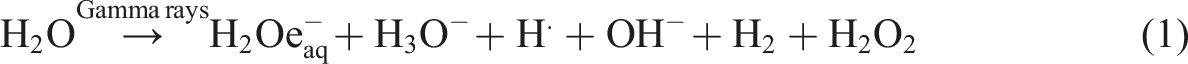Abstract
The synthesis of Poly(PVA/St/Ag NPs) nanocomposite by using of gamma radiation was carried out. The progress of the reaction was examined by using different techniques such as Fourier transform infrared (FTIR), transmission electron microscopy (TEM), UV, XRD, and scanning electron microscopy (SEM). The FTIR show the successful preparation of the Poly(PVA/St/Ag NPs) nanocomposite by gamma radiation at a dose of 5 kGy. The TEM analysis displays the particle size distribution of Ag NPs and it is observed that the Ag NPs size was in the range of 21-30 nm. The SEM images show a good distribution of silver nanoparticles in the Poly(PVA/St/Ag NPs) nanocomposite. matrix, but with little agglomerations or aggregates observed on the surface of the Poly(PVA/St/Ag NPs) nanocomposite. The XRD analysis indicates that amorphous regions are enhanced in the Poly(PVA/St/Ag NPs) nanocomposite. The coating of Citrus fruits by of Poly(PVA/St/Ag NPs) nanocomposite showed better performance in inhibition of the growth of P. digitatum on citrus fruits than free Ag NPs at concentration of 30 ppm. The incorporation of Ag NPs in Poly(PVA/St/Ag NPs) nanocomposite has extremely obvious antifungal activities against P. digitatum due to the nanometer range of Ag NPs that can interact with P. digitatum surface and/or its core where it enters inside the cell, as a result, cellular metabolism is inhibited causing death of P. digitatum and subsequently exhibits antifungal activities.
Keywords
Introduction
According to the U. N. statistics, about 50% of vegetables and fruits are lost each year. 1 In industries, coatings are being used to try and prevent postharvest loss. Wax coating is considered as the most popular method for preserving fruits and vegetables. On occasion, wax is used with chemicals that are known to cause cancer. Lately, numerous edible films that use natural polymers have been developed to significantly improve food’s shelf life, which serve as a barrier between the environment and the food to inhibit direct contact between the food, air gases, and bacteria. This lowers the rate of respiration, which keeps the food fresh for a longer period. Yet, edible biofilms are expensive and only available on an industrial scale; local fruit and vegetable dealers are unable to purchase such expensive biofilms. Due to the development of nanotechnology in the food and agricultural industries, there are now many chances to preserve and package food without harming the environment or human health.1,2
Different types of edible biopolymers 3 and semi-synthetic polymers 4 are available for food packaging, 5 such as Arabic gums, starch, chitosan, pectin, corn, alginate, cellulose, and its derivatives…etc. 6 With the involvement of nanotechnology and various techniques like dip coating, spray coating, and electrospun nano-fiber coating, thin edible coatings are applied to perishable fruits and vegetables. 7 Coatings have been applied to perishable fruits and vegetables by using several edible thin films made from biopolymer composites, and dip-coating techniques.
Banana fruit coated with a carrageenan composite (blend with carboxymethyl cellulose) had a 6-day increase in shelf life over the control; 8 on the other hand, banana ripening was slowed for 12 days by a rice starch edible coating mixed with sucrose esters while kept at 20 ± 2°C compared to the untreated control. To increase the shelf-life of banana fruit from nine to 19 days, gamma-irradiated plasticized PVA, CMC, and tannin composites were used. 9
A common food coating and packaging material is Starch may be obtained from a variety of plant sources, which is inexpensive, and degrades naturally. Starch films have poor water vapor barrier qualities, and are highly water sensitive due to their hydrophilic nature. 10 Starch is frequently combined with other biopolymers to create coatings and films with improved characteristics to overcome this challenge.11,12 To achieve this, many studies to PVA/St blend films have been conducted.13,14
PVA is a synthetic hydrophilic polymer that is nontoxic, biocompatible, and biodegradable, making it an environmentally acceptable material.15,16 PVA is one of the most recommended synthetic plastics used for promoting the properties of other biopolymers to gain high strength and film-forming capability. 17 PVA is frequently used as a cold and hot water-soluble film for a variety of packaging or coating applications, including the packaging of food goods, detergents, pharmaceuticals, and agricultural chemicals. 18 PVA has also received FDA approval for close contact with food products. PVA has recently been requested by the FDA to be approved for use in a water-soluble edible film comprising dry food ingredients (GRAS Notice no. 676, 2018). PVA films are transparent, have a high tensile strength, and are flexible in addition to having good oxygen and odour barrier qualities. According to several scientists, starch-PVA blended films had a variety of advantages over pure starch films because they formed interpenetrated polymer networks that improved the composite films’ mechanical and water barrier qualities. 2
Moreover, for enhancing antimicrobial activities and thermal properties the presence of silver nanoparticles (Ag-NPs), is extremely useful for food processing and packaging.19,20
Citrus (Citrus spp.) is one of the most important fruit crops in the Mediterranean basin. Egypt is one of the major citrus producing countries in the world. In Egypt, The area of citrus crop cultivation increased rapidly with the reclamation of new lands in the desert. The citrus crop in Egypt is now about 344,789 feddans distributed in all governorates and produced about 2,594,853 tons. 21 Postharvest disease causes serious losses to the fruits during transit and storage. Citrus fruits are produced in subtropical regions distant from consumer markets and often must be stored for the market economy as dictates. A months-long delay between harvest and consumption may result in postharvest losses due to pathological and physiological diseases. 22
Green mould (P. digitatum Sacc.) is one of the predominant pathogens on citrus fruits worldwide. 23 Essential oils from different species of the genus Cymbopogon are known for their antimicrobial activity.24,25 Also citral solutions at the concentration of 15,000 μL/L citral in 400 μL/L. triton x emulsifier completely inhibits the growth of P. digitatum, Penicilluim italicum and Geotrichum candidum, Postharvest spoilage fungi of citrus.
Consequently, in this work, the major objective is to use of Poly(PVA/St/Ag NPs) nanocomposite coatings incorporating silver nanoparticles were applied as an antimicrobial edible coat for Citrus fruits. The antifungal efficacy of these coatings is applied as a curative treatment against Penicillium digitatum.
