Abstract
Three cyclotriphosphazene derivatives were used as flame retardants for ABS resin in this study. These compounds were synthesized by the reaction of hexachlorocyclotriphosphazene with 2,2'-dihydroxybiphenyl, 1,8-dihydroxynaphthalene and 9,10-dihydroxyphenanthrene respectively. The activities of these compounds as flame retardants for ABS were characterized by UL94 and LOI tests. The 9,10-dihydroxyphenanthrene derivatives showed the best flame-retardant activity. The effects of these compounds to the physical properties of ABS were also measured. The synergistic effect of antimony (V) oxide or novolac to these flame retardant compounds were discussed.
Introduction
Because a big fire may bring about damage to human life and property, intense efforts are required to prevent a fire. One of the main weaknesses of plastics is that the general polymers are flammable materials. It is difficult to alter a plastic that has flammable properties to a flame retardant material. Therefore, many researchers have studied the effects of changing plastics to flame-retarded materials. Flammable materials may not be used in the construction of public buildings by law.
ABS is a copolymer of acrylonitrile, butadiene, and styrene. Presently it is widely used, because the weakness of polystyrene is improved and reinforced well by acrylonitrile and butadiene. Because butadiene can give a rubbery character to a copolymer, the weak impact resistance which is polystyrene flaw is improved. In addition, the stiffness is improved by adding acrylonitrile. Therefore, ABS has been used in the manufacture of TVs, refrigerators, telephones, toys, and other equipment. It is known that ABS is one of the most difficult polymers to alter to a flame retardant state1–5.
There are many methods to convert a flammable polymeric material into a flame retardant state. One of the most economic and simple methods is to add a flame-retardant material to a polymeric material. Currently many chlorinated compounds serve as examples of flame retardant materials. However, many researchers have opted for non-chlorinated flame retardants because chlorinated compounds may produce toxic gases during a fire. Therefore the cyclotriphosphazene received the attention as a flame retardant material6–13.
In our previous study, we reported that three cyclotriphosphazene derivatives could be used as flame retardants for ABS 10 . We found that the best one was the 2,3-dihydroxynaphthalene derivative. In this study three more cyclotriphosphazene derivatives were synthesized to use as flame retardants for ABS. The flame retardant property was compared with the results from our previous study.
Materials and Method
Materials and instruments
Hexachlorocyclotriphosphazene (HCCP), 2,2'-dihydroxybiphenyl, 1,8-dihydroxynaphthalene, 9, 10-dihydroxyphenanthrene, tetrahydrofuran, and trimethylamine were purchased from Aldrich Chemical (St. Louis, MO, USA). Pure ABS was obtained from Cheil Industry (Seoul, Korea); its density and tensile strength were 1040 kg m−3 and 392 kg cm−2, respectively. Novolac was synthesized from phenol, oxalic acid and 37% formaldehyde using reported methods 14 .
Thermogravimetry (TGA) was performed using TGA 4000 Thermogravimetric Analyzer (Perkin Elmer). The physical properties of the samples were measured using a universal test machine SFM-50KN (United Testing Systems, Concord, Ontario, Canada). Limiting oxygen index (LOI) was determined using MKM JD-14 (MKM Machine Tool Co., Jeffersonville, IN, USA).
Synthesis of cyclophosphazene derivatives
BPCP (2,2'-dihydroxybiphenyl derivative of cyclotriphosphazene) 15 , NTCP (1,8-dihydroxynaphthalene derivative of cyclotriphosphazene) 16 , and PTCP (9,10-dihydroxyphenanthrene derivative of cyclotriphosphazene) 17 were synthesized with HCCP using already-reported methods.
Sample manufacture
A Haake Rheocorder System 90 was used to manufacture the experimental samples, and a Fuse Mp-50 press was used for compression molding. The Rheocorder was preheated to 180° and 50 g of an ABS resin sample was placed into the system. The synthesized flame retardants were mixed with this resin for 10 min. The mixed resin was then moved into the press for molding through compression. The press was preheated to 190° and the mixed resin sample was pressed to form the plate. The average thickness of this plate was 3.0±0.5 mm, and this plate was cut for the UL94 test and LOI experiment, respectively.
UL94 test
The plate-shaped sample was cut for the vertical UL94 test to 120±1 mm in length, 13.0±0.5 mm in width, and 3.0±0.5 mm in thickness. The sample was heated by a Bunsen burner flame for 10 s at the starting stage of the UL94 vertical test. The burning sample was removed from the fire, and the time until the fire extinguished was recorded and labeled as first after-flame time. After the fire had extinguished completely, the sample was heated again by a Bunsen burner flame for 10 s, and then it was removed form the fire. The time until the fire extinguished was recorded again, and labeled as second after-flame time. Five specimens were processed in this way and ten afterflame times (5 first afterflame times + 5 second afterflame times) were recorded. The results were categorized into 3 classes, V-0, V-1, and V-2. The V-0 class indicates that the after-flame time did not exceed 10 s, and a summation of 10 after-flame times for five specimens did not exceed 50 s. Additionally, a piece of cotton placed under the tested sample did not ignited during the test. The V-1 class indicated that the afterflame time did not exceed 30 s, and a summation of 10 afterflame times for five specimens did not exceed 250 s. Additionally, a piece of cotton placed under the tested sample did not ignite during the test. The V-2 class indicated that the after-flame time did not exceed 30 s, and a summation of 10 after-flame times for five specimens did not exceed 250 s. Additionally, a piece of cotton placed under the tested sample could be ignited during the test.
LOI experiment
A plate-shaped sample was cut to 120±1 mm in length, 6.5±0.5 mm in width, and
3.0±0.5 mm in thickness for the LOI experiment. A line was drawn at 50 mm from
the top of the specimen. The specimen was placed perpendicularly into the LOI
test-equipment. The time elapsed between igniting the top of the specimen and
allowing the flame to spread 50 mm downwards, had to be three minutes. Oxygen
and nitrogen concentrations were controlled to set this three-minute period.
Simply put, oxygen concentrations were adjusted upwards or downwards if the
times were greater or less than 3 minutes, respectively. Ten specimens were used
in each experiment, and the average results were obtained.
Results and Discussion
The chemical structures of the cyclotriphosphazene derivatives (BPCP, NTCP, and PTCP)
used in this study are shown in
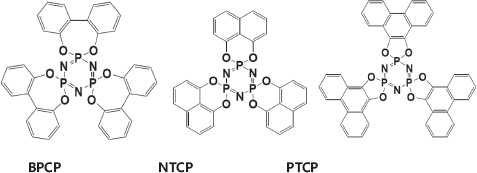
The molecular structure of the flame retardants
The thermal stability of the cyclotriphosphazene derivatives in this study was
estimated by using TGA, and the results were shown in

