Abstract
Polypropylene (PP) burns very rapidly due to its wholly aliphatic hydrocarbon structure. Studies have shown that the rapid decomposition and flammability of PP are very high when compared with wood and other cellulosic materials. This has limited the application of PP and necessitated the need to inculcate flame-retardant (FR) behaviour to PP to further widen its scope of application especially in areas where FR is paramount. With the advent of nanotechnology, increasing inclusion of FR nanofillers in PP has demonstrated propensity to repressing critical flammability parameters such as heat release rate, peak of heat release rate, rate of carbon monoxide production, smoke production rate and total mass loss rate while simultaneously increasing limiting oxygen index, time of ignition and total peak of heat release rate. This efficiently represses PP flammability and provides greater opportunity to minimize loss and risks to life in actual fire scenario through creation of equal layers of carbonaceous char in the condensed phase capable of suppressing the thermal decomposition caused by oxygen and heat to PP matrix, thus effectively cutting-off the fire path. This article reviews recent developments in FR of PP composites, nanocomposites and nano-biocomposites. Market structures are also presented.
Introduction
The inclusion of nanofillers into polypropylene (PP) has created PP nanocomposites. The nanosize, high aspect ratio and large surface area of nanoparticles have effectively improved the mechanical, thermal, morphological and fire-resisting behaviour of PP nanocomposites in comparison with pristine PP. 1 Two factors which have played significant roles in fire mechanism of PP nanocomposites during burning are protective barrier formation and increment in melting viscosity. 2 Generally, polymer nanocomposites (PNCs) exhibit two fire-retardant mechanisms via formation of a protective layer during burning and melt viscosity increment. The uniform distribution of nanoparticles in the PP matrix during processing is vital in attaining effective fire-repressing behaviour and effective suppression of dripping occurring during PP combustion is also essential in achieving flame retardant (FR) in PP nanocomposites. Despite the type of nanofiller utilized during formation of PP nanocomposite, the formation of a protective layering is essential to attaining FR during burning of these materials.
The ability of nanofillers to uniformly homogenize in the PP matrix during compounding is a vital factor for inculcating efficient fire suppressing attributes since it is an important parameter for improved structural stability for these materials at extreme temperatures despite the achievement of FR. This is because dripping flaw during burning is effectively suppressed. Generally, parameters influencing polymer FR include oxygen indexing (OI), heat release rate (HRR), flame suppression, flame propagation, flash ignition temperature and so on.
The type of smoke evolved during PP burning is affected by factors such as oxidation reaction, rate of combustion or pyrolysis and chemical constituents. 3 Investigation into the type of smoke evolved by PP composites during combustion is attained by gravimetric or optical procedures while the toxicity is attained by analytical and biological tests. 4
Some polymers tend to burn very slowly, some faster; others give off more smoke, some less; and others melt and flow on application of heat, while some undergo charring. PP pyrolysis can be reduced through usage of suitable additives, though some of these inclusions also generate smoke and impact negatively on the polymer behaviour. 5 Intumescent systems are composed of three fundamental factors, namely acid origin, a char-initiator and a blowing propagator. 6 FR in PP nanocomposites has been achieved through laboratory investigations of PP nanocomposites fire behaviour; fire-repressing agents, mechanism of operation and attainment of FR synergies in PP nanocomposites. The versatility of applications of PP nanocomposites as demonstrated in Figure 1 is very broad. Thus, attainment of a superior FR behaviour in these materials is essential to enlarging their scope of usage. Figure 2 elucidates some areas of applications of FR PP nanocomposites.
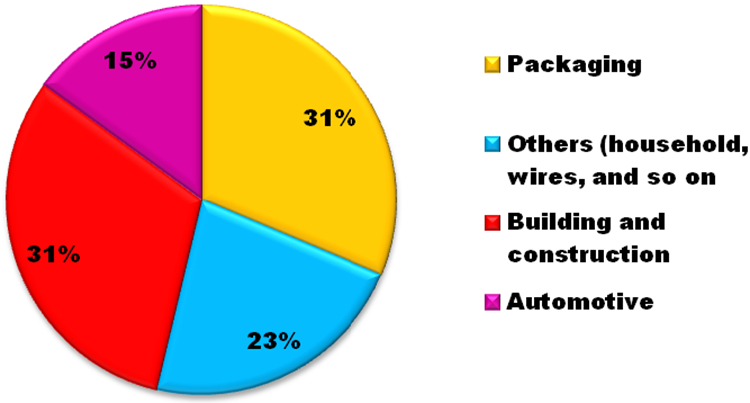
Scope of application of PP nanocomposites.
Some specific areas of applications of FR PP nanocomposites.
Studies have revealed that PP FR has been attained via inclusion of halogen atoms; aluminium trihydrate (ATH), magnesium hydroxide (MH), intumescent systems and other mechanisms. 7 Thus, the aim of this review is to elucidate comprehensive insight into recently attained eco-benign FR routes in PP composites, nanocomposites and nano-biocomposites while highlighting novel application areas and future market prospects.
Pyrolysis of PP
Due to its wholly aliphatic hydrocarbon structure, PP exhibits rapid combustion. Two main agents are involved in the combustion route of PP: (i) agent of oxidation (combustive agent) and (ii) single agent/agents of reduction (combustibles). Oxygen in the air is the main combustive agent or the oxidizing agent. 8 The combustion process of PP is initiated by a heat source which escalates the temperature to the extent of causing scissions in the polymer chain. Fuels in form of combustible gaseous volatiles are then emitted into the air by volatile part of the polymer through diffusion. 9 When the pyrolysis activation energy temperature is attained or the auto-ignition temperature is achieved, the gaseous combination ignites thereby evolving heat. Another fuel ignition source is the flashpoint which is a lower temperature at which the fuel reacts with an external heat origin, spark, intense energy or fire. 10 The burning time of PP composites and nanocomposites is a function of the degree of heat released during fuel combustion. When the level of heat evolved attains a specific peak, fresh reactions of decomposition are ignited in the solid-state resulting in formation of more combustibles. 11 This cycle of burning is thus sustained in this pattern and referred to as a fire triangle as shown in Figure 3.

Principles of PP pyrolysis.
Stages of PP pyrolysis
The stages in PP actual fire scenario are elucidated in Figure 4 in PP temperature versus time curve depicting ignition as the origin of fire initiation.
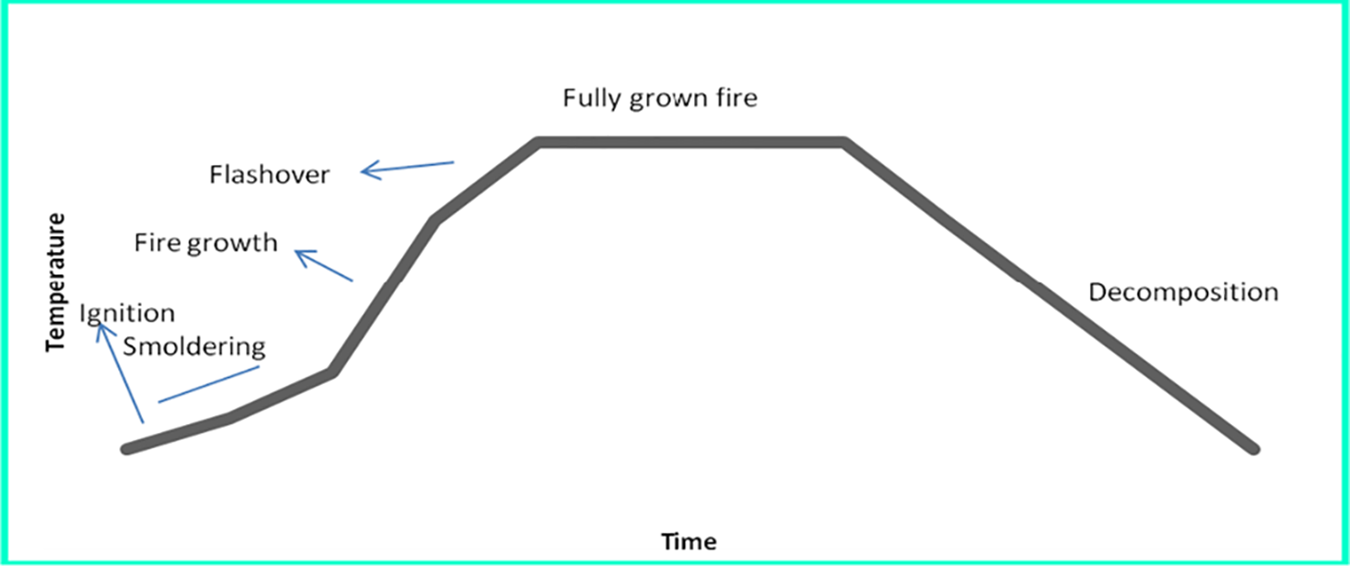
PP temperatures versus time curve (stages of PP fire pyrolysis).
Here, a heat source combines with fuel in presence of oxygen to start fire. This initiates the growth of a degradation process that is flammable releasing products, which, in turn, combine with oxygen from the air to form flame and heat. 12 The flow of volatile and flammable degradation products is sustained through the transfer of heat back to the fuel surface. Fire being a dynamic process maintains equilibrium between the heat transfer rate to the fuel, loss of heat from fuel and degradation products evolvement. 13 Thus, the fire may extinguish or propagate depending on existing conditions.
