Abstract
Biodegradable starch-based films are an environmentally friendly solution to reduce the use of petroleum-derived polymers. Thus, this work aims to obtain corn starch films by incorporating 5% w/w of cellulose nanocrystals (CNC), different concentrations of titanium dioxide nanoparticles (TiO2) (0, 0.25, 0.5, 1, and 2% w/w), and 40% w/w of glycerol and sorbitol (plasticizers), in a 1:1 ratio. The films showed a high potential for UV-light barrier, with an increase of 307% incorporating only 2 wt% of TiO2 in relation to the film without incorporation of TiO2. The use of TiO2 increased the white pigmentation capacity of the samples and decreased the lower water solubility – a reduction of approximately 28% using 0.5 wt% TiO2 compared to the CNC control sample was observed. The incorporation of TiO2 nanoparticles increased the tensile strength by adding 1 wt% of TiO2 nanoparticles with 34% increase in the tensile strength of the film without incorporation of TiO2. The thermal stability of the films with 1 wt% TiO2 and 5 wt% CNC increased by 85°C. Thus, the addition of CNC and TiO2 enabled an improvement in the physical/thermal/chemical properties of the films, making them possible alternatives for use in food packaging.
Introduction
The properties provided by polymers such as flexibility, strength and durability have revolutionized the field of materials due to their wide applicability compared to conventional ceramics and metals. However, most polymers are obtained from non-renewability sources, causing serious environmental problems mainly due to the accumulation of these plastic wastes in the environment.1–4 It is estimated that up to 53 million tons of plastic may enter aquatic ecosystems per year by 2030, considering mitigation actions and targets set by governments. 5 The entry of this material in both marine and freshwater systems, as well as in terrestrial systems, causes a series of environmental risks, either because of accumulation in the environment or the formation of microplastics. 6 Microplastics are emerging pollutants that can be easily ingested by animals and, when present at the nanoscale, have permeability characteristics in some biological membranes. 7 Although there are not enough studies regarding the effects on living beings, there are hypotheses that these particles affect food safety and human health, mainly when ingested by affecting stomach and intestine, for example. 8
As an alternative to this environmental problem, studies related to innovations in biodegradable polymeric materials, from renewable sources and that present characteristic like those commonly used, are being carried out. In this scenario, polymeric films obtained from natural materials such as polysaccharides have been constantly improved.4,9,10 Among the numerous possibilities, starch films are the ones that stand out the most due to the abundance of starch in nature and can be extracted from different residues such as fruit peel and even from foods such as corn, cassava, rice1,11–13 and leftovers of harvesting and industrial processes. 14
The starch gelation allows to produce films, but its properties are not sufficient to obtain a product with desirable properties as tensile and water resistance, flexibility, and durability. Therefore, the mixture of other components improves the properties of the films allowing the use of these materials in packaging industry.2,15 In this sense, different plasticizers and proportions have been studied aiming to improve the mechanical results, permeability to water vapor, among other specific characteristics.16,17 Another alternative to produce films is to use nanocellulose, both in the form of nanofibers or nanocrystals, which also serves as a reinforcing filler.18–20 Cellulose nanocrystals (CNCs) have been highlighted for their applicability in films mainly due to their high crystallinity that creates a more tortuous path for the molecules, creating a barrier effect. 21
With the constant enhancement of technology and science, the entry of these biodegradable films into the packaging market are a reality. Thus, metallic nanoparticles such as zinc, silver, and copper, as well as titanium dioxide (TiO2), have been added in starch films due to the improving of chemical and physical properties as antimicrobial action and barrier resistance to provide greater protection to the food.22–25 Also, physical-chemical characteristics such as ultraviolet light absorption, high refractive index, and photocatalytic activity are observed in the films.26–28
Some studies can be found on literature regarding the production and characterization of starch-based films. Peighambardoust et al. 24 prepared starch-based films (St) containing single nanoparticles or a combination of Ag, ZnO and CuO (NPs) using the casting method. The physical-mechanical and antibacterial properties of films obtained were evaluated. The results showed that the St-ZnO film presented greater tensile strength compared to St/Ag and St/CuO films with 2% by weight of each nanoparticle. Furthermore, the film containing a combination of Ag-ZnO-CuO NPs showed higher tensile strength and Young's modulus than the St/Ag film. Li et al. 29 evaluated the combined effects of titanium dioxide nanoparticles (TiO2-N) and clove oil on physical-chemical, biological, and structural properties of chitosan/starch films using the casting method. The results indicated that the incorporation of TiO2-N improved the performance of the films, increasing the tensile strength and antioxidant activity, and decrease water vapor permeability. Xiong et al. 30 developed starch/titanium dioxide (TiO2) film by casting method. The mixture was composed of 60% starch and 40% glycerol, and nano-TiO2 incorporation was from 2.5 to 10% by weight compared to the blend. The results showed that the moisture content reduced from 23 to 13% and the contact angle increased from 43 to 85°, indicating that nano-TiO2 enhance the water resistance and the hydrophobicity to starch/TiO2 films. The start temperature of thermal degradation changed from 119 to 174°C, indicating better thermal stability of the starch/TiO2 bionanocomposites. In addition, there was a considerably increase in the elongation at break, from 109 to 293%, proving improvement in mechanical properties. Liu et al. 31 investigated the effects of nanotanium dioxide (nano-TiO2) on high amylose starch in polyvinyl alcohol (PVA) films prepared by the casting method. The results showed that at a concentration of 0.6% of nano-TiO2, the film demonstrated the higher tensile strength (9.53 MPa), and the elongation at break was observed as 49.50%. The antimicrobial activities of the films showed that there were inhibitory zones around the circular film disk, which can be attributed to the addition of TiO2 nanoparticles. Iacovone et al. 32 studied the effect of adding titanium dioxide nanoparticles (TiO2) on morphology, physicochemical properties, and biodegradation under industrial composting in cassava starch-based films obtained by extrusion at different screw speeds (80 and 120 r/min). The films produced at 120 r/min showed fully processed starch and homogeneously distributed nanoparticles, leading to greater flexibility than those obtained at 80 r/min. The incorporation of TiO2 led to an increase in modulus storage of all films. Furthermore, the films produced at 120 r/min with TiO2 showed effective protection against UV light, higher hydrophobicity, and faster biodegradation, resulting in a promising material for food packaging applications.
Given the importance of developing alternative materials to petroleum derived polymers, this work aims to develop a biodegradable starch film, combining the characteristics of glycerol and sorbitol plasticizers, cellulose nanocrystals, and titanium dioxide nanoparticles. The material was evaluated regarding morphological, mechanical, optical, and thermal properties aiming potential applications in food packaging industry. The combination of cellulose nanocrystal with dioxide titanium seems to be very promisor in the field of packing due to the achieve of characteristics obtained.
Materials and methods
Materials
In this work, modified corn starch (AMD 12) was obtained from the partial acid hydrolysis of corn from Adicel Indústria e Comércio Ltda. (Liberdade city, Brazil), with a maximum moisture content of 14% and pH from 4.5 to 6.0. The microcrystalline cellulose (MCC) used was from Sigma-Aldrich (Barueri city, Brazil), (lot MKCJ3222) with an average diameter of 20 μm. The plasticizers, glycerol, sorbitol, and titanium dioxide were purchased from Sigma-Aldrich (Barueri city, Brazil). The titanium dioxide was used as an aqueous suspension (40% by mass) consisting of a rutile and anatase phases.
