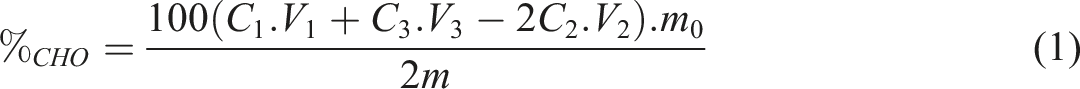Abstract
Dispersion in the melt is a very serious issue that affects properties of composites based on cellulosic fillers as these materials tend to form agglomerates. The aim of this study is to test the efficiency of various methods to improve the dispersion of cellulosic fillers in PBAT polymer. First, periodate oxidation treatment was carried out on the fillers in an attempt to reduce the amount of hydroxyl groups responsible of the agglomeration of cellulosic fillers in composite materials. Secondly, as several studies have demonstrated the positive effect of using tert-butanol (TB) as a freeze-drying medium for preventing the aggregation of cellulosic particles, this method was tested to produce composites with reduced amount of agglomerates. Finally, the addition of a third component as a compatibilizer which has a similar chemical structure to cellulose such as starch was also tested. No significant improvement of mechanical properties was noticed in using TB as a freeze-drying medium or with periodate oxidation treatment on the cellulosic fillers at least in improving dispersion in PBAT composites. However, the addition of starch as a compatibilizer has proved its effectiveness through the creation of a percolating network and a better dispersion of the fillers in the polymer matrix.
Introduction
Composite materials made of cellulosic fillers in a polymer matrix are generally obtained by solvent casting, in situ polymerization or melt mixing.1,2 Each technique generates composites with different properties. Solvent casting is not usable from an industrial point of view if it uses organic volatile solvents that could be toxic and harmful to the environment and also because it is a time consuming technique. 3 In-situ polymerization technique is not widely used in the case of the production of bio-composites especially when the polymerization temperature is so high that it may reach the degradation temperature, near 230°C for cellulosic fillers.4,5 Melt mixing consists of adding fillers to molten polymer with shearing stress and deformation in order to homogenize the two components. The relative simplicity of the process makes it really useful on an industrial scale 6 provided that agglomerates are avoided. Indeed, agglomerates are particularly detrimental to the mechanical properties of the composite because they act as stress concentrators that weaken the composite properties. In comparison to other methods, achieving proper dispersion during melt mixing might be difficult, as cellulosic fillers tend to form agglomerates through strong and numerous hydrogen bonds between the surface hydroxyl groups.7-10 Therefore, agglomeration can be avoided by reducing the probability of contact between the cellulosic fillers or the amount of hydroxyl groups. Fewer hydroxyl groups at the surface of the cellulose can be obtained by chemical modification of the surface of the fillers, protecting them from water absorption and, consequently, improving matrix adhesion, dispersion, crystallinity and mechanical properties of the material.11,12 Many chemical modifications of the surface of cellulose are possible considering the reactivity of the hydroxyl groups, such as oxidation, esterification, amidation, carbamation, etherification, nucleophilic substitution, etc… . 13 Oxidation treatments lead to the formation of functional groups, either carboxylic acid or aldehyde functionalities, 14 and result in huge changes of physical and chemical properties.15,16 The most known method to produce aldehydes is periodate oxidation to induce selective cleavage of vicinal 2-3 diol of the glucopyranose ring and formation of two aldehydes replacing the hydroxyl groups.13,17,18 Syamani et al. 19 used such modified cellulose fibers in poly (lactic acid) (PLA) polymer in order to improve adhesion between the polymer matrix and cellulose fibers. 4 h was claimed to be the optimal reaction time regarding the tensile strength of the composites, while further oxidation induced embrittlement. No comparison with composites using untreated cellulose fibers was carried out in this work. Actually, most of research work investigated the chemical method and its applications, 17,20-25 but very few dealt with polymer composites 19,26 and none of them using poly (butylene adipate-co-terephthalate) (PBAT) as a matrix.
Addition of a compatibilizer or a plasticizer which can interact with both the polymer and filler can also be used to overcome the lack of compatibility between a hydrophobic matrix such as PBAT and a hydrophilic filler such as cellulose. Moustafa et al. 27 used twin-screw extrusion to produce composites based on PBAT plasticized with polyethylene glycol (PEG), with a lignocellulosic filler (LCF) obtained from coffee grounds. They noticed that the addition of 15%-wt of PEG plasticizer to the PBAT/LCF composites promoted better homogeneity and dispersion of the filler in the polymer matrix. It was concluded that PEG plasticizer had a significant compatibilizing effect as chemical interaction between hydroxyl groups of the filler particles and PEG plasticizer was achieved. Nagarajan et al. 28 extruded biocomposites consisting of two polymers (PHBV/PBAT) reinforced by switchgrass fibers. Addition of lignocellulosic fibers, up to 40%-wt, has been investigated and optimal filler content was 25%-wt. Improvement of the mechanical properties with a higher filler content, up to 30%-wt, required the use of polymeric diphenylmethane diisocyanate, PMDI, as a compatibilizer. Infrared spectroscopy suggested a possible interaction between the PHBV/PBAT/PMDI polymer mixture and the cellulose fibers. Reis et al.29,30 introduced microcrystalline cellulose (MCC) in a PBAT matrix using thermoplastic starch (TPS) as a compatibilizer. Better dispersion was obtained by dispersing the cellulose particles in water and adding them to a mixture of starch and glycerol before the preparation of the PBAT based composites reinforced with MCC/TPS by extrusion. The films obtained were homogeneous with neither agglomerates nor porosities and MCC particles could not be observed. It was asserted that dispersed cellulose was covered with starch due to the chemical similarity of the two components. Fourati et al. 31 have also reported the excellent compatibility between TPS and micro-fibrillated cellulose (MFC). The positive effect of starch on the properties of other composites of polymer with cellulose has also been reported but it is seldomly explicitly mentioned as a compatibilizing effect except in PLA. 32,33
Several studies have demonstrated that using tert-butanol (TB) as a freeze-drying medium during the production of cellulose nano-fibers or nano-crystals prevents their aggregation34-38 but seldomly to produce composites. Petersson et al. 39 studied composites based on PLA and cellulose nanocrystals prepared by solution casting. CNC were either transferred to tert-butanol or modified with a surfactant before freeze drying. Differences in the flake-like structure of CNC after the freeze-drying process proved that tert-butanol was able to reduce aggregation and, as a result, the TB-CNC whiskers were more loosely bonded compared to the untreated CNC. The homogeneity of the PLA/TB-CNC material was improved in comparison to the PLA/CNC. The study showed that PLA/TB-CNC material contained smaller agglomerates which were more uniformly dispersed.
This work was inspired by the research mentioned above, to test different ways to improve the dispersion of various cellulosic fillers (micro-fibrillated cellulose and cellulose nanocrystals) in composites based on PBAT prepared through melt processing methods. Chemical modification of the surface of the cellulosic filler by periodate oxidation, use of tert-butanol as a freeze-drying medium and of corn starch as compatibilizer were contemplated in order to reduce the formation of agglomerates of cellulosic fillers during the composite preparation.
