Abstract
Nanocomposites composed of polypropylene (PP), organoclay, ethylene acrylic acid (EAA), and maleic anhydride-grafted polypropylene (PP-g-MA) were prepared using the melt mixing technique, and their thermal stability properties were investigated. PP-g-MA and EAA were used as compatibilizers in the nanocomposites. The effects of different concentrations of organoclay on the physical properties of nanocomposites were investigated. The kinetics of PP/organoclay nanocomposite degradation were investigated by thermogravimetric analysis, the activation energy of nanocomposite system was confirmed by the Kissinger method, and the performance of nanocomposites was investigated by differential scanning calorimetry, ARES rheometer, and transmission electron microscopy. The test results show that the addition of organoclay can improve the thermal stability of PP/organoclay nanocomposites, and the activation energy is slightly improved; rheological analysis shows that as the organoclay is gradually added to the composite system; the fluidity of PP and the processing properties are improved. In addition, the mechanical properties were measured, and it was found that an appropriate organoclay content can effectively improve the mechanical properties of nanocomposites.
Introduction
Polypropylene (PP) was the first synthetic stereo-regular thermoplastic polymer for industrial application, because of its outstanding properties and low cost, and it is now widely used in our daily life. 1 PP is easily flammable due to its chemical constitution, so flame retardancy is an important parameter for improving the performance of PP. 2 This is essential for delaying the production and spreading of flames or fires. To improve PP’s competitiveness in engineering, the resin application needs to be modified. Clay is one of the most studied nanomaterials in the recent 20 years. Elementary clay platelets consist of a 1-nm-thick layer made of two tetrahedral sheets of silica fused to an edge-shared octahedral sheet of alumina or magnesia. Stacking of the layers leads to a regular van der Waals gap between the layers called the interlayer or gallery. 3 The clay structure, with layers of high aspect ratio, imparts excellent barrier properties, which in return provides low gas permeability, enhanced chemical resistance, and flame retardancy.4,5 Due to the hydrophilic surface of the clay and the hydrophobic polymer, modification to the natural clay is essential in most polymer/clay composites to increase the surface compatibility. 6 Pristine clay is obtained through modifications made to organoclay using special surfactants. The surface energy, interlayer distance, and thermal stability of these modified clays strongly depend on the chemical structure and type of cation included in the surfactant. 7 Montmorillonite (MMT) is a naturally occurring 2:1 phyllosilicate, which can be defined as an aluminosilicate layer mostly present in clays and frequently used in the preparation of polymer nanocomposite. To ensure suitable intercalation, the surface of MMT platelets has to be modified.
There are several ways of modifying MMT, but the most common method is to use a cation; isomorphic cation substitution results in an excess of negative charges within the layer of inorganic cations (e.g. Ca2+, Na+, Mg2+) and can be exchanged with organic cations, rendering the silicate surfaces organophilic.8,9 The clay minerals have reactive hydroxyl groups that can react with silanol (Si–OH) groups of the hydrolyzed silane to form siloxane (Si–O–Si) bond. 10 Treatment of MMT with quaternary ammonium, phosphonium, and imidazolium ions is a widely used method, which allows the substitution of some metal ions present in the structures of MMT by cation exchange with large organic groups.11–13 In general, an interplay of entropic and enthalpic factors determines the outcome of whether an organically modified MMT will be dispersed, intercalated, or exfoliated in a polymer. Dispersion of MMT in a polymer requires sufficiently favorable enthalpic contributions to overcome any entropic penalties.14–16 Clay has many aspects of application value: incorporation of organic montmorillonite (OMMT) increases stress relaxation of natural rubber and nitrile rubber nanocomposites, the addition of nanoclay increases the thermal stability of the elastomer blends, incorporations of nanostructured sodium bentonite and sodium fluorohectorite reinforce the transport properties of natural rubber and carboxylated styrene butadiene rubber nanocomposites, and the addition of halloysite nanotubes for investigation of the uptake and toxicity will have better physiological effects.17–20 Samal et al. 21 reported of PP-based nanocomposites from quaternary ammonium salt modification sodium MMT and cloisite 15A using maleic anhydride-modified PP-grafted maleic anhydride (PP-g-MA) as a compatibilizer; this was observed to achieve excellent improvements in the mechanical properties, and nanocomposites showed higher thermal degradation temperature on heat loading. Attharangsan and Saikrasun 22 comparatively investigated PP and polyethylene (PE) filled with carbon nanotube (CNT) and OMMT as a heat stabilizing filler using thermogravimetric analysis (TGA); from TGA data and the calculated kinetic parameters, the incorporation of CNT and OMMT into PP and PE significantly improved the thermo-oxidative stability of the composites. Araújo et al. 23 reported four different types of quaternary ammonium salt modification clays. Incorporation of organoclay improved the thermal stability of PE, and the flammability resistance of PE/clay nanocomposites was improved due to the barrier effect of the organoclay during burning. PP/organic MMT composites and the resulting enhancement of thermal stability are closely related, not only due to the organic modifications of clay but also due to the organoclay content when using the same genres of clay under similar experimental conditions.
The main objective of the research reported in this article was to explore the influence of organoclay concentration on the thermal stability, crystallization behavior, morphology, and mechanical behavior of PP/PP-g-MA/ethylene acrylic acid (EAA)/organoclay composites. The introduction of a small amount of clay (≤5 wt%) has the potential to improve the barrier properties, thermal properties, mechanical properties, processability, and so forth.24–26 Hence, the compatibilized nanocomposites containing 1, 3, 5, and 7 wt% of organoclay were studied to reveal the effect of the filler concentration on the physical properties of such systems. Various organoclay contents in similar conditions were used as the reference samples. In this study, PP-g-MA and EAA were introduced into the PP matrix to enhance the interfacial interaction between PP and organoclay. Additionally, the mechanical properties of the prepared composites were systematically investigated to elucidate the effect of compatibilizers and organoclay content on the properties of composites.
Experiments
Materials
PP was obtained from China National Petroleum Corporation (Daqing Refining and chemical branch Co., Ltd, China); PP, nominal T30S, had a melt mass flow rate of 3.5 g/10 min; the PP pellets were dried in flowing air at 60°C, 12 h before use. OMMT was obtained from Nanocor, Inc. (Nanomer, PGN). OMMT, nominal I.44P was purchased. The OMMT was modified by alkylammonium salts with a melt flow index value of 30 g/10 min (190°C, 2.16 kg). PP-g-MA (grafting at 1%) were obtained from Arkema Group (Paris, France). EAA copolymer, with a content of acrylic acid of 20.5 wt%, was supplied by DuPont-Dow Chemicals. PP-g-MA and EAA were used as the compatibilizers. All materials were used without any further purification.
Preparation of PP-based nanocomposites
The purchased organoclay was prepared by cation exchange method according to the previous work. 27 The organoclay preparation procedures using organic quaternary ammonium salt are shown in Figure 1.
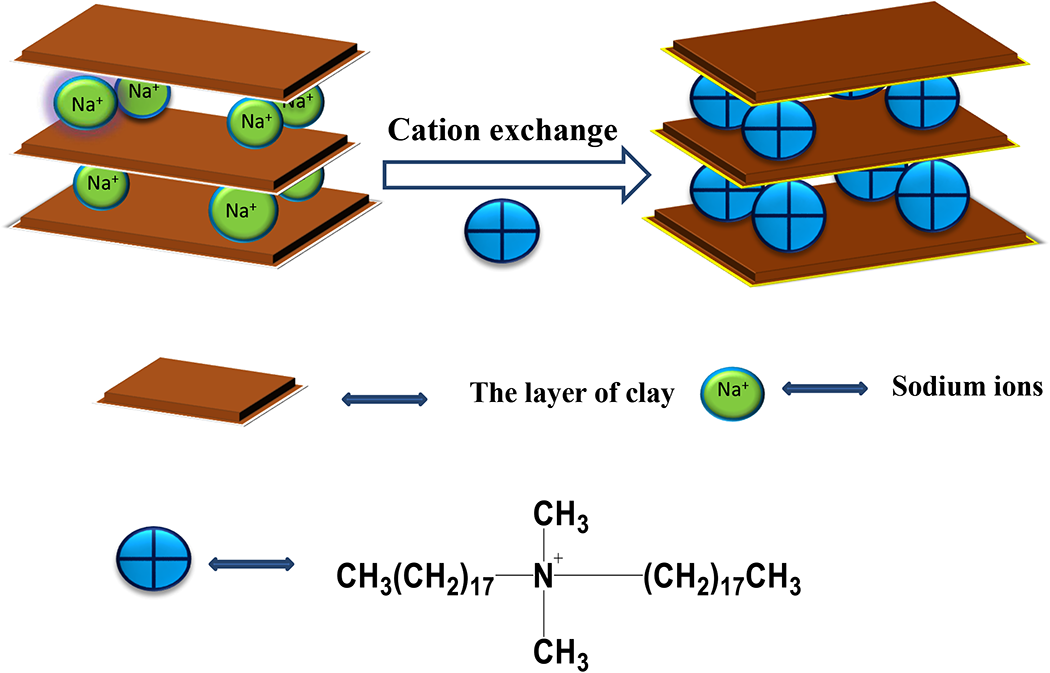
Preparation of intercalated functionalized organoclay by cation exchange method.
