Abstract
In polymer nanocomposites, graphene is possibly the most promising nanofiller. Graphene produces impressive properties for polymers at very low filler content, which makes it highly interesting in building high-performance materials compared to other classes of polymer nanocomposites. Graphene-modified polymer nanocomposites have attracted much attention in scientific literature because of the need of superior materials with desirable properties such as electrical, mechanical, thermal, flame retardant, and gas barrier. Frequent studies have been attempted to produce graphene–polyamide (G-PA) nanocomposites with novel and improved properties. Based on this review, one can identify the synthesis technique and preparation for G-PA nanocomposites, which can further be useful in numerous applications.
Introduction
Since the discovery of graphene by Geim and Novoselov, demand of graphene is increasing with wide array of applications, encompassing unique and remarkable properties like mechanical, electrical, thermal, physical, and optical. Graphene is considered as one of the most promising new materials. 1 However, researchers across the globe are still looking for a new dimension to produce lightweight, low-cost, and high-performance composite materials. Graphene have been used as one of the assuring nanofiller material to produce graphene–polyamide nanocomposites (G-PA). G-PA comprises a class of materials that are multiphase hybrid structures, where one of the phases has one, two, or three dimensions (1-D, 2-D, or 3-D) of less than 100 nm. Industrial and academic research on G-PA was pursued to provide added superior properties compared to the conventional polymer. Graphene provides substantial properties enhancements to G-PA, even at very low amount of nanofillers content. G-PA have been widely reported in the scientific literature. 2 Graphene is 2-D nanostructure in a hexagonal lattice or honeycomb lattice. Graphene is regarded as the fundamental building block for other allotropes, as shown in Figure 1. 3 Graphene is a 2-D honeycomb lattice of carbon atoms, stacked up into many layers graphite, rolled up into 1-D carbon nanotube, and it can be wrapped up into zero-dimensional (0-D) fullerene.
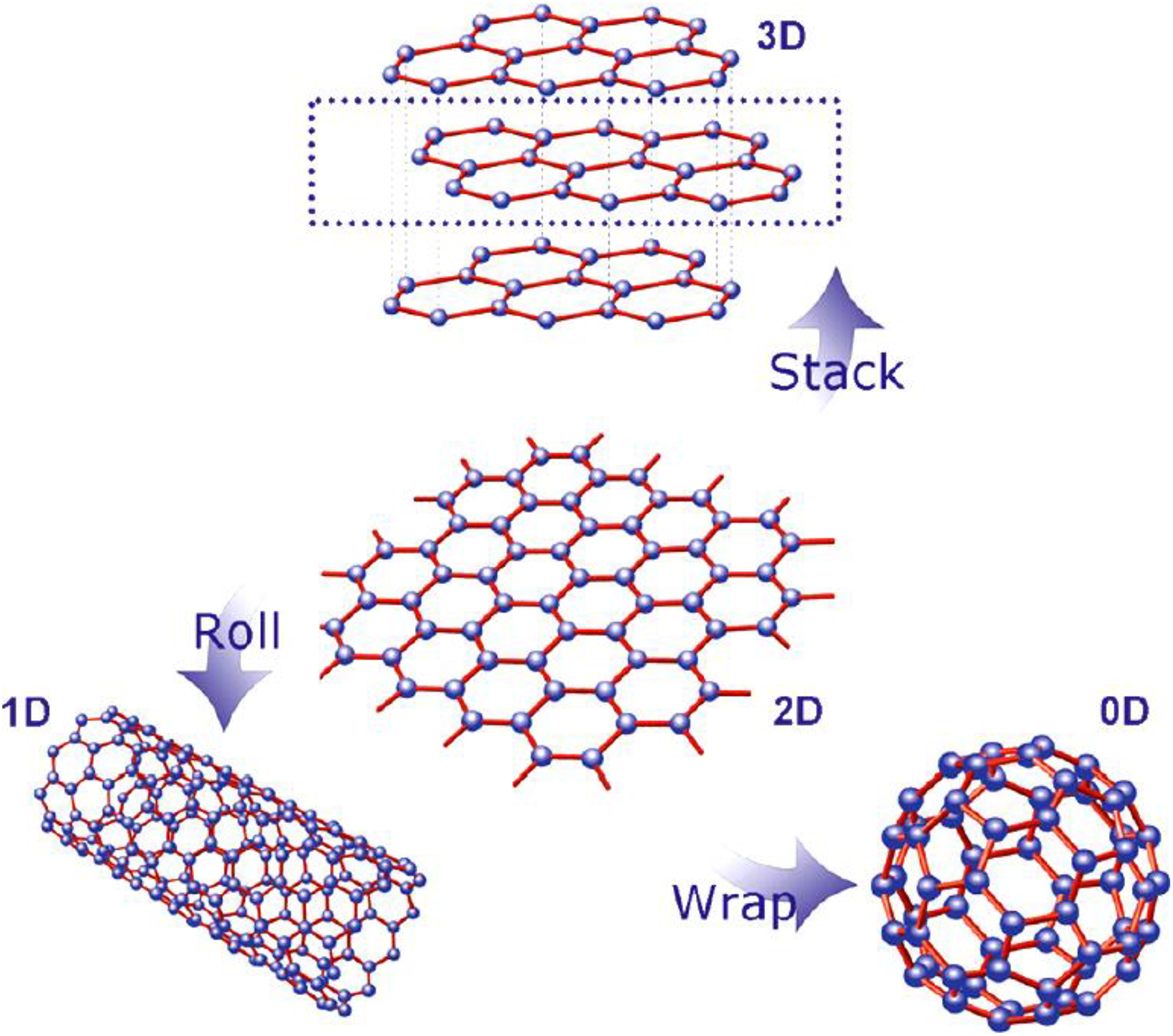
Graphene is regarded as the fundamental building block for other carbon allotropes.
Graphene consists of carbon atoms in a honeycomb lattice on a 2-D plane. Three atomic orbitals from carbon atoms are hybridized into sp2 orbitals. These sp2 orbitals form covalent bonds with the carbon atoms. The carbon atoms separated by a distance of 1.42°A from each other and this hybridized sp2 orbitals form hexagonal planar structure called honeycomb lattice (Figure 2). Graphene possesses excellent properties to its crystal structure. Due to the unique arrangement of atoms through sp2 hybridization, graphene has many characteristics, such as thermal conductivity 5000 W m−1 K−1, electrical conductivity 6000 s cm−1, strength 130 GPa, electron mobility 200,000 cm2 Vs−1, Young’s modulus 1.0 TPa, specific surface area 2630 m2 g−1, and optical transmittance approximately 98%. 2
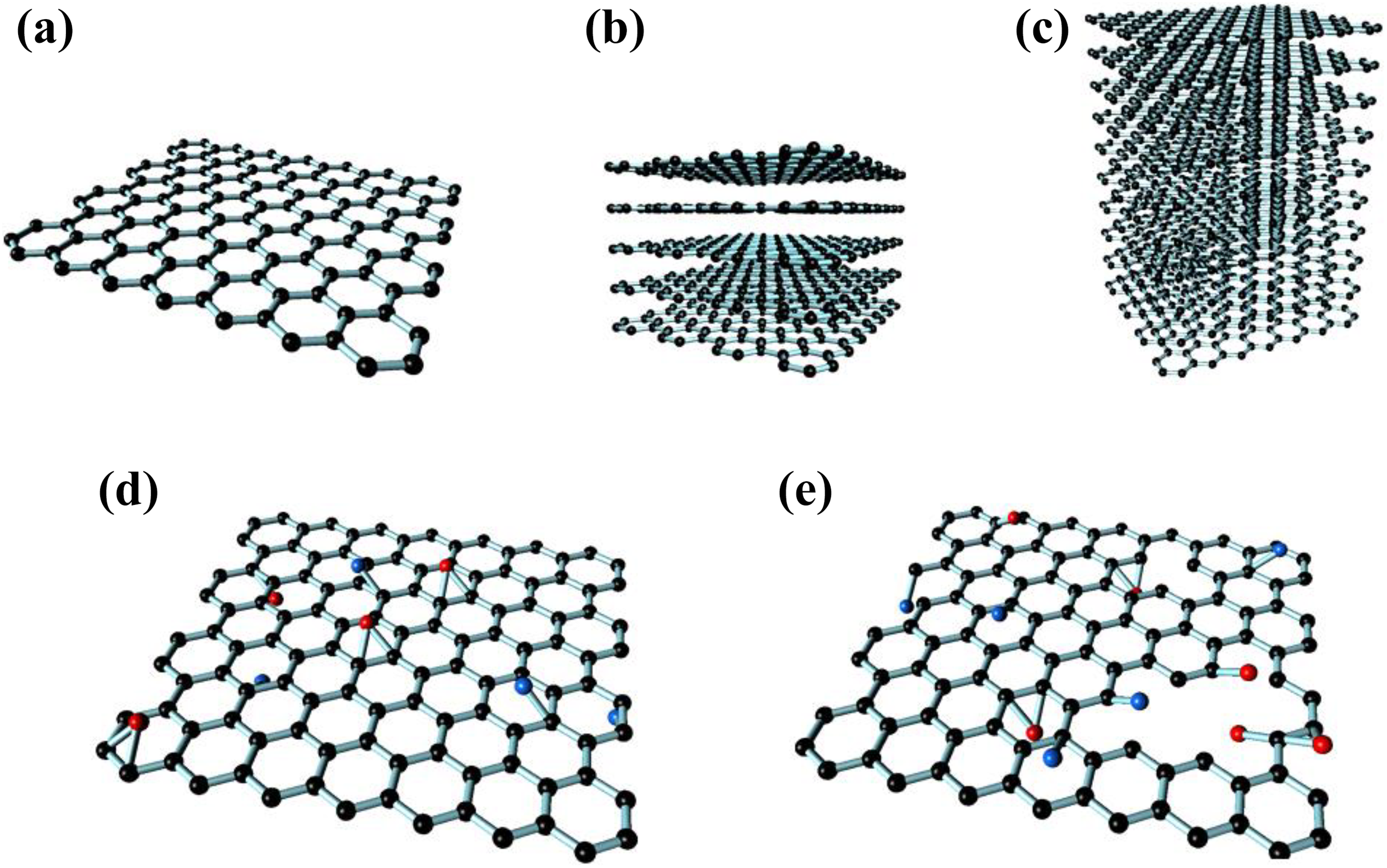
Graphene derivatives. (a) Graphene, (b) few-layer graphene, (c) graphite, (d) reduced graphene oxide (rGO), and (e) graphene oxide (GO).
Polyamide (PA) is an attractive thermoplastic having repeating amide –CO–NH– linkages with better process ability and chemical resistance and is broadly used in engineering plastic. PA can be classified into two types such as aliphatic PA and aromatic PA (Figure 3). PAs are among the most important as they posed important material properties like high strength and stiffness. However, it has some weaknesses such as low thermal stability, poor dimensional stability, and low electrical conductivity as well as exceptionally its mechanical performance is varied with aromatic and aliphatic PA. Therefore, many works have been undertaken to improve its comparative properties through the nanofillers. PA known as nylons are used as fibers (80–90%), as engineering plastics, adhesives, and rubbers. PA can be obtained from ring opening polymerization, respectively, or polycondensation of diamine with dibasic acids. This review is a summary of the recent synthesis on G-PA nanocomposites.
