Abstract
The preparation of biodegradable composite films from blending chitosan in maize starch via solution casting was explored and the effect of the ratio of chitosan/starch on the physicochemical, mechanical properties and antimicrobial activity of the films was investigated. The interaction through hydrogen bonding between starch molecules and chitosan and the reduction of crystallinity in blend films were revealed in FTIR and XRD results. The blend films with incorporation of proper amounts of chitosan to a maize starch matrix displayed an enhancement in tensile properties and water vapor barrier performance, an improvement in rigidity and antimicrobial activity accompanied by higher transparency. The film with 1/3 of chitosan/starch ratio appeared homogeneous structure, 21.54 ± 0.78 MPa of tensile strength and (2.81 ± 0.26)×10−10 g·m−1·s−1·Pa−1 of water vapor permeability which were 1.52 times higher and 60.9% lower than those of starch film, respectively. The deteriorated comprehensive performance of the films could be caused by excessively high chitosan content because of an incomplete miscibility between starch and chitosan. The chitosan/starch blend film possessed reasonable thermal stability and it could potentially be useful for food packaging applications.
Introduction
The traditional packaging materials are assembled with non-renewable resources and drastically generate serious environmental problem. 1 Consequently, there is a growing demand for bio-based raw resources to deal with the disposal of packaging waste. 2 Many attempts have been made progress to find green packaging materials from natural resources and the carbohydrate based polymers were considered as environmentally feedstock to substitute the fossil materials.3,4 Starch is one of the most competitive candidates for this respect because of its advantage of being an inexpensive renewable source, available worldwide, plentiful and relatively easy to manage.5,6 The processing conditions and properties of starch film strongly depend on the ratio of amylose and amylopectin and increasing the amylose ratio could generally enhance the above-mentioned properties.7,8 Maize starch contains a relatively high amount of amylose, at about 28% and appropriates for preparation of the biodegradable film substrate. However, universal acceptance of starch films for packaging application is restricted by its poor mechanical properties and inefficient barrier properties.9,10 The exploitation of the improved performance of starch films has been caused via modification starch properties and/or incorporation other materials.11-13
Chitosan is a natural and second most abundant polymer material composed of D-glucosamine units linked by β-(1–4) bonds and N-acetyl-D glucosamine and it is readily acquired from chitin. 14 Its biological characteristics have been found, such as biocompatibility, biodegradability, and antimicrobial activity. The films blended with chitosan and other polymers are applicable materials for food packaging and medical purposes. It is a feasible solution to blend chitosan in the starch films to make improvements to the mechanical and water vapor barrier performance, provide the antimicrobial activity of blend films, as well as to decrease the consumption and facilitate the biodegradability of chitosan film.15-17 In the present research the maize starch and chitosan were chosen as base materials to develop biodegradable and biocompatible blend films for food packaging applications, in which glycerol was used as a plasticizer. This study shows the functional properties and characterization of the blend films and the possible molecular miscibility between starch and chitosan has also been investigated.
Experimental
Materials
The maize starch was supplied by Shandong Lechang Food Co., Ltd. (China), while the chitosan (95% deacetylated) was purchased from Jinan Haidebei Marine Biological Engineering Co., Ltd. (China). As analytical grade reagents, glycerol was purchased from Tianjin Fuchen Chemical Co., Ltd. (China) and acetic acid (purity 36%) was purchased from Shanghai Zhongqin Chemical Co., Ltd. (China).
Preparation of films
The chitosan solution was obtained by dissolving chitosan powder in 50 mL of 1% acetic acid aqueous solution at 50°C with stirring. The fraction of the chitosan used was set at 0, 1/6, 1/5, 1/4, 1/3, and 1/2 of the maize starch dry basis, respectively. 3.0 g of maize starch was dispersed in 50 mL of deionized water and stirring for 60 min at 90°C to achieve a complete starch gelatinization. A series of chitosan/starch blend films were produced by admixing gelatinized starch and the chitosan solution, with an addition of glycerol at 40% level on the maize starch dry basis, stirring at 50°C for 40 min. The cooled paste was poured evenly onto a level glass plate and the films, when dried, were peeled from the casting plates and preserved in a culture chamber maintaining 25°C and 53% relative humidity (RH) for following experimental test. All samples were performed in triplicate.
Characterization methods
XRD analysis of the films was carried out using a Rigaku D/max-2500 X-ray diffractometer (Rigaku Corporation, Tokyo, Japan) operating with Cu-Ka radiation at 40 kV and 20 mA. The patterns were recorded over the 2θ range of 5–55°. FTIR spectroscopy measurements of films were carried out on a FTIR spectrometer (VERTEX 70, Bruker Scientific Instruments Co., Germany). The spectra in a wavenumber range from 400 to 4000 cm−1 were recorded, with 32 accumulated scans and 4 cm−1 of spectral resolution.
The morphologies of the surface and cross section of all films were studied adopting a scanning electron microscope (ULTRA Plus, Carl Zeiss AG, Germany) operating at a voltage acceleration of 25 kV. Surface and cross-section micrographs were taken at 5,000× magnification.
The thermogravimetric analysis (TGA) data were collected using STA449F3 thermo analyzer (Netzsch Co., Germany). The weight loss of films was determined over the temperature range of 20–700°C.
Film properties
Thickness and moisture content
A hand-held digital micrometer (Mitutoyo, Tokyo, Japan) with a definition of 0.001 mm was used to determine the thickness of the films. At six to ten different locations on each film sample the thickness was determined randomly and the report was given as mean ± standard deviation.
A gravimetric method was employed to ascertain the moisture content of the prepared films. Dry the weighed film samples in an oven at 105 ± 1°C for 24 h and measure the dry weight of the samples. The weight loss expressed as a percentage was computed and the moisture content of films was obtained. Three specimens of per sample were tested and the mean values are presented.
Mechanical properties
The tensile properties analysis was performed using a Universal Material Testing Machine (AGS-500N Shimadzu Co., Japan) referring to ASTM standard method D882-95.
18
Cut the film samples into strips 10 mm wide and 100 mm long and place them between the grips. Each type of film was determined in triplicate at a crosshead speed of 50 mm/min. The tensile strength (TS), elongation at break (EAB) of the preconditioned films were calculated by the following Equation (1) and Equation (2) respectively,
Moisture absorption and Water vapor permeability (WVP)
The difference weight of films before and after conditioning at relative humidity of 75% was tested to evaluate the moisture absorption of films. The film pieces of 2 cm ×2 cm size were first placed in a desiccator filled by calcium chloride until reaching a stable weight and then put in a sealed vessel containing saturated salt solutions of NaCl at 25°C to ensure a RH of 75%. The specimens were weighed after 48 h exposure and the results of moisture absorption of the films were determined by equation (4) :
ASTM E96-00 standard
19
was adopted to determine the WVP of films. Special glass vials containing calcium chloride desiccant were covered with film specimens and sealed the vial mouths with double side tape. All vials were placed in a desiccator with saturated solution of sodium chloride inside to keep 75% RH. The weight gain of the whole vials attached with the films was periodically measured every 2 h during the 24 h. Plot the curve of changes in the weight versus time and the water vapor transmission rate was obtained by the slope of the linear region divided by the film area. The WVP was calculated by the way of the water vapor transmission rate multiplied by the thickness of the film and divided by the pressure difference between the inner and outer surfaces, expressed as equation (3). The WVP values of the films were determined in triplicate.
