Abstract
Chitosan-reinforced starch-based biodegradable composite films were prepared by solution casting. The chitosan content in the films was varied from 20% to 80% (w/w). Tensile strength (TS) and tensile modulus (TM) of the starch-based composites were improved significantly with the addition of chitosan. Water vapor permeability (WVP) and oxygen transmission rate (OTR) of chitosan-reinforced starch-based films showed a significant reduction compared to native chitosan film and indicated better barrier properties to water vapor and oxygen. The water uptake of the films pointed out better hydrophobic character due to the incorporation of chitosan in starch-based films. Thermal stability was also found to increase with the addition of chitosan in starch-based films and was confirmed by thermogravimetric analysis (TGA) and differential thermal analysis (DTA). Fourier transform infrared (FT-IR) spectroscopy supported the molecular interactions due to the reinforcement of chitosan in starch-based films. Surface and interface morphologies of chitosan film and starch/chitosan composite film were examined by scanning electron microscope (SEM) and suggested sufficient homogenization of starch and chitosan in the biodegradable composite films.
Introduction
Petroleum-based synthetic polymers, like polyethylene, polypropylene, and polyvinyl chloride, are used as packaging materials due to their excellent thermomechanical properties and low cost. But these materials are not biodegradable. Therefore, research is in progress to develop alternative packaging materials that are environment-friendly, cheap, lightweight, possess good thermomechanical properties, and provide a good barrier to moisture and gas. In recent years, there has been a marked increase in interest in the use of biodegradable materials for packaging applications. A number of blends using biopolymers can be the alternative to the currently used synthetic polymeric materials. The most common and potential biopolymers are starch, chitosan, alginate, gelatin, and so on. It is expected that these biodegradable polymeric materials will reduce the use of synthetic nonbiodegradable polymeric materials and thus reduce the environmental pollution. The use of biodegradable polymers for packaging offers a solution to the problem of accumulation of nondegradable waste materials. 1 –5
Starch is a naturally occurring polymer, a truly renewable resource, and it is a biodegradable polymer because it can readily be metabolized by a wide range of microorganisms. The most important sources of starch are wheat, potato, rice, tapioca, and corn. Starch is a polysaccharide; it is composed of glucose monomers. Starch is a potentially interesting biodegradable polymeric material due to its availability, low cost, and renewability. Moreover, the use of starch in the plastics industry can reduce dependence on synthetic polymers. Starch is also a type of carbohydrate polymer and consists of a large number of glucose units joined together by glycosidic bonds. Starch is a widely useful polymeric material for making biodegradable plastics, but pure starch-based films possess low mechanical properties. To improve the drawbacks, blending of starch or its derivatives with various thermoplastic polymers is being investigated. 6 –10
Among the biodegradable films, a considerable attention has been given to chitosan because of its unique properties. Chitosan (β-(1, 4)-2-amino-2-deoxy-d-glucopyranose) is a biopolymer derived by deacetylation of chitin, which is the second most abundant biopolymer in nature after cellulose. Chitin is present in the exoskeleton of arthropods such as insects, crabs, shrimps, lobsters, and certain fungal cell walls. A significant research work has been done using chitosan in different fields. 11 –13 The presence of active groups in chitosan molecules favored its use in many fields. Chitosan has been reported as a potential polymeric material of food packaging, especially as edible films and coatings due to its excellent film forming properties. Because of reactive amino and hydroxyl functional groups, chitosan is frequently blended with other polymers or cross-link to improve their functional properties by inducing inter- or intramolecular cross-linking in the polymer matrix. 14 –16
The objective of the present research was to make biodegradable composite films for packaging applications using chitosan and starch via solution casting. Chitosan was used as the reinforcing agent in starch-based films, and the thermomechanical properties were evaluated. Water vapor permeability (WVP) and oxygen transmission rate (OTR) of the films were determined. Surface and interface morphologies of the composite films were investigated by scanning electron microscopy (SEM).
Materials and methods
Materials
Chitosan from crab shell (powder form; viscosity of 200 cP) and starch (from potato, powder form) were purchased from Sigma-Aldrich Chemie GmbH, Germany. The amylose and amylopectin contents in the starch were 25% and 75% respectively.
Preparation of biopolymeric films by casting
At first, 1% chitosan solution (w/w) was prepared using 2% (w/w) acetic acid solution. Films of chitosan were prepared by casting the chitosan solution onto flat silicon-coated Petri dishes and allowed to dry for 24 h at room temperature and at 35% relative humidity (RH). Generally 35% RH is used for casting process for rapid drying of the films. Dried films were peeled off manually using spatula and stored in the desiccator prior to characterization. Starch was dissolved in deionized hot (70°C) water with constant stirring. Then the starch and chitosan solutions were mixed together at different proportions. Composite films were prepared by solution casting with the same parameters mentioned above. Thickness of films was maintained at 100 μm.
Film thickness
Film thickness was measured using a Mitutoyo Digimatic Indicator (Mitutoyo MFG, Tokyo, Japan) at five random positions around the film, by slowly reducing the micrometer gap until the first indication of contact.
Mechanical properties of the films
Mechanical properties such as tensile strength (TS), tensile modulus (TM), and elongation at break (eb) of the films were investigated by the Universal Testing Machine (Hounsfield series S testing machine, H50 KS-0404) with a crosshead speed of 1/mm at a span distance of 25 mm. The dimensions of the test specimen were 60 mm × 15 mm × 0.01 mm. The thickness of the film was 100 µm. The experiment was carried out according to the European standard (ISO/DIS 527-1:2010).
Water uptake tests of the films
Water uptake test of the films was carried out in deionized water at room temperature (25°C). The test was carried out for 60 min. Samples were taken into glass beakers containing 100 ml of deionized water. At set time points, the samples were taken out from the beaker and wiped properly and then reweighed. The water uptake was measured by a weight difference methodology. The equation for water uptake was as follows: Water uptake (%) = (W wet − W dry)/W dry ×100, where W wet indicated the weight of the film after immersion in water and W dry represented the weight of the film before immersion. The geometry of the sample was 60 mm (length) × 30 mm (width) × 0.01 mm (thickness).
Analysis of thermal properties of the films
The thermal test of the films was taken using computer-controlled TG/DTA 6300 system controlled with an EXSTAR 6000 STATION, Seiko Instrument, Inc., Japan. The TG/DTA module uses a horizontal system balance mechanism. All the experiments were performed in the nitrogen atmosphere. Sample weights were 10–15 mg, and heating rate was 10°C/min within the temperature range of 50°C–600°C.
