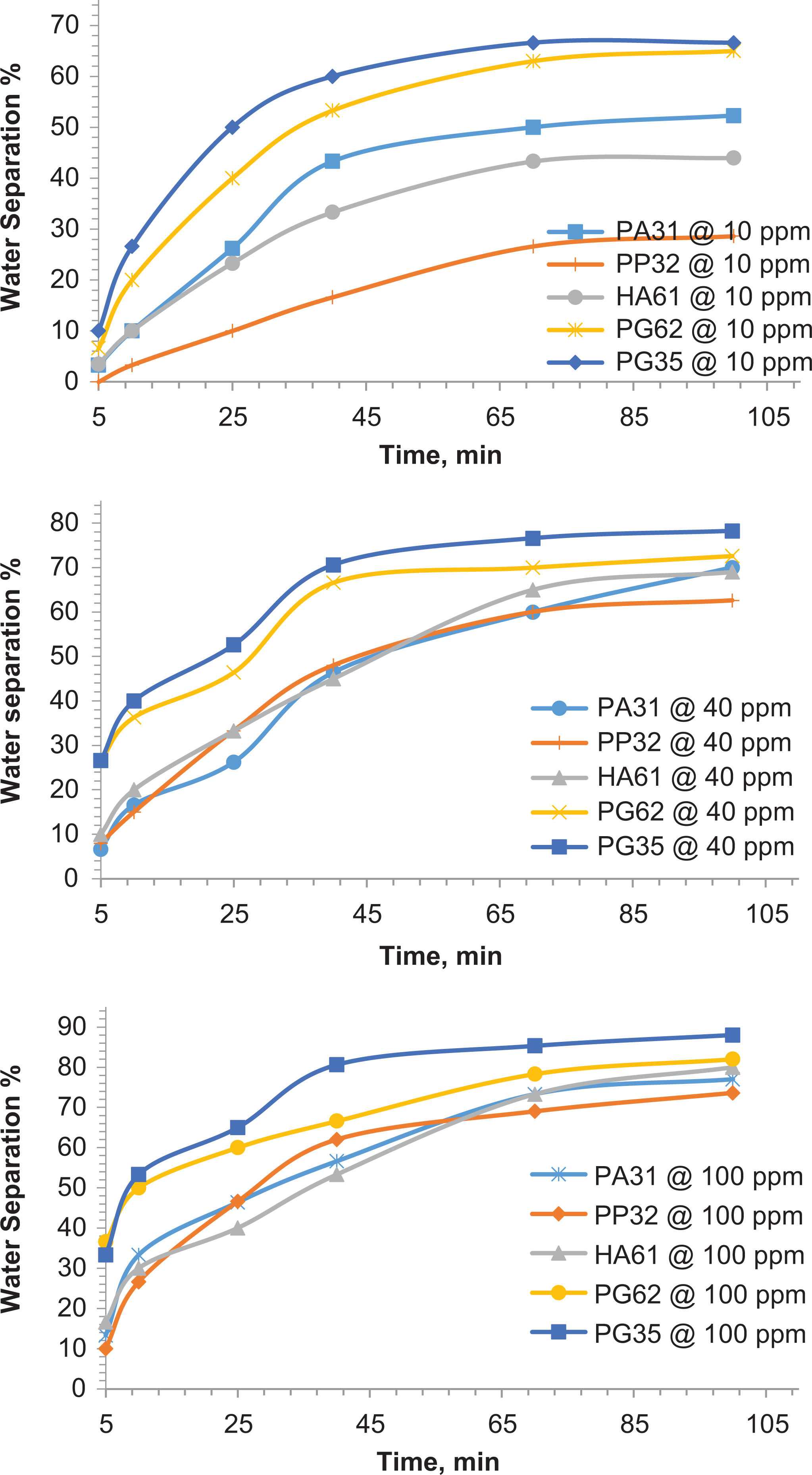
Optimizing demulsifiers dose for breaking DGH2 crude oil emulsion at 55°C.
The demulsification process initiated after 10 min from adding the potential demulsifier and the WSE was 10–45%. The total water content was obtained after about 40–60 min in both KD1 and DGH2 crude emulsions. The WSE increased after 40 min and, finally, appeared to stabilize after 95–100 min. The graphical results illustrated that when adding 100-ppm dose of polymer demulsifier PG35, a maximum WSE of 97% for KD1 and 88% for DGH2 is obtained.
All of the added demulsifiers exhibited minimal WSE at 10-ppm dose at 55°C. However, KD1 and DGH2 crude emulsion conferred a quantitative variation in the demulsification performance. Demulsification is, however, affected by numerous parameters, some of them related to the characteristics of the continuous phase, such as viscosity, and asphaltenes content, and others depend on the hydrophile–lipophile balance (HLB) in the chemical structure of the demulsifier molecules. In this regard, these variations may be explained on the basis of the asphaltene content in the studied samples and the chemical structure of the demulsifier molecules, as we discuss in the following.
DGH2 crude emulsion (asphaltene content 6.0 w%) exhibited low demulsification efficiency upon adding potential demulsifiers at different dosages. High content of asphaltene aggregates as in the case of DGH2 crude emulsion is expected to have only few voids at the water–oil interface. Therefore, only few demulsifier molecules can approach the interface and replace asphaltene molecules. More voids, on the other hand, are expected for KD1 crude emulsion which have lower content of asphaltene aggregates (3.1 w%). This effectively increases the amount of demulsifier molecules at the interface. Thus, the rate of film thinning and the rate of film drainage are accelerated and demulsification efficiency improved.
Alternatively, the varied performance of used W/O emulsion breakers can be interpreted regarding the HLB in their chemical structures. The HLB values for the used polymers and surfactant molecules were calculated using the following equation 18

