Abstract
The primary objective of this research is to fabricate highly hydrophilic composite hydrogels to be used as a humidity sensor. A series of interpenetrating network copolymer hydrogels based on sodium acrylate (SA) and N-isopropylacrylamide (NIPAAm) monomers has been synthesized under the effect of gamma irradiation. The hydrogels were loaded with calcium chloride and charcoal as fillers. For comparison, hydrogels based on pure monomers were also synthesized. The pure hydrogels and composite hydrogels were characterized by different analytical methods such as gel content analysis, FT-IR spectra, swellability in water, thermogravimetric analysis (TGA), scanning electron microscopy (SEM), water vapor uptake and humidity absorption. The results obtained indicated that the copolymer and composite hydrogels with CaCl2 and charcoal Showed significant improvement in swellability in water, thermal stability and water vapour uptake and humidity absorption. The findings revealed also that the gel fraction is largely dependent on the irradiation dose and hydrogel composition. The addition of CaCl2 and charcoal in the composite hydrogels strongly enhances the humidity absorption capacity of the dried gel and increases the humidity absorption capability in comparison to the hydrogels based on pure monomers.
Introduction
Hydrogels are a kind of crosslinked polymer that can absorb substantial quantities of water and other liquids, often up to a hundred times their dry weight, without dissolving. 1 The hydrogels are categorized into three groups: synthetic, semi-synthetic, and natural.2,3 Synthetic hydrogels that are derived from monomers are appealing owing to their tunable chemical and physical characteristics relative to natural polymers.4,5 Also, synthetic hydrogels have higher water retention capacity than hydrogels based on natural polymers.6,7 Hydrogels may also be sensitive to stimuli and reply to the surrounding environment, such as pH, existence of electrolytes, temperature, ionic strength, and electric and magnetic field. 8 Those behavioral changes are called stimuli-sensitive hydrogels or smart hydrogels, owing to their ease of control and broad applicability across various domains, including in chemical processing, biotechnology, and medical fields. 9 Stimuli-responsive polymers have garnered significant attention due to their unique ability to react to changes in their surrounding environment.
Different preparation techniques were adopted as physically cross-linked, 10 chemical crosslinking, 11 grafting polymerization, 12 and radiation crosslinking. 13 In this contest, radiation crosslinking is powerful technique broadly employed due to the absence of chemical additives. Additionally, the sterilization and modification can be accomplished in a single step. 14 The radiation method primarily depends on generating free radicals within the polymer after exposure to a high-energy source, including an electron beam, X-ray, or gamma ray.15,16
Radiation polymerization is a process in which ionizing radiation (such as electron beams, gamma rays, or ultraviolet light) is used to initiate and control the polymerization of monomers into polymers. 17 This technique is widely used in the manufacture of hydrogels, biomaterials and coatings due to its ability to achieve polymerization without the need for catalysts or chemical initiators. 18 Gamma rays play an important role in various modifications of the structural and optical behavior of polymers. The use of ionizing radiation is a very convenient method for preparing superabsorption hydrogels. 19 The synthetic radiotherapy method does not require an initiator, catalyst, or cross-linker because ionizing radiation is highly active.20,21 Radiation therapy has several other advantages, such as easy process control; the technology is environmentally friendly because it does not produce any residues or environmental pollutants. 22 Gamma rays play a crucial role in modifying the properties of hydrogels, affecting their structure, function, and performance. Ionizing radiation (such as gamma rays and electron beams) generates free radicals in polymers, which leads to the formation of covalent bonds and enhances the gel fraction. The cross-linking density affects water absorption; high cross-linking reduces swelling, while partial decomposition can increase it. Radiation can also modify the response of hydrogels to environmental stimuli.23,24
In this work, the synthesis of interpenetrating and superabsorbent hydrogels based on poly(sodium acrylate-co-N-isopropylacrylamide), filled with CaCl2 and charcoal, using gamma irradiation has been studied. These hydrogels exhibit the capability to absorb and hold substantial amounts of water or aqueous solutions. It is commonly used in products including sanitary napkins, diapers, and other absorbent materials. 25 In this regard, N-isopropylacrylamide is a monomer that is often used in the synthesis of temperature-responsive hydrogels. This polymer has a lower critical solution temperature (LCST) of about 32°C. Under the LCST, the hydrogel is hydrated and swollen, while beyond the LCST, the hydrogel collapses and releases the absorbed water.26–29 The synthesis of a novel humidity sensor (HR) utilizing a composite hydrogel lies in the preparation of a superabsorbent material composed of poly(sodium acrylate-co-N-isopropylacrylamide), integrated powerfully with CaCl2 and charcoal, employing the gamma irradiation technique. 30 Gamma irradiation has emerged as a method for synthesizing hydrogels with distinct properties and functionalities. By leveraging this radiation-induced copolymerization process, a composite hydrogel with unique characteristics suitable for humidity sensing applications is envisaged. 31 The incorporation of poly(sodium acrylate-co-N-isopropylacrylamide) ensures good water absorbency and responsiveness to environmental stimuli, crucial for accurate humidity detection. 9 The aim for the presence of CaCl2 within the composite is to enhance its sensitivity to changes in humidity levels. Calcium chloride is hygroscopic in nature and facilitates efficient water uptake. 32 Additionally, activated carbon can be used as a humidity control material due to its adsorption properties. Additionally, it has a strong affinity for water molecules and can effectively adsorb moisture from the surrounding environment, the integration of charcoal serves to improve the sensor’s stability. 33 The high surface area and porosity of activated carbon, combined with the hydrogel matrix, can provide an effective material for capturing the humidity, overall, activated carbon can be a useful material for humidity control, helping to maintain a dry environment, protect moisture sensitive items, and improve air quality in various settings. 34
Experimental
Materials
N-isopropylacrylamide (NIPAAm) was supplied by Sigma-Aldrich Co. (USA). Acrylic acid (98%) was purchased from Advent Chembio PVT LTD (India). Anhydrous calcium chloride CaCl2 was supplied by Wuhan Chemical Factory, Wuhan (China). Charcoal and sodium hydroxide were obtained from El-Goumhouria Co., Egypt.
Preparation of (Sodium Acrylate-co-N-isopropylacrylamide)/CaCl2/charcoal Composite hydrogels
Preparation of sodium salt of acrylic acid (SA) and N-isopropylacrylamide (NIPAAm) mixture solutions
In this procedure, the sodium acrylate (SA) was produced by dissolving 9 g of acrylic acid in a solution containing 1 g of sodium hydroxide dissolved in 9 mL of distilled water under continuous stirring at 50°C for 60 minutes. Then, 1 g of NIPAAm was dissolved in 9 mL of distilled water. The sodium salt of acrylic acid was added to the NIPAAm solution under continuous stirring at 35°C for 30 minutes.
Preparation of SA-co-NIPAAm copolymer hydrogel
The weight percent ratio of SA and NIPAAM monomers was 9:1. Then, the reaction mixture was allowed to cool down to room temperature and exposed to gamma irradiation in different doses.
Preparation of (SA-co-NIPAAm)/CaCl2/charcoal composite hydrogels
Hydrogel compositions and their designation.
Gamma irradiation
Gamma irradiation was conducted at the cobalt-60 cell within the Canadian facility at the National Center for Radiation Research and Technology in Cairo, Egypt. The irradiation is carried out at different doses with a dose rate of 0.8 kGy/h.
