Abstract
Polymer based materials capable of exhibiting both elasticity and electrical conductivity have been widely used in electronics, electrotechnics, and the automotive sector However, conventional conductive elastomers show limitations for applications which require stretchability, conformability in complex shapes and for dynamic movements. These limitations are caused by the incorporation of rigid, non-elastic electrically conductive fillers. The emergence of expanded graphite provided a promising solution to replace the non-stretched conductive fillers with stretched fillers. Expanded graphite being electrically conductive and mechanical flexibility serves as an excellent filler in the production of highly flexible, stretchable, and electrically conductive elastomers. So, considerable interest of researchers has been enthusiastic to expanded graphite-based elastomers, named as new-generation materials, because of their extraordinary stretchability while maintaining electrical and mechanical properties. The first half of the review focuses on a systematic evolution from carbon-based fillers to graphitic materials, ultimately leading to expanded graphite. It highlights the major intercalation and exfoliation strategies used to convert expandable natural graphite into EG, including modern approaches employing advanced oxidizing systems and blowing agents. Emphasis is placed on recent and innovative methodologies for producing EG with tailored properties suitable for stretchable, electrically conductive elastomeric architectures. The second half of study, creates a bridge between expanded graphite (stretchable electrically conductive materials) and their applications as a filler in flexible matrix (elastomers). As we know that expanded graphite has ability to enhance electrical conductivity, thermal stability, and mechanical resilience in flexible elastomers, which makes it quite a fitting material for different applications such as sensors, energy storage devices, and electromagnetic interference shielding. A comprehensive literature survey compiles the most recent studies exploring the integration of expanded graphite into stretchable elastomer systems.
Keywords
Introduction
Expandable graphite (EGp) is a type of graphite intercalation compound (GIC) that is fabricated by physical or chemical method. 1 Some other substances are inserted between layers of graphite. Under the traditional heat or microwave irradiation 2 the intercalated material is instantly decomposed and gasified, generating a force to push away the graphite layers, thus expanding the spacing between the graphite layers, forming a loose and porous worm-like material, which is expanded graphite (EG). It was first discovered in 1968 by the American company Carburet and has since been further developed in China over the last five decades. 3 EG exhibits various extraordinary graphite properties including conductivity, chemical resistance, as well as thermal stability. Such qualities enhance the suitability of EG applications in environmental engineering along with material science. To produce EG, natural graphite is typically treated with strong acids like phosphoric acid (H3PO4), sulfuric acid (H2SO4), or nitric acid (HNO3), which intercalate into the graphite layers. 4 Among these, concentrated sulfuric acid (∼98%) is especially effective due to its strong oxidizing power.5
The synthesis of EG can be classified into two types: physical and chemical methods. Physical techniques include methods such as microwave irradiation,6,7 ultrasonic treatment,8,9 and thermal expansion at varying temperatures. 10 These methods physically alter the graphite structure without significant chemical modifications. Chemical methods, on the other hand, involve more reactive processes, such as Electrochemical Intercalation, 8 Acid Intercalation and Thermal Expansion, 11 Liquid Phase Exfoliation 12 Chemical Vapor Deposition, 13 Chemical Oxidation (Hummers’ Method). 14 Each synthesis route has its own advantages and drawbacks, and the choice depends on the intended application and performance requirements of the final material. Among the various heating techniques, microwave radiation is especially notable due to its rapid, uniform heating and short processing time. It operates by inducing ionic movement under an oscillating electric field, leading to collisions that generate internal heat. 15 Recently, the research has focused on graphene nanoplatelets as well as nano-expanded graphite (nEG) based polymeric nanocomposite formation. 16 This interest surged after the Nobel Prize-winning discovery of freestanding graphene layers by Novoselov and Geim in 2010.
During the last few years, one major application of EG is in flexible conductive films and stretchable elastomeric composites. Both flexible graphene as well as nano-graphite based films consist of various applications such as fluid impermeability, resistance to high temperatures and low thermal expansion, high electrical and thermal conductivity, chemical resistance along with resilience. 17 In fact, the demand for polymer materials that are both elastic as well as electrically conductive at the same time has been constantly increased in the hi-tech and novel applications including flexible electronics, 18 biomedical fields,19,20 soft robotics, 21 smart sensors 22 and actuators, in automotive and in remote controllable micro/nano robots etc. 23 However, less elastic electrically conductive elastomers are limited in most of the applications where high stretch ability, conformability in complex shapes and dynamic movements are needed. 24 The limitations in stretchability of elastomers are caused by the incorporation of rigid, non-elastic electrically conductive fillers. 24
The discovery of EG provided a promising solution to replace the non-stretched conductive fillers with stretched fillers (EG). EG being electrically conductive and mechanically flexible, serves as an excellent filler in the production of highly flexible, stretchable, and electrically conductive elastomers. 25
Hence, there was an immense need to compile an intensive review that merges all available methodologies for the preparation of EG and its subsequent utilization as a reinforcing material, particularly in stretchable and flexible elastomers. Initially, a concise overview was provided on carbon and its derivative fillers, including graphite, graphene, and activated carbons, followed by a detailed discussion on the progression from traditional carbon-based fillers to EG. Most novel and recent methodologies to prepare the EG with unique properties are required for developing electrically conductive, stretchable elastomeric structures. The second half of study, creates a bridge between expanded graphite (stretchable electrically conductive materials) and their applications as a filler in flexible matrix (elastomers). A comprehensive literature review was conducted to compile all recent research studies highlighting the use of EG in stretchable elastomers. 25
Evolution from carbon-based fillers to expanded graphite (EG)
Carbon
Carbon is the second most abundant element in the biosphere and plays a central role in a wide range of scientific, industrial, nanotechnological, and biomedical applications. In recent years, significant research efforts have been directed toward the efficient synthesis of carbon-based materials and the exploration of their functional uses. The unique tetravalent nature of carbon, together with its ability to adopt different hybridization states, gives rise to an exceptional diversity of structural forms. As a result, carbon exists in multiple allotropes, including graphite, diamond, graphene, and fullerenes. Variations in orbital overlap and bonding configurations among these allotropes lead to distinct physical and chemical characteristics, enabling their use in diverse technological applications. 26
Although pure carbon allotropes have valuable properties such as high thermal conductivity, chemical stability, they still lack tunability, processability, and surface reactivity needed for modern applications such as in polymer reinforcement, electromagnetic shielding and energy storage devices. 27
Graphite
There are two types of graphite, broadly classified as natural or synthetic. Additional categories include carbon fibres, graphite blocks, graphite powders, graphite electrodes, and different types of synthetic graphite. Graphite is widely used in mining, battery manufacturing, lubrication, and numerous other sectors.
28
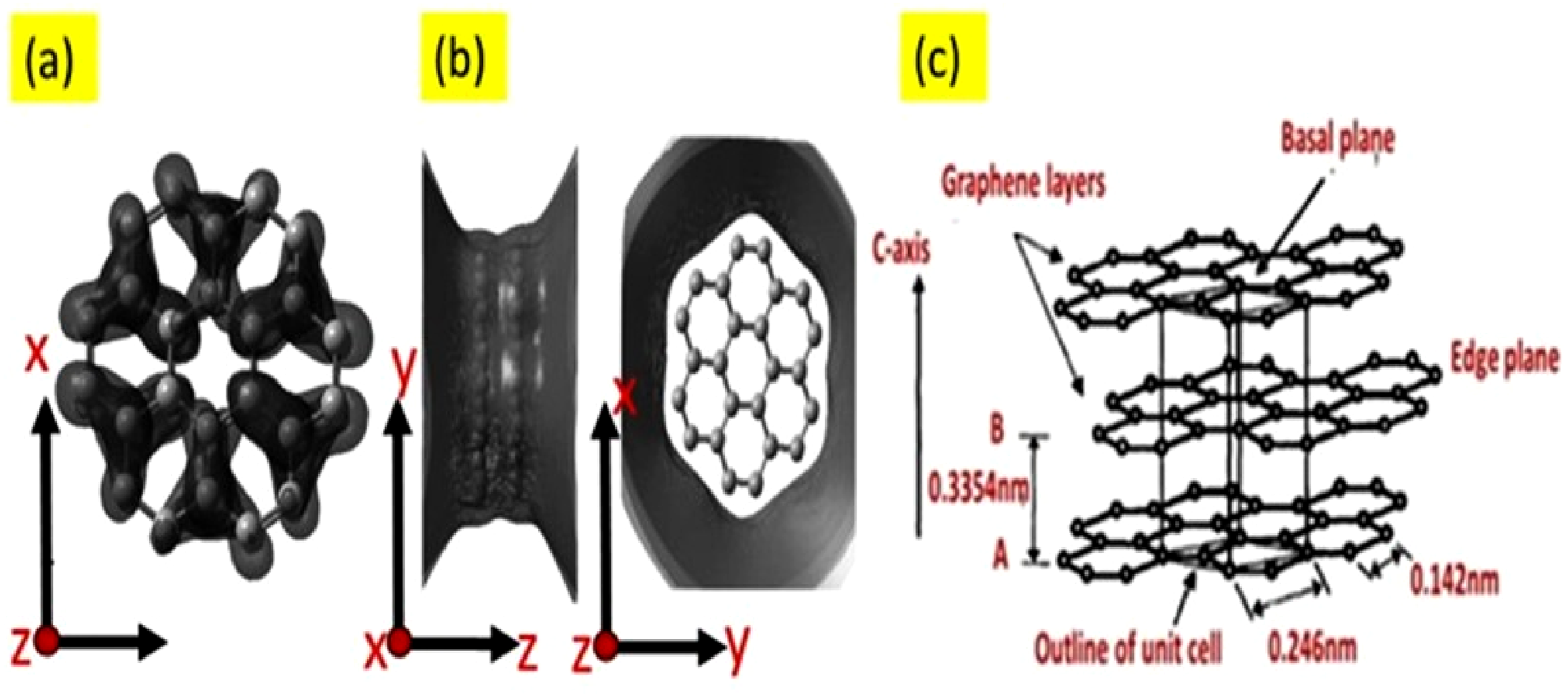
The unsaturated p orbitals in graphite promote sp2 hybridization of carbon atoms, which explains why density functional theory (DFT) simulations of its molecular orbital diagram display a trigonal planar structure (Figure 1(a)). The simulated electrostatic potential field of the graphite structure perpendicular to the graphene planes is presented in Figure 1(b). Due to the short-range nature of van der Waals interactions between these planes, graphite cannot form strong interactions with other elements or compounds along the c-axis without disrupting its multilayer structure.
28
The density of hexagonal graphite is about 2250 kg/m2. The space present between the layers is about 0.335 nm while 0.142 nm is the bond length between both C-C atoms (Figure 1(c)). (a) Molecular orbital for graphite (sp2), (b) graphite electrostatic potential,
28
(c) crystal structure of graphite showing ABAB stacking sequence and unit Reprint with permission from [Ayesha Kausar], [Poly (methyl methacrylate) nanocomposite reinforced with graphene, graphene oxide, and graphite: a review], published by [Taylor & Francis], [2019].
29
Graphene
Graphene is a two-dimensional layer of sp2-hybridized carbon atoms arranged in a hexagonal close-packed crystal lattice, forming nanosheets with exceptional properties. As a result of its exceptional qualities, such as ballistic transport, high thermal conductivity, high current density, optical conveyance, as well as super hydrophobicity at the nanoscale, graphene is employed as a functional material. 30
Despite these outstanding features, producing and utilizing high-quality graphene remains challenging due to issues such as particle aggregation, low solubility, and complex manufacturing processes. Graphene’s high electron mobility (25 m2 V−1 s−1) makes it a highly electrically conductive material. Common fabrication methods include graphene include ball milling, chemical exfoliation, thermal chemical vapour deposition (CVD), chemical synthesis, sonification, microwave synthesis, and the graphite oxide production in which a graphite precursor is oxidised in protonating fluids and then chemically reduced. 8
These properties have enabled the applications of graphene in many fields. For example, graphene-based breath sensors have been developed to detect volatile biomarkers such as NH3 for renal failure and CH3COCH3 for diabetes, enabling non-invasive disease diagnosis. 31
Expandable graphite (a precursor)
Expandable graphite (precursor for expanded graphite formation), a category related to the GIC (graphite intercalation compound) which are formed through intercalation process, has applications in a number of industries, including military applications, high-power batteries, fire retardants, oil-absorbing materials, airproof materials, electrodes, and more. 32 When heated beyond a specific threshold, EGp rapidly exfoliates into a worm-like, highly expanded structure, a process driven by the vaporization of intercalants as described by Chung. 33 Both EGp and its exfoliated form exhibit broad utility. Such a procedure originates from the intercalant’s vaporisation. Both expanded graphite as well as its exfoliated form have a broad area of applications. Expandable graphite (precursor) may function as an intumescent flame retardant for particular polymers, since polyethylene as well as polyurethane foams are the main substrates for EG. 34 EGp possesses in-plane electrical conductivity that is quite identical with natural flake graphite. Therefore, in addition to the effects previously mentioned, natural graphite as well as EGp graphite have the potential for the purpose of enhancing fire retardancy properties and adding electrical as well as thermal conductivity to their polymer substrate. 35
Expanded graphite
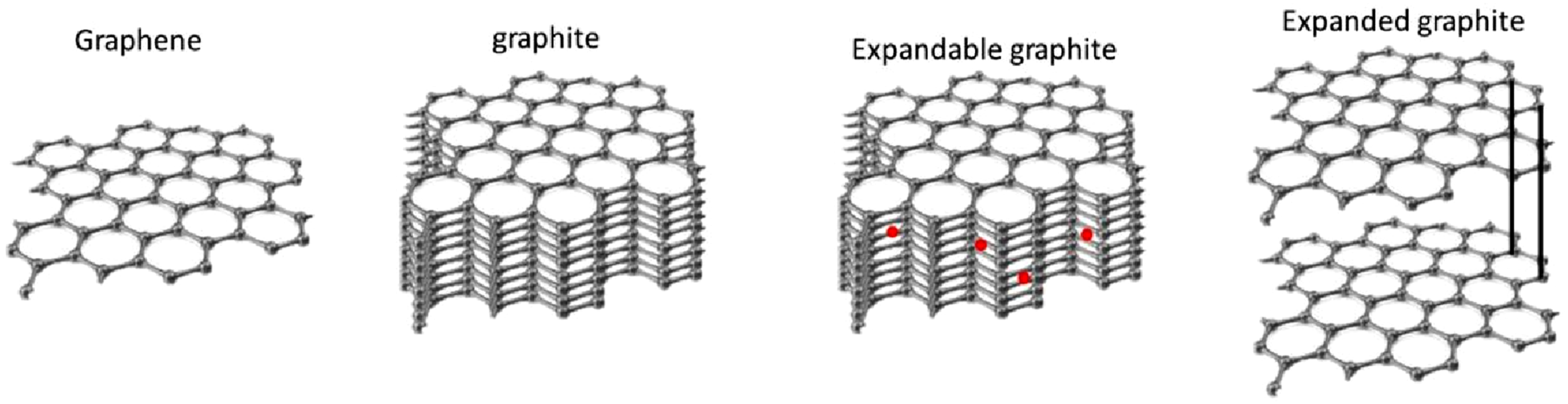
It is similar to the intermediate stage of graphene. The manufacturing of expanded graphite requires expandable graphite (EGp). In final products that are often not fully exfoliated, stacked graphitic layers may still be present in a section of the structure. The intercalated graphite expands as a result of heat treatment.
36
When EGp forms, the space between the layers expands from about 0.335 nm to roughly 0.8 nm, and EG’s interlayer spacing is hundreds of times longer along the c-axis than the predecessor. Conventional manufacturing may leave trace impurities, especially sulfur from acid intercalants, which can be undesirable for many applications.
37
Many publications describe EG as a worm-like structure (see Figure 2).
38
It is well known that the conjugated chemical bonding structure breaks when a graphene sheet is functionalised, reducing its conductive qualities. (a) SEM analysis of graphite (b) Expanded graphite, (c) expanded graphite after 6 min ball milling. Reprint with permission from [Handayani M], [The Development of Graphene/Silica Hybrid Composites: A Review for Their Applications and Challenges], published by [MDPI], [2021].
39
Reprint with permission from [Xueqing Yue], [In-plane defects produced by ball-milling of expanded graphite], published by [ELSEVIER], [2010].
38

The basic physical properties of EG are modelled by using classical concepts of disordered matter physics. EG is less expensive than graphene, has a low density (10–40 kg/m3), and anisotropic electrical and thermal characteristics (more thermally insulating than graphite). Excellent thermal shock suppression, chemical resistance, low coefficient of surface friction, negative Poisson ratio (auxetic material), and low permeability are all attributes of compressed EG.
40
It can function at temperatures between 200 and +3500°C. Compacted EG has large well-connected pores, enabling good accessibility to several chemical reagents. The physical structures of different kinds of graphite is presented in Figure 3.
41
Illustration of the physical structure of (a) graphene 1 D, (b) Structure of graphite stacking, (c) Structure of expandable graphite (d) Structure of expanded graphite. Reprint with permission from [Jindu], [Green Preparation of Expandable Graphite and Its Application in Flame-Resistance Polymer Elastomer], published by [ACS], [2017].
41
Preparation of expandable graphite as a precursor of expanded graphite
As discussed in the prior segment “Evolution from carbon-based fillers to EG,” natural graphite goes through an intermediate stage called expandable graphite before EG is formed. Expandable agents, such as gases or acids, intercalated after the generated expandable graphite precursor was subjected to additional processes and treatments, such as heat or chemical operations. This process enables the graphite to expand when heated.
