Abstract
The aim of the present study was to produce a blend of bacterial cellulose (BC) and poly(3-hydroxybutyrate) (PHB) from the combination of pure BC membranes and 30% PHB in acetic acid. Clove essential oil (CLO) was then added as an antimicrobial agent. BC membranes were produced from Gluconacetobacter hansenii in a modified Hestrin–Schramm medium containing corn steep liquor. The scanning electron microscopic analyses revealed a visible white lining on the BC surface due to the deposition of PHB. Other analyses such as oil permeability, flexibility, and water solubility, which showed no trace of oil permeation through the films, resistance to folding (more than 100 times), and hydrophobicity, respectively, demonstrated the improvement of the material due to the blend of the polymers. When compared with the pure PHB polymer membrane, the addition of the essential oil led to a substantial reduction of 65% in microbial growth and better mechanical and thermal properties, since the traction resistance value increased by 3.9 times while the maximum degradation rate was 10°C higher. The new material, composed of BC/PHB with the addition of CLO, has attractive properties for use as a biocompatible, biodegradable, active food packaging wrap.
Introduction
The packaging of food is essential to the containment, protection, and preservation of products and provides a medium for communicating information about the product, brand and so on. 1 However, the crucial problem with regard to the use of plastics for packaging is its postconsumption fate, as packaging is by far the major contributor (63%) of plastic waste products. 2 Moreover, some materials are difficult to reuse. Indeed, it is estimated that less than 14% of plastic packaging materials are recyclable. 3
Since most plastics are nonbiodegradable, these materials remain in the environment for long periods of time and continue to be a threat to human health and the environment. 4 Therefore, composites formed by biodegradable polymers obtained using renewable resources are an attractive alternative to petroleum-based plastics, since the degradation of these products leads to nontoxic waste that does not harm the environment. This growing need for environmental preservation has intensified the use of renewable resources in the manufacturing of polymeric films, such as packaging wrap, for maintaining the quality of packaged foods. However, these biopolymers have limitations with regard to use as disposable packaging, such as low thermal stability during processing and little flexibility. To overcome these limitations, a blend of two polymers [one with a structure in the form nanometric filament (bacterial cellulose (BC)) and one with a property similar to a synthetic polymer (poly(3-hydroxybutyrate (PHB))] offers a promising alternative since BC shows mechanical and structural properties, while PHB is a polymer with thermoplastic properties. Several studies have focused on the development of completely biodegradable blends manufactured in a sustainable manner 5 and those that combine cellulose and polyhydroxyalkanoates (PHA) are particularly important due to their intrinsic properties of biodegradability 6 and biocompatibility, as it will be described below.
Cellulose is the most abundant polymer on earth and the growing interest in this substance is due to its fibrillar nature and potential as a reinforcing material in composites. Moreover, cellulose is biodegradable, sustainable, and renewable. It has long been produced from plant sources. However, BC, which is produced in large quantities by bacteria of the genus Gluconacetobacter, is particularly attractive due to its purity and highly crystalline nanostructure. 7
Several studies have described quantities of cellulose produced by species of Gluconacetobacter cultivated in different media obtained with the substitution of the carbon and/or nitrogen components and a wide variety of carbon and nitrogen sources, along with the inclusion of additional supplements, has been investigated for this purpose. 8,9
BC has been produced from agricultural and industrial waste products, including food waste, wheat straw, fruit scraps, glycerol residue, and cotton-based textile waste products. Such waste products provide cheap raw materials for the production of BC while offering benefits to the environment. 10 The use of such waste materials enhances the sustainability of cellulose production by microorganisms and diminishes environmental pollution associated with the discarding of industrial waste.
PHA constitutes a family on intracellular biopolymers synthesized by bacteria as intracellular granules for storing carbon and energy. One member of this family—PHB —is produced by Ralstonia eutropha. PHB is a biocompatible, biodegradable, thermoplastic polymer with plastic properties that offer the possibility of replacing currently used nondegradable polymers such as polyethylene and polypropylene. PHB has a high fusing point (∼175°C), but its thermal instability and fragility are the main disadvantages for use in potential applications. However, BC is one of the substrates that can confer strength to PHB, as improves its mechanical properties through adsorption mechanisms. 11
BC blends with different polymers have been studied for applications in different fields. 12 Examples of such blends include chitosan, 13 polyurethane, 14 and PHA. 15 -17 PHB/corn starch blends have been used in packaging 18,19 and PHB/chitosan/ketoprofen microparticles have been used for the controlled release of drugs. 20 PHB/cellulose ester blends have been studied, 21 with completely miscible amorphous blends obtained using a PHB content of up to 50%. (PHB was found to crystallize at higher concentrations.) Jiang et al. 22 developed two-layer films using PHB and cellulose paper, reporting reductions in moisture absorption and the roughness of the surface compared with cellulose paper. Therefore, the possibility of obtaining a biodegradable material with better mechanical and barrier properties has clearly been demonstrated.
