Abstract
In situ copolymerizations of styrene and methyl methacrylate (MMA) by atom transfer radical random copolymerization in the presence of mesoporous diatomite were performed to synthesize various poly (styrene-co-methyl methacrylate)/diatomite composites. Special characteristics of the mesoporous diatomite were evaluated by X-ray fluorescence, thermal gravimetric analysis (TGA), transmission electron microscope, and nitrogen adsorption/desorption isotherm. Conversion and molecular weight determinations were carried out using gas chromatography and size-exclusion chromatography, respectively. Due to the pendant hydroxyl groups on the surface of the diatomite platelets and polarity change of the reaction medium, addition of 3 wt% mesoporous kieselgur platelets leads to increase of conversion from 69% to 81%. In addition, polydispersity index values of the poly (styrene-co-methyl methacrylate) chains increases from 1.14 to 1.39 by addition of 3 wt% mesoporous diatomite platelets. Increasing the thermal stability of the nanocomposites is demonstrated by TGA. Differential scanning calorimetry shows an increase in glass transition temperature from 66.4°C to 72.5°C by adding 3 wt% of mesoporous diatomite platelets.
Introduction
Polymer (nano)composites can be widely employed in many diverse applications, such as transportation vehicles, construction materials, electronics, and consumer products. 1,2 Nanocomposites present unique properties that are originated from inherent characteristics of organic and nanoinorganic components; polymer (organic phase) as a host material results in suitable flexibility and processability of the products and nanoinorganic filler(s) leads to appropriate rigidity and thermal stability of the products. 2,3 Unique benefits of nanocomposites in comparison with the traditional (micro)composites are generally attributed to the high surface area of the nanofillers. Increment of interfacial areas between the polymer matrix and the filler is one of the main advantages of nanofillers. 4 Various procedures have been employed for the preparation of polymer-based nanocomposites. Among the three main pathways for the preparation of nanocomposites (solution, melt intercalation, and in situ polymerization), in situ polymerization have attracted great attention. 5,6
Diatomite or diatomaceous earth is a kind of silicate material. It is a dust varying in color depending on composition, from white–gray to yellow–red. Pristine diatomite is an insulator, noninflammable, insoluble in water, extremely stable in air, and contains up to 80% voids in its structure. 7,8 Large specific surface area, highly porous structure, chemical inertness, high absorption capacity, low density, and low cost are some unique properties of diatomite platelets. 8,9 Since diatomite presents a unique combination of physical and chemical properties, it can be deserved for various applications in many diverse areas, such as nanofabrication, optics, water purification, solar cells, biosensing, drug delivery, polymer-based composites, adsorbent, and a flocculant for pollutants encountered in wastewater. 10,11
Controlled/living radical polymerization (CRP) methods have attracted great attention during the last decades. Stable free-radical polymerization, reversible addition–fragmentation chain transfer polymerization, and atom transfer radical polymerization (ATRP) are the three most common types of CRP procedures. Among the CRP methods, ATRP possesses several unique advantages over others, such as applicability for a wide range of monomers and polymerization systems, remarkable tolerance to different functional groups, capability for synthesis of the pure block copolymer, and commercial availability of its reagents (e.g. ligands and alkyl halides). 12 –15
A review of the literature indicates that the application of diatomite as filler to synthesize polymer/diatomite composites has attracted considerable attention. Karaman et al. have prepared polyethylene glycol (PEG)/diatomite composite as a novel form-stable composite phase change material (PCM) in which the PCM was prepared by incorporating PEG in the pores of diatomite. 16 Li et al. have synthesized conducting diatomite by polyaniline on the surface of diatomite. Linkage of polyaniline on the surface of diatomite is attributed to the hydrogen bond between the surface of diatomite and polyaniline macromolecules. 17 Li et al. have also prepared fibrillar polyaniline/diatomite composite by one-step in situ polymerization. According to their results, the polyaniline/diatomite composite can be applied as fillers for electromagnetic shielding materials and conductive coatings. 18 In addition, other studies such as investigating the effects of extrusion conditions on die-swell behavior of polypropylene/diatomite composite melts and crystallization behaviors and foaming properties of diatomite-filled polypropylene composites have been performed. 19,20
In the current study, we take unique advantages of normal ATRP method to synthesize tailor-made random poly (styrene-co-methyl methacrylate) nanocomposites. The ATRP process was applied to the mixture of monomer and pristine diatomite platelets to prepare well-defined nanocomposite using in situ polymerization. We have tried to investigate the effect of diatomite loading on the kinetics parameters of the polymerization, thermal stability, and glass transition temperature (T g) of the products. Evaluation of diatomite platelets as an appropriate replacement of natural clay platelets is also performed by examining kinetics and thermal properties of the prepared nanocomposites.
Experimental
Materials
Diatomite earth sample was obtained from Kamel Abad-Azerbaijan, Iran. It was dispersed in 100 ml distilled water by magnetic stirring. Then, the particles were separated with filter paper and dried at 100°C for 8 h. Styrene (St; Aldrich, Switzerland, 99% pure) and methyl methacrylate (MMA, Merck, Germany, 99% pure) were passed through an alumina-filled column, dried over calcium hydride, and distilled under reduced pressure (60°C, 40 mmHg). Copper (I) bromide (CuBr, Aldrich, Switzerland, 98%) was washed with glacial acetic acid, filtered, and finally washed with ethanol. N,N,N′,N″,N″-pentamethyldiethylenetriamine (PMDETA, Aldrich, Switzerland, 99%), ethyl alpha-bromoisobutyrate (EBiB, Aldrich, Switzerland, 97%), anisole (Aldrich, Switzerland, 99%), tetrahydrofuran (THF, Merck, Germany, 99%), and neutral aluminum oxide (Aldrich, Switzerland, 99%) were used as received.
Synthesis of random poly (styrene-co-methyl methacrylate) via normal ATRP
Normal ATRP of St and MMA was performed in a 200-ml three-necked lab reactor, which was equipped with a reflux condenser, nitrogen inlet valve, and a magnetic stir bar that was placed in an oil bath. A typical batch of copolymerization was run at 110°C with the molar ratio of 150:1:1:1 for [M]:[EBiB]:[CuBr]:[PMDETA] giving a theoretical copolymer molecular weight of 15,320 g mol−1 at 100% conversion. At first, St (13.34 ml, 0.11 mmol), MMA (6.2 ml, 0.05 mmol), CuBr (0.167 g, 1.1 mmol), PMDETA (0.24 ml, 1.1 mmol), and anisole (10 ml) were added to the reactor. Then, it was degassed and backfilled with nitrogen three times, and then left under N2 with stirring at room temperature. When the majority of the metal complex had formed, the reaction temperature was increased to 110°C over 5 min. Subsequently, initiator (EBiB, 0.17 ml, 1.1 mmol) was injected into the reactor to start the copolymerization. Samples were taken at the end of the reaction to measure the final conversion.
Preparation of poly (styrene-co-methyl methacrylate)/mesoporous diatomite composites via in situ normal ATRP
For the preparation of nanocomposites, a desired amount of the pristine diatomite platelets was dispersed in 5 ml of St, and the mixture was stirred for 18 h. Then, the remaining 8.34 ml of St was added to the mixture. Subsequently, copolymerization procedure was applied accordingly. Designation of the samples with the percentage of pristine diatomite platelets is given in Table 1. In this designation, NPSM refers to the neat poly (styrene-co-methyl methacrylate) and PSMD X implies various nanocomposites with X percentages of pristine diatomite platelets loading.
Designation of the samples.