Methodology
Production of Cellulose nanocrystalss
A suspension containing 4.5% (w/w) MCC in deionized water was grounded in a Supermass Colloider type ultrafine friction mill (MKCA6-2, Masuko Sanguo, Japan). A total of 150 passes at a rotation of 1500 r/min (central electrical control box) was used and the final suspension was refrigerated at 4°C.
The principle of this method consists in the breakdown of the cell wall structure due to the shear forces generated by the grinding stones. The pulp was passed between a static grinding stone and a rotating grinding stone, rotating at about 1500 r/min. The nanofibers that make up the cell wall in a multilayer structure will thus be individualized from the pulp. 33 This method, as well as the parameters used, were cited by several authors34–38 to whose reported the formation of cellulose nanofibers, with diameter ranging from 50 to 90 nm and apparent density estimated at 0.414g cm−3, confirmed by the results of transmission electron microscopy (TEM) and Field Emission Gun – Scanning Electron Microscope (FEG-SEM) techniques.
Starch film preparation
Identification and composition of the developed starch films.
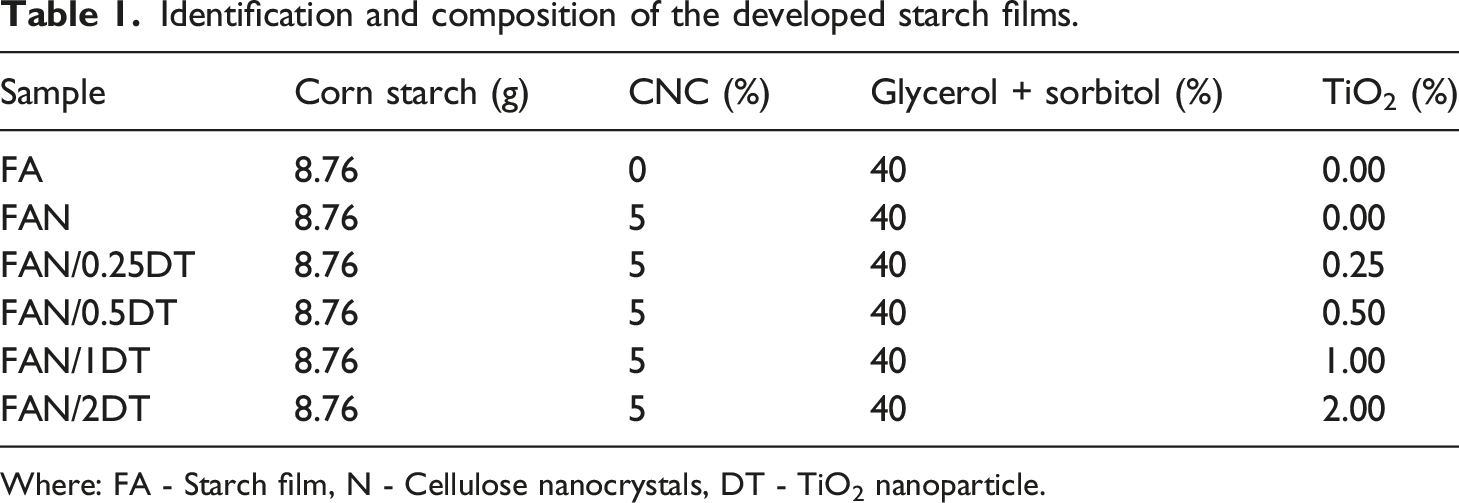
Where: FA - Starch film, N - Cellulose nanocrystals, DT - TiO2 nanoparticle.
Characterization
Scanning electron microscopy
The morphological characterizations of the starch films were carried out in a scanning electron microscope (SEM) (CARL ZEISS LS-10, Germany) at an accelerating voltage of 10 kV. The analysis was conducted under low vacuum and without surface metallization of the sample, using the backscattered electron detector (BSD). The surface porosity of the films was calculated using the OriginLab 2020b. More details and the complete procedure can be found on Abdullah & Khairurrijal. 42
Water Solubility
The percentage of water solubility (WS) was determined according to Oleyaei et al.
43
through the percentage of film dry matter remaining after immersion in deionized water for 24 h. Film samples (dimensions of 20 × 40 mm) were dried in a tray dryer (DeLeo) for 24 h at 100°C. After drying, the films were immersed in deionized water at room temperature (20 ± 2°C) for 24 h, with occasional stirring. The samples were removed from the water and dried in a tray dryer until reaching constant mass. Thus, the water solubility was calculated according to Equation (1), and the test was performed in triplicate.
Moisture content of films
The percentage of moisture content of films (
Thickness
The thickness of the starch films (T) was obtained with a micrometer (Pantec Digital 0–25 mm), and the result was evaluated from the average of 3 measurements. The readings were performed at 5 points on the film, being dispersed at 4 ends and one at the center of the film.
Tensile strength analysis
A DMA Q800 (TA Instruments) from the Polymer Laboratory of the University of Caxias do Sul was used for the stress–strain analysis. The parameters for the tests were: isothermal temperature of 25 ± 2°C, with a ramp force of 0.5 N/min, and a force range between 0.001 and 18 N, using the clamp film tension. The dimensions of the rectangular samples were approximately 10 mm × 5.3 mm. The film thickness (average of three measurements) was measured using a micrometer.
Color
The methodology used was described by Arifin et al.
44
in which a CM-5 spectrophotometer (Konica Minolta, Japan) was used to measure the color value of the samples. L*, a* and b* are the colorimetric parameters of the standard color surface. The results were presented in terms of 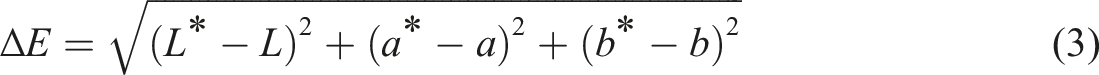
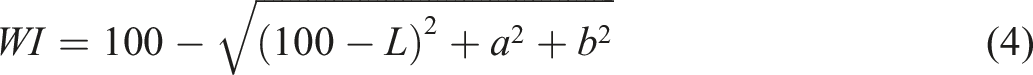
Opacity
The UV-Vis transmission spectra of the samples were analyzed from 200 to 800 nm in a DU530 UV/Vis spectrophotometer (Beckman, United States). Films were inserted into a cuvette and air was applied as a reference. The opacity of the films was obtained using Equation (6).
46


Fourier transform infrared spectroscopy
The starch films were performed using an infrared spectrophotometer (Perkin Elmer Frontier, USA) with an attenuated total reflectance (ATR) accessory. Each spectrum was obtained using 32 scans ranging from 4000 to 400 cm−1 at a resolution of 4 cm−1. The intensity ratios of peaks 1047/1022 and 3300/1149 were evaluated according to Piñeros-Hernandez et al. 47
Thermogravimetric analysis
The analysis was performed in a thermogravimetric analyzer (Shimadzu TGA-50) using approximately 10 mg of each sample. The heating rate was performed at 10°C/min from 25 to 700°C using N2 gas atmosphere at a rate of 50 mL/min.
