Abstract
The present study aimed to fabrication and characterization the hydroxypropyl methylcellulose (HPMC) films containing different concentrations of sodium alginate (0.5, 1, and 3% v/v) and
Introduction
The food packaging industry has increasing attention to biodegradable active packaging because of consumer demand for nutritive, healthy food products, and extended shelf life, as well environmental concerns.1,2 The biopolymer blending is a suitable method for preparation of the biodegradable active packaging materials with improved properties. 3 The biopolymer blend films can comprise three possible combinations: polysaccharide/polysaccharide, protein/protein, and polysaccharide/protein blends. 4 Various studies have reported better properties for edible films produced by combining selected biopolymers, compared to the films made of just one component.5,6
Hydroxypropyl methylcellulose (HPMC) is a semi-rigid and synthetic gum that possess suitable properties for food packaging usage, including biocompatibility, low cost, and good film-forming ability which is attributed to its linear structure and nonionic nature.7,8 HPMC films are transparent, odorless, tasteless, oil-resistant with excellent mechanical properties and very efficient oxygen, lipid and carbon dioxide barrier properties.9,10 However, they are highly permeable to water vapor on account of the inherent hydrophilic nature of HPMC, which is a main drawback for its application in food packaging.
7
To overcome this drawback, previous studies suggested the blending of HPMC with other biopolymers to development of blend films with desirable properties.11,12 Sodium alginate is typically extracted from brown seaweed (
Furthermore, the providing of antioxidant and antibacterial activity in the bio-based films and improving of their water barrier properties by incorporation of essential oils have been reported in various studies.8,18–20
To our knowledge, no study has been conducted on the production of HPMC bio-films containing sodium alginate (AL) and
Materials and methods
Materials
HPMC, AL, 2, 2-diphenyl-1-picrylhydrazyl (DPPH) were obtained from Sigma-Aldrich Co. (St. Louis, USA). Glycerol, tween 80, magnesium nitrate, anhydrous calcium chloride, and All the chemicals used for extraction were purchased from Merck Chemicals Co. (Darmstadt, Germany). The aerial parts of
Extraction of DEO
Firstly, the aerial parts of the plant were dried and powdered. Then, 50 g of powdered aerial parts were hydrodistilled at 100°C for 3 hours by a Clevenger apparatus. The essential oil was dried under anhydrous sodium sulfate and stored at 4–5°C in dark glasses covered with aluminum foil for 24 h before use. 26 It should be noted that the antioxidant activity of obtained essential oil at different concentrations (4–20 mg/mL) was investigated DPPH assay and the concentration of essential oil that reduces the 50% of DPPH free radical (IC50) was 7.85 ± 3.37 mg/mL.
Preparation of films
One gram of HPMC powder was mixed with 50 ml distilled water (2%, w/v) and agitated by a magnetic stirrer (1200 rpm) at 65°C for 2 h. AL with three different concentrations (0.5, 1, and 1.5% w/v) was provided by dispersing in 50 ml distilled water and stirring the solution with a magnetic stir (1200 rpm) at 40°C for 1 h. Afterward, the prepared HPMC solution and AL solution were mixed together, and the DEO at three different concentrations (1, 3, and 5% v/v of film solution) was added to the final solution. Glycerol at a level of 50% w/w of HPMC was added to the film-forming solution as a plasticizer then was stirred for 30 minutes at 40°C. The solutions were cast evenly onto rectangular glass containers (20 × 12 × 1 cm3) and dried at room temperature for 48 h. The dried films were conditioned at 50% relative humidity and 25°C in a desiccator for 72 h before testing.13,17 The prepared film samples were coded as Table 1.
The prepared of HPMC based film samples.
