Abstract
Surface of pristine silica aerogel nanoparticles was hydrophobically modified with hexamethyldisilazane (HMDS). Then, the resultant modified nanoparticles were used for in situ atom transfer radical copolymerization of styrene and methyl methacrylate. Surface area and structural characteristics of the HMDS-modified silica aerogel nanoparticles (H-SANs) were examined by nitrogen adsorption/desorption isotherm. Evaluation of size distribution and morphological studies were also performed by scanning electron microscopy and transmission electron microscopy. Conversion and molecular weight determinations were carried out using gas chromatography and size exclusion chromatography, respectively. Adding of H-SAN by 3 wt% results in decrement of conversion from 98% to 79%. In addition, molecular weight of copolymer chains decreases from 20,372 g·mol−1 to 16,487 g·mol−1. However, polydispersity index values increases from 1.38 to 1.76. Linear increase of ln(M0/M) with time for all the samples shows that polymerization proceeds in a living manner. Increasing thermal stability of the nanocomposites is demonstrated by thermal gravimetric analysis. Differential scanning calorimetry shows a decrease in glass transition temperature from 67.4°C to 59.8°C by the addition of 3 wt% of the H-SAN.
Keywords
Introduction
Polymer nanocomposites generate a great deal of attention from many scientists in different areas because of their interesting features.1–3 Low loading of nanofillers in the neat polymer matrix leads to considerable improvement in several properties of nanocomposites in comparison with neat polymer and also conventionally filled composites.3,4 Nanocomposites combine the advantages of the inorganic part and also organic part. During the last decades, silica/polymer nanocomposites as a popular family of nanocomposites have attracted much attention.5–8 Synthesis of silica/polymer nanocomposites is generally performed by three main procedures: (i) direct mixing of polymers with silica particles or silica precursors; (ii) direct mixing of monomer or monomers with silica particles followed by polymerization; and (iii) mixing of monomer or monomers with silica precursors followed by simultaneously polymerization of monomers and precursors.7,9
Nanoporous materials have attracted much interest due to their applications in various fields such as catalysis, chromatography, separation, biomaterials, microelectronics, and photonic materials.10,11 Materials on account of their pore diameter are divided into four groups: microporous (pore diameter less than 2 nm), mesoporous (pore diameter ranged from 2 nm to 50 nm), macroporous (pore diameter more than 50 nm), and nanoporous materials with pore diameter less than 100 nm.12–14 Nanoporous silica aerogels, as their name implies, are comprised of less than 10% silicon dioxide and more than 90% air. 15 Nanoporous silica aerogels possess unique features such as ultralow densities, large surface area, high degree of porosity, and extremely low thermal conductivity.16,17 Aerogels are employed for many diverse applications such as commercial thermal and acoustic insulator, adsorbent, chromatographic packing, and so on.18,19
Although free radical polymerization (FRP) can provide many advantages, disability to control over molecular weights of the products, weakness to produce block copolymers, and (co)polymers with narrow molecular weight distribution are some drawbacks of this method.20–22 Circumventing of these problems can be performed using controlled radical polymerization (CRP). CRP as a popular producer pathway to produce well-defined polymers is mainly categorized into three major types, namely nitroxide-mediated polymerization (NMP), reversible addition fragmentation chain transfer (RAFT), and atom transfer radical polymerization (ATRP).23–25 Among CRP methods, ATRP has attracted much attention because of its inherent characteristics; for example, commercial availability of its reagents, capability to produce block and gradient copolymers, and application for various monomers, polymerization systems, and media are some unique features of ATRP.26–28
Reviewing related literature indicates that some studies have been done on the synthesis and investigating properties of silica aerogel/polymer nanocomposites.29–33 In addition, different initiation techniques of ATRP such as reverse atom transfer radical polymerization (RATRP) and activators generated by electron transfer (AGET) ATRP in the presence of some nanofiller(s) have been previously studied.34,35 Random copolymerization of styrene (St) and butyl acrylate by RATRP in miniemulsion system is also studied. 36
In this study, hydrophilic silica aerogel nanoparticles (SANs) were modified by hexamethyldisilazane (HMDS) as a surface modifier agent and hydrophobic SANs were obtained. Abundant advantages of reverse ATRP were employed to synthesize well-defined poly (styrene-co-methyl methacrylate) nanocomposites. Effect of HMDS-modified silica aerogel nanoparticles (H-SANs) on the kinetics parameters of ATRP (conversion, molecular weight, and polydispersity index (PDI) values) and thermal properties of the products is discussed in detail.
Experiment
Materials
St (Aldrich, Switzerland, 99%) and methyl methacrylate (MMA, Merck, Germany, 99%) were passed through an alumina filled column to remove inhibitors. Copper(I) bromide (CuBr, Aldrich, Switzerland, 98%), N, N, N′, N″, N″-pentamethyldiethylenetriamine (PMDETA, Aldrich, Switzerland, 99%), ethyl alpha-bromoisobutyrate (EBiB, Aldrich, Switzerland, 97%), anisole (Aldrich, Switzerland, 99%), tetrahydrofuran (Merck, Germany, 99%), neutral aluminum oxide (Aldrich, Switzerland, 99%), tetraethoxysilane (Merck, Germany), n-hexane (Merck, Germany), ethanol (Merck, Germany, 99%), oxalic acid (Aldrich, Switzerland, >99%), ammonium hydroxide (Tabriz Petrochemical Company, Iran), and HMDS (Merck, Germany) were used as received.
Preparation of pristine (hydrophilic) and hydrophobic SANs
Synthesis of SAN and also preparation of the H-SAN were performed according to the procedure as reported previously.16,37
Atom transfer random radical copolymerization of St and MMA and preparation of its nanocomposites
Typical batch of copolymerization was run at 110°C with the molar ratio of 200:1:1:1 for [M]:[EBiB]:[CuBr]:[PMDETA], giving a theoretical molecular weight of 20,425 g·mol−1 at 100% conversion. At first, St (13.34 ml), MMA (6.2 ml), anisole (10 ml), CuBr (0.12 g, 0.87 mmol), and PMDETA (0.18 ml, 0.87 mmol) were added into the reactor, and the reactor was degassed and back-filled with nitrogen three times and stirring was continued at room temperature. The solution turned green color since the CuBr2/PMDETA complex was formed. After that, reaction temperature was increased to 110°C during 5 min. Subsequently, ATRP initiator (EBiB, 0.12 ml, 0.87 mmol) was injected into the reactor to start the normal ATRP. After 9 h, copolymerization process was stopped by opening the reactor and exposing the catalyst to air. For preparation of nanocomposites, a desired amount of H-SAN was dispersed in 8.34 ml of St and the mixture was stirred for 22 h. Subsequently, copolymerization procedure was applied accordingly. Schematic procedure for the synthesis of tailor-made random poly (styrene-co-methyl methacrylate) chains via normal ATRP in the presence of H-SAN is presented in Figure 1.