Results and discussion
Morphological evaluation of films
Figure 1 shows the surface morphology of the different starch films analyzed by SEM. Samples using CNC and TiO2 showed small differences regarding the surface roughness. It is observed the formation of agglomerates at some degree, mainly for the FAN sample compared to the FA. Also, no cracks or pores on the surface were observed for all samples. However, the FA sample, without the incorporation of nanofillers, presented a smoother surface (at least visually), without roughness or defects. According to Rico et al.
48
this is indicative that the processing conditions were adequate, with complete starch gelatinization. SEM of the developed starch films.
Figure 2 presents the method to obtain the surface porosity of the films, where the surface roughness was quantitatively mesaured. Table 2 presents the results of surface porosity following Abdullah & Khairurrijal
42
procedure. These characteristics were also observed by Alizadeh-Sani et al.
25
which their films showed good homogeneity and uniformity. By increasing nanofiber content, there is a trend to form cellulose nanofiber (CNF) clusters in the bionanocomposites, as observed for whey protein, CNF, TiO2 nanoparticles, and rosemary essential oil. The authors attribute the formation of agglomerates to the tension of the nanofibers, the lack of dispersion on the surface, and the reduced adhesion of the nanofibers to the polymeric chains due to the reduction of intermolecular forces between the different matrices. TiO2 has also been reported as a cause of agglomeration in films and biocomposites, which also affects the properties of the material and may be related to the high surface energy of the particles, as well as its low affinity for starch.40,49 This is explained by the strong attraction of Van der Waals forces on particles with smaller sizes. Thus, nanomaterials have significant interactions between their molecules, causing agglomerations. Thus, it is known that factors such as the drying and agitation process responsible for the dispersion of nanomaterials directly affect the formation of agglomerates.50,51 Schematic representation of the surface porosity of the FAN sample. The lines highlighted in yellow are referred to the parameter obtained using Originlab. Surface porosity of the samples studied.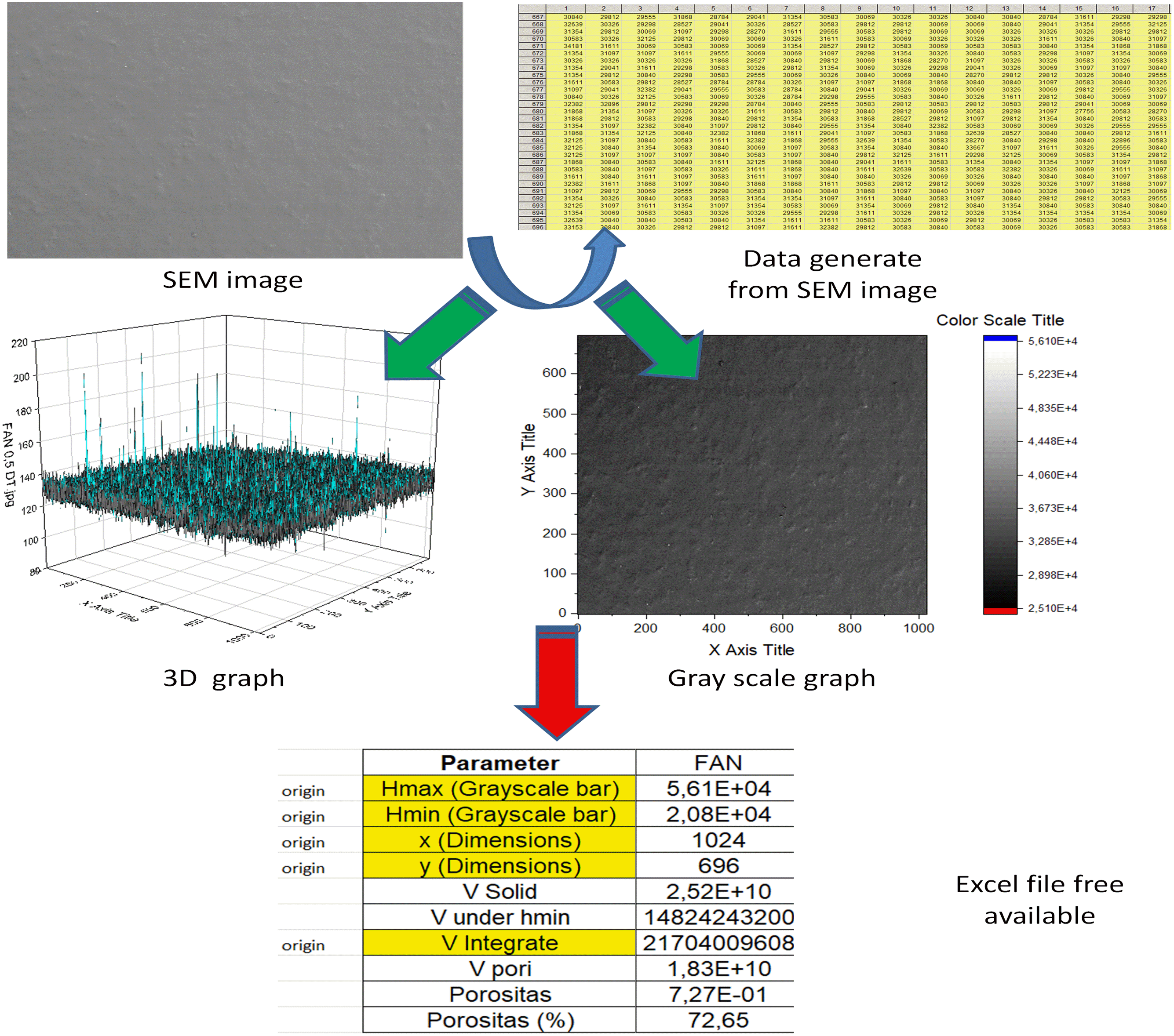
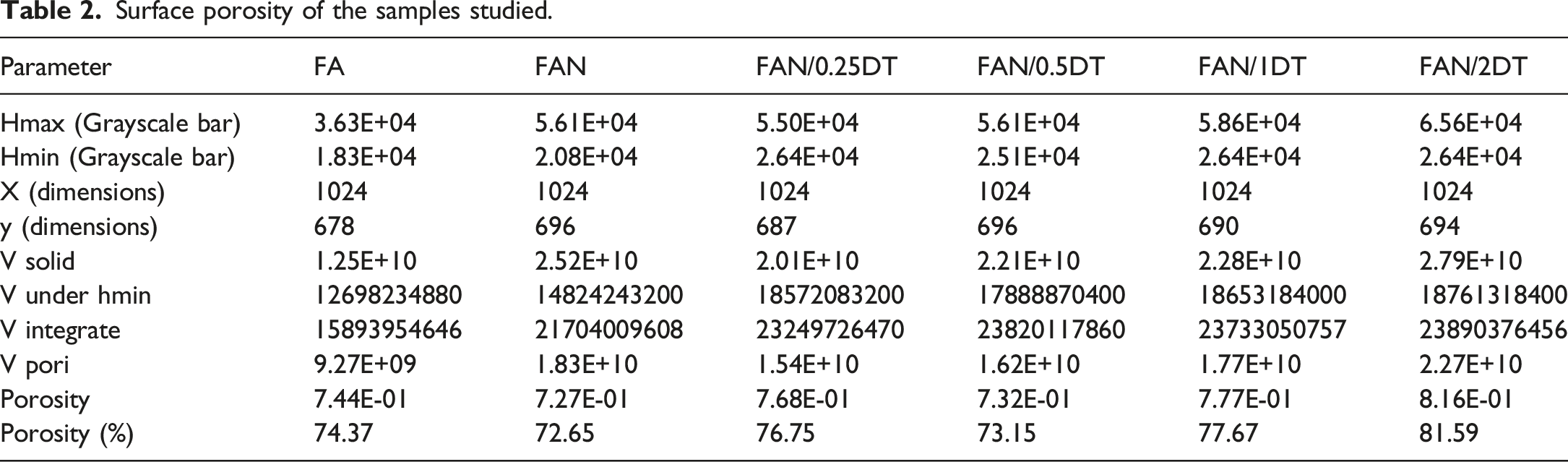
According to Table 2, the quantitative analysis showed that there is a trend to increase the surface porosity by increasing the TiO2 nanoparticle. This difference is not easily noted by visually comparing the samples. The increasing in the surface porosity by increasing the TiO2 nanoparticle seems to make sense due to the possibility to form agglomerates. Also, the slight decrease in porosity compared FA to the FAN, despite the incorporation of cellulose nanocrystals. This type of analysis showed to be very useful, mainly because the errors related to the visual analysis are eliminated.
