Abstract
Conventional methods of eliminating water from crude oil such as the chemical injection have both economic and environmental impacts; thus, this study proposed an economic and environmentally friendly demulsifier. The bottle test method was used to study the performance of the natural extract and commercial demulsifier on a crude oil sample. The GC-MS profile of the extract was in agreement with previous reports on composition of oil extracted from rice bran using hexane, ultrasound assisted extraction and conventional solvent extraction with ethanol. Varying degrees of saturated and unsaturated fatty acids as well as retention times as observed, was a function of total time of scanning, according to NIST08 library of mass spectra. The performance of the demulsifier was expressed in terms of percentage of water separated from 100 ml samples of the oil samples. For both the demulsifiers, the performance increased with increase in volume of the demulsifier, separation time and operating temperature. The extracted demulsifier performed better than the chemical demulsifier under all the experimental conditions adopted in this study. Based on the parametric evaluation, it was observed the results from software corroborated the results obtained from experiments in terms of the observations of the combined effect of temperature and volume which showed the most significant influence on demulsification of the emulsified crude. The highest efficiency of the bio-demulsifier was obtained with a volume of 5 mL of the extract, at a temperature of 70°C and separation time of 60 min. A water separation efficiency of 85.6% was obtained as compared to the chemical demulsifier, which gave an efficiency of 80.2%.
Introduction
Flow assurance provides an economic approach to the production and transportation of fluids from a hydrocarbon well to a process plant. Knowledge of fluid characteristics, operating conditions and properties is important to avoid the formation and precipitation of unwanted solids (that is, scales, hydrates, waxes, asphaltenes) and emulsions during the extraction and transportation of crude oil (Saliu and Della Pergola, 2018). The transition from green to mature fields further aggravates the problem of flow assurance. As the oil field matures, the percentage of water produced increases. In some cases, operators pump water into mature oil field reservoirs to improve oil recovery. Water-in-crude oil emulsion results, which further complicates the flow situation (Ese and Kilpatrick, 2008). Most of the commercially available anti-flocculating hydrate inhibitors become less effective as the water-cut increases (Abullah et al., 2016). In the end, the emulsion produced must be fragmented to separate the crude oil from water. The breaking of these emulsions by physical or chemical methods can be very expensive, especially for crude oil emulsions that contain emulsion-stabilizers, such as naphthenic acids or asphaltenes (Nydén et al., 2003). Therefore, it is important to develop profitable/innovative flow management strategies for controlling emulsions in order to minimize economic risks during the productive life of the field, and to avoid an increase in overhead costs that may accrue from crude oil processing and refining (Grace, 1992; Okoro et al., 2019). Chemical demulsification approach is the most often applied technique in the petroleum industry among mechanical, thermal, electrical, filtration and chemical-flotation treatment methods (Atta et al., 2016; Efeovbokham et al., 2013).
Adewunmi and Kamal (2019) studied the effects of counter ion and water type of ionic liquids for demulsification of water-in-oil emulsions. They concluded that the salts present in the sea water had propensities to accelerate the destabilization of the emulsions; and that the ionic liquid used can adequately collapse the effect of the natural emulsifiers (resins and asphaltenes) (Igwilo et al., 2017). Some nanoparticles have been identified as good candidates for efficient and effective separation of crude oil emulsions but their suspension in the oil phase after separation has been a setback hence, the need to look into possible ways of addressing the problem (Huang et al., 2017; Liang et al., 2014; Liu et al., 2015; Lü et al., 2017; Ortiz et al., 2019; Zhang et al., 2017). Hazrati et al. (2018) also considered the application of ionic liquids in demulsification of water-in-oil emulsion while Roodbari et al. (2016) studied the role of a nonionic polymer as a demulsifying agent for heavy crude oil/water emulsion. They observed that high molecular weight alkenes’ chains, esters and groups of ketone present in the polymer enhanced the performance of the demulsifiers and at the end of the process, no residual counter ion was present in the crude oil. Hippmann et al. (2018) used natural Alginite as a demulsifying agent. The study showed that Alginite is an effective natural demulsifier in crude oil emulsions and can serve as a worthy substitute to conventional demulsifiers. But like nanoparticles, a separation process and thermal recycling treatments are needed to remove and re-use Alginite after demulsification.
Concern about the decreasing profitability in the petroleum sector has created renewed interest in cost reduction and profit enhancement. Demulsification of crude oil emulsions on-site can be an expensive operational issue. The removal of produced water from crude oil is the primary goal of demulsification (Romanova et al., 2019). There is, therefore, a need for a demulsifier which shows the same or superior properties and efficacy as other chemicals which are adopted as standards for operations of this sort, however, they must be environmentally friendly. There is no doubt about the need for a natural demulsifier that can replace the natural emulsifiers for adequate separation of water-in-oil emulsions in the petroleum industry. This will lead to development of thin interfacial films that will promote the aggregation of small water drops and amalgamate into bigger droplets. The objective of this study is to address at least one of the above or other disadvantages associated with the prior art, by using a bio-waste extract to formulate a demulsifier that effectively separates water from crude oil without altering the chemical and physical properties of the hydrocarbon. The efficiency of the proposed demulsifier was also investigated and compared with a field applicable chemical demulsifier.
Methodology
Materials
The reagents for the extraction process are HNO3, n-hexane, ethanol, n-butanol and xylene of 99% purity and manufactured by Sigma-Aldrich chemical company. All chemical reagents used in this study are of analytical grade. The 4 L of water-in-oil emulsion sample was obtained from oil production facilities in Bayelsa (“Field X”), Niger Delta region. The emulsion sample from the field was steady at room temperature for more than 12 weeks without any obvious phase separation. The physical characterization of the produced emulsion is shown in Table 1.
Properties of the crude oil emulsion sample used for this study.

This study is geared towards culturing a plant-based demulsifier from Waste brown oil extracts of rice husks (Okoro et al., 2019). This will provide the petroleum industry with a cheaper, more environmentally friendly, and less toxic alternative for the demulsification process. The isolated rice husk was collected from a mini rice milling machine in Nasarawa state, Nigeria. The collected sample (1000 g) was passed through an electric grinder machine which was adopted for the extraction process and harmonized by sieving.
Methods
Figure 1 demonstrates the experimental methods utilized in this study and a short depiction of every procedure used.

Experimental short depiction of steps adopted in this study.
Extraction of waste brown oil from rice husk
The reflux method was employed in this study (Figure 2); the samples were promptly exposed to microwave heating at 150°C for 3 min to prevent against microbial spoilage and hydrolytic rancidity. The dried samples were homogenized with n-hexane at a proportion of 1:2 (w/v) for 15 min at 9500 r/min. The mixture was filtered through and the residue was re-extracted twice following the same procedure to get the defatted rice husk (Sanbongi et al., 1998). The defatted rice husk was dried in an oven at 50°C for 3 h so as to remove any remaining solvent left. The defatted rice husk sample was sieved by a 0.6 mm sieve mesh and kept at room temperature prior to the extraction of the waste brown oil.

The reflux method set-up for the study.
The rice husk was homogenized and refluxed with 50% aqueous ethanol, to obtain the crude alcoholic extract (CAE) in a ratio of 1:15 (w:v). The resulting mixture was filtered by Whatman No. 2 filter paper to remove the solvent. The CAE (1 g) was expended in 25 mL of the distilled water to remove any residual lipids and divided in 125 mL n-butanol. The aqueous layer of the 125 mL n-butanol was homogenized at 9500 r/min for 15 min. The mixture was then kept at room temperature until both solvent layers were present. This partitioning procedure was repeated twice and the n-butanol Waste brown oil fraction (BSF) was pooled and concentrated to dryness under reduced pressure using a Rotavapor R210.