Fourier transform infrared (FT-IR) spectroscopy
FT-IR spectra of the films were recorded using a Spectrum One spectrophotometer (Perkin-Elmer) equipped with an attenuated total reflectance (ATR) device for solid analysis and a high linearity lithium tantalate (HLLT) detector. Spectra were analyzed using the Spectrum 6.3.5 software. Films were stored at room temperature for 72 min in a desiccator containing saturated NaBr solution to ensure a stabilized atmosphere of 60% RH at 20°C. For FT-IR investigation, films should be stabilized at 60% RH, otherwise noise peaks could arise due to higher RH. Films were then placed onto a zinc selenide crystal, and the analysis was performed within the spectral region of 650–4,000/cm with 16 scans recorded at a 4/cm 10 resolutions. After attenuation of total reflectance and baseline correction, spectra were normalized with a limit ordinate of 1.5 absorbance units. Resulting FT-IR spectra were compared in order to evaluate the effects of starch filling in the chitosan-based films, based on the intensity and shift of vibrational bands.
WVP
The WVP tests were conducted gravimetrically using an ASTM procedure. 3 Films were mechanically sealed onto Vapometer cells (No. 68-1, Twhing-Albert Instrument Company, West Berlin, New Jersey, USA) containing 30 g of anhydrous calcium chloride. The cells were initially weighed and placed in a Shellab 9010L-controlled humidity chamber (Sheldon Manufacturing Inc., Cornelius, Oregon) maintained at 25°C and 60% RH for 24 h. According to ASTM procedure, 3 60% RH must be maintained. The amount of water vapor transferred through the film and absorbed by the desiccant was determined from the weight gain of the cell. The assemblies were weighed initially and after 24 h, for all samples and up to a maximum of 10% gain. Changes in weight of the cell were recorded to the nearest 10− 4 g. WVP was calculated according to the combined Fick and Henry laws for gas diffusion through coatings and films, according to the equation: WVP (g.mm/m2.day.kPa) = Δw.x/A.ΔP, where Δw is the weight gain of the cell (g) after 24 h, x is the film thickness (mm), A is the area of exposed film (31.67 × 10−4 m2), and ΔP is the differential vapor pressure of water through the film (ΔP = 3.282 kPa at 25°C).
OTR
The OTR was measured using an OX-TRANS® 1/50 (MOCON®, Minneapolis, USA) machine. During all experiments, temperature and relative humidity (RH) were held at 23°C and 0% RH. For OTR measurements, OTR is highly dependent on percentage RH. Biopolymers are susceptible to higher RH, so in this investigation 0% RH was used. The experiments were done in duplicate and the samples (dimension: 50 cm2) were purged with nitrogen for a minimum of 2 h, prior to exposure to a 100% oxygen flow of 10 ml/min.
SEM
Film samples (5 × 5 mm) were deposited on an aluminum holder and sputtered with gold–platinum (coating thickness, 150–180 Å) in a Hummer IV sputter coater. SEM photographs were taken with a Hitachi S-4700 FEG-SEM (Hitachi Canada Ltd., Mississauga, Ontario, Canada) at a magnification of 40,000×, at room temperature. The working distance was maintained between 15.4 and 16.4 mm, and the acceleration voltage used was 5 kV, with the electron beam directed to the surface at a 90° angle with a secondary electron imaging (SEI) detector.
Statistical analysis
For each measurement, five samples in each replicate were tested. Analysis of variance and Duncan’s multiple-range tests were used to perform statistical analysis of all results, using PASW Statistics Base 18 software (SPSS Inc., Chicago, Illinois, USA). Differences between means were considered to be significant when p ≤ 0.05.
Results and discussion
Mechanical properties of chitosan-reinforced starch-based composite films
Chitosan (20%–80% by wt) was added in starch-based films to investigate the effectiveness of chitosan as reinforcing filler. TS values of starch-based films was improved significantly (p ≤ 0.05) with the addition of chitosan. Figure 1 shows the effect of chitosan on TS values of starch-based films. For 20%, 30%, 40%, 50%, and 80% addition of chitosan, the TS values were observed to be 27, 30, 38, 47, and 53 MPa, respectively. The starch-based films became brittle below 20% chitosan content. So, in this investigation, the minimum amount of chitosan was maintained at 20% (by wt). It may be mentioned here that only starch could not form films by solution casting. The TS of chitosan films was found to be 56 MPa (denoted 100% chitosan in figure). Chitosan has excellent film forming capability. 14 So, chitosan containing starch-based films were prepared easily by solution casting. Starch is comparatively cheaper than chitosan. So, biodegradable composite films were successfully prepared using starch and chitosan. The 50% chitosan content starch-based films were almost transparent, but below 50% chitosan containing films were opaque in nature.
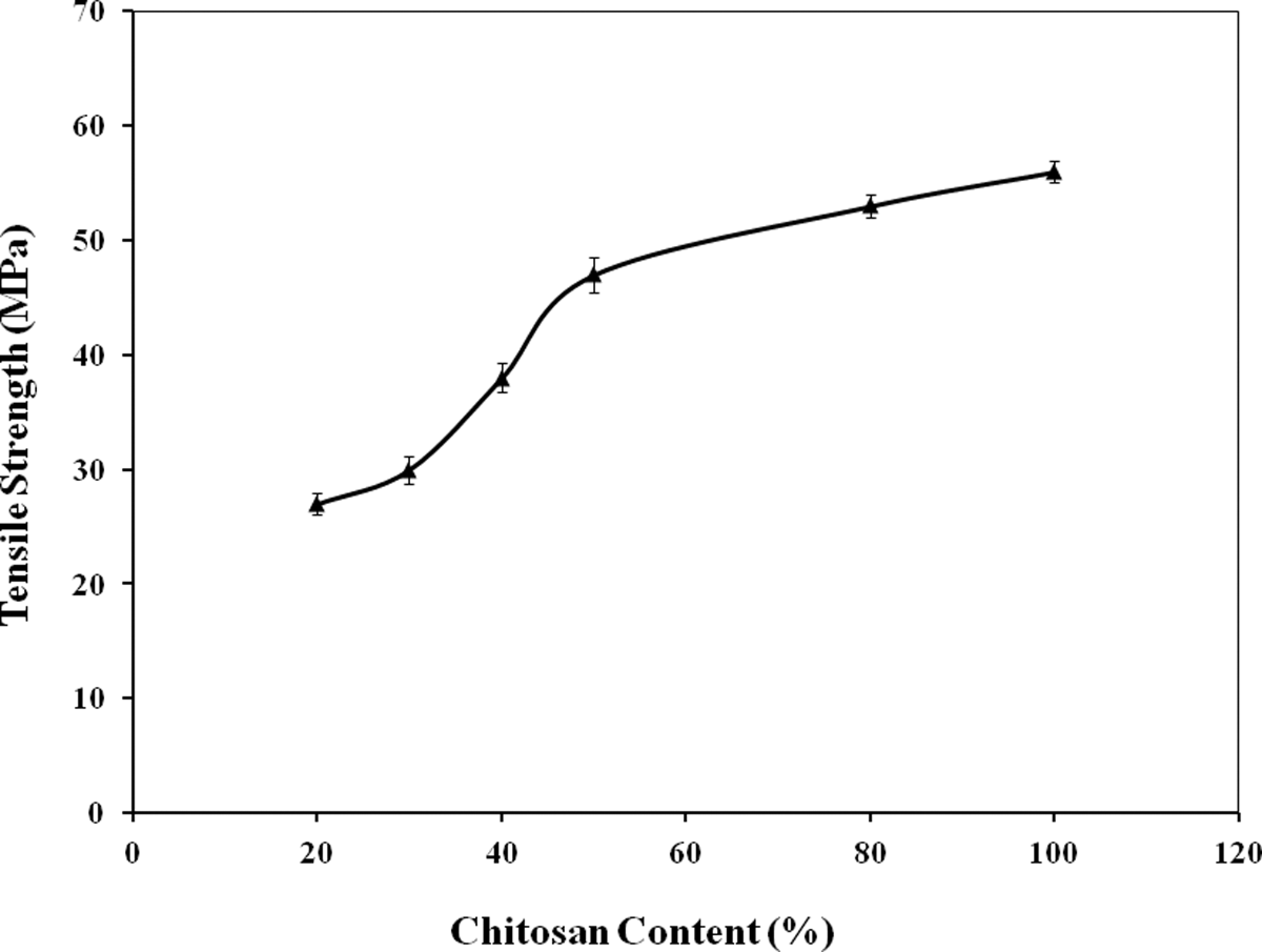
Effect of chitosan on the tensile strength of starch-based films.