Experimental
Materials and methods
Materials
Polyvinyl alcohol (PVA) with molecular weight 115,000 (C2H4O)n provided by Loba Chemie, India. Starch (St) soluble, purchased from Qualikems Fine Chem Pvt. Ltd, India. Silver nitrate (AgNO3) with minimum assay 99% (EMPLURA) purchased from Merck Specialties Private Limited, Mumbai, India.
Preparation of Poly (PVA/St/Ag NPs) nanocomposite
The antimicrobial of Poly(PVA/St/Ag NPs) nanocomposite was prepared via gamma radiation-induced in Situ polymerization. Firstly, Poly(PVA/St/Ag NPs) nanocomposite at the ratio (2:1) was prepared by dissolving 2 wt % of starch (0.66 g) in 96 mL bi-distilled water at 70°C via stirring using a magnetic stirrer. After the complete dissolution of starch, a weight of PVA (1.34 g) was added to the solution, then raising the temperature to 90°C via continuous stirring until complete dissolution. Finally, 0.1 M of silver nitrate was added. The produced solution was exposed to gamma radiation at a gamma irradiation dose of 5 kGy and the irradiation dose was 0.66 kGy/h. After irradiation, a detected volume from the gel solution was poured into a plastic petri dish to get a film to be used in SEM, EDX, FTIR, and XRD measurements.
In the synthesis of the Poly(PVA/St/Ag NPs) nanocomposite the solvent was an aqueous solution which when exposed to Gamma rays created hydrated electrons and primary radicals and molecules
26
as shown in equation (1):
The solvated electrons H2Oe–aq and H. atoms are strong reducing agents so that in the following step they easily reduce silver ions down to the zero-valent state as shown in equations (2) and (3):
Finally the produced mixed Ag NPs were incorporated within the Poly(PVA/St) blend to form Poly(PVA/St/Ag NPs) nanocomposite as follows:
Characterizations of poly (PVA/St/Ag NPs) nanocomposite
Scanning electron microscopy
The surface topography and structural features of the prepared Poly(PVA/St/Ag NPs) nanocomposite films were examined using scanning electron microscopy (SEM) (ZEISS-EVO 15-UK). EDX was utilized to identify and investigate the elemental composition. EDX attached to scanning electron microscopy (SEM) (ZEISS-EVO 15-UK).
Transmission electron microscope
To display composite particle size distribution and geometry High-resolution Transmission Electron Microscope (HRTEM) model (JEOL/JME-2100, Japan) was employed and operated at 200 kV for Poly(PVA/St/Ag NPs) nanocomposite. To image the Poly(PVA/St/Ag NPs) nanocomposite on HRTEM, the sample solution was sonicated then approximately 10–20 μL of this solution was dropped on a 3 mm copper grid, drying at room temperature. The copper grid was inserted into High-resolution transmission electron microscope.
UV-Vis spectrophotometry
The prepared Poly(PVA/St/Ag NPs) nanocomposite is characterized by measuring optical absorbance (A) in the wavelength range 200–1000 nm using double beam spectrophotometer model JASCO 670 UV–Vis–NIR. For this measurement, the sample solution of the Poly(PVA/St/Ag NPs) nanocomposite was diluted by using bi-distilled water (0.1 mL of sample/1 mL of bi-distilled water) to be used for the measurement of the absorption spectra.
FTIR analysis
FTIR Model Cary 630 FTIR spectrometer was used to record the FTIR analysis of original PVA, St, and a casted films of the Poly(PVA/St/Ag NPs) nanocomposite in the wavelength range 4000–400.
XRD analysis
XRD of different samples PVA, Starch, Silver nitrate, and PVA/St/Ag NPs film) were performed according to XRD spectrophotometer, Model (X’pert Pro), Manufacture (PAN analytical) equipped with X-ray tube [Cu target, 40 KV (Voltage), 30 (mA) (current)], the X-ray data were recorded in a range from 4 to 80 (degree) 2θ with continuous scanning mode and scanning speed 8 (deg./min).
Fungal species
Pencillium digitatum Sacc. was isolated from orange fruit (Citrus Sinesis L.) showing green rot collected from local markets of Sharkia governorate. The collected samples were carefully kept in clean and sterile plastic bags at room temperature. For isolation, samples were introduced and carefully washed with running tap water, dried between filter papers, and surface sterilized with 1% sodium hypochlorite for 2 min then the samples were washed thoroughly with sterilized distilled water several times and dried between sterilized filter paper fungal isolation was carried out from the inner tissues neighboring the infected ones. Segments were separately transferred to Petri-dishes containing potato dextrose agar (PDA) media and incubated for 7 days at 27°C (Waller). 27 The fungus was identified according to Raper and Thom. 28
Pathogenicity test
The isolated fungal strain was investigated for its pathogenicity to healthy orange fruits (Citrus Sinesis L.) healthy mature fruits were carefully washed, rinsed with sterile water, and dried between sterilized filter paper. Surface disinfested was carried out by soaking in 2% sodium hypochlorite solution for 2 min and then in 70% ethylalcohol for 1 min rewashed and dried and the inoculation was carried out according to the method described by Palou, 29 and Plotto. 30 The severity of infection was calculated as a percent of the external rotted area of the infected fruits in proportion to the total area of the same fruit Morcos. 31
Culture media
Media used were prepared according to Gams. 32 The fungus was isolated and maintained on potato-dextrose agar (PDA) medium and identified on Czapek’s medium. Growth inhibition, other physiological studies and samples for microscopical studies were carried out on Czapek’s broth medium both with and without the essential oil. PDA consists of potato extract, 230 mL; glucose, 20g; water, 770 mL. Potato extract was prepared by adding 100g potatoes (Peeled and sliced in a mincer) to 300 mL tap water; it was left overnight at 4°C and filtered through cloth.
In vitro effect of Ag NPs on P. digitatum
Different concentrations of Silver Nanoparticles were added individually to 250 mL Erlenmeyer flasks each contain sterilized Czapek’s medium to obtain the following concentration 00, 10, 20, and 30 ppm Then plates from each concentration were prepared. The plates were inoculated singly with disks (3 mm diam.) of fungal growth taken from 7 days old culture of P. digitatum. Three replicates were prepared from each concentration of both treatments and linear growth of the fungus was measured when the control plates reached full growth at 27 ± 2°C.
