Abstract
In the present work, corn starch was grafted with oleic acid in the presence of redox initiators. Starch nanocrystals (SNCs) were synthesized using acid hydrolysis method and nanocomposite films were prepared by incorporation of SNCs into polyvinyl alcohol/starch (PVA/S) and PVA/grafted starch (PVA/GS) matrix, separately. It was observed that SNCs exhibited a platelet-like shape and in the size range of 200 nm. The increased crystallinity of SNCs indicated the removal of the amorphous and waxy region after acid hydrolysis of starch. Scanning electron micrographs of nanocomposite films showed good dispersion of SNCs within the PVA/GS matrix compared to the PVA/S matrix. The tensile strength of the nanocomposite films increased significantly with the reinforcement of SNCs into PVA/S matrix. Water uptake (%) of PVA/GS nanocomposite film decreased by 38.25% compared to the PVA/S nanocomposite film. Reinforcement of SNCs with PVA/S matrix improved the biodegradability of the films and was higher than PVA/S film. PVA/S nanocomposite film showed 32.4% weight loss after 120 days of soil burial degradation test. Thus, the obtained nanocomposite films can provide a potential alternative to the synthetic and nondegradable packaging films used worldwide.
Introduction
In today’s world, the utilization of nonbiodegradable and petrochemical-based plastic packaging materials has been a major environmental concern due to their poor biodegradability. 1 According to the literature, 200 million tons (approximate) per year are produced all over the world. 2 Therefore, the interest toward the utilization of biodegradable materials has been more intense during the last few decades. Biodegradable polymers can be classified into two categories: artificially produced biodegradable polymers and natural biopolymers. Synthetic biodegradable polymers such as aliphatic polyesters, polyhydroxyalkanoates (PHAs), and polyglycolic acid are derived from petroleum-based resources. However, natural polymers such as starch, cellulose, and proteins are produced from natural resources such as plants and animals. 3 Natural biopolymers have several advantages over synthetic biodegradable polymers such as sustainability, renewability, abundancy, and low price as they are natural products. Starch has been preferred over other biopolymers as it is easily available, cheap, and biodegradable. 4 But the films composed entirely of starch lack in terms of mechanical properties. 5 Starch can be blended with other biopolymers or other synthetic polymers to produce fully biodegradable or partially degradable films with improved packaging properties. For the preparation of completely/partially biodegradable film, starch can be blended with other biopolymers such as poly β-hydroxyalkanoates, polylactic acid (PLA), and polyvinyl alcohol (PVA). 6 -8
PVA has several advantages such as good film-forming properties, high thermal stability, and strong conglutination. 9 Due to the presence of hydroxyl groups in PVA and starch, the formation of intermolecular and intramolecular hydrogen bonds takes place. 10 It is also responsible for higher water uptake of the films. The water-resistant properties and compatibility of PVA/Starch (PVA/S) films can further be improved by chemical modification of starch. Grafting is the most common method used for improving the properties of natural polymers such as starch and cellulose. 11 However, the blend of PVA/S is assumed to be biodegradable since both components are biodegradable in the microbial environment. 12 Oleic acid is one of the long-chain fatty acids and can be used for grafting of starch. Kohli et al. performed the grafting of barley husk with oleic acid and reported the improvement in properties of cellulosic material after grafting. 13 To further increase the mechanical properties of PVA/S-based films, reinforcement of various micro- and nanofillers such as cellulosic fibers, synthetic fibers, nanofibrils, and nanocrystals with PVA/S matrix can be approached. 14 -16
Recently, starch nanocrystals (SNCs) have received great attention due to their biocompatibility, availability, low cost, and compounded with many different kinds of polymer matrices. 17,18 Condes et al. reported that the incorporation of SNCs with amaranth protein resulted in an increase in the mechanical properties of nanocomposite films. 19 In the literature, very few studies have reported the properties of nanocomposite films prepared by the incorporation of SNCs within starch-based matrix. However, no study has reported the properties of PVA/grafted starch (PVA/GS) and SNCs-based nanocomposite films after reinforcement of SNCs within the polymer matrix. In the present study, SNCs were prepared and characterized using field-emission scanning electron microscopy (FESEM) and X-ray diffractometer (XRD). Nanocomposite films were synthesized by reinforcement of SNCs into PVA/S and PVA/GS matrix, separately. Morphological analysis of the films was performed using scanning electron microscopy (SEM). The evaluation of mechanical properties, water-resistant properties, and biodegradability of the nanocomposite films was performed.
