Abstract
Composites consisting of a polymeric matrix and renewable resources have attracted scientific attention due to the idea of sustainable development. The objective of this study was to blend the polyvinyl alcohol (PVOH) and oil palm ashes (OPA) using solution casting approach. The results revealed that increasing the OPA loading in PVOH film would decrease both tensile strength and elongation at break of the blended films. The morphology of the tensile fractured surface was examined which supported the result obtained from tensile properties. Also, the thermal characteristics of PVOH/OPA films were studied. The glass transition temperature and crystallization temperature were increased when the OPA loading was increased. Thermogravimetric analysis indicated that the thermal stability of PVOH/OPA film was higher than the unfilled PVOH film.
Introduction
The packaging industry has been revolutionized with petrochemical-based plastic such as polyethylene (PE), polypropylene, polystyrene, polyvinyl chloride, and polyethylene terephthalate. The petrochemical-based polymer has been widely used as packaging materials owing to the fact that they are available in huge quantities yet economical and favorable functionality characteristics such as good mechanical strength, low oxygen, and aroma permeability as well as heat seal-ability. 1 Yet most of the packaging plastics end up in the waste stream after single use only. 2 However, withdrawing plastic from packaging would complicate the issues instead of solving them as it will increase in terms of weight and volume of non-plastic packaging. 3 Biodegradability concept that emphasized both user and ecological-friendly attribute has advocated the paradigm shift arises from growing environmental awareness. 1
Polyvinyl alcohol (PVOH) also known as hydrolyzed ethylene-vinyl acetate is a promising example of biodegradable polymer matrix due to its unique properties such as water solubility, low toxicity, and film orientation characteristics 4,5 and the drawn film possesses high strength and modulus. 6 -8 The reason for this fact may account for the hydroxyl groups that produce more effective hydrogen bonding between the adjacent chains and consequently has a profound effect on the mechanical properties. 9 PVOH is also resistant to grease, oil, and solvent meeting the packaging requirement. 10 The PVOH with an average molecular weight as high as 106 could be completely decomposed by soil bacteria, for instance, Pseudomonas as reported by Chen et al. 11 However, the vinyl polymer in which the backbones are joined by the carbon–carbon linkages resulted in lower rate of biodegradability. 12
It is suggested that the incorporation of renewable agriculture waste in PVOH could enhance the biodegradability and cost reduction in conjunction with green environmental policy. There is a massive amount of biomass waste produced by oil palm plantation annually. These ashes are resulted from the on-site processing of empty fruit bunches, oil palm fronds, mesocarp fiber, and palm kernel shell in the boiler fuel to provide steam for electricity generation in oil palm mill plantation. 13 Since agriculture constitutes a much hefty of a solid waste burden than polymer, compositing seems the right approach to these. 14 To the best of our knowledge, there is no study involving the oil palm ashes (OPAs) blended with the PVOH for the production of biodegradable polymeric materials. Besides, disintegration of biodegradable PVOH/OPA packaging material used as agriculture mulch may decompose and becomes a fertilizer and soil conditioner, which will facilitate better crop yield while allowing us to preserve the dwindling petroleum resources. 1
In this study, our efforts in the preparation of biodegradable polymer films are focused on the varying compositions of OPAs and PVOH, followed by examining their effects on the tensile properties and thermal characteristics. Also, the characterization of PVOH/OPA films was studied by Fourier transform infrared (FTIR) spectroscopy and scanning electron microscopy techniques.
Experimental
Materials
The polymeric component of this study was PVOH, 99+% hydrolyzed with an average molecular weight of 89,000–98,000 g mol−1 and a density of 1.269 g cm−3, was purchased from Sigma–Aldrich (M) Sdn. Bhd (Malaysia) with product number and batch number of 341584 and #21897 LJ, respectively. The Chemical Abstract Service registry number was 9002-89-5. The agricultural waste in this study was OPAs, which was collected from United Oil Palm Mill Industry located in Nibong Tebal, Penang, Malaysia. The collected OPA particles were sieved with a 75-µm mesh to separate the unburned residues and larger particles such as sand and pebbles as shown in Figure 1, followed by oven-drying at 80°C for 24 h to expel moisture.

Process of raw OPA preparation.