Fire growth hazards
There are three basic fire hazards exhibited by fire growth. These hazards are heat; smoke and toxic gases, mainly carbon monoxide (CO) and others. 14 This process is intricate and incorporates many chemical reactions and transport procedures in the condensed, interfacial and gaseous phases. An external heat source can be used to generate heat through thermal energy which could be by convection, conduction or radiation via chemical reaction inculcated in the material through fermentation or oxidation or any similar appropriate route. This could also be through the initiation of exothermic combustion reaction. 15
Generally, for polymers, the energy required for combustion initiation is not equal and is determined by the physical properties of the polymer. 16 When heat is applied to PP, it softens, undergoes melting and subsequently followed by dripping due to its semi-crystalline structure. 17 The propensity of PP to store energy is a function of its enthalpy of fusion, heat storage ability and rate of crystallinity. Hence, temperature increase in PP is a function of heat flow; exothermic reaction induced thermal conductivity and specific heat. 18 This is unlike amorphous polymers with nil melting point and heating results in polymer decomposition. The dissociation of the covalent bond due to thermal decomposition of PP is an endothermic reaction requiring energy-input and the energy supplied to the system has to be higher than the energy covalently bonding the atoms (200–400 kJ/mol for many C–C polymers). 19 The mechanism of decomposition is mainly a function of availability or oxygen absence in the solid and gaseous phase and on the weakest bonds. Thermal decomposition is the aftermath combination effects of heat and oxygen. 20
Pyrolysis induces chain scissions in the polymer backbone chain, which, in turn, initiates non-oxidizing thermal degradation. This is determined by various parameters such as catalyst residues, presence of oxygen atoms in the backbone chain, chemically induced defects in the polymer backbone chains and the presence of weak bonds in the backbone chain. There are two routes by which chain scission can occur; through free radical formation which propagates the chain reaction occurring in both oxidizing and non-oxidizing conditions 21 ; and by transfer of atoms of hydrogen and the creation of double-stabilized molecules where one of the molecules contains a carbon–carbon double bond that is reactive. In thermally oxidizing conditions, the polymer undergoes reaction with oxygen present in air to form varying low-molecular weight products like aldehydes, carboxylic acids, ketones, alcohol and so on. In PP, this degradation releases species such as H* and OH*. 22
PP Fire retardancy
The main aim of PP flame suppression systems is retardation of PP combustion path as aforementioned. There are two major paths of fire retardation in PP viz-a-viz chemical and physical routes. The chemical route occurs in the condensed or gaseous state. 23 The physical route occurs via cooling through creation of a protective barrier or dilution of fuel. 24 The FRs function by intruding into various procedures occurring in PP combustion like thermal degradation propagation, ignition, pyrolysis and heating. 25
Physical path of PP pyrolysis
FR systems in this route function via endothermically induced decomposition with reduced temperature by heat elimination. Here, the reaction medium is cooled below the temperature of PP combustion. FR systems operating in this path include MH and ATH. 26 These FRs commence their flame suppression action through the liberation of water vapour at about 200–300°C, respectively. This reaction is endothermic in nature and referred to as ‘heat-sink’. The decomposition of the FRs results in liberation of gases which are inert in nature such as NH3, CO2 and H2O, which, in turn, dilutes the combination of combustible gases, thereby inhibiting reagent concentration and ignition possibility. 27
Also, certain FRs create a protective barrier or gaseous blanket between PP as the solid matrix phase where thermal degradation occurs and the gaseous layer where combustion takes place. This protective blanket or inhibiting barrier restricts the transfer of combustible oxygen and volatile gases, thereby repressing the quantity of gases liable to induce decomposition of PP. 28 In addition, fuel gases can be physically removed from oxygen, thereby inhibiting the combustion process. 29
Chemical route
Chemical route of FR process in PP occurs either in the condensed or in the gaseous state. In this phase, the combustion mechanism of free radicals for instance in halogen FRs is stopped through the co-option of halogen-free radical releasing FRs containing the CL* and Br* in the gaseous phase. 30 These free radicals have the propensity to chemically combine with species like H* and OH*, which are highly very reactive, thereby forming molecules that are inert or less reactive. This action modifies the combustion route resulting in a significant reduction in the exothermic reaction and a corresponding reduction in temperature and fuel evolved.
There are basically two types of FR initiated chemical reaction in the condensed state, namely rupturing of the polymer chain resulting in dripping of PP and migration from the fire zone. 31 The flame suppressant can also initiate the creation of expanded carbonaceous char layer or vitreous blanket or protective inhibiting barrier at the surface of PP by chemical modification of the decomposing PP chain.
Fundamentally, flame resistance was incorporated into polymers through the following methods; application of flame resistance polymers composed of halogen atoms like polyvinyl chloride and fluoropolymers, 32 or co-opting flame suppressing agents like brominated organic compounds, MH and ATH or inclusion of intumescent systems to the polymers to repress flammability. The flame mechanism of PP nanocomposites is attributed to the formation of a high-performance carbonaceous char on the surface during combustion, which insulates the underlying material, thereby slowing down the mass loss rate (MLR) of the decomposition products. 33
Two classifications of FRs are possible, namely additive and reactive FR. FR are said to be additive when they are co-opted during the process of polymer transformation and initially do not commence fire retardation until attainment of higher temperatures or when fire starts. Such fire retardants include organic compounds such as macromolecules, hybrids, nanoclays and some other nanofiller. Reactive FRs are normally incorporated into the PP chain as monomers or polymer precursors during processing or by chemically grafting into polymer backbone chain during post-reaction procedure. 34
Bench flammability tests
PP flammability is assessed through HRR, ignitability and rate of fire spread. Relative to the end application, these flammability parameters are determined using laboratory bench test methods. There are various small-, medium- or large-scale flammability test used industrially or in research laboratories for flammability test of materials during product development or testing flammability for already produced materials. PP nanocomposite fire repression abilities has been attained through inclusion of FRs in the polymer chains via graft polymerization or by compounding with a polymer having high thermal and FR behaviour. 35 PP nanocomposites intumescent flame suppression mechanism occurs via formation of many foamy cellular oriented chars on PP surface inhibiting the polymer from combusting. 36 The gaseous combustion products emitted to the fire zone are minimized by the foamy char via efficient cutting-off of heat and oxygen from the pyrolysis zone. 37
Fire suppression additives like intumescent agents attain fire repression through their ability to create a stabilized thermal residue under the influence of external heat flow, which inhibits the heat and mass transfer of oxygen from the flame to the polymer and flammable gases from the polymer to the flame. Metal hydroxide fillers are able to function as flame suppressors because of their endothermic decomposition which emit water that cools the flaming zone and due to their inherent inert properties capable of diluting products of pyrolysis. 38 The metal oxides formed on the substrate after degradation, in addition with the charring products are capable of insulating the material from the heat origin and enhance smoke repression. 39 Intumescent flame retardants (IFRs) have been used in improving the flame suppression properties of PP recently due to their halogen-free constituents, environmental friendliness, low smoke emission and null dripping ability.
During burning, a multicellular inflated foamy char is generated by IFR system inhibiting the heat and mass transfer from the flame to matrix polymer, thereby suppressing flammability of PP nanocomposites. Usually, the multicellular foamy char is incomplete and not thermally stable, which results in a weak flame resistance. This restricts the commercial application of IFRs, thereby arousing interests in co-option of synergistic agents to IFR system to efficiently enhance flame retardancy and better opportunity of minimizing loss and hazards in actual fire situation. The mechanism of IFR has been attributed to the creation of a foam multicellular char on the surface of the polymer, thereby inhibiting the polymer from combustion. The char foam acts as an effective barrier against the movement of heat and oxygen, thereby reducing the diffusion of gaseous pyrolysis products to the combustion zone. 40
In a bid to improve the intumescent flame retardancy of PP, alumina silicates including clays has been applied as synergistic IFRs. 41 Halogen-free IFRs are preferred because of their low toxicity, low smoke emission, nil corrosive gas emission and nil dripping during fire. 42 The utilization of halogenated fire suppressive agents has been limited by environmental regulators because of their lack of environmental friendliness such as generation of toxic gases and high levels of smoke. This has triggered the need for eco-friendly replacement for IFRs.
Cone calorimeter (CCT) is a standard test method using an oxygen consumption method applied in the determination of flammability parameters of PP. 43 Here, PP composite test samples burn in ambient air conditions by an external heat flux (0–100 kW/m2) induced by a spark ignition origin. The CCT is one of the most effective techniques for determining the flammability characteristics of polymers and their composites. 44 CCT provides data on time of ignition (TOI) (s), HRR (kW/m2), peak of heat release rate (PHRR) (kW/m2), effective heat of combustion (EHC) (kJ/g), average EHC (kJ/g), specific extinction area, smoke production rate (SPR), CO yield, total heat evolved (THE), total heat (kW/m2) and total MLR (g/s/m2). 45
CCT is a modern device used in studying the fire behaviour of small samples of various materials in a condensed phase. The apparatus enables the samples to be exposed to various heat fluxes across its surface. The name CCT originated from the conical shape of the radiant heater which almost forms a homogeneous heat flux over the surface of the sample under scrutiny. It is the main bench instrument in the area of fire testing. The CCT operates on the principle that the maximum heat of combustion of any organic material is directly related to the quantity of oxygen required for combustion. 46
Time to ignition (TTI), measured in seconds (s), in the CCT with a known heat flux is a critical parameter. Ignition is attained when an external heat flux; Q more than a critical heat flux is applied for a given duration of time, tign. On application of heat flux, conduction of heat into the substrate occurs and the rate of this conduction determines TTI. The CCT determines the TRP as a function of thickness of substrate, heat flux applied, effects of insulation and so on. 46 These values may vary from those determined through calculation using the aforementioned parameters determined using the CCT.
The EHC in kW/m2 is the average chemical heat of combustion determined in CCT. It is the heat of combustion expected in a fire where there is incomplete combustion and a function of irradiance level and time corresponding to the heat released from the volatile portion during solid matter combustion. EHC can be calculated using the equation: EHC = HRR/MLR. 47 EHC is function of the fuel chemistry, the fire mechanism, ventilation rate and the combustion efficiency. 48 The CO (g/g) and CO2 (g/g) production process is taken as a two-step reaction. 49 CO and other gaseous volatiles are produced during the first-step pyrolysis reaction which can be expressed as Arrhenius law. 50 The second step is the oxidation reaction of CO if enough oxygen is available. HRR, the SPR and the rate of toxic gas release are considered most critical fire risk and hazard. 50 Others are smoke obscuration, flammability of generated volatiles and ease of extinction. It is well known that HRR causes rapid ignition and flame propagation. 51
The MLR is determined using ṁ = (Qx + Qy − Qz)/Zv, where Qx = emitted heat flux from flame, Qy = external heat flux and Qz = loss in heat flux. This implies that MLR is a function of applied heat flux depending on flame heat flux, loss of heat flux and gasification latent heat, Zv. 52 Thus, for pristine PP and composites undergoing self-combustion in an open environment lacking thermal feeding from the environment, the heat flux is nil and MLR is determined by the difference of flame heat flux minus loss of heat flux and divided by the gasification latent heat.
Thus, for PP and composites undergoing combustion in the presence of another burning substrate or subjected to reradiation from heat trapped inside the enclosure in which the substrate is combusting, the external heat flux is more than zero and MLR is greater than aforementioned equation. Hence, materials combusting in enclosures will have the propensity to burn more rapidly than those in open environment. Thus, MLR determines HRR, SER and rate of gas evolvement and is vital to fire hazards posed by burning materials.
The HRR (kW/m2) is the HRR per unit surface area, Qs given as function of MLR, ṁ, combustion heat, ΔHc and the efficiency of flame combustion, fc. 53 Hence, the HRR per unit surface area is determined by QS = ṁ.fc.ΔHc, where, ṁ is the MLR as given above, fc is combustion efficiency and ΔHc is combustion heat. The HRR is a function of two substrate critical parameters, notably the combustion heat divided by the gasification latent heat, ΔHc/Zv. 54 This ratio, called the ratio of combustion, assists in materials ranking according to their combustion behaviour and describes their overall burning behaviour. 55 This characteristic is experimentally determined using the CCT. The HRR is one of the most critical fire parameters since it determines fire growth rate (FGR), heat growth and quantity of smoke and toxic gas generation. These parameters determine fire safety and hazard in real-life fire situation.
With regards to the SRR, there exists difficulty in establishing smoke release rate through calculation because of inherent complexities. Smoke is emitted when combustion is not complete and is related to the chemical constituents of the substrate, the combustion environment notably oxygen and temperature levels. 48 A correlation between the smoke released and amount of oxygen present has been investigated 56 and established that decreasing oxygen level results in higher smoke release. Thus, the smoke emission rate per unit area SR is a function of the product of MLR (ṁ) and percentage (%) smoke conversion by mass (Smc) given by SR = ṁ.Smc. This means that materials having high rates of smoke conversion, Smc, but low MLR will exhibit lower smoke formation, thereby reducing hazards accruable from smoke emission when compared to materials exhibiting lower conversion rate of smoke but higher MLR. This is a factor FR polymers posits to the end user in comparison to the behaviour exhibited by the pristine polymer because flame suppressants have the tendency to reduce overall MLR at a specific heat flux but increase the percentage conversion of fuel to smoke.