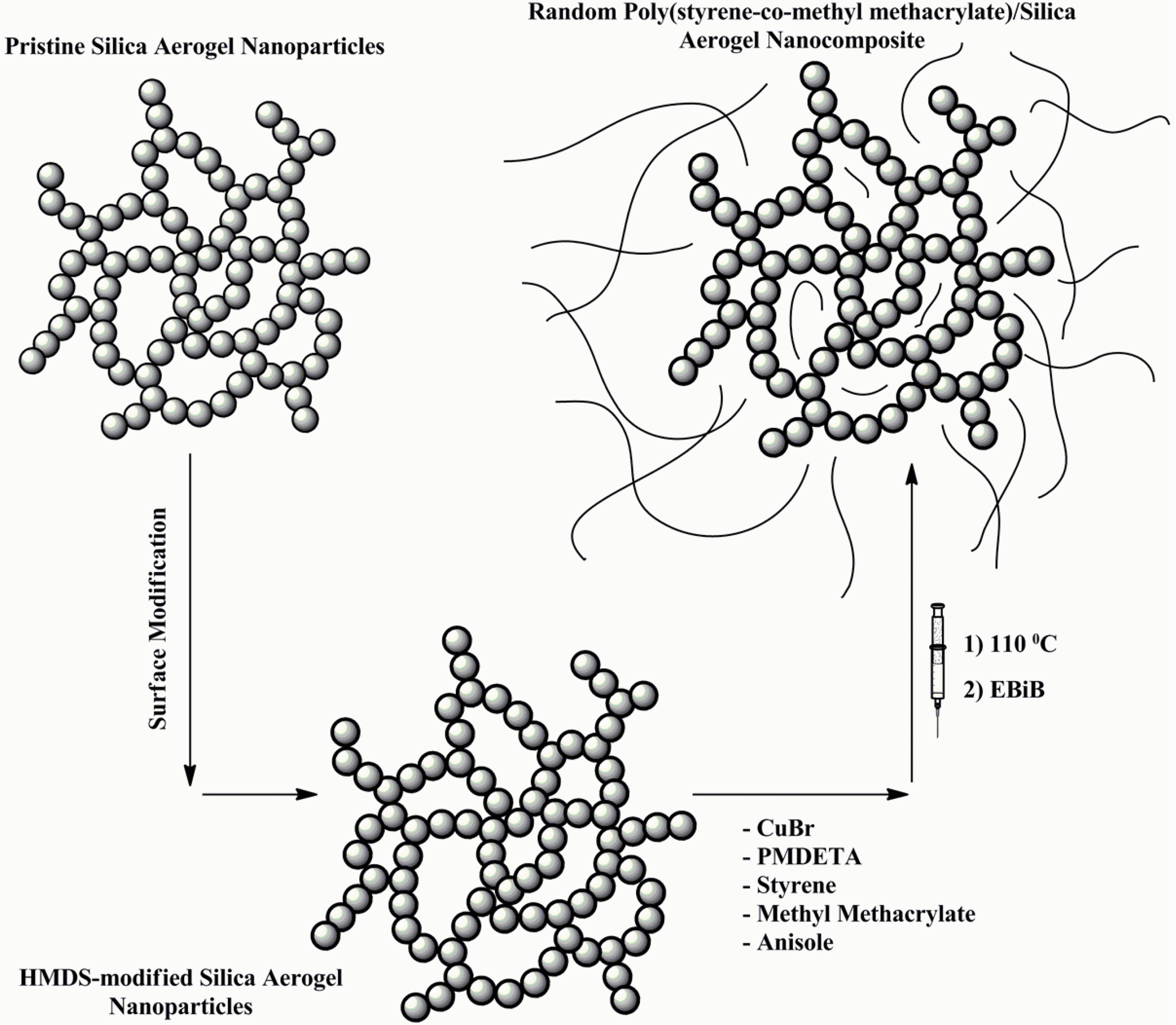
General procedure for the preparation of poly (styrene-co-methyl methacrylate)/H-SAN by normal atom transfer radical polymerization (ATRP).
Designation of the samples with the percentage of H-SAN is given in Table 1. In this designation, RCSM refers to random copolymer of St and MMA and RCSMN “X” implies different nanocomposites of random poly (styrene-co-methyl methacrylate) with various percentages of H-SANs loading.
Designation of the samples.
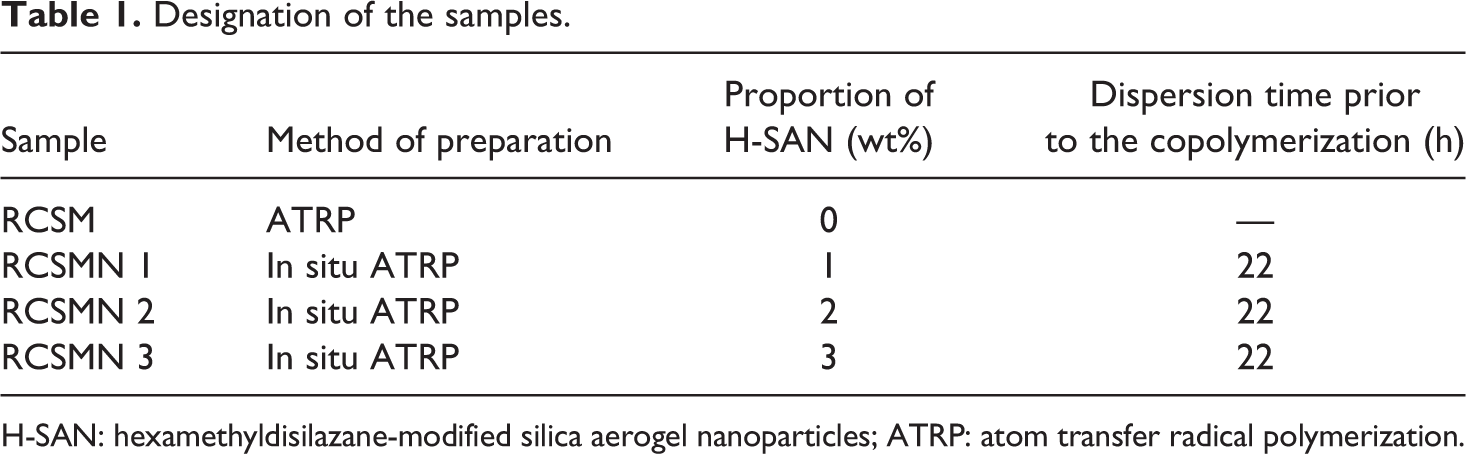
H-SAN: hexamethyldisilazane-modified silica aerogel nanoparticles; ATRP: atom transfer radical polymerization.
Characterization
Materials porosity was characterized by nitrogen adsorption/desorption curves obtained with a Quntasurb QS18 (Quntachrom, USA) apparatus. The surface area and pore size distribution values were obtained with the corrected Brunauer–Emmett–Teller equation. Surface morphology of the samples was examined by scanning electron microscope (SEM, Philips XL30, The Netherlands) with acceleration voltage of 20 kV. Also, transmission electron microscope (TEM, FEG Philips CM, The Netherlands) with an accelerating voltage of 200 kV was used. Gas chromatography (GC) is a simple and highly sensitive characterization method and does not require removal of the metal catalyst particles. GC was performed on an Agilent-6890N (USA) with a split/splitless injector and flame ionization detector, using a 60-m HP-INNOWAX capillary column for the separation. The GC temperature profile included an initial steady heating at 60°C for 10 min and a 10°C min−1 ramp from 60°C to 160°C. The samples were also diluted with acetone. The ratio of monomer to anisole was measured by GC to calculate monomer conversion throughout the reaction. Size exclusion chromatography (SEC) was used to measure the molecular weight and molecular weight distribution. A Waters 2000 ALLIANCE with a set of three columns of pore sizes of 10,000, 1000, and 500 Å was utilized to determine polymer average molecular weight and PDI. Thermal gravimetric analysis (TGA) was carried out with a PL thermo-gravimetric analyzer (Polymer Laboratories, TGA 1000, UK). Thermograms were obtained from ambient temperature to 700°C at a heating rate of 10°C min−1. Thermal analysis was carried out using a differential scanning calorimetry (DSC) instrument (NETZSCH DSC 200 F3, Netzsch Co, Selb/Bavaria, Germany). Nitrogen at a rate of 50 ml min−1 was used as purging gas. Aluminum pans containing 2–3 mg of the samples were sealed using DSC sample press. The samples were heated from ambient temperature to 180°C at a heating rate of 10°C min−1.