In vivo effect of Ag NPs on P. digitatum
Healthy orange fruits were sterilized through immersion in 70% ethanol for 1 min left to dry at room temperature and inoculated separately by disks (3 mm diam.) of P. digitatum through a small scratch in the middle surface of fruits. The fruits were dipped in the Poly(PVA/St/Ag NPs) nanocomposite solution, containing Ag NPs with concentrations of 0, 10, 20 and 30 ppm. The treated fruits were packaged in plastic net bags, put in perforated sterilized carton boxes, and stored for 4 weeks at room temperature for each treatment three replicates were used, each replicate containing 10 fruits.
Scanning electron microscopy
Mycelia of P. digitatum grown in PD broth medium fumigated with 3 μL/mL C. citratus essential oil and that from non-fumigated (control) were fixed in 2.5% glutaraldehyde at 4°C for 24 h and post-fixed in 1.0% osmium tetraoxide for 1 hr at room temperature Harley and Fergusen. 33 The specimens were then dehydrated with ascending concentrations of acetone, critical point dried, and finally sputter coated with gold. The examination and photographing were done through Joel scanning electron microscope (JSM – 1200 EX).
Result and discussion
Scanning electron microscopy (SEM) of Poly(PVA/St/Ag NPs) nanocomposite coated film
Figure 1 shows the surface morphology of prepared Poly (PVA/St/Ag NPs) nanocomposite film which loaded by silver nanoparticles. The Figure 1 reveals porous surface morphology. The pores noticed on the surface acquired an irregular shape with a large diameter. Also, the image displays a good distribution of silver nanoparticles, but some agglomerations or aggregates are observed on the surface.
34
shows SEM image for of Poly(PVA/St/Ag NPs) nanocomposite film at cross-section mode.
EDX spectroscopy of poly (PVA/St/Ag NPs) nanocomposite coated film
Energy dispersive spectroscopy (EDS), which analyses the energy and intensity distribution of X-ray signals produced by the electron beam striking the surface of the specimen, is used to perform an elemental analysis of the sample surface of Poly(PVA/St/Ag NPs) nanocomposite film. The EDX analysis confirms the presence of Ag NPs within the matrix of Poly(PVA/St/Ag NPs) nanocomposite as shown in Fig. (2) which presents around 3.0 keV that corresponds to the binding energy of Ag lines.
35
Also Figure 2(a) illustrates the good distribution of silver nanoparticles on the surface of Poly(PVA/St/Ag NPs) nanocomposite coated film. However, we can observe that some agglomerations on the surface.
36
(a) surface morphology scanning of Poly(PVA/St/Ag NPs) nanocomposite, (b) EDS (Energy Dispersive Spectrometer) element analysis of Poly(PVA/St/Ag NPs) nanocomposite.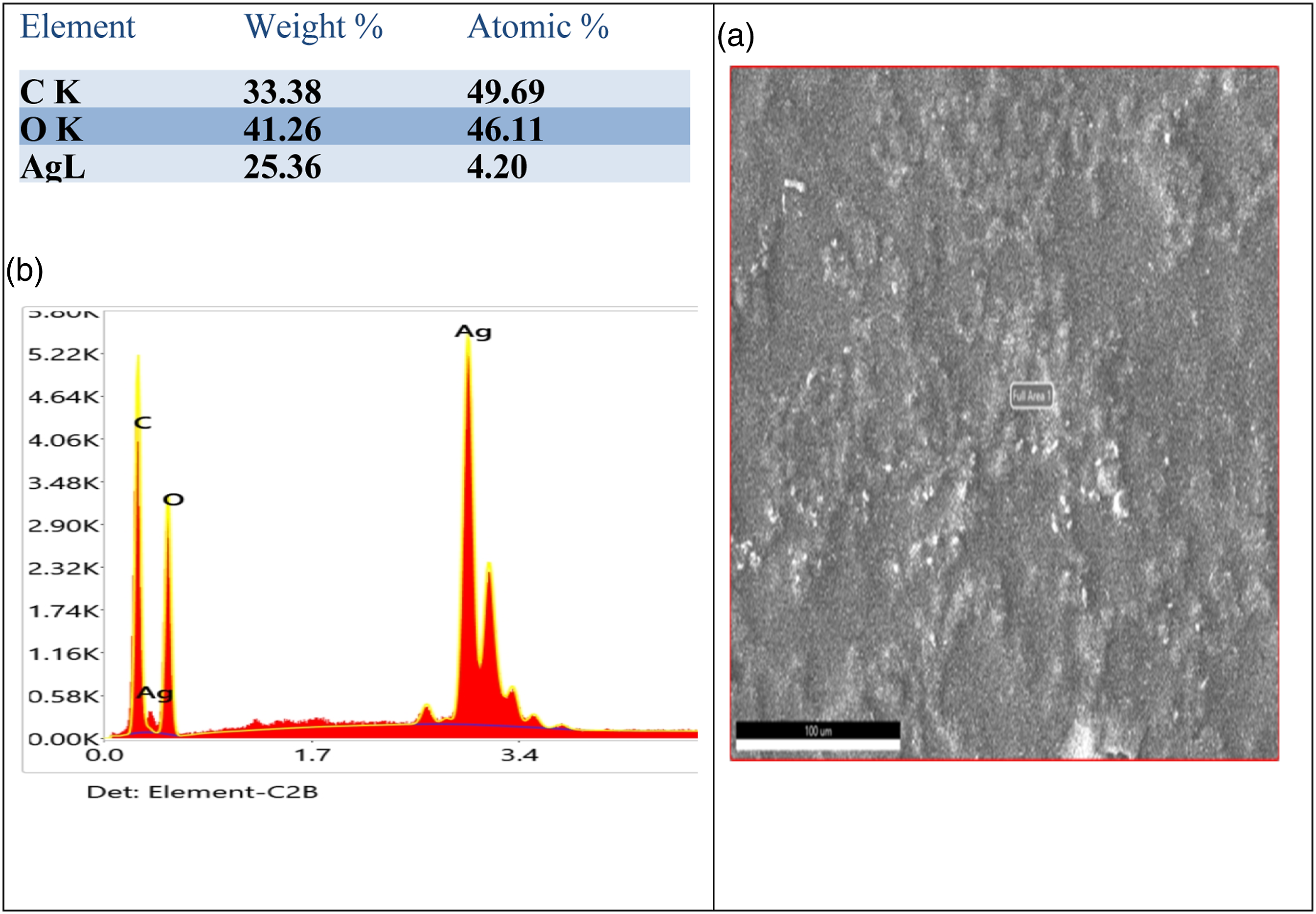
UV-Vis spectrophotometry
The fact that the silver salts incorporated in the Poly(PVA/St/Ag NPs) nanocomposite are easily reduced by gamma radiation and instantly change color to a dark brown indicates that the synthesis of silver nanoparticles in the whole of the Poly(PVA/St/Ag NPs) nanocomposite network can be predicted. The growth of silver nanoparticles within the crosslinked network was evidenced by the nano-gel solution’s dark brownish-black color.