Bottle test method
This experiment was adopted to investigate the effect of the extracted demulsifier on the produced emulsion. Using a micropipette, 1–8 mL portions of the extracted demulsifier were separately injected into the bottle test samples containing the emulsions. Then, the bottle was shaken evenly until a homogeneous solution was obtained. The volume of water separation was measured in accordance with ASTM D4006-81 and ASTM D1401-98 (Roodbari et al., 2016). At the desalination stage, the bottles were immersed into a water bath at 70°C and the efficiency of the extracted demulsifier was calculated at 5, 10, 20, 30, 60 and 120 min. The volume of the separated water at the lower part of the bottle was measured.
Chemical analysis of the extracted demulsifier
Samples were analyzed using gas chromatography/mass (GC-MS) and the compound seen were detected/identified based on their respective retention times to that of n-hexane from related studies and spectra of certified compounds based on their retention time in n-hexane. Further, the compounds were identified and authenticated using mass spectra compared to similar data obtained from the Wiley library version 7.0 N. The standard library of EI mass spectra is that produced by the American National Institute of Standards and Technology (NIST), which currently contains mass spectra for over 120,000 compounds.
Results and discussion
Extracted demulsifier constituents
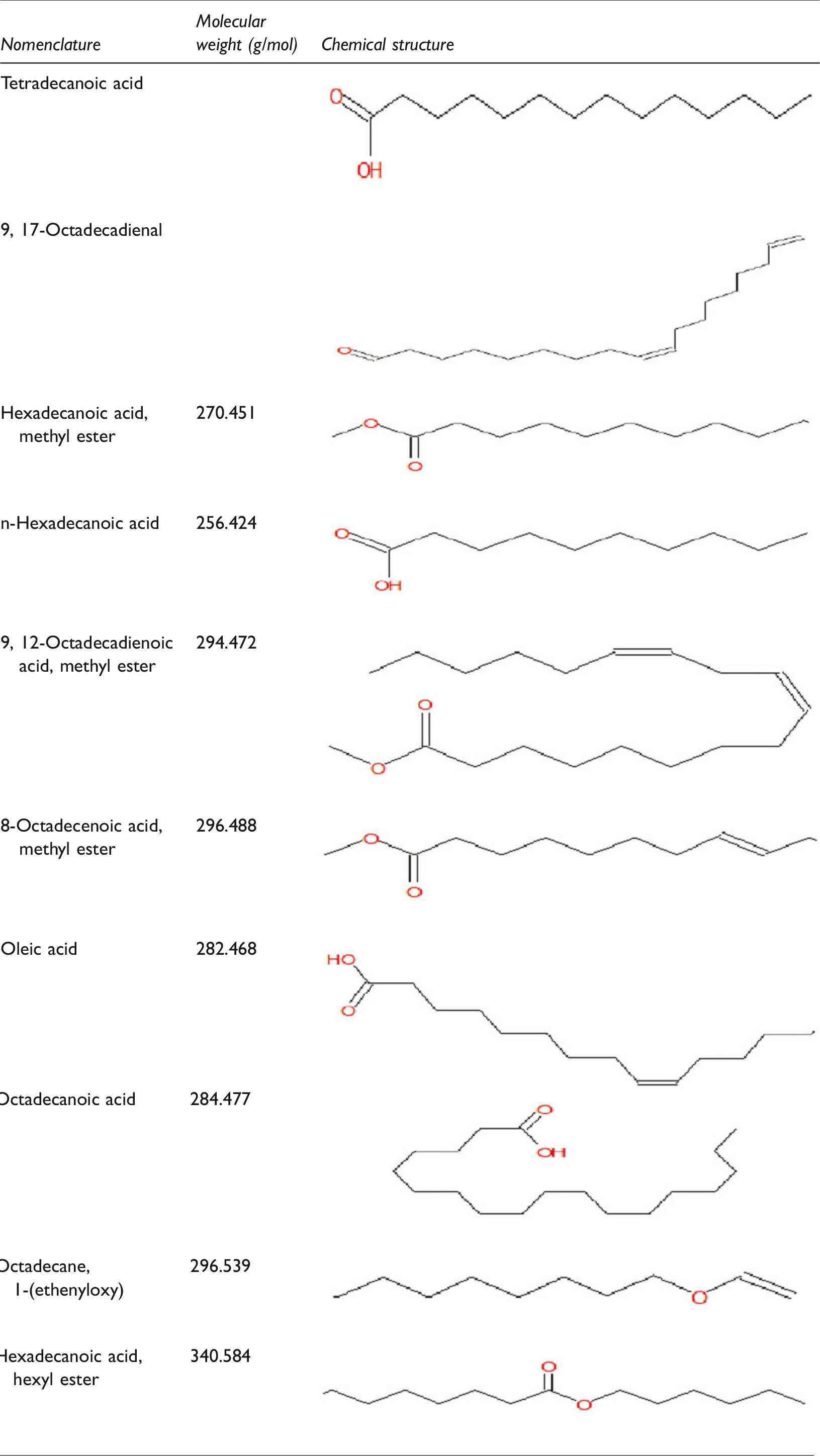
Interfacing a GC system to an MS instrument is relatively straightforward as the compounds eluting from a GC column would have already been volatilized and merely require separation from the carrier gas and ionization before mass analysis. It is thus possible to perform a search against a library of standard MS spectra and, under the right circumstances, identify the analyte. This is a process that has the ability to identify an unknown analyte from a known compound library. The GC-MS analysis shows that the main compositions and constituents of the extracted demulsifier are octadecanoic acid (49.884%) and n-hexaneoic acid (22.951%) which are able to break water-in-oil emulsion. Table 2 shows the summary of the extracted demulsifier investigated in this study. The present demulsifier is green, environmentally friendly and can be used without restriction on-site because of its source. It also supports the move towards more environmentally acceptable chemicals for the petroleum industry, which are less hazardous than the standard or conventional chemicals.
GC-MS analysis summary of the extracted green demulsifier in this study.
GC-MS results
The GC-MS spectra of the extracted green demulsifier as presented in Figure 3 revealed the abundance of the constituent saturated and unsaturated fatty acids and their retention times. Saturated fatty acids of n-hexadecanoic acid (palmitic acid) with a percentage composition of 22.951 and octadecanoic acid (stearic acid) with a percentage composition of 5.096 showed peak retention times at 14.391 and 16.340 min respectively. Also, the unsaturated fatty acids of 9,12-octadecadienoic acid (linoleic acid) and 9-octadecenoic acid (oleic acid) showed percentage compositions of 36.982 and 35.941 at the 16.129 and 16.292 min retention times respectively.