where M H and M L are assigned as formula weights of the hydrophilic and lipophilic portions in the polymer molecule, respectively; HLB scale 0–20. By inspection of the graphical results in Figures 4 and 5, the demulsification performance of our W/O emulsion breakers can be regarded as follows: PG35 > PG62 > HA61 > PA31 > PP32. Returning to Table 2, the polymer PG35 has the lowest HLB, that is, more lipophilic rather than hydrophilic, while polymer PG62 (0.443 HLB value) showed less affinity toward the oil fraction (i.e. less lipophilic). These dual features (HLB) encouraged well disperses of the asphaltene molecules at the oil–water interface from one side and hydrophilic bridges between water droplets from the other. Consequently, the rate of IFT gradient due to emulsifier loss at the interface is reduced, film thinning and film drainage accelerated, and water coalescence is triggered after about 10 min from adding the polymer.
Meanwhile, the lipophilic skeleton of polymer PP32 places this demulsifier in the latter case based on the WSE compared with the other used polymer demulsifiers (Figures 4 and 5). The lower demulsification performance of PP32 is a consequence of its lipophilic structure, which decreases the solubility of this polymer in aqueous phase (dispersed phase), reduces the hydrophilic bridges between water droplets, and stops interfacial film rupturing.
The calculated HLB values of demulsifer molecules PA31 and HA61 were 0.874 and 0.591, respectively. Regarding the HLB values, these molecules can be classified as hydrophilic or water soluble emulsion breakers. These demulsifiers, however, exhibited less WSE compared to PG35 and PG62 at the same demulsification conditions. This behavior is mainly due to low solubility in oil, that is, solvation of asphaltene aggregates at the interface is inefficient, leading to less water coalescence.
Effect of salinity
Figure 6(a) and (b) shows the effect of changing brine concentration on demulsification efficiency of KD1 and DGH2 crude emulsions. The bottle test method was employed using synthetic emulsion composed of 70% crude and 30% brine solution of different concentrations (0.5, 2, and 3.5 w/v%). The screening process was conducted at 55°C after injecting a potential demulsifier at optimal dose. Polymer demulsifiers PG62 and PG35 were chosen due to their high efficiency in water separation. The results revealed better water separation with increasing salinity for both of the studied crude emulsions. KD1 crude emulsion reported a WSE of 97% at 3.5-w/v% brine concentration, whereas DGH2 crude emulsion showed 85% WSE after 45-min settling time and 100-ppm demulsifier dose.
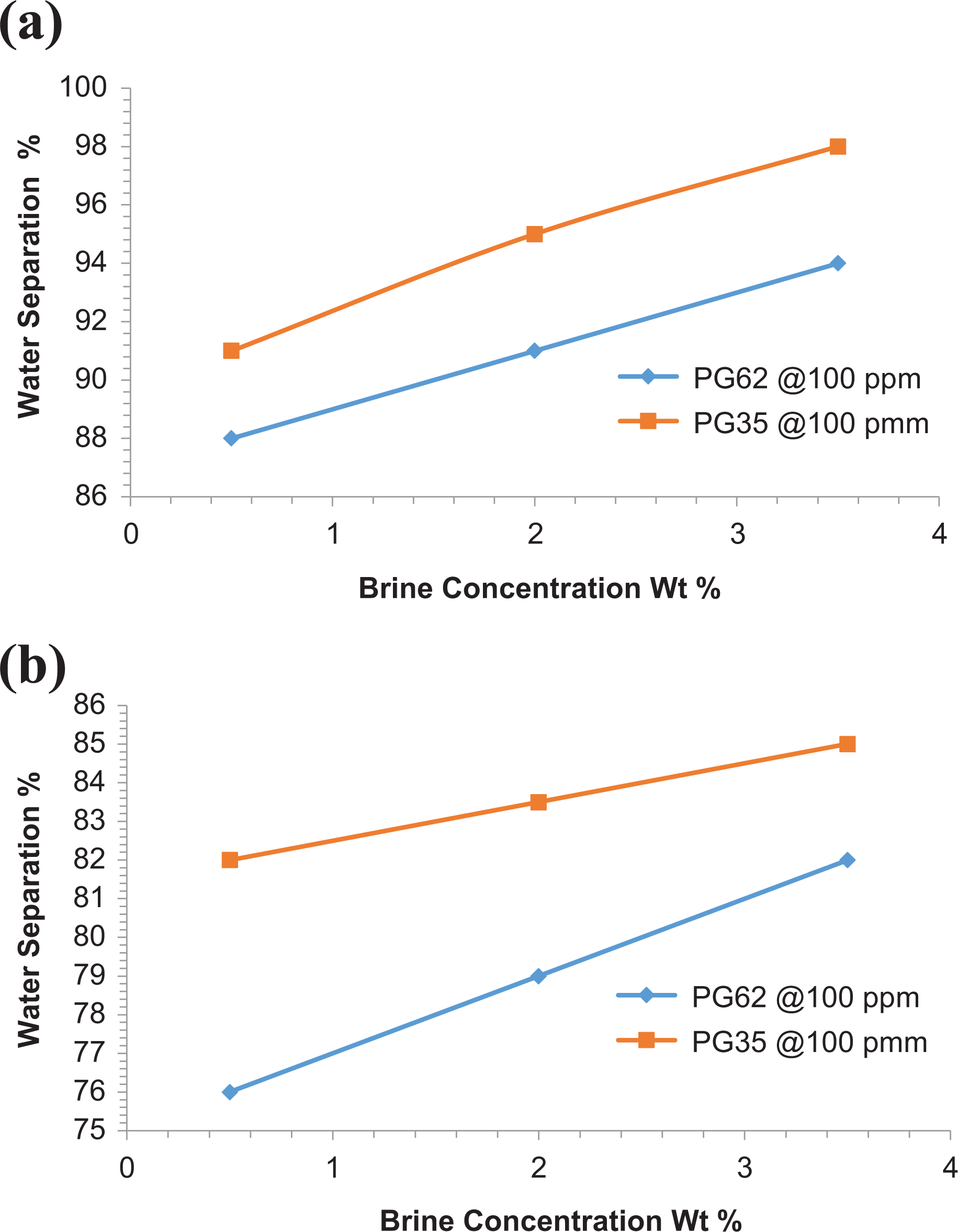
Effect of salinity on the W/O emulsion stability of (a) KD1 and (b) DGH2 at 100 min settling time and 100-ppm demulsifier dose.
Increasing brine salinity, however, can result in increasing water density and subsequently increases the differential density between water and oil. Thus, the entire structure of the W/O interfacial film is altered, making water coalescence easier and increases emulsion breakdown. These results are in very good agreement with that obtained by Fortuny et al. 19 who studied the effect of salinity on the stability of crude emulsion and Moradi et al. 20 who indicated that emulsion stability enhances at lower ionic strength of the aqueous phase.
Effect of temperature
In this study, we examined the effect of increasing temperature on the characteristics of W/O interfacial film and emulsion breaking of the studied samples. The bottle test method was carried out at different temperature intervals (30, 40, 55, 65, and 75°C). The demulsifiers were injected at 100-ppm dose and homogenized with synthetic emulsion for 5 min, and then lifted for settling for a period of 100 min. The results demonstrated a significant increase in the demulsification efficiency when the temperature increased from 30°C to 55°C (Figure 7(a) and (b)).

