Abstract
This study provides a comprehensive analysis of PVA/CMC hydrogels, investigating their wettability, swelling behavior, antimicrobial efficacy, and surface morphology. Essential oils, including argan, clove, and aloe vera, were incorporated via the freeze-thaw method, significantly influencing the physicochemical properties of the hydrogels. The intrinsic hydrophobic nature of essential oils played a crucial role in modulating swelling characteristics, with PCO200 series exhibiting the highest swelling ratio due to lower crosslinking density, whereas clove oil-loaded hydrogels demonstrated the lowest swelling capacity, indicative of their enhanced hydrophobicity. Contact angle measurements further confirmed this trend, with PCO135 displaying the highest recorded value (83.20° ± 1.66°) and PCO235 following closely (79.86° ± 1.60°), underscoring the pronounced hydrophobic effect of clove oil compared to other formulations. SEM analysis revealed a well-defined macroporous structure in essential oil-containing hydrogels, with pore distribution and morphology varying depending on the oil type and concentration. FTIR spectroscopy identified distinct characteristic peaks corresponding to the functional groups of the incorporated oils, elucidating their molecular interactions within the hydrogel network. Antimicrobial analysis highlighted clove oil’s robust activity, with enhanced effects in specific formulations against bacteria such as
Introduction
Hydrogels are functional polymer materials with a three-dimensional cross-linked network structure, allowing them to swell rapidly while remaining insoluble in water. Polyacrylic acid (PAA), polyacrylamide (PAM), and polyvinyl alcohol (PVA) are commonly used in hydrogel formulations due to their hydrophilic nature, ease of processing, and machinability. However, compared to PAA and PAM, PVA is more widely preferred because it is relatively inexpensive, commercially available, and exhibits superior biocompatibility and environmental friendliness. Additionally, PVA contains abundant hydrophilic side groups, enhancing its ability to form stable hydrogel networks. Hydrogels formed by blending PVA with natural polymers further improve biocompatibility, making them highly suitable for biomedical and environmental applications.
1
Polyvinyl alcohol (PVA) is widely used in polymeric hydrogels due to its hydrophilicity, biocompatibility, and ease of processing. However, its poor mechanical properties often necessitate blending with natural polymers capable of intermolecular crosslinking with its hydroxyl groups. Cellulose, chitosan, and starch are commonly incorporated into hydrogels because of their biocompatibility and biodegradability. Among these, carboxymethyl cellulose (CMC), a cellulose ether, is produced by treating alkaline cellulose with the sodium salt of monochloroacetic acid and is a water-soluble cellulose derivative. CMC dissolves in water-based solutions, including weak acids such as acetic acid (AAc).
2
When added to acetic acid, the carboxymethyl groups in CMC interact with acetic acid molecules via hydrogen bonding, enhancing its solubility.
2
The dissolution behavior depends on factors such as the acetic acid concentration and the number of carboxymethyl groups present in the cellulose chain. Additionally, mixing facilitates the dissolution of polar polymers like CMC in acetic acid. Acetic acid is non-toxic, affordable, readily available, and exhibits antimicrobial activity against various microorganisms.3–6 Sodium carboxymethyl cellulose (NaCMC) is an ionic cellulose ether with multiple carboxyl groups and a double-helix structure.
7
NaCMC, an ionic water-soluble polymer, exhibits high solubility in aqueous solutions due to the electrostatic repulsion forces generated by its ionized carboxylate (-COO-) groups. These repulsion forces are further enhanced in distilled water, leading to increased chain mobility and dissolution. However, the high viscosity of NaCMC in water can make it challenging to obtain a homogeneous solution. In contrast, when NaCMC is introduced into an acetic acid (AAc) environment, its dissolution behavior changes significantly due to the hydrogen bonding interactions between the carboxymethyl groups and acetic acid molecules. The extent of solubility is influenced by the concentration of acetic acid and the number of carboxymethyl groups present in the cellulose chain, while stirring facilitates the dissolution of NaCMC, a polar polymer, in acetic acid. Moreover, in an acetic acid medium, the protonation of carboxylate (-COO-) groups leads to their conversion into the non-ionized (-COOH) form, reducing electrostatic repulsion and resulting in a more compact polymer structure. This structural compaction contributes to the formation of a denser and more tightly cross-linked hydrogel network, enhancing its mechanical strength and stability.8,9 Additionally, acetic acid not only acts as a solvent but also improves the antimicrobial properties of the hydrogel due to its natural antimicrobial activity against various microorganisms, making the resulting hydrogels more resistant to microbial contamination. Building upon the solvent-dependent dissolution behavior of NaCMC, its integration with PVA in hydrogel formulations introduces additional structural and functional considerations. Crosslinking can occur between the hydroxyl groups of PVA, located on the outer regions of the macromolecular helices, and the carboxymethyl groups of CMC. Due to their biocompatibility, biodegradability, and water-absorbing properties, PVA and CMC are frequently employed in hydrogel preparation.
10
When blended in appropriate ratios, these polymers facilitate the formation of hydrogels with desirable mechanical strength, swelling capacity, and stability. The hydrogel formation performance of PVA/CMC systems is influenced by several factors, including the degree of crosslinking, the molecular weights of PVA and CMC, and the polymer solution concentrations, all of which play a crucial role in determining the final properties of the hydrogel. Typically, PVA/CMC hydrogels form rapidly under mild crosslinking conditions, exhibiting high water absorption capacity, a high swelling ratio, and excellent mechanical properties. The choice of crosslinking method significantly impacts the final hydrogel properties. Chemical crosslinking agents such as glutaraldehyde, formaldehyde, and epichlorohydrin can be used to enhance the mechanical strength and stability of PVA/CMC hydrogels. Alternatively, physical crosslinking techniques, such as freeze-thawing, offer a reversible and biocompatible approach to hydrogel formation.11,12 The freeze-thaw (F-T) method is one of the most commonly employed physical crosslinking techniques for PVA/CMC hydrogels. This method involves subjecting a PVA/CMC polymer solution to multiple cycles of freezing and thawing, promoting the formation of physical cross-links between polymer chains. The resulting hydrogel exhibits a porous structure with high water content and superior mechanical properties. Studies conducted on the preparation and characterization of PVA/CMC hydrogels using the F-T method have demonstrated its effectiveness in hydrogel synthesis.11,13 For example, El Salmawi et al. prepared PVA/CMC hydrogels using the F-T method. They investigated the effects of PVA/CMC concentration thawing time on the physical and mechanical properties of the hydrogels. They found that the swelling ratio increased as the mixture increased CMC composition. This hydrogel with the composition 80/20 (CMC/PVA) was used as a superabsorbent in soil for agriculture, resulting in increased water retention in soil containing this hydrogel.
11
Shin et al., studied the preparation of PVA/CMC hydrogels containing various concentrations of CMC from 2% to 10% by weight, using cyclic freezing/thawing and subsequent y-ray irradiation to evaluate the effect of CMC content on the cell growth rate of the hydrogels. The fastest cell growth was observed for PVA/CMC hydrogels containing 6% CMC by weight, as determined by the pore size of the hydrogels, the metronidazole drug release rate, the scratch test, the highest compressive force. This shows that they are very suitable for wound dressing that requires rapid regeneration.
14
In another study, Alsahag et al., were developed smart therapeutic PVA/CMC aerogel dressing able to sense the healing. Red-cabbage extract was include into this PVA/CMC hydrogel solution by freeze- thawng method. Also, anthocyanin was integrated as a water-soluble direct dye at various concentrations into carboxymethyl cellulose/polyvinyl alcohol. When the effects of anthocyanin content on the morphology, stiffness, air permeability and mechanical behavior of the airgel-like dressing were investigated by various analytical methods, they found to exhibit good comfort, antimicrobial and colorfastness properties.