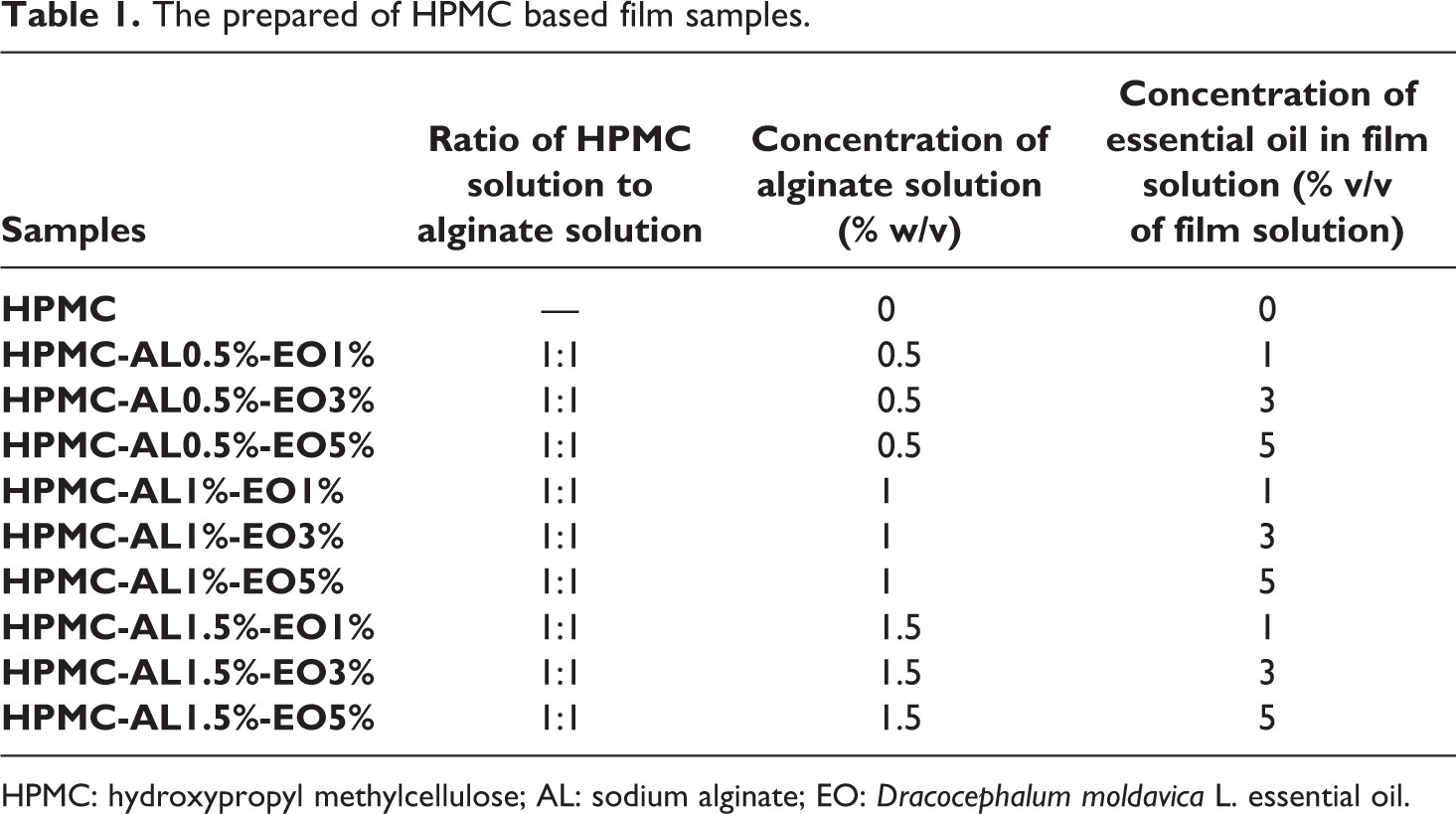
HPMC: hydroxypropyl methylcellulose; AL: sodium alginate; EO:
Characterization of films
Fourier transform infrared (FTIR)
To study the formation of possible structure interactions between the HPMC, sedum alginate, and essential oil, the neat HPMC, HPMC-AL1%-EO5%, HPMC-AL1.5%-EO5% film samples were selected to FTIR analysis. FT-IR spectrum of were obtained using FT-IR spectroscopy (Equinox 55LS 101, Bruker, Ettlingen, Germany) in the range of 400–4000 cm−1 at 0.5 cm−1 resolution.
Scanning electron microscopy (SEM)
The surface morphology of films was studied employing a SEM (Tescan VEGA 3 SBH, Brno, Czech). The film samples coated under vacuum with gold in an argon atmosphere prior to observation using a direct current sputtering technique (DST1, Nanostructured Coating Co., Tehran, Iran).
Film thickness
The thickness of film samples was determined using a digital micrometer (Mitutoyo, Kawasaki, Japan, accuracy of 0.01 mm). Measurements were performed at five randomly points of each film sample to calculate the average value., then the average values were evaluated and applied in the assessments of the mechanical properties and water vapor permeability.
Mechanical properties
Mechanical characteristics of films comprising elongation at break (EB) (%) and tensile strength (TS) (MPa), were measured using a TA. XT Plus Texture Analyzer (Stable Microsystems, Surrey, UK) according to ASTM standard method D882-10 (2010). The film samples were cut into dumbbell shape (8 cm × 0.5 cm) and were mounted in two grips at 50 mm. The initial grip and cross-head speed were fixed at 30 mm and 8 mm/min, respectively. 13
Moisture content (MC)
Prior to the experiments, films were put in a desiccator containing magnesium nitrate (52.9% RH at 25 ± 1°C) for at least 48 h. MC was measured by calculating the weight loss of about 0.2 g of each sample after oven-drying to constant weight at 105°C for 6 h. MC values were determined as follows 13 :
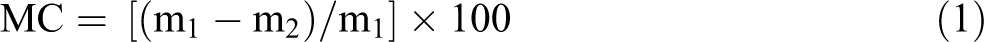
where m1 is the initial mass (g) of samples; m2 is the dry mass of the sample (g). MC (%) is moisture content of the samples expressed as g water/g wet base.