Chemical intercalation process
The chemical intercalation process is well known for being straightforward and providing both porosity and a high rate of expansion. Expandable graphite is a naturally found filler material that consists of a flake-shaped morphology. This method initially treats graphite with a range of intercalation chemicals, like nitric acid, and sulphuric acid, acetic acid usually to impart expansion behaviour. Normally, the expansion brought on due to higher temperatures causes their volume to grow by a factor of 300. This characteristic of expandable graphite makes it possible for it to function as a product with good flame resistance. Expandable graphite (precursor) is extensively utilised as a barrier filler, lubricant, conductive additive, radiation shielding material, electromagnetic reagent, insulation product as well as a pigment, in addition to its fire retardancy uses. 42
Secondary intercalating reaction and an ultrasound effect
After the initial intercalation procedure, a secondary intercalation step is performed to ensure better expansion in the structure of expandable graphite. This includes an intercalating reaction that follows as well as an ultrasonic effect. Before adding sulfamic acid (NH2SO3H) and potassium permanganate (KMnO)4 to deionised water and stirring for 5 minutes, 300 μm natural graphite (NG) particles were vacuumed and dried for the first intercalating reaction. The second intercalating method involved mixing a mixture with sodium nitrate (NaNO3) and perchloric acid (HClO4) and stirring it for 60 min. Dehydration, grinding, washing, and filtering were all applied to the mixture. The resulting EG particles had a first expandable temperature of about 150°C as well as an average size of about 25 μm, and also zeta potential of −20.5 mV at a pH of 6.5. 8
Green synthesis process to prepare the expandable graphite precursor
In order to minimize or completely get rid of sulphur content of EG, a number of processes have been offered yesteryear. The majority of these methods unveil new hazardous pollutants and take on HClO4,43,44 HCl (hydrochloric acid), 45 HNO3 (nitric acid), 46 H3BO3 (boric acid), 47 etc. So, the preparation of expandable precursor through the green synthesis method has been affirmed. This method has proven significant as it operates without chemical oxidizing agents but only using dilute acid concentrations. 41
In the testing process, firstly, 3 g of natural flake graphite (NFG) are peroxidised with 0.8 mL of hydrogen peroxide (H2O2) at room temperature for 60 min, agitated ultrasonically and stirred mechanically concurrently.
48
In accordance with Figure 4, 1 g if EG-1 is exposed to second peroxidation procedure (similar to the earlier peroxidation procedure) in order to get expandable graphite bearing low Sulphur content. Thereafter, concentrated phosphoric acid (3 mL) is added slowly and agitated for diverse temperatures (60°C) and intervals (60 min). The product obtained is then rinsed along with deionized water which is then dried to obtain green expandable graphite (EG-2).
48
For the preparation of EGs, several other green methods could also be used such as heat treatment,
49
vitamins,
50
glucose,
51
polyphenols,
52
ginseng,
53
proton pumps,
54
hydrogen-rich water,
55
flash light,
56
pulsed laser irradiation,
57
photocatalytic process.
58
Illustrating the process of preparation of the expandable graphite precursor with a low sulphur content. Reprint with permission from [Morsy, Ashraf], [Utilizing a blend of expandable graphite and calcium/zinc stearate as a heat stabilizer environmentally friendly for polyvinyl chloride], published by [Wiley Online Library], [2024].
48

Synthesis of precursor expandable graphite though chemical oxidation
Hummer’s method, also known as ‘chemical oxidation method’ is simple and highly efficient method to obtain intended product with high product yield. However, it can be environmentally toxic as it involves some of the chemical oxidizing agents.
At first, under the influence of mechanical stirring, NFG (3 g) was peroxidized by H2O2 (0.4−0.8 mL) for 60 min along with ultrasonication at normal room temperature in this process. Then this was added with 8-12 mL of H2SO4 gradually while being stirred at several temperatures (20−60°C) as well as times (60−140 min). The final mixture was then filtered, washed again and again using deionized water, after that it was dried at about 65°C for 12 h to get first intercalation compounds (EG-1). 1 g of EG-1 was again peroxidized (similar to earlier peroxidization procedure) termed as the second peroxidation treatment 41 Subsequently, concentrated H3PO4 (3 mL) was added gradually and stirred at various temperatures (20−60°C) and time (20−180 min). The final product was rinsed with deionized water and then dried, successfully achieving expandable graphite (EG-2) with low sulphur concentration. 41
The surface chemistry of expanded graphite can be further tailored using multicomponent reactions similar to the Ugi four-component assembly process (Ugi-4CAP), which allows covalent Mult functionalization of graphene oxide in aqueous media. It introduces a novel covalent functionalization method for carboxylated graphene oxide using the Ugi four-component assembly process (Ugi 4-CAP), where amine, aldehyde, isocyanide, and acid react in a one-pot procedure to produce hydrophobic, hydrophilic, or amphiphilic multifunctional graphene composites. The immobilization and biocatalytic activity of Bacillus thermocatenulatus lipase on the functionalized graphene demonstrated the effectiveness of this approach. Such synthetic strategies could enable the introduction of hydrophilic or hydrophobic groups onto expanded graphite surfaces, improving its dispersion, compatibility, and interaction with elastomer matrices. 59
Preparation of low-temperature expandable graphite
In order to develop sustainable expandable graphite precursor by means of some significant environment friendly approaches for the further synthesis of EG are needed. A higher production yield is offered by eco-friendly low temperature synthesis method of expandable graphite precursor. 48 Two stages are involved in this process i.e. peroxidation and deep oxidation and intercalation. During the first stage, peroxidation of natural flake graphite is done in H2O2 solution, then deep oxidation and intercalation. The next second stage involves KMnO4 and HClO4 in a water bath which is then followed by acetic acid normal water washing. The final product is dried for 4 h at 20-60°C which result in low-temperature expandable graphite. 1
Mechanism of expansion (expandable graphite to expanded graphite)
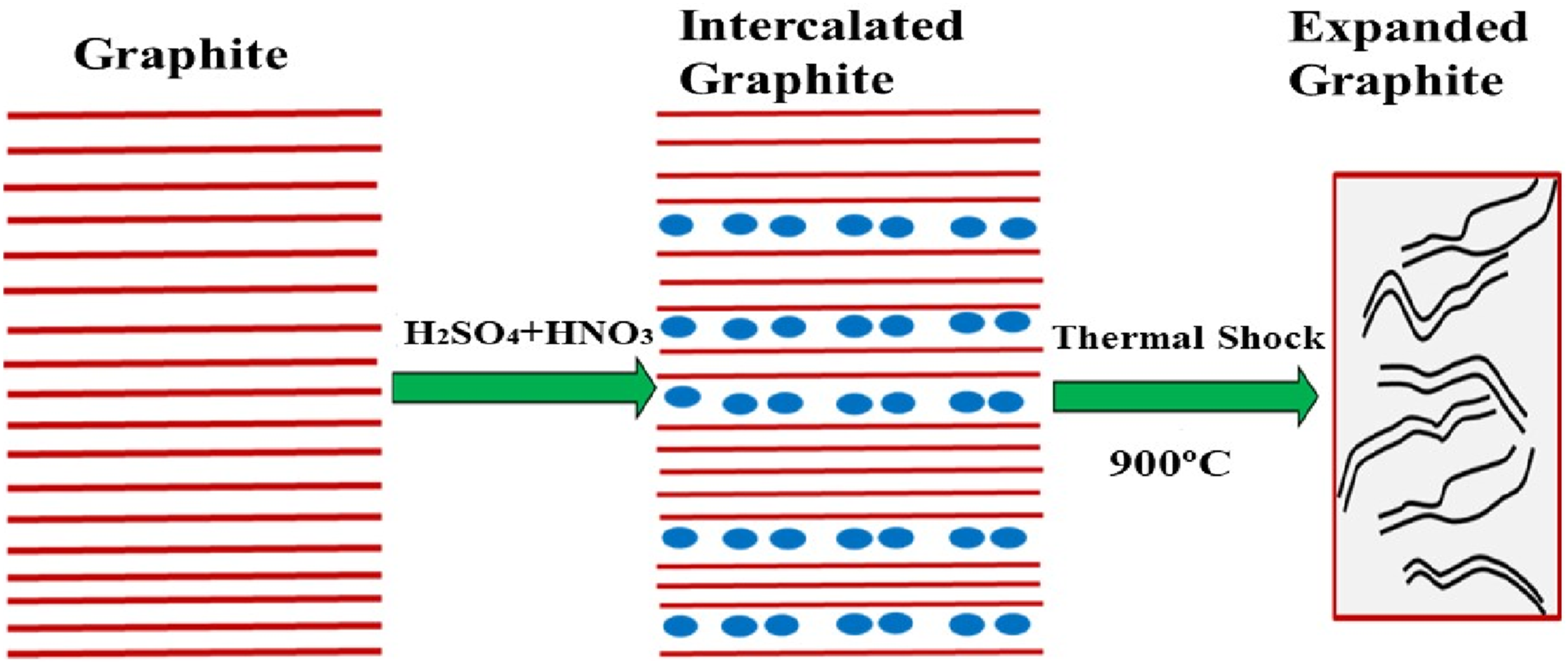
The conversion of expandable graphite (EGp) to expanded graphite (EG) occurs via rapid thermal expansion of the intercalated precursor. EGp is produced by inserting intercalants (e.g., acids, oxidizers) between the graphene layers of natural graphite.
60
Upon heating, the intercalants vaporize, generating a sudden ∼100× increase in their volume. This high internal pressure forces the adjacent graphite layers apart, producing the characteristic worm-like expanded structure. Figure 6 displays the scheme for the expansion agent in its metastable residence present between the adjacent graphene layers. The dimensions of the interlayer are about 3.355 Å (6.7 Å/2). After exfoliation, the interlayer spacing value becomes higher than 3.35 Å due to the protection process.61,62 In pristine graphite, the interlayer spacing is up to 3.35 Å; after expansion, the spacing increases significantly due to the separation of layers and creation of porous channels. This process is illustrated schematically in Figure 5. Schematic of expandable graphite expansion. Reprint with permission from [Seredych, Mykola], [Adsorption of bovine serum albumin on carbon-based materials], published by [MDPI], [2018].
63
Comparison of oxidizing and acidic intercalants in the preparation of expanded graphite and their effect on EG.
Moreover, another factor is the intercalation depth and interlayer interactions. Which describe the different stages of intercalations by observing; how deeply the intercalants penetrate to create the intercalated layers between numbers of graphene. During this step the expansion starts due to gas generation (works as intercalants) and decreases the van der Waals forces between layers and increasing interlayer spacing.42,69,70 In-fact the heating converts intercalants into gas forms such as SO3, CO2, O2, H2O vapor, etc. which in turn generates the interlayer pressure that causes expansion. More heating cause to create large pores and enhances sorption property of resultant materials. The techniques of XRD and Raman spectroscopy have been successfully used to observe the dynamic change in interlayer spacing during expansion.70,71
Synthesis techniques of expanded graphite
Expanded graphite (EG) is produced by intercalating natural graphite with oxidizing agents to form expandable graphite (EGp), followed by rapid expansion. The first observation of EG was reported by Schafhautl in 1841 during the treatment of graphite with sulfuric acid. 72 After that many researchers started their study on the synthesis and applications of EG. Nowadays, the method of preparing EG can be divided into two categories: chemical and physical approaches. 73 The rapid heating due to thermal expansion will cause the emission of gases, forming worm-shaped porous structure with high surface area and low density. 64
Ultrasonication usually exfoliate the graphite layers to form a thin-sheet like structure with improved dispersibility. Microwave heating offers faster, more uniform expansion and can better preserve pore structure and conductivity. Electrical heating applies direct current to generate internal heat, controlling the degree of exfoliation and particle morphology. 64 Mechanical milling is a top-down approach that fractures graphite into expanded particles with increased reactivity and surface roughness. Finally, explosive combustion and blending harness rapid gas release under high pressure to create ultra-porous EG with enhanced adsorption properties. 74
Physical methods to prepare the expanded graphite
Expanded graphite is manufactured using various physical approaches. The usually used physical methods involve low/high-temperature, 6 microwave method, 6 ultrasonic methods, 9 thermal expansion method, 10 Electrical heating, 42 and several other techniques. Among the aforementioned approaches, high/low-temperature expansion is quite often utilized. However, it may have some limitations which include inert gas protection, high equipment requirements as well as excessive energy consumption. The microwave approach is quite fast and effective for the synthesis of EG, nevertheless, it may require higher energy and attaining additional scale-up yield is also quite difficult which eventually restricts its industrial applications. Several physical approaches involve.
Thermal expansion process
In this method, graphite is initially intercalated along with some oxidizing agents such as sulfuric acid, nitric acid to GICs. Then this intercalated graphite is continuously heated at a very high temperature of about (800–1000°C).
6
The rapid intercalant evaporation produces gas pressure which leads to graphite layer expansion. Nowadays low/high-temperature thermal expansion technique is typically preferred.
10
But this approach also has some limitations such as extreme heat energy loss, need for inert gas protection as well as specialized equipment to endure high temperature and high pressure.
10
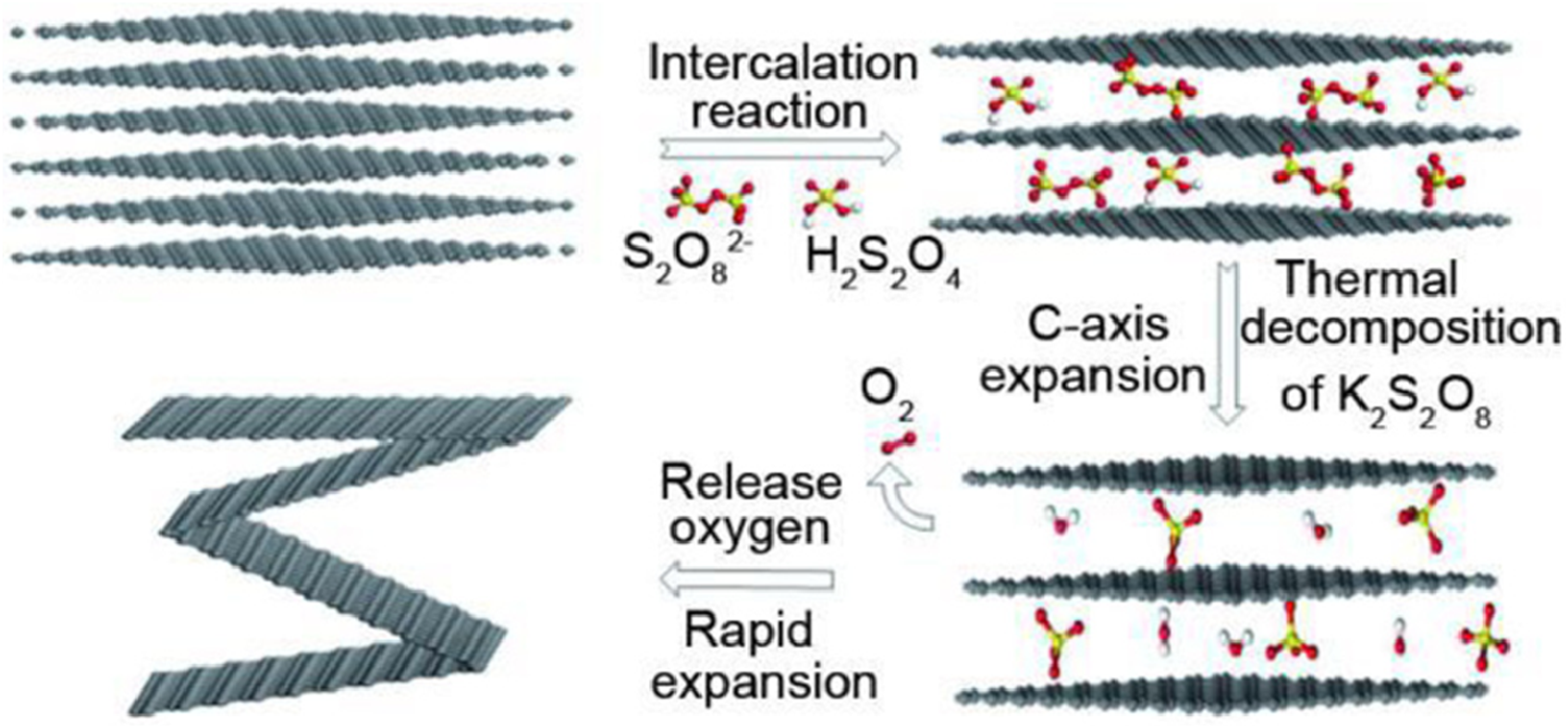
Bo Hou et al. prepared EG by exfoliating flake graphite using K2S2O8 under concentrated sulfuric acid, which can be used as both an efficient expander as well an oxidant. As Figure 6 displays, it opens the edge sides of the graphite, allowing sulfuric acid to inter its inner layer spacing that helped in intercalation along with the emission of gas as graphite decomposes and swells. The formed EG was a worm-shaped structure.
10
However, the electrical conductivity of FG decreases with its expansion volume, due to the influence of different factors including the enhance in contact resistance happening between both layers and porosity as well as deterioration in the orientation of EG. Therefore, under an optimum expansion rate, the electrical conductivity increased up to 5.47 × 104 S/m. Hence, under the ideal expansion rate, the resultant EG showed enhanced electrical conductivity, making it appropriate for advanced applications in many conductive materials as well as the formation of high-quality graphene.