15
PVA and CMC-based hydrogels have a wide range of applications in biomedical fields. PVA is widely used in biomaterials such as wound dressings due to its high water retention capacity, flexibility, and biocompatibility, while the hydrophilic and biodegradable nature of CMC provides wound healing acceleration and antimicrobial properties.10,16 The combination of PVA and CMC enhances mechanical strength, supporting wound healing and providing effective antibacterial protection against pathogens such as
Plant-based essential oils are preferred over synthetic antibacterial agents because they are generally considered to be safer and more environmentally friendly. Synthetic antibacterial agents can have negative effects on the environment and may contribute to the development of antibiotic-resistant bacteria. In contrast, essential oils are derived from natural sources and are biodegradable. Additionally, essential oils are known for their diverse range of therapeutic properties, including their ability to improve skin health and reduce inflammation, making them a popular choice for use in skin care and wound healing applications. Overall, the use of essential oils in hydrogels provides a natural and effective way to enhance their antimicrobial properties, making them useful in a variety of applications. In some of these studies, PVA/inulin-based films were produced using the solvent casting method, reinforced with Pickering emulsions, and given antimicrobial properties. Niaouli essential oil (NEO), stabilized with β-cyclodextrin (β-CD), enhanced antibacterial activity, with films containing 1:3 and 1:5 β-CD/NEO ratios showing strong inhibition against
In this research, PVA was dissolved in distilled water, while CMC was dissolved not only in distilled water but also in 1% acetic acid to enhance its antimicrobial properties. Unlike conventional hydrogel synthesis methods that typically require chemical cross-linkers, this study employed a freeze-thaw process to achieve physical cross-linking without the use of toxic chemical additives. This approach not only ensures the development of an environmentally friendly and biocompatible material but also enhances the mechanical integrity of the hydrogel network. Additionally, the incorporation of essential oils such as argan oil, aloe vera oil, and clove oil imparted bioactive properties, providing antimicrobial functionality and potential wound-healing applications. The structural, morphological, and chemical properties of the synthesized hydrogels were comprehensively characterized using SEM, XRD, and FT-IR, while swelling behavior and contact angle measurements were conducted to evaluate their hydrophilicity and stability. Furthermore, antimicrobial activity tests were performed against common bacterial strains to assess their potential for biomedical applications. This study builds upon the work of Bayraktar et al., 21 further refining the material properties and expanding its functional scope to include bioactive capabilities, thereby making a significant contribution to the advancement of hydrogel-based materials.
Materials and methods
Materials
Polyvinyl alcohol (PVA; typical average MW = 146000–186000; 98.0–98.8% hydrolyzed), Sodium carboxymethyl cellulose (CMC; typical average Mw = ∼250000) were purchased from Sigma-Aldrich Co. Acetic acid (glacial 100%, pro analysis) was purchased from Merck (Darmstadt, Germany) was used as the solvent. Tween 80 were purchased from Merck (Darmstadt, Germany). Clove oil was purchased from Arifoğlu Spıce and Food Ind. Trade. Ltd. Sti., also Argan oil and aloe vera oil were purchased from Smart Chemistry Trade and Consulting Ltd. Sti. (Çiğli, Izmir). These materials were used as received without further purification and functionalization.
Method
Preparation of PVA/CMC/oil nanocomposite hydrogels
In our previous study, PVA/NaCMC hydrogels were synthesized with varying CMC ratios, and optimal compositions of 1.8%, 3.5%, and 7% were identified. 21 For hydrogel formation, CMC was dissolved in two different solvents, 1% acetic acid (AAc) and distilled water (Dw), while PVA was solubilized in distilled water. The hydrogel was obtained through a freeze-thaw process with three cycles, ensuring physical crosslinking. Based on these findings, the 3.5% CMC composition was selected for further studies due to its favorable properties.
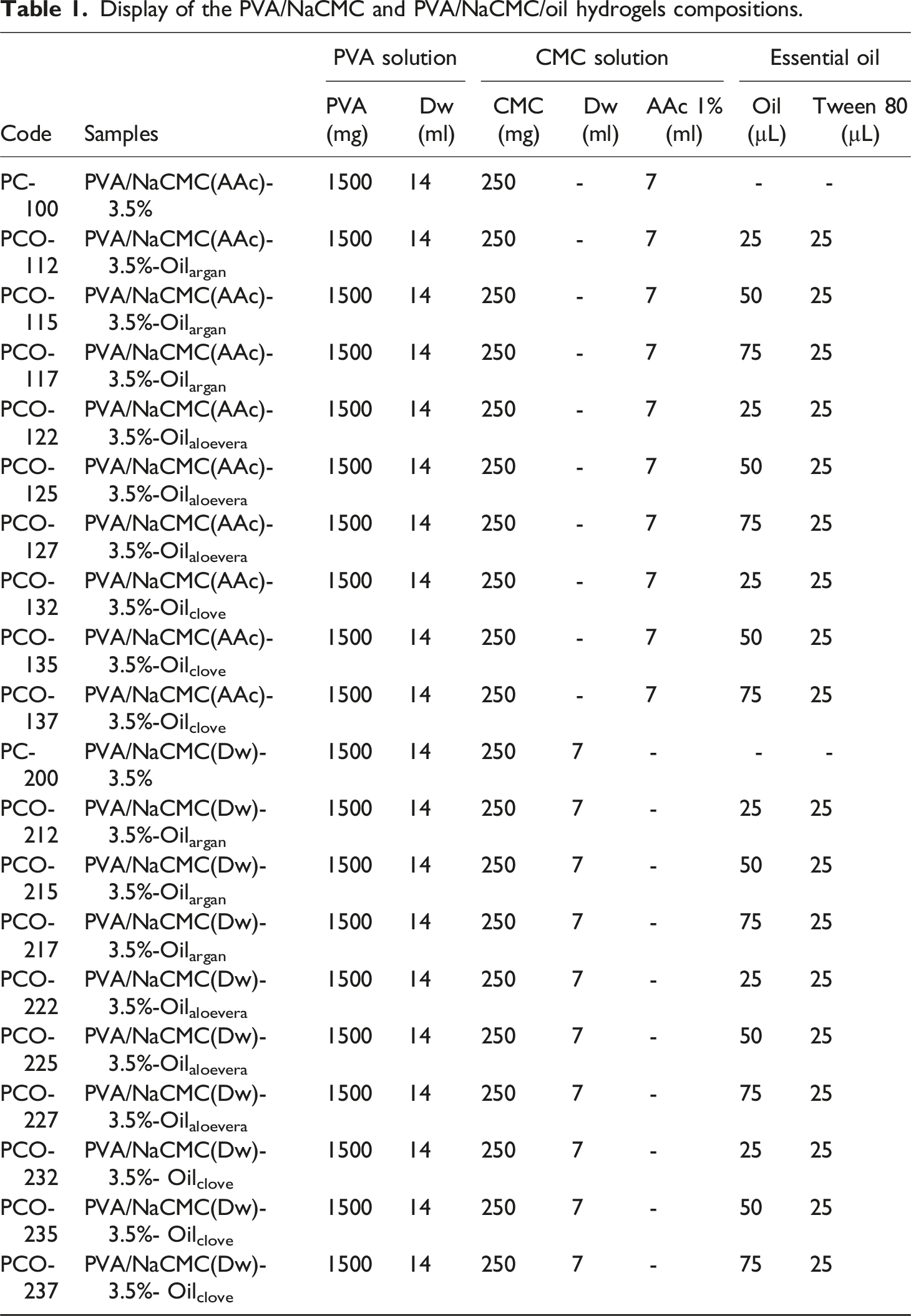
The hydrogels formulated with 3.5% CMC, designated as PC-100 and PC-200, as well as PCO (PVA/CMC-3.5%-Oil), were prepared as shown in Table 1. Firstly, CMC was dissolved in 1% AAc and distilled water, while PVA was dissolved in distilled water. To prepare the PC (PVA/CMC-3.5%) hydrogel, precise amounts of PVA and CMC solutions were mixed according to our previous study.