The growing demand for minimally processed foods and fresh products raises issues regarding the quality and safety of these products. Therefore, new methods have been developed to inhibit microbial growth in foods while maintaining the freshness, quality, and safety of the product. One of the most widely used methods is antimicrobial film. 23 Antimicrobial packaging reduces the growth rate of microbes and increases the shelf life of foods. Active packaging is attracting the interest of the food and packaging industries, as low levels of total additives in the packaging system rather than conservatives added directly to food products can help meet the growing demand for products without conservatives. 24
Therefore, the aim of the present study was to develop a novel polymeric material composed of a BC/PHB blend activated with clove essential oil (CLO) so that the film can serve as a biodegradable antimicrobial active packaging material for use in the industry of perishable foods.
Experiments
Materials
PHB in powder form (brand name: biocycle; molar mass: 563,110.00 g mol−1; density: 1.23 g mL−1; and fusing point: 178.4°C) was acquired from PHB Industrial S.A. (Serrana, São Paulo, Brazil) Sodium hydroxide (NaOH), chloroform, and acetic acid were acquired from MERTEC (Brazil).
Microorganism
The bacterium Gluconacetobacter hansenii UCP UCP1619 deposited in the Culture Bank of the Centre for Environmental Sciences of Universidade Católica de Pernambuco was used as the BC producer.
Growth medium and standard maintenance of microorganism
The method used to maintain the viability of the microorganism and produce it in a preestablished standard medium was based on studies conducted by Hestrin–Schramm (HS) 25 and adapted by Hungund and Gupta 26 and Gomes et al. 27 The HS medium used for the growth of the bacterium was composed of 20.0 g L−1 of glucose, 5.0 g L−1 of peptone, 5.0 g L−1 of yeast extract, 2.7 g L−1 of Na2HPO4, and 1.15 g L−1 of citric acid at pH 5.0. Moreover, 15 g L−1 of agar was added to the HS medium for the maintenance of the bacterium. Both media were sterilized at 121°C for 15 min and the maintenance medium was distributed among Petri dishes and Falcon tubes for the transference of the microorganism. The microorganism was kept refrigerated in a Falcon tube.
Production and purification of BC
BC was produced in a modified HS medium containing 1.5% glucose, 2.5% corn steep liquor, 0.27% Na2HPO4, and 0.15% citric acid at pH 5.0, as described by Costa et al. 28 The modified HS medium was sterilized at 121°C for 15 min.
The preinoculum was prepared by growing the bacterium at 30°C under static conditions for 48 h in 100 mL of liquid HS medium, pH 6.0, in 500-mL Erlenmeyer flasks.
The inoculum was prepared through the transference of 3% preinoculum in modified HS medium, followed by static cultivation at 30°C for 48 h. The cell suspension was inoculated at 3% in modified HS medium in 500-mL Schott flasks containing 100 mL of the production medium, followed by static incubation at 30°C for 6 days. All experiments were performed in triplicate.
After cultivation, the fermentation broth and BC pellicles were collected and cleaned by immersion in a 4% NaOH solution for 2 h to eliminate the retained cells. The BC was then weighed and the yield was calculated. The samples were placed in a desiccator prior to weighing. 10
Preparation of BC/PHB blend by dissolution
The blend was prepared by placing 10 g of the PHB polymer in a reagent flask, to which acetic acid (150 ml) was added, followed by heating until complete dissolution (∼90°C), similarly as described by Anbukarasu et al. 29 The recipient with the solution was placed in a water bath. Previously weighed, purified, dried BC membranes were immersed in the solution. The BC/PHB blend was prepared with 30% PHB. After resting, the blend was washed with deionized water and dried in an oven at 50°C until reaching a constant mass. Visually, the end product exhibited a close bond in the blend of the two components. All dried samples were kept in a desiccator.
