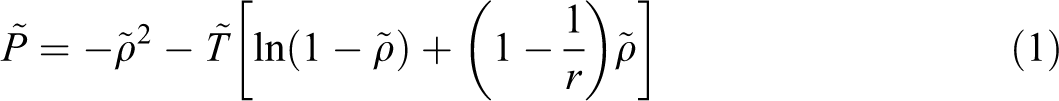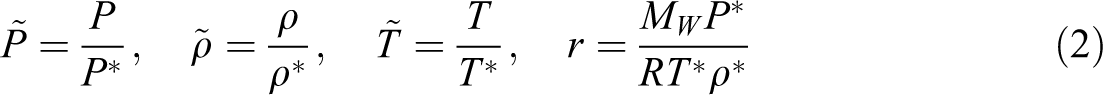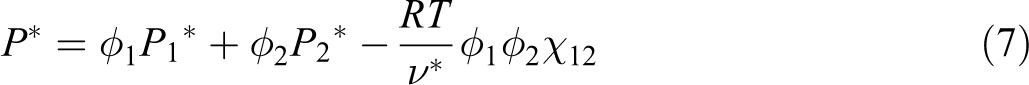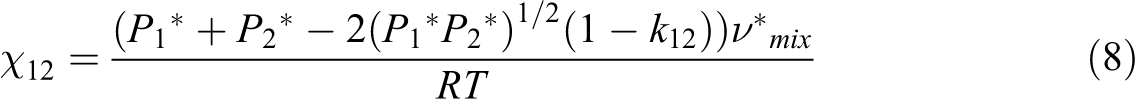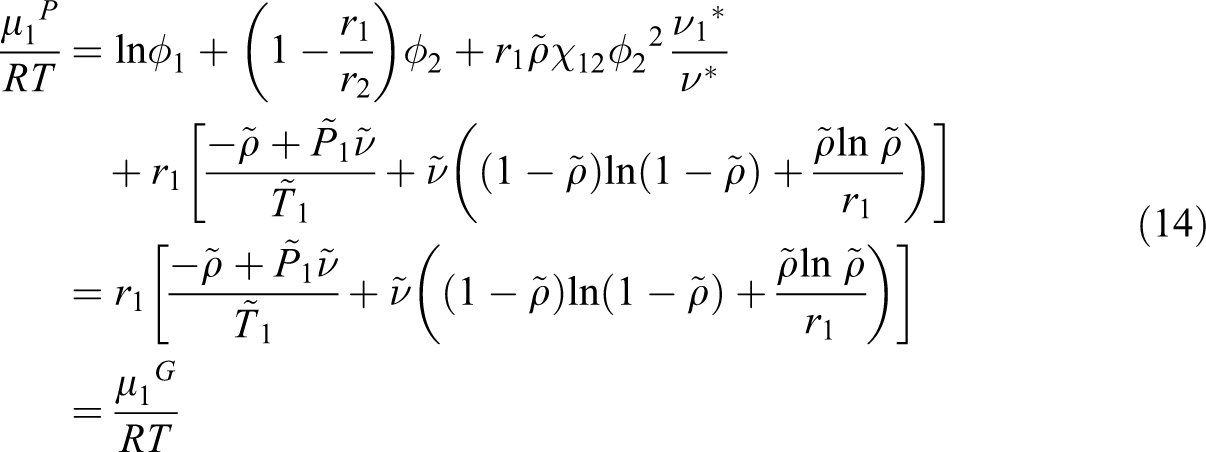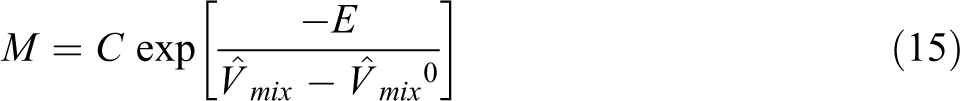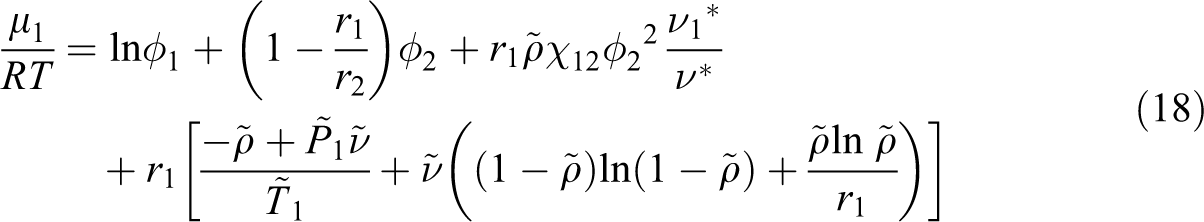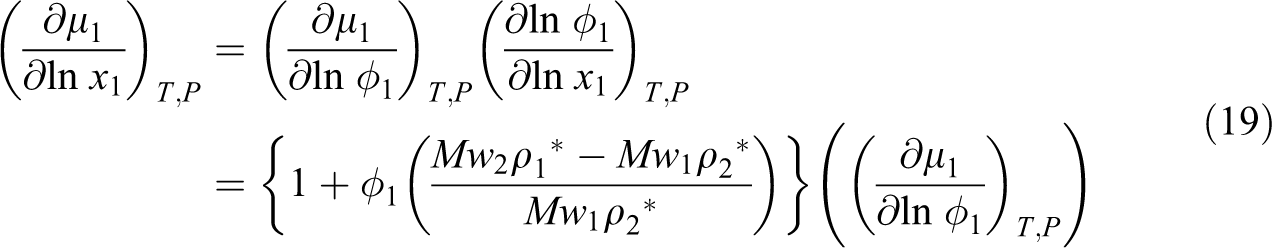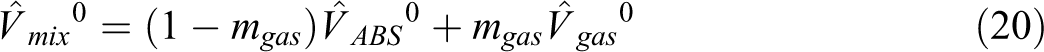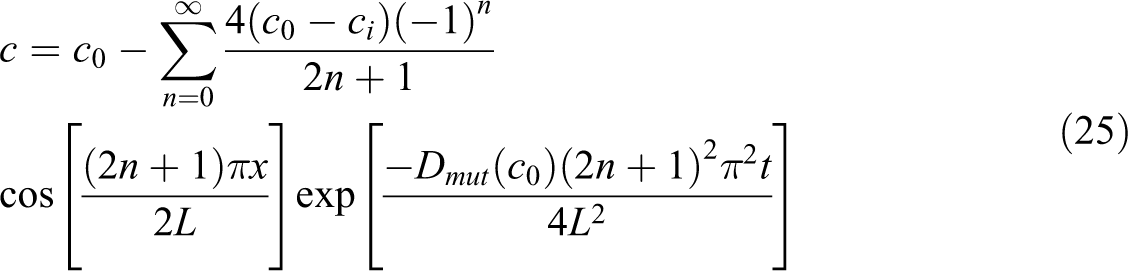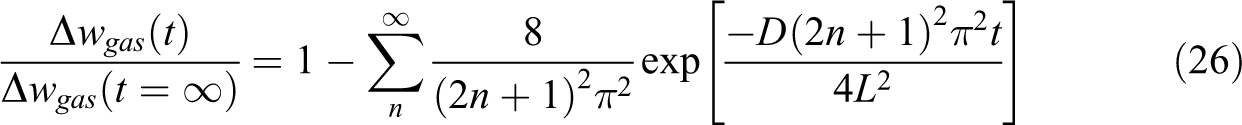Abstract
In this work, we used three gases (CO2, N2 and normal hexane) for diffusivity measurements in Acrylonitrile butadiene styrene (ABS). We proposed a diffusion model that the diffusion coefficients of each gas in ABS could be estimated from the specific volume of ABS/gas mixture and chemical potential of gas in ABS. The solubility and diffusivity of three gases into ABS were determined by a magnetic suspension balance. The results showed that the solubility and diffusivity of three gases increased with increasing of pressure. Also it was determined that N2 has a lowest solubility and the highest diffusivity in ABS in all temperature and pressure ranges. It was shown that there was a suitable overlapping between the experimental and predicted values from the proposed model, in which the proposed model could successfully estimate the diffusion coefficient of mentioned gases in ABS in all temperature and pressure ranges.