Strong localization and stabilization allowed the decreased particles to become restricted inside the network of the Poly(PVA/St/Ag NPs) nanocomposite. In addition, linear polymeric chains help stabilize particles by adhering to their surface and forming bonds with the carbonyl and hydroxyl functional groups of (PVA/starch) nano-gels. UV-vis spectral spectroscopy is used to verify whether silver nanoparticles are successfully formed in the gel network. 37
Figure 3 manifests the UV–vis spectra of silver nanoparticles via in situ polymerization of PVA/starch nano composite network using gamma radiation. As shown in Figure 3, a distinctive unique UV-Vis absorption peak around 413 nm which due to the characteristic surface Plasmon resonance (SPR) effect of quantum-size silver nanoparticles formed in PVA/St network.37,38 UV–Vis spectra of prepared silver nanoparticles.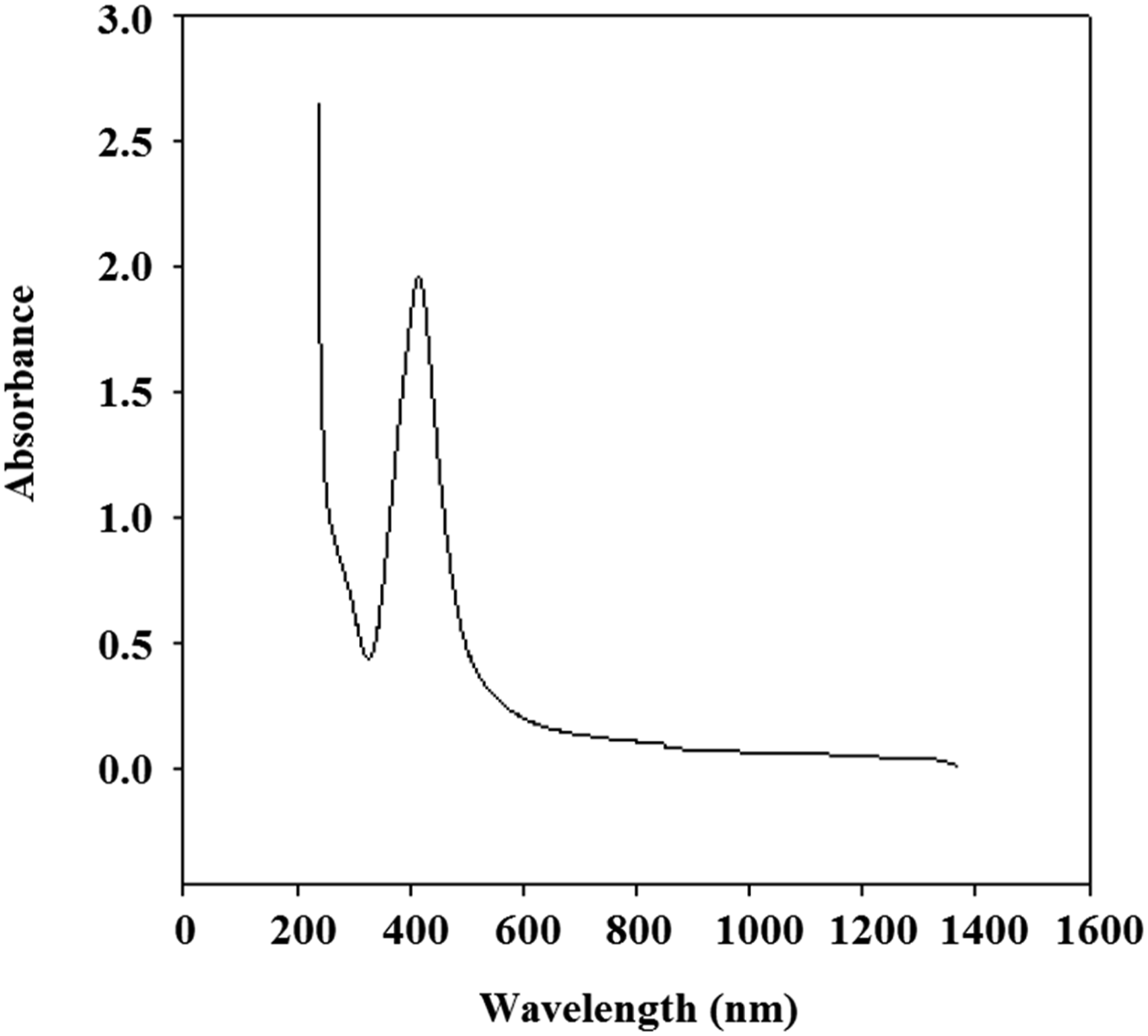
High-resolution transmission electron microscopy (HRTEM)
High-resolution TEM is employed to provide high resolution that can structurally analyze the Poly(PVA/St/Ag NPs) nanocomposite samples at the atomic level. As a result, it is a highly helpful tool for researching any nano-scale structures. In the present work, HRTEM was used to study the size, shape, and diffraction of the silver nanoparticles.
Figure 4(a)–(c) depicts the HRTEM images of the formed silver nanoparticles in Poly(PVA/St/Ag NPs) nanocomposite matrix. The images show the orderly arrangement of the silver nanoparticles, which is an indication of the successful preparation of the Ag NPs in the Poly(PVA/St/Ag NPs) nanocomposite matrix using gamma irradiation. From these images, it is noticed that silver nanoparticles have various shapes including cubic, pyramidal, octahedron, truncated pyramid, and cuboid morphology.
39
HRTEM images of silver nanoparticles (a, b, and c) HRTEM images show different shapes of silver nanoparticles, (d) size distribution of silver nanoparticles, (e), (f) SAED pattern of silver nanoparticles sample.