Opacity measurement
On the basis of Oleyaei et al.
20
the rectangle pieces of films were placed directly in a spectrophotometer cell and their absorbance were tested at 600 nm using UV-7504 spectrophotometer (Shanghai Metash Instrument Co., China) with an empty cell as the reference. The absorbance was used to calculate the opacity of the films according to equation (5):
Antimicrobial tests
The antibacterial activities of films were estimated by the agar diffusion method and the optical density method respectively. Using the method reported by Arias et al. 21 agar diffusion tests were carried out to assay the antimicrobial activity of the films against E. coli and S. aureus. Sample film disks with diameter of 2 cm were placed under UV light for 1 h to sterilize, then transferred onto Mueller Hinton agar plates inoculated with E. coli or S. aureus. The plates were nurtured at 37°C for 24 h. The width of the inhibition zones formed (if any) in the areas surrounding the sample film disks was observed. The antibacterial activity of the film samples was evaluated by the inhibition zone area on the agar media.
An optical density method was also used to evaluate the antibacterial activity of the films against E. coli and S. aureus as described by Zhai et al. 22 0.50 g dry film samples were added into 50 ml medium suspensions including E. coli or S. aureus culture. The suspensions, with and without films inside, were cultivated in a shaker at 37°C and 200 rpm for 24 h, and the optical density values of the suspensions at a wavelength of 580 nm were metered. It is under aseptic condition to do all the operations and the tests were performed in triplicates.
Results and discussion
FTIR studies
FTIR spectroscopy was a useful tool for detecting the possible interactions between the components in chitosan/starch films. The spectra of starch, chitosan and chitosan/starch blend film were shown in Figure 1. The bands appeared at 1160-800 cm−1 was regarded to involve the bending mode of C-H bonds, the vibration modes of C-C and C-O stretching. It was the typical region of polysaccharide bands. The two weak bands located at around 2900 cm−1 correspond to the stretching vibration of the aliphatic C-H in the -CH2 groups. For the spectra of starch film, the broad band appearing at 3434 cm−1 corresponded to the hydroxyl groups forming hydrogen bond which associated to the complex vibrational stretches involving free intramolecular and intermolecular bound hydroxyl groups. The characteristic peak occurred at 1635 cm−1 was corresponded to the presence of bound absorbed water in the non-crystalline regions of starch. The asymmetric stretching vibrations of the O-H groups in the glycerol could also be connected with this peak.
23
Most of the characteristic peaks in the spectra of chitosan film covered nearly the same regions as those of starch. The relatively broad band located at 3403 cm−1 was assigned to the N-H stretching vibrations, which was an overlap of the O-H stretching vibrations peak in the same position.
24
To a certain extent, this band was also correlated with intermolecular hydrogen bonding in the chitosan molecules. The peaks appeared at around 1600 cm−1 and 1550 cm−1 was associated with amide-I and the amide-NH2 absorption band, respectively. FTIR spectra of (1) starch, (2) chitosan and (3) chitosan/starch films.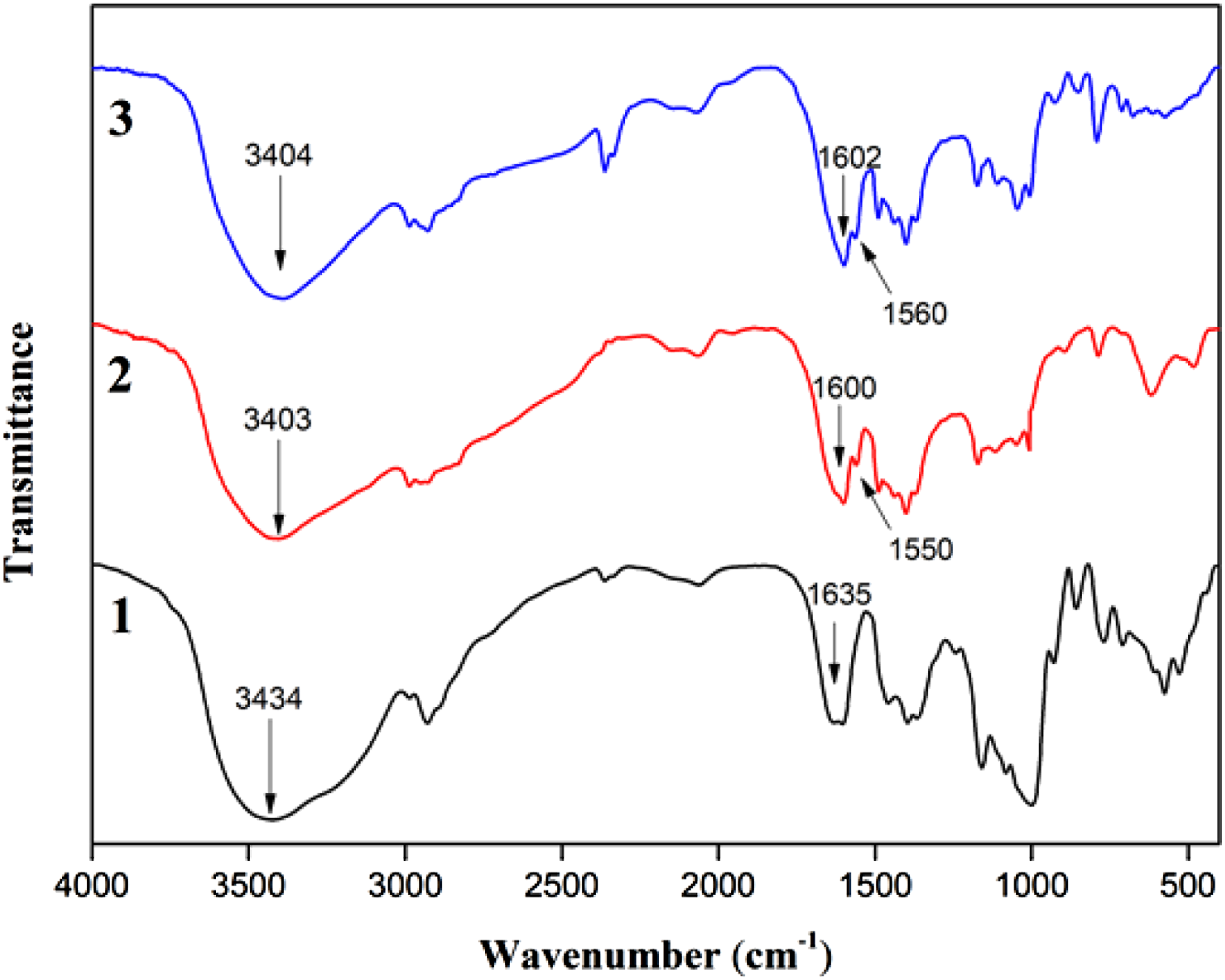
Compared with the spectra of chitosan/starch film, the amide-I peak of chitosan shifted from 1600 cm−1 to 1602 cm−1 and the amide-NH2 peak of chitosan shifted from 1550 cm−1 to 1560 cm−1. The results indicated that there were interactions between the hydroxyl groups of starch and the amino groups of chitosan. 25 The characteristic peak of the hydrogen bonds in chitosan shifted from 3403 cm−1 to 3404 cm−1. This suggested that the intermolecular and intramolecular hydrogen bonding formed between chitosan and starch. 26
XRD analysis
Figure 2 presented the XRD patterns of starch, chitosan and chitosan/starch blend films. The diffraction pattern of the starch film appeared peaks around 5.6°, 15.2°, 17.2°, 19.9°, 22.3° and 24.2° which correspond to the typical B-type starch diffraction pattern.