As per rate of emission of toxic gases and the quantity of gases emitted to the environment, this is similar to the rate of smoke emission and function of the chemical constituents of the fuel and burning conditions of the substrate. 57 Polymers emit CO during combustion, and those containing nitrogen will emit nitrogenous products especially when the supply of air is limited. Thus, a correlation for the rate of emission of CO can be thus presented; Eco = ṁ.Sco. 58 This implies that increasing FR results in higher conversion to CO while reducing the MLR. Hence, in actual fire circumstance, the external heat flux is likely to vary as fire in an enclosure propagates due to the effect of reradiation and ignition of other materials, which result in varying levels of HRR, gaseous toxicity and smoke emission. 59
LOI and UL-94-V fire tests
Limiting oxygen index (LOI) is used in polymer and composite characterization. It is defined as the minimum concentration of oxygen expressed as percent volume in a mixture of oxygen and nitrogen that will support flaming combustion of a material initially at room temperature (ASTM D2863-77). 60
UL-94-V (Underwriters laboratories) is standard small-scale bench flammability test equipment for polymer flammability that determines the propensity to either self-extinguish or propagate the flame once the specimen is ignited. The three ratings V-2, V-1 and V-0 show that the material was tested in vertical position and self-extinguished within a specified time after the ignition source was removed. 61 The polymer is mainly rated according to the recorded flaming time of the specimen as well as if the dripping occurs and ignites the cotton placed under the test specimen.
Usually, additional FR is required in combination with the PNC to pass the UL-94 test. The propensity to ignition and FR of polymers as characterized by LOI tests are done at ambient temperature and the values of LOI reduce with increasing temperature. Thus, the capacity to ‘self-extinguish’ may not be a criterion because some materials having very high LOI values at ambient temperature may burn under intensive flame situations without self-extinguishing.
Also, high LOI values that may not be correct may be given by the melting and dripping of polymer during LOI testing. Drops of flaming polymer can cause self-extinguishing by transferring out the fire from the sample surface. Other limitations of the LOI fire test were confirmed through the flammability tests carried out on flame suppressing systems composed of montmorillonites (MMT). 62 The results obtained from LOI tests conflicted with data obtained from UL-94-V test and CCT data for copolymers of ethylene vinyl acetate flame suppressed using magnesium-di-hydroxide, Mg(OH)2 or a combination of Mg(OH)2 and MMT. 62 The presence of MMT resulted in the creation of a protective layer inhibiting flame spread inside the nanocomposite ascribed to effective dispersion of MMT in the polymer matrix as elucidated in Figure 5, while Figure 6 elucidates a schematic illustration of the mechanism of MMT flame retardancy.
Effective intercalation of nanoparticles in the polymer matrix.
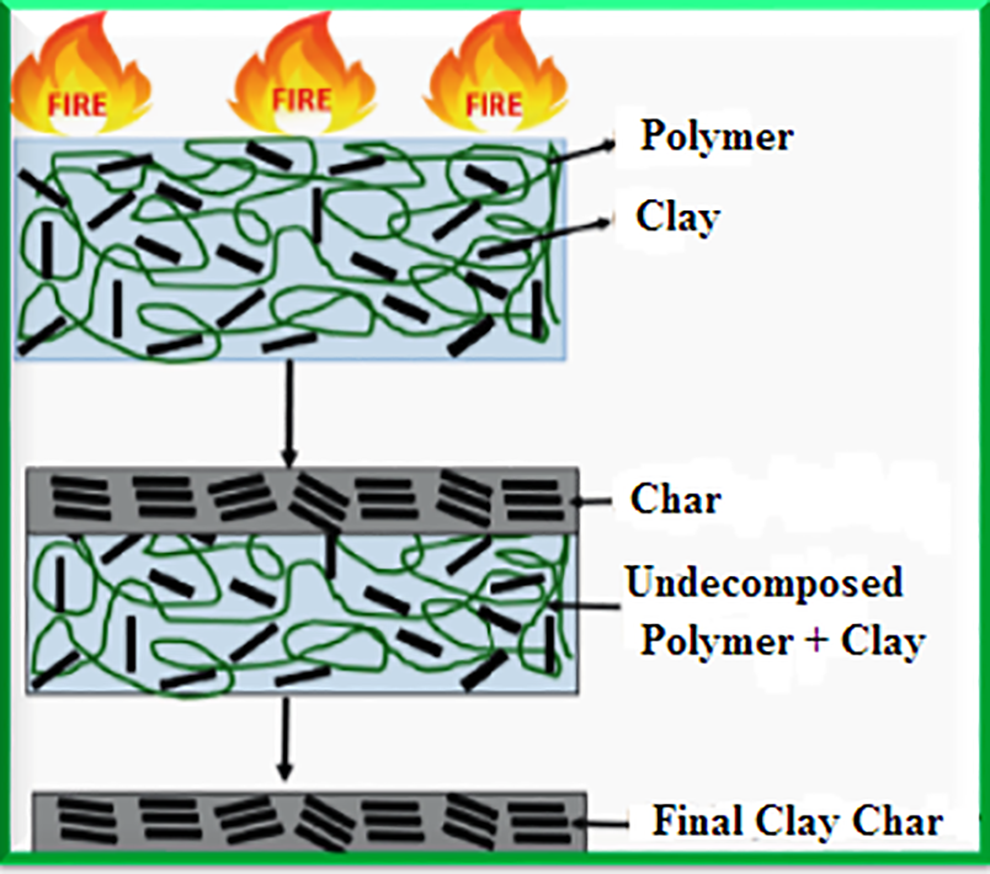
Mechanism of MMT flame retardancy.
Some limitations in UL-94-V include its enhanced performance for easily flowing samples than for samples with high cohesion. The CCT gives a more comprehensive flammability result. HRR is a broadly acceptable parameter for adjudging polymer FR. 63
Fire suppressing agents
Inorganic fillers
Inorganic fillers possess the propensity to affect the flammability of polymers, namely reduction in the composition of combustible emissions; modification of the thermophysical and thermal conductivity of evolved materials and variations in the viscosity of evolved products. And all these modifications influence flammability characteristics of the polymer. 64 However, specific minerals are used as flame suppressing agents on purpose due to their high temperature attributes. Metal hydroxides especially MH and aluminium hydroxide (AH) are specifically applied as FR additives. 65 Others are zinc borates and hydroxycarbonates. 66 Physically, during fire situations, these FR act by suppressing the rise in temperature through endothermic energy absorption. Their mechanism of FR lies in the formation of a protective ceramic char or vitreous barrier layer through the release of non-flammable molecules of H2O and CO2 which dilute the combustible gases. 67
ATH and MH
ATH, 2Al (OH)3, is an effective low-cost filler for reducing flammability of composites. However, a high loading of this filler (>50%) is required to attain adequate FR. 68 The decomposition of ATH commences at about 220°C and requires the composite processing temperature to be below 200°C. The decomposition of ATH occurs as shown; 2Al (OH)3 → Al2O3 + 3 H2O and is endothermic in nature.
ATH absorbs heat from the polymer at a temperature below which most composite polymers begin to degrade because the endothermic peak occurs at 300°C. Oxygen access to the polymer composite surface is inhibited by the emission of water and dilution of evolved flammable gases. 69 FR is believed to be enhanced by ATH through char formation in the condensed phase. MH, Mg (OH)2, is another endothermic FR which undergoes an endothermic reaction as follows: Mg (OH)2 → MgO + H2O.
Unlike ATH, it can maintain stability at 330–340°C and, therefore, is able to withstand high-processing temperature. The draw back is the possession of similar processing temperature with some polymers like polyester, vinyl esters and epoxies and hence a less efficient FR in comparison to ATH. Similar to ATH, MH absorbs heat from the matrix polymer, thereby retarding flaming and water evolved during decomposition, while also minimizing the concentration of flammable gases and H* and OH* radicals in the flame. Magnesium oxide released from the reaction also constitutes insulation. 70 A study of the effect of MH on saw dust and rice husks-filled PP composites reported a reduced horizontal burning rate (ASTM D 635) of about 50% for both fillers. 67 LOI values of composites containing FRs were higher than those without FRs.
Intumescent systems
Intumescent systems are additives used in thermoplastic and thermosetting fillers as char propagators. 71 CCT studies of the influence of hybrid MH/MMT on FR of polyamide 6/polypropylene (PA6/PP) nanocomposites revealed that partly replacing MH with MMT (2 and 4 wt%) carried out at filler content of 30 wt% reduced values of PHRR and THE as a result of the formation of a protective surface, and insulative sheet by MMT within the MgO layering on the material surface during combustion. The char residue was enhanced by the migration of MMT sheets to the surface of char. The thermal stability and FR of PA6/PP/MH/MMT nanocomposites were enhanced with increasing inclusion of MMT. 71 An intumescent system is composed of an agent of dehydration, which on thermally decomposing, releases a carbonaceous substance, containing large quantities of carbon and hydroxyl groups, which esterifies with the acid and undergoes decomposition to form carbon, acid, water, carbon dioxide and a foam emitting substance, which releases large amounts of non-flammable gases. 72 The intumescent char is then obtained from the carbonizing agent and non-flammable gases decomposition products. 73 An insulating layer capable of inhibiting heat transfer and access of oxygen is thus formed by the char.
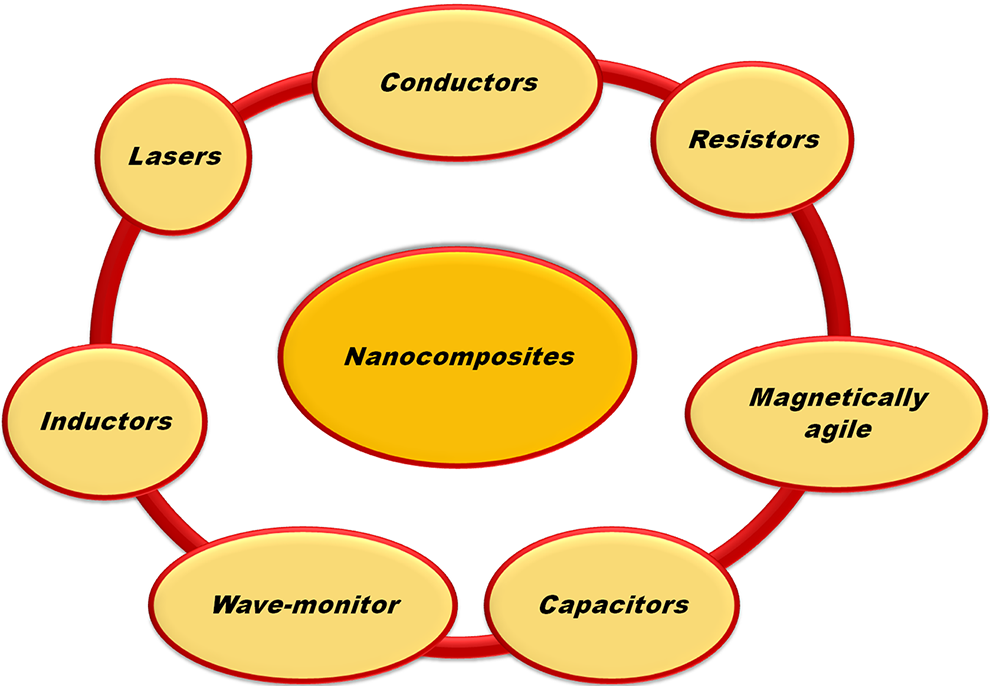
Flame retardancy using nanomaterials
Nanomaterials have evolved over the years to become an ecobenign route of FR. Some nanomaterials used in PP FR are schematically shown in Figure 7.