Figure 4(d) displays the particle size distribution of Ag NPs. It is observed that the Ag NPs size was in the range between 21 and 30 nm. 40 The SAED pattern is illustrated in Figure 4(e) and (f) the characteristic concentric rings that are associated with the crystalline structure of silver nanoparticles are visible as bright spots in the electron diffraction pattern. From the SAED pattern Figure 4(e) and (f), it was found that distances between two bright spots values are 12.271, and 22.591/nm, which are related to (0.16 (nm) d-spacing value, 54.2ᵒ (2ϴ) XRD peak position), (0.09 (nm), and 99.7ᵒ (2ϴ)), respectively.37,38
Fourier transform infrared analysis (FTIR)
For the identification of organic compounds, infrared spectroscopy has commonly been a significant tool. With the introduction of FTIR, a common technique for the analysis of complicated mixtures as well as for the study of surface and interfacial phenomena was established. Finding the chemical functional groups in the sample is the major goal of FTIR investigation.
Figure 5(a) exhibits FTIR of pure PVA. For pure PVA the bands at the range (3253.9 – 3309) cm−1 are apportioned to the stretching vibration of the intramolecular bonded -OH groups of PVA.36,37 At approximately 2927.67 cm−1, an asymmetric stretching vibration band corresponding to the CH2 has been found. The C=O and C=C stretching modes, respectively, have been attributed to the peaks at 1715.57 and 1655 cm−1.
38
FTIR of (a) pure PVA, (b) pure starch and (c) Poly(PVA/St/Ag NPs) nanocomposite prepared via gamma radiation at dose of 5 kGy.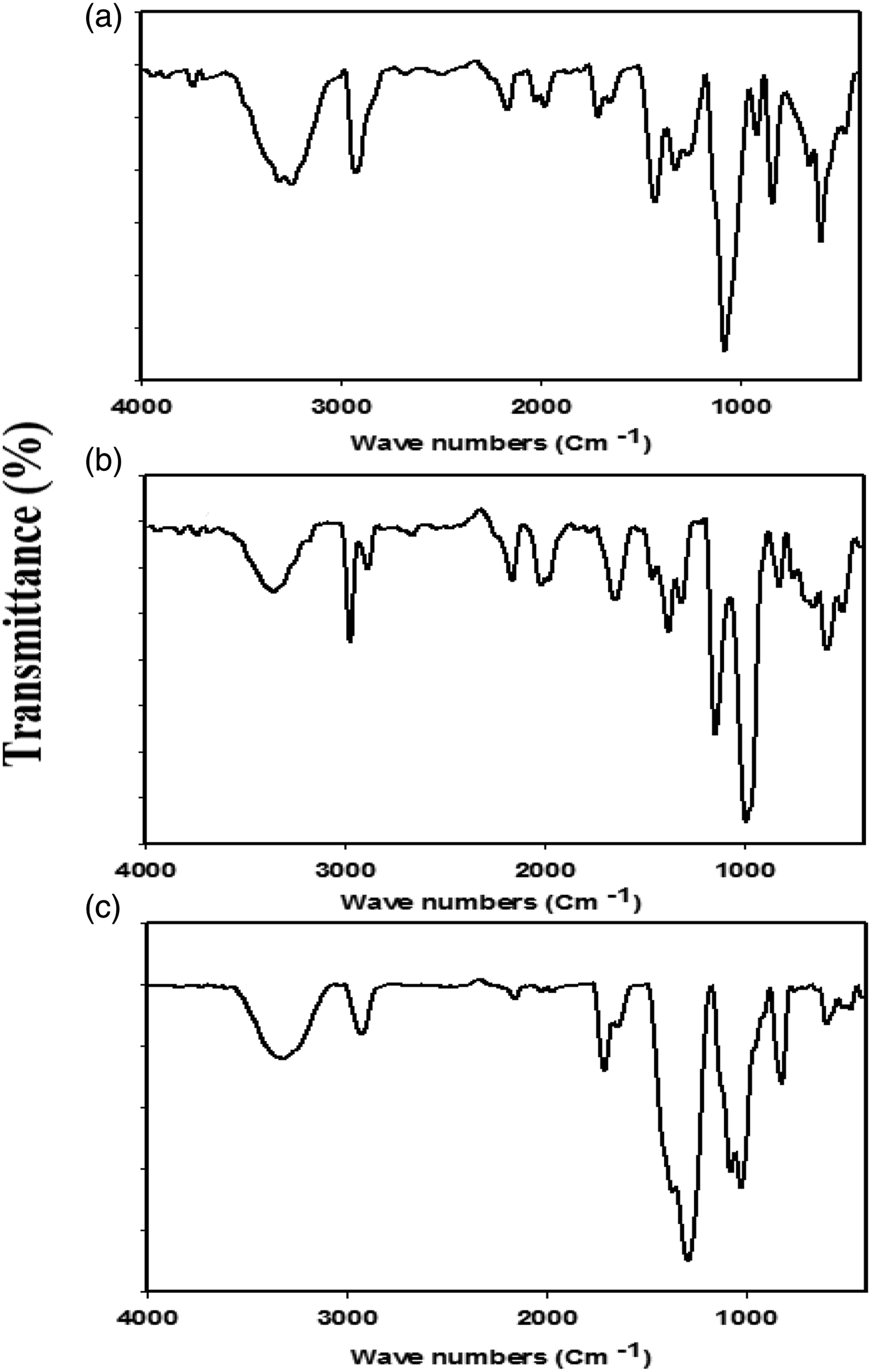
Symmetric bending of CH2 has been identified as the cause of the absorption peak at 1432 cm−1. Moreover, CH wagging is related to the IR peak at 1257 cm−1. The C-O groups found in the PVA backbone are being from C-O stretching, as indicated by the band at about 1082 cm−1. The C-C stretching vibrations of the significant absorption planar zigzag carbon backbone are seen at 835.6 cm−1. The peak at 644 cm−1 corresponds to the (OH) group’s wagging mode, whereas the peaks at 921 cm−1 and 1320.77 cm−1 belong to the (CH+OH) group’s bending mode. 41
Figure 5(b) shows FTIR of pure starch. For pure starch, the presence of absorption band appeared around 3356.7 cm−1 is assigned to -OH groups. The existence of the band around 2887.48 and 2975.35 cm−1 is referred to C-H stretching. The peak centered at 1145.96 is attributed to C-O-C asymmetric stretching. One more, the characteristic C-O-C ring vibration on starch gives rise to an absorbance peak appeared at around 987 cm−1. 42 The O-H bending would raise a band appeared around 1313.29 cm−1. Whereas the appearance of the peak found at 1384.34 cm−1 is suggested to C-H symmetric bending. Furthermore, the band noticed at 1463.79 cm−1 point out CH2 symmetric deformation. In addition, the peaks at 1837.7, 1715.57, and 1655 cm−1 have been ascribed to the C=O and C=C stretching modes respectively. 42
The C-C stretching vibrations of the planar zigzag carbon backbone are appeared around 835.6 cm−1. 43 The uncommon CO2 peaks at 1973.25, 1996.6, 2020, and 2166 cm−1 were observed in PVA and starch IR spectra which might be result from measuring conditions. 44
Figure 5(c) shows the FTIR of the Poly(PVA/St/Ag NPs) nanocomposite. Most of the observed peaks are similar to the peaks found in the spectra of PVA and St Only one new peak appeared at 1034.7 cm−1 for C-O-C stretching vibration. Moreover, the intensity of most of the observed peaks increased after preparing a blend of PVA and starch. Increasing the intensity of the IR peaks is an indication of the successful crosslinking process. Last but not least, a small shift of most of the IR bands of Poly(PVA/St/Ag NPs) nanocomposite was observed which due to the coordination bonds between the OH groups and Ag NPs. 45
X-ray diffraction analysis (XRD)
The X-ray diffraction (XRD) of pure PVA polymer is shown in Figure 6(a). There are two diffraction peaks at 2θ = 19.67° and 39.03°, which indicates that the PVA is semi-crystalline in nature.