10
The mechanistic scheme of K2S2O8 chemical intercalation and expansion. Reprint with permission from [Bo Hou], [Rapid preparation of expanded graphite at low temperature], published by [ELSEVIER], [2020].
10
Another investigation compared programmed heating and thermal shock methods for expanding intercalated graphite. Bulk density ranged from 0.017 to 0.018 g/cm3 for the lowest-yield samples, increasing to 0.058 g/cm3 at an 89.2% yield. Programmed heating produced EG with superior texture characteristics and a high specific surface area of ∼299 m2/g at a moderate expansion temperature of 400°C, indicating a favorable balance between porosity and structural integrity. 75
Ultrasonication
The ultrasonic approach to EG preparation involves the cautious selection of solvents and exhibits quite a good yield. 9 The graphite intercalation compounds (GICs) can work as both electron donor reagents including reducing agents like alkali metals which lead to metallic graphitised and electron acceptors containing oxidizing agents like halogens or oxoacids which lead to graphite salts. 76
In an alternative study, both graphite mixture as well as ultrasonic solvent were placed inside the flask that is present in the ultrasonic cleaning bath, which usually occurs at room temperature. Several experimental conditions such as ultrasonic solvent, ultrasonic power usually having nominal power of 500 Wand 250 W and ultrasonic time were provided. The ultrasonic medium was quite important for different factors including the transfer of energy, allowing sound waves to interrupt van der Waals forces while enhancing the interlayer spacing. The graphite mixture was then washed with water for neutralization, dehydrated and lastly. The formed expandable graphite or GICs were then heated at about 900°C to achieve expanded graphite. The expanded volume was calculated by simply taking 1.0 g of the expandable graphite in the graduated cylinder. The final EG included diamond-like pores. Hence, such a process not only explains how ultrasonic energy increases the penetration of the intercalating agent along with the separation of layers but also impacts the porosity as well as the morphology of the resulting EG. 8
In another research work, graphene was formed using the expanded graphite through the ultrasonic approach. It can be used to convert expanded graphite (EG) into few-layer graphene (FLG) via cavitation-induced exfoliation. In one approach, EG was subjected to controlled heating and ultrasonic treatment to increase cavitation energy at the graphite surface, followed by shear forces in a milling chamber that delaminated the flakes into ultrathin layers. 77 Another study prepared graphene with promising supercapacitor performance by sonicating EG in various organic solvents. A highly dilute suspension minimized reaggregation of exfoliated sheets, improving sheet quality but resulting in a low overall yield. Although this method produces high-quality graphene, yield limitations remain a challenge for scale-up. 77 In a recent study, graphitic materials have application in drug delivery mechanism., Graphene aerogel nanoparticles synthesized through vitamin C mediated reduction and sonication enabled the in-situ drug loading and pH-responsive anticancer release. 78
Microwave heating
Microwave irradiation offers a rapid, uniform, and energy-efficient approach for converting graphite intercalation compounds (GICs) into expanded graphite (EG). By directly heating intercalants, microwaves induce their vaporization and generate the pressure needed for layer separation. 7 But this technique is restricted by by its high energy consumption and many challenges to scale up for the industrial production.
Another research involves a microwave-assisted hydrothermal step using flake graphite, H2SO4, and K2S2O8 as oxidative intercalation, followed by 40 s of microwave heating at 800 W.
The resulting EG reached an expansion volume (EV) of about 455 mL/g, with high graphitization, minimal structural damage, and excellent performance in conductive foils, oil sorption, and flame-retardant applications. The manufactured EG also shows effective adsorption of oils such as hydraulic oil, vegetable oil and outstanding flame-retardant qualities. Such a synthesis method is preferred because it is green, cheap, safe and quite effective for preparing EG. 79
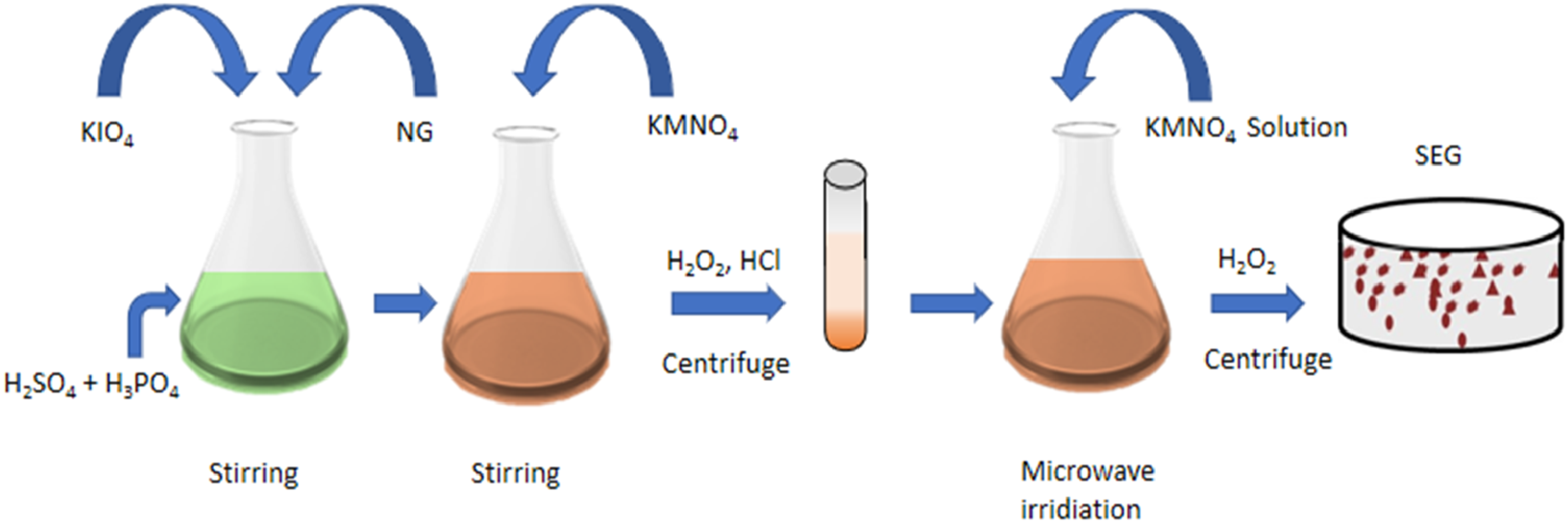
A green method was used to prepare EGs in which Ammonium Persulfate (NH4)2S2O8 and Sulphuric acid (H2SO4) were utilized as intercalate agents followed by the microwave irradiation. This method produces great expansion volume of about 267 L/kg using microwave irradiation process at 500 W for 60s.80,81 In an alternative research work, a newly graphite-based product also known as slight-expanded graphite (SEG) was prepared. Such a process only needs a minimal amount of conc. H2SO4 as well as using fewer toxic chemicals such as Potassium periodate (KIO4) and KMnO4 while disregarding the utilization of HNO3, K2Cr2O7, or KClO4. In this, first natural graphite was converted into graphene oxide using an intercalation process and then the formed product was microwaved to achieve SEG (shown in Figure 7).
82
The diagram of the preparation process of SEG. Reprint with permission from [Liu, Zhen-Xue] [Microwave-assisted fabrication of slight-expanded graphite under normal temperature], published by [Materials Science and Technology], [2020].
82
Electrical heating
Electrical heating during the synthesis of EG is highly potential for scale production of EG because of its techno-economic and eco-design characteristics. 42 At high temperatures, the thermal expansion of graphite occurs by the intercalation of ions (examples: SO42−, NO3−, Li+, Na+, K+, etc.) and oxidizing agents, which helped in the exfoliation process. Finally, the obtained TEG, an intumescent form of graphite, has been used in the preparation of composite materials with various conducting polymers (examples: epoxy, poly(styrene-co-acrylonitrile), polyaniline, etc.) and metal chlorides (examples: FeCl3, CuCl2, and ZnCl2) for hydrogen storage, thermal energy storage, fuel cells, batteries, supercapacitors, sensors, etc. The important characteristics of TEG are that it possesses a highly porous structure, low apparent density (0.002–0.02 g cm−3), high mechanical strength (10 MPa), thermal conductivity (25–470 W m−1 K−1), electrical conductivity (106–108 S cm−1) and low cost. The porosity and expansion ratio of graphite layers could be controlled in accordance with the needed values through the regulation of temperature and the use of suitable intercalation ions. 42
Recent work has demonstrated a programmable heating method in which GICs are heated at controlled rates (20°C/min from room temperature to 400–700°C). Compared with thermal shock, programmable heating at 400°C and 2°C/min yielded higher specific surface areas (184–699 m2/g), improved textural control, and higher EG yields (78–90%), while increasing bulk density. This approach provides flexibility in the development of EG for specific functional and structural requirements. 83
Mechanical milling
Mechanical approaches for the production of expanded graphite (EG) rely on the application of external forces to delaminate graphite layers, thereby minimizing or eliminating the need for high-temperature treatment or chemical intercalation. This strategy offers clear environmental advantages by reducing energy consumption and preventing residual intercalant contamination in the final material. 84 The experimental study investigates graphene nanoplatelet synthesis through a combination of expanded graphite compound preparation and mechanochemical processing. This method uses cold expansion on graphite impregnated with ammonium persulfate and sulfuric acid solution. A base treatment (urea) reduces the acidity and redox potential of the system before organic components are added to the expanded graphite compound. The resulting GNPs exhibited improved dispersibility due to glycerol’s stabilizing effect. 84 In an alternative study, two EGs named as EG-1 and EG-2 were produced through the continuous heating process of expandable graphite along with providing high milling energy (in a planetary way) under atmospheric air. The evolution of such ball-milled materials was evaluated by XRD and HRTEM on a micro-level. 85 The results of XRD showed that changes in the degree of average thickness of the crystallite with EG-2 c-axis are lower compared to EG-1 throughout the process. Furthermore, HRTEM results showed a few curved graphene planes in both samples of EG. But, EG-1 had sharply curved graphene planes and EG-2 showed smooth textured curved graphene planes along with high angles of bending.
A dry, mechanochemical approach has been developed to synthesize exfoliated graphite using environmentally benign reagents. In this method, equal parts of graphite flakes and calcium carbonate were mechanically milled, followed by mild treatment with vinegar-based acetic acid. Milling and acid treatment reduced graphite thickness up to 5 nm while retaining micro-meter-scale lateral dimensions. The specific surface area increased dramatically from 4 to 363 m2/g, with a high material yield of 92%. This method provides effective adsorption for methylene blue which frequently contains textile wastewater. 86
Explosive combustion and blending method
The explosive combustion method alongside the blending method stands out as a leading approach to produce EG. The natural flake graphite mixture containing nitric acid and sulphuric acid underwent 6 h of stirring followed by washing and subsequent drying steps to remove impurities. Researchers incorporated red phosphorus into GIC to trigger the rapid expansion process that resulted in EG formation. A mechanical blending process combined Ag2O, CuO, and ZnO materials to create composites that received oven drying below 60°C for 3 h. The pores in the EG exhibited outstanding development, forming interconnected network patterns. The proposed scheme is shown in Figure 8.
84
Proposed formation mechanism of EG sample. Reprint with permission from [Melezhik, A. V.], [Mechanochemical synthesis of graphene nanoplatelets from expanded graphite compound], published by [Springer], [2016].
84
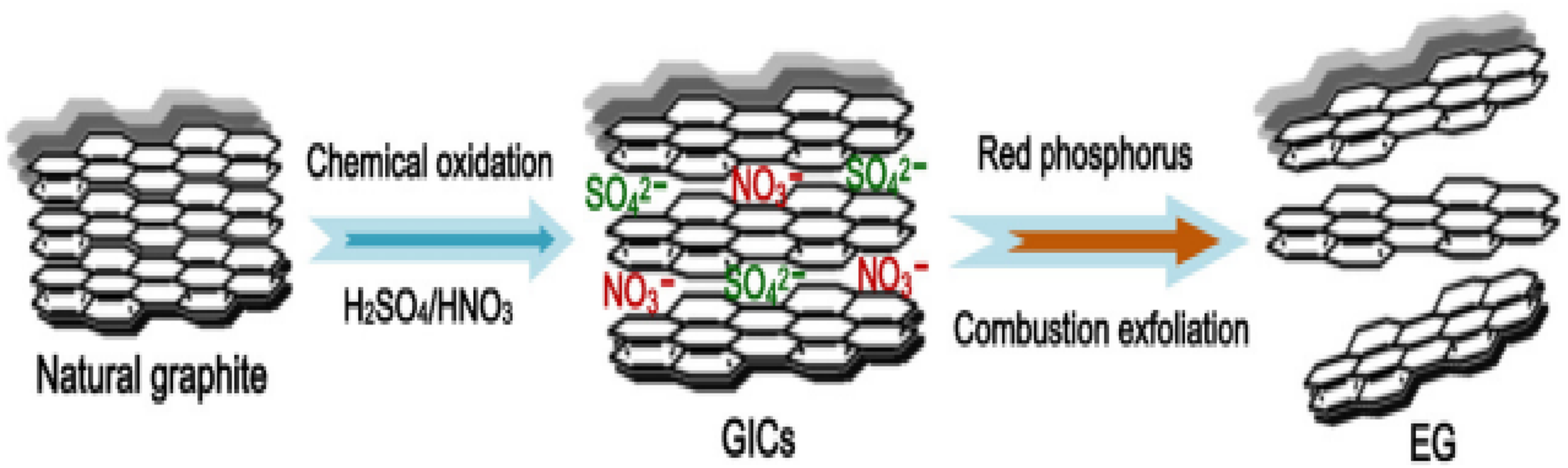
According to an additional study, the first natural flake was dried, mixed with both nitric as well as sulfuric acid, then stirred evenly for the purpose of obtaining GICs. The formed material was then washed, neutralized, dehydrated as well as dried a second time for the purpose of getting expandable graphite. 87 After that, red phosphorus was mixed into GIC which needs to be placed in an explosive container. Throughout this process, GIC expanded continuously to give EG. Therefore, the nitric acid in this process acts as both an intercalating agent as well as an oxidizer that promotes the work of sulfuric acid. The GIC in this process was exfoliated by continuous heating at a very high temperature, resulting in the breakdown of the chemical bonds between the compounds and their functional groups. Hence, the remaining compounds degrade and expand significantly to form a consistent porous EG material. 87
The summary of recent research shows that EG can be easily prepared via the method of explosive combustion shown in Figure 9. The separately placed red phosphorus received an appropriate dosage inside expandable graphite which entered an explosive container. The explosive combustion reaction rapidly expanded the expandable graphite raw material and yielded an expanded volume of 270 ml/g at 800 C when processed.
88
Table 2 describes about different types of physical methods for the preparation of expanded graphite and their effect on obtained properties. Preparation of magnetic expanded graphite by explosive combustion. Reprint with permission from [Hung, Wei-Che], [Preparation and infrared/millimeter wave attenuation properties of magnetic expanded graphite by explosive combustion], published by [Materials Express], [2016].
88
Different types of physical methods for the preparation of expanded graphite and their effect on obtained properties.

Chemical methods to prepare the expanded graphite
Expanded graphite is synthesized from graphite via a sequence of chemical procedures that incorporate intercalating agents, followed by the application of thermal energy to facilitate the expansion of the material. The primary chemical methodologies encompass processes of chemical oxidation, 86 electrochemical methods, 91 acid intercalation and thermal expansion, 12 liquid phase exfoliation, 12 Chemical vapor deposition, 92 hydrothermal method, 93 and chemical oxidation (Hummers’ method). 94 Several other techniques related to chemical methodologies are also used for preparing expanded graphite.