Materials and methods
Materials
Corn starch (carbohydrates 98%, amylose 23.6%, and moisture <0.3%) was obtained from Sukhjit Starch and Chemicals Limited (Phagwara, Punjab, India). PVA ( average molecular weight is 14,000 g/mol) used in the study was obtained from S.D. Fine Chemicals Limited (Mumbai, Maharashtra, India). Oleic acid (extra pure), dimethyl sulfoxide (DMSO), ferrous ammonium sulfate (FAS), potassium persulfate (KPS), and sulfuric acid were procured from Loba Chemie Pvt. Ltd (Mumbai, Maharashtra, India). All the chemicals were used as received.
Methods
Grafting of starch
Graft copolymerization of starch was done by taking a mixture of water/DMSO (1:3) as a solvent medium. Starch was soaked overnight at 30°C in the solvent to swell and to activate reactive sites on the surface. Initiators FAS and KPS were added to the reaction flask in a definite molar ratio. 20 Further, oleic acid was added dropwise into the reaction media and the concentration of oleic acid was maintained to be 7.92 mol L−1 × 10−3. The reaction mixture was heated at 50°C for 150 min with continuous stirring (300 r min−1). Grafted copolymers of starch were extracted with ethanol using a Soxhlet extractor to remove any soluble impurities or unreacted chemicals. The sample was dried in a hot air oven at 50°C for 24 h and stored at room temperature in an airtight container.
Preparation of SNCs
The preparation of SNCs with corn starch was conducted by acid hydrolysis method using sulfuric acid (H2SO4). The acid hydrolysis procedure was performed using a Quick Sonic bath-type ultrasound apparatus (frequency 40 kHz). Initially, the temperature inside the ultrasound bath was adjusted to 40°C. Dispersion of starch with 15% (w/w) concentration was prepared in 3.16 M sulfuric acid solution. The dispersion was placed immediately at a height of 20 cm from the transducer and submitted to sonication at 100% amplitude for 30 min. Treated samples were washed many times, followed by successive centrifugation with distilled water until neutral pH (7.0) is achieved and then freeze-dried to obtain the SNCs.
Film preparation methods
PVA/S was prepared by dissolving PVA (5 g) in hot water (∼90°C). Starch (5 g) was gelatinized in hot water, mixed with PVA solution, and the resulting mixture is set for continuous stirring using a mechanical stirrer (800 r min−1) for 45 min at 50°C to form a homogeneous gel solution. Glycerol was used as a plasticizer and added to the mixture with continuous stirring. The total amount of polymeric mixture was maintained to 100 g by adding distilled water. The suspension was poured onto the glass petri dish and dried in a hot air oven at 45°C for 24 h. The fully dried films were peeled off from the petri plates.
For PVA/S and PVA/GS-based nanocomposite films, SNCs were mixed in a varying proportion with PVA/S and PVA/GS blend mixture separately. The composition of the films is presented in Table 1. The schematic representation of the preparation method of nanocomposite films is shown in Figure 1. Then, the films were prepared by the casting method and dried in a hot air oven for 24 h at 50°C.
Composition of PVA/S nanocomposite films reinforced with SNCs.