TGA of BPCP, NTCP, and PTCP
All of these cyclotriphosphazene derivatives (BPCP, NTCP, and PTCP) showed the weight decrease in 400–500°, and an especially abrupt decrease was observed at about 450°. These compounds showed a little difference thermal stability and the difference was only 5–10°. The order of the thermal stability was BPCP < NTCP < PTCP. However, the residual amount of each compounds at 600° showed large differences. It was 12% in case of BPCP, 23% in case of NTCP, and 27% in case of PTCP. The residual material is char, and it is known that the formation of char is related to flame retardant mechanism.
The synthesized cyclotriphosphazene derivatives were mixed with ABS resin to form
the sample. The flame retardant property of this sample was estimated by a UL94
test and LOI experiment, and the results are shown in
UL94 and LOI tests for ABS flame-retarded by BPCP, NTCP, and PTCP

UL94 and LOI tests for ABS flame-retarded by BPCP, NTCP, and PTCP

Average flaming time from ignition to extinguishment (first run / second run)
In
In all of the cases (10, 15, and 20 wt%), the order of flame retardant property is same as PTCP > NTCP > BPCT. These results are agreed well with the thermal stability results and the results of the formed char amount after heating at 600°.
The operating mechanism of the flame retardant is very complicated because it includes several mechanisms, and each mechanism can affect to other mechanisms. Therefore, all of the flame retardant mechanism could not be determined by estimating only one or two properties. For one reason or another, the results of this study showed that both the thermal stability of flame retardant and formation of char are one of the important factors to the flame retardant property.
Some researchers found that mixing with other compounds enhanced the synergistic effects of the flame retardant properties. The synergistic effect can help to reduce the amount of the flame retardant compound. Therefore, two compounds i.e., antimony(V) oxide and novolac, and their synergistic effects were tested in this study. It was known that they enhanced the synergistic effect for flame retardant property frequently in other cases. Antimony(V) oxide is an inorganic compound and novolac is an organic compound.
In this study, the total amount of flame retardant was fixed 20 wt% and the
mixing ratios of another compound to the flame retardant were 5:1 or 4:1. The
results are shown in
UL94 and LOI test for ABS flame-retarded by BPCP, NTCP, and PTCP with
antimony(V) oxide and novolac
a