Results and discussion
Weak interaction between the polymer matrix and nano-fillers is one of the main factors which can restrict an appropriate dispersion of nanofillers in the polymer matrix. 38 Surface modification including modification reaction (e.g. silation reactions) and grafting methods (chain polymerization on the surface of nanoparticles) are two powerful pathways to overcome this problem. 39 In this study, hydrophilic SAN are modified with HMDS
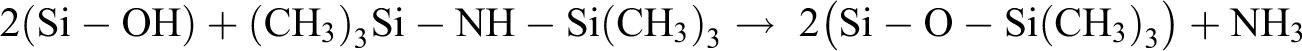
Nitrogen adsorption/desorption isotherm of the H-SAN is shown in Figure 2. According to this figure, the H-SANs reveal IV isotherm type according to the IUPAC classification and therefore the synthesized sample can be considered as a mesoporous material. 40
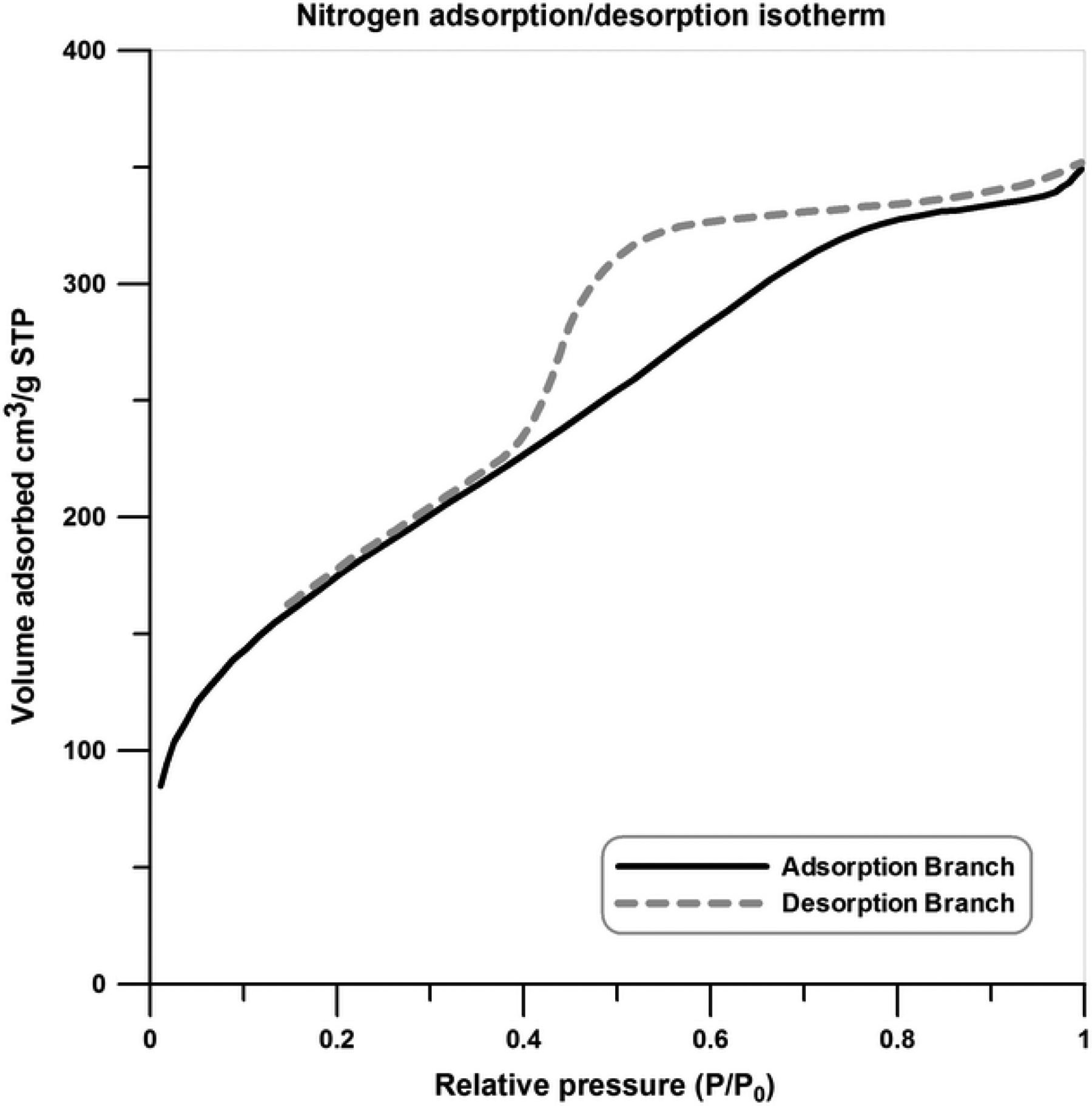
Nitrogen adsorption/desorption isotherm of the H-SAN.
Surface area and average pore diameter of the synthesized aerogel (H-SAN) are calculated 674.49 m2 g−1 and 3.77 nm, respectively.
SEM and TEM images of the H-SAN are represented in Figure 3. According to the Figure 3(a), H-SANs are generally monodispersed. These spherical nanoparticles present spongy structure and their average diameter is estimated less than 40 nm. These results are in coincidence with TEM image that nanoporous and spongy structure of the H-SAN can be observed (Figure 3(b)).

(a) SEM image and (b) TEM image of the H-SAN.
Transition metal complex in its lower oxidation state (e.g. CuI/L) is the main reagent of normal ATRP that is responsible for homogeneous cleavage of alkyl halide bond in the ATRP initiator (R–X). 41 General mechanism for normal ATRP is presented in Figure 4.
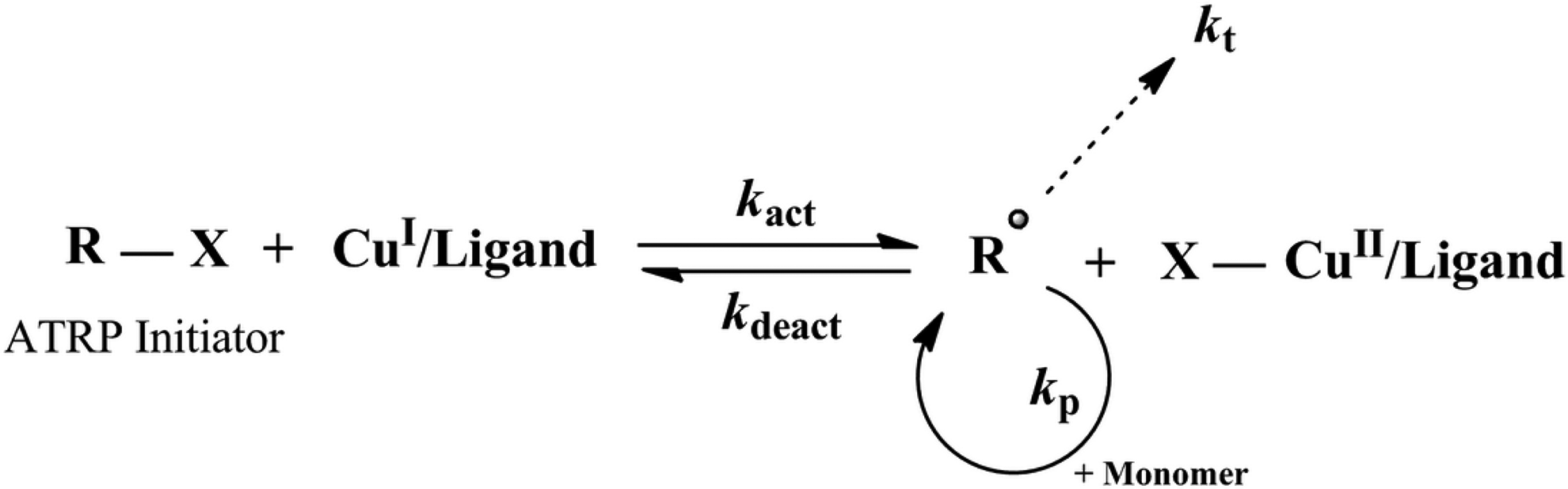
General mechanism of normal atom transfer radical polymerization (ATRP).
