Abstract
This work examined the effects of filler content and chemical treatment on the biodegradation of poly(lactic acid) (PLA)/coconut shell (CS) biocomposites in a diastase enzyme-containing buffer medium. CS was treated with two distinct chemical treatments: maleic acid and silanation with 3-aminopropyltriethoxysilane (3-APE). The CS was incorporated into PLA composites and their biodegradation patterns were studied. Both of the treated PLA/CS biocomposites exhibited lower biodegradation rates than the untreated biocomposites due to their enhanced interfacial adhesion, which reduced the area exposed to enzyme hydrolysis. Scanning electron micrographs taken after 30 days of biodegradation displayed surface roughening on both of the treated biocomposites, with fewer voids compared to the untreated biocomposites. The differential scanning calorimetry indicated that the glass transition temperature and melting temperature values of the treated biocomposites increased but that crystallinity declined. The crystallization temperature peak apparently disappeared due to the polymer chain alignment and rearrangement of the shorter PLA chains caused by the degradation. Fourier transform infrared analysis revealed the structural changes in the biocomposites after biodegradation, indicating the presence of soluble lactic acid as was confirmed by ultraviolet–visible spectroscopy analysis.
Introduction
Plastic waste has been a major environmental issue since the third industrial revolution several decades ago. This waste constitutes approximately 10–11% of the world’s total waste; therefore, the management of plastic waste will require several complementary practices. 1 Biodegradable plastic technology is regarded as an attractive solution to alleviate this issue, particularly due to its proven history of success. 2,3 One of the commercial biodegradable plastics is poly(lactic acid) (PLA). 4 This bioplastic is a transparent and crystalline polymer that is synthesized either by the condensation polymerization of lactic acid or by the ring-opening polymerization of lactide (the cyclic dimer of lactic acid). 5 The lactic acid is produced by chemical synthesis or through the fermentation of carbohydrates such as starch, glucose, and xylose. PLA has such desirable physical properties as a high melting point (175°C), high strength, and ease of processing. PLA is currently used in large number of fields, including agricultural, biomedical, and packaging applications, showing its high potential to replace non-biodegradable plastics. 6,7 However, the market of PLA is limited due to its high production cost and relatively poor mechanical properties. 8
PLA-natural fiber biocomposites have been developed to reduce the PLA production material costs via the incorporation of inexpensive fibers and also to increase the material toughness and moduli because of the reinforcing effect from the natural fiber. 9,10 However, the highly hydrophilic nature of these fiber hinders the fiber–polymer interaction due to the poor compatibilization on the interface. This problem is often alleviated by the chemical treatment of the fibers or by the addition of a functionalized polymer to the composite system. 11 Most of these chemical treatments aim to compatibilize the hydrophilicity of the fiber with the polymer to aid the polymer wetting. Some chemical treatments covalently bond the two components together or add surface roughness to the fiber to aid mechanical interlocking. A large variety of chemicals have been used in this compatibilization, but the success depends on the type of fibers, the polymer used, the choice and method of treatment, and the optimum level of modification achieved. 12,13
Many studies have been conducted on the biodegradation and biodegradability of PLA biocomposites, with particular emphasis on microbial and enzymatic degradation. 14,15 Proteinase K from Tritirachium album, subtilisin, a microbial serine protease has been employed in the PLA biodegradation. 16 Several mammalian serine proteases such as α-chymotrypsin, trypsin, and elastase have also been used to degrade PLA. 17 Among the known enzymes, diastase, a class of hydrolases that is widely distributed in microbes, plants, and animals has been shown to degrade starch-based material through a hydrolytic mechanism. 18 However, the use of this enzyme in PLA biodegradation has not yet been reported. Diastase is a group of starch-digesting enzymes that includes α- and β-amylase. The enzyme α-amylase hydrolyses starch chains at random locations for producing a variety of dextrins, and β-amylase splits the reducing sugar maltose from the end of the starch chain. 19 The degradation of starch mainly takes place on the ester bond along the molecular chain. PLA is composed of ester bond linkages, hence the biodegradation is expected to occur at the ester bond along the backbone chains.
This study continues the focus of previous works that investigated the effects of coconut shell (CS) powder content and chemical treatment on the mechanical and thermal properties of the PLA/CS biocomposites. 20,21 The present work examines the biodegradation behavior of PLA/CS biocomposites with and without chemical treatment in a diastase containing solution. Evaluations of the weight loss, chemical degradation, and thermal and morphological properties of the PLA/CS biocomposites after enzymatic degradation are also provided.
Materials and method
Materials
The PLA used in this study was supplied by TT Biotechnology Sdn. Bhd., Penang (Malaysia). The melt flow index of the PLA was 5.6 g/10 min (180°C/2160 g), and its density was 1.27 g cm−3. Commercial-grade diastase (sourced from malt) was purchased from Sigma Aldrich, Penang. CS powder was produced from CSs collected from a market in Perlis. The CS was cleaned manually and crushed into small pieces. These pieces were then ground into fine powder and dried at 80°C for 24 h. The average particle size of the resulting CS powder, as measured by a Malvern Particle Size Analyzer (Perlis, Malaysia), was 38 µm. The chemical reagents, that is, maleic acid, 3-aminopropyltriethoxysilane (3-APE), acetic acid, sodium acetate, and ethanol, were supplied by Fluka (Penang, Malaysia).
CS filler treatments
Two different chemical treatments applied were the mixing with maleic acid and the silanation with 3-APE. The maleic acid-treated CS filler was produced by immersing the CS filler in a stirred solution of maleic acid (3% by weight of filler) and ethanol for 2 h at ambient temperature before drying. The silanated CS filler was obtained by performing a pre-hydrolysis reaction with 3-APE to form silanol and then silanating with the CS filler to form Si–O–C linkage. The pre-hydrolysis reaction was conducted by reacting 3-APE with aqueous ethanol for 1 h at ambient temperature. Afterward, the CS filler was added and stirred slowly for the next 2 h. The filler was then removed, rinsed, and dried at 80°C for 24 h. The formulations of the untreated and treated PLA/CS biocomposites are shown in Table 1.
Composite composition and treatment.
