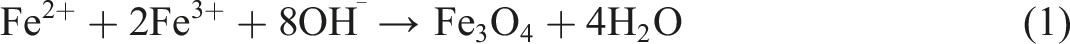Abstract
By using a traditional roll mill, nitrile butadiene rubber (NBR)/magnetite nanocomposites for electromagnetic interference shielding applications were successfully prepared. The synthesized magnetite nanoparticles were analyzed using X-ray diffraction (XRD), Fourier transform infrared (FTIR), transmission electron microscope (TEM), and energy dispersive X-ray (EDX) techniques. The results from these techniques emphasis the preparation of Fe3O4 with a diameter range between 3.8 nm and 19 nm. Before and after gamma irradiation at different doses the impact of adding different contents of magnetite nanoparticles in NBR was carefully examined through mechanical and electrical measurements for all samples at room temperature. The mechanical parameters and the electrical properties of NBR were enhanced after adding Fe3O4 nanoparticles. Electromagnetic interference shielding (EMI) for all fabricated nanocomposites before and after gamma-ray irradiation under the same conditions of pressure, humidity and temperature was performed as a promising application for these materials in practical life. The electromagnetic shielding effectiveness (SE) of the prepared samples was measured in the X-band of the radio frequency range. There are three global maxima around 9.4 GHz, 10.4 GHz, and 11.4 GHz. Subsequent reinforcement of Fe3O4 nanoparticles into NBR produced higher shielding effectiveness for radio frequency signals. Furthermore, applied gamma radiation doses improved the shielding properties of the fabricated nanocomposites.
Keywords
Introduction
With the rapid advancement of electronic and communications technology, as well as the expanding microwave industry, electromagnetic radiation has become a new source of contamination in contemporary society, ranking as the third most common pollutant. 1 Certain electronic gadgets emit electromagnetic waves that can interfere with other equipment’s functionality and lead to faults in medical devices. 2 The electromagnetic radiation produced during the operation of 5G-communication technology and other linked devices endangers people’s health and the correct functioning of minuscule and intricate electronic circuits and components.3,4 High-performance electromagnetic interface shielding is an effective way to diminish the adverse impact of these radiations. 5 The development of electromagnetic interference (EMI) and shielding effectiveness (SE) materials for harmful waves is gaining more attention in an effort to mitigate its effects. There are two forms of EMI/SE materials, namely metal-based and polymer-based. Steel, copper, and aluminum are examples of metal EMI shielding materials with the drawbacks of being physically inflexible, heavy, and prone to corrosion. Recently, polymer nanocomposites received much more attention regards to its light weight, electrically conducting, low cost, flexibility, and corrosion resistance.6,7
Several rubbers are frequently employed for the production of these types of composites, such as natural rubber, styrene-butadiene rubber, acrylonitrile butadiene rubber (NBR), and ethylene propylene diene monomer.8,9 In the current investigation, we utilized NBR because of its excellent low temperature flexibility, heat resistance, swelling resistance, and particularly exceptional oil resistance because of the polar nitrile groups. 10 Hybridization, or the introduction of high-strength conductive reinforcement fillers, is one technique for improving the mechanical and electrical performance of the rubber matrix. 11 A crucial technique for creating a charge carrier channel within an insulating polymer matrix is the use of conductive additives such as graphite, conducting organic polymers, carbon black, carbon fiber, carbon nanotubes, and magnetite (Fe3O4) nanoparticles.12,13 Within the past 10 years, authors have developed an interest in magnetic nanopowder-reinforced polymer composites. 14 Nano-Fe3O4 an excellent choice to fabricate magnetic polymer composite for EMI shielding at very low loading. 15 Therefore, composites of polymer/Fe3O4 nanoparticles can be currently used in high-performance micro and millimeter stealth materials, stealth construction materials and radiation-shielding cell phone materials. 16 Nasouri et al., found that the magnetic properties and electrical conductivity of the polyvinylpyrrolidone (PVP)/Fe3O4 composite were improved with an increase in the Fe3O4 nanoparticles’ concentration and at 4 wt% Fe3O4 nanoparticles can be used as an effective EMI shielding material. 17 El-Nemr et al. found that, the dielectric and mechanical properties of NBR/Fe3O4 composites depend on the concentrations of Fe3O4. 18 When a polymer is exposed to high-energy radiation, it absorbs energy and becomes electronically excited, or ionized. These excited molecules can then initiate chemical processes that produce reactive products that can cause deterioration and crosslinking within the macromolecules. 19 Ionizing irradiation (γ-rays) produces secondary electrons after absorbing energy in a substance. Radiation technology is a potential approach for the synthesis of multiple membranes, hydrogels, and composites. Many research articles have been documented exploiting the irradiation of polymeric materials for use in various technologies and applications. 20 Tarawneh et al. found that, the irradiation increased the interactions between CNTs and montmorillonite, resulting in better dispersion of the nanofiller. 21 This finally resulted in improved mechanical and thermomechanical characteristics. NR exhibits a linear rise in tensile modulus as the irradiation dosage increases from 0 to 250 kGy, mostly owing to an increase in crosslinking density, as confirmed by Moustafa et al. 22