Similarly, TM values of starch-based films was also improved significantly (p ≤ 0.05) with the incorporation of chitosan. The effect of chitosan content on the TM values of starch-based films is presented in Figure 2. For 20%, 30%, 40%, 50%, and 80% addition of chitosan, the TM values were observed to be 345, 395, 430, 550, and 750 MPa, respectively. The increased TM values of the chitosan-reinforced starch-based films may be attributed to the increased stiffness of the films by the addition of chitosan. It is to be noted here that TM is a measure of the stiffness of an elastic material and is a quantity used to characterize materials. It is defined as the ratio of the uniaxial stress over the uniaxial strain in the range of stress. In solid mechanics, the slope of the stress–strain curve at any point is called the tangent modulus. The tangent modulus of the initial, linear portion of a stress–strain curve is called TM (or Young's modulus). It is experimentally determined from the slope of a stress–strain curve created during tensile tests conducted on a sample of the material. 17 So, it was found that addition of chitosan improved the stiffness and strength of starch-based films.
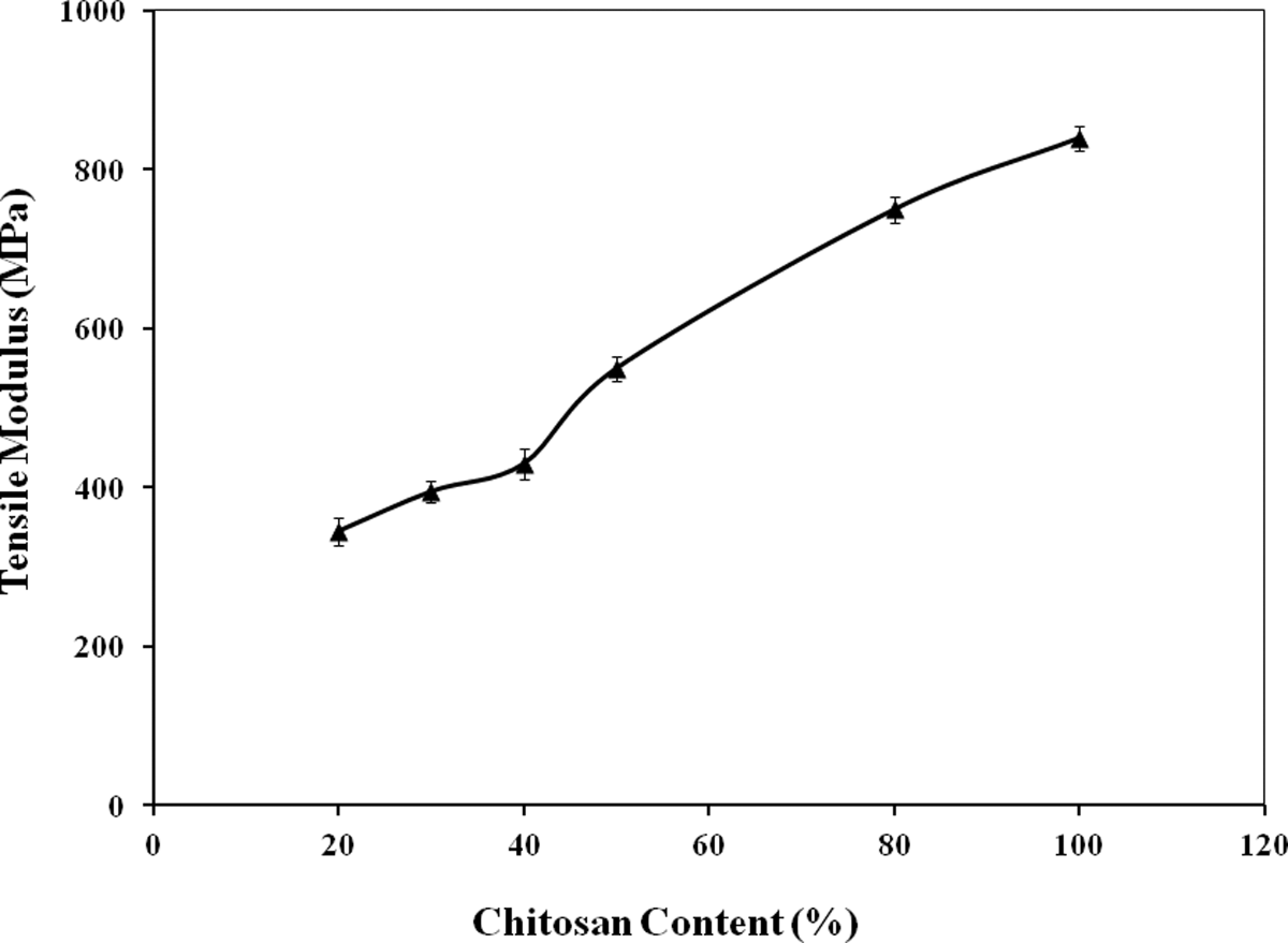
Effect of chitosan on the tensile modulus of starch-based films.