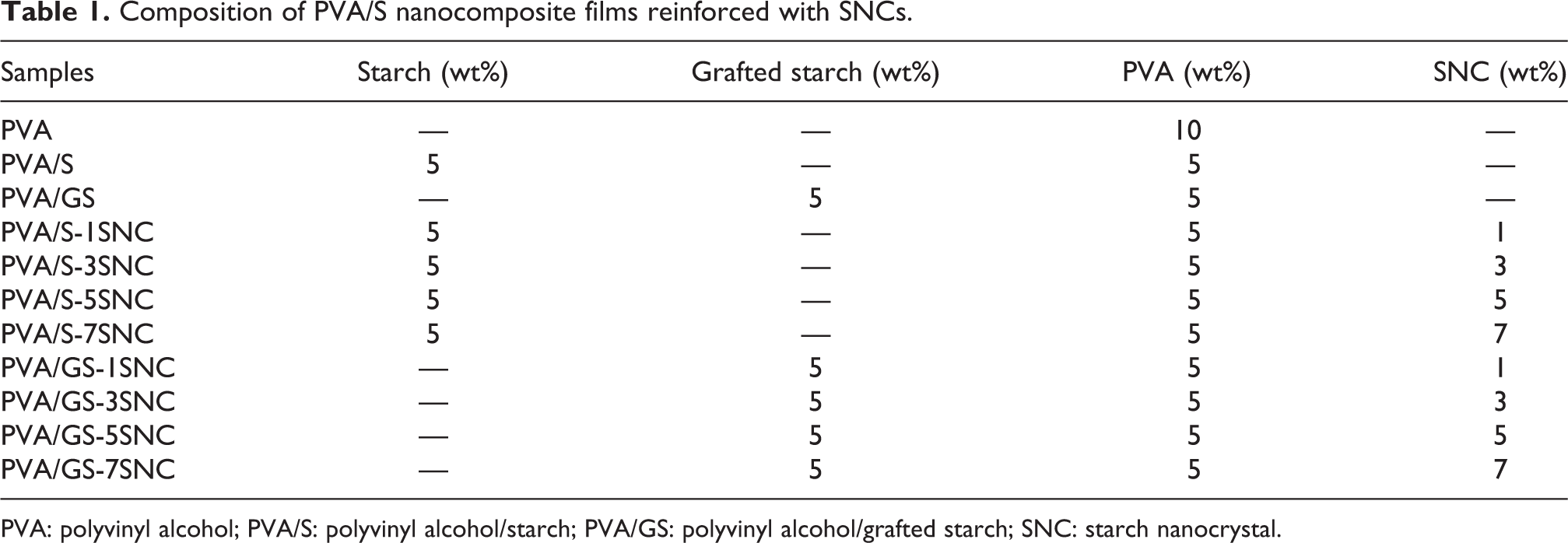
PVA: polyvinyl alcohol; PVA/S: polyvinyl alcohol/starch; PVA/GS: polyvinyl alcohol/grafted starch; SNC: starch nanocrystal.