PP, PP-g-MA, EAA, and OMMT (1, 3, 5, and 7 wt%) were first dry mixed (shaken in a bag to combine), followed by melt-extruding with a twin-screw extruder (CTE-20, Coperion Machinery Co., China); the mass percentage of OMMT, PP-g-MA, and EAA was 1:1:1. The six heating zones were set to 170°C, 190°C, 210°C, 210°C, 215°C, and 210°C, and the screw speed was set at 240 r min−1. The purchased composites of OMMT were dried in a vacuum oven at 80°C for 12 h to remove any residual moisture, followed by molding on an injection molding machine (SE-130; DongHua Machinery Co., Ltd, China) at 210°C into various specimens for testing and characterization; the injection pressure was set at 120 bar, and the cooling time was set at 30 s. A flowchart of PP-based nanocomposite preparation for test is shown in Figure 2. 27
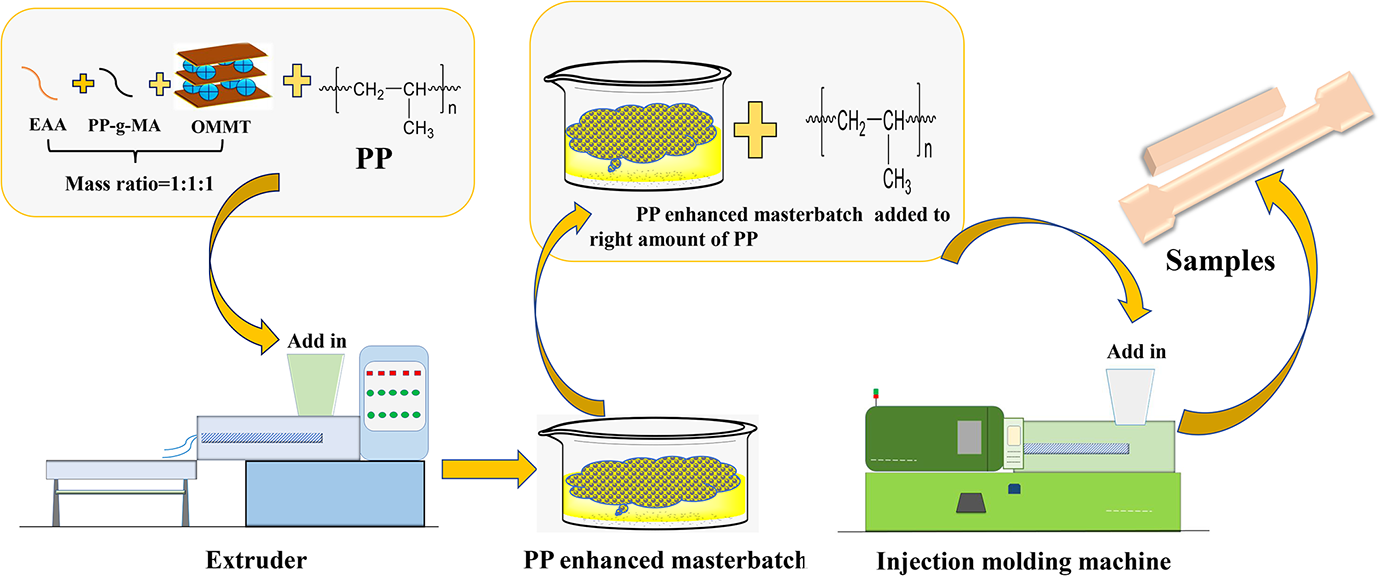
Scheme of the experimental for PP-based nanocomposites preparation.
Characterization methods
Characterization of thermal stability
The thermal stability of PP-based nanocomposites was investigated by TGA; TGA of the powders was performed on a TGA Q50 apparatus (TA Corporation New Castle, Delaware, USA) with a heating ramp of 10, 20, 30, and 40°C min−1 up to 700°C under flowing nitrogen (flow rate: 60 mL min−1). The mass of the samples used was approximately 9 mg.
Characterization of crystallization and melting behaviors
The crystallization behavior of the samples was investigated by differential scanning calorimetry (DSC) analysis (DSC Q10; TA Instruments Corp., USA) with a nitrogen flow rate of 60 mL min−1. About 6–8 mg of samples was accurately weighted and encapsulated in a typical aluminum pan. An empty sealed pan was used as the reference. It was conducted in isothermal crystallization procedures, in which the composite samples were initially heated to 200°C and held at 200°C for 5 min to erase the thermal history, and then cooled to 50°C at 10°C min−1. Finally, the samples were heated to 200°C again at 10°C min−1. The samples were tested in a random order to avoid any systematic error in the results. The crystallinity (χc) of the PP was calculated using the following two relationships. For neat PP28,29:
where ΔHm is the experimental heat of fusion (melting enthalpy) of crystallization obtained from the integral area of the melting thermogram and
where Wf is the weight percentage of the fillers in the composites.
Rheological measurements
The linear viscoelastic behavior of the PP-based nanocomposites was analyzed by a dynamic oscillatory rheometer in the melt state. A rotation rheometer (ARES-G2, TA Instruments Corp., USA) equipped with a 25-mm diameter parallel plate geometry was employed for the rheological tests. The samples were directly loaded and molded between the plates, and rheological tests were carried out at 190°C with a gap distance of 0.8–1 mm. The linear viscoelasticity test has a strain setting of 0.1% and a scanning frequency range of 0.1–500 rad s−1.
Characterization of morphology
To clarify the dispersion and morphologies of MMT in composites, a transmission electron microscope (TEM; JEM-1200EX, JEOL, Japan) was used and operated at an accelerating voltage of 120 kV. The samples for TEM observation were ultrathin-sectioned using a microtome equipped with a diamond knife. These sections (about 100 nm in thickness) were cut from Izod bars perpendicular to the flow direction and successively collected in a trough filled with water, followed by placing them on a 200-mesh copper grid.
Characterization of mechanics
Tensile and flexural tests were performed according to the ASTM D-638 and ASTM D-790 standard, respectively, using a universal testing machine (WDW-10C; Shanghai Hualong Test Instruments Co., Ltd, China) at room temperature. The crosshead speed was set at 50 mm min−1 for tensile tests and 2 mm min−1 for flexural tests. The specimen dimensions of tensile and flexural tests are shown in Figure 3. For each specimen, the data reported were the average of five to seven specimens. Notched Izod impact tests were performed on a ZBC-4 Impact Pendulum (Shenzhen SANS, China) at 21°C according to the ASTM D-256 standard. The notches (depth 2.5 mm and radius 0.25 mm) were machined after injection molding. Experimental errors were calculated from five specimens for the impact tests. The average deviations based on five specimens were of the order of 5%.
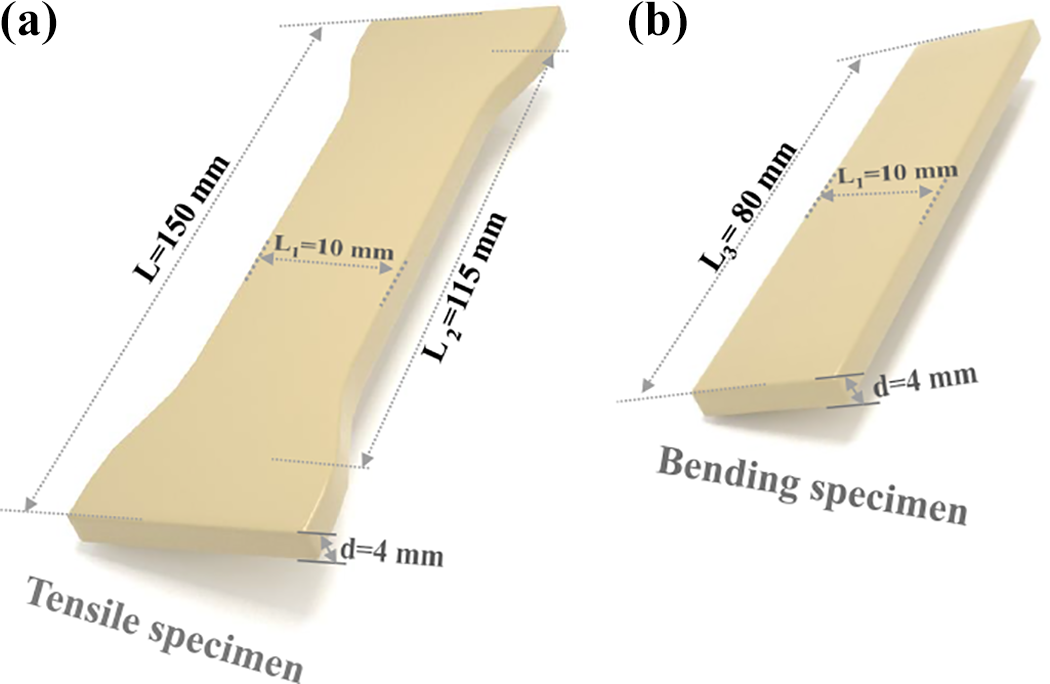
Specimen dimensions of (a) tensile and (b) flexural tests.
Results and discussion
Calculation of activation energy of nanocomposites
The activation energy of the PP/organoclay nanocomposite system is confirmed by the Kissinger method at different nanocomposite compounding ratios. First, the effect of OMMT content on the thermal stability of PP composite material was analyzed. The data at 10°C, 20°C, 30°C, and 40°C temperatures are listed in detail in Tables 1, 2, 3, and 4, respectively.
TG and DTG results of PP/EAA/PP-g-MA/OMMT nanocomposites (10°C min−1).