Electrochemical Intercalation
The electrochemical method for producing expanded graphite (EG) has significance for its speed, efficiency, and eco-friendly nature, even though it typically requires graphite paper or foil, adding extra processing steps. 8 In recent study, natural graphite (NG) was intercalated by both chemical (CI) and electrochemical (EI) methods. Following expansion, milling, and blending (1:1) with pristine NG, EI-derived materials exhibited superior electrochemical performance when employed as conductive cathodes in alkaline batteries. EI samples outperformed CI samples due to higher intercalate content, better particle integrity, greater expansion, porosity, and surface area, leading to improved exfoliation. 12 Furthermore, these graphitic materials have applications in nano-bubble formation. The ability of GO dispersions to nucleate stable micro and nano-bubbles, broadened their utility in fluid dynamics and gas liquid interfacial engineering. 95 Additionally, core shell Au@GO nanoplatforms prepared with GO coating and sequential PEG/arginine functionalization exhibit efficient miRNA loading and NIR-triggered photothermal release. 96
Acid intercalation and thermal expansion
Acid intercalation introduce chemical species (intercalates) into the layered structure of graphite, forming graphite intercalation compounds (GICs), which serve as precursors for EG. 73 Conventional procedures are costly, energy-intensive, and not environment friendly. A simplified, efficient approach uses K2S2O8 in concentrated H2SO4 at 80°C for 5 min, achieving up to 150 mL/g expansion with minimal sheet damage. The resulting worm-like EG, analyzed via XRD, SEM, FT-IR, and Raman, and formed into flexible graphite film, showed high electrical conductivity (5.47 × 104 S/m). Effectiveness was attributed to mild oxidation and oxygen release during K2S2O8 decomposition. 73 In a recent study, −10 mesh expanded graphite (EG) exhibited an expansion volume of 80 mL g-1 at 1150°C, whereas commercially available +50 mesh EG achieved approximately 400 mL g-1 under comparable thermal conditions. The finer EG displayed a well-developed mesoporous architecture, with pore volume increasing at elevated temperatures. This enhanced porosity is advantageous for applications requiring high surface accessibility, including adsorption, heterogeneous catalysis, and electrochemical energy storage. 13
Liquid phase exfoliation
This procedure entails the mechanical segregation of graphite layers within a liquid medium, typically involving a solvent or surfactant solution. 12 Multi-layer graphene was synthesized using hexagonal graphite (HG) as the precursor. The HG was combined along with an acidic mixture, subsequently dried, and passed for a thermal treatment. The resultant EG underwent sonication for 1 h, while being blended with an organic solvent. Upon completion of these investigations, it was determined that nano-sized graphene sheets had been successfully produced. 12
Another approach integrated laser ablation of a graphite substrate in liquid, with controlled target composition, laser wavelength, and ablation medium to produce high-quality few-layer graphene nanosheets more than10 layers. Characterization via FE-SEM, TEM, XRD, UV–vis-NIR, Raman, and XPS showed micro-meter-sized sheets deposited on flexible graphite substrates, offering potential for electrochemical, hydrophobic, and fundamental research applications. 97
Chemical vapor deposition (CVD)
Chemical vapor deposition (CVD) grows carbon layers, including expanded graphite (EG), by thermally decomposing gaseous hydrocarbons (e.g., methane, ethylene) more than at 1000°C. 92 Chemical vapor infiltration was employed to create carbon/carbon-based composites utilizing exfoliated graphite as well as pyrolytic carbon. Two distinct types of exfoliated graphite, compacted to densities ranging from 0.05 to 0.4 g/cm3, served as the preform. The method explains how pressure, temperature, duration affecting infiltration, morphology, and composite properties, analyzed via Raman, SEM, and nitrogen adsorption. 92
Hydrothermal method
The hydrothermal method for creating expanded graphite involves synthesizing graphene oxide, which is then expanded to produce EG. This approach is particularly useful for achieving a high expansion rate while being remain as environmentally non-toxic. 98
A separate study describes a sulfuric acid assisted method for the synthesis of expanded graphite (EG), in which natural graphite (NG) is treated with sulfuric acid, followed by the controlled addition of nitric acid and subsequent heating in a Teflon-lined autoclave. After hydrothermal processing, the product is thoroughly washed and dehydrated, then rapidly thermally shocked at 1000°C for 30 s to induce graphite expansion. To evaluate the effectiveness of this approach, ultrasound irradiation was additionally employed and compared with the hydrothermal method, as illustrated in Figure 10. The final product was washed, dehydrated, and dried in an oven at approximately 60°C to obtain EG.
93
Plot of expanded volume versus acid ratio (H2SO4/HNO3) for EGs obtained using the conventional liquid-phase, the ultrasound irradiation, and the hydrothermal methods. Reprint with permission from [Kuan, Chen‐Feng], [Preparation of expandable graphite via H2O2‐hydrothermal process and its effect on properties of high‐density polyethylene composites], published by [Wiley Online Library], [2012].
93
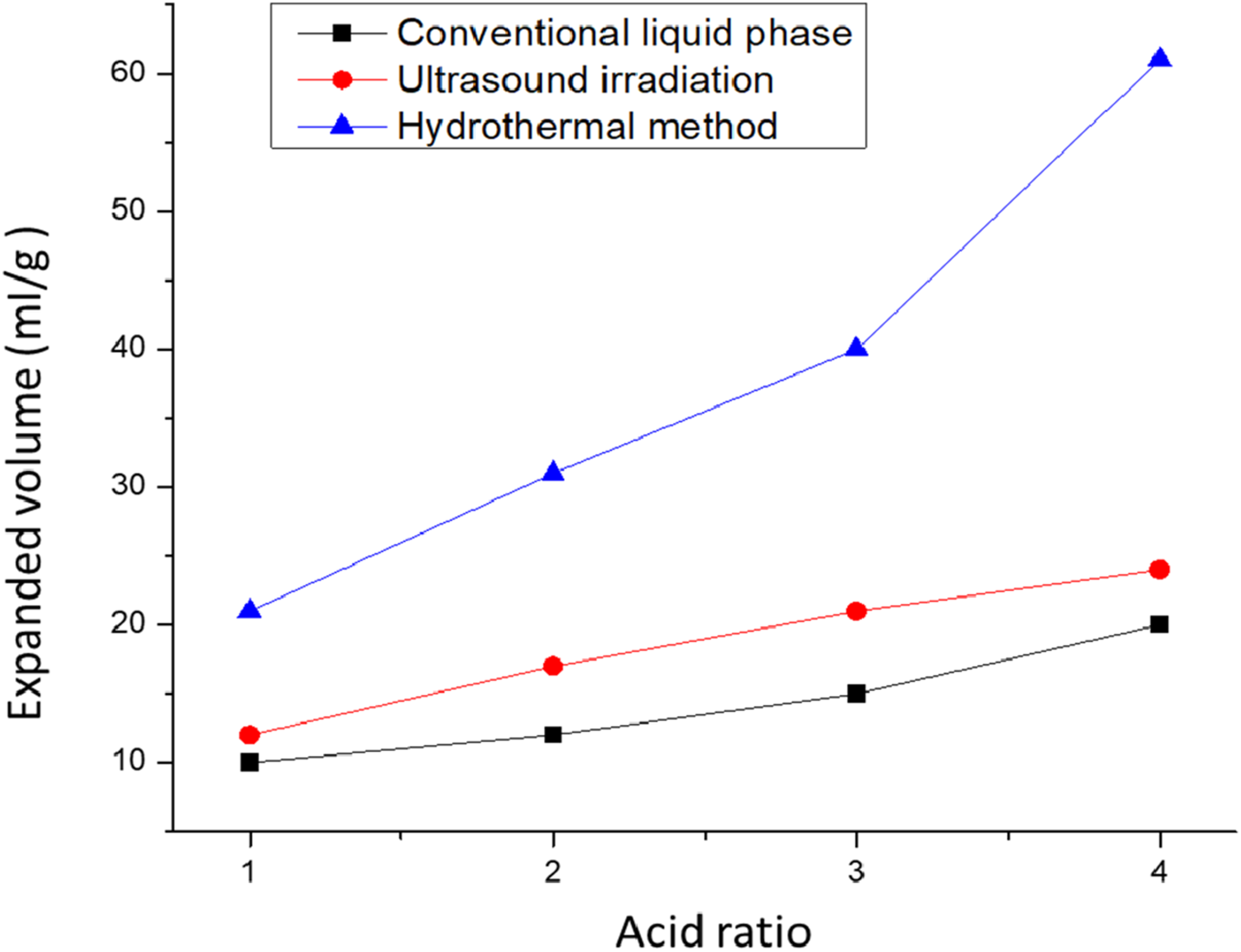
According to supplementary research, EG was effectively synthesized using the H2O2-hydrothermal approach. Subsequently, the H2O2-HEG (hydrothermally produced EG) was evaluated against EGs derived from traditional liquid phase synthesis (CEG) and those generated through ultrasound irradiation (UEG). The expansion ratio of the EG produced via the H2O2-hydrothermal procedure surpassed that of the other methods when utilizing a similar concentration of an acid mixture. The findings indicate that H2O2-hydrothermal method is superior in achieving a greater expanded volume of the EG. Both FTIR as well as XPS analyses validated the occurrence of oxidation along with intercalation reactions among the reactants and NG. 93
Chemical oxidation (hummers’ method)
The process of chemical oxidation is the most commonly used as compared to all the previously stated techniques. 94 At the beginning of another study, graphite was oxidized along with a combination of both potassium permanganate (KMnO4) as well as sulfuric acid. This reaction yields graphite oxide, which exfoliates on quick heating to form EG. Afterward, the chemical residuals are then eliminated through washing and neutralization. However, this method requires the usage of strong oxidants during the synthesis, resulting in the production of environmental pollutants, which restricts its additional applications. 10 A scientist named Hummers, in 1958, described the most widely used method now as: the oxidation of graphite using KMnO4 along with NaNO3 in concentrated H2SO4.99,100
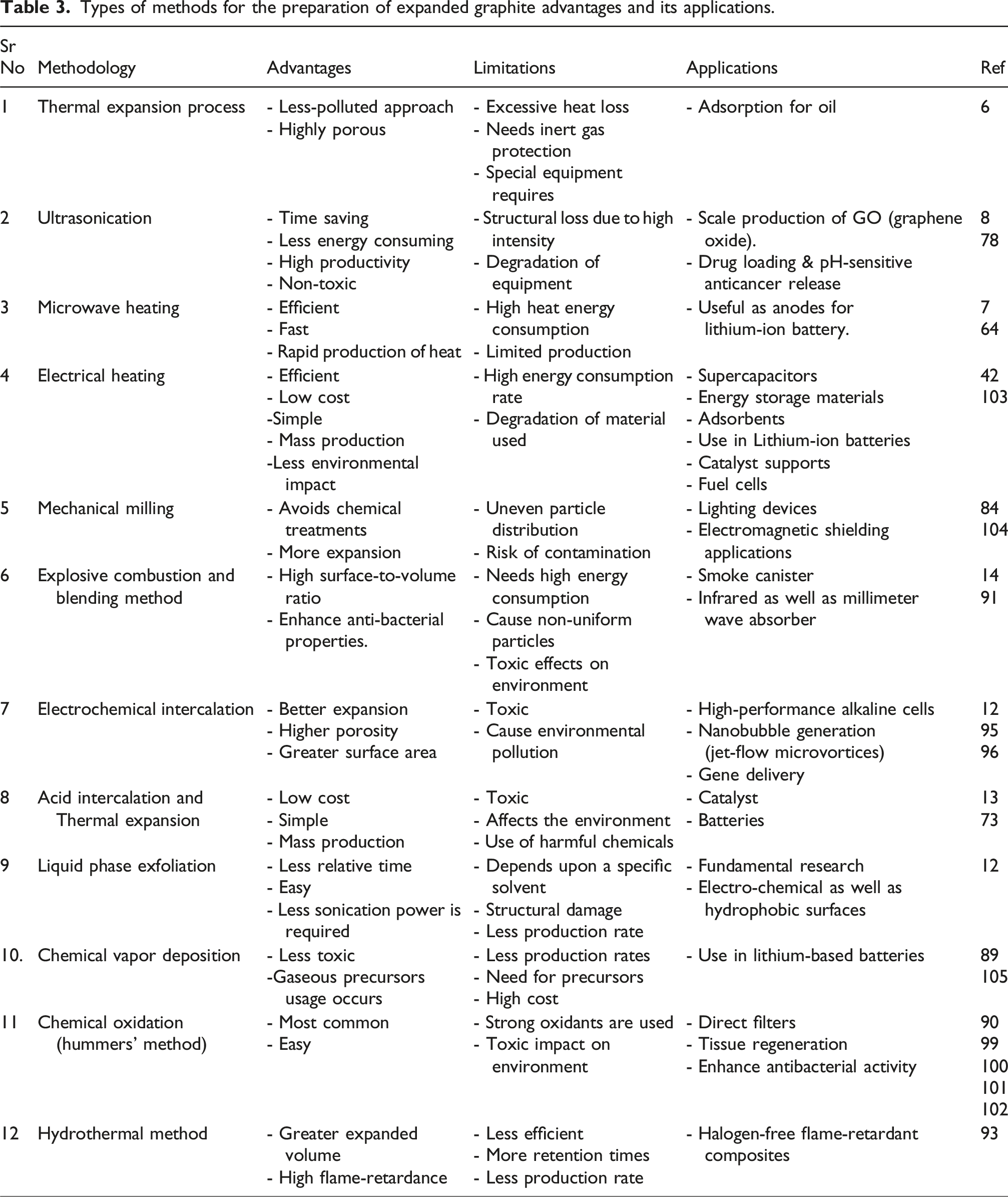
Types of methods for the preparation of expanded graphite advantages and its applications.
The synthesis of elastomeric materials with properties such as high flexibility and dielectric permittivity has gained considerable attention in recent years, particularly for applications in dielectric electroactive polymers. Expanded graphite (EG), even at low concentrations (up to 5 wt%), is emerging as a promising filler for extremely soft elastomers, where traditional fillers would compromise softness and mechanical stability. 106
EG as a filler can improve the conductivity, thermal stability, and mechanical properties of elastomers. The interfacial interactions between EG and the elastomer matrix (such as physical embedding, chemical bonding, interfacial polarity matching, etc.) depends upon the interaction of EG surface functional groups, particle size, and layer number. These factors determine the interfacial bonding strength and stress transfer efficiency. In fact, the EG prepared by oxidation methodology may have some functional groups present on its surface such as –OH, –COOH, –C = O etc. These groups remain active to form different types of hydrogen and covalent bonds with polar sites or epoxide rings available on ENR. 107 Another way to improve the interfacial adhesion and interaction is the hybridization of filler geometries. Where EG can be used with the combination of other carbon based filles to make the hybrid filler system to improve the thermal conductivity and mechanical properties of polyamide-based composites. 108
It also has been observed that the limited exfoliation of EG particles can also impact on the functional and mechanical properties of EG embedded composites. The limited amount of exfoliation causes the aggregation of filler materials due to poor interfacial interactions. The resulting product is therefore a macro-composite as shown in Figure 11. Hence, the process of melt compounding is selected to exfoliate the graphites into constituent layers, which are loosely bonded with each other by weak van der Waals forces. They are therefore amenable to delamination into few and multi-layer graphene nanoplatelets, under the action of the shear forces that develop during melt compounding. The incorporation of exfoliated fillers into ENR showed excellent improvement in the mechanical, electrical and thermal properties into resulting composites. (a) Graphite surface before exfoliation, (b) expanded graphite exfoliated by melt compounding method. Reprint with permission from [Quang Binh Ho], [Exfoliation of graphite and expanded graphite by melt compounding to prepare reinforced, thermally and electrically conducting polyamide composites], published by [ELSEVIER], [2019].
109

Juliean et al. conducted a study to improve the mechanical performance of expanded graphite reinforced polymer. The chemically developed (silane and polydopamine functionalized) expanded graphite were incorporated into ultra-high molecular weight polyethylene (UHMWPE). The resultant composite with enhanced interfacial adhesion between EG and polymers were characterized against mechanical and physical properties. 110 For better understanding, practical strategies could be used to enhance interfacial adhesion between expanded graphene (EG), graphene oxide (GO) and polymer matrices, which have been widely validated across carbon, rubber, graphene material in literature. Silane coupling agents particularly includes 3-aminopropyl-triethoxysilane (APTES), 3 methacryloxypropyltrimethoxysilane (MPS), and sulfur-containing TESPT/Si-69, which made covalent or condensation linkages with GO/EG surfaces, improving dispersion, crosslink density, and filler matrix stress transfer.111,112 Graphene oxide grafted through APTES notably enhances mechanical reinforcement, while bis[3 (triethoxysilyl)propyl] tetrasulfide (TESPT)/Si-69 is well established in rubber systems for chemically bridging carbon fillers with elastomer chains and reducing hysteresis. Covalent functionalization (–NH2, –SH, –COOH), achieved via controlled oxidation, plasma, or acid treatments, enables amidation or esterification reactions and promotes strong interfacial bonding. The amination pathways further interact favorably with epoxide groups in ENR. 112 Hybrid filler architectures (EG + CNTs, EG + MXene, EG + activated carbon) generate hierarchical conductive pathways where CNTs bridge inter-sheet gaps and MXenes add surface redox functionality, collectively lowering percolation thresholds and enhancing cyclic durability. 113 Polymeric grafting through diazonium chemistry, or compatibilizers such as polymers grafted through maleic anhydride improves interfacial wetting and entanglement, reinforcing stress transfer across the interface. 114 Among coupling agents, APTES provides effective aminosilane grafting, 112 TESPT/Si-69 enhances sulfur-cured elastomer bonding, 112 and vinyltrialkoxysilanes introduce hydrophobic or polymerizable linkages. 115 Selection of functional groups should match matrix chemistry (e.g., aminosilanes for epoxide-rich ENR; sulfur silanes for NR), while careful control of grafting conditions (pH, moisture, solvent) ensures reproducible functionalization.