Film preparation
Calculated amount of PVOH/OPA aqueous solution (10%, w/v) was prepared by dissolving PVOH/OPA in 100 mL deionized water, as presented in Table 1, the PVOH was first heated on a hot plate stirrer with magnetic bar with constant stirring at a temperature of 80–90°C until the PVOH was fully dissolved. The OPAs were then added, and stirring was continued for another 2 h. The homogenous PVOH/OPA aqueous solution was poured onto the clean glass plate, which was placed on a leveled flat surface, and the mixture was dried overnight to constant weight. The unfilled PVOH and PVOH/OPA cast films were then kept in a sealed PE bag and stored in a desiccator for 2 days prior to any characterization.
Formulation of PVA/OPA film preparations.
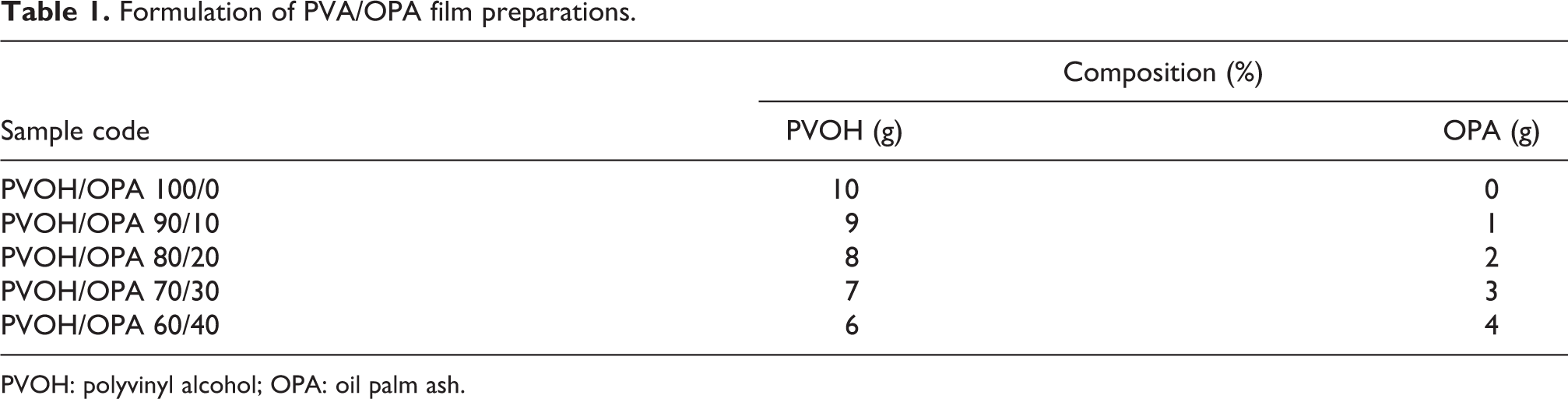
PVOH: polyvinyl alcohol; OPA: oil palm ash.
Evaluation of tensile properties
For tensile test, five dumbbell shaped specimens (average thickness = 0.10 mm) were cut from the thin films with a dumbbell cutter (model: Leader Technology Scientific (M) Sdn. Bhd, Malaysia) and tensile test was conducted according to the ASTM D882 standard using a universal tensile testing machine (model: Tinius Olsen, Salfords, UK) at a crosshead speed of 20 mm min−1. The data for tensile strength and elongation at break were evaluated from stress–strain determination, and average values of five repeated tests were taken for each composition.
FTIR spectroscopy
FTIR was used to obtain some qualitative information about the functional groups and chemical characteristics of the PVOH/OPA films. FTIR spectra were obtained and recorded using a Perkin Elmer Spectrometer (model RX1, Norwalk, CT, USA) in the range of 550–4000 cm−1 at 4 cm−1 resolution. For each spectrum, 16 scans were co-added.
Differential scanning calorimetry
The glass transition temperature (T g), melting point (T m), and crystallization temperature (T c) of films were carried out using differential scanning calorimeter (DSC) (Mettler Toledo; model DSC823e, Schwerzenbach, Switzerland). Slices of PVOH/OPA films with a total weight of 7–8 mg was weighted and hermetically sealed in an aluminum pan. An empty aluminum pan was used as the reference cell. The crystallization and melting behavior of PVOH/OPA films were determined at a rate of 10°C min−1 from 25°C to 250°C under nitrogen atmosphere and at a flow rate of 50 mL min−1.