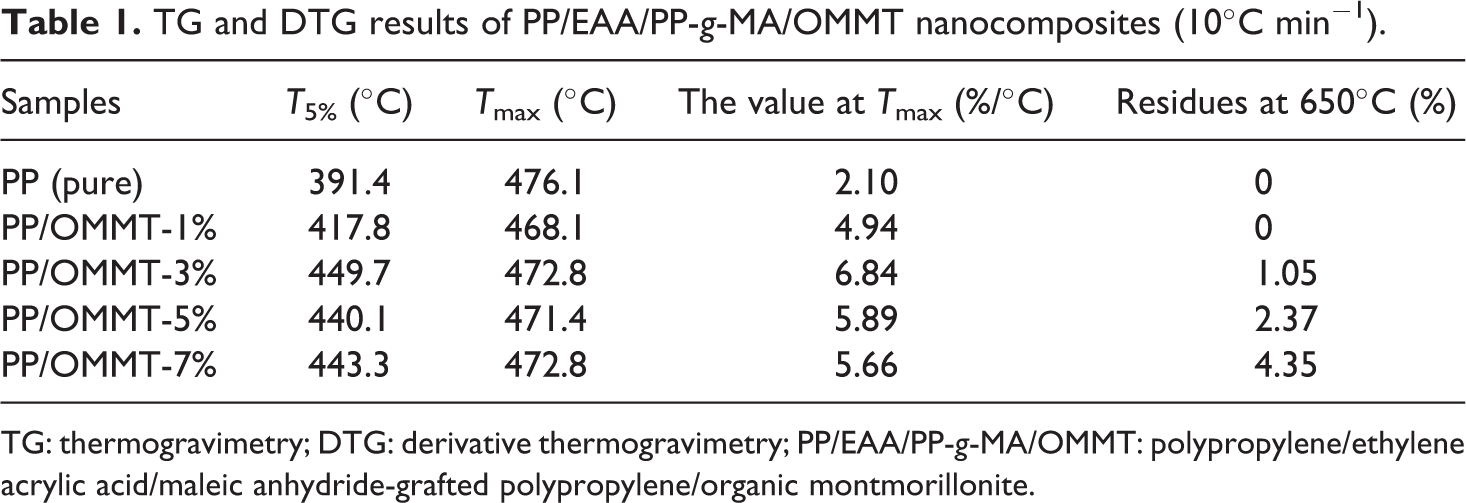
TG: thermogravimetry; DTG: derivative thermogravimetry; PP/EAA/PP-g-MA/OMMT: polypropylene/ethylene acrylic acid/maleic anhydride-grafted polypropylene/organic montmorillonite.
TG and DTG results of PP/EAA/PP-g-MA/OMMT nanocomposites (20°C min−1).
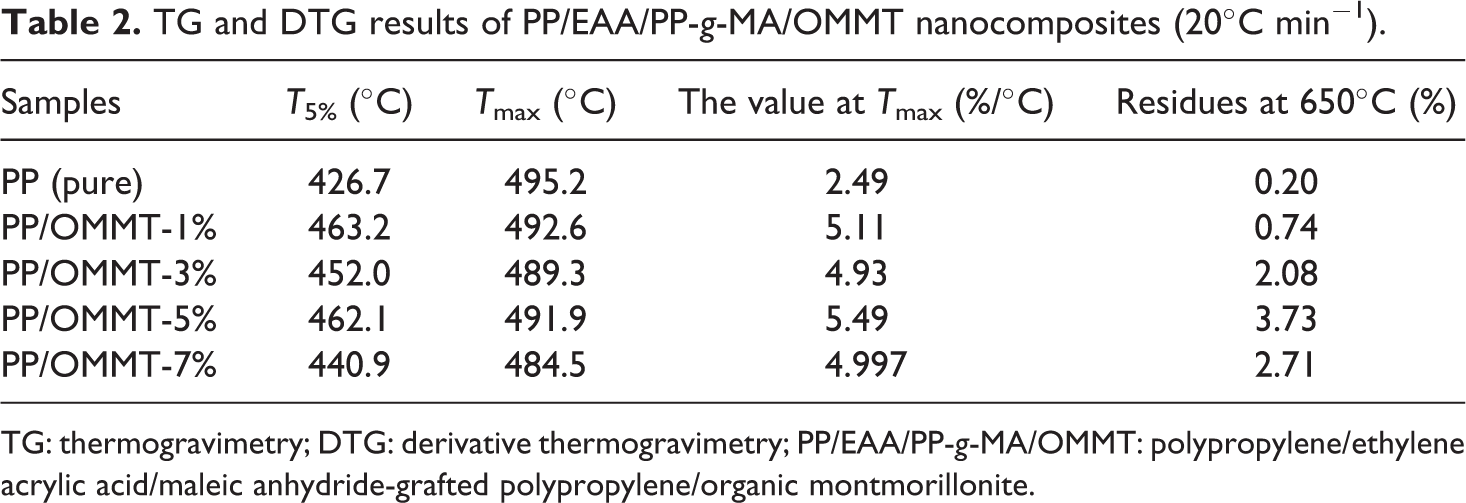
TG: thermogravimetry; DTG: derivative thermogravimetry; PP/EAA/PP-g-MA/OMMT: polypropylene/ethylene acrylic acid/maleic anhydride-grafted polypropylene/organic montmorillonite.
TG and DTG results of PP/EAA/PP-g-MA/OMMT nanocomposites (30°C min−1).
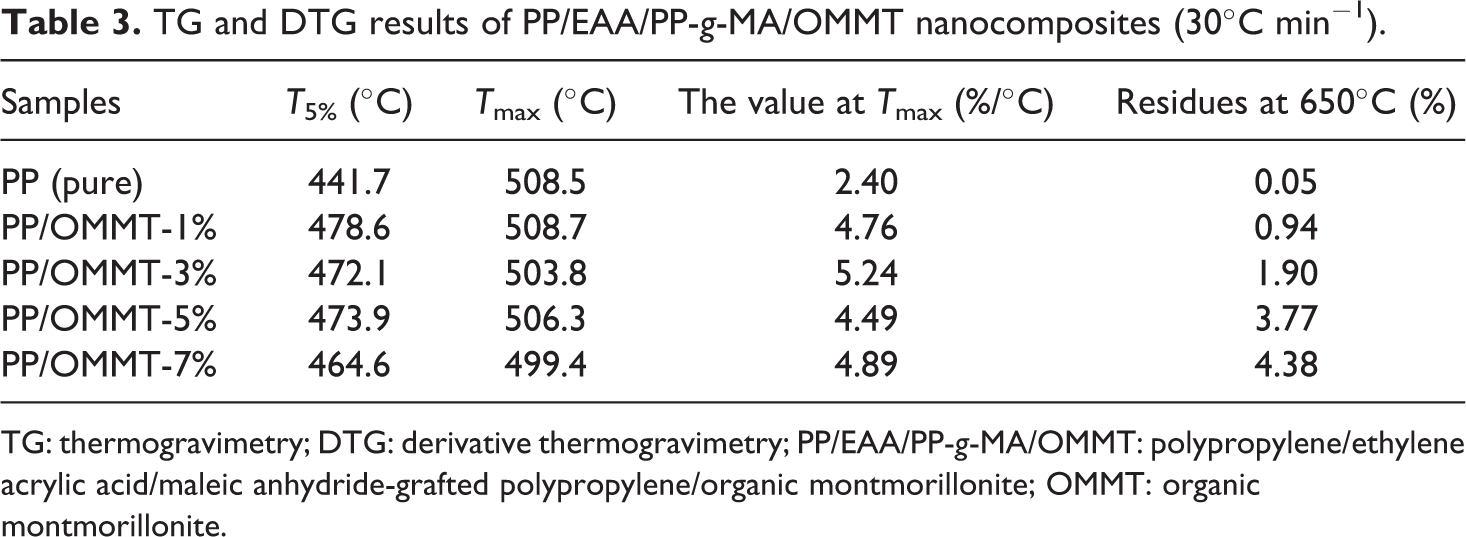
TG: thermogravimetry; DTG: derivative thermogravimetry; PP/EAA/PP-g-MA/OMMT: polypropylene/ethylene acrylic acid/maleic anhydride-grafted polypropylene/organic montmorillonite; OMMT: organic montmorillonite.
The TG and DTG results of PP/EAA/PP-g-MA/OMMT nanocomposites (40°C min−1).
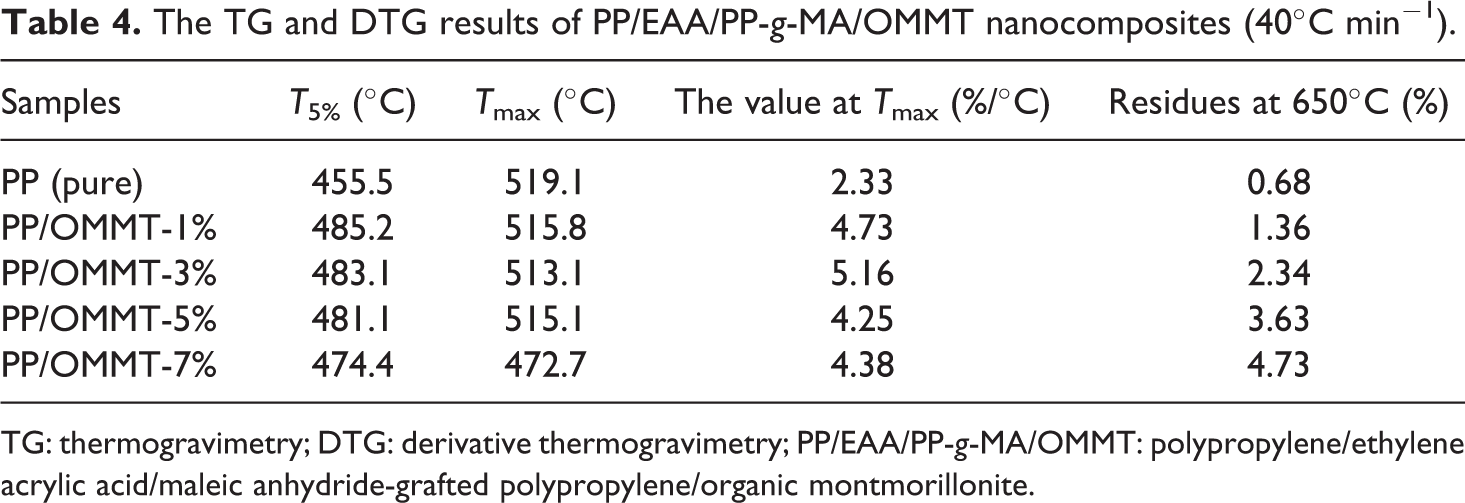
TG: thermogravimetry; DTG: derivative thermogravimetry; PP/EAA/PP-g-MA/OMMT: polypropylene/ethylene acrylic acid/maleic anhydride-grafted polypropylene/organic montmorillonite.