The objective of this research is to synthesize an electrically conductive nanocomposite formulated from the synthetic rubber matrix, acrylonitrile butadiene rubber (NBR), reinforced with magnetite nanoparticles, to serve as innovative electromagnetic interference shielding. Fe3O4 filler was preferably selected due to its superior mechanical and electrical qualities. The synergistic effects of gamma irradiation and the magnetite content on the composites’ network structure were studied. The effect of Fe3O4 nanoparticles contents and the gamma irradiation on the mechanical and electrical properties of the prepared samples was investigated. The suitability of the examined nanocomposites for shielding against electromagnetic interference was scrutinized.
Experimental part
Materials
Ferrous chloride tetra-hydrate (FeCl2⋅4H2O), ferric chloride hexahydrate (FeCl3⋅6H2O and ammonium hydroxide (NH4OH, 26 % ammonia) were obtained from Sigma Aldrich. A commercial NBR with density = 0.98 g/cm3, acrylonitrile content = 34 %, Mooney viscosity ML(1+4) at 100°C = 50 was supplied by Alexandria Trade Rubber Co., Egypt.
Magnetite nanoparticles synthesis
The co-precipitation method was used to create the Fe3O4 nanoparticles. The reaction conditions were carefully regulated in order to produce the desired nanoparticles. To make this preparation, Ferric and ferrous chlorides were mixed in a 2:1 M ratio. The Fe2+ and Fe3+ aqueous solutions were prepared in distilled water, then heated up to 70°C for 10 min. Solution spin speed was accelerated and concentrated NH4OH was drop wisely added until pH = 11 was reached. The production of magnetite nanoparticles was indicated by the shift in solution color from bright orange to black particles. A powerful magnet was used to separate the particles from the generated solution after it had been repeatedly cleaned with distilled water. The magnetite black powder was dried overnight at 100°C using hot air oven. The synthesis steps were showed in Figure 1. According to Thong et al., the process of creating magnetite nanoparticles may be explained as follows:
23
Synthesis steps of magnetite nanoparticles (Fe3O4).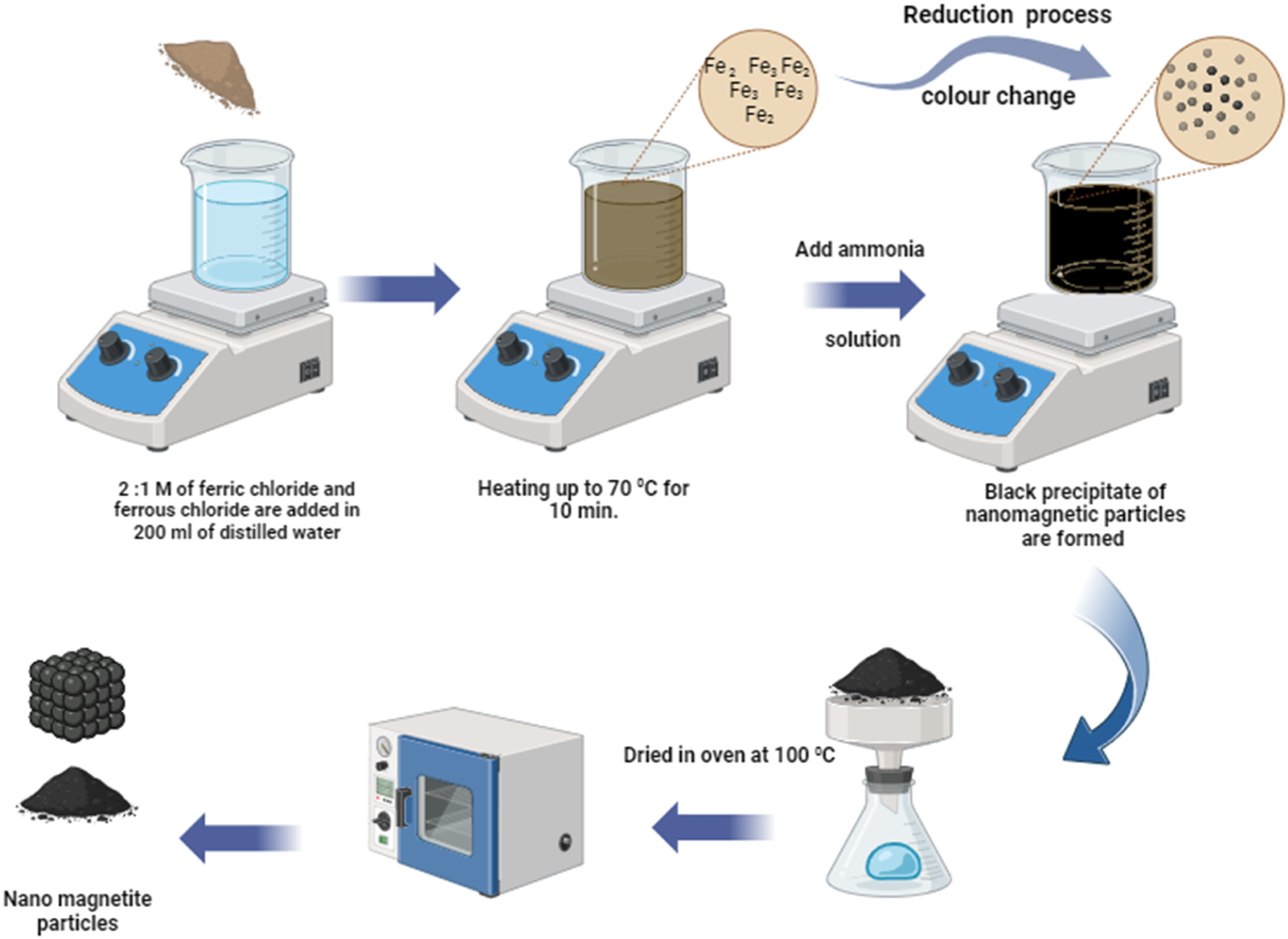
Nanocomposites preparation
Using a two-roll mill at room temperature, the produced nanomagnetite (Fe3O4) was added to NBR in varying contents (5, 10, and 15 phr) with other ingredients. The nanocomposites were forced into a 2-mm-thick sheet at 16 MPa pressure using a hot press for 10 min at 130°C, as depicted in Figure 2. Preparation of NBR/Fe3O4 nanocomposites.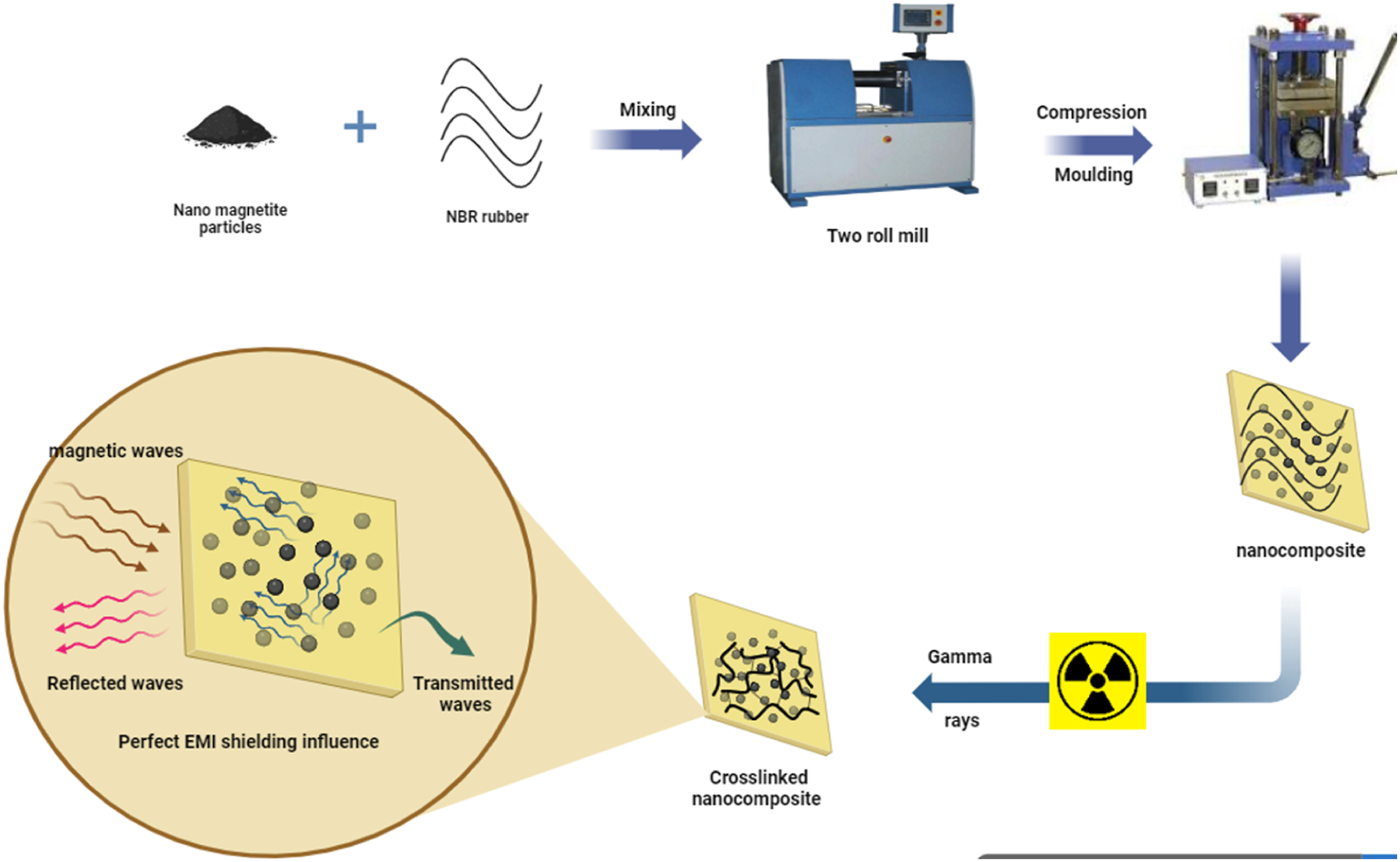
Exposure to gamma rays
The prepared nanocomposites were exposed to gamma radiation at a dosage rate of 0.7 kGy/h using Cobalt-60, Indian radiation exposure unit, at the National Centre for Radiation Research and Technology, housed in the Egyptian Atomic Energy Authority (EAEA), Cairo, Egypt.
Characterization
Using Cu-kα radiation at a wavelength of 0.1541 nm at room temperature and a generator voltage of 40 kV and generator current of 30 mA, the Philips X’pert X-ray diffraction pattern (XRD) between 2θ of 5° – 80° was obtained at a scan rate of 2 °/min on the diffractometer. Scherrer’s formula was employed to establish the crystallite particle size (D):
24
The constant k is assumed to be 0.935, λ is the wavelength of X-rays (λ = 0.1541 nm), and β2θ is the peak’s full width at half its maximum (FWHM). The Fourier transform infrared (FTIR) spectra of Fe3O4 nanoparticles and the prepared nanocomposites were determined (400 - 4000 cm−1) using VERTEX 70, which is based on a wide range DLaTGS detector and beam splitter. Fracture sample morphological observations were examined using scanning electron microscopy (SEM) (ISM-5400, JEOL, and Japan). To verify the filler particles size (Nanomagnetite, Fe3O4) features, transmission electron microscopy (TEM) analysis using the JEM-100 CX at an acceleration voltage of 80 kV was carried out.
Mechanical measurements
In accordance with ASTM D 638-14, the tensile strength (TS), elongation at break (%), and elastic modulus were assessed for NBR/Fe3O4 nanocomposites of concern, averaging the results of five specimens/each. The tensile testing machine Zwick, model Z010, Germany, was used along with a load cell of 1 kN at a crosshead speed of 500 mm/min.
Electrical measurements
Dielectric constant (ε`), dielectric loss (ε``), and the electrical AC conductivity (σ
AC
) were measured for neat NBR rubber and its nanocomposites at two different radiation doses (50 and 100 kGy), at room temperature, within the frequency range of 10 - 106 Hz, using a four terminal HP 71 8144A precision RLC meter. Circular samples of diameter 20 mm and thickness of 1.7 mm were used. Both surface areas (A) of samples were coated with high-purity silver paint to form the electrodes. ε` and ε`` were calculated from the following equations:
25
Electromagnetic interface shielding
Shielding effectiveness was measured using a vector network analyzer and a proper wave guide. This measurement uses the R&S ZVA 67 VECTOR NETWORK ANALYZER operates in the range starts at 10 MHz to 67 GHz and a wave guide operates in the X-band from 8 GHz to 12 GHz. The measuring setup was shown schematically in Figure 3. Measuring setup of shielding effectiveness concept.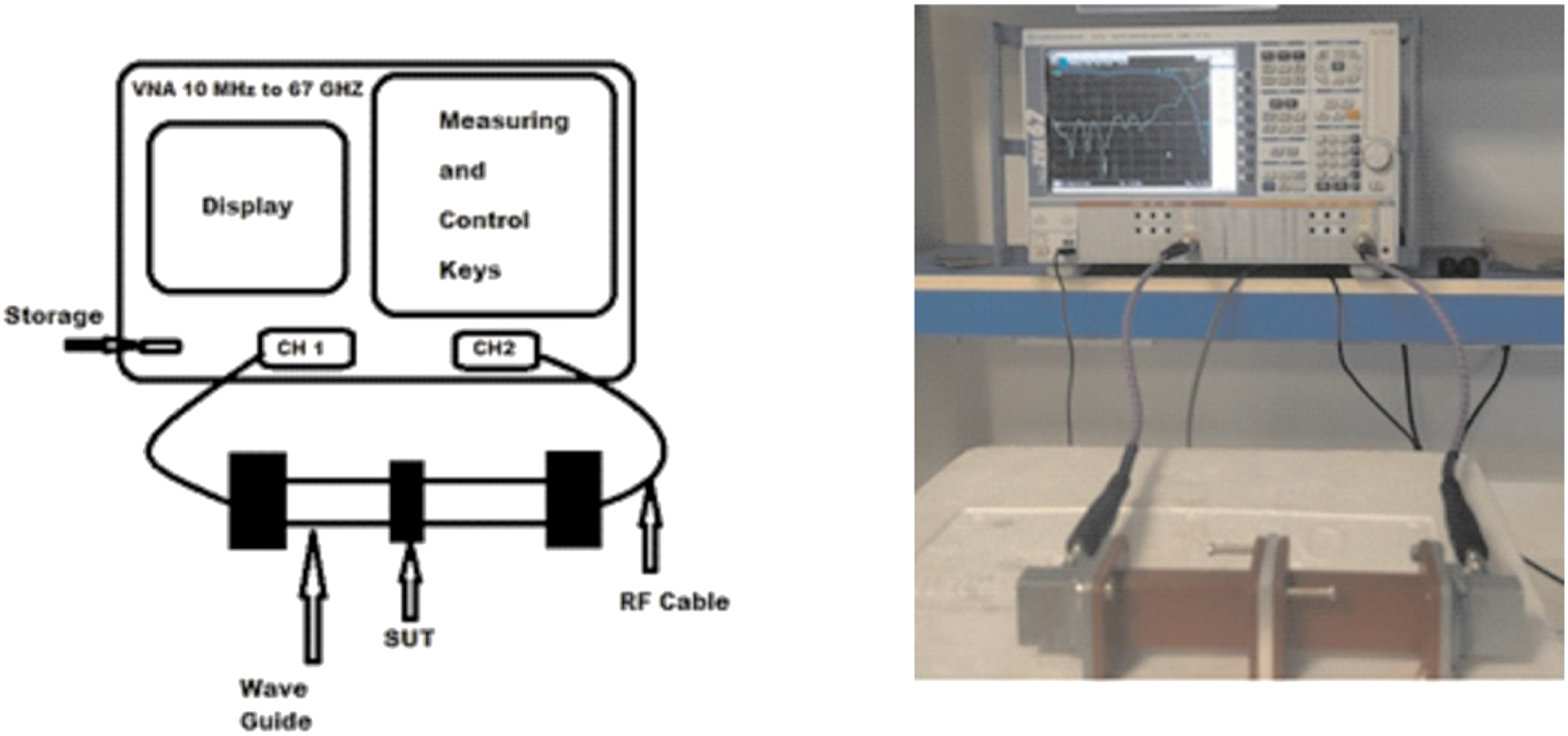
Results and discussion
Characteristics of Fe3O4 nanoparticles
The prepared nano-Fe3O4 particles’ XRD is displayed in Figure 4. The X-ray pattern shows distinct peaks that are exactly equivalent to Fe3O4 nanoparticles at approximately 30.3° (220), 35.5° (311), 43.325° (400), 53.7° (422), 57.2° (511), and 62.9° (440). These six distinctive peaks indicate the purity and crystallinity of the artificially prepared Fe3O4 nanoparticles. When Scherrer’s formula [Equation (1)] is applied, the particles size of Fe3O4 particles are about 10 nm. The synthesized Fe3O4 lattice constant was determined to be 8.357 Å, which is in good agreement with previously published results.