Incorporation of CLO to BC/PHB blend
Ten percent of CLO was added to the BC/PHB blend as an antimicrobial agent. First, the blend (1 g) was immersed in 150 mL of ethanol at 60°C for 10 min. The wet blend was then weighed to calculate the mass of incorporated ethanol. Next, a solution containing 3.0 g of the essential oil in 20 mL of ethanol was prepared. The wet blend was immersed in the solution, which was heated for 5 min until the evaporation of the ethanol solution. The blend containing the essential oil was placed in an oven at 50°C until complete drying and the difference in mass between the blend with and without oil was calculated to determine the percentage of oil incorporated.
Oil permeability
Three grams of soybean oil were placed in a glass tube (inner diameter: 25 mm and outer diameter: 27 mm) and a piece of the BC/PHB blend (50 × 50 mm2) was pressed firmly to the open end to seal the tube. The tube was inverted and placed on filter paper over a glass slide to determine whether the oil permeated the blend over time. Observations were made of at least five samples after 3 days of storage. 30,31
Flexibility
The BC/PHB film was folded 100 times along the same line to test its flexibility. Flexibility of the film was considered good if no groove was visually detectable. The number of folds until failure was recorded and scored as follows: <20 times = poor, 20–49 times = fair; 50–99 times = good and ≥100 times = excellent. 31
Water solubility
The BC/PHB film water solubility was measured by placing the test films into deionized water at 25°C and then the time for completely dissolving the film was recorded. Three samples of each film were tested. 32
Antimicrobial activity of BC/PHB blend containing essential oil
For the determination of the antimicrobial potential of the CLO in the BC/PHB blend, Luria–Bertani (LB) medium was inoculated with a 1% (v/v) Escherichia coli UCP 1517 solution. Under sterile conditions, a piece of the sterilized blend (2 × 2 cm2) was transferred to a test tube inoculated with 5 mL of the fresh E. coli suspension. The BC/PHB blend without the addition of the antimicrobial agent (CLO) and the LB medium inoculated with the microorganism were used as the controls. The assays were performed in triplicate. All tubes were incubated at 30°C. Samples were collected at predetermined time intervals (6, 12, 24, and 30 h) and the optical densities were read at 600 nm. 33 The count of microorganisms was performed by the plating method in counting agar. After inoculation and solidification of the medium, plates were incubated at 37°C for 48 h and cells counted as CFU mL−1.
Scanning electron microscopy
Samples of the BC/PHB blend, pure BC membranes, and pure PHB were dried and coated with metal (SANYU ELECTRON, Shanghai, China). Scanning electron microscopy (SEM) of the surfaces of the samples was performed using a JEOL scanning electron microscope (model JSM-5600, Tokyo, Japan) operating at 27 kV.
Mechanical properties
For the characterization of the mechanical properties of the BC/PHB blend, resistance to traction at maximum load (MPa), deformation until breakage (ε; mm%), and Young’s elastic modulus (E) were determined based on Rethwisch and William. 34 Samples of the dried blend were cut into rectangular strips (10 × 30 mm2). Mean film thickness was 0.10 mm. The traction test was performed at room temperature at a velocity of 5 m min−1 and a static load of 0.5 N using a universal testing machine (EMIC DL–500MF, Brazil), following the ASTM D882 method. The Bluehill Lite™ program was used to calculate the tension–deformation ratio and elastic modulus.
X-Ray diffractometry
The X-ray diffractometry (XRD) patterns of the blends were measured using a Phillips X’pert MPD diffractometer (Phillips, Netherlands) with copper K α radiation. The percentage of crystallinity was calculated as x (%) = (I max − I min)/I max × 100%, in which I max is the height of the peak at 2θ = 22.5° and I min is the valley between the peaks at 2θ = 22.5° and θ = 16.3°. 27
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was performed using a Mettler Toledo analyzer (Mettler-Toledo Ind. e Com. Ltda., Barueri, São Paulo, Brazil) on samples weighing approximately 8 mg. Each sample was scanned along a temperature range from room temperature to 600°C, under nitrogen atmosphere with a heating rate of 10°C min−1 and a flow rate of 20 mL min−1 to avoid thermoxidative degradation of the sample. 35
Attenuated total reflectance and Fourier-transform infrared spectroscopy
Prior to the analysis, the blends were placed in a desiccator containing silica gel for 2 weeks at room temperature to obtain more dehydrated films. The films were digitized with a Bruker Fourier transform infrared (FTIR) spectrometer (Equinox 55 Model, Bruker Co., Ettlingen, Germany). The samples were measured in a horizontal attenuated total reflectance device through a crystalline cell plate (45° ZnSe, 80 mm in length, 10 mm in width, and 4 mm in thickness; PIKE Technology Inc., Madison, Wisconsin, USA). Analyses were conducted to identify functional groups in the samples and analyze the interactions among the BC/PHB blends in a spectrophotometer between 4000 cm−1 and 400 cm−1. All spectra were recorded after 32 scans with a resolution of 4 cm−1. 36
Statistical analysis
The data from each test were analyzed statistically. Analysis of variance was used to evaluate the significance in the differences between factors and levels. The comparison of means was performed using Tukey’s test to identify which group differed significantly from other groups (p < 0.05). All data were expressed as mean and standard deviation. Principal components analysis (PCA) was performed to determine correlations between the BC and the PHB films.
