Abstract
Composite materials are produced using thermoplastic starch reinforced with cellulose microfibrils. The cellulose microfibrils are isolated from two different sources and their reinforcement capacity was evaluated. Vegetable cellulose (VC) microfibrils are isolated from vascular bundles of banana rachis, while bacterial cellulose (BC) microfibrils are produced by Gluconacetobacter genus bacteria using pineapple peel juice as the culture media. For this study, both the materials were obtained from Colombian agroindustrial wastes. Composite films were characterized using different techniques, including mechanical tensile testing, attenuated total reflection Fourier transform infrared spectroscopy, and thermogravimetric analysis. The purpose of this study is to assess the effect of different processing methods and cellulose microfibrils content in the composite material behavior. The results showed that the mechanical properties were increased when cellulose microfibrils were added before gelatinization. Significant increments in Young’s modulus and tensile strength of both VC and BC composites were obtained with respect to starch matrix.
Keywords
Introduction
Materials obtained from renewable resources have gained much attention in the last few decades due to the demand for alternative raw materials to replace fossil resources. 1,2 For instance, composites based on biopolymers such as starch and cellulose are attractive materials because of their sustainability, environmental friendliness, and adequate mechanical properties. 3,4
Starch is one of the most studied and promising raw materials for the development of composites, 5 because it is readily available, cheap, and biodegradable. 6 In the 1970s, native starch was transformed into thermoplastic starch (TPS) using plasticizers such as glycerol, 7,8 sorbitol, 9 and sugars. 10 However, the use of TPS is limited in industry, due to its brittleness and poor mechanical properties. 11 Experimentation has been directed at three basic methods to improve the characteristics of TPS. These methods are (1) the evaluation of different processing methods through the chemical modification of starch (esterification and cross-linking), 12 (2) blending of starch with synthetic polymers, 13 –15 and (3) the use of natural fibers like cotton, hemp, sisal, jute, and flax as reinforcing agents in starch. 16,17 In the first two cases, these procedures were used to develop materials with good mechanical properties but their biodegradability was affected. 18 Recently, much attention has been paid to the production of fully degradable composite materials that contain cellulose microfibrils. 19 –21 This is because cellulose is the most abundant biopolymer in nature, and it can be synthesized by plants, animals, and a large number of microorganisms. Similarly, cellulose, like starch, are polymers composed of glucose units linked by β-1,4 and α-1,4 glycosidic bonds. This ensures a good interface between them. 22
Nevertheless, the characteristics of cellulose microfibrils depend on the natural resource from which they are extracted. 23 For example, they can be obtained from agroindustrial waste. Colombia is a country with a considerable level of agricultural production, because of its rich flora and adequate climatic conditions. For instance, the cultivation of different Musaceae species results in 88 wt% of agricultural residues (only 12 wt% is marketable) consisting of fibrous residues, such as rachis. Rachis is composed of cellulose, which can be used as a raw material for isolating novel reinforcing elements for composite materials. 24,25
Another interesting source of cellulose is the bacterial origin. Bacterial cellulose (BC) is chemically equivalent to plant cellulose. It exhibits higher crystallinity, water-holding capacity, mechanical strength, and it is more pure. Moreover, it does not contain lignin, hemicellulose, or other natural components, which makes it an appealing raw material that can be used as a reinforcing component in other materials. 26 As a result, various starch-based composites have been manufactured using different natural fibers. 27 However, there is a limited amount of literature available on cellulose microfibrils, which is obtained from agroindustrial wastes such as rachis and pineapple peel juice that are used as culture media in its production.
The purpose of this study is to assess the effect of different processing methods and cellulose microfibrils content in the composite material behavior. Mainly, variations on processing methods are based on the moment that cellulose microfibrils are added into the starch matrix. Composite films were characterized using different techniques including tensile mechanical testing, attenuated total reflection Fourier transform infrared (ATR-FTIR) spectroscopy, and thermogravimetric analysis (TGA).
Experimental
Materials
Potato starch with 32.8 wt% amylase and 67.1 wt% amylopectin was provided by Almicor Industries Ltd (Bogotá, Colombia). United States Pharmacopeia (USP) grade glycerol and potassium sorbate Generally Recognized as Safe (GRAS)-type additives were provided by Protokimica (Medellín, Colombia). Vegetable cellulose (VC) microfibrils of 5–10 nm in diameter were isolated from the vascular bundles of banana rachis according to the method described by Zuluaga et al. 28 BC microfibrils of 20–70 nm diameter ribbons were produced from the fermented juice of pineapple peels as described by Castro et al. 26
Composite film formation
Composite films were obtained by casting. Depending on the type of cellulose microfibrils, different processing methods were defined. Table 1 and Figure 1 describe these methods. Mainly, changes were necessary due to the higher capacity to absorb water of BC with respect to VC samples. In order to compare the production methods, it was necessary to produce two different TPS matrices, labeled TPS and TPS30, because of their varying starch, glycerol, potassium sorbate, and water contents, as shown in Table 1. The TPS matrix was produced according to the method presented by Famá and coworkers 29 and reinforced with VC microfibrils, while TPS30 matrix was elaborated using the experimental details described by Woehl et al. 30 and reinforced with BC microfibrils.