26
The chitosan presented a characteristic peak at about 2θ = 21°, which agrees with previous publications.
13
The chitosan/starch blend films with low chitosan content displayed one broad peak at about 20° and the characteristic peak of chitosan and starch did not appear, which indicated that the blend of chitosan and starch was miscible, the intermolecular hydrogen bonding between the two limited the molecular chain segment movements and restrained the crystallization process.
27
However, the crystalline peaks were found in the blend film when the ratio of chitosan/starch was above 1/3 and further addition of chitosan made the composite film less amorphous, which was also verified in the research of Yusof et al.
28
The reappearance of crystalline peaks suggests that there was a phase separation between the two main components of the films, which was consistent with the results of the tensile strength and elongation at break. XRD patterns of samples.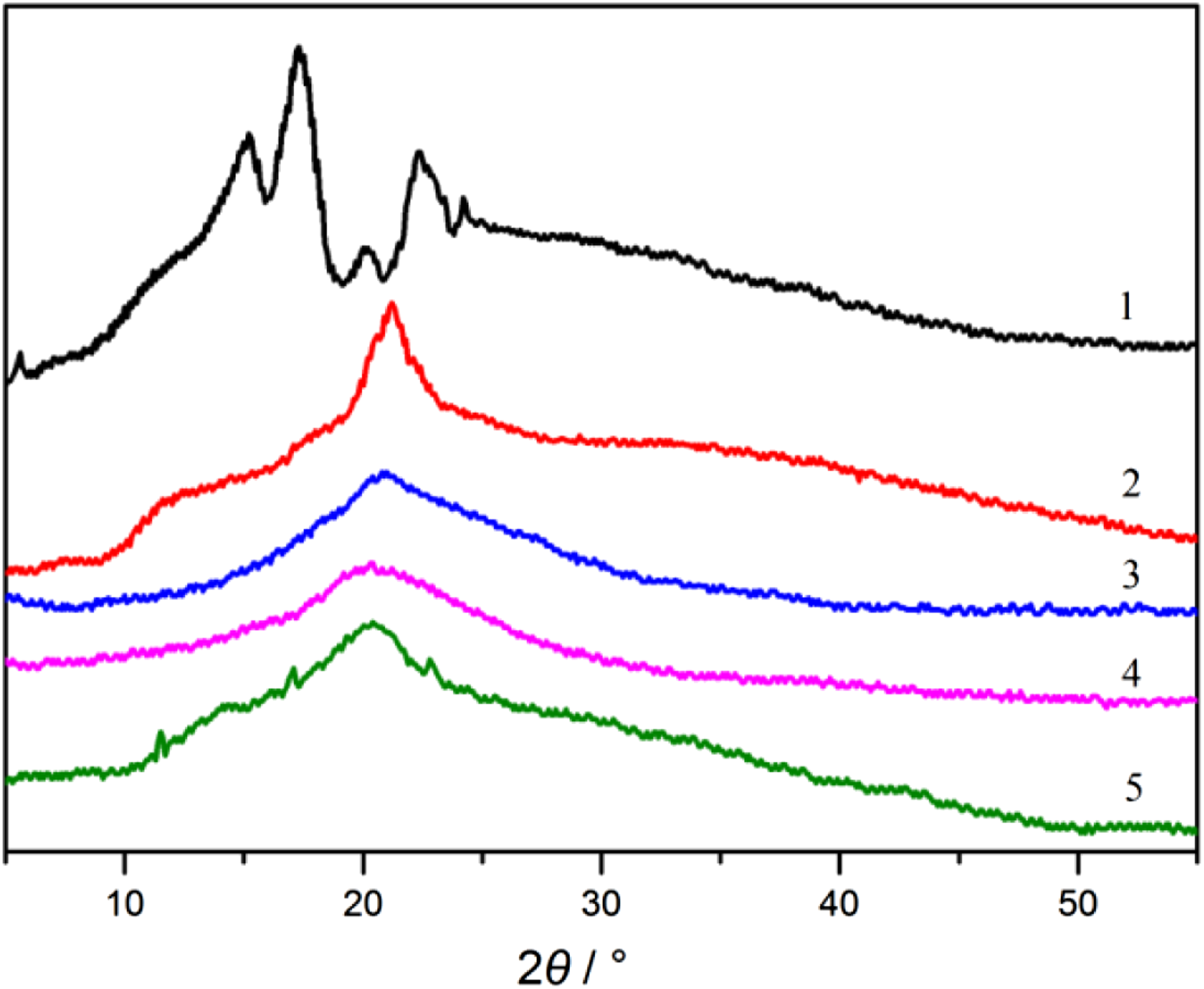
Film microstructure
The micrographs at surface and cross-section of the films were shown in Figure 3. The surface and cross-section of starch film (0) were uneven and coarse structure which is in agreement with its relative low TS and high WVP, as shown below. The structure of surface and cross-section for the composite film (1) was rough and not uniform. Although the flatness and uniformity of the structure of surface and cross-section for film (2) and (3) were improved with an increase of chitosan content, there were many small cracks distributed on their surface. It suggested that the chitosan and starch were not completely miscible. For the film (4) with 1/3 of chitosan/starch ratio, the surface was smooth, compact and continuous and there was homogeneous structure without obvious pores and cracks except for several particles. Its cross-section also presented relatively high structural integrity. The compact matrix without phase separation demonstrated there was intense interfacial adhesion between the chitosan and starch and this provided better mechanical and barrier properties to the film. The surface and cross-section structure of the film (5) showed quite bumpy and heterogeneous zones with some pores and cracks, which was probably due to the crinkling of the starch links in the surroundings of excessive chitosan
29
and the formation of crystallization in the starch and chitosan.
30
It confirmed that there was not good compatibility between the two components at greater chitosan/starch ratio and it restraint the physical properties of the films depended on its microstructure. For example, its TS and transparency were not high, as shown below. SEM images of the surface (S) and cross-section (C) of films.
Thickness and moisture content
Thickness and moisture content of the film samples.