Water solubility (WS)
The WS of the films was calculated as the percentage of soluble matter to initial dry matter of the films according to the reported by Nisar et al. 30 Film pieces were dried at 105°C for 6 h in the oven to obtain the initial dry matter (W1). Then, the films were put in 50 ml distilled water and, stirred for 24 h at 250 rpm. The samples were filtered using a Whatman filter paper No. 1 and dried for 24 h at 105°C. Finally, the weight of the final dried sample is the weight of the insolubilized dry matter of final dry weight (W2). WS was assessed by applying the following equation:
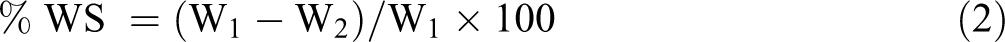
Water vapor permeability (WVP)
The WVP of the films was investigated according to the ASTM E96-05 (ASTM, 2005) standard method. The test film was sealed on the top of cups with a diameter of 7 mm containing 10 ml distilled water (100% RH; 2.337 × 103 Pa vapor pressure at 20°C). Then, they were put into a desiccator which was kept at 20°C and <0.2% RH. The weight loss measurement of the permeation cell indicated the water transferred through the film and adsorbed by the desiccant. The cups were weighed at intervals of 24 h for 6 days. The water vapor transmission rate (WVTR) was defined as the slope of the linear part of the curve (g/h) divided by the transfer area (m2). 31 Finally, the WVP (g/m·h·Pa) of films was calculated as follows:

where P is the saturation vapor pressure of water (Pa) at the test temperature (25°C), R1 is the RH in the desiccator, R2 is the RH inside the vial and X is the average thickness of film samples (m).
Antioxidant activity
The antioxidant activity of the films was assessed by the DPPH radical scavenging assay. Firstly, 25 mg of each film sample was dissolved in 3 ml of distilled water by continuous stirring for 2 min, and 1 ml of film extract solution was added to 0.2 mL of DPPH solutions (0.04 g/L) in ethanol. The mixture was vortexed vigorously and kept in the dark room for 30 min at room temperature. The reduction in absorbance at 517 nm was determined with UV–Vis spectrophotometer (Ultrospec 2000, Pharmacia Biotech, Cambridge, UK). Finally, the antioxidant activity was measured as percentage of DPPH free radical scavenging activity using the following equation. 32
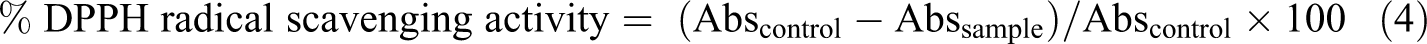
Color properties
Color values (
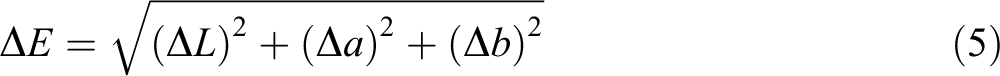
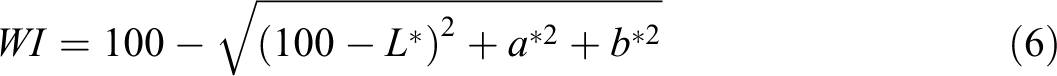
Statistical analysis
Statistical analysis was carried out based on Analysis of variance (ANOVA) and Duncan’s mean comparison tests at 5% significant level by SAS 9.1 software (SAS Institute Inc., Cary, NC).
Results and discussion
FTIR analyses
The results of FTIR spectroscopy the neat HPMC, HPMC-AL1%-EO5%, HPMC-AL1.5%-EO5% film samples are shown in Figure 1. The spectrum of control film (neat HPMC) showed several specified peaks including: (1) The peaks ranging 3116–3849 cm−1 that corresponded to hydroxyl groups (O–H) 33 ; (2) the peak at 2926 cm−1 that related to the C–H stretching vibrations which is attributed to the –CH3 arising from the substitution of hydroxyl groups by methoxyl and hydroxypropyl groups 7 ; (3) the peak at 1696 cm−1 that represented the existence of carbonyl groups (C=O) present in the glucose unit of cellulose 7 ; and (4) the specified peak at 1461 cm−1 which depicted the bending vibration of methylene (–CH3). 34 The addition of sodium alginate and DEO in the HPMC based films caused to some changes in the spectra of film samples. In the spectra of HPMC-AL1%-EO5% film sample, the peaks at 2926, 1696 and 1461 cm−1 were shifted to higher wavenumbers. As shown in the spectra of HPMC-AL1.5%-EO5% film, the peaks at 2926 and 1696 cm−1 were shifted to the higher wavenumbers, while the peaks at 1461 and 1251 cm−1 were shifted to lower wavenumber. These spectral changes can be attributed to the possible interaction (hydrogen bonds) between HPMC, sodium alginate and DEO. In accordance with these results, Okeke and Boateng 34 reported that hydrogen bonding in COO groups was observed at higher sodium alginate concentration in the HPMC based films which this phenomenon evidenced by shifting of the peak at 1647 cm−1 for COO– asymmetric stretching and the shifting of the peak at 1455 cm−1 for COO– symmetric stretching.