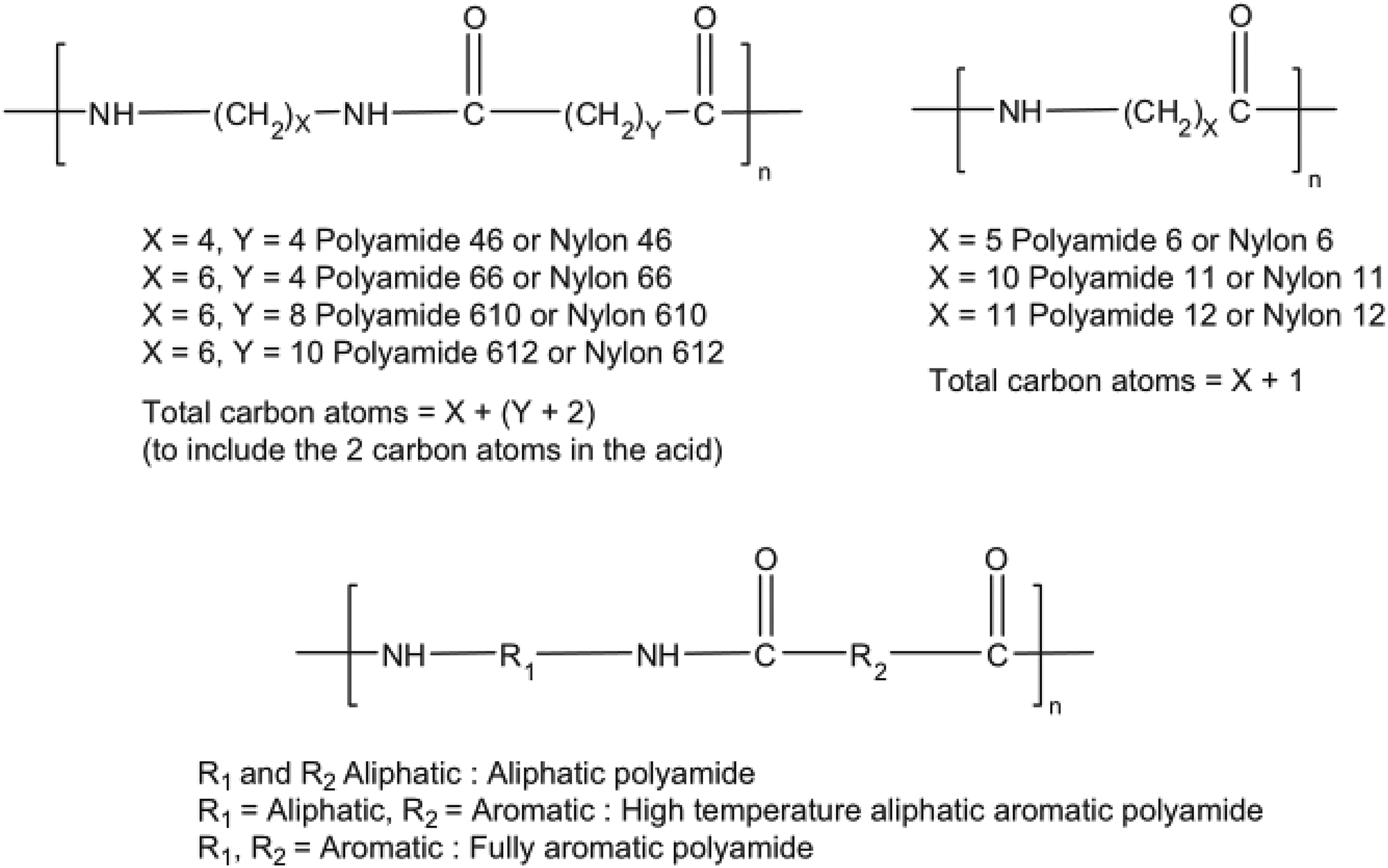
Polyamide nomenclature aromatic and aliphatic.
Preparation techniques for G-PA nanocomposites
In recent years, significant signs of progress have been made in synthesis and performances of G-PA. Graphene-filled PA materials have been considered as a stimulating route for creating a new type of high-performance material that combines the advantages of polymer and the merits of graphene. These materials provide unique behavior and improved properties at low nanoparticles content compared to the neat polymer. 6 Basically, the selection of the synthetic techniques depends on uniform dispersion of graphene, exfoliation, and the interfacial interaction between the graphene and polymer. At present, solution blending, in situ polymerization, and melt blending are three main methods to prepare G-PA nanocomposites. 2 From the mentioned techniques, in situ polymerization and solution mixing have been used for the chemical interaction between the polymer and the graphene. Use of hazardous chemicals and low yield with high cost are two major demerits for solution mixing. To overcome this problem, scientists and researchers got fascinated toward melt mixing technique as it is solvent-free and environment-friendly, which can fabricate G-PA nanocomposites at a large scale. Melt route has been modified to influence the dispersion quality. 7 Therefore, for better dispersion of graphene in polymeric material, a combined solution and melt route has been designed. 8 G-PA nanocomposite obtained by combination approach has shown improved.
Moreover, the description summarizes the important preparation techniques of G-PA nanocomposites in recent years as mentioned in Table 1. Hence, we can see an obviously increase and improvement in the mentioned properties, which have been captivated by combination approach to work in this field fruitfully.
G-PA nanocomposites.
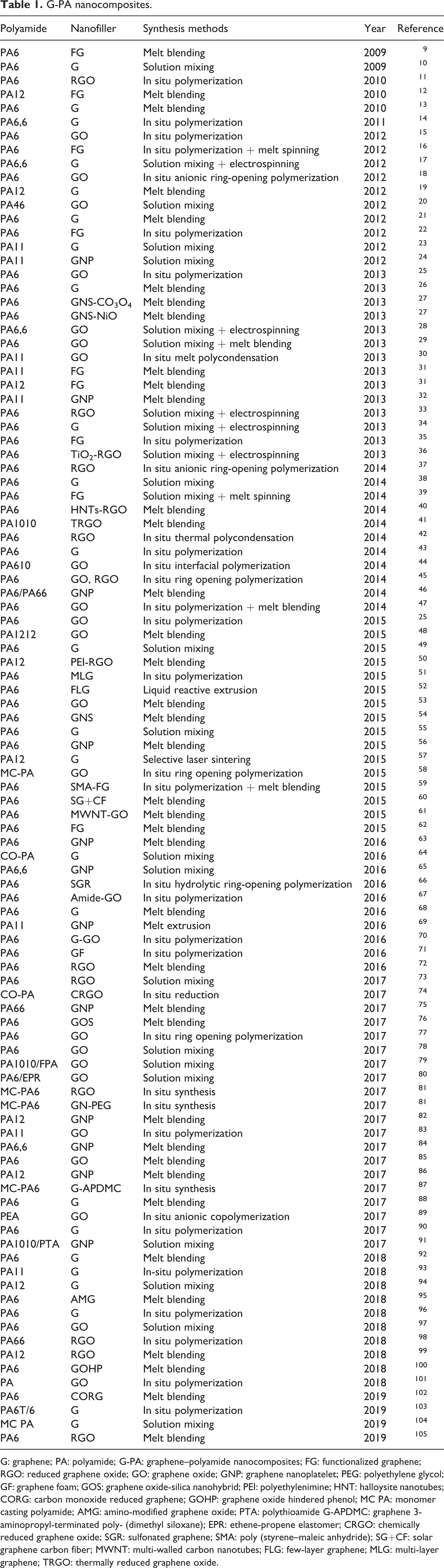
G: graphene; PA: polyamide; G-PA: graphene–polyamide nanocomposites; FG: functionalized graphene; RGO: reduced graphene oxide; GO: graphene oxide; GNP: graphene nanoplatelet; PEG: polyethylene glycol; GF: graphene foam; GOS: graphene oxide-silica nanohybrid; PEI: polyethylenimine; HNT: halloysite nanotubes; CORG: carbon monoxide reduced graphene; GOHP: graphene oxide hindered phenol; MC PA: monomer casting polyamide; AMG: amino-modified graphene oxide; PTA: polythioamide G-APDMC: graphene 3-aminopropyl-terminated poly- (dimethyl siloxane); EPR: ethene-propene elastomer; CRGO: chemically reduced graphene oxide; SGR: sulfonated graphene; SMA: poly (styrene–maleic anhydride); SG+CF: solar graphene carbon fiber; MWNT: multi-walled carbon nanotubes; FLG: few-layer graphene; MLG: multi-layer graphene; TRGO: thermally reduced graphene oxide.
In situ polymerization
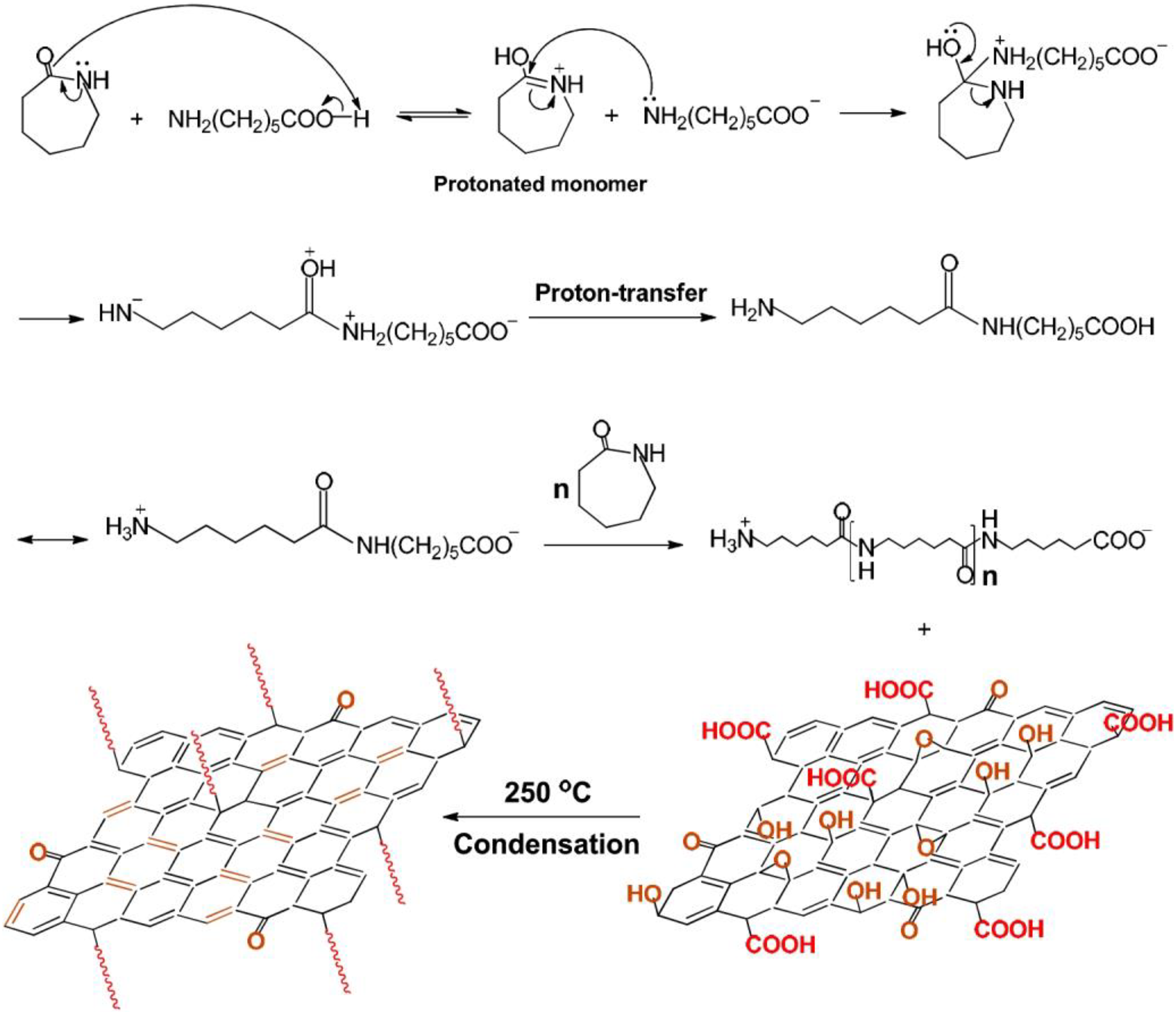
In situ polymerization is a suitable technique for the preparation of polymer nanocomposites based on graphene. In this method, monomers present in and out of the graphene interlayers to polymerize to form polymer nanocomposites. In situ polymerization is the important technique for graphene/PA6 nanocomposites. Xu et al. 11 Prepared graphene/PA6 nanocomposites using in situ ring-opening polymerization of caprolactam in the presence of GO. This technique opens a new possibility to fabricate graphene-based nanocomposites of polymers and offers wide range of applications. The nanocomposite prepared by condensation reaction between carboxylic graphene oxide (GO) and amino ends of PA6 chains. Graphene showed good solubility and compatibility to PA6. The blue bonds in the PA6-grafted graphene represent restored conjugated bonds through high-temperature reduction and the red curves represent the grafted PA6 macromolecular chains (Figure 4).