Characterizations
Gel content (%)
Samples of the dry hydrogels were carefully weighed (Wo), immersed in distilled water at 80°C for extraction, and subsequently dried until a stable weight (W1) was achieved in a vacuum oven. The gel fraction estimated employing the equation below: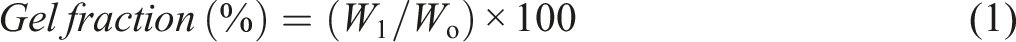
Swelling degree at equilibrium
A specified dry weight of the hydrogel (Wd) was submerged in distilled water at room temperature for varying periods. Each time, the samples were taken out, placed on filter paper to absorb any excess surface water, and then weighted (W
FT-IR spectroscopy
The FT-IR spectra were acquired employing an FT-IR spectrophotometer (Mattson 100 Unicam, England), covering the wave number range of 4000-500 cm−1.
Morphology (SEM)
The alteration in surface morphology was studied utilizing a high-resolution scanning electron microscope (HRSEM, ZEISS EVO 15 SEM, UK). Before scanning, the samples were covered with a fine layer of gold.
Thermogravimetric analysis (TGA)
The TGA thermograms were conducted using a Shimadzu–50 device (Kyoto, Japan) with a heating rate of 10°C/minute and a nitrogen flow rate of 20 mL/min, covering a temperature range from the temperature of the room to 600°C. To establish the rate of thermal decomposition reaction, the primary TGA thermograms were utilized.
Moisture uptake tests
The tests for moisture absorption were conducted using a humidity chamber created with an air purification system, an electric fan, a humidifier connected via a tube, a humidity gauge, and a balance. Before conducting moisture absorption tests, each sample of gel desiccants was sliced into uniformly sized pieces in terms of thickness and diameter, and then subjected to a freeze-drying procedure. The moisture absorption abilities of each gel desiccant were determined through the use the following equations.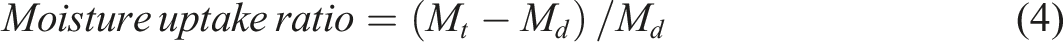
Vapor water uptake
The transport characteristics of water vapor were assessed using samples measuring 15 mm in thickness and 50 mm in diameter. The hydrogel samples were insulated in epoxy resin along their sides and placed inside a metal cup filled with silica gel beneath them. The cups were inserted into a climate-controlled chamber where the temperature was maintained at 25°C and the relative humidity was kept at 50%. The samples were periodically weighed to monitor their mass increase. 35
Results and discussion
Gel content
The Gel content is the insoluble fraction of a crosslinked polymer network and it is assessed following the removal of the soluble fraction by extraction in a suitable solvent. Conversely, gel content reveals the degree of crosslinking existing in the hydrogel. A high level of crosslinking will cause the structure to become stiff, decreasing the hydrogel’s capacity to absorb water. Figure 1 shows the gel fraction as a function of absorbed dose for the hydrogels designed from H1 toH6 as shown in Table 1. It is evident that the gel content passes through three regions, regardless of the composition of the hydrogel. A sudden rise in the gel content occurs with a low dose of 5 kGy, and the gel content tends to rises with a rise in irradiation dose up to 20 kGy and reaching maximum stable values of 88.0, 91.0, 92.5, 94.5, 95.3 and 97.5% for the hydrogels designed H1, H2, H3, H4, H5 and H6, respectively. Grounded in these findings, several points can be highlighted: (1) The copolymer and composite hydrogels showed a relatively higher gel content than the hydrogels made from pure monomers. (2) It can be observed that the introduction of both CaCl2 and charcoal into the copolymer hydrogel resulted in the highest gel content. The presence of CaCl2 and charcoal increases the voids within the polymers, thereby facilitating the production of additional free radicals. In addition, charcoal and calcium chloride are able to absorb humidity due to their porous structure and high surface area. Charcoal is particularly effective at absorbing moisture from the air. This is due to the porous structure where charcoal has a network of tiny pores that trap humidity from the air as moisture molecules adhere to the surface of the charcoal through physical absorption. Gel fraction of as a function of absorbed dose for the hydrogels designed from H1 toH6 as shown in Table 1.
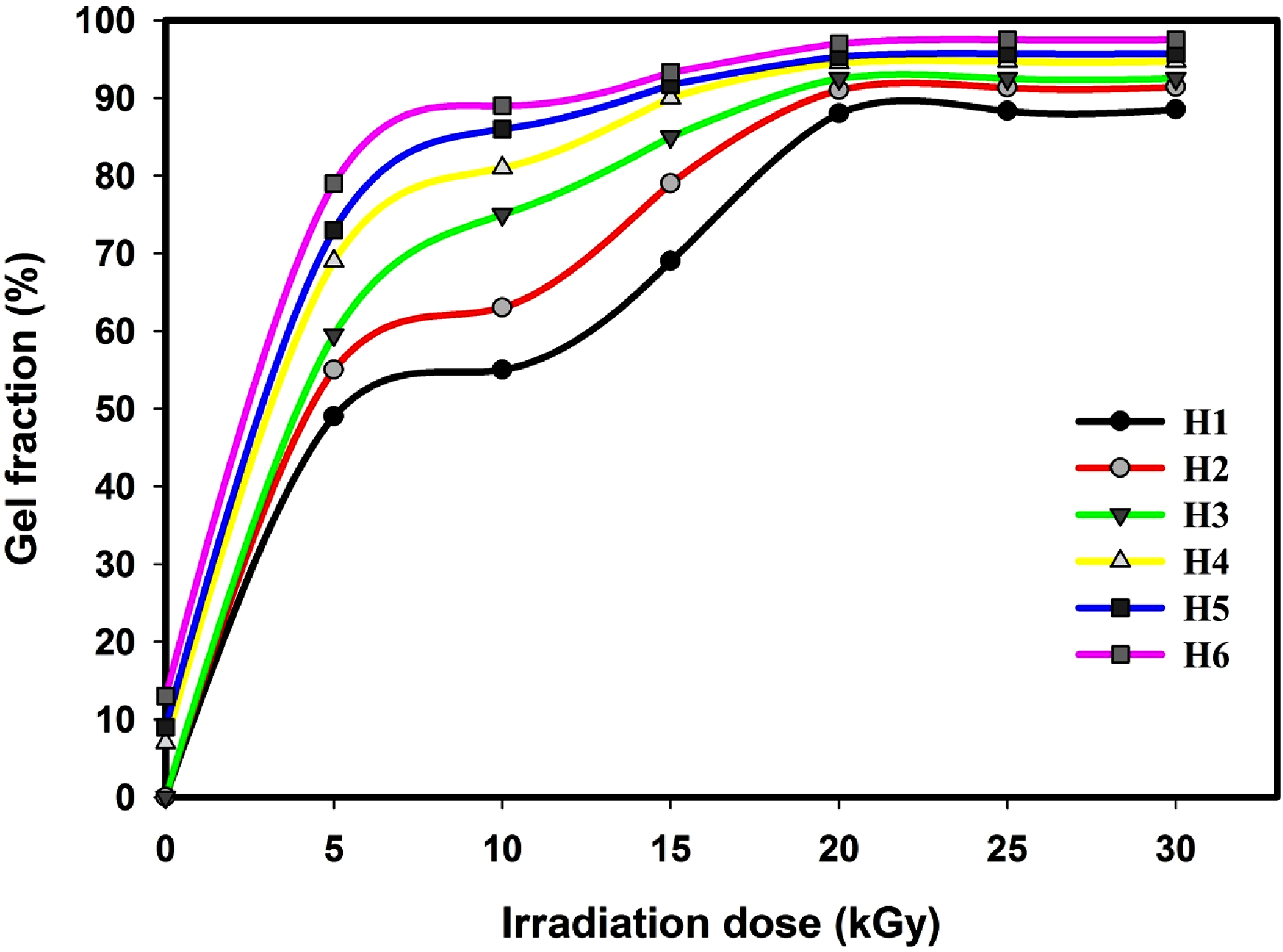
The crosslinking of PSA-co-NIPAAm in aqueous solution through gamma radiation can be succinctly described as outlined below: (1) Gamma radiation is absorbed by the polymers PSA and NIPAAm and the solvent H2O, the radicals –(-C•H▬C•H▬CONH
(2) To form a crosslink point, two polymer radicals with n and m repeat units combine.