Water solubility, moisture content, thickness, and mechanical properties
Properties of the developed starch films: water solubility (WS), moisture (M), thickness (T), tensile strength at break and maximum elongation.
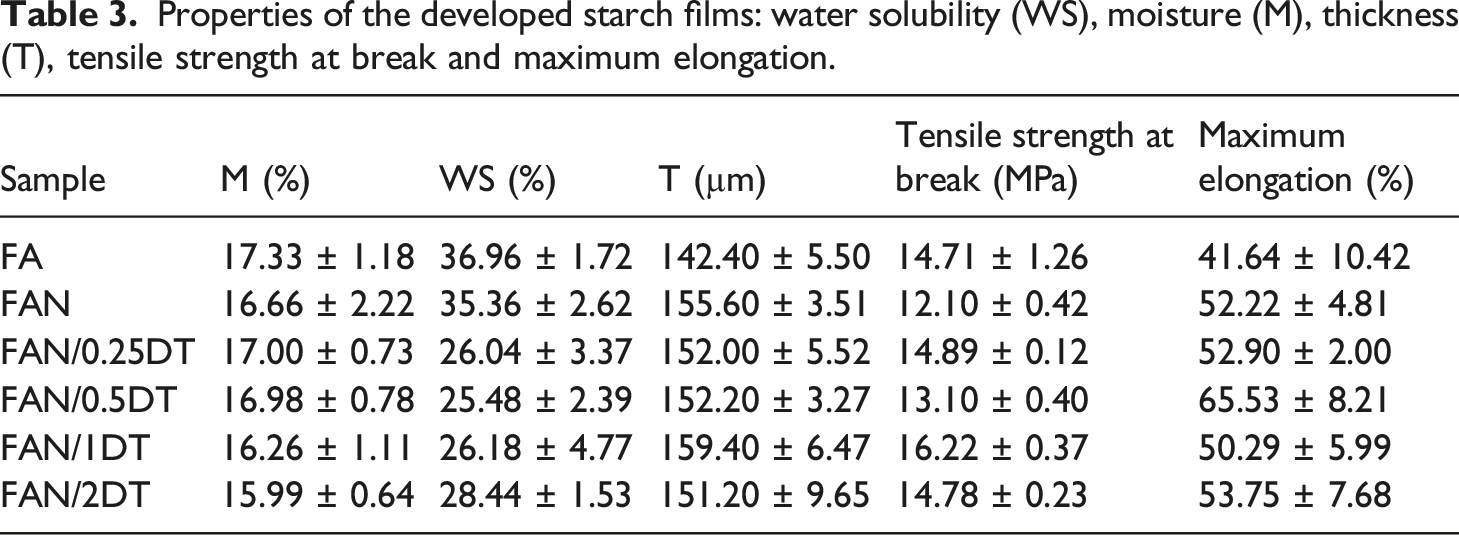
Moisture content is crucial in the films because water acts as a plasticizing agent, which facilitates the mobility of molecules and ensures greater flexibility of the films.52,53 It is observed that the incorporation of TiO2 and CNC did not alter the moisture values found for the different films (Table 3). This behavior was also evidenced by Rong et al. 54 in starch films obtained from hyacinth beans with the incorporation of TiO2 nanoparticles and Mesona chinensis Benth. Goudarzi et al. 40 also found an insignificant difference in moisture contents for the samples of starch films with and without the presence of TiO2. Other moisture results presented in the literature ranged from 18.16 to 16.39% in starch films with concentrations of 0 to 5 wt% TiO2 respectively, 40 and 17.00% in starch films containing 5 wt% CNC from grape pomace. 55
The WS results showed that with the increase in TiO2 incorporation, there is a decrease in the solubility of the film in water, which may be related to the low solubility of TiO2 molecules compared to the other components of the film, according to Oleyaei et al. 43 A similar result was observed by Hajizadeh et al. 56 who evaluated starch films with nanoparticles of TiO2 and SiO2 with modified Ag. The decrease in solubility was linked to the addition of nanoparticles, an important characteristic for application in food packaging. 1
The thickness increased with CNC. However, different concentrations of TiO2 did not interfere in the thickness. These thickness values agreed with studies in the literature. Goudarzi et al. 40 evaluated starch films with TiO2 in concentrations from 0 to 5 wt% and obtained thicknesses of 0.14 to 0.11 mm, respectively. Coelho et al. 55 prepared starch films with CNC from grape pomace and obtained a result of 0.08 to 0.11 mm in concentrations from 1 to 15 wt% of CNC. According to Arifin et al. 44 the increase in thickness due the presence of CNC is justified by its structure of long chains that serve as filling for the starch film, and that provide a strong interaction with the matrix.
To meet demand and be used as food packaging, starch films need reasonable tensile strength and elongation at break, 57 as they are subject to various types of stress mechanical during use. 40 According to Bangar et al. 58 the mechanical characteristics of starch films depend on intrinsic properties, such as shape, arrangement, quantity, nanoparticle size, and capacity of the biopolymer matrix to transfer stress.