Tables 1 to 4 show that neat PP itself has higher temperature properties: its T5% temperature increases with increasing heating ramp can reach approximately 391.4–455.5°C, Tmax can reach approximately 476.1–519.1°C, and the residual after thermal degradation is approximately 0–0.68%, indicating that PP itself is not char. The quality was loss of a gas phase substance. The addition of OMMT firstly increased T5% of the PP material: as the fraction of OMMT increased (1, 3, 5, and 7), T5% of the PP/EAA/PP-g-MA/OMMT composite was delayed at approximately 25–45°C. As the heating ramp increased, the overall Tmax is about 5–10°C ahead of time, which is almost no change, indicating that the thermal stability of PP added to OMMT was slightly increased. This is mainly because during the processing, OMMT decomposes after the matrix, and its decomposition products defer the decomposition of the PP matrix. Meanwhile, we know that the temperature T5%, Tmax, and residues of a large amount of clay (>5 wt%) for PP/organoclay nanocomposites may slightly decrease when the clay content is 7 wt%.
In addition, we use the Kissinger method to calculate the activation energy of the composite. 30 The principle of this method is calculated based on the temperature corresponding to the peak value (Tmax) of the DTG curve at different heating rates as

In equation (3), Tmax is the maximum thermal weight loss rate temperature (K), A is pre-factor, and R is the gas constant equation to 8.314 J (mol·k)−1.

Plotting equation (4) shows that the activation energy E can be obtained by plotting
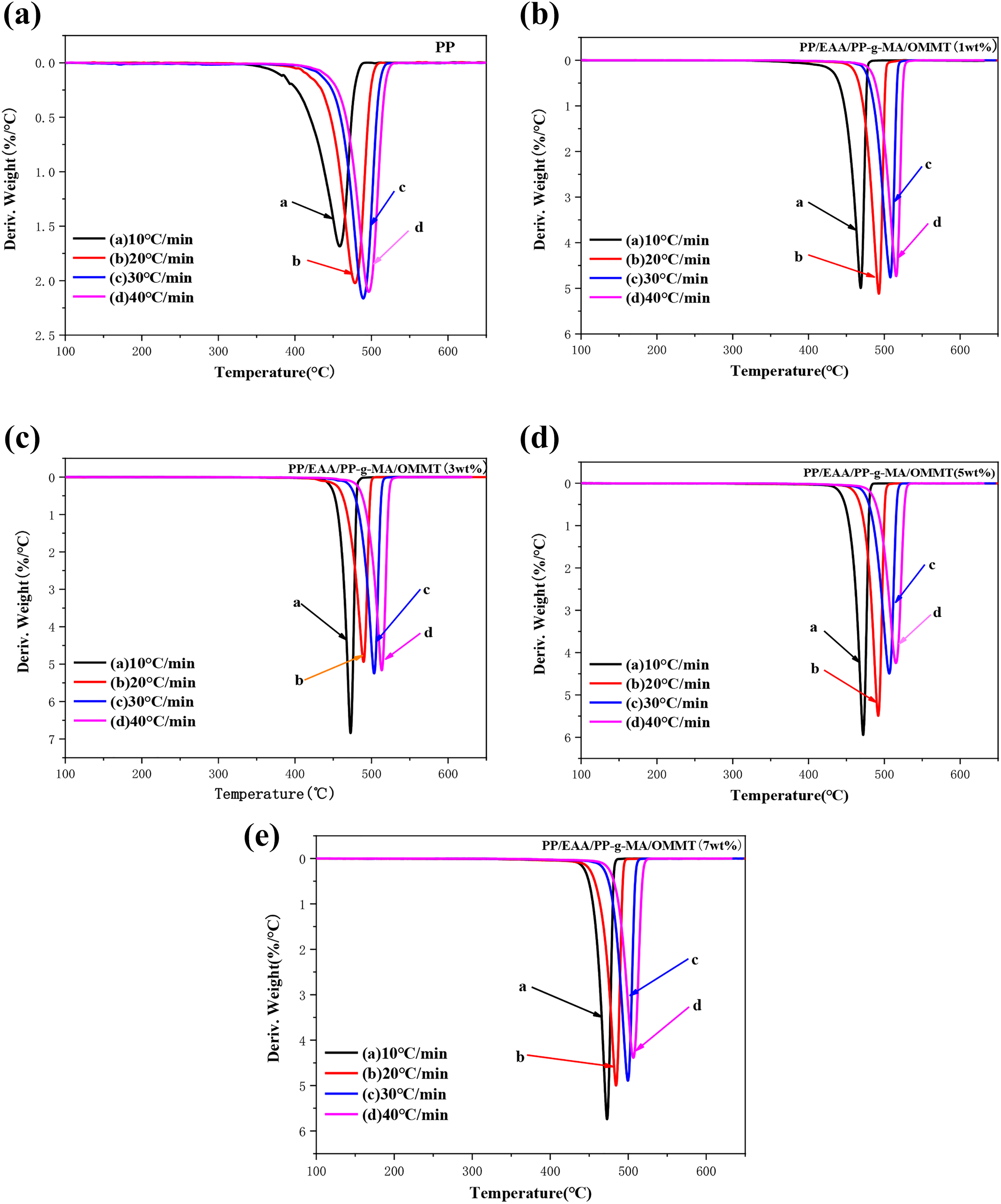
(a to e) DTG curves of PP, and each component PP/EAA/PP-g-MA/OMMT nanocomposites at different heating rates under nitrogen atmosphere.
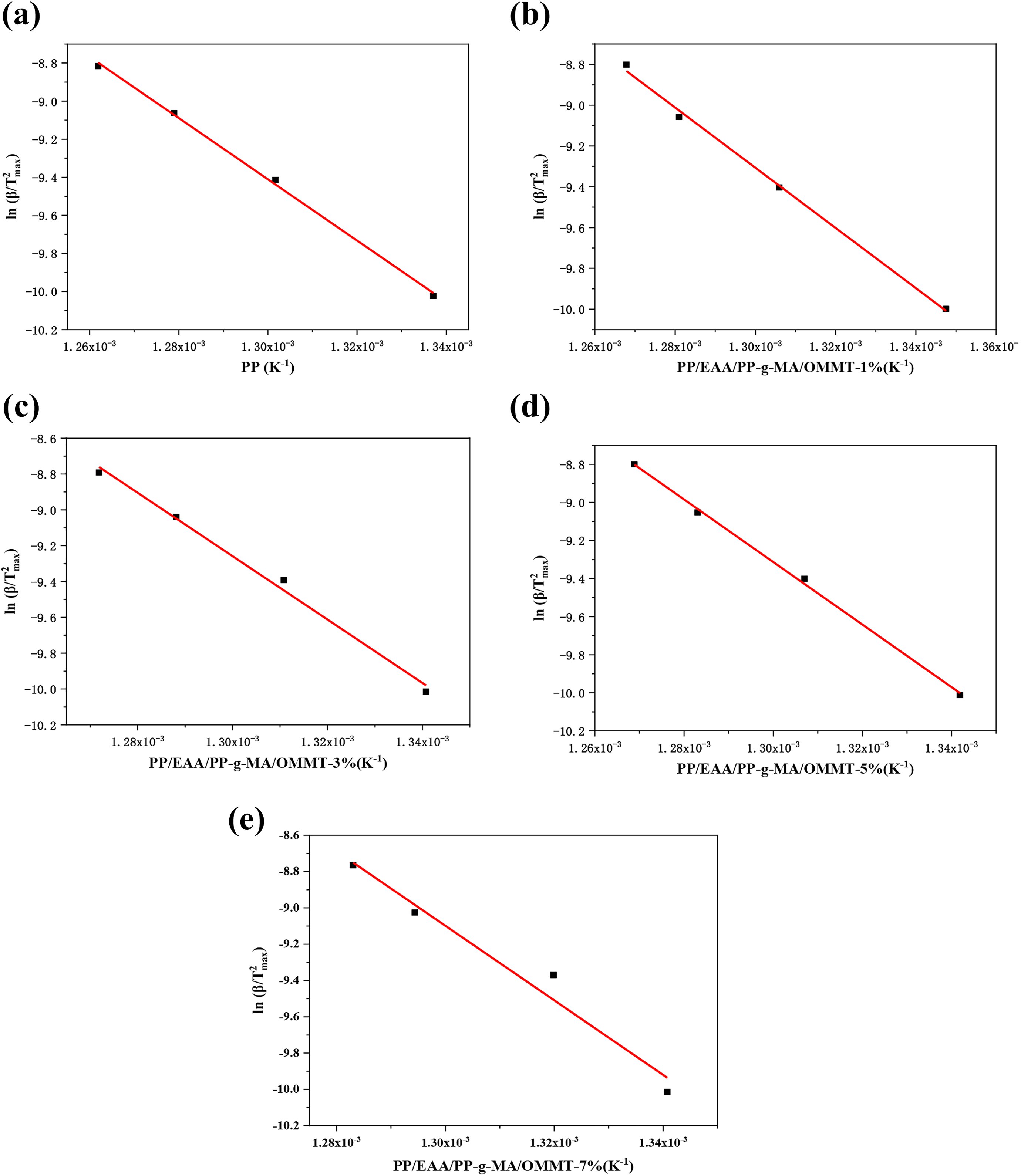
(a to e) Kissinger linearity diagram of the composite at different heating rates for pure PP and PP/EAA/PP-g-MA/OMMT nanocomposites.
Figure 5 shows the
Table 5 shows that the activation energy of neat PP is at 133.79 kJ mol−1. For the PP/EAA/PP-g-MA/OMMT composite system, the activation energy E of the reaction shows a decreasing trend first and then increases. It is shown that during the degradation process of the composite material, the degradation of OMMT forms a carbon layer on the surface of the substrate to cover the surface of the substrate, which delays the decomposition of the matrix. But when OMMT amount is 1 wt% the activation energy is lower than neat PP, indicating that the carbon layer formed in the PP/EAA/PP-g-MA/OMMT composite is not dense enough to function as a thermal barrier. The alkylammonium cations in the organoclay could suffer thermal degradation following the Hofmann mechanism, which leads to volatilization or degradation of the corresponding olefin matrix. 31 As regards the PP matrix, PP-g-MA and EAA affect the PP matrix and OMMT to enhance the thermal stability of PP nanocomposites. This addition of EAA and PP-g-MA promoted the insertion of PP molecular chains between OMMT platelets; the enhanced thermal stability is attributed to the strong interaction of the PP matrix and OMMT surface through chemical linkage between compatibilizer and OMMT, which in turn mediates the surface polarity of the clay and clay–polymer interface. 21 Analysis of combined TGA data shows that the binding effect prevents PP random chain scission, and the OMMT platelets exhibit better reinforcing effect. The effects of EAA/PP-g-MA on the PP-based nanocomposites are shown in Figure 6.