Swelling characters
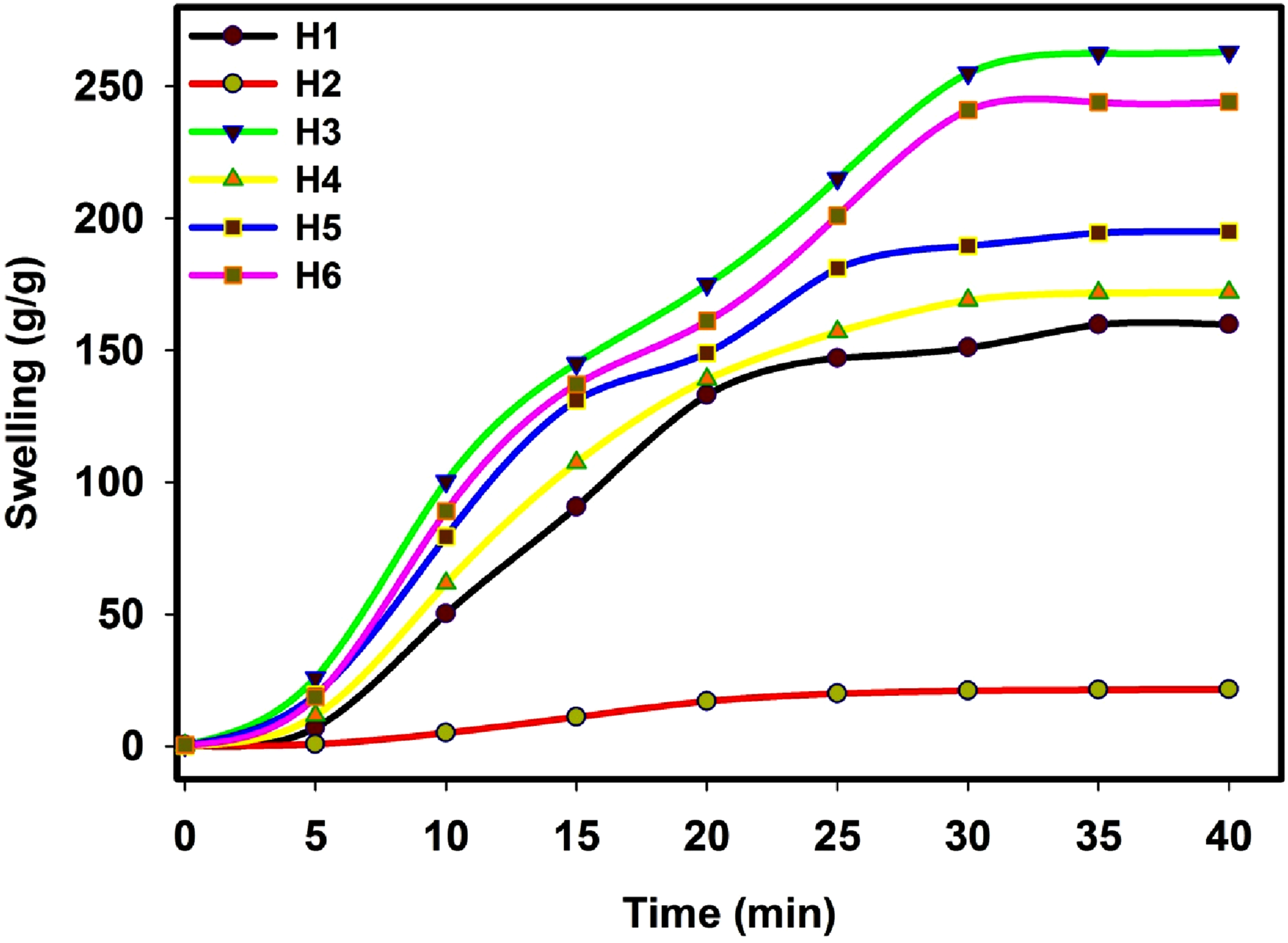
The swelling ratio indicates the extent of the crosslinking in a polymer. Figure 2 reveals the swelling in water of the hydrogels designed from H1 toH6 as shown in Table 1; all of these hydrogels were produced using a consistent gamma irradiation dose of 20 kGy. From the data presented in Figure 2, a few points could be indicated: (1) The swelling ratio substantially rises over time, peaking approximately after 30min, where the hydrogel based on pure PSA displayed the lowest water swelling. This is understandable because PSA monomer is highly sensitive to gamma radiation than PNIPAAm monomer and when the crosslinking density increases, that is the structure became compact, the water swelling decreases. (2) The hydrogel based on (PSA-co- PNIPAAm) copolymer showed the highest water swelling in accordance with the gel content6 behavior. The copolymer hydrogel (H3) can absorb ∼263-fold the weight of water and thus the elevated swelling ratio may be related to the formation of a porous structure, which enhances the water transparency into hydrogels. The porosity favors a better water diffusion through the hydrogel network. In addition, the high swelling behavior of H3 hydrogel is due to the combination of hydrophilic groups and sodium ions in the PSA the polymer chain that enable to absorb and retain water effectively. Moreover, the existence of NIPAAm with sodium acrylate resulted in the production of hydrogels with unique properties, which can absorb a substantial quantity of water in a short period. (3) In case of H6, H5 and H4, the swelling ratio was 244, 194, and 172 fold the weight of water, respectively. As depicted in Figure 2, the swelling ratio of the (PSA-co-NIPAAm)/CaCl2 (H4) composite hydrogel filled with CaCl2 is first increased with time from 0 to 6h to reach ∼172 g/g. The (PNaAc-co-PNIPAAm)/charcoal (H5) composite hydrogel showed a comparable change trend. The final equilibrium-swelling ratio is higher than that of (PSA-co-NIPAAm)/CaCl2 composite hydrogel (about 194 g/g). When CaCl2 and charcoal are added, the equilibrium-swelling ratio of the Poly(PSA-co-NIPAAm)/CaCl2/charcoal composite hydrogels (H6) is found to reach 244 g/g. This is primarily due to the presence of CaCl2 and charcoal increased the hydrophilic of the composite hydrogel resulting in the expansion of the networks as well as the electrostatic repulsion of the same charged counterparts in the network structure, which led to a rise in swelling ratio and the size.32,36 Swelling in water of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced using a consistent gamma irradiation dose of 20 kGy.
