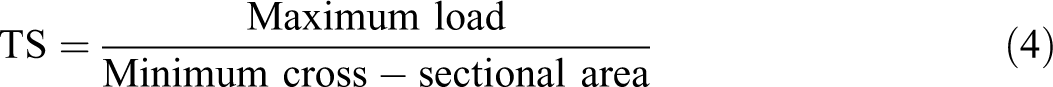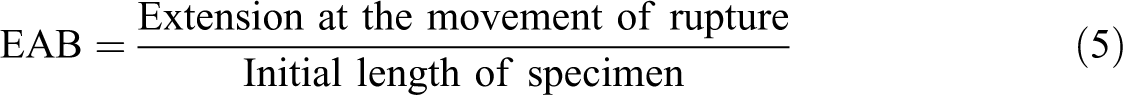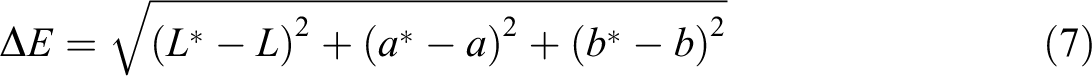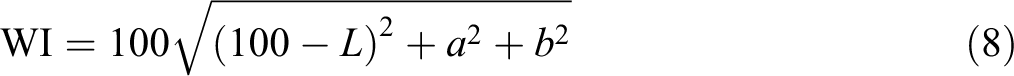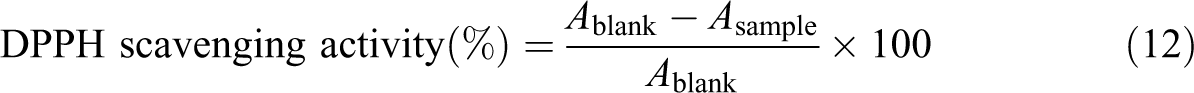Abstract
The current research presents the physicochemical, antioxidant, mechanical, and antimicrobial features of biocomposite films on the basis of carboxymethyl cellulose (CMC) combined with essential oil (EO) of Mentha pulegium (MEO) (1, 2, and 3% v/v). Adding EOs enhanced the penetrability of water vapor and the solubility of films in water, while decreased absorption of moisture and level of moisture. Incorporation of MEO enhanced opacity, total color differences, and whiteness index. Generally, adding MEO into the CMC biocomposite reduced considerably young modulus and tensile strength, while enhanced elongation at the composite films break (p < 0.05). The films incorporating MEO had excellent total phenolic and antioxidant features; this impact was significantly developed when the MEO was enhanced. Films comprising MEO controlled the microorganism’s growth efficiently. Fourier-transform infrared spectroscopy was used to determine the physicochemical features of the films and the features of the films were corresponded to their microstructure, which was found using scanning electron microscopy.
Introduction
Among the materials used in packaging, polymers, especially plastics, are in great request. They show a large number of worthwhile features, including softness, transparency, heat seal capability, and excellent strength to weight ratio. Furthermore, they are usually cheap ingredients, display effective mechanical features including tear and tensile strength (TS), and they are excellent barrier to oxygen and heat. 1 However, a large number of the plastic materials used for packaging foods are almost nondegradable and have serious environmental pollution-related problems. Bio-based polymeric hydrogels used for packaging food have been extensively interested by scientists as an alternative method to manage the problem corresponding to discarding waste plastic materials. 1
Bio-based packaging materials have been derived primarily from annually renewable sources and many (but not all) are biodegradable. Regarding to ASTM D7075-04 standard, bio-based materials are defined as materials containing carbon-based compound(s) in which the carbon comes from contemporary (nonfossil) biological sources. Bio-based polymers generally have a smaller carbon dioxide footprint and are associated with efforts to create more sustainable, environmentally friendly products. 2
Among those ingredients, polysaccharides are taken into account as attractive source because of their excellent film-forming feature and excellent mechanical and gas barrier features comparing with other biodegradable ingredients. Carboxymethyl cellulose (CMC) is water-soluble cellulose ether derivative achieved through chemical modification from natural cellulose. 3 CMC is a main industrial polymer, which is widely used in flocculation, textiles, detergents, papers, foods, and medicine. 4 CMC is applied primarily as it is nonallergenic and nontoxic and has a large viscosity. The several carboxylic and hydroxyl groups in CMC allow water binding and moisture sorption features, and so CMC hydrogels show a great water content, excellent biodegradability, and a variety of applications because it is cheap. 5
Microbial pollution has been still a problem even in advanced countries, as it decreases shelf life and safety of food products. Antimicrobial substances can control the growth of microbes via incorporation into packaging materials that, in turn, can decrease the growth speed and highest microbe’s population and extend the objective microorganism lag phase. 6 Essential oils (EOs) of aromatic and medicinal herbs are biologically active, mostly possessing antioxidant and antibacterial features. 7 EOs categorized as Generally Recognized as Safe (GRAS) 8 , which causes they have been widely used in food industry as additives, particularly as they have been applied as medicines from ancient times. Moreover, EOs are hydrophobic in nature and incorporating EOs could progress the water vapor barrier feature. 9 One of the main applications of nanocomposites is active packaging. Active packaging has been successful in shelf life extension of food and, subsequently, reduction of food waste due to microorganism’s activities. 10 So, the use of EOs in structure of package can give it an active packaging feature. Mentha pulegium (MEO), or Pune, are medicinal and aromatic herbs of the Labiate family that is rapidly recognized because of its unique minty smell. These herbs are rich in terms of polyphenolic compounds and most of them are well known because of their antibacterial, antioxidant, and antifungal features. 11
As biodegradable films are brittle and stiff, plasticizers are required to reaching the favorable structural features. Glycerol is a plasticizer, which is widely used for preparing the films. Glycerol has frequently been applied as a plasticizer because of its compatibility with amylose and as such it progresses the mechanical features of films by reducing intermolecular forces and interfering with amylose packing. On the other hand, plasticizers normally raise a film’s hydrophilicity, which in turn enhances water vapor penetrability, while this may be developed through incorporating lipidic additives like several EOs. 12
Consequently, this research was aimed to improve an antimicrobial film on the basis of CMC and MEO to investigate the impact of incorporating MEO on the antimicrobial features of composite polysaccharide films and to assess several chemical, mechanical, physical, barrier, antimicrobial, and antioxidant features of these films to study their potential for applying in packaging foods.