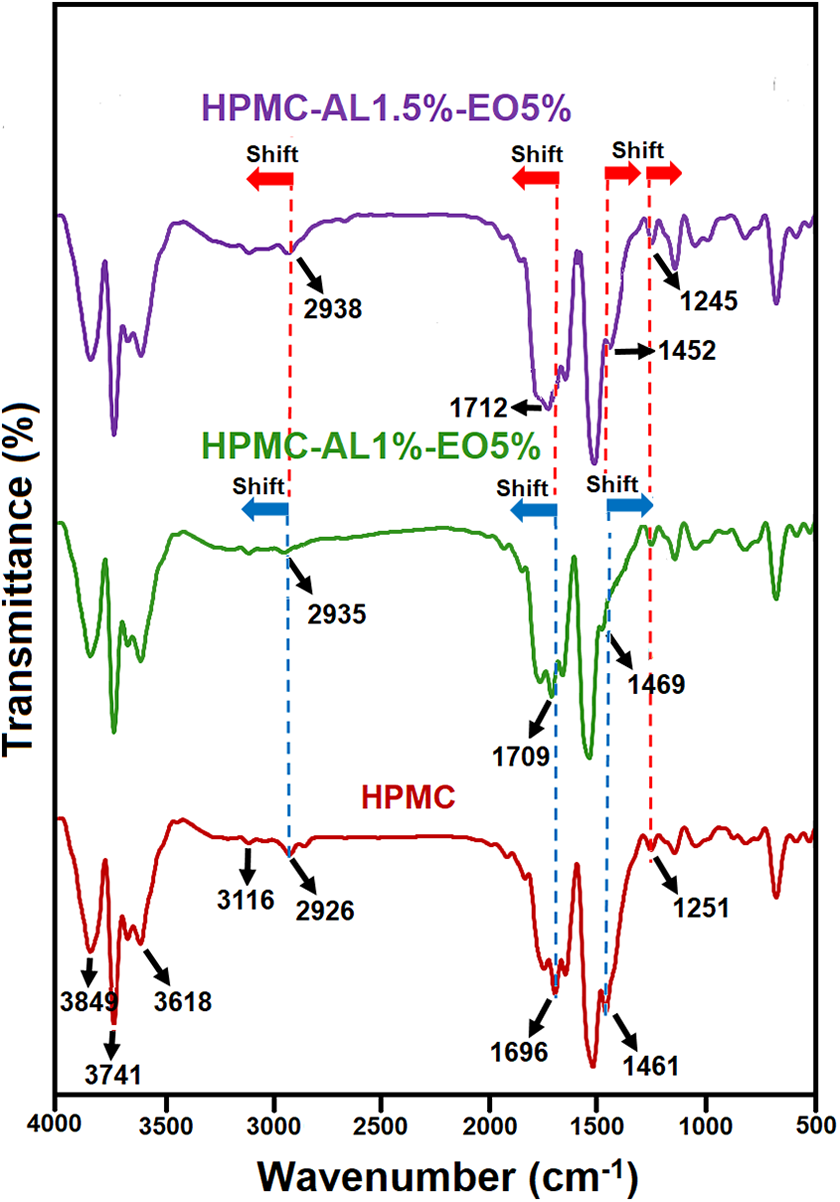
Fourier transforms infrared (FT-IR) spectra of neat HPMC, HPMC-AL1%-EO5%, HPMC-AL1.5%-EO5% film samples.