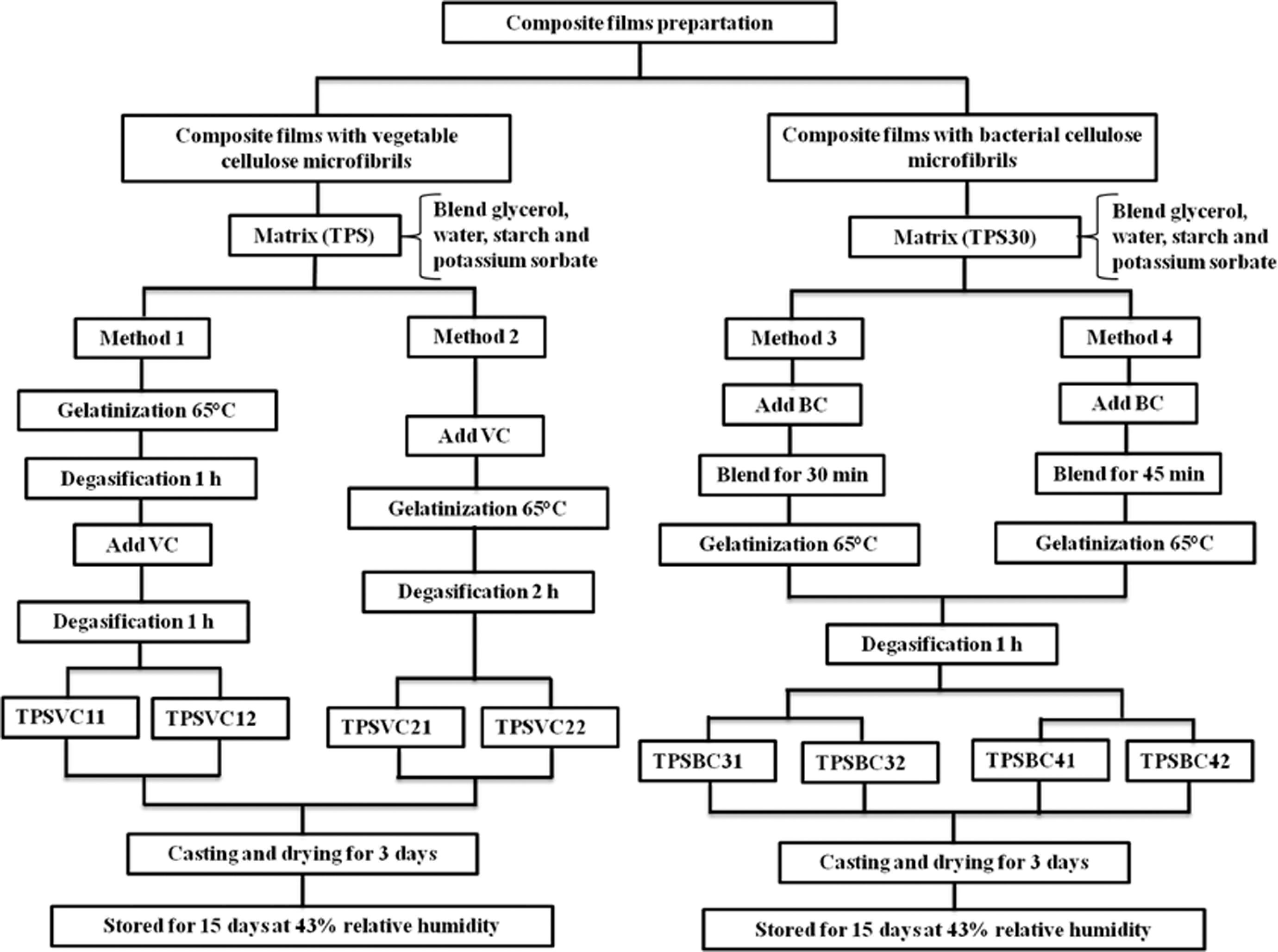
Scheme for the composite film processing methods.
Codification and composition of composite films.
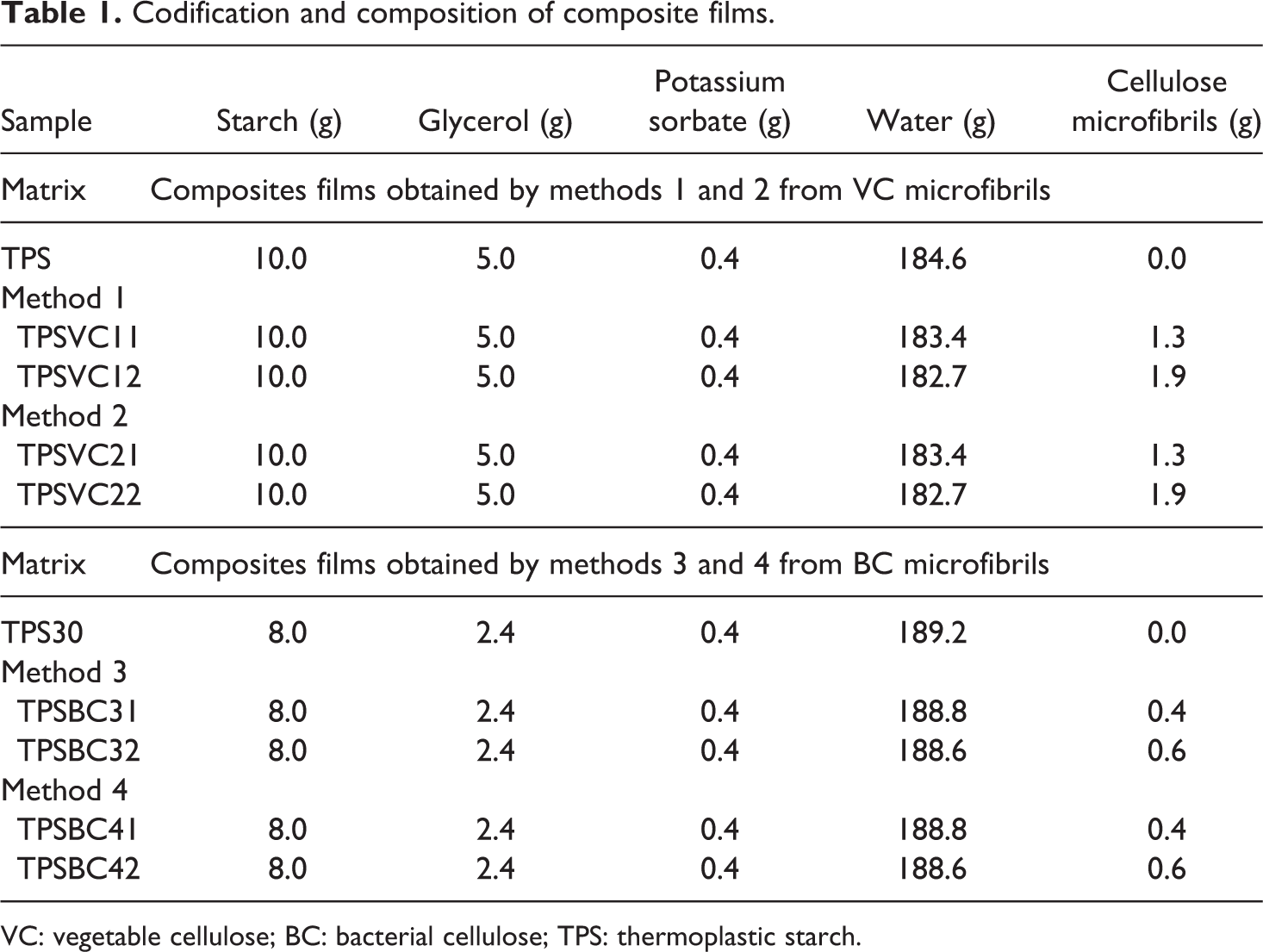
VC: vegetable cellulose; BC: bacterial cellulose; TPS: thermoplastic starch.
Mechanical properties
The tensile testing was performed using an Instron 5582 Universal Testing Instrument, equipped with a 50-N load cell, and all the samples were tested in accordance with the ASTM D882-09 standard, with a cross-head speed of 25 mm min–1, with 5 mm × 19 mm rectangular strips. The mechanical tensile data were gathered for nine different specimens and then averaged.
ATR-FTIR spectroscopy
Infrared spectroscopy experiments were performed using an FTIR Nicolet 6700 series spectrometer equipped with a single-reflection ATR and type IIA diamond crystal tungsten carbide. The diamond ATR had an approximate sampling area of 0.5 mm2 and applied a consistent, reproducible pressure applied to every sample. The infrared spectra were collected with a 4-cm–1 resolution and a total of 64 scans were performed.
Thermal properties
Thermogravimetric analyses were performed using a Mettler Toledo TGA/SDTA 851e instrument in a nitrogen atmosphere at 40 ml min–1 and a heating rate of 10°C min–1. The samples were heated from 30°C to 600°C.
Results and discussion
Mechanical properties
The tensile properties of the TPS and the composite films reinforced with different VC microfibrils content are presented in Table 2. It was possible to observe that with 1.9 g of VC, the tensile strength increased to 7.4 MPa and the Young’s modulus increased to 100.9 MPa. However, using the processing method 2, the tensile strength increased to 10.2 MPa and Young’s