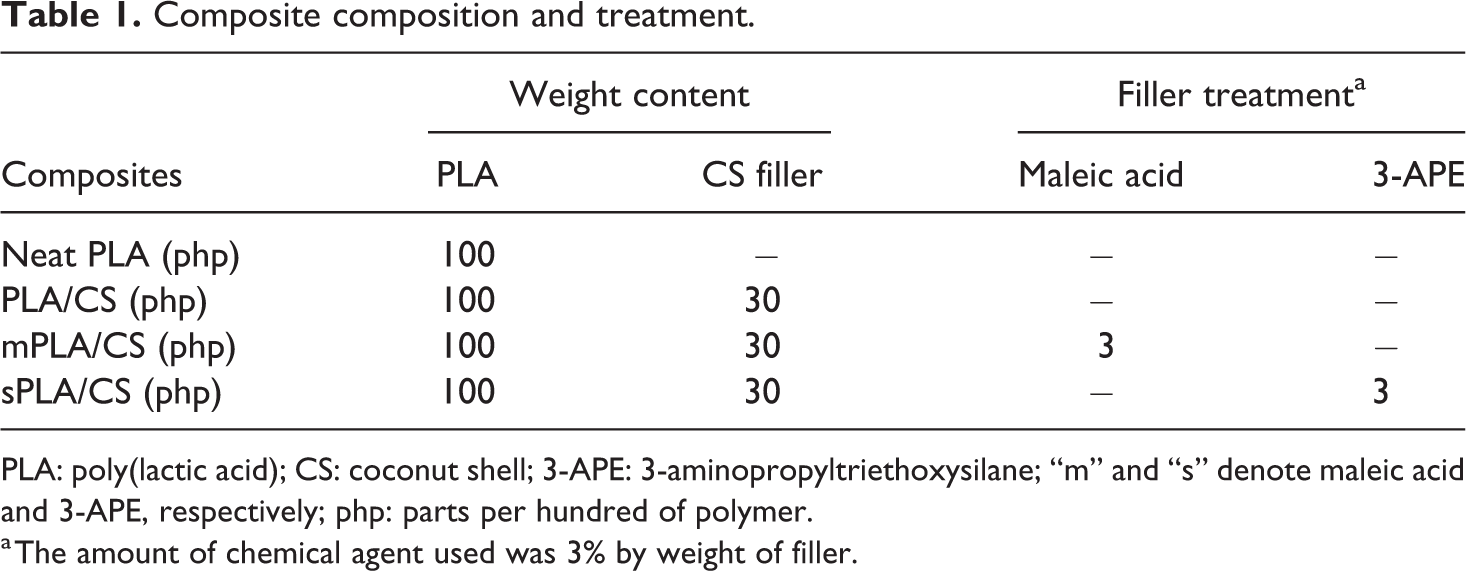
PLA: poly(lactic acid); CS: coconut shell; 3-APE: 3-aminopropyltriethoxysilane; “m” and “s” denote maleic acid and 3-APE, respectively; php: parts per hundred of polymer.
a The amount of chemical agent used was 3% by weight of filler.
Preparation of PLA/CS biocomposites
The PLA/CS biocomposites were compounded using a Brabender Plastrograph mixer (Perlis, Malaysia) Model EC PLUS at 180°C and a rotor speed of 50 r min−1. First, PLA pellets were loaded into the mixing chamber for 1 min until melted. The CS filler was then added and mixed continuously for 7 min. The total mixing time was 8 min. The PLA/CS biocomposites were compressed into a tensile bar using a Gotech compression-molding machine model GT 7014A (Perlis, Malaysia). The compression-molding cycle involved 1 min of preheating at 180°C, followed by compression under 9.8 MPa for 1 min. After compression, the sample was cooled under the same pressure for 5 min.
Preparation of substrate
The biocomposite specimens were cut into small squares (6 × 5 × 1 mm3) and placed into a relative humidity (RH) cabinet set to 50% RH at 25°C for 1 week to allow for moisture content equilibration. The equilibration of samples was performed to ensure that each specimen was maintained under uniform storage conditions. The moisture content within each sample remained unchanged after equilibration, demonstrating that all samples had reached equilibrium.
Enzymatic degradation of PLA/CS biocomposites
The enzymatic degradation test was conducted in a diastase containing solution with an activity of 480 KNU g−1. A buffer solution (pH 7.3) was prepared in a beaker by adding 5 ml of 0.2-M acetic acid to 45 ml of 0.2-M sodium acetate solution. The samples were immersed in the solution for 40 days in an incubator (New Brunswich Scientific, Perlis, Malaysia) at 37°C. The samples were removed every 5 days and washed thoroughly with distilled water before a conditioning step in an oven at 60°C for 24 h.
Weight loss analysis
Each specimen was removed periodically, wiped with tissue and dried until a constant weight at 60°C in a vacuum oven. The percentage of weight loss was measured using an electronic balance and calculated using equation (1)
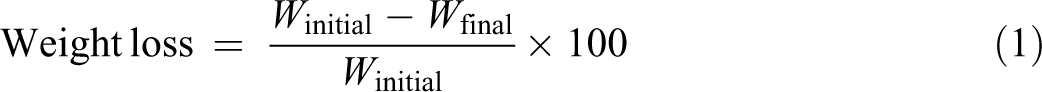
where W initial and W final are the weights of the sample before and after the enzymatic degradation test, respectively. The mean value of three measurements for each compound was recorded.
Tensile test
Tensile tests were conducted according to ASTM D 638-91 on an Instron 5582. A minimum of five dumbbell specimens of each composition, 1 mm thick were cut from the molded sheets with a Wallace die cutter. A crosshead speed of 20 mm min−1 was used, and the test was performed at 25 ± 3°C.
Differential scanning calorimetry
Differential scanning calorimetry (DSC) analysis was conducted using a DSC Q 1000 (Perkin Elmer TA Instrument, Malaysia). Samples were scanned from 25°C to 250°C at a heating rate of 20°C min−1 under nitrogen flow of 50 ml min−1. The glass transition temperature (T g), crystallization temperature (T c), melting point (T m), and enthalpy of each PLA/CS biocomposite were automatically calculated by the instrument software. The crystallinity of each composite (X c) was manually calculated using equation (2)

where
SEM analysis
The morphologies of the untreated and treated PLA/CS biocomposites before and after degradation were investigated with a scanning electron microscope (SEM) JEOL model JSM 6260 LE (Perlis, Malaysia). The fracture ends of the specimens were mounted on aluminum stubs and sputter coated with palladium to avoid electrostatic charging during examination.
FTIR analysis
Fourier transforms infrared (FTIR) spectroscopy, using the attenuated total reflection mode, was performed on the PLA/CS specimens before and after enzymatic degradation test (Perkin Elmer 1600 Series). Samples were scanned from 650 cm−1 to 4000 cm−1 with a resolution of 4 cm−1. The carbonyl index of each sample was calculated using the equation (3)
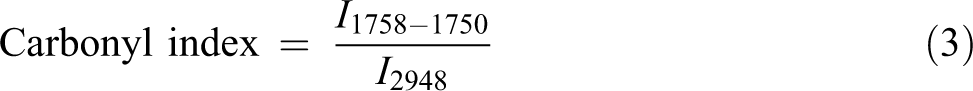
where I represents the peak intensity. In this study, the peak at 2948 cm−1 was chosen as the reference peak.
UV-vis spectroscopy
Ultraviolet–visible (UV-vis) spectroscopic analysis was conducted using Varian Cary 20 Scanning (Perlis, Malaysia) to determine the presence of soluble degraded lactic acid. The buffer medium before and after a specific biodegradation time were transferred into a quartz cuvette. Absorption spectra were recorded from 200 nm to 350 nm.
Results and discussion
Weight loss analysis
Figure 1 shows the percentage weight loss of neat PLA and the untreated PLA/CS biocomposites as a function of time elapsed in the biodegradation test. Both neat PLA and the untreated PLA/CS biocomposites were further degraded as time elapsed, as indicated by the increasing weight loss. The biodegradation rate of neat PLA was remarkably slower than that of the untreated PLA/CS biocomposites. The incorporation of the CS filler increased the biodegradation rate of the PLA biocomposites with respect to neat PLA. The percentage weight loss of the untreated PLA/CS biocomposites increased approximately twofold over that of neat PLA, suggesting that the presence of the CS filler increased degradation in the PLA composites. The CS filler is lignocellulose material composed of cellulose, hemicellulose, and lignin. These substances contribute to the high polarity character of the PLA/CS biocomposites, leading to decreased tensile strength as reported previously. 20 The increased biodegradation rate of the PLA/CS biocomposites is likely due to the presence of hydroxyl groups belonging to the CS filler, which, when dispersed in PLA, played a vital role in accelerating the hydrolytic degradation of the PLA matrix. 23 Conversely, the biodegradation rates of the PLA/CS biocomposites treated with maleic acid and 3-APE are lower than those of the untreated biocomposite, indicating that the treatments improved the barrier properties of the materials and restricted the diffusion of the enzyme. The enhanced interfacial adhesion between the PLA matrix and the CS filler due to the coupling effects of maleic acid and 3-APE reduced the area exposed to enzyme hydrolysis, resulting in decreased biodegradation rates.