21
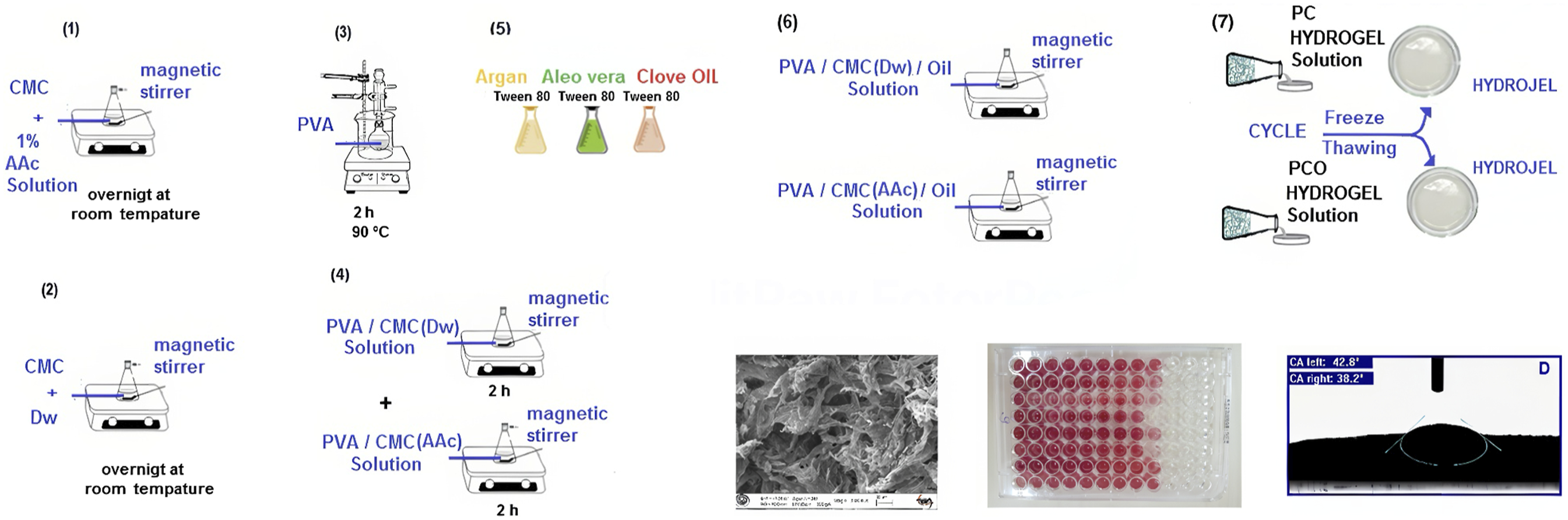
For the PCO (PVA/CMC-3.5%-Oil) hydrogel, essential oil solutions of clove, argan, and aloe vera were incorporated in specific proportions as indicated in Figure 1. Different volumes of oils (15, 25, and 50 µL) were tested, with each homogenized in 50 µL of Tween 80 using ultrasonication in a water bath for 15 minutes before being added to the PC solutions. The sample coding system used in this study is as follows: AAc-based Samples (100 series) PC-100: PVA/NaCMC(AAc)- 3.5% Argan Oil: PCO-112 (15 µL), PCO-115 (25 µL), PCO-117 (50 µL) Aloe Vera Oil: PCO-122 (15 µL), PCO-125 (25 µL), PCO-127 (50 µL) Clove Oil: PCO-132 (15 µL), PCO-135 (25 µL), PCO-137 (50 µL) Dw-based Samples (200 series) PC-200: PVA/NaCMC(Dw)- 3.5% Argan Oil: PCO-212 (15 µL), PCO-215 (25 µL), PCO-217 (50 µL) Aloe Vera Oil: PCO-222 (15 µL), PCO-225 (25 µL), PCO-227 (50 µL) Clove Oil: PCO-232 (15 µL), PCO-235 (25 µL), PCO-237 (50 µL). Display of the PVA/NaCMC and PVA/NaCMC/oil hydrogels compositions. Prepation of PVA solution (3), prepation of 1.8, 3.5, 7% (W/W) changing amounts of CMC with 1% acetic acid and distilled water (1-2), prepation of PVA/CMC(Dw) and PVA/CMC(Dw) solutions (4), essantial oil (5), prepation of PVA/CMC(Dw)/OIL and PVA/CMC(Dw)/OIL (6), PC and PCO solutions pour into sterile petri dishes (7).
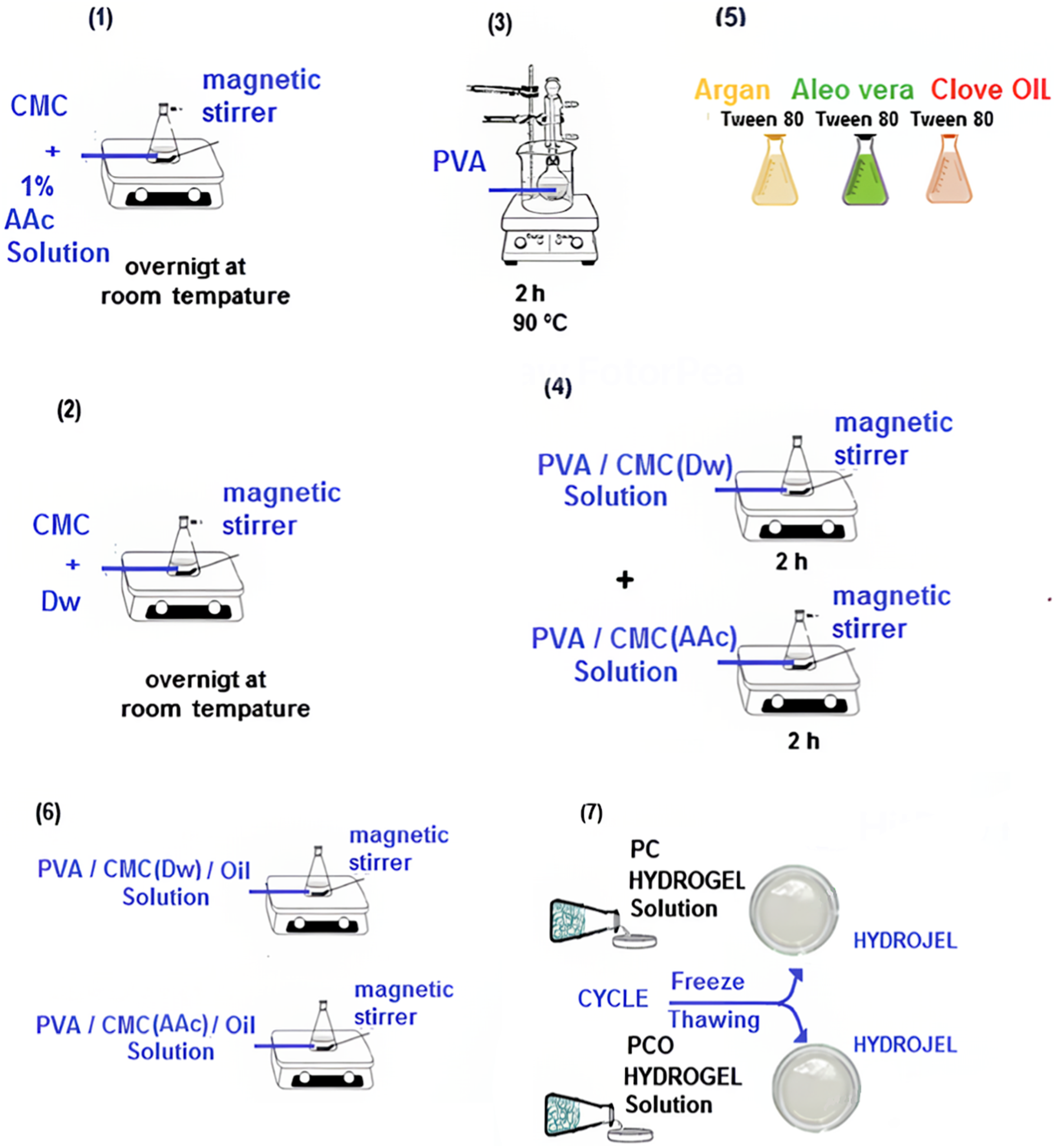
Subsequently, the solution mixture, designated as PCO, was subjected to vigorous magnetic stirring for 30 minutes to ensure homogeneity, following which it was cast into Petri dishes. The ensuing mixtures were subjected to a cyclic freeze-thaw method involving freezing at −20°C for 18 hours and subsequent thawing at 20°C for 6 hours, as illustrated in Figure 3, depicting the physical crosslinking process. This freeze-thaw cycle was repeated three times to ensure the attainment of desired hydrogel properties, as established by Lim, Lee et al., Shin, Lee et al., Afni, Taba et al.14,40,41
Characterization
Swelling test
The determination of the swelling degree for the hydrogel samples was conducted following the gravimetric method, a well-established technique in hydrogel characterization. The water uptake and swelling behavior of the prepared hydrogels were assessed by quantifying the weight differential (ΔW) between the initial weight (WD) of hydrogel specimens, each measuring 2 × 2 cm2, after 24 hours of drying at room temperature, and the subsequent weight (WS) recorded after immersing the samples in distilled water at room temperature for specific time intervals, namely, 1, 3, 5, 7, 12, and 24 hours. Swelling test measurements were conducted on samples from three different regions for each hydrogel formulation, and each test was repeated three times. The reported values represent the average results along with standard deviations to reflect data variability. This process was rigorously repeated thrice to ensure precision and accuracy. To minimize surface moisture, excess water on the hydrogel surface was meticulously blotted using filter paper before obtaining the wet mass (WS). The degree of swelling (SD) was meticulously calculated using equation (1), as previously reported by Părpăriţă, Cheaburu et al.
42
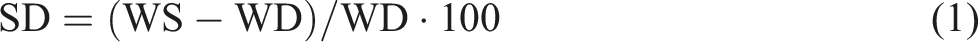