The incorporation of 5 wt% of CNC caused a reduction in the tensile strength of the films with only starch (FA). In a study carried out by Yang et al. 57 using cellulose nanocrystals (0.1 to 3 wt%) in starch films, the authors observed that the tensile strength of the composite film increased by CNC content. But a decreasing of the tensile strength was observed at higher CNC amounts. When adding 1 wt% of TiO2 nanoparticles an increase of 34% in the tensile strength of the film in relation to the control sample (FAN) was observed. The elongation at break increased by 25% compared to the starch sample (FA) with the addition of 5 wt% CNCs. The increase in flexibility may be due to the increased synergy of a flexible starch matrix with the CNCs, and the effective transfer of stress through good interfacial adhesion between the components.9,19
The incorporation of TiO2 nanoparticles increased the tensile strength and elongation at rupture of the films, independentely of the content used. The FAN/1DT sample had the higher tensile strength value (16.22 MPa) and the FAN/0.5DT sample the higher elongation at break value (65.53%). A similar behavior was observed by Goudarzi et al. 40 when evaluating wheat starch films with different concentrations of TiO2 (1, 3, and 5 wt% of dry base). The increase in tensile strength and elongation at break, with respect to the starch film (FA), can be attributed to the reduction of crosslinking in the nanocomposite matrix and the increase in the polymer chain mobility. 40 Menezes et al. 45 suggested that adequate amounts of TiO2 nanoparticles can provide improvement in mechanical properties through electrostatic interactions and hydrogen bonds.
Color
Color analysis of the developed starch films.
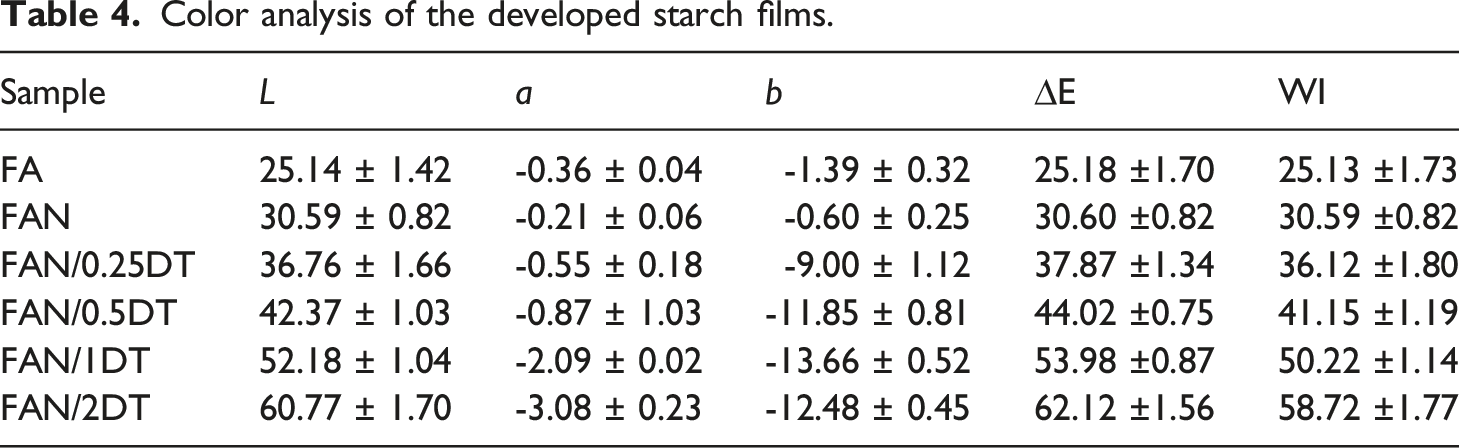
The visual characteristic of films is very important for the packaging industry since food packaging is the main way of presenting and protecting products. In Figure 3 is observed a tendency for the appearance of white and blue at higher TiO2 concentrations. The FA and FAN films had the highest transparency and as the TiO2 concentration increases, the films become more opaque. Similar behavior was observed by Alizadeh-Sani et al.
25
when analyzing n whey protein isolate films with nanocellulose and TiO2 (0.5, 1 and 1.5% w/w based on whey protein isolate). Visual analysis of the developed starch films.
Similar behavior was reported by Li et al. 29 in their research, where the authors attributed the increase in tinting of the films to the whitening characteristic of TiO2. Likewise, Zhang et al. 59 observed a higher value of L for the chitosan film in the presence of TiO2 particles, when compared to the control. Furthermore, the values of “a” did not show great variations, as also observed by Goudarzi et al. 40 in starch films with addition of TiO2.