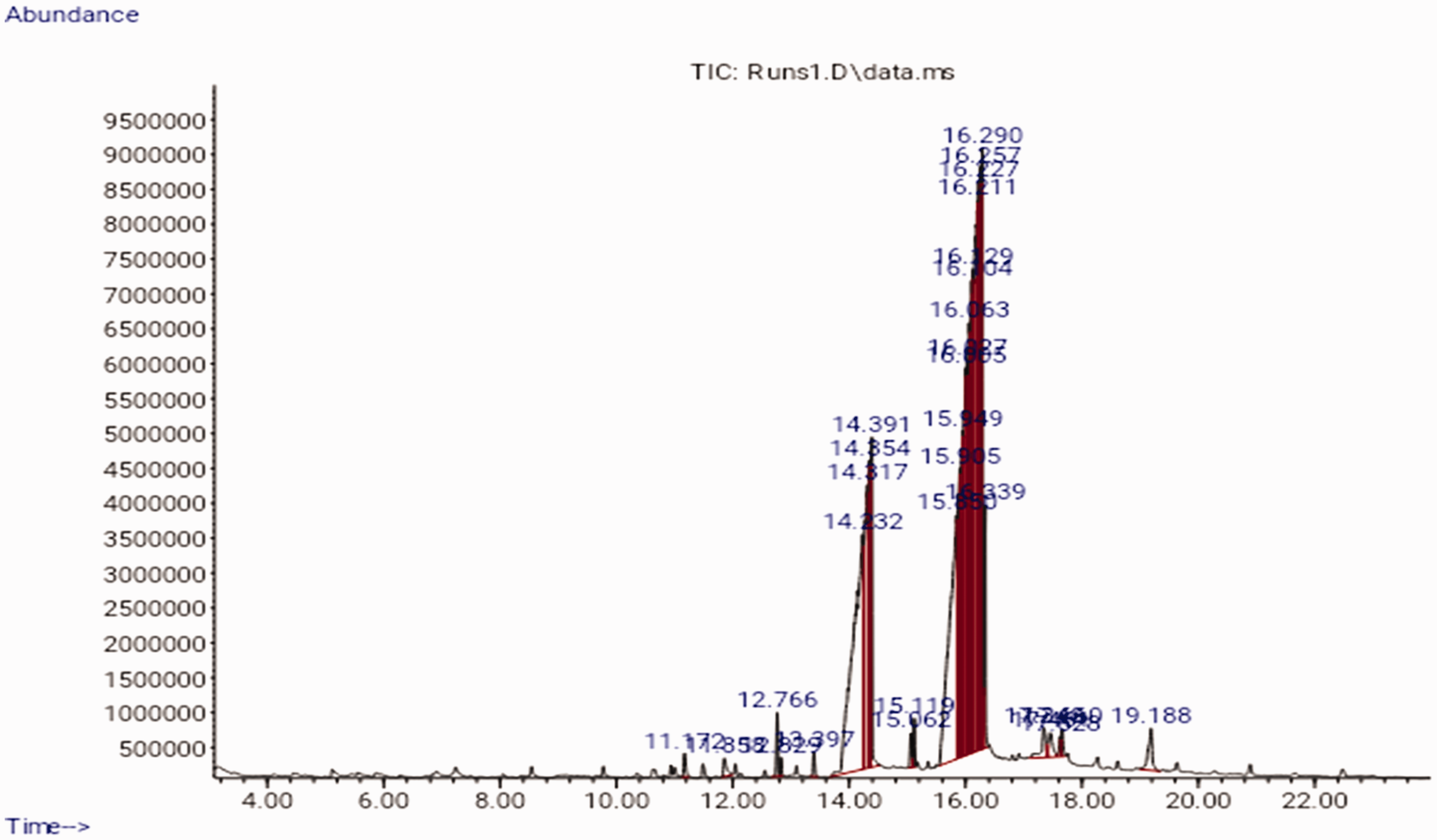
GC-MS chromatography of the extracted green demulsifier.
Other constituent fatty acids include 9,17-Octadecadienal (0.56%, 12.766 min), hexadecanoic acid, methyl ester (0.837%, 13.397 min) and Oleic acid, eicosyl ester (0.976%, 19.187 min), all of which have variously been reported as surface-active agents. More so, their structures and molecular weights are shown in Table 2, confirmed by the fragmentation patterns of 9,12-octadecadienoic acid (294.472 g/mol), 9-octadecenoic acid (282.468 g/mol), n-hexadecanoic acid (256.424 g/mol) and octadecanoic acid (284.477 g/mol) (Figure 4).
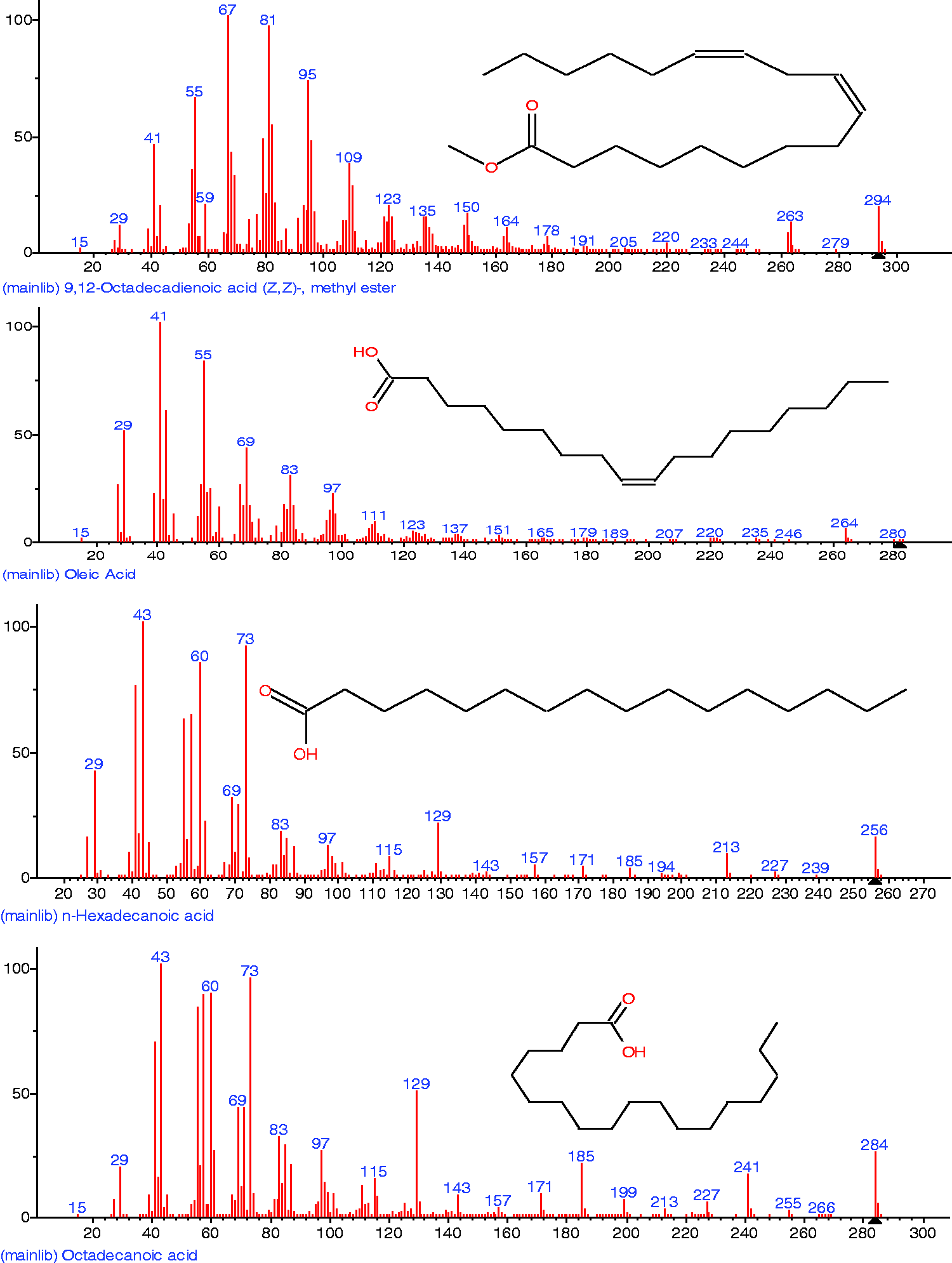
Fragmentation patterns and chemical structure of selected constituent fatty acids of the extracted green demulsifier.
Our GC-MS profile were in agreement with previous reports of Krishnan et al. (2015) on composition of oil extracted from rice bran using hexane, ultrasound assisted extraction and conventional solvent extraction with ethanol. Varying degrees of saturated and unsaturated fatty acids as well as retention times as observed, were a function of total time of scanning, according to NIST08 library of mass spectra.
Also worthy of note, is the high molecular weight of the extracted green demulsifier, which as succinctly put by Roodbari et al. (2016) on the high molecular weight of Tweens, would improve demulsifier performance; with researchers opining that increased concentrations (though not below 500 ppm) and percentage of non-ionic polymers in demulsifiers can lead to better separation and thus, increased efficiency of demulsifiers (Pena et al., 2004; Zaki et al., 1996).