SD represents the degree of swelling (dimensionless), WS denotes the weight of the hydrogel sample after swelling (grams), WD signifies the initial weight of the dry hydrogel specimen (grams).
Contact angle measurement
The assessment of hydrogel wettability involved the utilization of a DataPhysics OCA contact angle instrument, equipped with a camera, in accordance with established research protocols. In this methodology, a controlled volume of 5 µl of distilled water was delicately dispensed onto the hydrogel’s surface using a micro syringe. A brief incubation period of 5 seconds allowed for equilibration, after which the contact angle formed by the water droplet on the hydrogel was meticulously captured by the instrument’s camera. The measurements were conducted under standard room temperature conditions, with the resulting contact angle (CA) values accurately documented. To ensure precision and reliability, this procedure was replicated at least three times on distinct areas of the hydrogel. The obtained contact angle values were then averaged, and standard deviation calculations were performed to reflect data variability.43,44
Antimicrobial activity screening by broth micro dilution test
In this study totally eight microorganisms (
Mueller-Hinton Broth (100 µL) was placed into each 96 wells of microplates. Chemicals and hydrogel samples were initially prepared and 100 µL of sample were added into first wells of microplates and two fold dilutions of the chemicals were made by dispensing the solutions to the remaining wells. Then, 100 µL of culture suspensions were inoculated to each well. Gentamicin was used as positive control. The sealed microplates were incubated at 35°C for 18 h. Microbial growth was determined by adding 20 µL of 2,3,5-Triphenyl-tetrazolium chloride (0.5%) after incubation to each well and incubating for 30 minute at 37°C. The lowest concentration of the extracts that completely inhibit macroscopic growth was determined as minimum inhibitory concentrations (MICs). 46
Scanning electron microscopy (SEM)
The examination of hydrogel morphology was conducted using a scanning electron microscopy (SEM) apparatus, specifically the Jeol JSM-6510 LV model. To prepare the SEM samples, the hydrogel specimens were subjected to controlled fracturing under cryogenic conditions within a liquid nitrogen environment. Subsequently, these fractured samples were securely affixed onto sample stubs, and a thin gold coating was meticulously applied under a vacuum of 10−2 bar prior to microscopy. The SEM analysis focused on scrutinizing the surface morphologies of the hydrogels, providing valuable insights into their structural characteristics.
Fourier-transformed infrared spectroscopy (FT-IR)
The examination of crosslinking and the identification of functional groups within the hydrogel samples were undertaken using Fourier-transform infrared spectroscopy (FT-IR) conducted with a Bruker instrument purchased in Waltham, Massachusetts, USA. To prepare the hydrogel specimens for FT-IR analysis, they were meticulously cut into square pieces measuring 5 × 5 mm2 and subsequently dried at room temperature. The FT-IR analyses were conducted with precision, employing 16 scans for each sample, spanning the wavenumber range of (4000–400) cm−1. This analytical approach facilitated the comprehensive investigation of crosslinking bonds and the characterization of functional groups within the hydrogel matrix.