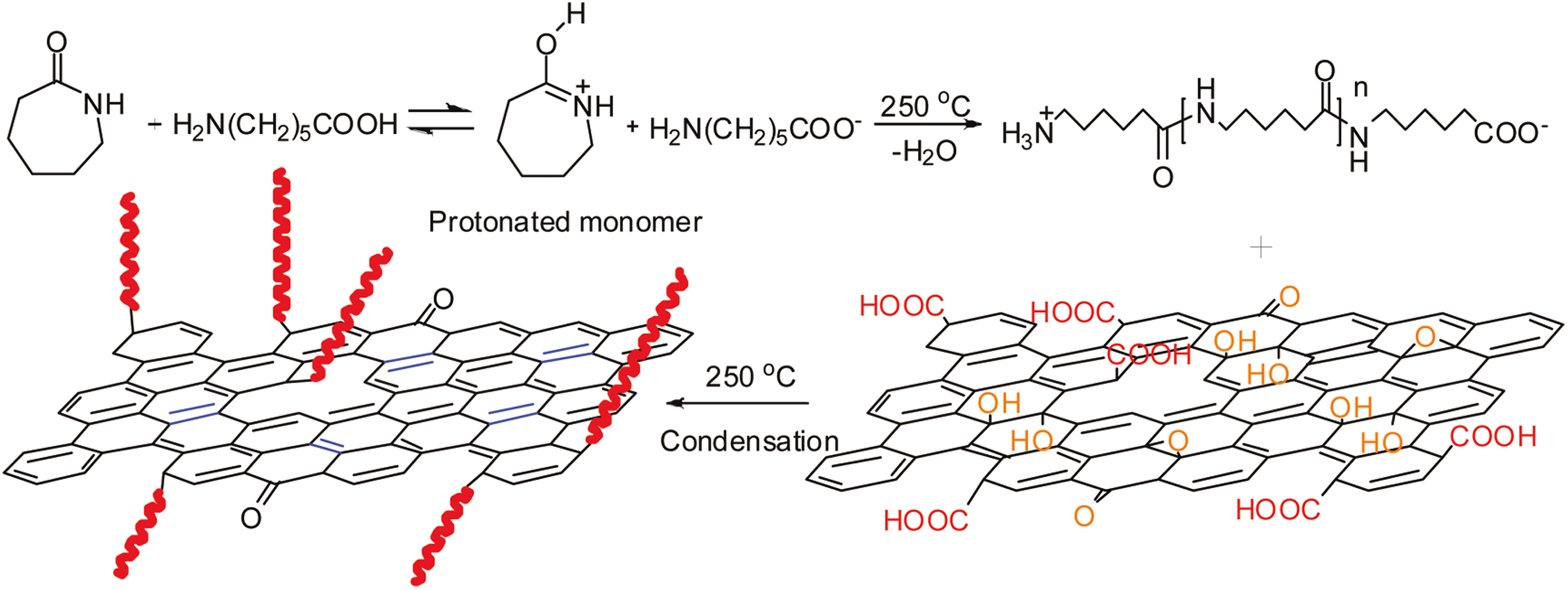
Schematic of PA graphene composites by in situ ring-opening polymerization of caprolactam in the presence of GO.
Das et al. 14 have reported a novel in situ polymerization technique to develop localized polymer coatings on the surface of dispersed pristine graphene sheets and successfully coat the graphene noncovalently with PA610 and PA66, and the stability of the graphene dispersion in water is enhanced. Zheng et al. 15 have also reported the in situ polymerization of GO/PA6 nanocomposites. These nanocomposites show a low percolation threshold of approximately 0.41 vol% and high electrical conductivity of approximately 0.028 S m−1 with only approximately 1.64 vol% of GO. This method showed potential for the mass production of electrically conductive polymer/reduced graphene oxide (RGO) nanocomposites. Dixon et al. 25 developed GO-PA6 nanocomposites by in situ polymerization technique. The nanocomposite prepared by direct synthesis of the polymer in the presence of the GO/water dispersion. High levels of interfacial interaction improved degradation temperature and tensile strength 64.9 MPa at 0.65 wt% was obtained.
Zhang et al. 18 prepared GO-reinforced PA6 composites via in situ anionic ring-opening polymerization. A two-step new and facile method was employed in the synthesis of PA6-GO nanocomposites. The ε-caprolactam (CL) was firstly fixed onto the GO sheets coupling by 4,4′-methylenebis (phenyl isocyanate), and then PA6 was grafted from the GO surface by in situ anionic ring-opening polymerization. The polymerization processing was effectively carried out at relatively low reaction temperature (150°C) and in a short reaction time (20–40 min) by using a caprolactam magnesium bromide (C1) initiator in combination with a difunctional hexamethylene-1,6-dicarbamoylcaprolactam (C20) activator (Figure 5).
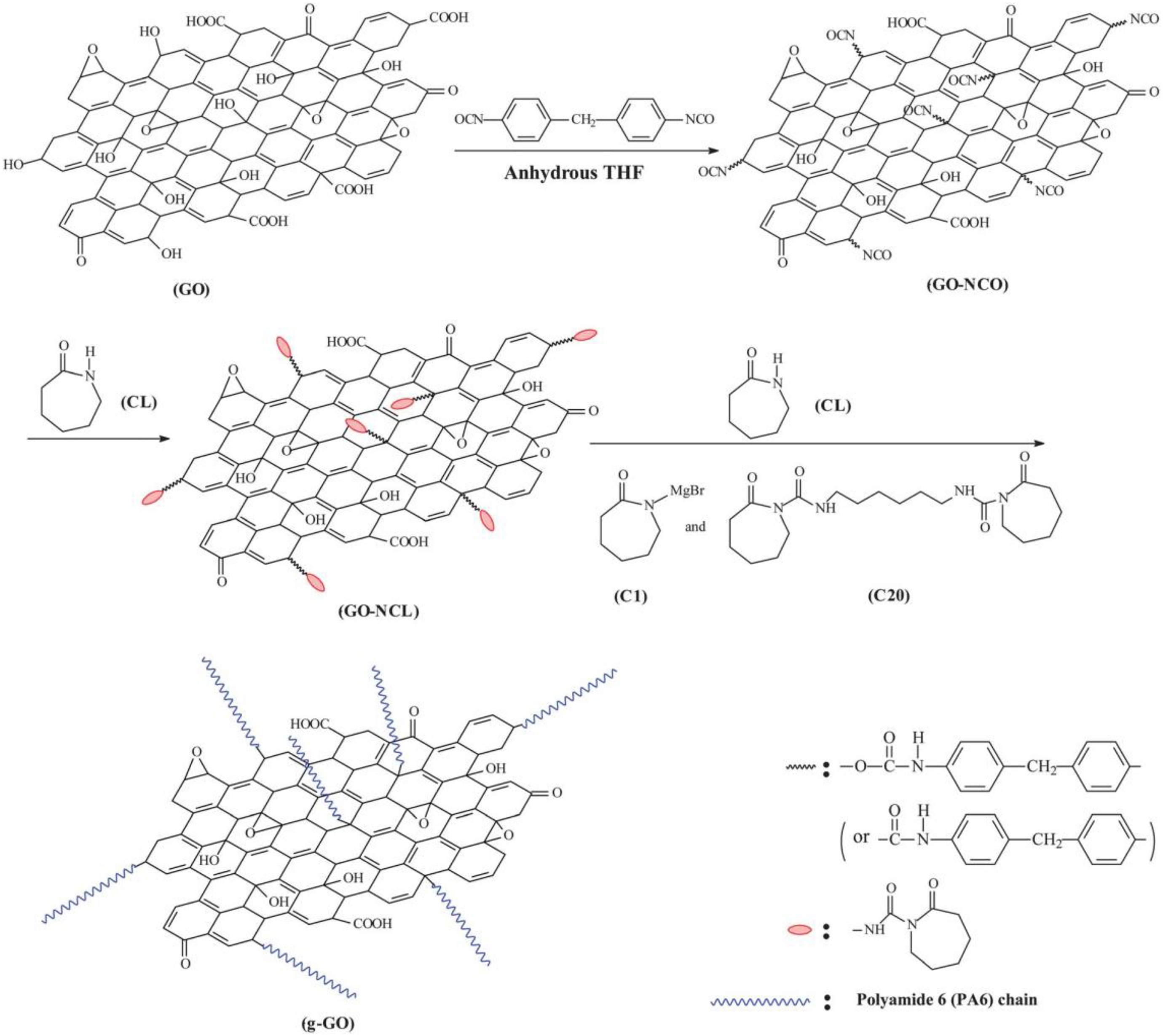
Schematic representation of graphene oxide with caprolactam and subsequent grafting of PA6 by in situ anionic ring-opening polymerization.
Ding et al. 37 reported the thermal conductive PA6/graphene (PG) composite was synthesized by in situ ring-opening polymerization reaction using CL as the monomer, 6-aminocaproic acid as the initiator, and RGO as the thermal conductive filler. The uniform dispersion and interconnected structure of RGO sheets in the nanocomposite helped to improve the λ from 0.196 W m−1 K−1 of neat PA6 to 0.416 W m−1 K−1 of PG-10.0 composite. The application of PG in thermal interface materials, connectors, and thermal management systems. The brown bonds in PG composite represent the restored sp2 C after the thermal reduction at 250 C and the red curves represent the grafting PA6 chains (Figure 6).
Schematic representation of synthesis of graphene/PA6 nanocomposites by in situ polymerization with simultaneous thermal reduction from GO to RGO.
Lee et al. 44 developed PA610/PA610-grafted GO composites by using acyl-functionalized GO and in situ interfacial polymerization. The PA610 grafted on GO enhanced the dispersion of GO in the PA610 matrix and the interfacial adhesion between GO and PA610. The thermal properties of PA610/GO-g-PA610 and PA610/GO composites were improved with increasing GO-g-PA610 and GO contents, respectively, compared with pure PA610. O’Neill et al.45,67 presented an efficient method to prepare PA6 nanocomposites containing highly dispersed oxidized graphene by in situ polymerization. The technique apparently maintained the single-layer nature of GO in the nanocomposite by direct synthesis of the polymer in the presence of the GO/water dispersion. Nguyễn et al. 47 reported the preparation of PA6 and GO nanocomposites. PA6-grafted GO was prepared using an in situ polymerization method with GO dispersed in an CL monomer with 6-aminocaproic acid as the initiator and in situ polymerization with GO dispersed in an CL monomer with 6-aminocaproic acid as the initiator. The MB chips were well compounded with the virgin PA6 chips with good processability.
Fu et al. 51 have reported the effective method to prepare PA6 multilayer graphene nanocomposites via in situ polymerization. The mechanical and thermal properties of PA6 nanocomposites were investigated with low unmodified multilayer graphene content of 0.01–0.5 wt%. The dispersion of multilayer graphene sheets in the polymer matrix was studied in extensive detail. The mechanical properties of the nanocomposites were significantly enhanced; the flexural modulus, flexural strength, and impact strength increased by approximately 97%, approximately 69%, and approximately 76% relative to pristine PA6. Furthermore, multilayer graphene can increase the crystallization speed and improving the crystallization temperature of PA6/multilayer graphene nanocomposites. Wang et al. 58 have prepared MC PA/GO-CTAB composites by in situ ring-opening polymerization. The tensile strength of the composites was greatly improved even though at very low containing of GO-CTAB. The mechanical properties and the thermal decomposition temperature were increased at various degrees. Wang et al. 66 developed PA6/sulfonated graphene composites using sulfonated graphene by in situ hydrolytic ring-opening polymerization of CL. The PA6 chains were covalently grafted onto sulfonated graphene sheets by the condensation reaction between the active amino groups at PA6 chain terminals and the sulfonic acid groups on sulfonated graphene sheets (Figure 7). These composites have simple preparation process, good mechanical properties, high-thermal conductivity, and excellent processability, providing great promises for the large-scale application in thermal conductive materials. Li et al. 71 demonstrated that 3-D graphene structures constructed by graphene foam were introduced into PA6 for enhancing the thermal-conductive and anti-dripping properties of PA6 composites.