Results and discussion
BC membrane yield
The yield in terms of hydrated membranes was 370 g L−1 and in terms of dried membranes was 9.5 g L−1, showing the presence of 97% of water. This result obtained is in agreement with Costa et al. 28 and Lin et al. 37 After removal from the culture medium, the purification step with NaOH favored a color transformation as well as the removal of metabolites and possible residue from the culture medium adhered to the surface of the membranes.
General characteristics of BC/PHB blends
Immediately after the removal of the solution containing PHB, the formed blends were somewhat transparent, indicating the absorption of the solution by the BC. After immersion in water, the BC became opaque due to the sudden precipitation of the PHB among the cellulose fibers.
After drying the BC/PHB blend, the amount of PHB incorporated (30%) was determined by the difference in weight between the dry cellulose and the blend after the incorporation of the PHB. Membranes of approximately 150 µm in thickness were obtained, while the pure BC showed 70 µm in thickness. The pure BC and membrane of the BC/PHB blend had a milky and white coloration, respectively, as shown in Figure 1.

Color and appearance of (a) BC film and (b) BC/PHB blend.
Oil permeability, flexibility, and water solubility
The BC/PHB films were tested with regard to oil permeability to ensure that oil would not leak through the films when used for wrapping oily products. Besides leaking, permeability would also affect the appearance, flavor, and properties of products. The results revealed that the soybean oil exhibited no trace of permeation through the films. BC/PHB films have hydrophilic hydroxyl and carboxyl groups that impede the adsorption of oil molecules to the surface. Hu et al. 30 and Chen et al. 31 report similar results for potato starch films with the addition of glycerol as a plasticizer and cellulose sulfate (NaCS) films plasticized with glycerol, respectively. The BC/PHB blends also exhibited excellent flexibility, as remaining intact after being folded more than 100 times. Regarding solubility, the films obtained by the BC/PHB blend were insoluble in water for the time evaluated using the method employed in this study.
SEM characterization of BC/PHB blends
Figure 2 shows the images of the pure dried PHB film, pure dried cellulose film, and BC/PHB blend with a 30% polymer content. Figure 2(a) shows the surface of the pure cellulose film, revealing a network of randomly arranged cellulose nanofibers. The BC exhibited an interlaced structure of ultrathin nanofibrils. Figure 2(b) shows the surface of the pure PHB, revealing a rugged granular structure with microparticles. Figure 2(c) shows the BC/PHB blend, revealing cellulose microfibers filled with PHB as well as PHB microparticles associated with the cellulose microfiber network. The PHB is on the outer surface of the fiber, covering the cellulose nanofibers on the surface, indicating that the PHB was incorporated with the fibrils of the BC. Ruka et al. 17 and Barud et al. 15 report similar results regarding the surface structure of cellulose and PHB blends analyzed using SEM.

SEM of (a) dried BC film, (b) dried PHB film, and (c) BC/PHB blend (magnification: ×1500).