26
XRD of Fe3O4 nanoparticles.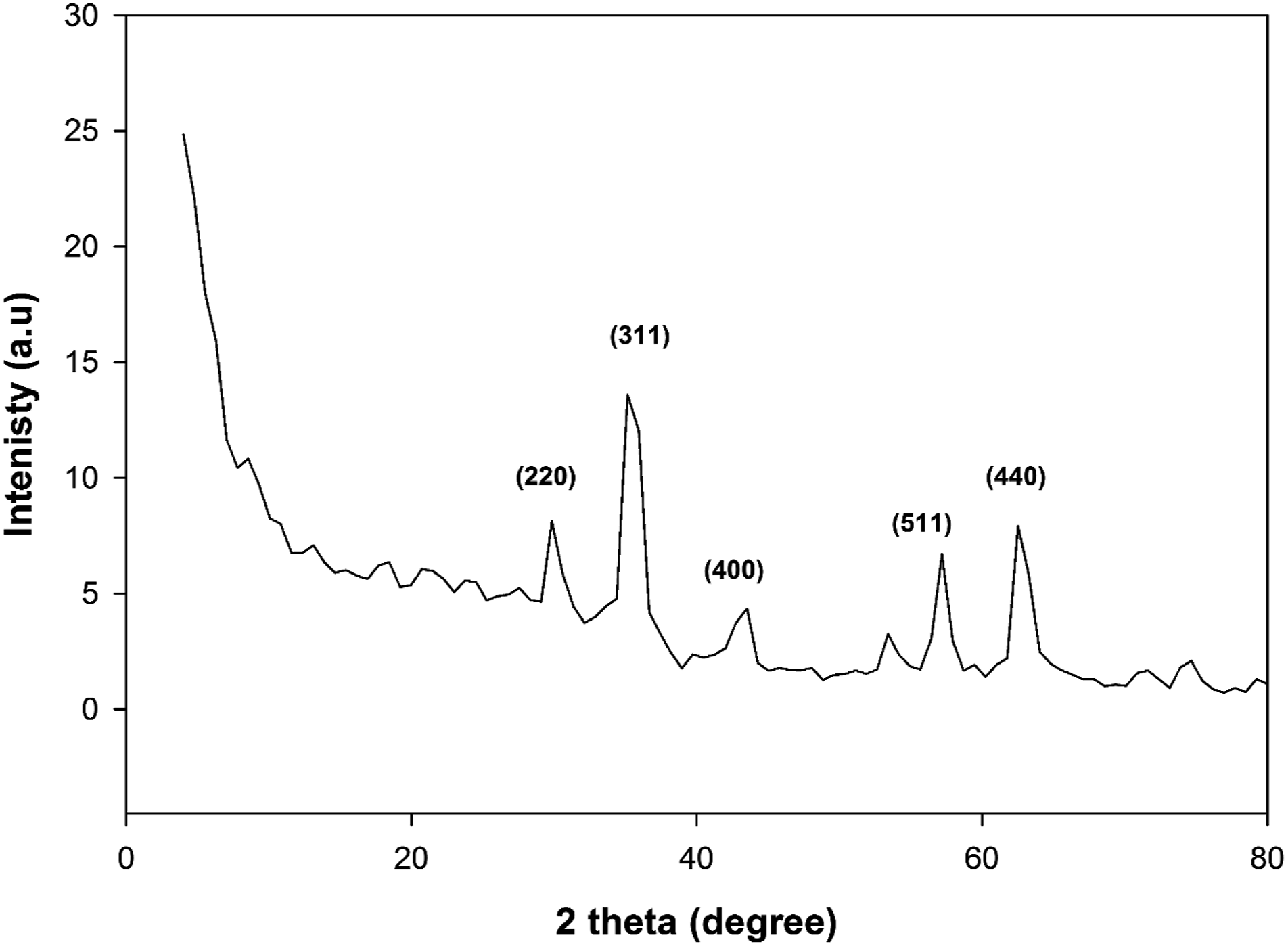
The FTIR spectrum of scattered Fe3O4 nanoparticles in deionized water is displayed in Figure 5. The two peaks at approximately 550 and 600 cm−1 are associated with Fe–O vibrations for magnetite nanoparticles.
27
On the other hand, the two detected peaks, 1637 and 1038 cm−1, which are linked to –OH bending vibrations in conjunction with Fe atoms, show that Fe3O4 nanoparticles were successfully synthesized.
28
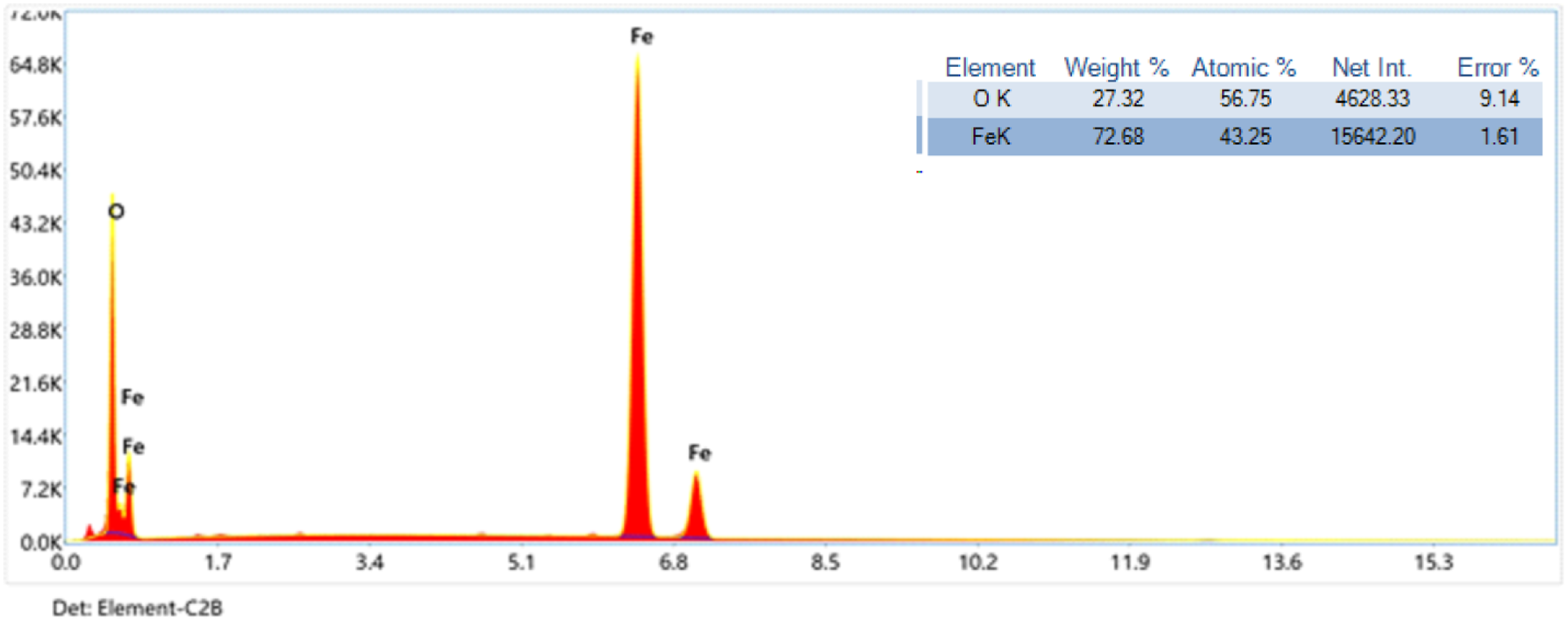
The large peaks at 3078 cm−1 correspond to the O-H group’s stretching vibration. The obtained Fe3O4 nanoparticles powder’s SEM image, which displays a spherical-like particle shape, is shown in Figure 6(a). The TEM image of Fe3O4 nanoparticles is displayed in Figure 6(b), which indicate a diameter particles size of about 3.87–16 nm as previously resulted from XRD (Figure 4) by Scherrer’s equation. Figure 7 displays the strong peaks of Fe and O atoms by EDX spectrum. The Fe3O4 nanoparticles formed by the co-precipitation synthesis were contained of 72.68 wt% (Fe) and 27.32 wt% (O). These results emphasis the purity of the prepared Fe3O4 nanoparticles. FTIR spectrum of Fe3O4 nanoparticles. (a) SEM and (b) TEM images of Fe3O4 nanoparticles. EDX spectrum of Fe3O4 nanoparticles.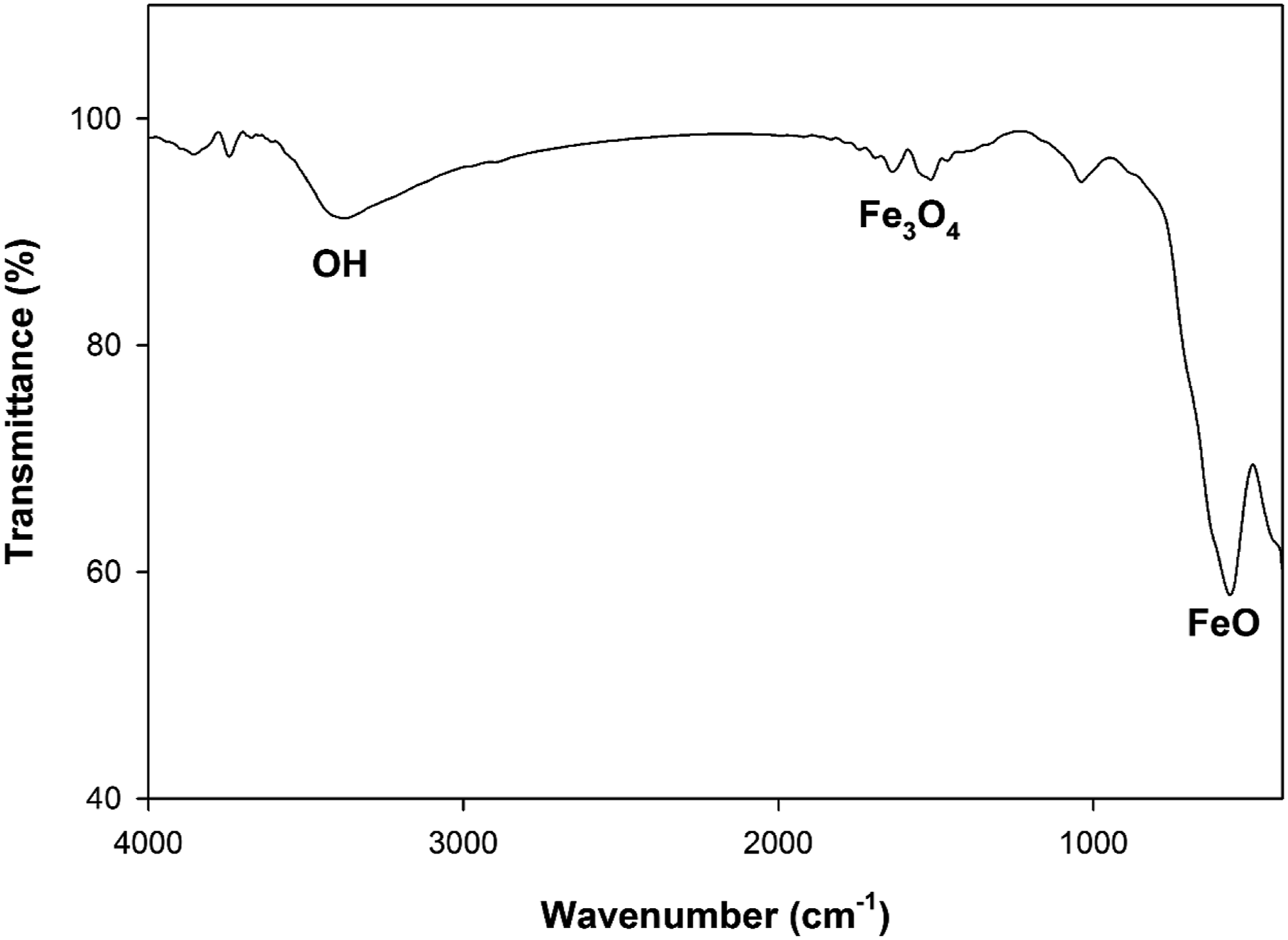

Characterization of NBR/Fe3O4 nanocomposites
Figure 8 shows the XRD patterns of pure NBR and NBR/Fe3O4 nanocomposites filled with different contents of Fe3O4 nanoparticles (5, 10, and 15 phr). The amorphous character of NBR is represented by the large diffraction peak achieved at 2θ = 18.72° with an interplanar spacing for of 4.73 Å.
29