Demulsification performance of the demulsifier
The performance of the demulsifier was investigated under different conditions such as volume, separation time and temperature. After the preliminary tests, the demulsifier was compared with that of a field applicable chemical demulsifier under similar conditions (Figure 5). The volume of water separated from the emulsion system as a function of operating conditions was used to measure the performance of the demulsifier. This was expressed as
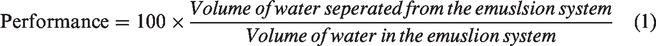

Demulsification at 70°C for the oil field produced-water-in-oil emulsion system.
Eight distinct volumes of the extracted demulsifier were used to investigate its impact on the field-produced crude oil emulsion system. The volumes used were 1, 2, 3, 4, 5, 6, 7, and 8 mL. Figures 4 to 8 show the effects of separation time and volumes on performance at different operating temperatures ranging from 30 to 70°C. A definite pattern was observed for the demulsifier in the test efficiency variables; with an increase in the separation time, the performance of the extract improved. This can be attributed to more time available for the phases to separate. Separation time is essential for such procedures in sizing separators, hence the significance of determining its impact. An increase in the demulsifier volume had a major impact at all temperatures as the demulsifier performance improved. The volume impact is reflective of the demulsifier effectiveness. It can be deduced from the experimental analysis (Figures 6 to 10) that the operating condition is 5 mL for 60 min separation time at temperature of 70°C.
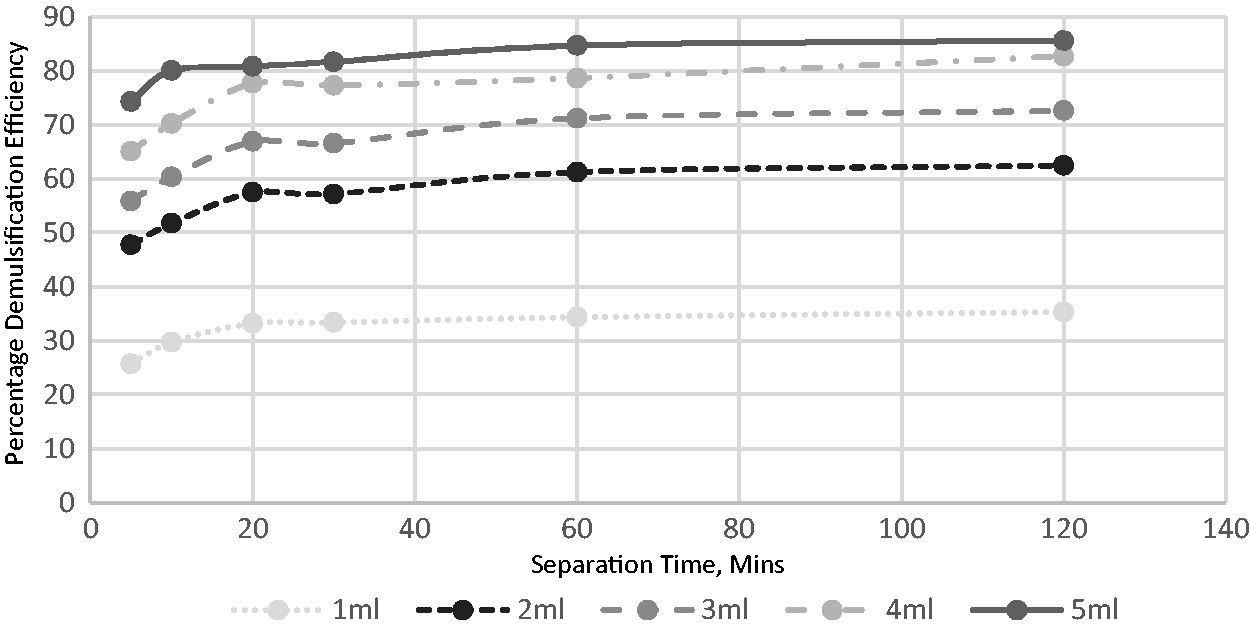
The effect of demulsifier volumes and separation time for breaking the emulsion at 70°C.
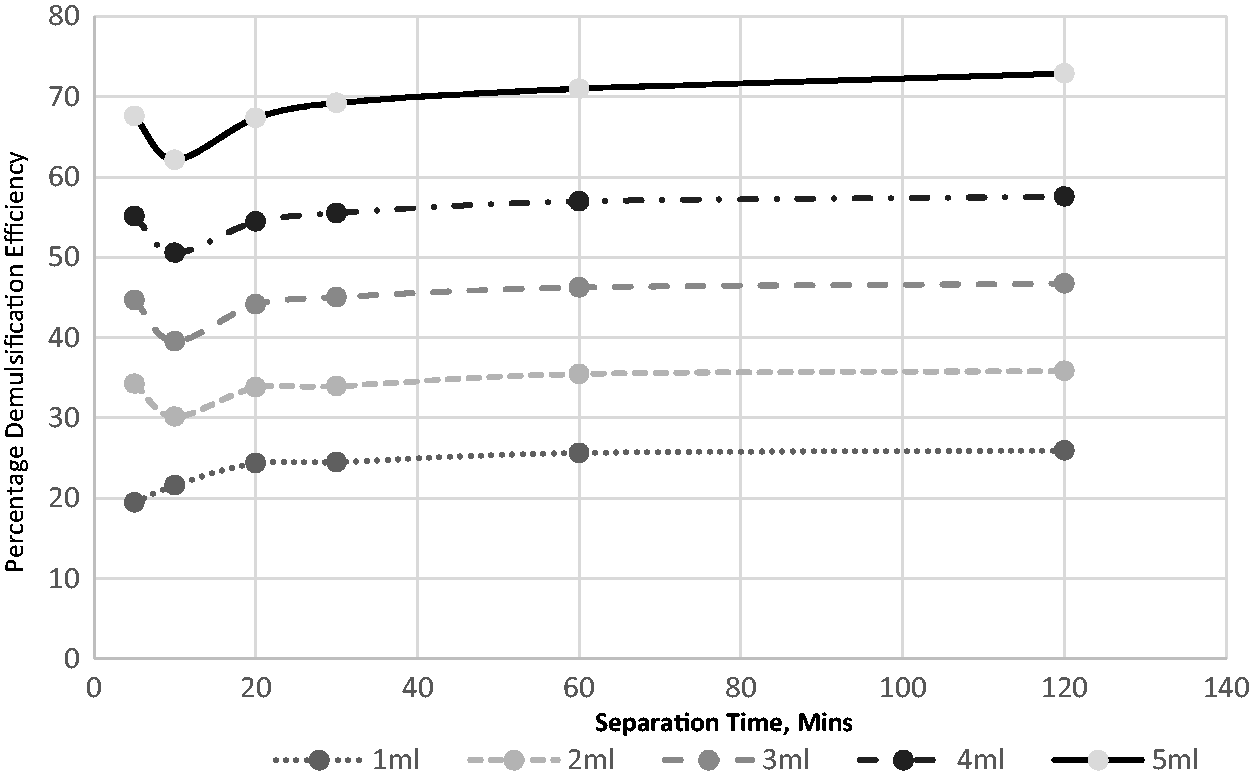
The effect of demulsifier volumes and separation time for breaking the emulsion at 60°C.