modulus increased to 145.5 MPa. Comparing the TPS and the composite films obtained from the methods 1 and 2, the tensile strength increased 4.9 and 6.8 times and the Young’s modulus 6.1 and 8.9 times with the methods 1 and 2, respectively. The difference in the resulting of the same content of VC is attributed to the moment in which the cellulose microfibrils are added during the processing of the material (before the gelatinization). Prior to gelatinizing, the viscosity of the starch solution is not so high; thus, the incorporation and homogenization of the VC are improved. In addition, the strong interaction by hydrogen bonds between the VC and the starch could improve the interface and affect the crystallization of the TPS. 2,31 The mechanical behavior of BC composites is also presented in Table 2; in this case, the BC was added before the gelatinization of the TPS30 with two different stirring times (methods 3 and 4). The values of mechanical properties of the composite films prepared by methods 3 and 4 did not show any significant change; however, the reinforcing capacity of BC was evidenced by a significant improvement of mechanical properties compared with TPS30 matrix. The BC content in the composite films varied from 0.0 g to 0.6 g (starch weight), and while using method 3, both the tensile strength and the Young’s modulus increased from 3.3 MPa to 9.2 MPa and 31.0 MPa to 409.6 MPa, and using method 4, they increased form from 3.3 MPa to 9.4 MPa and from 31.0 MPa to 412.9 MPa, respectively. These results confirm that the BC content was responsible for the increased mechanical properties of the composite films.
Mechanical properties of matrices and cellulose microfibril composites.