Schematic representation of PA6/sulfonated graphene composites by in situ polymerization process.
Hocker et al. 83 prepared GO-based PA11 via in situ polymerization. These nanocomposites had elevated resistance to hydrolytic degradation. The results show 40% increase over PA11’s equilibrium molecular weight when aged in water at 100°C and 120°C, concentration 1 mg°g−1 GO in PA11, and slight decrease in the amount of PA11 crystallinity prior to aging. The crystallinity of the PA11 was similarly affected by a reduction in chain mobility during annealing due to the GO nanoparticles and highly asymmetric nano-planar sheet structure. Li et al. 87 have produced MC PA6/graphene (GN)–3-aminopropyl-terminated poly-(dimethyl siloxane) (APDMS) nanocomposite via in situ polymerization by incorporation of polyethylene glycol (PEG) as a solid lubricant. Addition of PEG led to more effective grafting of PA6 molecules on GN surface. The tensile strength increased 12% and impact strength increased 20.6% with 0.5 wt% GN addition, while over 75% reduction in specific wear rate and 13% reduction in friction coefficient were obtained with 0.7 wt% GN addition. This work provides outstanding anti-wear MC PA6 nanocomposites to expand the applications of GN in the engineering plastic (Figure 8).
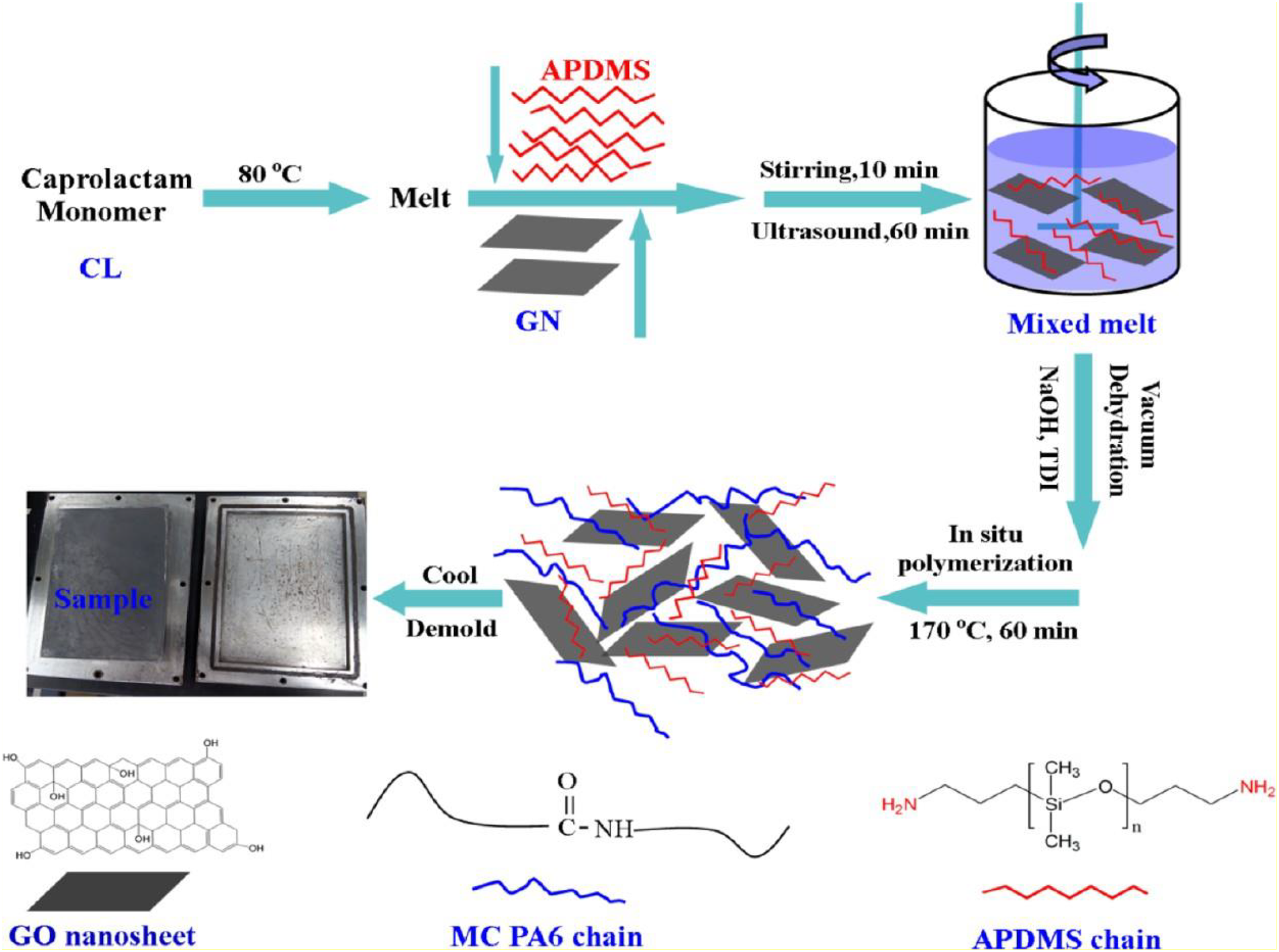
Synthesis of MC PA6/GN-APDMS nanocomposite via in situ polymerization.
Yousefian-Arani et al. 101 developed semi-aromatic PA-based GO nanocomposites by in situ polymerization. In this preparation, GO or SGO nanosheets mixed into the PA matrix, during interfacial polymerization. There were significant improvements observed in thermal stability, electrical conductivity, and dielectric constant of PA by adding 0.1% SGO in the PA matrix. Duan et al. 98 have produced PA66/RGO nanocomposite by a one-step process of in situ polymerization. The nanocomposites produced by a condensation reaction between the carboxylic acid groups on the GO surface and the terminal amino ends of PA66 chains. The Young’s modulus and tensile strength of the nanocomposites were increased to 660 MPa and 84 MPa, respectively, which offered a wider application of the PA66 materials.
Wang et al. 96 have prepared PA6 nanocomposite with low graphene content by in situ polymerization. A 3-D graphene network was created in PA6 through the reduction and self-assembly of GO, and then PA6 nanocomposites with low graphene content were fabricated through in situ polymerization. The 3-D graphene network can suggestively improve the thermal conductivity of PA6. In the case of the PA6 with only 0.25 wt% graphene, its thermal conductivity is 0.69 W m−1 K−1, about 2.88 times of that of the neat PA6. Moreover, the mechanical properties and water resistance of PA6 also have increased with the incorporation of the 3-D graphene network (Figure 9).
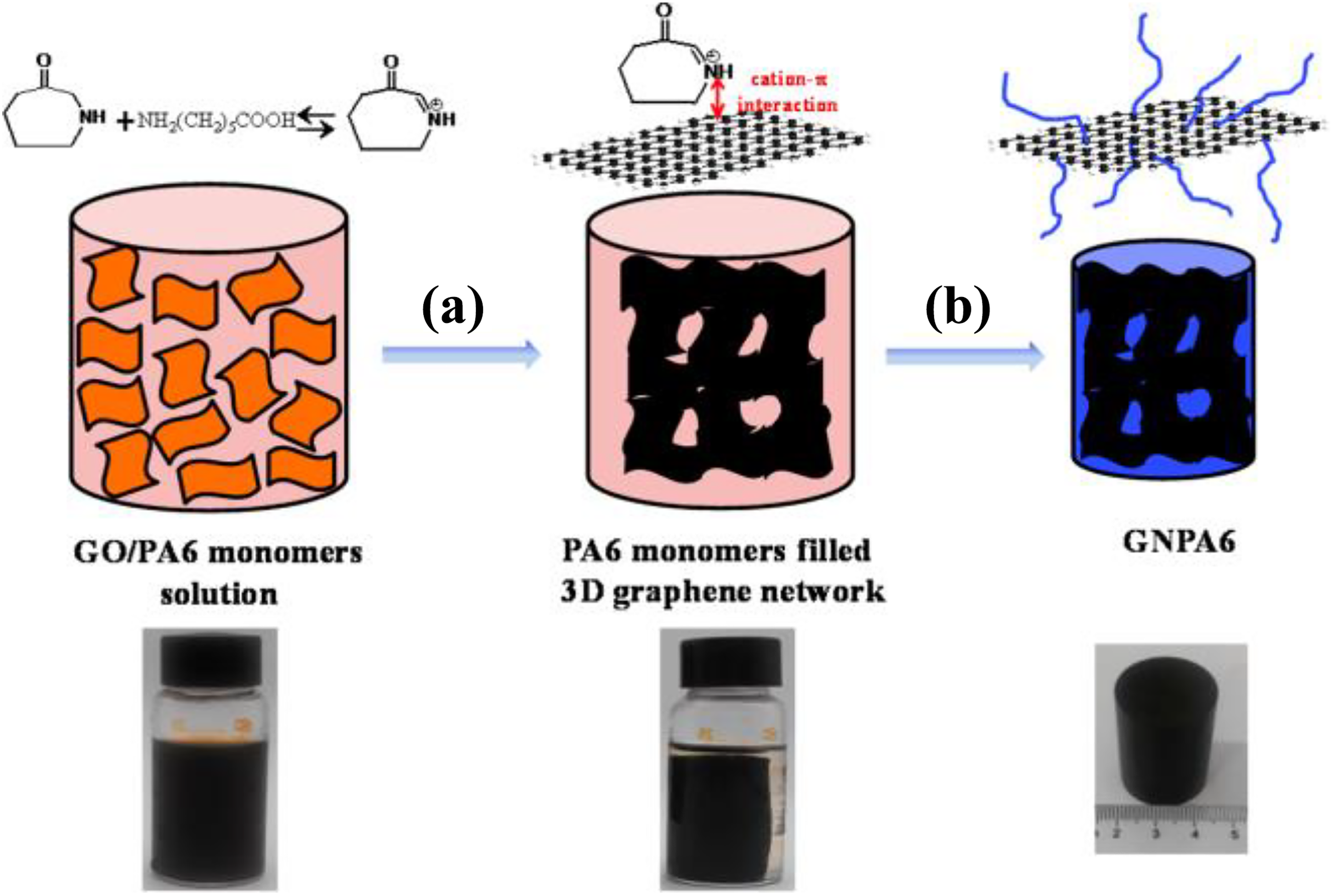
Schematic representation of GNPA6 nanocomposites: (a) fabrication of PA6 monomer filled 3-D graphene network by self-assembly of reduced GO and (b) in situ polymerization of PA6 in the resultant 3-D graphene network.
Zhuang et al. 104 prepared monomer-casting PA/graphene nanocomposite by in situ polymerization (Figure 10). The pre-dispersion method led to good dispersion of graphene in MC PA. The results of nanocomposites indicated that thermal conductivity and thermal stability increased.