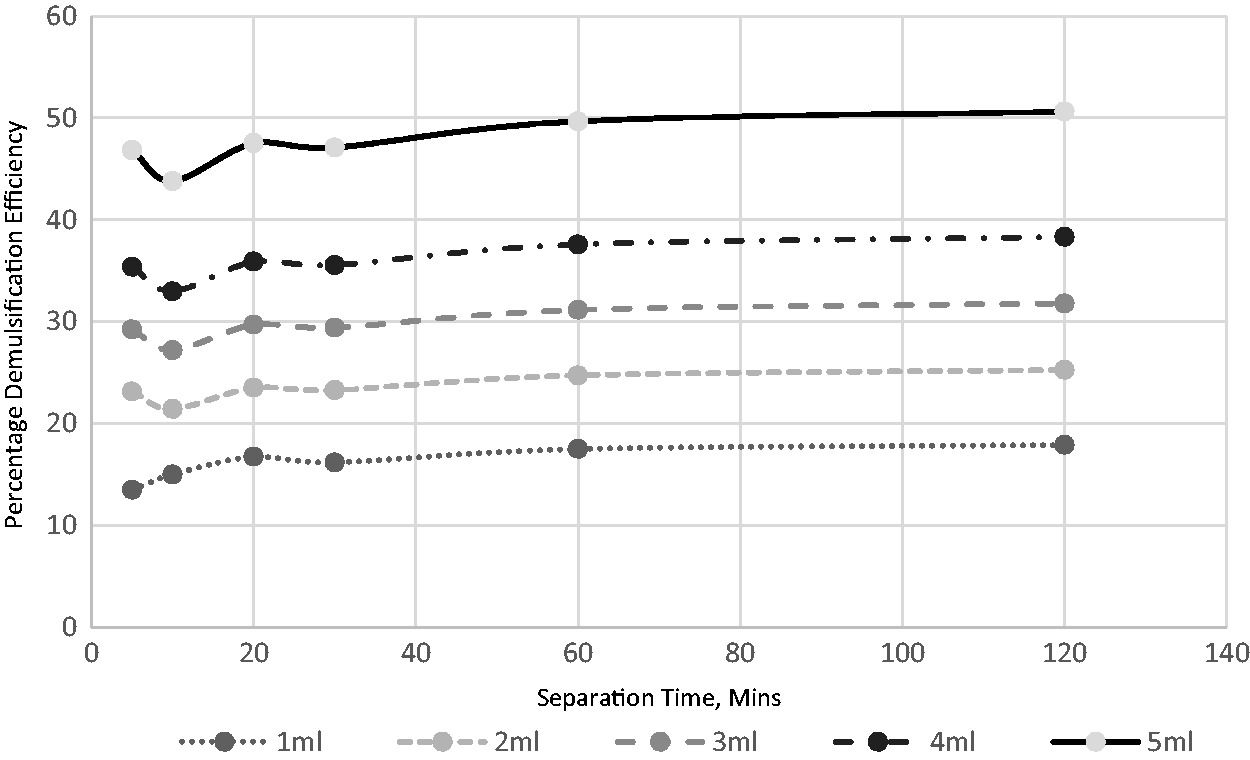
The effect of demulsifier volumes and separation time for breaking the emulsion at 50°C.
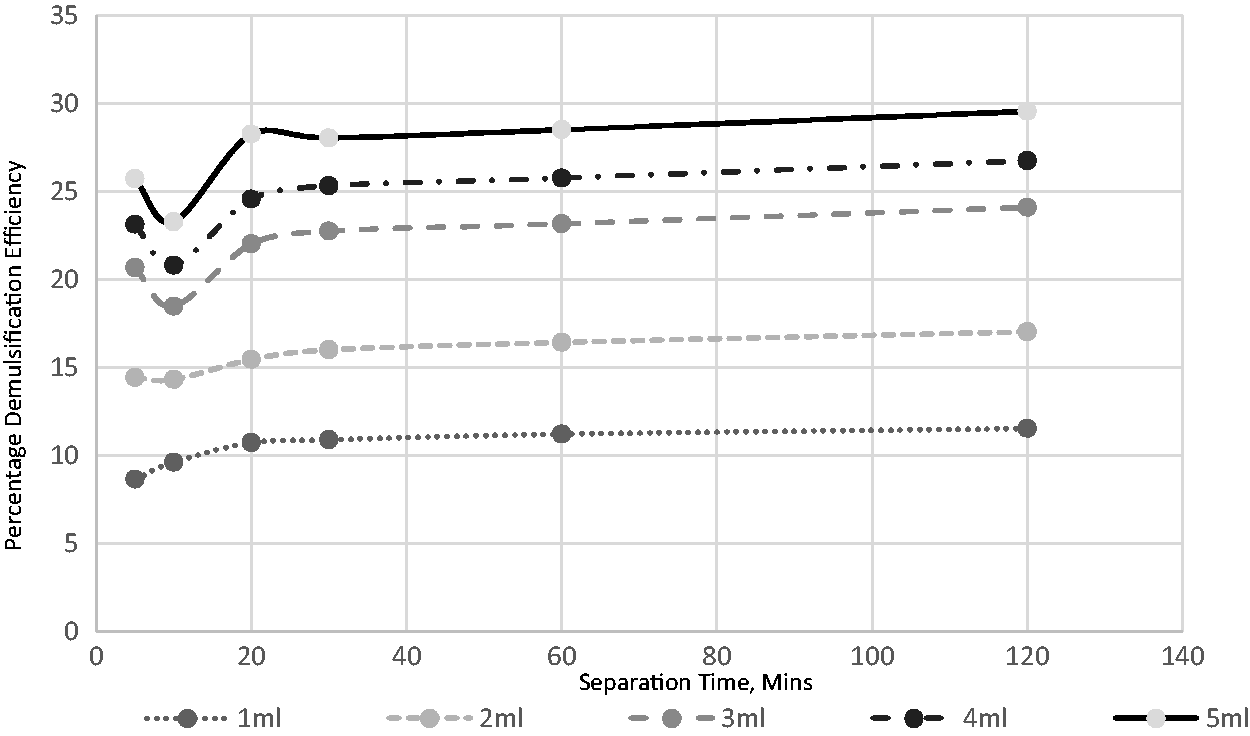
The effect of demulsifier volumes and separation time for breaking the emulsion at 40°C.
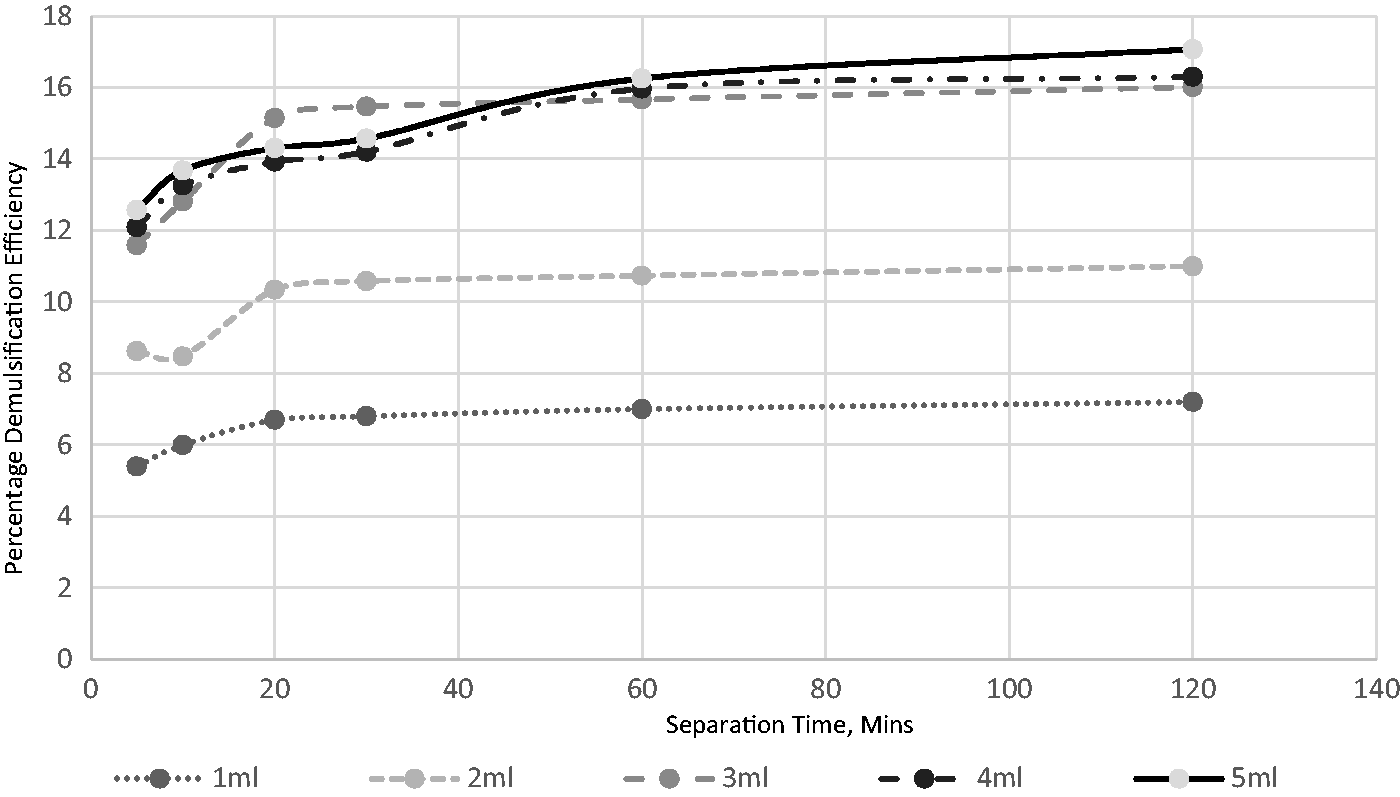
The effect of demulsifier volumes and separation time for breaking the emulsion at 30°C.