Materials and methods
Materials
Poly (butylene adipate-co-terephthalate) PBAT (Ecoflex®, BASF Germany) was in pellet form with a solid density of 1.25–1.27 g/cm3 and a melting temperature of 119°C. This polymer also shows two separate glass transitions attributed to the aliphatic (−32 ± 1°C) and the aromatic sequences (40 ± 1°C) (see supplementary materials Figure C). The proportions of each co-monomer are 44 mol-% of butylene terephthalate and 56 mol-% of butylene adipate. Micro or nano-fibrillated cellulose (MFC or NFC) was obtained from INOFIB, France. They are referred to as enzymatic freeze-dried cellulose nano-fibers by the producer. Cellulose nano-crystals were purchased from the University of Maine, USA. These nano-fillers are obtained by an acid hydrolysis in sulfuric acid carried out on wood pulp. According to the producer, CNC present a density of about 1.5 g/cm3 in the dry powder form, as well as a length and diameter of about 200 nm and 10 nm, respectively. Corn starch was purchased from Roquette, France.
Preparation of oxidized MFC (O-MFC) and CNC (O-CNC)
Oxidized celluloses were prepared by oxidizing 6 g of either MFC or CNC in water (300 g) with sodium metaperiodate (NaIO4). The oxidation of 30% of the reactive surface hydroxyl groups was found to be enough to improve the dispersion while avoiding a loss of properties of the cellulosic filler. The amount of metaperiodate required for this was calculated by two different methods. Calculation considering the average length and diameter of CNC particle, respectively 200 nm and 20 nm, and the number of cellulose units on the surface of each particle leads to the value of 0.7 g. The other calculation was based on prior literature work 40 that have used surface phosphitylation coupled with 31P NMR and FT-IR analysis to estimate the amount of accessible hydroxyl groups at the surface of the CNC. This amount was 3.1 ± 0.1 mmol/g for CNC with estimated dimensions of 4.8 nm in thickness and 110 nm in length and represents 16.7 mol-% of the total OH groups contained in the CNC. In this case, the required amount of periodate was found to be 0.5 g. Finally, 0.7 g was used to oxidize CNC or MFC. The pH of the reaction was fixed between four and five by adding two to three drops of acetic acid. Suspensions were magnetically stirred for 4 h at 40°C in the dark. Ethylene glycol (0.5 mL) was added at the end to quench the oxidation reaction. The mixture was dialyzed against deionized water for 3 days to remove any unreacted reagents and by-products. The resulting suspension was then freeze-dried to be ready for use in composite preparation.
Chemical titration and determination of aldehyde group (CHO) content
0.2 g (m) of cellulose was dissolved in 10 mL of water and 10 mL (V1) of NaOH aqueous solution (C1= 0.25 mol/L). The mixture was put in water bath at 70°C for 2 min. Then, 15 mL (V2) H2SO4 aqueous solution was added (C2 = 0.125 mol/L) and well shacked. Then, two to five drops of phenolphthalein were added into the solution and titrated with 0.1 mol/L (C3) NaOH aqueous solution until it became pink to purple and did not fade in 30 s (V3). The calculation formula is shown in equation 1:
41
Preparation of MFC coated with starch (S-MFC)
Surface coating of cellulose fillers (MFC) was obtained by dispersing 2.5 g of cellulose in distilled water for 1 day. 7.1 g of starch was then added and the suspension was magnetically stirred for 30 min at 80°C to make the mixing procedure easier. The mixture was finally freeze-dried to be ready for use in composite preparation.
Preparation tert-butanol freeze-dried MFC (T-MFC)
Tert-butanol (TB) freeze-dried fillers were obtained by the following procedure: MFC were first dispersed in tert-butanol and magnetically stirred for 1 day at 35°C, at a temperature above the boiling point of TB. A conventional freeze-drying device was adopted to remove tert-butanol by sublimation for 3–4 days until the mass remained constant for at least 24 h.
Composite preparation
All composites were prepared in the melt using a PolyLab HaakeTM OS Rheomix. The temperature was fixed at 150°C above the melting point of the polymer matrix (120°C) and under the degradation temperature of the cellulosic components (230°C). Preliminary tests have shown that the best mechanical properties of the composite materials were obtained with a rotation speed of 120 rpm, equivalent to a shear rate of 60 s−1, and mixing time of 600 s.
The composite material was then compression molded at 150°C and 100 bars during 3–5 min to obtain plates or films with various thicknesses depending on the intended characterization: 2 mm for mechanical and morphology analysis or 0.5 –1 mm for rheological characterization.
Thermogravimetric analysis (TGA)
Thermogravimetric analysis of the T-MFC was conducted in a TGA Mettler Toledo analyzer. A heating rate of 10 °C/min and a sample weight of 20 mg in an alumina crucible were used. Thermogram was recorded over a temperature range of 30–500°C in nitrogen atmosphere with a flow rate of 80 mL/min.
Trasmission electron microscopy (TEM)
TEM observation was performed on MFC and O-MFC fillers for comparison purpose. 1%-wt of filler was dispersed in water and diluted three times. Ultrasound stirring was applied to separate the fibrils or the nano-crystals as much as possible. It was carried out with an ultrasound energy input of about 160 kJ/kg of liquid during 10 min by intervals of 30 s followed by 10 s of cooling in an ice bath to avoid overheating.
For TEM observation, a drop (10 µL) of the treated cellulose suspension was deposited onto a copper grid with a waiting time of 20 min to sediment the filler particles and then stained with a Uranyless (ready-to-use mix of lanthanides aqueous solution) for 1 minute to create a better contrast.
Hitachi H800 TEM was used at an accelerated voltage of 100 kV and under various magnifications.
Scanning electron microscopy (SEM)
The morphology of the composite samples was observed using a mini scanning electron microscope HIROX SH-4000 M in high vacuum operated at 15–20 kV. The specimens obtained before and after compression molding were frozen in liquid nitrogen, fractured and gold-coated by sputtering during 60–70 s. SEM micrographs on MFC and T-MFC were obtained by spraying a small quantity of the filler on a carbon tape; the samples are then gold-coated by sputtering during 60 s.
Tensile tests
Tensile tests were performed using a Shimadzu tensile device with specimens obtained by compression moulding and cut to dumbbell shape according to ISO 527-2 standards (58 x 5 x 2 mm3). Specimens were dried under vacuum for 10 h at 80°C to homogenize the crystallization history and to evacuate humidity. For each composition, at least five specimens were tested with 10 mm/min crosshead speed at room temperature. A statistical analysis with a two tails Student’s test (t-test) was carried out on series, assuming equal variances, in order to compare the values of modulus, breaking stress and elongation at break, the confidence interval was set to 5%.