Antimicrobial activity of BC/PHB blend containing CLO
The results of the antimicrobial tests with E. coli are displayed in Figure 3. The samples of the BC/PHB blend containing CLO achieved a visible reduction in the growth rate of the bacterium, especially in the first hours of incubation, compared with the samples of the culture medium and blend without the essential oil. The antimicrobial activity of CLO has been attributed to the presence of its main functional components (i.e. eugenol and carvacrol) since these hydrophobic compounds are capable of penetrating the bacterial cell membrane to disrupt the cell structure. 38 With the usage of essential oils, when applied as an antimicrobial agent, the mechanism of action happens at a membrane level along with inactivation of both the enzyme and the genetic material. 39 The results indicate that the BC/PHB film with CLO is a promising antimicrobial additive for food packaging. The major component of CLO is eugenol, which has an inhibitory effect on the growth of many pathogenic microorganisms. 40
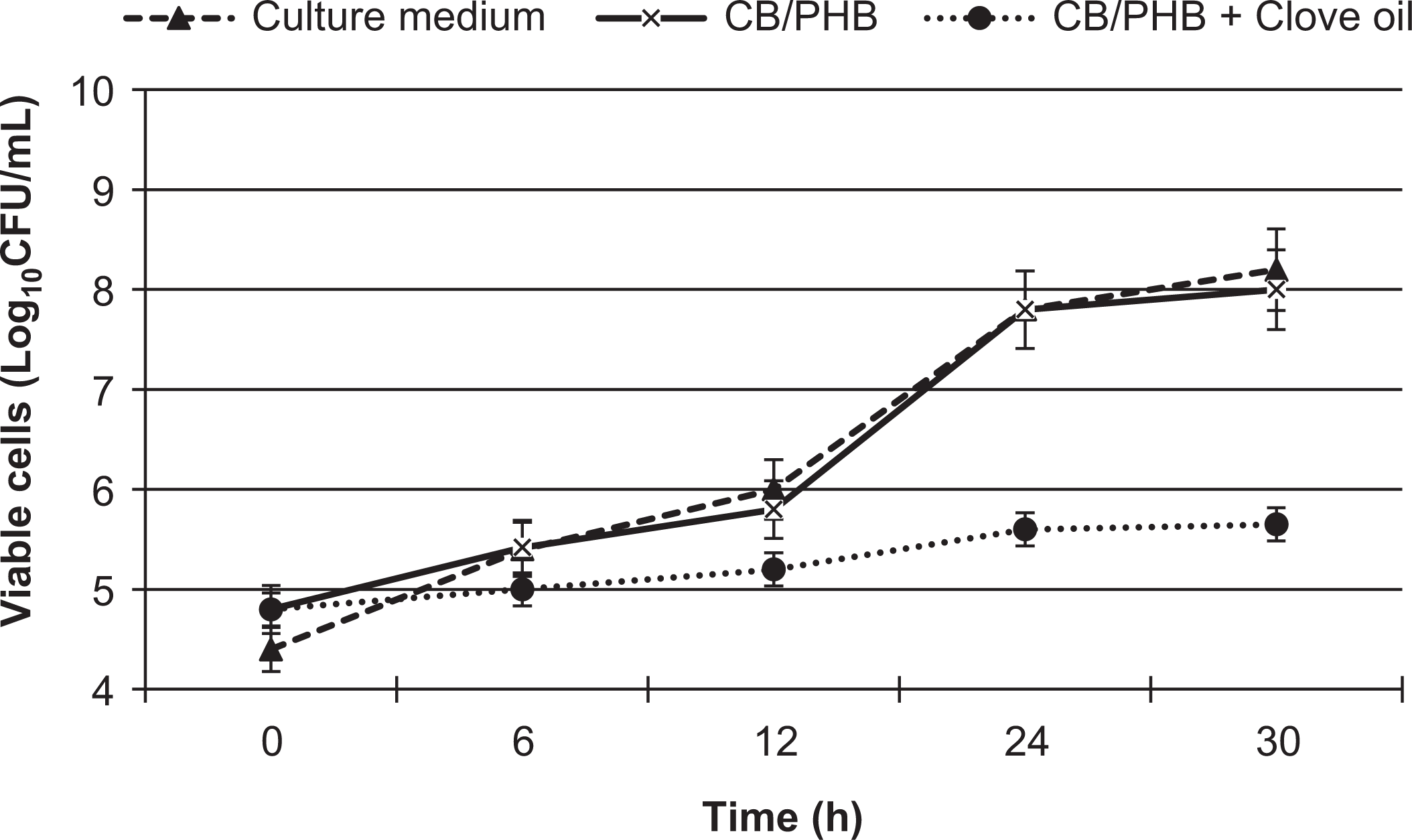
Growth of Escherichia coli in samples of BC/PHB blend containing CLO over time. Samples of blend without essential oil and culture medium used as controls.