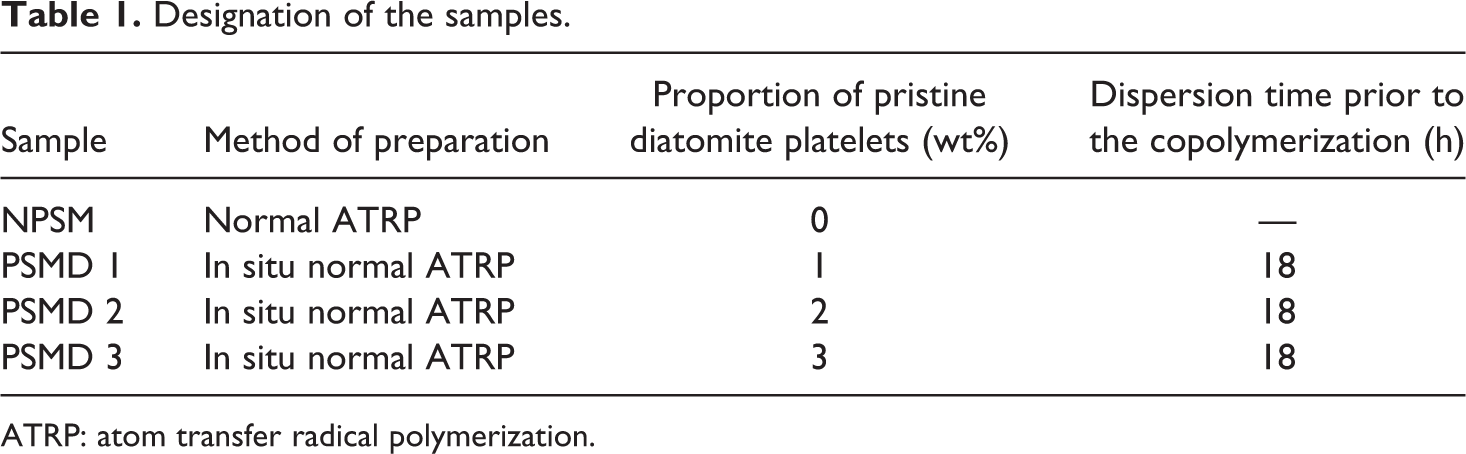
ATRP: atom transfer radical polymerization.
Separation of copolymer chains from pristine diatomite platelets and catalyst removal
For separating poly (styrene-co-methyl methacrylate) chains from the pristine diatomite particles, nanocomposites were dissolved in THF. By ultracentrifugation (10,000 r min−1) and then passing the solution through a 0.2-μm filter, poly (styrene-co-methyl methacrylate) chains were separated from pristine diatomite particles. Subsequently, copolymer solutions passed through an alumina column to remove catalyst species.
Characterization
Characterization of the pristine diatomite sample for its chemical composition was performed by an X-ray fluorescence spectrometer (XRF, Philips 2400, Netherlands). Materials porosity was characterized by N2 adsorption/desorption curves obtained with a Quntasurb QS18 (Quntachrom) apparatus (USA). The surface area and pore size distribution values were obtained with the corrected Brunauer–Emmett–Teller equation. Pore size distributions were also calculated by the Barrett–Joyner–Halenda method. The transmission electron microscopy (TEM), Philips EM 208 (Eindhoven, Netherlands), with an accelerating voltage of 120 kV was employed to study the morphology of the pristine diatomite sample. Gas chromatography (GC) is a simple and highly sensitive characterization method and does not require removal of the metal catalyst particles. GC was performed on an Agilent-6890 N with a split/splitless injector and flame ionization detector, using a 60-m HP-INNOWAX capillary column for the separation. The GC temperature profile included initial steady heating at 60°C for 10 min and a 10°C min−1 ramp from 60°C to 160°C. The samples were also diluted with acetone. The ratio of monomer to anisole was measured by GC to calculate monomer conversion throughout the reaction. Size-exclusion chromatography (SEC) was used to measure the molecular weight and molecular weight distribution. A Waters 2000 ALLIANCE system (Milford, USA) with a set of three columns of pore sizes of 10,000, 1000, and 500 Å was utilized to determine polymer average molecular weight and dispersity. THF was used as the eluent at a flow rate of 1.0 ml min−1, and calibration was carried out using low-dispersity polystyrene standards. Thermal gravimetric analysis (TGA) was carried out with a PL thermogravimetric analyzer (TGA 1000; Polymer Laboratories, UK). Thermograms were obtained from ambient temperature to 700°C at a heating rate of 10°C min−1. Thermal analysis was carried out using a differential scanning calorimetry (DSC) instrument (NETZSCH DSC 200 F3; Netzsch Co, Selb/Bavaria, Germany). Nitrogen at a rate of 50 ml min−1 was used as purging gas. Aluminum pans containing 2–3 mg of the samples were sealed using DSC sample press. The samples were heated from ambient temperature to 220°C at a heating rate of 10°C min−1.
Results and discussion
Pristine diatomite platelets are employed to synthesize well-defined random poly (styrene-co-methyl methacrylate) nanocomposites and evaluating the effect of these platelets on the kinetics of copolymerization. In addition, considerable effects of these platelets on thermal properties of the products are investigated. General procedure for the synthesis of tailor-made random poly (styrene-co-methyl methacrylate) chains via normal ATRP in the presence of the pristine diatomite platelets is graphically presented in Figure 1.
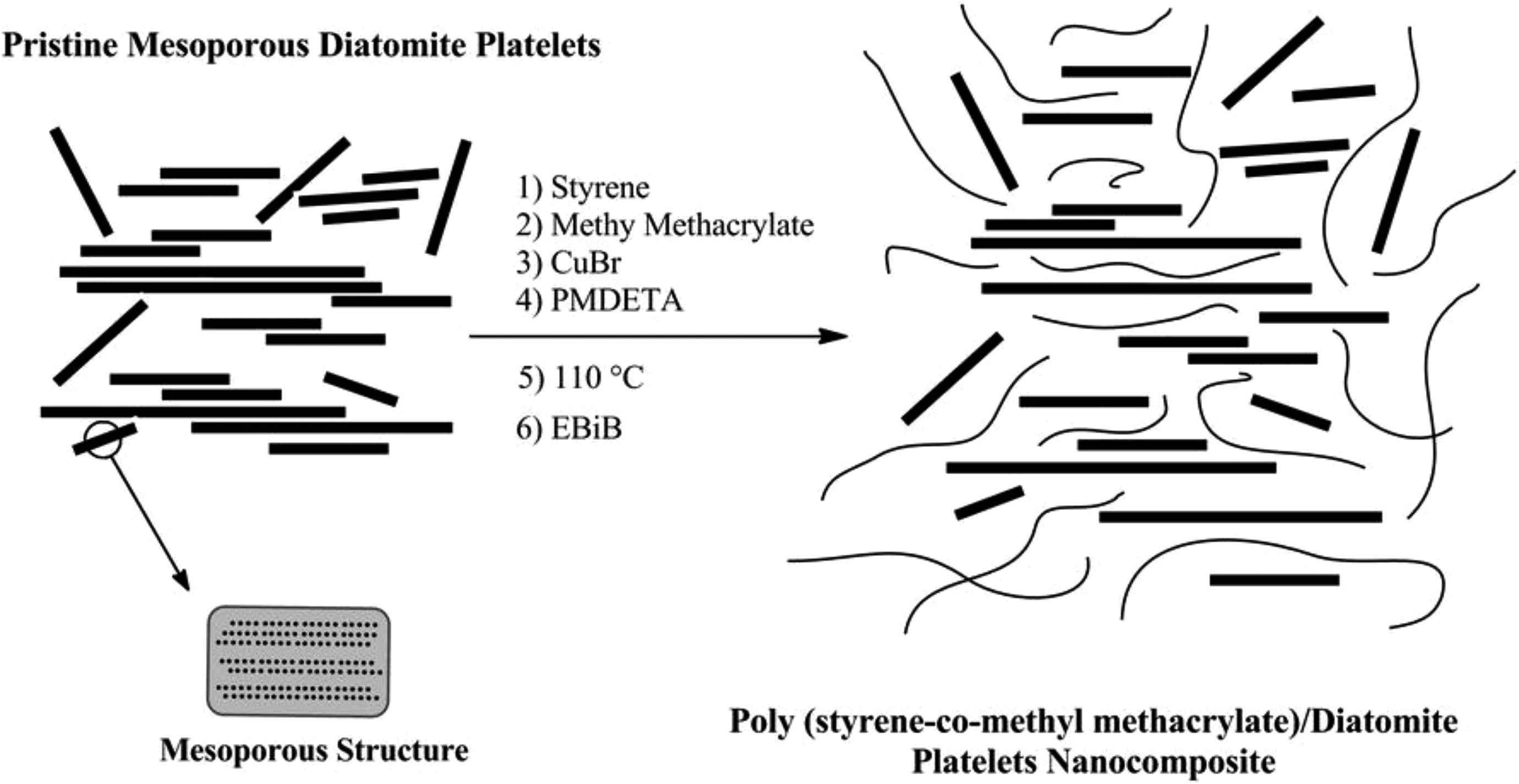
General procedure for the synthesis of poly (styrene-co-methyl methacrylate)/diatomite nanocomposites via normal ATRP.