This present study assessed the effects of heating on the volume of demulsifiers used for the destabilization and separation of water from crude oil emulsion. Figures 6 to 10 demonstrate that there is a link between demulsification temperature and efficiency for a specified demulsifier volume. In all cases, the water yield from the separation process increased as the temperature increased from 30 to 70°C. This indicates the significance of temperature in the demulsification of the produced crude oil emulsion system.
An increase in temperature favors the breaking of bond and thus, aids in the demulsification process (Figures 6 to 10). There are four benefits achieved through heating, and these include to dissolve the paraffin crystals, increase the relative density of the water and crude oil, increase droplets and reduction in viscosity (Akbari et al., 2016). The selection of high environmental temperature for chemical demulsification will affect the estimation of the actual volume of demulsifier needed for the demulsification process in the field (Table 3). Often, the environmental temperature of the separation process is within the range of 30 to 70°C (Roodbari et al., 2016). The process optimization is attached as supplementary document.
Concentrations and their Efficiencies for different temperature.
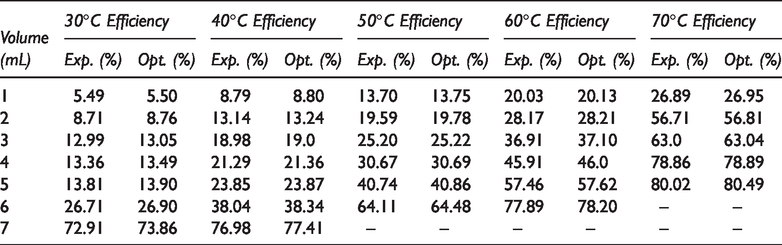
Table 3 shows that in order to obtain the best results, an increase in temperature affects the volume of demulsifier needed to achieve the separation. For the volume of demulsifier used, it was observed during the bottle test experiment that at lower volumes (in drops to about 2 mL), the demulsifier could not effectively break the emulsion at lower temperatures, but the actual demulsification began for demulsifier volumes of 3 to 7 mL depending on the temperature range. Above 7 mL and at 30 to 40°C, a binding effect (anti-bonding) was observed which affected the demulsification process efficiency negatively (Graham et al., 2008). Also, above 6 mL for 50 to 60°C and 5 mL for 70°C, a binding effect (anti-bonding) was observed. The optimization equations for the different temperatures (30 to 70°C) are presented as equations (2) to (6). The optimum conditions were 5 mL at 70°C within 60 min of experimental time.
The optimization equations at 30, 40, 50, 60 and 70°C are presented as equations (2) to (6), respectively.
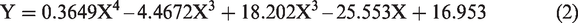
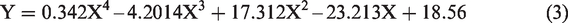
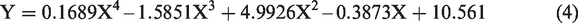
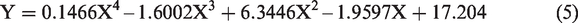
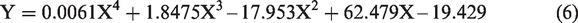
Comparing the extracted demulsifier with field applicable chemical demulsifier
The commercial and extracted demulsifier performance is contrasted under comparable circumstances as shown in Figure 11 (5 mL and 70°C). The field applicable chemical demulsifier sample was collected from the “Field X” production facility in Bayelsa and transported to Covenant University. Both demulsifier samples showed similar trends in terms of demulsification performance and time/temperature (Figure 11) i.e. with an increase in time and separation temperature, the performance of the commercially available and extracted demulsifiers improved. However, the level of efficiency obtained during demulsification is dependent on the nature and source of the demulsifier. It was observed in this study that at the operating conditions, the performance of the extracted demulsifier was better than the chemical demulsifier.
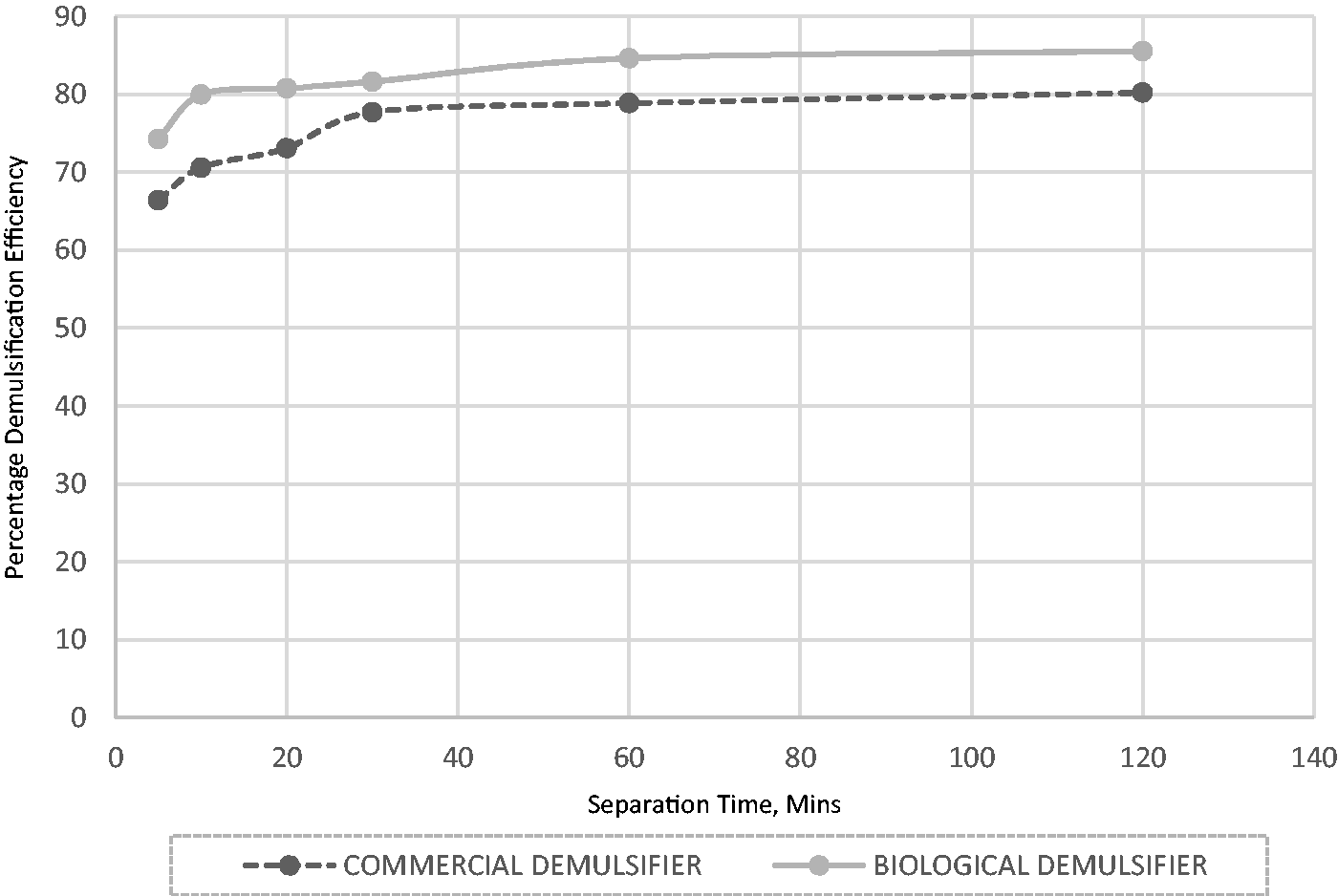
Comparison of the demulsifiers at 5 mL volume and temperature of 70°C.