Some nanomaterials applied in FR of PP.
Modified silicate layers are homogenized at nanoparticles level in the polymer matrix to attain interestingly unique properties including enhanced thermal and FR. 74 The silicate-layered FR of nanocomposites depends on its structure and formation of a char layer. Transmission electron microscopy (TEM) images of PP/nanoclay depicting effective dispersion of nanoparticles of clay in PP nanocomposite are shown in Figure 8, while Figure 9 shows coloured scanning electron microscope (SEM) of nanographene revealing graphene sheets, respectively.74,75

TEM of PP/nanoclay (a) 50 kX and (b) 100 kX. 74

SEM micrograph of nanographene. 75
As already mentioned, hazards emanating from fire occur due to combination of various parameters including smoke toxicity, smoke obscuration, flame spread, released volatiles flammability, ignition, ease of flame out, and HRR. Nevertheless, most dangerous fire hazards are toxic gases, smoke and heat. An increased HRR causes rapid ignition and flame spread. In addition, HRR checkmates the intensity of fire and hence more important than other fire parameters such as ignition, smoke toxicity and flame spread. 76
In real-life fire condition, the time for individuals to safely escape from fire depends on the HRR. 77 The emission of smoke is another critical fire hazard. This is because dense smoke dishevels victims and hinders ease of fire-fighting personnel from rescuing fire victims. The degree of toxicity of fire gases is controlled mainly by CO contents and is responsible for fire most fatalities.
Hence, it becomes imperative to formulate enhanced FR polymers to reduce fire hazards in real fire situation. The FR property of polymers such as PP is improved through inclusion of FRs such as ammonium phosphate, MH, ATH, bromine-based organic compounds or intumescent systems, which hinder pyrolysis of these materials. 78 However, inclusion of these FR into PP, sometimes introduces some negative effects. For instance, MH, APP and ATH can only enhance polymer FR at very high filler loadings of up to 60%.79,80,81 And this high level of filler loadings results in high density, difficulties in compounding, extrusion and reduction in mechanical properties and absence of flexibility in the end product. Additionally, usage of halogen-oriented compounds such as bromine have developed environmental issues and thus banned in several countries due to emission of toxic gases. 82 Additionally, IFRs are not cost-effective and viable in applications requiring electrical conductivity. However, advent of PNC has generally eliminated most IFRs deficiencies. 83
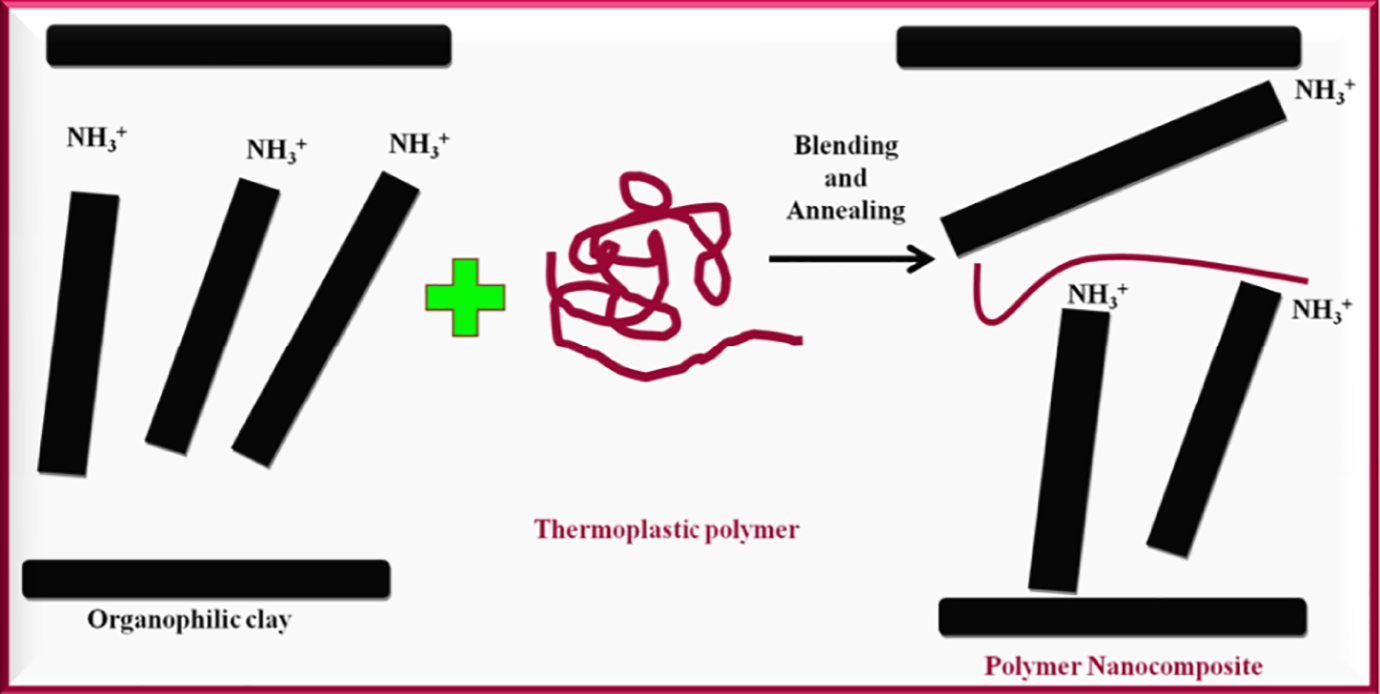
Nanocomposites are double-phased materials incorporating appropriate fillers at nanometre level (10−9 nm) into the polymer matrix. 84 In comparison to the pristine PP, PP nanocomposites exhibit improved properties at low filler loading of between 0.1% to 10 wt%. Layered silicates align themselves in a stack orientation having a gap inclusion within them and referred as ‘interlayer’ or gallery. 85 Commonly used layered silicates for intercalating polymers are MMT, saponite and hectorite. 86 The layered silicates that are modified through ionic exchanges are called organoclays and combine with polymers to form polymer-layered silicate nanocomposites. Layered silicates incorporated in nanocomposite synthesis have thickness of about 1 nm. 86 Figure 10 shows TEM images of some nanomaterials used in PP nanocomposites. The dimension of these layers laterally varies up to multi-microns and hence the ratio of length to thickness or aspect ratio of these layered fillers is very high in the range of values above 1000.

TEM images of (a) nanosilica particles, (b) Halloysite nanotubes, (c) MWNTs, and (d) nano-alumina nanoparticles. 86
Synthesis of nanocomposites
There are several processes applied in the fabrication of nanocomposites.
In situ polymerization
During in situ intercalative polymerization, graphene or modified graphene is initially swollen within the liquid monomer. Polymerization is then initiated using a suitable initiator applying heat or radiation.82,87,88 PNC prepared using these methods include (polystyrene)/graphene, 88 (polymethylmethacrylate)/expanded graphite (EG), 89 (polyethylene terephthalate (PET))/layer double hydroxide (LDH).16,33 This was the earlier route used in synthesizing (PA6)/clay nanocomposites. 90
Here, a monomer in liquid state or a monomer solution swells the modified silicate layers whereby monomer migration into layered silicate environment takes place, thereby creating suitable condition for polymerization to occur in the intercalated sheet layers. The polymerization reaction is initiated using a heat, radiation or appropriate initiator. The initiator is positioned within the interlayer before monomer swelling.
Solution intercalation
Solution intercalation involves the polymer or prepolymer been soluble in the solvent system while graphene or modified graphene is dispersed in a suitable solvent like water, acetone, chloroform, dimethyl formamide or toluene. 91 The polymers then absorb on to the delaminated sheets while the solvents evaporate. 92 PNC like polyethylene-grafted maleic anhydride/graphite is fabricated through this route. This method is similar to in situ polymerization. 93 Subsequently, polymer dissolved in solvent is added to the solution and intercalation occurs within the layers of clay. The final procedure is solvent removal by vacuum vaporization. This route of nanocomposite fabrication involves the use of large quantities of solvents and not environmentally friendly.
Melt intercalation
In melt intercalation, graphene or modified graphene is mixed with the polymer matrix while in molten state. A thermoplastic polymer is mechanically mixed with graphene or modified graphene at elevated temperatures using conventional methods like extrusion and injection moulding. 94 The polymer chain is then intercalated or exfoliated to form nanocomposites. PNC fabricated using this method include PP/ EG, 95 HDPE/EG 96 and so on.
There is a significant loss of conformational entropy by polymer chains during melt intercalation. The major reason for this system is the contribution of polymer/ graphite nanoplatelets (xGNPs) and polymer/organoclay interaction during blending and annealing procedure. 97 Industrially, melt intercalation is increasingly utilized in fabrication of PNCs.
PNC structures
The homogeneous and even distribution of nanofiller in the polymer matrix is a major factor in property determination of PNCs. 98 The quality of dispersion is categorized into three types, namely immiscible or phase separated, intercalated and exfoliation98,99 as schematically elucidate in Figure 11.