FT-IR analysis
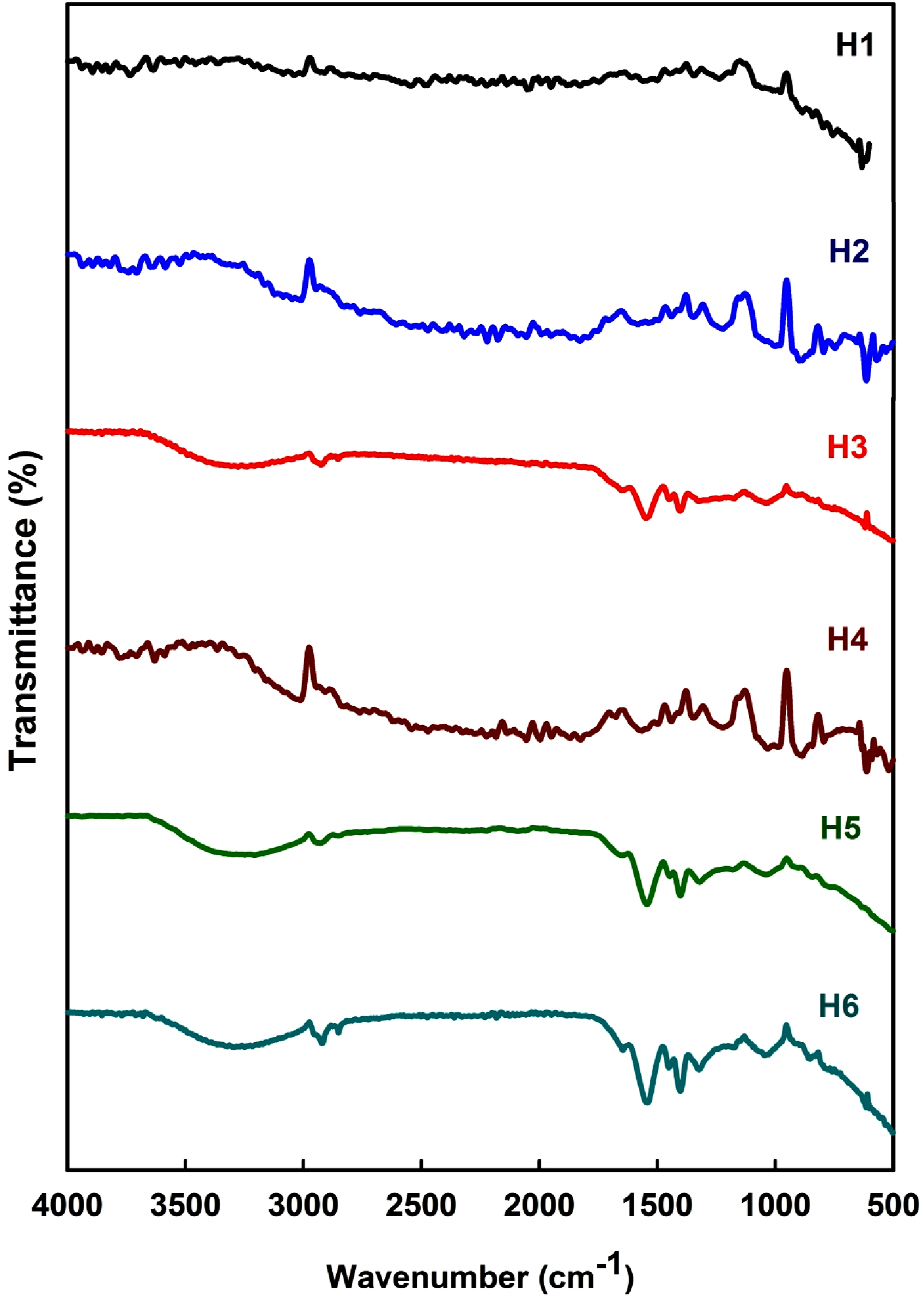
Figure 3 reveals the FT-IR spectra of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced using a consistent gamma irradiation dose of 20 kGy. From the data in Figure 3, a few points could be addressed: (1) A wide band corresponding to stretching vibrations due to the hydroxyl groups (free OH group and hydrogen bonding occurs in the range of wave number 3441.12 cm−1. This band is most clear in the H1 spectrum due to its capability to absorb water. The band at 2937.9 cm−1 in the instance of the H1 spectrum results from the asymmetrical and symmetrical stretching vibrations of -C-H. (2) The band at 1740 cm−1 arises from the stretching vibrations of the C = O occurs only in the case of H1. Following that, a band of asymmetrical stretching vibrations of the carboxyl –COO- is also properties of the H1 hydrogel. (3) For the hydrogel (H2), the band at 2977 cm−1 is due to the stretching vibrations of the -CH(CH3)2 group. Furthermore, the bands at 1377 cm−1, 1395 cm−1 and 1545 cm−1 corresponding to the -CH(CH3)2 and the secondary amide groups in the structure of PNIPAAm. Moreover, the typical double peaks at 1650 cm−1 can be ascribed to the amide I band (C = O stretching) and 1540 cm−1 to the amide II band (N–H in-plane bending vibration) of PNIPAM. (4) For the hydrogel (H3), (PSA-co-NIPAAm), the prominence of the characteristic peak at 1675 cm−1, distinctly noticeable in PSA, diminished progressively, while new absorption bands emerged within the range of 1673 cm−1 and 1651 cm−1. This suggests that novel intermolecular hydrogen bond interaction in the hydrogel was formed, providing evidence that the hydrogen bonds originally present in the PSA hydrogel have been substituted by those forming among PSA and PNIPAAm. (5) For the composite hydrogels H4, H5 and H6, the previous bands are shifted toward lower wavenumbers could prove an interaction of carboxyl groups of the polymer with the calcium ions or charcoal. In (PSA-co-PNIPAAm), the bands at 1563 cm−1, 1560 cm−1 and 1557 cm−1 are ascribed to (CH2) vibrations occurs in the case of the hydrogels H4, H5] and P6. However, the bands at 1411, 1407, 1405 and 1403 cm−1 are ascribed to the symmetric stretching vibrations of –COO- can be seen. (6) These findings are the consequence of the hydrogen bonding among the amide group of the PNIPAM network and the carboxyl group of the PSA chains, signifying that (PSA-co-NIPAAm) is prepared successfully. In addition, the FT-IR confirms the strong interactions between (PSA-co-NIPAAm) and calcium and ions, and charcoal. FT-IR spectra of the hydrogels designed from H1 toH6 as shown in Table 1; all of these hydrogels were produced using a consistent gamma irradiation dose of 20 kGy.
Scanning electron microscope (SEM)
Figure 4 reveals the SEM micrographs of the fracture surfaces of hydrogels designed from H1 toH6 as shown in Table 1; all of these hydrogels were produced using a consistent gamma irradiation dose of 20 kGy. It is evident that the morphology of the fracture surfaces of hydrogels is characterized by the presence of porous along the entire structure, irrespective of the kind of hydrogel, that confirming the formation of three-dimensional network structure. It is hypothesized that these pores function as areas where water can permeate and serve as points of interaction between external factors and the gel’s hydrophilic groups. These pores were generated due to the formation of NH3 gas and the evaporation of water throughout the hydrogel synthesis.37–39 The absorption of water into hydrogels is largely attributed to the existence of pores on their surface. However, the SEM microphage of the hydrogel based on pure PNIPAAm (H2) showed that the pore structure is more compact, and its pore size is significantly smaller than that of the hydrogel based on pure PSA (H1). This suggests that the addition of the NIPAAm monomer led to a rise in the pore size of the PSA hydrogel. On the other hand, it can be observed that the pores on the fracture surface of the copolymer hydrogel (SA-co-NIPAAm) (H3) hydrogel are large. It is expected that the increase in the size of pores could allow for greater solution infiltration due to the elevated absorption levels observed in H3, H5 and H6. In H4, H5 and H6 hydrogels reinforced by CaCl2 and charcoal particles, the desperation and interfacial compatibility of particles in the hydrogel are significant to the mechanical property. This finding indicates that the CaCl2 and charcoal particles have excellent interfacial compatibility with (SA-co-NIPAAm) hydrogel due to forming covalent bonding with (NaAc-co-NIPAAm). SEM micrographs of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced employing a consistent gamma irradiation dose of 20 kGy.
Thermogravimetric analysis (TGA)
TGA was utilized to assess the thermal stability properties of the copolymer and composite hydrogels under investigation. The documented dissociation energies of the covalent bonds C–H, C–C, C = O, C–O, C–N and O–H are 414, 347, 741, 351, 293 and 464 kJ/mol. 40 Based on these values, the calculated average total dissociation energies for pure AAc and NIPAAm, considering the various covalent bonds that make up the polymer molecules, are 336.5 kJ/mol and 396.1 kJ/mol, respectively. It can therefore be inferred that the creation of networks incorporating AAc and NIPAAm is likely to produce hydrogels with thermal stability that falls between that of AAc and NIPAAm, depending on the composition.