UL94 and LOI test for ABS flame-retarded by BPCP, NTCP, and PTCP with antimony(V) oxide and novolac a

Total amount of flame retardant (flame retardant + additive) is 20 wt%
Antimony(V) oxide (AO), novolac (NO)
Flame retardant: additive
Average flaming time from ignition to extinguishment (first run / second run)
In
Using novolac, there was no synergistic effect for the flame retardant property
when NTCP or PTCP was used. These results were almost similar with the results
of using antimony(V) oxide. But a meaningful synergistic effect of novolac was
shown in the case of BPCP. The AFT value was reduced to 2-3 s, and LOI value was
increased as 1.5, comparing with the value as only BPCP was used. Because both
mixing ratio 5:1 and 4:1 showed the synergistic effect, the effect of mixing
ratio to flame retardant property was checked. The results are shown in
UL94 and LOI test for ABS flame-retarded by BPCP with novolac a

Total amount of flame retardant (flame retardant + additive) is 20 wt%
Novolac (NO)
BPCP: NO
Average flaming time from ignition to extinguishment (first run / second run)
In
It was known that several flame retardant mechanisms worked together when the
flame retardant was used. Using very flammable material, the fire is delivered
to the neighboring material quickly after ignition. At this stage, the
activation energy which is needed to ignite is delivered to the neighboring
material and the materials burn continuously. There are two kinds of
representative role of the flame retardant: the first is the non-flammability
itself, and this material prevents the delivery of the activation energy; the
second is the remaining of non-flammable material after burning. One
representative remaining material is char. It was known that novolac retained
char well after burning. We think that the synergistic effect of novolac is due
to the char formation from novolac using BPCP. In the case of using NTCP or
PTCP, there were no synergistic effects. The reason is that NTCP or PTCP forms
char well after burning, so the char formation of novolac is not meaningful. In
the
It is known that adding a flame retardant to a resin can lower the physical
properties of the resin, so we measured the physical properties of the flame
retardant ABS. Tensile strength and elongation were measured, and the results
are shown in
Tensile strength and the elongation of ABS flame-retarded by BPCP, NTCP,
and PTCP
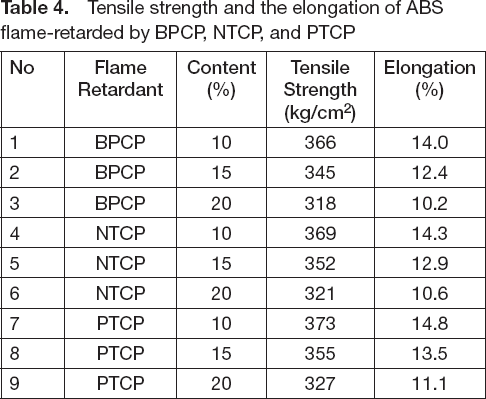
Tensile strength and the elongation of ABS flame-retarded by BPCP, NTCP, and PTCP

In
PTCP showed the best flame retardant property among the flame retardants used in this study. A combination of our current and prior results 10 yielded the following total order for the flame retardant properties of the samples i.e., PNCP > BPCP > CTCP > NTCP > HNCP > PTCP. PTCP showed the best performance among all of the compounds in our study for not only flame retardant property but also physical property. We think that the reason for the results is that the PTCP has the phenanthrene ring structure. A phenanthrene ring has greater unsaturation. Therefore the formed char amount at 600° is also high. Additionally, PTCP has the highest thermal stability among the flame retardants used in this study. Therefore, PTCP showed the best performance as a flame retardant.
Conclusion
Cyclotriphosphazene derivatives (BPCP, NTCP, and PTCP) were synthesized by the reaction of hexachlorocyclotriphosphazene with 2,2'-dihydroxybiphenyl, 1,8-dihydroxynaphthalene and 9,10-dihydroxyphenanthrene respectively. These three compounds were tried to use as flame retardants for ABS. The flame retardance was measured by UL94 and LOI tests. PTCP showed the best flame retardant activity among these three compounds, and it also showed the best tensile strength and elongation. Novolac showed the synergistic effect on flame retardance activity only in case of BPCP.