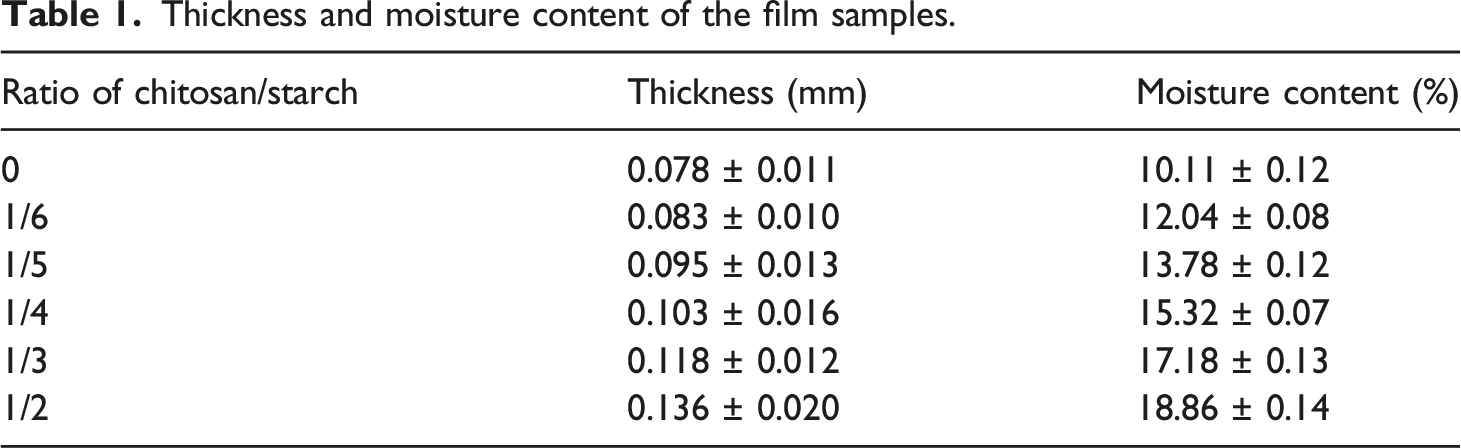
The differences in the polymer packing derived that the water holding capacity had undergone different changes in the process of the drying of the films. The lower value of thickness and moisture content of the starch film is due to the neutral character of the chains in maize starch give rise to the thinner hydration layers. The chitosan chains are positively charged and contain wider hydration layers with many water molecules, these highly retained water molecules take part in the film structure to restrain the chain approximation and lead to thicker films. The thickness and moisture content of the film increased with an increasing content of chitosan due to the more open matrix with the greater hydration level of the films. 31
Mechanical properties
The determination of mechanical properties of films is necessary because sufficient mechanical strength and ductility are essential for packaging materials to keep its intact ability during using. The mechanical properties of chitosan/starch blend films are given in Figure 4, which demonstrated the TS, EAB and YM. Mechanical properties of films. (a) TS, (b) EAB and (c) YM.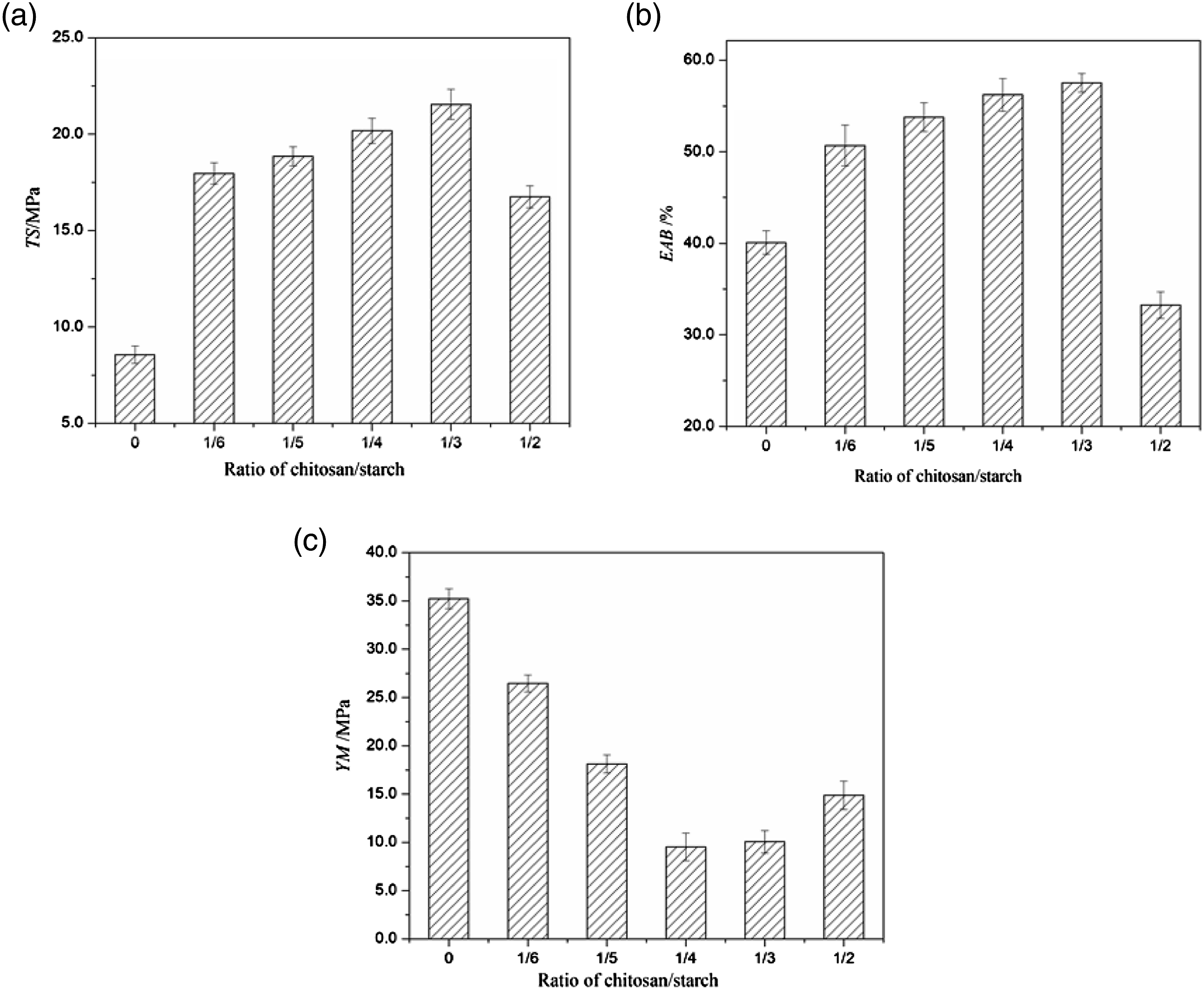
There was an increasing TS values of the blend films with the addition of chitosan, reaching a maximum of 21.54 ± 0.78 MPa, 2.52 times that of the starch film, for the film with a chitosan/starch ratio of 1/3. Then the TS of the film declined as the ratio of chitosan/starch increased to 1/2. The rise of the TS is due to the charged amino groups in chitosan caused the polarization of other chain bonds and the strengthening of attractive forces between the chains, accompanying the enhanced van der Waals forces and hydrogen bonds in a cooperative network structure, the more intense cohesion forces generated in the chitosan than those in the starch. The cooperative van der Waals forces increased with the incorporation of chitosan in the starch films as a result of chitosan’s greater capacity to form hydrogen bonds. 31 The gelatinization destroyed the ordered crystalline structures of the starch molecules and NH3+ was generated from the protonation of the amino groups of chitosan in the acetic acid solution, resulting in the NH3+ forming the inter-molecular hydrogen bonding interaction to easily form hydrogen bonds with the OH groups being exposed. 13 The interplay between chitosan and starch could reinforce the TS of blend films, nevertheless, the enlargement of chitosan content in the film-forming solutions brought a large number of amino groups. It was hardly to form a homogeneous system with the chitosan content surpassed a certain value and it led to weak interactions at the chitosan-starch boundaries and the poorer TS of the films. 32 This was proved by the heterogeneous microstructure and phase separation of the film at 1/2 of chitosan/starch ratio in SEM, as shown in Figure 3(5).