In the immiscible or phase-separated phase, the polymer matrix does not possess the ability to intermix and penetrate into the single layers of graphene sheets or silicate layers. A phase distinction exists between the graphene or silicate layers and the polymer matrix. The polymer chains are not able to penetrate through individual graphene sheets or silicate layers. Therefore, the composite may be referred to as a micro-composite because the nanofiller particulate sizes are greater than 100 nm. 99
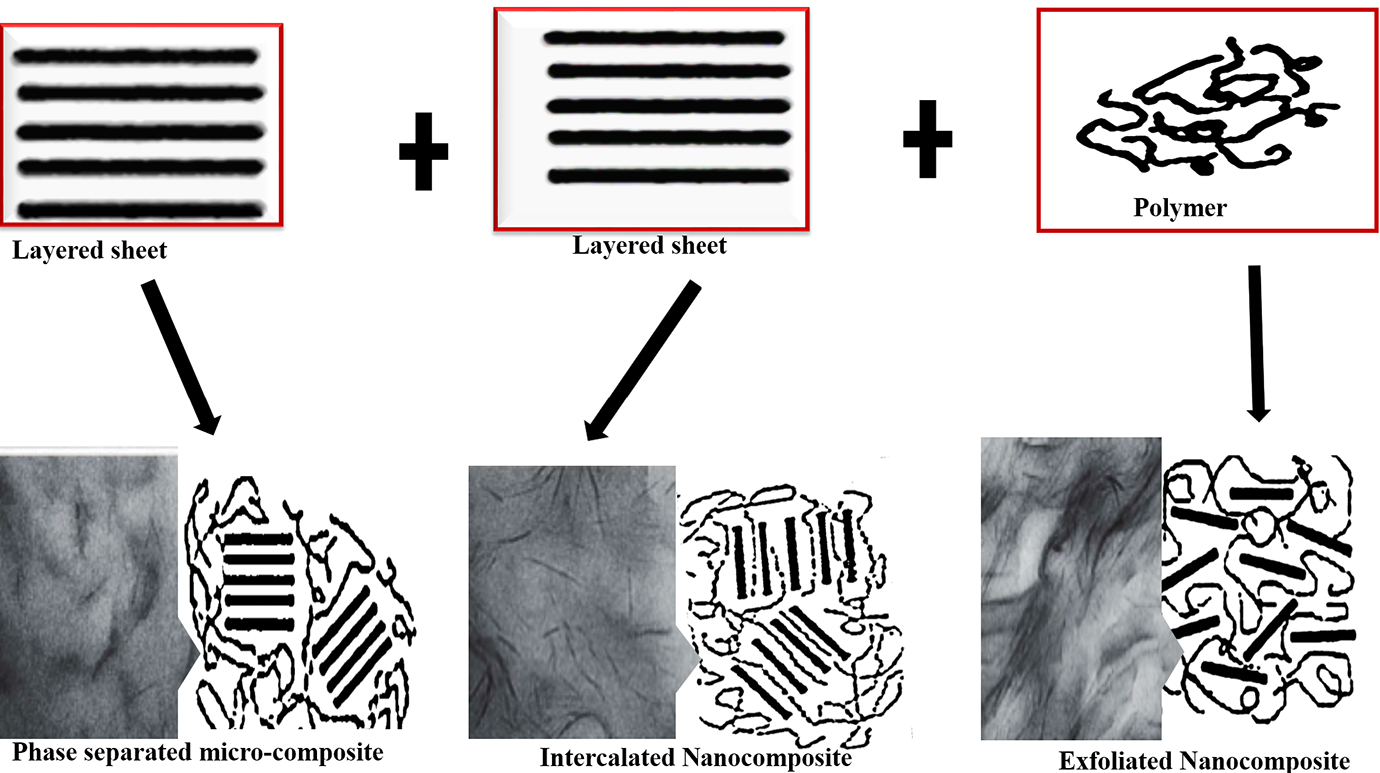
PNC dispersion structures.
The significance of this phase is that the effect of reinforcement of the layered graphite or layered silicate will not attain a maximum level, thereby minimally improving property enhancement or completely degrading the properties. The intercalated structure entails an improvement in the extent of penetration of individual graphene layers by the polymer chain. They are fabricated when a mono- or multichains of polymer are intercalated or properly aligned within the graphene or silicate sheets resulting in a properly arranged multilayer morphology of alternating graphene sheets or silicate layers and polymer matrix. The intercalated structures are referred to as nanocomposites because the nanofiller particle sizes are less than 100 nm. 49 In comparison to the immiscible or phase separated micro-composite, there are significant improvement of PNC properties accruable from this phase of reinforcement.
The phase of exfoliation/delaminated or reinforcement phase involves a homogeneous interaction between the polymer matrix and the graphene or silicate layers. Here, there is complete penetration of polymer chains into isolated graphene layers and uniform distribution of polymer matrix within individual isolated silicate or graphene layers. The significance of the delaminated phase is the maximization of the interfacial interaction between the graphene and silicate layers and the polymer matrix due to the provision of a large surface area of graphene sheets or silicate layers available for polymer interaction. Thus, the effective reinforcement of graphene sheets or layered silicate is excellent with superior property enhancement.
The exfoliation phase entails a nanocomposite composed of large ratio of surface area to volume, lower stress concentration, improved FR, and mechanical and physical properties. 100 CCT studies of the influence of hybrid MH/MMT on FR of PA6/PP nanocomposites revealed that partly replacing MH with MMT (2 and 4 wt%) carried out at filler content of 30 wt% reduced values of PHRR and THE as a result of the formation of a protective surface and insulative sheet by MMT within the MgO layering on the material surface during combustion. The char residue was enhanced by the migration of MMT sheets to the surface of char. The thermal stability and FR of PA6/PP/MH/MMT nanocomposites were enhanced with increasing inclusion of MMT. 101 This is elucidated in Figure 12.
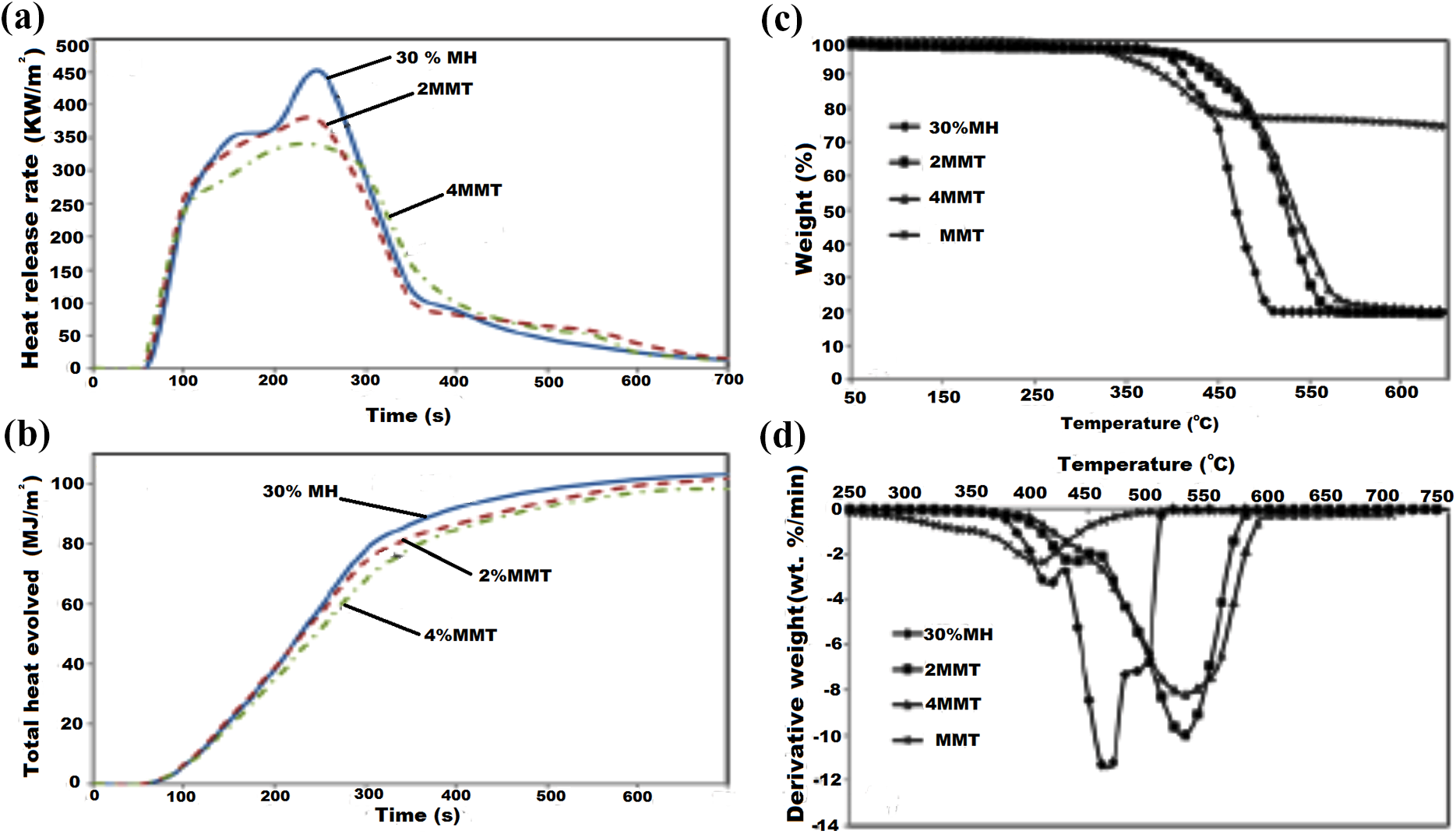
(a) HRR and (b) THE curves of 30% MH, 2MMT and 4MMT, respectively; (c) TGA and (d) DTG curves of MMT, 30% MH, 2MMT and 4MMT. 101
FR of PP and PP/blends nanocomposites
Enhancement of PP FR has been attained through inclusion of various LDHs. However, all LDHs incorporated in PP system were organically modified. This is because PP is a non-polar material and inherently exhibits inferior compatibility with the intercalated hydrophobic LDHs. Conclusively; LDHs used alone are not enough and require other materials to attain the needed FR behaviour. 12
The influence of covalently modified carbon nanotubes (CNTs) combined with C 60 (abbr. C-60-d-CNT) on thermal; flame retardancy and mechanical behaviour of PP have been studied. Inclusion of 1.0 wt% C-60-d-CNT significantly reduced PHRR by 71% in comparison with PP, while minimizing the combustion process. The enhanced thermal and FR behaviours were ascribed to the free radical trapping impact of C-60 and CNTs network. 21
As amphiphilic nanomaterials, graphene oxide sheets (GOSs) can be utilized as surfactants in varying technological aspects. Moreover, the use of GOSs in compatibilizing immiscible polymer blends offers prospects because in as much as it exploits their amphiphilic behaviour, it also explores their unique attributes. Thus, a research has shown that GOSs polymer functionalization can highly enlarge their scope of compatibilization. 22
The covalent functionalization of CNTs incorporated to IFR have been successfully fabricated and characterized. Through adjustment of the ratio of CNTs to FR, functionalized CNTs diameter was efficiently maintained within 20–90 nm. Inclusion of functionalized CNTs enhanced the FR of PP/PPMA, while significantly improving the mechanical behaviour of the polymeric materials. This was ascribed to enhanced interfacial adherence and stress transfer. 23
A new IFR was mixed with PP to fabricate novel IFR composites (PP/IFR) with good FR behaviour. Results revealed that IFR could minimize PP degradation rate; the created intumescent char was composed of irregularly aligned carbon and graphitic structures and contain P, N, O and C elements. Fourier transform infrared (FTIR) demonstrated that network structures of P–O–P and P–O–C were created. Energy-dispersive X-ray spectroscopy (EDS) demonstrated that some P elements were in connection with polyaromatic rings and could create large connected network. 24
The development of novel rigid PP composite foams reinforced with synthetic hydromagnesite obtained from magnesium carbonate derived from an industrial waste has been prepared and characterized. Ammonium polyphosphate (APP) was combined with hydromagnesite and layered nanoparticles and resulted to enhanced thermal stability. Enhanced flame retardancy, via CCT, was observed in specimens composed of intumescent additive. 25
Thus, novel nanocomposites have been fabricated using PP and some LDHs via new solvent-mixing technique. SEM analysis reveals that the LDH nanoparticles were evenly distributed within the PP matrix. X-ray diffraction (XRD) analysis reveals that LDHs were fully exfoliated. CCT analysis indicates that PP/Zn2 Al-borate nanocomposites exhibited superior FR behaviour in comparison with PP/Mg3Al borate nanocomposites 15 wt% inclusion of Zn2 Al borate LDH in pristine (unmodified) PP resulted in minimized PHRR of 63.7%. Solvent mixing was adjudged superior to melt-mixing technique. 34
A highly effective single-component polymeric IFR, piperazine-treated APP (PA-APP) was prepared. The LOI of PP with inclusion of 22 wt% PA-APP attained 31.2%, which improved by 58.4% in comparison with that of PP with same amount of APP, while the vertical burning test (UL-94) passed the V-0 rating. CCT results revealed that PP/PA-APP composite exhibited superior performance in comparison with PP/APP composite. 35