Activation energies of pure PP and PP/EAA/PP-g-MA/OMMT nanocomposites at different weight loss rates as determined by Kissinger method.
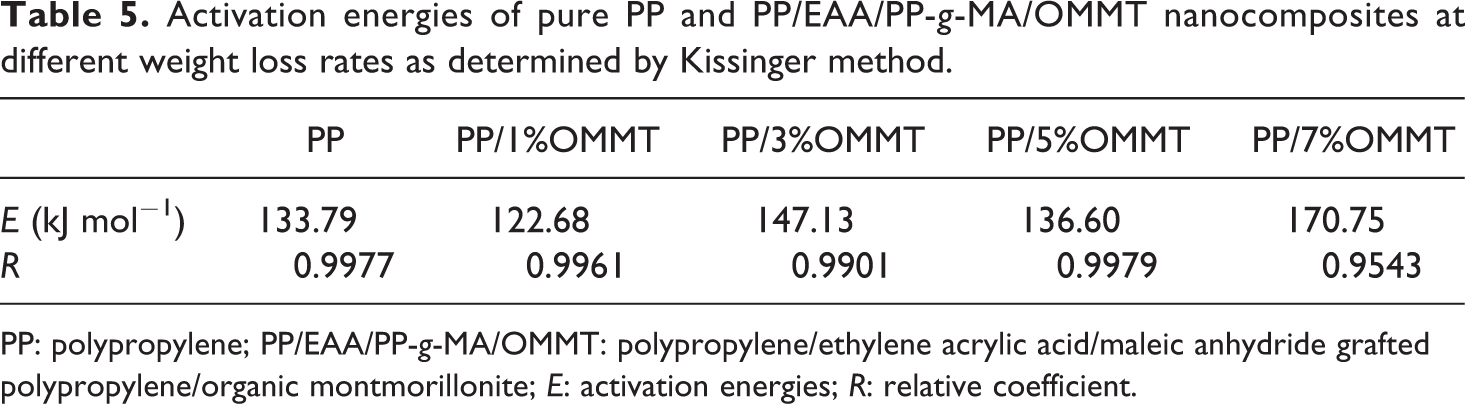
PP: polypropylene; PP/EAA/PP-g-MA/OMMT: polypropylene/ethylene acrylic acid/maleic anhydride grafted polypropylene/organic montmorillonite; E: activation energies; R: relative coefficient.
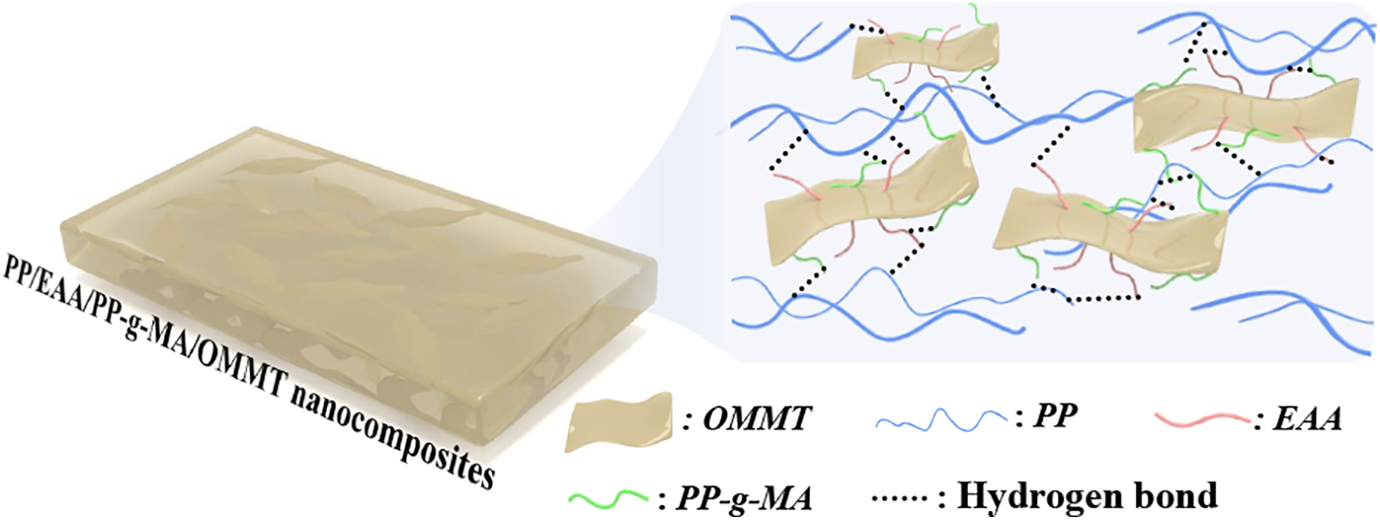
Schematic representation showing effect of compatibilizer on the PP-based nanocomposites.
When the OMMT addition amount is 3, 5, and 7, the activation energy is higher than the activation energy of neat PP, indicating that the compact carbon layer formed by the thermal degradation of the composite material under this component can well protect the matrix and delay the decomposition. When the OMMT content in PP/EAA/PP-g-MA/OMMT composites is 5 wt%, the activation energy decreases, which is considered to be the high activation degree of the thermal degradation reaction of the system. The reason is that OMMT is heated up to form a reticular silicate structure and ablation recombination, which is conducive to the formation of a graphite structure of carbon layer. To protect the overall thermal degradation stability of the system, the system needs to form a more compact carbon layer, blocking the heat transfer and diffusion of small combustible substances. The real mechanism of increase in thermal stability may be attributed to the degradation of OMMT forming a carbon layer on the surface of the substrate to cover the surface of the substrate, which delays the decomposition of the matrix. Meanwhile, the addition of EAA and PP-g-MA promotes insertion of PP molecular chains between the OMMT platelets, which makes OMMT to disperse more uniformly in the matrix, and the interface between PP matrix and OMMT to become stronger.
Crystallization and melting behaviors
The crystallization behavior of PP/EAA/PP-g-MA/OMMT composites was investigated by DSC analysis. Figures 7 and 8 show the DSC melting and cooling curves, respectively, of the PP/EAA/PP-g-MA/OMMT composites. From these curves, several parameters can be obtained such as melting temperature (Tm), which was defined as the maximum temperature of transition endothermic curve from second heating scan of DSC melting curves (see Figure 7). Tm was evaluated from the second scan to ensure that the results were free from prior thermal history effects. Crystallization temperature (Tc) was defined as the maximum temperature of the transition exothermic curve (see Figure 8). The samples were used to study the isothermal crystallization behavior of the nanocomposites. All the parameters are summarized in Table 6.
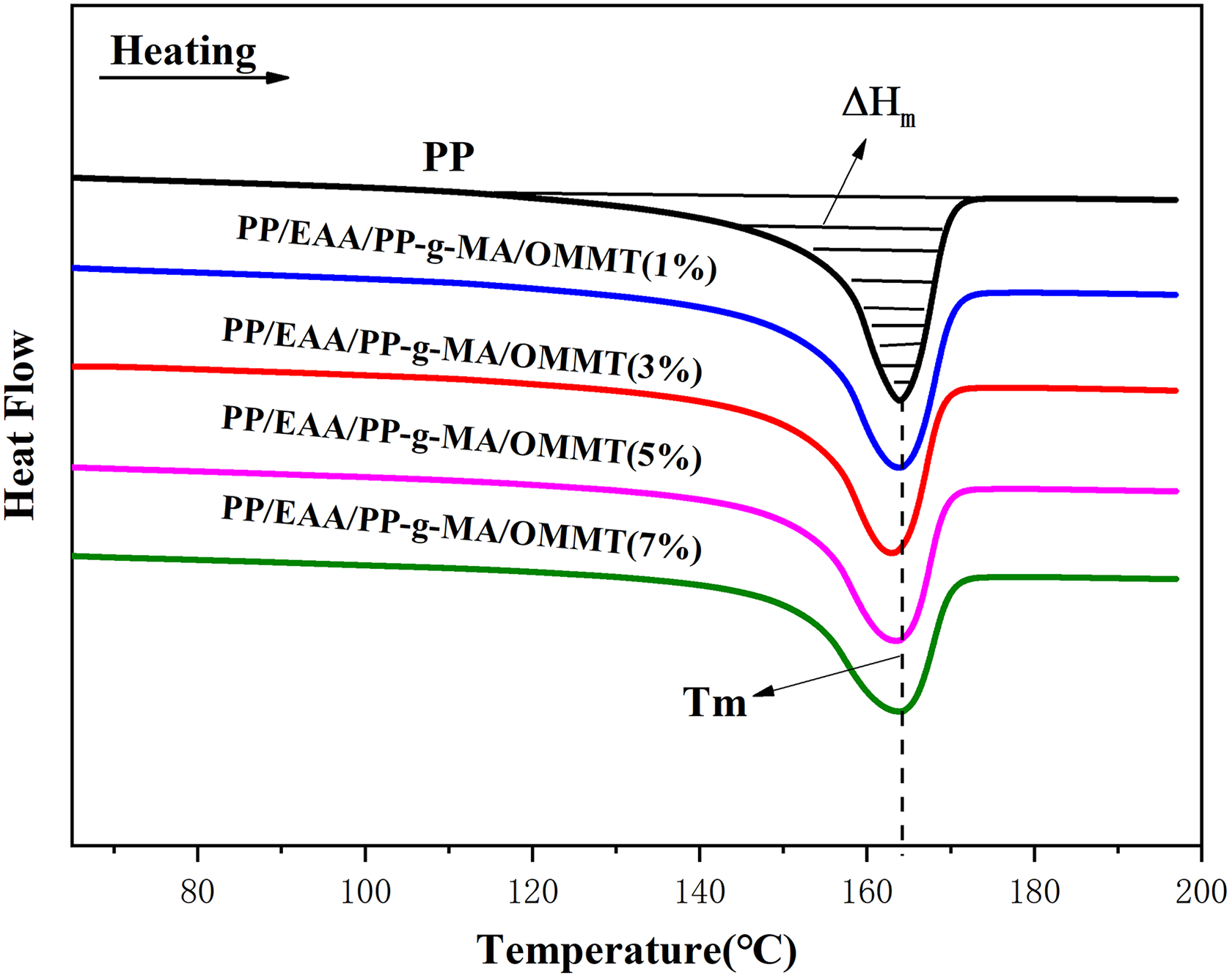
Heating scans of pure PP, and the PP/EAA/PP-g-MA/OMMT nanocomposites with different OMMT loadings.
