Abstract
Current challenges associated with titanium (Ti) and its alloys in biomedical applications motivate researchers to find alternatives. Many researchers suggest the potential benefits of nanoparticles (NPs) due to their novel characteristics. Considering this, different percentages of silver nanoparticles (AgNPs), zirconium dioxide nanoparticles (ZrO2NPs), and hydroxyapatite nanoparticles (HANPs) have been reinforced with titanium dioxide (TiO2) to form titanium-based nanocomposites by applying sintering method. The developed nanocomposite samples (sample 1 and sample 2) have been characterized by hardness, surface roughness, Fourier Transformed Infrared Spectroscopy (FTIR), Field Emission Scanning Electron Microscopy (FESEM), and X-ray diffraction analyses (XRD). Maximum hardness 49 in Rockwell hardness in C scale has been obtained from sample 2 and minimum hardness 42 from sample 1: the highest average surface roughness, 6.5 µm, received from sample 1 and minimum average surface roughness 5.2 µm from sample 2. Higher average surface roughness helps to improve the osseointegration properties of the materials. The FTIR spectra identified different functional groups (-OH, C-H, and Ti-OH). The NPs are uniformly distributed in the samples. The presence of micropores is identified, which may influence the mechanical properties of the materials. Sharp peaks formed in the XRD spectra confirm the crystallinity of the developed samples.
Keywords
Introduction
Oral diseases are prevalent among public health diseases globally with very high incidence and prevalence. 1 Both local and general health can be jeopardized by oral disorders such as dental caries and periodontitis. Dental caries destroy tooth hard tissues, and tooth-supporting tissues are destroyed by periodontitis. 2 Around 11.2% of people globally are affected by periodontitis at any stage of their life cycle. 3 Other diseases, such as Alzheimer’s, diabetes, and atherosclerosis, are also related to periodontitis. 4 A person’s oral health can be maintained, and general health can be promoted through an achievable plan such as restoration using different materials.
The restoration materials are exposed to abrasion, high masticatory force, saliva, and oral microbiota, which can also cause secondary caries. 5 The missing teeth are currently substituted by dental implants in conventional treatment.6,7 Besides, the average surface roughness of dental materials plays a crucial role in the adhesion and accumulation of biofilm. Rougher surfaces help improve dental materials’ osseointegration properties by improving bone-to-implementation contact.8–10 Moreover, the artificial dental materials should have percentages of crystallinity close to natural dental materials. Titanium and its alloys are the most widely used materials as dental implants worldwide due to their favorable biocompatibility. 11 However, failure in osseointegration and bone loss due to the generation of ions into peri-implant tissues is a common problem with titanium. 12 Therefore, developing novel dentistry materials continuously for dental applications to obtain desired properties is necessary.
Nanomaterials contribute to the development of dental materials and thus are helping to overcome the current defects.13,14 Nanomaterials have significant advantages compared to bulk materials in interacting with other particles, making them useful for diagnostic and therapeutic purposes. 15 Nanomaterials can directly interact with the bacterial cell wall, show superior antibacterial activity, and keep the surrounding environment safe. 16 Nanomaterials are less harmful than other materials and more effective against bacteria.17–19 Including nanomaterials with other particles can contribute to treating infections related to teeth. 20 Commonly used nanomaterials in dentistry are Ag, ZnO, ZrO2, CuO, TiO2, HA, graphene, etc.
Titanium-based nanocomposites have been fabricated in this research work, reinforcing silver nanoparticles (AgNPs), zirconia nanoparticles (ZrO2NPs), and hydroxyapatite nanoparticles (HANPs). AgNPs are extensively used in different applications such as industrial, commercial, energy storage, medical, and agriculture. 21 AgNPs have excellent antimicrobial properties, making them applicable in bacteria-rich environments.22–25 Excellent biocompatibility made ZrO2NPs applicable in orthopedic applications, especially for replacing damaged and sick portions of teeth, bones, and joints and repairing them. Due to its low toxicity, it can be reinforced with other materials and can be used in biomedical applications such as theragnosis, biolabeling, and bioimaging.26,27 HANPs have a chemical structure similar to human bone tissue, making them excellent candidates for treating bone-related diseases.28,29 It can store and release different types of ions, which helps kill bacteria and keep the surrounding environment safe. 30
There is a growing concern about the biological safety of NPs. The toxicity of NPs depends on the synthesis process. However, green synthesis is considered a safe process. Literature suggests that green synthesized AgNPs and ZrO2NPs are biologically safe with zero cytotoxic effect.31,32
Observing the excellent and promising properties of AgNPs, ZrO2NPs, and HANPs and their potential applications, they have been reinforced with TiO2 to fabricate nanocomposites for dental applications. Different physical and morphological analyses have characterized the fabricated nanocomposites. This paper’s novelty is that these nanoparticles have novel properties and have not been used before with TiO2. Moreover, the obtained data from the characterization techniques show potentiality in biomedical fields.