During the copolymerization process, monomers conversion with time can present various behaviors. Thus, examining the variation of conversion with time for the copolymerization system can be considered as an important parameter. Variation of monomers conversion versus time for the neat poly (styrene-co-methyl methacrylate) and its different nanocomposites are presented in Figure 5. According to the results, by proceeding of the reaction, an increment in the conversion is obtained (for the neat copolymer and its different nanocomposites). Also, copolymerization of St and MMA without the H-SAN results in higher conversion in various time steps. On the other hand, by increasing H-SAN content lower conversions are obtained (conversion: RSCMN 3 < RSCMN 2 < RSCMN 1 < RSCM). These results indicate that the presence of the H-SAN can result in negative effect on the copolymerization rate and final conversion of the monomers. Negative effect of the H-SAN on the copolymerization may be attributed to the three main factors. (i) Impurity role of the H-SAN: it is demonstrated that the existence of some nanofillers in the copolymerization media can disturb the ATRP equilibrium and therefore products with lower conversion, lower molecular weights, and higher PDI values can be prepared. (ii) Restriction of the mobility: due to the porous structure of the H-SAN, mobility of the radicals and growing macro-radicals in the solution can be restricted and therefore copolymerization rate and monomers conversion can be decreased. It is clear that by increasing H-SAN content, more restriction will be happened and therefore higher decrement in the conversion can be obtained. (iii) Irreversible reactions: it is demonstrated that growing radicals in the copolymerization media can irreversibly react with various functional moieties. Thus, growing radicals can irreversibly react with the moieties (such as unmodified hydroxyl groups) on the surface of the H-SAN and therefore can present negative effect on the copolymerization rate and conversion.42–44 By comparing these results with the previous study (simultaneous reverse and normal initiation technique [SR&NI] ATRP of St and MMA in the presence of the H-SAN), it is clearly demonstrated that normal and SR&NI initiation technique present similar behavior for the variation of monomer conversion with time by the addition of 1–3 wt% of the H-SAN. 45
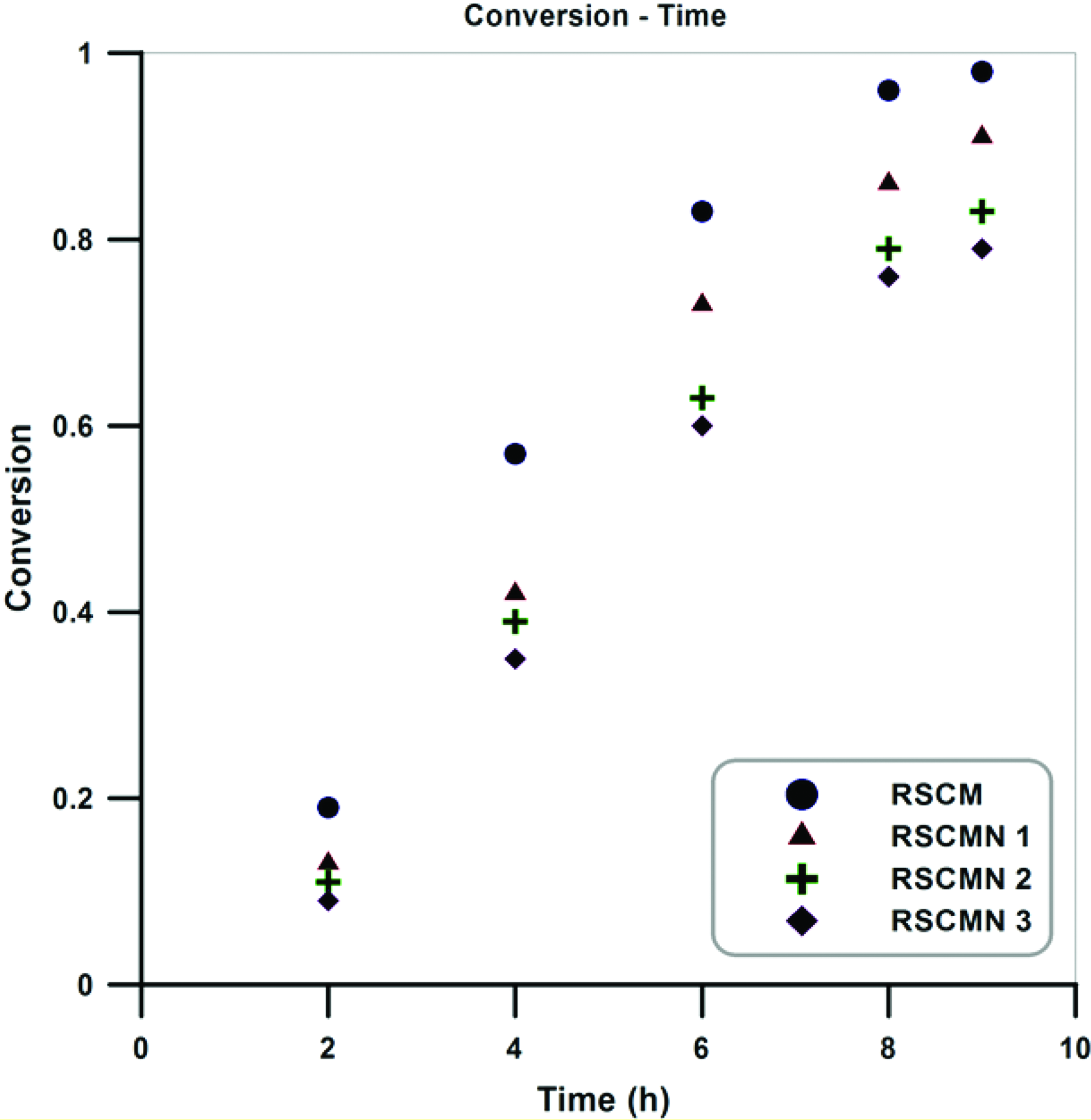
Evolution of monomer conversion with time for the RSCM and RSCMN 1–3.
Variation of number-average molecular weight (Mn) with conversion during the copolymerization is represented in Figure 6. Theoretical line in this figure is displayed to present theoretical Mn values for the various conversions. As expected, in the neat poly (styrene-co-methyl methacrylate) and different nanocomposites by increasing conversion copolymer chains with higher Mn were obtained. According to the results, RSCM has higher Mn values in comparison with the different nanocomposites (RSCMN 1–3). On the other hand, addition of H-SAN in the copolymerization media results in decrement of Mn of the products. By increasing H-SAN content, more decrement in Mn is observed (Mn: RSCM > RSCMN 1 > RSCMN 2 > RSCMN 3). This negative effect of the H-SAN on the molecular weight of the poly (styrene-co-methyl methacrylate) chains can be also attributed to the three abovementioned reasons (impurity role, restriction of the mobility, and irreversible reactions).42–44 By comparing these results with the previous study (SR&NI ATRP of St and MMA in the presence of the H-SAN), it can be concluded that normal and SR&NI initiation technique present similar behavior for the variation of molecular weight with conversion by the addition of 1–3 wt% of the H-SAN. 45
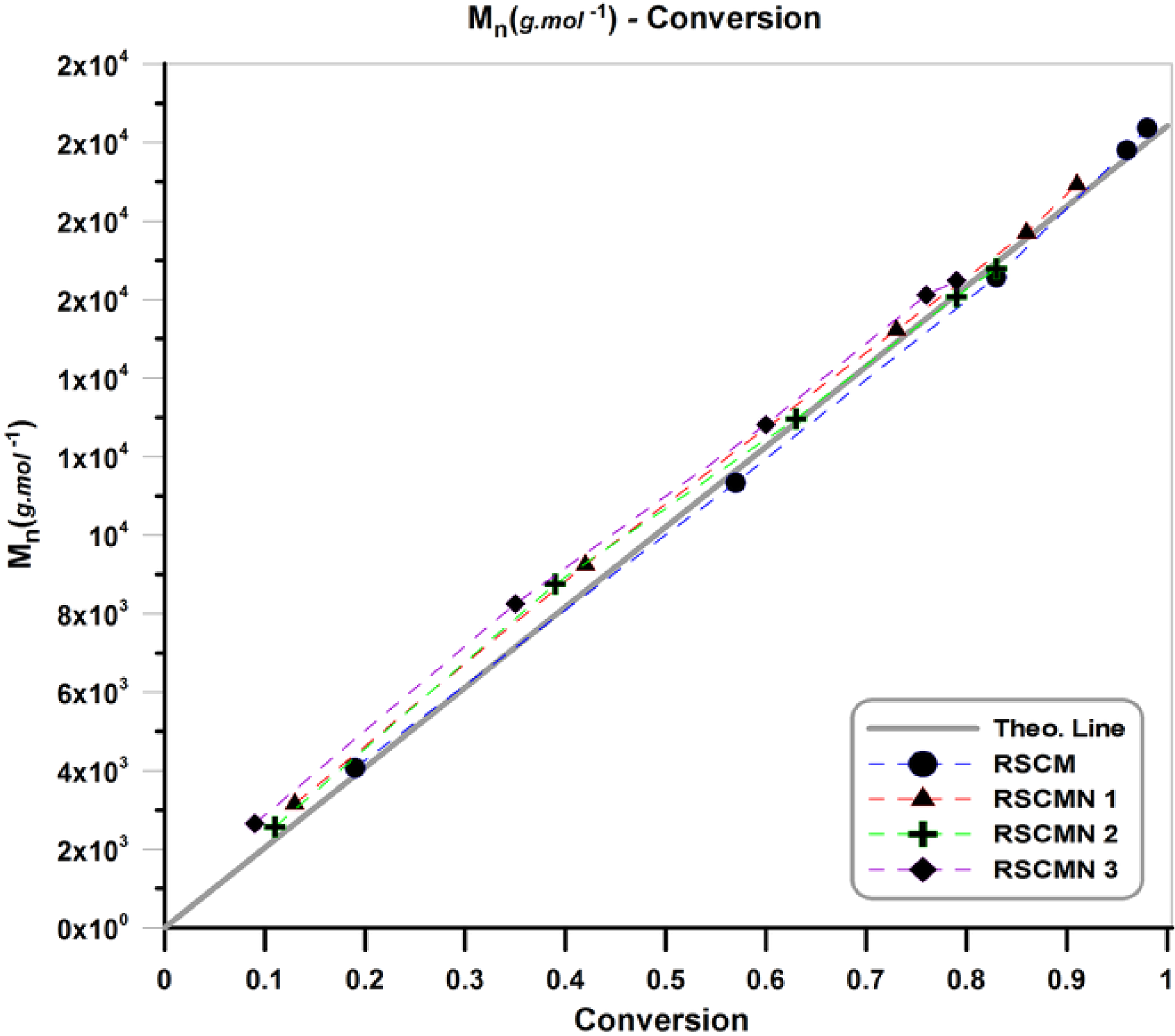
Variation of molecular weight versus conversion for the RSCM and RSCMN 1–3.