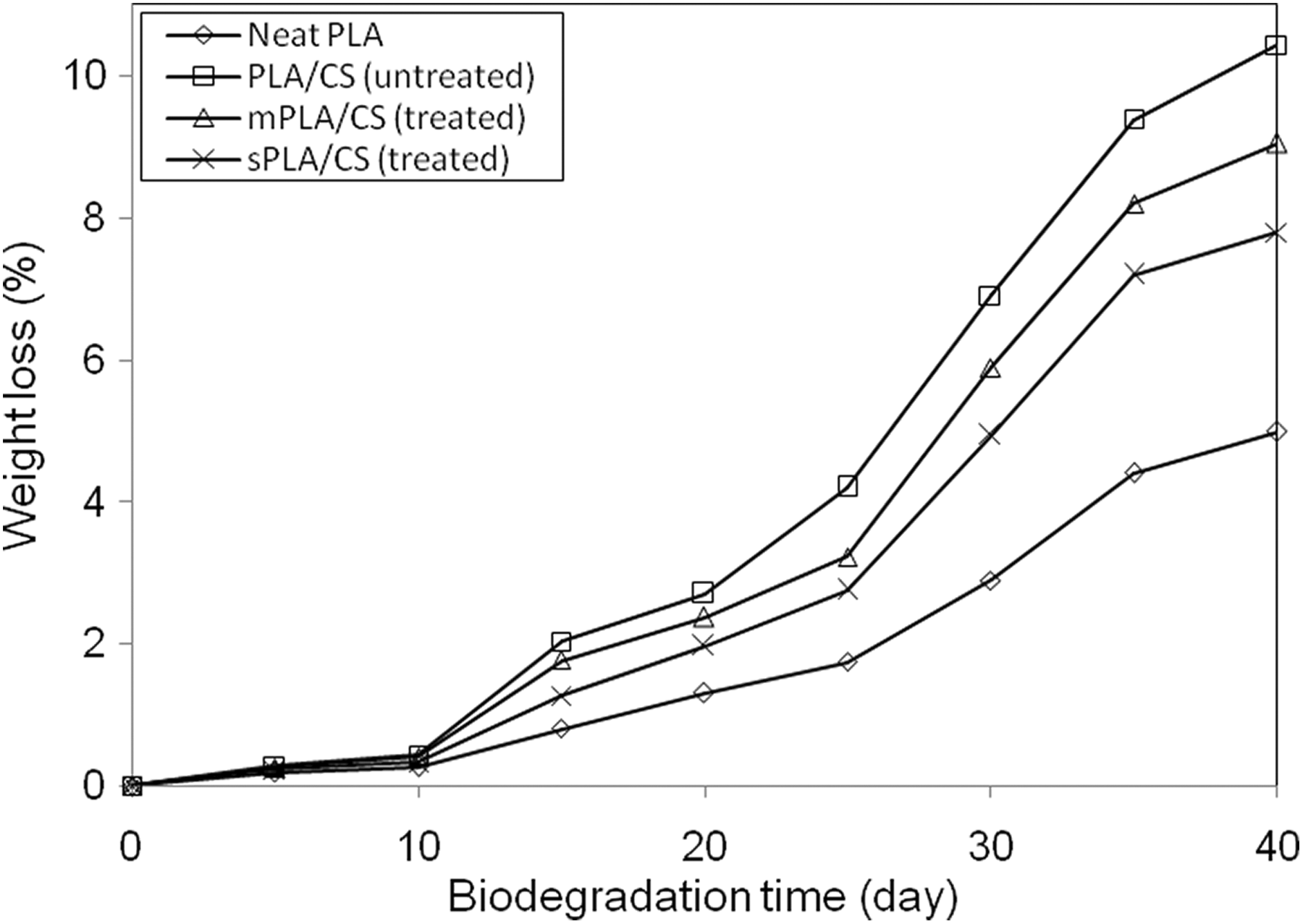
Percentage of weight loss of neat PLA and PLA/CS biocomposites as a function biodegradation time.
Additionally, the PLA/CS biocomposite treated with maleic acid was more susceptible to biodegradation than the PLA/CS biocomposite treated with 3-APE. During the enzymatic test, the water penetrated into the biocomposites, subsequently reacting with the maleic acid to form an acid group through hydrolyzation. This acid group accelerated the chain scission of PLA, resulting in a biocomposite with higher biodegradability. 24 The schematic hydrolyzation of maleic acid-treated CS powder is shown in Figure 2.
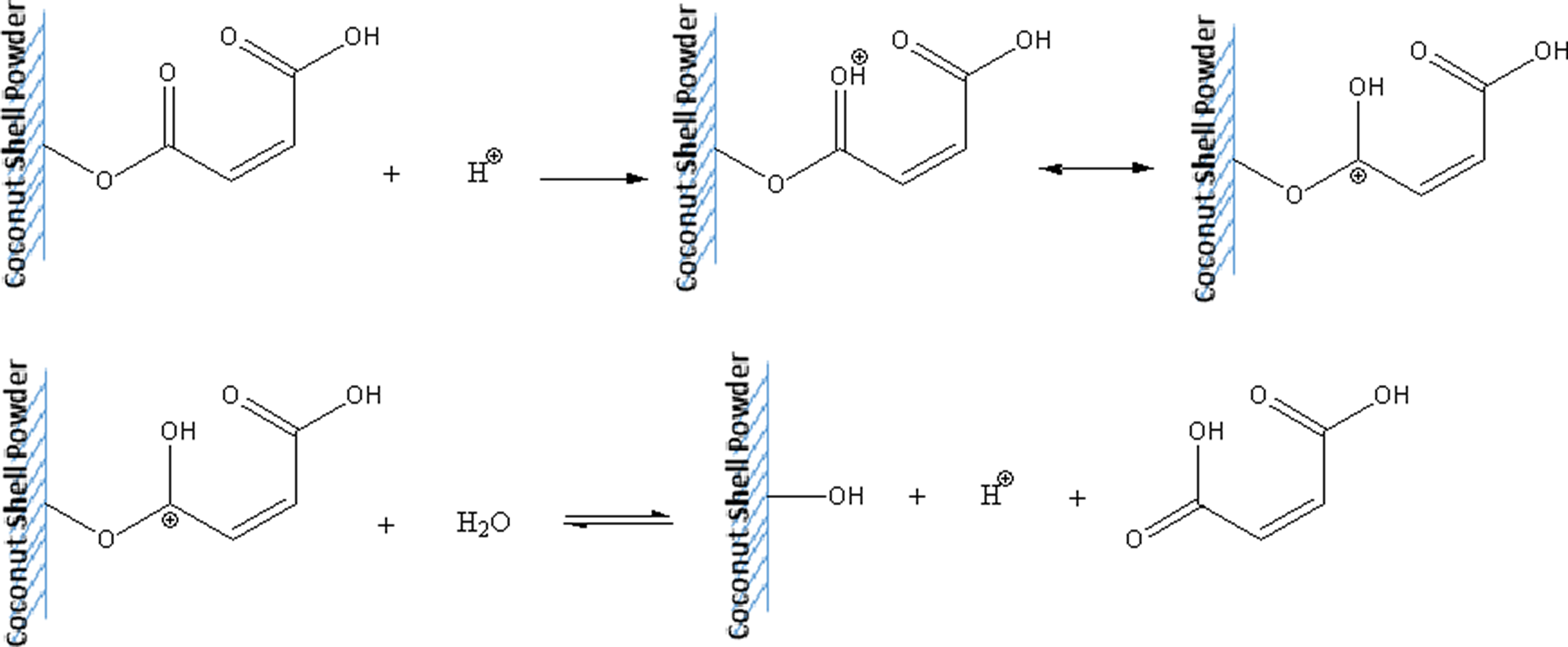
Schematic hydrolyzation of maleic acid-treated CS powder during enzymatic degradation. CS: coconut shell.