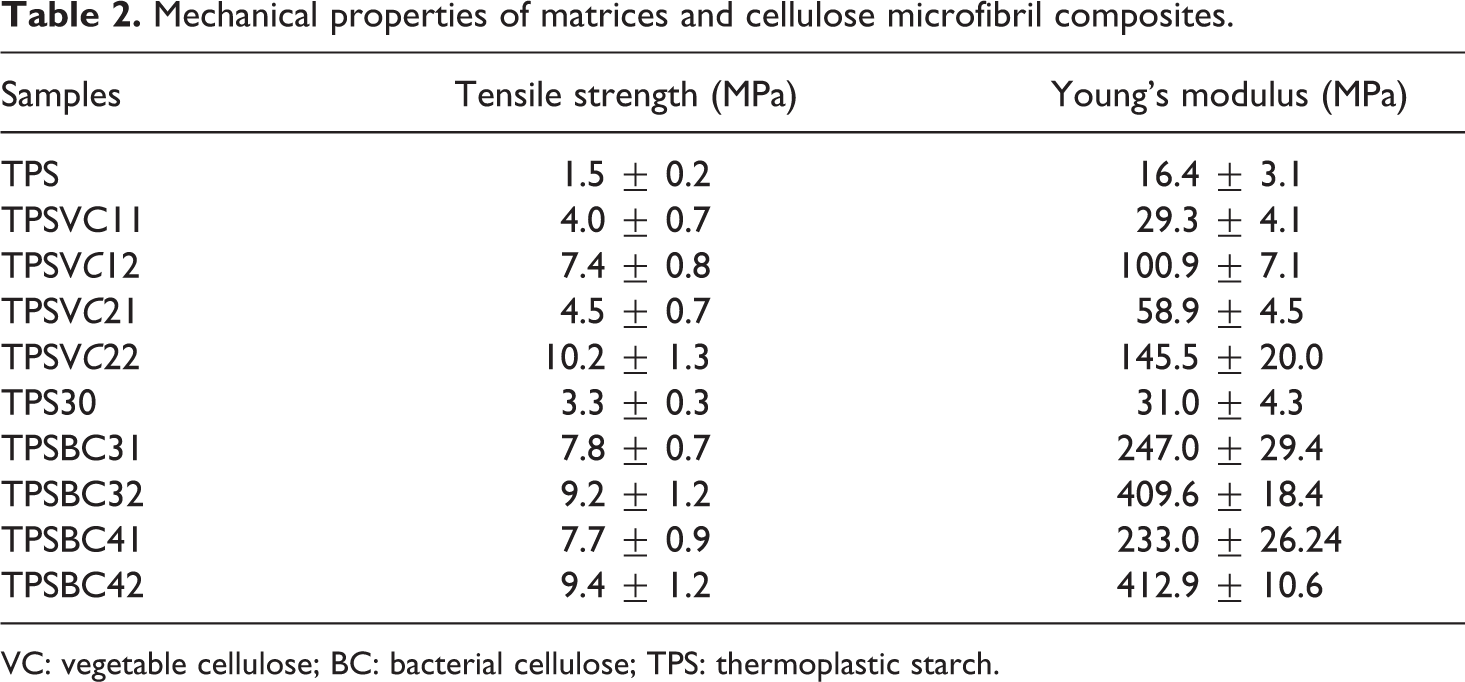
VC: vegetable cellulose; BC: bacterial cellulose; TPS: thermoplastic starch.
On the other hand, the results obtained after mechanical testing showed that the variations in the material formulation and processing methods increased the tensile strength and Young’s modulus, as seen for the TPS and TPS30 matrices. This is because the TPS formulation contained a higher amount of plasticizer (glycerol) compared with TPS30. 2,32,33 Finally, the results are encouraging because they are comparable with the mechanical properties of the low-density polyethylene 34 and reflect their potential to be used in food packaging with the outcome of reduced environmental impact.
ATR-FTIR spectroscopy