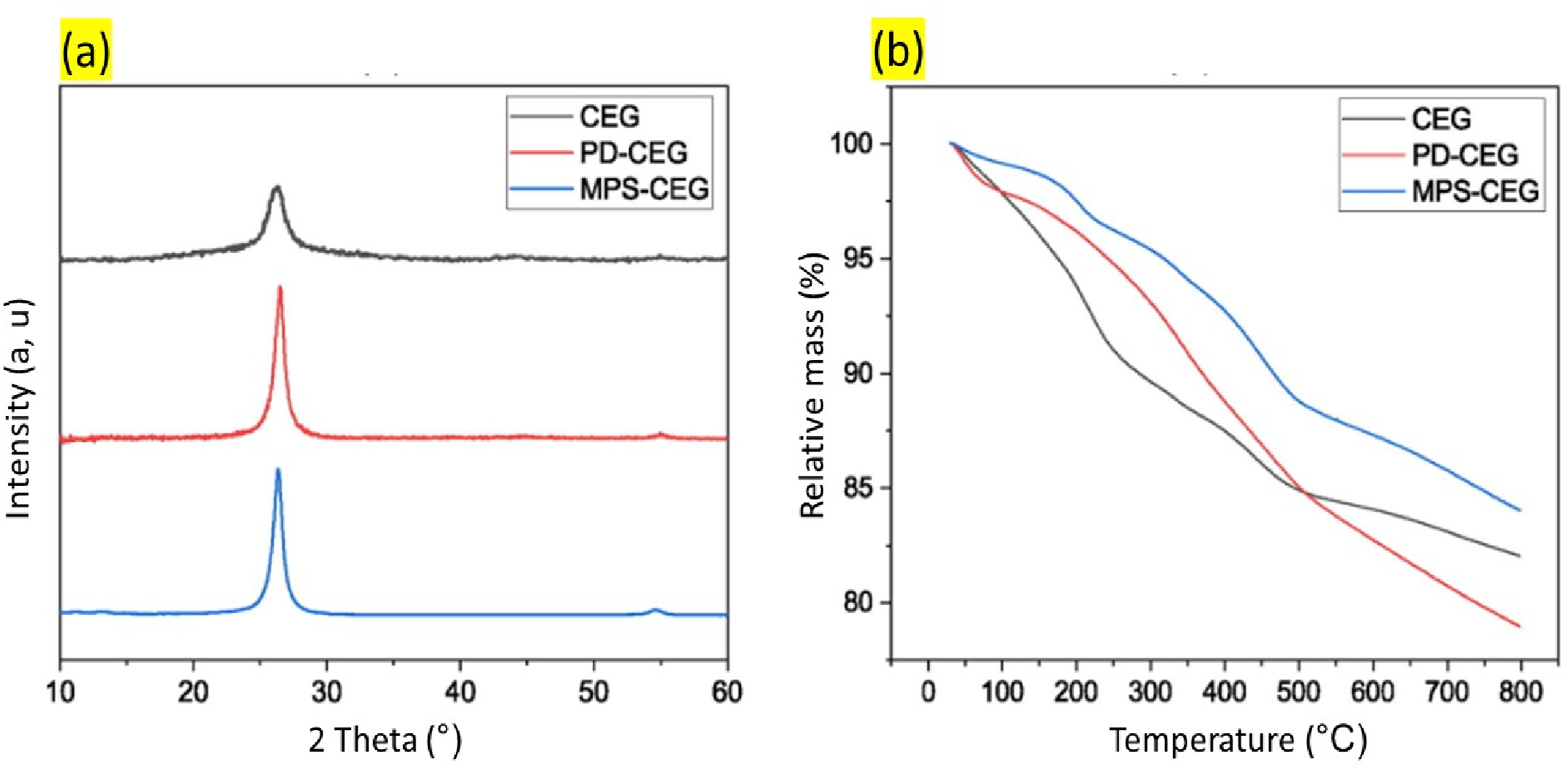
The addition of functionalized CEG enhanced the mechanical properties of the UHMWPE matrix with an increase in micro-hardness of up to 25% and storage modulus of up to 58%. XRD of the composite provided a broad 002 diffraction beak at 26.4°. This peak corresponds to that of unexpanded graphite. The weak intensity and wide nature are related to the oxidation (due to chemical modification) of the starting graphite. While, the reduction of chemically modified EG by the functionalization process allows for the partial restoration of the initial graphitic lamellar structure, giving rise to an enhanced 002 peak intensity. The mass loss curves as found by TGA can be found in Figure 12. The mass loss of the pure composite indicated an oxygen content of 15 wt%, closely matching the XPS data. Although TGA is considered a powerful tool to determine the degree of functionalization, the evident reduction of CEG makes determining the degree of functionalization challenging.
116
Characterisation results of chemically modified expanded graphite polymers (a) XRD diffraction pattern, (b) TGA mass loss curves. Reprint with permission from [Julian Somberg], [Chemically expanded graphite-based ultra-high molecular weight polyethylene nanocomposites with enhanced mechanical properties], published by [ELSEVIER], [2022].
110
Critical evaluation of eco-friendly and green synthesis strategies for expanded graphite
In recent years, numerous studies have proposed environmentally friendly approaches for preparing expandable graphite (EGp) and expanded graphite (EG). However, many of these reports focus primarily on demonstrating feasibility at the laboratory scale, with limited discussion of practical constraints such as scalability, process stability, cost implications, and environmental trade-offs. A meaningful evaluation of “green” synthesis therefore requires moving beyond descriptive claims and examining whether these alternatives can realistically compete with conventional acid–oxidant systems in industrial contexts.
Practical feasibility for expanded graphite production
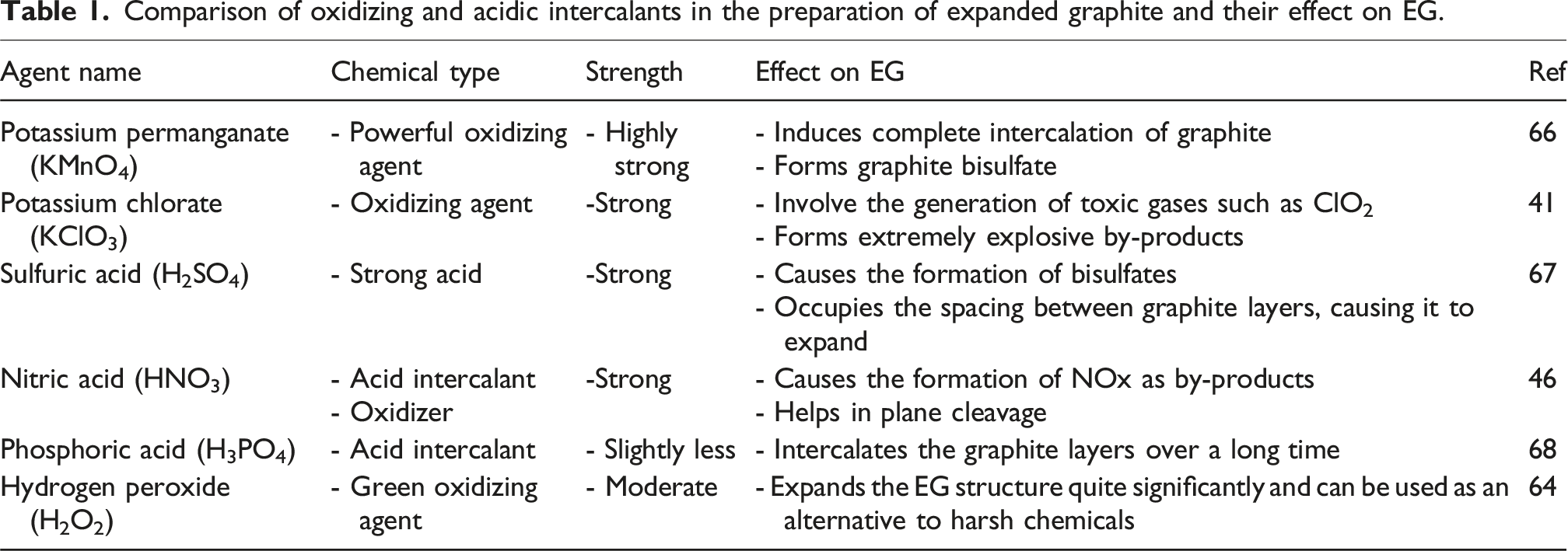
Conventional EG production is typically based on concentrated acid intercalation systems involving H2SO4, HNO3, KMnO4, or KClO3. These methods consistently provide high intercalation efficiency and large expansion volumes.99,100 However, they are associated with acidic effluents, sulfate residues, NOx emissions, and manganese-containing wastes, all of which raise environmental and regulatory concerns. To address these issues, alternative oxidants such as hydrogen peroxide (H2O2), ammonium persulfate ((NH4)2S2O8), and hydrothermal-assisted intercalation routes have been explored.48,93
Hydrogen peroxide has been considered as a green oxidant due to its decomposition into H2O and O2, and avoiding persistent toxic residues. 64 However, although it’s a chemically milder in nature, H2O2-based systems often require controlled temperature, prolonged reaction time, or secondary intercalation steps to achieve comparable expansion ratios to sulfuric acid-based systems. This suggests that complete replacement of strong mineral acids may compromise intercalation depth unless process conditions are carefully controlled. Similarly, ammonium persulfate-based intercalation combined with microwave irradiation has exhibited rapid expansion with relatively lower toxic emissions. 80 Yet persulfates remain strong oxidants and require controlled handling. While such approaches mitigate specific environmental risks, they do not eliminate oxidative chemical inputs altogether. 80 Mechanical and mechanochemical strategies further reduce liquid reagent consumption. For instance, dry milling followed by mild acetic acid treatment has produced materials with surface areas up to ∼363 m2/g and yields near 92%. 86 However, these routes mainly decrease particle thickness and increase surface area rather than generating the characteristic worm-like, high-expansion morphology obtained through thermal exfoliation. Consequently, for applications demanding high expansion volume, such as flame-retardant systems or compressible conductive structures, purely mechanical approaches may not provide a full substitute. Compared with the classical Hummers or acid permanganate methods99,100 green strategies significantly reduce heavy metal residues and NOx emissions. However, conventional systems still provide superior and reproducible expansion volumes, making them attractive for industrial flame-retardant and large-scale conductive filler applications. Thus, environmentally friendly approaches often prioritize chemical safety and effluent reduction, whereas traditional acid systems prioritize expansion efficiency and process robustness. The optimal choice depends on application-specific performance requirements.
Comparative assessment for scalability, energy, cost and environmental impact
Each synthesis strategy for EG involves trade-offs between throughput, energy intensity, environmental hazard, and operational complexity. Acid–thermal intercalation remains the most mature and industrially scalable method. Lan et al. (2019) demonstrated that H2SO4/HNO3 intercalation followed by thermal expansion can produce high-quality EG while preserving graphitic structure. Despite its robustness, this route depends on concentrated acids and high-temperature treatment, increasing both environmental burden and neutralization costs. Mechanical milling and electrical heating reduce chemical waste but may require higher energy input or result in lower expansion volumes. 117 Microwave-assisted processes, as reported by Mendoza-Duarte et al., enable rapid and uniform volumetric heating within seconds to minutes, achieving expansion volumes around 150–165 mL·g-1.118,119 Hydrogen peroxide–based intercalation improves oxidation controllability and reduces reliance on harsh oxidants, yet expansion performance is highly sensitive to reagent ratios and milling conditions. 64 Hydrothermal methods, described by Wu et al. (2014), operate with comparatively benign reagents and offer stable intercalation behavior but typically yield moderate expansion ratios (∼150–160 mL·g-1) and depend on autoclave systems, limiting throughput. 120 In general, hydrothermal and green-modified peroxide routes are more attractive for small to medium scale production with lower chemical waste; microwave-assisted methods excel in rapid proof-of-concept production with minimal structural damage; while optimized acid-intercalation combined with programmed thermal expansion remains the most practical option for large-scale EG manufacturing but requires strong waste-management strategies.
Environmental trade-offs associated with graphite expansion and exfoliation cannot be judged solely on the basis of reagent selection. A process described as “green” does not automatically imply negligible environmental impact. Instead, sustainability must be evaluated through a comprehensive life-cycle lens that considers chemical toxicity, energy demand, water consumption, by-product formation, and post-treatment requirements. For example, high-temperature thermal expansion (typically 800–1000°C) minimizes chemical inputs but imposes substantial energy demand per kilogram of product, particularly when operated in batch furnaces without heat recovery systems. In comparison, hydrothermal systems function at comparatively lower temperatures; however, they rely on pressurized reactors, which extended the reaction durations, and repeated washing steps and can ultimately increase cumulative energy consumption per batch and water usage. Thus, lower reaction temperature does not automatically translate into lower environmental impact. 117 Oxidant substitution strategies present similarly nuanced considerations. Replacing permanganate-based systems (e.g., KMnO4) with hydrogen peroxide (H2O2) effectively eliminates manganese-containing sludge and lowers hazardous solid waste generation. Nevertheless, peroxide-driven processes often require extended reaction durations, higher oxidant-to-graphite ratios, or additional stabilization steps. These adjustments may increase operational energy requirements and wastewater volumes, partially offsetting the environmental gains associated with removing heavy metals. Microwave-assisted expansion further underscores this complexity. Although rapid volumetric heating significantly shortens reaction times, overall electricity consumption depends on reactor design, scale, and power density. Without careful scale-up optimization, high instantaneous power demand may counterbalance the benefit of reduced processing time. 121
From a life-cycle-oriented perspective, meaningful sustainability improvements are unlikely to arise from isolated chemical substitutions alone. Rather, progress depends on integrated process optimization. Promising directions include the use of milder intercalation chemistries, such as H2O2-assisted or electrochemical systems, to limit toxic oxidant loads; programmable thermal expansion within moderated temperature ranges (≤400–700°C) to balance energy efficiency and expansion performance; and the implementation of water recycling and acid recovery systems to reduce freshwater consumption and effluent discharge. Environmental improvement should therefore prioritize holistic process engineering, encompassing effluent management, closed-loop washing, heat recovery, and energy-efficient expansion control, and to lower cradle-to-gate impacts while preserving material functionality. 122
Standardized characterization protocol
A standardized characterization framework is critical for meaningful comparison of expanded graphite (EG) reported across the literature, as EG properties are highly sensitive to precursor characteristics, intercalation chemistry, and expansion pathways. Recent studies demonstrate that even EG materials described as nominally similar can differ by more than an order of magnitude in expansion volume (EV) and specific surface area (SSA). These variations directly influence porosity, accessible surface area, and effective percolation behavior when EG is incorporated into elastomer matrices. 123 To improve inter-study consistency and enable robust cross-comparability of reported performance metrics, a minimal yet application-oriented characterization protocol is therefore essential. As a primary descriptor, the expansion volume (EV, mL g-1) of each EG batch was determined using a standardized 1 g sample in a graduated cylinder. EV serves as a direct indicator of exfoliation efficiency and shows strong correlation with accessible porosity and pore connectivity. Prior work has shown that EV changes from ∼120 to >300 mL·g-1 can shift SSA and adsorption/thermal behavior by nearly an order of magnitude, underscoring the need for consistent reporting.121,123 Furthermore, the material should be evaluated through its specific surface area (SSA, m2·g-1, N2-BET) with clearly stated degassing conditions and porosity distribution (BJH or NLDFT) to determine the micro and mesopores available for interfacial bonding. Raman I_D/I_G ratios reported with the laser wavelength, power, baseline method, and whether peak-height or integrated area values. These parameters are essential for assessing defect density relative to graphitic order. XPS analysis, including the carbon to oxygen (C/O) ratio and detailed chemical speciation, provides insight into surface oxygen functionalities that govern chemical coupling. XRD measurements of d002 spacing and Scherrer domain size further clarify the extent of residual stacking. Morphological characteristics such as lateral flake size and thickness distribution obtained from SEM or TEM imaging should also be included. Finally, electrical (four-point probe) and thermal conductivity must be reported with complete details of sample geometry and testing conditions, including temperature and relative humidity.
The pre-sampling details such as intercalant chemistry, expansion temperature to time ratio, microwave/electrical heating or milling, post-treatments, solvents, sonication energy, provide normalized metrics (conductivity per unit SSA or per wt% filler), and give statistical dispersion (mean ± SD, n ≥ 3) and, where possible, an inter-laboratory reference or commercial standard to enable meta-analysis and percolation modelling. This minimal set is motivated by (a) observed correlations between EV/SSA and adsorption or thermal transfer performance in EG samples (case: EVs of ≈123–300 mL·g-1 corresponding to SSA changes from ≈7 to ≈72 m2·g-1 that altered adsorption/thermal behaviour), and (b) demonstrations that extreme intercalation/expansion routes can produce very large SSA (hundreds of m2·g-1 to >800 m2·g-1) but with tradeoff in defect content and stack preservation that change electrical/thermal transport, therefore process history must be recorded to interpret EV/SSA/I_D/I_G jointly. 117 The large lateral flakes with low I_D/I_G ratios should be preserved when maximizing in plane conductivity, such as in EMI shielding or Joule heating applications. In contrast, when improved mechanical coupling and fatigue resistance are required, it becomes advantageous to increase the accessible surface area and introduce controlled surface chemistry, such as thin silane layers or polydopamine coatings to enhance interfacial bonding. In many cases, hybrid architectures combining expanded graphite with CNTs or graphene nanoplatelets offer the most balanced performance, as each filler contributes a complementary function, with planar fillers supporting efficient conduction while CNTs or similar nanofillers provide crack bridging and reinforcement. 122
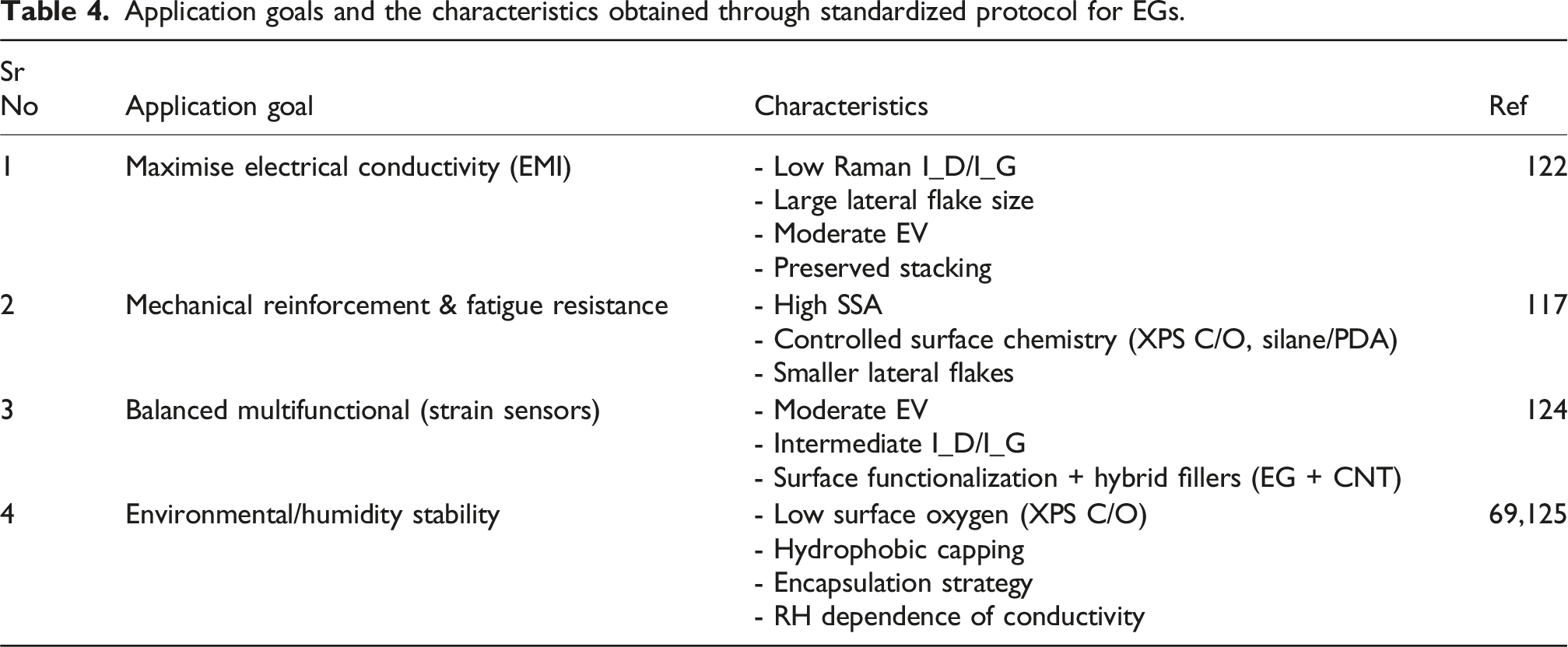
Application goals and the characteristics obtained through standardized protocol for EGs.