Loss of mechanical properties is one of the most relevant practical criterion to determine the degradation of a biodegradable material. Table 2 tabulates tensile strength, elongation at break and Young’s modulus of neat PLA, untreated PLA/CS biocomposite, and PLA/CS biocomposite treated with maleic acid and PLA/CS biocomposite treated with 3-APE at 0 and 30 days biodegradation time. Before immersion, it can be observed that tensile strength of neat PLA decreased upon the addition of 30 php CS filler, presumably caused by poor wettability, dispersion, and filler–matrix adhesion between hydrophilic CS and hydrophobic PLA matrix. Furthermore, elongation at break was also seen to decrease resulted by the decreased chain mobility of the PLA matrix; however, the Young’s modulus improved. The presence of maleic acid and 3-APE coupling agent on the CS surface remarkably increased the tensile strength and Young’s modulus of the treated biocomposites as compared with the untreated biocomposite but lower in elongation at break. From Table 2, it can also be seen the obvious decrease in the tensile strength, elongation at break, and Young’s modulus after 30-day biodegradation time. The action of enzyme in the solution caused the partial disruption of filler–matrix bonding, voids formation, and a subsequent random chain scission in the polymer chains led to a reduction in the tensile properties.
Tensile strength, elongation at break, and Young’s modulus of neat PLA, PLA/CS biocomposites with and without treatment at 0 and 30 days biodegradation.
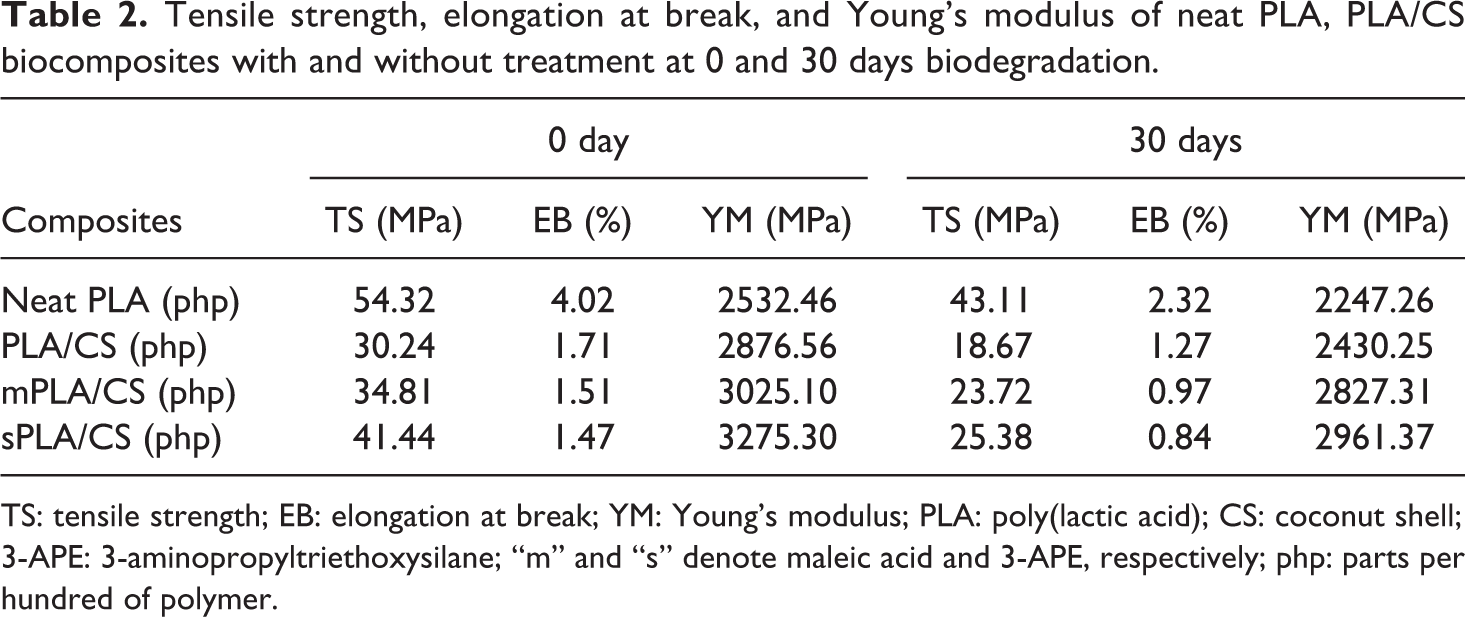
TS: tensile strength; EB: elongation at break; YM: Young’s modulus; PLA: poly(lactic acid); CS: coconut shell; 3-APE: 3-aminopropyltriethoxysilane; “m” and “s” denote maleic acid and 3-APE, respectively; php: parts per hundred of polymer.
SEM analysis
Figure 3(a) to (d) shows the SEM micrographs of neat PLA, untreated PLA/CS biocomposite, PLA/CS biocomposite treated with maleic acid, and PLA/CS biocomposite treated with 3-APE before and after biodegradation for 30 days, respectively. All of the samples exhibited a relatively smooth and clear surface at day 0 of biodegradation. After degradation for 30 days, visible erosion was observed on each surface due to the biodegradation of PLA matrix, which caused permanent damage to the material. The untreated PLA/CS biocomposite and both of the treated PLA/CS biocomposites exhibited significantly more surface erosion, corresponding to their higher biodegradation rate. In the untreated PLA/CS biocomposite (Figure 3(b)), the majority of the matrix was degraded, leading to the formation of large holes on the surface. The erosion of the treated PLA/CS biocomposites (Figure 3(c) and (d)) was slower than that of the untreated biocomposite. The surface of the treated biocomposites became rougher but exhibited fewer voids after 30 days of biodegradation. This result may be attributable to the stronger interfacial adhesion between the PLA matrix and the CS filler in treated biocomposites, reducing the area exposed to enzyme hydrolysis. Taken together, the SEM micrographs provide clear evidence for the biodegradation of neat PLA and the PLA/CS biocomposites.

SEM micrographs of (a) neat PLA, (b) PLA/CS biocomposite, (c) PLA/CS biocomposite treated with maleic acid, (d) PLA/CS biocomposite treated with 3-APE before and after 30 days biodegradation test.