Dynamical mechanical analysis (DMA)
Measurements of the variation of the dynamic modulus with temperature at constant frequency were performed using a dynamical mechanical thermal analyser Q800 (TA instruments) on films (0.1–0.3 mm thickness). Experiments were performed with the film under tension. A static pre-load force (0.1 N) was applied to the sample prior to the dynamic oscillation to prevent film buckling. During measurement, the instrument was programmed to maintain the static load at 140% of the force required to oscillate the sample. First, strain sweeps were made on each sample composition to determine the linear viscoelastic range, and to define the composition dependent constant strain to be applied for further experiments. A wide range of temperatures (−50°C to +150°C) was investigated during temperature sweeps at a constant heating rate of 3 °C/min and oscillating frequency of 1 Hz was used.
Shear rheology
Dynamic frequency sweep tests in the melt were conducted using ARES G2 Rheometer (TA instrument). The shear storage modulus G’ and the shear loss modulus G” were measured as a function of angular frequency (between 0.01 and 100 rad/s) at 150°C, using parallel plates of 25 mm of diameter and a gap between 0.5-1 mm. Dynamic strain sweep tests were conducted between 0.01% and 100% at two different angular frequency (1 and 100 rad/s) in all samples to select the strain amplitude within the linear viscoelastic region of the material. The frequency sweeps were performed starting from the highest frequency.
Results and discussion
Effect of oxidation by NaIO4
Aldehyde content on oxidized cellulose is a key parameter that influences the surface properties of the MFC or CNC filler and its adhesion to polar polymer matrix and in turn the mechanical properties of the resulting composite. Therefore, the aldehyde content of oxidized cellulose fillers, after oxidation treatment with NaIO4, was quantified by chemical titration. The calculation of the required amount of metaperiodate to obtain 30% of oxidized hydroxyl groups is made considering the ones on the surface of cellulosic fillers; however, the chemical titration giving the aldehyde content is carried out considering all tested cellulose which means considering hydroxyl groups on the surface and in volume. This explains why a lower percentage of aldehyde content is obtained by chemical titration than the targeted percentage. According to literature, 23 metaperiodate oxidation reaction is more effective on amorphous materials than crystalline materials as hydroxyl groups are much more accessible. The aldehyde content is 0 mol-% in untreated cellulose42,43 and were found to be 15 ± 3 mol-% and 13 ± 3 mol-% respectively for O-MFC and O-CNC fillers and showed no