Materials and methods
Materials
Sodium CMC (NaCMC), molecular weight 250 kDa with degree of substitution 0.7 and Tween 80, was purchased from Sigma-Aldrich (St. Louis, MO, USA). Glycerol was purchased from Merck Chemical Co. (Darmstadt, Germany). MEO was supplied by Exir Golesorkh Pharmaceutical Co. (Mashhad, Iran). Mueller–Hinton agar, Mueller–Hinton Broth and Lactose Broth, and Folin–Ciocalteu reagent were bought from Merck Chemical Co. Standard gallic acid and 2,2-diphenyl-1-picrylhydrazyl (DPPH) were purchased from Sigma Chemical Co (Steinheim, Germany). All other chemical materials were purchased from Merck Chemicals Co.
Preparation of films
Films were prepared as described by the method of Rhim with some modifications. 13 On one hand, 1.5 g of CMC was dispersed in 90 mL of distilled water which was subjected to magnetic stirring at 800 r min−1 and 75°C on a hot plate to obtain a 1.5% (w/v) polysaccharide suspension, then 0.9 mL of glycerol (60% v/w of the CMC weight) was added as a plasticizer. Control films (lacking EOs) were cast from this solution. In quantities proportional to the EO (0.35, 0.7, 1.05% v/v), Tween 80 was added as an emulsifier, and stirring was continued for a further 2 h at 50°C. On the other hand, different concentrations of MEO (1, 2, and 3% v/v) were dispersed in 10 mL of ethanol with magnetic stirring for 1.5 h at 300 r min−1 in room temperature. In next step, CMC and EO dispersions were mixed at 800 r min−1 in 40°C for 24 h. Next day, the film solution was homogenized by homogenizer Ultra-Turrax T-25 (IKA T25-Digital Ultra Turrax, Staufen, Germany) for 10 min at 20,000 r min−1 and then the homogenized mixture underwent ultrasonication for 7 min, which removed the air bubbles formed as a result of agitation. 14 Finally, film solutions were cast by pouring the mixture onto polystyrene Petri dishes (12 cm in diameter) placed in an oven and dried at 35°C for 30 h. Dried films were peeled off the casting surface and stored in aluminum sheets at room temperature.
GC-MS analysis of MEO
Analysis of gas chromatography (GC)–mass spectrometry (MS) was performed on Agilent model of 5977A/7890B with electron impact ionization (70 eV) (Agilent, Santa Clara, CA, USA). Helium was applied as the carrier gas at a fixed flow of 1 mL min−1. The injection port was applied in the split mode (1:30) and its temperature was kept at 270°C. The temperature of transfer line was kept at 280°C. An Ultra Alloy capillary column (30 m × 0.25 mm i.d. and 0.25 µm film thickness; 5% phenyl methyl polysiloxane) was used for the separations. The temperature program of oven was set from 60°C to 200°C at the speed of 5°C min−1; the ending temperature was kept for 1 min.
In order to do additional identification, their mass spectra fragmentation patterns were matched with those kept in the Wiley/NBS mass spectral library and other available mass spectra. 15 Furthermore, the relative percentage corresponding to the component MEO was computed using normalization of the base GC–mass peak.
Determination of physical properties of films
Thickness
The thickness of the film was measured using a handheld micrometer (Mituto, Tokyo, Japan) with a sensitivity of 0.001 mm. The results were reported as a mean, at least 10 accidental points for every film. The averages were computed and applied in determining penetrability and mechanical features of the films.
Moisture content
In order to determine moisture content (MC) of films, the weight of films was measured prior and following drying at 110°C in a Memmert UF 55 oven made in Germany until reaching to a constant weight (dry specimen weight).
Water solubility
The water solubility (WS) was measured based on the technique of Ojagh et al. 16 Briefly, the primary solid content was determined with drying specimens at 110°C until achieving constant weight (W 0), then film specimens were immersed in 50 mL of deionized water under fixed agitation speed of 200 r min−1 for 6 h at 25°C. Then, the filtration of remained pieces of film was performed and dried at 110°C in a hot air oven until a final constant weight was achieved (W 1). Percent of WS was computed from the first and last dry weights, relative to the first dry weight, with equation (1)
where W 0 represents the first weight corresponding to the film expressed as dry matter and W 1 refers to the unsolved film dry weight.