FTIR spectroscopy was used to verify the distinct chemical interactions of the matrices and the composite films developed. Figures 2 and 3 show the fingerprint region for the range 1400–800 cm–1 of the composite materials reinforced with VC and BC, respectively. To clearly determine the peaks, the second derivatives of the acquired spectra were calculated.
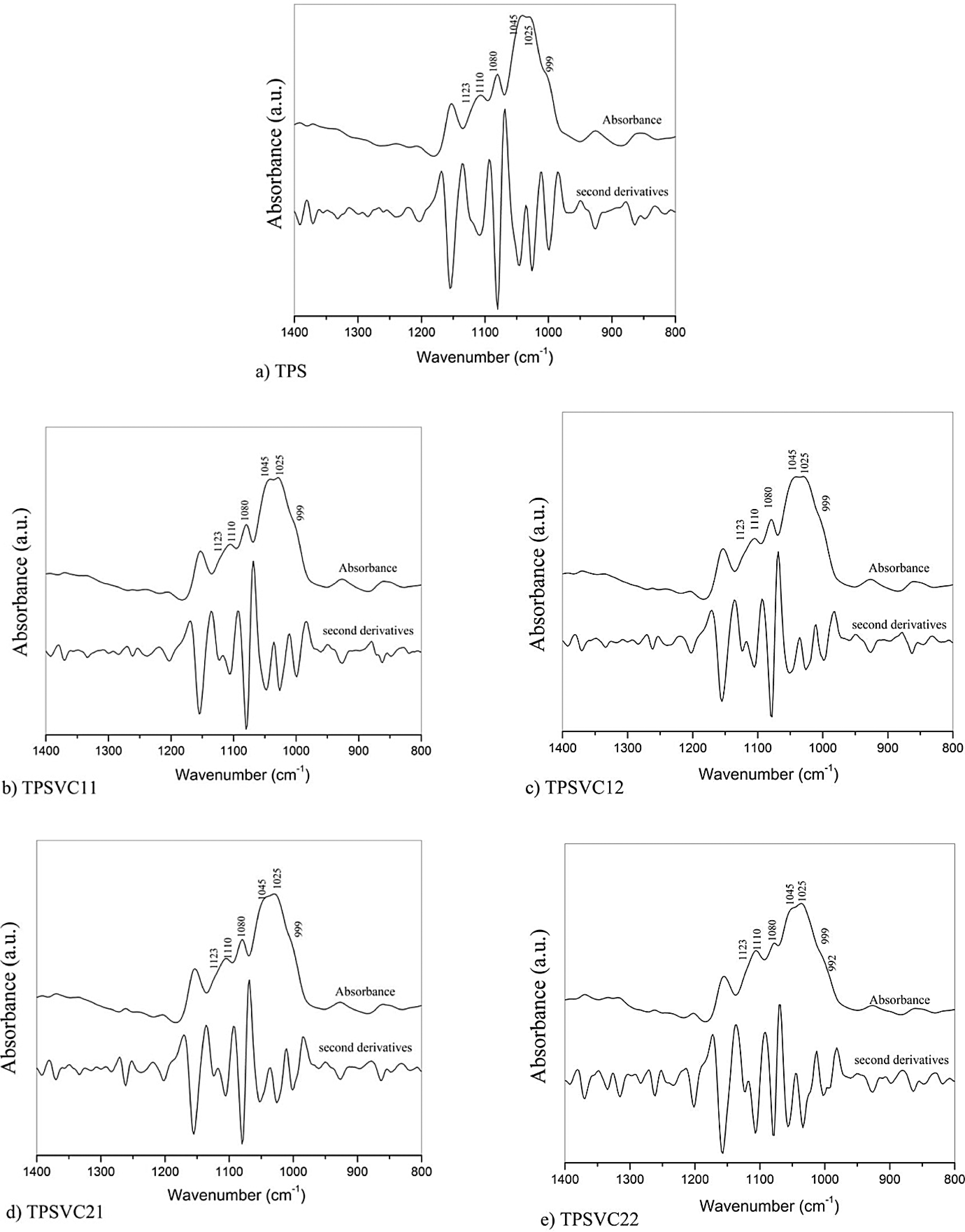
FTIR spectra of TPS and composites films reinforced with VC: (a) TPS, (b) TPSVC11, (c) TPSVC12, (d) TPSVC21, and (e) TPSVC22. VC: vegetable cellulose; TPS: thermoplastic starch; FTIR: Fourier transform infrared.
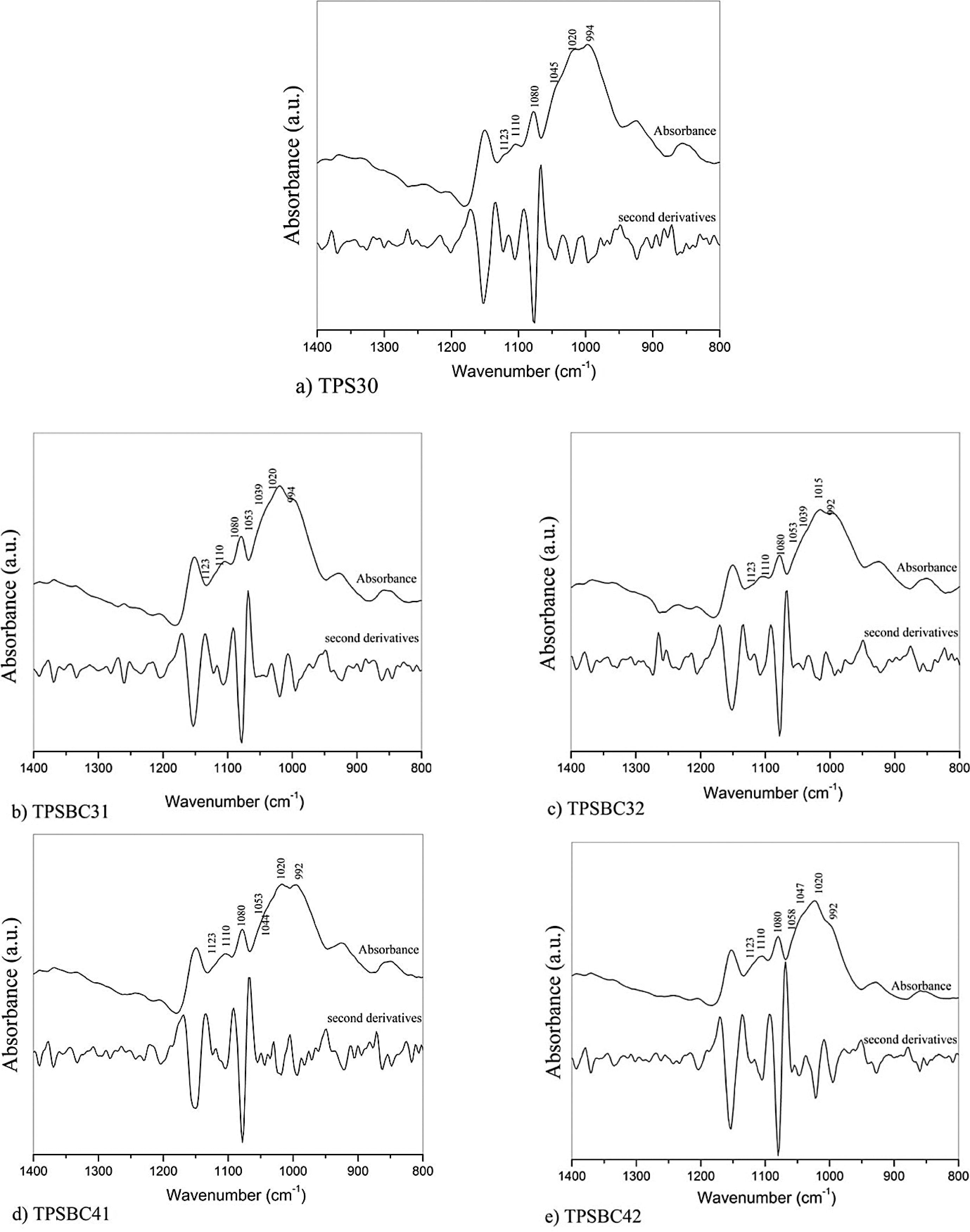
FTIR spectra of TPS30 and composite films reinforced with BC: (a) TPS30, (b) TPSBC31, (c) TPSBC32, (d) TPSBC41, and (e) TPSBC42. BC: bacterial cellulose; TPS: thermoplastic starch; FTIR: Fourier transform infrared.