Methodology
Materials
In this research, titanium-based nanocomposite samples have been fabricated using cold sintering methods to reinforce AgNPs, ZrO2NPs, and nano-HA. TiO2 was used here as base material, collected from Sisco Research Laboratories Pvt. Ltd. Maharashtra, India, with a purity of 98%. We have synthesized AgNPs, ZrO2NPs, and nano-HA. The synthesis process and the properties of the NPs can be found in the literature.33–35
Development of the nanocomposite samples
Ratio of the constituents in sample 1 and sample 2.

Schematic diagram for sample preparation.
Characterization of the nanocomposite samples
Rockwell hardness test in C scale has been performed with the developed materials to observe the properties of the developed materials by employing a motorized Brinell Rockwell and Vickers hardness tester with model number HBRV-187.5. A Surtronic S128 roughness tester developed by TAYLOR HOBSON has observed the surface roughness profile. The fabricated nanocomposite samples were undergone by FTIR analysis to find the presence of different functional groups. A Shimadzu, Japan-made Fourier Transformed Infrared Spectroscopy with model number IRPrestige-21, has been used to perform the test from 4000 cm−1 to 500 cm−1. An SEM analyzer of Zeiss Sigma 300 VP made by GEMINI has been employed to observe the microstructure of the fabricated nanocomposite samples. The SEM images have been captured at different scales at an accelerating voltage of 5 kV. The crystallinity of the fabricated nanocomposite samples has been characterized by an X-ray diffraction analyzer made by Bruker, Germany. The test was performed using Cu Kα radiation, where the voltage and current were 40 kV and 35 mA, respectively.
Results and discussion
XRD analysis
The XRD spectra of the developed nanocomposite samples are shown in Figure 2 (a) and (b). It is seen from the Figure that the intensity of the spectrum increased significantly with the increase of AgNP content in the sample. Sharp peaks formed in the spectra confirm crystal formation in the samples. The characteristic peaks at 2Ѳ = 25°, 27°, 36°, 41°, 44°, 55°, 56°, 62°, and 69° corresponds to (1 0 1), (1 1 0), (1 0 1), (1 1 1), (2 1 0), (2 1 1), (2 2 0), (0 0 2), and (1 1 2), respectively, for sample 1; 2Ѳ = 25°, 27°, 36°, 41°, 44°, 54°, 56°, 62°, and 69° corresponds to (1 0 1), (1 1 0), (1 0 1), (1 1 1), (2 1 0), (2 1 1), (2 2 0), (0 0 (2), and (1 1 2), respectively, for sample 2. XRD spectra of the developed nanocomposite samples (a) sample 1 and (b) sample 2.
Hardness and average surface roughness analysis
Figure 3(a) shows the hardness results obtained from the developed nanocomposite samples after the hardness test. Both samples showed excellent hardness properties due to the reinforcement of the NPs. However, sample 2 shows improved hardness due to the higher percentages of AgNP reinforcement, the smaller sizes of NPs, and the physical crosslinks form, ultimately enhancing the nanocomposite’s hardness.
36