Scanning electron microscopy (SEM)
SEM analysis was applied to observe the morphological characteristics of the films. SEM micrographs of HPMC (a), HPMC-AL0.5%-EO1% (b), HPMC-AL0.5%-EO5% (c), and HPMC-AL1.5%-EO5% (d) films at magnifications of 2 µm are shown in Figure 2. As can be noticed, the control film possesses a rough and uneven structure with a fairly harsh crack. By incorporating of 0.5% alginate into the films, the number of cracks were decreased notably and became smaller compared to the control film. Decrease in the number of cracks as a consequence of addition of alginate enhanced the mechanical resistance of the treated films which is confirmed by results of tensile strength. On the other hand, the addition of 1.5% alginate at the constant concentration of DEO (5%) into the HPMC film resulted in the formation of holes and more cracks. In fact, the presence of the essential oil generates the non-uniformity in the structure of the HPMC, which could be attributed to the volatility of the DEO compounds and the effect of oil in water emulsion. Furthermore, the presence of a large number of cracks at higher concentrations of alginate may be due to its non-uniform distribution within HPMC polymer. In accordance with our results, Bazzaz et al. reported that the

Scanning electron microscopy (SEM) images of pure HPMC (a), HPMC-AL0.5%-EO1% (b), HPMCAL0.5%-EO5% (c), and HPMC-AL1.5%-EO5% (d).
Thickness
Thickness is an important parameter for measurement of mechanical properties and water barrier properties of films. 35 The thickness of film samples is shown in Figure 3(a). The results showed that with increasing the alginate content, the thickness of films was significantly increased, while no significant (P > 0.05) difference resulted by the addition of DEO to the samples in thickness. Therefore, the highest thicknesses belonged to films containing 1.5% alginate with various concentrations of essential oil (0.172–0.173 mm), which shows a significant difference with the thickness of the control films (0.143 ± 0.005 mm). This finding can be related to the increasing of dry matter content by addition alginate, which the same results were reported in previous studies. 15 Additionally, Choi et al. reported the thickness of HPMC films revealed no significant differences after the incorporation of essential oils. 36
Mechanical properties
Mechanical properties are influenced by several factors, including interactions between film compounds, temperature, microstructural characteristics, and chemical conditions. 37 The TS and EB, parameters representing the mechanical properties of the films are summarized in Figure 3(b) and (c). Solely HPMC films exhibited a TS of 8.8 MPa and EB of 14.94%. An improvement in the mechanical properties of the prepared blend films in comparison to pure HPMC film was observed. By incorporation of sodium alginate, TS significantly (p < 0.05) increased. However, with an increase in its loading ratio to 1.5%, TS leveled off. The highest values of TS (16.13 MPa) was obtained for the HPMC-AL0.5%-EO1% blend films. The results also indicated that upon blending with sodium alginate, the EB values of the blend films generally increased compared to the pure HPMC films (p < 0.05). Improved mechanical properties of the blend films could be attributed to the presence of specific intermolecular interactions between HPMC and alginate. Similar results were reported for the blend films of gelatin and sodium alginate, 13 and HPMC and whey protein isolate. 11 Their results demonstrated that the formation of the crystalline domain and hydrogen bonds that is favorable for increasing the mechanical properties of blend films. 38 Within the concentration range under consideration, the further increase of the alginate ratio to 1.5% led to a decrease in the TS. The decrease in TS due to the increase of alginate concentration in the formulation of the films has been noted by other studies. 