Figure 5 reveals the TGA thermograms and the corresponding rate of the thermal decomposition reaction curves of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced using a consistent gamma irradiation dose of 20 kGy. Table 2 presents the percentage of weight loss at various heating temperatures and the temperature of the maximum rate of the thermal decomposition (Tmax) of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced using a consistent gamma irradiation dose of 20 kGy. From the data illustrated in Figure 5 and Table 2, several conclusions can be drawn: (1) The TGA thermograms revealed three distinct stages of decomposition, for all the hydrogels, either prepared from pure monomer al the copolymers. In the first stage up to ∼350oC, is linked to the removal of water that has adhered to the surface of particles, commonly referred to as bound water or moisture absorption. However, during this stage, the hydrogel based on NIPAAm showed higher thermal stability in terms of weight loss (%) over all the hydrogels based on SA-co-NIPAAm copolymer or composites in accordance with the data in Table 2. (2) The primary thermal phase takes place in the second region, around 350 °C–450 °C, where there is a substantial weight reduction. (3) According to the temperatures at which different weight loss (%) occurred, the thermal stability of the various hydrogels may be ordered as follows: TGA thermograms and the corresponding rate of the thermal decomposition reaction curves of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced employing a consistent gamma irradiation dose of 20 kGy. Weight loss (%) at various temperatures of heating and the temperature of the maximum rate of the thermal decomposition (Tmax) of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced employing a consistent gamma irradiation dose of 20 kGy.
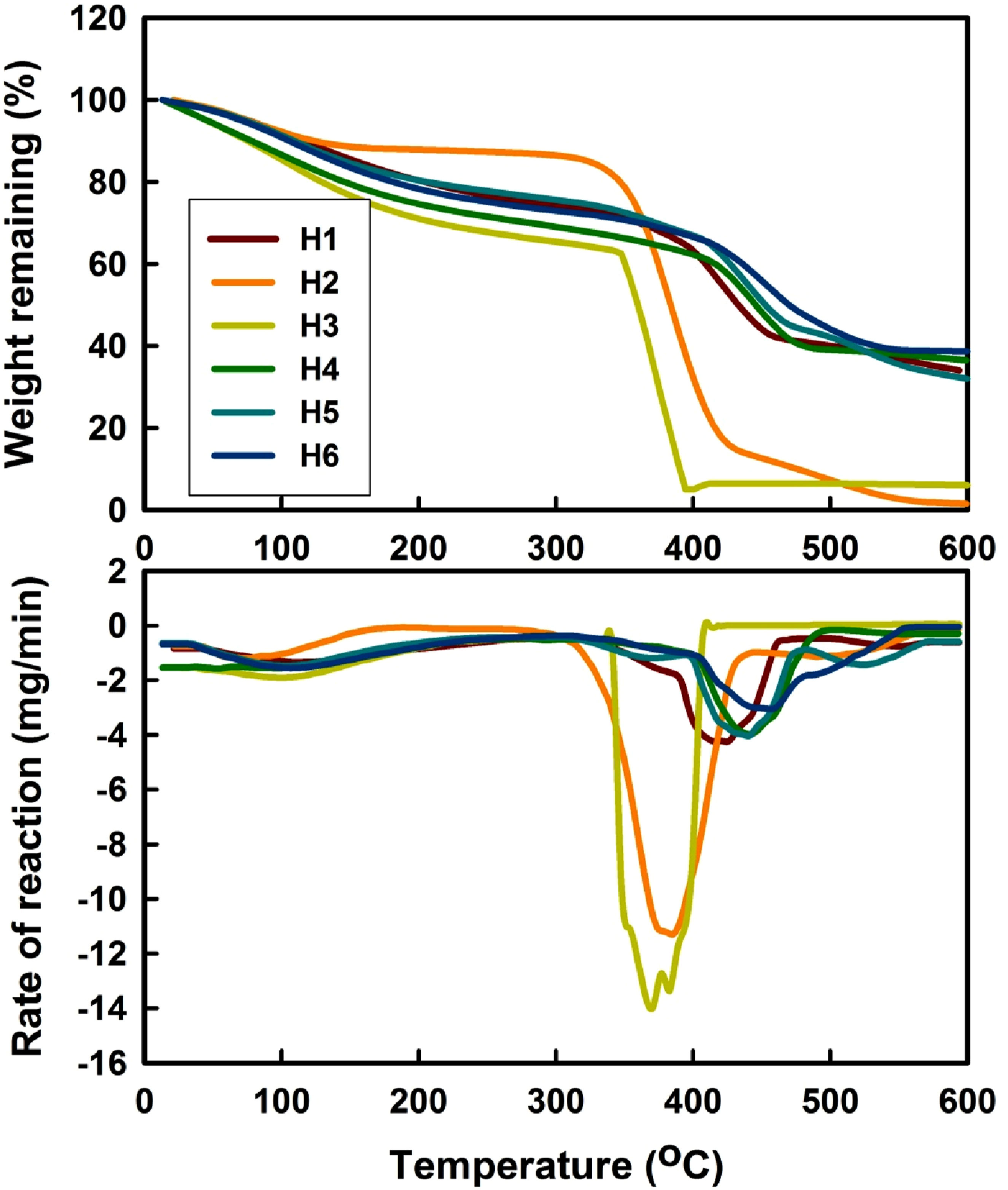
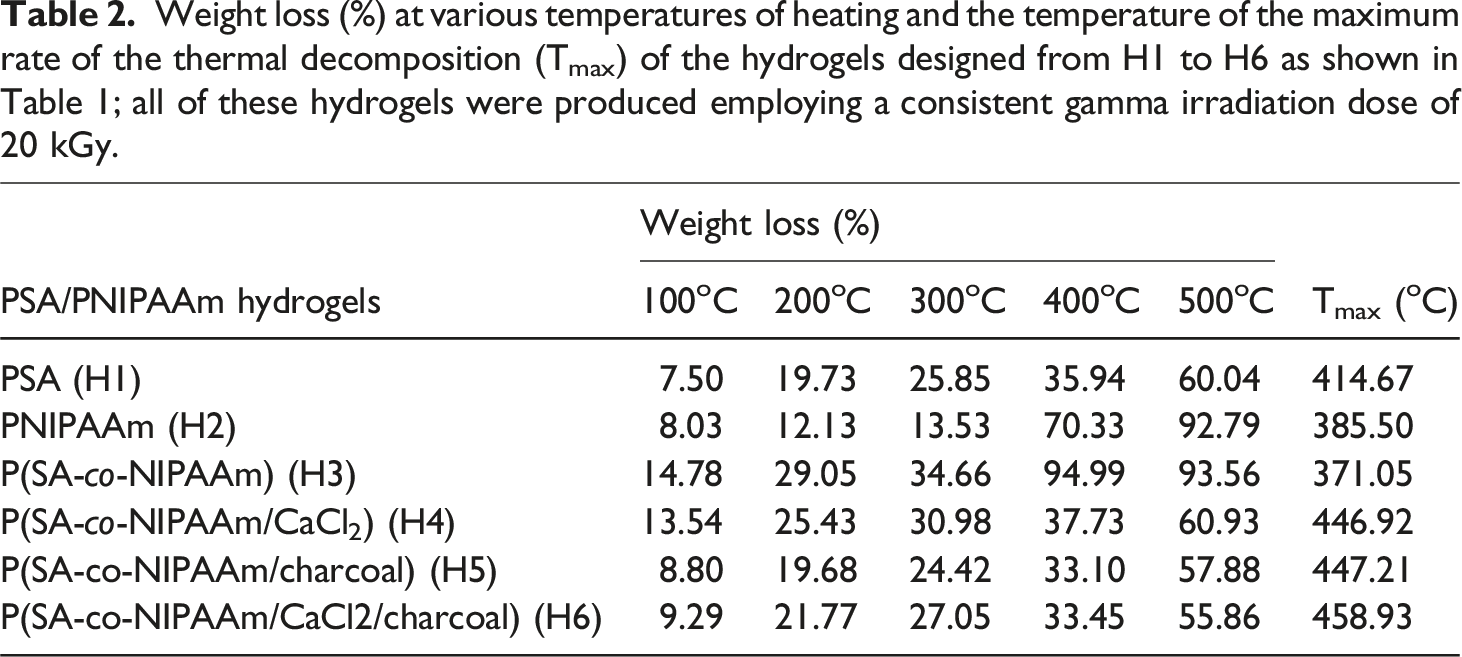
H2 > H1 > H5 >H6 >H4 >H3 (4) Based on the temperature of the maximum rate of the thermal decomposition (Tmax), the thermal stability of the different hydrogels may be arranged as follows:
H6 (458.93oC) > H5 (447.21oC) ≥ H4 (446.92oC) > H1 (414.67oC) > H2 (385.50oC) >H3 (371.05oC)
Vapor water uptake
Figure 6 shows the water vapor absorption of the hydrogels designed from H1 to H6 as shown in Table 1; all of these hydrogels were produced employing a consistent gamma irradiation dose of 20 kGy. Considering this data, several points may be addressed: (1) It is supposed that the small number of water molecules of vapor water is adsorbed on its hydrophilic moieties. (2) The enhancement in the vapor water uptake at the equilibrium of the different hydrogels can be arranged as follows: Water vapor uptake of the hydrogels designed from H1 toH6 as shown in Table 1; all of these hydrogels were produced employing a consistent gamma irradiation dose of 20 kGy.