Thermogravimetric analysis
The thermal degradation analysis of PVOH/OPA films was tested by thermogravimetric analysis (TGA) (Mettler Toldedo; model TGA/SDTA851e, Schwerzenbach, Switzerland). About 4–5 mg of sample was weighted and sealed in an aluminum pan. The weight loss was measured as a function of temperature, which was heated from 25°C to 700°C. A heating rate of 10°C min−1 was used under nitrogen atmosphere and at a flow rate of 50 mL min−1. The weight change of sample as a function of temperature was recorded. The temperature at a certain weight loss of films and percentage of residue were determined.
Morphological studies
Tensile fractured surface of unfilled PVOH and PVOH/OPA films were scanned with a scanning electron microscopy (FESEM) (JEOL: model JSM 6701-F, Akishima, Japan) was conducted at an accelerating rate of 2.0 kV. The tensile fractured films were mounted on aluminum stubs and sputter coated with a thin layer of platinum to avoid electrostatic charging and poor resolution during examination. The SEM images observed from tensile fractured surfaces were used to support the changes in tensile properties.
Results and discussion
Morphological studies
The biodegradable PVOH/OPA-blended films were successfully produced by solution casting method. Figure 2(a) depicts the morphology of the unfilled PVOH film under SEM observation. The unfilled PVOH film was clear and appeared smooth, but the phase structure was changed when the OPA filler was incorporated and tend to become rougher upon increasing of OPA loading. This was attributed to the coarsely dispersed (as highlighted with white circle) of OPA filler in PVOH matrix causing the surface of PVOH/OPA films appeared rougher when compared to unfilled PVOH film.

Surface morphology of PVOH/OPA (a) 100/0, (b) 80/20, and (c) 60/40 films.
Tensile properties
The tensile strength and elongation at break of the unfilled PVOH film and PVOH/OPA films with respect to varying loading of OPA filler are shown in Figures 3 and 4, respectively. Both tensile strength and elongation at break of PVOH/OPA films decreased when the OPA filler loading was increased. The data of tensile strength showed an unfilled PVOH exhibit the highest tensile strength (57.87 MPa), whereas the PVOH/OPA 60/40 films possess the lowest tensile strength (24.66 MPa). Elongation at break of PVOH/OPA films showed a similar trend as tensile strength results with a significant reduction from 137.7% (unfilled PVOH film) to 14.54%. The reason for reduction in both tensile strength and elongation at break may be due to the agglomeration of the OPA filler. With the incorporation of the OPA filler, OPA-OPA interaction is more pronounced than PVOH-OPA interaction, thereby suggesting the phase separation arising from the coalesced and coarsely dispersed OPA and consequently reduced the compatibility of PVOH and OPA. Another word of saying, the OPA filler could act as a stress concentrator and initiates the crack when external forces are applied to it. This can be proved by the morphological study on the tensile fractured surface of PVOH/OPA film later.

The dependence of the tensile strength on the varying compositions of the PVOH/OPA films.
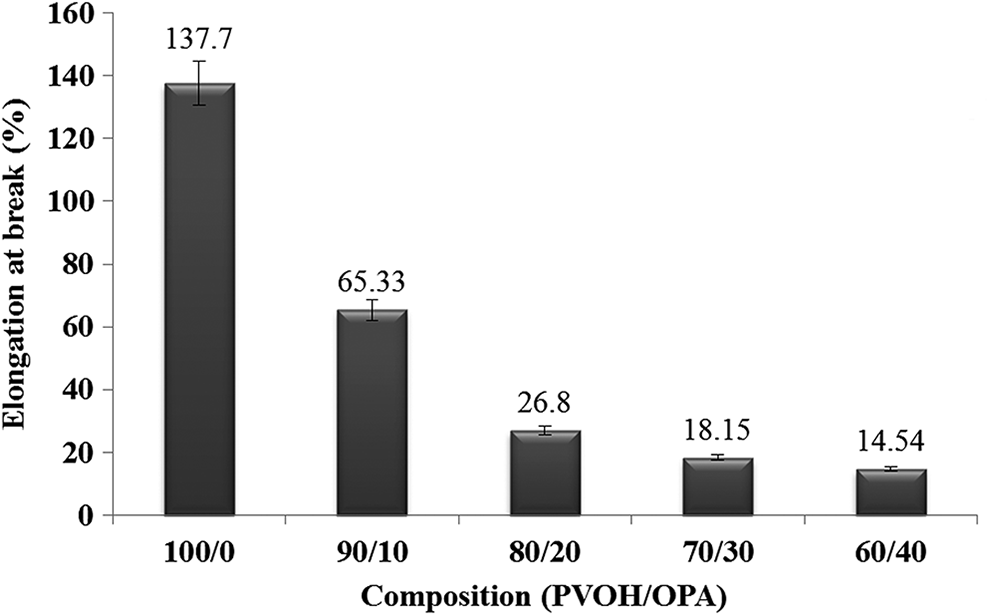
The dependence of the elongation at break on the varying compositions of the PVOH/OPA films.