39 The mechanical properties of polymer blends are influenced by the degree of interaction at the interfaces of the components, distribution, and intensity of inter- and intra-molecular interactions. 39 Additionally, structural discontinuities in the polymeric network might contribute to the inferior mechanical properties of the obtained films. 15 The partial miscibility of alginate into the HPMC matrix could result in the formation of micro-domains within the polymeric matrix. Furthermore, Almasi et al. have suggested that the biopolymers might play the role of impurities and raise the stress concentration points in the matrix which could initiate fracture and decrease the mechanical properties of blend films. 40 According to SEM images, the small pores and cracks or fractures available on the surface of the films (Figure 2) could corroborate the lower mechanical properties observed for this blend films. The amount of EB decreased with the addition of DEO, so that the lowest percentage of elongation was observed for films containing the highest amount of DEO. These results can be explained by reduction of cohesiveness within film matrix due to the increase of structural discontinuities by the incorporation of DEO.25,30 The decrease of EB by the incorporation of different essential oil to a bio-polymeric matrix was also reported by other authors.35,41 However, there is also a discrepancy between the reported effects of different essential oils on the EB values of HPMC films in the literature, which can be attributed to the different compounds and different percentages of components in each essential oil. Therefore, different results were observed in the application of various essential oils, so a similar trend of decrease in mechanical properties by incorporation of essential oils cannot be observed in all films. In this regard, Sánchez-González et al. reported that the incorporation of tea tree essential oil into HPMC edible film decreased film’s tensile strength and elastic modulus, however, it didn’t show any significant effect on the EB. 42
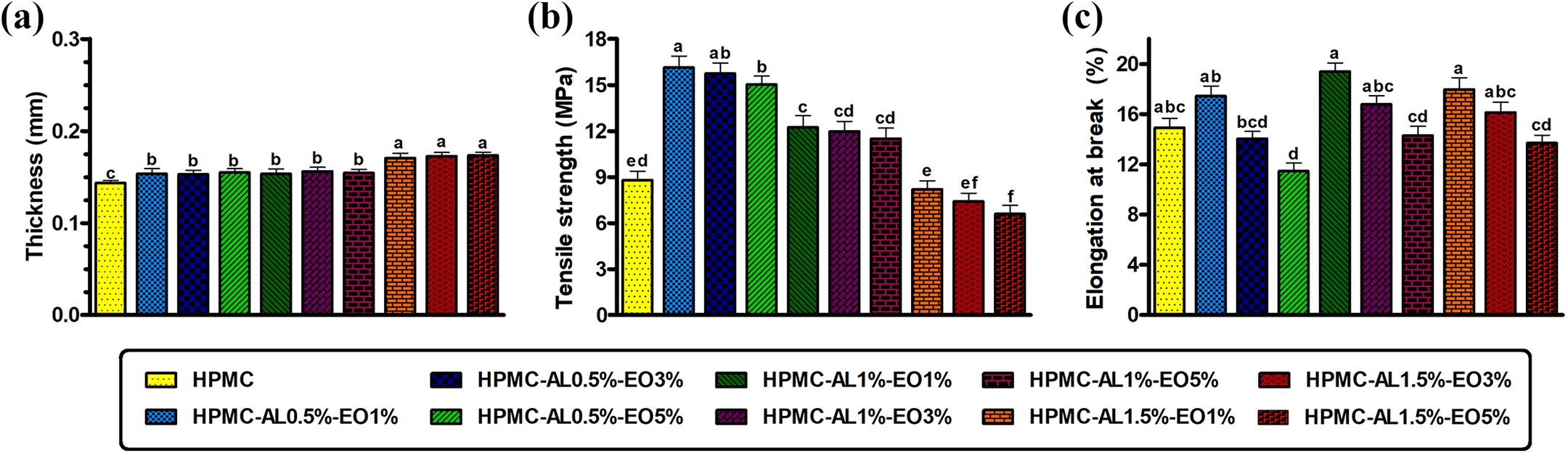
Thickness (a), tensile strength (TS) (b) and elongation to break (EB) (c) profiles of film samples. Data are expressed as mean ± standard deviation (n = 3) and different letters show significant difference at the 5% level in Duncan’s test (p < 0.05).
Moisture content (MC)
The results of MC measurement are shown in Table 2. As a result, the MC was significantly influenced by alginate while it was not affected by essential oil (p > 0.05). The results revealed that there is a significant (p < 0.05) reduction in the MC of HPMC films with the increase of alginate concentration; so that the lowest MC was observed in the film containing 1.5% alginate. The highest and the lowest MC belonged to pure HPMC film (33.21%) and HPMC-AL1.5%-EO5% blend film (6.30%), respectively. As a result, at the constant concentration of alginate, the MC of the films was not significantly affected by enhancing the concentration of DEO from 1 to 5%, which indicates that DEO does not influence the MC of the films. According to previous studies, the addition of essential oils in the biopolymer-based film may be caused to the reduction of MC in the films due to the hydrophobic nature of essential oils.8,29 The observed results in this study can be attributed to the levels of DEO and these levels of essential oil weren’t enough to changes in MC. Furthermore, the decrease in the MC of the films by addition of alginate is probably due to the strong hydrogen bond formation between the alginate and the HPMC film matrix. 17 In other words, water molecules cannot break these strong bonds appropriately, and diffuse through the film matrix. Hence, the MC was reduced. 43 The interesting point in this regard is that the results of MC measurement are in line with the results of the thickness measurement. So that, the film samples with higher thickness showed lower MC which this phenomenon can be attributed to the presence of more interactions in thicker samples.