With the rise of strength and modulus, the Eb values of the starch-based films decreased monotonously due to chitosan addition (Figure 3). The Eb values of 20%, 30%, 40%, 50%, and 80% chitosan content films were 23%, 22%, 19%, 16%, and 12%, respectively. Chitosan acted as a reinforcing agent in starch-based biodegradable films. Thus, higher content of chitosan can render the films stiffer. As a result, a decrease in Eb values was observed. Similar results were reported by Pinotti et al. 18 who indicated the reduction in methylcellulose (MC) film flexibility with increasing chitosan concentration. Khan et al 19 reported that chitosan incorporation (5%–36% by wt) in MC-based films significantly improved the strength of the films with a reduction in viscoelasticity. The 50% chitosan containing starch-based films were considered as the optimum because the films had good strength (47 MPa), modulus (550 MPa), and optimum Eb (16%) values. Moreover, the appearance of the films was quite transparent.
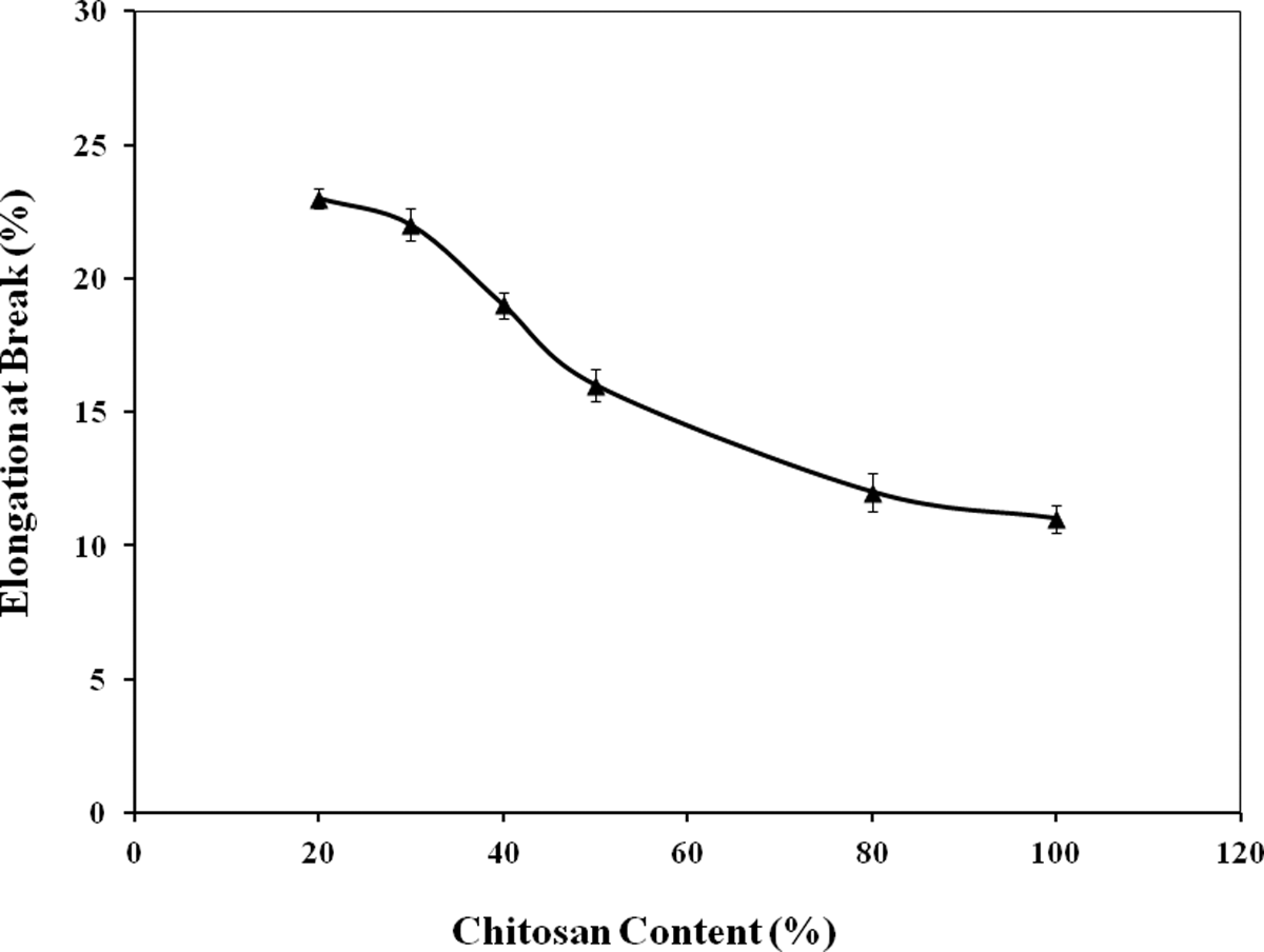
Effect of chitosan on elongation at break (%) of starch-based films.
Barrier properties of the composite films
The WVP of chitosan films and chitosan-reinforced starch-based composite films was measured. The results are presented in Figure 4. The values of WVP for chitosan films was found to be 3.30 g.mm/m2.day.kPa. The values of WVP increased monotonously with an increase in chitosan in starch-based films. The WVP values of 20%, 30%, 40%, 50%, and 80% (by wt) of chitosan-reinforced starch-based composites were 2.00, 2.12, 2.32, 2.45, and 3.10 g.mm/m2.day.kPa, respectively. It was clearly reveled that chitosan-reinforced films had lower WVP values compared to native chitosan film. For 50% chitosan containing films, the WVP reduced to 25.75% from native chitosan film. Reduction in WVP values indicated better barrier to water vapors of the films. The presence of chitosan in starch-based films could increase the tortuosity of the two biopolymers (chitosan and starch), leading to slower diffusion processes and hence, to a lower permeability. 20 The barrier properties are enhanced if the filler is less permeable and has a good dispersion into the matrix. 21 Owing to the higher amount of hydrogen bonds, most of the biodegradable polymeric films are hydrophilic, which is responsible for poor barriers to water vapor. 22 The presence of chitosan in the starch-based composites is responsible for lower WVP values compared to control chitosan films. The WVP values of the chitosan-reinforced starch-based composites indicated excellent barriers to water vapor.