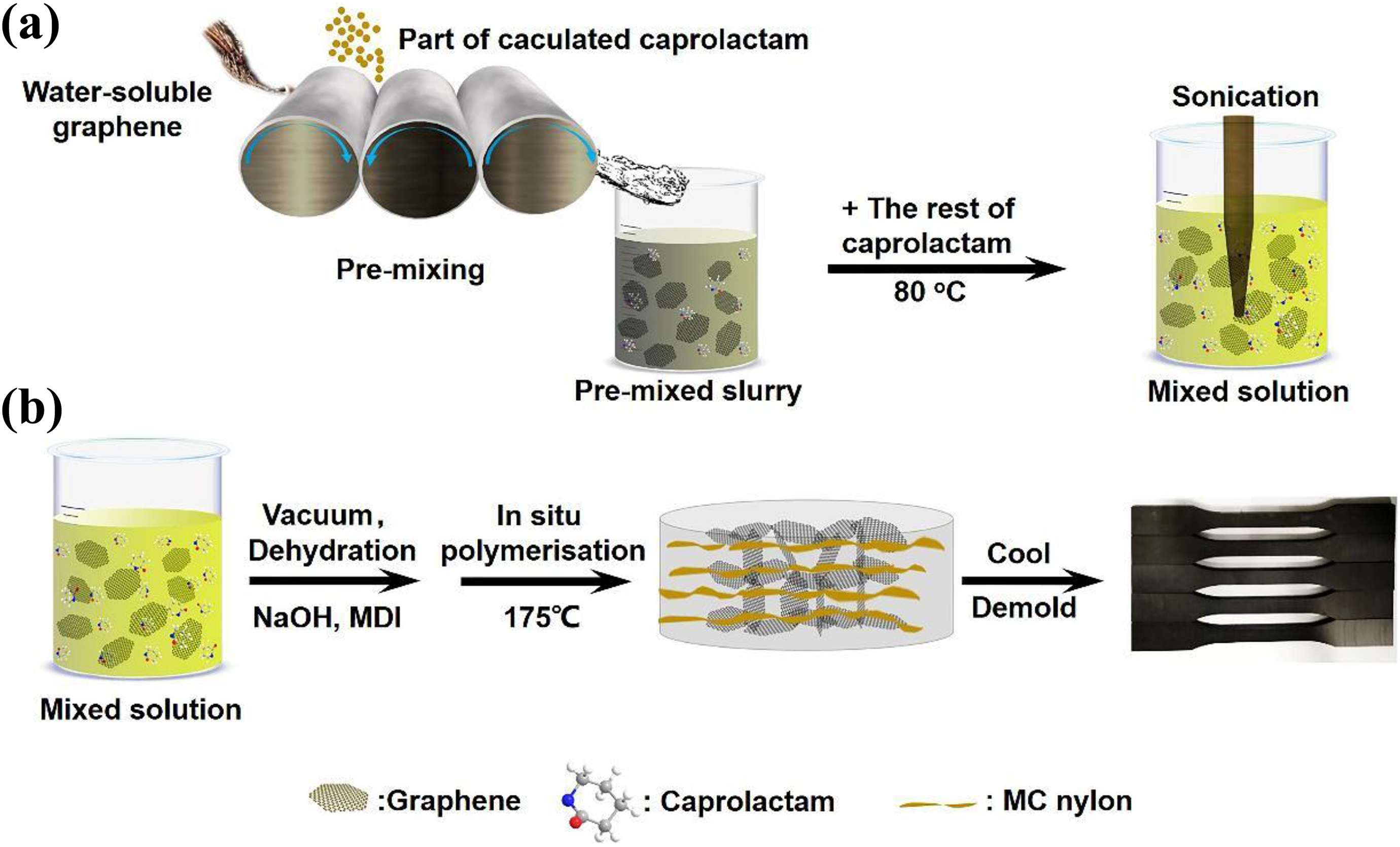
Schematic representation of MC-PA/graphene nanocomposite by in situ polymerization: (a) pre-dispersion process and (b) preparation of nanocomposites.
Wang et al. 103 produced semi-aromatic PA6T-co-6(PA6T/6)/GO composites by in situ polymerization (Figure 11). PA6T chain grafted on graphene sheets by strong hydrogen bonding produces polymer-bridged graphene network structure which enhanced mechanical properties of nanocomposites at small amount of GO (2 wt%).
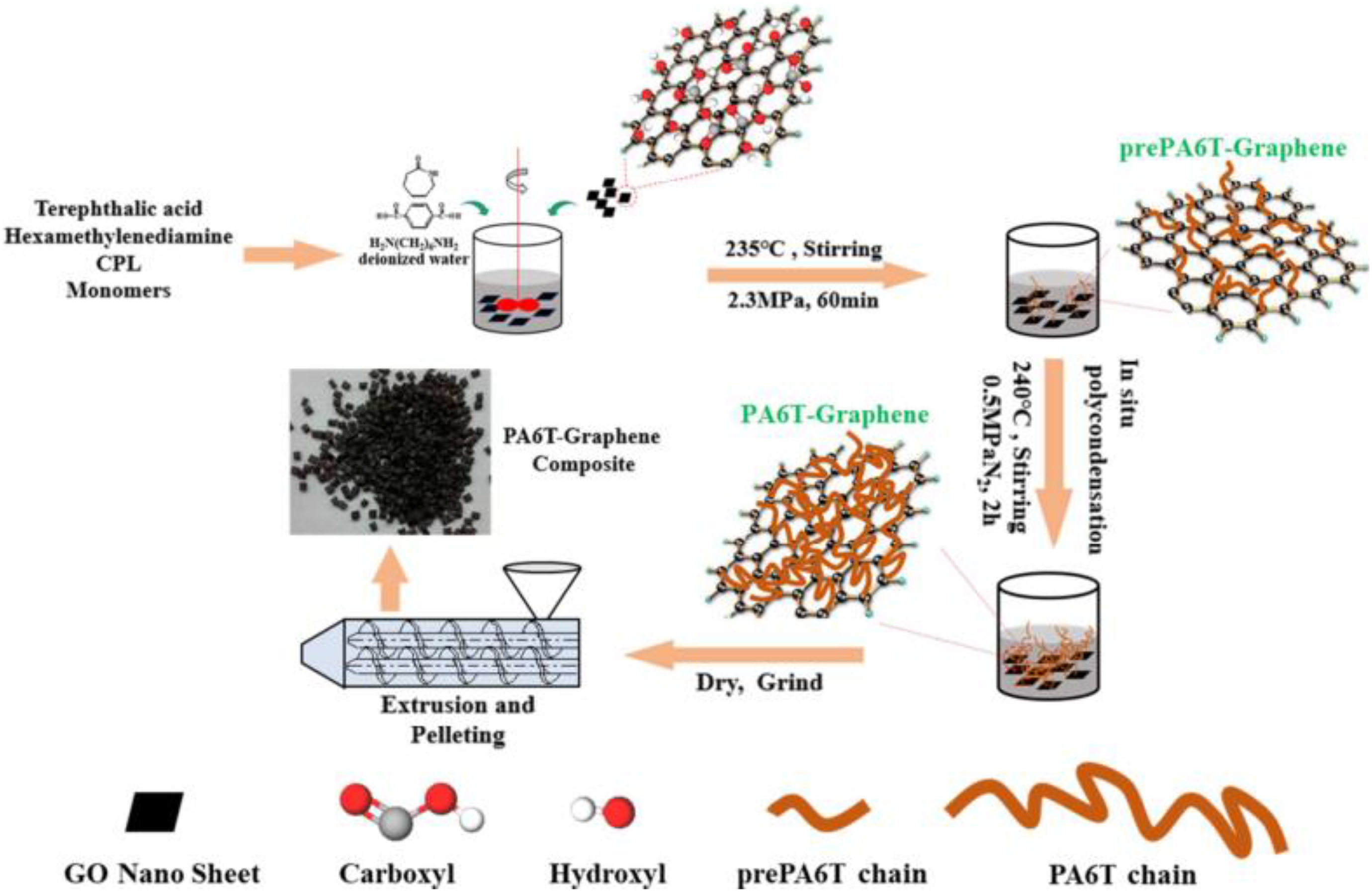
Schematic representation of PA6T-graphene nanocomposite by in situ polymerization. PA: polyamide.
Melt blending
Melt blending is one of the useful techniques to produce G-PA nanocomposites. In melt blending, graphene is mixed up with polymer by mechanically or using extruder, melt spinning to prepare G-PA nanocomposites. Rafiq et al. 12 prepared ultra-toughened functionalized graphene (FG)/PA12 composite by melt blending and investigated the effect of FG on the mechanical properties of PA12. With small amount of FG caused significant improvement in the toughness of the PA12 due to increase in γ-phase of PA12. Tensile strength and elongation at break of the PA12 is improved by approximately 35% and approximately 200%, respectively. Yan et al. 19 prepared electrically conductive PA12/graphene binary nanocomposites with maleated polyethylene-octene rubber by melt blending. Zhang et al. 21 synthesized graphene/PA6 composites by melt processing. With the use of graphene, the crystalline temperature improved due to heterogeneous nucleation and thermal properties of PA6 also increased.
Hong et al. 27 prepared graphene/PA6 nanocomposites by melt mixing. The graphene nanosheets coated with Co3O4 and NiO (denoted as GNSeCo3O4 and GNSeNiO) and then melt blend with PA6. The GNSeCo3O4 and GNSeNiO are well dispersed in PA6 and significantly decrease the gaseous products from PA6. The thermal stability of the composites was improved as evidenced by the increase in decomposition temperature and reduction in maximum decomposition rate. jin et al. 31 prepared high performance of graphene/PA11 and PA12 nanocomposites by direct melt compounding. The ultimate tensile strength, fracture toughness, elongation at break, and impact failure energy of the PA12 were significantly increased by approximately 35%, approximately 75%, approximately 200%, and approximately 85%, respectively, when only 0.6 wt% FG was incorporated, and the tensile and fracture toughness of PA11 were slightly improved compared to PA12. Barrier resistances of the PA11 were improved.
Gaikwad et al. 32 developed PA11/nanographene platelets (NGPs) nanocomposites by melt blending with using co-rotating twin-screw extruder to produce nanocomposite master batch pellets. The tensile modulus and flexure modulus with increases in the wt% of NGPs within PA11 matrix. There was no change in the flammability properties with increase in the NGP wt%. Co-rotating twin-screw extrusion and high shear action involved in mixing resulted in uniform dispersion of NGPs in PA11. Li et al. 40 synthesized a novel RGO decorated with halloysite nanotubes (HNTs-d-rGO) hybrid composite as the additive in PA6. The flame-retardant properties of PA6 were significantly improved. Xu et al. 46 developed PA6/PA66/GNP thermal composites by melt-blending method. The thermal conductivity of composite containing 50 wt% of GNP was measured as 5.03 W m–1 K–1 at 30°C, indicating an increase of more than 15 times compared with that of the neat PA6. Cai et al. 48 prepared PA1212/GO composites by two-step melt-blending process. GO solution was mixed with an ethanol-soluble PA solution then melt-compounded with a PA1212. With the addition of GO, crystallinity in the composites increased slightly.
Roy et al. 50 prepared PA12/polyethylenimine (PEI)-RGO composites by melt mixing process. The thermal stability of PA12 significantly improved with the addition of PEI-RGO and glass transition temperature increased. Mayoral et al. 56 prepared PA6/graphene nanoplatelet composites via twin-screw extrusion. A small increase in crystallinity and a maximum enhancement of 420% in Young’s modulus were achieved at 20 wt% loading of GNPs. Liu et al. 62 reported graphene non-covalently functionalized with amino-terminated dendronized PA (DPA-GNs) filled into the PA6 to prepare nanocomposites. The functionalization of graphene enhanced dispersion and interface interaction between graphene and PA6. The thermal conductivity of DPA-GNs/PA6 composite approximately increased by 200% with low graphene content, compared with neat PA6. The decomposition temperatures of DPA-GNs/PA6 were approximately increased by 20°C. Wu et al. 68 prepared graphene/PA6 nanocomposites by masterbatch melt mixing with a kneader mixer. The rheological behavior and viscous activation energy of these nanocomposites were investigated. Graphene/PA6 masterbatch (10 wt%) produced by twin-screw extruder. The viscosities of the graphene/PA6 composites improved with increasing graphene concentration. As the concentration of graphene increased, the flow behavior changed from Newtonian to pseudo-plastic flow. They found most suitable processing parameters for maximizing the exfoliation stress level, the range from 25 kPa to 30 kPa at a 100 s−1 shear rate and 240°C.