Opacity
Food contact with ultraviolet (UV) light can negatively influence the quality of the product, due to the release of free radicals responsible for oxidation, destruction of vitamins and proteins, in addition to decreasing the antioxidant capacity.
60
In this way, particles that have blocking capacity, such as TiO2, become allies in the elaboration of new materials.
61
Figure 4 presents the behavior of the tested starch films, in the range from 200 to 800 nm. UV-Vis spectra of developed starch films.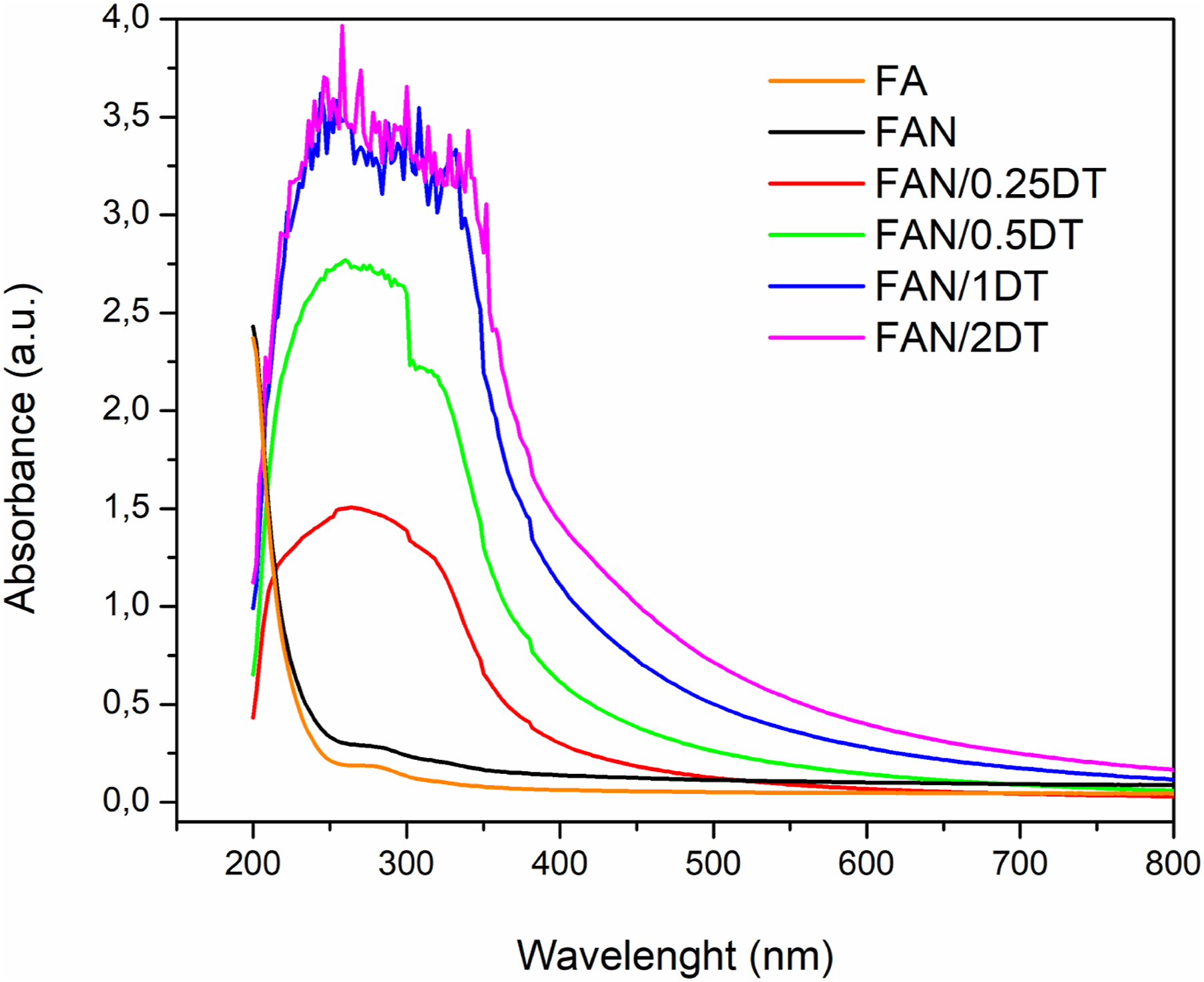
It is observed that the incorporation of nanocellulose (FAN) caused an increase in the L value and of opacity in the films. This can be attributed to the occupation of cellulose free volume space in the starch network, due to the interaction between the starch matrix and CNCs, thereby increasing light scattering and increasing of L.14,20 It is noticed that samples with higher TiO2 content showed greater absorption of UV light. Furthermore, there is a significant improvement in blocking the light beams of samples containing nanoparticles compared to samples composed only of starch, plasticizer, and CNC. TiO2 acts in such a way as to absorb the energy of the photons to excite the electrons in the valence band, to reach the conduction band after overcoming the gap it has in the range of 385 nm. Also, its high refractive index leads to diffuse reflection of light, which justifies the reduced transparency of films.31,62
Transmittance (%) and opacity of the developed starch films.
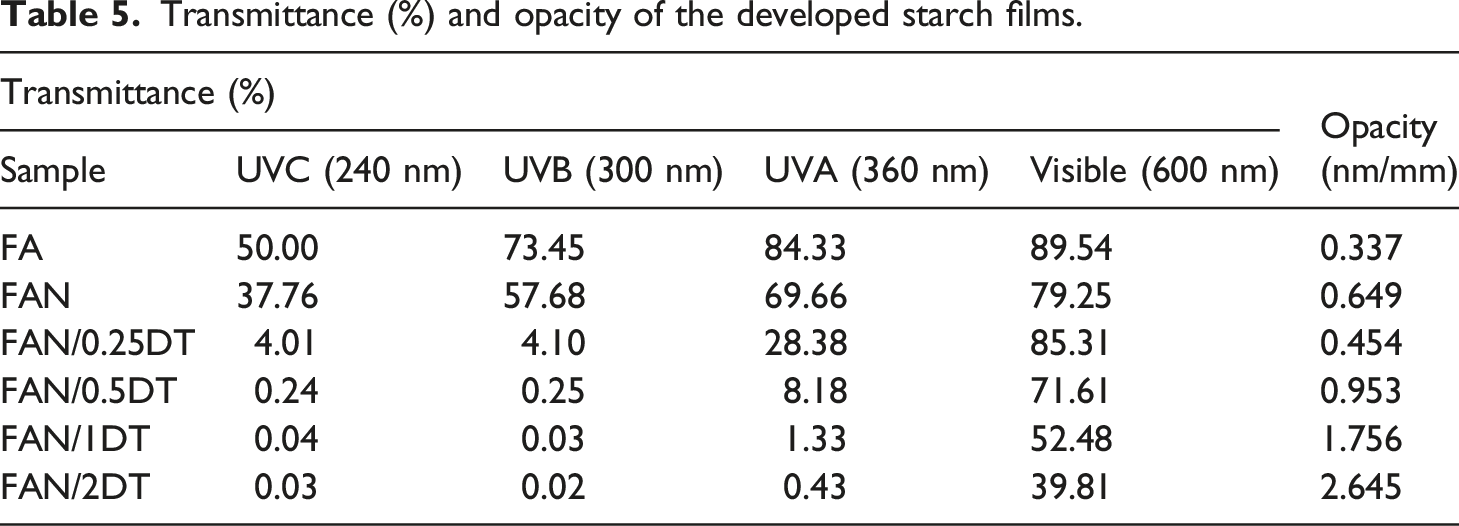
Observing the results, is concluded that the incorporation of 2% by mass of TiO2 nanoparticles promotes better opacity properties and, therefore, prevention of damage caused by UV-visible radiation. Oleyaei et al. 43 found in starch films with addition of 0, 0.5, 1, and 2 wt% TiO2 opacity from 0.32 to 6.70 nm/mm with transmittance of 0.02% (UVC, UVB and UVA) and 5.61% (Visible) at the highest TiO2 concentration. Noorian et al. 62 observed in films of starch/TiO2 and essential oil of Mentha piperita, a significant reduction in transmittance between the control sample (starch) and the other films containing 1, 3, and 5 wt% of TiO2. Yousefi et al. 46 found better opacity results with the incorporation of 1, 2, and 4 wt% TiO2, with values of 1.558, 1.835, and 3.45 nn/mm, respectively, in starch, montmorillonite, and TiO2 films.