Variation of PDI values with conversion for the neat poly (styrene-co-methyl methacrylate) and its various nanocomposites is displayed in Figure 7. Since the copolymerization system (ATRP) is a member of CRP family, it could be predicted that PDI values present a decrement by proceeding of the reaction (by increasing of conversion). Although the addition of nanofillers can affect CRP equilibrium, they cannot change the living nature of the system and therefore a decrement in PDI values by increasing conversion are observed in the pure poly (styrene-co-methyl methacrylate) (RSCM) and its different nanocomposites (RSCMN 1–3). According to Figure 7, PDI value of the RSCM is lower than RSCMN 1–3. In the nanocomposites, by increasing H-SAN content, higher PDI values are obtained (PDI: RSCM < RSCMN 1 < RSCMN 2 < RSCMN 3). Increment of PDI values by increasing H-SAN content can be attributed to the impurity role of these nanoparticles. The occurrence of termination and transfer reactions between the propagating (macro)radicals and the H-SAN can be considered as the main reason of emerging high PDI values in the nanocomposites samples.42–44 Comparing these results with the previous study indicates that normal and SR&NI initiation technique present similar behavior for the variation of PDI values with conversion by the addition of 1–3 wt% of the H-SAN. 45
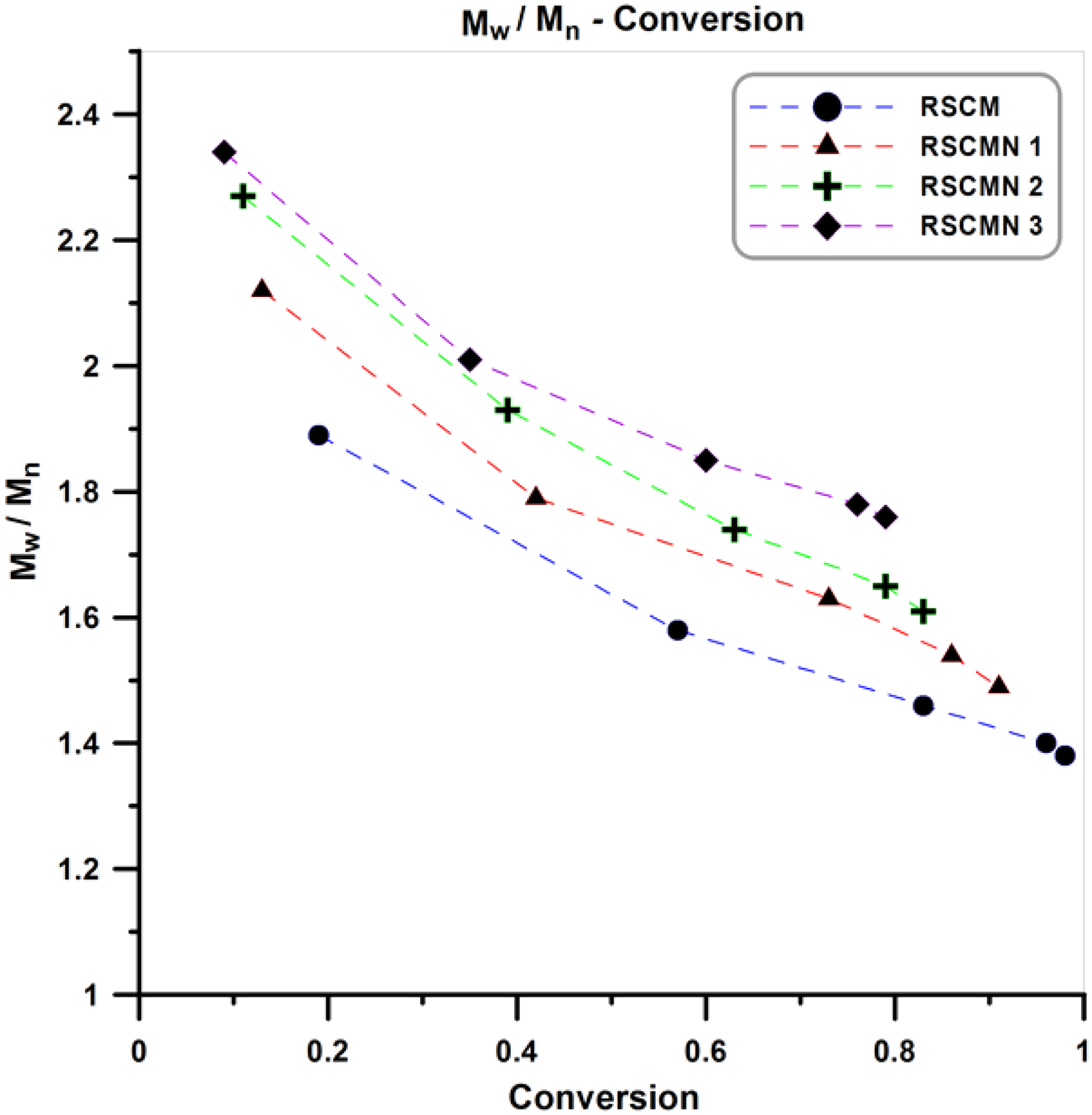
Variation of PDI values with conversion for the RSCM and RSCMN 1–3.
Figure 8 shows pseudo-first-order kinetic plots for the synthesized nanocomposites and the neat poly (styrene-co-methyl methacrylate). Results indicate that the slope of the curves (RSCM and RSCMN 1–3) remains fairly constant in the whole of the copolymerization and therefore it can be concluded that steady-state kinetics for all of the samples is obtained. According to the figure, RSCM and RSCMN 1–3 present linearly increment of ln(M0/M) with time in which constant radical concentration in the copolymerization media can be concluded.42,43 Results also indicate that by increasing H-SAN content a considerable decrement in the copolymerization rate will be obtained. Comparing these results with the previous study indicates that normal and SR&NI initiation technique present similar kinetics behavior by the addition of 1–3 wt% of the H-SAN in the polymer matrix. 45
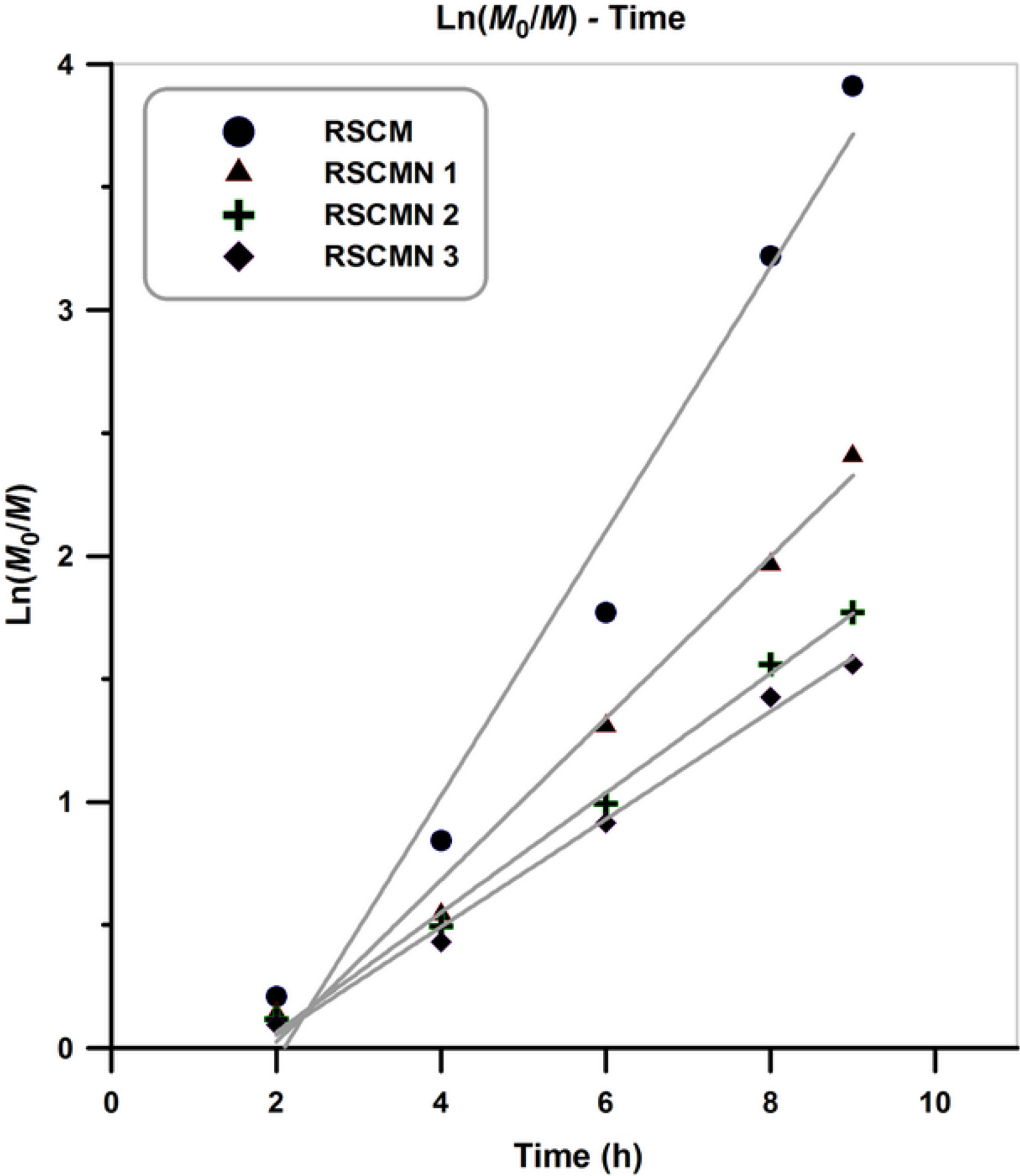
Kinetic plots with respect to the reaction time.