Rashmi et al. 69 developed production technique for multifunctional bio-based PA11/graphene nanocomposites by melt extrusion processes via masterbatch dilution. Addition of graphene improves the electrical conductivity, crystallization temperature, degree of crystallinity, tensile strength, and dielectric constant of PA11. The tensile strength increases by approximately 14–25% and modulus by approximately 35–56% for 0.5–5 wt% GNP, respectively. With extensive usage of carbon-based nanofillers will cause emissions to the environment and increase in human exposure to nanofillers. Therefore, manufacturing of carbon-based nanofillers in plastic parts often a key issue. This production process of a GNP masterbatch in PA is dust-free processing method with lower risks compared to the bulk carbon nanofillers as well as an easier method for handling the material. Cho et al. 72 prepared the PA/reduced graphene nanocomposites by melting blending. The chemical compatibility between RGO and PA enhanced by using titanate coupling agent (TCA). Covalent bonds can be formed by the chemical reaction between hydroxyl groups on the GO and alkoxy group of titanate molecules. The TCA molecule can replace of water of hydration at the RGO surface, which can improved thermal conductivity. With using RGO loading level (5 wt%), PA/TCA RGO composite has 53% higher thermal conductivity. Moreover, PA/TCA-RGO composite material can be used in thermal management for electronic as well as photonic applications. The LED lamp combined with PA/TCA-RGO composite as heat sink also displays better strength due to its excellent thermal dissipation.
Scaffaro and Maio
76
have prepared GO-silica nanohybrid (GOS)/PA6 nanocomposites by melt blending process. GOS was prepared in water and used as a filler for PA6-based nanocomposites. This method can be considered green because the reactions in aqueous environment which reduces the time and avoid toxic solvents. GOS prepared by 150 mL of water under mild operative conditions (
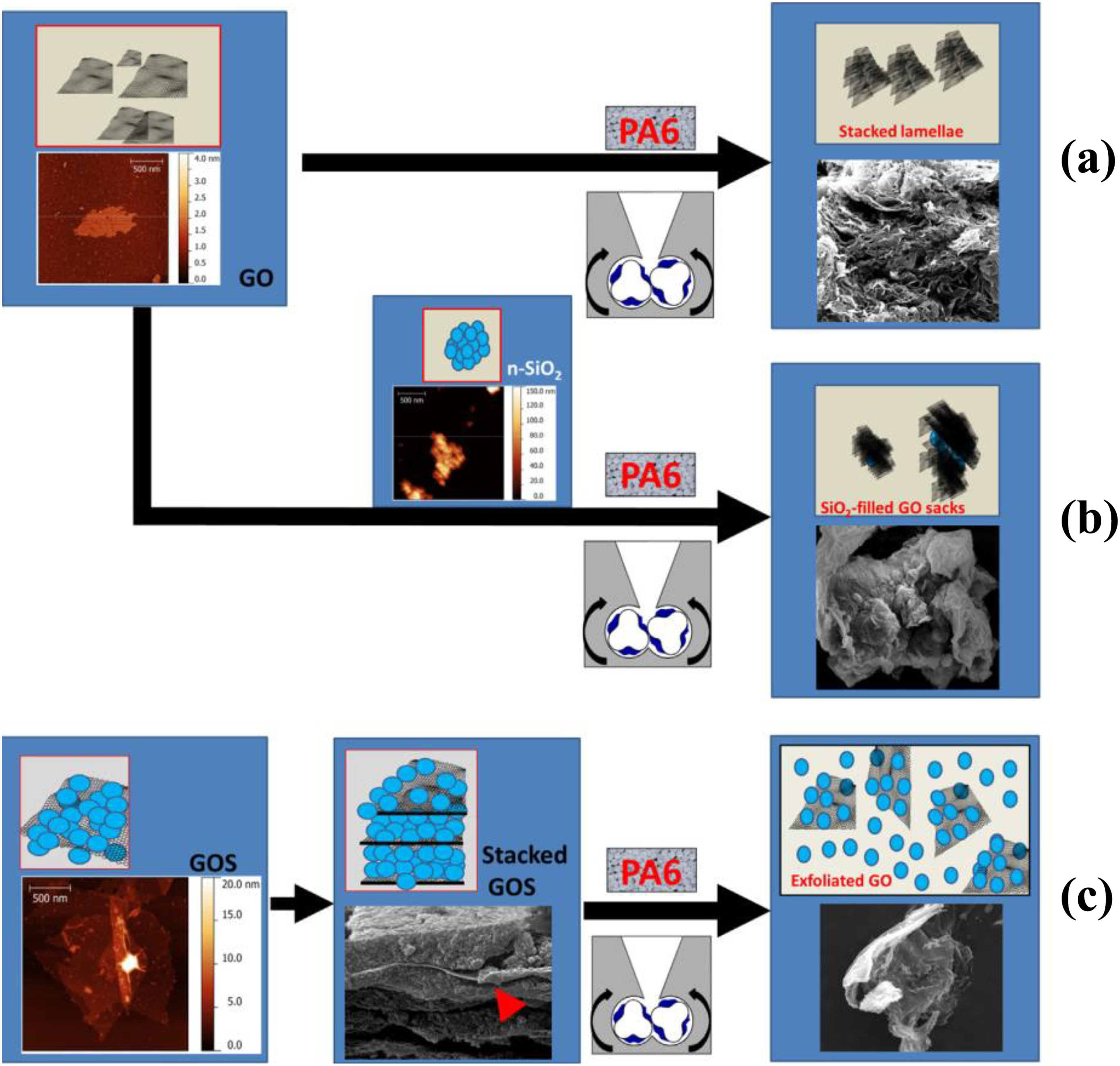
Schematic representation of nanocomposites (a) (direct melt mixing of PA6 and GO), nanocomposites (b) (melt mixing of PA6, GO and nanosilica), and nanocomposites (c) (synthesis of lasagna-like self-assembled GOS nanohybrids and melt mixing with PA6).
Zhu et al. 82 developed 3-D printable PA12/GNPs nanocomposites by melt blending. The thermal conductivity (k) and elastic modulus (E) of 3-D printed PA12/GNPs parts increased by 51.4% and 7%, respectively. The ultimate tensile strength, thermal conductivity, elastic modulus, and higher thermal stability of PA12/GNPs are improved. The crystallinity decreased by using GNPs content of 6 wt%. Furthermore, raster angle of 8° printed PA12/GNPs parts showing the reinforcement efficiency. Song et al. 88 prepared PA6/graphene (PG) composites by melting, stirring through an internal mixer. They demonstrated the bridge effect of silicon carbide whiskers (SiCw) on the enhancement of thermal conductivity in the experimental results. The thermal conductivity of PG composites increased with SiCw, particularly in the through-plane direction. The mechanical properties of the composites were synchronously enhanced. The thermal conductivity enhancement of the through-plane direction can reach as high as 199%, the maximum can reach 508% related to the PA matrix.
Scaffaro and Maio 92 developed two-step synthesis technique for PA6 graphene nanocomposites by solvent-assisted melt blending (Figure 13). The nanocomposites prepared by three two-step methods and two different loading content of GO. Among the three techniques, method III was quick and involved less consumption of water.
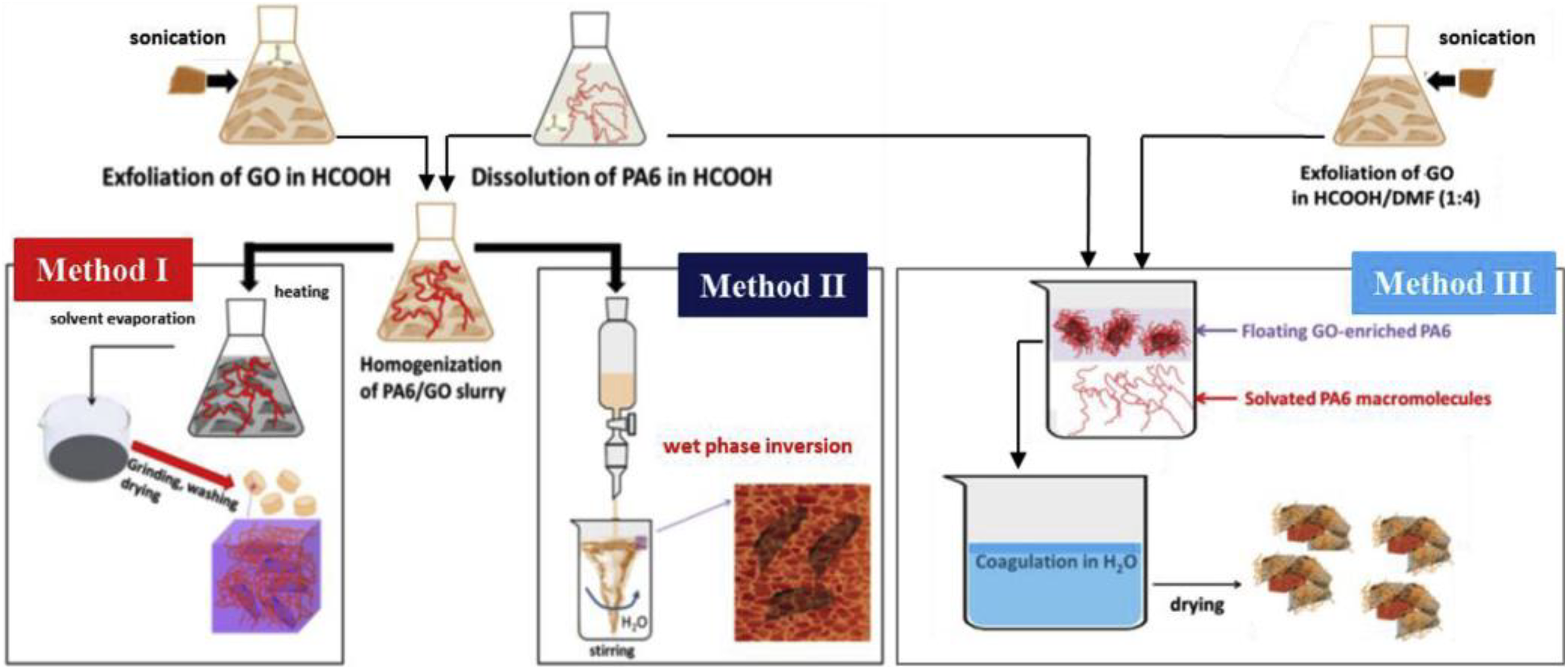
Schematic representation of preparation of PA6 graphene nanocomposites.
Chen et al. 95 prepared N-(2-aminoethyl)-3-aminopropyltrimethoxysilane (AAPS)-modified GO PA6 nanocomposite by melt blending (Figure 14). The nanocomposites revealed that the AMG owned improved dispersity, high thermal stability, tensile strength, and tribological properties such as friction behavior and wear rate.