Studies of the effects of increasing inclusions of graphene, EG, carbon black (CB) and multiwall carbon nanotubes on pyrolysis, reaction to mini fire, burning trend and on electrical, thermal and rheological behaviours of FR PP have been conducted. FR, mechanical and thermal behaviours of the material was improved. 38
Inclusion of wood powder enhances mechanical behaviours of polymers whilst also escalating their burning speed. To improve FR of wood–plastic composites (WPC), varying FRs, including APP, MPP and AH were included to WPCs. On inclusion of 10 wt% of APP to PP, FR was not improved. Wood powder escalated the burning pattern of PP. Synergy between wood powder/APP was affirmed in minimizing FR. Wood powder enabled the formation of foamed char layering by APP during burning. The protective char layering can minimize heat and oxygen diffusion within WPCs. 39
ATH has been included in PP to fabricate a FR composite. PP/ATH composites exhibit superior FR on comparison with pristine PP. 40
PP/APP-II/pentaerythritol (PER) and PP/microencapsulated APP-I with MF(MFAPP)-II/PER composites have been fabricated and their FR, thermal stability and microstructure of the formed composites investigated based on LOI, UL-94, thermogravimetry (TG), EDS, SEM and CCT. In comparison with PP/APP-II/PER composites, the PP/MFAPP-II/PER composites exhibited superior LOI value and passed the V-0 rating. TG, EDS, SEM, and CCT results demonstrate that MFAPP-II is advantages towards formation of a compacted and stronger intumescent char, which minimizes the maximum rate decomposition temperature (Tmax), HRR, total heat release (THR) and mass loss (ML) for PP/MFAPP-II/PER composites, in addition to enhancing the thermal stability, compatibility and distribution of MFAPP-II in PP/MFAPP-II/PER composites. 43
EG and IFR composed of triazine char-forming agent (CFA) and APP was selected in wood powder-PP composites. The WPC containing 25 wt% EG/IFR (2:3) exhibited the most superior strength in the flexural, Izod impact and thermal stability. The mixture demonstrated significant enhancements in the FR behaviour as revealed by minimizations in HRR and the smoke production release rate. SEM results demonstrated that a compact and thick char layering was created in the mix, thereby restraining heat transfer flow and flammable gases in the condensed phase. Hence, the mix of EG/IFR confirmed as a potential FR system for WPC. 44
PNC exhibits significant improvement in properties when compared to the pristine polymer with the content of layered silicate or xGNPs in the range of 0.1–10%. There are reported improvements in mechanical properties such as tensile, compression, bending and fracture; barrier properties such as permeability and solvent resistance; optical properties and ionic conductivity. Other known properties enhanced by the incorporation of nanocomposites are flame retardancy, thermal conductivity and stability at very low loading levels. These properties are improved due to the formation of thermally insulating and low permeability char as a result of fire and polymer degradation. Moreover, enhanced IFR can improve distribution and result in highly enhanced FR and mechanical behaviour when compared with those not uniformly distributed. The FR behaviour is shown in Figure 13. 102
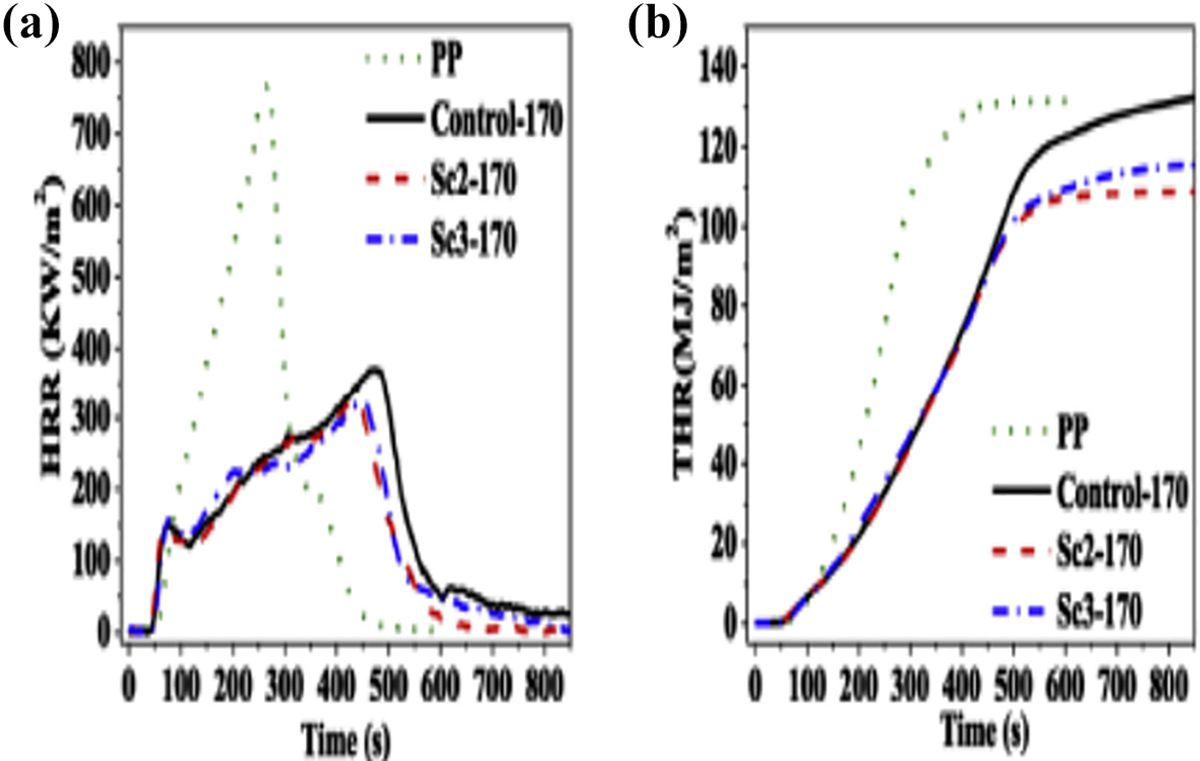
CCT: (a) HRR and (b) THR curves as a function of time of pristine PP and varying defoamed PP/IFR composites. 107
APP is not an effective FR for PP when applied singly. Ethanolamine (ETA) has been used to modify APP chemically through ion-exchange reaction. 53 The produced ethanolamine-modified APP (ETA-APP) was used in retarding PP flammability such that LOI value attained 35.0% and the vertical burning test (UL-94) passed the V-0 rating at a loading of 35 wt% ETA-APP. Additionally, CCT results revealed that the HRR, THR, MLR, the SPR and TSP of PP/35 wt% ETA-APP composite reduced by 77.2%, 88.5% and 77.9% for THR, TSP and the FGR, respectively, in comparison with PP composed of equal amount of APP. 53
A study had compared FR and degradation behaviour of PP/kenaf fibre composites using three different APP FRs. 36 UL-94-V tests and CCT results affirm that level of APP inclusion and the particles high aspect ratio are critical to obtaining a homogeneous FR blend, which tends to reduce sustained combustion, while minimizing the composites PHRR as elucidated in Figure 14.
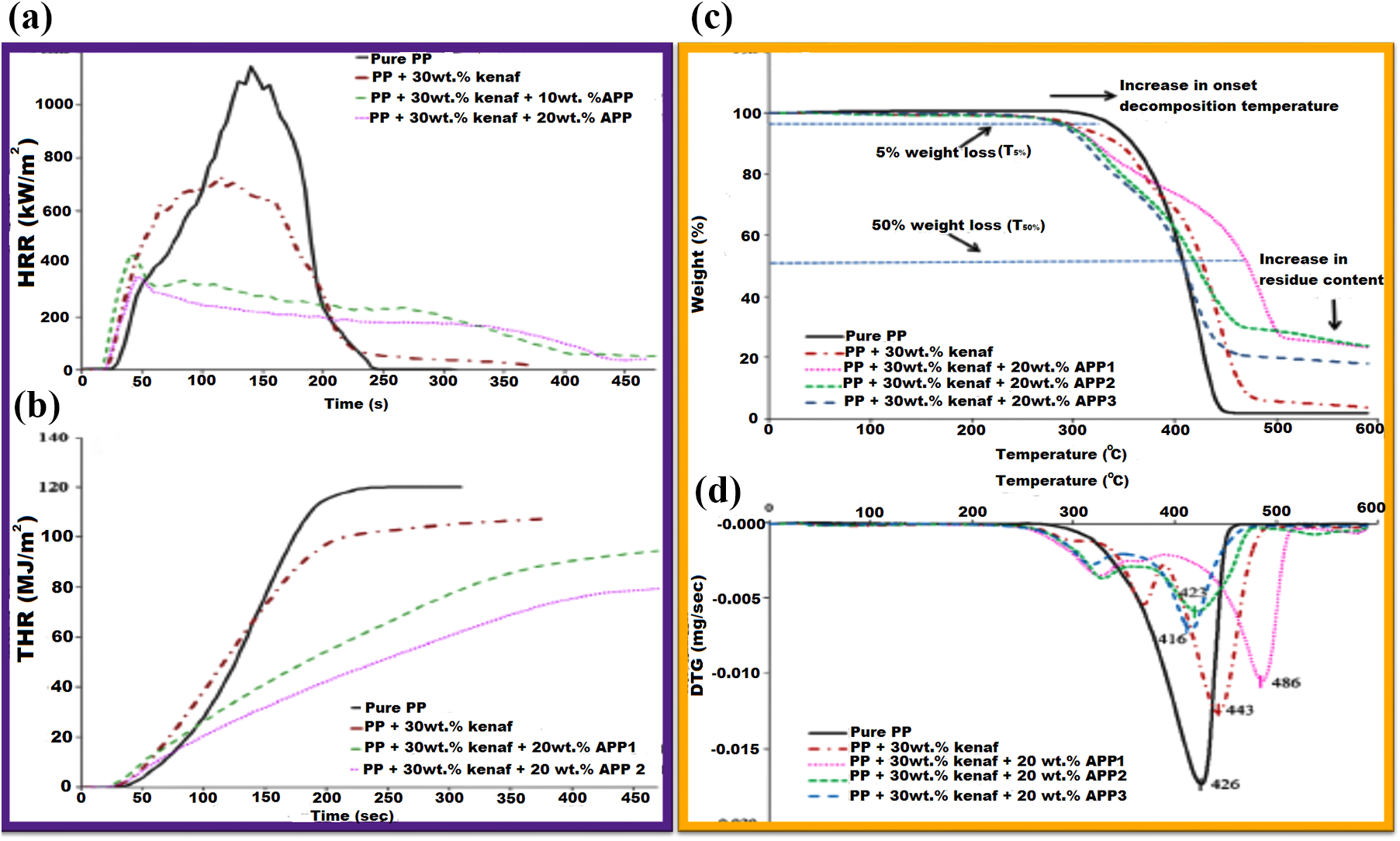
(a) CCT test results in PP/kenaf composites with FR APP1: (a) HRR, (b) THR, (c) TGA and (d) DTG curves of FR samples. 36
An investigation using CCT, LOI and UL-94 to understand the flammability behaviour of xGNP/PP/PE nanocomposites via melt extrusion revealed that co-opting GNPs into PP/PE enhanced the flame repression tendencies of PP/PET/GNP nanocomposites tremendously. Results from CCT exposed appreciable minimization of PHRR; MLR and very high TI due to GNPs inclusion in PP/PET blend. Improvement in flammability parameters was ascribed to the growth of aligned, dense and organized char layers on the surface of the PP/PET/GNP nanocomposite. It was also discovered that effective thermal conductivity of the nanocomposites was increased by 80% with increasing GNP loading from 10.2 W/m.K for the unreinforced PET/PP blend to 18.7 W/m.K for the 7 phr-reinforced PP/PET/GNP nanocomposites. Thermal percolation threshold was attained at 3 phr GNP loading and ascribed to the creation of effective interwoven network of heat transmission links by GNP which was collaborated by morphological studies. DSC data indicated improvement in crystallization temperature but reduced degree of crystallinity with melting point mostly unaffected and equal dispersion of GNPs in polymatrix as a result of a network of intermeshed GNP sheets at 3 phr. 103
An investigation into the development and characterization of xGNPs-oriented amorphous thermoplastic nanocomposites revealed a significant enhancement of flammability and mechanical properties with increased loading of xGNPs. Generally, all the thermoplastics exhibited about 3% increase in flexural modulus in comparison to pristine matrices, with percolation threshold at only 1 wt% of xGNPs. However, the degree of dispersion of xGNPs in the matrix was not proportional to results obtained, though the observed difference in samples was much lower than the error bars of the tests. 104 Another study revealed that the dispersion of small quantities of xGNPs in polymers resulted in the enhancement and improvement of thermal conductivity, thermal stability, tensile strength and young’s modulus, gas barrier and electrical and flame retardancy of the PNCs. 105 It concluded that two-dimensional xGNPs has initiated an alternative route to the production of high performance; low cost and lightweight polymeric nanocomposites for diverse applications.