To evaluate the filler distribution and fractured mode of PVOH/OPA films after the tensile test, the tensile fractured surface of the unfilled PVOH film, PVOH/OPA 80/20, and PVOH/OPA 60/40 films were observed using SEM, as shown in Figure 5. The fractured surface of the unfilled PVOH was rougher with more tearing lines and angular cracking. This explained that more forces are required to fracture the unfilled PVOH specimen. It is not surprising to note that the agglomeration of OPA was clearly observed on the fractured surface of PVOH/OPA 80/20 (Figure 5(b)) and PVOH/OPA 60/40 specimens (Figure 5(c)). Besides, the micro-voids could be clearly observed in the PVOH/OPA films, especially at higher OPA loading (40 wt%) due to detachment of OPA fillers and less effective cross-sectional area of OPA in the PVOH matrix when external forces exerted on the specimen. This indicated the deterioration of tensile strength and elongation at break of PVOH/OPA films at higher OPA loading.

Tensile fracture surface of PVOH/OPA (a) 100/0, (b) 80/20, and (c) 60/40 films.
Infrared spectroscopy
Figure 6 shows the FTIR spectra of unfilled PVOH films and PVOH/OPA films at varying OPA content. No absorption band was detected at 3600 cm−1. This indicated that there are no free hydroxyl groups 15 that exist in unfilled PVA film and PVA/OPA blended film; however, the broad hydroxyl –OH group was observed at around 3257–3264 cm−1. This may be due to the hydroxyl groups of PVOH engaged with the inter- and intra-molecular hydrogen bonding. Similar finding was reported by Pu-You et al. 16 The hydroxyl band became less broad and shifted to a higher value since the introduction of the OPA filler suggest the interruption of PVOH hydrogen bonding by OPA filler. Besides, there absorption band at 1645–1655 cm−1 was observed, indicating the existence of the carbonyl group. The existence of carbonyl group is mostly due to the incomplete removal of acetate groups during hydrolysis of PVOH. 17 Furthermore, –C–O, –C–O–C, and –C–O–Si medium peak was observed around 1011–1079 cm−1. The appearance of absorption peak at 1079 cm−1 in the unfilled PVOH film is the C–O absorption peak of PVOH itself. It can be seen that the absorption peak became broader and shifting to lower frequency when the OPA loading was increased. This might be due to the interaction of OPA with PVOH.