The value of EAB is an index signifying the flexibility and stretch ability of films. It was observed that the blend films elongated significantly more than the starch film, which are typically very brittle. The EAB of the films increased with rising of chitosan content from 40.07 ± 1.28% to 57.54 ± 1.01% up to chitosan/starch ratio of 1/3, and then decreased at 1/2 of chitosan/starch ratio. The increased EAB of the films may be due to a plasticizing role of chitosan is conducive to the extension and plastic deformation of films, the mobility of polymer chains was promoted with the interaction of plasticizer-polymer and the overall flexibility of films was enhanced. 32 The reduction of EAB for the films at 1/2 of chitosan/starch ratio is may be owing to the increased crystallinity of the starch in the blend films. 13 It is also related to its heterogeneous microstructure as shown in SEM.
The rigidity of the material was denoted by the YM and a larger value of YM involved a more inflexible film. The increase of chitosan/starch ratio brought about a significant decrease of the YM, relating to the more elastic films and a slightly increase of the YM occurred in the film incorporated greater amount of chitosan. The decreased YM with the addition of chitosan was perhaps due to the decreased crystallinity of starch and chitosan in the composite films. 33 The molecular chain segment movements were restricted by the intermolecular hydrogen bonding between chitosan and starch and the crystallization process was restrained. 27 However, the noticeable crystallization appeared in the films at the higher chitosan content and the films became less amorphous with adding too much chitosan.
Water vapor barrier performance
It is an important function for films to prevent the transmission of the moisture between packaged contents and the surrounding air or between two components of product. Therefore, the blend films should have a lower WVP. The values of WVP and moisture absorption of films were shown in Figure 5. It presented a WVP value of (7.19 ± 0.32) ×10−10 g·m−1·s−1·Pa−1 for the starch film and those values in the range of (2.8∼4.9)×10−10 g·m−1·s−1·Pa−1 for chitosan/starch films. The result illustrated that the water vapor barrier performance of composite starch films was improved by adding chitosan. The WVP of composite films was decreased with increasing the ratio of chitosan/starch from 1/6 to 1/3 and the value of the film at 1/3 of chitosan/starch ratio was (2.81 ± 0.26) ×10−10 g·m−1·s−1·Pa−1, which was 60.9% lower than the starch film. Then by further increasing chitosan/starch ratio to 1/2 the WVP of film was increased. The higher WVP value of starch film may be explained that the interactions with water molecules were enhanced via the larger number of free hydroxyl groups in starch, consequently, the transfer of water vapor through the films was promoted. The hydrophilic glycerol had the effects of weakening the hydrogen bonds between molecules and expanding the distance between the molecules, it relaxed the starch network and made the adsorption and desorption of water molecules beneficial.
34
However, the incorporation of chitosan resulted in the hydrogen bonds formed between chitosan and starch molecules and the availability of the hydrophilic groups reduced, imparting greater hydrophobicity to the films and decreasing the WVP of the films. Furthermore, the presence of acetyl groups in the chitosan pyranose ring made it less hydrophilic compared to the starch and the stronger hydrogen bond led to glucopyranosyl linkages in chitosan could provide better packing structure or higher crystallinity than glucopyranosyl linkages in starch. Difficulties arise in the absorption of moisture or water and hydrolysis.
35
Therefore the interaction of the film matrix with water molecules was reduced by the way of incorporation of chitosan, resulting lower WVP of the starch/chitosan films.
36
The film at 1/2 of the chitosan/starch ratio have relatively poor water vapor barrier characteristics, this trend could be explained by the excess chitosan brought too much hydrophilicity NH3+ groups in the films.
13
Water resistance of films. (a) WVP and (b) moisture absorption.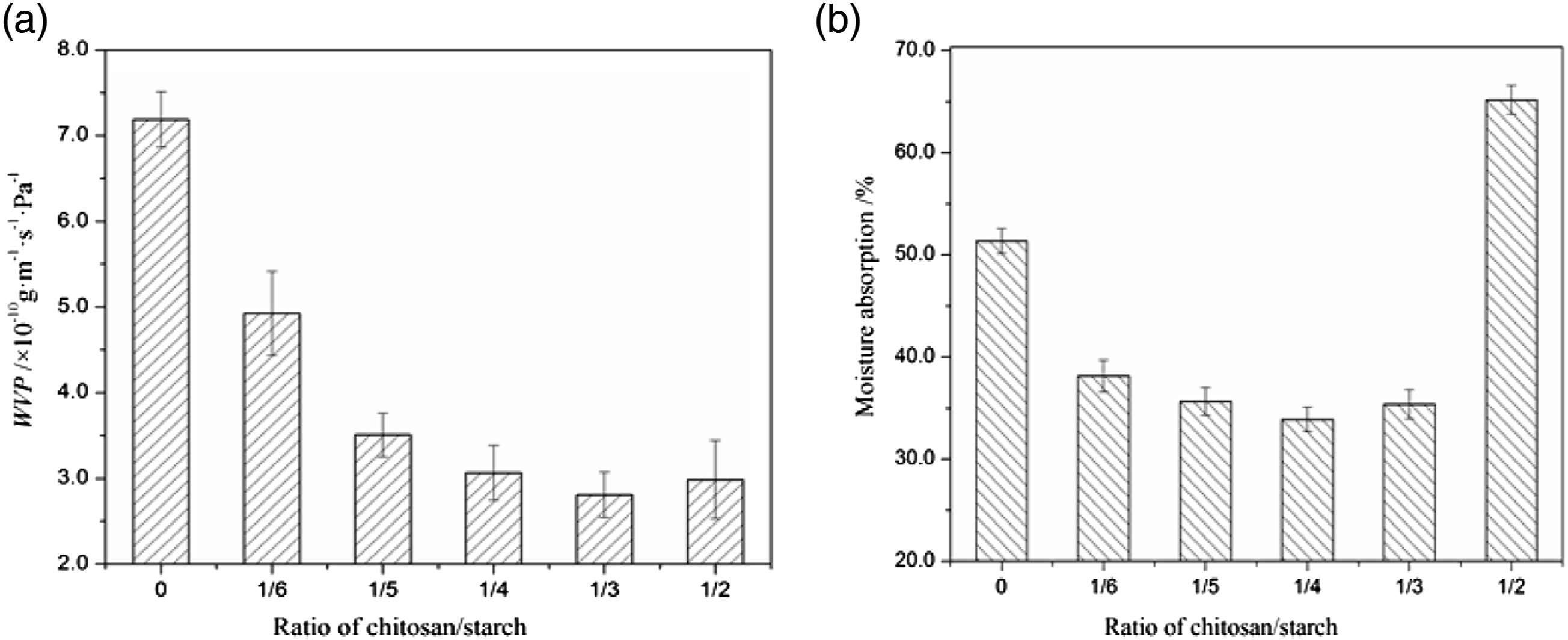
A similar tendency was observed for the moisture absorption of the films. The decrements of the moisture absorption of the films presented as the chitosan/starch ratios increased from 1/6 to 1/4 and the further increase of chitosan caused a rise of moisture absorption. The greater hydrogen bonding interactions among the chitosan and starch declined the starch hydrophilic groups’ availability and the free sites for water vapor absorption in the films were reduced, resulting in the lower moisture absorption of the films. 37 The increase of moisture absorption at higher ratios of chitosan may be associated with the greater number of hydrophilic groups in films.