Differential scanning calorimetry
The effect of enzymatic degradation on the thermal properties of the PLA/CS biocomposites was investigated using DSC analysis. Figure 4(a) to (d) show DSC thermograms of neat PLA, untreated PLA/CS biocomposites, PLA/CS biocomposite treated with maleic acid, and PLA/CS biocomposite treated with 3-APE, respectively, at 0 and 30 days of biodegradation, and the results are tabulated in Table 3. Neat PLA had a T g of 57.7°C and a T m of 151.8°C at day 0 (Figure 4(a)), values were consistent with the literature. 25 After 30 days of biodegradation, both T g and T m of neat PLA increased, while the crystallinity degree (X c) value slightly declined. This phenomenon indicates that biodegradation occurred progressively on the intermediate phase of the PLA without significantly changing the crystalline lamellar thickness. In meantime, the increase in T g may be attributed to the reduced mobility of PLA molecular chain and hydrolysis of the amorphous region. 26
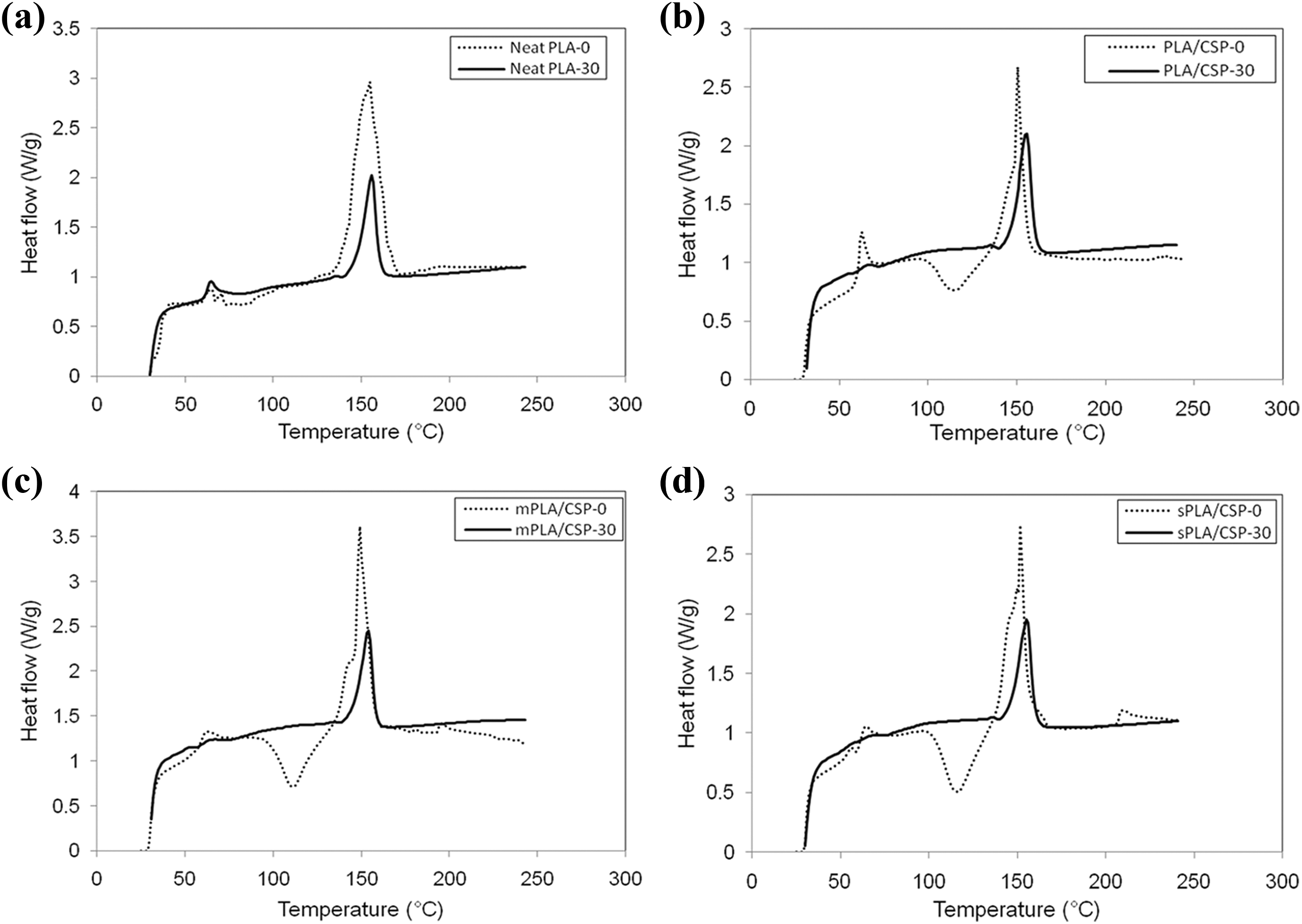
DSC curves of (a) neat PLA, (b) PLA/CS biocomposite, (c) PLA/CS biocomposite treated with maleic acid, (d) PLA/CS biocomposite treated with 3-APE at 0 and 30 days immersed in diastase containing buffer solution.
Summary of T g, T c, T m, and X c of neat PLA, PLA/CS biocomposites with and without treatment at 0 and 30 days biodegradation time.
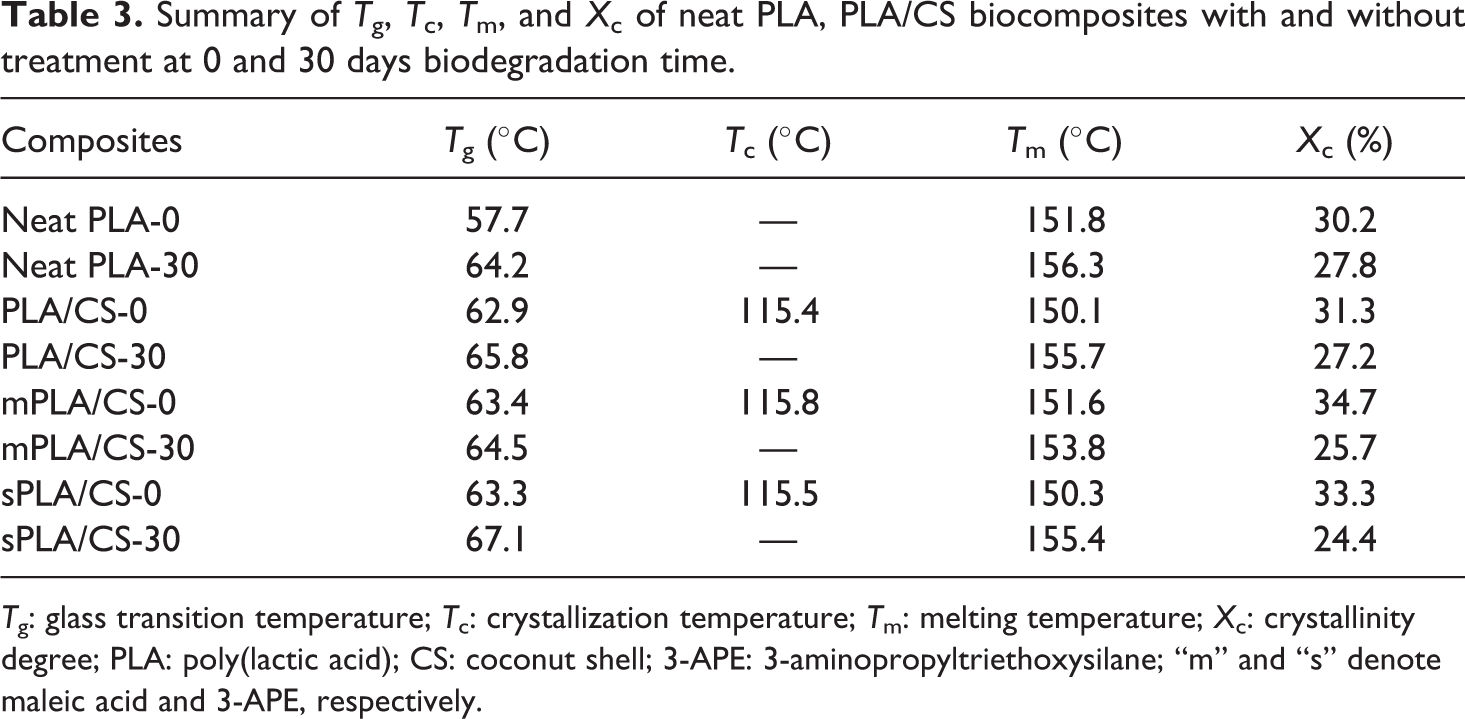
T g: glass transition temperature; T c: crystallization temperature; T m: melting temperature; X c: crystallinity degree; PLA: poly(lactic acid); CS: coconut shell; 3-APE: 3-aminopropyltriethoxysilane; “m” and “s” denote maleic acid and 3-APE, respectively.