Water vapor permeability
Water vapor permeability (WVP) tests were performed at 25°C and 75% relative humidity (RH) with the standard ASTM E96/E96M, 17 considering the modification offered by Ghanbarzadeh et al. 18 Films were cut, set, and closed over a circular opening of 0.000576 m2 in a permeation cell, which was kept in a desiccator at 25°C. The cell was fully filled with calcium anhydride (CaCl2—0% RH), and the system was located in a desiccator comprising a saturated sodium chloride solution (NaCl—5% RH).
The RH outside the cell was always more comparing with inside, and water vapor transport was measured from the weight gain of the permeation cell at a stable state of transfer. Eight weights were determined at 12 h intervals during a week to the nearest 0.0001 g. Variations in the cell weight were recorded and drawn versus time. The slope of all lines was computed using linear regression (Microsoft® Office Excel 2016) with r 2 > 0.99.
WVP (g m−1 h−1 Pa−1) was computed as equation (2): water vapor transmission rate (WVTR) was obtained by dividing the slope (g h−1) by the transfer area (m2).
where P refers to the saturation vapor pressure corresponding to water (Pa) at the experiment temperature (25°C), R 1 represents the RH in the desiccators, R 2 refers the RH in the cup, and x represents the thickness of the film (m). The driving force [P(R 1−R 2)] is 1753.55 Pa under these conditions.
Moisture absorption
Moisture absorption (MA) was determined based on the modified method of Ghanbarzadeh et al. 18 Firstly, conditioning of the dried sheets 20 × 20 mm was performed for 24 h at 0% RH (calcium sulfate) and they were weighed for giving (W 0). Following weighing, they were conditioned in a desiccator comprising calcium nitrite saturated solution at 25°C for ensuring an RH of 55%. The specimens were weighed at desired intervals until reaching the equilibrium state (Wt ). The MA of the specimen films was computed as following equation (3)
where W 0 and Wt represent the first weight of each specimen and the weight of the specimen following t time at 55% RH, respectively.
Mechanical properties
The TS, Young’s modulus (YM), and elongation at break (EAB) of the films were measured after the processes outlined in the ASTM standard technique. 19
All of the examined film strips (100 × 10 mm) equilibrated at 25°C and 53% RH in desiccators comprising Mg(NO3)2 saturated solutions for 48 h before starting experiments, which were located between the grips of the texture analyzer TA.XT2i (Stable Micro Systems, Surrey, England). The first separation of grip was examined at 50 mm and the crosshead rate of 25 mm min−1. Three replicates were performed for all film samples. The values of TS, EAB, and YM were computed using equations (4) to (6)
Color measurements
Measured technique of film color was explained by Datta et al., the specimen of films cut and located over the scanner glass (Canoscan LIDE 100, Japan) with a resolution of 300 dpi. 20 The analysis of scanned image was determined with the software Image J (1.4.3.67 version). Lightness (L), redness (+a) or greenness (−a), and yellowness (+b) or blueness (−b) of the films were determined. Films were determined on the surface corresponding to the white standard plate having color coordinates of L* = 98.416, a* = −0.007, and b* = −1.995.
All experiments were carried out in triplicates three times. Equations (7) to (9) were used to calculate the total color difference (ΔE) and whiteness index (WI) and yellowness index (YI) of specimens, respectively 21
Opacity
The film samples’ opacity (OP) was assessed based on the technique of Siripatrawan and Harte by determining the absorbance at 600 nm with a spectrophotometer model DR5000 HACH (Colorado, USA). 22 The films were cut into a rectangle piece and located in a spectrophotometer test cell directly. An unfilled test cell was utilized as the control specimen. The OP was computed with the following equation
where Abs600 is an absorbance value at 600 nm and x refers to the thickness of the film (mm).
Total phenolic content
To determine the total phenolic (TP) content, the technique explained with Siripatrawan and Harte was utilized, 22 based on the following equation
where T represents total content of phenolic compound (mg g−1 dried film, in gallic acid equivalents), C refers to the level of gallic acid attained from the calibration curve (mg mL−1), V represents the film extract volume (mL), and M is the dried film weight (g).
DPPH radical scavenging activity
The radical scavenging behavior of films was measured using DPPH free radical, based on technique with Noronha et al. using small changes. 23,24
About 100 mg of film specimens were cut into small pieces and dissolved, and 2.9 mL of the DPPH solution (0.1 mM of methanol solution) and 0.1 mL of film extract solution were mixed. The resultant solution was strongly agitated and stored in the dark at ambient temperature for 30 min. Absorbance was recorded at 515 nm by means of a UV–visible spectra photometer (DR5000 HACH). The percentage of DPPH radical scavenging activity was computed with the following equation
where
Antibacterial activity
The microorganisms utilized in this research were two gram-positive Bacillus cereus and Staphylococcus aureus and two gram-negative Escherichia coli and Salmonella enterica prepared from the Iranian Research Organization for Science and Technology (Tehran, Iran). Stock cultures corresponding to the examined bacteria were grown for 24 h in Mueller-Hinton Broth (MHB) at 30°C prior to the tests.