Opacity of films
Opacity or transparency of films is an important quality to affect the appearance of the contained goods and it also can be used to reveal the miscibility of a polymer mixture.
38
Generally, most food packaging required low opacity or high transparency of the films in the visible range. As shown in Figure 6, the chitosan/starch films were more transparent than the starch film and their opacity values decreased with increasing chitosan/starch ratio down to 1/3. We suggest this was a result of the intermolecular hydrogen bonds formation increasing the compatibility of the chitosan and starch in the film matrix. The better transparency of the film at 1/3 of chitosan/starch ratio was consistent with the more complete miscibility among starch and chitosan than that in the other films, as shown by SEM results. An increase of opacity of the film with 1/2 of chitosan/starch ratio demonstrated the weak interactions between the chitosan-starch and poor compatibility in a polymer mixture. Opacity of films.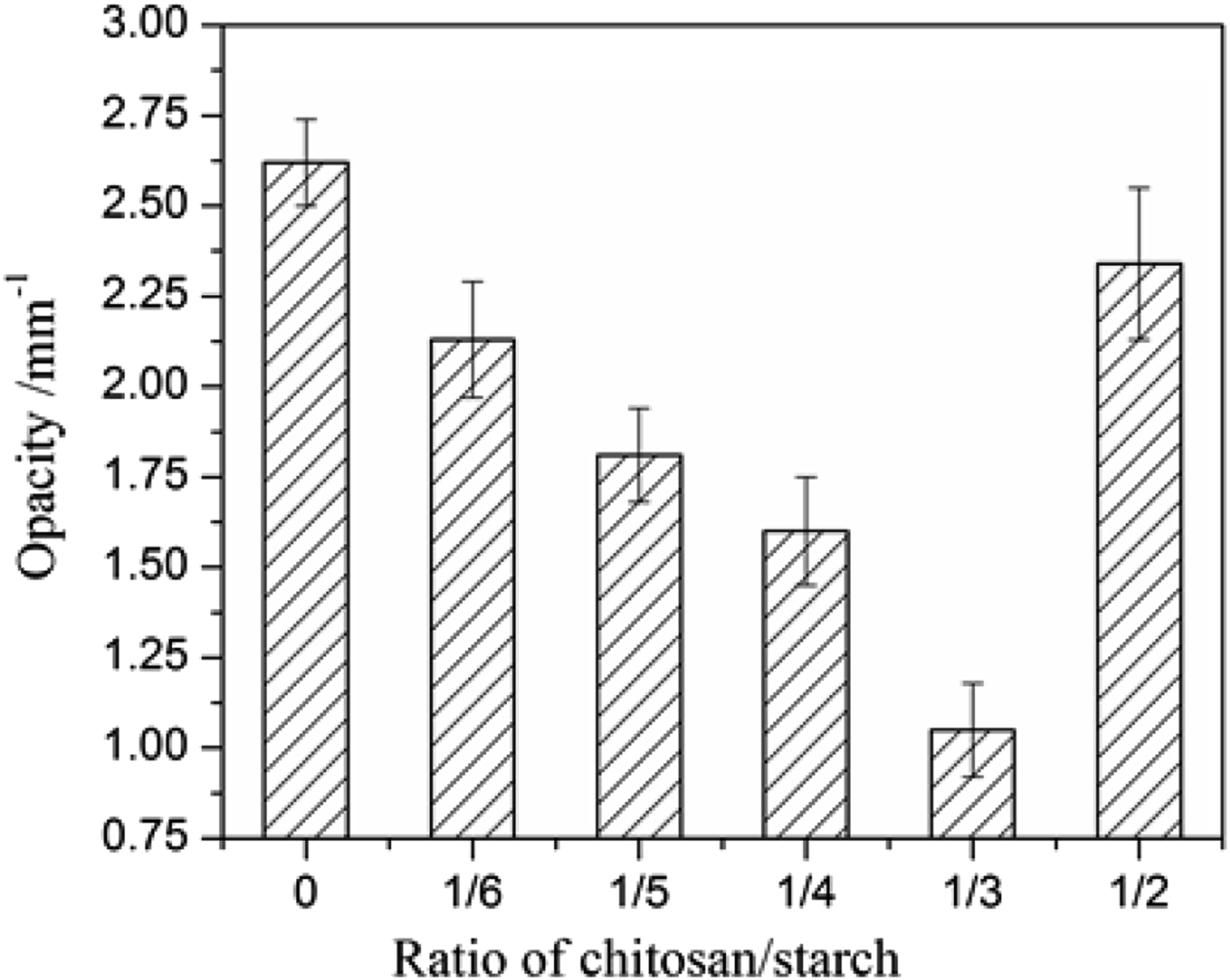
Thermal Analysis
The TGA profiles of starch film and chitosan/starch blend film are shown in Figure 7, respectively. As it is shown, the weight loss occurred between 20∼200°C was associated with the evaporation of free water and bound water from the films. The subsequent weight loss in the temperature range of 200∼300°C corresponded to the thermal decomposition of starch in and degradation of glycerol in the films. The degradation of starch and the release of chitosan as well as plasticizer glycerol with lower molecular weight appeared around 300∼500°C. There was a pyrolysis of carbonated compounds took place in the films beyond 500°C and the inorganic were left in the residual samples. TGA curves of (1) starch film and (2) chitosan/starch film.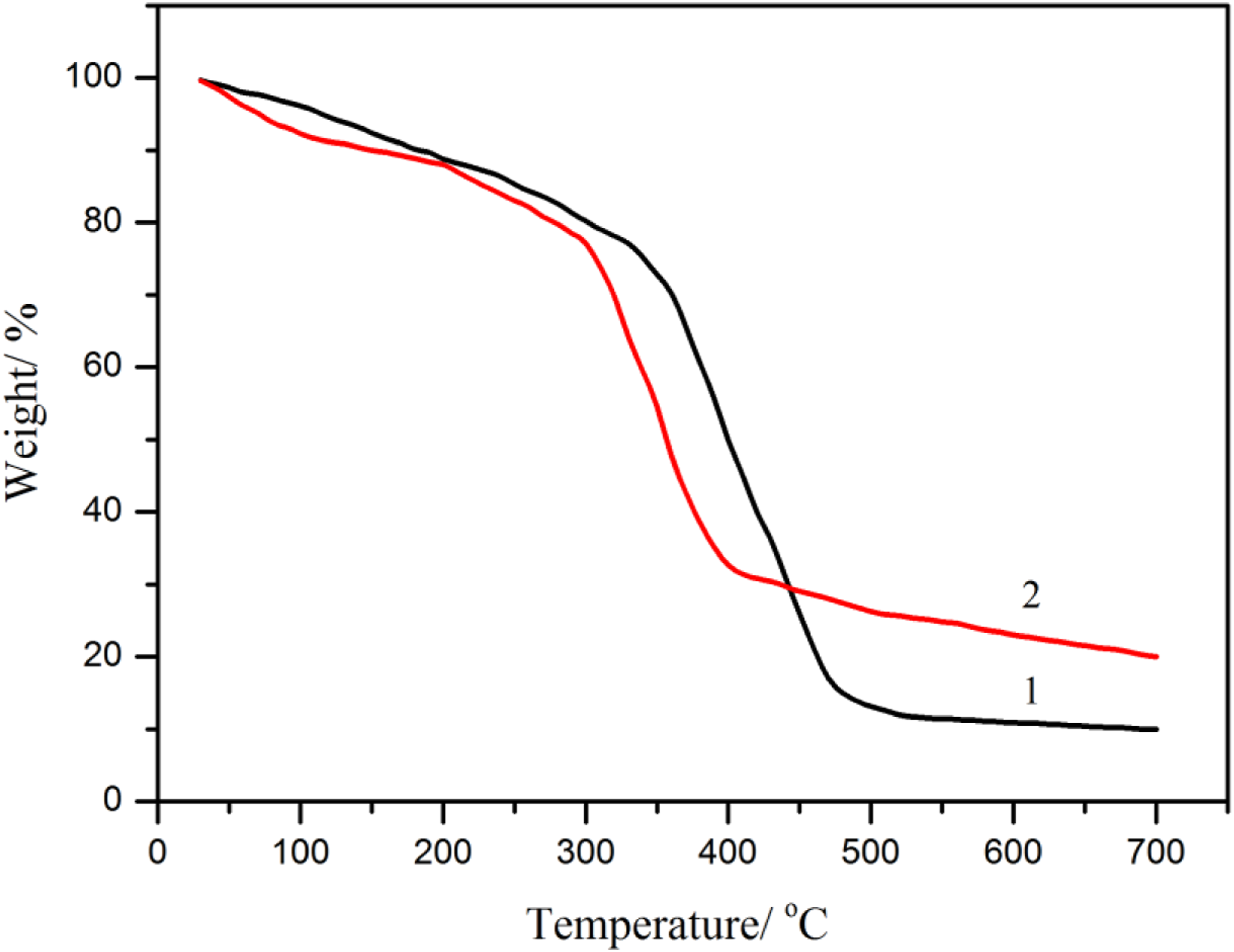
The chitosan/starch composite film had a lower decomposition temperature, which might be related to the acetic acid disrupted the inter- and intra-molecular hydrogen bonds to cause the thermal degradation. 39 Another explanation could be the decreased crystallinity in the chitosan/starch composite film, as previously discussed in the XRD analysis. The more flexible molecular movement in the polymer chains would arise in the film with lower crystallinity and thereby it had a lower degradation temperature. 40 The operating temperatures of most applications associated with the blend films are well below their thermal stability.