Figure 2 shows that a slight change occurred at the peaks. The band at 1123 cm–1 was asymmetric for phase ring stretching, as well as for C–C and C–O stretching. 35 –37 This intensity showed a variation associated with the addition of VC to all of the composite films. The band at 1110 cm–1 was caused by stretching the C–O bond, which was mainly attributed to the association of primary alcohols in cellulose 38 , this also indicate an intimate connection by hydrogen-bonding in the VC/TPS system. Peaks were identified at 1081 cm–1 and 1023 cm–1 and were the characteristic of the stretching of C–O bonds in the anhydrous-glucose ring of the starch. 39 –42 Changes in these bands were attributed to the amount of short range ordering of the composite films when adding each component and the distinct chemical interaction (hydrogen bonding) between the components. 43 –45 Furthermore, the broad band at 1045 cm–1 was related to the starch crystallinity, and the band that occurred at 1020 cm–1 was the characteristic of amorphous starch, which for TPS and the composite films displayed a change in the intensity related to the added VC. 43 The bands at 999 cm–1 and 992 cm–1 revealed the presence of several conformations of these primary alcohols. 36 However, only the 992 cm–1 band appeared to comply with this conjecture for the TPSVC22.
Figure 3 illustrates four peaks at 1053 cm–1, 1045 cm–1, 1020 cm–1, and 994 cm–1. The bands at 1045 cm–1 and 1020 cm–1 are sensitive to the changes in starch crystallinity. 43 The band at 1053 cm–1 shows a minimum that has been interpreted corresponding to the weakening or breaking of the hydrogen bonds, which is caused by adding BC. 36 The band at 1045 cm–1 reflects C–C stretching, which is sensitive to the amount of ordered or crystalline starch. The band at 994 cm–1 can be related to the intermolecular hydrogen bonding of the hydroxyl group at C6, because the starch is water sensitive. 36 However, the composite films show a change in wavelength resulting from interactions with the cellulose microfibrils because of its affinity with starch. This shows that even though the cellulose microfibrils improved the mechanical properties of the composite, it changed the chemical interaction of the matrix that was produced. Moreover, the presence of the C–O–C pyranose ring induces a vibration in the skeletal region between 1076 cm–1 and 1023 cm–1. 31,43