Plots of monomer conversion versus time for the neat poly (styrene-co-methyl methacrylate) and its different nanocomposites are shown in Figure 2. According to the results, the typical behavior as in pure poly (styrene-co-methyl methacrylate) was observed in all the nanocomposites. In addition, copolymerization rate and conversion in the nanocomposites are higher than the neat poly (styrene-co-methyl methacrylate) and by increasing diatomite platelets content, the copolymerization rate and conversion were also increased. Pendant hydroxyl groups on the surface of the diatomite platelets can possibly cause a polarity change into the reaction medium in which exerts a rate acceleration effect on the polymerization system and therefore leads to increment of the polymerization rate. 21,22
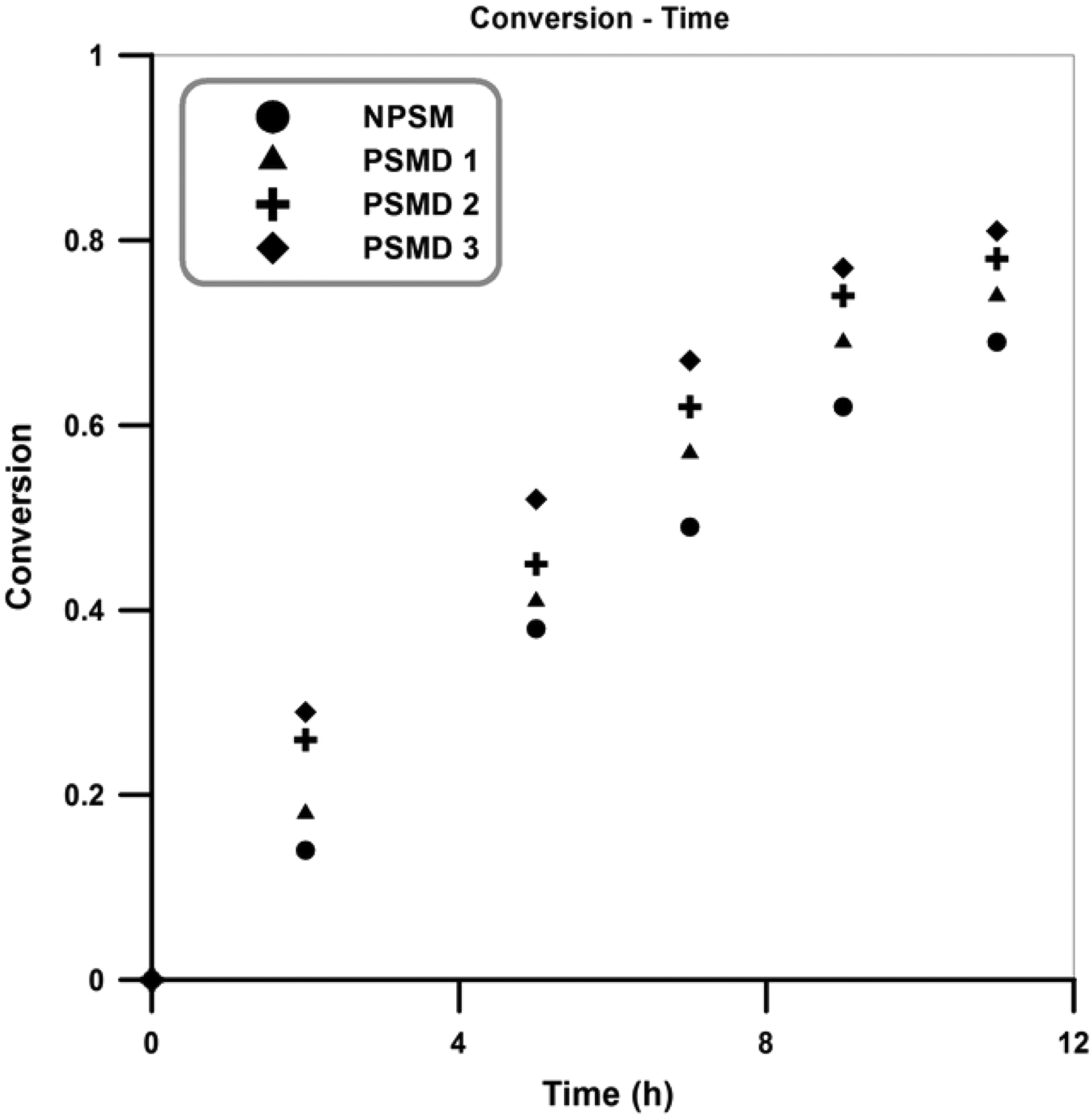
Evolution of monomer conversion with time for the neat copolymer and its nanocomposites.
Figure 3 presents the variation of number-average molecular weight (M n) versus conversion during the copolymerization. Two main results can be concluded from these plots: (i) M n increases by monomer conversion; (ii) addition of diatomite platelets results in higher M n values compared with the neat poly (styrene-co-methyl methacrylate). Higher M n values of the nanocomposite samples are attributed to the polarizing effect of hydroxyl groups on the surface of diatomite platelets. 21,22 Moreover, the experimental results are in good agreement with the theoretical molecular weight that well-controlled copolymerization processes are appropriately demonstrated. 21
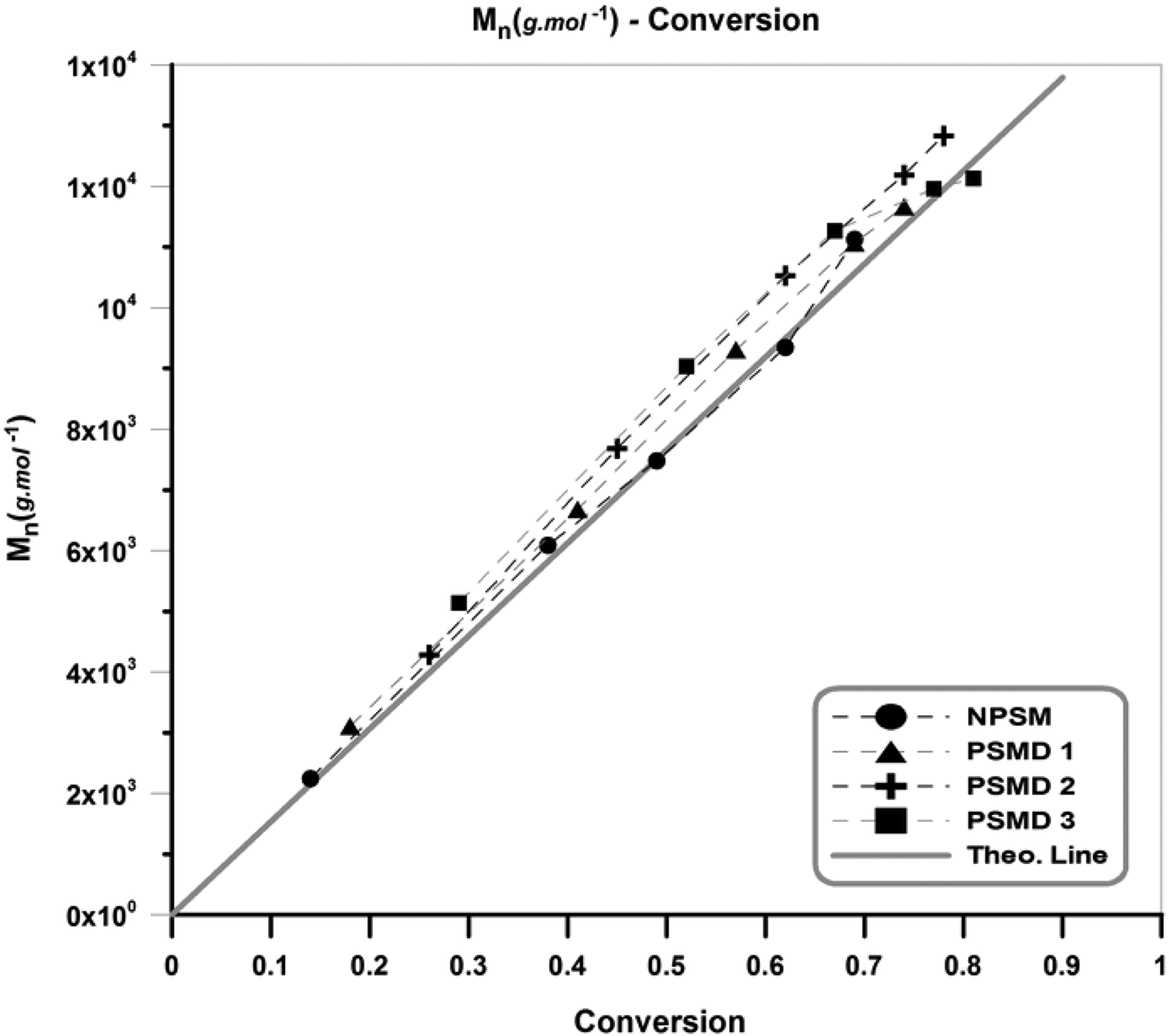
Molecular weight versus conversion for the neat copolymer and its various nanocomposites.