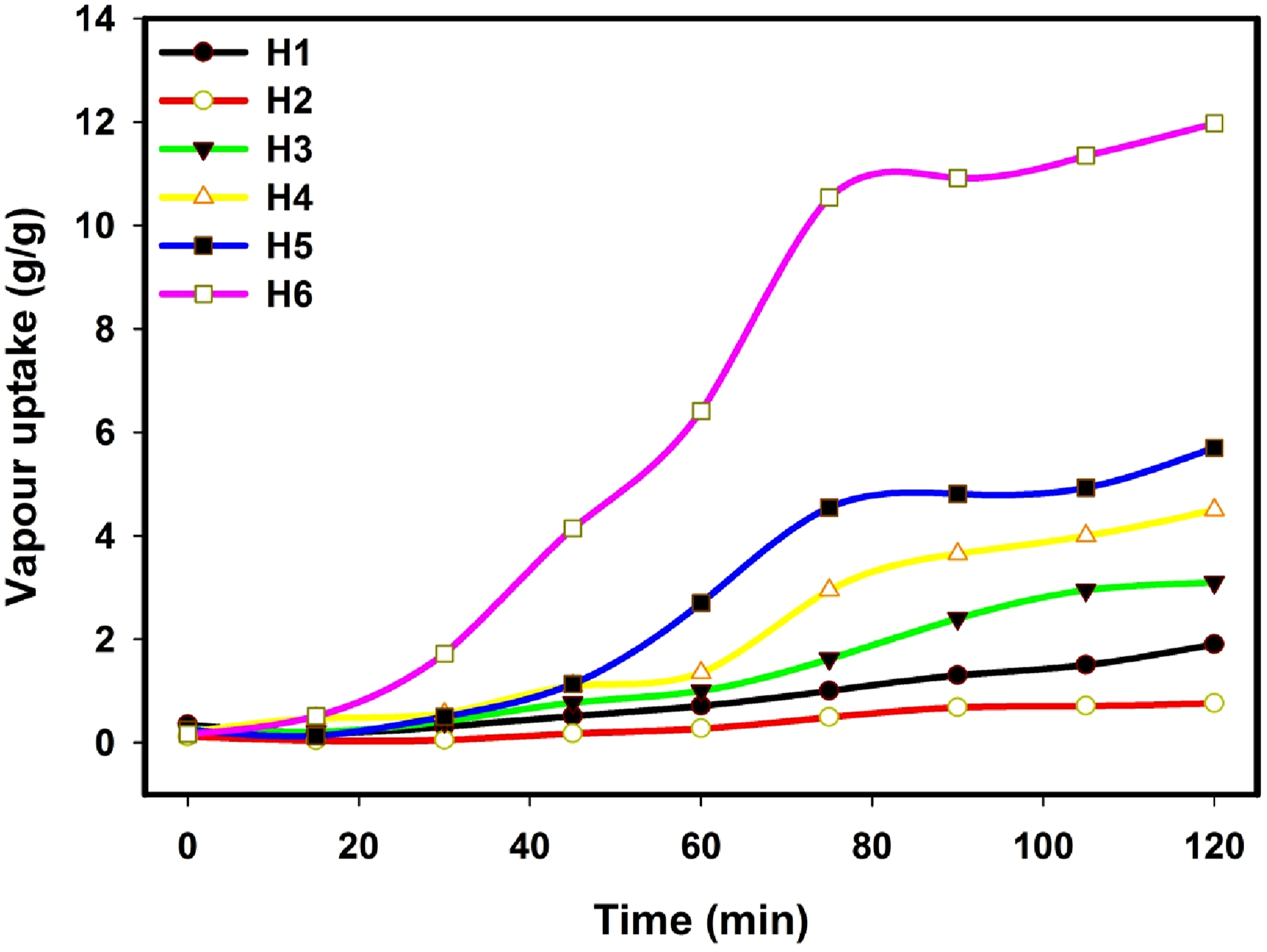
H6 (11.97 g/g) > H5 (5.7 g/g) > H4 (4.5 g/g) >H3 (3.1 g/g) >H1 (1.9 g/g) >H2 (0.76 g/g) (3) According to the above values of the vapor water uptake at equilibrium, it can be concluded that the hydrogels based on the copolymer possessed higher values than the hydrogels based on pure monomers. (4) The large vapor water uptake leads to an expansion in the surface area or the capacity for absorption in composite hydrogels. The presence of calcium chloride and charcoal in the hydrogel (H6) acts as a general deliquescent salt employed daily. When exposed to air, this salt can turn into clumps and become a saturated solution. This process, known as deliquescence, occurs when the partial pressure of water vapor in the surrounding environment differs from the vapor pressure at the surface of salts.41,42
From the above, PNIPAAm is a thermosensitive hydrogel that exhibits water absorption behavior depending on the temperature of the medium. At less than 40°C, it exhibits a hydrophilic state. PNIPAAm absorbs water and forms a hydrogel due to the hydrogen bonding between its amide groups and water molecules. At temperatures above 40°C, it exhibits a hydrophobic and shrinking state. PNIPAAm repels water and shrinks, becoming insoluble due to increased hydrophobic interactions. The temperature-sensitive mechanism is dominated by the hydrogen bonding between the amide groups of PNIPAAm (-CONH) and water, keeping the polymer swollen and hydrated. The hydrophobic behavior is dominated by the hydrophobic isopropyl groups (-CH(CH3)2) which leads to the collapse of the polymer and the expulsion of water. This is what happened in the case of sample (H2) as well as the presence of PNIPAAm in sample (H3). However, due to the property of charcoal and CaCl2 to absorb moisture and water vapor and their presence in sample (H6), sample (H6) had the highest absorption of moisture and water vapor.