significant difference within the uncertainties. In fact, other parameters can interfere and play and important role such as the difference of dimension of the fillers. As CNC fillers are smaller fillers than MFC, their specific surface is higher which leads to more hydroxyl groups accessible on the surface. These two effects are mutually compensating, resulting in an equal aldehyde content for MFC and CNC fillers.
TEM micrographs in Figures 1(a) and (b) show a comparison between MFC fibrils before and after oxidation. The surface treatment has a visible positive effect in separating the fibrils as it reduces the number of hydrogen bonds that can occur. However, the micrographs also shows that fibrils were degraded during the oxidation treatment and this results in reduction of the dimensions of most of the fibrils from 5-15 µm to 1–3 µm in length and from the 50–400 nm to 10–50 nm in diameter. This will lead to lower mechanical properties, stiffness and breaking strength. Residues of NaIO4 can also affect the composite properties because they may induce oxidation of the polymer matrix. Oksman et al.
44
compounded microcrystalline cellulose treated with N, N-dimethylacetamide containing lithium chloride as fillers in polylactic acid matrix by extrusion and showed that the treatment also deteriorated the mechanical properties of the composite material. Possible residues of DMAc/LiCl combined with high temperature during processing were suspected to be the reasons of degrading the PLA which in term affected mechanical properties. TEM micrographs of MFC: (a) without oxidation treatment (b) after oxidation treatment with NaIO4 and SEM micrographs of: (a) PBAT-5-O-MFC (X 1000) (b) PBAT-5-O-CNC (X 1000).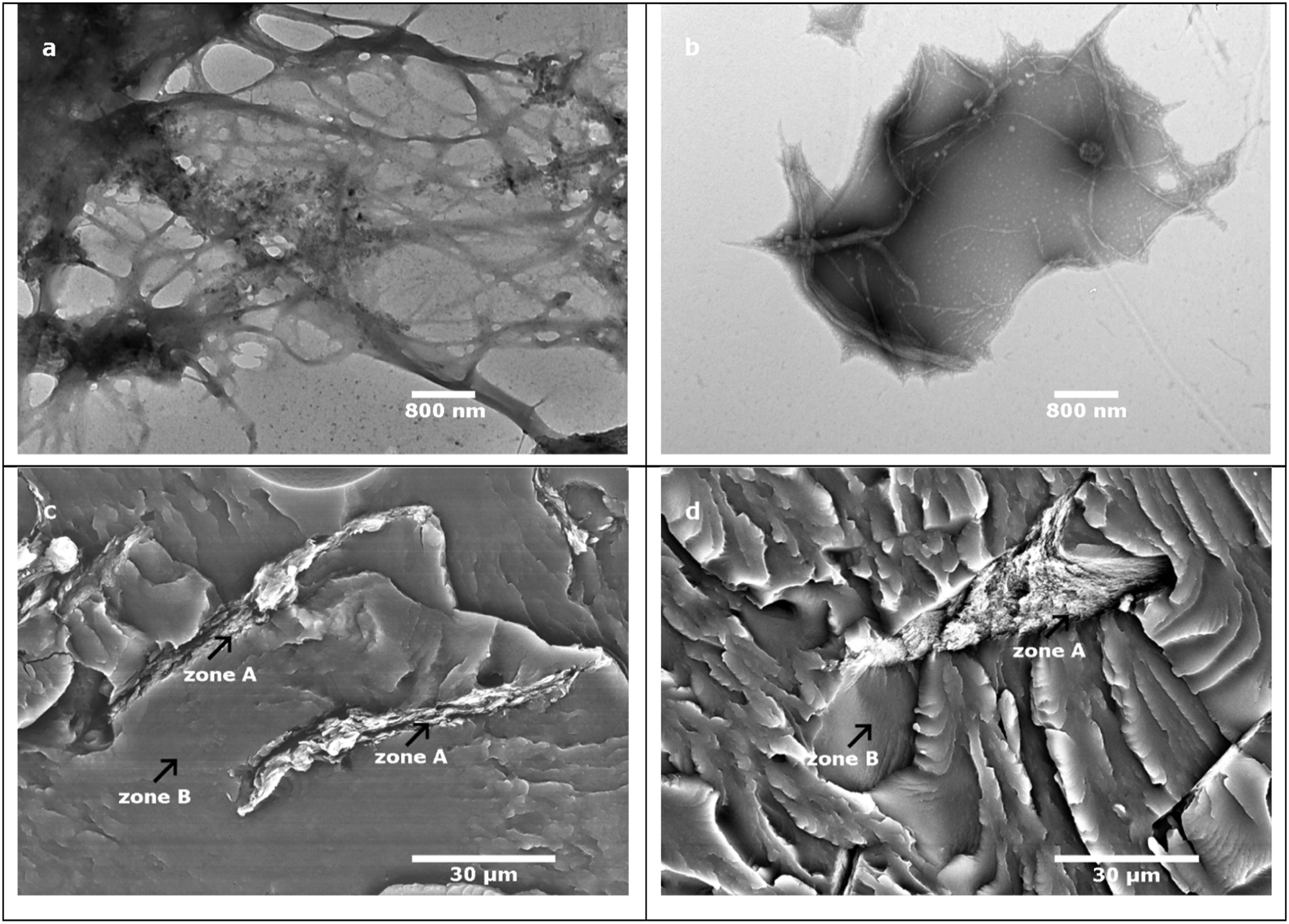
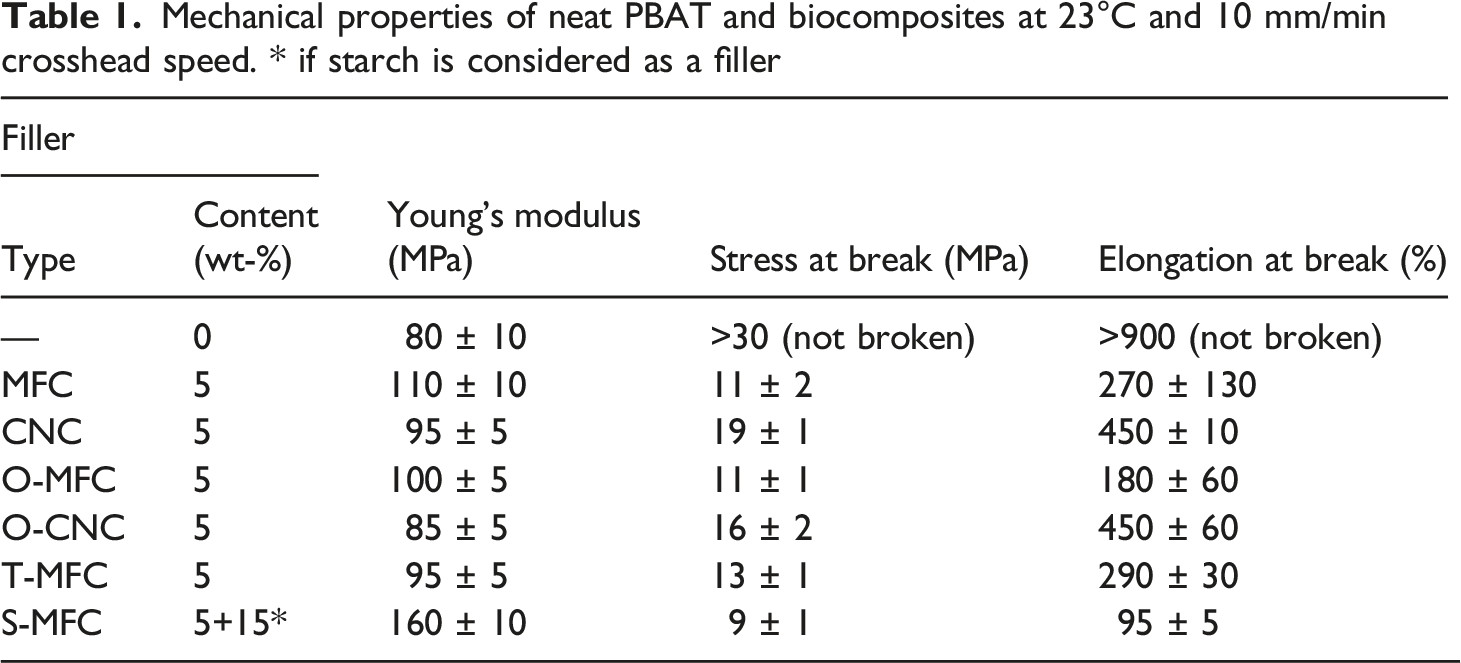
Mechanical properties of neat PBAT and biocomposites at 23°C and 10 mm/min crosshead speed. * if starch is considered as a filler
Dynamic mechanical analysis in solid state and in the melt do not show any plateau at high temperature or low frequency respectively (see Figure A and B in supplementary materials). Only composite made with 5%-wt O-MFC tested in oscillatory shear in the melt showed a high modulus at low frequency, but this is suspected to be an artifact because O-MFC agglomerates were so large, in the order of size of the gap thickness (0.5 mm), that they interfere with the flow. No percolating network was created in relation to a better dispersion of the cellulosic fillers in the polymer matrix.
Effect of dispersion of MFC in tert-butanol (T-MFC)