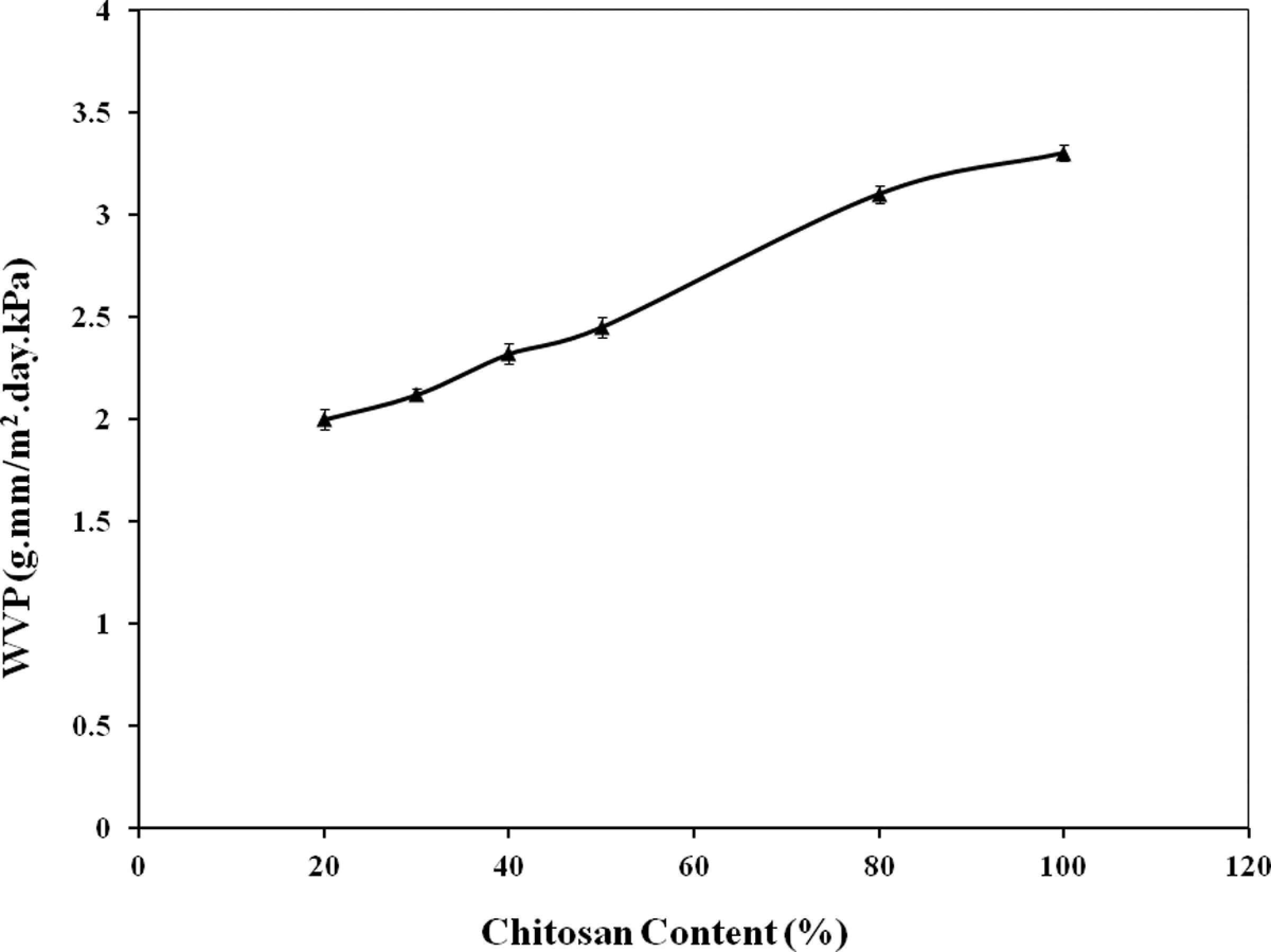
Water vapor permeability (WVP) of chitosan-reinforced starch-based films.
The OTR of chitosan films and starch-based composite films with chitosan was measured and the results are depicted in Figure 5. This is clear that chitosan containing composite films showed lower OTR values than that of the native chitosan films. The OTR value of chitosan film was found to be 2.2 cc/m2.day. Incorporation of 20%, 30%, 40%, 50%, and 80% (by wt) of chitosan in starch-based films, the OTR values were found to be 1.44, 1.56, 1.70, 1.82, and 2.12 cc/m2.day, respectively. Therefore, 50% chitosan-reinforced starch-based composites showed a 17.27% reduction in OTR values. The OTR is defined as the quantity of oxygen gas passing through a unit area of the parallel surface of a film per unit time under predefined oxygen partial pressure, temperature, and relative humidity. 4 The transfer of oxygen from the environment to food has an important effect on food quality and shelf life. Oxygen causes food deterioration such as lipid and vitamin oxidation, leading to sensory and nutrient changes. The OTR is very important since oxygen gas influences the rates of oxidation and respiration in the enclosed food, as fruits and vegetables. Generally synthetic polymers (PCL, polypropylene, polyethylene, etc.) have higher OTR values. 23–24 Addition of chitosan in starch-based films formed a barrier to oxygen and as a result the OTR values decreased.
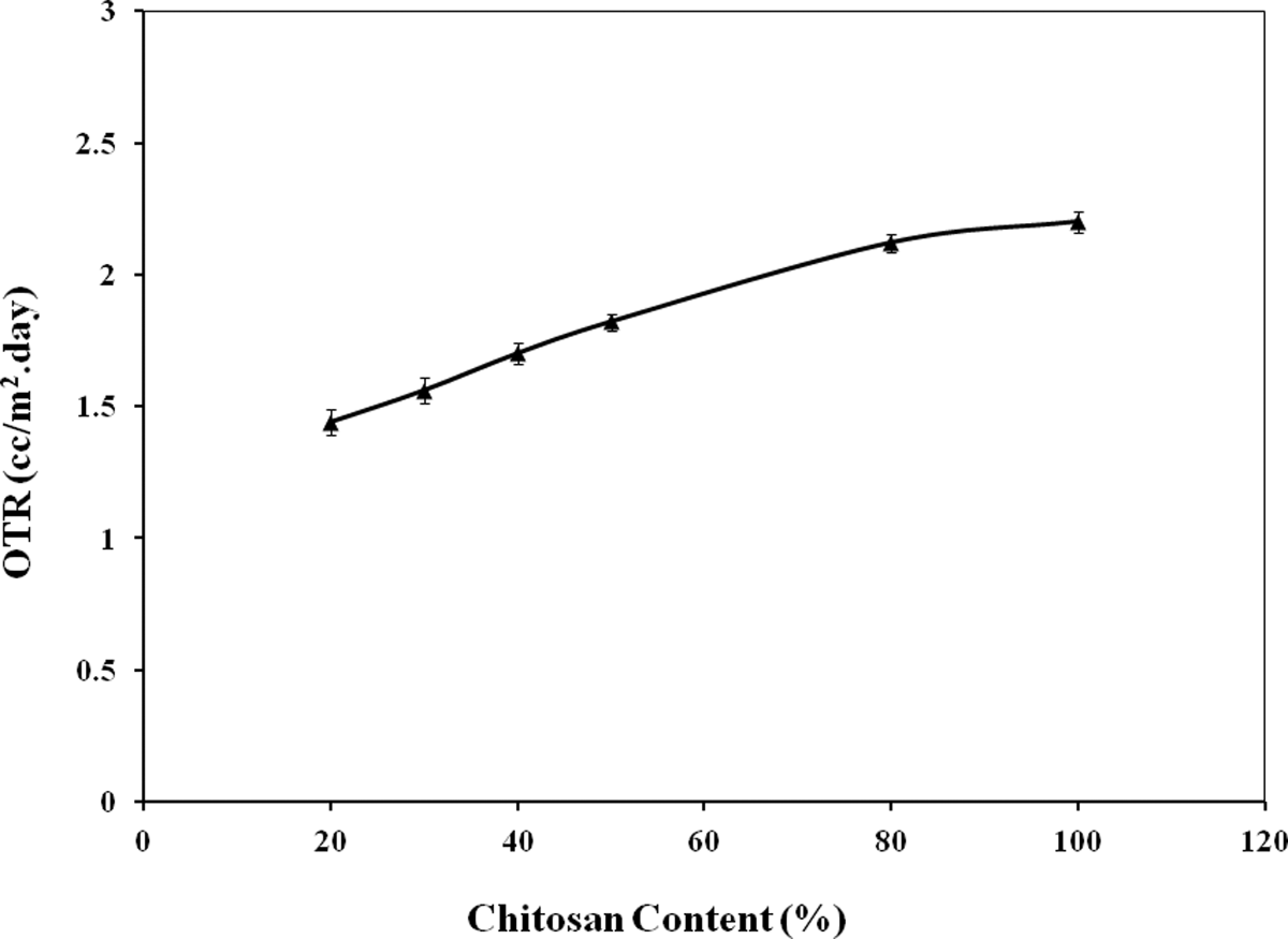
Oxygen transmission rate (OTR) of chitosan-reinforced starch-based films.