Fourier transform infrared spectroscopy
FTIR-ATR spectra of the films are shown in Figure 5. It is observed that all the samples showed the same behavior, demonstrating that there was no formation of a new bonds/chemical reactions when compared to the control sample FA. FTIR-ATR spectra of the developed starch films.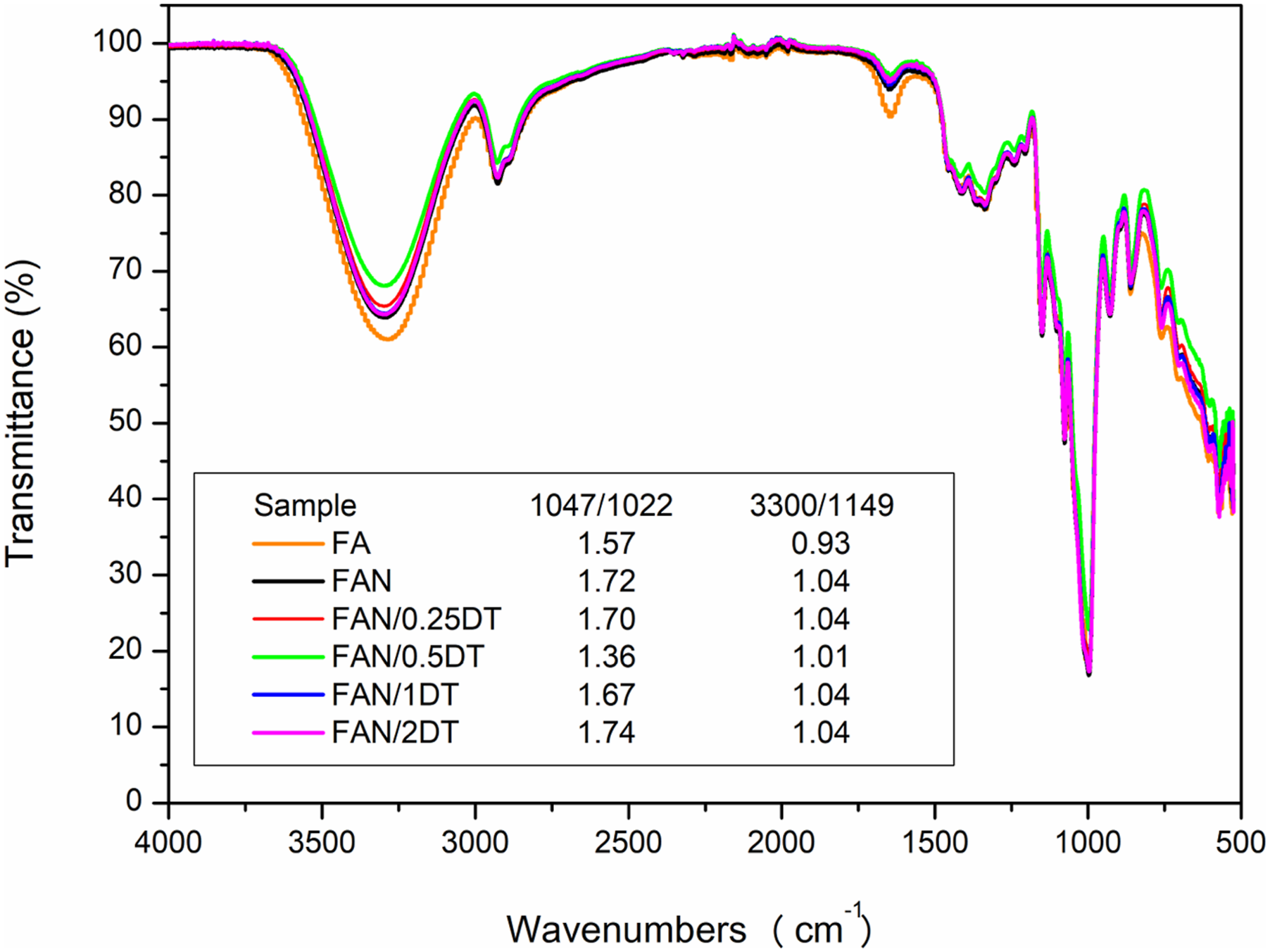
The band observed at 3280 cm−1 is characteristic of OH elongation, 63 indicating the formation of intramolecular hydrogen bonds in starch, plasticizers, and CNC. 9 The peak located at 2933 cm−1 corresponded to the C-H elongation. At 1640 cm−1, an elongation of C=O is observed, and at 1149 cm−1 it corresponds to the elongation of the C-O bond. Similar results were found by Amin et al. 64 in starch films with addition of TiO2. The peak at 1635 cm−1 is also related to the presence of water absorbed in the amorphous regions of the starch and the asymmetric stretching vibrations of the O-H groups in the glycerol. 3
Although there were no additional peaks, intensity spectra changes were observed. Thus, the degree of crystalline order in starch materials can be evaluated through the intensity ratio between peaks 1047/1022, as these bands are related to the crystalline and amorphous organization of starch, respectively.
Lower values indicate the formation of amorphous material. 65 In this sense, a maximum value of 1.74 was observed in the FAN/2DT sample, which indicates the formation of an ordered structure, unlike the FAN/0.5DT sample, which resulted in 1.36, which may be related to the unlikely recrystallization of starch in this condition, an effect also observed by Kochkina & Butikova 66 in starch/PVA/TiO2 films. Except for the latter, all other samples showed values greater than the FA control (1.57), demonstrating that the addition of CNC and TiO2 was beneficial to the structure of the films.
The intensity ratio 3300/1149 serves as a parameter to evaluate the strength of interactions between molecules. Hydrogen bonds are responsible for such interactions, and therefore, the smaller the ratio between the peak of the OH group (3300) and the C-O of the C-O-H bond (1149), the smaller the number of available hydroxyls. 47 All samples containing CNCs and TiO2 showed higher values than the FA sample, because the greater the availability of OH groups, the greater the probability of forming hydrogen bonds with water.
Thermogravimetric analysis
TGA results are shown in Figure 6. Table 6 presents the mass loss temperature with 5 (T5%) and 20% (T20%), in addition to presenting the residue at 650°C. TGA result of the developed starch films. TGA result of the developed starch films. where: T5% represents the loss of 5% of the mass of the sample and T20% represents the loss of 20% of the mass of the sample.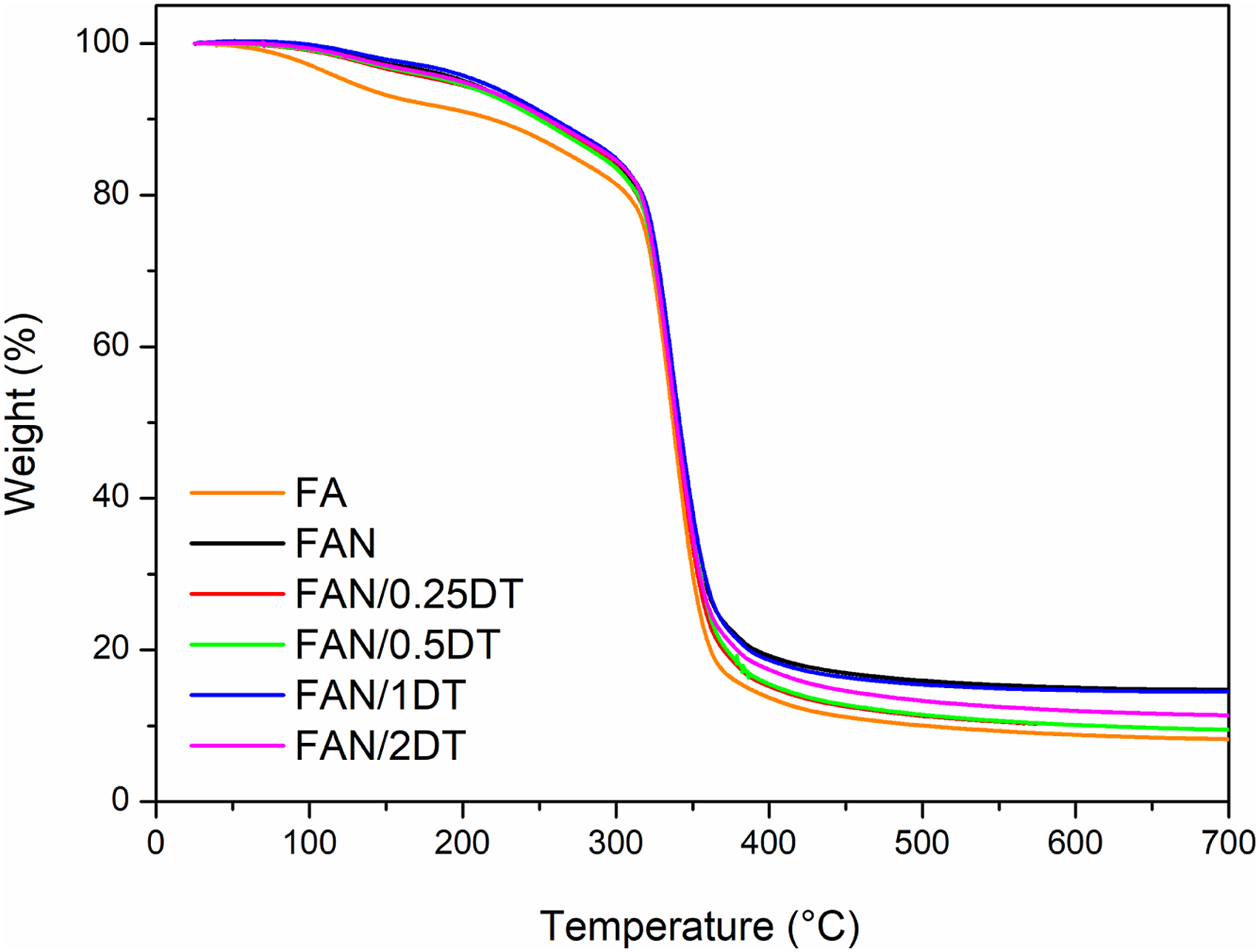
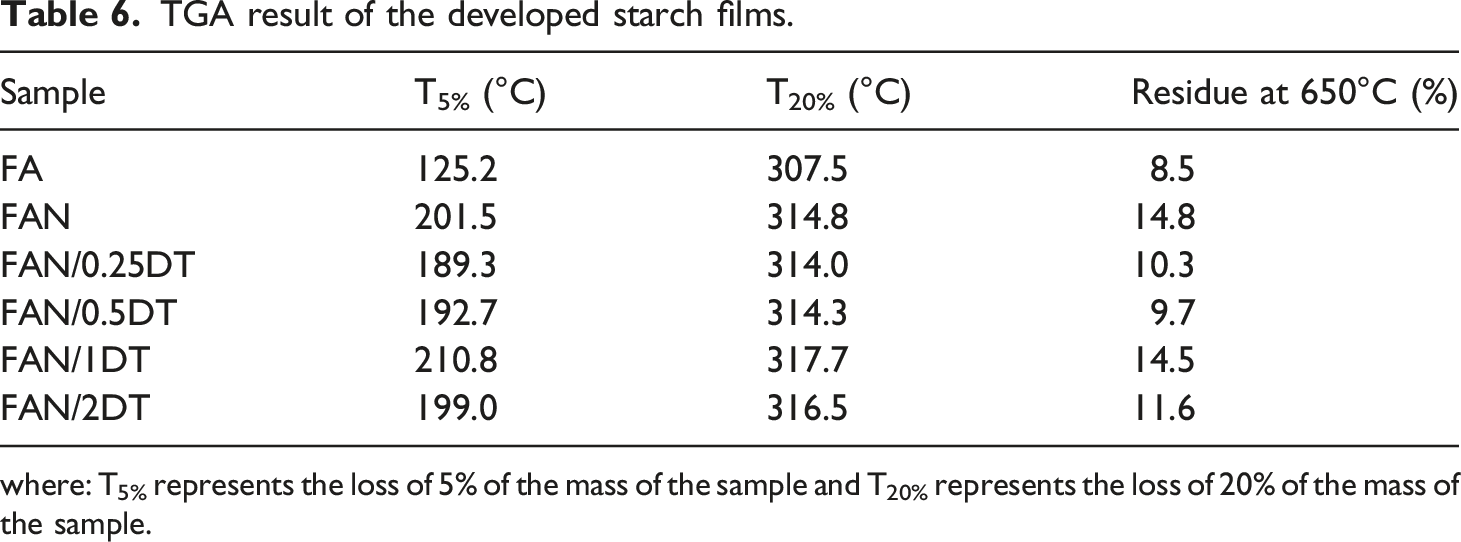