Keywords
Introduction
Recently, supercritical carbon dioxide (sCO2) and supercritical nitrogen (sN2), due to their economical availability and not having any environmental impact, are being commonly used as blowing agents.1–4 Carbon dioxide (CO2) is a preferable choice for solubility and diffusivity applications,5,6 due to its good properties like; environmentally safe and inexpensive.7–9 Solubility and diffusivity of CO2 in molten polymers were affected by temperature and pressure.10–13 Some models have been proposed to determine the diffusion coefficients of gases in polymers that the free volume is accepted theory for developing a prediction model of diffusion coefficients.14–17 Among different methods for measuring the solubility and diffusivity, the Magnetic Suspension Balance method is popular and was used in different works.18–20 For instance, Li investigated the solubility and diffusivity of CO2 and N2 in thermoplastic polyurethane in pressures up to 19.3 MPa. CO2 solubility was one magnitude higher than that of N2, and their solubility had an inverse dependence on temperature. 21 Solubility and diffusivity of supercritical CO2 in poly(l-lactide)-hydroxyapatite (PLLA–HA) and poly(d, l-lactide-co-glycolide)-hydroxyapatite (PLGA–HA) composite materials were measured and the results suggest that dense CO2 could be used as a “green” processing agent for composite biomaterials. 22 Also in another study the diffusion coefficients of carbon dioxide (CO2) in poly (ethylene-co-acrylic acid) over temperatures of 373–473 K and pressures of up to 20 MPa were determined. 23 Solubility and diffusivity of CO2 in ionic polyimides were investigated and it was found that solubility is weakly related to the theoretical surface area, and the diffusivity is correlated to the fractional free volume. 24 As well, different parameters like; the diffusivity, permeability and solubility of different gases in poly(tetrafluoroethylene) were measured and reported. 25 In our recent work the solubility of CO2 in Polystyrene (PS) at different temperatures and pressures using a magnetic suspension balance (MSB) was determined and it was found that the bubble growth is a diffusion controlled phenomenon. 26 Also we investigated the foaming process of PS/PMMA blends and copolymers by our designed bath system and the cellular structures of produced polymer foams were fully illustrated.27–30 Most of the models developed so far have been expressed in a form of an exponential function of either the free volume or the free volume fraction. In order to apply the models to estimating the diffusion of gas in a polymer, one must estimate the specific free volume of the polymer/gas mixture and to determine model parameters, such as the pre-exponential coefficient, an activated energy, etc. by performing diffusion experiments for each pair of polymer and gas.
The MSB (Rubotherm and Bell Japan) consists of a measuring chamber and a balance (Mettler AT261, Switzerland) in which the balance is located outside the chamber under atmospheric conditions. 26 In the measuring chamber, the sample was hooked up to a so-called suspension magnet, which consists of a permanent magnet, a position sensor and a device for coupling/decoupling the measurement load (sample). A high pressure and temperature condition was realized in this measuring chamber. In this system, an electromagnet is attached to the under-floor weighing hook of the balance and situated outside the chamber so as to have the suspension magnet in a freely suspended state controlled by an electronic control unit. Using this magnetic suspension, the weight of the sample in the chamber could be transmitted to the balance without direct contact. The MSB used in this study can measure the mass of two samples with only one suspension magnet by using the device for coupling/decoupling measuring load. By changing the position of the suspension magnet, three different measuring modes can be realized; zero position, the first sample measuring (is related to gas sorption measurement) and the second sample measuring (is related to fluid density measurement). 26
In this study, three gases as, CO2, N2 and normal hexane, are used for investigating their diffusivity in ABS. At the first the solubility and diffusivity of each gas in ABS were determined at temperatures of 382 K and 455 K and pressures up to 12 MPa by using the magnetic suspension balance (MSB). For diffusivity prediction, we proposed a diffusion model that the diffusion coefficients of each gas in ABS could be estimated from the specific volume of ABS/gas mixture and chemical potential of gas in ABS.
Experimental
Material
The Acrylonitrile butadiene styrene, (ABS) was supplied from Tabriz petrochemical company (SD-0150). The weight and number average molecular weight of used ABS were 196000 and 89000, respectively. The density of used ABS is 1.070 g·cm−3. The glass transition temperature was about 105°C obtained by the differential scanning calorimetry (DSC). Super critical CO2, N2 and high pressure of n-hexane were used as gases. The solubility and diffusivity of gases in ABS were determined using the MSB system.
Experiments
The experimental solubility and diffusivity data for different gases were calculated by the MSB system under desired temperature and pressure range.
31
The PVT data were obtained by a high-pressure apparatus (GNOMIX, Inc., USA) with a temperature from 320 to 500 K and pressure from 0.2 to 50 MPa.