T-MFC were obtained after dispersion in TB and freeze-drying. Statistical tests on the mechanical properties for composites made with T-MFC show no significative improvement in contradiction to studies demonstrating the positive effect of using TB as a freeze-drying medium for preventing the aggregation of cellulosic particles.34,35 The Young’s modulus as shown in Table 1 even decreased from 110 MPa for composites made with MFCs to 95 MPa for composites made with T-MFCs though Figure 2(b) demonstrated smaller aggregates of T-MFC comparing to regular MFC (Figure 1(a)). Additionally, it was very difficult to avoid any residue of TB. Crystallized TB that was not evacuated during the freeze-drying process as observed on Figure 2(b). It seems that smaller MFC aggregates do not induce any benefit on the dispersion process in PBAT based composites. Besides, on the opposite, TB residues may have adverse effects on composite properties. In particular, the actual weight fraction of MFC filler in the polymer matrix is not controlled and may be reduced. SEM micrographs of: (a) MFC, (b) T-MFC dispersed in TB after freeze-drying (X 1000) and SEM micrographs of composites made with: (c) MFC, (d) T-MFC dispersed in PBAT after freeze-drying (X 1000).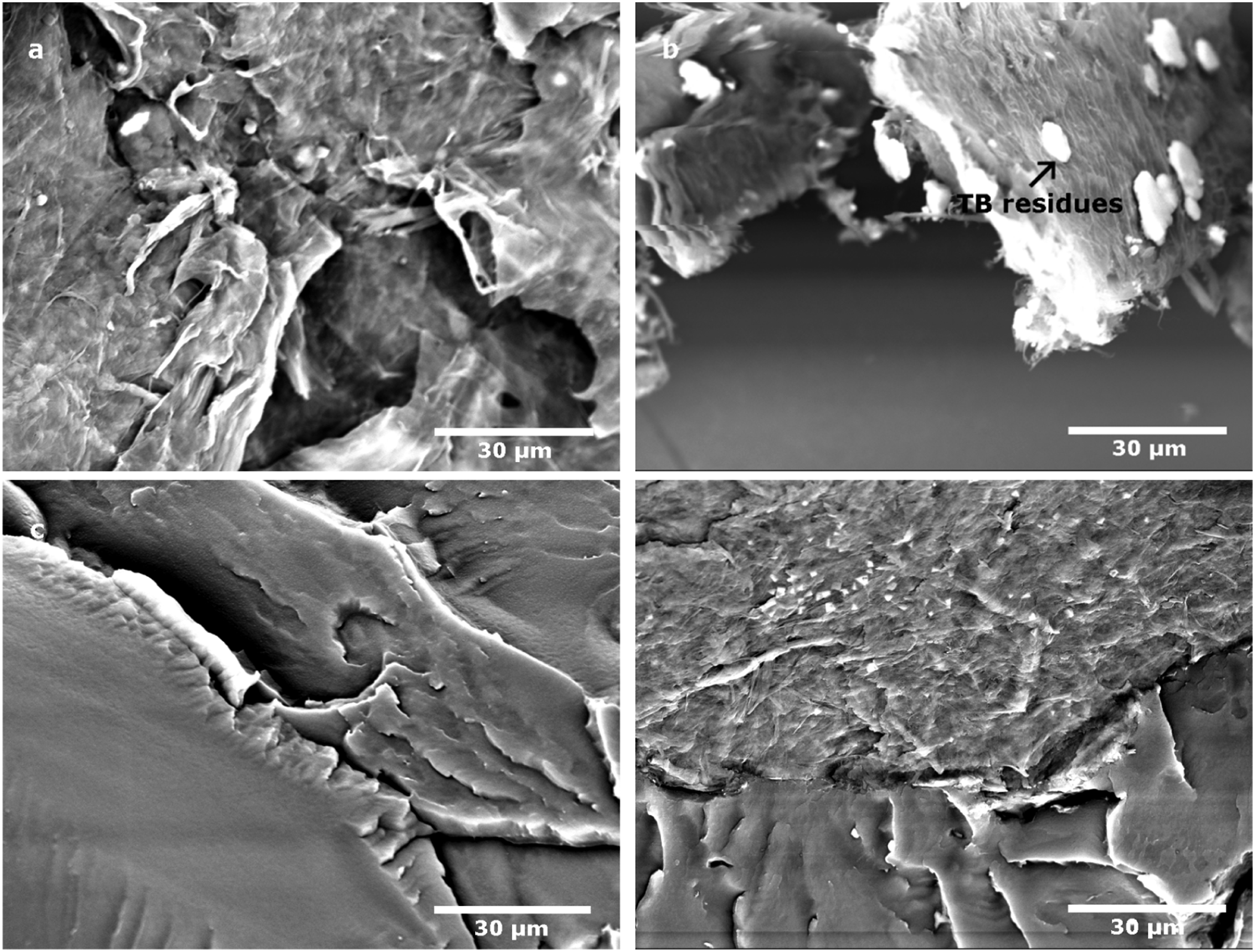
ATG-DTG curves (Figure 3), showing the variation of the weight fraction of T-MFC with temperature and the derivative, display two events corresponding to the vaporization of the TB remaining on MFC fibrils. 4% of the initial mass is lost below 150°C and a second weight loss of 3% occurs between 190 and 270°C. This loss occurring at temperatures much higher than the boiling point of TB (81°C
45
) suggests that TB is tightly bounded to MFC fibrils and that its vaporization during freeze-drying is not efficient. As a consequence, the actual weight fraction of MFCs in composites made with T-MFC is reduced by 0.4 wt-% and becomes significantly lower than 5wt-%. No significant variation of the elongation at break and the ultimate tensile strength was observed (Table 1). ATG (a)-DTG (b) curves of T-MFC dispersed in TB after freeze-drying. The arrows indicate the two events of vaporization of the remaining TB on MFC fibrils.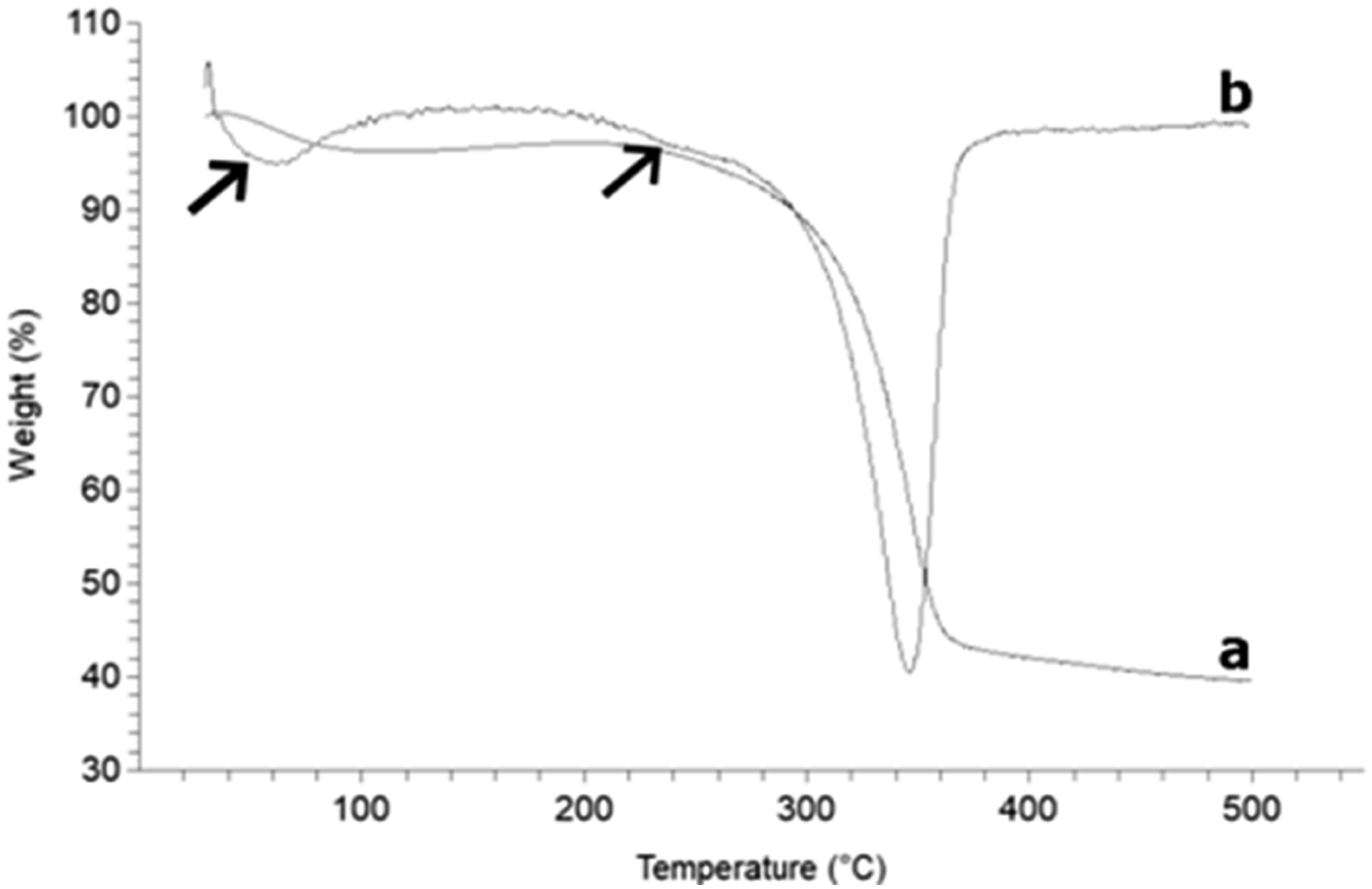
Effect of addition of starch (S-MFC)
SEM micrographs of S-MFC dispersed in swollen starch and then freeze dried (Figures 4(a) and (b)) show starch grains between MFC sheets. Figure 4(c), 4(d), 4(e) and 4(f) show composite materials reinforced with S-MFC. Fibrils in composites made with S-MFC are observable in many zones of the sample. This is not the case in composites made with regular MFC (Figure 2(a)). Better dispersion is achieved after previous dispersion in a starch suspension. In fact, usually MFC are always well dispersed in water but, after freeze drying, -OH groups present on the surface of cellulose are responsible for their re-agglomeration back to the initial state.