Studies of synergistic FR effect of xGNPs/MMT on PNCs have been investigated. 106 Results revealed that increasing concentration of xGNPs improved the FR of the nanocomposites. SEM micrographs revealed that xGNPs and MMT were trapped inside the char layers when the blend was subjected to high heat flux which indicated that xGNPs and MMT are efficient thermal barrier irrespective of the complete combustion of the matrix polymer. Thermogravimetric analysis (TGA) results revealed the effectiveness of xGNPs as inhibitors to ML. Another investigation into the post-flammability residual bending characteristics of xGNPs/MMT-reinforced PNCs revealed that xGNPs influenced the flammability behaviour of the PNCs. This FR attribute exhibited by xGNPs was related to their inherent thermal conductivity. This study also posited that rate of increase of temperature on the surface of the hybrid nanocomposites was reduced with increasing concentration of xGNPs. This phenomenon was attributed to the formation of a thermal barrier as a result of the homogeneous dispersion of xGNPs-MMT within the PNC. 41
A comparative investigation of the property enhancement of xGNPs, CB and CNTs inclusion within PNCs revealed that the rate of dispersion of these carbon allotropes in the matrix determined accruable property enhancements. Overall, xGNPs were found to impact most on properties especially flammability. This report concluded that xGNPs inhibited dripping and improved heat absorption which led to delayed TOI and enhanced resistance to flammability relative to the formation of a protective char residue during combustion. 107
Another recent investigation into property improvement by xGNPs inclusion into various PNCs and varying techniques of formulating and dispersing xGNPs in PNCs revealed that co-opting xGNPs into PNCs generally improved the rheological, electrical, mechanical, thermal and barrier properties of PNCs. This report also reviewed applicability prospects of xGNPs/PNCs and challenges in further developments of PNCs. 95
The effect of xGNPs and other carbon additives in combination with antimony trioxide FR PP was investigated. To enhance the flame retardancy of PP, xGNPs was formed by thermally reducing graphite oxide (TRGO). The incorporation of xGNPs into the blend resulted in increased flame retardancy, mechanical, electrical and thermal properties. Studies revealed that even without compatibilization during melt extrusion, the blend exhibited a homogeneous dispersion of xGNPs in the matrix resulting in increased flame retardancy, improved mechanical, electrical and thermal properties. 108
The thermal degradation and flammability properties of PP/CNT nanocomposites have been investigated. 109 Report from this research indicated that CNT was well dispersed without any organic treatment on the CNT surface and without incorporation of any compatibilizer. Results indicated that thermal properties of CNT improved with CNT addition. Flammability properties were also affected as HRR value of PP was also affected. They author further investigated the mechanism of fire retardancy in PP/CNT nanocomposites. 110 It was posited that FR was attained through the formation of a homogeneous network-structured floccules layer, which blanketed the entire surface covering all gaps and cracks. According to this study, the layer re-emitted much of the incident radiation back into the gas phase from its hot surface and thus decreased the transmitted flux to the receding PP layers below it. This eventually reduced the pyrolysis rate of PP. To further understand this phenomenon, the researchers assessed the thermal conductivity of PP/CNT nanocomposites. It was revealed that thermal conductivity of PP/CNT nanocomposite increased with increasing CNT concentration especially above 160°C. This report also posited that the TOI and the PHRR of the PP/CNT attained percolation thresh hold at 1 wt%. 111
A novel PP nanocomposite was formed by co-opting IFR, CNTs and graphene into the PP matrix. 58 TEM results which was supported by XRD results revealed a homogeneous and uniform dispersion of IFR, CNTs and xGNPs in the PP matrix. TGA results indicated that the incorporation of IFR, CNTs and xGNPs into the PP matrix enhanced thermal stability and char yields of the blend. The PP/IFR/CNTs/RGO nanocomposites, incorporated 18 wt% IFR, 1 wt% CNTs and 1 wt% xGNPs attained LOI value of 31.4% and UL-94-V0 grade. Data from CCT indicated that combustion behaviour, PHRR and ASEA reduced simultaneously with increasing addition of IFR, CNTs and xGNPs. PP/IFR/CNTs/RGO nanocomposites exhibited an 83% reduction in PHRR and 40 s delayed TOI in comparison to pristine PP. 112
Another research into thermal and flammability properties of PP/CB nanocomposites prepared by melt compounding studied the effect of nanofiller loadings on the thermal and flammability properties of PP. 113 Results revealed enhanced flame retardancy and thermal stability of PNC in oxygen and air was recorded with increasing filler loading level of CB. This improved mechanism was attributed mainly to trapping of peroxy radical by CB nanoparticles at high temperature resulting in the formation of a ball-like gelled cross-linked network that inhibited heat and mass transfer. Results obtained from FTIR, rheology and gel measurements supported this thermal-oxidation cross-linking reaction. The possibility of applying CNT in improving electrical and thermal conductivity of polymers was articulated. 113
The mechanical, electrical, thermal and flammability property of pristine CNT was identified as the reason for the interest in the use of CNT in fabricating PNCs. The percolation threshold was identified as a parameter for the determination of electrical property due to the formation of interconnected network of CNT equating to a dramatic increase in electrical conductivity. It was also concluded that the high aspect ratio and small diameters of CNT led to attainment of enhanced electrical and thermal properties by the addition of small quantities of CNT (∼1 wt%). The report posited the prospects of enhancing the flammability, thermal, electrical and mechanical properties of PNCs with the co-option of CNTs in future. 114
A very recent study fabricated MH flame-retarded PA6/PP nanocomposites with MH weight replacement using MMT, xGNPs and CNT were prepared by melt extrusion using a counter rotating twin-screw extruder followed by injection moulding. 115 Results from SEM and FTIR analyses revealed the presence of interfacial copolymer PA6-g-PP enhanced compatibilization between PP and PA6. SEM micrograms revealed that the particle size of PP decreased in the presence of MH, MMT, CNT and xGNPs. CCT data revealed a reduction in PHRR and THE values of PA6/PP composites due to the inclusion of MH. 115 The synergistic combination of MH and MMT, and xGNPs and CNT has further reduced the PHRR and THE values of PA6/PP composite while SEM revealed that the nanocomposites had a compact residue ranging from xGNPs, followed by MMT and subsequently by CNT. LOI values improved with increasing MH content and further improved in the presence of MMT, GNP and CNT. TGA analysis revealed a decreasing thermal stability of PA6/PP while the weight replacement of MH with nanofillers (MMT, xGNPs and CNT) improved it. XRD analysis revealed that MMT layers are well dispersed in nanocomposites. TEM images revealed agglomerated CNT while MMT and xGNPs were well dispersed. The improvement in Young’s and flexural modulus values was countered by a drop-in strength, impact strength and elongation at break values improved further while the tensile and flexural strength values remained steady in the presence of MMT, xGNPs and CNT. The MH replacement with nanofillers (MMT, xGNPs and CNT) has shown the potential combined use of MH nanofillers to improve the flame retardancy of PA6/PP while maintaining its mechanical strength. It can be concluded that the combined use of MH with xGNPs exhibited the best performance in comparison to MMT and CNT. 115
In a recent investigation, organic-vermiculite (OVMT) was prepared from vermiculite, with high aspect ratio and regularly arranged platelets and intercalated by octadecyl trimethyl ammonium bromide. It was subsequently applied as a synergistic agent on the FR of PP/IFR system. 116 Results of LOI and UL-94-V testing revealed that low loading of OVMT improved the FR and retarded dripping for PP/IFR composites. The maximum LOI value was 32.9% with 1 wt% loading of OVMT in the composite. The advantageous combination of thermal and oxygen insulation at low content of OVMT significantly improved FR of the composite.
An investigation into the flame retardation of epoxy/organophilic montmorillonites (epoxy/OMMT) nanocomposite prepared by epoxy intercalated into the interlayer of OMMT and FR PP obtained with epoxy/OMMT nanocomposites and triphenyl phosphate (TPP) mixture as FR agent. 117 It was revealed that there is a remarkable synergistic FR effect of novolac epoxy/OMMT (NE/OMMT) and TPP on PP. FR PP with OI value of 36.5% and PHRR of 654 KW/m2. NE has more oxirane groups than BAE, which makes it more susceptible to generate carboxylic acid during combustion. The carboxylic acid can react with phosphoric acid generated from TPP and form thermally stable char to shield the matrix from heat and reduce the permeability of oxygen and flammable gas resulting in better FR. It can be seen that FR PP with NE/OMMT-TPP has a much higher residue than PP with BAE/OMMT-TPP which can further prove that NE is better char.