There are multiple peaks for NBR/magnetite nanocomposites that are entirely matched to Fe3O4 nanoparticles at 30.5° (220), 35.5° (311), 43.2° (400), 53.4° (422), 57.3° (511), and 62.6° (440). This suggests that there is a great dispersion of Fe3O4 nanoparticles inside the NBR matrix.30,31 XRD pattern of NBR and NBR/Fe3O4 nanocomposites.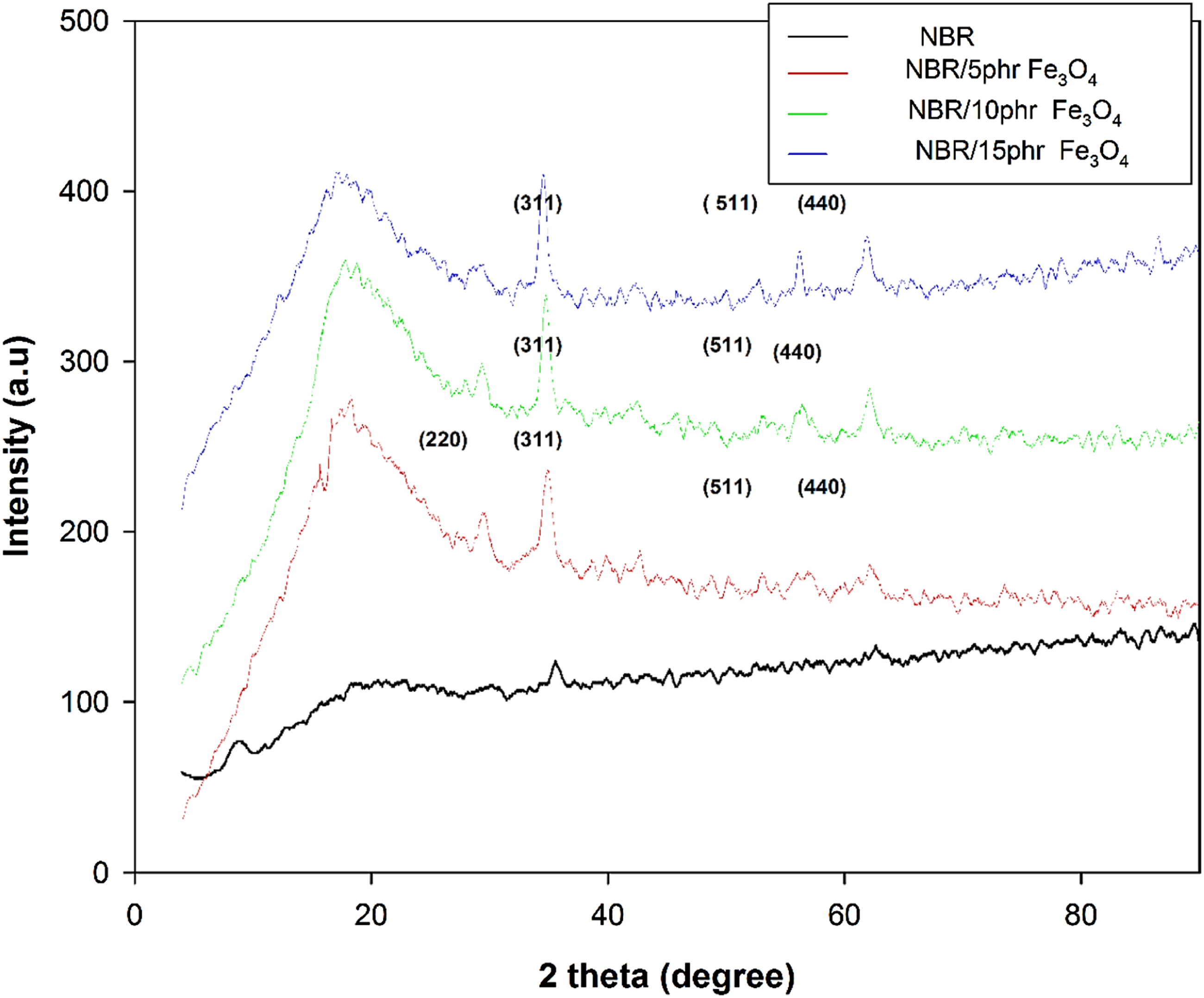
Mechanical part
The tensile properties is a respectable technique to determine the ability of the material to withstand tensile load prior to failure.
32
The relation between the tensile strength (TS) and the irradiation dosage for the NBR/Fe3O4 nanocomposites filled with different content of magnetite nanoparticles (5, 10, and 15 phr) is shown in Figure 9. The interfacial compatibility between the NBR matrix and Fe3O4 nanoparticles, which efficiently allowed stress transfer inside the matrix and resulted in an improved reinforcing effect, may be the cause of the load’s notable elevation of TS values up to 10 phr. It can be clearly observed that, the tensile strength start to decrease with further increase in Fe3O4 content (above 10 phr) in the composite. This can be owing to greater filler-filler contacts, which led to agglomerations when additional nanomagnetite was added. Segmental motion becomes limited as a result of the dilution influence’s weak connection between the rubber and filler.
33
The tensile strength increased with the increase of the irradiation dose and reached to optimum value at 50 kGy and behind that, the tensile strength started to decrease. All NBR/Fe3O4 nanocomposites had the same behavior where they reached the optimum tensile strength values at 50 kGy. The decrease in tensile strength happened at 100 kGy may be attributed to the predomination of degradation reaction at the high irradiation dose.