Thermal properties
TGA of the composite films was carried out to assess their thermal stability and degradation. Figure 4 shows the results of the TGA analysis for the composite materials reinforced with VC microfibrils. The mass loss from 40°C to 140°C (onset temperature for thermal decomposition) was related to the evaporation of both water and glycerol. 2 However, the mass loss was different at the onset temperature for TPS, TPSVC12, and TPSVC22 (mass loss of 12.0 wt%, 9.0 wt%, and 6.0 wt%, respectively), as shown in Figure 4. This difference in the percentage of mass loss is associated with the thermal stability of the cellulose. However, changes in the mass loss for same concentration of VC microfibrils are attributed to stronger hydrogen bonds that form when VC are added before to the gelatinization of TPS. This facilitated the interaction between them.
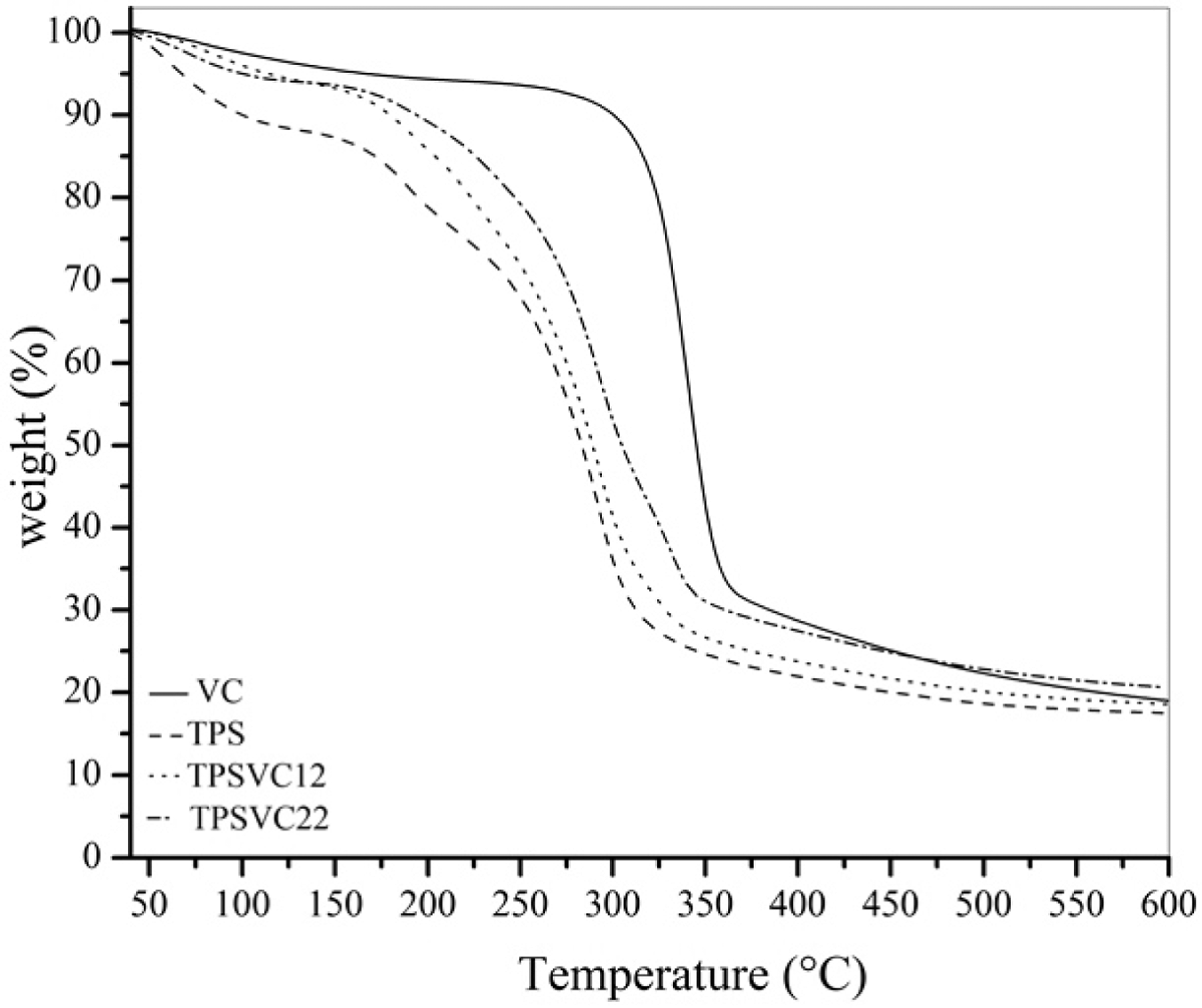
TGA curves for TPS, TPSVC12 and TPSVC22. VC: vegetable cellulose; TPS: thermoplastic starch; TGA: thermogravimetric analysis.
Cellulose microfibrils shift the maximum degradation temperature of the matrix to higher temperature. This behavior is related with the VC content and its reinforcing effect in the final material.
Table 3 presents the results of degradation temperature to 25 wt% (T 25 wt%) and 75 wt% (T 75 wt%) of VC, TPS, TPSVC12, and TPSVC22. The main difference between the processing methods 1 and 2 was the need to apply more energy to degrade 25 wt% and 75 wt% of material obtained through method 2 (TPSVC22). Hence, thermal stability of the TPSVC22 composite mainly relies on the stronger hydrogen bonds formed during the gelatinization of material.
Thermal properties of TPS, TPS30, VC, BC, and cellulose microfibrils composites.
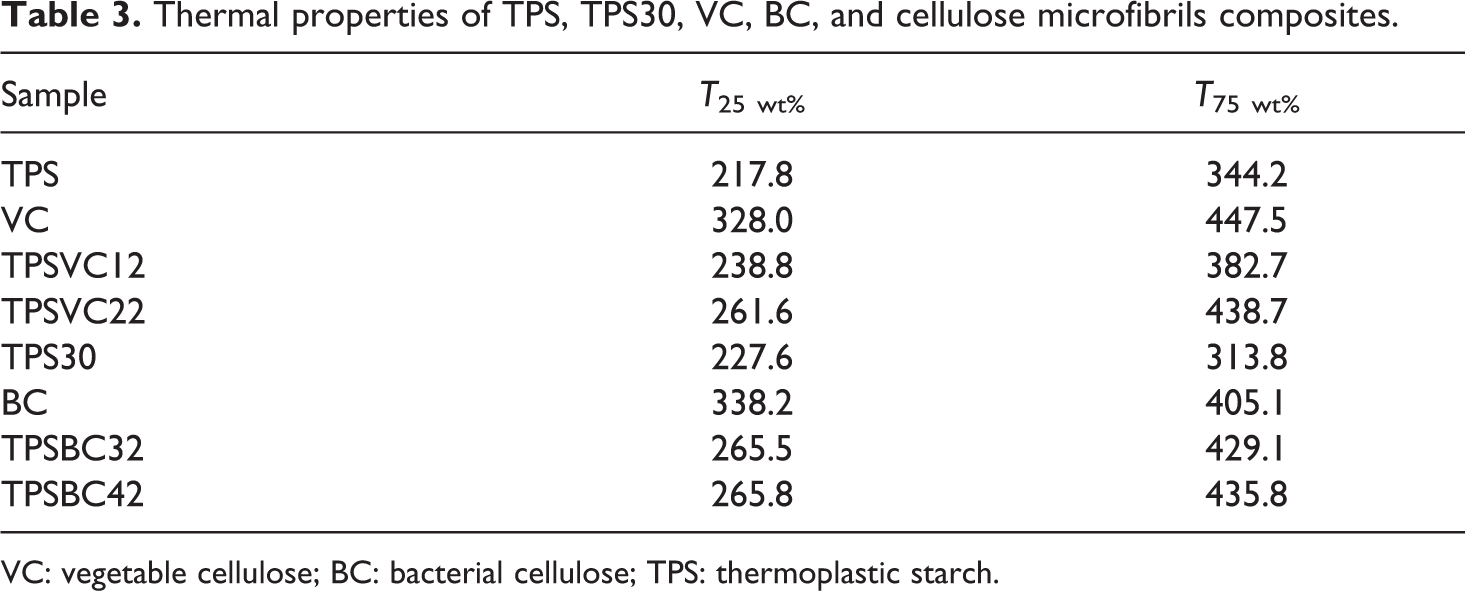
VC: vegetable cellulose; BC: bacterial cellulose; TPS: thermoplastic starch.