Another important aspect for an active packaging material is to favor the migration of the active ingredient so that can interact with the food product, minimizing or impeding microbial proliferation. Therefore, the BC/PHB blend with CLO is adequate for this purpose, as the migration of the oil began in the first 6 h of contact with the bacterial cells, favoring an approximately 65% reduction in microbial growth at the end of 30 h. The incorporation of different substances into a polymeric material for the development of antimicrobial food packaging materials has been described. Dobre et al. 33 report similar results for a poly(vinyl alcohol)/BC blend containing sorbic acid as the antimicrobial agent. Potassium (2E,4E)-hexa-2,4-dienoate was used as the antimicrobial agent in composite materials with polyvinyl alcohol and BC powder, as described by Jipa et al. 41 Silver particles have been described as antimicrobial agent in composite materials based on biocellulose membranes 42 and the release and antimicrobial activity of potassium sorbate supported in tapioca starch–glycerol films were also studied. 43
Mechanical tests
The results of the traction resistance and deformation tests of the BC membrane, BC/PHB blend, and BC/PHB blend impregnated with CLO are displayed in Figure 4. The tension upon breakage of the BC film was 83% higher than that of the PHB film. The BC/PHB and BC/PHB + CLO blends had traction resistance values closer to that found for the pure BC membrane. In contrast, the traction resistance value of the pure PHB was 12.62 MPa. The efficiency of the three-dimensional network of microfibrils and the efficient reinforcement for polymers and resins have been described in the literature. 13 The findings show that the BC and BC/PHB films had similar values for tension and deformation upon breakage but higher than those of PHB alone. Moreover, a slight increase in specific deformation was found when the blend was 30% PHB and 70% BC, further demonstrating the greater stretching of this novel material.

Tension maximum (MPa) and deformation (%) of samples of PHB, BC, BC/PHB blend, and blend impregnated with CLO (BC/PHB/CLO).
The incorporation of PHB was also evidenced in the SEM images, as shown above. This behavior may be due to the evidence dispersion of PHB throughout the BC network. The combination of PHB granules and cellulose networks results in a denser fiber structure with enhanced mechanical strength due to the interfacial adhesion and the formation of strong interactions between the BC chains and PHB, as reported by Fernandes et al. 13 According to Barud et al., 15 traction resistance diminishes beginning with a PHB content of 50% blended with the BC membrane, since PHB is a rigid, brittle polymer with weak mechanical properties. The addition of BC to PHB proved to be advantageous, resulting in an improvement in the mechanical properties of this polyester. Biocomposite scaffolds with better characteristics for tissue engineering applications were obtained from PHB and microfibrillated bacterial cellulose composites. The composite scaffold produced displayed better mechanical properties than the pure matrix. 44