46
Starch grains act as spacers between cellulose sheets, preventing them from sticking (through hydrogen bonding) and therefore minimizing the formation of agglomerates after freeze-drying. This loose structure enables the improvement of the dispersion in the matrix. Additionaly, Figures 4(c) and (e) show an empty cavity of one starch grain torn off during the cryo-fracture of the composite sample surrounded by cellulose fibrils which confirms the affinity between cellulose and starch grains and their role as spacers between MFC sheets. SEM. Micrograph of: (a,b) MFC dispersed in starch after freeze-drying, (c,d,e,f) composites reinforced with S-MFC dispersed in starch after freeze-drying.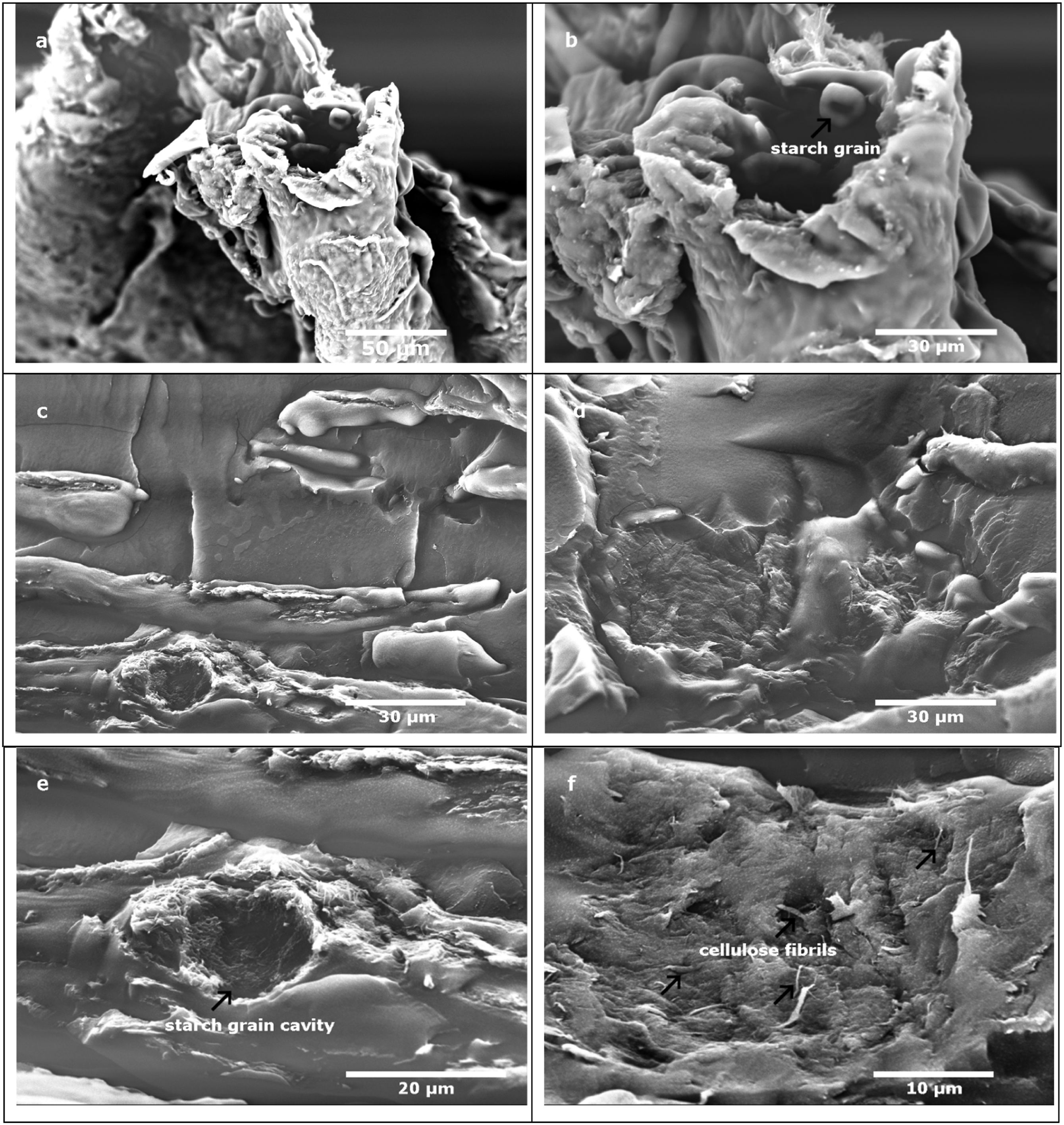
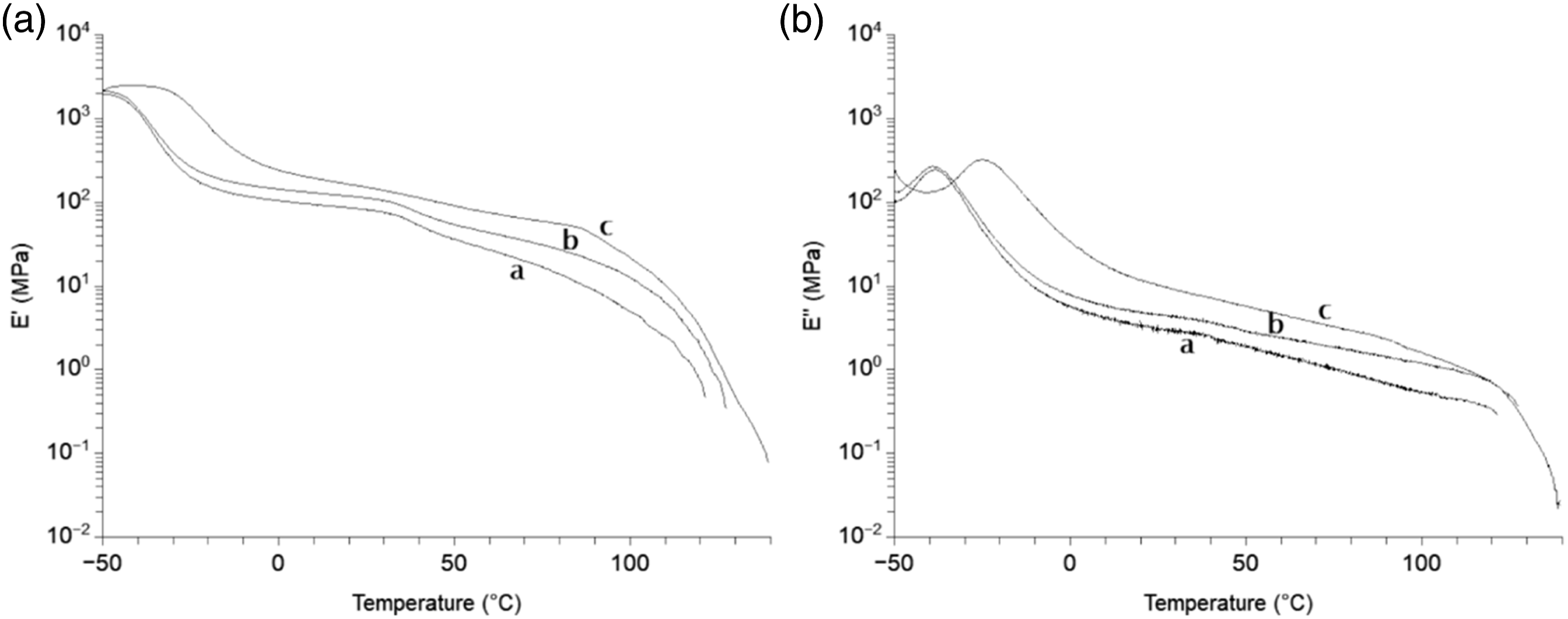
Figure 5 presents the variation of the storage modulus and loss modulus versus angular frequency of composites with 5 wt-% MFC and composites made with 5wt-% S-MFC in the melt at 150°C. A plateau of the modulus can be observed at low frequencies for composite materials reinforced with 5wt-% S-MFC that have undergone the dispersion process in starch compared to the other composite material obtained with the conventional process. Dynamic mechanical analysis results in the solid state are presented in Figure 6; a low modulus can be observed even above the melting temperature, up to 140°C for composites made with S-MFC. None of these results were observed for composite materials reinforced with O-MFC or T-MFC. In fact, these results confirm the formation of a percolating network for composites with 5wt-% S-MFC in agreement with the mechanical properties and especially the high and statistically significant increase of the Young’s modulus which is partly due to the addition of a second reinforcement (starch granules) and secondly to better dispersion of the cellulose fibrils and the resulting percolating network. Storage modulus G’ (a) and