Is observed from Figure 6 the presence of three events of mass loss for the films. The first stage (from 50 to 120°C) is related to the evaporation of loss of water through evaporation and the sublimation of low molecular weight substances. Still at this stage, it is noticed that there is a more accentuated mass loss of the FA film compared to the others due to the smaller number of solids in the sample. The second stage is characterized by starch degradation due to depolymerization and decomposition, from 200 to 400°C. This is in accordance with Chen et al. 67 that evaluated corn starch films in the presence of nanocellulose, and Paiva et al. 68 that evaluated corn starch films with chitosan. The thermal degradation of glycerol occured up to 250°C and of sorbitol in the range of 270 to 400°C. 48 The third stage of degradation occured above 400°C and is related to the degradation of starch and cellulose polysaccharides. 69
The initial degradation temperature of the samples with 5% mass loss is presented at Table 5. Therefore, the addition of CNC and TiO2 improved the thermal stability of the films, but there was no correlation between the increase in TiO2 content. The same behavior occurred in the percentage of 20% mass loss. The FAN/1DT film presented greater thermal stability among all films. Regarding CNC, this result can be explained by the increase in crystallinity and char formation that hinder the release of volatile by-products, thus increasing thermal stability. 70 Residues from pyrolysis at 700°C are related to non-volatile materials and materials the effect of the char formed by degraded starch. 71 So the increase in the percentage of residues is related to the incorporation of more solids in relation to the dry mass of starch, causing a reduction in the amount of water in the sample.
Conclusion
In this work, the development of corn starch films with cellulose nanocrystals and titanium dioxide nanoparticles has been successfully produced. The corn starch films with CNCs and TiO2 improved most properties analyzed. A reduction of solubility in water from 35.36% to 25.48% in the condition of 0.25 wt% TiO2; increased UV light blocking in all ranges tested, especially the film with 2 wt% TiO2 which increased blocking by 307%. However, the samples showed a high white coloration, characteristic of the presence of metallic oxide and rough surface with the incorporation of TiO2 and CNCs. Furthermore, better thermal stability of the films was observed with an increase in the initial degradation temperature. It is observed too that the incorporation of TiO2 nanoparticles increases the tensile strength and elongation at rupture of the films. Thus, it was observed that the use of nanoparticles of TiO2 and CNCs provided obtaining starch films with superior properties, demonstrating potential for use as biodegradable films in food packaging.
Footnotes
Acknowledgements
The authors thank Universidade do Vale do Taquari_Univates and Universidade de Caxias do Sul (UCS) for supporting this research, and FAPERGS for financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Credit authorship contribution statement
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Aluisie Picolotto, Cleide Borsoi, Danielli Dallé and André Luis Catto. The first draft of the manuscript was written by Cleide Borsoi, Heitor Luiz Ornaghi Jr and Lilian Vanessa Rossa Beltrami and all authors commented on previous versions of the manuscript. Ademir José Zattera, funding acquisition and Resources. All authors read and approved the final manuscript.