X-Ray diffractometry
Figure 5 displays the XRD of the BC, PHB, BC/PHB, and BC/PHB/CLO. As expected, the pure PHB and pure BC films exhibited characteristics of semicrystalline polymers, and this structure was maintained in the BC/PHB blend and BC/PHB/CLO films.
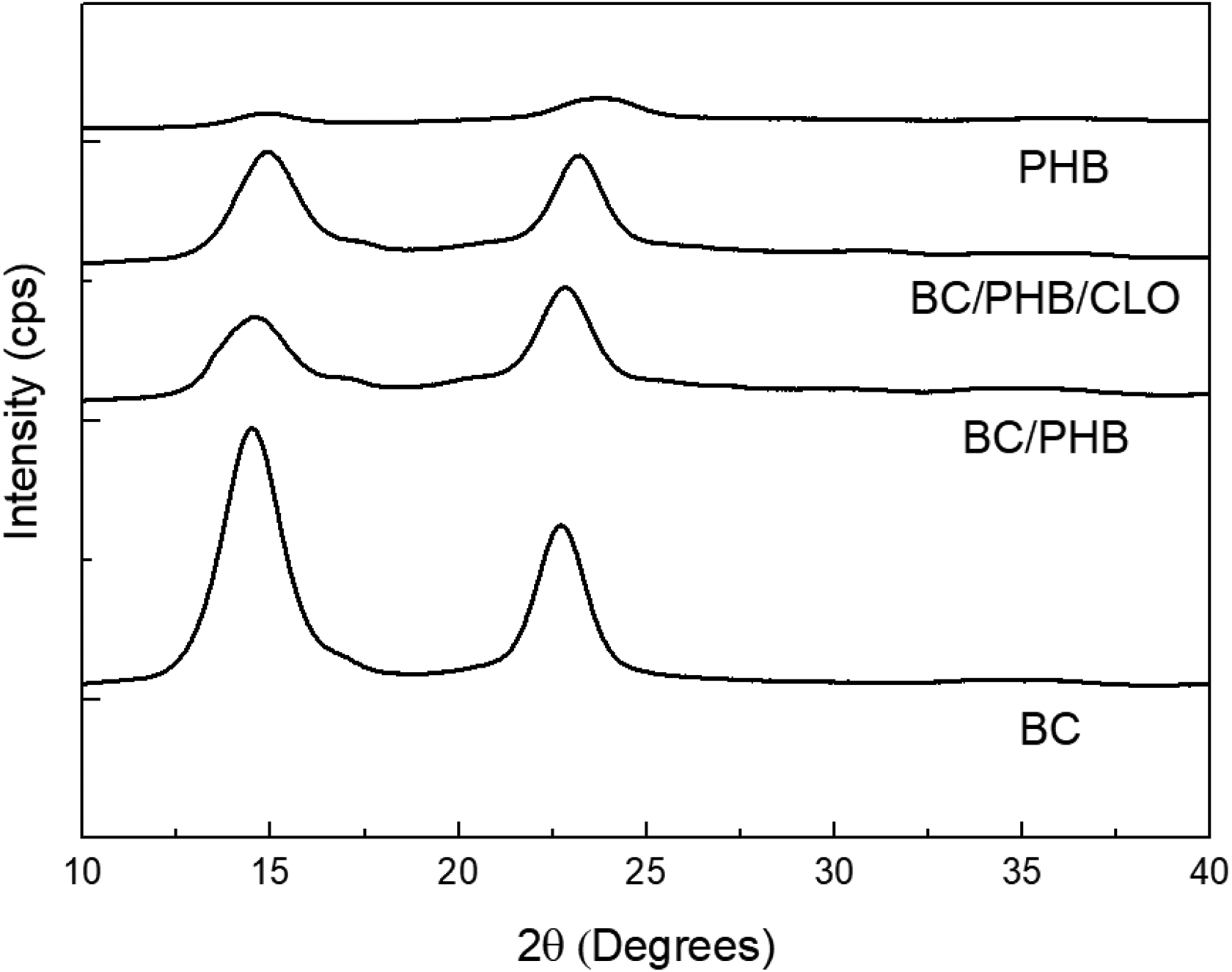
XRD of samples of PHB, BC, BC/PHB blend, and blend impregnated with CLO (BC/PHB/CLO).
The crystallinity index (CI) was calculated, where the BC had a higher percentage of crystallinity compared with the pure PHB and the blend. The CI was 86% for pure BC, 31% for the pure PHB, 50% for the BC/PHB blend, and 55% for the blend containing the essential oil. These results show that the incorporation of PHB to the cellulose matrix exerted a positive effect on the property of the cellulose, diminishing its crystallinity. This change in crystallinity directly affected the mechanical properties of the membrane, reducing the tension and generating a more elastic material in comparison to pure BC.
Thermogravimetric analysis
The thermal stability of the BC and PHB films, BC/PHB blend, and blend impregnated with CLO (BC/PHB/CLO) was investigated through TGA. The results are displayed in Figure 6 and Table 1.

TGA of samples of PHB, BC, BC/PHB blend and blend impregnated with CLO (BC/PHB/CLO).
Thermal decomposition of samples of BC, PHB, BC/PHB blend, and blend impregnated with oil essential oil (BC/PHB/CLO; data obtained from TGA curves).

BC: bacterial cellulose; PHB: poly(3-hydroxybutyrate); CLO: clove essential oil; TGA: thermogravimetric analysis.
The BC film lost mass in two stages: (1) during the evaporation of the water remaining in the film and (2) due to pyrolysis of the polymer. The PHB film lost mass in only one stage (during its thermal decomposition). All mass loss percentages, according to the degradation stages, are described in Table 2.
Mass loss percentages of samples of BC, PHB, BC/PHB blend, and blend impregnated with CLO (BC/PHB/CLO; data obtained from TGA curves).

BC: bacterial cellulose; PHB: poly(3-hydroxybutyrate); CLO: clove essential oil; TGA: thermogravimetric analysis.
For the blends, the degradation of the polymeric material occurred in two distinct stages: one related to the BC and the other related to the PHB. These findings are in agreement with data described by Barud et al., 15 who also found an increase in the degradation temperature of the polymeric blend, indicating a possible increase in the stability of the material due to the crystallization of the PHB among the microfibrils of the BC. An additional degradation stage occurred for the film with the addition of CLO (between 121°C and 188°C), which is due to the oxidative reaction of the major component of the oil (eugenol). 45 Martinez-Sanz et al. 46 showed that no improvement of the mechanical properties of PHBV was observed after BC nanowhiskers addition for nanocomposites from PHBV copolymer (7% and 40% valerate content) and 1–3 wt% BC nanowhiskers obtained by solution casting, but better thermal stability and water and oxygen barrier properties were observed for the nanocomposite with low amount of BC.