Effect of alginate gum and
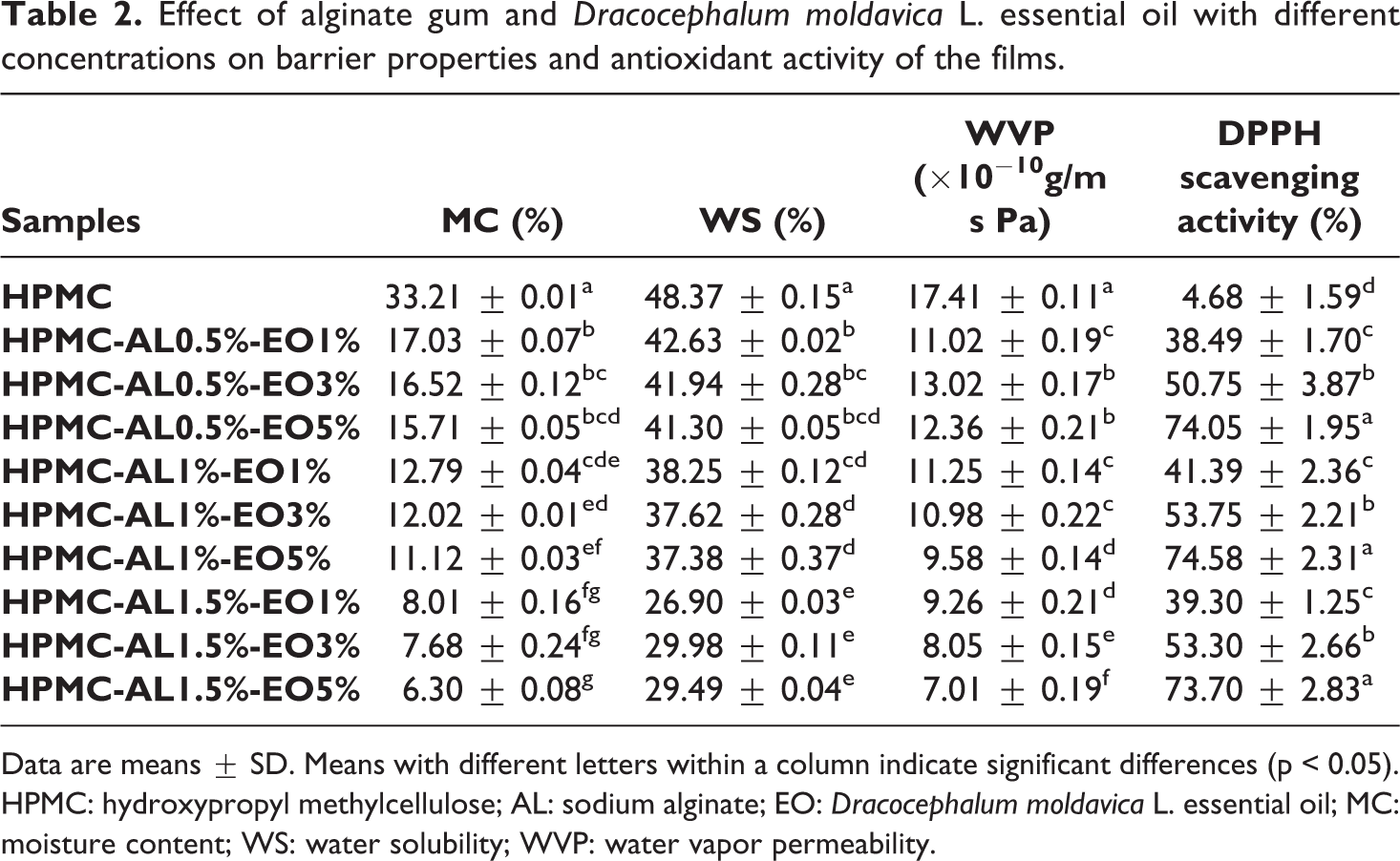
Data are means ± SD. Means with different letters within a column indicate significant differences (p < 0.05). HPMC: hydroxypropyl methylcellulose; AL: sodium alginate; EO:
Water solubility (WS)
As shown in the results, the WS of the films were significantly (p < 0.05) decreased by increasing the concentrations of alginate, while showed no significant (p > 0.05) difference by incorporation of DEO (Table 2). Thus, the highest solubility (48.37%) was obtained for pure HPMC film and the lowest WS was observed for HPMC-AL1.5% with 1–5% of EO blend films. These results can be explained by the formation of interactions between HPMC, alginate, and essential oil which caused to decreased the availability of hydroxyl groups, and consequently, caused to increase the hydrophobic nature of the films. 44 These results approved the results of MC measurement and indicated that the addition of alginate and DEO increased the water barrier properties of HPMC based films.