Application of graphite in different elastomers
Expanded graphite (EG), due to its exceptional electrical conductivity, mechanical flexibility, high surface area, and chemical stability, has been increasingly integrated into a variety of elastomeric systems. Its incorporation enhances electrical performance, self-healing capability, absorption properties, flame retardancy, sensing functionality, and dielectric behavior, enabling the development of advanced materials for applications ranging from wearable electronics to environmental remediation.
Applications of expanded graphite in electrically conductive elastomers
Expanded graphite is a porous, multi-layered graphene structure having high electrical conductivity, flexibility, and has low cost, making it a promising conductive filler in electrically conductive elastomers.
126
Conductive fillers such as expanded graphite (EG), carbon nanotubes, metal powders, and carbon fibers are used to create stretchable, highly conductive materials that possess high elasticity or stretch ability.
127
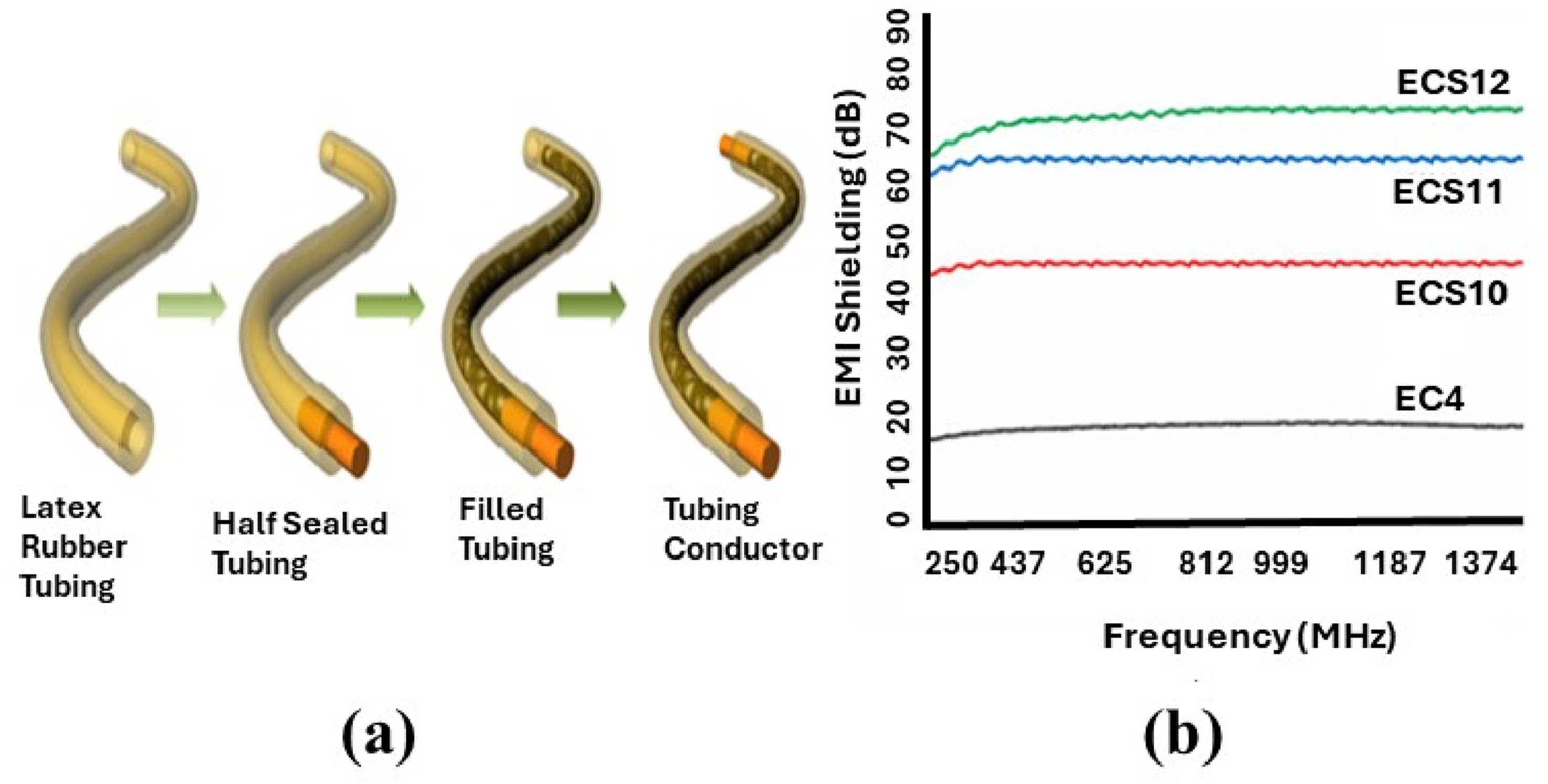
In one study, EG was used as a packed conductive filler in elastic natural rubber (NR) tubing to achieve exceptional elasticity, mechanical strength, and tunable electrical properties. This yielded stretchable electrodes, connectors, resistors, and strain sensors capable of monitoring joint and muscle movement. For the purpose of compressible as well as macro confined conductive components, the expanded graphite was sealed inside an elastic natural rubber tube which is displayed in Figure 13(a).
126
The structure schematic of the tubular conductor (TC). Reprint with permission from [Darabut, Alina Madalina], [Influence of thermal treatment on the structure and electrical conductivity of thermally expanded graphite], published by [ELSEVIER], [2022]
129
and EMI shielding of conductive elastomers. Reprint with permission from [Azam Ali], [Development of silver plated electrically conductive elastomers embedded with carbon black particles obtained from Kevlar waste source], published by [ELSEVIER], [2022].
128
Recently, EGs were synthesized and used as smart materials. In a study conducted by Omid et al., Graphite particles with a pyramidal geometry were generated by introducing controlled defects into high-purity graphite flakes, followed by intensive sonication. After sodium intercalation, these particles underwent instant exfoliation, when dispersed in water, ejecting graphene flakes at jet velocities reaching upto 7000 m/s, which generated thrust forces of at least ∼0.7 L (pN) proportional to swimmer size. This mass-ejection mechanism produced forward swimming speeds of ∼177–400 µm/s, with microscale swimmers displaying the highest velocities. The jetting behavior was attributed to the rapid formation and explosive release of hydrogen nanobubbles trapped between the Na-intercalated layers, while no propulsion was observed in organic solvents until water was introduced. When integrated with TiO2 nanoparticles, EG swimmers became remotely guidable, UV irradiation increased electron transfer into the graphene sheets, amplifying negative charge and accelerating the propulsion rate, while an external magnetic field enabled directional steering through charge-dependent Lorentz forces. Overall, the results highlight EG as a multifunctional smart material whose intrinsic electronic properties, interlayer reactivity, and compatibility with photocatalytic control make it uniquely effective for autonomous, fuel-free, and remotely tunable micro/nanoscale locomotion. 23
Another approach incorporated conductive activated carbon fillers into silicone elastomers, followed by silver electroplating to enhance surface conductivity. Electrical resistivity and EMI shielding were measured under various conditions (post-carbon infill, silver plating, stretching, and repeated stretching cycles). At 0% strain, resistivity reached as low as 12 Ω·mm with EMI shielding of 74 dB (1500 MHz). At 60% strain, resistance rose to 156 Ω·mm. Thermal stability was evaluated through TGA and Ohmic heating, and mechanical durability was confirmed under moisture exposure and tensile testing. These elastomers are promising for applications such as flexible electrodes in medical devices (e.g., EMG, ECG, TENS) and wearable sensors (Figure 13(b)). 128
Azam Ali et al. demonstrated that coating Nylon 6/Spandex yarn with carbon nanotubes (CNTs) significantly enhances electrical conductivity. SEM analysis characterized the CNTs, while electrical testing in relaxed and stretched states showed low resistance under high stretch loads. The CNT coated yarns exhibited strong heating performance, reaching up to 280°C after 60 min at 5 V, and maintained conductivity after repeated heavy washing, indicating excellent durability and CNT retention. 130 In another study, a series of immiscible polymer blends were formulated to enhance the flexibility, durability, and electrical performance of 3D-printed conductive monofilaments on textiles. The approach combined Low density Polyethylene (LDPE) based conductive composite loaded with CNTs (Carbon nanotubes) and structured carbon black with a propylene-based elastomer, where maintaining co-continuity between phases and directing filler placement remains crucial. Two-step melt processing enabled CNTs and carbon black to concentrate at the LDPE/PBE interface, improving phase continuity and significantly boosting mechanical strength, elasticity, melt flow, and conductivity compared to one-step extrusion. The optimized 60% were comprised of LDPE, KB (carbon black) and CNT; 40% PBE (Propylene-Based Elastomer) blend provides the strongest overall performance, closely matching textile behavior and demonstrating strong potential as a printable, flexible conductive material for next-generation functional textiles. By tailoring polymer blends and controlling filler distribution at phase interfaces, EG-based composites achieved improved flexibility, strength, and electrical performance compared to traditional conductive polymers. The selective localization of CNTs and carbon black around EG-rich regions enhanced co-continuity and conductivity. 131
Applications of expanded graphite in self-healing elastomers
An elastomer is flexible, rubber-like material which can withstand under the effect of force, experiences considerable deformation and rapidly recovers its estimated original shape and size on pulling back the force. Additionally, it can expand volume when comes in interaction along with various liquids and also can heal itself. 132
For the purpose of meeting practical applications as well as self-healing properties and required levels of mechanical strength are really significant for highly sensitive and flexible sensors. But it is still a challenging task to balance two earlier-mentioned parameters. In the current study, the researchers introduce low-cost, large scale and easy approach for the purpose of fabricating autonomously self-healing strain sensors possessing acceptable mechanical properties. 133 Embedding memristive nanomaterials (e.g., metal oxides, conductive polymers, graphene, MXenes) inside elastomer matrices creates composites that combine electrical memory with mechanical flexibility.134,135 Ionic memristors in elastomers including soft ionic conductors (like hydrogels or ionogels) inside elastomer substrates, can exhibit memristive ionic transport, mimicking biological synapses. 136 Mechanical tunability: The resistive state of an elastomer-based memristor can change not only with electrical input but also with mechanical deformation (stretching or compression), enabling multifunctional sensing and computation.
Lili Yang et al. reported that elastomers are such type of flexible materials that can bear force, deform to some extent and then rapidly recover to gain their real shape on the removal of force applied. They are capable of self-healing and expanding in the presence of some liquids.
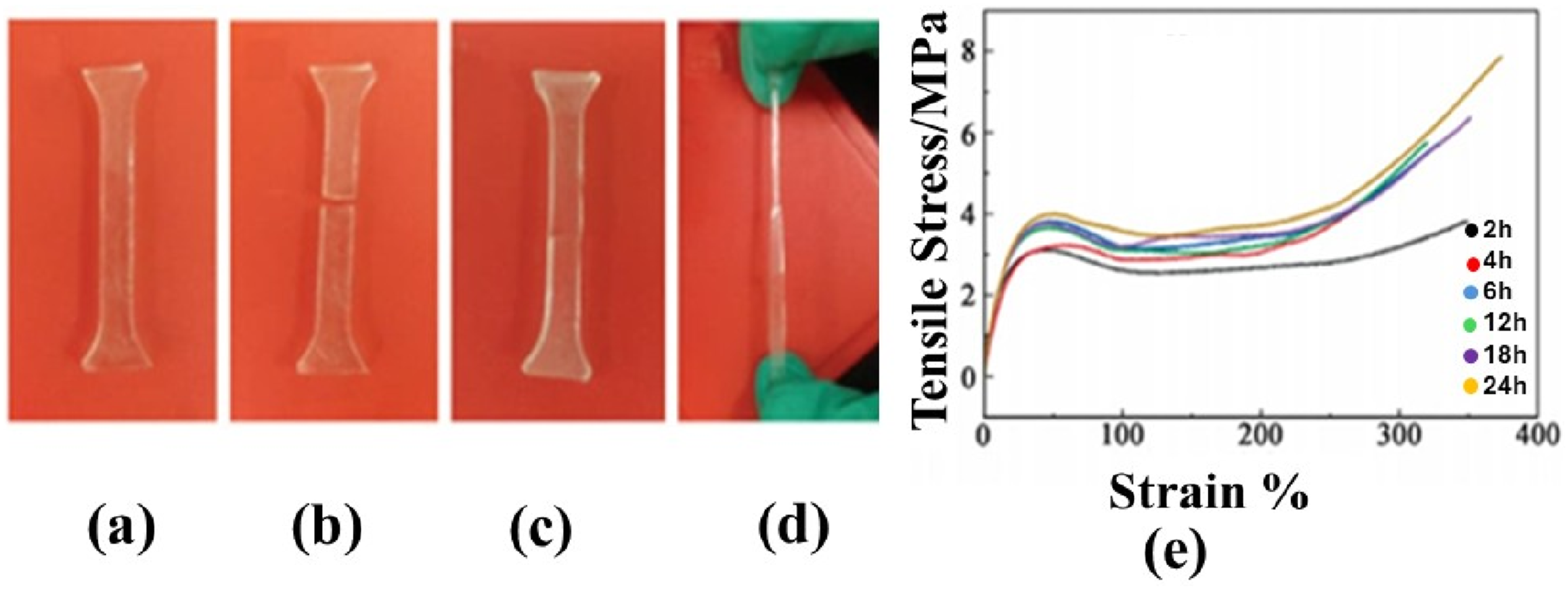
132
In a study, an elastomer was cut into a smaller piece and then set in the oven for 24 h at a temperature of 80°C. When it was withdrawn from the oven and a tensile test was done, it was seen that the sample remained intact even when necking phenomenon took place. Figure 13 explains the process. To evaluate tensile strength at 80°C temperature, the tensile process was conducted a few times on the self-healing PUSS-6 elastomers and the noted results are demonstrated in Figure 14.
132
Photos of elastomer self-repair and its evaluation (a) Original sample (b) After cutting off (c) After self-healing (d) Artificial constant pulling and (e) Tensile stress-strain curves of PUSS-6 after self-repair at different times. Reprint with permission from [Yang, Lili], [Research progress of elastomer materials and application of elastomers in drilling fluid.], published by [MDPI], [2023].