FTIR spectroscopy
Figure 7 shows relevant regions of the FTIR spectra. The main bands observed for the pure BC are attributed to the stretching of the OH group (3286 cm−1), CH stretching of alkanes, asymmetrical CH2 stretching (2921 cm−1), and the deformation of C–OH and CH (1399 cm−1).

Spectra of PHB, BC, BC/PHB blend, and blend impregnated with CLO (BC/PHB/CLO).
The main bands observed in the PHB spectra are attributed to C–O (1282 cm−1) and C=O (1850 cm−1) elongations and peaks at 2853 cm−1, referring to the elongation of CH2, and 2926 cm−1 and 1400 cm−1, referring to the elongation of CH3. These results are in agreement with data described by Barud et al. 15 The spectra obtained for the blends basically exhibited the bands observed for the two individual polymers weighted by the relative content. The region between 2000 cm−1 and 3500 cm−1 seems to be affected little by the relative content of the two components, whereas the changes from 1500 cm−1 to 2000 cm−1 seem to be related to the change in the crystallinity of the cellulose determined by the XRD analysis.
A band in the region of 1510 cm−1 is seen for the BC/PHB blend containing CLO. This peak is attributed to C=C bonds of the aromatic group of eugenol, which is the major component in CLO. 47
PCA is a multivariate statistical tool that can be used to complete the infrared spectrum, due to the vibration characteristics of the chemical structure, as it decomposes the multivariate matrix through the linear combination and transforms it into a multidimensional region of space, which are the main components, so there are groupings of samples by its similarity of chemical bonds. 48 Thus, PCA was used to complement the results of the FTIR analysis and demonstrate differences among the polymeric films, blend, and blend with the addition of CLO. The graphs of the scores (Figure 8) show that the first principal component explained 96% of the spectral information and the second principal component explained more than two 2%. Clusters were formed as a function of the formulation of the films, demonstrating chemical differentiation of the matrix of the pure BC and PHB films, BC/PHB blend, and BC/PHB blend impregnated with CLO. Therefore, it can be suggested that the essential oil was incorporated throughout the entire polymeric matrix, which is a good indication that the antimicrobial agent would be present throughout the entire packaging.

Graphic representation of PCA of PHB, BC, BC/PHB blend, and BC/PHB blend impregnated with CLO (BC/PHB/CLO).
When compared while other polymer blends, the BC/PHB with CLO found to have an elasticity degree of 18.9 ± 0.3%, and a decomposition with maximum degradation rate at 364°C, whereas the PHB/PLA with natural limonene blend showed 8.0 ± 0.2% elasticity and degradation rate at 345°C. 49 The bacterial growth inhibition level for the BC/PHB/CLO blend reduced significantly by 3.2 log CFU g−1 after 30 h, while a BC/nisin composite reduced by 3.3 log CFU g−1 after 14 days. 50
Conclusions
BC produced in an alternative medium constitutes an input with high aggregated value not only from the scientific standpoint but also from the environmental, economic, and social standpoint. The BC and PHB polymers demonstrated compatible interaction for the formation of a stable blend under the conditions evaluated in this work. The blend formed presents favorable mechanical and thermal characteristics for use in food packaging. CLO was favorable as an antibacterial agent for future application of blends as an active food packaging and did not compromise the properties of the polymers. In the near future, the combination of natural biodegradable polymers, such as BC and PHB, can furnish new biotechnological products that meet the needs of the world market, which seeks safe, environmentally friendly options.
Footnotes
Acknowledgements
The authors are grateful to the laboratories of Universidade Católica de Pernambuco [Catholic University of Pernambuco], Universidade Federal de Pernambuco [Federal University of Pernambuco], and Instituto Avançado de Tecnologia e Inovação [Advanced Institute of Technology and Innovation], Brazil.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from the Brazilian fostering agencies Fundação de Amparo à Ciência do Estado de Pernambuco (FACEPE [State of Pernambuco Science Assistance Foundation]), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES [Coordination for the Advancement of Higher Education Personnel]—Code 001), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq [National Council for Scientific and Technological Development]), and the Research and Development Program of the Agência Nacional de Energia Elétrica (ANEEL [National Electrical Energy Agency]).