The antimicrobial behavior corresponding to the films with MEO was assessed qualitatively following an agar diffusion analysis. Specimens were slice into 6 mm diameter pills by means of a sterile punch and then they were located on plates comprising Mueller-Hinton agar (MHA) which had been planted before with 100 μL of an overnight broth culture comprising almost 108 CFU mL−1 of usual food pathogens. The incubation of plates was performed for 24 h at 37°C. On every plate, one disk was the control EO-free film with the other three pills contained three replicates of every formulation. The total area of zone was computed then subtracted from the area of film pill and this area difference was stated as the “inhibition zone.” 25
Fourier-transform infrared spectroscopy
Fourier-transform infrared (FTIR) spectroscopy was carried out for analyzing functional groups and for providing an insight into the structural features of CMC. The spectrum was acquired using Thermo Nicolet Avatar 370 (Thermo Nicolet, USA). In order to prepare plates, all powders were combined with KBr. The measurement of FTIR spectra was carried out in the wave number ranging from 400 cm−1 to 4000 cm−1.
Scanning electron microscopy
The films microstructure was found with scanning electron microscopy (SEM; Leo 1450VP, Germany). Next, specimens were gold covered by means of a sputter coater model Polaron-SC7620 (England) for 180 s under 5 mA. All specimens were studied with an accelerating voltage of 20.0 kV.
Statistical analysis
The data were analyzed statistically with SPSS statistical software version 16 (SPSS Inc., Chicago, IL, USA). Analysis of variance continued with Duncan’s multiple range tests were used for determination of any significant differences among the treatments at confidence level of 95%.
Results and discussion
EO composition
GC-MS analytical data of major compounds in MEO are shown in Table 1. Twelve compounds were detected and the results show predominant components of the total peak area are Pulegone (59.88%), Menthofuran (15.74%), Eucalyptol (7.97%), and α-Terpineol (5.98%), respectively.
EO composition of MEO.

EO: essential oil; MEO: Mentha pulegium essential oil.
Physical properties of films
Thickness
Thin films were simply isolated from the cast plate. The average of films thickness with different levels of MEO changed from 0.1410 mm to 0.1835 mm (Table 2) and average of control film thickness (without MEO) was 0.0880 mm. Thus, the outcomes revealed that the thickness of films enhanced with raising the content of MEO. This enhance can be ascribed for blocking microdroplets of MEO into the matrix of film. Furthermore, enhance in thickness with adding MEO could be because of enhance in solid content of the films. Similar outcomes were observed by Ojagh et al. for film prepared from chitosan and cinnamon EO. 16
Physical and mechanical properties of CMC films formulated with different concentrations of MEO.a
MC: carboxymethyl cellulose; MEO: Mentha pulegium essential oil; WVP: water vapor permeability; SD: standard deviation; MC: moisture content; WS: water solubility; MA: moisture absorption; TS: tensile strength; EAB: elongation at break; YM: Young’s modulus.
aValues are given as mean ± SD. Different letters in the same column indicate significant difference (p < 0.05) according to Duncan’s multiple range test.
Moisture content
As presented in Table 2, control film has smaller MC than other specimens. Adding MEO considerably (p < 0.05) enhanced the amount of the moisture comparing with the control film, which is ascribed to breakup of the film network. This phenomenon enhanced the water molecules amount that was presented between polymer chains using hydrogen bonding. 26
If the concentration of MEO within the CMC matrix enhanced from 1% to 3%, the amount of moisture reduced considerably (p < 0.05). These outcomes can be ascribed to interaction between the MEO components and the hydroxyl groups corresponding to CMC, which may decrease the accessibility of hydroxyl groups for interacting with molecules of water, so causing a more water-resistant film. Ojagh et al. reported same outcomes. 16
Water solubility
In the current research, the solubility for the control films showed a great WS factor. Similar outcome was observed by García et al. 27 Generally, control film was very hydrophilic, so absorbed water rapidly and caused in swelling.
A significant (p < 0.05) enhance in solubility of water was found by the addition of MEO to the film (Table 2). The greater solubility for oil incorporated films may be allocated to the reemerging polysaccharide interactions with this EO which decline the interactions that had stabilized the structure of polysaccharide network. 28 Moreover, the existence of EO may limit the interaction of glycerol with polysaccharide matrix which would permit an enhance in its leaching phenomenon. 29 Similarly, the inclusion of clove EO to gelatin–chitosan film matrix enhanced their WS. 28
Water vapor permeability
In the current research, the WVP corresponding to the control films was 1.534 ± 0.1910
Though the existence of MEO enhanced the hydrophobicity ratio of films, it resulted in an enhance in the water vapor transfer procedure. This can be accredited to decrease of film cohesion because of the existence of MEO microdroplets. The same outcomes have been found by Bonilla et al. for the impact of EOs on features of films based on chitosan. 30
Moisture absorption
As presented in Table 2, adding MEO at levels 2% and 3% (v/v) did not significantly impact (p < 0.05) the MA of these films comparing with the control films. Though, greater MA was reported for the films comprising 1% MEO. Similar outcomes were reported by Salarbashi et al. for characterizing soluble soybean polysaccharide film combined with EO intended for food packaging. 31
Mechanical properties
Generally, the mechanical property of the film depends on the type of EO. Analysis of the TS (resistance to elongation), EAB (stretching capacity), and YM of packaging materials results in better forecasts about their mechanical features for applying in foodstuffs.