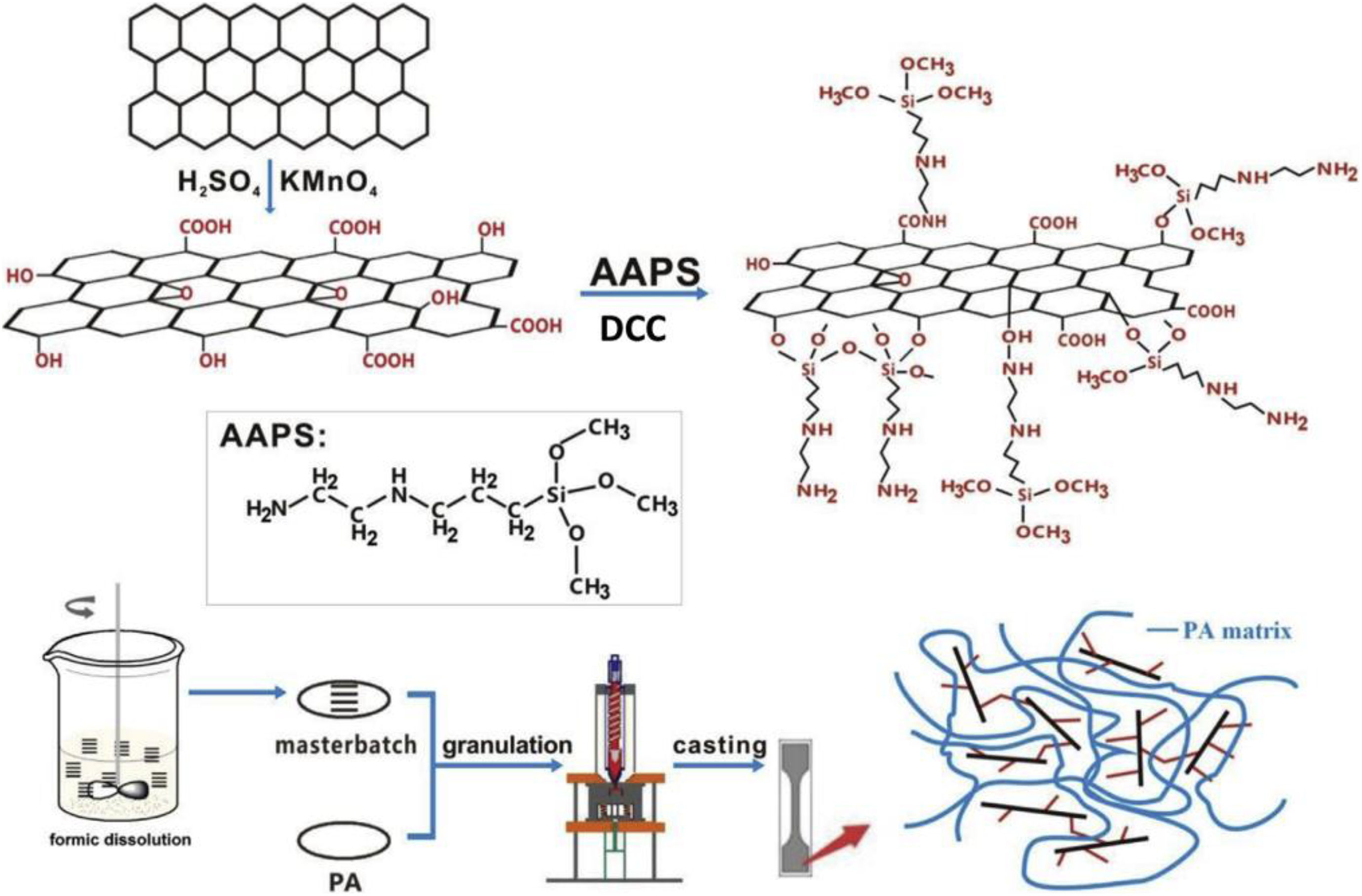
Schematic representation of the synthesis of AMG(AAPS)/PA nanocomposites.
Li et al.
100
prepared PA6/GO-
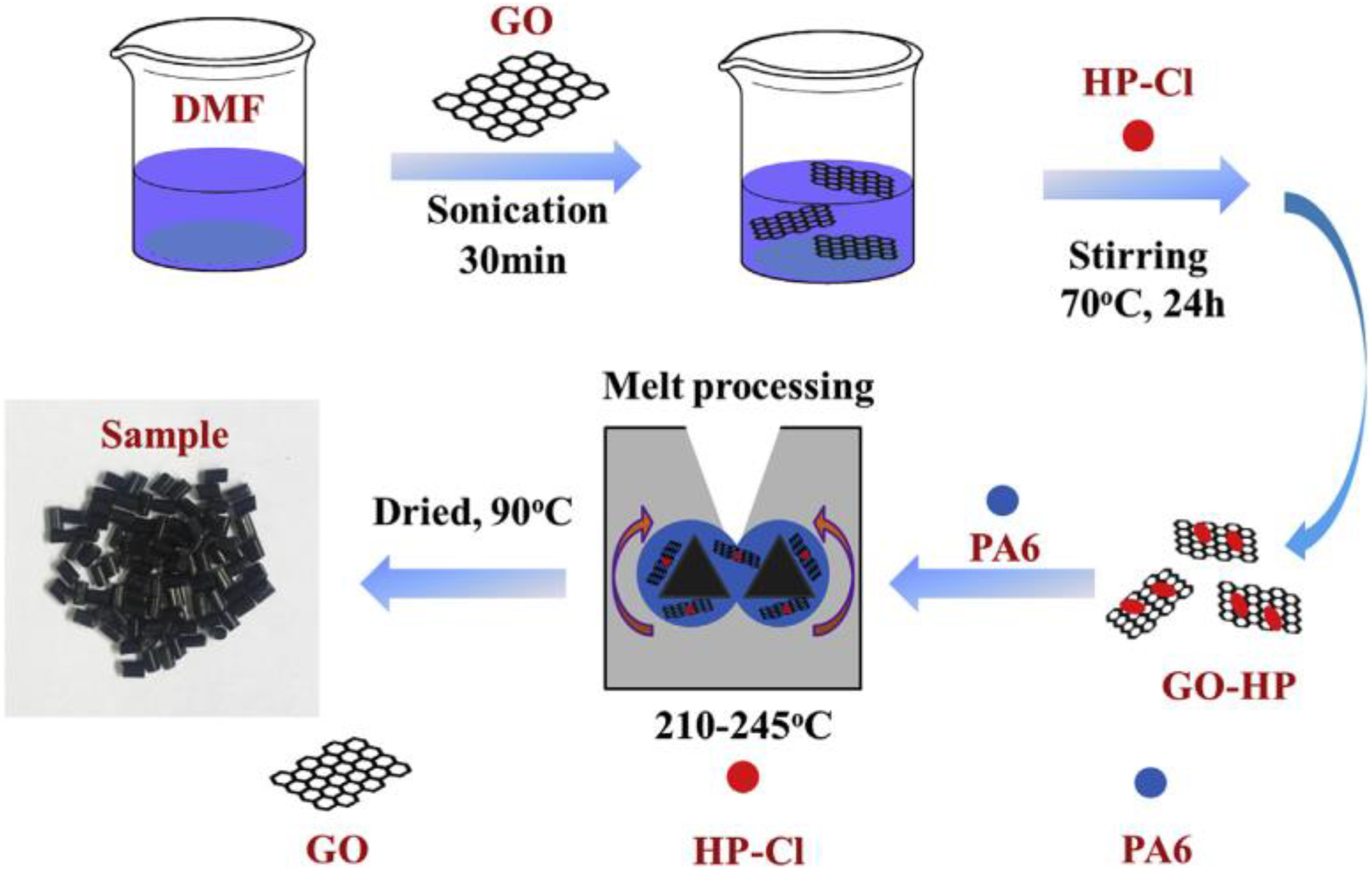
Schematic representation of preparation of PA6/GO-HP nanocomposites.
Solution mixing
Perhaps the simplest applied processing technique for graphene-modified polymer nanocomposites is the solution mixing or solvent blending technique, which involves mixing of polymer and graphene in a suitable solvent. The nanocomposite is produced by precipitating or casting a film. During the solvent blending process, graphene dispersed in a suitable solvent with the polymer. The solvent removed by evaporation or distillation process. Chiu and Huang 20 prepared PA46/GO nanocomposites by solution mixing technique. The morphological observations showed the compatibility between PA46 and GO. With the small amounts of GO facilitate the nucleation and overall crystallization of PA 46. The stiffness of PA46 increased with GO. Pan et al. 23 presented preparation of PA11/graphene coatings. The wear life of composite coatings increased compared to the neat PA11 coating. Jacobs et al. 24 prepared PA11/NGP nanocomposites by solvent blending. The addition of NGPs showed an initial increase in thermal stability and crystallization temperature. Fathali et al. 38 developed new method of PA-decorated graphene sheet as a potential antimicrobial agent. The thermal stability and antibacterial properties of PA film increase with graphene. Hou et al. 39 synthesized amine FG/PA6 nanocomposites by solution mixing (Figure 16). Prepared nanocomposites GO, GO–NH2, and GO–(CH2)6 NH2 showed that the nature of the functional groups affects the grafting chemistry of PA6 to graphene.
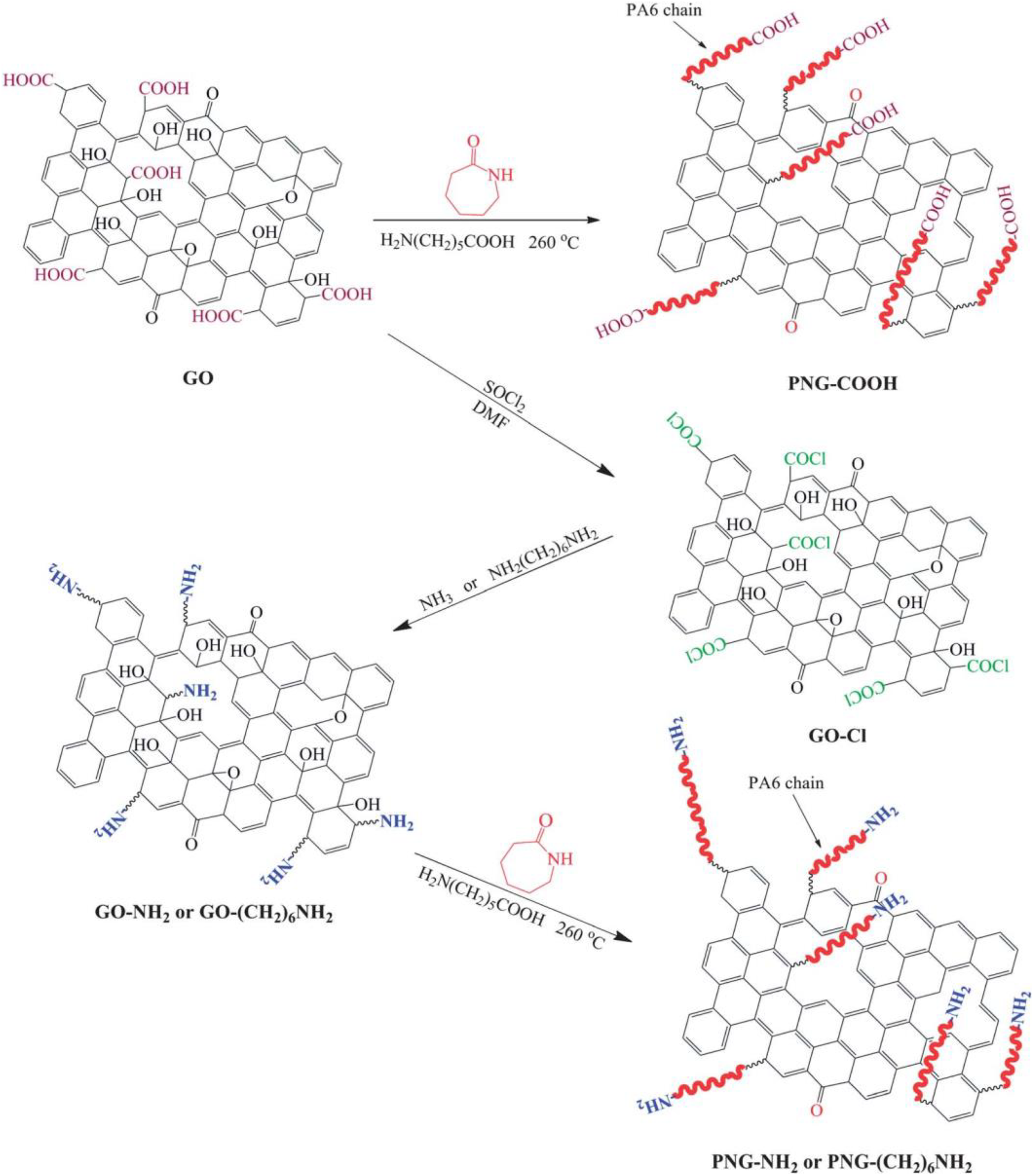
Synthesis of amine FG/PA6 nanocomposites.
Gong et al. 49 prepared PA6/graphene composites modified through polymeric modification of graphene by solvent blending. Song et al. 55 have prepared PA6/graphene nanocomposites by solution blending and find effect of polymer modifier chain length on thermal conductive property of PA6/graphene nanocomposites (Figure 17).