Results
Swelling test
Some researchers investigating the degradation behavior of PVA/CMC hydrogels have reported a decline in the swelling rate after the 6th hour.47–49 Accordingly, this study systematically examines time points both before and after this critical period. The selected intervals (1, 3, 5, 7, 12, and 24 hours) facilitate a comprehensive assessment of the swelling reduction rate, providing crucial insights into the long-term stability of the hydrogels. This research contributes valuable findings regarding the swelling behavior of PVA/CMC hydrogels, particularly in the presence of essential oils. Given their inherent hydrophobic nature, essential oils significantly influence hydrogel swelling by reducing the available free volume for water absorption within the polymer network. This effect leads to a complex interplay of factors governing the swelling process, with oil concentration emerging as a key determinant. As illustrated in Figure 2, the results demonstrate that PCO200 hydrogels exhibit a higher swelling capacity than PCO100 hydrogels. This distinction is attributed to the higher degree of crosslinking in PCO100 hydrogels, which restricts water absorption by diminishing the available free volume within the polymer matrix. Conversely, PCO200 hydrogels, characterized by a lower crosslinking density, exhibit greater water uptake, resulting in an increased swelling capacity. These findings are further corroborated by FTIR analysis, which reveals a higher -OH spectrum intensity, and SEM images, which display a greater pore density within the hydrogel structure. Furthermore, as evidenced in Figure 2, the swelling behavior of hydrogels follows a distinct trend: an initial rapid swelling phase, followed by a stabilization period between the first and third hours. Subsequently, a gradual decline in swelling is observed, culminating in notable structural degradation by the 24th hour. Notably, this trend is consistently observed in PCO hydrogels, further supporting the relationship between crosslinking density and hydrogel stability over time. Swelling degrees of oil-doped PVA/CMC hydrogels.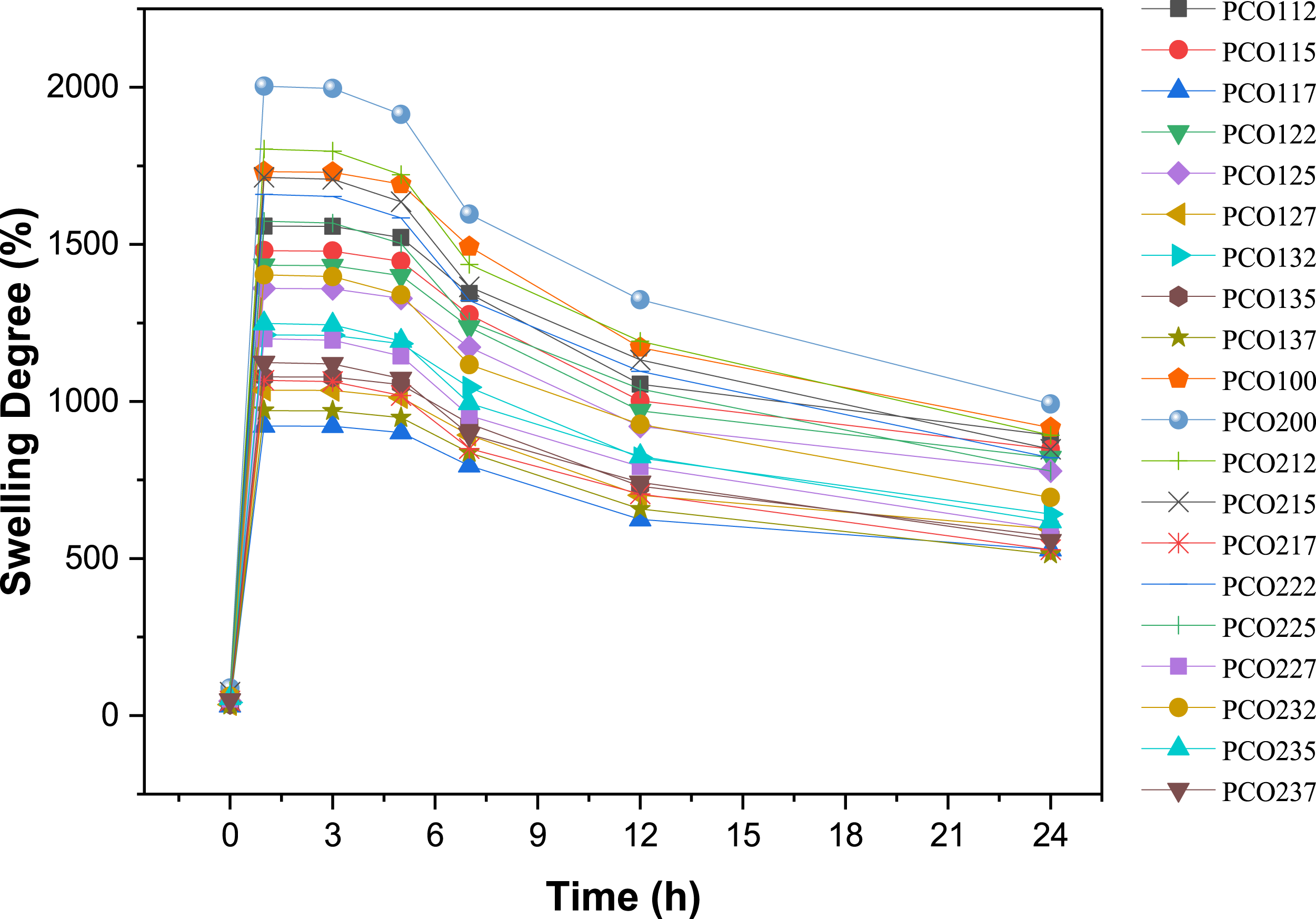
The effect of acetic acid on the crosslinking mechanism of PVA/CMC-based hydrogels was evaluated, and its influence on swelling behavior was examined. In the PC100 series, the use of acetic acid led to the protonation of CMC, strengthening hydrogen bonding with PVA and resulting in a higher crosslinking degree. While an increase in crosslinking generally reduces the swelling ratio, some samples retained a significant water absorption capacity. For instance, the PC100 sample exhibited a swelling ratio of 1748% at 1 hour, which decreased to 975% after 24 hours. Similarly, the PC115 sample showed an initial swelling of 1490%, decreasing to 950% at 24 hours. In contrast, the PC117 sample initially reached a swelling ratio of 1875%, but this value dropped to 650% after 24 hours. This trend indicates that increased crosslinking limits water retention capacity by reducing the hydrophilic character of the hydrogel. On the other hand, in the PC200 series, where distilled water was used, the lower crosslinking degree contributed to higher water absorption capacity. The swelling ratios were generally higher compared to the PC100 series. For example, the PC200 sample exhibited a swelling ratio of 1997% at 1 hour and still maintained a relatively high value of 1075% at 24 hours. Similarly, PC212 and PC215 samples showed high swelling ratios. In samples with lower crosslinking degrees, such as PC217 and PC237, water absorption was initially higher, but structural stability decreased over time. Overall, the observed trend indicates that crosslinking degree significantly influences swelling behavior, and the differences between the PC100 and PC200 series confirm the role of the solvent in the crosslinking mechanism. These findings are presented in Table S1 and Figure 2, providing experimental validation of the relationship between crosslinking and swelling behavior.
The absence of oil additives leads to the hydrogels reaching their maximum equilibrium swelling ratio. On the contrary, hydrogels containing clove oil exhibit reduced swelling compared to those with argan and aloe vera oils, underlining the influence of clove oil’s higher hydrophobicity. The lowest degree of swelling was observed in PCO117,127 and 137 hydrogels. Furthermore, a higher oil ratio, specifically 75 microliters, results in a more pronounced reduction in swelling, while minor differences are observed between 25 and 50 microliters. It is important to highlight that hydrogels containing clove oil generally exhibit lower swelling compared to those containing argan and aloe vera oils, a testament to clove oil’s higher hydrophobic nature. 50 Considering various hydrogel types, our results indicate a consistent swelling pattern among PCO 100, 112, 115, 117, 122, 125, 127, 132, 135, 137 and PCO 200, 212, 215, 217, 122, 225, 227, 232, 235, 237 hydrogels. However, PCO 100, 112, 115, 117, 122, 125, 127, 132, 135, 137 hydrogels exhibit superior antimicrobial activity, alongside their water absorption capabilities, making them suitable for potential use as wound dressings, particularly for wound hydration. Additionally, Table S1 provided the swelling ratio (%) over time at specific intervals (1, 3, 5, 7, 12, and 24 hours) along with standard deviation values. The measurements were performed by collecting data from three different regions of each hydrogel sample, and the results were presented in Table S1 as mean values with standard deviations. In essence, the incorporation of essential oils in PVA/CMC hydrogels holds promise for mitigating swelling due to their intrinsic hydrophobic properties. However, the extent of this effect hinges on factors such as oil concentration and compatibility with the hydrogel matrix.