Table 2 indicates the effect of incorporating EO on the mechanical features of CMC-based films. The incorporation of MEO into the CMC films considerably decreased in TS when MEO amount was enhanced. This can be ascribed to the complex structures generated between the CMC chains and lipids, which decrease the cohesion corresponding to the polymer network forces, so decline the resistance to breakage of specimen films. 32
On the other hand, incorporating MEO increased the elongation compared with the control film. It seems that the above-mentioned variations in the interaction balances showed a plasticizing influence, even at small amounts of EO, which makes the film more stretchable (high EAB values). This observation coincides with the outcomes found for hydroxyl propyl methylcellulose comprising oleic acid. 32
Table 2 indicates that the EO caused a considerable decrease in the YM and, so, the films generated are less rigid. Variations in mechanical features as influenced by polyphenolic compounds were also found for other biopolymeric films, containing murta leave extract in gelatin films of tuna-fish 28 and antioxidant borage extract of commercial gelatin films of fish. 33
Color measurements
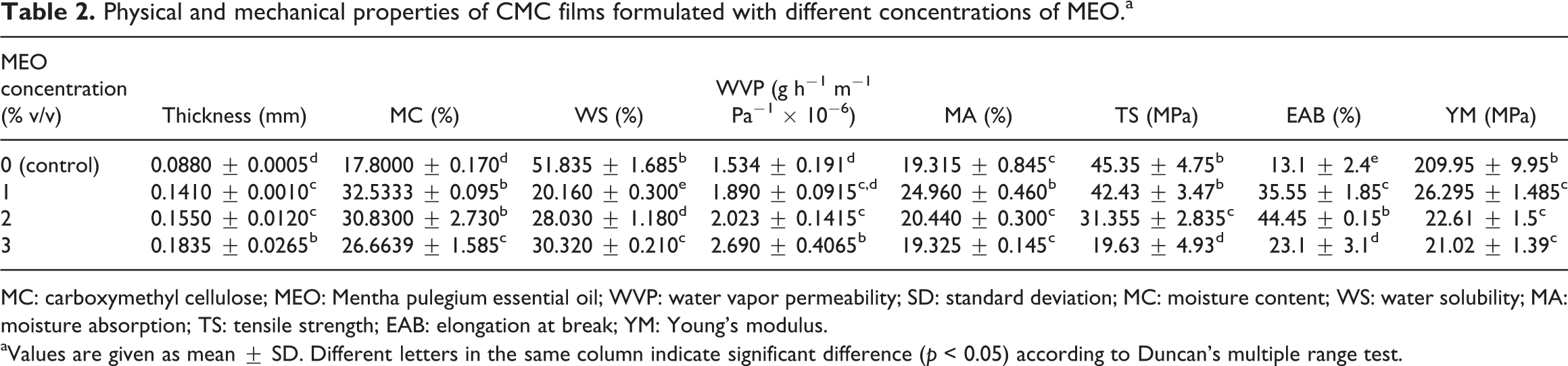
In general, CMC films with no MEO indicated larger L values but smaller b values that are significant (p < 0.05) and smaller a values that are not significant (p < 0.05) comparing with those films with MEO.
The outcomes in Table 3 indicated that L values slightly reduced (p < 0.05) with the addition of 1% MEO, while a value did not alter considerably (p > 0.05). There were no significant differences between L and a value presented the MEO levels enhanced from 1 to 3%.
Effect of different concentrations of MEO on optical properties of CMC films.a
CMC: carboxymethyl cellulose; MEO: Mentha pulegium essential oil; SD: standard deviation; WI: whiteness index; YI: yellowness index; OP: opacity.
aValues are given as mean ± SD. Different letters in the same column indicate significant difference (p < 0.05) according to Duncan’s multiple range test.
The ΔE value indicated same trend to variations in L values. This outcome is in agreement with results where combined κ-carrageenan films became denser with incorporating EOs. 34 This impact is possible because of enhance in diffuse reflectance provoked using light scattering in the lipid droplets which drops both the intensity of light scattering and the WI of film.