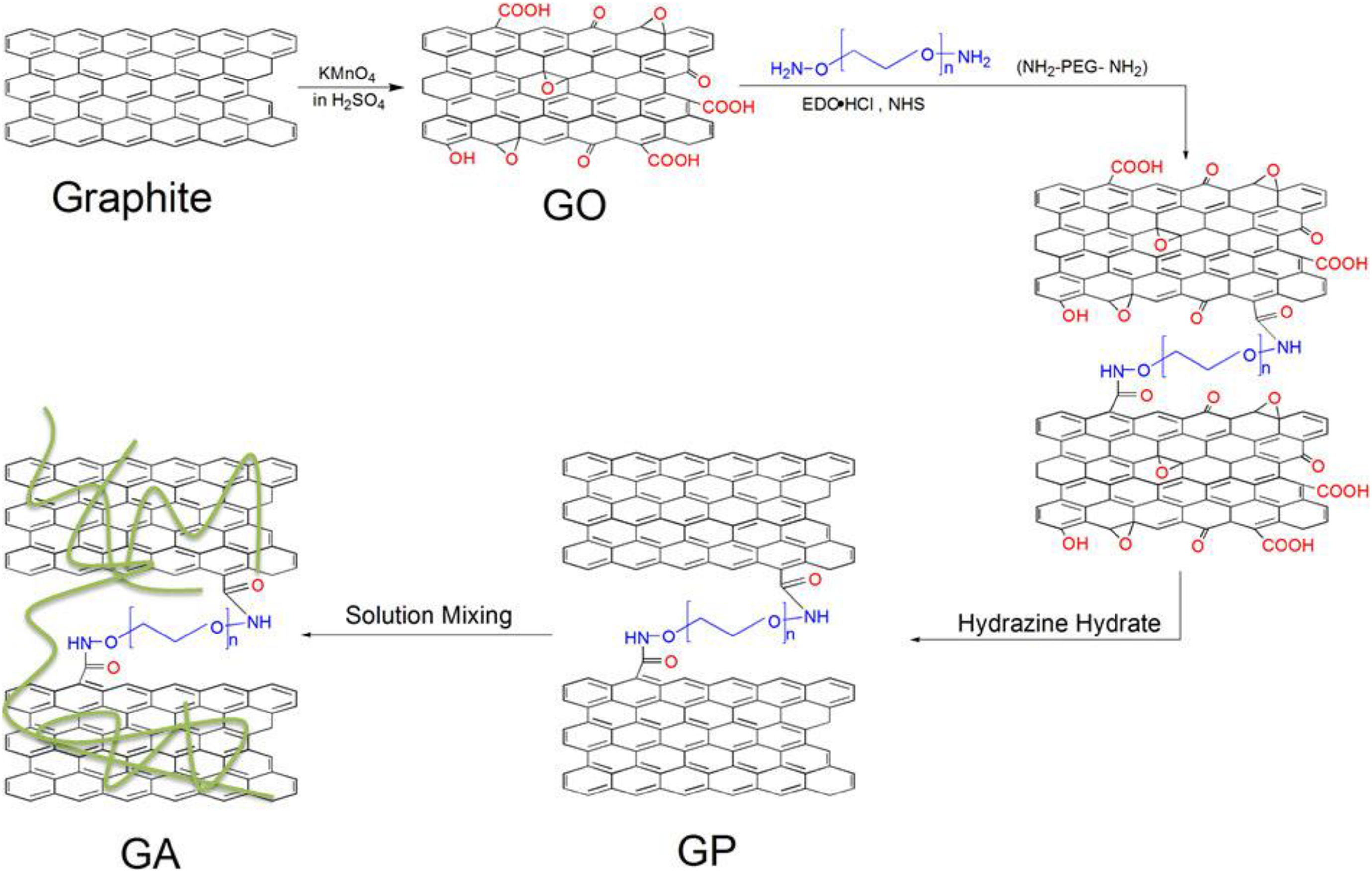
Synthesis of GA composites (GA represents PA 6).
Liu et al. 64 developed graphene/CO-PA nanocomposites by solution mixing method. They have prepared the simple method to make CO-PA nanocomposites with perfect properties. de Melo et al. 97 prepared GO-reinforced PA6 nanocomposites by solution mixing. Evie et al. 65 have prepared PA66/graphene nanoplatelet composite films obtained from a new solvent by solution blending technique. Ma et al. 73 reported preparation of highly conductive composites with segregated structure based on PA6 and reduced GO by solution mixing. Allahbakhsh et al. 78 prepared 3-aminopropyl-triethoxysilane-functionalized rice husk and rice husk ash-reinforced PA6/GO sustainable nanocomposites. Kausar 91 prepared PA1010/PTA blend reinforced with GNP by solution mixing method. The nanocomposite revealed optimum tensile strength (40 MPa), impact strength (1.9 MPa), and flexural modulus (1373 MPa) to manufacture automotive part (Figure 18).
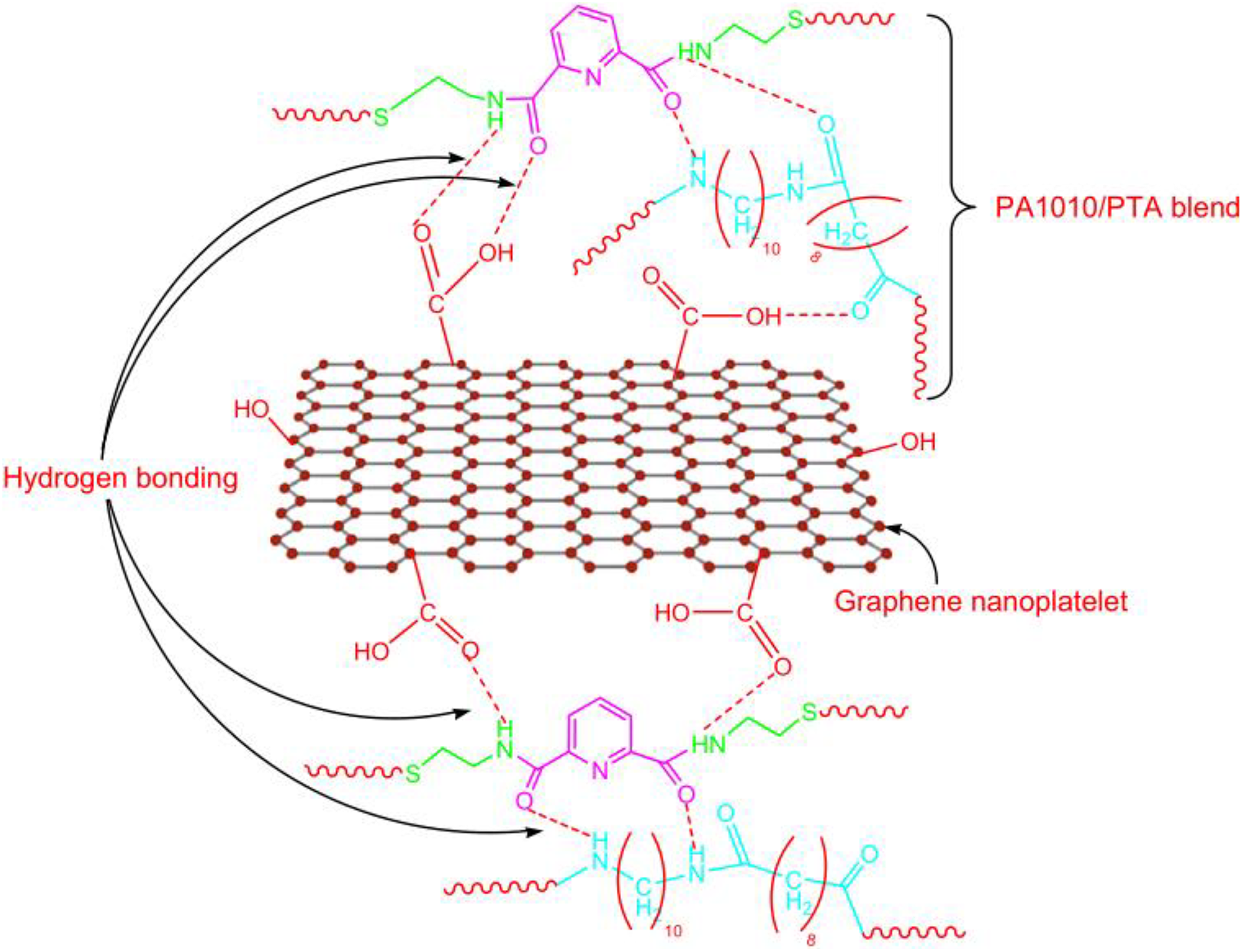
PA1010/PTA/GNP nanocomposite.
Conclusion
The preparation of G-PA nanocomposites has been reviewed. There are three main methods to prepare G-PA nanocomposites such as in situ polymerization, solvent blending, and melt blending. In situ polymerization and solution blending produce composites with a high degree of consistency for research purposes, but these processes were very difficult to scale up, while the environmental concerns also need to be considered. On the other hand, making use of the conventional melt mixing process, nanocomposites can be produced that may still be performing enough for some applications. In situ polymerization and solvent blending can achieve a fine dispersion of graphene in polymer matrices nevertheless use of hazardous chemicals and removal of solvent lead to aggregation, which shows potential risks as these two methods are environment unfriendly. Melt blending is environmentally friendly and suitable for mass production, but the dispersion of graphene in the polymer matrix is not good. It has been shown that in the graphene structure, the number of layers, the specific surface, and the chemical functionalization of these fillers, all play a major role in the final performance of the G-PA nanocomposites.
Moreover, the variety of preparation methods that have been presented in this review offers different pathways, depending upon the perceived application of the material. The scaling-up of the production of high-quality graphene is a determinant factor for the production on commercial scale of these nanocomposites with useful properties. So far, the combination of graphene and PA has been playing a ubiquitous role in the field of research encompassing significant enhancement in the mechanical, thermal, electrical, and barrier properties. As future unfolds, the advancement in the achievement of the interaction between graphene and PA without modification process and cost of graphene. In this review, potential synthesis of G-PA nanocomposites has been highlighted. It concludes significant properties, synthesis, and preparation method. In the field of material science, G-PA nanocomposites are one of the most fortunate developments. Hence, we can say undoubtedly that G-PA nanocomposites are the materials for future innovation.
Future scope of work
A respectable amount of work has been done and important conclusions have been drawn. Continued overwhelming response from academicians and researches, graphene provides an exciting platform to study engineering, physics, chemistry, and materials science of unique 2-D systems as well as offers a route toward user friendly end products conducting polymer graphene composite. A reviewed research may lead to a new dimension for high-performance nanocomposite materials for a wide range of applications such as energy devices, electronics, automobiles, sports industries, aerospace, defense, solar cell, fuel cell, supercapacitor, gas detection, lithium ion batteries and functional conducting electrodes for technical relevance, and so on. Despite these interesting developments, a lot remains to be done about both essential understanding and the improvement in intrinsic methods, synthesis as well as properties. Appropriate surface modification of graphene to develop strong interaction between matrix and nanofillers is yet the blistering area of research. There are countless challenges that need to be addressed like to prepare single-layer graphene, development of fine interaction between graphene and matrix without any modification process, design of appropriate method for processing, and property improvement at very low loading levels are among the foremost argues. Although, some important parameters such as low-cost material with excellent quality and high production demand on large scale is yet to come. There is still room for broad fundamental research for basic understanding of the subject and their future commercial utilization. Besides the laboratory scale research to introduce graphene in PA for product applications, it is essential to scrutinize that how nanomaterials behave under industrial processing conditions. However, research is never ending task thus more future work on G-PA nanocomposite can be ultimately advanced by upgraded methods and technologies.
Footnotes
Acknowledgements
The authors are thankful to the Department of Chemistry, Gujarat University, Ahmedabad, for providing the essential facilities. UGC-Info net and Information and Library Network Centre, Gujarat University are acknowledged for providing the resource facilities.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