RSCM and RSCMN 1–3 display monomodal peak in their SEC traces (Figure 9). According to this figure, RSCM presents narrow distribution in comparison with the RSCMN 1–3. Also, by increasing H-SAN loading, broaden distribution is obtained (molecular weight distribution: RSCM < RSCMN 1 < RSCMN 2 < RSCMN 3).
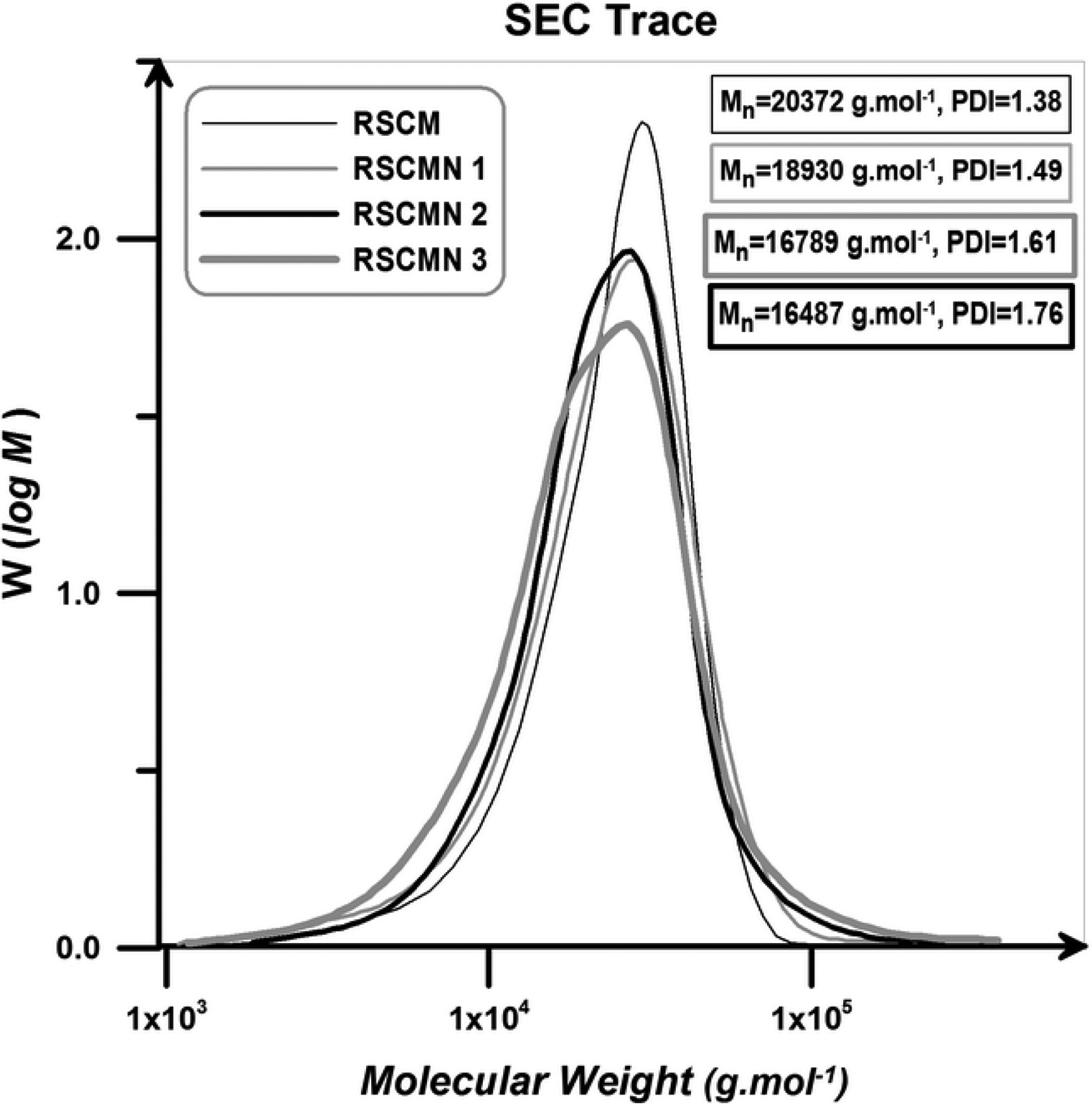
SEC traces of the RSCM and RSCMN 1–3.
SEC traces demonstrate that ATRP of St and MMA in the presence of H-SAN results in random poly (styrene-co-methyl methacrylate) with broaden molecular weight distribution. On the other hand, the absence of the H-SAN in the copolymerization media leads to random copolymer with lower PDI values. As it mentioned above, H-SAN can disturb the equilibrium of the ATRP due to its three main roles (impurity role, restriction of the mobility, and irreversible reactions).42–44 According to the extracted data in Table 2, by the addition of 3 wt% of the H-SAN monomer conversion decreases from 98% to 79%. Meanwhile, PDI values present an increment from 1.38 to 1.76.
Molecular weights and PDI values of the extracted copolymers resulted from SEC traces.
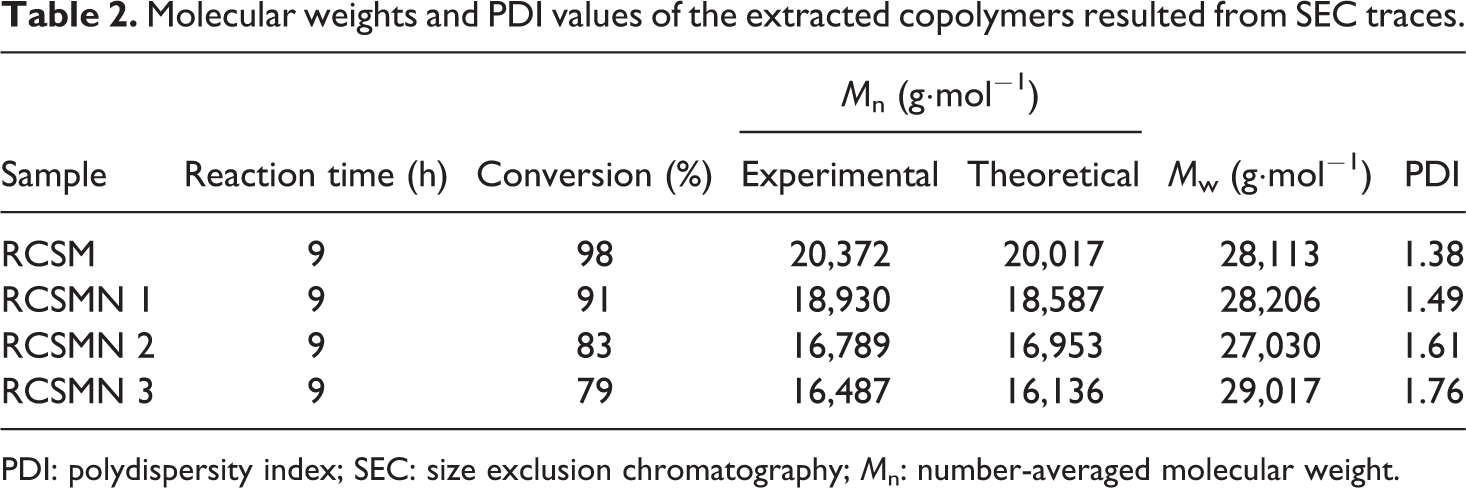
PDI: polydispersity index; SEC: size exclusion chromatography; Mn: number-averaged molecular weight.