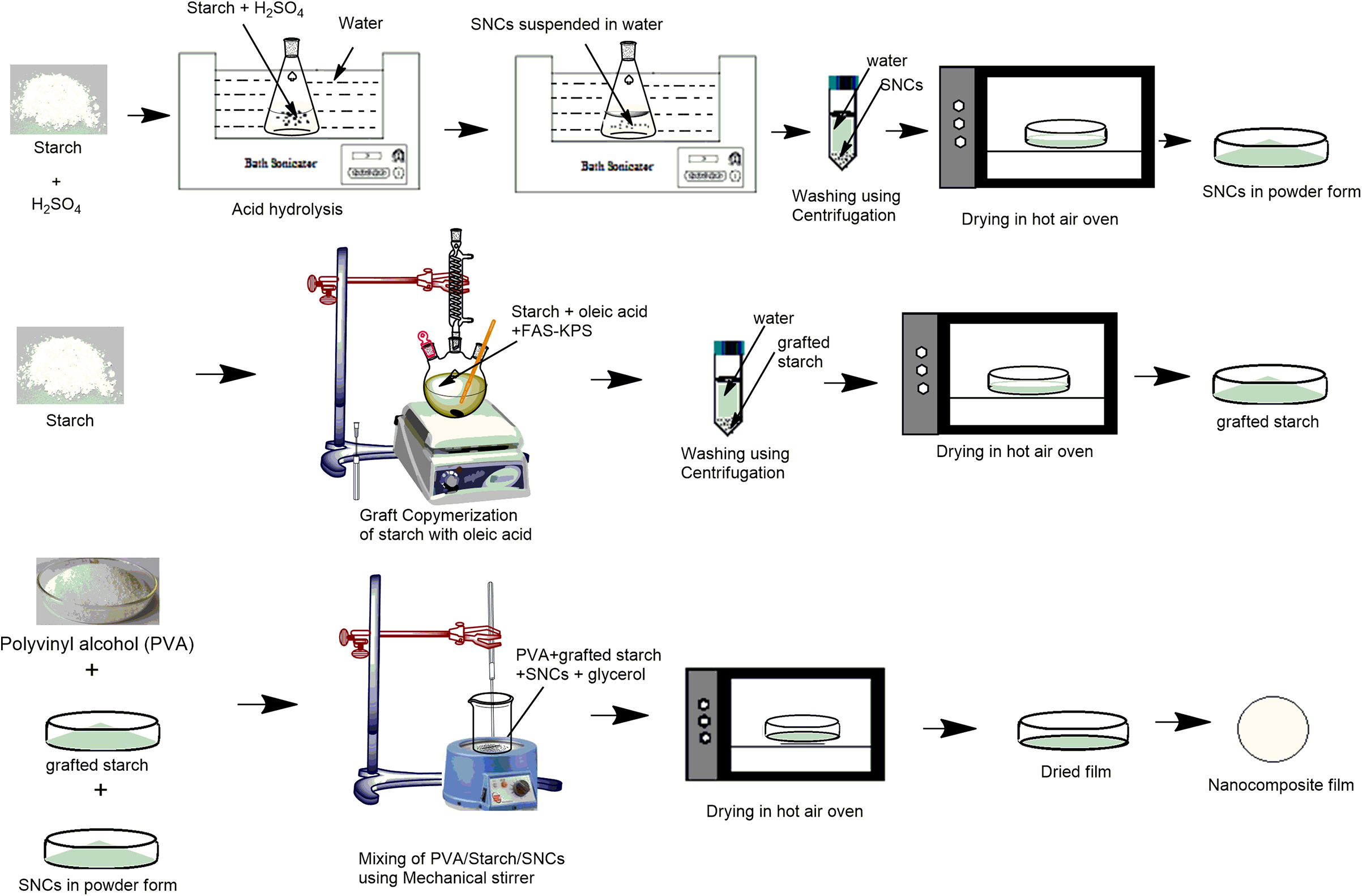
Schematic representation of the preparation method of nanocomposite films.
FTIR spectroscopy
Starch, graft copolymers of starch, and SNCs were characterized by using PerkinElmer Fourier transform infrared (FTIR) spectrophotometer in the range from 4000 cm−1 to 400 cm−1 at a resolution of 4 cm−1 using potassium bromide pellets.
Field-emission scanning electron microscopy
SNCs were characterized using FESEM (QUANTA 200 FEG, FEI, the Netherlands). Tungsten filament coated with zirconium dioxide layer was used as an electron source. A drop of diluted suspension of SNCs (with a solid content of about 0.02 wt%) was deposited on a silicon wafer, evaporated at room temperature, and then coated with a thin carbon layer limited to 3 nm applied by sputtering.
XRD studies
XRD studies were carried out to know about the crystalline and amorphous regions of the starch. XRD spectra of starch, graft copolymers of starch, and SNCs were recorded by X-ray diffractometer (XPERT-PRO PW 3064, Phillips, Japan). The radiation used is copper
Scanning electron microscopy
Surface morphology of starch, grafted starch, PVA/S films, and PVA/GS nanocomposite films reinforced with SNCs was observed with SEM (LEO 435 VP). The range of accelerating voltage was up to 30 kV. The samples were first sputter coated with a thin layer of gold and then observed at different magnifications.