Water vapor permeability (WVP)
As the results presented in Table 2, the presence of alginate and its inclusion with DEO made a significant reduction in WVP of HPMC films (p < 0.05). The increasing of alginate concentration from 0.5 to 1.5% significantly (p < 0.05) reduced the WVP of films. Thus, the lowest value of WVP related to HPMC-AL1.5%-EO5% blend film, which was equal to 7.01 × 10−10 g/m·h·Pa. Moreover, WVP of the films was affected by the interaction between alginate and DEO. In other words, the DEO probably intensified the barrier effect of alginate which means that at the same concentration of alginate, films containing a higher concentration of DEO exhibited lower WVP. In fact, the incorporation of DEO into the film formulation caused to a reduction in the hydrophilic nature of the films and an improvement in the water vapor barrier characteristics. 41 It has been noted that these results are in line with the results of MC and WS measurements. Furthermore, these findings appeared to be quite consistent with results of FTIR and SEM where approved the interactions between HPMC, alginate and DEO and formation of pores, cracks, and holes in film structure. This finding is in accordance with those reported by previous studies.17,38,39
Antioxidant activity
Incorporation of antioxidants into food packaging materials may help to keep the quality of food products, especially in control of the fatty components oxidation. Table 2 exhibits the antioxidant capacity of the HPMC films incorporated with alginate and DEO in comparison with the control film. As expected, pure HPMC films presented a slight scavenging activity (3%). The antioxidant activity of the films significantly (p < 0.05) enhanced by increasing the essential oil content. The highest antioxidant activity of film samples was associated with the film samples containing 5% of DEO. Additionally, the antioxidant activity of films showed no significant (p > 0.05) difference by increasing the alginate level. Therefore, the antioxidant activity of film samples can be attributed to the high antioxidant activity of DEO. 21 According to previous literature, the main compounds in DEO are geranyl acetate, geranial, neryl acetate, geraniol, neral, and nerol which the antioxidant activity of DEO can be related to these compounds.23,24 Similar results were reported in another study in case of HPMC films formulated with clove essential oil. 44
Color properties
Color is recognized as an imperative factor affecting consumer acceptance of a food product.
36
The color parameters of the films are shown in Table 3. The
Color parameters of HPMC-based films incorporated with different concentrations of alginate gum and
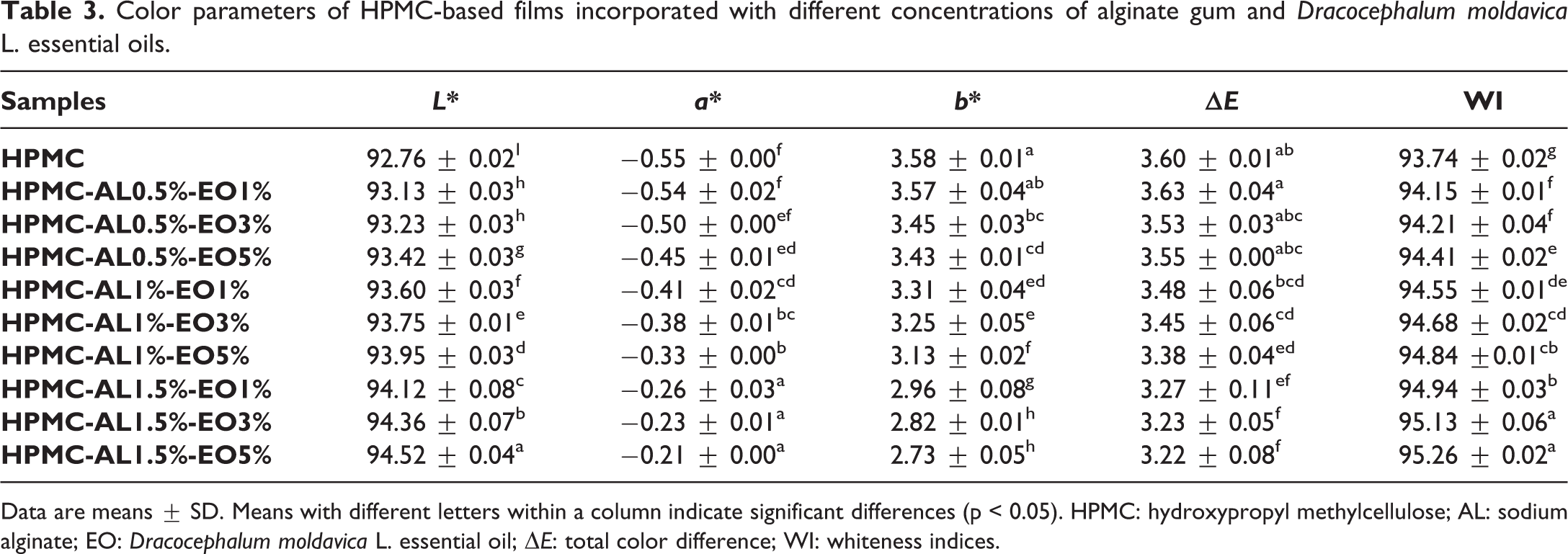
Data are means ± SD. Means with different letters within a column indicate significant differences (p < 0.05). HPMC: hydroxypropyl methylcellulose; AL: sodium alginate; EO:
Conclusion
The HPMC-based films incorporated with alginate and DEO were developed and characterized. FTIR patterns confirmed that there was an interaction between alginate groups with the hydroxyl group of HPMC. SEM images showed a decrease in the number of cracks by the addition of alginate and formation of the microporous holes on the films’ surface by the addition of DEO. The blend films showed higher antioxidant activity, improved water barrier properties, as well as appropriate mechanical properties compared to pure HMPC films. Generally, the prepared blend films exhibited high potential for use as a suitable alternative to synthetic packaging.