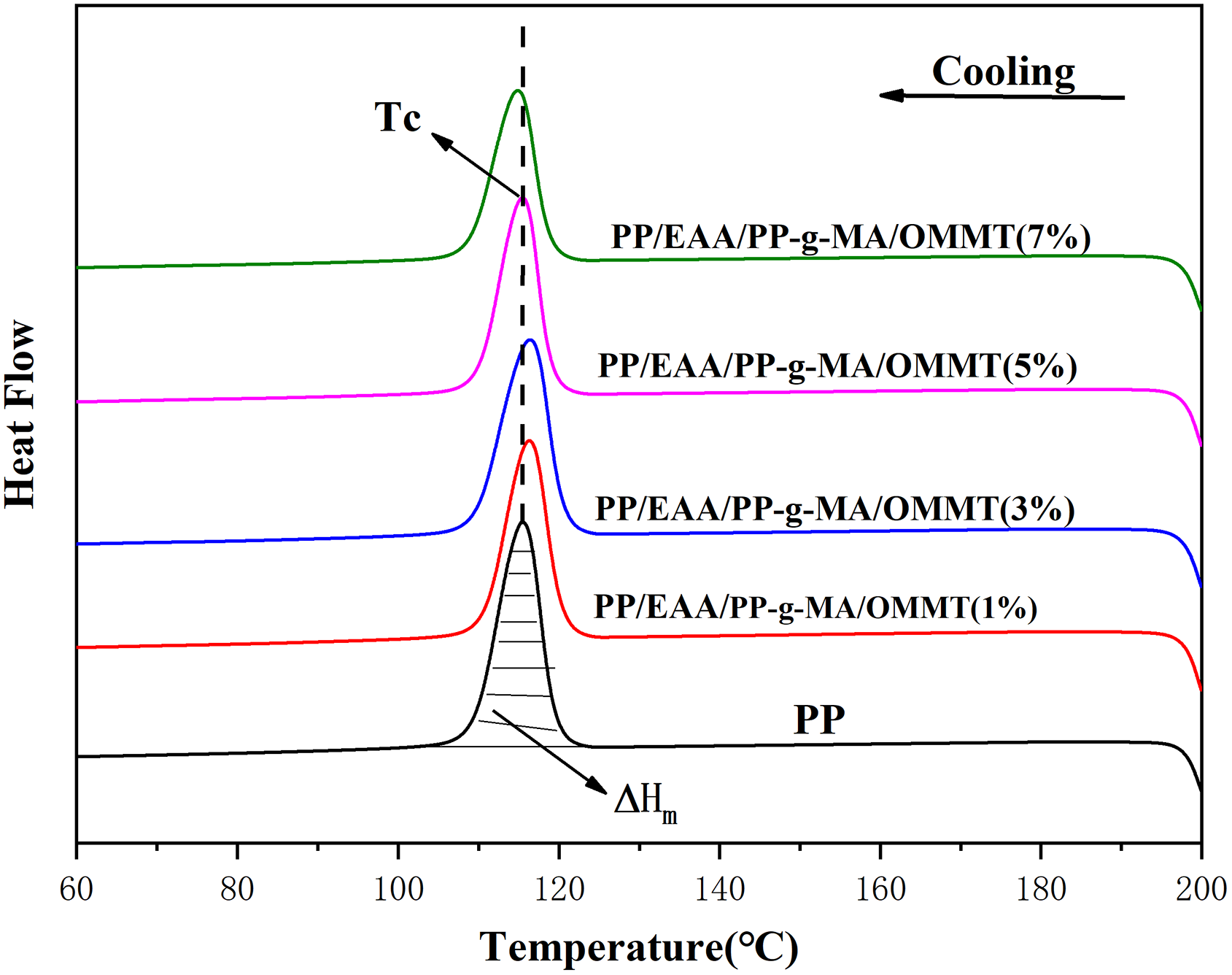
Cooling scans of pure PP, and PP/EAA/PP-g-MA/OMMT nanocomposites with different OMMT loadings.
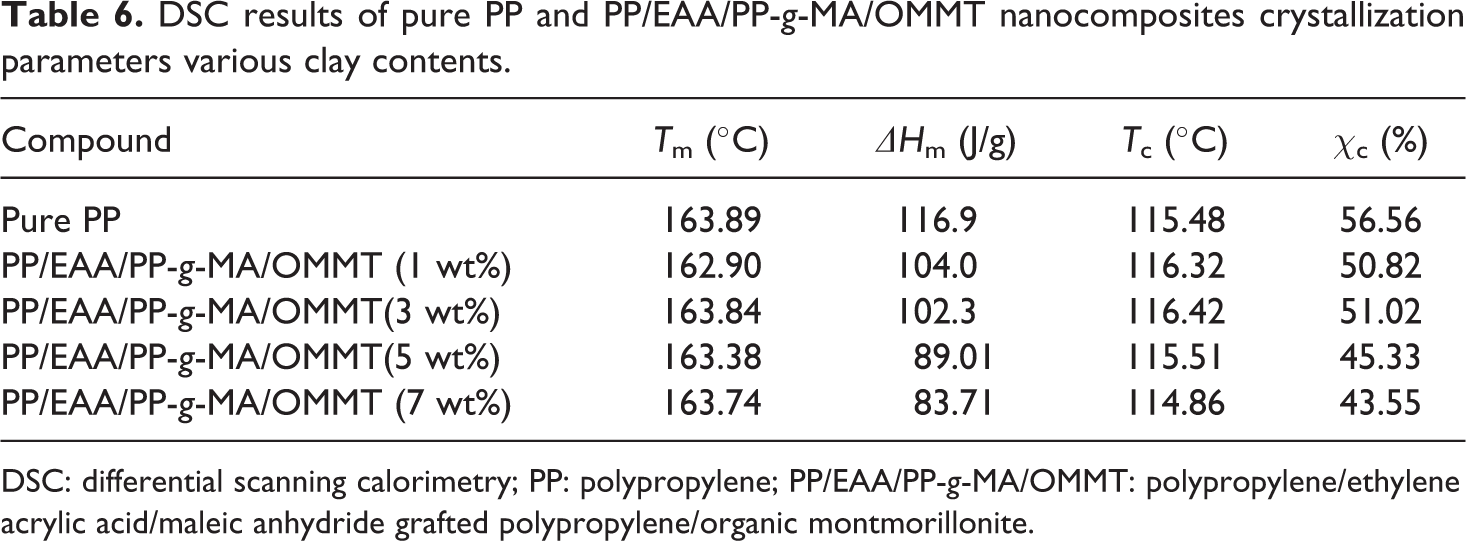
DSC results of pure PP and PP/EAA/PP-g-MA/OMMT nanocomposites crystallization parameters various clay contents.
DSC: differential scanning calorimetry; PP: polypropylene; PP/EAA/PP-g-MA/OMMT: polypropylene/ethylene acrylic acid/maleic anhydride grafted polypropylene/organic montmorillonite.
Table 6 shows that the melting temperature of PP/EAA/PP-g-MA/OMMT composites is not significantly affected by changes in the OMMT content. The similar result is reported in other literatures.32,33 The total heat of fusion (ΔHm) of PP, estimated by the area integration of the melting peaks under the endothermic region of the DSC thermograms, decreases from the incorporation of OMMT, EAA, and PP-g-MA, indicating that the degree of crystallinity is decreased with the addition of OMMT, EAA, and PP-g-MA. On the other hand, the percentage of crystallinity (χc) of all the nanocomposites samples is lower than that of neat PP (Table 6). This decrease can be attributed to OMMT platelets which act as a physical obstruction to the diffusion of the polymer molecules and the growing crystallite and impede the local lamellar crystallization. 34 However, very slight increase of χc is reached at 3 wt% of OMMT content. Thereafter, increasing the OMMT content resulted in a gradual decrease in χc (Table 6). The explanation for such behavior is that this increase can be attributed to the dispersed organoclay platelets in the PP matrix, which enhances the crystallization process of PP by acting as a “nucleating agent”. 35 However, the lowest amount (1 wt%) of OMMT does not have sufficient EAA and PP-g-MA to bind the PP molecular chains, enhancing the crystallization process of the PP nanocomposites. The amount of OMMT added is not obvious to the molecular chain of PP, so it has less influence on its crystallinity.