Mechanical properties
Tensile strength (TS) and elongation percent (%E) of the PVA/S and PVA/GS nanocomposite films were performed on a computerized Instron Universal Testing Machine (Lloyd Instruments, England, UK) in accordance with the American Society for Testing and Materials (ASTM) D638 method. It was operated at a crosshead speed of 20 mm min−1 at 30°C. The samples were cut into dumbbell shape with an overall length of 4 cm, width of the grip section of 1 cm, length of the narrow section of 2 cm, and width of narrow section of 0.5 cm. The average thickness of the samples was 1 mm, and the tensile stress was applied until the failure of the sample.
Water uptake (%)
The water uptake (%) of the nanocomposite films was measured according to ASTM D570-98. The samples (dimensions 25 × 25 mm2) were first dried in a hot air oven for approximately 24 h at 50°C, weighted (
The water absorption capacity is calculated as:

where
Biodegradation studies
PVA/S and PVA/GS films reinforced with SNCs were studied for biodegradability. The samples (3 × 10 cm2) were buried at 10 cm deep in the soil taken from sewage sludge in Jalandhar City (Punjab, India) filled pot. The microbial concentration and humidity were maintained constant, and the soil was kept moist by sprinkling sewage water. The degradation was determined after every 15 days by taking out the samples carefully from the soil, washing it gently with water to remove the soil, and then drying it at 50°C in oven to remove the moisture present. The weight loss percentage (%) of the specimen is calculated as follows:

where
Results and discussion
Grafting of starch was performed with oleic acid in the presence of redox initiators FAS-KPS, and the reaction mechanism is shown in Figure 2. Starch is made up of glucose units that contain reactive hydroxyl groups on their carbons. C2, C3, and C6 are the most reactive hydroxyl groups to get modified. Initiators FAS and KPS initiated the reaction by generating sulfate free radicals which further reacted with water to produce hydroxyl free radicals (equations (1) and (2) in Figure 2). These free radicals reacted with starch and monomer, that is, oleic acid, and produced macro radicals and monomer radicals, respectively (equations (3) to (5) in Figure 2). The reaction between macro radicals and monomer radicals resulted in graft copolymers of starch (equation (6) in Figure 2). The grafting reaction was terminated by homopolymerization as shown in equation (7) in Figure 2.

Reaction mechanism of grafting of starch with oleic acid.
FTIR spectra
The FTIR spectra of starch and graft copolymers of starch are shown in Figure 3(a) and (b). In the spectra of starch (Figure 3(a)), a strong and broad absorption band appeared at 3253 cm−1 and was attributed to the vibrational stretching, associated with free, inter- and intramolecular of bounded –OH groups on the starch. In the spectra of native starch, peaks at 1157, 1080, and 997 cm−1 were due to the –CO bond stretching. 21 The characteristic peaks of the anhydrous glucose ring (O–C stretching) were present at 1157 and 1080 cm−1. 22 The absorption band at 2932 cm−1 was present due to the C–H stretching in the glucose unit of starch.

FTIR spectra of (a) starch and (b) graft copolymers of starch.
In the FTIR spectra of graft copolymers of starch (Figure 3(b)), an increase in the intensity of absorption peak present at 2927 cm−1 corresponding to the methyl and methylene C−H stretching associated with fatty acid substituents was observed on grafting. 21 Spectra of grafted starch showed a new peak at 1708 cm−1 attributed to the carbonyl groups, indicating the occurrence of grafting.
Field-emission scanning electron microscopy
The surface morphology of SNCs was analyzed by FESEM as shown in Figure 4. It was observed that SNCs exhibited a platelet-like shape and found to be in the size range of up to 200 nm. The tendency of SNCs to self-aggregate is favored by their platelet morphology, and interactions via hydrogen bonding is favored due to the presence of surface hydroxyl groups. 23

FESEM image of starch nanocrystals.