31
Al the experiments were repeated three times. For determining the specific volume of ABS with CO2, N2 an n-hexane,
where,
The superscript * indicates the property of binary mixture, the subscript 1 is related to each gas and 2 for ABS. v1* and v2* represent the characteristic molar volumes of gas and ABS, respectively, k12 is a binary interaction parameter between ABS and each gas, and m1 and m2 are weight fractions of gas, x1 and x2 are mole fractions. 31 Finally, we used the equation of phase equilibrium, which equates the chemical potentials of gas in the two phases:
Considering the SL equation of state and above equation, the following equation of chemical potential for each gas is written:
For diffusivity prediction of each gas into ABS, the model of Maeda and Paul with the Fujita model was combined and we developed a proposed model. In this model it is expected that the absolute specific free volume of the mixture determines the mobility of both ABS and gas (CO2, N2 and n-hexane), and it is shown by
where M is the mobility of gas relative to the ABS,
The relationship between Dself and diffusion coefficient D, was obtained from 31
where x2 is mole fraction of polymer and
Then, term
For estimating the diffusion coefficients of gas, the values of
where
Fick’s second diffusion law was given for estimating the diffusion coefficient from the weight data by the following equation:
In which the analytical solution of the above equation with boundary conditions was given by
where L is the thickness of the sample. ci and co, respectively, are the concentration of gas at the initial state and that at the surface of the sample equilibrium with the CO2 gas pressure during the measurement. The solution of the equation could be written by
where Δwgas(t) is the weight difference of dissolved gas in ABS at time t. The left hand of above equation is the relative amount of dissolved gases in ABS. We calculated the diffusion coefficient by fitting the Equation (26) to the experimental data versus the time.
Results and discussion
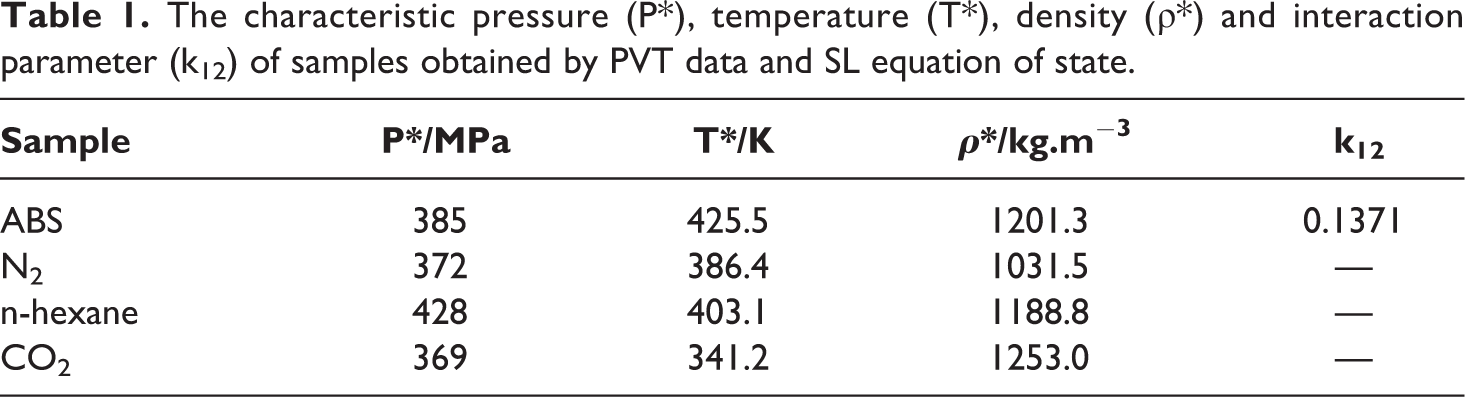
Using the SL equation of state, all the characteristic properties of samples, in the pressure range of 0–40 MPa, are listed in Table 1. The solubility of different gases (CO2, N2, and n-hexane) in ABS at different pressures and temperatures is shown in Figure 1.
The characteristic pressure (P*), temperature (T*), density (ρ*) and interaction parameter (k12) of samples obtained by PVT data and SL equation of state.