By comparing the results of this study with the previous study (SR&NI ATRP of St and MMA in the presence of the H-SAN), some useful conclusions can be obtained. (a) In general, normal and SR&NI initiation techniques present relatively similar kinetics behavior in the case of addition of 1–3 wt% of the H-SAN. (b) Application of normal ATRP results in higher final conversion in the same loadings of the H-SAN in comparison with the SR&NI ATRP. (c) It is very interesting that variation of the final conversion by the addition of 3 wt% of the H-SAN is relatively equal for both of the systems (19 and 21% decrement in conversion by adding 3 wt% of the H-SAN). (d) Both of the systems present similar behavior in the case of the variation of the molecular weights. (e) As it expected, application of SR&NI ATRP results in lower PDI values in comparison with the normal ATRP (for the neat copolymer and the corresponding nanocomposites). (f) According to the PDI values, both of the systems present equal variations in PDI values by the addition of 3 wt% of the H-SAN (0.38 and 0.36). 45
TGA thermograms of the RSCM and RSCMN 1–3 in the temperature window of 25–700°C in addition to their corresponding differential thermogravimetric (DTG) are displayed in Figure 10. As expected, by increasing H-SAN content, char values of the prepared nanocomposites should be increased (char values: RSCM < RSCMN 1 < RSCMN 2 < RSCMN 3).
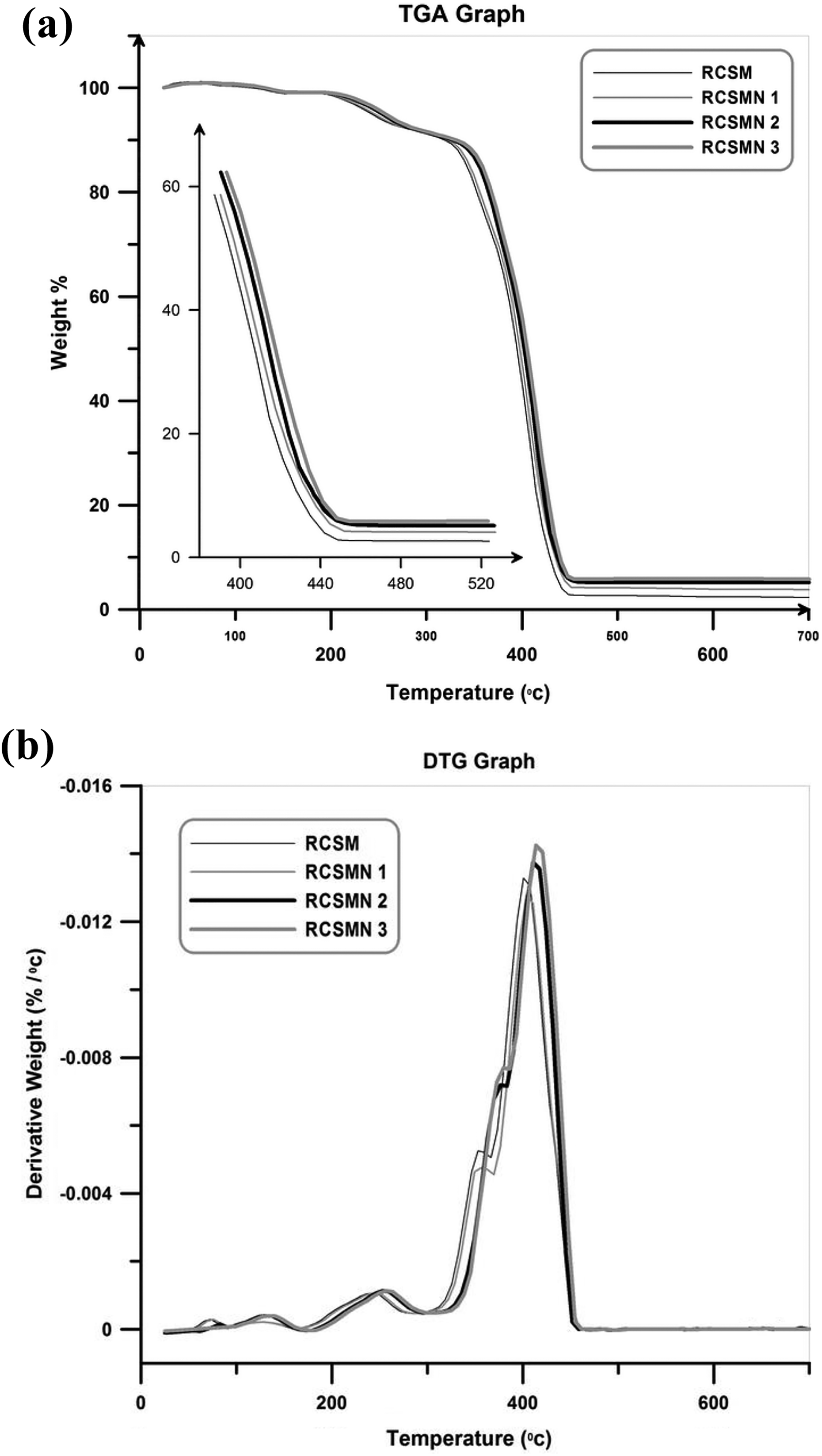
(a) TGA and (b) DTG thermograms of the RSCM and RSCMN 1–3.
Figure 10 indicates that thermal stability of the RSCM is lower than various nanocomposites. Also, it can be concluded that by increasing H-SAN content, more improvements in thermal stabilities can be obtained (thermal stability: RSCM < RSCMN 1 < RSCMN 2 < RSCMN 3). Except the main degradation step in the TGA graphs (at the temperatures above 350°C) that is attributed to the degradation of the synthesized poly (styrene-co-methyl methacrylate) and its nanocomposites, some other degradation can also be recognized. These degradations in the lower temperatures are ascribed by evaporation of water molecules and volatile materials such as residual St, MMA, and HMDS.
TGA results demonstrate that incorporation of H-SAN in the copolymer matrix leads to improvements in thermal stabilities. This improvement can be ascribed using inorganic nature of the H-SAN. H-SAN as inorganic nano-filler can result in higher thermal stabilities of the RSCMN 1–3 in comparison with the RSCM.9,38 Absorption of the introduced gas (in oxygen atmosphere) in the porous structure of the incorporated nano-filler (modified SANs is classified as mesoporous materials) may be considered as another factor for increment of thermal stabilities of the nanocomposites in comparison with the RSCM.
According to Figure 10, addition of the H-SAN in the copolymer matrix results in slight improvements in thermal stabilities (this improvements are really higher in the case of clay or diatomite platelets). This can be attributed to the three-dimensional network structure of the H-SAN. Due to this structure, SAN cannot be appropriately dispersed in the copolymer matrix and therefore cannot significantly improve thermal stabilities of the nanocomposites. 44 Extracted data from TGA thermograms and DTG curves are also summarized in Table 3.
Extracted data from TGA and DTG thermograms of the RSCM and RSCMN 1–3.

TGA: thermal gravimetric analysis; DTG: differential thermogravimetric.
Evaluation of the effect of H-SAN on the chain confinement and also determination of glass transition temperature (Tg) are performed by DSC analysis. DSC curves of the RSCM and RSCMN 1–3 are presented in Figure 11. Although the samples are heated from room temperature to 220°C to remove their thermal history, cooling path of the analysis in the temperature range of 20–180°C is displayed in this figure.