X-Ray diffraction
XRD was done to examine the change in crystalline and amorphous regions of the starch after grafting and acid hydrolysis treatment.
It is evident that starch gave peaks at 17.97° and 23.19° with relative intensities of 1757 (counts) and 1533 (counts), respectively (Figure 5(a)) while in grafted starch, peaks were at 18.17° and 23.13° with relative intensities 1425 (counts) and 1169.5 (counts), respectively (Figure 5(b)). The value of

X-Ray diffractogram of (a) starch, (b) grafted starch, and (c) SNCs.
Surface morphology
Scanning electron micrographs of starch, grafted starch, pure PVA/S film, PVA/S and PVA/GS nanocomposite films reinforced with 5% SNCs are shown in Figure 6.

Scanning electron micrographs of (a) starch, (b) grafted starch, (c) PVA/S film, (d) PVA/GS film, (e) PVA/S-5SNC, and (f) PVA/GS-5SNC.
The micrographs of starch showed spherical and smooth granules of starch. However, grafted starch showed rough surface with deposition over the surface. The surface of PVA/S film was rough, and the starch granules were visible in the matrix. However, grafting of starch with oleic acid resulted in the homogenous and smooth surface of the PVA/S blend and the starch granules were embedded within the PVA matrix. However, scanning electron micrographs of PVA/GS-5SNC film showed a smooth surface and the SNCs were well dispersed into the matrix compared to PVA/S-5SNC film.
Mechanical properties
TS and % E of various blend films and nanocomposite films are presented in Table 2. TS of PVA film was 42.41 MPa which decreased to 10.15 MPa with the addition of starch in the PVA/S film. The %E of pure PVA film was 209.33%, and it decreased to 61.76% with the addition of 50% starch in the film.
Tensile strength and % elongation at break of PVA/starch nanocomposite film.
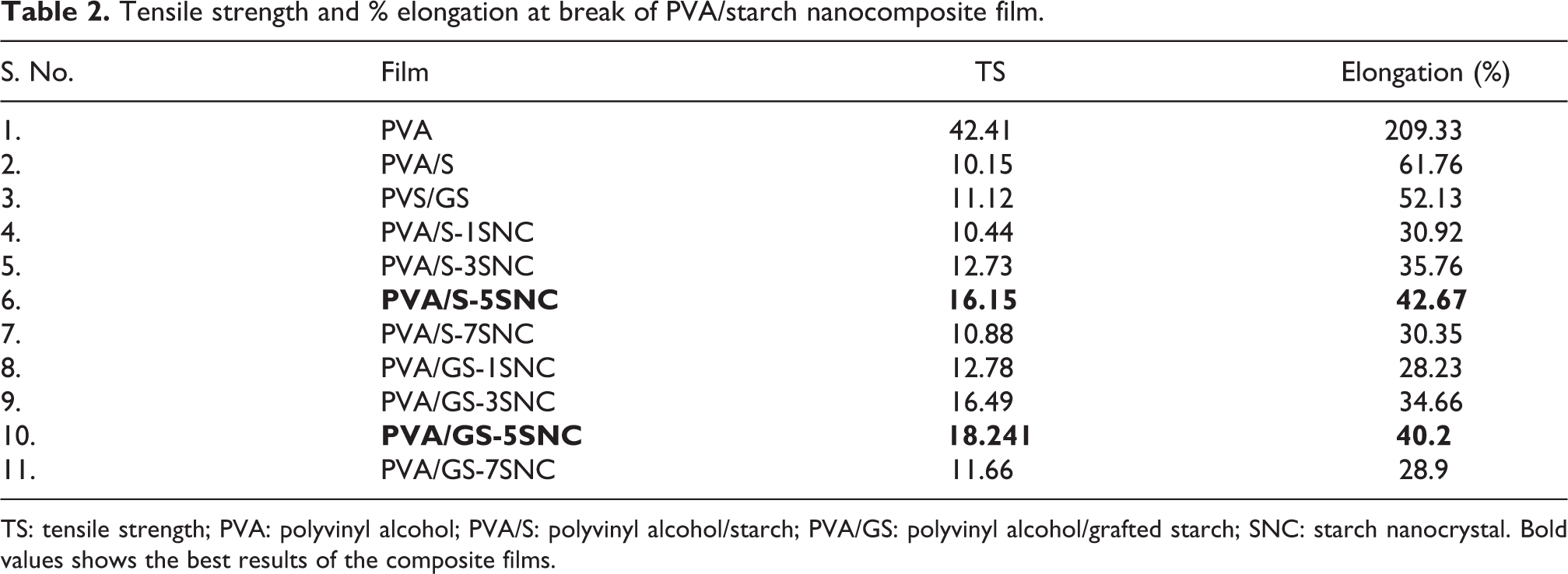
TS: tensile strength; PVA: polyvinyl alcohol; PVA/S: polyvinyl alcohol/starch; PVA/GS: polyvinyl alcohol/grafted starch; SNC: starch nanocrystal. Bold values shows the best results of the composite films.