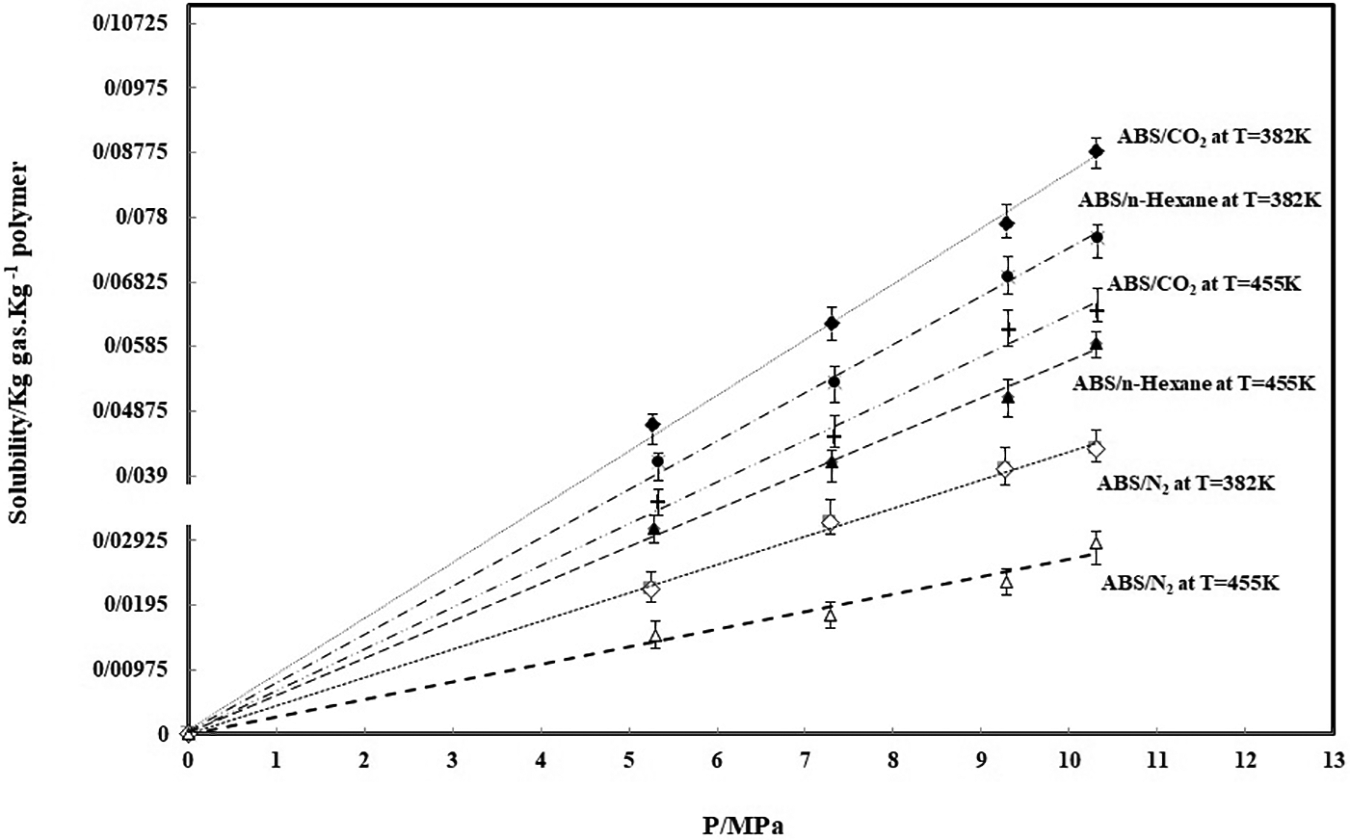
The solubility of three gases in ABS at T = 382 K and 455 K (The solid lines represent estimates by SL equation of state).
As a whole, the solubility of different blowing agents in ABS calculated by the MSB, increases with increasing of selected pressure. It is clear that with temperature increment, the solubility of three gases in ABS is decreased. The solubility data for ABS/N2 is the lowest at two temperatures and three pressures. These data for ABS/CO2 and ABS/n-hexane are comparable in all pressure ranges. The solid lines are the prediction of the SL equation.
The absolute specific free volume, for ABS/CO2, ABS/N2 and ABS/n-hexane mixtures was calculated using SL equation of state and Equation (20), at two temperatures, and the results are plotted in Figure 2. The absolute specific free volume, increases with increasing of gas weight fraction in ABS. This behavior will affect the diffusivity results.