Evolution of polydispersity index (PDI) values with the conversion for the neat poly (styrene-co-methyl methacrylate) and its various nanocomposites are presented in Figure 4. As expected for controlled living radical polymerization, PDI values are decreased with conversion (for the neat copolymer and its nanocomposites). Also, PDI values of the nanocomposites are higher than the neat poly (styrene-co-methyl methacrylate). This can be ascribed to the impurity role of the diatomite platelets. The occurrence of termination and transfer reactions between the propagating (macro)radicals and diatomite platelets can be considered as the main reason for emerging high PDI values in the nanocomposites samples. 22,23
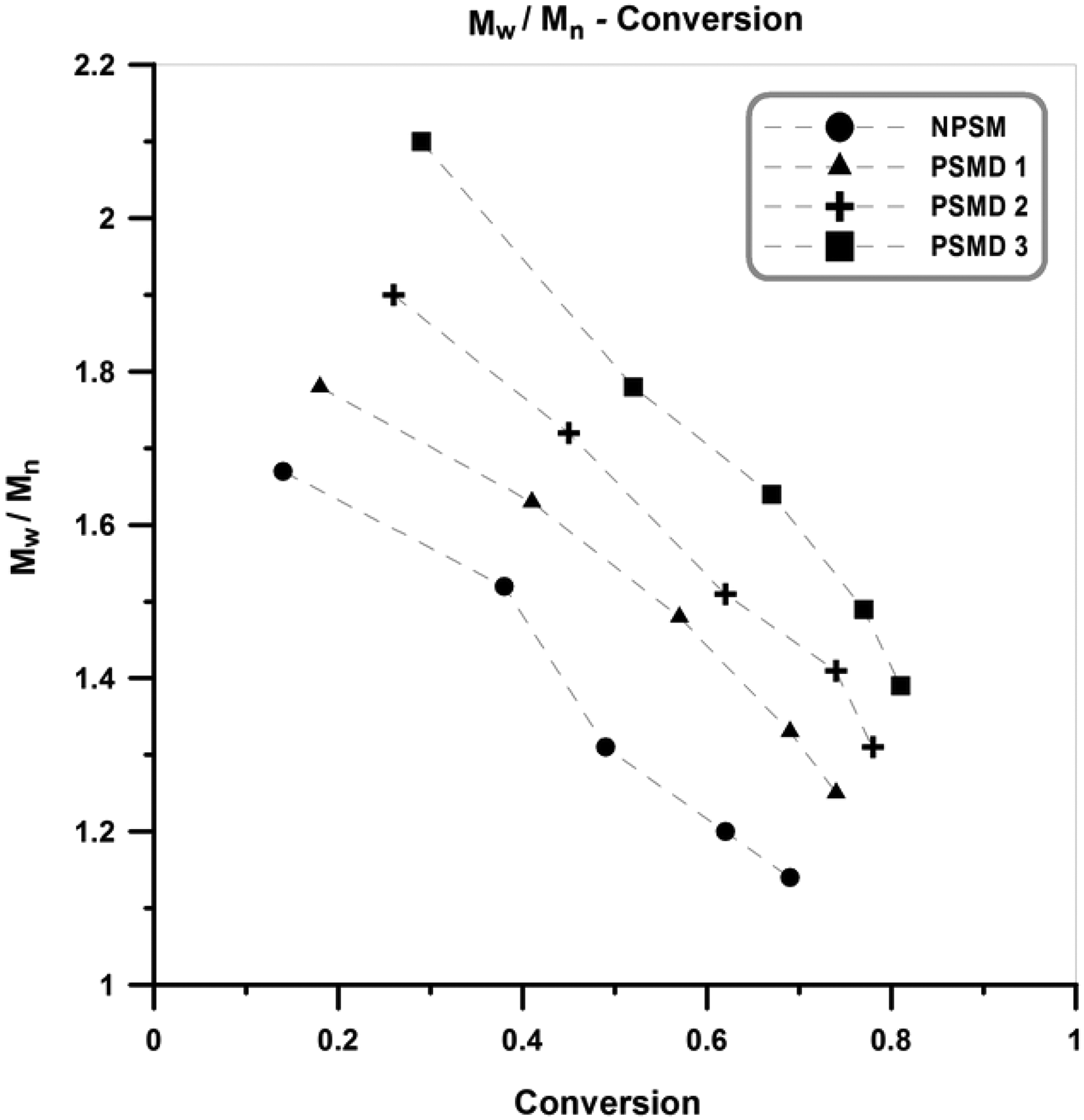
PDI values versus conversion for the neat copolymer and its different nanocomposites. PDI: polydispersity index.
Pseudo-first-order kinetic plots for the synthesized nanocomposites and the neat poly (styrene-co-methyl methacrylate) are represented in Figure 5. Since the slope of the curves remains fairly constant in the whole of copolymerization, it can be concluded that the steady-state kinetics is obtained (for all the experiments). In all the samples, L n(M 0/M) increases linearly with reaction time in which constant radical concentration in the copolymerization medium can be concluded. 22 According to the results, the rate of the copolymerization increases by adding diatomite platelets content.