34
Variation of tensile strength with radiation dose of NBR and NBR/Fe3O4 nanocomposites.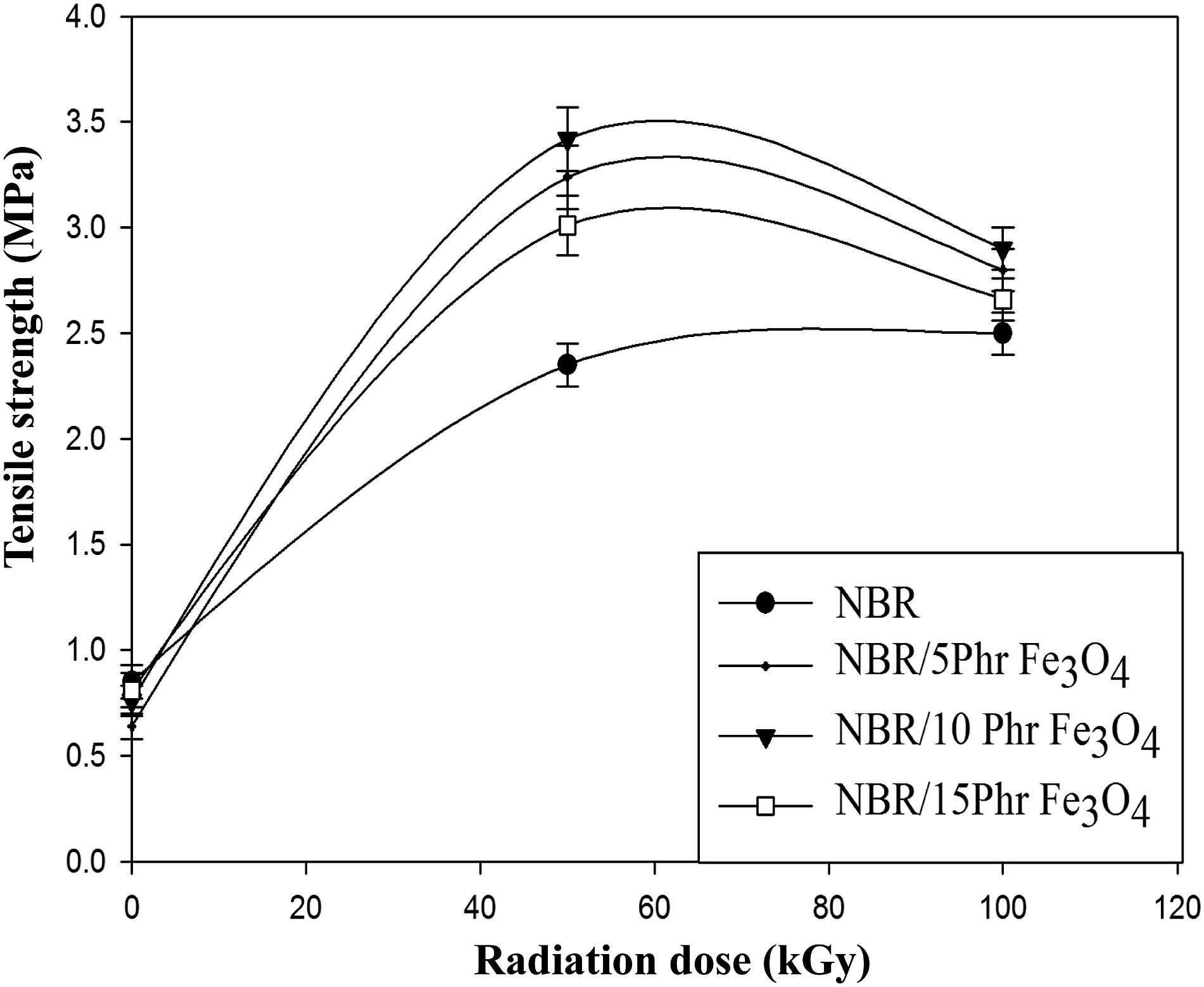
As the content of Fe3O4 nanoparticles increases the values of the elongation at break (%) decreases as clear in Figure 10. Strong interfacial bonding between NBR and the magnetite nanoparticles may limit segmental movement of the NBR matrix, which in turn prevents Fe3O4 nanoparticle pullouts from the matrix and reduces rubber mobility. This decrease is due to the agglomeration of filler particles or simply the result of physical contact between adjacent agglomerates. The agglomerate is a domain that can behave like a foreign body in the composites; this agglomerate reduces the movement of macromolecular chain and initiate failure under stress.
35
Additionally, because of radiation-induced crosslinking within the matrix, the elongation values decrease with increase the radiation dose up to 100 kGy. This can be contested by the fact that comparatively smaller doses function as plasticizers.
36
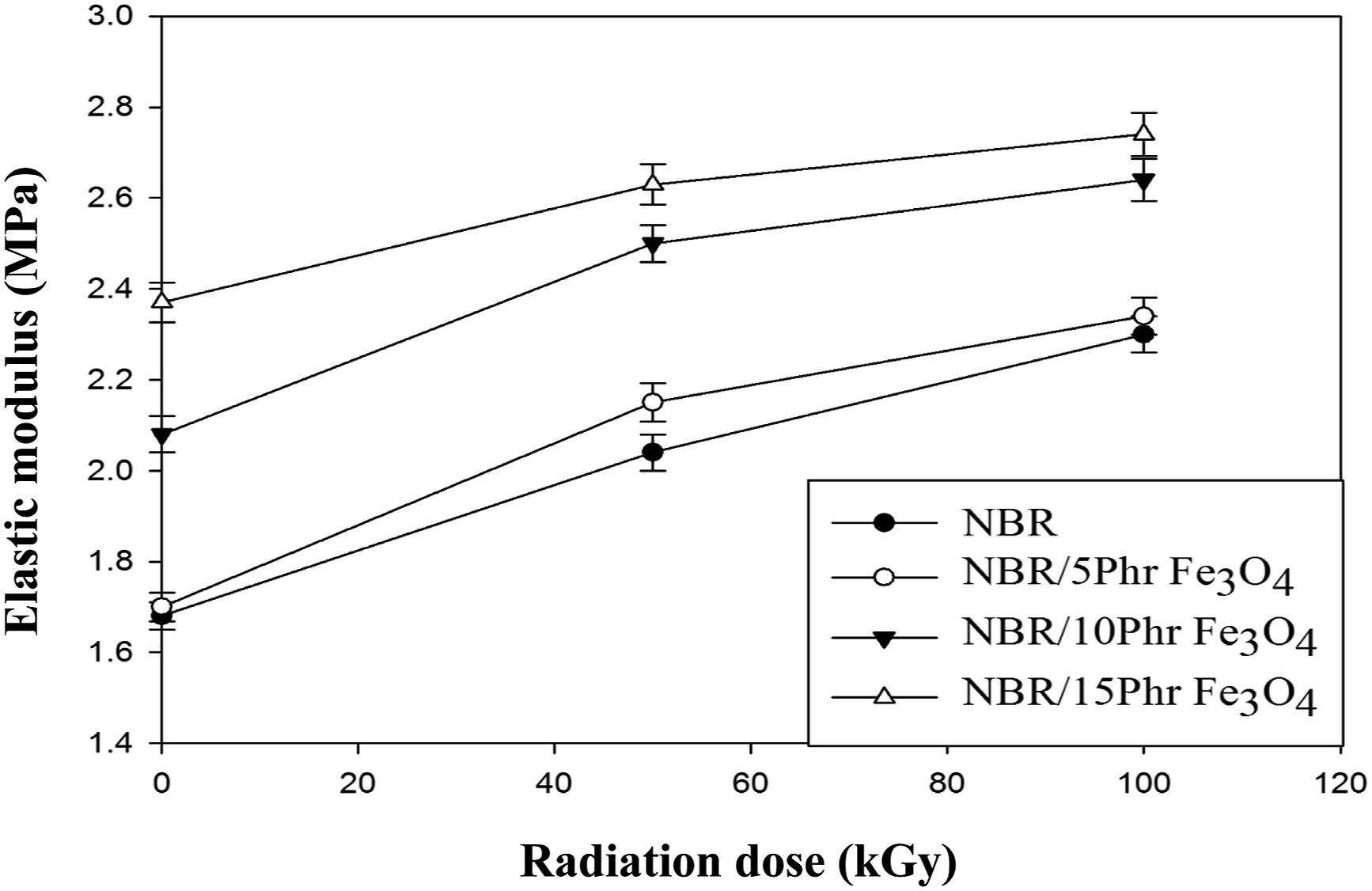
Figure 11 demonstrates the variation in the elastic modulus and the radiation dosage at 50 and 100 kGy of the prepared nanocomposites with varying Fe3O4 nanoparticle concentrations. Elastic materials generally tend to contract when force is applied.