Figure 5 represents TGA curves for composite films reinforced with BC. Differences in the onset mass loss between TPSBC32 and TPSBC42 composites were not observed. In addition, Table 3 presents the results of degradation temperature of BC, TPS30, TPSBC32, and TPSBC42, at 25 wt% (T 25 wt%) and 75 wt% (T 75 wt%) of material degradation. The difference in the energy applied to degrade 25 wt% and 75 wt% between TPSBC32 and TPSBC42 was not observed. However, changes were evident when the composites are compared with the TPS30, due to increased that generates BC in the thermal stability of composite film.
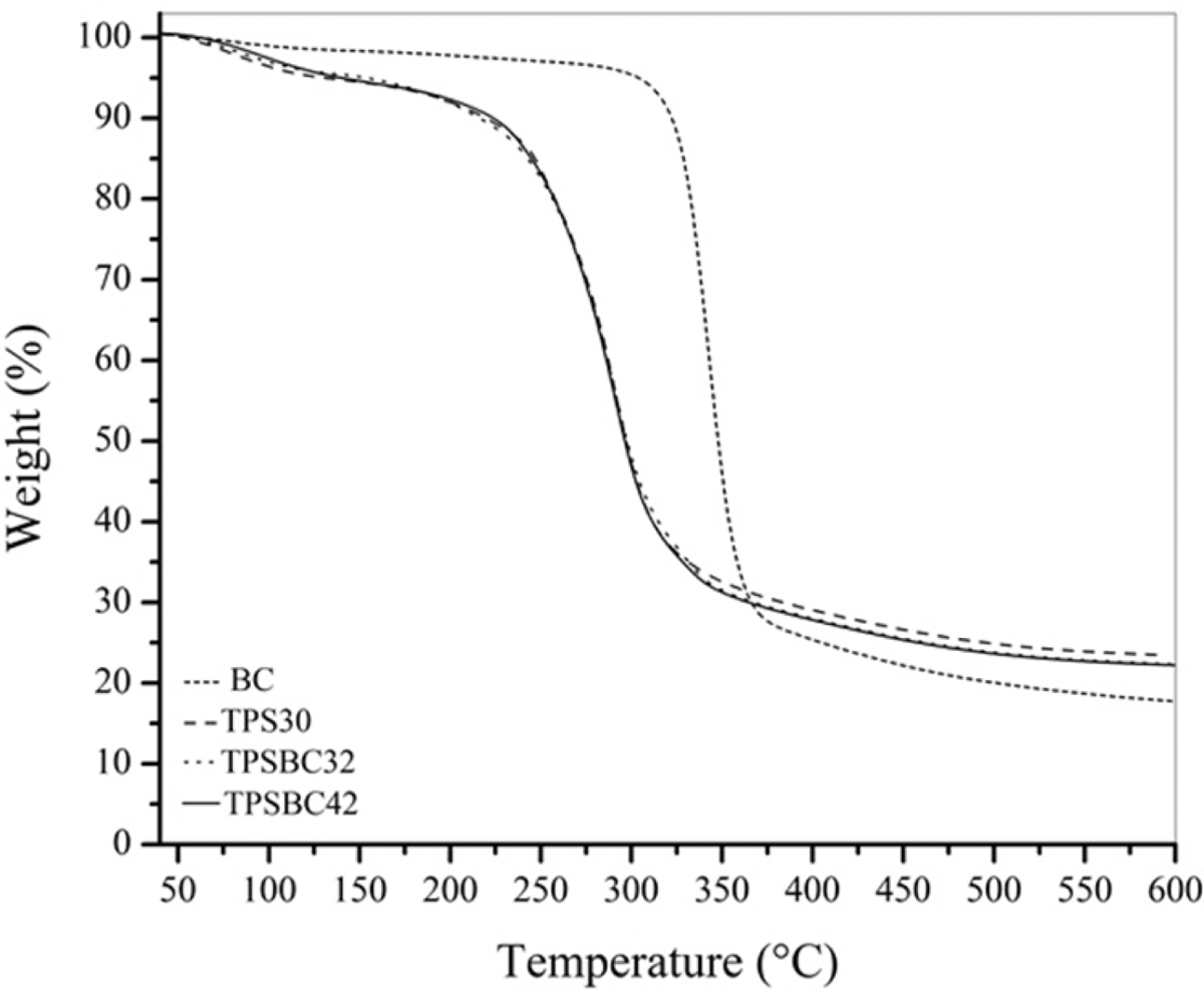
TGA curves for TPS30, TPSBC32 and TPSBC42. BC: bacterial cellulose; TPS: thermoplastic starch; TGA: thermogravimetric analysis.
Conclusions
Different processing methods were applied to obtain potato starch/cellulose microfibrils composite films. Overall, the mechanical strength of all the composite films was dependent on the processing method. When the cellulose microfibrils are added before gelatinization, a better homogenization of the reinforcement in the matrix is achieved, hence there is a greater miscibility between components. Furthermore, there are interactions between the elements through the formation of hydrogen bonds, generating an interface with excellent characteristics. The processing method and the presence of the cellulose microfibrils in the composite affect the mechanical behavior of materials. The miscibility generated between the reinforcing and matrix when the microfibrils are added before gelatinization could also be evidenced by the results of the TGA. More energy is required to degrade composite materials than their respective matrices, which agrees with the results of mechanical tests, there is a good homogenization of the components and excellent interaction between the reinforcement and the matrix by the hydrogen bonds. These materials are promising candidates for food packaging and edible film applications.
Footnotes
Acknowledgements
The authors thank Santiago Betancourt and Adriana Restrepo for their valuable contributions to the TGA and tensile strength experiments.
Funding
This work was supported by the Departamento Administrativo de Ciencia, Tecnología e Innovación (Colciencias) and Centro de Investigación para el Desarrollo y la Innovación (CIDI) for the financial support.