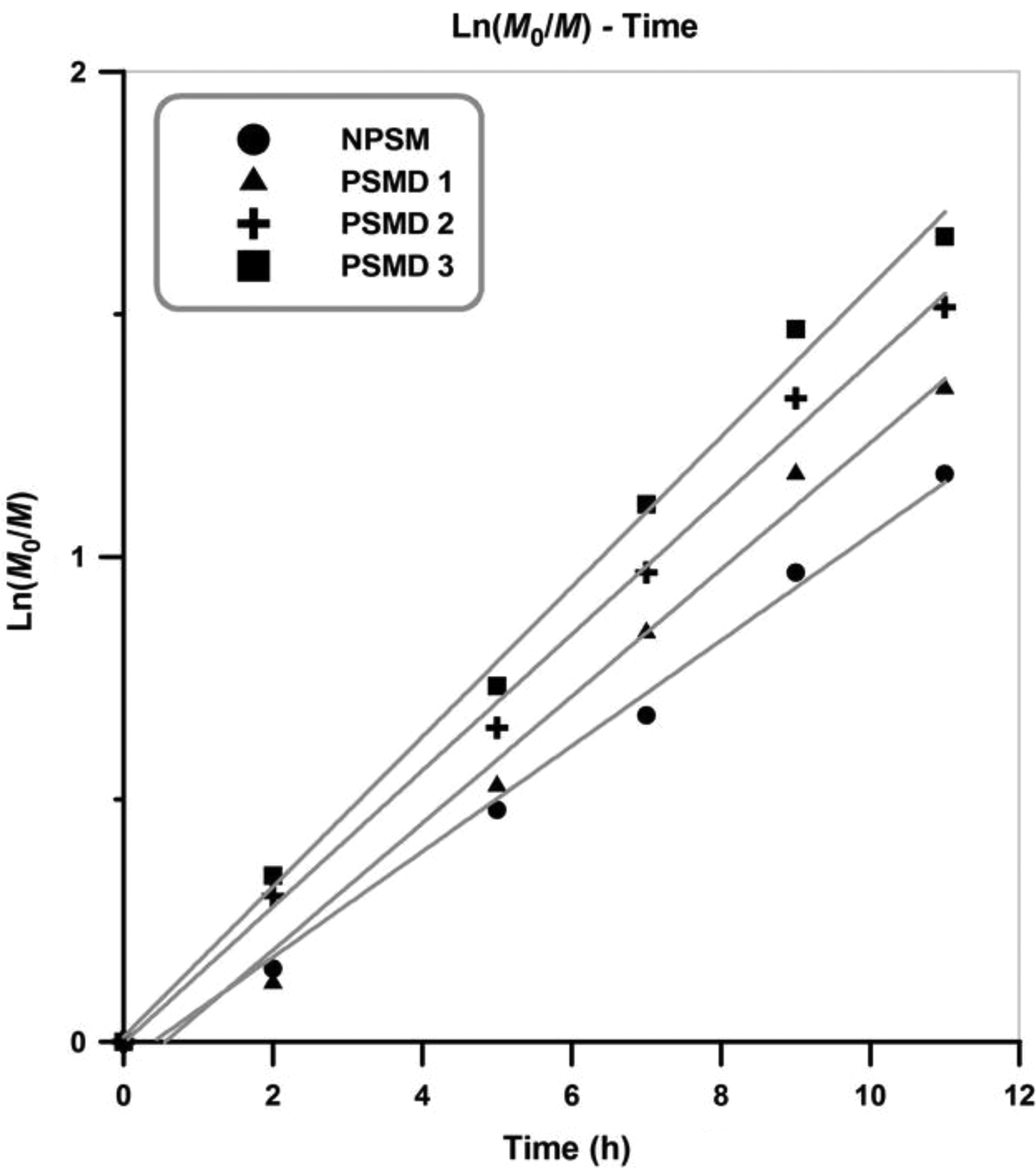
Kinetic plot with respect to reaction time.
Figure 6 represents SEC traces of the samples. According to this figure, SEC traces of the neat poly (styrene-co-methyl methacrylate) and all of the nanocomposites display monomodal peaks corresponding to the molecular weight values predetermined by the molar ratio of monomer to the initiator. Pure poly (styrene-co-methyl methacrylate) reveals narrow distribution and low PDI value which can demonstrate that successful normal ATRP is established.
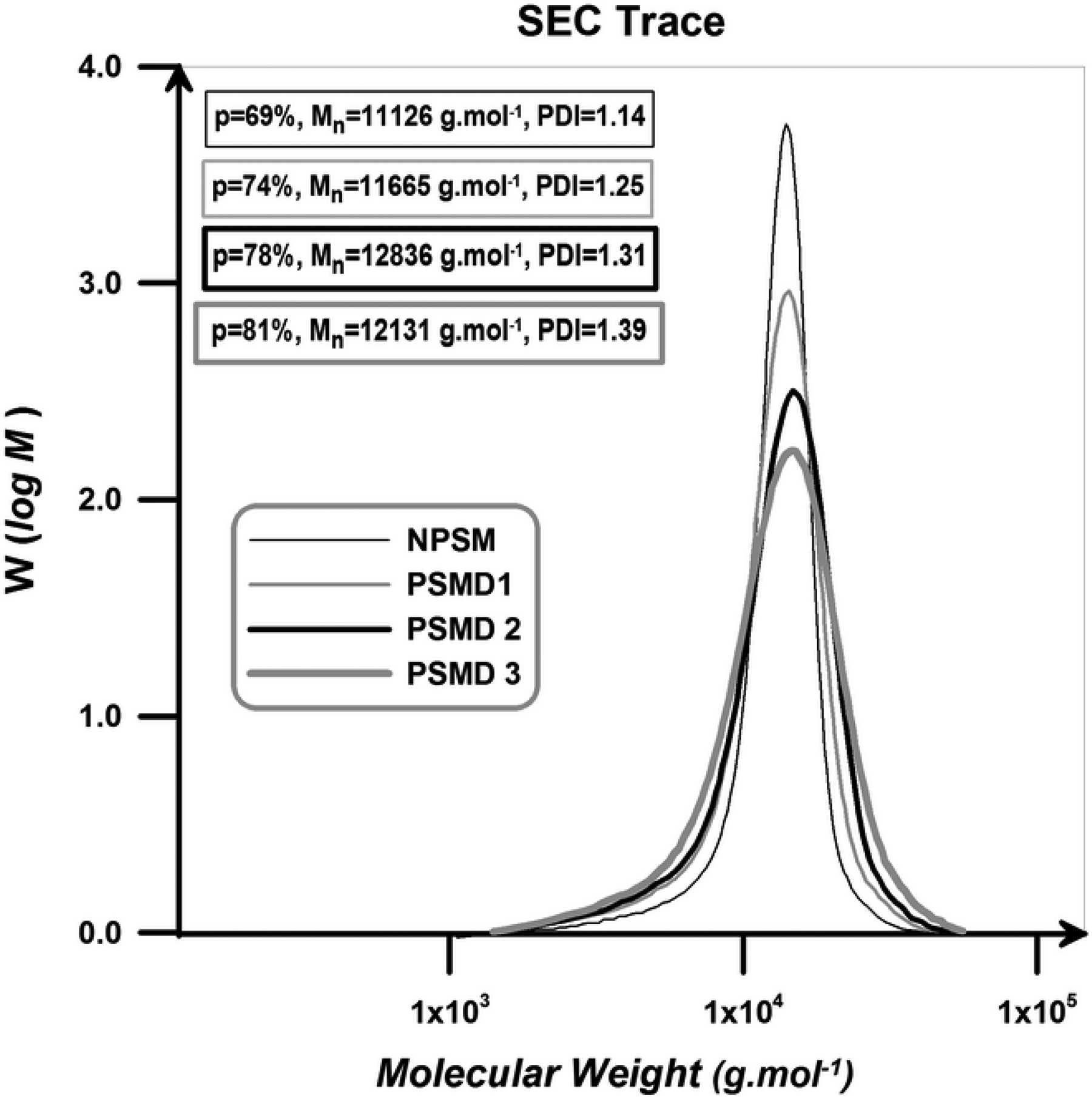
SEC traces of the neat poly (styrene-co-methyl methacrylate) and its nanocomposites. SEC: size-exclusion chromatography; MMA: methyl methacrylate.