SNCs were added to the PVA/S mixture in varying proportions (1, 3, 5, and 7%). TS is increased after the addition of SNCs in the matrix. With an increase of SNCs content from 1% to 5% in the PVA/S film, TS of the films increased to 16.15 MPa. TS of the nanocomposite film (PVA/S-5SNC) was much higher and was almost 59.1% of the PVA/S film. TS of the nanocomposite films decreased further with the addition of SNCs content from 5% to 7% in the PVA/S matrix. %E of PVA/S film was 61.76% which decreased to 42.67% with an increase in the SNCs loading from 1% to 5% in the films. There was an increase in the %E of PVA/S nanocomposite films on increasing the SNCs loading from 1% to 5%, which further decreased with the addition of SNCs.
The reason for the increase in the TS of the nanocomposite films with increasing the content of SNCs from 1% to 5% might be the denser structure and greater stiffness of SNCs. 25 The initial increase in %E with the increase in SNCs content from 1% to 5% was linked to the increased movement of polymer chains with increased interactions of SNCs and PVA/S matrix. 26 TS and %E of the nanocomposite films further decreased on increasing the content of SNCs from 5% to 7% in the films because of the larger number of the hydroxyl groups present in the SNCs, which caused the SNCs to link together leading to micro-phase separation and aggregation of SNCs. 27
TS of the PVA/GS nanocomposite films were higher as compared to the PVA/S nanocomposite films. TS of the PVA/GS-5SNC was 18.241 MPa which was 12.9% higher as compared to the PVA/S-5SNC. %E of the PVA/GS nanocomposite films increased to 40.2% on increasing the SNCs content from 1to 5% which further decreased on increasing the SNCs content from 5% to 7% in the film. The grafting of starch has a considerable effect on the mechanical properties of the PVA/GS nanocomposite films. After grafting, homogeneity and compatibility of PVA/S blend improved which increased the TS of the nanocomposite films. The better compatibility of PVA/GS was also shown by SEM images of the nanocomposite films.
Water uptake (%)
Water uptake (%) of PVA/S nanocomposite films as a function of time (days) is shown in Figure 7. In the PVA film, water uptake (%) was found to be around 193% after 1 day which increased up to 202.45% after 6 days. However, it was observed that water uptake decreased in the case of PVA/S films. Water uptake (%) of PVA/S film was observed to be 141.82%, which was lesser as compared to PVA film. Both starch and PVA are hydrophilic materials, and the water uptake (%) was lesser in PVA/S film due to the presence of intermolecular bonds between PVA and starch.

Water uptake (%) of (a) PVA/S and (b) PVA/GS-based nanocomposite films.