37
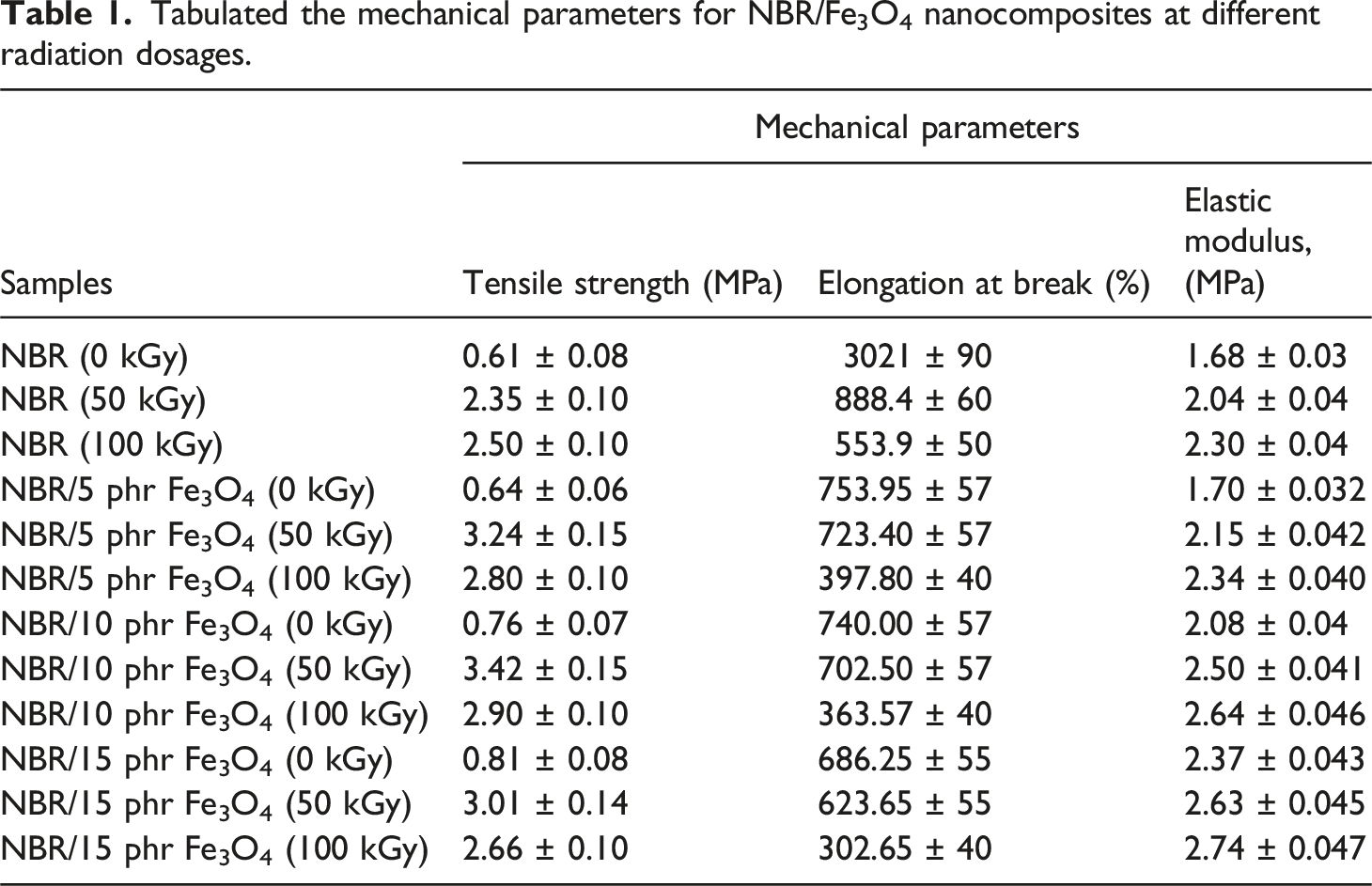
Because Fe3O4 nanoparticles interact and disperse sufficiently inside the rubber phase to make it stiffer, the modulus increased gradually as the loading of Fe3O4 nanoparticles increased. As the dose of γ radiation was increased to 100 kGy the elastic modulus raised continuously. Table 1 represents the results of all mechanical parameters for the prepared nanocomposites including the average and standard deviation. Variation of elongation at break (%) with radiation dose of NBR and NBR/Fe3O4 nanocomposites. Variation of elastic modulus with radiation dose of NBR and NBR/Fe3O4 nanocomposites. Tabulated the mechanical parameters for NBR/Fe3O4 nanocomposites at different radiation dosages.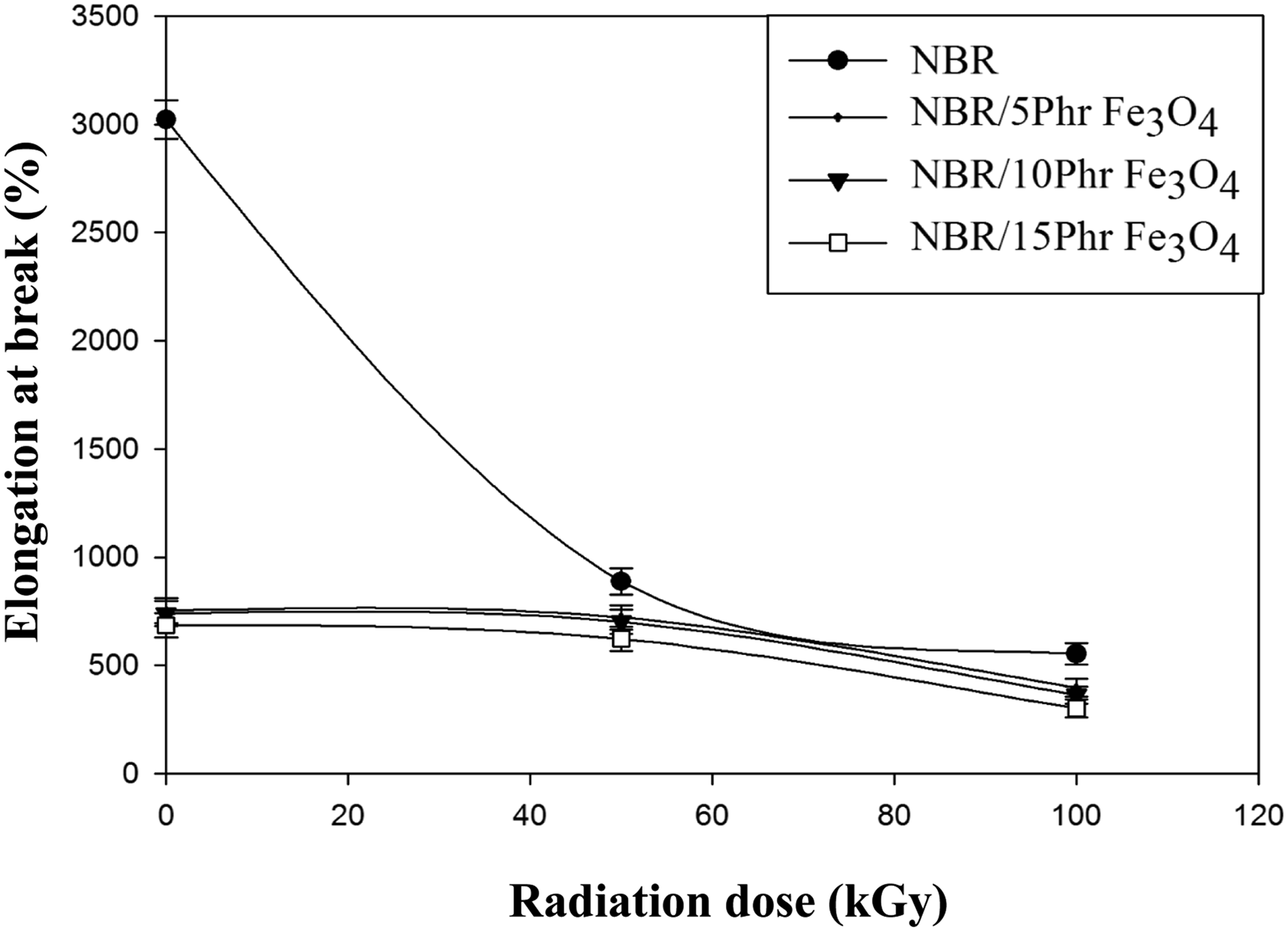
Electrical part
Frequency has a considerable influence on the dielectric properties, particularly at low frequencies. Because NBR contains polar C≡N groups, it possesses good electrical characteristics and a high degree of polarity.
38
At room temperature, Figure 12(a) depicts the relationship between the dielectric constant (ε`) and frequency ranging from 10 to 107 Hz for unirradiated pure NBR and these filled with different percentages of Fe3O4 nanoparticles (5, 10, and 15 phr) nanocomposite films. It was observed that ε′ decreases continuously as frequency increases. Due to Maxwell-Wagner interfacial polarization, ε′ is relatively large in the low-frequency region. This polarization can be seen in heterogeneous media with various phase compositions. As a result, charges accumulate up at the phase boundaries, the dipoles align in the field, and the dipoles reverse when the field changes direction, producing a high ε`.
39
the response time of orientation polarization is the reason of the decrease in ε′ at the high frequency range. The increased mobility of bounded charges caused by the increase the frequency hindered the dipole orientation, as a result the orientation polarizability dramatically reduces. Figure 12(b) shows the frequency dependence of the dielectric loss (ε``) for the NBR/Fe3O4 nanocomposites. ε`` illustrates electromagnetic energy dissipation that is emitted in the air as thermal energy. The relaxation in the interfacial polarization caused by the migration and trapping of charge carriers at the interfaces causes a maximum dielectric loss at about 15 Hz for all samples in the low-frequency regime.
40
The second observed peak at 106 Hz is due to the relaxation orientational polarization. Because the Fe3O4 nanoparticles bonded strongly with the NBR matrix, the molecular networks in the NBR restrict the mobility of molecular chains and are sluggish due to this segmental motion.
41
The orientation polarization’s contribution to the dielectric loss is minimal. From the figures, it is also notable that, ε′ and ε`` almost increase by increasing Fe3O4 nanoparticles content as compared to pure NBR. The presence of permanent electrical dipoles in the matrix, which result from charge pairs generated by the Fe ions (cations) and non-bridging oxygen (anions), may be responsible for the increase in electronic contribution to polarizability. Therefore, there will be greater numbers of these permanent dipoles when the Fe3O4 nanoparticles concentration rises, adding to the dipolar polarization Figure 12(c) shows the variation of the AC electrical conductivity, σAC, for unirradiated NBR nanocomposites loaded with different concentrations of Fe3O4 nanoparticles as a function of frequency at room temperature. σAC decreases with the increase of frequency for pure NBR and all nano-magnetite loadings. The presence of system charge carriers that absorb energy and move from one conductive site to another is what causes the apparent increase in σAC with frequency.