Water uptake of the films
The water uptake behavior of chitosan film and chitosan (50% by wt)-reinforced starch-based composite films are shown in Figure 6. It was found that water uptake of starch/chitosan composite films were much lower compared to native chitosan films. Both type of films absorbed water very rapidly. After 2 min, chitosan film absorbed 160% water, whereas the starch/chitosan film absorbed 96% water. At 10 min, chitosan reached 176% and starch/chitosan arrived at 125% of water uptake and both the films showed a gradual decrease in water uptake and indicated the loss of its mass. After 30 min, the water uptake of chitosan and starch/chitosan films showed 155% and 101% of water uptake. Chitosan is water soluble as the salts of various acids present in
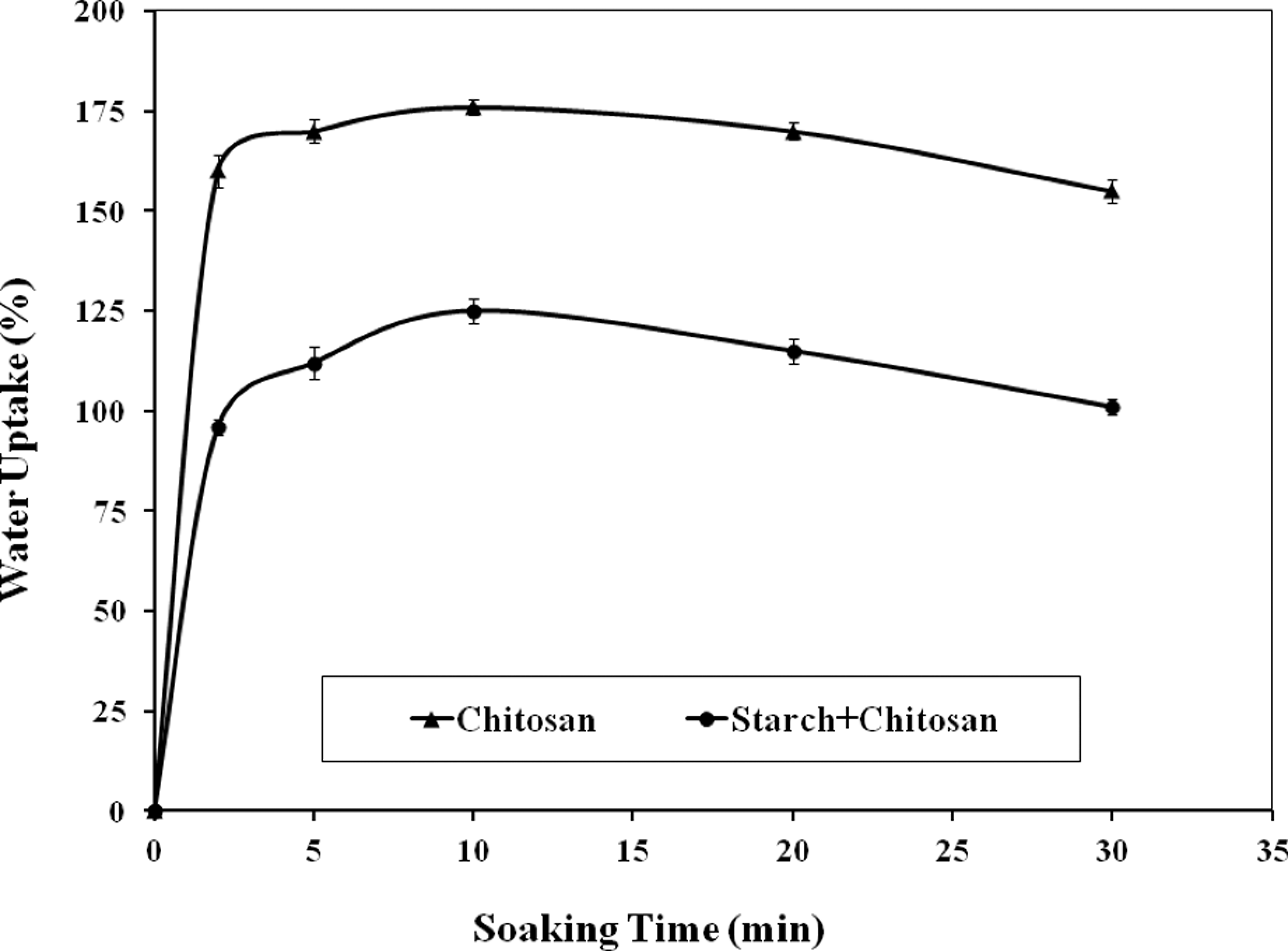
Water uptake of chitosan and chitosan-reinforced (50% by wt) starch-based films.