46
Also, both peaks are due to the intermolecular interaction of hydrogen bonding for PVA chains.
46
XRD analysis of (a) pure PVA, (b) pure starch and (c) AgNO3, and (d) Poly(PVA/St/Ag NPs) nanocomposite prepared via gamma radiation at irradiation dose of 5 kGy.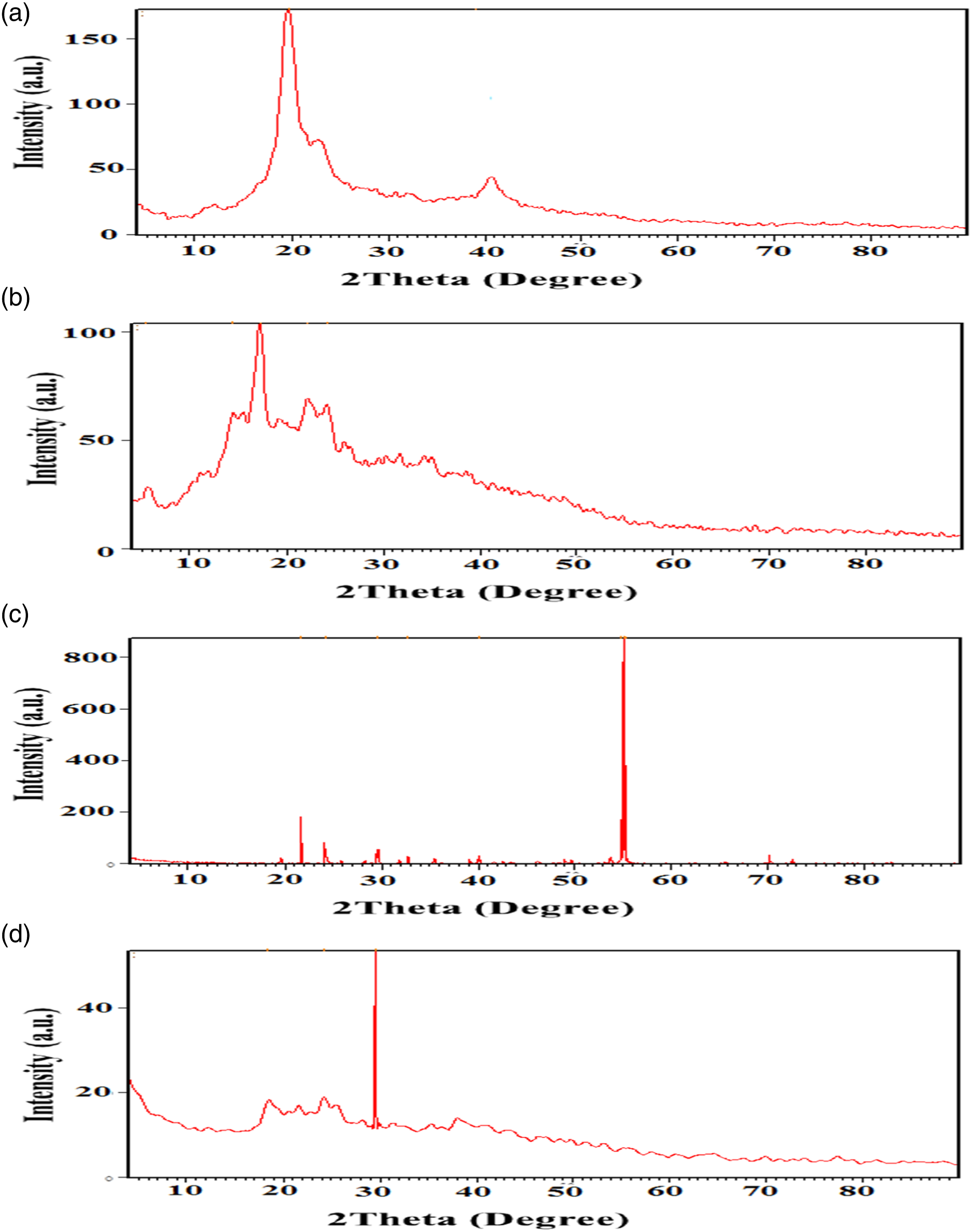
The X-ray diffraction of pure starch polymer is shown in Figure 6(b). Starch is a semi-crystalline material that contains both the crystalline and amorphous phases, due to its principal components which are the amylose and the amylopectin. 47 The amylose has a linear structure, which produces the crystalline region of the starch, whereas the amylopectin has a branched structure which is responsible for the amorphous phase of the starch. There are five diffraction peaks (2θ) for starch that appeared at 2θ = 5.49°, 14.35°, 17.25°, 22.15°, and 24.23° which indicated that Starch has semi-crystalline in nature. 47 Also the slight difference between our data and other published articles is due to the difference in ratio between amylose and amylopectin, determined by different starch provenience. 47
The X-ray diffraction of the original AgNO3 is shown in Figure 6(c). It is clear that pure AgNO3 exhibits several sharp intense crystalline peaks appeared at 2θ = 21.7°, 24.2°, 29.6°, 32.7°, 40.0°, 54.8°, 55.1° and 55.29°.