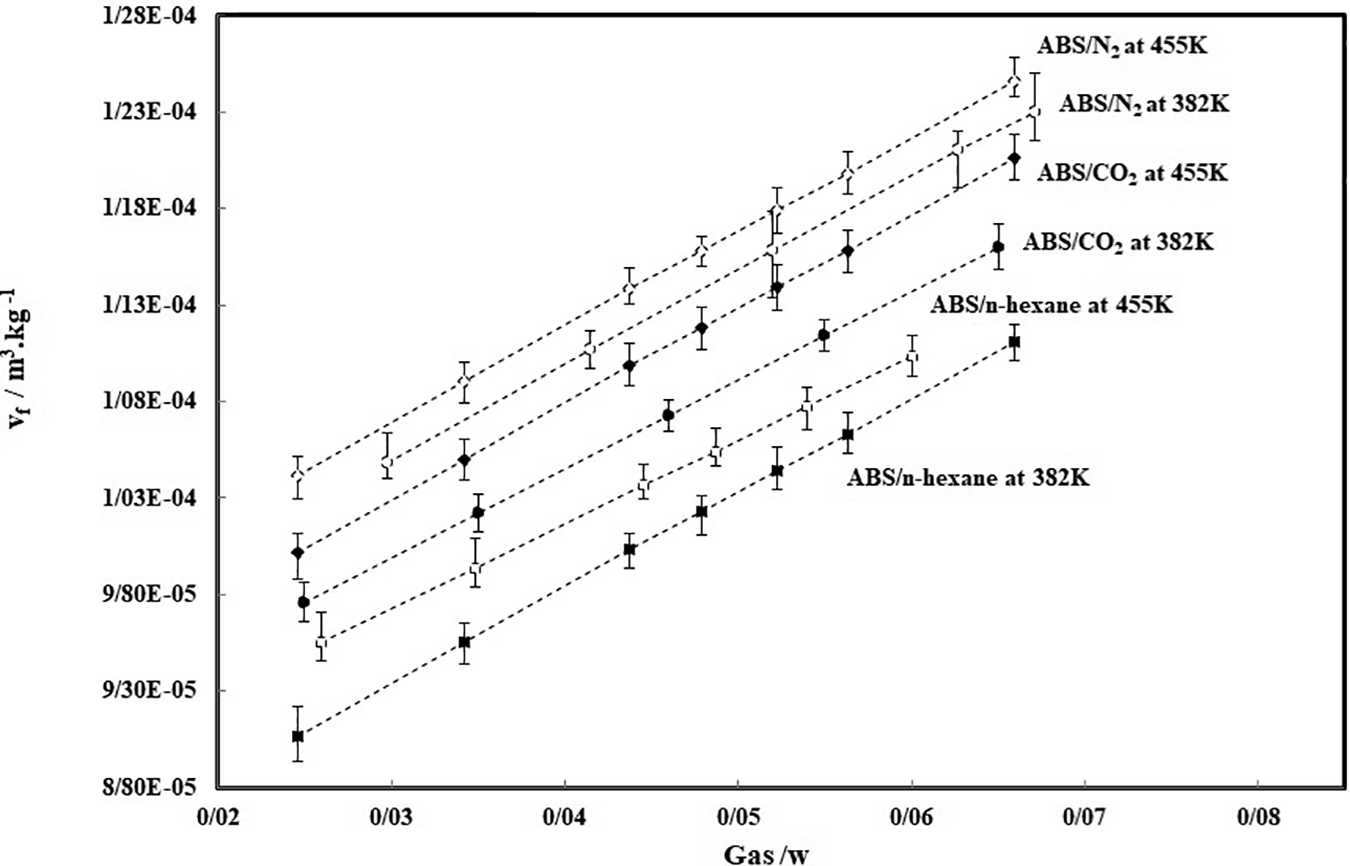
Calculated absolute specific free volume (vf) of ABS/gas.
The relationship between dissolved gas (CO2, N2 and n-hexane) in ABS with sorption time was determined is shown for ABS/N2 in Figure 3. The solid line represents the calculation results by Equation (26) and the symbols are the experimental data.
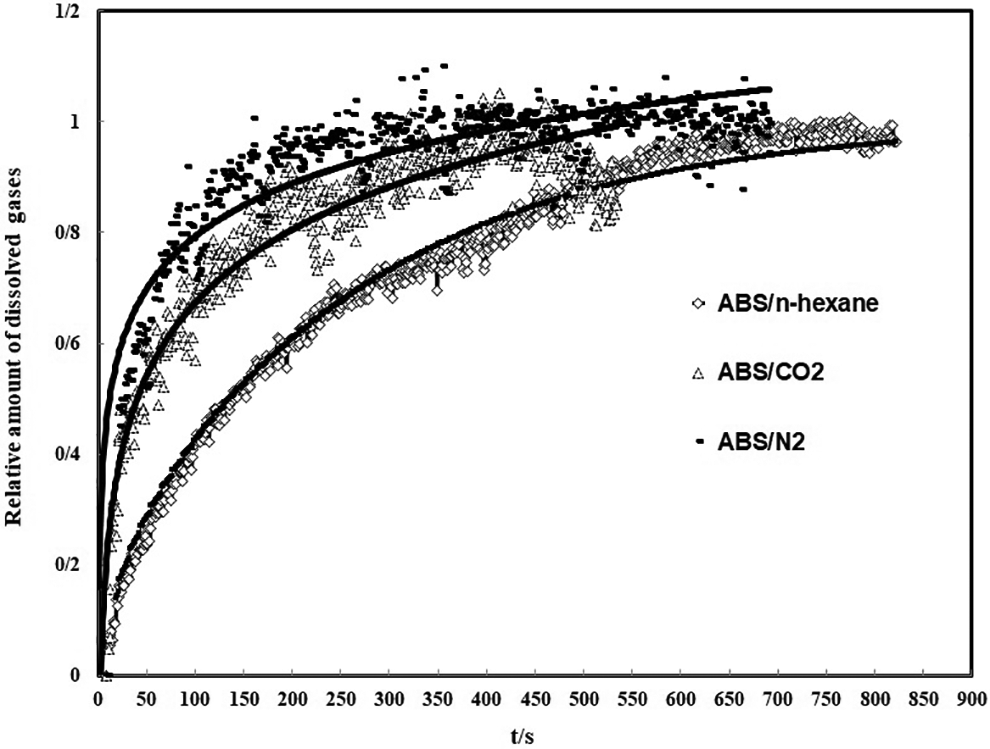
Relative amount of dissolved three gases versus sorption time for ABS, by the step change in pressure from 9.3 to 10.3 MPa at 382 K. The solid lines are predicted by Equation (26).