Factors that influenced the demulsifier effect on the demulsification process
The Pareto graph was used to determine which variable(s) had the most statistically important effects on the demulsification process. It indicates the absolute value of the standardized effect of separation time, temperature, and volume from maximum effect to minimum effect. The reference blue row on Figure 12 assisted in determining which impacts (single or mixed) are significant to the response. On the Pareto chart, bars that cross the reference red line (1.978) are statistically significant. From the Pareto chart (Figure 10), it was observed that (AC) combined effect of temperature and volume had the greatest impact on the volume of water expelled from the emulsion, followed by (A) temperature, then demulsifier volume (C) and then time (B). Thus, these factors are statistically significant at the 0.05 level with the current model terms.
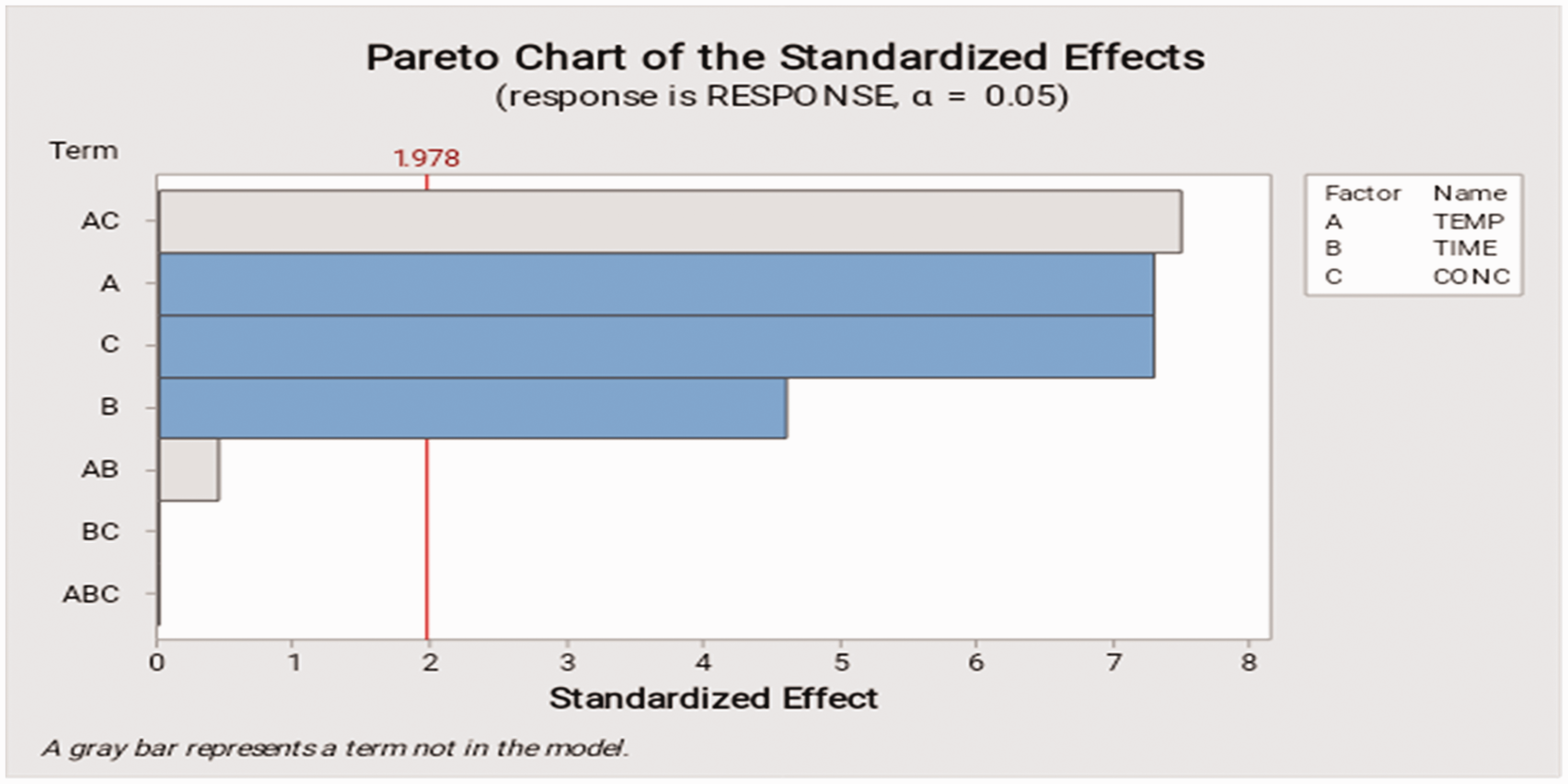
Pareto chart of the standardized effects.
A contour chart was further introduced in this section (Figure 13); it is a chart used to determine three variables prospective interconnections. The plot shows the effects of time, volume and temperature on the volume of water expelled from the crude oil emulsion. Figure 13(a) shows the combined effect of time and temperature. The dark green region revealed the ratio of time and temperature that will cause the highest volume of water to be expelled from the crude oil emulsion while the dark blue region showed the combination with the least effect. Figure 13(b) shows the combined effect of temperature and volume; and Figure 13(c) shows the effect of volume and time. The dark green shows the range of temperatures and volume in which their combined effect will be significant; while the dark blue region shows the range where the combined effect of temperature and volume will be insignificant.