Antibacterial assay
The antibacterial activity of films against E. coli and S. aureus was investigated and Figure 8 presented the results of the agar diffusion assays. There were not any obvious inhibition zone around the starch films and E. coli and S. aureus grew onto the surface of the films. The starch film had scarcely inhibitory function on E. coli or S. aureus. However, clear, narrow and small inhibition zones surrounding the chitosan/starch film disks were observed with growth the of microbial colonies in the agar being prevented in the areas contacting with the films and the antimicrobial effect for E. coli was better than that for S. aureus. The results indicated that the starch films incorporating chitosan displayed antimicrobial property against both E. coli and S. aureus. Pictures of the inhibitory zones of the films. (a) E. coli, Starch film, (b) E. coli, Chitosan/starch film, (c) S. aureus, Starch film, (d) S. aureus, Chitosan/starch film.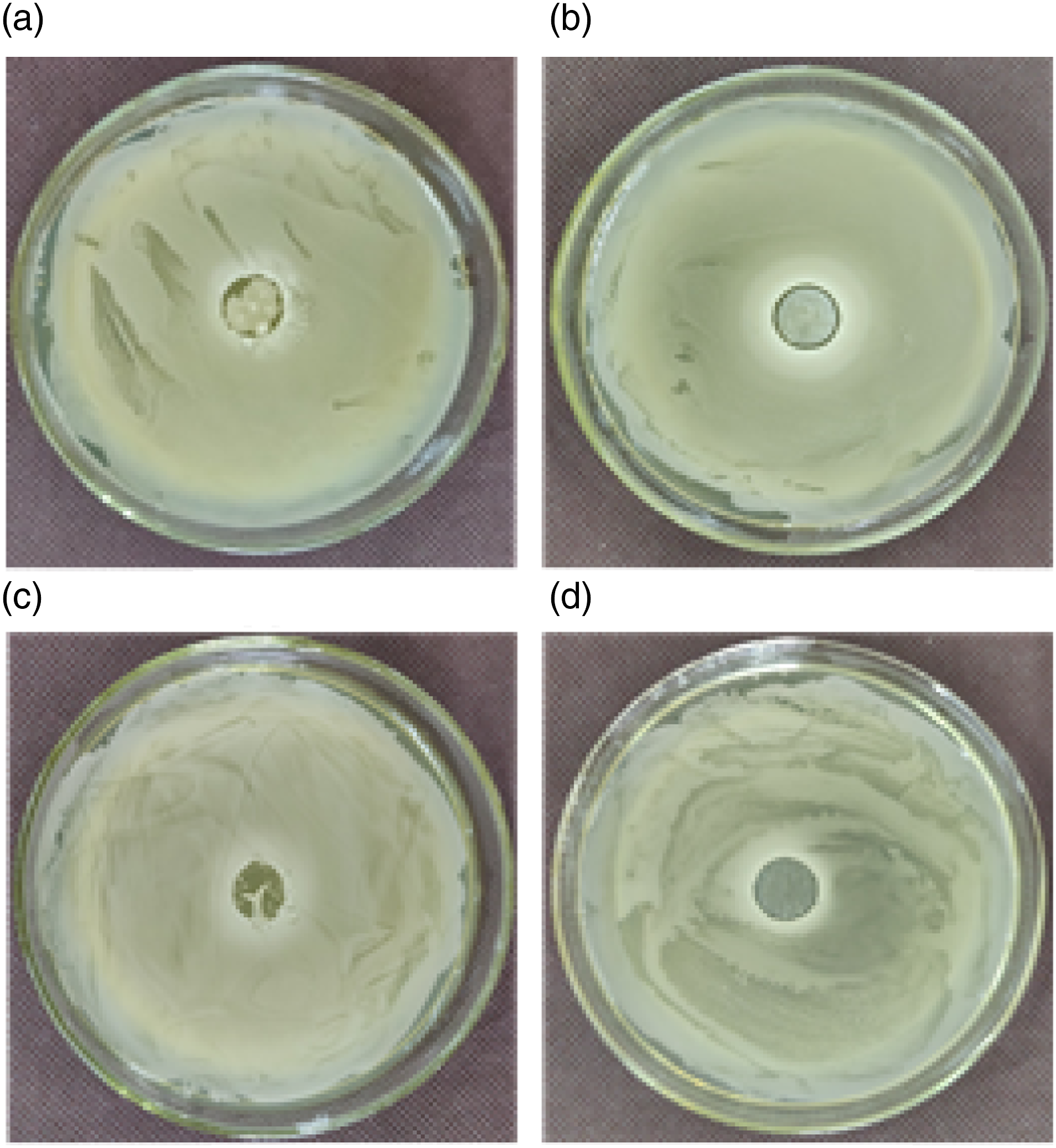
According to the optical density method the inhibitory effects on E. coli and S. aureus of the films was expressed and the results were shown in Figure 9. It was found that the optical density of the suspension with chitosan/starch films inside was lower compared to the pure medium or that with starch film inside and the values of optical density reduced as the chitosan/starch ratio increased in blend films. When the chitosan/starch ratio exceeded 1/3, an increase of optical density was observed for the film, but it was still lower than that for the starch film. Antimicrobial activity of films.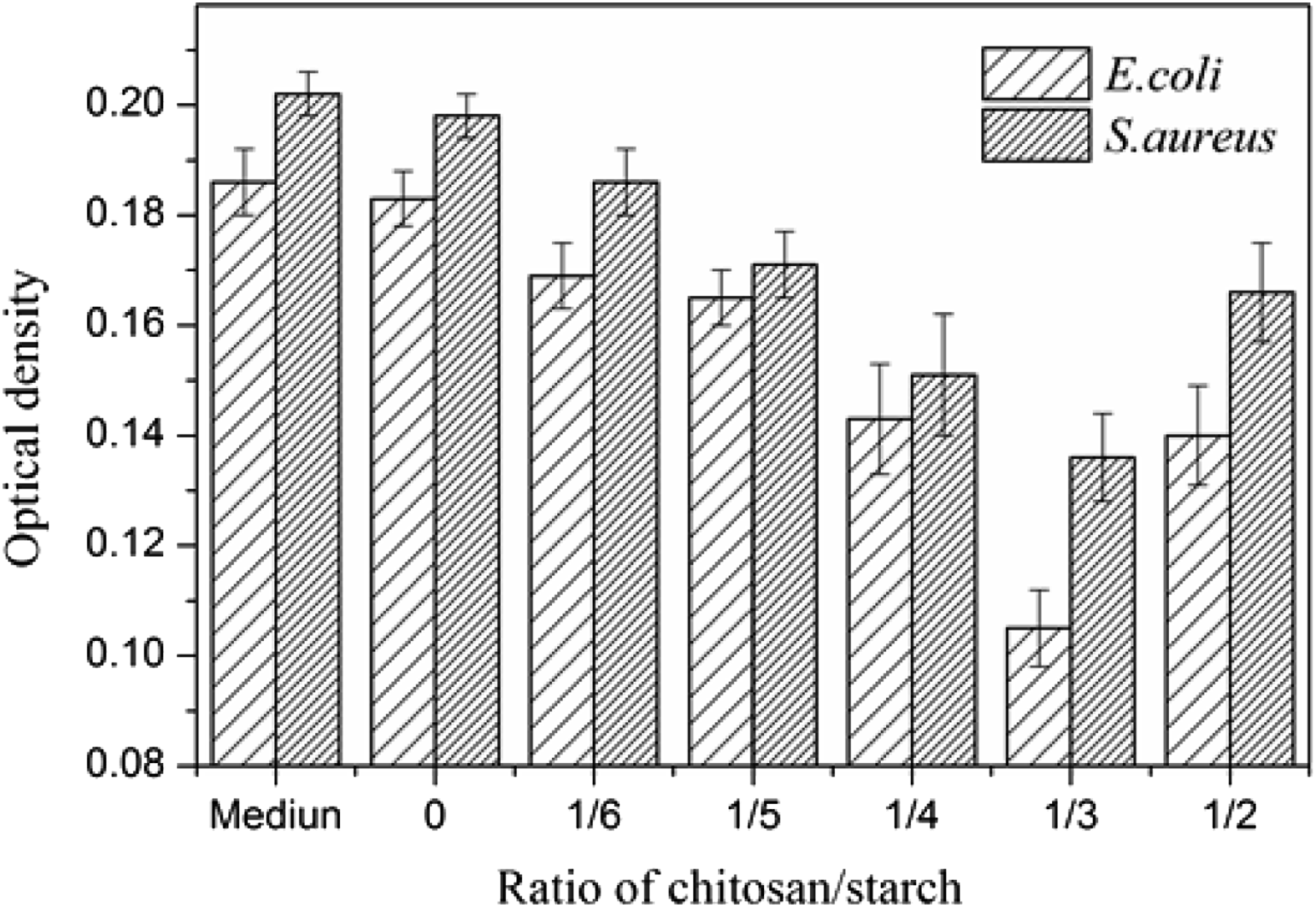