Contact angle measurement
The combination of PVA/CMC’s natural affinity for water (hydrophilicity) with the water-repellent (hydrophobic) characteristics of essential oils yielded varying contact angles, contingent on the proportion of hydrophilic and hydrophobic components.
30
The introduction of hydrophobic oils caused an increase in the contact angle, signifying reduced wetting or enhanced hydrophobic properties.
51
Notably, clove oil, renowned for its high hydrophobicity, induced the most substantial elevation in the contact angle, followed by argan and aloe vera oils. In essence, the assessment of contact angles in PVA/CMC hydrogels serves to evaluate their wetting behavior, with lower angles denoting higher hydrophilicity and higher angles indicative of increased hydrophobicity. Figure 3 provides a visual representation of the contact angle test results on PVA/CMC hydrogels.52–54 It’s evident that the incorporation of hydrophobic oils led to a notable rise in the contact angle, with the hydrogels containing clove oil (PCO135 and PCO235) displaying the highest contact angle values. Furthermore, as shown in Figure 3, hydrogels incorporating argan (PCO115 and PCO215) and aloe vera (PCO125 and PCO225) oils exhibited no comparable difference in observed contact angle values. The wettability of prepared PVA/CMC hydrogels align well with prior research on hydrophilic and hydrophobic modifications in biopolymer-based materials. According to previous studies,21,55 PVA and CMC are inherently hydrophilic polymers, with water contact angles below 40°, primarily due to the presence of hydroxyl and carboxyl functional groups, respectively. The higher hydrophilicity of carboxymethyl cellulose is attributed to its abundant carboxyl functional groups, which facilitate strong interactions with water molecules. Our control samples (PCO100: 36.97° ± 0.74° and PCO200: 31.13° ± 0.62°) exhibit water contact angles consistent with these expectations, confirming their hydrophilic nature. However, when essential oils were incorporated, the water contact angle increased significantly, demonstrating the impact of hydrophobic modifications. Previous studies
56
reported a water contact angle of approximately 55° for hybrid hydrogels, positioning them within the optimal range for promoting cell attachment and proliferation. This hydrophilic-to-moderate hydrophobic transition aligns with our results for PCO115 (72.51° ± 1.45°), PCO125 (73.68° ± 1.47°), PCO135 (83.20° ± 1.66°), PCO215 (62.83° ± 1.26°), PCO225 (66.15° ± 1.32°), and PCO235 (79.86° ± 1.60°). These values confirm that the integration of essential oils modulates surface wettability, rendering the hydrogels moderately hydrophobic, which may enhance their potential for biomedical applications such as wound dressing and controlled drug release. Comparative studies on essential oil-modified hydrogels further support our findings. Previous research demonstrated that increasing the proportion of an essential oil nanoemulsion within carboxymethyl cellulose-based films reduced their water contact angle from 79.10° to 31.30°, reinforcing that oil incorporation alters hydrophilicity. Likewise, another study
26
emphasized that increasing essential oil concentration led to enhanced surface hydrophobicity in oil-stabilized biopolymer films, although factors such as surface roughness and porosity influenced final water contact angle values. Our findings mirror this trend, where the highest contact angles were observed in hydrogels containing clove oil (PCO135: 83.20° ± 1.66°, PCO235: 79.86° ± 1.60°), followed by those with argan and aloe vera oils. Furthermore, additional studies57,58 indicated that the incorporation of hydrophobic oil into PVA and CMC solutions led to structured materials with increased water contact angles due to their porous structure. This aligns with our results, suggesting that not only the intrinsic hydrophobicity of the essential oils but also potential structural modifications in the hydrogel matrix contribute to the observed contact angle variations. Contact angles of oil-doped PVA/CMC hydrogels.
SEM
The structure and surface morphology of PVA/CMC/oil hydrogels were analyzed based on the SEM images presented in Figure 4. The SEM images of all hydrogels, including those with different solvents and 50 µL of various oils, exhibited a distinct textural contrast.
58
Regardless of the type of oil, these hydrophobic surfaces displayed a darker appearance or a pronounced color contrast, which can be attributed to reduced electron interactions in hydrophobic regions. Additionally, contact angle analysis indicated that hydrogels containing clove oil exhibited a higher tendency for water repellency, observed in the PCO135 and PCO235 SEM images as rounded structures with high contact angles. The effect of CMC and PVA composition on surface morphology has been previously investigated.
21
It has been reported that the interfacial surface of PVA/CMC hydrogels is initially porous but becomes smoother upon oil incorporation.21,59 These findings align with our results, confirming that oil addition induces changes in surface texture. Previous studies have also observed that hydrogels exhibit a uniform morphology without phase separation.56,57 Similarly, in our study, the inclusion of oils altered the microstructure and modified polymer-oil interactions compared to oil-free PVA/CMC hydrogels.
21
Furthermore, SEM images revealed differences in oil distribution compared to earlier PVA/CMC findings, suggesting that the incorporation of essential oils affects the homogeneity of polymer matrices.
26
In conclusion, these findings demonstrate that oil incorporation alters the surface morphology of PVA/CMC hydrogels, influencing their porosity, phase behavior, and hydrophobicity. SEM images of oil-doped PVA/CMC hydrogels.
FT-IR
The FTIR spectra of PVA/CMC hydrogels and their hydrogels containing essential oil show distinct differences in Figure 5. The FTIR spectrum of PVA/CMC hydrogels indicates characteristic wave numbers of PVA and CMC. The wave numbers 1160 cm−1 and 1097 cm−1 are typical of PVA, while wave numbers 1599 cm−1 and 1055 cm−1 are typical of CMC.
41
Common peaks in PVA/CMC hydrogels included O-H stretching (around 3283 cm−1), C-H stretching (around 2937 cm−1), and C = O stretching from carboxylic groups (around 1637 cm−1).60–63 The incorporation of essential oils introduces new spectral features. Eugenol, a major component of clove oil, exhibits a characteristic C = C stretching vibration at 1600 cm−1.
64
Fatty acids in argan and aloe vera oils contribute peaks in the 2800-3000 cm−1 region (C-H stretching), while esters present in these oils display a strong C = O stretching peak at 1737 cm-1.
65
The presence of sterols and polysaccharides is confirmed by peaks in the 900-1160 cm−1 range, corresponding to C-O-C and C-O-H stretching vibrations.66–68 Beyond peak identification, molecular interactions such as hydrogen bonding play a crucial role in validating oil incorporation. The broad absorption band at 3450-3190 cm−1 (-O-H stretching) and the distinct peak at 1640 cm−1 (intramolecular H-bond hydroxyl group) suggest strong hydrogen bonding interactions within the modified hydrogel network. Additionally, spectral shifts and intensified bands in the 1500-900 cm−1 region, associated with C–C and C–O stretching and bending vibrations, indicate structural modifications resulting from essential oil integration.