loss modulus G’’ (b) in the melt at 150°C versus angular frequency of: (✳) neat PBAT, (▴) composites with 5 wt-% MFC, (•) composites with 5 wt-% S-MFC dispersed in starch and freeze-dried. Tensile storage modulus E’ (a) and loss modulus E’’ (b) versus temperature with oscillating frequency of 1 Hz for: (a) neat PBAT, (b)composites with 5 wt-% MFC, (c) composites with 5 wt-% S-MFC dispersed in starch and freeze-dried.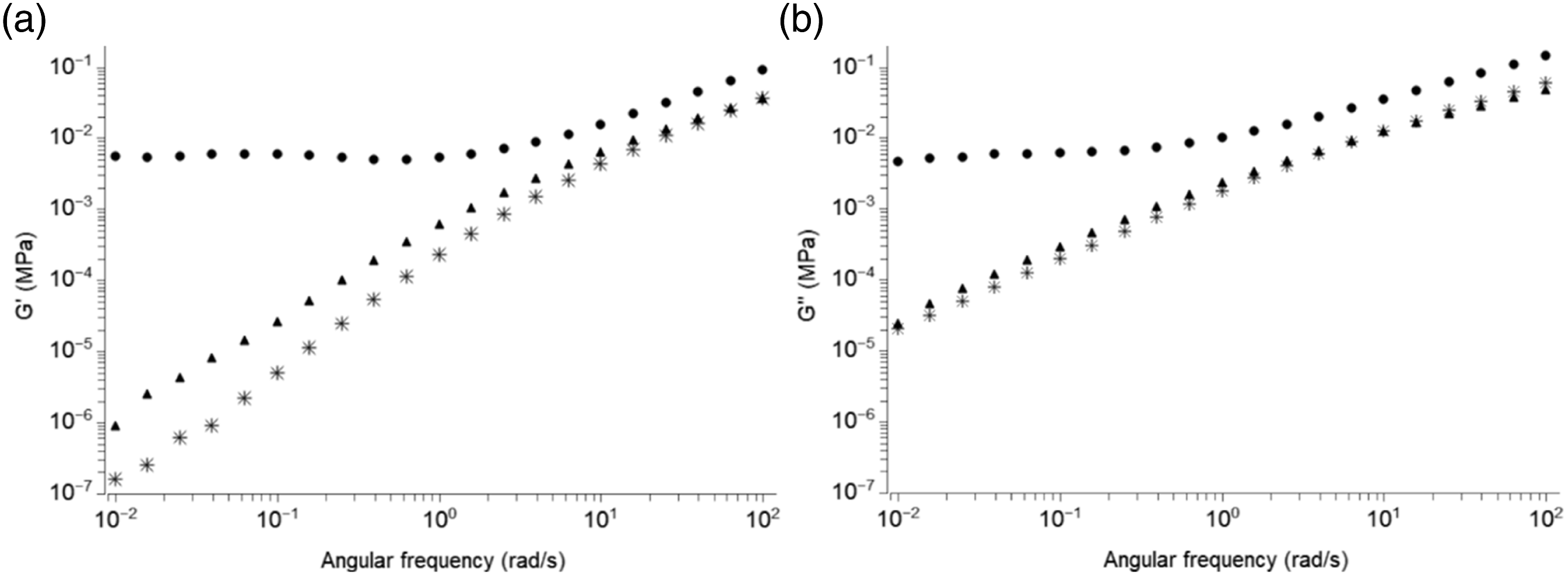
In order to rationalize the effectiveness of the reinforcement and of the adhesion, the C and A factors respectively defined in the papers by Pothan et al. 47 and by Kubat et al. 48 were calculated from DMA results (Figure 6).
Parameter C defined as in equation 2, compares the relative drops of the storage modulus when the material passes the glass transition for the composites and the polymer matrix. Its lowering is a trace of an effective filler.
And c and m refer to composite and matrix respectively.
Similarly, parameter A defined in equation 3 is used to assess the adhesion between the filler and the matrix in the composite since macromolecular mobility in the vicinity of the filler surface should be reduced compared to that in the bulk matrix.
WFor calculation of C, temperatures of −50°C and +50°C were chosen below and above the glass transitions of both aliphatic and aromatic parts of PBAT (−32 ± 1°C and 40 ± 1°C). The decrease from 0.7 to 0.4 of the C coefficient upon addition of starch shows the effectiveness of the reinforcement with its addition. On the opposite, the A parameter was similar in all cases in the whole range of temperature. If the A parameter can be considered reliable in the case of the addition of starch, this would confirm that starch improves the compatibility between the matrix and the filler mostly through its interaction with cellulose by enhancing the dispersion rather than adhesion between cellulose and PBAT.