Humidity absorption
Figure 7(b)) shows the humidity absorption of various hydrogels. It is important to highlight that the hydrogels designed from H1 to H6 as shown in Table 1; measured at constant temperature of 35oC and relative humidity (RH) of 75%. The samples of the hydrogel were positioned on a glass Petri dish and equilibrated for moisture absorption evaluations. It can be found that the dried H1, H2, H3, H4, H5 and H6 hydrogels absorbed 0.31 g, 0.186 g, 0.477 g, 1.33 g, 1.75 g and 2.63 g of humidity per 1 g, respectively. The addition of CaCl2 and charcoal in the composite hydrogels designed H4, H5 and H6 strongly enhances the humidity absorption capacity of the dried gel and increases the humidity absorption capability in comparison to the hydrogels based on pure monomers. In the beginning, the absorption rate was relatively fast because it occurred in the existence of CaCl2 and charcoal on the surface of the material and interpenetrating polymeric network, leading to the best humidity absorption. In addition, the presence of CaCl2 and charcoal together in hydrogel networks (H6) could be covered with water molecules by their adsorption onto the polymer chains, potentially making the networks more hydrophilic.
42
(a) 1. Sample H6 before and after humidity absorption. 2. The humidity absorption tests setup. (b) The humidity absorption of H1, H2, H3, H4, H5 and H6 at 35oC and 75% RH. (c) The humidity absorption of the H6 composite hydrogel at various temperatures.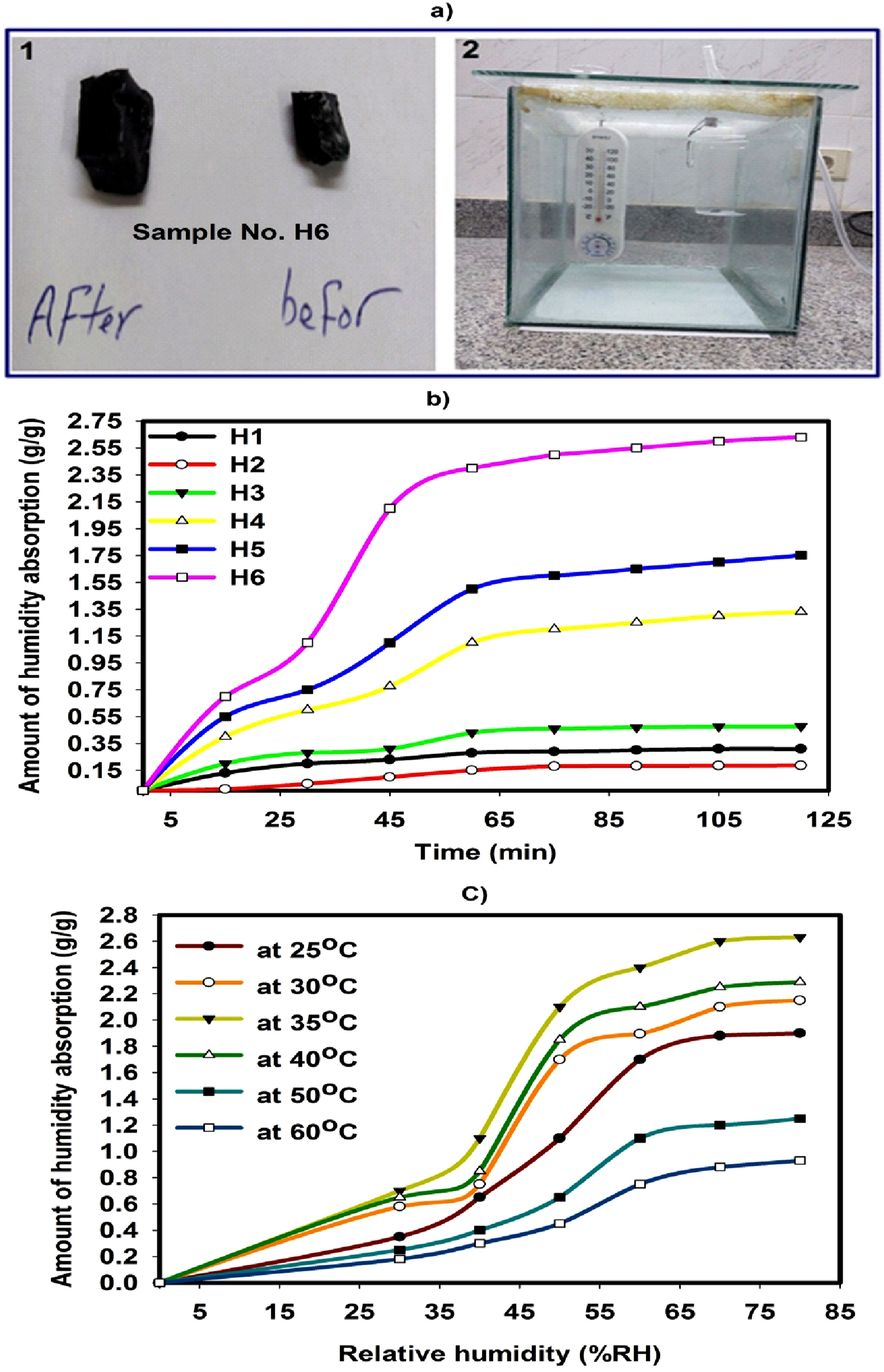
Figure 7(c)) illustrates that the dried composite hydrogel’s absorption of moisture (SA-co-NIPAm)/CaCl2/charcoal measured at different temperatures, different relative humidity and prepared to a consistent dose of 20 kGy of gamma radiation. The humidity absorption is found to increases gradually with increasing temperature until 40oC and relative humidity. It’s crucial to recognize that the isotherms for humidity absorption at temperatures above 40°C varied significantly compared to those at temperatures below 30°C. This could be ascribed to the transformation of the H6 chains from being hydrophilic to hydrophobic. From the isotherms of humidity absorption below 30°C, it is evident that the H6 composite hydrogel’s absorption capacity increased gradually at relative humidity of less than 40% RH, but showed a rapid increase once the relative humidity exceeded 40% RH. The variation in the absorption mechanism arises from a temperature-sensitive alteration in the hydrophobic properties of the H6 chains. Consequently, the capacity of the P6 composite hydrogel to absorb moisture can be controlled by adjusting the temperature; however, it does not exhibit swelling when submerged in water.