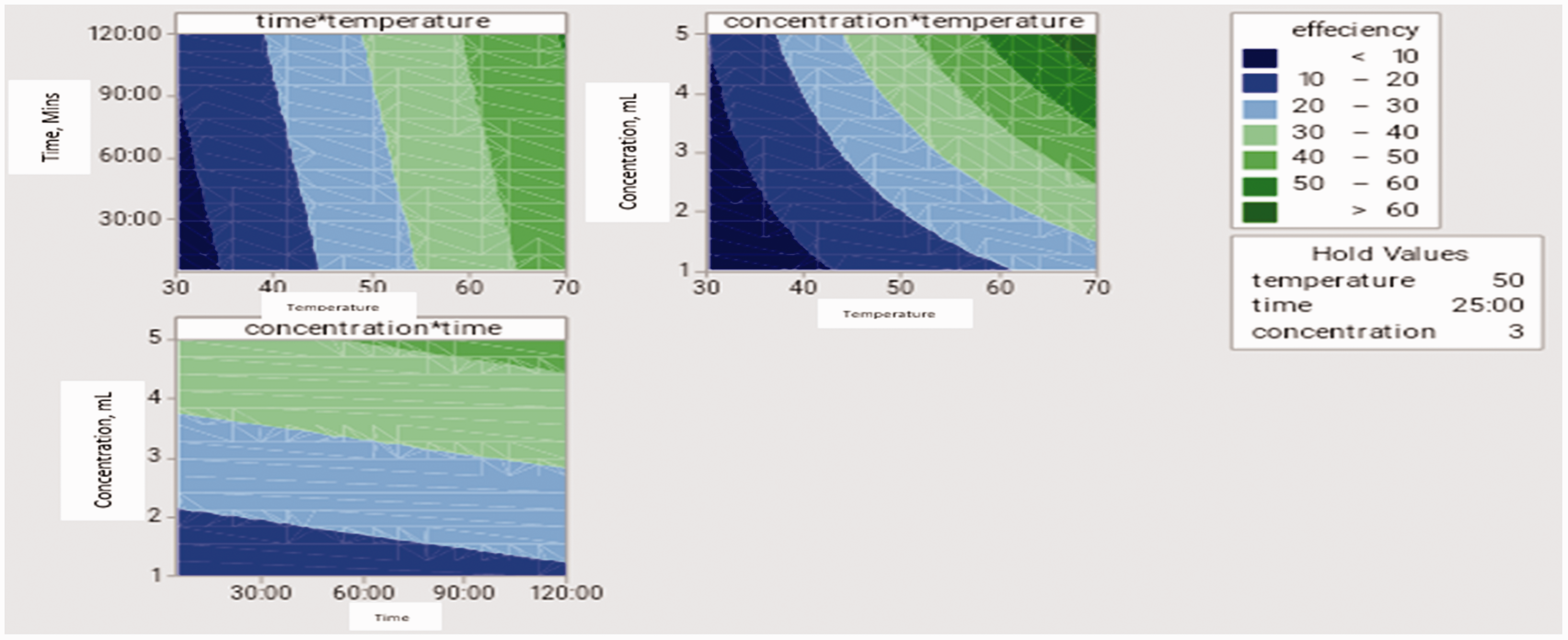
Contour plots for the changes experienced by interfacing factors.
The normal effects plot (Figure 14) was used to compare the relative magnitude and the statistical significance of both the main and interaction effects; because the Pareto plot presents the entire value of the effects, but cannot define which effects decrease or increase the response. The normal presented in Figure 14 was used to survey the direction and magnitude of the effects at the same time. The factors that have significant effects are shown in red. The farther a factor is from the red line, the more significant the effect it has on the corresponding response. From Figure 14, it was observed that temperature (A) has the most significant effect of 44 with a corresponding magnitude of 82%. Volume (C) was the next most significant factor with a significant effect of 27 and magnitude of 61%; and this is followed by time with an effect of 6 and a magnitude of 17%.
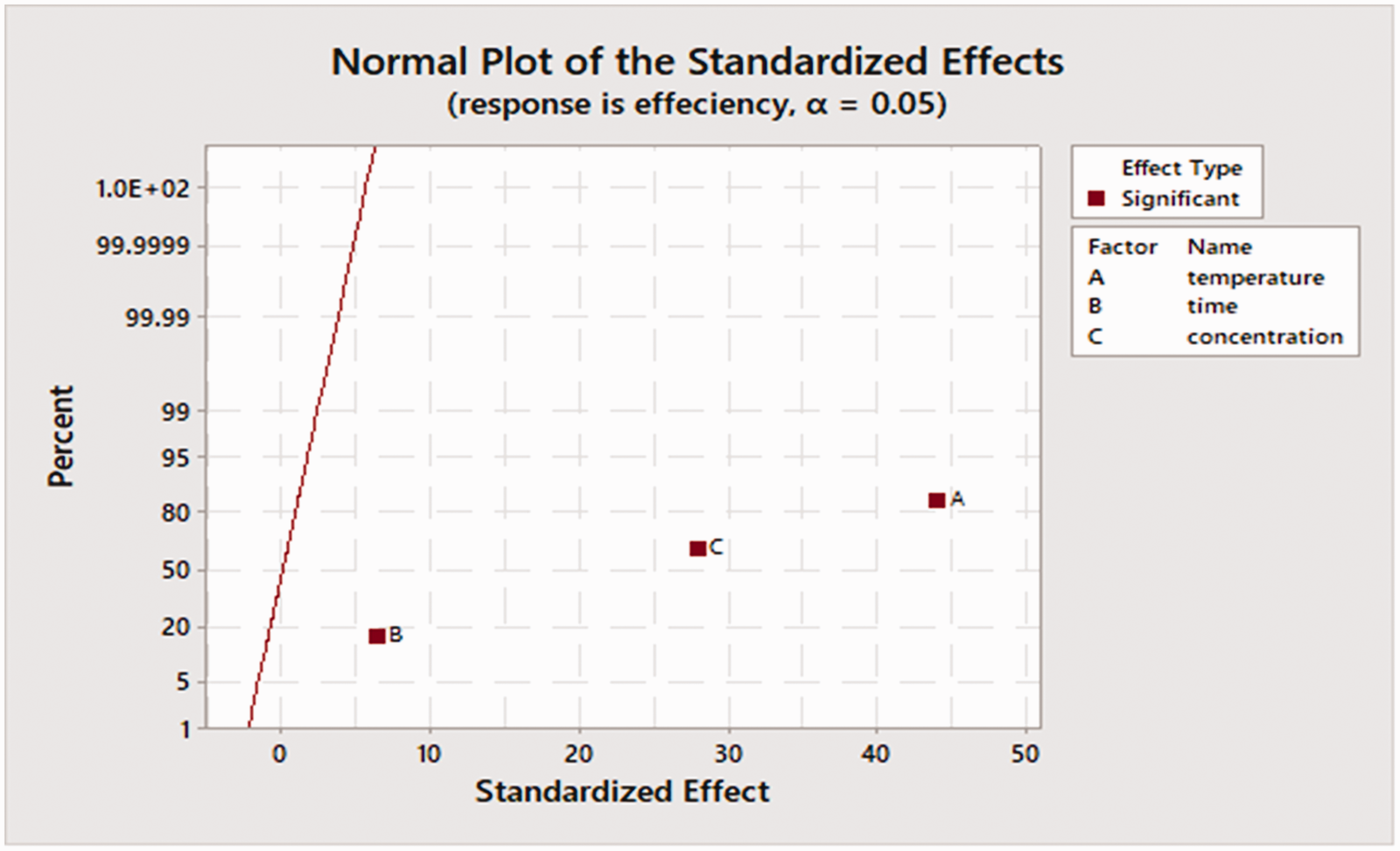
Normal plot.
Effects away from 0 on the X-axis have greater magnitude and are more statistically significant; thus, showing the magnitude, direction, and the importance of the effects. Figure 14 shows that the influence of the separation temperature is much more important than the separation time within the experimental conditions of this study. Separation temperature (A), time (B), and volume (C) have a positive standardized effect and are statistically significant at the 0.05 level. This means that the response increases when the process changes from a low level to a high factor level.
Conclusion
It has been shown in this study that rice husk can be fully utilized in the petroleum industry as a natural alternative to chemical demulsifiers. From the experimental results, one can conclude that:
The performance of the demulsifier increased as temperature and separation time increased. The overall efficiency was improved with an increase in separation time at all volumes and temperatures considered. The effect of temperature on performance was greater than the corresponding effect of volumes. The operating condition is 5 mL for 60 min separation time at temperature of 70°C.
Supplemental Material
EEA905080 RESPONSE SUPPLEMENTARY FILE - Supplemental material for Evaluation of a naturally derived waste brown oil extract for demulsification of crude oil emulsion
Supplemental material, EEA905080 RESPONSE SUPPLEMENTARY FILE for Evaluation of a naturally derived waste brown oil extract for demulsification of crude oil emulsion by Emmanuel E Okoro, Chinedu G Nnaji, Samuel E Sanni, Eze F Ahuekwe and Kevin C Igwilo in Energy Exploration & Exploitation
Footnotes
Acknowledgements
The authors would like to thank Covenant University Centre for Research Innovation and Discovery (CUCRID) Ota, Nigeria for its support in making the publication of this research possible.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