Higher percentages of AgNPs provide more crosslink, which gives the nanocomposites better hardness. (a) Hardness and (b) average surface roughness analysis of the developed nanocomposite samples.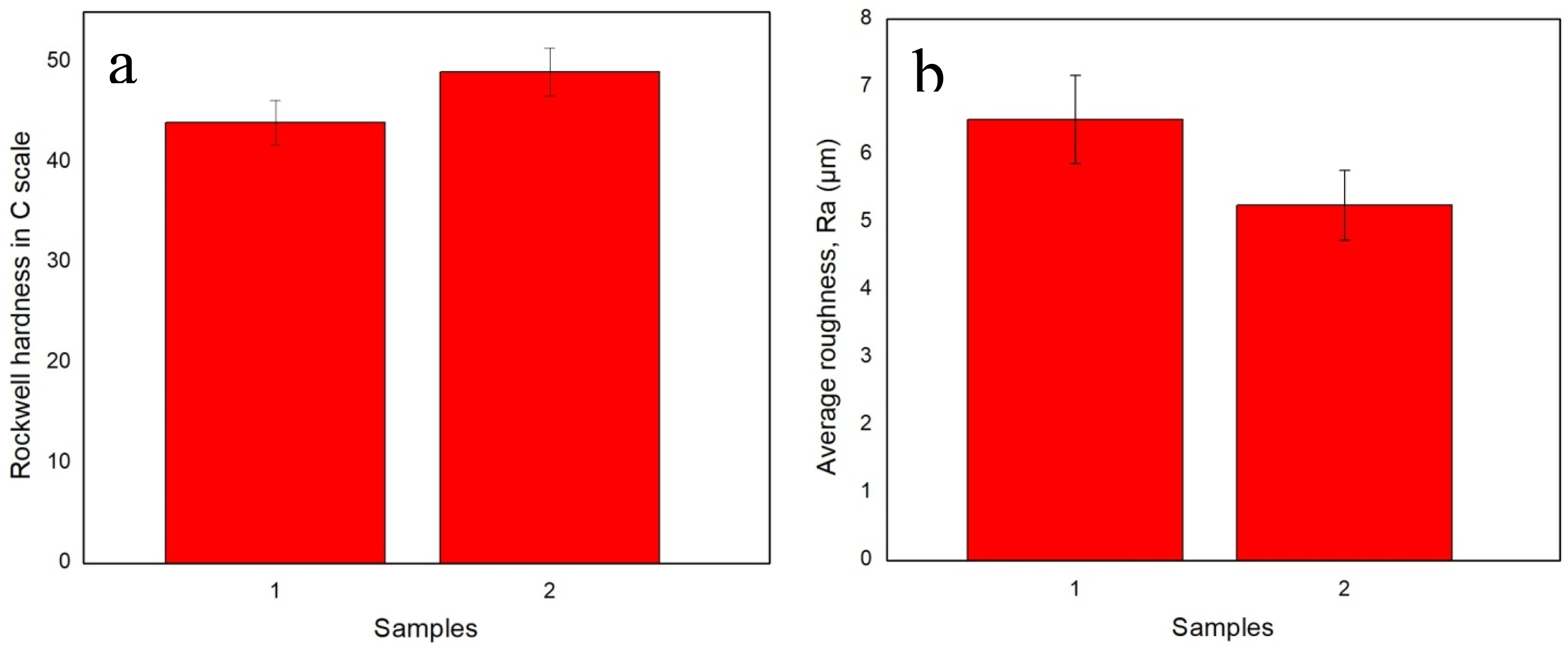
Comparison of average surface roughness with the data available in the literature.

FTIR analysis
Figure 4 shows the FTIR spectra of the developed nanocomposite samples from 4000 cm−1 to 1000 cm−1. The spectra have identified the presence of different functional groups. The functional groups of the spectra obtained can establish a correlation between the biological and chemical processes.
54
The spectra show that the transmittance of the nanocomposite samples increased due to the reinforcement of higher percentages of AgNPs (Figure 4(b)). The presence of Ti-OH has been identified by the broad peak at around 1718 cm−1 by both samples,
55
making the samples biocompatible.
56
The sharp peak at around 2875 cm−1 is responsible for the presence of C-H. This improves the antimicrobial performance of the developed nanocomposite samples.
57
The broad peak at around 3406 cm−1 confirms the presence of –OH.
58
The presence of –OH improves the osseointegration performance of the materials.
59
FTIR analysis of the developed nanocomposite samples.
FESEM analysis
Figures 5(a)–(d) and 6(a)–(d) show the FESEM images of the developed nanocomposite samples at different resolutions. It is observed from the pictures that the NPs are uniformly distributed in the samples. The size and shape of the NPs are visible in the images of Figures 5(a)–(d) and 6(a)–(d). Besides, microcavities are visible at higher resolutions. The presence of microcavities may affect the mechanical properties of the nanocomposites. Microcavities are less visible in sample 2, which can be a reason for the higher hardness of that sample. SEM images of the developed nanocomposite samples at (a) 20 µm, (b) 1 µm, (c) 500 nm, and (d) 300 nm. SEM images of the developed nanocomposite samples at (a) 20 µm, (b) 1 µm, (c) 500 nm, and (d) 300 nm.

Conclusion
AgNPs, ZrO2NPs, and HANPs have been reinforced with TiO2 in this research work to develop titanium-based nanocomposites using the sintering method. Different mechanical and morphological analyses characterized the developed nanocomposite samples. The obtained data indicates superior hardness from the developed nanocomposite samples, which ensures their longevity. Higher surface roughness favorable for osseointegration was achieved from both samples. Osseointegration helps to overcome implant failure. The functional groups identified in the FTIR analysis can improve biocompatibility, antimicrobial performance, and osseointegration, which are essential for dental materials. SEM images show the proper distribution of the NPs, as well as their structure and shape. The crystallinity of the samples has been identified by the sharp peaks formed in the XRD spectra. However, the developed nanocomposites show favorable mechanical and morphological characteristics, and in vitro and in vivo tests are required to find their applications in biomedical sectors.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