The antibacterial mechanism could be interpreted as the electrostatic interactions likely occurred between NH3+ generated by the protonation of NH2 groups of chitosan and anionic groups of microbial cell membranes, causing the seepage of proteinaceous and intracellular constituents of the microorganisms. 24 An enhancement of antimicrobial activity of the blend films appeared with an increment of chitosan based on the antimicrobial capacity of chitosan. But the antimicrobial efficacy of chitosan was compressed by the reinforced actions between starch and chitosan, which reduced the availability of NH3+ positions and inhibit NH2 groups bonding to the cell membrane, depressing its antimicrobial activity. 41
Conclusions
Biodegradable starch films blended with chitosan were fabricated by a way of solution casting via incorporating various levels of chitosan. A comprehensive investigation about the influence of chitosan content on mechanical, antimicrobial properties, water vapor barrier performance, and microstructure features of the composite films was conducted. The appropriate amount of chitosan was shown to improve the overall performance of the composite films. The film at 1/3 of chitosan/starch ratio exhibited better rigidity flexibility and its TS and WVP were 21.54 ± 0.78 MPa and (2.81 ± 0.26)×10−10 g·m−1·s−1·Pa−1, which were 1.52 times higher and 60.9% lower than those of the starch film, respectively. FTIR and XRD analyses of starch/chitosan films revealed the formation of hydrogen bonds between chitosan and starch and the reduction of crystallinity in blend films, SEM indicated that excessive chitosan in the blend systems sabotaged the miscibility between starch and chitosan. The blend films displayed reliable thermal stability and significant antimicrobial performance against E. coli and S. aureus, with the antimicrobial efficiency of 43.5% and 32.7% for E. coli and S. aureus, respectively. The obtained results suggest that it may have a potential possibility to formulate renewable, biodegradable and antimicrobial films with blending maize starch and chitosan for various food packaging applications.
Footnotes
Acknowledgements
The authors wish to acknowledge the Institute of the School of Chemical and Biological Engineering, Lanzhou Jiaotong University for the support provided for our experimental research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.