Opacity
The control films showed higher transparent (smaller OP value) comparing to those comprising MEO (Table 3). The OP of the film specimens considerably enhanced (p < 0.05) with raising MEO level. This is because of dispersing oil droplets in the polysaccharide-based matrices. 35 Furthermore, this phenomenon is corresponded with the light scattering provoked with lipid droplets (with a diverse refractive index) distributed over the network of film. 36
Incorporating EOs decreased light transmittance at 600 nm (visible region) and at 280 nm (UV region). Therefore, this kind of films shows barrier features against UV light and could be used to possibly prevent oxidation of food. Moreover, Gómez-Estaca et al. found an enhance in OP of the film as a result of adding antioxidant in fish gelatin films comprising borage extract. 33
Total phenolic content
Phenolic compounds experience a complex redox reaction with phosphotungstic and phosphomolybdic acids available in the Foline Ciocalteu reactant. The development of color is because of the electrons transfer at basic pH for reducing the phosphotungstic and phosphomolybdic acid complexes. 37
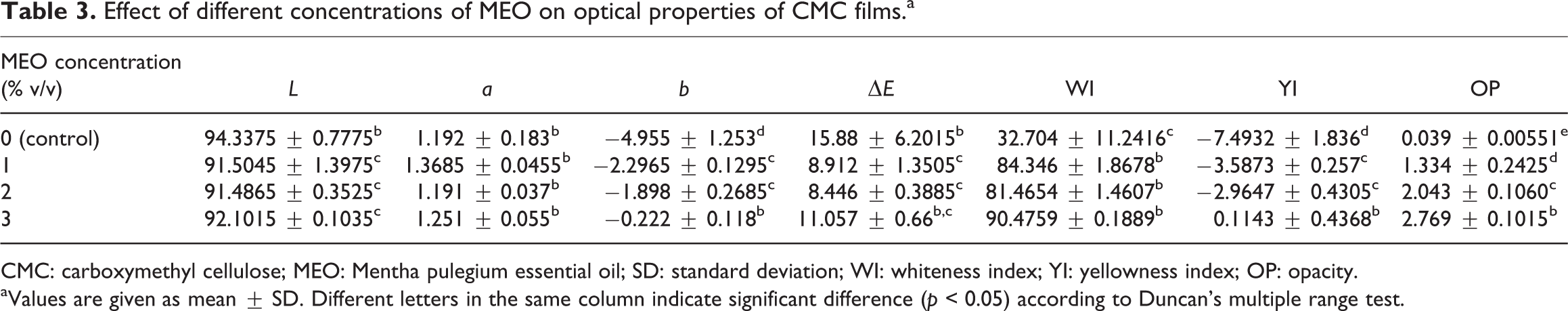
The outcomes of this research indicated that TP of films enhances considerably with enhancing MEO level (Figure 1(a)). The maximum amount of TP (0.2673 mg gallic acid/g film) was found in films at a level of 3% MEO.
(a) TP content and (b) DPPH scavenging activities of films.
DPPH radical scavenging activity
Based on Moradi et al., the antioxidant power degree for edible films is usually proportional to the concentration of added antioxidant additives. 38 Also, based on observation of Pirouzifard et al., increasing the concentration of EO (Salvia afficinalis) in the film composition caused a significant increase in the antioxidant activity of active film based on potato starch films (p < 0.05). 24
In the DPPH test, the antioxidants cause reduction of the DPPH radical to compound which is yellow, and the reaction extents are affected by the hydrogen donating potency of the antioxidants. 39 The outcomes indicated that DPPH scavenging behavior corresponding to the films significantly enhanced (p < 0.05) with increasing MEO concentration as presented in Figure 1(b). The impact of EO amount on the films’ antioxidant behavior was expected and has been discussed earlier in the article. 33,38 Our results verified this and indicated that MEO-containing films at a level of 3% had the maximum antioxidant activity (27.51%). The outcomes proposed that incorporation of MEO into CMC film developed antioxidant behavior of the film.
Antibacterial activity
The antibacterial behavior of CMC films combined with MEO against two gram-negative and two gram-positive bacteria is presented in Table 4. The outcomes presented MEO’s considerable inhibitory impacts on gram-positive and gram-negative bacteria. The control film did not present an inhibition zone around the film disk. The outcomes indicated that antibacterial impacts enhanced with raising MEO level. MEO at concentration 3% showed the maximum inhibition zone against all examined bacteria but did not indicate considerable difference (p < 0.05) with specimen films comprising 2% MEO.
Antibacterial activity (inhibitory zone, mm2) of CMC film disks incorporated with various concentrations of MEO against gram-positive and gram-negative bacteria.a
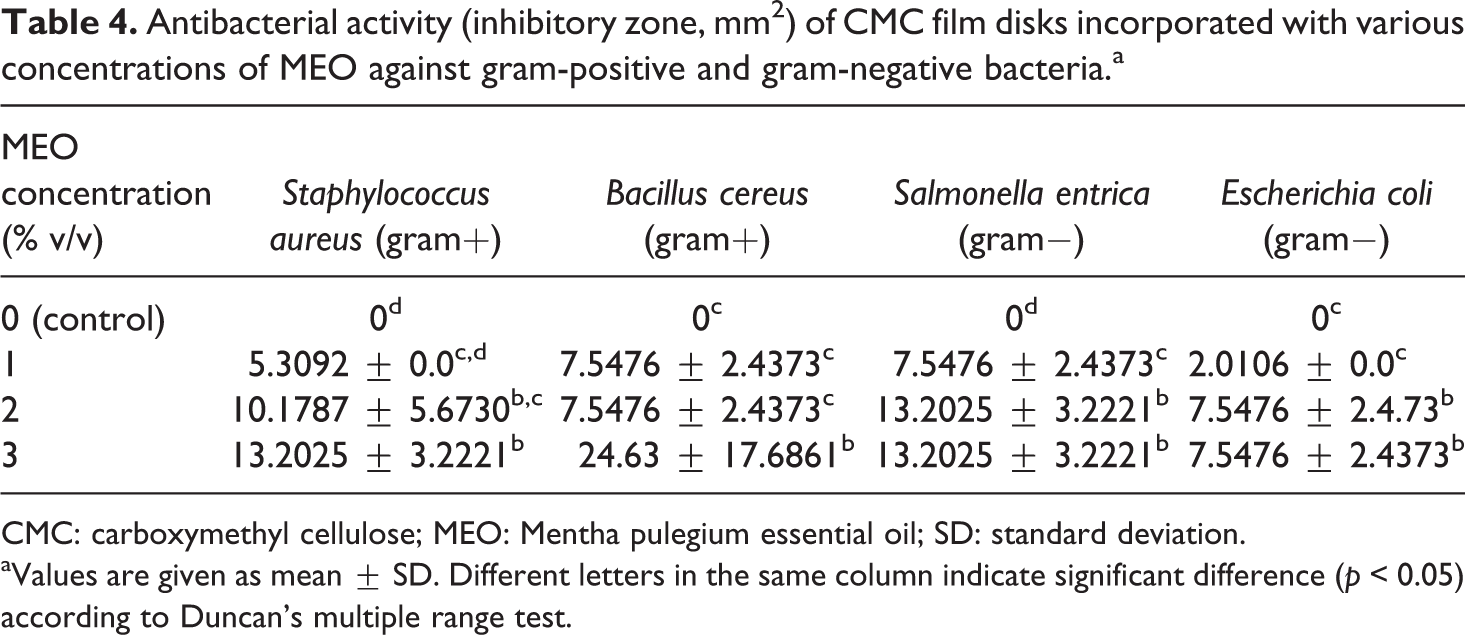
CMC: carboxymethyl cellulose; MEO: Mentha pulegium essential oil; SD: standard deviation.