Figure 7 shows the melting curves of pure PP and its nanocomposites. For PP and its compounds a distinct peak is observed. The melting endotherm in the temperature range of 120–170°C is attributed to the melting of the original crystallites formed at the isothermal crystallization temperature Tm. As shown in Figure 8, a composite consisting of PP and OMMT exhibits remarkable Tm that is almost constant at about 163.5°C, compared with that of pure PP. The increase in Tm and the decrease in χc are more pronounced in the presence of EAA and PP-g-MA (Figure 7 and Table 6). This is due to the high level of dispersion of OMMT platelets in the PP matrix. Figure 8 shows the crystallization temperature. All the samples show an exothermic peak that can be correlated with the crystallization of PP in each sample. In addition, the corresponding temperature is known as the crystallization temperature Tc. It is clear from Figure 8 that the exothermic curve in the temperature range of 100–130°C is shifted to the higher temperature region reached at 1 wt% and 3 wt% of OMMT content in the PP matrix. Thereafter, increasing the OMMT content resulted in a gradual shift to the lower temperature region. This is probably because the addition of EAA and PP-g-MA promotes dispersion of OMMT platelet in the PP matrix and makes the PP crystal grains more perfect, thereby increasing the melting temperature of the composite. Moreover, after the addition of the compatibilizer, the compatibility between the OMMT platelet and the PP matrix is good, which limits the crystal nucleation of the PP matrix and reduces the crystallization temperature. As the amount of OMMT increases, the OMMT platelet which restricts PP crystal nucleation also increases, while the crystallization temperatures do not significantly alter.
Morphological studies
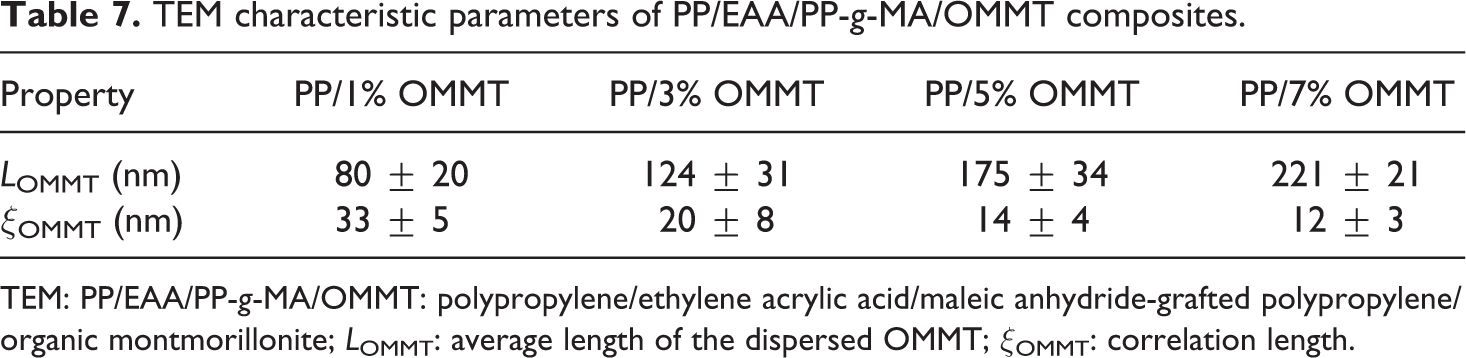
TEM analysis was carried out for more supportive information about the state of dispersion of clay in the nanocomposites and microstructures of the nanocomposite material. Figure 9(a) to (d) shows TEM micrographs of the inner surface of the nanocomposites at magnifications of 10,000× and 200,000×, in which the dark line represents the silicate layers, while the brighter area stands for the PP matrix. Relatively, distribution and dispersion were bad for clay platelets with increased clay contents in the polymer matrix (see Figure 9), but had better overall dispersion of clay in the PP matrix. There are still some areas that show a large aggregate with clay contents increased, and most clay platelets show fairly poor dispersion in the polymer matrix. This full dispersion is probably why EAA and PP-g-MA are critical for facilitating the insertion of PP between the OMMT platelets, which consequently acts as an obstacle for mobility and flexibility of the polymer chains to fold and join the crystallization growth. 36 This could be attributed to the high shear stress of the polymer melt during processing and surface modification of nanoclay particles. Thus, using masterbatch yielded a good distribution of OMMT in the composite. 33 The change in correlation length (ξOMMT) and the average length of the dispersed clay (LOMMT) layers are analyzed from the TEM images using Image J software and are tabulated in Table 7. The correlation length of the dispersed nanoclay was found to be the maximum for 1 wt% of nanoclay loading (33 nm). This suggests that the nanocomposite with 1 wt% of OMMT has the maximum dispersion with a partially exfoliated structure in the blend matrix. With increase of OMMT loading for 3, 5, and 7 wt%, the correlation distance decreases to 20, 14, and 12 nm, respectively. The increase in the average length of the dispersed OMMT platelets show that as the OMMT loading is increased, OMMT particles get aligned over so as to increase the effective length of OMMT. 37 Owing to the strong interaction between hydroxylated edge–edge groups, OMMT particles are sometimes flocculated in the PP matrix. As a result of this flocculation, the length of the clay particles increases enormously and hence the overall aspect ratio. 3

TEM micrographs of the nanocomposites of PP/EAA/PP-g-MA/OMMT composites with different OMMT contents: (a) 1 wt%, (b) 3 wt%, (c) 5 wt%, and (d) 7 wt%.
TEM characteristic parameters of PP/EAA/PP-g-MA/OMMT composites.
TEM: PP/EAA/PP-g-MA/OMMT: polypropylene/ethylene acrylic acid/maleic anhydride-grafted polypropylene/organic montmorillonite; LOMMT: average length of the dispersed OMMT; ξOMMT: correlation length.
Rheological characteristics of nanocomposites
Figure 10(a) and (b) shows the storage modulus (G′) and loss modulus (G″) of pure PP and PP-based nanocomposites as a function of the scanning frequency. It can be seen from the figure that at a higher frequency, the G′ and G″ of the composite material are greatly reduced and decrease with the increase of OMMT content. However, at lower frequencies, the difference between G′ and G″ is small as both are gradually increasing. The increase is very slight at higher frequency of G′ and G″ reached at 3 wt% of OMMT content.
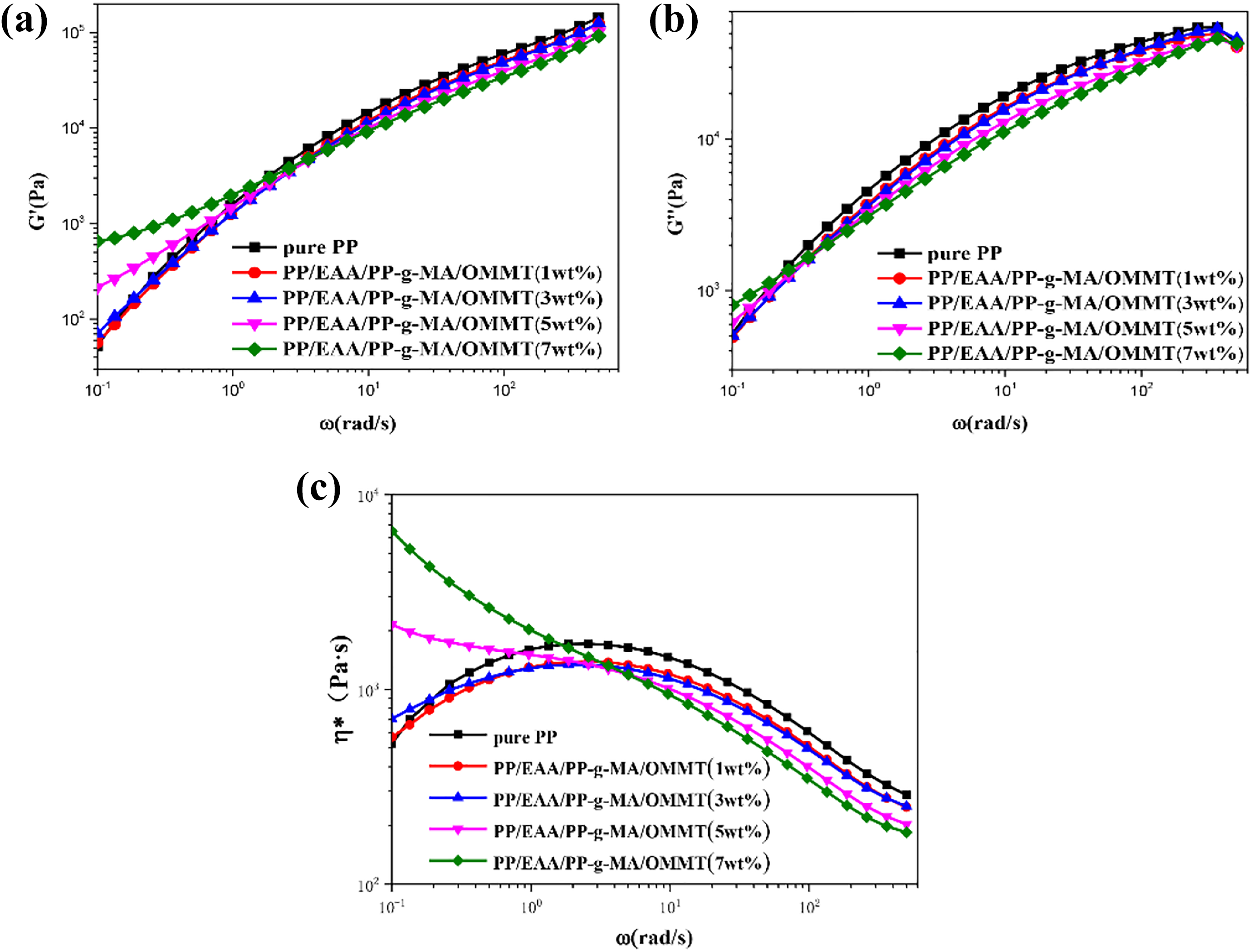
Frequency dependence of (a) storage modulus (G′), (b) loss modulus (G′′), and (c) complex viscosity (η*) for pure PP, and PP/EAA/PP-g-MA/OMMT composites with different OMMT contents.