Normal ATRP of St and MMA without diatomite platelets results in well-defined poly (styrene-co-methyl methacrylate) chains with low PDI value. By adding the diatomite platelets, final conversion and molecular weight were increased. By the addition of only 3 wt% of the diatomite platelets, final conversion increases from 69% to 81%. The positive effect of the diatomite platelets on the molecular weight of the samples is generally attributed to the pendant hydroxyl groups on the surface (and pores) of the diatomite platelets. It is demonstrated that polar solvents (especially hydroxyl containing ones like water, phenol, and carboxylic acids) exert a rate acceleration effect on the copolymerization systems for increasing radical activation rate and also reducing radical recombination rate. Pendant hydroxyl groups on the surface of the diatomite platelets can possibly cause a polarity change into the reaction medium. In addition, negatively charged surface (pendant hydroxyl groups on the surface of the diatomite platelets) could absorb and gather positively charged catalyst (Cu ions at this work) and consequently enhances the chain growth rate. The accelerating effect of other nanofillers, such as clay nano-sheets and MCM-41 nanoparticles on polymerization rate was also achieved elsewhere. 21,22 PDI values of the copolymer chains increase by the addition of diatomite platelets loading. Diatomite platelets act as an impurity in the copolymerization medium and therefore cause the molecular weight distribution of the resultant copolymers to be increased; PDI value increases from 1.14 to 1.39 by 3 wt% loading of diatomite platelets. 24 Table 2 presents the extracted data from SEC traces of the neat poly (styrene-co-methyl methacrylate) and its nanocomposites.
Molecular weights and PDI values of the extracted copolymers resulted from SEC traces.
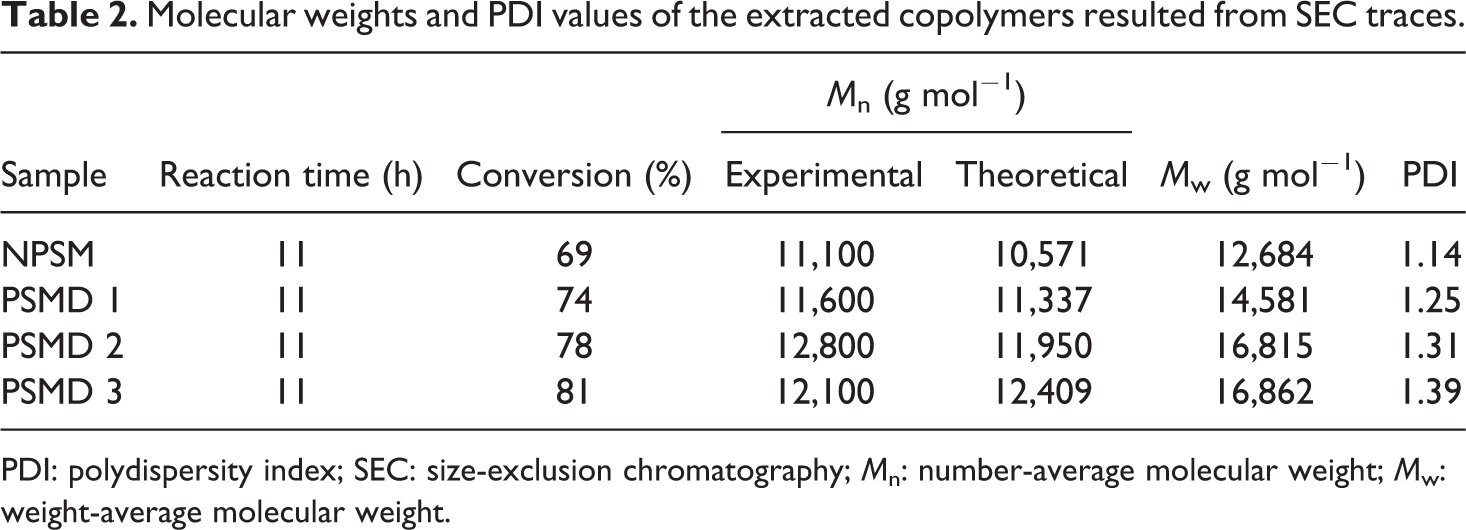
PDI: polydispersity index; SEC: size-exclusion chromatography; M n: number-average molecular weight; M w: weight-average molecular weight.
Theoretical molecular weight is calculated by using equation (1):
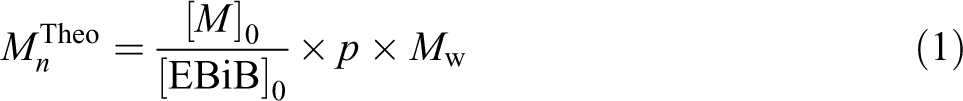
where [M]0 and [EBiB]0 are the initial concentration of the monomers and ATRP initiator, respectively. Conversion is denoted by p and the symbol of the average molecular weight of the monomers is M w.
Thermal stability of the neat poly (styrene-co-methyl methacrylate) and its various nanocomposites are studied using TGA. TGA thermograms of weight loss as a function of temperature in the temperature window of 25–700°C in addition to their corresponding differential thermogravimetric (DTG) curves are shown in Figure 7.
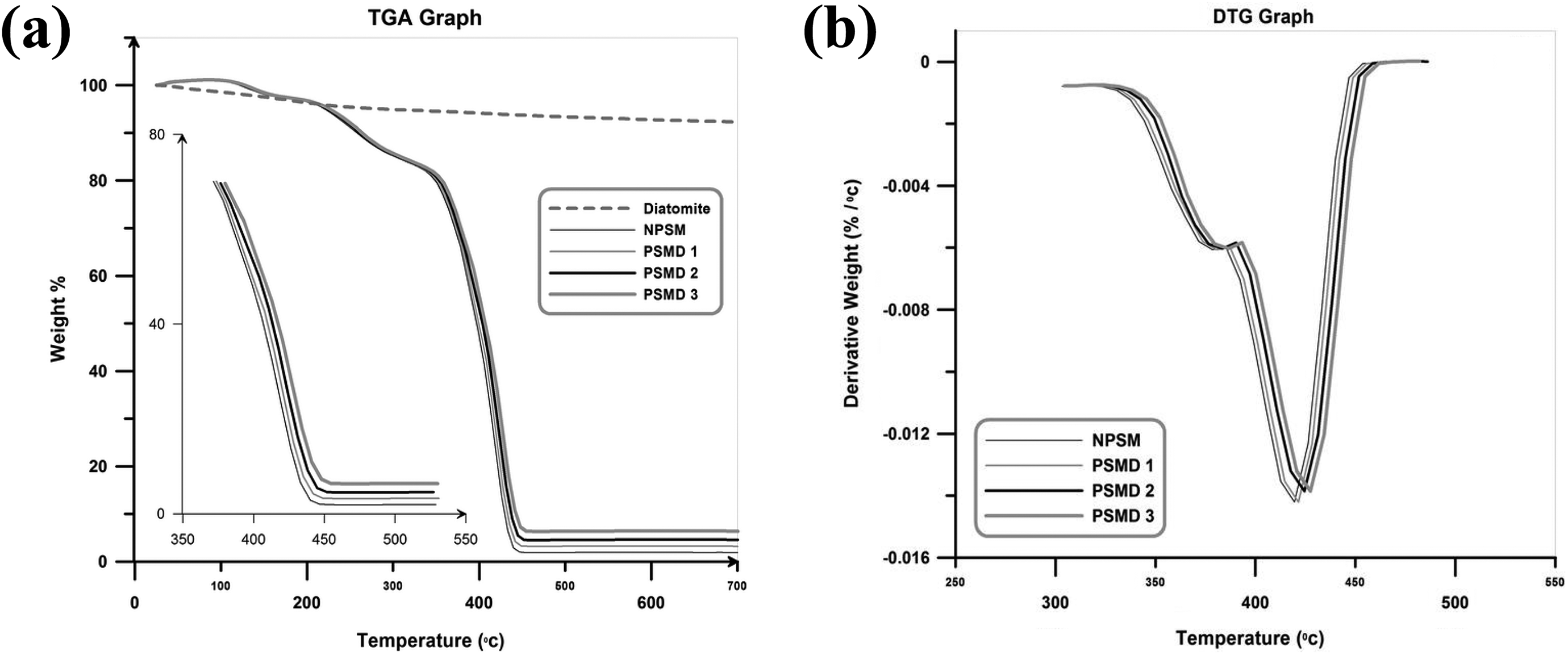
(a) TGA and (b) DTG thermograms of the neat poly (styrene-co-methyl methacrylate) and its nanocomposites. TGA: thermal gravimetric analysis; DTG: differential thermogravimetric; MMA: methyl methacrylate.