26
Comparative FTIR analyses confirm the successful incorporation of essential oils into the PVA/CMC hydrogel matrix. The observed spectral changes, including peak shifts, intensity variations, and new absorption bands, demonstrate interactions between essential oil components and the hydrogel network, leading to modifications in both structural and chemical properties. FT-IR spectrum of oil-doped PVA/CMC hydrogels.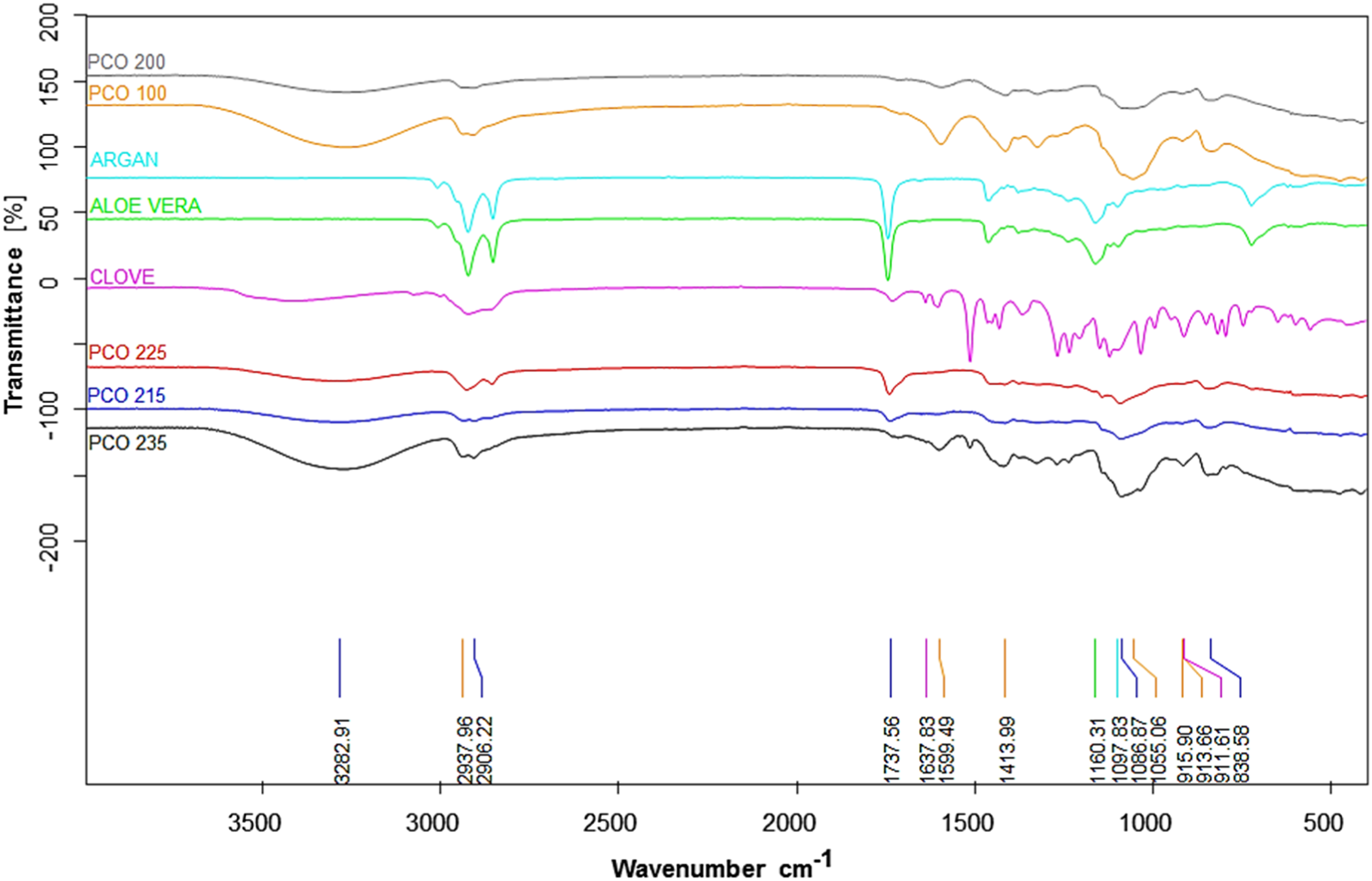
Antımicrobial activity test
Antimicrobial activity results of tested compounds and hydrogels.
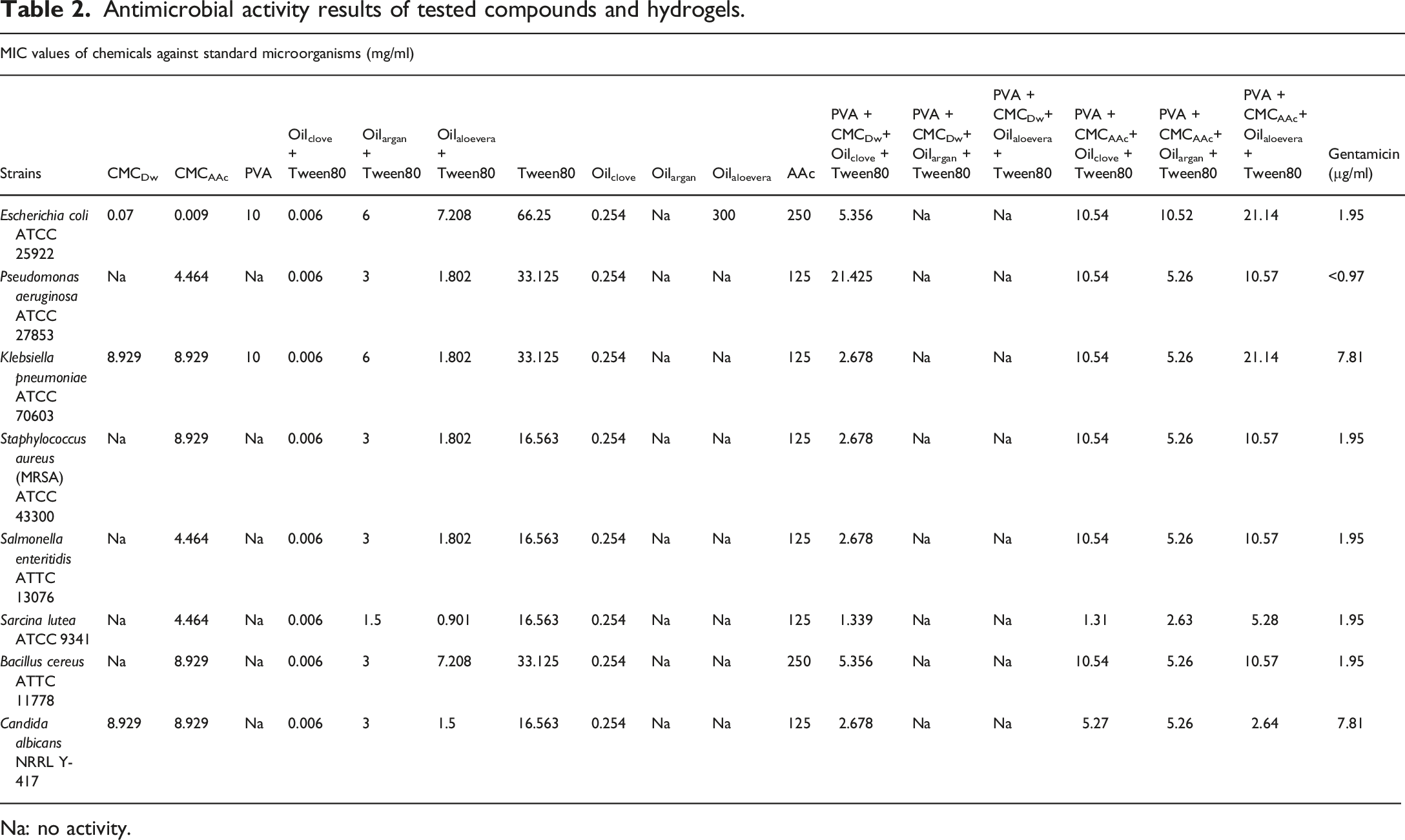
Na: no activity.