Structural characterization by FT-IR
The FT-IR analysis was carried out to investigate the molecular interactions due to the addition of chitosan in starch-based films. Figure 7 represented the FT-IR spectra of native chitosan film (a) and chitosan-reinforced starch-based composite film (b). The position of the peaks of chitosan film spectrum is similar to those described by different authors. 26 –28 The transmission peaks of the chitosan films are mainly assignable to (i) amide I vibrational mode at 1,638/cm and (ii) amide II at 1,543/cm, and the peak at 2,960/cm corresponding to symmetric and asymmetric C–H vibrations. The stretching C–N vibration appeared at 1,210/cm and stretching C–O band came at 1,150/cm. Some differences were observed in the FT-IR spectrum after addition of chitosan in starch-based film. Both amide I and amide II peaks shifted slightly to higher frequencies and the peaks became sharp. The stretching C–N vibration peak shifted to 1,230/cm and a new peak appeared at 1,150/cm which indicated hydrogen bonding between starch and chitosan. The stretching vibration for C–O peak also shifted to higher frequencies (from 900 to 950/cm) after the addition of chitosan in starch-based film, which revealed that C–O stretching vibration of chitosan coupled with C–H bending vibration of starch. In general, the FT-IR spectra of chitosan-reinforced starch-based composite films allowed determining the chemical interactions between the two polymers, based on the intensity of identified IR vibrations and their shift related to hydrogen bonding. The obtained results showed significant interactions related to the functional groups of both polymers. The literature 19 also mentioned similar type of phenomenon when chitosan was incorporated in MC-based films. Finally, structural changes were also observed due to the reinforcement of chitosan in starch-based films which supported the higher mechanical strength and better barrier properties of the biodegradable films containing chitosan and starch.
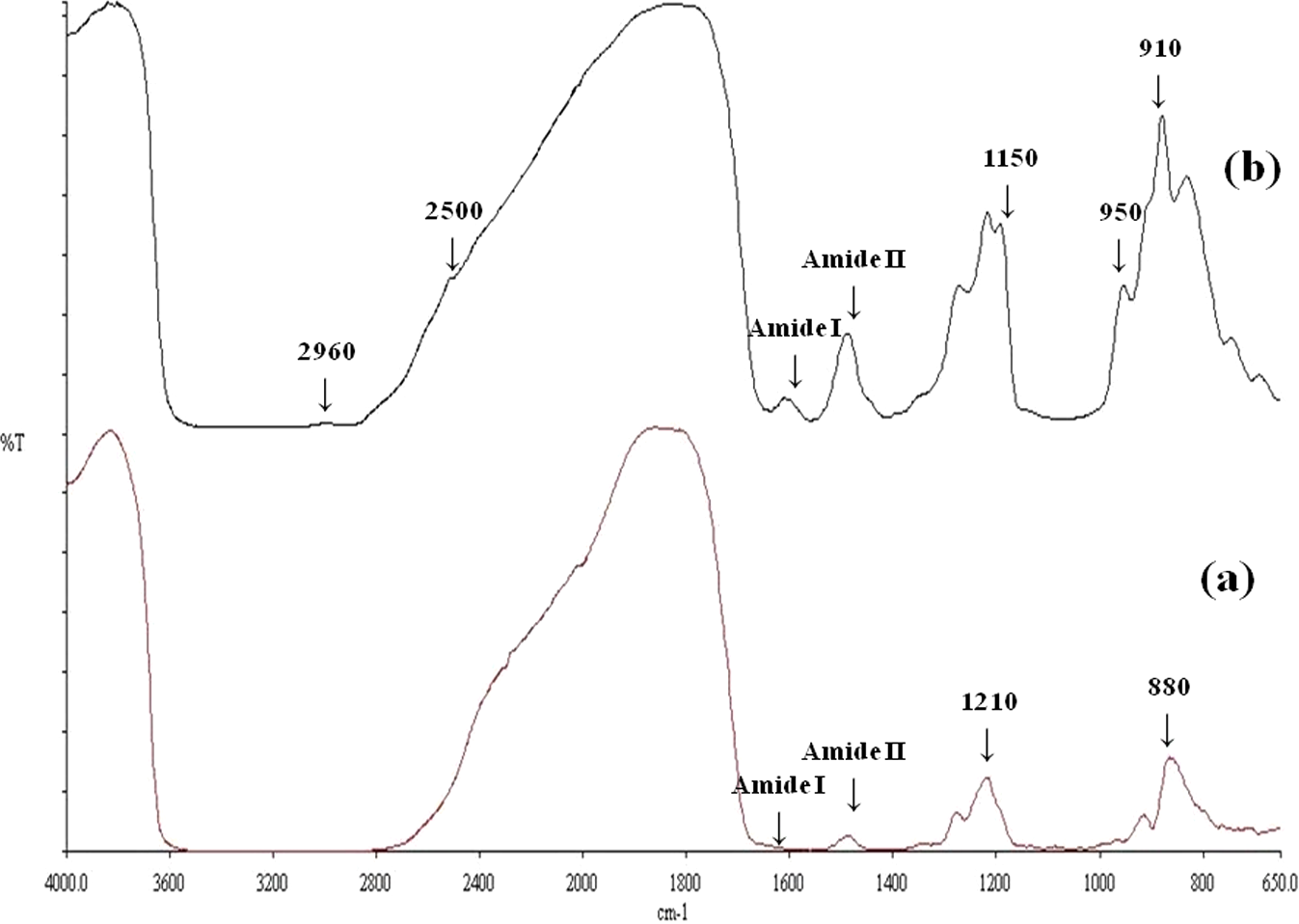
Fourier transform infrared (FT-IR) spectra of chitosan (a) and starch/chitosan (50:50) (b) films.
Thermal properties of the composite films
To investigate the effectiveness of chitosan reinforcement in starch-based films, thermal properties were also investigated. Figure 8 (a)-(b) showed the thermogravimetric analysis (TGA), differential thermal analysis (DTA), and differential thermogravimetric (DTG) thermograms of chitosan films (a) and chitosan reinforced (50%) starch-based composite films (b). Samples were heated from room temperature (25°C) to 600°C. The top line (green colour) of the figures (a-b) indicated TG thermograms. It was found that chitosan film exhibited a major degradation step at 284°C, but the starch-chitosan composite degraded at 304.8°C. These degradation steps corresponded to the thermal decomposition of native chitosan and starch by breaking their polymer network. A minor degradation occurred at 144.1°C for chitosan and 211.9°C for starch–chitosan, which represented the loss of bound water from biopolymers. From these TG thermograms, it is clearly revealed that melting temperatures of chitosan-reinforced starch-based films improved significantly compared to native chitosan films. The DTA thermograms showed that chitosan exhibited two exothermic peaks (74.1°C and 289.3°C), but the starch–chitosan composite displayed two exothermic peaks at 94.5°C and 302.8°C. The DTA also confirmed that starch–chitosan films are much more stable than pure chitosan films. The DTG curve showed one major peak at 283.7°C for chitosan, the degradation rate was found to be 0.409 mg/min. On the other hand, composite film (starch–chitosan) displayed the peak at 304.9°C and the rate was 0.728 mg/min which is higher than chitosan. It indicated that after 300°C, the rate of degradation of starch–chitosan was higher than native chitosan which revealed that at higher temperature starch degraded rapidly. Finally, from these thermograms, it is clear that chitosan improved the thermal stability of starch-based biodegradable films. It was reported 19 that chitosan improved the thermal stability of MC-based biodegradable films. Therefore, our results are in accordance with the published data.