This explains the increase of the Young modulus without improvement of the tensile strength noticed in Table 1. A secondary reason is that improving dispersion favors a percolating network of fillers which has a favorable effect on the modulus but in turn reduces the ductility of the material preventing any chance to reach the optimal tensile strength at high deformation. Consequently, the material remains fragile.
Surface tension at 25°C and its component for the ingredients and diagram showing the distance in the component space.

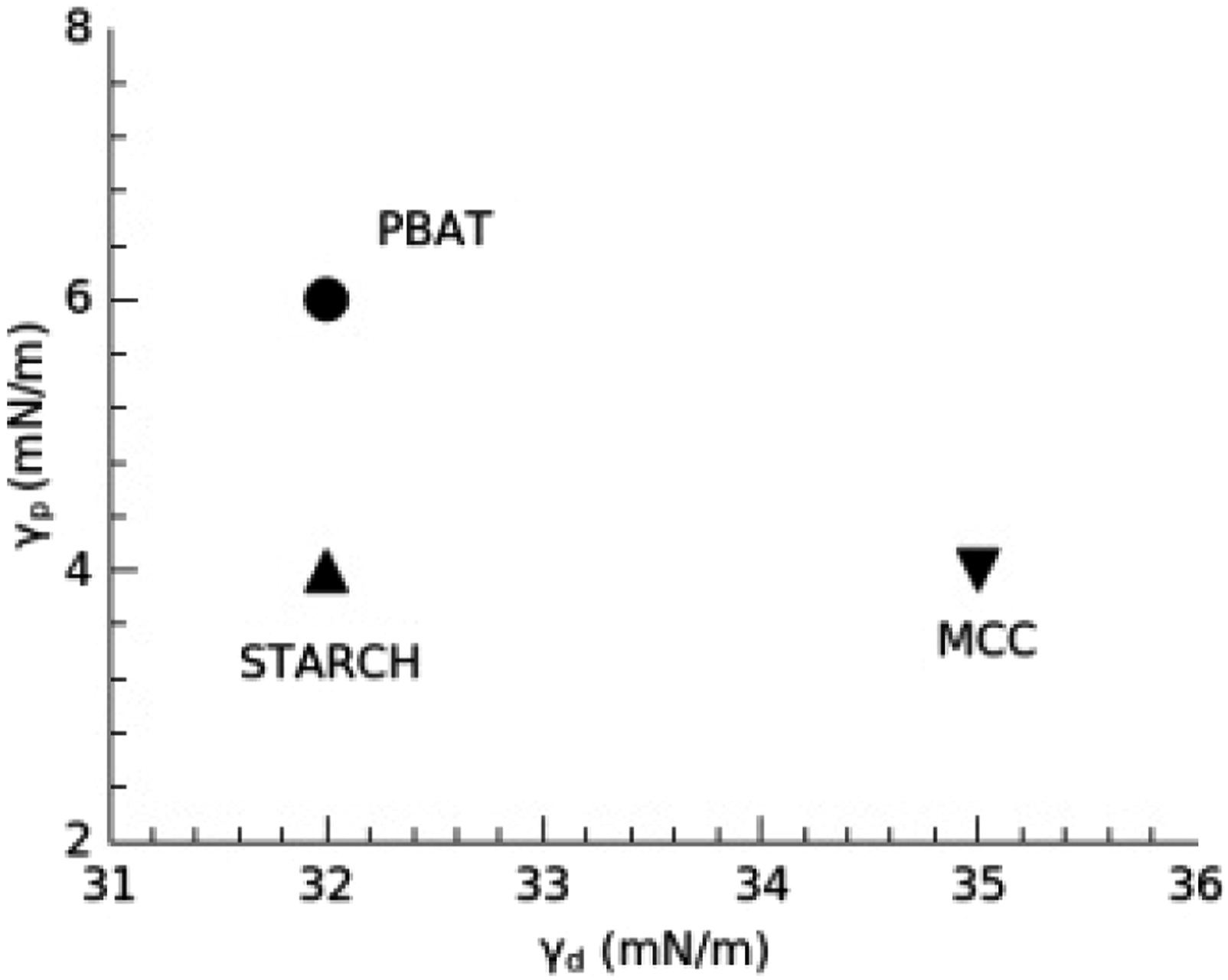
Location of the composite ingredients according to their surface tension at 25°C, presented in the polar and dispersive components space.
Conclusion
This study aims at comparing the relative efficiency of various chemical or physico-treatments of nano and micro cellulosic fillers to improve their dispersion in a polybutylene terephatalate matrix during melt processing. Chemical modification of the surface of the cellulosic fillers by periodate oxidation was first considered in order to reduce the amount of hydroxyl groups as they are responsible for the strong interactions between cellulosic fibrils or nanocrystals (hydrogen bonds) and in turn of their agglomeration in the composite material. This method did not show any significant improvement neither on mechanical properties and nor on dispersion. Actually, the chemical treatment strongly degraded the cellulosic filler; reduction of their dimensions and consequently of their shape factor was demonstrated by TEM imaging.
The use of tert-butanol instead of water as a freeze-drying medium of the cellulosic filler was inspired by literature data as a suitable method to prevent agglomeration of cellulosic fibrils or nano-crystals. Microscopic observations of MFC fillers showed a reduction of the size of agglomerates of cellulosic fillers with the use of tert-butanol but no significant improvement of the properties of PBAT composites. As already observed in other polymer-filler systems, the reduction of size of the agglomerates makes them more difficult to break and higher shear stresses are required during the melt. In addition, it is also very difficult to remove all the tert-butanol during the freeze-drying.
The most successful and efficient method to enhance interaction and dispersion in the polymer matrix was the addition of starch as a compatibilizer in PBAT/cellulose composites. Starch was chosen as a compatibilizer due to its chemical similarity with cellulose. However, starch interacts with both components and it also has a shielding effect to reduce interaction and agglomeration of the cellulosic fillers. This method has proved its effectiveness through the creation of a percolating network confirmed by the presence of a secondary plateau at low frequency in dynamic mechanical analysis in the melt, the increase of the Young’s modulus and a better dispersion of the cellulosic filler.
Supplemental Material
Supplemental Material - The effect of periodate oxidation, tert-butanol treatment, and starch addition in improving melt dispersion of cellulosic fillers in PBAT
Supplemental Material for The effect of periodate oxidation, tert-Butanol treatment, and starch addition in improving melt dispersion of cellulosic fillers in PBAT by Mariam Keskes, Melinda Desse, Mohamed Jaziri and Christian Carrot in Journal of Thermoplastic Composite Materials
Footnotes
Acknowledgement
The authors would like to express their gratitude to Nora Mallouk (CMES) for TEM observations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Campus France (P744702G) and EMORI program.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.