The X-ray diffraction of the Poly(PVA/St/Ag NPs) nanocomposite is shown in Figure 6(d), and it is similar to the original PVA, St and AgNO3 as mentioned above. The Poly(PVA/St/Ag NPs) has three main peaks that appeared at 2θ = 18.32°, 24.21°, and a sharp peak at 29.5°. The Poly(PVA/St/Ag NPs) presented in an amorphous structure due to the semi-crystalline structure of both starch and PVA which being destroyed during the dissolving process. 48 The peak appeared at 2θ of 29.5° is belonged to the remaining crystalline phase of both PVA and St. 48 The sharpening of the peak that appeared at 29.5° was probably due to more molecular chains rearranged during the little crosslinking process at lower irradiation dose conditions of 5 kGy. This indicates that amorphous regions are enhanced in the Poly(PVA/St/Ag NPs) nanocomposite.
Antifungal activity of Ag NPs
Effect of Silver-Nanoparticles on the growth of P. digitatum Sacc the causal agent of the green mold of citrus fruits.

aThe values are mean of three replicates ± standard deviation (p<.01).

Photographs showing linear growth inhibition of P. digitatum by 30 ppm silver nanoparticles on the PDA medium.
Effect of silver-nanoparticles on severity of infection of green mold of citrus fruits
Effect of Silver nanoparticles on the severity of infection of the green mold of citrus fruits caused by Penicillium digitatum at different storage periods (weeks).
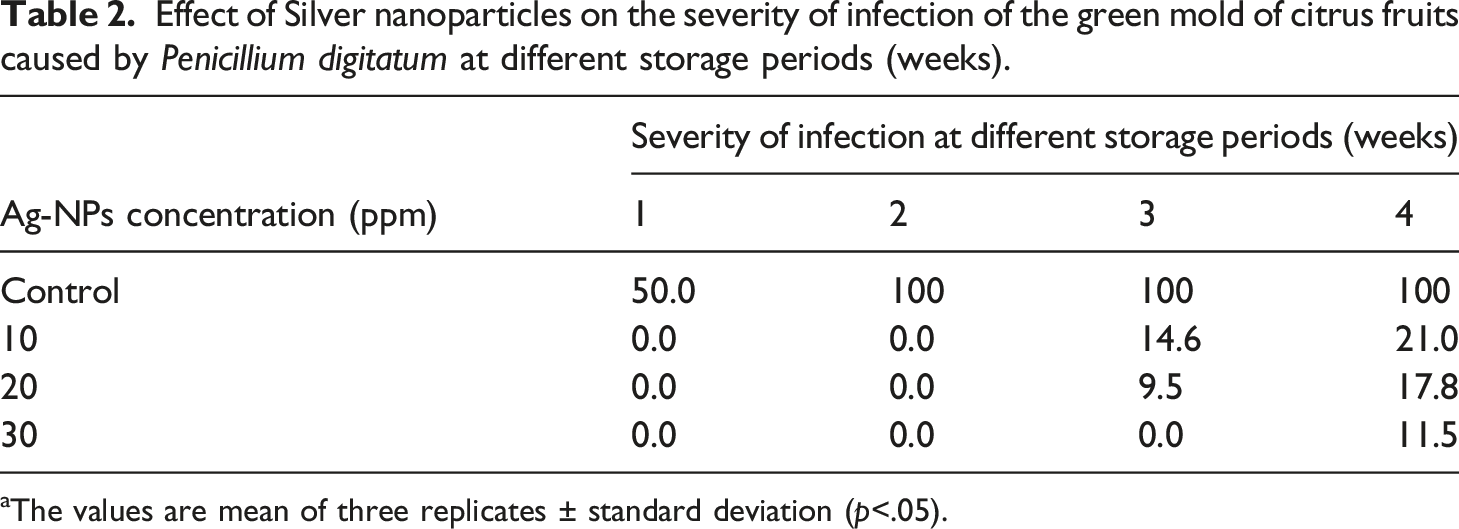
aThe values are mean of three replicates ± standard deviation (p<.05).
Effect of coating citrus fruits with the poly (PVA/St/Ag NPs) nanocomposite on the weight loss (%)
Effect of Coating citrus fruits with the Poly(PVA/St/Ag NPs) nanocomposite solution on the weight loss (%) at different storage periods (weeks).
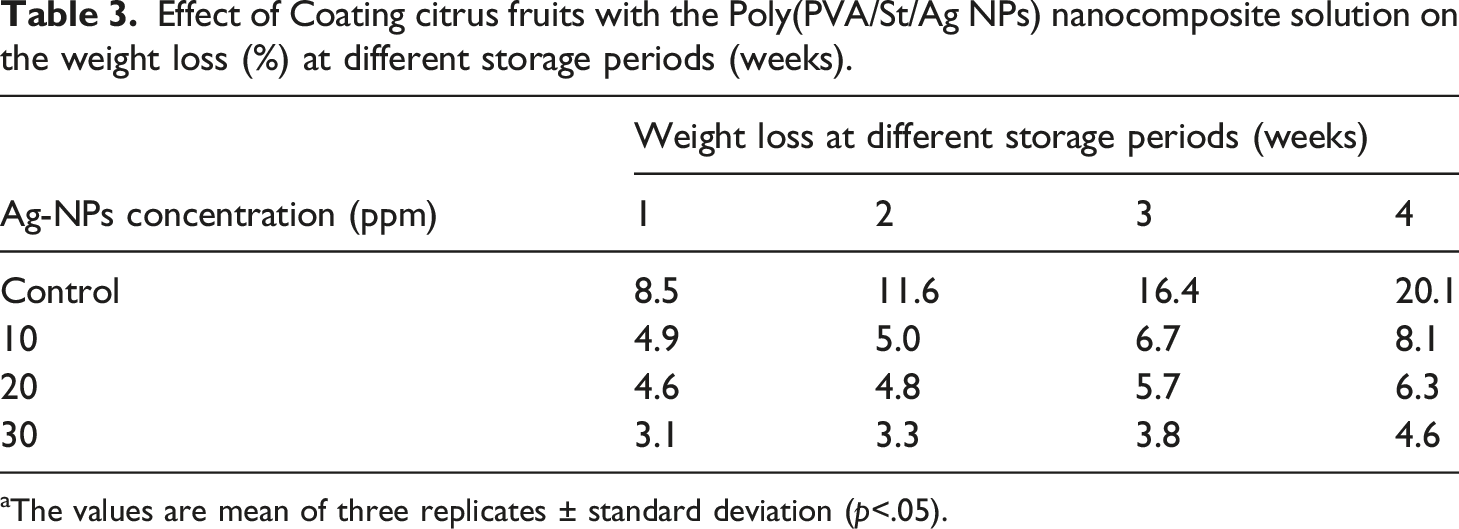
aThe values are mean of three replicates ± standard deviation (p<.05).