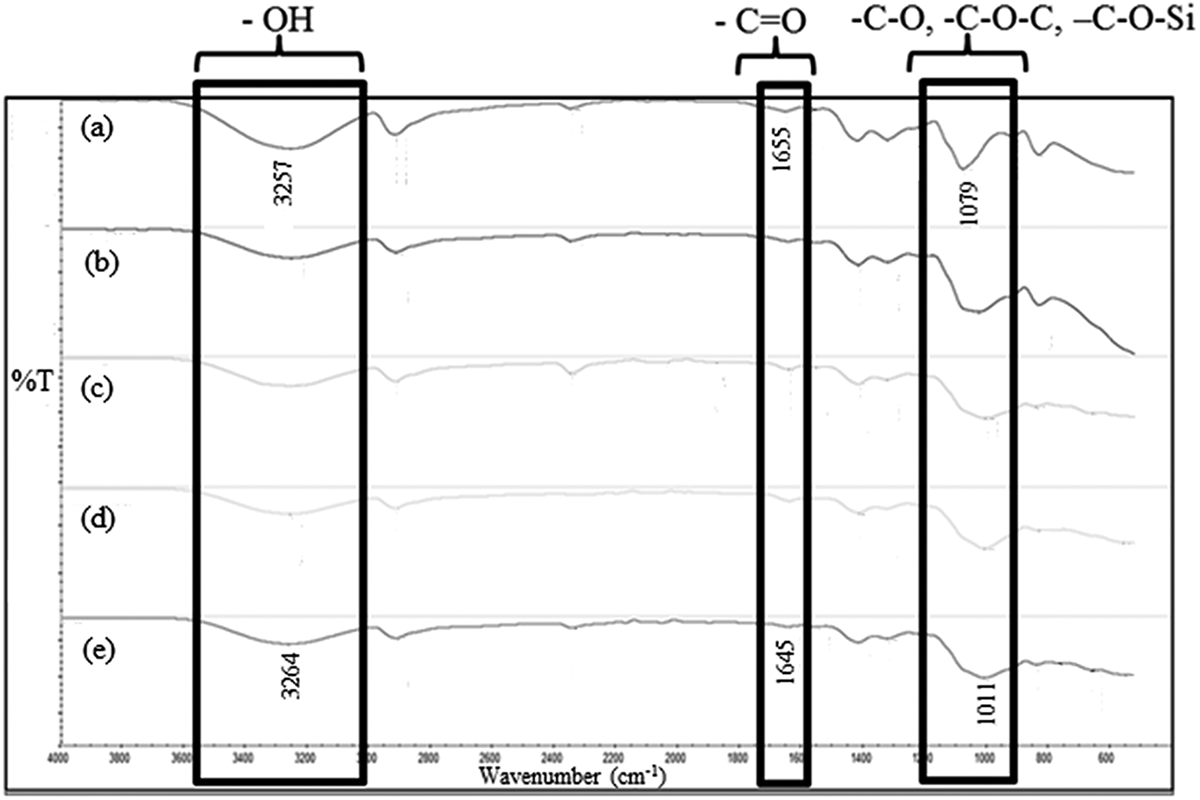
FTIR spectra for PVOH/OPA film (a) PVOH/OPA 100/0, (b) PVOH/OPA 90/10, (c) PVOH/OPA 80/20, (d) PVOH/OPA 70/30, and (e) PVOH/OPA 60/40.
DSC of unfilled PVOH film and PVOH/OPA films
Table 2 summarizes the thermal behavior of PVOH/OPA film with different loading of OPA filler. The unfilled PVOH film shows the T g at 74.78°C. With the incorporation of the OPA filler, the T g takes place at a higher temperature. The T g showed an increased trend as the OPA filler loading increased. This plausibly implies that decrease of conformational mobility of the PVOH polymeric chain due to the rigidity of OPA filler. Another reason may be attributed to the agglomeration and non-well dispersion of the OPA filler as shown in Figure 5. This reflects the significant increase of the T g from 74.78°C (unfilled PVOH film) to 89.08°C (PVOH/OPA 60/40 film). T g associated with the amorphous/disordered fraction of the films while T m referred to the crystalline/order region of the film. T m was reduced significantly and the peak tends to broader when 10 wt% of the OPA was added, as shown in Figure 7. However, beyond the 10 wt% of OPA, the T m of PVOH/OPA film was increased as increasing of OPA loading. This implies that the OPA filler do affect the T m of the PVOH film. T c increased which indicated that the OPA could act as a nucleating agent. The increment of T g and T c indicated that OPA played a role in reducing the mobility of polymer chains and promoting crystallization. It was interesting to note that both T m and T c were reduced when 10 wt% of the OPA was added. This may be attributed to the physical hindrance of 10 wt% of OPA affecting the crystallization of PVOH polymeric chains.
Thermal behavior of PVOH/OPA films.

PVOH: polyvinyl alcohol; OPA: oil palm ash; T g: glass transition temperature; T m: melting temperature; T c: crystallization temperature.