Water uptake (%) of PVA/S nanocomposite films reinforced with SNCs is shown in Figure 7(a). After the addition of SNCs in the PVA/S film, there was 38.25% decrease in the water uptake as compared to the PVA/S film. The water uptake of PVA/S-5SNC film was observed to be 112.17% after 1 day which increased up to 117.8% after 6 days of exposure to water. A likely reason for the improvement in water-barrier properties after reinforcement of SNCs with PVA/S matrix was good dispersion of SNCs throughout the matrix which made the films more compact and hydrogen bonding between the polymer matrix and SNCs. 28 The water uptake (%) of the nanocomposite films increased further with an increase in SNCs content from 5% to 7% due to the aggregation of SNCs within the polymer matrix.
Water uptake (%) of PVA/GS nanocomposite films as a function of time (days) is shown in Figure 7(b). Water uptake (%) of PVA/GS film was 112.67% after 1 day of immersion in water which increased to 119.86% after 6 days, which was 21.96% lower than PVA/S film. The water uptake (%) of PVA/GS-5SNC nanocomposite film was 82.86% after 6 days of exposure to water. Grafting of starch with oleic acid led to 34.93% decrease in the water uptake of PVA/GS-5SNC as compared to the PVA/S-5SNC. There was a significant improvement in the water-resistant properties of the nanocomposite films prepared with grafted starch than of starch due to the grafting of hydroxyl groups present in starch with oleic acid. Namazi et al. also performed the grafting of starch with long-chain fatty acid (C8, C12, and C16) and reported the hydrophobic modification of starch after grafting. 29
Biodegradability
PVA, PVA/S, PVA/S, and PVA/GS films were exposed to the soil for 120 days under biodegradable environmental conditions. After burying the films in the soil, it was observed that the size of the films was reduced, and the films became more hard and fragile. The weight loss was observed after exposure which shows film’s deterioration due to the microbes present in the soil. PVA film showed 19.26% loss in weight after 120 days, while PVA/S film showed a higher degradation rate of 25.78% (Figure 8). The degradation rate was maximum in PVA/S nanocomposite films (PVA/S-7SNC) and showed a weight loss of 32.4% after 120 days of soil burial. The degradation rate of PVA/S nanocomposite film was higher due to the ability of the composite to absorb a higher amount of moisture. The water absorbed by the films caused the swelling of the films and allowed the growth of microbes on the surface. Further, it was observed that the degradation rate decreased in PVA/GS nanocomposite films reinforced with SNCs due to the reduction in hydrophilicity.

Biodegradability of (a) PVA/S and (b) PVA/GS-based nanocomposite films.
Conclusions
SNCs were prepared by acid hydrolysis method and characterized by FESEM and XRD techniques. SEM images of the PVA/S and PVA/GS nanocomposite films showed good dispersion of SNCs within the polymer matrix. However, the surface of the PVA/S nanocomposite film was smoother than PVA/GS nanocomposite film. PVA/GS nanocomposite film with 5% SNCs showed a maximum TS of 18.24 MPa. PVA/GS nanocomposite film (PVA/GS-5SNC) showed 12.9% increase in the TS as compared to the PVA/S nanocomposite film (PVA/S-5SNC). %E also increased with increasing content of SNCs from 1% to 5%. Water uptake (%) of the films decreased after the addition of starch and SNCs into the PVA matrix. Further, there was 59% and 35% decrease in the water uptake (%) of the PVA/GS nanocomposite film when compared to PVA/S film and PVA/S nanocomposite film, respectively. Increased crystallinity of the SNCs and their good dispersion into the PVA/S matrix resulted in lower water absorption into the matrix. The addition of starch and SNCs enhanced the biodegradability of the nanocomposite films. The degradation rate was higher (32.4) after 120 days in PVA/S nanocomposite films. PVA/S-based nanocomposite films can be a potential alternative to the packaging films prepared by using petroleum-based nondegradable polymers.
Footnotes
Data accessibility statement
The raw/processed data required to reproduce these findings cannot be shared at this time as the data also forms part of an ongoing study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