Effect of rising demulsification temperature on (a) KD1 and (b) DGH2 crude emulsions at 100-min settling time and 100-ppm demulsifier dose.
This increment trend was found to decline above 55°C for both samples. However, increasing the temperature also affects the viscosity and density of oil, water, W/O interfacial film, and emulsifying and demulsifying solubility in the oil and water phases. In this view, heating crude oil emulsion is expected to decrease the interfacial film viscosity; the increased thermal energy of the water droplets enhances dispersion of asphaltene aggregates, and consequently, more voids allow the demulsifiers to penetrate the internal phase and adsorb onto water droplets. This results in a rate of faster film thinning and film drainage, thus increasing water coalescence.
The decline in demulsification efficiency above 55°C can be interpreted by considering the increase in vapor pressure of low molecular weight hydrocarbons in the studied samples. This results in decreasing the diffusivity of the polymer demulsifiers at the W/O interfacial film and as a result decreases water separation rates.
Polymer blends as demulsifiers
The effect of using blends of demulsifiers on the demulsification efficiency of the studied samples has been investigated. The polymer blend demulsifiers were obtained by mixing two demulsifiers in two different volumetric ratios: (1:1) and (1:3); PG62: PG35, which both presented the highest WSE as individual demulsifiers. A dose of 100 ppm of the new demulsifier formulation was injected into the synthetic crude emulsion, homogenized, and lifted for settling for a period of 100 min. The obtained results are shown in Figure 8.