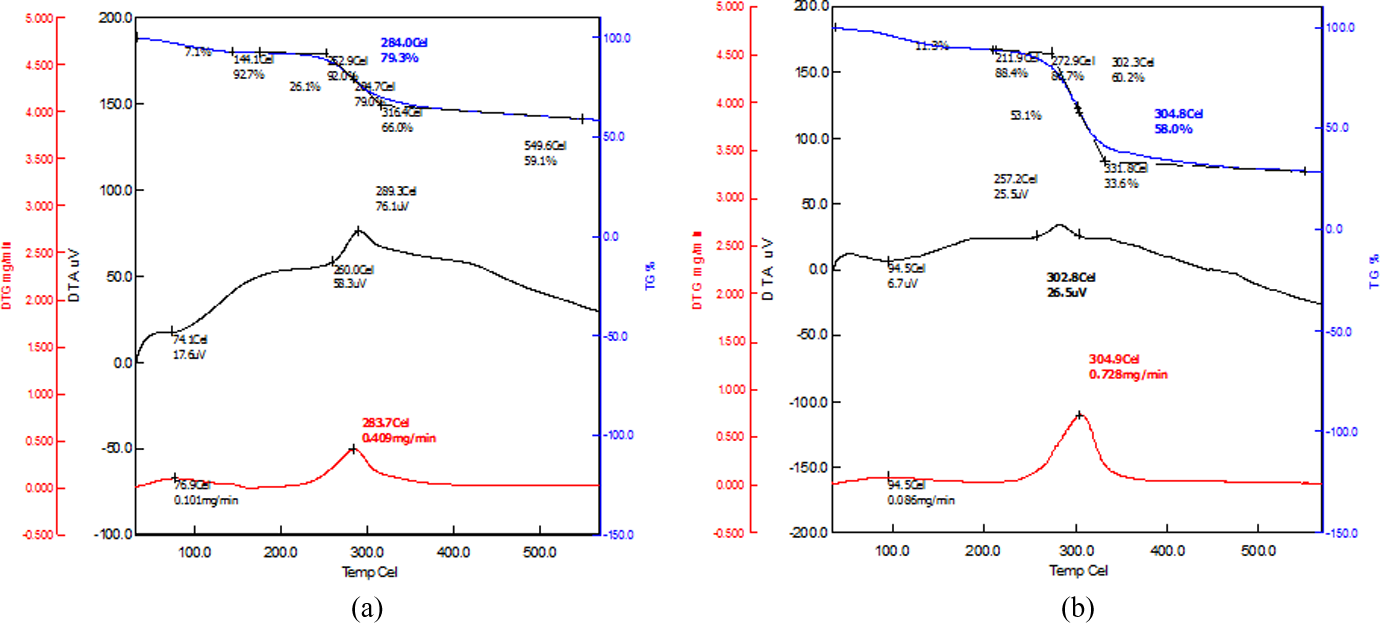
Thermograms of (a) chitosan and (b) chitosan-reinforced (50% by wt) starch-based films.
Investigation of morphological properties using SEM
Figure 9 represented the surface morphology of chitosan (a), and chitosan-reinforced (50% by wt) starch-based (b) films. The surface of chitosan films appeared a homogenous, smoother, and denser, with no gross defects. The smooth and homogenous surface of the films is an indicator of the structural integrity of the observed films, and thus good mechanical properties were obtained. It also indicated better solubilization and homogenization of chitosan in aqueous medium. But chitosan-reinforced starch-based films showed rough and irregular surface with bubbles as compared to pure chitosan films. Figure 10 showed the interface morphology of chitosan (a) and chitosan-reinforced starch-based (b) films. Similar phenomenon was also observed in the interface as like as surface. From this SEM observation, it was found that sufficient homogenization occurred between starch and chitosan in the biodegradable composite films. Pinotti et al. 18 reported that the surface of the MC-based biodegradable films became opaque due to chitosan addition.

Surface morphology of (a) chitosan film and (b) chitosan-reinforced (50% by wt) starch-based film.

Interface morphology of (a) chitosan film and (b) chitosan-reinforced (50% by wt) starch-based film.
Finally, chitosan-added starch-based films were prepared by solution casting. Both strength and modulus improved with the rise in chitosan content in starch-based films. It was found that chitosan-reinforced films had lower WVP values than that of native chitosan film. Similarly, chitosan-containing composite films also showed lower OTR values than that of the native chitosan films. It was also noticed that water uptake of starch/chitosan films was much lower than that of chitosan films. Chitosan significantly improved the thermal stability of starch-based biodegradable films. From this investigation, this is clearly revealed that chitosan acted as a good reinforcing agent in starch-based films. It was reported 29 that chitosan/corn starch blended films (50:50) had the TS value of 24.7 MPa. Here, we observed 47 MPa for the same concentration of chitosan and starch. We used potato starch and, moreover, better homogenization could improve the strength of the films. The TS value of of chitosan reported here (56 MPa) is almost similar to the published value (60 MPa). It was also mentioned that WVP values improved with the increase in chitosan content in starch-based films. So, our results are in accordance with the published values. Chitosan is a positively charged polymer which has strong film forming capability. Chitosan showed strong cationic characteristics and that can easily react with negatively charged polymer/molecules. 30 On the other hand, starch behaves like a negatively charged polymer. 31 As a result, the amino groups of chitosan might react with the hydroxyl groups of starch and thus improved the thermomechanical properties of the films.
Conclusion
Chitosan-reinforced starch-based biodegradable composite films were prepared by solution casting. Chitosan contributed to the improvement in mechanical (strength and modulus) and barrier properties (water vapor and OTR) in starch-based films, which indicated the potentiality of chitosan as reinforcing agent. It was also noticed that chitosan incorporation in starch-based films significantly improved the thermal stability of the films. Structural changes were proved due to chitosan addition in the films. The surface and interface morphologies indicated better homogenization of the two biopolymers (chitosan and starch). The collectivity of the above observations indicated that chitosan was performed as satisfactory filler for the production of biodegradable starch-based packaging films.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