To predict the diffusion coefficient of three gases in ABS at a given T and P, using Equations (16) and (17), the parameters C and E in Equation (17) have to be determined. At the first, the chemical potential of CO2, n-hexane, N2, and their derivative with the volume fraction were calculated for each polymeric sample by using the SL equation of state and Equation (18). The derivative of the chemical potential relative to ln ϕ was obtained. Then, the self-diffusion coefficient of gas for ABS was calculated by Equation (17) from the measured diffusion coefficient, the obtained chemical potential derivative, polymer weight fraction, x2, and temperature, T. The absolute specific free volumes of samples were also calculated. The resulting self-diffusion coefficients for ABS are plotted against the inverse of Vf as shown in Figure 4.
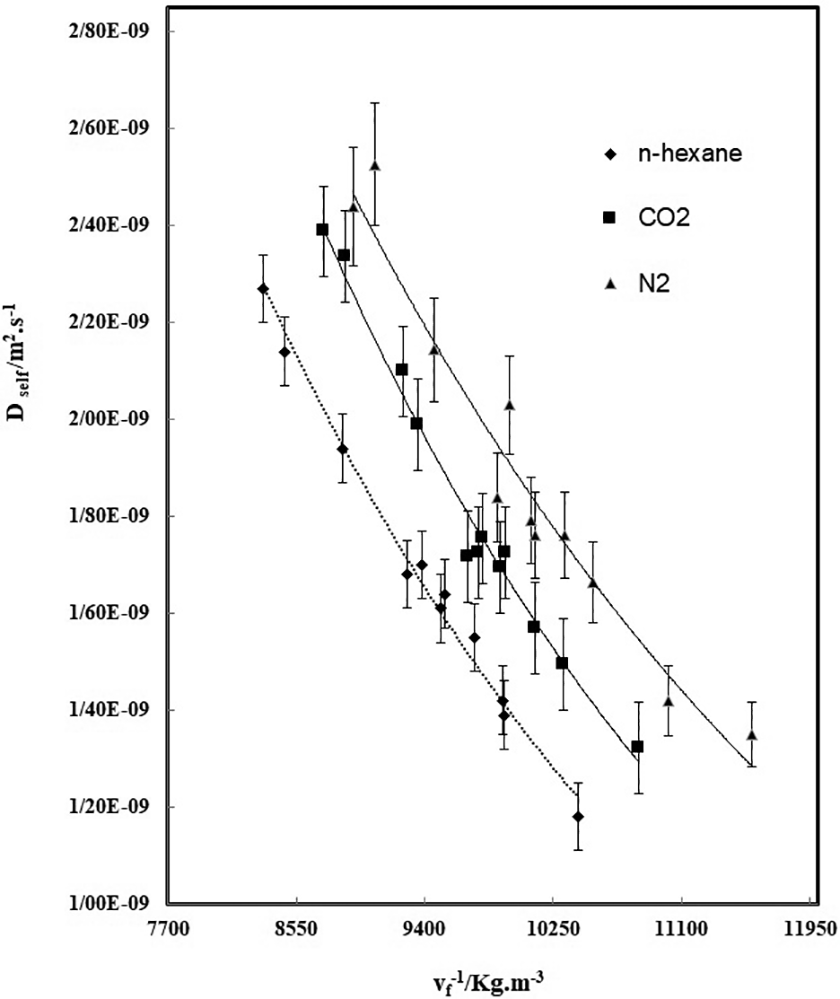
Self-diffusion coefficients of n-hexane, CO2 and N2 (Dself) against the inverse of absolute specific free volume of the mixtures at 382 K and pressures in the range from 5 to 12 MPa (the solid line represents estimates given by the proposed model).