The FR of PP composites containing melamine phosphate (MP) and pentaerythritol phosphate (PEPA) was characterized by CCT in another study.37,118 The combustion mechanism of the composites after CCT was studied using SEM, FTIR, X-ray photoelectron spectroscopy, XRD and Raman diffusion. Results showed that PP composite composed of MP/PEPA exhibited good FR. It was revealed that three stages of intumescences existed and categorized into outer, middle and inner char layer, according to their various structures and components.
Another study of the FR of PP/flax/APP/EG composites revealed that all materials exhibited two decomposition procedures. FTIR evolved gas analysis assigned the steps in ML to distinct decomposition processes of various compounds. Results indicated improved FR with inclusion of APP/EG in PP/flax blend. 119
The effect of novel charring–foaming agent on FR and thermal degradation of IFR PP was studied and polymer was synthesized using cyanuric chloride, ETA and ethylenediamine as raw materials. Results indicated that IFR PP system (IFR-PP system) consisting of CFA, APP and Zeolite 4A is an efficient FR for PP. 120 It was revealed that the weight ratio of CFA to APP is 1:2, in order words, the components of the IFR are 64 wt% APP, 32 wt% CFA and 4 wt% Zeolite 4A, and IFR exhibited the most efficient flame retardancy in PP systems. LOI values of IFR-PP attained 37, when the IFR loading is 25 wt% in PP. Results also revealed that when IFR loading is only 18 wt% in PP, the FR of PP-IFR passed the V-0 rating and LOI values attained 30.2. TGA micrographs and data revealed that CFA has superior char formation ability and a high initial temperature of thermal degradation. CFA char residue attained 35.7 wt% at 700°C. The char formation ability of APP-CFA system was promoted by the incorporation of APP. The char residue attained 39.7 wt% at 700°C while the calculated value was 19.5%. Hence, IFR influenced the thermal degradation behaviour of PP, improved Tmax of PP decomposition peak and promoted PP to char formation as supported by calculation and experimental evidence. The proffered reason was that endothermic reactions occurred in IFR charring procedure and the layers of char formed by IFR inhibited the transfer of heat within IFR-PP system. 121
The effect of compatibilizing agent on the FR performance of intumescent PP formulation using PA6 as charring agent was investigated. 122 It was confirmed that addition of APP/PA6 blends promoted FR. Though, the use of interfacial agent stabilized the formulation. It was revealed that the incorporation of EVA x as interfacial agent promoted FR of PP/APP/PA6 blend and a function of the type, weight and amount of interfacial agent applied. The effect of EVA x inclusion gave better FR for CCT and LOI, better mechanical properties and higher elongation at break, at a lower price. The kinetic study assisted in identifying the various steps of PP/PA6/APP/ethylene-vinyl acetate (EVA) degradation of the intumescent blend. 123 PP degraded first while the second step corresponded to the interaction between APP and PA6. The result was a material showing a slow level of degradation in TGA analyses. Hence, this is the step of intumescent shield development. At high heating rate, in TGAs, the two steps occur simultaneously, thereby promoting the development of the protective shield. Oxygen was acknowledged to promote the development of intumescent structure.
The effectiveness of APP in various biocomposites was investigated. 124 To make for effective comparative studies of FR, lignocellulosic biocomposites reinforced with fillers were prepared via PP, polyurethane (PUR) and biodegradable starch matrices. PP/wood flake interfacial adhesion was made possible using alkoxy-silane-based reactive surfactant. The salination enhanced both the compatibility and the thermal stability of the wood flake according to TGA. Studies using Raman spectroscopic analysis of the silanated products showed that improvement of thermal stability was the result of the reduction in the ratio of amorphous phase of cellulose. The phosphorous additives in FR PUR biocomposites made up of waste biofillers and recycled polyol proved to be very effective because both the matrix and the filler components participated in the FR mechanism. It was shown that FR was attained with as little as 10% inclusion of polyphosphate.
The pyrolysis procedure of PP, PP/nanoclay nanocomposites, acrylonitrile–butadiene–styrene (ABS) and ABS/metallic hydroxide nanorods (MHR)/graphene nanosheets (GNS) nanocomposites in a CCT underwent simulation using numerical codes, the Federal Aviation Administration (ThermaKin, USA). Initially, HRR and the surface temperature relative to time were comparatively analysed with experiment data. Using reasonably input parameters, the pyrolysis behaviours were predicted. Results revealed that the char residue of PP/nanoclay performed as a heat-transferring barrecade, while the char layering of ABS/MHR/GNS performed as a mass transfer barricade. 125
A recent investigation into the melt rheology, thermal, electrical and electronics properties of xGNPs included PP nanocomposite revealed a good improvement in these properties with increasing concentration of xGNPs into PP. SEM images revealed a not too homogeneous dispersion of xGNPs in PP and a cryofractured microstructure of PP/xGNPs at 5% and 15%, respectively. At 5 wt% loading, SEM images revealed that xGNPs were independent of each other. But as the loading increased, xGNPs closely interwove with each other resulting in a dense dispersion of xGNPs in the PP matrix at a loading of 15 wt%. A higher resolution investigation revealed a wrinkled agglomeration of xGNPs showing a partial exfoliation of xGNPs in the PP matrix. 126
TRGO, composed of four single carbon sheets, was mixed with APP and MH, respectively and incorporated into PP. The influence of the nanoparticles on varying FR systems and potential synergisms in pyrolysis, behaviour to small flame, fire and mechanical attributes was studied. As regards OI and UL-94 results, inclusion of increasing amounts of TRGO to PP/APP effected OI and UL-94 classification negatively. On the other hand, systems containing only low levels (≤1 wt%) of TRGO attained V-0 status in the UL-94 test and high OI (>31 vol%) with decreasing HRR as depicted in Figure 15. TRGO enhances MH residue-structure, thereby functioning as a strong synergist relative to OI and UL 94 status (from HB to V-0) as depicted in Figure 16. 127
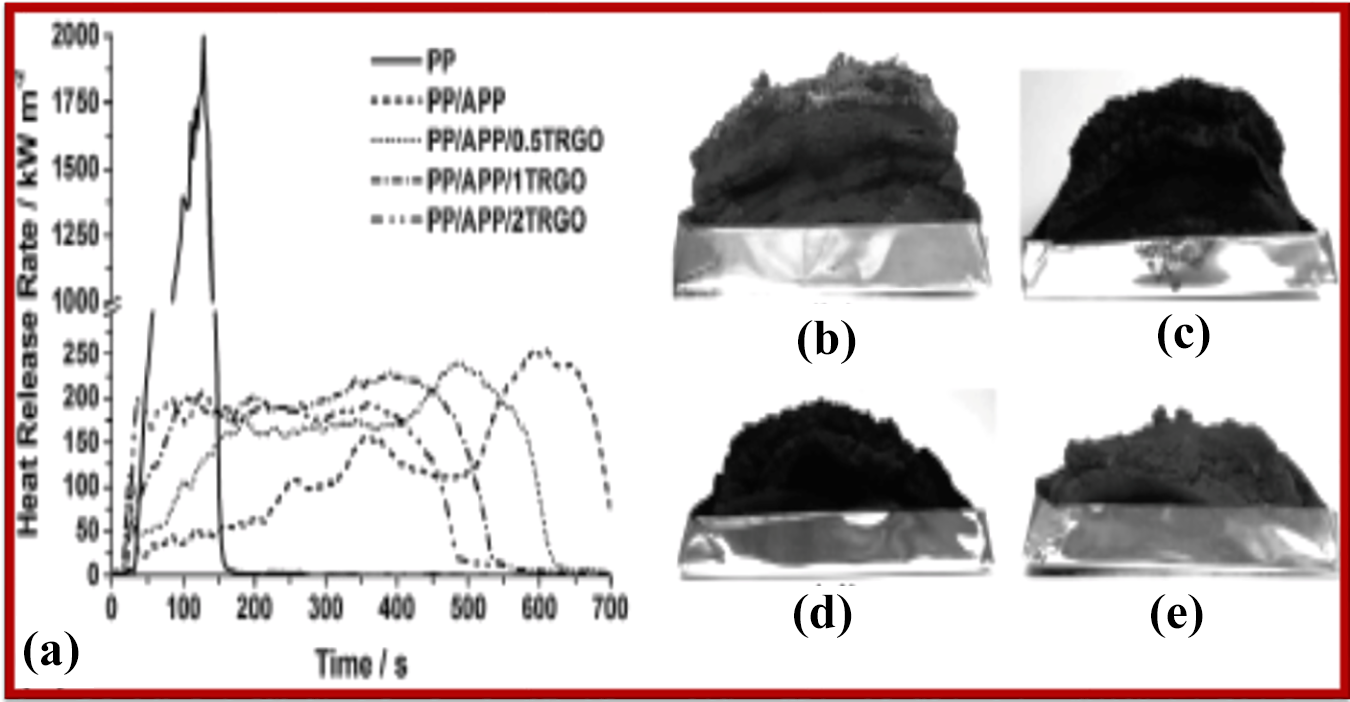
(a) HRR of PP, PP/APP and mixtures of PP/APP/TRGO; intumescent residue after burning of (b) PP/APP; (c) PP/APP/0.5TRGO; (d) PP/APP/1TRGO and (e) PP/APP/2TRGO. 127

Macroscopic surface structure and microscopic structure of CCT residues of (a) PP/53MH; (b) PP/53MH/1TRGO; (c) PP/59MH and (d) PP/59MH/1TRGO. 127
Finally, the properties of PP nanocomposites and nano-biocomposites have been studied and shown to be FR, electrically, and thermally conductive with enhanced mechanical behaviour in varying modes, and these have increased their scope of application in versatile areas of human endeavors including the aerospace, electrical/electronics, automotive, sensors, packaging, construction and building and so on. The potential of application of PP nanocomposites with further studies is endless.128–179
Conclusion and future outlook
PP is inherently a flammable material. Thus, FR is essential for their most common usage in energy, electronics, construction, aerospace, automotive and marine sectors. The use of traditional FRs to satisfy fire safety expectations is quite significant because of susceptibility to yielding POPs, which is of great environmental concern globally. With regards to environmental friendliness and other essential attributes, below standard fire behaviour presents a daunting challenge. Varying aspects of these challenges are elucidated in this article from the point of view of both materials and fire-exposure methodologies. It is imperative to essentially comprehend the combustion stages of PP nanocomposites in a qualitative and quantitative approach. This will enable insights into FR mechanisms, thereby facilitating accurate delineation of the material boundary parameters and transport behaviour. Essentially, this insight can enable integration with mathematical models and simulation techniques, thereby facilitating correct predictions of the materials applicability.FR is a critical parameter for PP nanocomposites essential for enlarging the scope of their applications especially in areas where flammability had previously hampered their usage. Successful FR attainment in PP nanocomposites pre-posits a potentially infinite scope of application for these materials in future.