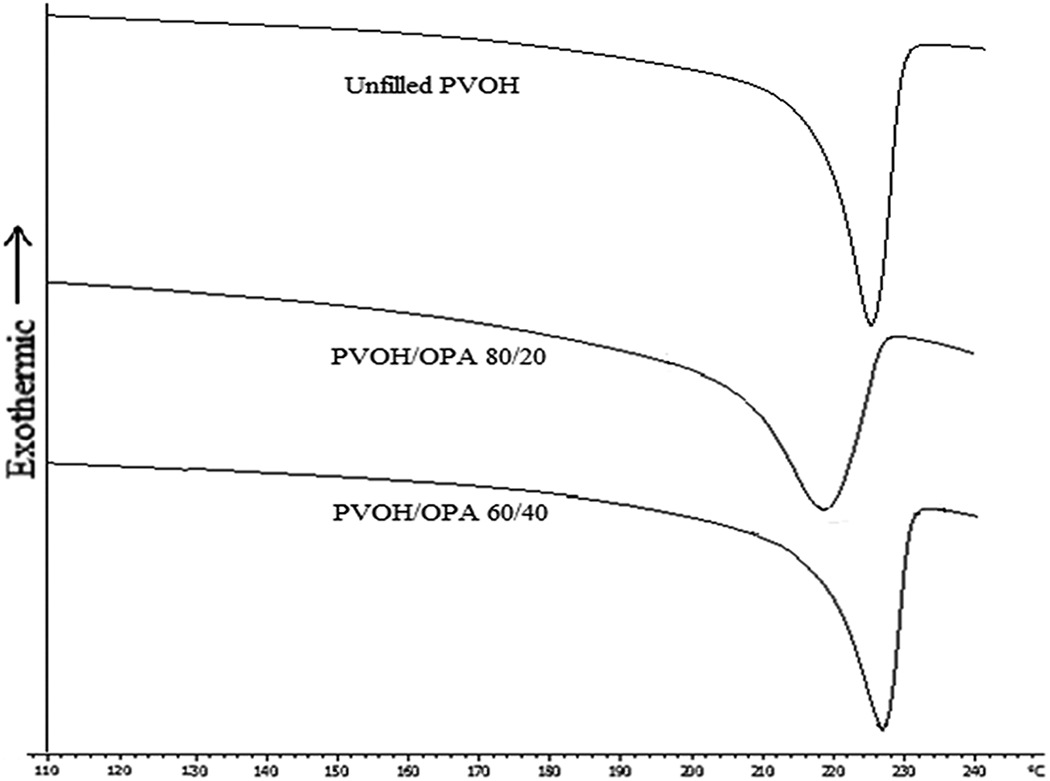
DSC melting thermograms of unfilled PVOH, PVOH/OPA 80/20, and PVOH/OPA 60/40 film.
TGA of unfilled PVOH film and PVOH/OPA film
In this part, the thermal decomposition temperature of the unfilled PVOH film and PVOH/OPA 60/40 film were measured by a thermogravimeter. As shown in Figure 8, the thermogravimetric curves of the unfilled PVOH film and PVOH/OPA 60/40 film generally shows three distinct mass loss regions. The first decomposition region is attributed to loosely bound water, and the weight loss is due to moisture vaporization of the adsorbed water by the film itself. The second decomposition region is described as the main decomposition of the PVOH matrix and the later decomposition region is due to the carbonization of organic matters. Table 3 summarizes the decomposition temperature of the the unfilled PVOH film and PVOH/OPA 60/40 film at 10%, 40%, and 60% weight loss. Ironically, the thermal decomposition temperature was increased with the incorporation of the OPA filler. The reason for this fact may account for the mineralogical composition that contained in OPA, which could act as a thermal barrier and retard the thermal motion. Besides, the PVOH/OPA 60/40 film showed higher residue percentage than the unfilled PVOH film. Undeniably, the OPA-filled PVOH film shows a better thermal stability than unfilled PVOH as a result of thermal resistance of OPA and development of blending toward high energy stability of the PVOH blend film.

Thermogravimetric analysis curve and derivatives of thermogravimetric curves of unfilled PVOH film and PVOH/OPA 60/40 film. PVOH: polyvinyl alcohol; OPA: oil palm ash.
Thermal decomposition temperature of unfilled PVOH film and PVOH/OPA 60/40 film.

PVOH: polyvinyl alcohol; OPA: oil palm ash.
Conclusions
In this study, the OPA was successfully blended with the PVOH film by solution casting method in the presence of water solely. The tensile properties depended on the OPA content, therefore the PVOH film prepared with the highest OPA content resulted in lower tensile strength and elongation at break. This was inconsistent with the SEM morphology, which shows agglomeration of OPA. However, incorporation of OPA increased the T g and T c of the PVOH film. Moreover, PVOH/OPA 60/40 film possesses higher thermal stability as well as a higher residue percentage compared to unfilled PVOH film.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the University Tunku Abdul Rahman Research Fund (Project no. IPSR/RMC/UTARRF/2015-C2/O03).