aValues are given as mean ± SD. Different letters in the same column indicate significant difference (p < 0.05) according to Duncan’s multiple range test.
Different mechanisms have been proposed for the antibacterial impact of EOs, including their attacking the phospholipid bilayer corresponding to the systems of cell membrane or disrupting enzyme, the fatty acid hydro peroxidase production, the cytoplasm coagulation, lipids and proteins damage, the proton motive force disturbance, and the genetic material corresponding to bacteria being compromised. 40
The first of these has been found to be a key antimicrobial mechanism because of the lipophilic nature of EOs, which allows them to act on the membrane of cell, leads to main morphological damages, disrupts membrane integrity, and reduces the number of cell. These outcomes are in accordance with the outcomes of several research. 41,42
Several investigations that have studied the behavior of EOs against food spoilage organisms and food-borne pathogens agree that, in the main, EOs is a low activity against gram-positive comparing with gram-negative bacteria. 40
During the antibacterial activity test, E. coli presented the maximum resistance, while B. cereus showed the maximum sensitivity to specimen films comprising MEO, with an inhibition zone of 24.63 mm2.
FTIR spectroscopy
The FTIR analysis was applied for identifying bands related with functional groups available in films. Figure 2(a) indicates the FTIR spectrum corresponding to CMC control film, and Figure 2(b) indicates the FTIR spectrum corresponding to films incorporated with MEO. In general, FTIR spectra corresponding to control film and films comprising EO showed analogous basic peaks but the amplitudes of peaks varied. The peaks visible at 800–1200 cm−1 area are carbohydrate’s fingerprint usually applied for comparing various structures. Furthermore, two bands at 923–853 cm−1, related with C–C bonds skeletal stretching vibration, are more obvious in the films combined with EO. 43 As presented, the peaks at 1112 cm−1 and 1054 cm−1 may relate to C–O stretching vibrations. Two absorption peaks are observed at 1300–1500 cm−1 that may be allocated to symmetric and asymmetric deformational vibrations corresponding to C–H in CH3 groups. Stretching intense bands are observed at 1608 cm−1 relating to C=O of the carboxyl groups of films. Hydrogen bonds (inter- or intramolecular) are usually identified with –OH stretching vibrations. Wide areas of absorption in the range of 3000–3500 cm−1 indicate several properties like free hydroxyl groups stretching bonds and O–H bands corresponding to carboxylic acid. 44 The wide absorption bands in the range of 2800–3000 cm−1 represent C–H absorption; these contain CH, CH2, and CH3 stretching and bending vibrations. 44
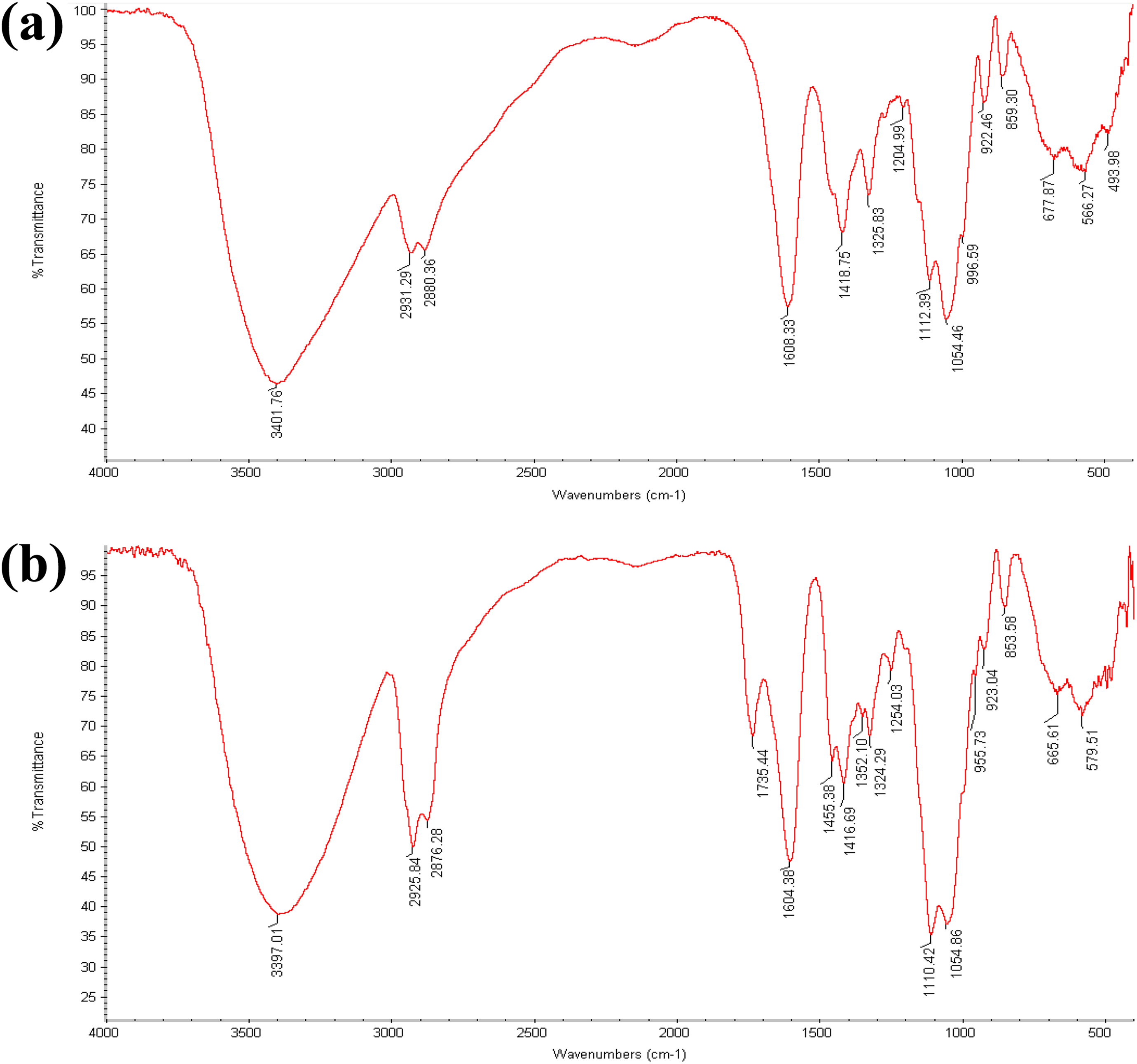
(a) FTIR spectrum of CMC control film and (b) FTIR spectrum of CMC film comprising 2% MEO (optimum concentration).