The effect of the blends of demulsifiers on the water separation efficiency of KD1 and DGH2 crude emulsion at 100-min settling time and 100-ppm dose.
The monitoring process using (1:1) PG62: PG35 blend demulsifiers exhibited pronounced increase in the trend of WSE, reaching 95% and 86.6% after 25 min; 99% and 94% after 100 min, added to KD1 and DGH2 crude emulsions, respectively. Nevertheless, (1:3) PG62: PG35 blend gives less demulsification performance in both samples. The increase in WSE as a result of blending may be attributed to the HLB of the new blend demulsifiers. However, the new blends have a synergetic action toward W/O emulsion. The lipophilic property of PG35 enhanced asphaltenes dispersion at the W/O interface, while the hydrophilic property of PG62 facilitates hydrophilic bridges between the droplets. This results in accelerating film thinning and film drainage and faster water coalescence.
IFT studies
For further analyzing the demulsification performance of the used demulsifiers, the dynamic IFT studies for our emulsion system with and without added demulsifiers was conducted by the Drop Volume Tensiometer method using stalagmometric instrument (Fischer Scientific, Hampton, New Hampshire, USA). For this purpose, the several drops of the liquid leaked out of the glass capillary of the stalagmometer are weighed. This method was first time described by Tate in 1864 who formed an equation, which is now called the Tate’s law

where W is the drop weight, r is the capillary radius, γ is the surface tension of the liquid, and f is the correction factor which is a function of the stalagmometer tip radius, volume of the drop, and a constant.
Dynamic IFT data for samples under study were collected and analyzed following a procedure published elsewhere. 21 Figure 9 shows the dynamic IFT for the blank emulsion system KD1 (70 v/v% of crude oil with 30 v/v% of brine water, 3.5 g/L) and after added demulsifiers.