132
Applications of expanded graphite in oil-absorbing and water-absorbing elastomers
Expanded graphite is an effective adsorbent for removing oil spills due to its high surface area and ease of adsorption. 47 Oil-absorbing and water-absorbing elastomers seal the leak channels by expanding into it. EG seals leakage channels, reduces fluid loss, and improves lubrication by forming protective films, while graphene-based nanocomposites enhance pore plugging without altering fluid rheology. Engineering. 132
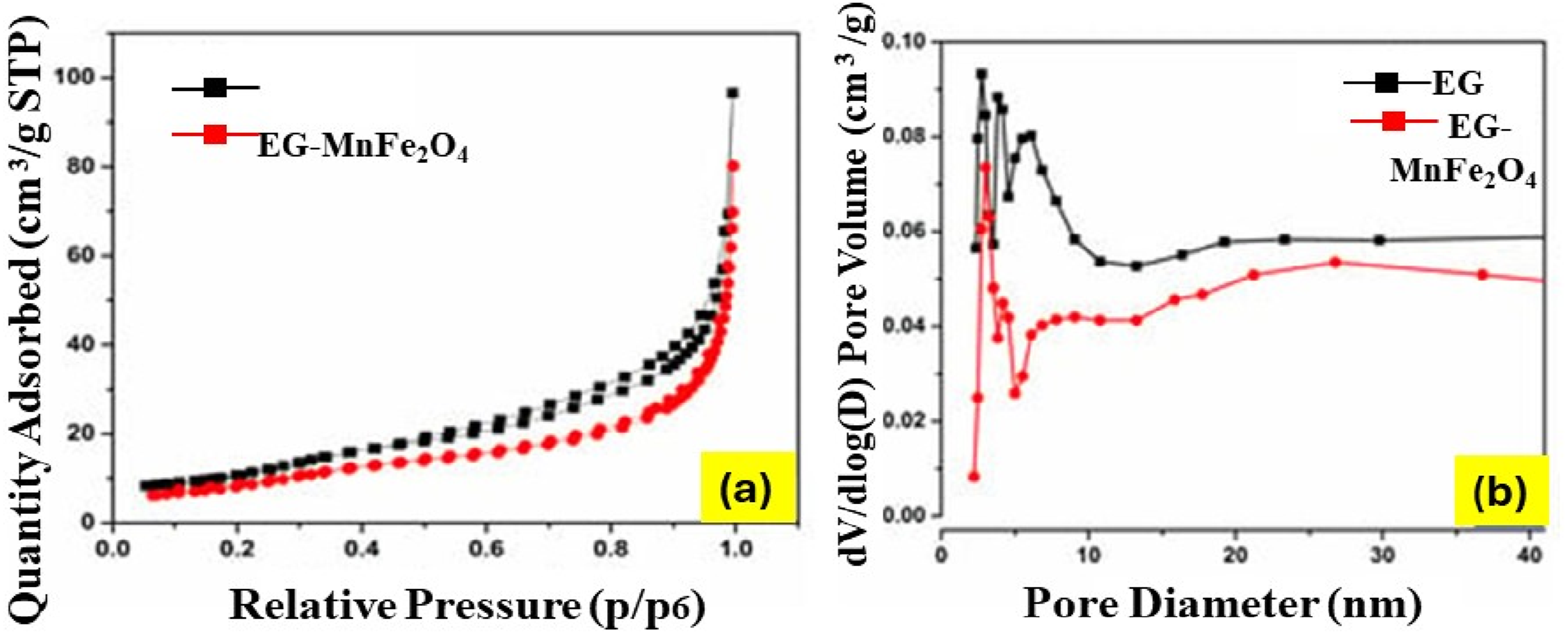
A novel approach was used to decorate EG layers with magnetic MnFe2O4 to enable easy recovery via magnetic separation. This research resulted in the assortment of adsorbents utilized on a large scale. The EG–MnFe2O4 composites, produced from Vietnamese graphite flakes, retained the worm-like EG structure and exhibited magnetism of 16 emu/g. They showed high adsorption capacitie2.20 ± 0.46 g/g for diesel oil and 33.07 ± 0.33 g/g for crude oil, making them promising, reusable materials for large-scale oil spill remediation as shown in Figure 15.
47
The nitrogen adsorption/desorption isothermal of MnFe2O4-decorated EG (A) and the corresponding distribution of the pore diameter (B). Reprint with permission from [Tuan Nguyen], [The preparation and characterization of MnFe2O4-decorated expanded graphite for removal of heavy oils from water], published by [MDPI], [2019].
47
Applications of expanded graphite in flexible EMI shielding elastomers
Electrical conductivity is a key factor in determining EMI shielding effectiveness. However, many traditional EMI shielding composites struggle to achieve high electrical and thermal conductivity simultaneously due to inefficient heat transfer pathways. Nevertheless, the effectiveness of enhancements is often restricted due to poor management of heat transfer pathways. Conversely, research indicates low molecular weight can effectively occupy voids in porous EG via polar interactions with EG surface groups, making them ideal fillers in EMI shielding elastomers. 137 Azam Ali et al. developed porous, electrically conductive activated carbon fabric from Kevlar waste through a single-stage carbonization and activation process under charcoal at 800–1200°C, utilizing a heating rate of about 300°C/h while providing no holding time. The resulting material, characterized by EDX, XRD, SEM, and BET, demonstrated promising EM shielding at both low frequencies (below 1.5 GHz) as well as high frequencies (2.45 GHz). 138
Ankur Katheria et al. highlighted the growing concern of electronic pollution, particularly electromagnetic interference (EMI), which can lead to equipment malfunctions due to the increasing use of electronic devices.
139
In contrast, polymer composites present a more cost-effective, lightweight, and corrosion-resistant solution. These composites achieve conductivity by incorporating fillers. TPEs with carbon-based fillers like EG offer a lightweight, corrosion-resistant alternative. High filler loadings are often required, but carbon nanoparticle-reinforced TPEs show promise for next-gen shielding. Azam Ali et al. addressed the limitations of traditional conductive homopolymers like polypyrrole as well as poly-3,4-ethylenedioxythiophene (PEDOT), which typically exhibit reduced mechanical properties. He improved electrical conductivity and mechanical strength by incorporating carbon particles into green epoxy resin. EMI shielding at 2.45 GHz was measured via the waveguide method. Joule heating tests showed surface temperatures reaching 55°C at 10 V. Aging tests confirmed durability in mechanical strength and moisture resistance.
140
A recent approach introduced an innovative method for producing polymer composites that exhibit both high thermal conductivity and electromagnetic interference (EMI) shielding. It blends EG with stearic acid, polyethylene wax, and linear low-density polyethylene (LLDPE) via powder mixing with thermal molding, achieving low interfacial thermal resistance. With 24.89 vol% EG, composites reached thermal conductivity of 19.6 W·m-1 K-1, EMI shielding effectiveness, and electrical conductivity up to 4000 S·m-1. Electrical conductivity increased proportionally with EG loading. Figure 16 in the study illustrates how the electrical conductivity of the EG/LLDPE-3D network composites varies with the incorporated content of EG, highlighting the relationship between the filler concentration and the electrical properties of the composites.
141
(a) Electrical conductivity of the expanded graphite incorporated composites versus filler contents in percentage (b)EMI Shielding versus filler contents in percentage (c) Comparison of SEA and SER on EG content at 10 GHz. (d) Illustration of the EMI shielding mechanism. (e) The skin depth of EG/LLDPE-3D network composites. Reprint with permission from [Wei, Baojie], [Polymer composites with expanded graphite network with superior thermal conductivity and electromagnetic interference shielding performance], published by [ELSEVIER], [2021].
141
Applications of expanded graphite in mechanical elastomers
Expanded graphite can be obtained from natural graphite. It has unique layered structure, good electrical conductivity and high surface area. It is usually incorporated into mechanical elastomers to enhance the properties of elastomeric materials and gained quite a considerable attention. 25
Meng, Qingshi et al. analyzed that to enhance the properties of polyvalent products use of nanofillers is essential, however uneven dispersion limits the performance. A new technique used like ball milling and silane coupling agent (APTES) for the purpose of boosting the properties of expanded graphite-based graphene nanosheets. It also increases the dispersion and compatibility in the PDMS matrix. The improved graphene/PDMS composite films involving even dispersion enhanced mechanical and electrically conductive properties as compared to unmodified graphene. This modified graphene enhanced the electrically conductive properties, an extensive sensing range, high sensitivity and reliable repeatability over 10,000 cycles. These properties make the material more flexible. Figure 17 displays various mechanical properties of different conductive films.
25
(a) filler contents verses Elongation (b) filler contents verses Young’s modulus and (c) filler contents verse tensile strength EG filled composite films. Reprint with permission from [Meng, Qingshi], [Durable, highly sensitive conductive elastomeric nanocomposite films containing various graphene nanoplatelets and their derivatives], published by [Wiley Online Library], [2023].
25
Due to amazing mechanical properties elastomer graphite is assumed as a new generation material. Observation shows that graphitic fillers like natural graphite flakes, expanded graphite (EG), graphite nano-platelets (GNP) and graphene depend upon rheological, mechanical properties of elastomer (also talks about particle size and filler interaction with matrix). 20
Applications of expanded graphite in flame-retardance elastomers
Expanded graphite expanded under high temperature and forms a quite stable worm worm-shaped harmless carbon layer. As a commonly used physical expansion flame retardant, it gained a main role in field of flame retardants. Due to its strong flame-retardant effect, it has been widely used in thermosetting plastics. Studies showed that only EG can enhance the flame retardancy of polyurethane elastomers, foam and coatings. 142
Jasomati Nayak et al. highlight that research on expanded graphite explains that having high sulfur content in EG is quite harmful, as it causes pollution. To gain improved compatibility, polyvinyl acetate (PVAc) was layered over EG which enhances the flame resistance as well as maintaining fly ash. The eco-friendly EG presented quite a good capability of applications in the polymeric flame-resistant materials. 41
The cone calorimeter test, the best method of bench scale to analyze the flammability properties of polymeric materials for the purpose of stimulating real fire scenarios in polymer-based materials. Both 48 curves of heat release rate as well as total smoke release curves at flux of about 35 kWm-2 are demonstrated in Figure 18. Anyhow, the flame resistant EG-g-PVAc/EVM composites presented a gradual drop of the HRR and TSR curves with the increase of combustion time and value of PHRR and TSR were also minimized by 84.3 and 94.5 %, respectively.
143
(a) Heat release rate verses time and (b) smoke release verses time. Reprint with permission from [Xiaoya Jiang], [Green preparation of expandable graphite and its application in flame-resistance polymer elastomer], published by [Industrial & Engineering Chemistry Research], [2017].
41
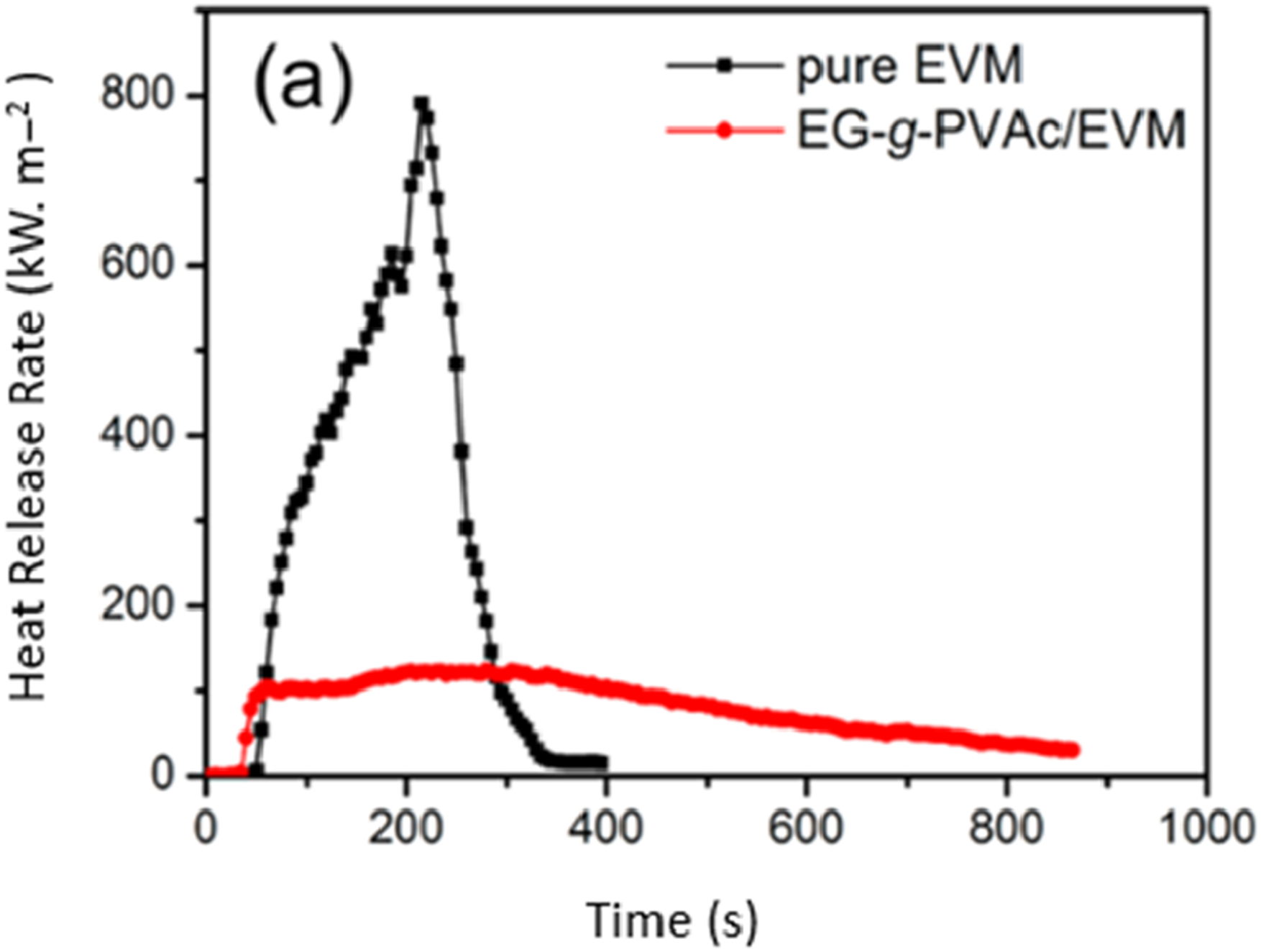
Applications of expanded graphite in wearable electrodes
Expanded graphite has emerged as a prominent material in wearable electrodes. A study reports a smart textile gas sensor created by e-jet printing ionic-liquid-functionalized Cu3(HHTP)2 MOFs (metal organic framework) onto electrospun PLA fibers for efficient nitric oxide detection. Functionalization with EMIM-Otf dramatically improved performance, increasing conductivity by 582× and boosting NO response from <5% to ∼570% at 100 ppm. The sensor detected NO across 5–300 ppm with a low theoretical detection limit of 3.7 ppm, while maintaining partial reversibility and stability under humidity. This study is parallel to the use of EGs, which offer high conductivity, tunable surface chemistry, and excellent integration with flexible substrates, the Cu3(HHTP)2 IL system leverages nanoscale structural control to achieve exceptional sensitivity and stability. Both approaches highlight how functional nanomaterials, whether MOFs or EGs, can be precisely deposited onto textile fibers through techniques such as e-jet printing, enabling lightweight, flexible, and highly responsive sensing platforms. Thus, the strategies used here align with broader EG driven trends in smart textile design, emphasizing material engineering, surface functionalization, and substrate compatibility to achieve advanced wearable sensing capabilities. 144
Expanded graphite (EG) continues to gain prominence in wearable electronics due to its scalable conductivity, low percolation threshold, and stable piezoresistive behavior when dispersed in elastomers for strain sensing and motion tracking (e.g., ENR, PDMS, Ecoflex). 145 In a recent research work, a highly stretchable strain sensor was developed by creating a continuous conductive graphene network with an exceptionally low percolation threshold. With the help of a binary rubber blend, the material formed a double-interconnected graphene pathway that enabled conductivity at just 0.3 vol%, far lower than traditional graphene composites. Near this threshold, the sensor withstood over 100% strain while maintaining strong signal sensitivity (gauge factor ≈82.5) and stable performance for around 300 cycles. This design strategy offers a practical route for producing flexible, reliable, and high-performance strain sensors suitable for wearable applications. 145 EG-based films also function effectively as flexible EMG/ECG electrodes, offering low surface resistivity and good skin conformability compared to conventional gel electrodes. 101 Relative to other fillers, EG’s 2D high-lateral-size morphology provides efficient planar conduction and excellent thermal pathways suited for Joule heating and EMI shielding, 42 whereas 1D carbon nanotubes (CNTs) form highly stretchable, crack-bridging networks that maintain conductivity even at large strains, making them ideal for ultraflexible, high-gauge-factor sensors. 42 MXenes, with their metallic conductivity and surface-rich chemistry, enable multifunctional sensing (strain, pressure, biochemical detection) but face environmental-stability limitations, particularly under humidity and oxidation unless encapsulated or hybridized. 145 In terms of durability, CNTs generally exhibit the strongest cyclic performance due to fiber entanglement and resilience, while EG provides stable 2D pathways when interfacial adhesion and dispersion are optimized, and MXenes require protective strategies to maintain long-term performance. 146 From a processing and cost perspective, EG is the most scalable and economical option, CNTs remain costlier with dispersion challenges, and MXenes, though promising, still require improvements in synthesis and stability. Overall, EG is preferred for robust electrothermal devices and cost-effective EMI shielding, CNTs or EG–CNT hybrids for highly stretchable strain sensors, 101 and MXene-based systems for high-sensitivity multimodal sensing when adequately stabilized.
Applications of expanded graphite in oil-sensing elastomers
Expanded graphite displays many outstanding properties including high and low-temperature resistance, corrosion resistance and self-lubricating capability without disturbing the microscopic molecular structure of graphite materials. It is quite light in weight, and has abundant pores as well as a soft structure which leads to some excellent properties including adsorption, compression resilience and sealing. Nowadays, it is quite common in various fields such as in environmental protection, electrical power, chemical catalysis, machinery, the military and other fields. The outstanding adsorption capability of expandable graphite is already well-known around the world. 147
In the recent research work, thin film elastomers (based on 10 w/w% content of expanded graphite) for particular sensor applications were manufactured. The formed materials were found to be oil-sensing materials in different modes. Such kinds of sensors work, when exposed to the oil and can be easily noted through examination of the changes that happen in the electrical resistance (R) related to the composites. This process can also include both geometrical variations as well as alternation in different exhibited properties of the materials which also involve specific electrical conductivity (resistivity). An innovative technique was applied to increase the response rate of the sensors which includes the utilization of stretched sensing films. A lightly stretched film having 4 % extra length than its original form displayed about 12.5 times quicker response in the oil absorption when compared with an un-stretched film. Figure 19 displays the results.