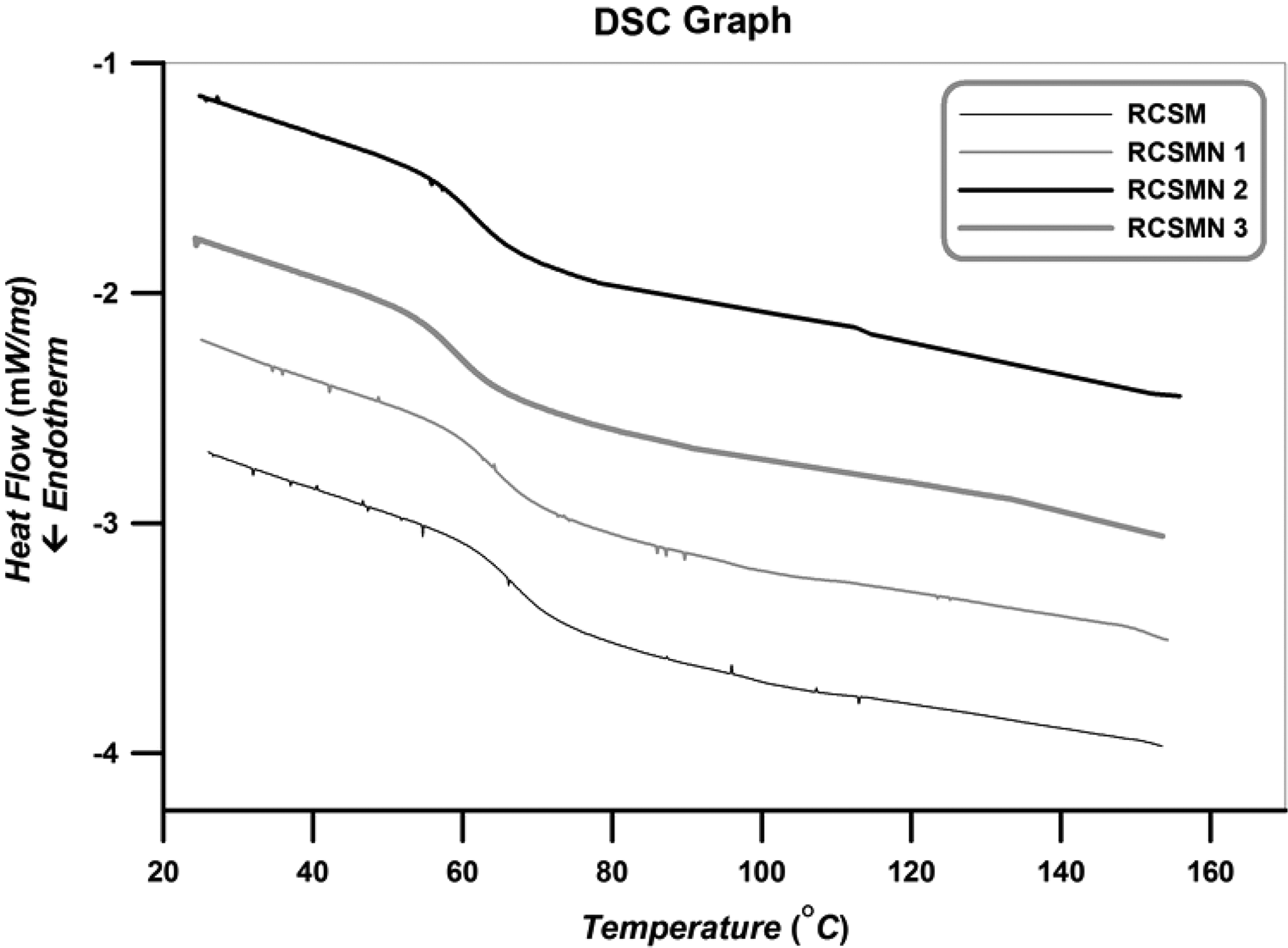
DSC thermograms of the RSCM and RSCMN 1–3.
According to Figure 11, an obvious inflection in the cooling path is occurred which shows Tg values of the RSCM and RSCMN 1–3. In addition, amorphous structures of the synthesized samples can be also concluded. Extracted Tg values of the samples from DSC graphs (in the cooling path) are summarized in Table 4.
Tg values of the RSCM and RSCMN 1–3.
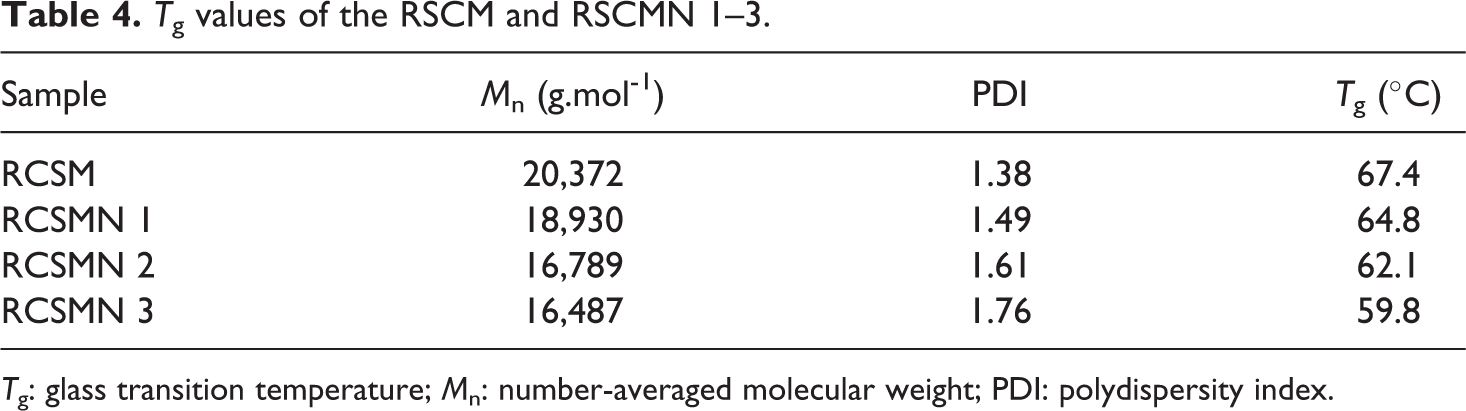
Tg: glass transition temperature; Mn: number-averaged molecular weight; PDI: polydispersity index.
According to Table 4, RSCM presents higher Tg value in comparison with the other samples (RSCMN 1–3). By incorporating H-SAN in the copolymer matrix, a decrement in Tg value was happened (Tg: RSCMN 3 < RSCMN 2 < RSCMN 1 < RSCM). Decrement in Tg values by adding H-SAN in the copolymer matrix is related to the reduction of the packing of copolymer chains. By dispersing H-SAN within the copolymer matrix, packing of the copolymer chains will be reduced and therefore copolymer chains can obtain more segment mobilities in which results in decrement of Tg values. In addition, due to the three-dimensional network structure of the H-SAN, these nanoparticles cannot appropriately dispersed within the copolymer matrix and therefore some free volume spaces for the dangling copolymer chains may be created. This is also responsible for more segment mobilities of the copolymer chains which in turn results in Tg reduction. Moreover, according to Table 2, by increasing H-SAN in the copolymer matrix, an increment in PDI value and also a decrement in Mn are observed. These parameters should be also considered since decrease in Mn and increment of PDI values may cause reduction of Tg values.44,46 Previous studies indicate that Tg values can be changed by the addition of low volume of different nano-fillers.47–49 Reduction of Tg value by incorporating alumina nanoparticles as a nano-fillers was also reported by Ash et al. 49
Conclusions
Random poly (styrene-co-methyl methacrylate) and its different nanocomposites in the presence of the H-SAN were synthesized by in situ normal ATRP. Pristine SANs were modified with HMDS and the resultant hydrophobic nanoparticles were used for in situ copolymerization. Porous and spongy structure, spherical shape, and high surface are some unique properties of the synthesized H-SAN. Normal ATRP of St and MMA in the presence of H-SAN results in a decrease of conversion from 98% to 79%. Moreover, molecular weight of the random copolymer chains decrease from 20,372 g·mol−1 to 16,487 g·mol−1 and PDI values increase from 1.38 to 1.76. Improvement in thermal stability of the nanocomposites and decreasing Tg values from 67.4°C to 59.8°C was also observed by the addition of 3 wt% of the H-SAN.