As it can be seen in Figure 7, the thermal stability of the neat poly (styrene-co-methyl methacrylate) is lower than all of the nanocomposites. In addition, the thermal stability of the neat poly (styrene-co-methyl methacrylate) is improved by introducing diatomite platelets, which by increasing diatomite platelets content, an increase in degradation temperature occurred. In general, by increasing temperature in the TGA graphs, three separated steps can be recognized; evaporation of the water molecules (at the temperature range of 100–150°C), degradation of volatile materials such as residual monomer and low-molecular-weight oligomers (at the temperature window around 180–350°C), and degradation of the synthesized copolymer and nanocomposites (at the temperatures above 380°C). Figure 8 graphically illustrates the extracted data from TGA graphs. Degradation temperature of the samples versus the amount of degradation is employed to show that the addition of diatomite platelets in the poly (styrene-co-methyl methacrylate) matrix results in an improvement of thermal stabilities of the nanocomposites (T X: temperature threshold at which X% of poly (styrene-co-methyl methacrylate) and its nanocomposites is degraded). Char values at 700°C are given in Table 3. As expected, the char values increase by increasing diatomite platelets content.
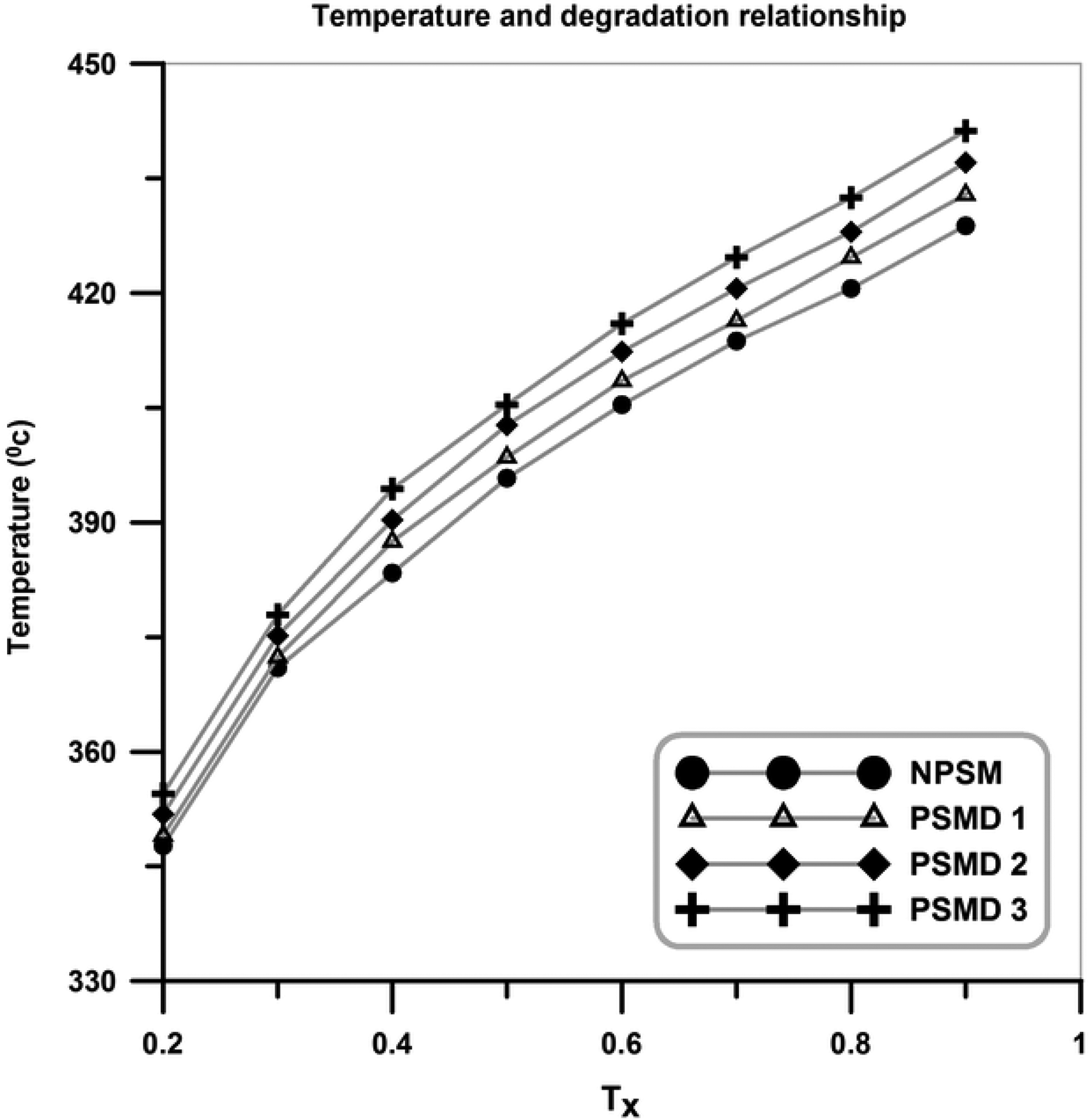
Graphical illustration of temperature and degradation relationship.
Char values and peak points (final degradation step) of the neat poly (styrene-co-methyl methacrylate) and its composites.

TGA: Thermal gravimetric analysis; DTG: differential thermogravimetry.
Improvement in thermal stability of the nanocomposites by adding diatomite platelets can be attributed to the high thermal stability of diatomite platelets and also the interaction between diatomite platelets and copolymer matrix. 25 Physical interaction between poly (styrene-co-methyl methacrylate) chains and the surface of the diatomite platelets is an important factor for increasing the thermal stability of the nanocomposites. 26 Additionally, hindrance effect of the diatomite platelets on the copolymer chains movement and restriction of oxygen permeation by these sheets are the other reasons for the higher thermal stability of the nanocomposites. Similar conclusions are also achieved in the case of polymer/clay nanocomposites. 27,28 According to the DTG curves, a shoulder in the final degradation step has occurred which can be attributed to the MMA segments of the copolymer. In the case of MMA, head-to-head linkages, chain unsaturation, and random scission can be achieved; therefore, its thermogram reveals several degradation steps. 29 Extracted data from TGA curves (char value) and DTG curves (peak point in final degradation step) are also summarized in Table 3. Pristine diatomite leaves 92.27% char after complete degradation at 700°C.
DSC analysis is employed to evaluate the effect of diatomite platelets on the chain confinement of the pure poly (styrene-co-methyl methacrylate) and its nanocomposites and also the determination of T g of them. DSC curves of the neat poly (styrene-co-methyl methacrylate) and its different nanocomposites are presented in Figure 9. Temperature range of 20–200°C is used to describe DSC results in the heating path. Diatomite platelets do not bear any transitions in this range of temperature; therefore, only thermal transition of copolymers is observed. In these experiments, samples are heated from room temperature to 220°C to remove their thermal history. Then, they cooled to room temperature to distinguish the phase conversion and other irreversible thermal behaviors. Finally, samples are heated from room temperature to 220°C to obtain T g values.