Scanning electron microscopy
Morphological changes occurred in the hyphae and conidiophores of P. digitatum due to growing the fungus in the PD broth medium containing 30 ppm silver nanoparticles as shown in (Figure 7). The growing healthy mycelia showed normal morphology with linear regular, homogenous and smooth cell wall hyphae, normal matulae, phialids, and long chain of conidia as shown in Figure 8(a). The conidia smooth walled ranging from subglobose to long cylindrical in shape or elliptical 4.0 μ occur, Conidiophere biverticillat asymmetrical with branches, matulae and phialides. The phialides are limited in the number of produced chains of elliptical conidia. This normal morphological structure varied in the presence of 30 ppm silver nanoparticles. Treatment with silver nanoparticles on P. digitatum caused irregular branching of hyphae in the apical part Figure 2(b) and the loss of the linearity. The effects of nanoparticles on the morphology of fungus hyphae revealed alterations in the morphology of hyphae which appeared several collapsed and squashed due to the lack of cytoplasm Figure 7(b). A massive increase in extruded amorphous–fibrillar material was marked in the innermost part of the mycelium. The fumigated spores with a sublethal dose of the oil-induced irregular shape and clear depressions on the surface of spores. These spores were originated from distorted phialides Figure 7(b). (a) Normal phialied and matulae. (b) Altered mycelia without spore information.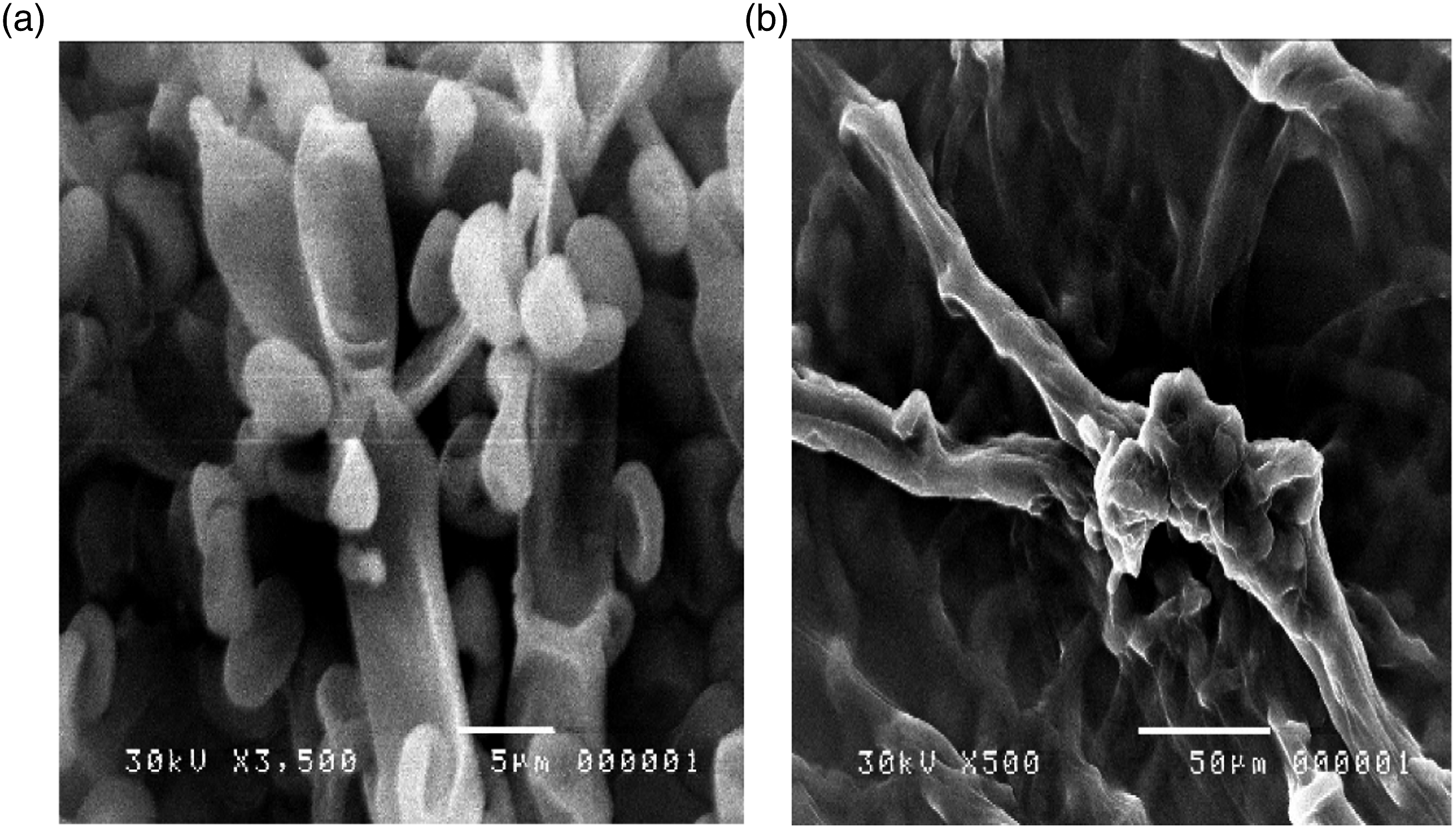
Conclusion
Radiation technique used successfully in the preparation and production of advanced antimicrobial Poly(PVA/St/Ag NPs) nanocomposite as coating materials for the Citrus fruits. The different analytical techniques such as FTIR, TEM, UV, and SEM showed successful formation of the Poly(PVA/St/Ag NPs) nanocomposite nanocomposite. The results of the present study indicate that AgNPs have a significant effect on the inhibition of the growth of P. digitatum. These nanoparticles caused complete growth inhibition at a concentration of 30 ppm. Also the Poly(PVA/St/Ag NPs) nanocomposite incorporated with concentration of 30 ppm Ag NPs showed higher fungicidal activity than free Ag NPs. The nanometer range of Ag NPs that can interact with P. digitatum protein surface and/or its core where it enters inside the cells, as a result cellular metabolism is inhibited causing the death of P. digitatum and subsequently exhibits antifungal activities.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.