Dynamic IFT profile versus time for KD1 crude emulsion without demulsifiers and after added 100 ppm of different demulsifiers at 55°C.
As illustrated in Figure 9, the blank emulsion system presented a slow decreases in the dynamic IFT within hundreds of seconds and then the reduction becomes less significant until the value of 45 mN/m. The dynamic IFT observations for the emulsion system with added demulsifiers at 100 ppm dose at 55°C elucidated considerable decreases in the IFT trend at early seconds. The dynamic IFT, however, exhibited varied reduction behavior upon added different demulsifiers. For instance, a much faster reduction in the IFT trend upon added demulsifiers with low HLB values was observed for polymers PG62 and PG35. It is also seen that the IFT cycles decrease very slowly even after 2 h, which implies that the system did not completely reach the equilibrium state. Previous studies suggested that such a reduction in the dynamic IFT is similar to diffusion-controlled process of proteins or polymers at liquid–liquid interface. 22 –24 The Gibbs–Duhem diffusion equation (equation (5)) has been established to describe diffusion-controlled process of our demulsifiers and to analyze our dynamic IFT data, as shown in the following

where γ 0 and γt are the IFT at t = 0 and an arbitrary time t, R is the gas constant with the value of 8.314 J/(mol·K), C 0 is the demulsifier bulk concentration, D is the diffusion coefficient of demulsifiers moving to the interface, and T is the absolute temperature in Kelvin. According to equation (5), for a diffusion process, a linearity between γt and t 0.5 should be achieved. Therefore, to better understand the diffusion process of our demulsifiers, we plotted IFT versus t 0.5 as shown in Figure 10.

Dynamic IFT profile versus t 0.5 for KD1 crude emulsion without demulsifiers and after added 100 ppm of different demulsifiers at 55°C.
The slope for the linear correlation between γt
and t
0.5 in equation (5) is defined as k and
For fixed demulsifier concentration, that is, 100 ppm, the interfacial diffusion coefficient D is related to k by equation (6). Its values under optimized demulsifier concentrations, that is, 100 ppm at 55°C are displayed in Table 3

Interfacial diffusion coefficient for used demulsifiers at optimized conditions, that is, 100 ppm at 55°C.

The obtained data in Table 3 show that the value of parameter (D) is higher for the polymers PG62 and PG35 which revealed a better reduction in the IFT trend, that is, these polymers diffuse more efficiently at W/O interface at optimized conditions. These results, however, agreed very well with the previous results (see Figures 4 and 5).
Conclusions
Demulsification of KD1 and DGH2 crude emulsions were successfully carried out. The sequence of used materials as W/O emulsion breakers was PG35 > PG62 > HA61 > PA31 > PP32. KD1and DGH2 crude oil samples elucidate a stable emulsion with increasing shearing speed and asphaltene content. High emulsion stability was ascribed to highly viscous DGH2 crude over KD1. High shear rate made water droplets smaller and provide larger surface for asphaltenes to bind with the droplets and prohibit coalescence. The demulsification of the studied W/O emulsions was found to be at optimum conditions of 55°C, 100-ppm demulsifier dose, and 3.5 mass% brine concentration. Polymers that have low HLB values presented better IFT reduction and higher diffusivity at the W/O interface. Increasing emulsion salinity improves the differential density between water and oil and encouraged phase separation. Rising emulsion temperature reduces interfacial film viscosity, provides more voids for demulsifiers to reach water drops, and accelerates water drop coalescence. In addition, vapor pressure of low-molecular-weight hydrocarbons increases above 55°C which may prevent demulsifiers from approaching the interfacial film and reduces demulsification efficiency. Blends of demulsifiers further increased the WSE to 99% in one case, attributed to a combination of lipophilic and hydrophilic activity of the selected polymers.
Footnotes
Acknowledgments
The authors highly appreciate the unlimited support introduced by KAR groups (Oil Refineries) quality control department for the help in laboratory measurements. Great thanks and gratitude also goes to the Baker Hughes and Petro Fann for their scientific support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