42
As Fe3O4 nanoparticles content increases a significant increase in σAC at any given frequency. This suggests that a rubbery network of continuous conductivity develops allowing more electrons to pass through the samples. (a) The dielectric constant (ε`), (b) The dielectric loss (ε``), and (c)T AC conductivity (σAC) as a function of frequency at room temperature for the pure NBR and these filled with different content of Fe3O4 (5, 10, and 15 phr) nanocomposites.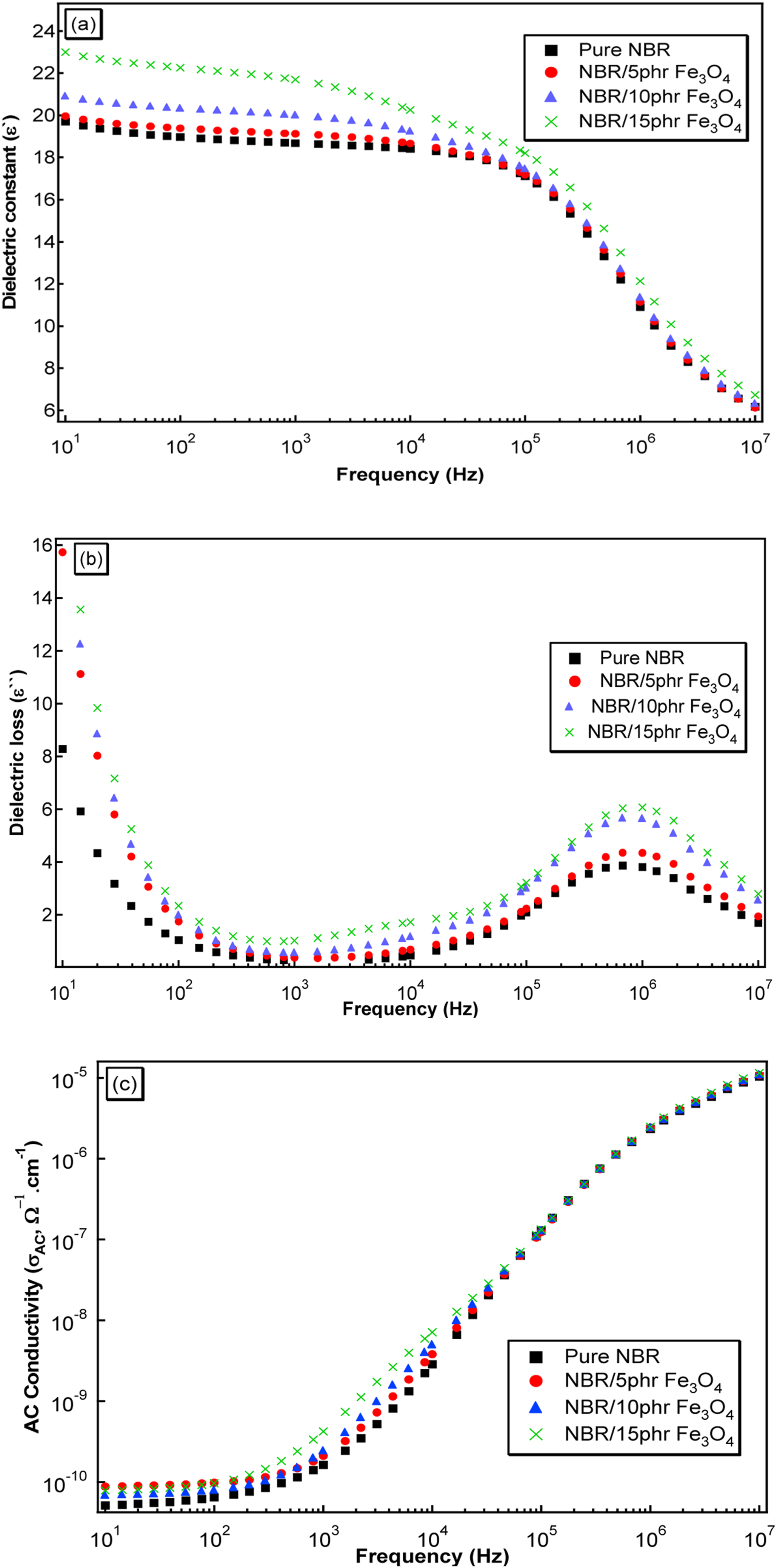
The effect of γ-radiation doses (50 and 100 kGy) on the electrical properties of the all prepared samples at 103 Hz is shown in Figure 13(a)–(c). Polymeric molecules are ionized and/or excited by γ-irradiation, which results in crosslinking or chain-scission.
43
The molecules’ or groups’ active energy to become polarized rises because of crosslinking. Thus, as samples are exposed to radiation the relaxation time and dielectric constant both decrease as shown in Figure 13(a). The rise in irradiation drops could be caused by an increase in free radicals, which could then react to reduce the number of dipoles in the nanocomposites.
44
In the upcoming generation of high-speed electrical devices, including electrical insulators and electrical lines, low-dielectric constant materials are anticipated to play tremendously significant roles.
45
Figure 13(b) shows the decrease of the dielectric loss, ε``, with increasing the γ-radiation dosage. Due to their direct relationship, the dielectric constant and the Ac conductivity show identical behavior following exposure to γ-radiation. The cross-linking that took place during irradiation causes the free charge carriers’ mobility to decline, which lowers the values of σAC as shown in Figure 13(c) (a) The dielectric constant (ε`), (b) The dielectric loss (ε``), and (c) The AC conductivity (σAC) as a function of Fe3O4 content at different γ-radiation doses (50, 100, and 150 kGy).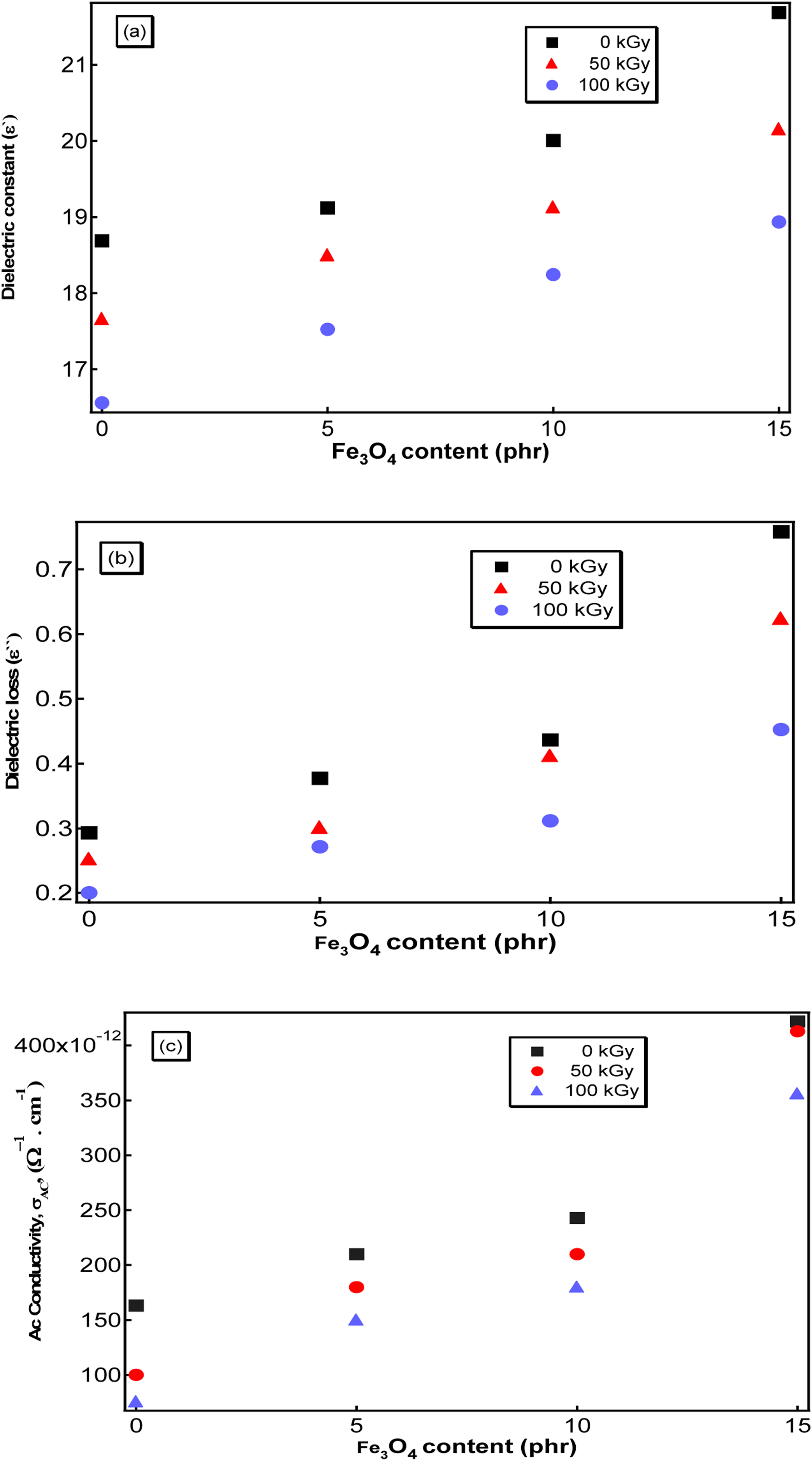
Electromagnetic interface shielding
A material’s effectiveness as an electromagnetic interface shielding material is reinforced by its capacity to attenuate the propagation of incident electromagnetic waves. 17 The attenuation of electromagnetic waves is performed by different mechanisms, namely reflection, absorption and even multiple reflections. 6 The power ratio between the incident and transmitted electromagnetic waves reflects the shielding effectiveness (SE) performance. The total shielding effectiveness (SETOT) is computed as the sum of the reflected shielding effectiveness (SER), the absorption shielding effectiveness (SEA), and the multiple shielding effectiveness (SEMR), which can be written as: SE TOT = SE R + SE A + SE MR , in which the third term is very small and can be neglected. 46 The S-parameter extracted from using the vector network analyzer is related to the reflection and transmission coefficients as follows: the transmission (T) equals the square of the absolute values of S12 or S21 and the reflection (R) equals the square of the absolute value of S11. In which, S11 represents the onward reflection coefficient; while S12 represents the inverse transmission coefficient then S21 represents the onward transmission coefficient.