On the other hand, the untreated PLA/CS biocomposite (Figure 4(b)), PLA/CS biocomposite treated with maleic acid and PLA/CS biocomposite treated with 3-APE (Figure 4(c) and (d)) showed T g values between 62°C and 63°C and T m values of approximately 151°C at day 0, considerably higher than those observed for neat PLA. A crystallization peak (T c) at 115°C was also observed in these biocomposites, indicating the formation of metastable crystalline polymers due to the addition of CS filler in the PLA composites. As immersion for 30 days, the T g and T m values of the untreated PLA/CS biocomposite increased from 62.9°C to 65.8°C and 150.1°C to 155.7°C, respectively. Similarly, both of the treated PLA/CS biocomposites also exhibited increases in T g and T m (see Table 3), with the biocomposite treated with 3-APE showing superior qualities compared to the biocomposite treated with maleic acid. However, the crystallinity degree of each biocomposite decreased over time due to their reduced proportions of PLA. Furthermore, the disappearance of the T c peak could be attributed to the change in polymer chain alignment and the more convenient rearrangement of the shorter PLA chains that are formed by biodegradation over time.
FTIR spectroscopy
Figure 5(a) to (d) display the IR spectra of neat PLA, untreated PLA/CS biocomposite, PLA/CS biocomposite treated with maleic acid, and PLA/CS biocomposite treated with 3-APE at 0 and 30 days of biodegradation, respectively. The presence of an absorption peak in the 3300–3110 cm−1 range corresponds to the stretching vibration of the hydroxyl groups (−OH) in PLA chain, the peak at approximately 2924–2948 cm−1 is associated with the symmetric deformational vibration of the CH groups in the main chain, and the band in the 1750–1754 cm−1 range is attributed to the stretch vibrations of the carbonyl groups (C=O) in the PLA chain. Similar transmittance bands were observed in untreated PLA/CS biocomposite and both of the treated PLA/CS biocomposites. It is worth noting that neither the addition of CS filler nor chemical treatment significantly altered the PLA matrix spectra.
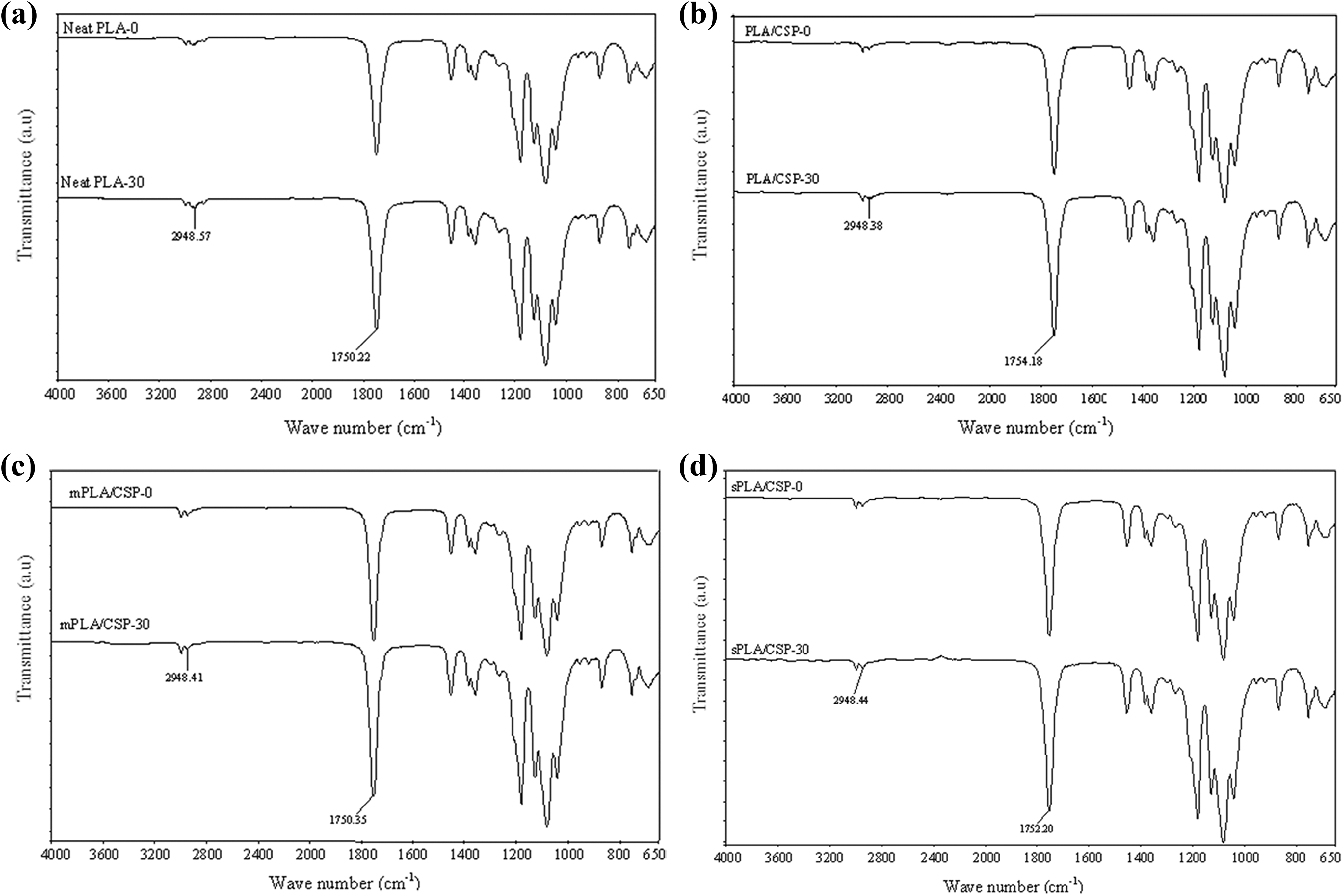
IR spectra of (a) neat PLA, (b) PLA/CS biocomposite, (c) PLA/CS biocomposite treated with maleic acid, (d) PLA/CS biocomposite treated with 3-APE before and after 30 days biodegradation time.
PLA is an aliphatic polyester, and its mechanism of hydrolytic degradation involves the scission of PLA ester groups and the formation of alcohol and carboxylic acid groups. 27 The infrared spectrum of neat PLA after 30 days of biodegradation exhibited a considerable decrease in the width of the IR peak related to the symmetric vibration of CH groups (2948 cm−1) along with an increase in the typical IR absorption of the carbonyl groups belong to carboxylic acids formed by hydrolysis (1758 cm−1). These results clearly confirm the impacts of biodegradation on the PLA polymer. Similar observations were obtained for the untreated PLA/CS biocomposite and both of the treated PLA/CS biocomposites (Figure 5(b) to (d), respectively).