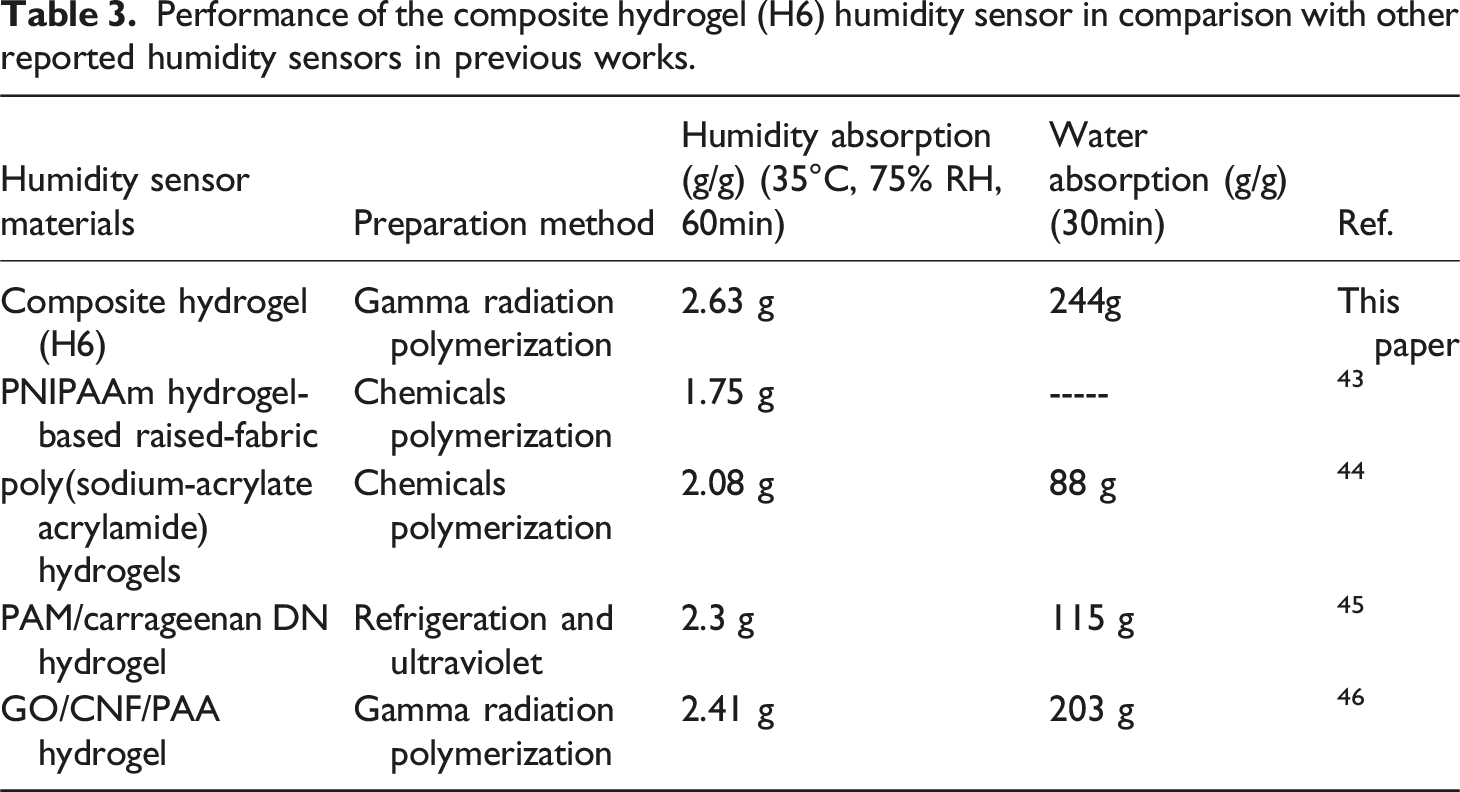
Performance of the composite hydrogel (H6) humidity sensor in comparison with other reported humidity sensors in previous works.
Conclusions
In this work, superabsorbent hydrogel composites have been successfully synthesized by gamma radiation copolymerization of copolymer based on sodium salt of acrylic acid and N-isopropylacrylamide. The hydrogels were loaded with calcium chloride and charcoal. The results obtained indicated that the copolymer and composite hydrogels with CaCl2 and charcoal achieved the highest improvement in swelling in water, thermal stability and water vapor uptake and humidity absorption. The findings revealed also that the gel fraction is largely dependent on the irradiation dose and hydrogel composition. All samples show a high absorption capacity for water vapor, which can be controlled by the chemical composition. Thus, it may be concluded that the composite hydrogels loaded with CaCl2 and charcoal could be utilized as a moisture sensitive material to construct a moisture sensor as show Scheme 1. The schematic diagram for preparation and application PSA-co-PNIPAAm/CaCl2/ charcoal composite hydrogels.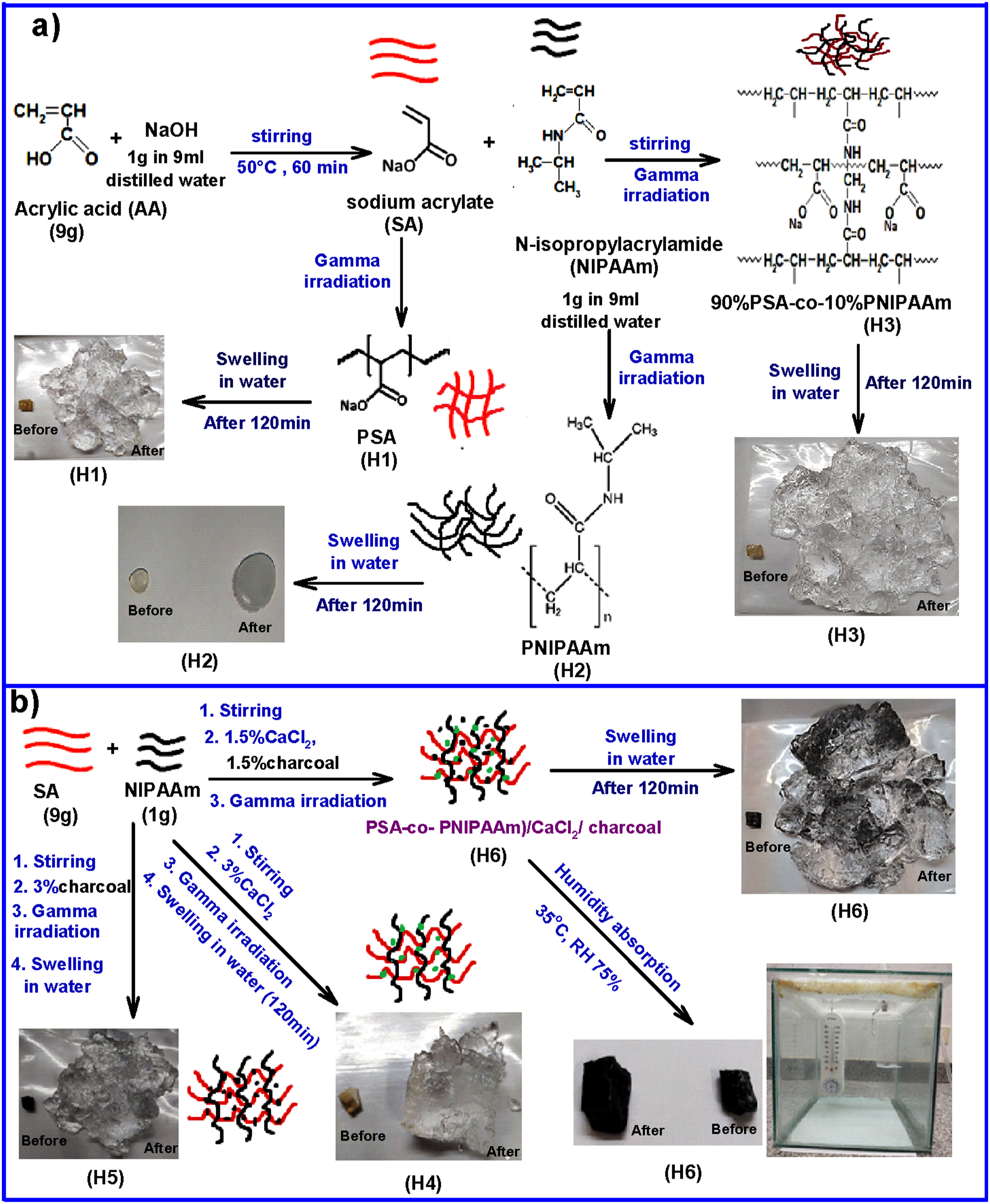
Footnotes
Acknowledgements
They also extend their sincere thanks to the National Center for Radiation Research and Technology, part of the Egyptian Atomic Energy Authority in Cairo, Egypt, for their financial backing. MA Elhady, Reham H Helal.
Author contributions
All contributors were involved in the development and planning of the study. Material preparation: Dr MA Elhady, and Dr Reham H Helal. Data collection and analysis: Dr MA Elhady, and Dr Reham H Helal. The first draft of the manuscript: Dr MA Elhady, and Dr Reham H Helal. Each author reviewed earlier drafts of the manuscript and provided feedback. All authors also read and gave their approval for the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The authors affirm that the data and resources can be accessed.