The total shielding effectiveness can be calculated as the sum of the reflection and absorption values and related to the S-parameters as follows:
46

The electromagnetic shielding effectiveness of all prepared samples was measured in the X-band range from 8 GHz to 12 GHz. Figure 14 illustrates the shielding effectiveness, SE, for pure NBR, NBR/5 phr Fe3O4, NBR/10 phr Fe3O4, and NBR/15 phr Fe3O4 nanocomposites before irradiation and after irradiation with 50 and 100 kGy gamma-ray. The responses of all samples behave in the same pattern, as each one has different local maxima and minima. There are three global maxima around 9.4 GHz, 10.4 GHz, and 11.4 GHz (the largest value). The rapid increase in the curve after 11.9 GHz. This figure gives a significant sight for the increase of the shielding effectiveness that as the radiation dose increased the attenuation of the received signal increased which gives better shielding.
47
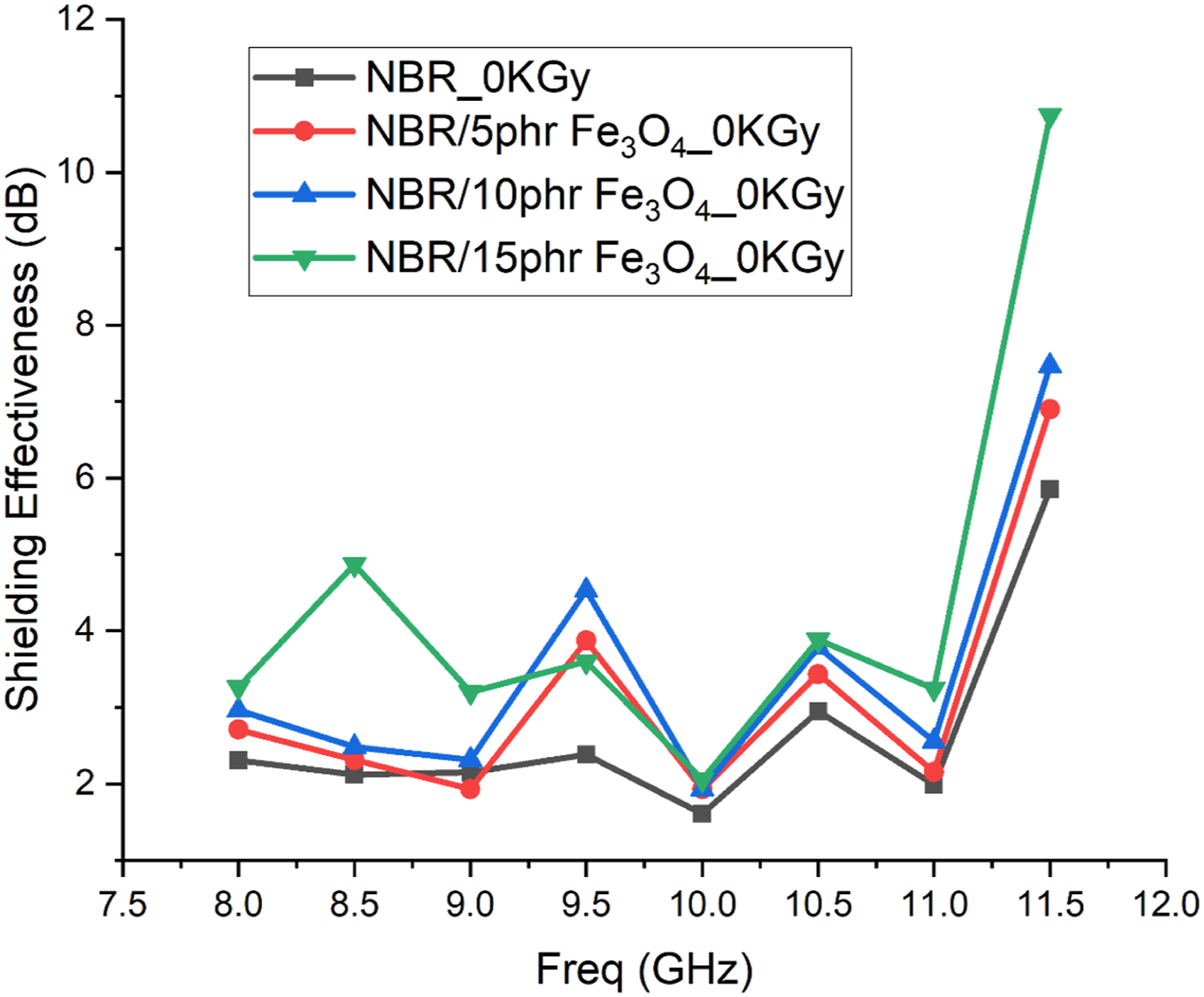
Obviously, SE slightly increased with radiation for all samples compared to the unirradiated samples. Meanwhile, Figure 15 shows the effect of magnetite nanoparticles concentration on the shielding effectiveness for the prepared samples before radiation. Regarding the curves in this figure, it is clear that the SE increased significantly with the increase in magnetite nanoparticles concentration before irradiation. A same response was achieved for the samples after irradiation. Pure NBR is the control sample, which shows a weak response. Whereas, NBR/15 phr Fe3O4 nanocomposite shows a good response for the shielding effectiveness. Represents the shielding effectiveness (dB) versus the frequency (GHz) for the pure NBR and NBR/Fe3O4 unirradiated nanocomposites. Represents the shielding effectiveness (dB) versus the frequency (GHz) for the unirradiated pure NBR and NBR/Fe3O4 filled with different concentration (5, 10, and 15 phr) nanocomposites.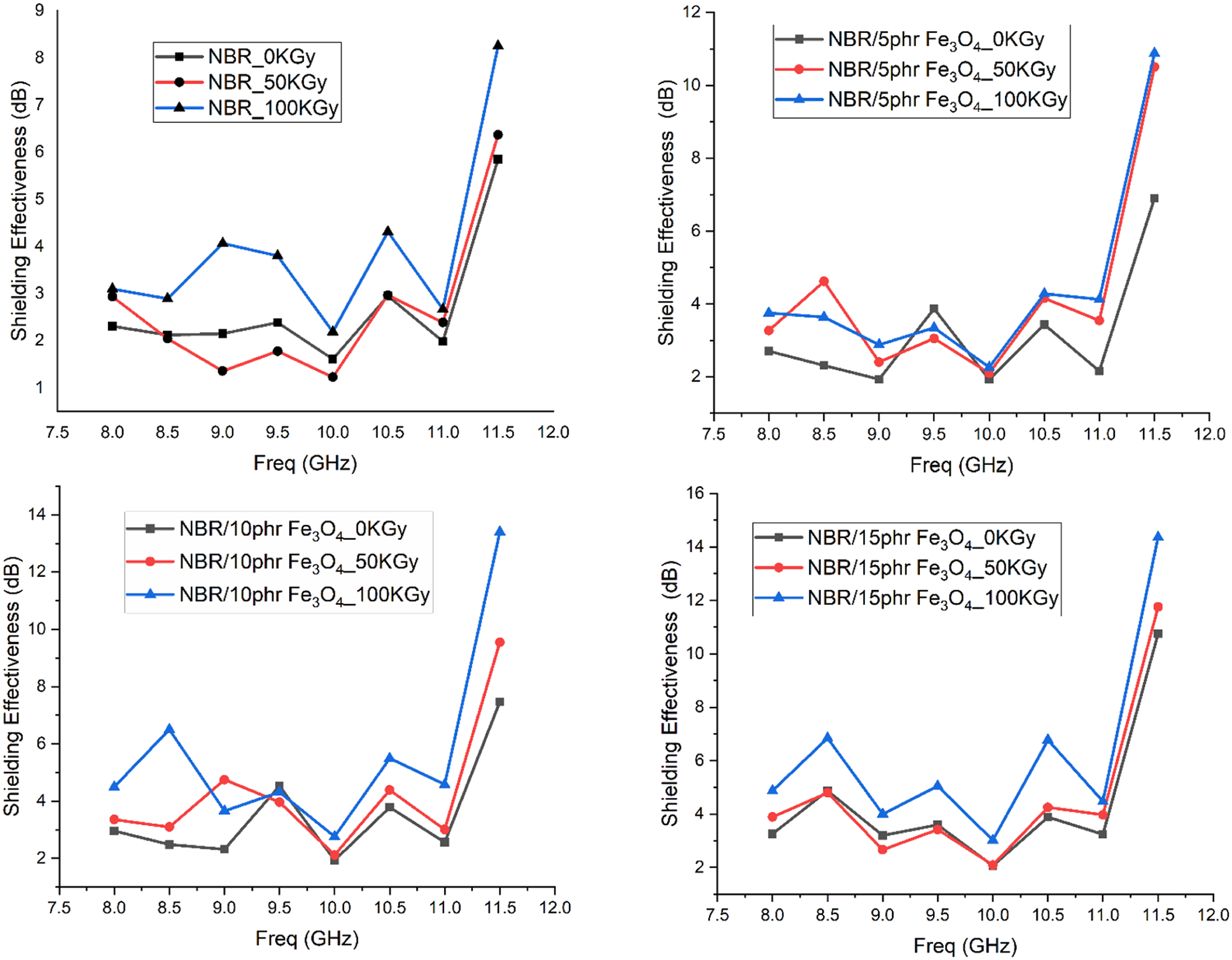
Conclusion
Various techniques including XRD, FTIR, SEM and EDX, were effectively used to characterize the prepared nanomagnetite. Fe3O4 nanoparticles allowed for significant dispersion within the matrix. Due to intermolecular radiation crosslinking within the composite matrix, the mechanical characteristics of the formed nanocomposites improved overall with increasing the loading of Fe3O4 nanoparticles. The existence of permanent electrical dipoles in the matrix, which emerge from charge pairs created by the Fe ions (cations) and non-bridging oxygen (anions), may be accountable for the increase in electronic contribution to polarizability. The addition of Fe3O4 nanoparticles increased the NBR matrix’s electrical conductivity. As γ-radiation doses increases, the dielectric parameters decrease. Because of the crosslinking that happened during radiation, the free charge carriers’ mobility is reduced, which lowers the values of σAC. It can be summarized, that elevation of Fe3O4 nanoparticles performs well when combined with inexpensive, flexible, and corrosion-resistant rubber. The SE enhanced as the concentration of Fe3O4 nanoparticles increased inside NBR matrix. These nanocomposites were used more effectively as an electromagnetic interface shielding material when the dose of gamma-ray irradiation were increased.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.