Additionally, the bands at 2948 cm−1 and 1758 cm−1 are essential reference peaks for the identification of the carbonyl indices (CI) of PLA/CS biocomposites, as shown in Table 4. All of the biocomposites exhibited increases in CI with increasing biodegradation time. The increase in CI values after biodegradation is attributed to scission in the PLA chain, which is associated with the formation of carboxylic groups. Similarly, the rupturing of the polymer main chain by enzymatic attack leads to depolymerization. Before degradation, the PLA/CS biocomposites demonstrated higher CI values in comparison to neat PLA, due to the presence of the carbonyl groups from the CS filler. These groups favor the hydrolytic degradation of the PLA matrix, resulting in higher biodegradation rates and CI values. The CI values for both of the treated PLA/CS biocomposites were lower than that of the untreated material, indicating the changes in barrier properties due to the enhanced interfacial adhesion between the PLA matrix and the CS filler. Based on the above observations, a mechanism of PLA biodegradation is proposed in Figure 6.
CI of neat PLA and PLA/CS biocomposites with and without treatments at various degradation time.
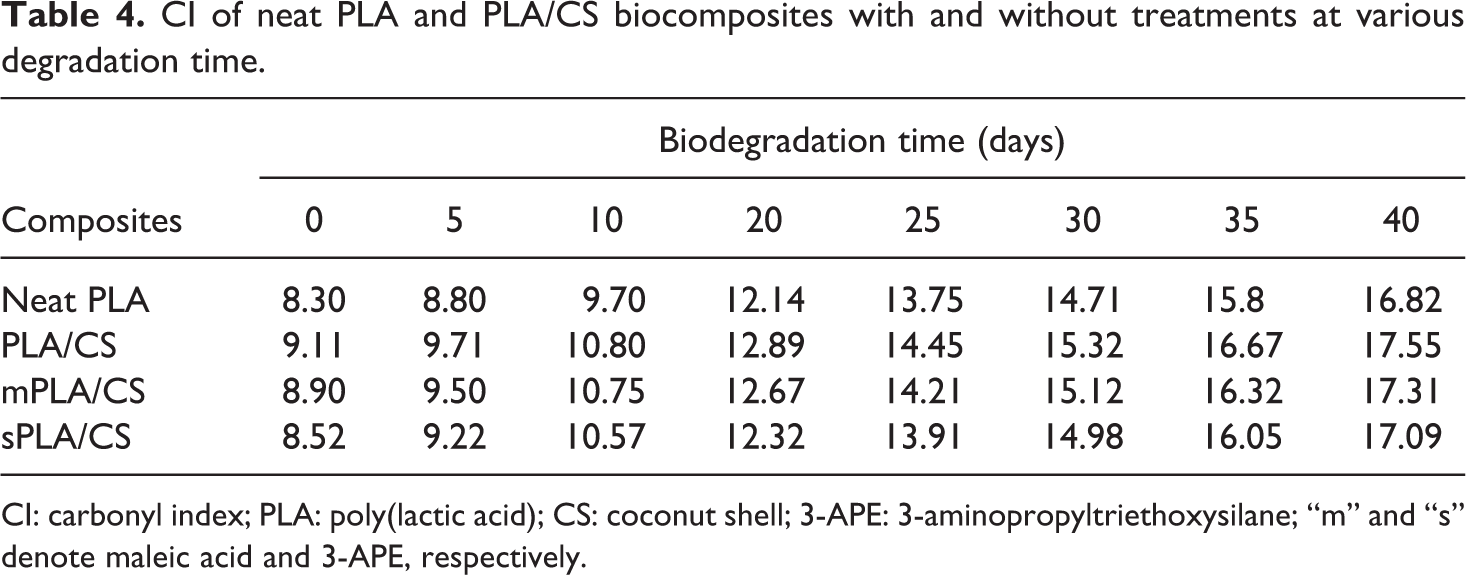
CI: carbonyl index; PLA: poly(lactic acid); CS: coconut shell; 3-APE: 3-aminopropyltriethoxysilane; “m” and “s” denote maleic acid and 3-APE, respectively.
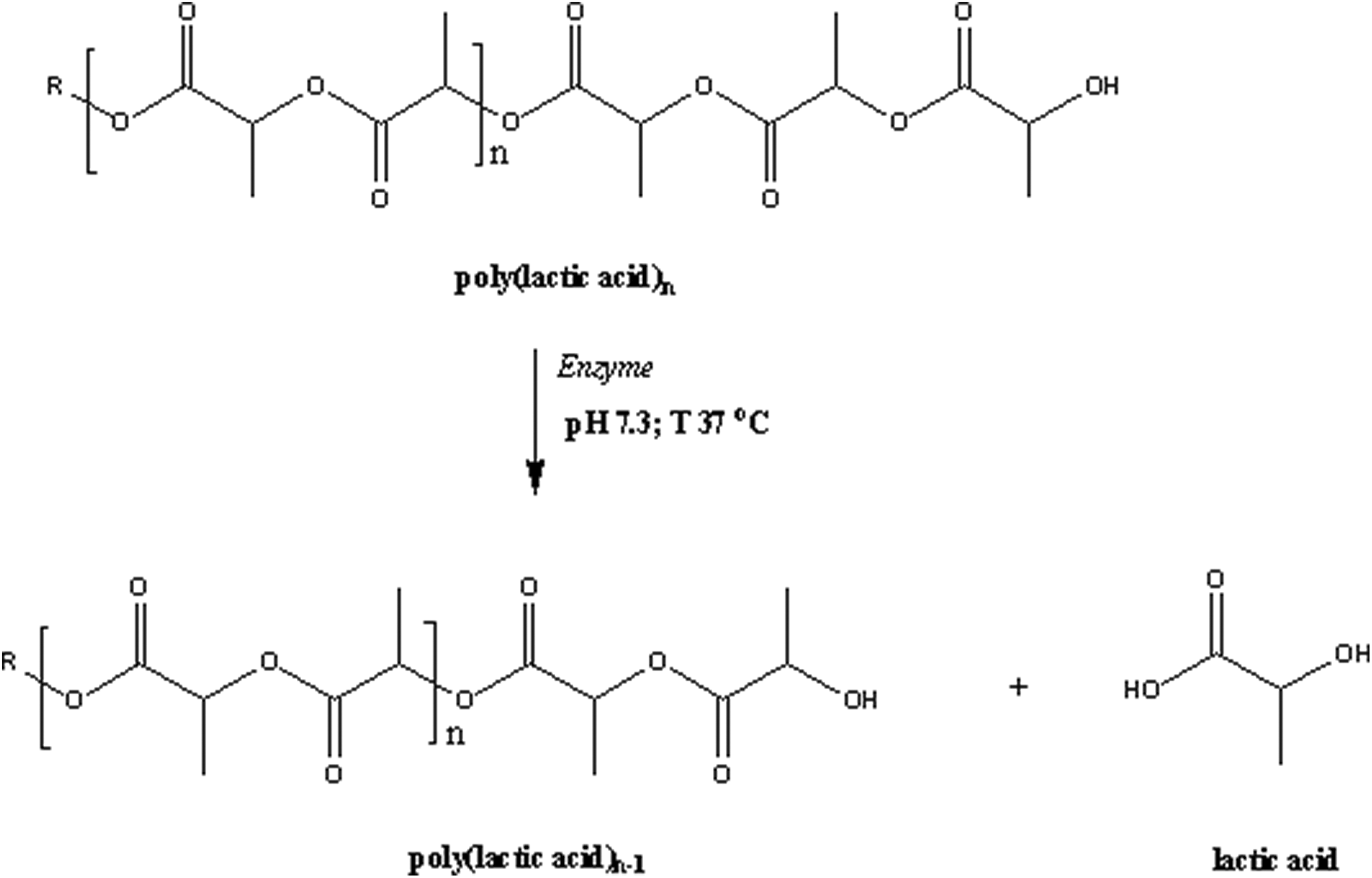
Proposed schematic mechanism of PLA biodegradation.