24
(a–c): SEM images of a coating film (a) and SEM micrographs of composite structures filled with 10 wt.% of expanded graphite (b) and the dependence of the normalized electrical current (I/I0) on time during oil absorption for both the samples in un-stretched and in stretched form (SIS/EG = 90/10 w/w). Reprint with permission from (c) [Krupa Igor], [Electrically conductive composites based on an elastomeric matrix filled with expanded graphite as a potential oil sensing material], published by [Smart materials and structures], [2014].
24

Applications of expanded graphite in dielectric elastomers
Usually, a systematic evolution from micro-composites to nanocomposites imparts the considerable significance of graphene nanocomposites. Several synthesis approaches for such composites involve latex compounding, melt intercalation, solution blending, as well as polymerization. Furthermore, it also provides information about how the properties related to the elastomeric composite vary when the used filler is changed which can be either GNP or EG. Both graphene and GNPs act as a multifunctional reinforcement material which usually utilized to enhance the dielectric properties as well as surface area of elastomers that makes them a outstanding filler. 20 According to additional research work, the method to form silicone elastomers incorporated with EG for the actuation applications is quite significant, as improper EG incorporation can cause problems like short-circuiting in the actuator. On the other hand, when the properly incorporated EG reacts along with the silicone matrix it minimizes the elastic modulus in some thin films having 200 µm width while the thick films having about 1 mm width showed no considerable variations. Furthermore, the mixing technique also plays an important role, the mixing done by the Speed Mixer displayed better electromechanical properties when compared with the simple mechanical mixing technique.
The process of dielectric relaxation in the fluoro-elastomer vulcanizates usually incorporated with exfoliated nano-graphite has been considered a work of enhancement in loading of both filler as well as blowing agent within 0.01–105 Hz frequency range. The enhancement in the loading of filler has a direct relation with the dielectric loss tangent which was confirmed according to the visco-elastic nature of the composites. Usually, the continuous changes that occur in the complex fragment of impedance having applied frequency displayed quite a distinct peak whose presence depended upon the concentration of filler used in the composite. Additionally, the enhancement in the loading of the filler also causes an increase within the non-semicircle nature of the Nyquist plots. The whole process was evaluated through a resistance-capacitance circuit model. The filler percolation limit within the composite has been calculated via different measurements of electrical conductivity which is displayed in Figure 20. The percolation limit has been confirmed to be happening between the loading of about 2.5 and 3.5 phr.
128
The generalized functional properties of expanded graphite and their role in elastomers have been discussed in Table 5. Effect of conductive filler incorporation into elastomeric structures conductivity at various frequencies. Reprint with permission from [Azam Ali], [Development of silver plated electrically conductive elastomers embedded with carbon black particles obtained from Kevlar waste source], published by [ELSEVIER], [2020].
128
Applications and functional roles of expanded graphite (EG) in elastomer composites.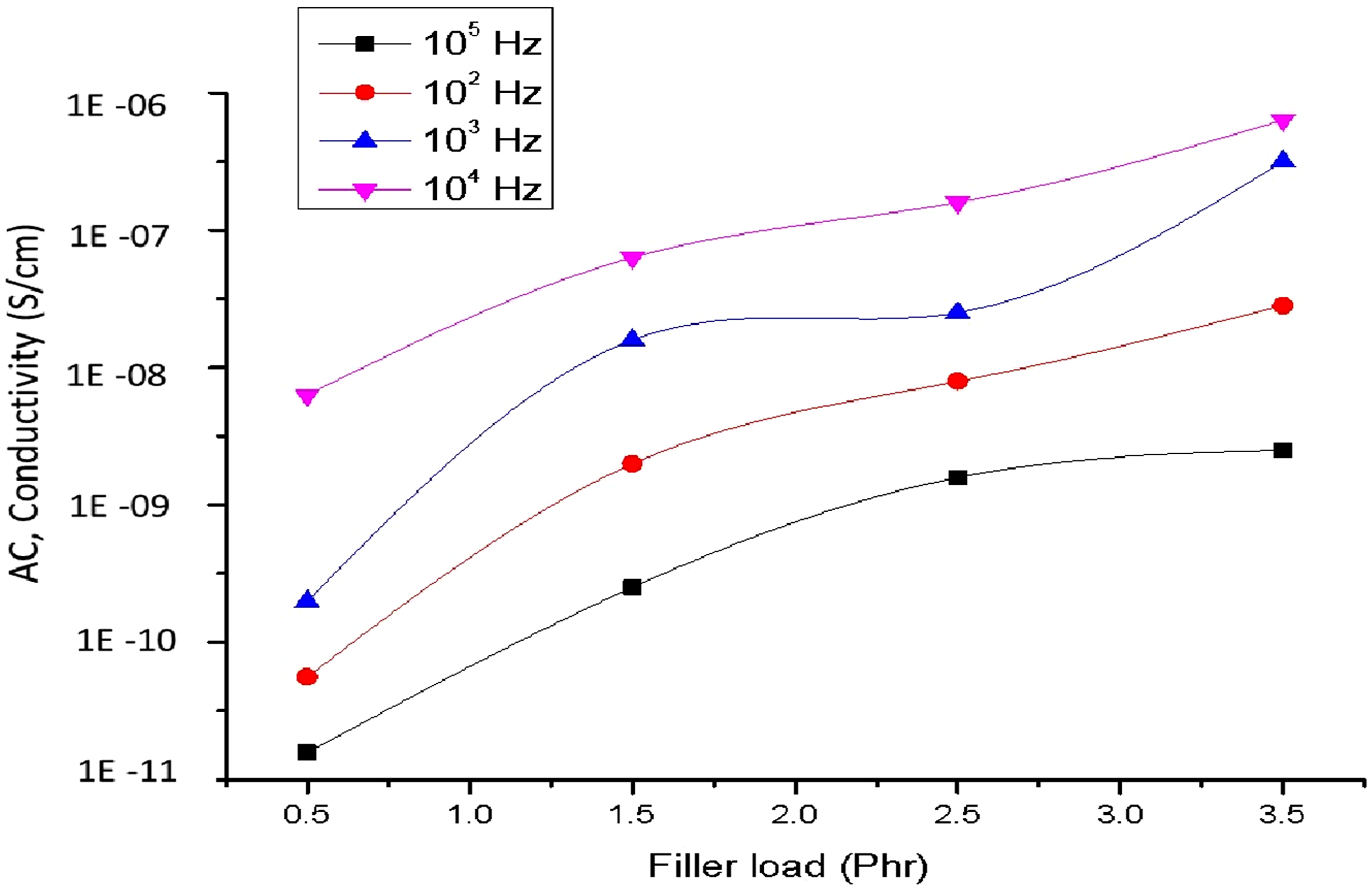
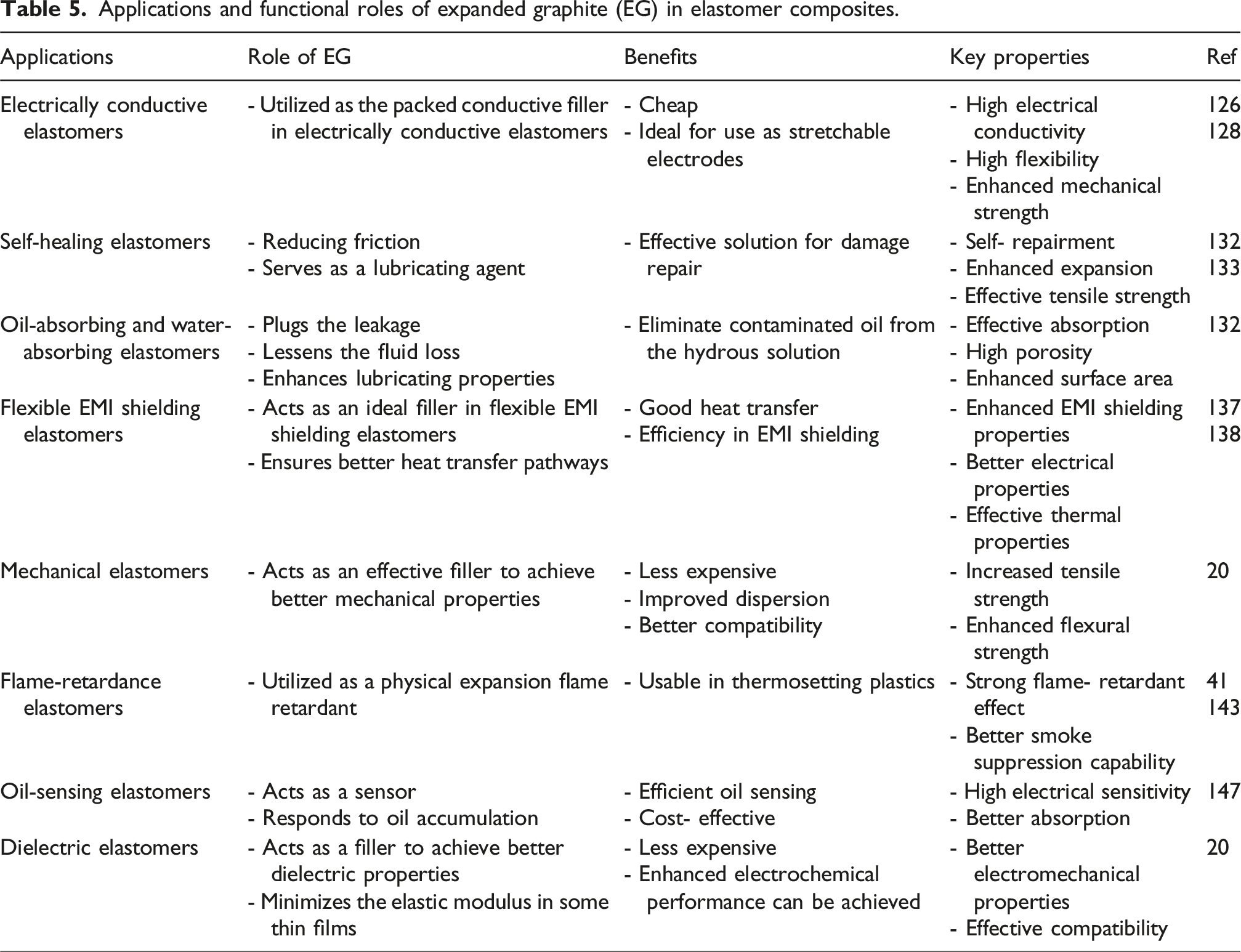
Durability performance of EG-based elastomers
Durability under cyclic mechanical, electrothermal, and environmental loading is a critical benchmark for evaluating the long-term reliability of expanded graphite (EG) based elastomers. Several studies demonstrate that EG-filled elastomers maintain highly stable conductive pathways during repeated deformation. It is due to its turbostratic capability, multilayer structure and strain capacity, through which reversible layer slide rather than the tunnelling dominated failure seen in CNT or graphene nanoplatelet systems. 148 For example, natural rubber and EG strain sensors preserved over 90% of their initial electrical conductivity after 5000 cyclic tensile cycles at 50% strain, indicating excellent fatigue resistance in dynamic applications. 129 Hybrid EG and CNT elastomers further enhance durability, showing <5% resistance drift after 3000 cycles, significantly outperforming composites formed by only CNT, which typically exhibit 20–40% drift due to tunneling instability. 148 Under electrothermal cycling, expanded graphite films demonstrate stable Joule heating output and show minimal resistance change over repeated heating cooling cycles because the sp2-rich graphitic domains resist oxidation and maintain contact integrity, Hou et al. (2020) reported negligible degradation even after 100 electrothermal cycles. 10 Environmental humidity also impacts composite stability; however, low-oxygen or hydrophobically modified EG composites show less than 5% conductivity variation across 30–90% RH, contrasting with MXene or CNT elastomers, which often experience 20–60% humidity-induced resistance drift due to water adsorption. 113 These findings suggest that EG based elastomers exhibit superior mechanical fatigue tolerance, electrothermal stability, and humidity robustness, making them promising candidates for wearable electronics, soft robotics, and long-term flexible device engineering.149,150
List of Abbreviations
Expanded graphite (EG), Expandable graphite (EGp), Graphite intercalation compounds (GICs), Nano-expanded graphite (nEG), Chemical vapor deposition (CVD), Natural graphite (NG), Natural flake graphite (NFG), Flake graphite (FG), Graphene oxide (GO), Expansion volume (EV), Fourier transform infrared spectroscopy (FTIR), Scanning Electron Microscopy (SEM), Slight expanded graphite (SEG), Thermally expanded graphite (TEG), Metal oxide (MO), Chemical intercalation (CI), Electrochemical intercalation (EI), Kish-based expanded graphite (KEG), Poly-3,4-ethylenedioxythiophene (PEDOT), Hexagonal graphite (HG), X-ray photoelectron spectroscopy (XPS), Transmission Electron Microscopy (TEM), Ultraviolet visible near infrared (UV vis-NIR) spectroscopy, Hydrothermal formed EG (HEG), Ultrasound irradiation formed EG (UEG), Conventional liquid phase synthesis (CEG), Natural Rubber (NR), Tubular Conductor (TC), Vibrating Sample Magnetometer (VSM), Energy Dispersive X-ray (EDS), Micro wrinkled reduced graphene oxide (MWrGO), Electromagnetic interference (EMI), Thermoplastic elastomers (TPEs), Polydimethylsiloxane (PDMS), Latex compounding method (LCM), Graphite nanoplatelets (GNP), Ethylene vinyl acetate copolymer (EVM), Limited oxygen index (LOI), Polyvinyl acetate (PVAc), Heat release rate (HRR), Total smoke release (TSR), Zeolitic imidazolate framework-8 (ZIF-8) and Polyurethane elastomer (PUE).
Conclusion
Expanded graphite (EG) has emerged as a highly versatile filler for flexible elastomers, offering a unique combination of high aspect ratio, hierarchical worm-like porosity, and tuneable interlayer spacing. These intrinsic structural characteristics enable EG to form long-range, deformable conductive networks at low loadings, enhancing electrical conductivity, thermal transport, and mechanical reinforcement while preserving matrix compliance. Various physical and chemical methods of EG preparation such as the hydrothermal method, chemical intercalation, mechanical milling, etc., have been described in this study. The direct interplay between structure, properties, and applications of expanded graphite is clearly evident. Parameters such as lateral flake size, expansion volume, and pore connectivity govern percolation thresholds and strain-tolerant electrical conduction, while surface chemistry controls interfacial adhesion and stress transfer. Additionally, defect density provides a balance between conductivity and mechanical flexibility. This structure–property relationship has been successfully leveraged in a range of applications, including stretchable electrodes, conformal EMI shielding, dielectric actuators, oil-sensing films, and multifunctional self-healing or strain-sensing devices. Despite these advantages, critical bottlenecks hinder broader adoption. EG dispersion and exfoliation remain inconsistent, particularly in viscous elastomer matrices, which leads to variability in conductive network formation. Interfacial adhesion is often inadequately engineered or quantified, limiting mechanical durability. Additionally, synthesis methods yield EG with widely varying expansion ratios, surface areas, and defect levels, yet studies frequently omit standardized characterization, impeding cross-comparison. Long-term stability under cyclic mechanical and electrothermal loading, as well as environmental exposure, is insufficiently reported, restricting reliability assessments for wearable and industrial applications. Furthermore, many EG synthesis routes rely on hazardous reagents or energy-intensive processes, raising concerns about scalability and environmental footprint. Future development should focus on establishing standardized EG property descriptors (e.g., expansion volume, specific surface area, interlayer spacing, Raman ID/IG ratio, in-plane conductivity), optimizing dispersion through advanced compounding techniques, and applying targeted surface functionalization to strengthen filler–matrix interfaces. Incorporating hybrid fillers and designing hierarchical networks can further enhance multifunctionality. Finally, rigorous durability testing under realistic operational conditions and adoption of greener, scalable synthesis routes will be essential. By strategically aligning EG’s structural attributes with desired properties and end-use requirements, it is possible to unlock its full potential in next-generation flexible elastomer technologies.
Future aspects
In future, the progress in EG based flexible elastomers will depend on focusing key limitations while enabling broader, real-world use. A major priority is the development of sustainable, low-cost, and scalable EG synthesis routes by replacing hazardous oxidants and intercalants with greener alternatives. The improvement of Icompatibility between EG and elastomer matrices is equally crucial; targeted interface engineering and surface functionalization can greatly enhance dispersion, interfacial adhesion, and long-term durability. Hybrid filler systems, where EG is combined with MXenes, graphene, or carbon nanotubes can offer exciting opportunities to achieve stronger electrical, thermal, and mechanical performance. At the same time, advanced methods of coating including electrospinning as well as 3D printing, can offer high chances for designing complex, hierarchical structures as well as anisotropic for the purpose of better functional properties of the elastomers 131 and smart wearable textiles 144 in various applications. To ensure the long-term reliability still remains an essential challenge. Comprehensive testing of fatigue resistance, electrothermal stability, and environmental durability is needed to support safe and stable operation in everyday conditions. The Incorporation of smart features such as self-healing, stimuli responsiveness, or shape-memory behavior may further extend the technology’s reach, particularly in biomedical and flexible electronic devices. Finally, researchers must evaluate environmental safety, biocompatibility, and toxicity to enable confident transition of EG-based elastomers into biomedical and commercial applications. Addressing these priorities will accelerate their journey toward practical, next-generation materials.
Footnotes
Funding
This work was supported by the Technology Agency of the Czech Republic (TAČR) under the project “Multifunctional textiles using the heat radiated from the human body” (Project No. FW12010015).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting this study are available from the corresponding author upon reasonable request.