In our previous study,
21
some starting materials such as PVA, CMC and AAc were evalutaed for their potential antimicrobial action. As the experiments were conducted in coordination, some data from the aforementioned study were used in this study. The clove oil with tween 80 revealed excellent antimicrobial activity against all test organisms with a MIC value of 0.006 mg/mL. In other word, clove oil and tween 80 demonstrates noteworthy antimicrobial activity, as indicated by the relatively low Gentamicin values across various organisms. Only clove oil combination manifested antimicrobial action against all test organisms at concentration of 0.254 mg/mL. Argan oil + tween 80 also exhibits some antimicrobial effect. Bu it was discussed as weak antimicrobial when compared with clove oil. Sarcina lute was affected by this component at a dose of 1.5 mg/mL. Except for
When the CMCDW evalutaed in our previous study (Bayraktar et al 2025), it was only effective on
The inclusion of clove oil and Tween80 in PVA + CMCDw significantly enhances its antimicrobial activity.69,70 This suggests that the incorporation of clove oil into hydrogels, when used in conjunction with CMCDw and Tween80, enhances their antimicrobial efficacy. In our study this hydrogel was effective on all tested micoorganisms at different concentration. The most sensitive bacteria was
The hydrogel, composed of clove oil, Tween 80, PVA and CMCAAc, exhibited effectiveness against all test microorganisms, particularly at a concentration of 10.54 mg/mL, with the exception of
In summary, Oilclove stands out as the most potent antimicrobial component among the tested oils. Clove oil appears to be a key contributor to this enhanced antimicrobial activity. Addition of argan oil to the hydrogel consisting CMCAAc boosted antimicrobial activity, too. Similary to the bacterial organisms,
Discussion
This research extends our work in the realm of PVA/CMC hydrogels. Contact angle, swelling behavior, and antimicrobial characteristics in hydrogels prepared through the freeze-thaw method were studied by incorporating argan, clove, and aloe vera essential oils. The surface morphologies of these hydrogels were examined using various analytical techniques to elucidate their chemical structure and chemical composition. The presence of essential oils, with their hydrophobic nature, significantly influences hydrogel swelling, with oil concentration being a key factor. Our findings demonstrate that PCO200 hydrogels exhibit higher swelling due to greater crosslinking, supported by various analytical techniques. The absence of oil additives leads to maximum swelling, while clove oil imparts the lowest swelling among the oils. Hydrogel type shows a consistent swelling pattern, with PCO 100, 112, 115, 117, 122, 125, 127, 132, 135, and 137 hydrogels exhibiting superior antimicrobial activity, making them suitable for potential use as wound dressings.
PC100 series hydrogels exhibited characteristic peaks associated with strong hydrogen bonding and enhanced crosslinking. The broad –OH stretching band (∼3300 cm−1) was more pronounced, indicating increased intermolecular interactions between PVA and CMC due to the protonation effect of AAc. This finding aligns with previous studies reporting that acidic environments enhance crosslinking by strengthening hydrogen bonds.21,59 In contrast, PC200 series hydrogels showed a relatively weaker –OH band, suggesting lower crosslinking density and higher water retention, consistent with literature on physically crosslinked PVA/CMC systems26,56. Oil-loaded hydrogels exhibited distinct spectral changes compared to non-oil samples. The presence of carbonyl (C = O) stretching (∼1740 cm−1) and C–H stretching (∼2850–2920 cm−1) bands was more prominent in argan, aloe, and clove oil-loaded hydrogels, confirming oil incorporation into the polymer matrix. Similar results were reported in studies where essential oils altered the hydrophilic-hydrophobic balance of hydrogel systems. 57 Additionally, oil addition led to a reduction in the –OH peak intensity, indicating a shift in hydrogen bonding interactions, which may contribute to increased hydrophobicity and altered swelling behavior.
Our findings align with previous research, indicating the promise of essential oil incorporation in mitigating swelling, contingent on factors like oil concentration and compatibility with the hydrogel matrix. In summary, the combination of PVA/CMC’s hydrophilicity with the hydrophobic characteristics of essential oils resulted in varying contact angles, reflecting wetting behavior. Hydrophobic oils, especially clove oil, led to significantly higher contact angles, while argan and aloe vera oils exhibited relatively similar contact angle values. This assessment, as depicted in Figure 3, provides a clear visual representation of the impact of hydrophobic oils on contact angles in PVA/CMC/Oil hydrogels, with lower angles indicating higher hydrophilicity and higher angles suggesting increased hydrophobicity. SEM images revealed the formation of macroporosity in all our PVA/CMC and PVA/CMC/Oil hydrogels during the freeze/thawing process, consistent with previous research. Additionally, SEM images provided insights into the interaction between the oil and the polymer matrix, as well as the dispersion of oil droplets in the hydrogels. Hydrogels containing 50 microliters of oil, including argan, aloe, and clove oil, displayed distinct textural contrast and hydrophobic surfaces. This contrast, attributed to reduced electron interaction in hydrophobic regions, was consistent with the results of contact angle analysis. Notably, hydrogels containing clove oil exhibited high contact angles, indicating strong water repellent as observed in the SEM images of PCO135 and
In the antimicrobial analysis the presented Table 2 encompasses an antimicrobial analysis of multiple organisms and chemical components. Oilclove, demonstrates noteworthy antimicrobial activity, as indicated by the relatively low Gentamicin values across various organisms. Argan and aloe vera oils have no antimicrobial effects. The inclusion of clove oil and Tween80 in PVA + CMCDw changed its antimicrobial activity and this activity was boosted. This observation suggests the hypothesis of a synergistic effect between clove oil and the other constituents, namely PVA+, CMCDw, and Tween80, resulting in an enhancement of the antimicrobial efficacy of hydrogels. The present study suggests a potential for clove oil to play a pivotal role in this augmented antimicrobial activity. The incorporation of argan and aloe vera oil, along with Tween80, into a blend of PVA and CMCAAC increased discernable antimicrobial properties. It was discussed in results section that hydrogels containing clove oil was effective on tested microorganisms at different concentrations and
Conclusions
This research advances the understanding of PVA/CMC hydrogels by investigating their contact angle, swelling behavior, and antimicrobial properties upon the incorporation of argan, clove, and aloe vera essential oils. The study’s pivotal findings demonstrate that the hydrophobicity of the essential oils significantly influences the hydrogel’s swelling capacity, with clove oil imparting the lowest swelling effect due to its strong hydrophobic nature, as evidenced by contact angle measurements and SEM images. Furthermore, the antimicrobial analysis indicates that clove oil and argan oil, particularly when combined with Tween80, substantially enhances the hydrogel’s ability to combat various pathogen bacteria such as methicillin resistant
Supplemental Material
Supplemental Material - Antimicrobial activity and characterization of PVA/CMC hydrogels prepared with essential oil
Supplemental Material for Antimicrobial activity and characterization of PVA/CMC hydrogels prepared with essential oil by Seyda Bayraktar, Buse Copur, Erdogan Gunes, Ahmet Uysal and Ahmet Okudan in Journal of Thermoplastic Composite Materials.
Footnotes
Author’s note
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Author contributions
Project Administration: [Ahmet OKUDAN]; Experimental Work: [Buse ÇOPUR, Şeyda BAYRAKTAR]; Antimicrobial Studies and Analysis: [Ahmet UYSAL, Erdoğan GÜNEŞ]; Writing-Original Draft: [Buse ÇOPUR, Şeyda BAYRAKTAR, Ahmet OKUDAN, Ahmet UYSAL, Erdoğan GÜNEŞ]; Writing-Review & Editing: [Buse ÇOPUR, Ahmet OKUDAN].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study presented was supported by Selcuk University Research Foundation (project number: BAP-20401100).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