UV-vis spectroscopy
The presence of soluble degraded lactic acid in the buffer media from neat PLA, untreated PLA/CS biocomposite, and both of the treated PLA/CS biocomposites after 30 days biodegradation was examined. Figure 7 shows the UV-vis spectra of the aqueous solution for each biocomposite, with those containing of aliphatic ester (lactic acid) possessing a maximum wavelength at 280 nm. This value is assigned to the absorption mode of aliphatic carboxyl groups, generated by the scission of the PLA main chain at the aliphatic ester bonds. 28 The higher increase in the absorption intensity of the untreated PLA/CS biocomposite over 30 days of biodegradation as compared to the neat PLA indicates greater amount of soluble lactic acid degraded from the PLA chain for the composite. Furthermore, the absorption spectra of the aqueous solutions obtained after the biodegradation of both of the treated PLA/CS biocomposites clearly demonstrated similar absorption patterns to those of neat PLA and the untreated PLA/CS biocomposites. It is likely that the intensity of the absorption peak was affected by the presence of CS filler as well as by the chemical treatment.
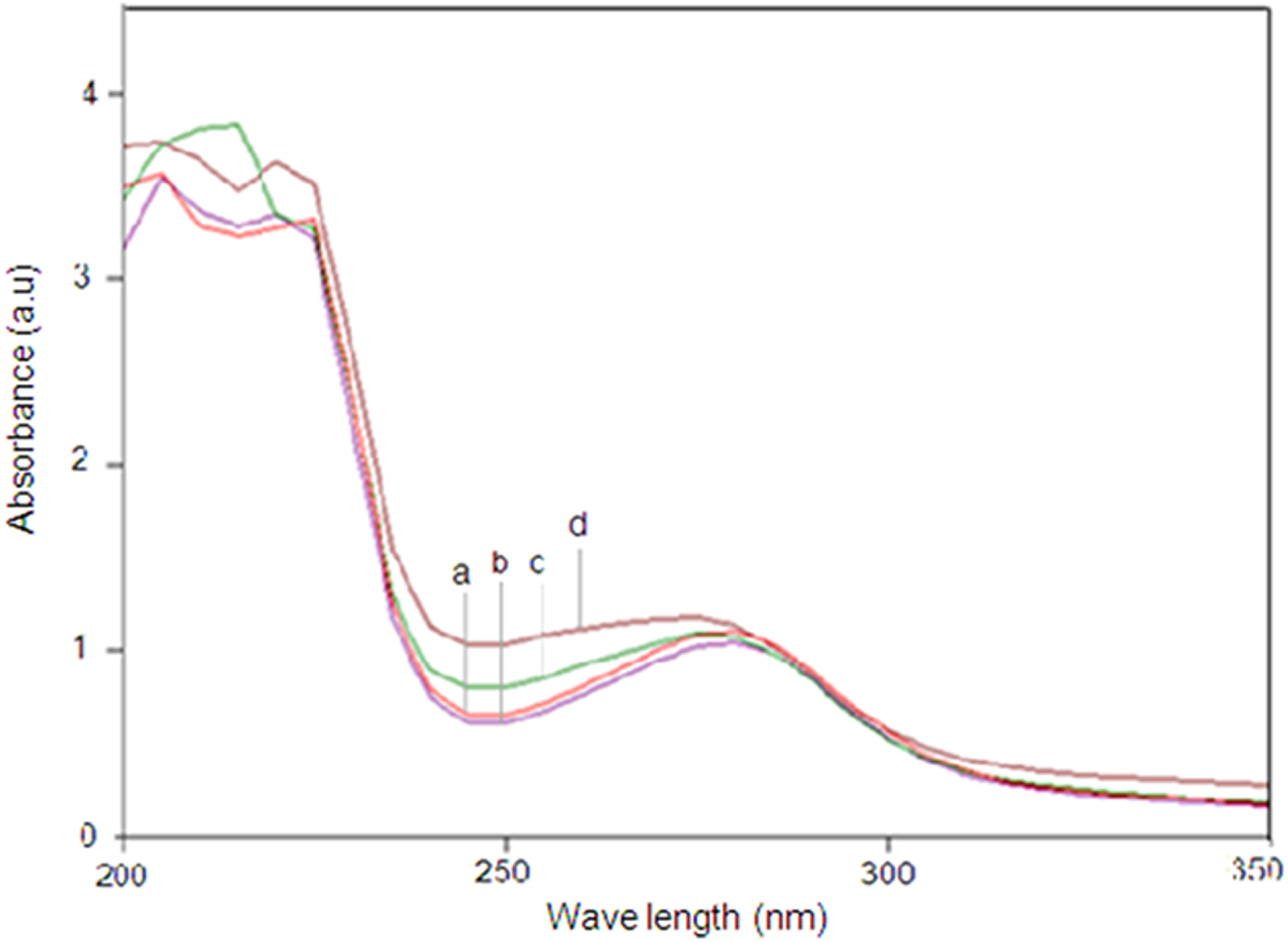
UV-vis spectra of (a) neat PLA, (b) PLA/CS biocomposite, (c) PLA/CS biocomposite treated with maleic acid, (d) PLA/CS biocomposite treated with 3-APE after 30 days immersed in enzyme solution.
Conclusions
Enzymatic degradation of PLA/CS biocomposites with and without treatments was conducted in a diastase-containing solution. The PLA/CS biocomposites were shown to degrade faster than the neat PLA over the extended immersion time. Both of the treated PLA/CS biocomposites showed lower biodegradation rates than the untreated PLA/CS biocomposite due to enhanced interfacial adhesion. SEM micrographs confirmed that roughening occurred on the surface of the treated biocomposites, but fewer voids were formed. The DSC results have demonstrated that the thermal properties were affected by biodegradation time. The T g and T m values both increased, but the X c was diminished. Furthermore, the T c peak disappeared after degradation, suggesting that the polymer chain realigned and that the shorter PLA chains formed by biodegradation rearranged into a more convenient pattern. The FTIR analysis demonstrated that there were structural changes in the PLA chain and both the treated and untreated PLA/CS biocomposites after degradation. Higher carbonyl index values were observed after biodegradation for all of the materials. UV-vis spectroscopy results revealed the presence of soluble degraded lactic acid after 30 days of biodegradation. The intensity of the absorption peak was affected by the presence of CS filler as well as by the chemical treatment. Overall, the results of this study revealed the effectiveness of diastase sp. on the PLA biocomposites biodegradation that will open a new protocol in the recovery of chemicals from waste bioplastics prior to resource sustainability and environmental protection.
Footnotes
Funding
The author(s) disclosed receipt of following financial support for the research, authorship, and/or publication of this article: This work is carried out under the financial support of Fundamental Research Grant Scheme (FRGS) (MOHE Ref. No: FRGS/1/2016/TK05/CURTIN/03/1) entitled “Structure, Morphology and Properties Relationship of Well-Defined Architecture Cellulose Nanowhisker Reinforced Chitosan-g-Polylactic acid Nanocomposites via Atomic Transfer Radical Polymerization (ATRP) in an Ionic Liquid” under cost center number of 001064.