The parameters C and E of Equation (16) can be determined by fitting of this equation to the plots of the diffusion coefficients of each gas in ABS obtained at 382 K and in the pressure range from 5 to 12 MPa, in which the solid line represents the fitting curve of Equation (16). Thus, whenever the absolute specific free volume of polymeric samples/gas mixture and the derivative of chemical potential of gas relative to the gas volume fraction in mixture were obtained, by using Equations (16) and (17) with the obtained C and E parameter values, the diffusion coefficient of gas under different temperatures and pressures could be estimated in a certain accuracy. The prediction results of diffusivity for samples are illustrated in Figure 5. The symbols are the experimental values and the solid lines represent the diffusion coefficient estimated by Equations (16) and (17) with the obtained parameters C and E. As it is clear, there is a good overlapping between the experimental and estimated values, in which the proposed model could successfully estimate the diffusion coefficient of gases in ABS at different temperatures and pressures. It is clear that the diffusion coefficients are increased with the temperature increment and the diffusivity of n-hexane in ABS is the lowest value. The diffusivity data for ABS/N2 is the highest that it is due to lower molecular weight of N2 compare to other two gases. As it is clear, there is a suitable overlapping between the experimental and predicted values, in which the proposed model could successfully estimate the diffusion coefficient of all gases in ABS at different temperatures and pressures ranges.
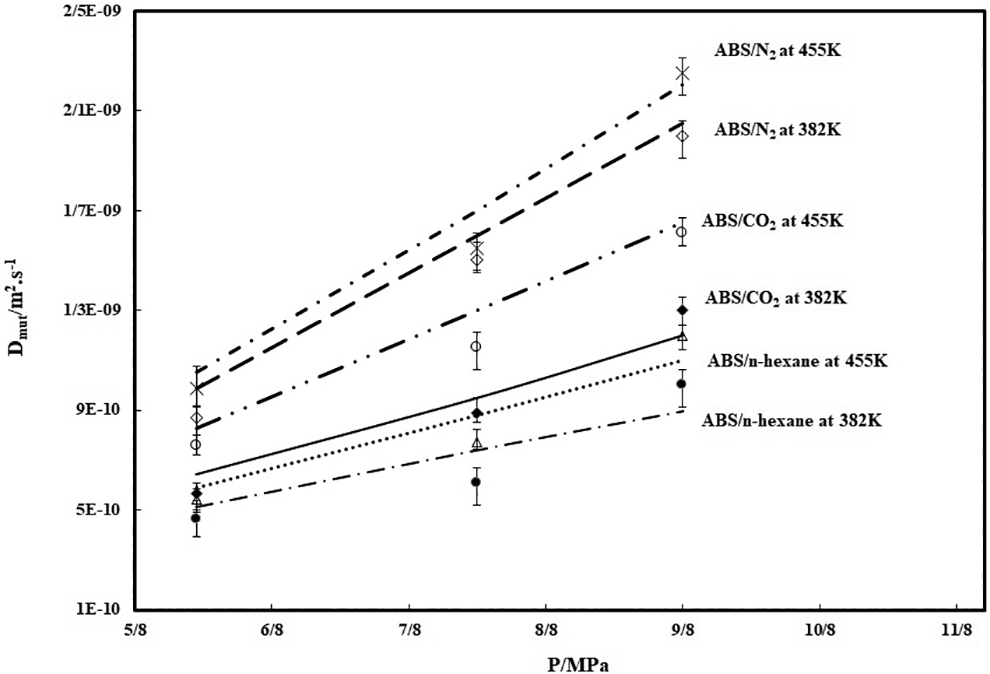
Diffusion coefficient of each gas in samples, at 393 K and at 423 K.
Conclusion
In this study, the solubility and the diffusivity of three gases (CO2, N2 and n-hexane) in ABS were experimentally measured at temperatures of 382 K and 455 K and pressures up to 12 MPa using the MSB. The solubility of all gases in ABS increased with pressure increment and decreased with temperature increasing. The relationship between the diffusion coefficient and the absolute specific free volume of mixtures was investigated to develop a diffusivity model of the diffusion coefficient. Combining Fujita’s model with Maeda and Paul’s model, we used proposed model to estimate the diffusion coefficient of gases in ABS. Whenever the absolute specific free volume of the mixture and the derivative of chemical potential of gases were known, the proposed model could estimate the diffusivity of different gases in ABS.