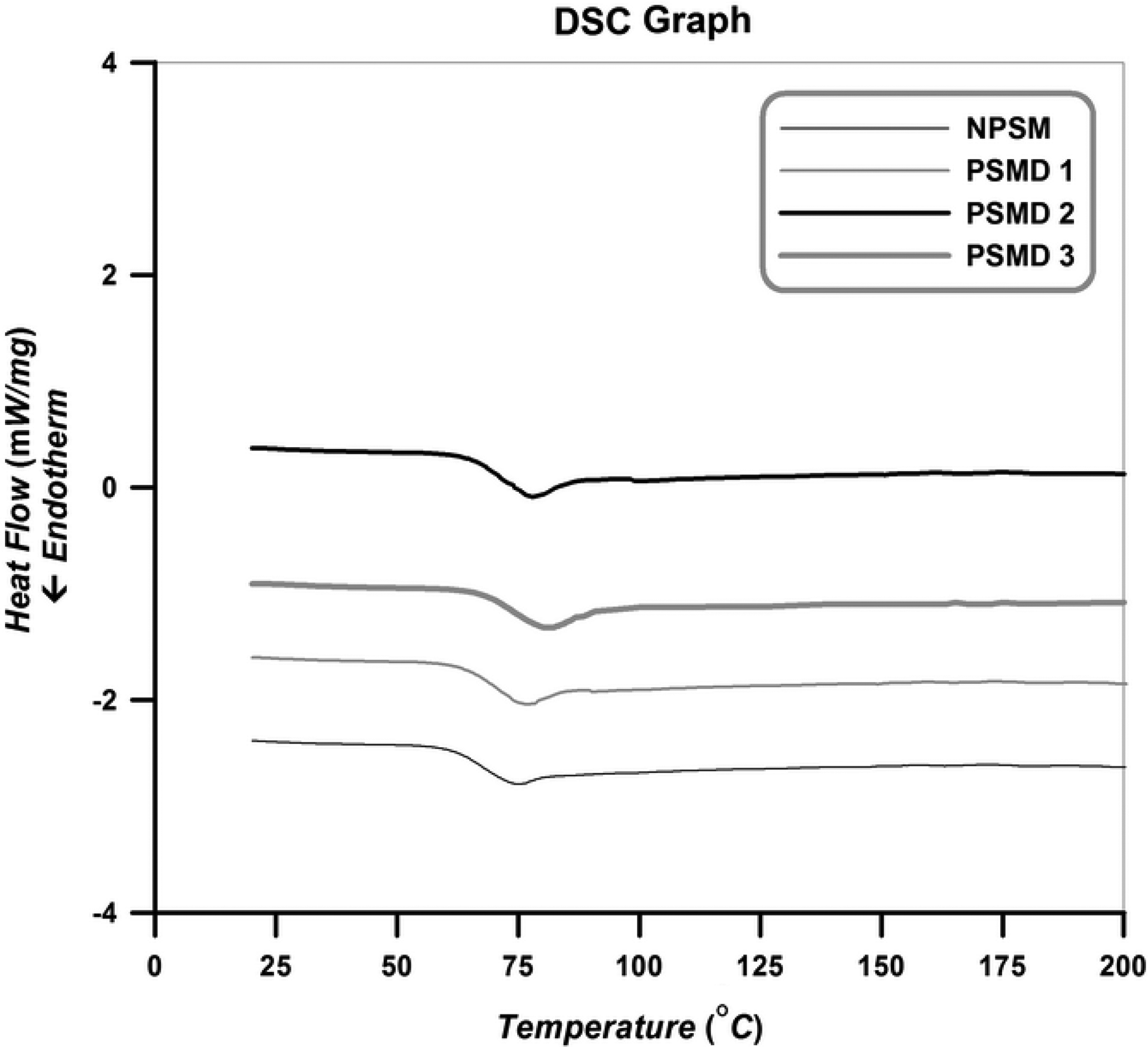
DSC curves of the neat poly (styrene-co-methyl methacrylate) and its different nanocomposites (heating path). DSC: differential scanning calorimetry; MMA: methyl methacrylate.
According to Figure 9, an obvious inflection in the heating path is occurred, which shows T g of the samples. The corresponding inflection in the cooling path has also appeared. Since there is not another peak in the cooling path, amorphous structure of the synthesized poly (styrene-co-methyl methacrylate) and its nanocomposites can be concluded. Extracted T g values of the samples from DSC curves are summarized in Table 4.
Extracted T g of the neat poly (styrene-co-methyl methacrylate) and its nanocomposites.
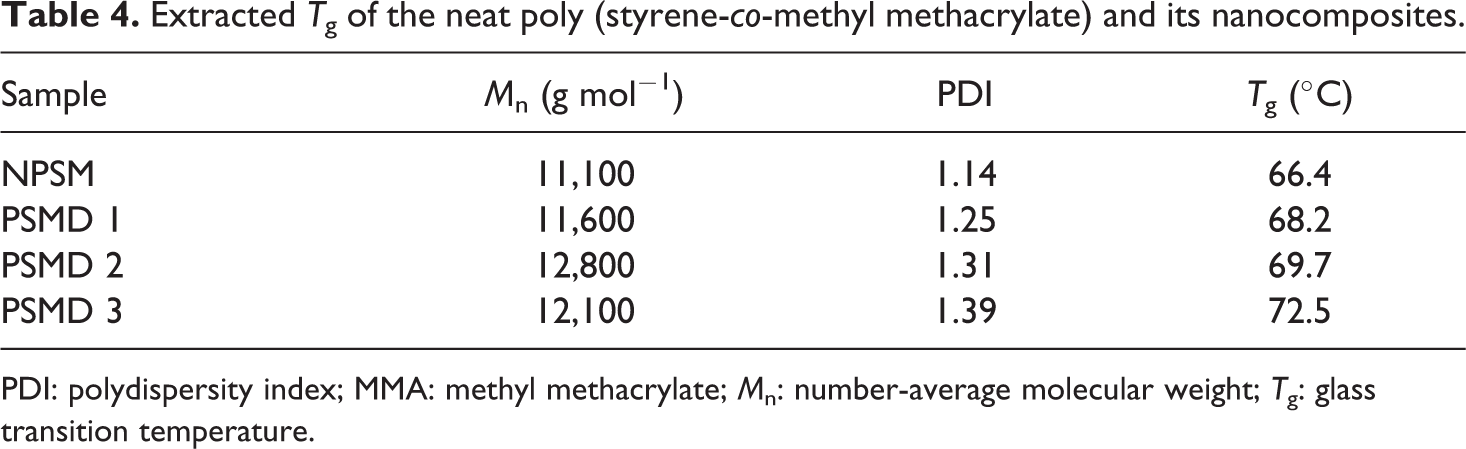
PDI: polydispersity index; MMA: methyl methacrylate; M n: number-average molecular weight; T g: glass transition temperature.
As it can be seen in Table 4, T g value of the neat poly (styrene-co-methyl methacrylate) is lower than all of the nanocomposites and an increase in T g values occurred by increasing of diatomite platelets loading. Increasing T g values by adding diatomite platelets content in the poly (styrene-co-methyl methacrylate) matrix can be attributed to the confinement effect of the diatomite platelets. The rigid two-dimensional diatomite platelets can restrict the steric mobility of the poly (styrene-co-methyl methacrylate) chains and cause the inflection in the DSC curves starts at higher temperatures. Similar conclusions are also reported in the case of polymer/clay nanocomposites. 27,28
Conclusions
Well-defined poly (styrene-co-MMA)/diatomite nanocomposites with different diatomite loadings are prepared via in situ ATRP. XRF results indicate that silica is the main constituent of the pristine diatomite. Mesoporous structures of the pristine diatomite with pore diameter around 40 nm are concluded from nitrogen adsorption/desorption isotherm and TEM images. In situ ATRP of St and MMA in the presence of mesoporous diatomite platelets leads to the increment of conversion from 69% to 81%. Moreover, molecular weight and PDI values increase from 11,100 g mol−1 to 12,100 g mol−1 and from 1.14 to 1.39, respectively. Improvement in thermal stability of the nanocomposites and increasing T g values from 66.4°C to 72.5°C were also observed by the incorporation of 3 wt% of mesoporous diatomite platelets.
Supplemental material
Supplemental Material, Supporting_Information - Influence of mesoporous diatomite on atom transfer radical random copolymerization of styrene and methyl methacrylate: Kinetics and thermal studies
Supplemental Material, Supporting_Information for Influence of mesoporous diatomite on atom transfer radical random copolymerization of styrene and methyl methacrylate: Kinetics and thermal studies by Khezrollah Khezri and Yousef Fazli in Journal of Thermoplastic Composite Materials
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