Two absorption peaks were found at 1735 cm−1 and 1455 cm−1 at spectrum corresponding to CMC films incorporated with MEO (Figure 2(b)). However, these peaks were not found at the spectrum corresponding to CMC control film (Figure 2(a)). The bending vibrations observed at 1455.38 cm−1 may associate with symmetric and asymmetric deformations corresponding to –CH3 group. A peak was found at 1735cm−1 in films incorporated with MEO because of the C=O stretching from keto esters, which is a feature of pulegone as found by Molinaro et al. 45
Scanning electron microscopy
Figure 3(a) and (b) showed SEM micrograph specimen films comprising 2% MEO and control film, respectively. According to the figures, the films exhibited almost a compact, continuous, and smooth microstructure without irregularities. The film images indicated that the addition of EO resulted in a heterogeneous structure in which the continuous polysaccharide network entrapped oil droplets. In general, the roughness of surface structure was more noticeable in films combined with EOs comparing to that observed in control film. CMC films combined with MEO indicated a loose texture with sponge-like structure, with pores distributed through the matrix of CMC. EO might be evaporated over drying causing the generation of micropores throughout the films. The presence of holes or pores might be corresponded to the volatility of the Mentha EO. Actually, these spaces were filled with EO evaporated from the surface of film. 36

(a) SEM surface images of CMC control film SE (left) and QBSD (right) and (b) SEM surface images of CMC film comprising 2% MEO (optimum concentration).
The pore and a larger number corresponding to spaces of film combined with MEO were in accordance with greater WVP of this film, comparing with CMC control film. Though the hydrophobic feature of EOs could impact the hydrophilic and hydrophobic behavior of the specimen films, the physical factors showed a main impact on the WVTR via the film.
The improving in surface coarseness with the existence of EOs ascribed to the migration of oil droplets upwards the specimen films. Also, holey structure related to extra volatilization through water evaporation. 36
Conclusion
The properties of the specimen films were greatly affected by MEO incorporation. Our findings revealed that MEO has a good potential for addition to CMC in the production of antimicrobial films and coatings with various applications in the food industry. Films comprising MEO exhibited a large inhibitory effect against four selected bacteria. CMC films incorporating MEO showed antioxidant activity. Furthermore, the incorporation of MEO caused a significant decrease in TS and YM, but increased the EAB of the CMC films. Aggregation and creaming of oil droplets happened to various extents (depending on the ratio of MEO added) during film drying. These phenomena could be related to the reduction in TS and higher WVP of films comprising MEO, as compared to the CMC control film. FTIR showed that the interaction between CMC polymer matrix and MEO happened. The SEM analysis results demonstrated that the MEO is well dispersed in the CMC polymer matrix. Our findings showed that the incorporation of MEO into CMC films has great potential in the production of active, biodegradable packaging. It is worth mentioning that investigations are needed to test the effectiveness of these films on selected food systems.