The main reasons for the decrease of G′ and G″ at higher frequency of PP/EAA/PP-g-MA/OMMT nanocomposites are as follows: this may be part of the quaternary ammonium salt molecules insertion MMT interlayer and coating in the MMT laminates in PP-based composites, as well as the strong capacity increasing effect of EAA and PP-g-MA. Under the shear effect, the PP molecular chain and OMMT laminates in the composites are prone to slip, resulting in the reduction of G′ and G″. When the clay content of PP-based nanocomposites is 3 wt%, G′ and G″ increase, which may be because the increase in the content of MMT to 3 wt% is more conducive to the intercalation of macromolecular chain into OMMT laminates. The MMT laminates restrict the movement away from the macromolecules and increase the restriction effect, so G′ and G″ increase. In addition, the polar end groups of EAA and PP-g-MA interact with the layered silicates aggravate the limiting effect. Therefore, the modulus of PP matrix composites containing 1 wt% OMMT is the same or slightly lower than that containing 3 wt% OMMT. Likewise, these are the main reasons for the increase of G′ and G″ at the lower frequency of PP/EAA/PP-g-MA/OMMT composites. Figure 10(c) shows the complex viscosity (η*) of pure PP and PP-based nanocomposites as a function of the scanning frequency. It can be seen from the figure that at a higher frequency, η* of the composite material is greatly reduced. The results show that the addition of PP-g-MA and EAA is beneficial to reduce the melt viscosity of PP-based nanocomposites, improve the fluidity of the composites, and improve the processing properties of PP-based nanocomposites. With the increase of OMMT content, η* at low frequency increases remarkably, which fully reflects the polar end groups of EAA and PP-g-MA interacting with layered silicates that aggravate the limiting effect.
Mechanical properties
The mechanical properties of the neat PP and PP/EAA/PP-g-MA/OMMT nanocomposites at various contents of OMMT are shown in Figure 11.
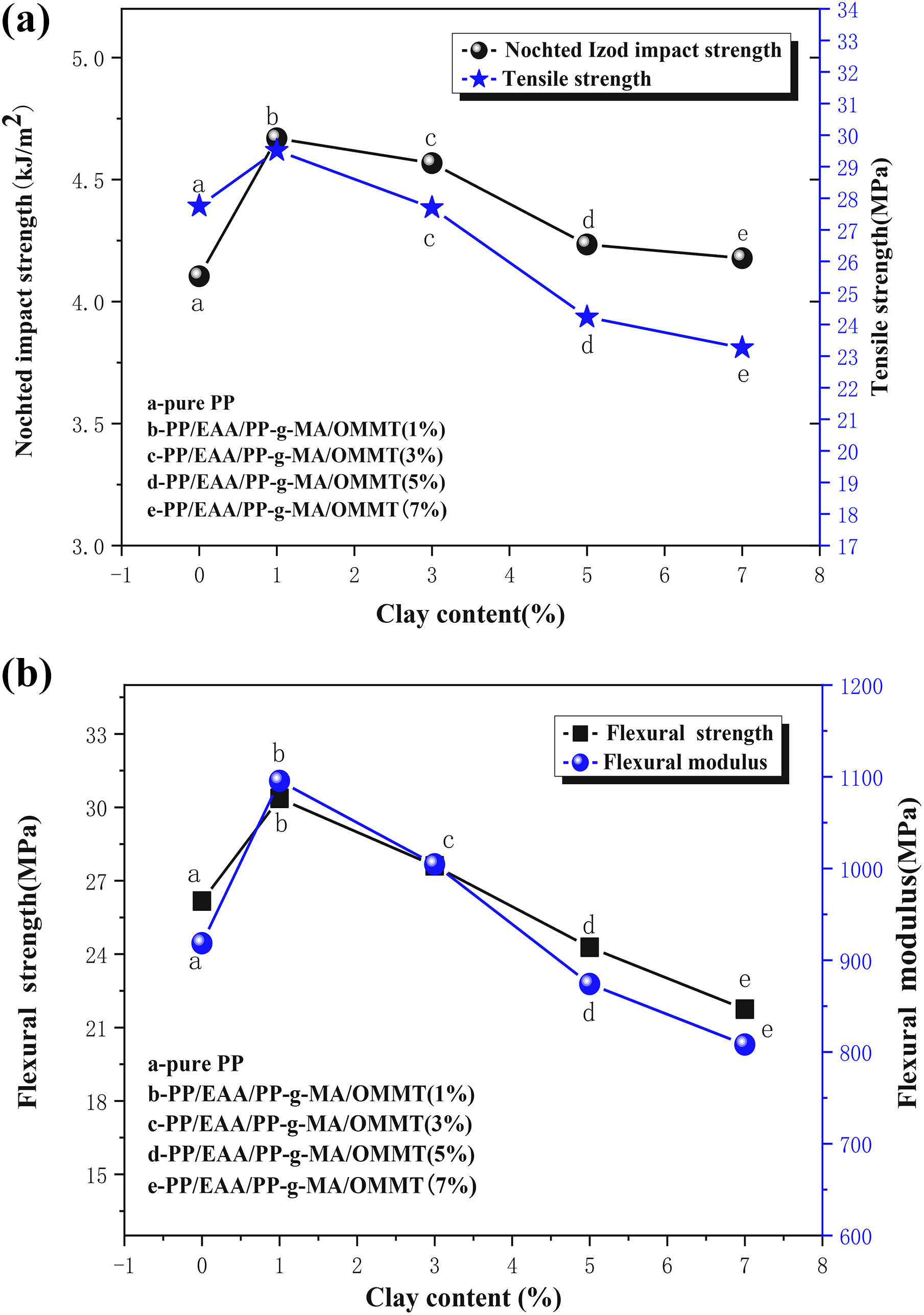
The mechanical properties for pure PP and PP/EAA/PP-g-MA/OMMT composites with different OMMT loadings: (a) notched Izod impact strength and tensile strength of the nanocomposites and (b) flexural strength and flexural modulus of the nanocomposites.
As seen in Figure 11(a), incorporation of OMMT into the PP matrix is found to enhance the tensile strength by acting as reinforcing filler because of its high aspect ratio and platelet structure. The PP/EAA/PP-g-MA/OMMT nanocomposite exhibits higher tensile strength, but increasing OMMT content results in a gradual shift to the lower tensile strength. The tensile strength shows an increasing trend with increasing amounts of OMMT up to 1 wt% and then decreases. With 1 wt% of OMMT, the nanocomposites having higher tensile strength value are related to contribution of better dispersion of OMMT in the nanocomposites. Besides, the compatibilizers can promote dispersion of OMMT into the PP matrix. When clay contents are up to 1 wt% the tensile strength decreases, which could be due to the aggregation or excess silicate layers dispersed in the matrix affecting its homogeneity and, consequently, destroying adhesion between the polymer matrix and clay particles at higher loading, thus decreasing the tensile strength of the nanocomposites.
The impact strength of neat PP and its nanocomposites is shown in Figure 11(a). It is evident that the addition of OMMT increases the impact strength of the PP matrix. A similar trend to that of the tensile strength can be observed. This indicates that the toughness of the PP matrix is increased with the presence of OMMT and compatibilizers. Owing to the strong interaction between clay platelets and the PP matrix in the presence of compatibilizers, the strength of the disk–disk interaction plays an important role in determining the stability of the clay particles and hence the enhancement of tensile strength of the nanocomposites. 38 Besides, the notched Izod impact strength shows an increasing trend with increasing amounts of OMMT up to 1 wt% and then decreases. The toughness of the PP matrix is related to the dispersion and interfacial interactions between PP and OMMT in PP-based nanocomposites. With 1 wt% of OMMT, the dispersion of silicate layers in the PP matrix is relatively uniform, which is able to absorb more energy before breaking. Higher filler loading could inhibit the uniform dispersion of clay particles and inevitably form particle aggregates that would easily result in stress concentration in the neighboring matrix, thus initiating the crack growth and decreasing the impact strength. 39 These results indicate that separately dispersed nanoclay exerts influence on the tensile strength of the composites, and has a reinforcing effect in improving the impact strength of the nanocomposites.
Similar to the tensile strength and impact strength, shown in Figure 11(b), it is also found that the flexural strength and flexural modulus of the composites with compatibilizers were higher. Clay contents were 1 wt% flexural strength and flexural modulus showed an increasing trend, and then showed a steady linear decline with increasing clay content. These properties could be attributed to OMMT dispersion in PP matrix and excessive dosage of OMMT used made the aggregation of OMMT to take place and hence decline the flexural strength and flexural modulus.
Conclusion
PP/PP-g-MA/EAA/organoclay nanocomposites were obtained using the melt mixing technique. TGA test results show that the addition of OMMT makes the thermal stability of PP/organoclay nanocomposite to slightly increase. When the clay content is higher than 1 wt% calculated by the Kissinger method, the activation energy of the initial degradation of the nanocomposite is improved and the char forming behavior of the material for nanocomposites is promoted to some extent. This may be due to the low thermal stability of modified OMMT by alkylammonium salts leading to restacking of silicate layers accelerated for OMMT lower contents is 1 wt% in PP matrix decomposition. TEM studies indicated that the addition of compatibilizer led to a certain amount of overall good dispersion of OMMT in the PP matrix and nanocomposite with 1 wt% of OMMT has the maximum dispersion with partially exfoliated structure in the blend matrix. DSC analyses revealed that the addition of OMMT led to decrease in χc, but the melting temperatures of the polymer do not alter markedly with increase in the OMMT loading. The results of dynamic rheological studies show that OMMT, EAA, and PP-g-MA can significantly reduce the G′ and G″ at a higher frequency of the PP composite. The results indicate that the compatibilizers effectively increased the tensile strength, flexural strength, and flexural modulus of the nanocomposites by enhancing the high dispersion degree of OMMT. Improved OMMT dispersion can help to enhance the mechanical properties of the PP/organoclay nanocomposite. It was much dependent on the composition of the PP blends. OMMT can effectively improve the activation energy of PP matrix, and excess OMMT will gradually reduce the dispersion of matrix. The physical and chemical properties of PP/organoclay composites can be effectively improved by improving the dispersibility of OMMT and selecting suitable compatibilizers and intercalation agents with high thermal stability.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guizhou Province Science and Technology Plan Projects (Grant Nos: [2016]2812, [2017]5623, [2017]1091, [2018]1012, [2019]4022) and the Guiyang Baiyun district Science and Technology Plan Projects (Grant No.: [2017]65).